Abstract
Hepatocellular carcinoma (HCC) is the most frequent primary liver cancer and represents the third and the fifth leading cause of cancer-related death worldwide in men and women, respectively. Hepatitis B virus (HBV) and hepatitis C virus (HCV) chronic infections account for pathogenesis of more than 80 % of primary HCC. HCC prognosis greatly varies according to stage at beginning of treatment, but the overall 5-year survival rate is approximately 5–6 %. Given the limited number of effective therapeutic strategies available, immunotherapies and therapeutic cancer vaccines may help in improving the clinical outcome for HCC patients. However, the few clinical trials conducted to date have shown contrasting results, indicating the need for improvements. In the present study, a novel combinatorial strategy, based on metronomic chemotherapy plus vaccine, is evaluated in a mouse model. The chemotherapy is a multi-drug cocktail including taxanes and alkylating agents, which is administered in a metronomic-like fashion. The vaccine is a multi-peptide cocktail including HCV as well as universal tumor antigen TERT epitopes. The combinatorial strategy designed and evaluated in the present study induces an enhanced specific T cell response, when compared to vaccine alone, which correlates to a reduced Treg frequency. Such results are highly promising and may pave way to relevant improvements in immunotherapeutic strategies for HCC and beyond.
Electronic supplementary material
The online version of this article (doi:10.1007/s00262-015-1698-0) contains supplementary material, which is available to authorized users.
Keywords: PIVAC 14, Liver cancer, Immunotherapy, Cancer vaccine, Metronomic chemotherapy, Combinatorial strategy
Introduction
Hepatocellular carcinoma (HCC) is a leading cause of death from cancer worldwide, estimated to be responsible for nearly 750,000 deaths in 2012 (9.1 % of the total). In most countries, HCC accounts for 70–85 % of primary liver cancer cases, with the burden of disease expected to increase in coming years. In particular, it represents the fifth most common cancer in men (554,000 cases, 7.5 % of the total) and the ninth in women (228,000 cases, 3.4 %) (http://globocan.iarc.fr/).
Chronic HBV and HCV infections are the major recognized risk factors for HCC worldwide [1], HBV being most common in eastern Asia and HCV in Mediterranean countries [2]. In particular, about one-third of all liver cancers occurring in the world are attributed to HCV with a mechanism which has not yet fully deciphered [3]. As a non-integrating virus, HCV is unlikely to have a direct role in HCC initiation. Indeed, since most HCV-associated HCC occurs in the presence of cirrhosis [4], chronic HCV infection may lead to cancer through the indirect mechanism of immune-mediated damage, resulting in liver cell turnover and scarring (fibrosis) [5, 6].
HCC prognosis is generally very poor, with a median survival of 14 months and less than 5 % of symptomatic patients surviving more than 2 years. In early-stage HCC patients, surgery (i.e., tumor resection and liver transplantation) represents the standard treatment with a 5-year survival rate in 70 % of treated patients [7–9]. However, most of the patients present with unresectable HCC or are not eligible for liver transplantation. In such case, only loco-regional non-surgical treatments [i.e. radiofrequency (RF), thermal and nonthermal ablation, trans-arterial chemoembolization (TACE)] can be applied with an extremely variable 3 to 5-year survival rates, according to the disease stage at the time of treatment [10]. Recently, sorafenib, an oral multi-kinase inhibitor with anti-proliferative and anti-angiogenic effects, has been approved for advanced HCCs with sufficient remaining liver function [11]. However, the effect of sorafenib is inadequate to control the progression of HCC. In this framework, immunotherapeutic interventions, including cancer vaccines, may represent a novel and effective therapeutic tool.
To date, a limited number of immunotherapy trials for HCC, including therapeutic vaccines, have been conducted with yet modest results [12–15]. This suggests that improvements in several aspects of the immunotherapy approaches need to be implemented [16–19].
Main causes for the failure of HCC cancer vaccines tested so far can be identified both in the selected antigens and in the non-addressed intrahepatic immunosuppressive environment [20, 21].
Among the antigens tested so far in HCC cancer vaccine approaches, human telomerase reverse transcriptase (hTERT), a catalytic subunit of telomerase, is highly expressed in more than 85 % of different human tumors, with little or no expression in normal somatic cells [22–24]. Moreover, HCC is characterized by somatic mutations in the telomerase reverse transcriptase promoter, leading to increased telomerase expression [25, 26]. Consequently, hTERT is an attractive tumor-associated antigen (TAA) which may be used to develop a universal vaccine for cancer immunotherapy [27–33]. Indeed, therapeutic hTERT vaccines have been developed to elicit telomerase-specific cytotoxic CD8+ T cells based on different strategies, including peptides, full-length proteins, mRNA-transduced dendritic cells (DCs) as well as viral vectors [34–38]. However, clinical trials of hTERT vaccines in cancer patients have proven of limited impact on overall survival [29], indicating that immune response elicited by hTERT alone is not sufficient to control tumor growth.
Considering that HCV chronic infection is responsible for more than 30 % of all HCC worldwide, targeting HCV antigens could represent a valid strategy to control HCC. Indeed, many studies propose that CD8+ T cells are the most important effectors in controlling HCV infection. In particular, a successful viral clearance strongly correlates with multi-specific and cross-genotype CD8+ T cell response to different HCV epitopes, which have been selected for the development of multiple HCV therapeutic vaccines evaluated in clinical trials (reviewed in [39]). Most of such epitopes are located in the core and NS3 structural proteins [40–44].
The intrahepatic immunosuppressive environment includes CD4+CD25+FoxP3+ regulatory T cells (Tregs) which evoke tumor immune evasion mechanisms that can be a major obstacle to the success of cancer immunotherapy [45–47]. Treatments aiming at limiting Tregs should be considered to improve cancer vaccine efficacy in HCC. In this framework, it has been shown that metronomic (low dose) chemotherapy with cyclophosphamide (CTX) or paclitaxel (PTX) in single or daily administration may significantly reduce the number of regulatory T cells [48–50]. Such effect results in improved T cell-mediated anti-tumor responses in both preclinical and clinical studies [50–52] as well as improved immunogenicity of vaccines [48, 53–55].
In this perspective, the overall objective of this study was to assess in a preclinical animal model a novel combinatorial strategy for HCC, combining a cancer vaccine with a metronomic chemotherapy. In particular, the cancer vaccine included the mouse telomerase reverse transcriptase (mTERT) tumor antigen as well as HCV-specific antigens (core and NS3), to elicit a broad T cell immunity; the metronomic chemotherapy included a multi-drug cocktail including taxanes (docetaxel and paclitaxel) and alkylating (cyclophosphamide) agents, to hit different targets of the immune-suppressive environment. Results showed that daily metronomic chemotherapy significantly enhanced the vaccine immunogenicity, and this effect strongly correlated with reduced Treg population.
Materials and methods
Mice
C57BL/6 (H-2b MHC) mice, 5–8 week old, were purchased from Harlan (Udine, Italy). All animals were housed at the Animal Facility of the Istituto Nazionale Tumori “Pascale” (Naples, Italy). Mice were maintained under specific pathogen-free conditions, and all procedures were in accordance with recommendations for the proper use and care of laboratory animals.
Multi-peptide vaccine
The multi-peptide cocktail vaccine used for the immunization included both HCV epitopes, derived from NS3 and Core viral proteins, and universal tumor antigen mTERT epitopes. Binding affinity for H-2Db allele was predicted by in silico modeling using different algorithms (i.e. SYFPEITHI, NetMHC 3.2). The best ranking peptides in terms of affinity values (<50 nM) were selected for immunization: in particular, for the HCV core protein: GGAARALAHGVRVLEDGVNY (145–164); for the NS3 protein: LLYRLGAVQNEVTLTHPITK (598–617); for the mTERT protein: PTRPVGRNFTNLRFLQQIKS (194–213). All peptide were synthesized at a purity of ≥80 % (Primm S.r.l., Milan, Italy). The peptide cocktail contained 20 µg per each peptide, emulsified with a combination of 50 µg of CpG and Montanide ISA 70 at a 30:70 ratio (w/o, vaccine/adjuvant).
Chemotherapy administration
Cyclophosphamide (CTX) (5 and 10 mg/Kg—low and high dose), paclitaxel (PTX) (2.5 and 5 mg/Kg—low and high dose) and docetaxel (DTX) (0.5 and 1 mg/Kg—low and high dose) diluted with phosphate-buffered saline (PBS) were administered via subcutaneous (s.c.) injection. The dose was extrapolated to human equivalent dose (HED) according to Reagan-Shaw et al. [56]. In experiment 1, chemotherapy was weekly administered; in experiment 2, it was daily administered until the end of the experiment.
Immunization experiments
Two immunization experiments were performed. The first one consisted of three vaccine s.c. administrations (week 0, 3, 6), with low- and high-dose chemotherapy once a week (Suppl. Fig. 1). The second one consisted of five weekly vaccine s.c. administrations, with daily high-dose chemotherapy (Suppl. Fig. 3). In both experiments, each experimental group consisted of five animals and control mice were treated with endotoxin-free phosphate-buffered saline (PBS). One week after the last injection, whole blood was collected by puncture of the retro-orbital vein. After euthanasia of mice, spleens were resected and processed into single-cell suspensions for immunological evaluation.
Antibodies for flow cytometry
PE-conjugated anti-mouse CD4 (clone RM4-5), PE/Cy7-conjugated anti-mouse CD8 (clone 53-6.7), APC-conjugated anti-mouse IFN-γ (clone XMG1.2), FITC-conjugated anti-mouse CD25 (clone 3C7), and Alexa Fluor 488-conjugated anti-mouse FoxP3 (clone 150D) antibodies were purchased from BioLegend (San Diego, CA). PerCP-eFluor 710-conjugated anti-mouse CD3 (clone 17-A2) and FITC-conjugated anti-mouse IL-4 antibodies were purchased from eBioscience (San Diego, CA).
Characterization of regulatory T lymphocytes
For Tregs detection, 5 × 105 splenocytes were resuspended in RPMI medium and incubated for 30 min at 4 °C in the dark with PE-conjugated anti-mouse CD4 and FITC-conjugated anti-mouse CD25. After washing and permeabilization, cells were incubated with Alexa Fluor 488-conjugated anti-mouse FoxP3 for 30 min at 4 °C in the dark. The staining was characterized by flow cytometry using FACScan hardware using CellQuest software (BD Biosciences, Mountain View, CA).
Intracellular cytokine staining
1 × 106 splenocytes, after red blood cells lysis with ACK lysing buffer (Lonza, Basel CH), were resuspended in RPMI medium and stimulated for 5 h at 37 °C, in the presence of 1 μL/mL Golgi Plug, with 20 μg of each peptide, or PMA (100 ng/mL) and ionomycin (1 μg/mL) (positive control), or PBS (negative control). Cells were then incubated with PE/Cy7-conjugated anti-mouse CD8 and PerCP-eFluor 710-conjugated anti-mouse CD3 for 30 min at 4 °C in the dark. As previously described, after washing and permeabilization, cells were incubated with APC-conjugated anti-mouse interferon gamma and FITC-conjugated anti-mouse IL-4 for 30 min at 4 °C in the dark. Cells were analyzed at a FACScan hardware using CellQuest software (BD Biosciences, Mountain View, CA).
Statistical analysis
Comparison between individual data points were performed with the unpaired two-sided Student’s t test. All P values were two-tailed and considered significant if <0.05.
Results
Cancer vaccine and weekly metronomic chemotherapy
The multi-peptide vaccine used in the study included epitopes from the NS3 and Core viral proteins as well as the universal tumor antigen TERT. According to parameters described in M&M, the HCV core145–164 epitope, the HCV NS3598–617 epitope, and the mTERT194–213 epitope were selected.
To test whether metronomic chemotherapy would improve the immune response against the multi-peptide vaccine, a protocol combining the vaccine with weekly administered high- or low-dose metronomic chemotherapy was performed in C57BL/6 mice (Suppl. Fig. 1).
The general status of animals in the experimental groups was followed up during the whole protocol, and the weight of each animal was recorded on the day of each immunization. Data clearly indicated that, regardless the treatment, all animals showed a good general status and a progressive increase in the weight during the whole immunization protocol (Suppl. Fig. 2). In particular, the average weight increase in the four groups was 10.02 % (range 5.32–12.71 %).
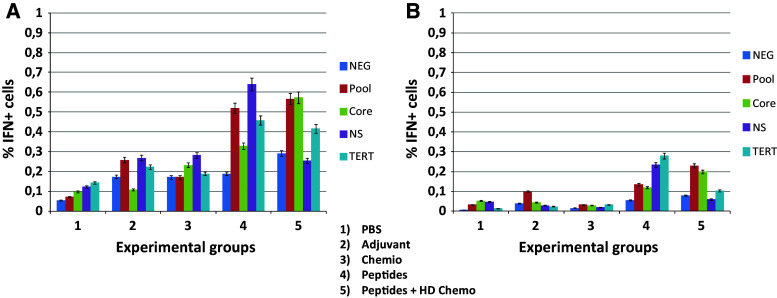
At the end of the protocol, animals were killed and immunological analyses were performed on resected spleens. Frequency of interferon-γ (IFN-γ)-secreting T cell populations was assessed in splenocytes pooled from each experimental group after ex vivo re-stimulation with the pool of vaccine peptides. The results showed that the percentage of IFN-γ-positive CD8+ T cells, and not of CD4+ T cells, was significantly higher in both experimental groups treated with weekly low- and high-dose metronomic chemotherapy (Fig. 1). Such result correlated with reduced percentage of Tregs in both low- and high-dose metronomic chemotherapy experimental groups (data not shown).
Fig. 1.
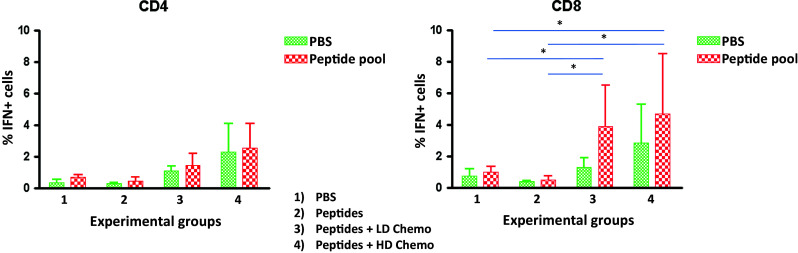
Evaluation of interferon-γ (IFN-γ)-secreting T cell populations after in vitro re-stimulation with peptide pool or PBS. At the end of the protocol combining the multi-peptide vaccine with weekly metronomic chemotherapy in C57BL/6 mice, single-cell splenocyte suspension cultures were obtained and re-stimulated ex vivo for 5 h. ICS for IFN-γ was evaluated in CD4+ and CD8+ T cell populations
Cancer vaccine and daily metronomic chemotherapy
Subsequently, a protocol was designed combining the multi-peptide vaccine, adjuvanted in Montanide and CpG, with daily administration of high-dose metronomic chemotherapy (Suppl. Fig. 3).
The general status of animals in the experimental groups was followed up during the whole protocol, and the weight of each animal was recorded on the day of each immunization. The data clearly indicated that, regardless the treatment, all animals showed a good general status. However, while animals in the control groups showed a progressive increase in the weight (13.2 % average weight increase), animals in the chemotherapy group showed a steady or decreasing weight (−2.3 % average weight variation). Interestingly, animals in the group treated with combination of chemotherapy + vaccine showed a 3.73 % average weight increase, suggesting a balancing effect played by the vaccine (Suppl. Fig. 4).
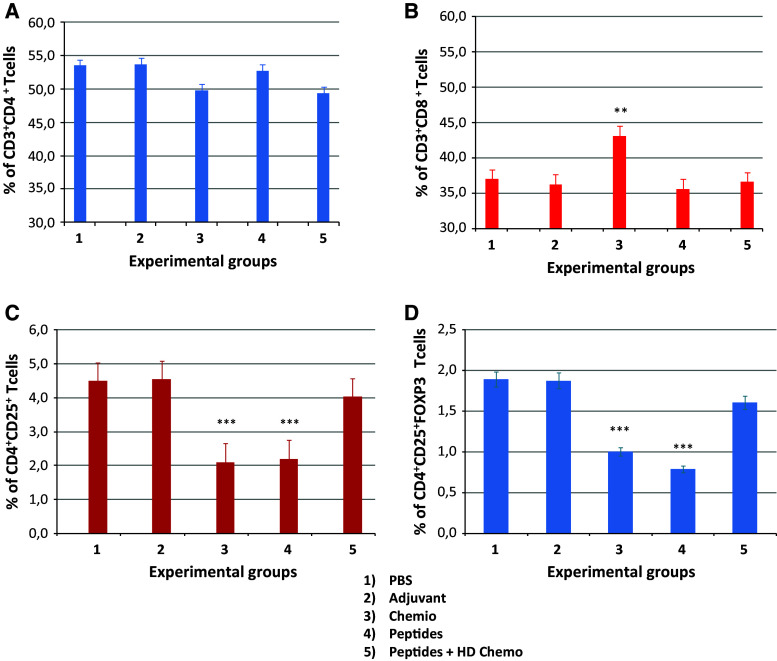
Daily metronomic chemotherapy induced a trend toward reducing CD4+ T cell percentage combined with a significant increase in the CD8+ T cell percentage, which was partially counterbalanced by vaccination (Fig. 2a, b). Nevertheless, the CD4+/CD8+ ratio did not drop below 1 which is the normal ratio in C57BL/6 mice at this age [57]. Furthermore, such ratio is considered as marker of an immune competence status in HIV-positive patients [58] (Suppl. Fig. 5).
Fig. 2.
Evaluation of T cell subpopulations at the end of the multi-peptide vaccine with daily metronomic chemotherapy protocol. At the end of the experimental protocol, in the spleens from each experimental groups were evaluated a CD3+CD4+, b CD3+CD8+, c CD4+CD25+ and d CD4+CD25+FoxP3+ T cell populations
Furthermore, a significant reduction in the percentage of CD4+CD25+ and CD4+CD25+FoxP3+ Treg population was observed in the spleens of the mice treated with either chemotherapy or vaccine. On the contrary, mice treated with the combination chemotherapy + vaccine did not show a significant reduction in Treg population (Fig. 2c, d).
In vivo effect of daily metronomic chemotherapy on antigen-specific immunity
Antigen-specific immune response was evaluated in splenocytes obtained from all animal groups 7 days after last immunization. Intracellular staining for IFN-γ production in CD8+ T cells was assessed by flow cytometry after ex vivo re-stimulation of splenocytes with the pool or individual vaccine peptides.
Results show an overall increase in IFN-γ-producing CD8+ T cells in mice treated with daily metronomic chemotherapy and multi-peptide vaccine (Fig. 3a). Such cells were specifically and significantly expanded by ex vivo re-stimulation only in splenocytes derived from animals treated with vaccine or the chemotherapy + vaccine combination. In particular, re-stimulation with either the peptide pool, the HCV core, or the TERT peptides induced a comparable >1.5-fold expansion of antigen-specific IFN-γ-producing CD8+ T cells in splenocytes of animals from both experimental groups (Fig. 3a). On the contrary, re-stimulation with the HCV NS3 peptide was effective in expanding antigen-specific IFN-γ-producing CD8+ T cells in splenocytes from animals treated with the vaccine only. The same ex vivo re-stimulation did not exert any expansion in splenocytes from the other three experimental groups (Fig. 3a).
Fig. 3.
Evaluation of interferon-γ (IFN-γ) a and IL-4 b producing CD8+ T cells after in vitro re-stimulation with either the peptide pool, the single peptides, or the PBS (Neg). At the end of the protocol combining the multi-peptide vaccine with daily metronomic chemotherapy, single-cell splenocyte suspension cultures were re-stimulated ex vivo for 5 h. ICS for IFN-γ and IL-4 were evaluated in the CD8+ T cell population
The percentage of interleukin-4 (IL-4)-producing CD8+ T cells showed a similar trend compared to IFN-γ, although at significantly lower scale, suggesting a Th1 skewing of immune response induced by the combinatorial strategy (Fig. 3b).
Multi-parametric analysis of immunological data
A multi-parametric analysis was performed to evaluate the agreement between the different immunological parameters identified in the present study. Percentage of CD8+, CD4+CD25+FoxP3+ T cells and their ratio were independently correlated with antigen-specific IFN-γ-producing CD8+ T cells in splenocytes of animals from the experimental group treated with combination of chemotherapy + vaccine. Results showed that the strongest correlation is found between IFN-γ-producing CD8+ T cells and Treg percentage as well as CD8+/Treg ratio (Table 1). As expected, Tregs are characterized by an inverse correlation, whereas the CD8+/Treg ratio is characterized by a direct correlation. Such results indicate that daily metronomic chemotherapy induces a Treg depletion strongly associated with an enhanced antigen-specific immune response.
Table 1.
Correlation coefficient (Pearson’s r) between CD8+, CD4+CD25+FoxP3+ T cells as well as their ratio and IFN-γ-producing CD8+ T cells, specific for each of the ex vivo re-stimulation epitopes
| Antigen-specific IFN-γ-producing CD8+ T cells | CD8+ | CD4+CD25+FoxP3+ | CD8+/Tregs |
|---|---|---|---|
| Pool | 0.823 | −0.997 | 0.984 |
| Core | 0.924 | −0.991 | 0.999 |
| NS3 | 0.867 | −0.999 | 0.994 |
| TERT | 0.806 | −0.993 | 0.977 |
Positive values indicate direct correlation; negative values indicate inverse correlation
Conclusion
In the current study, the effect of a strategy based on therapeutic peptide vaccine combined with a metronomic chemotherapy was evaluated in a mouse animal model. In particular, the therapeutic peptide vaccine was designed to target HCC and is a peptide cocktail containing the universal tumor antigen mTERT together with the HCV core and NS3 antigens. To our knowledge, such peptide combination has never been explored before for HCC, aiming at targeting both the replicating tumor cells and the underlying infectious causative agent. In the present study, the immunogenicity was evaluated in C57BL/6 animal model.
The combined metronomic chemotherapy was a mixture of CTX, PTX, and DTX administered in a weekly or daily schedule. CTX and PTX daily doses used in the tested combination have been previously reported to be independently effective in enhancing immune response to vaccine [48, 59]. On the contrary, DTX has been previously shown to increase antigen-specific T cell responses when combined to a vaccine with a schedule different from daily [60]. Enhancement of immune response to vaccine exerted by the combination of the three drugs, with the addition of doxorubicin, has been previously reported in a schedule characterized by single administration (i.e. 1 day before or 7 days after vaccination) [61]. The present study, therefore, represents the first observation of such chemotherapeutic combination strategy administered in a daily schedule.
The protocol combining the vaccine with weekly administered high- or low-dose metronomic chemotherapy showed no toxicity and induced an increased percentage of peptide-specific IFN-γ-positive CD8+ T cells (Fig. 2), directly correlated with a reduced percentage of Tregs. Likewise, the protocol combining the vaccine with daily administration of high-dose metronomic chemotherapy induced no toxicity. However, animals treated only with chemotherapy showed a steady or limited decreasing weight (−2.3 % average weight variation) compared with control groups. Interestingly, animals in the group treated with combination of chemotherapy + vaccine showed a 3.73 % average weight increase, indicating a partial balancing effect played by the vaccine and suggesting the absence of toxicity associated with the evaluated combinatorial strategy (Suppl. Fig. 4).
A significant reduction in the percentage of CD4+CD25+FoxP3+ Treg population was observed in the spleens of mice treated with either chemotherapy or vaccine, confirming results previously reported in the literature [48–50, 62–69].
Unexpectedly, such reduction was very limited in mice treated with the combination chemotherapy + vaccine compared with those in the PBS and adjuvant control groups (Fig. 2c, d). Explanation for such paradox effect is currently explored, evaluating how different parameters (i.e., administration routes and sites, doses) may impact on changes in the Treg population.
Immunogenicity of vaccination, alone or in combination with metronomic chemotherapy, was confirmed by identification of antigen-specific IFN-γ-producing CD8+ T cells in splenocytes of animals from both experimental groups. Re-stimulation with either the peptide pool or the individual epitopes induced, on average, an >1.5-fold expansion, suggesting that all three epitopes were able to elicit an immune response in immunized animals. Overall, the combination chemotherapy + vaccine treatment did not induce a significant increase in immunogenicity compared with vaccine only. Persistence of immune response in this experimental setting is not known, and consequently, timing of immune evaluation may have an impact on such result. Such aspect needs to be further evaluated.
Furthermore, immunological parameters identified in each animal within the combination chemotherapy + vaccine group show very strong correlation between IFN-γ-producing CD8+ T cells, specific for each of the ex vivo re-stimulation epitopes, and Treg percentage as well as CD8+/Treg ratio (Table 1). In particular, the strongest correlation coefficient was found with Treg percentage (negative coefficient), confirming the direct effect of reduction in Tregs on the CD8+ T cell response.
In conclusion, the present study describes a combination of novel multi-peptide formulation specific for HCV-related HCC with a novel multi-drug daily metronomic chemotherapy. Its efficacy is currently evaluated in a tumor challenge model to experimentally reproduce the tumor-associated immune-suppressive environment. Results indicate promising efficacy with high potential translational application of such combinatorial therapeutic strategy in human cancer vaccine clinical trials for HCC and beyond.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
The study was funded by European Union Framework Program 7 (EU FP7) Project Cancer Vaccine development for Hepatocellular Carcinoma—HEPAVAC (Grant Nr. 602893) and Italian Ministry of Health through Institutional “Ricerca Corrente”. Maria Tagliamonte and Annacarmen Petrizzo are HEPAVAC fellows.
Conflict of interest
The authors declare that they have no competing interests.
List of abbreviations
- ACK
Ammonium chloride potassium
- CTX
Cyclophosphamide
- DC
Dendritic cell
- DTX
Docetaxel
- HBV
Hepatitis B virus
- HCC
Hepatocellular carcinoma
- HCV
Hepatitis C virus
- HD Chemo
High-dose chemotherapy
- HED
Human equivalent dose
- HIV
Human immunodeficiency virus
- hTERT
Human telomerase reverse transcriptase
- ICS
Intracellular cytokine staining
- IFN-γ
Interferon gamma
- IL-4
Interleukin 4
- LD Chemo
Low-dose chemotherapy
- mTERT
Mouse telomerase reverse transcriptase
- Neg
Negative
- PBS
Phosphate-buffered saline
- PMA
Phorbol myristate acetate
- PTX
Paclitaxel
- RF
Radiofrequency
- s.c.
Subcutaneous
- TAA
Tumor-associated antigens
- TACE
Trans-arterial chemoembolization
- Tregs
Regulatory T cells
References
- 1.McGlynn KA, London WT. The global epidemiology of hepatocellular carcinoma: present and future. Clin Liver Dis. 2011;15:223–243. doi: 10.1016/j.cld.2011.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Franceschi S, Raza SA. Epidemiology and prevention of hepatocellular carcinoma. Cancer Lett. 2009;286:5–8. doi: 10.1016/j.canlet.2008.10.046. [DOI] [PubMed] [Google Scholar]
- 3.Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer. 2006;118:3030–3044. doi: 10.1002/ijc.21731. [DOI] [PubMed] [Google Scholar]
- 4.La Vecchia C, Negri E, Cavalieri DL, Franceschi S. Liver cirrhosis and the risk of primary liver cancer. Eur J Cancer Prev. 1998;7:315–320. doi: 10.1097/00008469-199808000-00007. [DOI] [PubMed] [Google Scholar]
- 5.Moriya K, Fujie H, Shintani Y, Yotsuyanagi H, Tsutsumi T, Ishibashi K, Matsuura Y, Kimura S, Miyamura T, Koike K. The core protein of hepatitis C virus induces hepatocellular carcinoma in transgenic mice. Nat Med. 1998;4:1065–1067. doi: 10.1038/2053. [DOI] [PubMed] [Google Scholar]
- 6.Anzola M. Hepatocellular carcinoma: role of hepatitis B and hepatitis C viruses proteins in hepatocarcinogenesis. J Viral Hepat. 2004;11:383–393. doi: 10.1111/j.1365-2893.2004.00521.x. [DOI] [PubMed] [Google Scholar]
- 7.Schwartz M, Dvorchik I, Roayaie S, Fiel MI, Finkelstein S, Marsh JW, Martignetti JA, Llovet JM. Liver transplantation for hepatocellular carcinoma: extension of indications based on molecular markers. J Hepatol. 2008;49:581–588. doi: 10.1016/j.jhep.2008.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mazzaferro V, Llovet JM, Miceli R, Bhoori S, Schiavo M, Mariani L, Camerini T, Roayaie S, Schwartz ME, Grazi GL, Adam R, Neuhaus P, Salizzoni M, Bruix J, Forner A, De CL, Cillo U, Burroughs AK, Troisi R, Rossi M, Gerunda GE, Lerut J, Belghiti J, Boin I, Gugenheim J, Rochling F, Van HB, Majno P. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: a retrospective, exploratory analysis. Lancet Oncol. 2009;10:35–43. doi: 10.1016/S1470-2045(08)70284-5. [DOI] [PubMed] [Google Scholar]
- 9.Llovet JM, Bruix J. Novel advancements in the management of hepatocellular carcinoma in 2008. J Hepatol. 2008;48(Suppl 1):S20–S37. doi: 10.1016/j.jhep.2008.01.022. [DOI] [PubMed] [Google Scholar]
- 10.Lencioni R, Crocetti L. Local-regional treatment of hepatocellular carcinoma. Radiology. 2012;262:43–58. doi: 10.1148/radiol.11110144. [DOI] [PubMed] [Google Scholar]
- 11.Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Haussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390. doi: 10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]
- 12.Reinisch W, Holub M, Katz A, Herneth A, Lichtenberger C, Schoniger-Hekele M, Waldhoer T, Oberhuber G, Ferenci P, Gangl A, Mueller C. Prospective pilot study of recombinant granulocyte-macrophage colony-stimulating factor and interferon-gamma in patients with inoperable hepatocellular carcinoma. J Immunother. 2002;25:489–499. doi: 10.1097/00002371-200211000-00005. [DOI] [PubMed] [Google Scholar]
- 13.Sangro B, Mazzolini G, Ruiz J, Herraiz M, Quiroga J, Herrero I, Benito A, Larrache J, Pueyo J, Subtil JC, Olague C, Sola J, Sadaba B, Lacasa C, Melero I, Qian C, Prieto J. Phase I trial of intratumoral injection of an adenovirus encoding interleukin-12 for advanced digestive tumors. J Clin Oncol. 2004;22:1389–1397. doi: 10.1200/JCO.2004.04.059. [DOI] [PubMed] [Google Scholar]
- 14.Shi M, Zhang B, Tang ZR, Lei ZY, Wang HF, Feng YY, Fan ZP, Xu DP, Wang FS. Autologous cytokine-induced killer cell therapy in clinical trial phase I is safe in patients with primary hepatocellular carcinoma. World J Gastroenterol. 2004;10:1146–1151. doi: 10.3748/wjg.v10.i8.1146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Takayama T, Sekine T, Makuuchi M, Yamasaki S, Kosuge T, Yamamoto J, Shimada K, Sakamoto M, Hirohashi S, Ohashi Y, Kakizoe T. Adoptive immunotherapy to lower postsurgical recurrence rates of hepatocellular carcinoma: a randomised trial. Lancet. 2000;356:802–807. doi: 10.1016/S0140-6736(00)02654-4. [DOI] [PubMed] [Google Scholar]
- 16.Butterfield LH, Ribas A, Dissette VB, Lee Y, Yang JQ, De la RP, Duran SD, Hernandez J, Seja E, Potter DM, McBride WH, Finn R, Glaspy JA, Economou JS. A phase I/II trial testing immunization of hepatocellular carcinoma patients with dendritic cells pulsed with four alpha-fetoprotein peptides. Clin Cancer Res. 2006;12:2817–2825. doi: 10.1158/1078-0432.CCR-05-2856. [DOI] [PubMed] [Google Scholar]
- 17.Butterfield LH, Ribas A, Meng WS, Dissette VB, Amarnani S, Vu HT, Seja E, Todd K, Glaspy JA, McBride WH, Economou JS. T-cell responses to HLA-A*0201 immunodominant peptides derived from alpha-fetoprotein in patients with hepatocellular cancer. Clin Cancer Res. 2003;9:5902–5908. [PubMed] [Google Scholar]
- 18.Lee WC, Wang HC, Hung CF, Huang PF, Lia CR, Chen MF. Vaccination of advanced hepatocellular carcinoma patients with tumor lysate-pulsed dendritic cells: a clinical trial. J Immunother. 2005;28:496–504. doi: 10.1097/01.cji.0000171291.72039.e2. [DOI] [PubMed] [Google Scholar]
- 19.Palmer DH, Midgley RS, Mirza N, Torr EE, Ahmed F, Steele JC, Steven NM, Kerr DJ, Young LS, Adams DH. A phase II study of adoptive immunotherapy using dendritic cells pulsed with tumor lysate in patients with hepatocellular carcinoma. Hepatology. 2009;49:124–132. doi: 10.1002/hep.22626. [DOI] [PubMed] [Google Scholar]
- 20.Buonaguro L, Petrizzo A, Tagliamonte M, Tornesello ML, Buonaguro FM. Challenges in cancer vaccine development for hepatocellular carcinoma. J Hepatol. 2013;59:897–903. doi: 10.1016/j.jhep.2013.05.031. [DOI] [PubMed] [Google Scholar]
- 21.Buonaguro L, Petrizzo A, Tornesello ML, Buonaguro FM. Translating tumor antigens into cancer vaccines. Clin Vaccine Immunol. 2011;18:23–34. doi: 10.1128/CVI.00286-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL, Coviello GM, Wright WE, Weinrich SL, Shay JW. Specific association of human telomerase activity with immortal cells and cancer. Science. 1994;266:2011–2015. doi: 10.1126/science.7605428. [DOI] [PubMed] [Google Scholar]
- 23.Hiyama E, Hiyama K. Telomerase as tumor marker. Cancer Lett. 2003;194:221–233. doi: 10.1016/S0304-3835(02)00709-7. [DOI] [PubMed] [Google Scholar]
- 24.Hastie ND, Dempster M, Dunlop MG, Thompson AM, Green DK, Allshire RC. Telomere reduction in human colorectal carcinoma and with ageing. Nature. 1990;346:866–868. doi: 10.1038/346866a0. [DOI] [PubMed] [Google Scholar]
- 25.Nault JC, Mallet M, Pilati C, Calderaro J, Bioulac-Sage P, Laurent C, Laurent A, Cherqui D, Balabaud C, Zucman-Rossi J. High frequency of telomerase reverse-transcriptase promoter somatic mutations in hepatocellular carcinoma and preneoplastic lesions. Nat Commun. 2013;4:2218. doi: 10.1038/ncomms3218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cevik D, Yildiz G, Ozturk M. Common telomerase reverse transcriptase promoter mutations in hepatocellular carcinomas from different geographical locations. World J Gastroenterol. 2015;21:311–317. doi: 10.3748/wjg.v21.i1.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Harley CB. Telomerase and cancer therapeutics. Nat Rev Cancer. 2008;8:167–179. doi: 10.1038/nrc2275. [DOI] [PubMed] [Google Scholar]
- 28.Shaw VE, Naisbitt DJ, Costello E, Greenhalf W, Park BK, Neoptolemos JP, Middleton GW. Current status of GV1001 and other telomerase vaccination strategies in the treatment of cancer. Expert Rev Vaccines. 2010;9:1007–1016. doi: 10.1586/erv.10.92. [DOI] [PubMed] [Google Scholar]
- 29.Beatty GL, Vonderheide RH. Telomerase as a universal tumor antigen for cancer vaccines. Expert Rev Vaccines. 2008;7:881–887. doi: 10.1586/14760584.7.7.881. [DOI] [PubMed] [Google Scholar]
- 30.Conforti A, Cipriani B, Peruzzi D, Dharmapuri S, Kandimalla ER, Agrawal S, Mori F, Ciliberto G, La MN, Aurisicchio L. A TLR9 agonist enhances therapeutic effects of telomerase genetic vaccine. Vaccine. 2010;28:3522–3530. doi: 10.1016/j.vaccine.2010.03.020. [DOI] [PubMed] [Google Scholar]
- 31.Dharmapuri S, Peruzzi D, Mennuni C, Calvaruso F, Giampaoli S, Barbato G, Kandimalla ER, Agrawal S, Scarselli E, Mesiti G, Ciliberto G, La MN, Aurisicchio L. Coadministration of telomerase genetic vaccine and a novel TLR9 agonist in nonhuman primates. Mol Ther. 2009;17:1804–1813. doi: 10.1038/mt.2009.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mennuni C, Ugel S, Mori F, Cipriani B, Iezzi M, Pannellini T, Lazzaro D, Ciliberto G, La MN, Zanovello P, Bronte V, Scarselli E. Preventive vaccination with telomerase controls tumor growth in genetically engineered and carcinogen-induced mouse models of cancer. Cancer Res. 2008;68:9865–9874. doi: 10.1158/0008-5472.CAN-08-1603. [DOI] [PubMed] [Google Scholar]
- 33.Peruzzi D, Gavazza A, Mesiti G, Lubas G, Scarselli E, Conforti A, Bendtsen C, Ciliberto G, La MN, Aurisicchio L. A vaccine targeting telomerase enhances survival of dogs affected by B-cell lymphoma. Mol Ther. 2010;18:1559–1567. doi: 10.1038/mt.2010.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wenandy L, Sorensen RB, Sengelov L, Svane IM, thor Straten P, Andersen MH. The immunogenicity of the hTERT540-548 peptide in cancer. Clin Cancer Res. 2008;14:4–7. doi: 10.1158/1078-0432.CCR-07-4590. [DOI] [PubMed] [Google Scholar]
- 35.Liao ZL, Tang XD, Lu MH, Wu YY, Cao YL, Fang DC, Yang SM, Guo H. Antitumor effect of new multiple antigen peptide based on HLA-A0201-restricted CTL epitopes of human telomerase reverse transcriptase (hTERT) Cancer Sci. 2012;103:1920–1928. doi: 10.1111/j.1349-7006.2012.02410.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Su Z, Dannull J, Yang BK, Dahm P, Coleman D, Yancey D, Sichi S, Niedzwiecki D, Boczkowski D, Gilboa E, Vieweg J. Telomerase mRNA-transfected dendritic cells stimulate antigen-specific CD8+ and CD4+ T cell responses in patients with metastatic prostate cancer. J Immunol. 2005;174:3798–3807. doi: 10.4049/jimmunol.174.6.3798. [DOI] [PubMed] [Google Scholar]
- 37.Rusakiewicz S, Dosset M, Mollier K, Souque P, Charneau P, Wain-Hobson S, Langlade-Demoyen P, Adotevi O. Immunogenicity of a recombinant lentiviral vector carrying human telomerase tumor antigen in HLA-B*0702 transgenic mice. Vaccine. 2010;28:6374–6381. doi: 10.1016/j.vaccine.2010.06.071. [DOI] [PubMed] [Google Scholar]
- 38.Adotevi O, Mollier K, Neuveut C, Dosset M, Ravel P, Fridman WH, Tartour E, Charneau P, Wain-Hobson S, Langlade-Demoyen P. Targeting human telomerase reverse transcriptase with recombinant lentivector is highly effective to stimulate antitumor CD8 T-cell immunity in vivo. Blood. 2010;115:3025–3032. doi: 10.1182/blood-2009-11-253641. [DOI] [PubMed] [Google Scholar]
- 39.Xue J, Zhu H, Chen Z. Therapeutic vaccines against hepatitis C virus. Infect Genet Evol. 2014;22:120–129. doi: 10.1016/j.meegid.2014.01.008. [DOI] [PubMed] [Google Scholar]
- 40.Wedemeyer H, He XS, Nascimbeni M, Davis AR, Greenberg HB, Hoofnagle JH, Liang TJ, Alter H, Rehermann B. Impaired effector function of hepatitis C virus-specific CD8+ T cells in chronic hepatitis C virus infection. J Immunol. 2002;169:3447–3458. doi: 10.4049/jimmunol.169.6.3447. [DOI] [PubMed] [Google Scholar]
- 41.Lauer GM, Barnes E, Lucas M, Timm J, Ouchi K, Kim AY, Day CL, Robbins GK, Casson DR, Reiser M, Dusheiko G, Allen TM, Chung RT, Walker BD, Klenerman P. High resolution analysis of cellular immune responses in resolved and persistent hepatitis C virus infection. Gastroenterology. 2004;127:924–936. doi: 10.1053/j.gastro.2004.06.015. [DOI] [PubMed] [Google Scholar]
- 42.Guglietta S, Garbuglia AR, Salichos L, Ruggeri L, Folgori A, Perrone MP, Camperio C, Mellace V, Maio G, Maio P, Capobianchi MR, Spada E, Gargano N, Scotta C, Piccolella E, Del PP. Impact of viral selected mutations on T cell mediated immunity in chronically evolving and self limiting acute HCV infection. Virology. 2009;386:398–406. doi: 10.1016/j.virol.2009.01.020. [DOI] [PubMed] [Google Scholar]
- 43.Urbani S, Uggeri J, Matsuura Y, Miyamura T, Penna A, Boni C, Ferrari C. Identification of immunodominant hepatitis C virus (HCV)-specific cytotoxic T-cell epitopes by stimulation with endogenously synthesized HCV antigens. Hepatology. 2001;33:1533–1543. doi: 10.1053/jhep.2001.25091. [DOI] [PubMed] [Google Scholar]
- 44.Cucchiarini M, Kammer AR, Grabscheid B, Diepolder HM, Gerlach TJ, Gruner N, Santantonio T, Reichen J, Pape GR, Cerny A. Vigorous peripheral blood cytotoxic T cell response during the acute phase of hepatitis C virus infection. Cell Immunol. 2000;203:111–123. doi: 10.1006/cimm.2000.1683. [DOI] [PubMed] [Google Scholar]
- 45.Liyanage UK, Moore TT, Joo HG, Tanaka Y, Herrmann V, Doherty G, Drebin JA, Strasberg SM, Eberlein TJ, Goedegebuure PS, Linehan DC. Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol. 2002;169:2756–2761. doi: 10.4049/jimmunol.169.5.2756. [DOI] [PubMed] [Google Scholar]
- 46.Zou W. Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol. 2006;6:295–307. doi: 10.1038/nri1806. [DOI] [PubMed] [Google Scholar]
- 47.Josefowicz SZ, Lu LF, Rudensky AY. Regulatory T cells: mechanisms of differentiation and function. Annu Rev Immunol. 2012;30:531–564. doi: 10.1146/annurev.immunol.25.022106.141623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Peng S, Lyford-Pike S, Akpeng B, Wu A, Hung CF, Hannaman D, Saunders JR, Wu TC, Pai SI. Low-dose cyclophosphamide administered as daily or single dose enhances the antitumor effects of a therapeutic HPV vaccine. Cancer Immunol Immunother. 2013;62:171–182. doi: 10.1007/s00262-012-1322-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ghiringhelli F, Menard C, Puig PE, Ladoire S, Roux S, Martin F, Solary E, Le CA, Zitvogel L, Chauffert B. Metronomic cyclophosphamide regimen selectively depletes CD4+CD25+ regulatory T cells and restores T and NK effector functions in end stage cancer patients. Cancer Immunol Immunother. 2007;56:641–648. doi: 10.1007/s00262-006-0225-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ghiringhelli F, Larmonier N, Schmitt E, Parcellier A, Cathelin D, Garrido C, Chauffert B, Solary E, Bonnotte B, Martin F. CD4+CD25+ regulatory T cells suppress tumor immunity but are sensitive to cyclophosphamide which allows immunotherapy of established tumors to be curative. Eur J Immunol. 2004;34:336–344. doi: 10.1002/eji.200324181. [DOI] [PubMed] [Google Scholar]
- 51.Wada S, Yoshimura K, Hipkiss EL, Harris TJ, Yen HR, Goldberg MV, Grosso JF, Getnet D, Demarzo AM, Netto GJ, Anders R, Pardoll DM, Drake CG. Cyclophosphamide augments antitumor immunity: studies in an autochthonous prostate cancer model. Cancer Res. 2009;69:4309–4318. doi: 10.1158/0008-5472.CAN-08-4102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lutsiak ME, Semnani RT, De PR, Kashmiri SV, Schlom J, Sabzevari H. Inhibition of CD4(+)25+ T regulatory cell function implicated in enhanced immune response by low-dose cyclophosphamide. Blood. 2005;105:2862–2868. doi: 10.1182/blood-2004-06-2410. [DOI] [PubMed] [Google Scholar]
- 53.Emens LA, Armstrong D, Biedrzycki B, Davidson N, Davis-Sproul J, Fetting J, Jaffee E, Onners B, Piantadosi S, Reilly RT, Stearns V, Tartakovsky I, Visvanathan K, Wolff A. A phase I vaccine safety and chemotherapy dose-finding trial of an allogeneic GM-CSF-secreting breast cancer vaccine given in a specifically timed sequence with immunomodulatory doses of cyclophosphamide and doxorubicin. Hum Gene Ther. 2004;15:313–337. doi: 10.1089/104303404322886165. [DOI] [PubMed] [Google Scholar]
- 54.Emens LA, Reilly RT, Jaffee EM. Augmenting the potency of breast cancer vaccines: combined modality immunotherapy. Breast Dis. 2004;20:13–24. doi: 10.3233/bd-2004-20103. [DOI] [PubMed] [Google Scholar]
- 55.Bass KK, Mastrangelo MJ. Immunopotentiation with low-dose cyclophosphamide in the active specific immunotherapy of cancer. Cancer Immunol Immunother. 1998;47:1–12. doi: 10.1007/s002620050498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J. 2008;22:659–661. doi: 10.1096/fj.07-9574LSF. [DOI] [PubMed] [Google Scholar]
- 57.Pinchuk LM, Filipov NM. Differential effects of age on circulating and splenic leukocyte populations in C57BL/6 and BALB/c male mice. Immun Ageing. 2008;5:1. doi: 10.1186/1742-4933-5-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Serrano-Villar S, Perez-Elias MJ, Dronda F, Casado JL, Moreno A, Royuela A, Perez-Molina JA, Sainz T, Navas E, Hermida JM, Quereda C, Moreno S. Increased risk of serious non-AIDS-related events in HIV-infected subjects on antiretroviral therapy associated with a low CD4/CD8 ratio. PLoS One. 2014;9:e85798. doi: 10.1371/journal.pone.0085798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chen CA, Ho CM, Chang MC, Sun WZ, Chen YL, Chiang YC, Syu MH, Hsieh CY, Cheng WF. Metronomic chemotherapy enhances antitumor effects of cancer vaccine by depleting regulatory T lymphocytes and inhibiting tumor angiogenesis. Mol Ther. 2010;18:1233–1243. doi: 10.1038/mt.2010.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Garnett CT, Schlom J, Hodge JW. Combination of docetaxel and recombinant vaccine enhances T-cell responses and antitumor activity: effects of docetaxel on immune enhancement. Clin Cancer Res. 2008;14:3536–3544. doi: 10.1158/1078-0432.CCR-07-4025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Machiels JP, Reilly RT, Emens LA, Ercolini AM, Lei RY, Weintraub D, Okoye FI, Jaffee EM. Cyclophosphamide, doxorubicin, and paclitaxel enhance the antitumor immune response of granulocyte/macrophage-colony stimulating factor-secreting whole-cell vaccines in HER-2/neu tolerized mice. Cancer Res. 2001;61:3689–3697. [PubMed] [Google Scholar]
- 62.Hueman MT, Stojadinovic A, Storrer CE, Foley RJ, Gurney JM, Shriver CD, Ponniah S, Peoples GE. Levels of circulating regulatory CD4+CD25+ T cells are decreased in breast cancer patients after vaccination with a HER2/neu peptide (E75) and GM-CSF vaccine. Breast Cancer Res Treat. 2006;98:17–29. doi: 10.1007/s10549-005-9108-5. [DOI] [PubMed] [Google Scholar]
- 63.Gates JD, Clifton GT, Benavides LC, Sears AK, Carmichael MG, Hueman MT, Holmes JP, Jama YH, Mursal M, Zacharia A, Ciano K, Khoo S, Stojadinovic A, Ponniah S, Peoples GE. Circulating regulatory T cells (CD4+CD25+FOXP3+) decrease in breast cancer patients after vaccination with a modified MHC class II HER2/neu (AE37) peptide. Vaccine. 2010;28:7476–7482. doi: 10.1016/j.vaccine.2010.09.029. [DOI] [PubMed] [Google Scholar]
- 64.Nakai N, Katoh N, Kitagawa T, Ueda E, Takenaka H, Kishimoto S. Immunoregulatory T cells in the peripheral blood of melanoma patients treated with melanoma antigen-pulsed mature monocyte-derived dendritic cell vaccination. J Dermatol Sci. 2009;54:31–37. doi: 10.1016/j.jdermsci.2008.11.007. [DOI] [PubMed] [Google Scholar]
- 65.Cipriani B, Fridman A, Bendtsen C, Dharmapuri S, Mennuni C, Pak I, Mesiti G, Forni G, Monaci P, Bagchi A, Ciliberto G, La MN, Scarselli E. Therapeutic vaccination halts disease progression in BALB-neuT mice: the amplitude of elicited immune response is predictive of vaccine efficacy. Hum Gene Ther. 2008;19:670–680. doi: 10.1089/hum.2007.127. [DOI] [PubMed] [Google Scholar]
- 66.Papewalis C, Wuttke M, Jacobs B, Domberg J, Willenberg H, Baehring T, Cupisti K, Raffel A, Chao L, Fenk R, Seissler J, Scherbaum WA, Schott M. Dendritic cell vaccination induces tumor epitope-specific Th1 immune response in medullary thyroid carcinoma. Horm Metab Res. 2008;40:108–116. doi: 10.1055/s-2007-1022565. [DOI] [PubMed] [Google Scholar]
- 67.Hus I, Schmitt M, Tabarkiewicz J, Radej S, Wojas K, Bojarska-Junak A, Schmitt A, Giannopoulos K, Dmoszynska A, Rolinski J. Vaccination of B-CLL patients with autologous dendritic cells can change the frequency of leukemia antigen-specific CD8+ T cells as well as CD4+CD25+FoxP3+ regulatory T cells toward an antileukemia response. Leukemia. 2008;22:1007–1017. doi: 10.1038/leu.2008.29. [DOI] [PubMed] [Google Scholar]
- 68.Hao S, Liu Y, Yuan J, Zhang X, He T, Wu X, Wei Y, Sun D, Xiang J. Novel exosome-targeted CD4+ T cell vaccine counteracting CD4+25+ regulatory T cell-mediated immune suppression and stimulating efficient central memory CD8+ CTL responses. J Immunol. 2007;179:2731–2740. doi: 10.4049/jimmunol.179.5.2731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ikezawa Y, Nakazawa M, Tamura C, Takahashi K, Minami M, Ikezawa Z. Cyclophosphamide decreases the number, percentage and the function of CD25+CD4+ regulatory T cells, which suppress induction of contact hypersensitivity. J Dermatol Sci. 2005;39:105–112. doi: 10.1016/j.jdermsci.2005.02.002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.