Abstract
Recently, several types of immunotherapies have been shown to induce encouraging clinical results, though in a restricted number of patients. Consequently, there is a need to identify immune biomarkers to select patients who will benefit from such therapies. Such predictive biomarkers may be also used as surrogates for overall survival (OS). We have recently found correlations between immunologic parameters and clinical outcome in prostate cancer patients who had been vaccinated with a HER-2/neu hybrid polypeptide vaccine (AE37) and received one booster 6 months post-primary vaccinations. Herein, we aimed to expand these retrospective analyses by studying the predictive impact of HLA-A*24 and HLA-DRB1*11 alleles, which are expressed at high frequencies among responders in our vaccinated patients, for clinical and immunological responses to AE37 vaccination. Our data show an increased OS of patients expressing the HLA-DRB1*11 or HLA-A*24 alleles, or both. Vaccine-induced immunological responses, measured as interferon γ (IFN-γ) responses in vitro or delayed-type hypersensitivity reactions in vivo, were also higher in these patients and inversely correlated with suppressor elements. Preexisting (i.e., before vaccinations with AE37) levels of vaccine-specific IFN-γ immunity and plasma TGF-β, among the HLA-A*24 and/or HLA-DRB1*11 positive patients, were strong indicators for immunological responses to AE37 treatment. These data suggest that HLA-DRB1*11 and HLA-A*24 are likely to be predictive factors for immunological and clinical responses to vaccination with AE37, though prospective validation in larger cohorts is needed.
Keywords: Prostate cancer, Cancer vaccine, Biomarkers, HLA, Immune response, Clinical response
Introduction
The relatively indolent nature even of progressed prostate cancer, compared with some other types of metastatic disease, allows time for an immune response to be generated, making it a good candidate for immune-based therapies [1]. Indeed, recent immunotherapy trials in prostate cancer have indicated that active immune responses can be achieved with therapeutic cancer vaccines, providing long-term clinical benefit for cancer patients [2, 3]. FDA-approved Sipuleucel-T, an autologous cellular-based immunotherapy which targets prostatic acid phosphatase, demonstrated its effectiveness in reducing the risk of death in metastatic castration-resistant prostate cancer [2]. Beyond Sipuleucel-T, several other active immunotherapies, involving targeting prostate-specific antigen (PSA) or prostate-specific membrane antigen (PSMA), are currently in development, making immunotherapy an established treatment approach for prostate cancer [1, 2, 4]. However, despite encouraging results from vaccination trials in prostate cancer, still there is a significant need to further improve clinical efficacy. The proper clinical design, as well as combining vaccines with other treatments, is essential for developing an effective prostate cancer vaccine strategy. Moreover, the methods of response evaluations in vaccination strategies should be standardized, and the best surrogate markers need to be chosen so as to optimize vaccine efficacy and select the best treatment schedules [5].
Major histocompatibility complex (MHC) products are important factors genetically restricting T cell responses to peptides also including those derived from tumor proteins [6]. Cytotoxic and helper T cells recognize processed antigenic peptides presented in the context of MHC class I or II molecules, respectively [6]. The polymorphism of MHC gene products directly affects the ability to bind specific peptide sequences, and, thus, it is likely that various HLA molecules differ in their ability to present tumor-specific endogenous antigens and to stimulate antitumor immune responses in individual patients [7]. Several investigators have demonstrated a relationship between susceptibility and resistance to various types of cancer including lung carcinoma, melanoma, head and neck carcinoma and epithelial ovarian cancer, and HLA phenotype [8–11], highlighting the potential of HLA alleles as prognostic factors and supporting the concept of immune surveillance in cancer patients. Moreover, various HLA alleles have been studied as biomarkers predicting responses to immune-based therapies in solid tumors as well as in haematological malignancies [12–14].
Tumors of the prostate in the majority of patients (>70 %) are positive for HER-2/neu (HER-2) expression, whereas HER-2 overexpression, found during the late stages of disease in castrate-resistant patients [15–17], makes HER-2 targeting an attractive treatment strategy for these patients. Work from our laboratory has shown that the HER-2(776–790) epitope (also named AE36) serves as a compelling tumor immunogenic epitope and that CD4+ T cells primed with the synthetic AE36 peptide help autologous cytotoxic T lymphocytes (CTL) for increased antitumor activity [18, 19]. We have shown that AE36 chemically linked to a tetra-peptide from the invariant chain of MHC class II molecules (Ii-key/AE36 hybrid peptide or AE37) induces more potent immunologic responses both in vitro and in vivo compared to the non-modified peptide AE36 [19]. In a phase I trial, we have immunized 29 prostate cancer patients with AE37 plus GM-CSF as an adjuvant. The vaccine was given in 6 monthly inoculations and was well tolerated with minimal toxicity. AE37-induced strong immunological responses in vivo (DTH) and in vitro (INF-γ production) could be measured in the majority of patients and were correlated with decreases in TGF-β plasma levels [20]. Long-term immunity to AE37 was still detectable 6 months post-vaccinations and could be considerably prolonged for an additional period of 36 months after a single AE37 booster inoculation [21]. Retrospective analyses revealed that preexisting IFN-γ immunity to the vaccine and plasma TGF-β levels correlated with DTH reactions and overall survival (OS) [22]. The data from our studies have demonstrated the important role of HER-2 as a therapeutic target in prostate cancer and implied a significant role for AE37 as a therapeutic vaccine that should be studied to prevent disease progression both at early and late stages (i.e., castrate-sensitive and castrate-resistant prostate cancer, respectively).
The goal of this study was to retrospectively assess the relevance of HLA molecules on the immunological responses to AE37 vaccination and on the long-term survival of vaccinated patients.
Materials and methods
Study design
A detailed description of the phase I clinical trial (EudraCT 2006-003299-37) design has been reported recently [20, 21]. In brief, androgen-dependent and androgen-independent prostate cancer patients with primary tumors expressing HER-2/neu + (score 1+ to 3+) and ECOG 0 or 1, were eligible. Exclusions criteria included patients with ECOG ≥ 2, active infection, severe cardiovascular comorbidity, acute/chronic HBV, HCV and HIV seropositivity, diagnosis of other primary solid/hematologic malignancy and any pathological comorbidity affecting patients’ compliance in the proposed clinical protocol. Patients received 6 monthly vaccinations of the AE37 peptide with GM-CSF as an immunoadjuvant. Of the 32 patients enrolled, 23 received one AE37 booster 6 months post-primary vaccine regimen, following the approval of St Savas Institutional Review Board and written informed consent. Analyses were performed by comparing data from immune monitoring performed at enrollment (R0) and 1 month after the 6th vaccination (R6). Long-term (LT) assessment was done at 6th month after the 6th vaccination. The booster injection was given on the same day of LT, and long-term booster (LTB) assessment was performed 1 month later.
HLA typing
The HLA typing was performed for HLA-A and HLA-DR loci by PCR–SSP (polymerase chain reaction—sequence-specific primers) using Olerup products (Olerup SSP, Saltsjobaden, Sweden), according to the manufacturer´s instructions.
Delayed-type hypersensitivity
Delayed-type hypersensitivity (DTH) reaction was tested at the indicated time points. The DTH reaction was assessed with 100 μg AE36, measured in two dimensions at 48 h and results are reported as an orthogonal mean [20]. Patients must have an induration of >5 mm post-vaccination to be considered as having developed a positive DTH reaction.
ELISPOT assay
The ELISPOT assay was performed as described previously [20]. In brief, freshly isolated PBMCs were cultured in quadruplicate at 2.5 × 105 cells per well, with 10 μg/ml of AE36 or medium alone (negative control) in precoated IFN-γ ELISPOT plates (Mabtech AB, Sweden). The plates were incubated for 40 h and developed as described by the manufacturer. Spots were enumerated using an ELISPOT analyzer (A.EL.VIS GmbH). Data are presented as specific spots (experimental spots minus negative control spots) per 106 PBMCs.
Plasma human TGF-β determination
Plasma (heparine) samples were collected at the indicated time points and stored until analysis at −70 °C. Commercially available immunoassay kits were used for measurements of the activated (immunoreactive form) TGF-β1, after acid treatment of plasma, according to the manufacturer’s instructions (human TGF-β1 instant enzyme-linked immunosorbent assay, Bender MedSystems GmbH).
Phenotypic characterization of Tregs
This was performed by using anti-CD45-PerCP, CD4-APC, CD25-FITC and CD127-PE (all purchased from BD Biosciences) as previously reported [20]. Tregs were defined as CD4 + CD25brightCD127low/neg.
Assessment of IDO activity
IDO activity was estimated by quantifying tryptophan (Trp) and its metabolite kynurenine (Kyn) as previously described [21]. Patient’s plasmas were tested in duplicates, and samples of three donors without cancer were also systematically included. Results are expressed as Kyn μM/Trp mM ratios (mean of duplicate measurements) [23].
Statistical analysis
GraphPad Prism version 5 software was used for statistical analysis of data. The nonparametric Mann–Whitney t test with a 95 % confidence interval was used for statistical evaluation of patient groups at different time points. Kaplan–Meier curves and log rank test were used for the evaluation of OS. Fisher’s exact test or Chi-square test was used for comparison of percent distributions among group profiles. For t test and survival analyses differences were considered significant when p value was ≤0.05. For Fisher’s exact test or Chi-square test, statistically significant differences were further evaluated after Bonferroni correction of the cutoff (α/n, where n = 7: IFN-γ, DTH, TGF-β, Tregs, IDO, HLA-A*24 and HLA-DRB1*11) and were considered highly statistically significant for p values ≤0.0071.
Results
Association between expression of two HLA antigens and clinical response to the AE37 vaccine
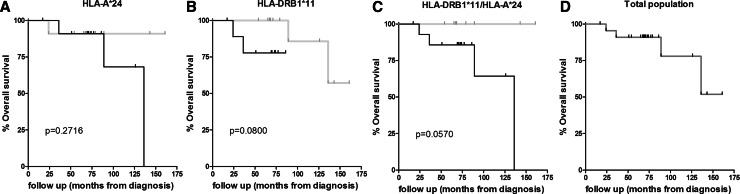
A total of 23 patients, who had received one booster of the AE37 hybrid peptide vaccine, 6 months post-primary vaccinations, were included in the analysis. Twelve of these patients developed increased immunity in response to AE37 vaccinations and were classified as high responders (HR) (Table 1 and [20, 21]). Interestingly, of the 12 HR patients, nine (75 %) were HLA-DRB1*11 positive (+) and seven (58 %) were HLA-A*24+ (Table 1). We have stratified our patients based on the expression of these HLA alleles and calculated the median OS (mOS) starting from the date of diagnosis (median follow-up 71 months; range, 17 to 161). Figure 1 shows that, in contrast to HLA-A*24, HLA-DRB1*11 was strongly associated with OS (Fig. 1a, b). When patients were categorized as having expressed HLA-A*24 and HLA-DRB1*11 vs neither HLA-A*24 nor HLA-DRB1*11, differences in OS reached almost statistical significance (Fig. 1c). Finally, there were no differences when mOS was analyzed for each group separately vs the total population.
Table 1.
Responder status, HER-2 status, and HLA typing of prostate cancer patients receiving the AE37 vaccine
| Patient # | Time points | HLA typing | HER-2 status | Responder status | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| R1 | R2 | R3 | R4 | R5 | R6 | LT | LTB | ||||
| 08 | a+++ | a+ | a+ | a++ | a+ | a+/−b | a +++/−b | a−/++b | A*24 A*68 DRB1*11 DRB1*14 | 1+ | R |
| 10 | ± | ± | − | + | + | +/++++ | ++/+ | −/+++ | A*01 A*30 DRB1*04 DRB1*11 | 1+ | HR |
| 11 | + | +++ | ++ | + | − | +/− | +/− | −/− | A*02 A*32 DRB1*04 DRB1*07 | 2+ | R |
| 12 | ++ | + | ++ | − | ++ | ++/− | +/+++ | +/++ | A*26 DRB1*11 | 1+ | HR |
| 13 | − | ± | − | − | −/− | −/− | −/++ | A*02 A*32 DRB1*04 | 3+ | NR | |
| 14 | − | + | + | − | + | −/+ | ++/++ | ++/+++ | A*24 A*33 DRB1*03 DRB1*11 | 1+ | HR |
| 15 | + | − | + | ++ | ++ | ++/+++ | ++/+++ | ++/+++ | A*24 A*29 DRB1*11 | 2+ | HR |
| 16 | ++ | ++ | ++ | ++ | ++ | ++/+++ | +++/+++ | ++/+++ | A*24 A*32 DRB1*11 DRB1*15 | 1+ | HR |
| 17 | − | ± | ++ | − | + | ++/− | +/− | +/+ | A*01 DRB1*11 DRB1*13 | 1+ | R |
| 19 | − | − | − | + | − | −/− | +/− | +/− | A*02 A*24 DRB1*01 DRB1*09 | 2+ | NR |
| 20 | + | + | ++ | ++ | + | ++/+++ | +/+++ | ++/+++ | A*02 A*03 DRB1*04 DRB1*12 | 2+ | HR |
| 21 | ++++ | ++ | +++ | + | + | −/++ | +++/+++ | ++/+++ | A*02 A*03 DRB1*15 DRB1*16 | 1+ | HR |
| 22 | + | − | − | − | − | +/++ | −/++ | −/++ | A*02 A*30 DRB1*15 | 1+ | R |
| 23 | − | +++ | − | − | − | −/+ | −/+ | −/+ | A*02 A*11 DRB1*04 DRB1*16 | 3+ | R |
| 24 | − | +++ | + | ++ | ++ | +/− | +/− | +/− | A*01 A*03 DRB1*11 DRB1*16 | 2+ | R |
| 25 | − | ++ | − | + | + | −/− | −/+ | −/+ | A*24 A*32 DRB1*04 DRB1*11 | 3+ | R |
| 26 | +++ | + | ++ | − | ++ | −/++ | +/+++ | −/++ | A*03 A*11 DRB1*01 DRB1*16 | 2+ | R |
| 27 | + | ++ | + | + | + | ++/++ | −/+ | +/+ | A*02 A*03 DRB1*11 DRB1*15 | 3+ | HR |
| 28 | − | − | − | ++ | + | −/− | +/+++ | ++/+++ | A*02 A*24 DRB1*16 | 3+ | HR |
| 29 | − | − | − | + | + | −/++ | −/++ | +++/++ | A*03 A*24 DRB1*11 DRB1*13 | 1+ | HR |
| 30 | ++ | + | + | + | + | −/+++ | −/++ | +/+++ | A*24 DRB1*03 DRB1*11 | 2+ | HR |
| 31 | − | ++ | + | ++ | + | ++/+++ | −/++ | +/+++ | A*02 A*24 DRB1*01 DRB1*11 | 2+ | HR |
| 32 | − | − | − | − | − | ++/− | NT | NT | A*24 A*32 DRB1*07 DRB1*10 | 1+ | ? |
R (responders): those who developed positive ELISPOT (i.e., SI > 1.55) (i) at 2 sequential time points between R1 and R5 and in at least one of R6, LT, or LTB or (ii) positive DTH in at least two of the R6, LT, and LTB
HR (high responders): as for “responders” plus (ii) positive DTH in at least two of the R6, LT, and LTB
NT not tested
a SI for IFN-γ-based ELISPOT at the indicated time points (no. of experimental spots/no. of background spots): 1.45–1.55: ±; >1.55–2: +; >2–4 : ++; >4–8: +++; >8: ++++
bDTH reactions: 5–10 mm: +; >10–20 mm: ++; >20–50 mm: +++; >50 mm: ++++
Fig. 1.
Association between clinical response and HLA expression in AE37-vaccinated patients. Kaplan–Meier analysis of time from primary diagnosis (a–d) by HLA expression, as indicated. p values for d versus a, versus b, versus c: 0.2716, 0.2482, and 0.1038, respectively. Correlation between expression of HLA antigens with clinical outcome. Kaplan–Meier plots comparing the overall survival of a HLA-A*24, b HLA-DRB1*11, and c HLA-DRB1*11/HLA-A*24 groups of patients and d total population with clinical outcome from disease diagnosis
Immunological responses in vitro and in vivo
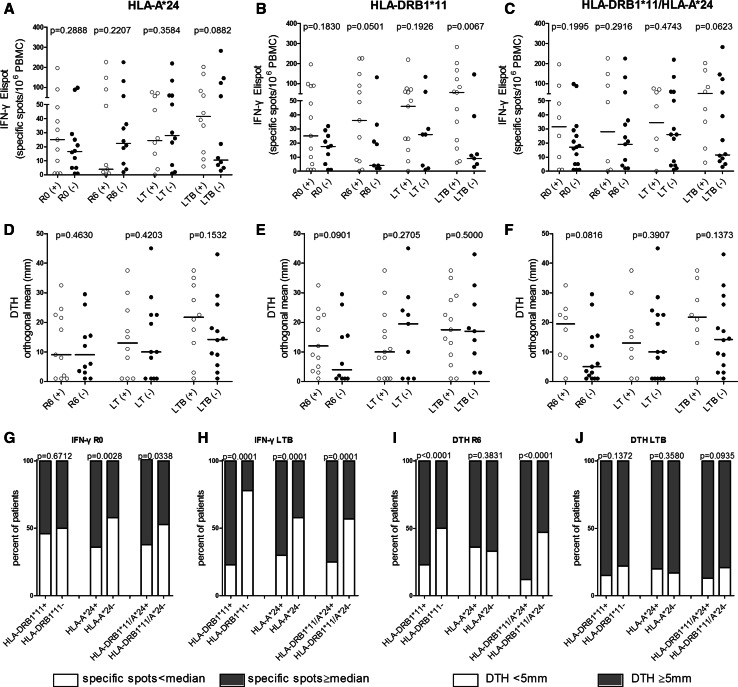
To understand how immunity develops during and post-vaccination as well as after booster, we first analyzed the levels of in vitro immunity in the three groups of patients, which was measured as frequencies of IFN-γ producing PBMCs in response to AE36 in the ELISPOT assay [20–22]. As shown in Fig. 2a, there was a strong trend for increased IFN-γ responses during LTB in HLA-A*24+ patients compared to HLA-A*24− ones. Such responses measured post-booster were even stronger among HLA-DRB1*11+ patients and highly significant when compared to those developed by HLA-DRB1*11− patients (Fig. 2b). Statistical significance was also reached when IFN-γ responses to AE36 were analyzed in HLA-DRB1*11+ versus HLA-DRB1*11− patients during R6 (i.e., 1-month post-primary vaccinations) (Fig. 2b). The difference in the levels of IFN-γ immunity was also profound in patients co-expressing HLA-DRB1*11 plus HLA-A*24 versus those being negative for both alleles, reaching levels close to statistical significance during LTB (Fig. 2c). There was statistically no significant increased preexistent IFN immunity to AE36, in groups of patients carrying the above HLA alleles, which may be indicative for “boosted” endogenous immunity.
Fig. 2.
Patients’ preexisting and AE37-induced in vitro and in vivo immunity in patients expressing (+) (open circles) or not expressing (−) (filled circles) the indicated HLA alleles (IFN-γ ELISPOT and DTH results are presented, respectively, in a and d for HLA-A*24, b and e for HLA-DRB1*11 and c and f for both allele expression, HLA-DRB1*11/HLA-A*24). R0: preexisting immunological responses; R6, LT, LTB: vaccine-induced immunity; see also “Materials and Methods”. Horizontal lines, median responses. g–j immune profile of patients; percentage of patients with preexisting or AE37-induced immunity above or below the respective median values (17 specific spots/106 PBMCs for IFN-γ immunity and 5 mm for DTH reactions)
In addition, interaction between treatment and HLA-DRB1*11 expression seemed to correlate with the intensity of DTH reactions, given the trends for increased responses observed among patients expressing HLA-DRB1*11 or co-expressing HLA-DRB1*11 plus HLA-A*24 (in both cases at R6; Fig. 2e, f, respectively).
We next estimated the proportions of patients in each HLA group according to (1) their preexisting endogenous IFN-γ immunity to AE36 which was measured before vaccinations at R0 and (2) their induced IFN-γ immunity after AE37 injections, at LTB, (including all six inoculations during primary vaccinations and the single booster). As a cutoff, we considered the 17 specific spots/106 PBMCs, which was the median IFN-γ immunity at R0 for the 23 patients who received the AE37 booster, as determined by Perez et al. [20–22]. Patients bearing either the HLA-A*24 or HLA-DRB1*11/HLA-A*24 alleles experienced statistically increased preexistent IFN-γ immunity to AE36 vs patients lacking these alleles (Fig. 2g) which was further enhanced at LTB (Fig. 2h). We could, however, emphasize that this result should not associate a link between preexistent immunity and outcome of vaccine-induced immunity, as HLA-DRB1*11+ patients developed strong AE36-specific IFN-γ immune responses only during vaccinations (Fig. 2h vs. g). We also made estimations for the proportions of patients who responded with increased DTH reactions post-primary AE37 vaccinations (R6) as well as post-booster (LTB). The cutoff for positive DTH responses was 5 mm [20–22]. As depicted in Fig. 2i, the vast majority of HLA-DRB1*11+ and of HLA-DRB1*11+/HLA-A*24+ patients developed positive DTH reactions at R6 displaying highly significant differences compared to the respective allele-negative patients. DTH responses after AE37 booster were almost similar in all patients (Fig. 2j).
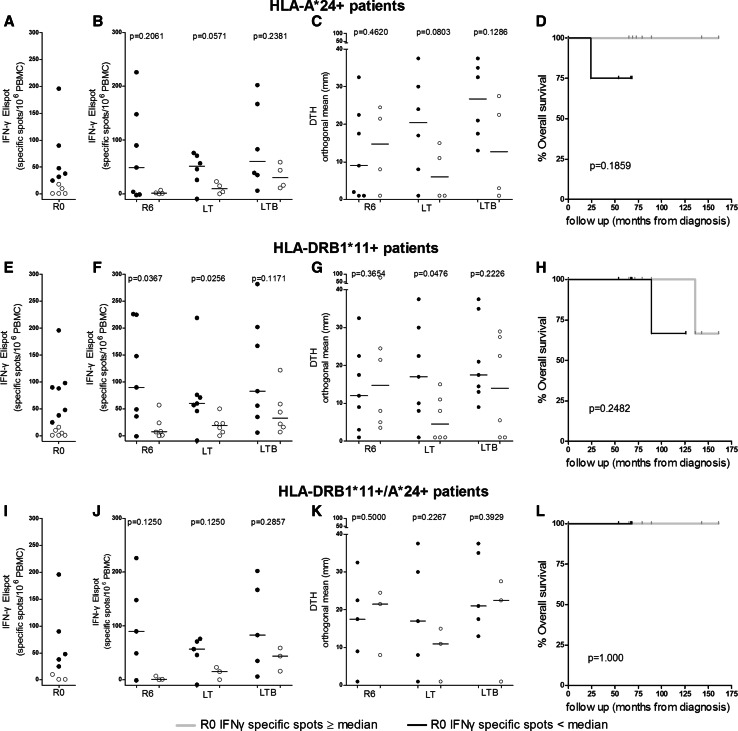
Levels of preexisting immunity dictate vaccine-induced immunological responses in vitro and in vivo, and clinical efficacy
In recent years, accumulating evidence suggests that hosts’ endogenous immune activation against autologous tumor is associated with patient survival [24]. In line with this, we next investigated if and to which extent preexisting IFN-γ immunity dictates the magnitude of in vitro as well as in vivo AE37-induced immunological responses in patients expressing HLA-DRB1*11, HLA-A*24, or both alleles. We considered patients with preexisting or no preexisting immunity those with values above/equal vs below the median IFN-γ value, respectively, which was determined by Perez et al. [21] (shown in Fig. 3a, e and i for each group). The group of HLA-A*24+ patients, who had preexisting IFN-γ immunity to AE36, exhibited higher vaccine-induced median IFN-γ responses (which almost reached significance at LT) over those with no preexisting IFN-γ immunity (Fig. 3b). Patients with IFN-γ preexisting immunity also developed higher DTH reactions with strong trend to significance at LT (Fig. 3c). A similar correlation was also observed between preexisting and vaccine-induced immunity among HLA-DRB1*11+ patients: preexisting IFN-γ immunity to AE36 could be boosted and maintained during vaccinations (reaching significance at R6 and LT) (Fig. 3f) and was also correlated with significantly higher DTH reactions measured during LT (Fig. 3g). This positive correlation between increased preexisting IFN-γ immunity to AE36 with vaccine-augmented IFN-γ and DTH responses was less pronounced when examined in patients co-expressing these two alleles (Fig. 3j, k).
Fig. 3.
Preexisting immunity predicts the magnitude of AE37 vaccine-induced specific T-cell responses and clinical efficacy. Patients were analyzed based on the expression of the indicated HLA alleles. In (a), (e), and (i), filled and open circles indicate patients with preexisting immunity above and below median responses, respectively. Vaccine-induced immunity at the indicated time points for these groups of patients is shown in (b, c), (f, g), and (j, k). Clinical responses for the same groups (weak lines, patients with preexisting immunity; bold lines, patients with no preexisting immunity) are shown in (d), (h) and (l)
There was also a trend for increased OS in HLA-A*24+ (Fig. 3d) and HLA-DRB1*11+ (Fig. 3h) patients having preexisting INF-γ immunity to the AE36 peptide estimated from first diagnosis with 95 and 90 % reduction in the relative risk for death (RRRD). No difference could be found among patients co-expressing both alleles, since no disease-related deaths were observed for this group.
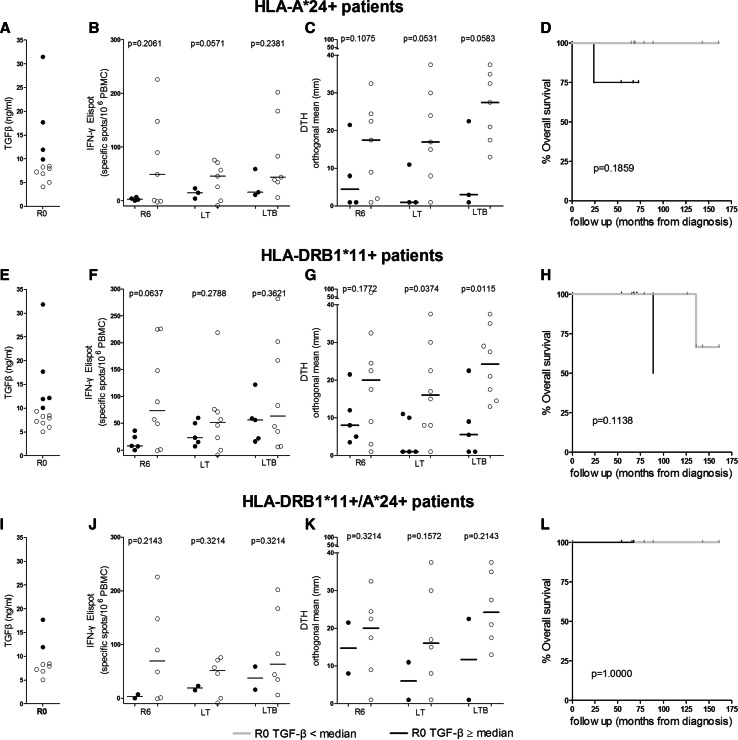
Preexisting TGF-β levels regulate vaccine-induced immunity in vitro and in vivo and clinical efficacy
We have also categorized our patients in two groups, based on their preexisting TGF-β plasma levels at R0 (i.e., pre-vaccination) as determined by Perez et al. [21]: those with values above/equal or below median (9.87 ng/ml; high or low TGF-β producers, respectively) (Fig. 4a, e and i). As shown in Fig. 4b, among HLA-A*24+ patients, low TGF-β producers had increased vaccine-induced frequencies of IFN-γ-producing T cells against the native AE36 peptide compared with high TGF-β producers, reaching levels almost close to significance at LT. Low TGF-β producers also developed robust DTH reactions reaching nearly significant differences at LT and LTB compared to high TGF-β producers (Fig. 4c). This inverse correlation was also observed when analyzing pre-vaccination TGF-β levels vs vaccine-induced immunity in HLA-DRB1*11+ patients: there was a strong trend for higher IFN-γ in the group of low TGF-β producers vs the high TGF-β producers at R6 (Fig. 4f), whereas the differences in DTH reactions among both groups became significant at LT and LTB (Fig. 4g). When patients were analyzed based on co-expression of HLA-DRB1*11 plus HLA-A*24, analogous immune profiles were observed (Fig. 4j, k). HLA-A*24+- and HLA-DRB1*11+-vaccinated patients with low levels of preexisting plasma TGF-β had RRRD of 16 and 33 %, respectively (Fig. 4d, h). There was no difference in OS among patients co-expressing both alleles (Fig. 4l).
Fig. 4.
Preexisting plasma levels of TGF-β predict the magnitude of AE37 vaccine-induced specific T-cell responses and clinical efficacy. See also legend to Fig. 3. Correlation between preexisting TGF-β levels and vaccine-induced immunity. a HLA-A*24+, e HLA-DRB1*11+, and i HLA-DRB1*11+/A*24+ patients with increased preexisting (filled circles) and low preexisting TGF-β levels (open circles). Levels of IFN-γ immunity (b, f, j) and DTH reactions c, g, k at the indicated time points for HLA-A*24, HLA- DRB1*11, and HLA-DRB1*11+/A*24+ patients, respectively. Correlation of TGF-β levels with OS for each group of patients from diagnosis (d, h, l)
Assessment of suppressor circuits
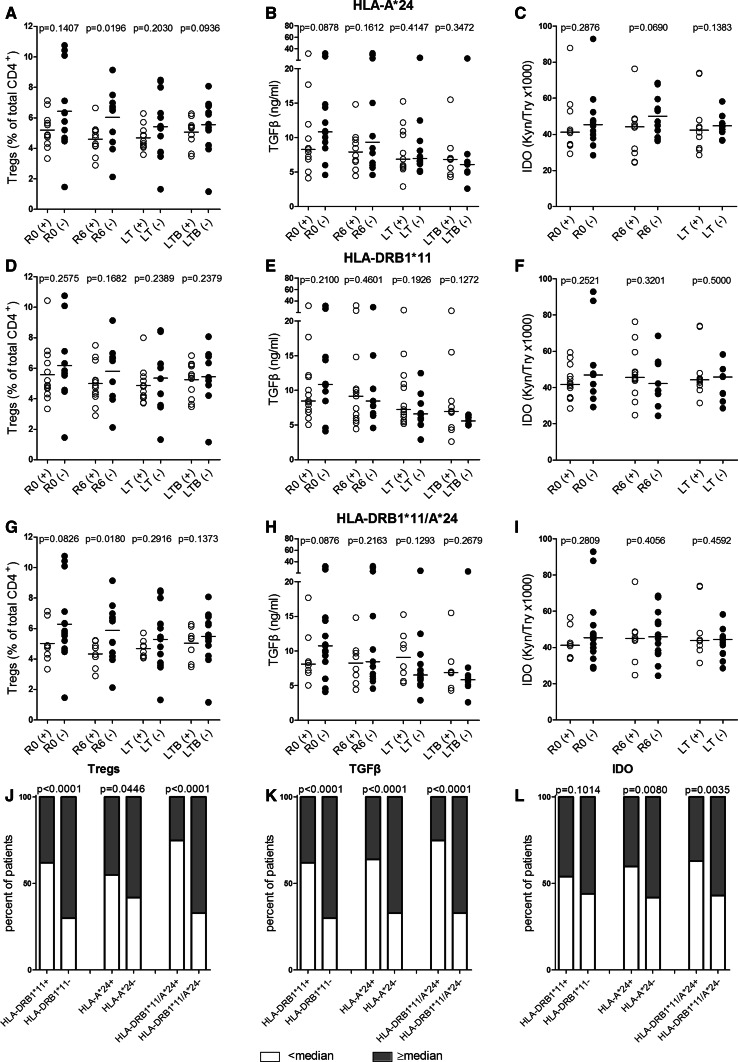
The association between the above HLA molecules, with preexisting immunity, and immunological responses to vaccination with AE37 was paralleled with decreased suppressor elements, including circulating Tregs, plasma TGF-β, and IDO activity. Analyses made in HLA-A*24+ patients revealed significantly lower numbers of Tregs measured at R6 as compared to HLA-A*24− individuals (Fig. 5a), and trends for decreased Tregs at LTB (Fig. 5a), for TGF-β at R0 (Fig. 5b), and for IDO activity at R6 (Fig. 5c).
Fig. 5.
Association of HLA with pre- and post-vaccination levels of suppressor elements. Levels of Tregs, TGF-β, and IDO activity were assessed in the indicated groups of patients and are shown in a–i (see also legends to previous Figs.). Suppressor profile is shown in j–l, as percentage of patients expressing the indicated suppressor elements above or below their respective median values at R0. Assessment of suppressive circuits. Circulating Tregs (a, d, g), TGF-β plasma levels (b, e, h) and IDO activity as the kynurenine-to-tryptophan ratio (Kyn/Trp*1000) (c, f, i) determined at the indicated time points for all groups of patients. Proportions of patients of each group according to pre-vaccination Treg frequency (j), TGF-β plasma levels (k), and IDO activity (l)
Almost equal levels for these suppressor elements could be detected in HLA-DRB1*11+ versus HLA-DRB1*11− patients at R0 and throughout vaccinations (Fig. 5d–f).
Finally, HLA-DRB1*11/HLA-A*24 expression was associated with trends of reduced Tregs (Fig. 5g) and TGF-β (Fig. 5h) at pre-vaccination and statistically significant decreases for Tregs at R6 (Fig. 5g), but with no differences in the levels of IDO activity (Fig. 5i).
The preexisting suppressor profile of groups analyzed above is shown in Fig. 5j–l. The proportion of patients with Treg frequencies above the cutoff median value of 5.53 %, as determined by Perez et al. [21, 22], was significantly lower among single (HLA-A*24 or HLA-DRB1*11) or double (HLA-DRB1*11 plus HLA-A*24) allele carriers versus those not expressing the respective allele(s) (Fig. 5j). Similarly, the same HLA allele carriers expressed significantly lower TGF-β levels (cutoff median value for TGF-β: 9.87 ng/ml [21, 22]) (Fig. 5k). The percentages of patients with IDO activity levels below the median Kyn/Trp cutoff ratio of 42.15 [21, 22] were significantly higher in patients expressing HLA-A*24 or both alleles (Fig. 5l).
Discussion
As strategies are being currently pursued for the establishment of immune-based cancer therapies, identification of host genetic factors related to the generation of protective antitumor immune responses is fundamental for understanding mechanisms underlying cancer-induced immunity and rational vaccine development. HLA alleles act as genetic restriction elements regulating T cell activation upon presentation of tumor peptide by professional APCs. Hence, the efficacy of a peptide-based therapeutic cancer vaccine may depend not only on its antigenicity per se, but also on the type of the restricting HLA allele, which determines the avidity of the trimolecular complex, consisting of HLA-peptide-TCR [25]. Such restrictions may limit the ability of an individual antigen to function as a T cell immunogen in the context of different HLA allele restriction elements. HLA molecules on the surface of host APCs present peptides from endogenous proteins or from exogenous processed antigens to CD8+ and CD4+ T lymphocytes [25]. Therefore, it is logical to postulate that individuals expressing different HLA class antigens may respond differently to tumor proteins or polypeptide vaccines composed of various immunogenic epitopes. Consequently, the evidence that subgroups of patients expressing certain HLA molecules have delayed tumor relapse might be explained by an immunological control.
In this study, we have retrospectively monitored our phase I AE37 vaccine trial through an assessment of HLA association with respect to both, immune and clinical responses to treatment. We have shown that both clinical and immunological responses to vaccination are more likely to be found among patients expressing HLA-DRB1*11 and/or HLA-A*24. By analyzing these groups of patients, we detected a frequent (and in some instances significant) vaccine-induced IFN-γ and DTH immunity in patients with preexisting IFN-γ immunity to AE36, which was correlated with clinical benefit. In contrast, preexisting TGF-β levels in the same patients were inversely correlated with vaccine-induced immunological responses both in vitro and in vivo as well as with clinical outcome. The AE37 vaccine represents an Ii-key modification of the immunogenic epitope of HER-2(776-790) (AE36) which, in this hybrid form, can be recognized in an accelerated and improved fashion by CD4+ T cells and also shows highly promiscuous binding to a series of MHC class II alleles with various affinities, as tested in binding and functional assays [26–28]. Moreover, according to prediction algorithms (SYFPEITHI, www.uni-tuebingen.de/uni/kxi/database.html; [7]), AE36 encompasses binding motifs for several MHC class I molecules, thus triggering not only CD4+ but also CD8+ T cells of different HLA restriction. To this end, we could recently demonstrate in our vaccinated prostate cancer patients increased percentages of both CD4+ and CD8+ T cells producing IFN-γ upon in vitro sensitization with AE36 [21, 22]. Thus, although the two alleles, HLA-A*24 and HLA-DRB1*11, defined herein correlate with both preexisting and vaccine-induced immunologic responses to AE36, they might be only a part of the total number of HLA alleles which regulate these types of immunity. In line with this, we have found, among our responders, patients who did not express either of these alleles (Table 1). In addition, since CTL responses to exogenous antigens are induced through cross-presentation by professional APC, another factor that may contribute to the selection of candidate responder MHC class I alleles, besides binding motifs, is the particular efficiency of cross-presentation of relevant CTL epitopes, in the context of the appropriate HLA class I allele [29].
In cancer, there are only some sporadic reports to demonstrate an HLA association with induction of immunity to vaccination. Vaccinating melanoma patients with melacin revealed an association between antitumor efficacy and host expression of two or three of the following class I antigens: HLA-A2, HLA-A28, HLA-B44, HLA-B45, and HLA-Cw3, with HLA phenotypes A2 and Cw3 showing the strongest association with clinical response [14]. Hoon et al. [30] reported a benefit for whole-cell vaccine treatment in HLA-A25-positive patients, as well as a favorable outcome of patients whose HLA expression matched the HLA phenotype expressed by the three melanoma cells lines that composed the vaccine (i.e., HLA-A3/11 and HLA-B7/44). Recombinant tumor antigens including several cancer testis antigens (e.g., MAGE-A3) but also differentiated and overexpressed antigens (HER-2) are presently among the most promising candidate cancer vaccines under evaluation [31–33]. However, the HLA class I restriction of total CTL responses induced by these vaccines have not been assessed systematically in most cases. Compelling evidence for the role of HLA alleles in regulating vaccine-induced immunological responses was provided by Bioley et al. [34], who demonstrated that cancer patients receiving injections with NY-ESO-1 developed CTL responses if they were expressing Cw3 or B35 alleles.
Our study results underscore the importance of type I adaptive immune response development as a positive prognostic factor. Type I CD4+ T cells release IFN-γ and support both the induction and effector phases of antigen-specific CD8+ T cells [35, 36] which are directly linked to tumor cell death [29]. One of the most common methods to assess type I adaptive immunity is the measurement of tumor-specific immune responses by IFN-γ ELISPOT and DTH [37]. Several recent studies, in a number of tumor types, have reported an association with immune responses measured by IFN-γ ELISPOT or DTH and improved clinical outcome after immune modulation [37]. Recently, we have demonstrated a trend in OS benefit for patients who had developed positive IFN-γ ELISPOT and DTH during vaccinations with AE37 [21, 22]. A similar trend was noted in patients who had preexisting IFN-γ ELISPOT responses. In the same reports, we have demonstrated an inverse correlation between preexisting TGF-β levels throughout and post-primary vaccinations as well as post-booster with vaccine-induced IFN-γ immunity, DTH reactions, and clinical outcome. Although these observations were made in total vaccinated patient population [22], in this report we show that these findings are more profound when retrieved from patients expressing HLA-DRB1*11 or HLA-A*24 or both alleles, further supporting the role of these alleles as restricting elements for vaccine-induced adaptive immunity to the endogenous AE36 peptide followed by clinical improvements. Furthermore, we have shown that these patients had preexisting IFN-γ immunity, implying similar HLA restrictions also for endogenous responses to AE36. However, no matter what the type of the HLA allele was, patients with increased preexistent TGF-β plasma levels had less vaccine-induced immunity and worse OS. This implies that less suppressed preexisting immunity in combination with previous (i.e., before vaccinations) standard therapies may effectively synergize with vaccine-induced immunity for clinical benefit. The results presented herein suggest a possible prognostic role of these HLA alleles for disease outcome in conjunction with standard treatments after diagnosis and vaccinations. The same also applies for preexisting increased IFN-γ immunity along with low TGF-β levels in HLA-A*24 and/or HLA-DRB1*11 carriers, whose combination could be considered as prognostic biomarker for increased OS. Our data are also suggestive for a predictive role of both HLA alleles as well as of IFN-γ and TGF-β for immunological responses to vaccination and clinical outcome [38].
Our study should only be considered at best hypothesis generating, as it is limited by the number of patients analyzed. Despite this limitation, can we hypothesize that the biomarkers identified herein may have prognostic significance in early-stage prostate cancer patients by slowing down their growth rates and thus delaying disease progression. Furthermore, will these biomarkers have any utility at later stages of disease when tumor biology may have been altered through the process of immunoediting? How, then, preexistent immunity will be able to deal with a tumor having different immune phenotype and oncogenic signalling pathways? Apparently, in such a case, the endogenous antitumor immunity will be too weak to perform sufficiently against tumor and will require exogenous support via immunotherapeutic modalities. Therapeutic vaccines are useful tools for reinstating the endogenous antitumor response by enhancing its effector functions. Consequently, quantitative and qualitative determinations of preexisting endogenous antitumor immunological responses during and post-immune system-oriented treatments may serve as biomarkers or surrogate endpoints for clinical responses. Therefore, the vaccines should have a powerful capacity to activate the immune system, for instance, by being polyvalent capable of stimulating a plethora of helper and/or cytotoxic T cell clones. Such multivalent vaccines, comprising of a variety of determinants representing immunogenic T cell epitopes, have been described in literature [39]. The AE37 vaccine is polyvalent since, according to prediction algorithms (SYFPEITHI), it encompasses MHC class I-binding motifs, which renders it suitable for directly triggering, CD4+ as well as CD8+ T cells [20]. Indeed, during primary vaccinations, we could demonstrate increased percentages of both CD4+ and CD8+ T cells producing IFN-γ upon in vitro sensitization with AE36. Another most representative example of a polyvalent vaccine is Provenge, an autologous cellular vaccine, the first to receive an FDA approval for metastatic castrate-resistant prostate cancer [40]. Recombinant virus-based as well as plasmid DNA-based vaccines are also polyvalent and have shown promising results in the field of active immunotherapy of cancer [41, 42]. Finally, we also demonstrate that the genetic background of patients determines, to a certain extent, their response status since HLA-DRB1*11+ and HLA-A*24+ patients had pre-existent immunity and rigorously responded to the vaccine both immunologically and clinically. To this end, however, we cannot exclude the possibility that additional HLA alleles may contribute to this as well, given the fact that polyvalent vaccines cover a broad range of HLA alleles functioning as restricting elements.
To conclude, HLA-A*24 and/or HLA-DRB1*11 genotype carriers respond better to the AE37 vaccine, both immunologically and clinically. However, these alleles may not yet be considered as favorable factors. The distribution of HLA antigens, also including HLA-A*24 and HLA-DRB1*11, differs worldwide as a result of populations migration and environmental selection. HLA-A*24 and HLA-DRB1*11 are among the predominating alleles in southern Europe (http://www.allelefrequencies.net/#). According to the National Tissue Typing Center in Greece, 29 % of individuals express HLA-A*24 and 39 % HLA-DRB1*11. These percentages are far below compared with those detected among our vaccinated patients. Thus, it would be quite interesting to explore in future studies the association of HLA-A*24 and HLA-DRB1*11 in a larger cohort of prostate cancer patients and their association with prognosis and survival after AE37 vaccinations.
Taking all the aforementioned into consideration, our studies underscore the importance of HLA-A*24 and HLA-DRB1*11 as positive prognosticators for immunological and clinical responses to AE37 vaccination. Our findings, relating elevated preexisting IFN-γ immunity to decreased TGF-β plasma levels and increased vaccine-induced immunity with prolonged survival, suggest that less suppressed endogenous immunity can be positively modulated by vaccinations. Additionally, the situation described in the periphery proposes that the vaccine supports the evolution of endogenous immunity within the tumor microenvironment. Given that all retrospective analyses, described above, were based on a relative small sample of vaccinated patients, the findings are hypothesis-generating only and a phase II trial is warranted for applying the identified biomarkers for validation.
Acknowledgments
We are indebted to our patients for their voluntary participation in this study. We also thank Joanne Kalogeropoulou for excellent technical assistance, Dr. Panagiotis Tzonis for helping in patients’ follow-up, and C. Zeyher for expert assistance with Trp/Kyn measurements. This study was supported by Antigen Express, Inc. Worcester, Massachusetts, and a grant from OPAP SA (Greek Organisation of Football Prognostics S.A.) to Michael Papamichail.
Conflict of interest
The authors declare that they have no conflict of interest.
Abbreviations
- APC
Antigen presenting cells
- CTL
Cytotoxic T lymphocytes
- DTH
Delayed-type hypersensitivity
- FDA
Food and drug administration
- GM-CSF
Granulocyte–macrophage colony-stimulating factor
- HER-2
Human epidermal growth factor receptor 2 or HER-2/neu
- HLA
Human leukocyte antigen
- IDO
Indoleamine 2,3-dioxygenase
- IFN-γ
Interferon gamma
- LT
Long term
- LTB
Long-term booster
- MHC
Major histocompatibility complex
- OS
Overall survival
- TGF-β
Transforming growth factor beta
References
- 1.Schlom J. Therapeutic cancer vaccines: current status and moving forward. J Natl Cancer Inst. 2012;104:599–613. doi: 10.1093/jnci/djs033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cheng ML, Fong L. Beyond sipuleucel-T: immune approaches to treating prostate cancer. Curr Treat Options Oncol. 2014;15:115–126. doi: 10.1007/s11864-013-0267-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.May KF, Jr, Gulley JL, Drake CG, Dranoff G, Kantoff PW. Prostate cancer immunotherapy. Clin Cancer Res. 2011;17:5233–5238. doi: 10.1158/1078-0432.CCR-10-3402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baxevanis CN. Outlining novel scenarios for improved therapeutic cancer vaccines: the PANVAC paradigm. Expert Rev Vaccines. 2012;11:275–277. doi: 10.1586/erv.11.193. [DOI] [PubMed] [Google Scholar]
- 5.Hoos A, Eggermont AM, Janetzki S, et al. Improved endpoints for cancer immunotherapy trials. J Natl Cancer Inst. 2010;102:1388–1397. doi: 10.1093/jnci/djq310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coulie PG, Van den Eynde BJ, van der Bruggen P, Boon T. Tumour antigens recognized by T lymphocytes: at the core of cancer immunotherapy. Nat Rev Cancer. 2014;14:135–146. doi: 10.1038/nrc3670. [DOI] [PubMed] [Google Scholar]
- 7.Rammensee H, Bachmann J, Emmerich NP, Bachor OA, Stevanovic S. SYFPEITHI: database for MHC ligands and peptide motifs. Immunogenetics. 1999;50:213–219. doi: 10.1007/s002510050595. [DOI] [PubMed] [Google Scholar]
- 8.Andersson E, Villabona L, Bergfeldt K, Carlson JW, Ferrone S, Kiessling R, Seliger B, Masucci GV. Correlation of HLA-A02* genotype and HLA class I antigen down-regulation with the prognosis of epithelial ovarian cancer. Cancer Immunol Immunother. 2012;61:1243–1253. doi: 10.1007/s00262-012-1201-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Helgadottir H, Andersson E, Villabona L, et al. The common Scandinavian human leucocyte antigen ancestral haplotype 62.1 as prognostic factor in patients with advanced malignant melanoma. Cancer Immunol Immunother. 2009;58:1599–1608. doi: 10.1007/s00262-009-0669-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lu CC, Chen JC, Jin YT, Yang HB, Chan SH, Tsai ST. Genetic susceptibility to nasopharyngeal carcinoma within the HLA-A locus in Taiwanese. Int J Cancer. 2003;103:745–751. doi: 10.1002/ijc.10861. [DOI] [PubMed] [Google Scholar]
- 11.So T, Takenoyama M, Sugaya M, Yasuda M, Eifuku R, Yoshimatsu T, Osaki T, Yasumoto K. Unfavorable prognosis of patients with non-small cell lung carcinoma associated with HLA-A2. Lung Cancer. 2001;32:39–46. doi: 10.1016/S0169-5002(00)00204-X. [DOI] [PubMed] [Google Scholar]
- 12.Bain C, Merrouche Y, Puisieux I, et al. Correlation between clinical response to interleukin 2 and HLA phenotypes in patients with metastatic renal cell carcinoma. Br J Cancer. 1997;75:283–286. doi: 10.1038/bjc.1997.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cortes J, Fayad L, Kantarjian H, O’Brien S, Lee MS, Talpaz M. Association of HLA phenotype and response to interferon-alpha in patients with chronic myelogenous leukemia. Leukemia. 1998;12:455–462. doi: 10.1038/sj.leu.2400965. [DOI] [PubMed] [Google Scholar]
- 14.Sosman JA, Unger JM, Liu PY, Flaherty LE, Park MS, Kempf RA, Thompson JA, Terasaki PI, Sondak VK. Adjuvant immunotherapy of resected, intermediate-thickness, node-negative melanoma with an allogeneic tumor vaccine: impact of HLA class I antigen expression on outcome. J Clin Oncol. 2002;20:2067–2075. doi: 10.1200/JCO.2002.08.072. [DOI] [PubMed] [Google Scholar]
- 15.Mellinghoff IK, Vivanco I, Kwon A, Tran C, Wongvipat J, Sawyers CL. HER2/neu kinase-dependent modulation of androgen receptor function through effects on DNA binding and stability. Cancer Cell. 2004;6:517–527. doi: 10.1016/j.ccr.2004.09.031. [DOI] [PubMed] [Google Scholar]
- 16.Ricciardelli C, Jackson MW, Choong CS, Stahl J, Marshall VR, Horsfall DJ, Tilley WD. Elevated levels of HER-2/neu and androgen receptor in clinically localized prostate cancer identifies metastatic potential. Prostate. 2008;68:830–838. doi: 10.1002/pros.20747. [DOI] [PubMed] [Google Scholar]
- 17.Shariat SF, Bensalah K, Karam JA, Roehrborn CG, Gallina A, Lotan Y, Slawin KM, Karakiewicz PI. Preoperative plasma HER2 and epidermal growth factor receptor for staging and prognostication in patients with clinically localized prostate cancer. Clin Cancer Res. 2007;13:5377–5384. doi: 10.1158/1078-0432.CCR-07-0330. [DOI] [PubMed] [Google Scholar]
- 18.Sotiriadou NN, Kallinteris NL, Gritzapis AD, et al. Ii-Key/HER-2/neu(776-790) hybrid peptides induce more effective immunological responses over the native peptide in lymphocyte cultures from patients with HER-2/neu + tumors. Cancer Immunol Immunother. 2007;56:601–613. doi: 10.1007/s00262-006-0213-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Voutsas IF, Gritzapis AD, Mahaira LG, Salagianni M, von Hofe E, Kallinteris NL, Baxevanis CN. Induction of potent CD4+ T cell-mediated antitumor responses by a helper HER-2/neu peptide linked to the Ii-Key moiety of the invariant chain. Int J Cancer. 2007;121:2031–2041. doi: 10.1002/ijc.22936. [DOI] [PubMed] [Google Scholar]
- 20.Perez SA, Kallinteris NL, Bisias S, et al. Results from a phase I clinical study of the novel Ii-Key/HER-2/neu(776-790) hybrid peptide vaccine in patients with prostate cancer. Clin Cancer Res. 2010;16:3495–3506. doi: 10.1158/1078-0432.CCR-10-0085. [DOI] [PubMed] [Google Scholar]
- 21.Perez SA, Anastasopoulou EA, Tzonis P, Gouttefangeas C, Kalbacher H, Papamichail M, Baxevanis CN. AE37 peptide vaccination in prostate cancer: a 4-year immunological assessment updates on a phase I trial. Cancer Immunol Immunother. 2013;62:1599–1608. doi: 10.1007/s00262-013-1461-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Perez SA, Anastasopoulou EA, Papamichail M, Baxevanis CN. AE37 peptide vaccination in prostate cancer: identification of biomarkers in the context of prognosis and prediction. Cancer Immunol Immunother. 2014;63:1141–1150. doi: 10.1007/s00262-014-1582-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sperner-Unterweger B, Neurauter G, Klieber M, Kurz K, Meraner V, Zeimet A, Fuchs D. Enhanced tryptophan degradation in patients with ovarian carcinoma correlates with several serum soluble immune activation markers. Immunobiology. 2011;216:296–301. doi: 10.1016/j.imbio.2010.07.010. [DOI] [PubMed] [Google Scholar]
- 24.Galon J, Mlecnik B, Bindea G, et al. Towards the introduction of the ‘Immunoscore’ in the classification of malignant tumours. J Pathol. 2014;232:199–209. doi: 10.1002/path.4287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rammensee HG. Chemistry of peptides associated with MHC class I and class II molecules. Curr Opin Immunol. 1995;7:85–96. doi: 10.1016/0952-7915(95)80033-6. [DOI] [PubMed] [Google Scholar]
- 26.Perez SA, von Hofe E, Kallinteris NL, Gritzapis AD, Peoples GE, Papamichail M, Baxevanis CN. A new era in anticancer peptide vaccines. Cancer. 2010;116:2071–2080. doi: 10.1002/cncr.24988. [DOI] [PubMed] [Google Scholar]
- 27.Salazar LG, Fikes J, Southwood S, Ishioka G, Knutson KL, Gooley TA, Schiffman K, Disis ML. Immunization of cancer patients with HER-2/neu-derived peptides demonstrating high-affinity binding to multiple class II alleles. Clin Cancer Res. 2003;9:5559–5565. [PubMed] [Google Scholar]
- 28.Sotiriadou R, Perez SA, Gritzapis AD, et al. Peptide HER2(776-788) represents a naturally processed broad MHC class II-restricted T cell epitope. Br J Cancer. 2001;85:1527–1534. doi: 10.1054/bjoc.2001.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Disis ML. Immune regulation of cancer. J Clin Oncol. 2010;28:4531–4538. doi: 10.1200/JCO.2009.27.2146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hoon DS, Okamoto T, Wang HJ, Elashoff R, Nizze AJ, Foshag LJ, Gammon G, Morton DL. Is the survival of melanoma patients receiving polyvalent melanoma cell vaccine linked to the human leukocyte antigen phenotype of patients? J Clin Oncol. 1998;16:1430–1437. doi: 10.1200/JCO.1998.16.4.1430. [DOI] [PubMed] [Google Scholar]
- 31.Baxevanis CN, Papamichail M, Perez SA. Therapeutic cancer vaccines: a long and winding road to success. Expert Rev Vaccines. 2014;13:131–144. doi: 10.1586/14760584.2014.852961. [DOI] [PubMed] [Google Scholar]
- 32.Baxevanis CN, Voutsas IF, Gritzapis AD, Perez SA, Papamichail M. HER-2/neu as a target for cancer vaccines. Immunotherapy. 2010;2:213–226. doi: 10.2217/imt.09.89. [DOI] [PubMed] [Google Scholar]
- 33.Cuppens K, Vansteenkiste J. Vaccination therapy for non-small-cell lung cancer. Curr Opin Oncol. 2014;26:165–170. doi: 10.1097/CCO.0000000000000052. [DOI] [PubMed] [Google Scholar]
- 34.Bioley G, Guillaume P, Luescher I, et al. HLA class I—associated immunodominance affects CTL responsiveness to an ESO recombinant protein tumor antigen vaccine. Clin Cancer Res. 2009;15:299–306. doi: 10.1158/1078-0432.CCR-08-1747. [DOI] [PubMed] [Google Scholar]
- 35.Schietinger A, Philip M, Liu RB, Schreiber K, Schreiber H. Bystander killing of cancer requires the cooperation of CD4(+) and CD8(+) T cells during the effector phase. J Exp Med. 2010;207:2469–2477. doi: 10.1084/jem.20092450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Toes RE, Ossendorp F, Offringa R, Melief CJ. CD4 T cells and their role in antitumor immune responses. J Exp Med. 1999;189:753–756. doi: 10.1084/jem.189.5.753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Disis ML. Immunologic biomarkers as correlates of clinical response to cancer immunotherapy. Cancer Immunol Immunother. 2011;60:433–442. doi: 10.1007/s00262-010-0960-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Baxevanis CN, Papamichail M, Perez SA. Prostate cancer vaccines: the long road to clinical application. Cancer Immunol Immunother. 2015;64:401–408. doi: 10.1007/s00262-015-1667-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Arens R, van Hall T, van der Burg SH, Ossendorp F, Melief CJ. Prospects of combinatorial synthetic peptide vaccine-based immunotherapy against cancer. Semin Immunol. 2013;25:182–190. doi: 10.1016/j.smim.2013.04.008. [DOI] [PubMed] [Google Scholar]
- 40.Di Lorenzo G, Buonerba C, Kantoff PW. Immunotherapy for the treatment of prostate cancer. Nat Rev Clin Oncol. 2011;8:551–561. doi: 10.1038/nrclinonc.2011.72. [DOI] [PubMed] [Google Scholar]
- 41.Gulley JL, Drake CG. Immunotherapy for prostate cancer: recent advances, lessons learned, and areas for further research. Clin Cancer Res. 2011;17:3884–3891. doi: 10.1158/1078-0432.CCR-10-2656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schweizer MT, Drake CG. Immunotherapy for prostate cancer: recent developments and future challenges. Cancer Metastasis Rev. 2014;33:641–655. doi: 10.1007/s10555-013-9479-8. [DOI] [PMC free article] [PubMed] [Google Scholar]