Abstract
Tumor or metastatic cells lose MHC class I (MHC-I) expression during cancer progression as an escape mechanism from immune surveillance. These defects in MHC-I may be reversible by cytokines or different agents (soft lesions) or irreversible due to structural defects (hard lesions). The nature of these MHC-I alterations might determine the success or failure of immunotherapy treatments. In this study, we have used an MHC-I-positive murine fibrosarcoma tumor clone, GR9-A7, which generates multiple lung and lymph node metastases with reversible MHC-I alterations after treatment with IFN-γ. Four different antitumor treatments were carried out after primary tumor excision to determine their capacity to inhibit spontaneous metastatic colonization of the GR9-A7 tumor clone. We found that 2 different immunotherapy protocols (CpG plus autologous irradiated-GR9-A7 cells and protein-bound polysaccharide K (PSK) and 1 chemoimmunotherapy (docetaxel plus PSK) induced eradication of metastases. In contrast, chemotherapy with docetaxel alone produced only partial reduction in the number of metastases. Flow cytometric analysis of lymphocyte populations showed an immunosuppression in GR9-A7 tumor-bearing host, which could be reverted by immunotherapy treatments. Our results suggest that irreversible or reversible MHC-I alterations in tumor target cells may determine its progression or regression independently of the type of immunotherapy used.
Keywords: MHC class I, Reversible defects, Metastases, Immunotherapy
Introduction
A variety of cancer immunotherapy protocols are widely used for activation of different branches of the immune system, especially in patients with metastatic disease, since primary tumors are usually surgically removed [1, 2]. New therapies have been developed based on the fact that tumor antigens recognized by antitumor lymphocytes are small peptides coupled with HLA class I molecules. Peptide-based vaccines have proven able to boost a specific antitumor T cell response, inducing specific CTLs able to recognize and eliminate autologous tumor cells in vitro [3, 4]. Nevertheless, the clinical effects of antitumor vaccines and cancer immunotherapies remain below expectations [5, 6]. To date, cancer immunotherapy has achieved a full response in < 5% of cases [7, 8].
Evidence has been accumulating that tumors generate sophisticated escape mechanisms to avoid attacks by different components of the immune system [9, 10]. Among those mechanisms, alterations in the expression of MHC-I molecules are frequently detected in primary tumors and metastatic lesions [11–13]. These molecules are known to play a crucial role in the presentation of tumor antigens to T lymphocytes and modulate NK cell function. Our group previously reported that the progression or regression of human melanoma metastases after immunotherapy correlates with the level of HLA class I antigen expression [14, 15]. Metastatic melanoma tissue samples were obtained from 2 patients undergoing autologous vaccination plus BCG and/or IFN-α treatment. Results demonstrated that regressing metastases expressed high levels of HLA-ABC molecules, while progressing lesions had low/intermediate levels of HLA class I and harbored structural defects (hard lesions) in MHC-I or β2-microglobulin genes. Based on these data, we hypothesized that metastatic tumor cells with reversible defects in MHC-I expression (soft lesions) might respond to immunotherapy by upregulating MHC-I expression and promoting T cell-mediated rejection [16, 17].
In the present study, we have tested this hypothesis in a preclinical murine metastatic model. GR9-A7 fibrosarcoma clone presents surface expression of H-2 Kd, Dd, and Ld MHC molecules and generates multiple spontaneous lung and lymph node metastases. These metastases present defects in MHC-I expression that are always reversible by IFN-γ treatment. Three different immunotherapy and a chemotherapy protocols were applied after excision of the primary tumor: (a) immunotherapy with CpG plus irradiated autologous tumor cells GR9-A7; (b) immunotherapy with protein-bound polysaccharide K (PSK) [18]; (c) chemotherapy with docetaxel; and (d) chemoimmunotherapy with docetaxel plus PSK. PSK obtained from Basidiomycetes, also known as Krestin, has been used as an agent in the treatment for cancer in Asia for over 30 years [19–21]. PSK is derived from the fungus Coriolus versicolor and has documented anticancer activity in vitro in experimental models [22] and in human clinical trials [23, 24]. These studies have suggested the efficacy of PSK as principally an immunomodulator of biological response.
We report here that a mouse tumor clone (GR9-A7) with high MHC class I expression generates multiple metastases with reversible MHC lesions. These metastases can be eradicated with different immunotherapy protocols.
Materials and methods
Cell lines
GR9 cell line is derived from a mouse fibrosarcoma induced by methylcholanthrene in BALB/c mice, which has been developed and extensively characterized in our laboratory [25]. It is composed of cell clones with distinct H-2 class I expression patterns and metastatic capacities [26]. Spontaneous metastasis assays were performed with different GR9 cell clones and one of them, the GR9-A7 clone, was selected for this study. GR9-A7 is a clone obtained by a limited dilution method from GR9 cell line. Cell lines were maintained in Dulbecco’s medium (Sigma–Aldrich, Madrid, Spain) supplemented with 10% fetal bovine serum (Life Technologies, Milan, Italy), 2 mM glutamine (Sigma–Aldrich), and antibiotics. In some experiments, cell lines were treated with 100 U/ml IFN-γ for 48 h (Sigma–Aldrich) or irradiated with a dose of 100 Gy.
Mice
Eight-week-old male BALB/c mice (Charles River Laboratories, Barcelona, Spain) were used in experiments. Breeding and care of animals were undertaken in compliance with European Community Directive 86/609/CEE and Spanish law (Real Decreto 1201/2005) for the use of laboratory animals. Housing and all experimental procedures involving animals were performed according to protocols approved by the hospital’s animal care committee and in compliance with the guidelines on animal welfare of the National Committee for Animal Experiments.
Spontaneous metastasis assay
1.25 × 106 GR9-A7 cells were injected into the footpad of syngeneic BALB/c mice. The growth of local tumors was monitored in all animals and recorded 3 times/week. For each tumor, the largest diameter was measured with electronic calipers. Tumors were excised when the largest diameter of each tumor reached 10 mm, at around 20–25 days postinjection. The model resembles metastatic development in humans where primary tumor is surgically removed. Before tumor excision, mice were anesthetized with 0.04 ml of diazepam (Valium, Roche, Madrid, Spain) and 0.1 ml of ketamine (Ketolar, Pfizer, Spain). Primary tumors were removed with sterilized instruments using electrocautery to minimize bleeding, and the wounds were closed using surgical clips and adhesive. After surgery, each animal was housed alone until recovery from anesthesia. On day 70 postinjection, when signs of illness started to evidence, animals were anesthetized and euthanized by cervical dislocation. Complete necropsy was done, and the number of metastases was counted. Local tumors and macroscopically visible metastatic nodules were excised, disaggregated, and adapted to tissue culture. Following, mice lungs were fixed in Bouin’s solution and micrometastases were counted.
Immunotherapy, chemotherapy, and chemoimmunotherapy protocols
Each of 4 different treatment protocols was applied in a group of 20 mice. The protocols used were as follows:
PSK: kindly provided by Kureha Chemical Ind. Co. (Tokyo, Japan). It is prepared by extracting cultured mycelia of Coriolus versicolor with hot water. The precipitate is separated from the clear supernatant with saturated ammonium sulfate, then desalted and dried. Protein-bound polysaccharide K was dissolved in saline solution and heated at 50°C for 30 min until a clear solution appeared. PSK solution was filtered by 0.22-μm filters (Millipore, Spain). PSK was administered intraperitoneally (i.p.) at 2.5 mg/mouse/week in 500 μl of saline solution.
CpG ODN 1826 plus irradiated GR9-A7 cells: 1 million GR9-A7 cells were collected in Dulbecco’s medium and irradiated with a dose of 100 Gy. After irradiation, cells were maintained in culture for 48 h, washed with PBS, and resuspended in 200 μl saline solution. Twenty micrograms of CpG ODN 1826 (InvivoGen, San Diego, CA, USA) were resuspended in 200 μl saline solution. Both solutions were i.p. injected weekly in each mouse.
Docetaxel: (Taxotere, 80 mg/2 ml, Sanofi Aventis, Barcelona, Spain) was diluted to 10 mg/ml according to the manufacturer’s instructions and further diluted to 625 μg/ml in sterile saline solution; 125 μg/mouse/week was i.p. injected.
PSK plus docetaxel: mice were simultaneously i.p. injected with PSK combined with docetaxel at the same doses as in the above monotherapies.
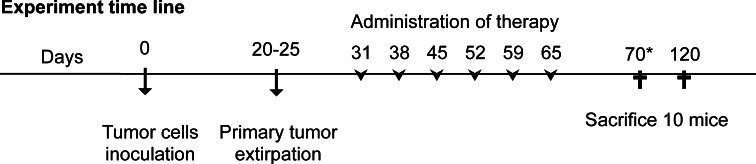
These agents were administered once a week during a 6-week period from day 7 after local tumor excision (i.e., days 31, 38, 45, 52, 59, and 65 after cell injection) (Fig. 1). Ten mice from each group were euthanized at 1 week after the end of the treatment (day 70 post-cell injection). The remaining 10 mice in each group were euthanized on day 120 post-cell injection (Fig. 1). In docetaxel group, all mice were euthanized on day 70 due to signs of illness. Twenty mice in a control group (injected with GR9-A7 cells) received i.p., two hundred microliter of saline solution. In this group, all mice were euthanized on day 70 post-cell injection. The same protocols were applied in tumor-free animals to evaluate the toxicity of the treatments and their effect on the survival rate.
Fig. 1.
Time schedule of the treatments. Therapies were administered weekly for 6 consecutive weeks by intraperitoneal injection, beginning 1 week after removal of the primary tumor. Ten mice were killed on day 70 to analyze treatment efficacy. Remaining 10 mice were killed on day 120 to evaluate the long-term effect. *In control and docetaxel group, all mice were killed on day 70
Analysis of MHC-I surface expression
MHC-I surface expression was analyzed by indirect immunofluorescence using FACS (FACSCanto; Becton–Dickinson) according to a standard protocol. Primary antibodies used were anti-H-2 Kd (K9–18), anti-H-2 Dd (34-5-8), and anti-H-2 Ld (28.14.8 and 30.5.7), all obtained from the ATCC (Rockville, MD). Fluorescein isothiocyanate (FITC)-conjugate antibody (anti-mouse FITC IgG/Fab, Sigma–Aldrich) at a 1:120 dilution was used as secondary antibody. Isotype-matched nonimmune mouse IgG and cells labeled with only the fluorescein-conjugated antibody were used as controls. A minimum of 1 × 104 cells were analyzed with Cell-Quest Pro software. Each cell line was firstly tested after only one culture passage and retested for 3 times more during different passages. All cell lines were analyzed in basal conditions and after treatment with IFN-γ for 48 h.
Real time RT–PCR
An mRNA isolation kit (Myltenyi-Biotech) was used to extract mRNA from GR9-A7 fibrosarcoma clone, from metastases with both high- and low- MHC-I surface expression. First-strand cDNA was synthesized with 100 ng of mRNA using a High Capacity Reverse Transcription Kit (Applied Biosystems, Foster City, CA) in a total volume of 20 μl. These cDNAs were diluted to a final volume of 100 μl. Real-time quantitative RT–PCR analyses for β2-microglobulin, H2-Kd, H2-Dd, H2-Ld, TAP1, TAP2, LMP2, LMP7, calnexin, calreticulin, and tapasin genes were performed in the 7500 Fast System (Applied Biosystems), using GADPH and β-actin genes as housekeeping genes. PCR reactions were performed in quadruplicate, and values obtained were expressed as means ± SD (standard deviation). Real-time quantitative RT–PCR was performed with the Power SYBR Green Master mix (Applied Biosystems). Primers and amplicon size for each gene are shown in Table 1. PCR conditions were 40 cycles of 15 s of denaturation at 95°C and 60 s at 60°C.
Table 1.
Primers used in real time RT–PCR
| Primer | Name | Sequence 5′ → 3′ | Product length (bp) |
|---|---|---|---|
| Gapdh | Forward | TCAAGAAGGTGGTGAAGCAGG | 117 |
| Reverse | CGATCGAAGGTGGAAGAGTGG | ||
| Actb | Forward | CAACACAGTGCTGTCTGGTGG | 62 |
| Reverse | CTCCTTCTGCATCCTGTCAGC | ||
| B2m | Forward | CTGGTGCTTGTCTCACTGACC | 214 |
| Reverse | GACCAGTCCTTGCTGAAGGAC | ||
| H-2 Kd | Forward | CCATCCACTGTCTCCAACACG | 112 |
| Reverse | CCACCTGTGTTCCTTCTCATC | ||
| H-2 Dd | Forward | GCCTCCTTCATCCACCAAGAC | 81 |
| Reverse | CACAGCTCCAAGGATGACCAC | ||
| H-2 Ld | Forward | CGTCCACTGACTCTTACATGG | 74 |
| Reverse | CCACAGCTCCAATGATGGCC | ||
| TAP1 | Forward | GCTGTTCAGGTCCTGCTCTC | 105 |
| Reverse | CACTGAGTGGAGAGCAAGGAG | ||
| TAP2 | Forward | AGGAGCCTGTGCTGTTCTCG | 116 |
| Reverse | CTATGAAGTCGTCTGCACAGG | ||
| LMP2 | Forward | CCTCTGCACCAGCACATCTTC | 93 |
| Reverse | CGTGTAGCTCCAGCTGGTAG | ||
| LMP7 | Forward | GGACCTCAGTCCTGAAGAGG | 116 |
| Reverse | CAACCGTCTTCCTTCATGTGG | ||
| Calnexin | Forward | GCAGCTGAAGAGCGTCCATGG | 154 |
| Reverse | TCATCCTTCACATCTGGCTGG | ||
| Calreticulin | Forward | AGCAGATGAAGGACAAGCAGG | 139 |
| Reverse | CCTCTCATCTTCTTCGTCCTC | ||
| Tapasin | Forward | CAGCTACCTCCAGTCACTGC | 193 |
| Reverse | CCTAGCACCTTGAGGAGTCC |
Analysis of lymphocyte populations
Splenocyte and peripheral blood lymphocyte populations were studied by direct immunofluorescence. Mice were anesthetized with 0.04 ml of diazepam and 0.1 ml of ketamine. Spleens were excised, cells were dissociated, and splenocytes were isolated. Blood was collected from the vena cava with a 25-G needle using 0.02 ml of heparin (Rovi, Madrid, Spain). Blood was centrifuged at 300×g for 15 min. Mononuclear cells from spleen and blood were isolated by gradient centrifugation at 500×g for 20 min on Ficoll Histopaque 1083 (Sigma–Aldrich), and they were washed 3 times with PBS. For direct immunofluorescence, the following labeled antibodies (Miltenyi Biotec, Madrid, Spain) were used: CD3ε-APC, CD4-FITC, CD8-PE, CD25-PE, FoxP3-APC, CD19-FITC, and CD49b-FITC. Isotype-matched nonimmune mouse IgGs conjugated with FITC, PE, or APC were used as controls. FcR Blocking Reagent was used to block unwanted binding of antibodies to mouse cells expressing Fc receptors. Immunofluorescence was done according to the manufacturer’s instructions (Miltenyi Biotec), using FoxP3 staining buffer to obtain optimal results in FoxP3 immunofluorescent staining. Cells were analyzed on a FACSCanto cytometer (Becton–Dickinson). Each sample consisted of a minimum of 5 × 104 cells and was analyzed with CellQuest-Pro software.
Statistical analysis
Data were expressed as mean ± SD. The paired Student’s t test was used to compare mean values. A significance level of P ≤ 0.05 was assumed for all statistical evaluation. SPSS 16.0.2 (SPSS, Chicago IL) was used for the data analyses.
Results
GR9-A7 fibrosarcoma cells originate multiple spontaneous metastases with reversible defects in MHC-I expression
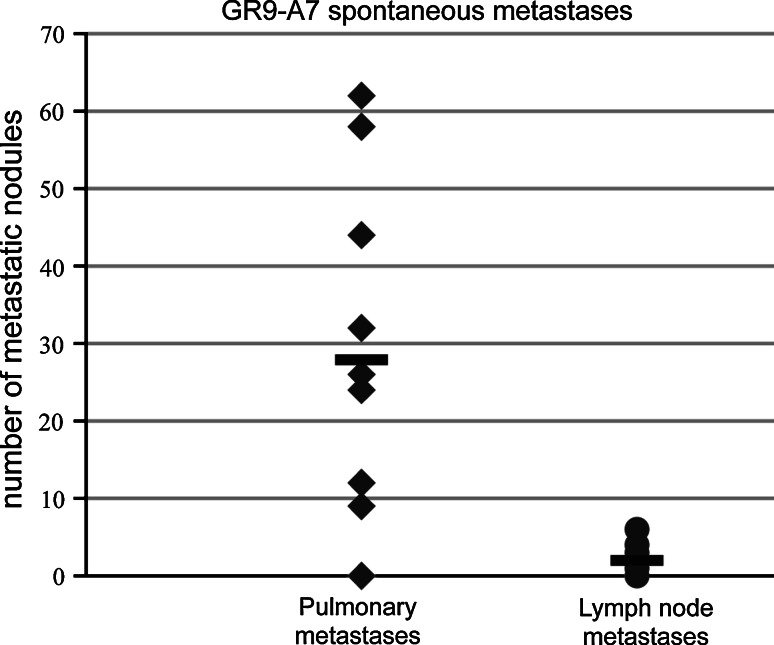
We performed spontaneous metastasis assays (as described in “Materials and methods”) in groups of 10 BALB/c mice using GR9-A7 fibrosarcoma clone cell line. Mice were killed on day 70 postinjection and complete necropsy was done. Ninety percent of animals presented metastases. Mice developed pulmonary metastases (PMs, range 9–62, mean of 28) and lymph node metastases (LNMs, range 0–6, mean of 2) (Fig. 2). Lymph node metastases were localized in axillaries nodes, para-aortic nodes, and superior mesenteric nodes. The experiment was repeated twice with similar results.
Fig. 2.
Spontaneous metastases in mice bearing GR9-A7 fibrosarcoma clone. Seventy days postinjection of GR9-A7 cells, 10 mice were euthanized and metastatic nodules were counted. Nine of ten mice developed metastases (range, 9–62 pulmonary metastases and 0–6 lymph node metastases). This experiment was repeated twice more with similar results
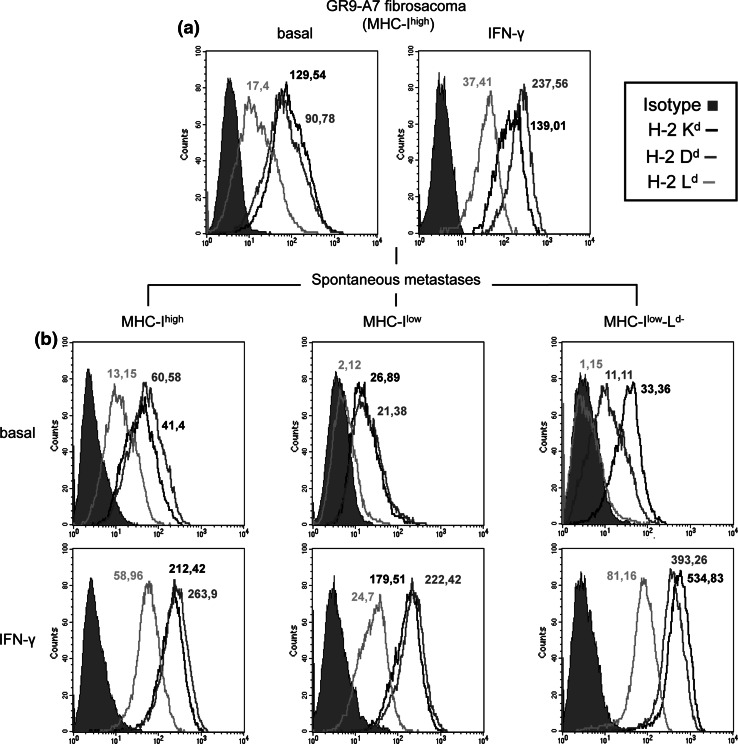
Seventy-seven PMs and 27 LNMs were adapted to tissue culture, and their H-2 class I surface expression was analyzed and compared with GR9-A7 clone. GR9-A7 is characterized by positive H-2 Kd, Dd, and Ld surface cell expression (Fig. 3a), and these 3 molecules were upregulated after treatment with 100 U/ml IFN-γ for 48 h (Fig. 3a). There were no differences between pulmonary and lymph node metastases. Similar or higher H-2 class I surface expression to that of the GR9-A7 fibrosarcoma clone was found in 29% of the metastases (MHChigh) (Fig. 3b), whereas lower MHC-I expression versus GR9-A7 tumor cells was found in 71% of metastases (MHClow), with half of them negative for Ld allele (MHClow-Ld−) (Fig. 3b). Each metastatic cell line was tested after the first culture passage and retested for 3 times more during different passages, and the results were always practically identical. The MHC-I defects found in the metastases were always reversible after in vitro treatment with 100 U/ml IFN-γ for 48 h (Fig. 3b).
Fig. 3.
MHC-I surface expression of GR9-A7 clone and GR9-A7 spontaneous metastases. a MHC-I phenotype of the GR9-A7 fibrosarcoma cell line in basal conditions and after treatment with IFN-γ (100 U/ml) for 48 h: H-2 Kd (black line), H-2 Dd (gray line), and H-2 Ld (light gray line); means of fluorescence are indicated. b MHC-I phenotypes of GR9-A7 spontaneous metastases: 3 different MHC-I phenotypes were found (MHChigh, MHClow, and MHClow-Ld−). A representative example of 3 independent experiments is depicted
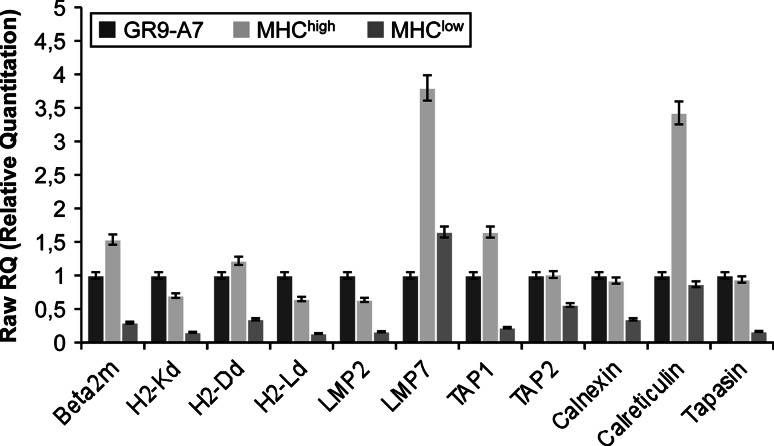
To study the possible mechanisms implicated in the different MHC-I expression of these metastases, we analyzed the transcriptional gene expression of antigen-processing machinery (APM), H-2 class I heavy chain, and β2-microglobulin genes. GR9-A7 fibrosarcoma clone was compared with 3 MHChigh metastases and 3 MHClow metastases. Data were expressed as mean ± SD of 3 metastases by group and the 3 independent experiments. The genes analyzed and primers used in real-time quantitative RT–PCR are given in Table 1. Data were normalized to GADPH and β-actin housekeeping genes. Figure 4 shows the results using the values for GR9-A7 cells as reference (assigned a relative value of 1). MHChigh metastases had higher expression of β2-microglobulin, LMP7, TAP1, and calreticulin genes (Fig. 4). There were no differences in the other genes versus GR9-A7. MHClow metastases presented a downregulation of all MHC-I-related genes except for LMP7 and calreticulin (Fig. 4). These results evidence a direct relationship between H-2 class I surface expression and MHC-I-related transcriptional gene expression in these metastatic cell lines.
Fig. 4.
Transcriptional expression of MHC-I and APM genes in GR9-A7 tumor cells and in its spontaneous metastases. APM, β2-m, and H-2 class I heavy chain genes expression was analyzed by real-time quantitative RT–PCR. GR9-A7 fibrosarcoma clone (black) was compared with 3 MHChigh metastases (light gray) and 3 MHClow metastases (gray). Data are expressed as mean ± SD of 3 metastases of each group. Data were normalized using GADPH and β-actin as housekeeping genes. Results are the average of 3 independent experiments
Eradication of metastases by immunotherapy and chemoimmunotherapy but not by chemotherapy treatments
The GR9-A7 fibrosarcoma clone produced multiple PMs and LNMs in spontaneous metastasis assays, with a downregulation of MHC-I surface expression in 71% of the metastases, including the absence of the Ld allele expression in 36% of the metastases. All of these alterations in MHC-I expression were reversible with IFN-γ treatment. We established a preclinical assay in mice with four different anticancer therapies: 2 immunotherapy protocols, CpG ODN 1826 (20 μg/mouse/week) + 1 million irradiated GR9-A7 cells, and PSK (2.5 mg/mouse/week); a chemotherapy protocol based on docetaxel (125 μg/mouse/week); and a chemoimmunotherapy protocol combining PSK plus docetaxel. The control group was also injected with GR9-A7 cells but received i.p., two hundred microliter of saline solution. All therapies were administered after primary tumor excision, only during metastatic development and not during local tumor growth. These therapies comprised weekly intraperitoneal injections administered during a 6-week period starting at 1 week after local tumor excision (Fig. 1). None of the therapies applied in this study had any toxic effect in tumor-free mice or affected their survival rate.
Firstly, we performed in vitro assays analyzing MHC-I expression in irradiated GR9-A7 tumor cells after 48 h in tissue culture, since irradiation of cells may induce an increase in MHC-I surface expression [27]. We found an increased expression of Kd, Dd, and Ld molecules in comparison with baseline MHC-I expression (data not shown). This increase in MHC-I expression might induce an increase in specific immune response against these tumor cells. PSK can produce a cytotoxic effect on tumor cells [28]; therefore, we tested in vitro cytotoxic activity of PSK on GR9-A7 cells at 2 different concentrations, 50 and 100 μg/ml, finding that PSK did not modify the proliferation rate of GR9-A7 cells (data not shown). Our group previously reported that PSK only slightly modified the proliferation rate of GR9-B9, another GR9 fibrosarcoma tumor clone [28].
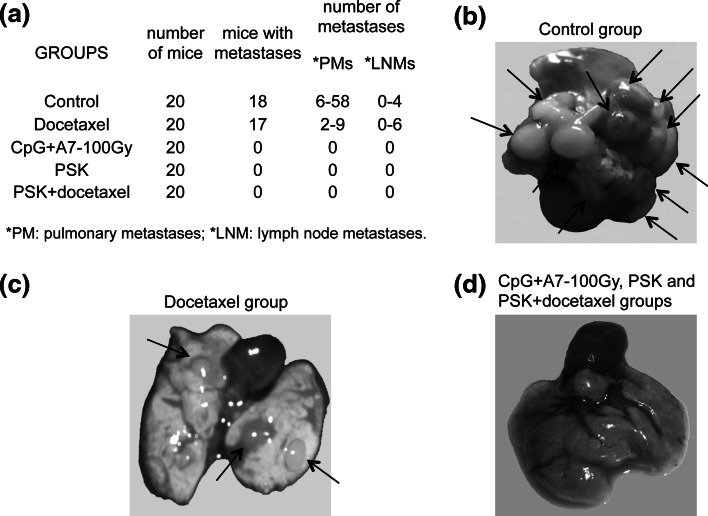
Twenty mice from control group and docetaxel group were killed on day 70 postinjection. In control group, 18 of 20 mice developed pulmonary metastases (range 6–58) and lymph node metastases (range 0–4) (Fig. 5a, b). Seventeen of twenty mice in the docetaxel group had metastases (2–9 PMs; 0–6 LNMs) (Fig. 5a, c). Ten mice from each immunotherapy and chemoimmunotherapy treatment group were euthanized at the end of the treatment (day 70 post-cell injection). No metastases were found in the mice from the CpG + A7–100 Gy, PSK, or PSK + docetaxel groups (Fig. 5a, d). The other 10 mice of these groups were euthanized at day 120 post-cell injection, and newly, all mice were metastasis-free. The experiment was repeated twice with the same results. In a subsequent assay, we found that the mice in these treatment groups remained alive and metastasis-free at 12 months. In brief, immunotherapy and chemoimmunotherapy showed complete inhibition of metastasis development. In contrast, chemotherapy treatment with docetaxel only partially reduced the number of metastases.
Fig. 5.
Spontaneous metastases in mice treated with the different therapies. a The table shows the number of mice/group, number of mice with metastases, and number of pulmonary (PMs) and lymph node (LNMs) metastases found/mouse. These results were reproducible in other 2 independent experiments. b–d Representative picture of lungs from mice of control group (b), docetaxel group (c), and CpG plus A7 irradiated cells, PSK alone or in combination with docetaxel groups (d)
Metastases obtained from docetaxel group were adapted to tissue culture, and MHC-I surface expression was analyzed. These metastases present the same 3 MHC-I phenotypes as found in control group (Fig. 3b) and at practically identical percentages: 31% MHChigh, 39% MHClow, and 30% MHClow-Ld−. According to these results, MHC-I expression of the metastases was highly similar between control and docetaxel groups, indicating that the MHC-I profile was not altered by docetaxel treatment for the mice.
Changes in lymphocyte populations in mice from different groups
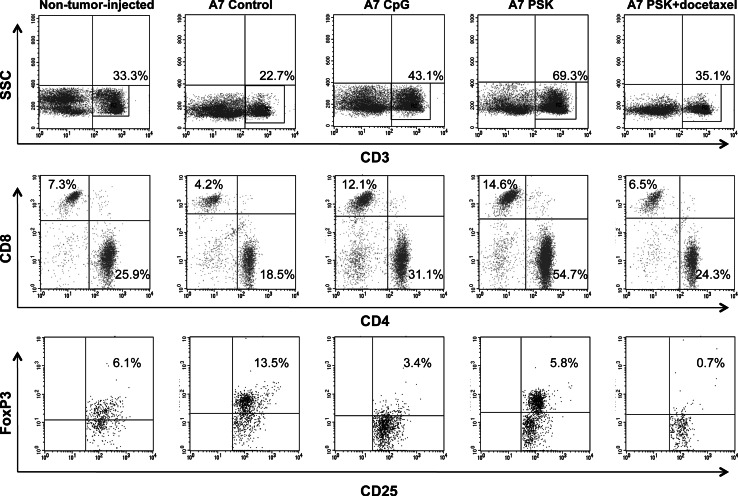
Ten mice from each group were killed at the end of treatment administration, on day 70 postinjection. Spleen and peripheral blood lymphocyte populations from these mice were studied by flow cytometry. Mice from control group showed statistically significant changes on the lymphocyte populations as compared to animals not injected with tumor cells (non-tumor-injected mice) (P < 0.05): decrease in CD3+ lymphocytes (25.3 vs. 31.5%), principally CD8+ T lymphocytes (4.9 vs. 9.1%); also decrease in NK cells (3.4 vs. 8.3%); and an increase of CD4+CD25+FoxP3+ regulatory cells (10.8 vs. 6.1% among CD4+) (Table 2). The immunotherapy and chemoimmunotherapy treatments eradicated metastases in the mice, showing statistically significant differences in lymphocyte populations between treated and control mice (P < 0.05) (Table 2). In the CpG + A7-100 Gy group, the treatment produced an increase in CD3+ (40.9 vs. 25.3%), CD3+CD8+ (12.4 vs. 4.9%), and CD3+CD4+ (28.5 vs. 20.4%) lymphocytes and a decrease in CD4+CD25+FoxP3+ lymphocytes (4.7 vs. 10.8% among CD4+) (Table 2). In the PSK group, the treatment increased CD3+ lymphocytes to 51.6%, CD3+CD8+ to 12.2%, and CD3+CD4+ to 39.4% and markedly augmented NK cells, CD3−CD49+ (9.9 vs. 3.4%) (Table 2). In the PSK plus docetaxel group, a strong decrease in CD4+CD25+FoxP3+ lymphocytes was detected (1.1 vs. 10.8% among CD4+) and an increase in CD3+ and CD4+ lymphocytes (32.2 and 26.5%, respectively) (Table 2). A representative experiment depicting these changes is shown in Fig. 6. It should be borne in mind that these data correspond to the systemic level and not to the tumor site. Lymphocyte populations were also analyzed in peripheral blood, finding similar differences to those found in spleen (data not shown).
Table 2.
Spleen lymphocyte populations in the different mice groups (%)
| Mice | CD3+ | CD4+ | CD8+ | CD3+CD4+CD25+FoxP3+a | CD3−CD19+ | CD3−CD49+ | CD3+CD49+ |
|---|---|---|---|---|---|---|---|
| Non-tumor injected | 31.5 ± 4.5 | 9.1 ± 2.4 | 22.4 ± 3.1 | 6.1 ± 1.8 | 50.7 ± 4.5 | 8.3 ± 1.0 | 2.5 ± 1.1 |
| A7 Control | 25.3 ± 6.9* | 4.9 ± 1.7* | 20.4 ± 5.3 | 10.8 ± 4.2* | 54.6 ± 5.9 | 3.4 ± 2.0* | 0.9 ± 0.5* |
| A7 CpG | 40.9 ± 5.4** | 12.4 ± 2.2** | 28.5 ± 3.9** | 4.7 ± 3.8** | 43.6 ± 12.3 | 3.4 ± 1.3 | 2.0 ± 0.8** |
| A7 PSK | 51.6 ± 17.7** | 12.2 ± 2.4** | 39.4 ± 15.3** | 9.5 ± 3.8 | 27.4 ± 15.7** | 9.9 ± 0.6** | 1.6 ± 0.1** |
| A7 PSK + docetaxel | 32.2 ± 2.9** | 5.7 ± 0.8 | 26.5 ± 2.2** | 1.1 ± 0.3** | 52.9 ± 5.3 | 2.7 ± 0.1 | 0.9 ± 0.1 |
Data are expressed as mean ± SD of 10 mice of each group
* P < 0.05 when A7 control group was compared to non-tumor-injected group
** P < 0.05 when each treatment group was compared to A7 control group
aPercentage among CD4+ cells
Fig. 6.
Changes in spleen lymphocyte populations in the different groups of mice. A representative experiment showing T lymphocyte populations (CD3+CD4+ and CD3+CD8+) and T reg cells (CD4+CD25+FoxP3+). All percentages are referred to total lymphocytes except for T reg cells that are among CD4+ cells
Discussion
The data presented in this paper indicate that the spontaneous metastatic colonization produced by a fibrosarcoma tumor clone (GR9-A7) was completely eradicated by different immunotherapy treatments but not by chemotherapy alone. To resemble the natural cancer progression in humans, we always initiated therapies after excision of large primary tumor when metastases are already established [29]. Importantly, all the metastases generated from GR9-A7 tumor cells had reversible defects in MHC-I expression, which could be restored after IFN-γ treatment. The MHC-I defects were due a coordinated transcriptional downregulation of several APM and H-2 class I heavy chains genes. We classified the metastases obtained in 3 groups according to the total MHC expression as high, low, or low-Ld− (when the Ld expression levels were undetectable). In all cases, the mean fluorescence obtained for Ld was lower that the level in the control GR9-A7 clone. All the studied metastases retained the capacity to upregulate the Ld gene after IFN-γ treatment (Fig. 3b). In the GR9-A7 metastatic system, the level of H-2 Ld expression could act as a restriction element for the presentation of the specific tumor antigen that influences the T cell activation.
We favor the idea that the level of MHC-I expression in a particular cancer cell target is a crucial factor that determines the capacity to activate specific antitumor T lymphocytes. In immunotherapy protocols with peptides derived from tumor antigens, the loss of expression of MHC-I molecules or the loss of the antigen itself predicts treatment failure [30–32]. Furthermore, previous results from our group indicate that irreversible “hard” defects in MHC-I expression play a major role in the appearance of progressing metastases after immunotherapy in melanoma patients [14, 15]. Similarly, we have recently reported that bladder tumor relapses after BCG immunotherapy present more profound irreversible “hard” MHC-I alterations [33]. In this context, the success or failure of immunotherapy to induce tumor rejection (specific immunization with tumor-derived peptides or polyclonal immune stimulation with immunomodulatory agents) might depend on the reversible “soft” or irreversible “hard” nature of preexisting MHC-I defects of the target tumor cell rather than on the type of therapy used. In the case of reversible lesions, MHC-I cell surface expression can be restored, leading to a CTL antitumor response and the regression of lesions [16, 17]. This could be the case for the metastases originated from GR9-A7 clone since all of them have the capacity to upregulate MHC-I after IFN-γ treatment. We also studied other clones of GR9 tumor model, with low MHC-I expression and irreversible MHC-I downregulation in metastases, in which immunotherapies did not eradicate metastatic colonization (unpublished observations).
We found an increase in some lymphocyte populations in immunotherapy and chemoimmunotherapy groups at systemic level. These data do not necessarily reflect events at the tumor site, but the metastases were totally eradicated in all of these mice, indicating that a strong immune response may also occur at metastatic sites. The immunotherapy used included PSK and CpG combined with autologous irradiated GR9-A7 cells. PSK is widely used in Asia and has dual anticancer effects, not only activating T cells, NKs, and dendritic cells [34–36], but also exerting a direct cytotoxic activity on tumor cells [28]. According to our assays, PSK had no cytotoxic activity on GR9-A7 tumor cells and therefore acted only as an immunomodulator, producing an increase in T and NK cells. GR9-A7 fibrosarcoma cells evidence in vitro sensitivity to antitumor T cells and resistance to NK cytotoxicity [37]. The action of PSK on T cells might induce the selection of metastatic colonies with low MHC-I phenotypes. In this context, we analyzed some metastases during the course of PSK treatment, in the halfway through the treatment, and all these metastases showed reduced MHC-I expression (unpublished observations). The activation of T and NK cells could locally release cytokines in the tumor microenvironment upregulating MHC-I molecules and, therefore leading to the elimination of these metastatic cells. Similarly, treatment with CpG ODN 1826 plus autologous irradiated GR9-A7 cells may produce in vivo a cytokine storm, mainly IFN type I [38, 39], which could markedly increase MHC expression in metastatic cells, favoring their elimination by CTLs. Other possibility is that activation of T cells and decrease in T reg cells might promote recognition of metastatic cells with low MHC-I expression.
In mice from PSK + docetaxel therapy group was detected a marked decrease in T reg CD4+ lymphocytes, which would further potentiate the immune response. Various clinical trials have shown that PSK enhances the antitumor effect of different chemotherapeutic agents [21, 40, 41]. It is also possible that docetaxel increases the PSK-induced immune response. Thus, several clinical and preclinical studies with chemoimmunotherapy have reported enhanced immune response and antitumor activity [42–46]. All of these data support the benefits of combined chemoimmunotherapy protocols. In our preclinical tumor model, the application of docetaxel alone did not block metastatic progression but partially reduced the number of metastases. Furthermore, the MHC-I phenotype of the metastases was similar to that of the control group, suggesting that the mechanism of action of this chemotherapeutic treatment is independent of the MHC-I phenotype.
Previous results from our laboratory showed that another tumor clone of this fibrosarcoma, GR9-B9 (H-2 class I negative but with capacity to upregulate Kd, Dd and Ld molecules after IFN-γ treatment), generated MHC-I-positive metastatic variants on immunodeficient nude/nude mice and MHC-I-negative metastases on immunocompetent syngeneic BALB/c mice [47]. These results using a deprived immune system showed that MHC-positive metastases progressed in the absence of T cells but were eliminated in immunocompetent mice, in which only MHC-negative metastases could progress. Interestingly, the metastases in nude mice were highly immunogenic when transplanted into immunocompetent mice [48]. Our previous and present results, alongside observations in human studies, suggest that the progression or regression of metastases after immunotherapy may depend not on the type of immunotherapy used but rather on the capacity to upregulate the MHC-I expression of tumor cells and on the immune status of the host [49].
The results presented in this paper may contribute to explain the apparently low effectiveness of current immunotherapeutic protocols. Analysis of MHC expression in primary tumors and metastases, especially the identification of irreversible versus reversible defects in MHC-I expression, might help to explain why some metastatic lesions are eradicated after immunotherapy and others are not. It will also open up the possibility of identifying metastases that would respond to immunotherapy and those that require gene therapy to replace the defective MHC-I gene or other mutated genes involved in antigen presentation.
Acknowledgments
The authors thank I. Linares and E. Arias for technical assistance and staff of the Departments of Hospital Pharmacy and Radiotherapeutic Oncology of the Virgen de las Nieves University Hospital for their collaboration. They also thank Dr. Natalia Aptsiauri for helpful discussion and Richard Davies for editorial assistance. C. Garrido was supported by the MEC (FPU, 1631) and A.M. García-Lora by FIS-Research Contract CP03/0111 and Stabilization Contract of Fundación Progreso y Salud. This study was partially funded by grants from the FIS (CP03/0111; PI 08/1265; RTICC, RETIC RD 06/020), Consejería de Salud and PAI (Group CTS-143 and projects CTS-695 and CTS-3952) from the Junta de Andalucía in Spain; by the ENACT project (LSHC-CT-2004-503306) and by the Cancer Immunotherapy project (OJ 2004/c158, 18234) of the European Community; and by Kureha Chemical Industry, Tokyo, Japan.
Conflict of interest
The authors declare that they have no conflict of interest.
Abbreviations
- MHC-I
Major histocompatibility complex class I
- H-2
Mouse leukocyte antigen
- HLA
Human leukocyte antigen
- PSK
Protein-bound polysaccharide K
- BCG
Bacillus of Calmette-Guérin
- MCA
Methylcholanthrene
- PMs
Pulmonary metastases
- LNMs
Lymph node metastases
Contributor Information
Angel García-Lora, Email: angel.miguel.exts@juntadeandalucia.es.
Federico Garrido, FAX: +34-958020069, Email: federico.garrido.sspa@juntadeandalucia.es.
References
- 1.Rosenberg SA. Progress in human tumour immunology and immunotherapy. Nature. 2001;411:380–384. doi: 10.1038/35077246. [DOI] [PubMed] [Google Scholar]
- 2.Schadendorf D, Algarra SM, Bastholt L, Cinat G, Dreno B, Eggermont AM, Espinosa E, Guo J, Hauschild A, Petrella T, et al. Immunotherapy of distant metastatic disease. Ann Oncol. 2009;20(Suppl 6):41–50. doi: 10.1093/annonc/mdp253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Godelaine D, Carrasco J, Lucas S, Karanikas V, Schuler-Thurner B, Coulie PG, Schuler G, Boon T, Van Pel A. Polyclonal CTL responses observed in melanoma patients vaccinated with dendritic cells pulsed with a MAGE-3.A1 peptide. J Immunol. 2003;171:4893–4897. doi: 10.4049/jimmunol.171.9.4893. [DOI] [PubMed] [Google Scholar]
- 4.Boon T, Coulie PG, Van den Eynde BJ, van der Bruggen P. Human T cell responses against melanoma. Annu Rev Immunol. 2006;24:175–208. doi: 10.1146/annurev.immunol.24.021605.090733. [DOI] [PubMed] [Google Scholar]
- 5.Anichini A, Vegetti C, Mortarini R. The paradox of T cell-mediated antitumor immunity in spite of poor clinical outcome in human melanoma. Cancer Immunol Immunother. 2004;53:855–864. doi: 10.1007/s00262-004-0526-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.de la Cruz-Merino L, Grande-Pulido E, Albero-Tamarit A, Codes-Manuel de Villena ME. Cancer and immune response: old and new evidence for future challenges. Oncologist. 2008;13:1246–1254. doi: 10.1634/theoncologist.2008-0166. [DOI] [PubMed] [Google Scholar]
- 7.Rosenberg SA, Yang JC, Restifo NP. Cancer immunotherapy: moving beyond current vaccines. Nat Med. 2004;10:909–915. doi: 10.1038/nm1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rosenberg SA, Sherry RM, Morton KE, Scharfman WJ, Yang JC, Topalian SL, Royal RE, Kammula U, Restifo NP, Hughes MS, et al. Tumor progression can occur despite the induction of very high levels of self/tumor antigen-specific CD8+ T cells in patients with melanoma. J Immunol. 2005;175:6169–6176. doi: 10.4049/jimmunol.175.9.6169. [DOI] [PubMed] [Google Scholar]
- 9.Ahmad M, Rees RC, Ali SA. Escape from immunotherapy: possible mechanisms that influence tumor regression/progression. Cancer Immunol Immunother. 2004;53:844–854. doi: 10.1007/s00262-004-0540-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Smyth MJ, Dunn GP, Schreiber RD. Cancer immunosurveillance and immunoediting: the roles of immunity in suppressing tumor development and shaping tumor immunogenicity. Adv Immunol. 2006;90:1–50. doi: 10.1016/S0065-2776(06)90001-7. [DOI] [PubMed] [Google Scholar]
- 11.Garrido F, Ruiz-Cabello F, Cabrera T, Perez-Villar JJ, Lopez-Botet M, Duggan-Keen M, Stern PL. Implications for immunosurveillance of altered HLA class I phenotypes in human tumours. Immunol Today. 1997;18:89–95. doi: 10.1016/S0167-5699(96)10075-X. [DOI] [PubMed] [Google Scholar]
- 12.Seliger B, Cabrera T, Garrido F, Ferrone S. HLA class I antigen abnormalities and immune escape by malignant cells. Semin Cancer Biol. 2002;12:3–13. doi: 10.1006/scbi.2001.0404. [DOI] [PubMed] [Google Scholar]
- 13.Chang CC, Campoli M, Restifo NP, Wang X, Ferrone S. Immune selection of hot-spot beta 2-microglobulin gene mutations, HLA-A2 allospecificity loss, and antigen-processing machinery component down-regulation in melanoma cells derived from recurrent metastases following immunotherapy. J Immunol. 2005;174:1462–1471. doi: 10.4049/jimmunol.174.3.1462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cabrera T, Lara E, Romero JM, Maleno I, Real LM, Ruiz-Cabello F, Valero P, Camacho FM, Garrido F. HLA class I expression in metastatic melanoma correlates with tumor development during autologous vaccination. Cancer Immunol Immunother. 2007;56:709–717. doi: 10.1007/s00262-006-0226-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Carretero R, Romero JM, Ruiz-Cabello F, Maleno I, Rodriguez F, Camacho FM, Real LM, Garrido F, Cabrera T. Analysis of HLA class I expression in progressing and regressing metastatic melanoma lesions after immunotherapy. Immunogenetics. 2008;60:439–447. doi: 10.1007/s00251-008-0303-5. [DOI] [PubMed] [Google Scholar]
- 16.Aptsiauri N, Carretero R, Garcia-Lora A, Real LM, Cabrera T, Garrido F. Regressing and progressing metastatic lesions: resistance to immunotherapy is predetermined by irreversible HLA class I antigen alterations. Cancer Immunol Immunother. 2008;57:1727–1733. doi: 10.1007/s00262-008-0532-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Garrido F, Cabrera T, Aptsiauri N. Hard and soft lesions underlying the HLA class I alterations in cancer cells: implications for immunotherapy. Int J Cancer. 2010;127:249–256. doi: 10.1002/ijc.25270. [DOI] [PubMed] [Google Scholar]
- 18.Fisher M, Yang LX. Anticancer effects and mechanisms of polysaccharide-K (PSK): implications of cancer immunotherapy. Anticancer Res. 2002;22:1737–1754. [PubMed] [Google Scholar]
- 19.Nakazato H, Koike A, Saji S, Ogawa N, Sakamoto J. Efficacy of immunochemotherapy as adjuvant treatment after curative resection of gastric cancer. Study group of Immunochemotherapy with PSK for Gastric Cancer. Lancet. 1994;343:1122–1126. doi: 10.1016/s0140-6736(94)90233-x. [DOI] [PubMed] [Google Scholar]
- 20.Munemoto Y, Iida Y, Ohata K, Saito H, Fujisawa K, Kasahara Y, Mitsui T, Asada Y, Miura S. Significance of postoperative adjuvant immunochemotherapy after curative resection of colorectal cancers: identification of responders incorporating the age factor. Oncol Rep. 2004;11:623–635. [PubMed] [Google Scholar]
- 21.Ohwada S, Ikeya T, Yokomori T, Kusaba T, Roppongi T, Takahashi T, Nakamura S, Kakinuma S, Iwazaki S, Ishikawa H, et al. Adjuvant immunochemotherapy with oral Tegafur/Uracil plus PSK in patients with stage II or III colorectal cancer: a randomised controlled study. Br J Cancer. 2004;90:1003–1010. doi: 10.1038/sj.bjc.6601619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Katoh R, Ooshiro M. Enhancement of antitumor effect of tegafur/uracil (UFT) plus leucovorin by combined treatment with protein-bound polysaccharide, PSK, in mouse models. Cell Mol Immunol. 2007;4:295–299. [PubMed] [Google Scholar]
- 23.Sakamoto J, Morita S, Oba K, Matsui T, Kobayashi M, Nakazato H, Ohashi Y. Efficacy of adjuvant immunochemotherapy with polysaccharide K for patients with curatively resected colorectal cancer: a meta-analysis of centrally randomized controlled clinical trials. Cancer Immunol Immunother. 2006;55:404–411. doi: 10.1007/s00262-005-0054-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ueda Y, Fujimura T, Kinami S, Hirono Y, Yamaguchi A, Naitoh H, Tani T, Kaji M, Yamagishi H, Miwa K. A randomized phase III trial of postoperative adjuvant therapy with S-1 alone versus S-1 plus PSK for stage II/IIIA gastric cancer: Hokuriku-Kinki immunochemo-therapy study group-gastric cancer. Jpn J Clin Oncol. 2006;36:519–522. doi: 10.1093/jjco/hyl048. [DOI] [PubMed] [Google Scholar]
- 25.Garrido A, Perez M, Delgado C, Garrido ML, Rojano J, Algarra I, Garrido F. Influence of class I H-2 gene expression on local tumor growth. Description of a model obtained from clones derived from a solid BALB/c tumor. Exp Clin Immunogenet. 1986;3:98–110. [PubMed] [Google Scholar]
- 26.Perez M, Algarra I, Ljunggren HG, Caballero A, Mialdea MJ, Gaforio JJ, Klein G, Karre K, Garrido F. A weakly tumorigenic phenotype with high MHC class-I expression is associated with high metastatic potential after surgical removal of the primary murine fibrosarcoma. Int J Cancer. 1990;46:258–261. doi: 10.1002/ijc.2910460219. [DOI] [PubMed] [Google Scholar]
- 27.Reits EA, Hodge JW, Herberts CA, Groothuis TA, Chakraborty M, Wansley EK, Camphausen K, Luiten RM, de Ru AH, Neijssen J, et al. Radiation modulates the peptide repertoire, enhances MHC class I expression, and induces successful antitumor immunotherapy. J Exp Med. 2006;203:1259–1271. doi: 10.1084/jem.20052494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jimenez-Medina E, Berruguilla E, Romero I, Algarra I, Collado A, Garrido F, Garcia-Lora A. The immunomodulator PSK induces in vitro cytotoxic activity in tumour cell lines via arrest of cell cycle and induction of apoptosis. BMC Cancer. 2008;8:78. doi: 10.1186/1471-2407-8-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ellis LM, Fidler IJ. Finding the tumor copycat. Therapy fails, patients don’t. Nat Med. 2010;16:974–975. doi: 10.1038/nm0910-974. [DOI] [PubMed] [Google Scholar]
- 30.Benitez R, Godelaine D, Lopez-Nevot MA, Brasseur F, Jimenez P, Marchand M, Oliva MR, van Baren N, Cabrera T, Andry G, et al. Mutations of the β2-microglobulin gene result in a lack of HLA class I molecules on melanoma cells of 2 patients immunized with MAGE peptides. Tiss Antigens. 1998;52:520–529. doi: 10.1111/j.1399-0039.1998.tb03082.x. [DOI] [PubMed] [Google Scholar]
- 31.Riker A, Cormier J, Panelli M, Kammula U, Wang E, Abati A, Fetsch P, Lee KH, Steinberg S, Rosenberg S, et al. Immune selection after antigen-specific immunotherapy of melanoma. Surgery. 1999;126:112–120. doi: 10.1016/S0039-6060(99)70143-1. [DOI] [PubMed] [Google Scholar]
- 32.Hoffmann TK, Nakano K, Elder EM, Dworacki G, Finkelstein SD, Appella E, Whiteside TL, DeLeo AB. Generation of T cells specific for the wild-type sequence p53 (264–272) peptide in cancer patients: implications for immunoselection of epitope loss variants. J Immunol. 2000;165:5938–5944. doi: 10.4049/jimmunol.165.10.5938. [DOI] [PubMed] [Google Scholar]
- 33.Carretero R, Cabrera T, Gil H, Saenz-Lopez P, Maleno I, Aptsiauri N, Cozar JM, Garrido F (2010) BCG immunotherapy of bladder cancer induces selection of HLA class I-deficient tumor cells. Int J Cancer [Epub ahead of print] [DOI] [PubMed]
- 34.Jimenez E, Garcia-Lora A, Martinez M, Garrido F. Identification of the protein components of protein-bound polysaccharide that interact with NKL cells. Cancer Immunol Immunother. 2005;54:395–399. doi: 10.1007/s00262-004-0601-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Okuzawa M, Shinohara H, Kobayashi T, Iwamoto M, Toyoda M, Tanigawa N. PSK: a protein-bound polysaccharide, overcomes defective maturation of dendritic cells exposed to tumor-derived factors in vitro. Int J Oncol. 2002;20:1189–1195. [PubMed] [Google Scholar]
- 36.Asai H, Iijima H, Matsunaga K, Oguchi Y, Katsuno H, Maeda K. Protein-bound polysaccharide K augments IL-2 production from murine mesenteric lymph node CD4+ T cells by modulating T cell receptor signaling. Cancer Immunol Immunother. 2008;57:1647–1655. doi: 10.1007/s00262-008-0498-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Algarra I, Ohlen C, Perez M, Ljunggren HG, Klein G, Garrido F, Karre K. NK sensitivity and lung clearance of MHC-class-I-deficient cells within a heterogeneous fibrosarcoma. Int J Cancer. 1989;44:675–680. doi: 10.1002/ijc.2910440420. [DOI] [PubMed] [Google Scholar]
- 38.Whitmore MM, DeVeer MJ, Edling A, Oates RK, Simons B, Lindner D, Williams BR. Synergistic activation of innate immunity by double-stranded RNA and CpG DNA promotes enhanced antitumor activity. Cancer Res. 2004;64:5850–5860. doi: 10.1158/0008-5472.CAN-04-0063. [DOI] [PubMed] [Google Scholar]
- 39.Kawai T, Akira S. Innate immune recognition of viral infection. Nat Immunol. 2006;7:131–137. doi: 10.1038/ni1303. [DOI] [PubMed] [Google Scholar]
- 40.Ohwada S, Ogawa T, Makita F, Tanahashi Y, Ohya T, Tomizawa N, Satoh Y, Kobayashi I, Izumi M, Takeyoshi I, et al. Beneficial effects of protein-bound polysaccharide K+ tegafur/uracil in patients with stage II or III colorectal cancer: analysis of immunological parameters. Oncol Rep. 2006;15:861–868. [PubMed] [Google Scholar]
- 41.Oba K, Teramukai S, Kobayashi M, Matsui T, Kodera Y, Sakamoto J. Efficacy of adjuvant immunochemotherapy with polysaccharide K for patients with curative resections of gastric cancer. Cancer Immunol Immunother. 2007;56:905–911. doi: 10.1007/s00262-006-0248-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Garnett CT, Schlom J, Hodge JW. Combination of docetaxel and recombinant vaccine enhances T cell responses and antitumor activity: effects of docetaxel on immune enhancement. Clin Cancer Res. 2008;14:3536–3544. doi: 10.1158/1078-0432.CCR-07-4025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kohlmeyer J, Cron M, Landsberg J, Bald T, Renn M, Mikus S, Bondong S, Wikasari D, Gaffal E, Hartmann G, et al. Complete regression of advanced primary and metastatic mouse melanomas following combination chemoimmunotherapy. Cancer Res. 2009;69:6265–6274. doi: 10.1158/0008-5472.CAN-09-0579. [DOI] [PubMed] [Google Scholar]
- 44.Arlen PM, Gulley JL, Parker C, Skarupa L, Pazdur M, Panicali D, Beetham P, Tsang KY, Grosenbach DW, Feldman J, et al. A randomized phase II study of concurrent docetaxel plus vaccine versus vaccine alone in metastatic androgen-independent prostate cancer. Clin Cancer Res. 2006;12:1260–1269. doi: 10.1158/1078-0432.CCR-05-2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nistico P, Capone I, Palermo B, Del Bello D, Ferraresi V, Moschella F, Arico E, Valentini M, Bracci L, Cognetti F, et al. Chemotherapy enhances vaccine-induced antitumor immunity in melanoma patients. Int J Cancer. 2009;124:130–139. doi: 10.1002/ijc.23886. [DOI] [PubMed] [Google Scholar]
- 46.Ramakrishnan R, Assudani D, Nagaraj S, Hunter T, Cho HI, Antonia S, Altiok S, Celis E, Gabrilovich DI. Chemotherapy enhances tumor cell susceptibility to CTL-mediated killing during cancer immunotherapy in mice. J Clin Invest. 2010;120:1111–1124. doi: 10.1172/JCI40269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Garcia-Lora A, Algarra I, Gaforio JJ, Ruiz-Cabello F, Garrido F. Immunoselection by T lymphocytes generates repeated MHC class I-deficient metastatic tumor variants. Int J Cancer. 2001;91:109–119. doi: 10.1002/1097-0215(20010101)91:1<109::AID-IJC1017>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 48.Garcia-Lora A, Martinez M, Algarra I, Gaforio JJ, Garrido F. MHC class I-deficient metastatic tumor variants immunoselected by T lymphocytes originate from the coordinated downregulation of APM components. Int J Cancer. 2003;106:521–527. doi: 10.1002/ijc.11241. [DOI] [PubMed] [Google Scholar]
- 49.Garrido F, Algarra I, Garcia-Lora AM. The escape of cancer from T lymphocytes: immunoselection of MHC class I loss variants harboring structural-irreversible hard lesions. Cancer Immunol Immunother. 2010;59:1601–1606. doi: 10.1007/s00262-010-0893-2. [DOI] [PMC free article] [PubMed] [Google Scholar]