Abstract
Cytokine-induced killer (CIK) cells show cytolytic activity against tumor. The purpose of this study was to evaluate the antitumor effect of dendritic cell (DC)-activated CIK cells in vitro and their clinical efficacy of DC-activated CIK cells in combination with chemotherapy (abbreviated below as chemotherapy plus DC + CIK) in patients with advanced non-small-cell lung cancer (NSCLC). A paired study was performed between 61 patients treated with chemotherapy alone (group 1) and 61 patients treated with chemotherapy plus DC + CIK cells (group 2). In group 2, 36 patients with adenocarcinoma and 18 patients with squamous cell carcinoma were analyzed for the survival rate. Compared to unstimulated CIK cells, DC-activated CIK cells significantly enhanced antitumor activity, increased the ratio of CD3+CD56+ cells, promoted cell proliferation and lessened cell apoptosis. In the paired study, the 1- and 2-year overall survival rates in group 2 were 57.2 and 27.0 %, which were significantly higher than that of group 1 (37.3 and 10.1 %) (P < 0.05). There was no significant difference in the survival rate between the adenocarcinoma and squamous carcinoma patients in group 2. The present study suggests that DC-activated CIK cell has enhanced antitumor effects and chemotherapy plus DC + CIK cells improved the clinical outcomes of chemotherapy for advanced NSCLC patients.
Keywords: Dendritic cell, Cytokine-induced killer cells, Adaptive therapy, Advanced non-small-cell lung cancer
Introduction
Lung cancer is one of the major causes of cancer deaths with a 5-year survival rate of only 15 % [1]. About 80 % of lung malignancies are non-small-cell lung cancer (NSCLC) [2], and more than 50 % of these patients have advanced local invasion and/or long-distance metastasis, which require further postoperative treatment including chemotherapy, radiotherapy, and immunotherapy [3]. Despite current advances in chemotherapeutic treatment, the prognosis for patients with lung cancer remains poor [4, 5]. One limitation of the current chemotherapy is the lack of the clinical evidence to increase patients’ survival time. The largest meta-analysis published to date has showed that chemotherapy only increases the 1-year survival rate by 10 % and median survival time by 6 weeks, as compared to non-chemotherapy treatment [6]. Therefore, developing a new and effective therapy is necessary to improve the survival time in NSCLC patients.
Immunotherapy is becoming a promising and effective therapy for cancer patients, especially for patients with later stage disease. It has been suggested that a number of cancer immunotherapy approaches for lung cancer be developed and tested [7]. Increasing evidence suggests that adoptive immunotherapy reduces tumor recurrence and the metastatic rates of malignant tumors, and [8], as a currently emerging adoptive immunotherapy, cytokine-induced killer (CIK) cells have demonstrated significant antitumor activity in both clinical trials and animal studies having cytotoxic effects against several different types of tumor cells, including Hodgkin disease, non-Hodgkin lymphoma, and hepatoma [7].
Cytokine-induced killer cells are a unique population of cytotoxic T lymphocytes with the characteristic CD3+CD56+ phenotype. CIK cells can proliferate abundantly in vitro and kill tumor cells directly [9]. Moreover, these cells can regulate and increase host cellular immune function in vivo [10]. The high lytic activity against tumor cells is mainly due to the high proliferative potential of CD3+CD56+ CIK cells [11, 12]. CIK cells are therefore ideal for immunotherapy against residual tumor cells. Importantly, the antitumor activity of CIK cells can be enhanced by activation with dendritic cells (DC), which are the most potent antigen presenting cells [13].
Therefore, the purpose of this study was to evaluate the antitumor effect of DC-activated CIK cells in vitro and the clinical efficacy of DC-activated CIK cells in combination with chemotherapy (abbreviated below as chemotherapy plus DC + CIK) in patients with advanced NSCLC. Our study suggests that DC + CIK cells have enhanced antitumor effects and that their combination with chemotherapy DC + CIK cells improves the clinical outcomes of conventional chemotherapy for advanced NSCLC patients.
Materials and methods
Patients
A paired study was performed to compare the clinical outcomes of advanced NSCLC patients received either chemotherapy alone (group 1) or chemotherapy plus DC + CIK cells (group 2). All the patients (28–82 years) were histologically or cytologically confirmed with NSCLC at stage III–IV. The characteristics of patients are summarized in Table 1. In group 2, the survival rate was analyzed between adenocarcinoma (36 patients) and squamous carcinoma (16 patients). The criteria for patient selection included: (1) patients aged between 18 and 80 years, (2) having an expected survival duration of longer than 3 months, (3) a Karnofsky performance status score higher than 40 %, (4) a white blood cell count higher than 3,500/μL, (5) platelet count > 100,000/μL, (6) serum AST/ALT < 2.0 of the upper normal limit, (7) no cardiac arrhythmias, congestive heart failure, or severe coronary artery diseases, and (8) pregnant and lactating women were excluded. Subjects for the two groups were matched for gender, age at onset of disease, pathology, tumor size and metastasis and stage at the first visit. All patients in the chemotherapy group were treated identically to the patients in the DC + CIK plus chemotherapy, including doses and cycles.
Table 1.
Characteristics of 122 patients enrolled in the paired study
| Group 1 | Group 2 | |
|---|---|---|
| Total number of patients | 61 | 61 |
| Gender | ||
| Male, n ( %) | 49 | 49 |
| Female, n ( %) | 12 | 12 |
| Age (years) | ||
| Median | 63 | 63.5 |
| Range | 29–80 | 28–82 |
| Pathology | ||
| Adenocarcinoma, n | 36 | 36 |
| Squamous cell carcinoma, n | 18 | 18 |
| Large-cell carcinoma | 7 | 7 |
| Stage, n | ||
| IIIa | 16 | 16 |
| IIIb | 11 | 11 |
| IV | 34 | 34 |
Informed consent was obtained from all the patients, and the local Ethnics Committee approved the protocol. After the enrollment, all the patients provided a complete medical history and underwent a physical examination. This study was approved by the State Food and Drug Administration of China (2006L01023).
Treatment
All patients were treated as we previously described [14]. Briefly, all patients in group 1 and group 2 received 4 cycles of Navelbine (25 mg/m2 body surface) at day 1 and day 8 and cisplatin (80 mg/m2) at day 1. For the 61 patients who received the chemotherapy plus DC + CIK cells, one month after completion of 4 cycles of chemotherapy, they received autologous CIK cells activated by antigen-loaded DC for four cycles with an interval of one month. For each treatment, the patients received two intravenous infusions of (12.53 ± 1.13) × 109 DC-activated CIK cells with a 1-day interval. Clinical examinations were performed by oncology specialists weekly or biweekly, and the curative effects were evaluated after each treatment.
Preparation of CIK and DC
Auto-CIKs were generated as described [8, 15]. Peripheral blood mononuclear cells (PBMCs) were collected from advanced NSCLC patients using the Cobe Spectra Apheresis System (CaridianBCT, USA) [14]. For culture of CIK, PBMCs were cultured in X-VIVO 20 serum-free medium (Cambrex, USA) containing 50 ng/ml of anti-CD3 antibody, 100 U/ml of recombinant human interleukin (IL)-1α (e-Bioscience, USA), and 1,000 U/ml of recombinant human interferon (IFN)-γ (PEPROTECH, USA). The cells were incubated in a humidified atmosphere with 5 % CO2 at 37 °C. After 24 h, 300 U/ml of recombinant human IL-2 (PEPROTECH, USA) was added to the medium. Every 5 days, IL-2 and IFN-γ were added to the medium. For culture of autologous DC, PBMCs were purified from NSCLC patients as described above and incubated for 2 h, and the adherent cells were cultured in X-VIVO 20 medium containing 1,000 U/ml of IL-4 and 500 U/ml of GM-CSF to facilitate DC growth. To stimulate DC, autologous tumor lysates (100 μg/ml) [16] were added at day 6 and co-cultured with DC for 24 h.
DC activation of CIK cells
After 7 days of culture, CIK cells were co-cultured with stimulated DC for another 7 days. CIK cells were harvested and analyzed for the phenotype, cytotoxicity, and secretion of cytokines. Safety tests were performed during the course of cell culture. All products were free of bacterial and fungal contamination, negative for mycoplasma and contained < 5 Eu endotoxin. Since mature DCs have short lives (only a few days), most DCs in the co-culture system died when CIK cells were harvested.
Identification of CIK cells and DC-activated CIK cells and cytotoxic examination
The cytotoxicity of CIK cells and DC-activated CIK cells was analyzed using CytoTox 96-well Non-Radioactive cytotoxicity assay according to the manufacturer’s instruction (Promega, USA). The target cells used for this assay were tumor cancer cell line A549, K562, and HepG2, as previously described [14]. The phenotypes were analyzed by flow cytometry (BD Aria, USA) with antibodies against CD3-PerCP5.5, CD4-FITC, CD56-PE, and CD25-APC (BD Bioscience, USA). Meanwhile, the supernatants were collected to analyze the level of the following cytokines by an enzyme-linked immunosorbent assay (ELISA): TGF-β, IFN-γ, and IL-10 (eBioscience, USA) after 14 days of culture.
Cell proliferation and apoptosis of DC-activated CIK cells
After 7 days of culture, CIK cells were collected, washed, resuspended with RPMI-1640, and incubated for 10 min at 37 °C after adding CFSE (final concentration was 0.25 μmol/L). Cells were then washed twice with PBS. CIK cells were co-cultured with stimulated DC and labeled with CFSE with the same method as CIK cells. Medium was changed every 2 days (half volume). After 7 days of culture, cells were collected and analyzed by flow cytometry. CIK cells and DC-activated CIK cells were collected and double stained with Annexin V/DAPI for apoptosis analysis (Biovision, USA).
Curative effects
Assessment of clinical response was carried out in all patients of groups 1 and 2, respectively. The primary endpoint was overall survival (OS). OS was defined as the time from the initiation of treatment until death from any cause. The secondary endpoints were the overall response rate (ORR) and disease control rate (DCR). Complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) were reported according to World Health Organization and International Union Against Cancer Criteria [8]. The ORR was the sum of CR and PR, whereas the DCR was the sum of CR, PR, and SD [17].
Immunologic assessment
Cytokine secretion and T cell populations were assessed in 10 patients from group 2. The PBMCs from 10 patients, who received 4 cycles of DC + CIK treatment after chemotherapy, were collected at the beginning of the first DC + CIK treatment. One month after the fourth DC + CIK fusion, PBMCs from the same patient were collected again. Cytokines secretion of IFN-γ, IL-10, TGF-β, and TNF-α were analyzed by ELISA, and the T cell populations CD3+, CD3+CD4+, CD3+CD8+, and CD4+CD25+ cells were analyzed using cytokine antibody assays and flow cytometric analysis.
Statistical methods
The data were calculated using SPSS16.0 (SPSS Inc., Chicago, IL) software package. T cell populations are expressed as mean ± SD and were analyzed by the Student’s t test. The overall survival rate was analyzed by the Kaplan–Meier method. Statistical significance was assessed using the log rank test. We also performed a Cox regression analysis, comparing age and sex with survival rate. A biostatistician performed all analyses, and statistical significance was indicated by P < 0.05.
Results
Induction and identification of DC-activated CIK cells
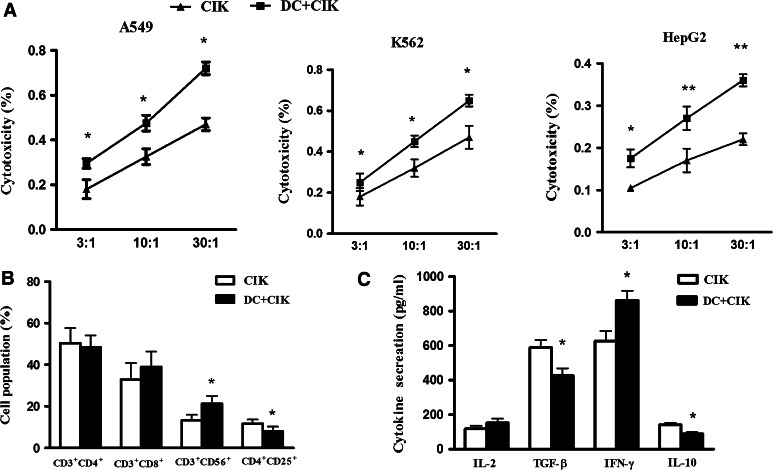
The cytotoxicity of DC-activated CIK cells and CIK cells against tumor cells A549, K562, and HepG2 was detected. The results showed that DC-activated CIK cells had antitumor effects on the cancerous cell lines: A549, K562, and HepG2. Furthermore, DC-activated CIK cells showed the strongest antitumor activity, as compared to unstimulated CIK cells, at ratios of effector to target cells (E:T) of 3:1, 10:1, and 30:1 (Fig. 1a). To determine the immunological effects of DC-activated CIK cells, the phenotype of the cells was characterized. The population of CD3+CD56+ cells increased in the DC-activated CIK group compared with CIK cells alone (P < 0.05). Conversely, the population of CD4+CD25+ cells was decreased in DC-activated CIK cells (P < 0.05, Fig. 1b). The secretion of the cytokine IFN-γ (P < 0.05) was increased, and the secretion of IL-10 (P < 0.05) and TGF-β (P < 0.05) was decreased by DC-activated CIK cells compared to CIK cells alone (Fig. 1c).
Fig. 1.
The analysis of characteristics of DC-activated CIK cells. PBMCs were collected from advanced NSCLC patients and were cultured in serum-free medium containing anti-CD3 antibody, IL-1α, and IFN-γ to obtain CIKs. After 7 days of culture, CIK cells were co-cultured with stimulated DC for another 7 days. CIK cells were harvested and analyzed for the phenotype by flow cytometry, and the secretion of cytokines was determined by ELISA. Every experiment was repeated three times. a The cytotoxicity of DC-activated CIK against A549, K562, and HepG2 cell lines compared to CIK cells alone. DC-activated CIK cells showed the strongest antitumor activity at E/T ratio of 3:1, 10:1, and 30:1. The statistics were calculated at each E/T ratio point between CIK and DC-activated CIK cells. * P < 0.05, ** P < 0.01. b The characteristics of the cell phenotype of DC-activated CIK cells compared with CIK cells alone. The population of CD3+CD56+ was increased in the DC-activated CIK group compared with CIK cells alone. Conversely, the population of CD4+CD25+ cells was decreased in DC-activated CIK (P < 0.05). * P < 0.05. c The analysis of the secretion of cytokines. The secretion of cytokine of IFN-γ was significantly increased, and the secretion of IL-10 and TGF-β were significantly decreased in DC-activated CIK cells compared to CIK cells alone. * P < 0.05
Cell proliferation and apoptosis of DC-activated CIK cells
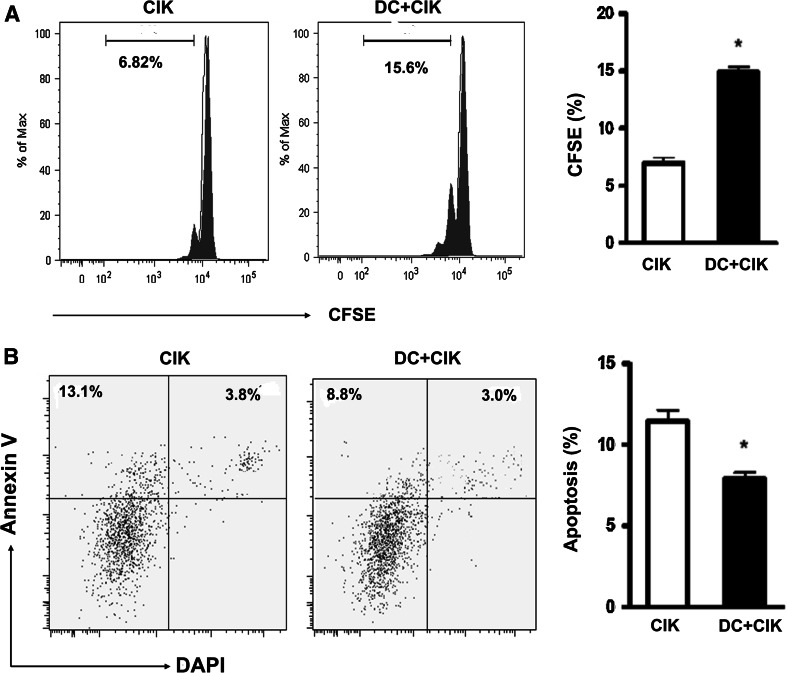
To detect the proliferation of DC-activated CIK cells, we used CFSE assay to examine the change in the proliferation of DC-activated CIK cells as compared to CIK cell alone. DC-activated CIK cells showed an improved proliferation rate (7 %) compared with CIK cell alone (15 %) (P < 0.05, Fig. 2a). We next used Annexin V/DAPI to test the cellular apoptosis of DC-activated CIK cells. After 2 week’s culture, CIK cells showed about 12 % of apoptotic cells, while the DC-activated CIK cells generated about 7.5 % of apoptotic cells (P < 0.05, Fig. 2b).
Fig. 2.
Cell proliferation and apoptosis of DC-activated CIK cells. After 7 days of culture, CIK cells and DC-activated CIK cells were labeled with CFSE (0.25 μmol/L). After another 7 days of culture, cells were collected and analyzed by flow cytometry. CIK cells and DC-activated CIK cells were collected and double stained with Annexin V/DAPI for apoptosis analysis. a DC-activated CIK cells showed an improved proliferation rate (7 %) as compared with CIK cell alone (15 %). b After 2 week’s culture, CIK cells showed about 12 % of apoptotic cells, while the DC-activated CIK cells generated about 7.5 % of apoptotic cells (P < 0.05). Every experiment was repeated three times. * P < 0.05
Immunological responses
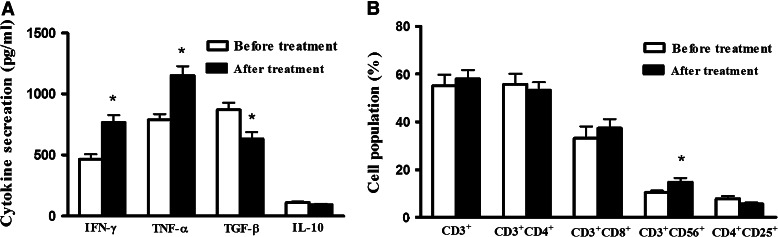
From the 70 advanced NSCLC patients who received the chemotherapy plus DC + CIK cell treatment, plasmas of 10 patients were analyzed for the production of IFN-γ, TNF-α, TGF-β, and IL-10 before and after therapy. There was a significant increase in the secretion of IFN-γ and TNF-α2, a decrease in TGF-β secretion, and no significant changes in IL-10 secretion (Fig. 3a). The T cell subgroups, including CD3+, CD3+CD4+, CD3+CD8+, CD4+CD25+, and CD3+CD56+, were analyzed before and after chemotherapy plus DC + CIK cell treatment. There were no obvious changes in CD3+, CD3+CD4+, CD3+CD8+, and CD4+CD25+ cells before and after therapy (P > 0.05). However, there was a statistical increase in CD3+CD56+ cell after chemotherapy plus DC + CIK cell treatment (P < 0.05) (Fig. 3b).
Fig. 3.
The immunological analysis of 10 patients before and after chemotherapy plus DC + CIK cell treatment. The peripheral blood was collected before and after chemotherapy plus DC + CIK cell treatment. a The analysis of cytokine secretion. There was significant increase in the secretion of IFN-γ and TNF-α, a decrease in TGF-β secretion, and no significant changes in IL-10 secretion. b The analysis of phenotype. There were no obvious changes in CD3+, CD3+CD4+, CD3+CD8+, and CD4+CD25+ cells before and after therapy (P > 0.05). There was a significant increase in CD3+CD56+ cell after chemotherapy plus DC + CIK cell treatment. Every experiment was repeated three times. * P < 0.05
Clinical outcomes
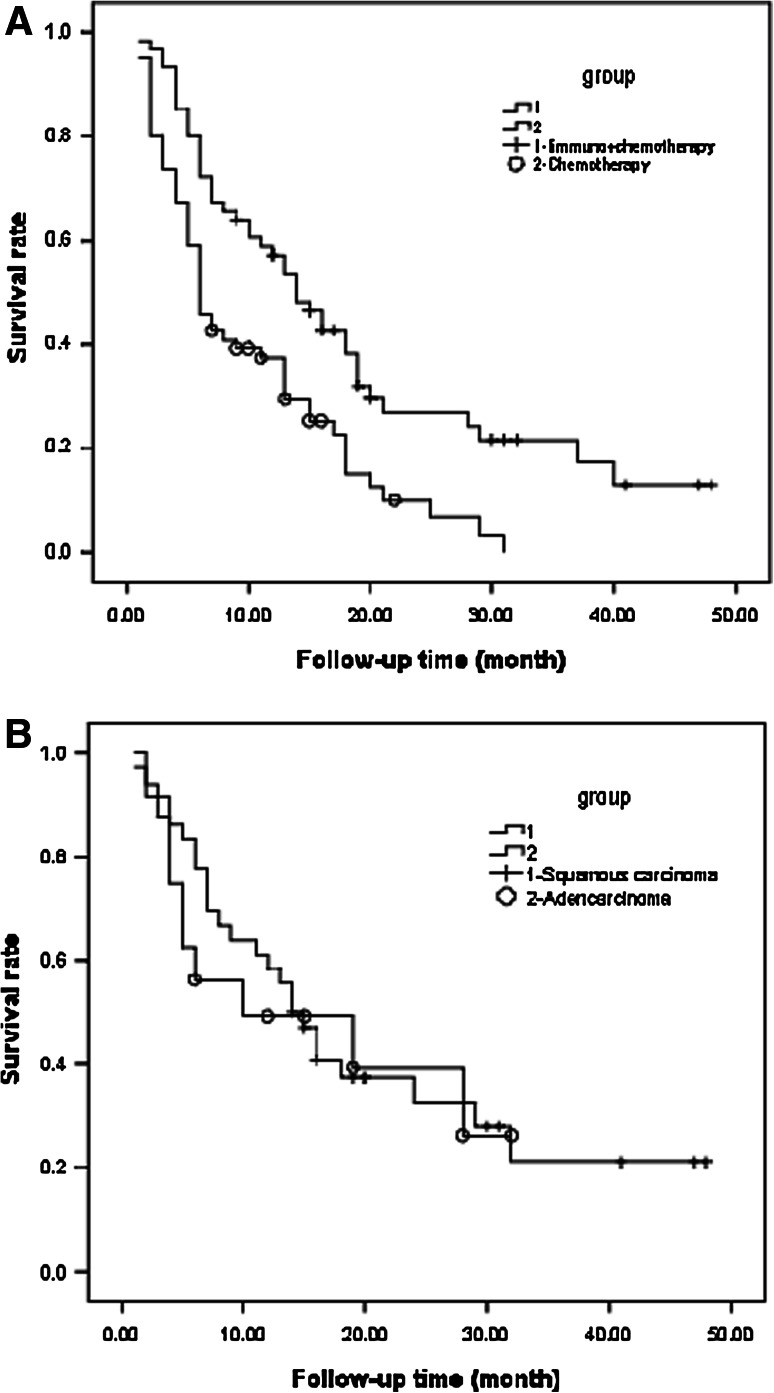
To compare the clinical effect of chemotherapy plus DC + CIK cells to chemotherapy alone, a paired study was performed on 122 patients where the 1- and 2-year overall survival rates were 37.3 and 10.1 % in group 1 and 57.2 and 27.0 % in group 2, respectively. Compared to chemotherapy alone, chemotherapy plus DC + CIK cells enhanced the clinical efficiency of chemotherapy for advanced NSCLC patients (P < 0.05, Fig. 4a).
Fig. 4.
The analysis of the overall survival in advanced NSCLC patients. a The survival curve of two groups in the paired study. One hundred and twenty-two patients were enrolled in this study. The 1- and 2-year overall survival rates were 37.3 and 10.1 % in group 1 and 57.2 and 27.0 % in group 2, respectively. Compared to chemotherapy alone, chemotherapy plus DC + CIK cells prolonged the overall survival of patients (P < 0.05). b The survival curve for adenocarcinoma and squamous carcinoma patients treated with chemotherapy plus DC + CIK cells. Thirty-six patients with adenocarcinoma and 18 patients with squamous carcinoma were enrolled in the group 2 of this study. The 1- and 2-year overall survival rates were 58.3 and 30.9 % for adenocarcinoma and 49.2 and 39.4 % for squamous carcinoma, respectively. There was no significant difference between the two types of pathology (P > 0.05)
Cox regression analysis of these 61 patients from group 2 showed that age (P = 0.09) and sex (P = 0.81) did not significantly impact the survivorship. We also compared the overall survival rate between 36 patients with adenocarcinoma and 18 patients with squamous carcinoma. The 1- and 2-year overall survival rates were 58.3 and 30.9 % for adenocarcinoma and 49.2 and 39.4 % for squamous carcinoma, respectively. There was no significant difference between the two types of pathology (P > 0.05) (Fig. 4b). Cox regression analysis of 36 adenocarcinoma patients showed that the overall survival rate was associated with age and the rate of proportional hazard increases by 4 % with one-year increase in age, assuming all other variables constant (P = 0.04). However, sex did not significantly impact survivorship (P = 0.50) for 36 patients with adenocarcinoma. For the 18 patients with squamous carcinoma, age (P = 0.94) and sex (P = 0.52) did not significantly impact survivorship.
For this cohort, the clinical response was assessed in all 61 patients in group 1 and 2, respectively. The ORR of group 1 and group 2 was 17 and 18 %, respectively (P > 0.05). The DCR of group 2 (69 %) was significantly higher compared with that of group I (49 %) (P < 0.05, Table 2).
Table 2.
Clinical outcomes of patients in two groups
| ORR (CR + PR) | DCR (CR + PR + SD) | CR | PR | SD | PD | |
|---|---|---|---|---|---|---|
| Chemotherapy alone (group 1) | 10 (17 %) | 30 (49 %) | 0 | 10 | 20 | 31 |
| Chemotherapy plus DC + CIK cells (group 2) | 11 (18 %) | 42 (69 %)* | 0 | 11 | 29 | 21 |
Assessment for clinical response was carried out in each 61 patients of group 1 and 2
ORR Overall response rate, DCR disease control rate, CR complete response, PR partial response, SD stable disease, PD progressive disease
* P < 0.05 compared with chemotherapy alone
During chemotherapy, the patients presented the usual side effects such as temporary fever, headache, chills or anemia, nausea, and vomiting. No severe adverse reactions, such as high fever, chills, or anemia, occurred in patients during or after DC-activated CIK cell transfusions were observed. Nonspecific symptoms, including fever, could be relieved naturally within 24 h without any medical treatment.
Discussion
Cellular immunotherapy, which directly or indirectly regulates the biological interaction between the host and the tumor [18, 19], is a potential strategy for the improvement of cancer treatment. Among them, CIK cells represent a promising cellular immunotherapy [20] since they may theoretically kill the residual chemotherapy-resistant tumor cells and hence be beneficial to patients with advanced disease. These cells have previously shown significant antitumor activity in preclinical experiments and animal tumor models [8, 13]. More recently, a study on the international registry on CIK cells (IRCC) reported that adjuvant immunotherapy with this cellular modality may prevent recurrence and improve quality of life and progression-free survival rates in patients with cancer [21]. DCs can further increase the populations of these hallmark-CD3+CD56+ effectors cells and at the same time decrease the proportion of CD4+CD25+ cells that have potent immune suppressive function [22]. Furthermore, our study indicates that antigen-loaded DC enhances the cytotoxicity of CIK cells against tumor cells. Therefore, we performed a clinical protocol to administer the chemotherapy plus DC + CIK cells in advanced NSCLC patients to compare efficiency as compared to the standard of care.
In the paired study, chemotherapy plus DC + CIK cells prolonged the overall survival rate compared with chemotherapy alone. It demonstrates that this combined treatment is more beneficial to the patient compared to chemotherapy alone. For the advanced NSCLC patients treated with CIK cells, it has been reported that the 1- and 2-year overall survival rates are 50.0 and 13.3 % in chemotherapy group and 58.6 and 20.6 % in CIK plus chemotherapy group, respectively [23]. However, their 1-year survival postchemotherapy in this study (50.0 %) was higher than our present study (37.3 %) likely due to the differences in the characteristics of patients and different chemotherapeutic regimens between their study and ours. However, the 1-year overall survival rate of the combined chemotherapy plus CIK therapy in that study was 58.6 % which is close to our observed 1-year survival rate (57 %) of chemotherapy plus DC + CIK cells. However, the 2-year overall survival rate (27.2 %) of our study of chemotherapy plus DC + CIK cells is higher than their study (20.6 %) of chemotherapy plus CIK indicating that DC-activated CIK combined with chemotherapy is more beneficial than CIK plus chemotherapy. Furthermore, our previous study showed that the 2-year overall survival rate for NSCLC patients after surgery is 94.7 % in DC-activated CIK in combination with chemotherapy group compared with the chemotherapy alone group (78.8 %) [14]. These data indicated that NSCLC patients who have undergone surgery may benefit more than the advanced NSCLC patients when treated with chemotherapy plus DC + CIK cell therapy compared to chemotherapy alone.
For the NSCLC patients, adenocarcinoma accounts for a significant percentage of cases worldwide, which accounts for about 40 % of cases. And 25–30 % of primary lung cancers are squamous cell carcinomas. So, we also compared the survival rate between adenocarcinoma and squamous carcinoma patients treated with chemotherapy plus DC + CIK cells. Although there was no statistical significance between the two types of pathology, the 1-year overall survival rate of adenocarcinoma patients was higher than that of squamous carcinoma patients. Meanwhile, the 2-year overall survival rate of adenocarcinoma patients was lower than that of squamous carcinoma patients. Cox regression analysis of 36 patients with adenocarcinoma showed that age significantly impact survivorship, but for 18 patients with squamous carcinoma, age and sex did not significantly impact the survivorship. However, in our present study, only 18 patients with squamous carcinoma were enrolled. Therefore, more patients will be needed to get more convincing data in the future study.
The immunological effects of chemotherapy plus DC + CIK cells in ten patients were investigated. There were no obvious changes in CD3+, CD3+CD4+, and CD3+CD8+ between the patients before and after therapy but statistical increase in CD3+CD56+ cells after therapy. However, we observed that CD4+CD25+ cells were decreased after treatment (P > 0.05). Tregs (CD4+CD25+) play a critical role in maintaining immune tolerance and suppressing autoimmune diseases [23]. It has been shown to inhibit cytotoxic lymphocyte-mediated immune responses. However, it is possible that a decrease in CD4+CD25+ cells may not represent Tregs because we did not examine Foxp3 expression, and CD4 and CD25 are markers shared with T helper cells. It is also likely that the decrease in CD4+CD25+ cells is not indicative of T helper cells because a reduction in T helper cells would not explain the immunological responses presented. In future studies, Foxp3 analysis will be included along with CD4 and CD25 to verify the reduction in Tregs. In the present study, only 10 patients were analyzed, and hence, further studies with more patients will be needed in the future studies. After chemotherapy plus DC + CIK cell therapy, there were statistical increase in the secretion of IFN-γ and TNF-α and decrease in TGF-β secretion and without significant changes in IL-10 secretion. IFN-γ, a hallmarker of Th1 cells, has been shown to enhance expression of the major histocompatibility complex (MHC) I molecule and improve cytotoxicity [24]. TNF-α, another Th1 cytokine produced by activated T cells, is able to induce tumor cell necrosis and enhance the activity of NK and T cells [25]. The serum levels of these two Th1 cytokines were increased after DC + CIK cell therapy, which indicated the activation of T cells and induction of immune response by the host. TGF-β can inhibit the proliferation and differentiation of T cells and B cells and reduce the activity of NK cells and Ag-presenting cells [14, 24]. So, our study indicated that chemotherapy plus DC + CIK cells inhibit production of cytokine which serve as immune suppressors.
In conclusion, the present study suggests that DC + CIK cells have enhanced antitumor effects and chemotherapy plus DC + CIK cells improved the clinical outcomes of conventional chemotherapy for advanced NSCLC patients. No severe side effects were recorded during DC-activated CIK cell treatment, indicating that chemotherapy plus DC + CIK cells is a safe form of therapy. In the future studies, a larger cohort randomized trial will be needed to investigate the clinical efficacy and to elucidate the detailed mechanism for this line of treatment.
Acknowledgments
This work was supported by a grant from major scientific and technological project of Science and Technology Commission Foundation of Tianjin (No. 043115211-2) and Health Department Fund project of Tianjin (No. 09KZ80).
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Lili Yang and Baozhu Ren contributed equally to this work.
Contributor Information
Xishan Hao, Email: xishanhao@gmail.com.
Xiubao Ren, Phone: +86-022-23340123, FAX: +86-022-23537796, Email: xiubaoren@gmail.com.
References
- 1.Rosti G, Carminati O, Monti M, Tamberi S, Marangolo M. Chemotherapy advances in small cell lung cancer. Ann Oncol. 2006;17(Suppl 5):v99–v102. doi: 10.1093/annonc/mdj961. [DOI] [PubMed] [Google Scholar]
- 2.Grilli R, Oxman AD, Julian JA. Chemotherapy for advanced non-small-cell lung cancer: how much benefit is enough? J Clin Oncol. 1993;11:1866–1872. doi: 10.1200/JCO.1993.11.10.1866. [DOI] [PubMed] [Google Scholar]
- 3.Choudhury A, Palma M, Mellstedt H. The future of cancer vaccines for non-small-cell lung cancer: ongoing trials. Clin Lung Cancer. 2008;9(Suppl 1):S37–S44. doi: 10.3816/CLC.2008.s.006. [DOI] [PubMed] [Google Scholar]
- 4.Mac Manus MP, Matthews JP, Wada M, Wirth A, Worotniuk V, Ball DL. Unexpected long-term survival after low-dose palliative radiotherapy for non-small cell lung cancer. Cancer. 2006;106:1110–1116. doi: 10.1002/cncr.21704. [DOI] [PubMed] [Google Scholar]
- 5.Penland SK, Socinski MA. Management on unresectable stage III non-small cell lung cancer: the role of combined chemoradiation. Semin Radiat Oncol. 2004;14:326–334. doi: 10.1016/j.semradonc.2004.07.002. [DOI] [PubMed] [Google Scholar]
- 6.Märten A, Ziske C, Schöttker B, Renoth S, Weineck S, Buttgereit P, Schakowski F, von Rücker A, Sauerbruch T, Schmidt-Wolf IG. Interactions between dendritic cells and cytokine-induced killer cells lead to an activation of both populations. J Immunother. 2001;24:502–510. doi: 10.1097/00002371-200111000-00007. [DOI] [PubMed] [Google Scholar]
- 7.Zhang YS, Yuan FJ, Jia GF, Zhang JF, Hu LY, Huang L, Wang J, Dai ZQ. CIK cells from patients with HCC possess strong cytotoxicity to multidrug-resistant cell line Bel-7402/R. World J Gastroenterol. 2005;11:3339–3345. doi: 10.3748/wjg.v11.i22.3339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ren X, Yu J, Liu H, Zhang P, An X, Zhang N, Hao X. Th1 bias in PBMC induced by multicycles of auto-CIKs infusion in malignant solid tumor patients. Cancer Biother Radiopharm. 2006;21:22–33. doi: 10.1089/cbr.2006.21.22. [DOI] [PubMed] [Google Scholar]
- 9.Schmidt-Wolf IG, Negrin RS, Kiem HP, Blume KG, Weissman IL. Use of a SCID mouse/human lymphoma model to evaluate cytokine-induced killer cells with potent antitumor cell activity. J Exp Med. 1991;174:139–149. doi: 10.1084/jem.174.1.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Verneris MR, Kornacker M, Mailänder V, Negrin RS. Resistance of ex vivo expanded CD3 + CD56 + T cells to Fas-mediated apoptosis. Cancer Immunol Immunother. 2000;49:335–345. doi: 10.1007/s002620000111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lu PH, Negrin RS. A novel population of expanded human CD3 + CD56 + cells derived from T cells with potent in vivo antitumor activity in mice with severe combined immunodeficiency. J Immunol. 1994;153:1687–1696. [PubMed] [Google Scholar]
- 12.Scheffold C, Brandt K, Johnston V, Lefterova P, Degen B, Schöntube M, Huhn D, Neubauer A, Schmidt-Wolf IG. Potential of autologous immunologic effector cells for bone marrow purging in patients with chronic myeloid leukemia. Bone Marrow Transplant. 1995;15:33–39. doi: 10.1038/jcbfm.1995.4. [DOI] [PubMed] [Google Scholar]
- 13.Leemhuis T, Wells S, Scheffold C, Edinger M, Negrin RS. A phase I trial of autologous cytokine-induced killer cells for the treatment of relapsed Hodgkin disease and non-Hodgkin lymphoma. Biol Blood Marrow Transplant. 2005;11:181–187. doi: 10.1016/j.bbmt.2004.11.019. [DOI] [PubMed] [Google Scholar]
- 14.Li H, Wang C, Yu J, Cao S, Wei F, Zhang W, Han Y, Ren XB. Dendritic cell-activated cytokine-induced killer cells enhance the anti-tumor effect of chemotherapy on non-small cell lung cancer in patients after surgery. Cytotherapy. 2009;11:1076–1083. doi: 10.3109/14653240903121252. [DOI] [PubMed] [Google Scholar]
- 15.Schmidt-Wolf IG, Finke S, Trojaneck B, Denkena A, Lefterova P, Schwella N, Heuft HG, Prange G, Korte M, Takeya M, Dorbic T, Neubauer A, Wittig B, Huhn D. Phase I clinical study applying autologous immunological effector cells transfected with the interleukin-2 gene in patients with metastatic renal cancer, colorectal cancer and lymphoma. Br J Cancer. 1999;81:1009–1016. doi: 10.1038/sj.bjc.6690800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fields RC, Shimizu K, Mulé JJ. Murine dendritic cells pulsed with whole tumor lysates mediate potent antitumor immune responses in vitro and in vivo. Proc Natl Acad Sci USA. 1998;95:9482–9487. doi: 10.1073/pnas.95.16.9482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981;47:207–214. doi: 10.1002/1097-0142(19810101)47:1<207::AID-CNCR2820470134>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 18.Wu C, Jiang J, Shi L, Xu N. Prospective study of chemotherapy in combination with cytokine-induced killer cells in patients suffering from advanced non-small cell lung cancer. Anticancer Res. 2008;28:3997–4002. [PubMed] [Google Scholar]
- 19.Herberman RB. Cancer therapy by biological response modifiers. Clin Physiol Biochem. 1987;5:238–248. [PubMed] [Google Scholar]
- 20.Sangiolo D, Martinuzzi E, Todorovic M, Vitaggio K, Vallario A, Jordaney N, Carnevale-Schianca F, Capaldi A, Geuna M, Casorzo L, Nash RA, Aglietta M, Cignetti A. Alloreactivity and anti-tumor activity segregate within two distinct subsets of cytokine-induced killer (CIK) cells: implications for their infusion across major HLA barriers. Int Immunol. 2008;20:841–848. doi: 10.1093/intimm/dxn042. [DOI] [PubMed] [Google Scholar]
- 21.Hontscha C, Borck Y, Zhou H, Messmer D, Schmidt-Wolf IG. Clinical trials on CIK cells: first report of the international registry on CIK cells (IRCC) J Cancer Res Clin Oncol. 2011;137:305–310. doi: 10.1007/s00432-010-0887-7. [DOI] [PubMed] [Google Scholar]
- 22.Gallimore A, Godkin A. Regulatory T cells and tumour immunity—observations in mice and men. Immunology. 2008;123:157–163. doi: 10.1111/j.1365-2567.2007.02748.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li H, Yu JP, Cao S, Wei F, Zhang P, An XM, Huang ZT, Ren XB. CD4 +CD25 + regulatory T cells decreased the antitumor activity of cytokine-induced killer (CIK) cells of lung cancer patients. J Clin Immunol. 2007;27:317–326. doi: 10.1007/s10875-007-9076-0. [DOI] [PubMed] [Google Scholar]
- 24.Wahl SM, Wen J, Moutsopoulos N. TGF-beta: a mobile purveyor of immune privilege. Immunol Rev. 2006;213:213–327. doi: 10.1111/j.1600-065X.2006.00437.x. [DOI] [PubMed] [Google Scholar]
- 25.Galliher AJ, Neil JR, Schiemann WP. Role of transforming growth factor-beta in cancer progression. Future Oncol. 2006;2:743–763. doi: 10.2217/14796694.2.6.743. [DOI] [PubMed] [Google Scholar]