Abstract
The radiolabeled somatostatin analogue, yttrium-90 DOTA-d-Phe(1)-Tyr(3)-octreotide (DOTATOC), is currently applied to treat advanced somatostatin receptor-positive tumors, e.g., neuroendocrine tumors of the pancreas, lung or gut. However, effects of this treatment on antimicrobial immune responses are not yet defined. In 20 patients treated with DOTATOC, cellular in vitro immune function was determined. Their antimicrobial lymphocyte responses were assessed by lymphocyte transformation test and enzyme-linked immunospot—measuring lymphocyte proliferation and on a single cell level production of pro- and anti-inflammatory cytokines (interferon-γ and interleukin-10)—prior to therapy, at day 1, day 7 and day 90 post-therapy. Proliferative lymphocyte responses and interferon-γ production after in vitro stimulation with microbial antigens were non-significantly suppressed at day 1 and significantly (p < 0.05) at day 7 versus pre-therapy. In vitro immune responses did not fully recover until day 90. In contrast, at day 1 interleukin-10 production was significantly (p < 0.05) increased. Taken together, we observed a decrease in pro-inflammatory immune responses after DOTATOC therapy. Patients with versus without bone metastases displayed significantly (p < 0.05) lower cellular immune responses toward several microbial antigens. Progressive disease and higher tumor burden could also be defined as factors associated with impaired immune function. Spearman correlation analysis indicated that cellular in vitro immunity was positively correlated with kidney function; better kidney function led to stronger immune responses. In conclusion, DOTATOC therapy caused a decrease in in vitro immune responses against microorganisms. The clinical impact needs to be evaluated in further studies.
Keywords: DOTATOC therapy, Antimicrobial immune response, Lymphocyte proliferation, ELISpot, Cytokine
Introduction
The number of indications for the application of radionuclides in tumor therapy is constantly growing. In particular, peptide receptor radionuclide therapy is increasingly used as a treatment modality for certain neoplasias such as neuroendocrine tumors. These tumors often exhibit an overexpression of somatostatin receptors compared to normal tissues. Those receptors can be targeted with different synthetic somatostatin analogues. Many of these peptidic analogues can be conjugated with chelators and labeled with radiometals. The basis of this treatment is then the binding of the radiolabeled compound to the receptor, the subsequent internalization of the receptor–ligand complex and the intracellular emission of ionizing radiation.
A somatostatin analogue that is frequently used in peptide receptor radionuclide therapy of neuroendocrine tumors is yttrium-90 DOTA-d-Phe(1)-Tyr(3)-octreotide (DOTATOC). It contains the chelating agent 1,4,7,10-tetraazacyclododecane-N,N′, N″,N‴-tetraacetic acid (DOTA) to label Tyr(3)-octreotide (TOC) with yttrium-90 (90Y) [1]. 90Y is an energetic beta particle emitter that requires enhanced stability of the radionuclide complex to minimize in vivo release of high levels of free 90Y and, in turn, bone marrow toxicity [2, 3]. Conjugates of 90Y and octreotide can be stabilized, e.g., by DOTA [4, 5]. Octreotide is an octapeptide that mimics natural somatostatin pharmacologically, though it is a more potent inhibitor of growth hormone, glucagon and insulin than the natural hormone. 90Y DOTATOC is considered a safe tool for receptor-mediated radiopeptide therapy of somatostatin receptor-overexpressing tumors [1]. In these tumors, intravenous administration of 90Y DOTATOC is a highly targeted and effective therapy where minimal side effects are described [1, 6].
Recently, it could be demonstrated that radioiodine therapy in patients with thyroid carcinoma led to a short-term increase in pro-inflammatory immune responses [7]. However, radioiodine-induced changes were moderate and only observed at day 1 post-therapy. We now addressed the question whether treatment with DOTATOC alters antimicrobial immune responses in a similar way.
We are not aware of any study investigating the effect of DOTATOC on immune defense in a clinical setting. But it has been known for many years that the immune system is highly sensitive to gamma rays [8–12] which cause, e.g., chromosomal aberrations [10], reduced reagibility of lymphocytes [11] or reduced levels of all lymphocyte subsets, with B lymphocytes and naïve (unprimed) T lymphocytes being most sensitive [12]. To assess the effect of irradiation, the kind of radiation, its specific activity and the activity distribution have to be considered [7, 12].
In the present study, DOTATOC induces continuous but inhomogeneous irradiation that acts not only locally but to some extent also on the whole body. The target tissue expressing the somatostatin receptor is exposed to a high dose of DOTATOC, whereas the exposure of the remaining tissue is low. Particularly, patient-specific bone marrow dosimetry is challenging [13]. Current literature suggests that DOTATOC therapy as applied in our study (4.0–5.9 GBq) leads to anemia, leukopenia and thrombocytopenia in approximately 60, 30 and 30 % of patients, respectively [14]. The 90Y dose considered the generally accepted maximum bone marrow dose of 2 Gy [15]. With a dose of 2 Gy, the probability for developing leukemia is approximately 2 % [16].
To measure immunoreactivity after intense gamma irradiation, the highly sensitive lymphocyte transformation test (LTT) has been used as a gold standard [7, 10, 11]. This test measures cell proliferation and determines the uptake of 3H thymidine by peripheral blood mononuclear cells (PBMCs) after in vitro stimulation. Various groups of stimuli can be used, e.g., mitogens which can stimulate naïve as well as memory T lymphocytes or microbial antigens (recall antigens) which can only stimulate memory T lymphocytes. Response to the LTT mitogen can determine the overall reactivity of lymphocytes (almost all T/B lymphocytes should proliferate if stimulated by mitogens), and the LTT antigen can determine whether microorganisms are recognized adequately by T lymphocytes. A proper in vitro reaction toward antigens indicates that in vivo infections should also be controlled effectively by proliferating T lymphocytes.
Recently, it was shown that the enzyme-linked immunospot (ELISpot), a sensitive method which determines cytokine production on a single cell level, is an excellent tool to quantify effects of irradiation on the immune system [7]. The pattern of cytokines produced allows suggesting whether the immune system is more in an inflammatory or an immunosuppressed condition. An inflammatory state is characterized by the production of, e.g., interferon (IFN)-γ, an immunosuppressed by the production of, e.g., interleukin (IL)-10. IFN-γ production is a prerequisite for the control of microorganisms, whereas IL-10 production facilitates the persistence of infections.
It was the aim of the present study to define for the first time how DOTATOC therapy affects lymphocyte function in patients with advanced somatostatin receptor-positive tumors in the short- and long-term (day 1–7 and day 90 post-treatment, respectively). Further, we considered potential influencing factors of cellular in vitro immunity.
Methods and materials
Patients
Twenty patients receiving DOTATOC therapy because of advanced somatostatin receptor-positive malignant tumors were consecutively included into this study from January to August 2010. Patient characteristics are detailed in Table 1. For treatment planning with 90Y DOTATOC, hybrid imaging of positron emission tomography together with computed tomography (PET/CT) was performed prior to therapy with gallium-68 (68Ga) DOTATOC (Fig. 1). Because of the disseminated nature of the disease, the tumor burden was estimated visually on an ordinal scale. The glomerular filtration rate was determined with technetium-99m diethylene-triamine-pentaacetate (DTPA) using the two-sample, two-compartment technique described by Russel [17]. The applied activity of 90Y DOTATOC was calculated according to the national guidelines (in press) and was influenced by neither tumor burden nor kidney function. Assays to detect cellular immunity were performed immediately prior to therapy (day 0) and at day 1, 7 and 90 post-therapy.
Table 1.
Characteristics of 20 patients with advanced somatostatin receptor-positive tumors receiving DOTATOC therapy
| Female/male | 8/12 |
| Median age (range), years | 66 (23–74) |
| Median activity of current therapy cycle (range), GBq | 4.8 (4.0–5.9) |
| Therapy cycle, no. | |
| 1st | 13 |
| 2nd | 2 |
| 3rd | 5 |
| Activity of all therapy cycles, no. | |
| >4 and ≤5 GBq | 11 |
| >5 and ≤10 GBq | 3 |
| >10 and ≤16 GBq | 6 |
| Primary tumor | |
| Pancreas | 5 |
| Lung | 4 |
| Gut | 5 |
| Paraganglioma | 2 |
| Thyroid | 2 |
| Other | 2 |
| With/without bone metastases | 13/7 |
| Glomerular filtration rate prior to therapya | |
| Within/below age-dependent norm | 17/3 |
| Median (range), (ml/min/1.73 m2) | 95 (55–112) |
| Response to therapy | |
| Stable disease | 10 |
| Progression | 10 |
aThe glomerular filtration rate was determined by radioisotope nephrography
Fig. 1.
68Ga DOTATOC positron emission tomography (PET) in 20 patients prior to 90Y DOTATOC therapy. The images were arranged according to the tumor burden
Lymphocyte transformation test
Cellular in vitro responses toward four mitogens [phytohemagglutinin, concanavalin A, pokeweed mitogen and anti-CD3 monoclonal antibody (OKT3)] and twelve microbial antigens (tuberculin, tetanus toxoid, diphtheria toxoid, Candida albicans, cytomegalovirus, Herpes simplex virus type 1, Varicella zoster virus, rubella virus, mumps virus, measles virus, influenza A and B virus) were measured using standardized assay formats as described previously in greater detail [7, 18]. In brief, PBMCs were collected after density gradient centrifugation, and 50,000 cells per 200 μL were incubated with mitogens for 3 days and antigens for 5 days, respectively. For the last 16 h, the cultures were labeled with 37 kBq 3H thymidine per culture. Cells were then harvested onto filter pads, and the incorporated radioactivity was quantified by liquid scintillation counting.
ELISpot assay
The secretion of IFN-γ and IL-10 was detected by ELISpot as also detailed previously [7]; 200,000 PBMCs per 200 μL culture were stimulated by four mitogens (same as for LTT) and 400,000 PBMCs per 200 μL culture by three microbial antigens [tuberculin, tetanus toxoid and Candida albicans]. After 2 days of cell culture, spot formation was detected by adding avidin–biotin peroxidase complex and the substrate 3-amino-9-ethyl-carbazole. Numbers of spots were analyzed by an ELISpot plate reader.
Statistical analysis
LTT data were presented as counts per minute (cpm), representing the uptake of 3H thymidine, and ELISpot data as spot numbers per cell culture. Cpm increment or spots increment was generated as mitogen stimulated or antigen stimulated minus autologous (unstimulated) reaction. In addition, the cumulative antigen score was generated by an algorithm described previously [18]; it sums up reactions toward twelve recall antigens. Reactions pre- and post-DOTATOC therapy were compared by paired t test or Wilcoxon matched pairs test as appropriate. Correlation of numeric clinical parameters and immune function was analyzed using Spearman correlation analysis. The influence of categorical parameters was tested by Mann–Whitney U test. All these data were analyzed by GraphPad Prism software (version 3.02).
Results
Immune response after DOTATOC therapy
In 20 patients receiving DOTATOC therapy, inflammatory T lymphocyte responses toward mitogens and microbial antigens continuously decreased until day 7 post-therapy (Fig. 2a, b). The data indicate a DOTATOC-induced impairment of antimicrobial immune responses. In detail, at day 1 after therapy lymphocyte responses were moderately suppressed. At day 7, lymphocyte proliferation (LTT) after stimulation with all four mitogens tested and with the microbial antigens Herpes simplex virus type 1, influenza A and influenza B virus was significantly lower than at day 0 (p < 0.05 each). Thereafter, at day 90, responses still tended to be lower than pre-therapy. This means that therapy induced a short-term decrease in specific lymphocyte proliferation.
Fig. 2.

Cellular in vitro reaction toward mitogens and recall antigens in patients with somatostatin receptor-positive tumors receiving 90Y DOTATOC therapy. Data represent mean and standard error of the mean (SEM), either pre-therapy (day 0, white bars), at day 1 (black bars), at day 7 (hatched bars) or at day 90 (striped bars) post-therapy. LTT mitogen responses (a, n = 18–20) and LTT antigen responses (b, n = 18–20) are given as counts per minute of 3H thymidine uptake (cpm), IFN-γ and IL-10 secretion to the ELISpot (c–f, n = 15–20) as spots per culture. The Wilcoxon matched pairs test was used for comparisons; the level of significance is indicated in comparison with day 0 (*p < 0.05, **p < 0.01). LTT lymphocyte transformation test, Aut. autologous, PHA phythohemagglutinin, ConA concanavalin A, PWM pokeweed mitogen, OKT3 anti-CD3 monoclonal antibody, PPD purified protein derivate (tuberculin), TET tetanus toxoid, CAN Candida albicans, HSV-1 Herpes simplex virus type 1, VZV Varicella zoster virus, INV-A influenza virus A, INV-B influenza virus B, IFN interferon, IL interleukin
IFN-γ secretion as determined by ELISpot was also significantly (p < 0.05 each) decreased at day 7 for the mitogens concanavalin A and pokeweed mitogen and the antigen Candida albicans (Fig. 2c, d). Furthermore, at day 1 IL-10 secretion to the ELISpot was increased (p < 0.05 each for phytohemagglutinin, pokeweed mitogen and tuberculin) (Fig. 2e, f).
Influence of DOTATOC therapy on white cell counts
DOTATOC therapy led to a significant (p < 0.05) decrease in PBMC numbers on day 1 and day 7 post-therapy [day 0: 9.7 ± 1.0, day 1: 8.2 ± 0.9, day 7: 7.1 ± 0.8 million PBMC per 10 mL peripheral blood, data represent the mean and standard error of the mean (SEM), n = 19–20]. On day 90, cell numbers were comparable to those on day 1 (8.2 ± 0.9 million PBMC per 10 mL blood, n = 18). White blood counts at day 7 versus day 0 indicated that lymphocytes were significantly reduced (0.7 ± 0.1 vs. 1.2 ± 0.2/nL, n = 15, p = 0.0002), whereas leukocyte and granulocyte numbers did not change significantly (5.8 ± 0.3 vs. 6.1 ± 0.3/nL and 4.3 ± 0.3 vs. 4.2 ± 0.3/nL). However, leukocyte numbers tended to decrease and the percentage of granulocytes tended to increase (74 vs. 69 %).
Correlation analysis of clinical parameters and cellular in vitro immune responses
To assess the dependency of immune function on clinical parameters, all patient characteristics as given in Table 1 were included into the analysis. Significant results are displayed in Fig. 3 and Table 2.
Fig. 3.

Immune responses toward microbial antigens are dependent on the presence of bone metastases in patients with 90Y DOTATOC therapy. In vitro immune responses toward tetanus toxoid and Candida albicans were determined immediately prior to therapy (day 0), at day 1, day 7 and day 90 post-therapy. Patients with bone metastases (n = 13, black bars) displayed significantly lower immune responses as compared to patients without bone metastases (n = 7, white bars). Immunity was determined by lymphocyte transformation test as proliferation (LTT, a, b) and by ELISpot as interferon (IFN)-γ production per cell (c, d). The Mann–Whitney test was used for comparisons between patient groups (*p < 0.05, **p < 0.005). Not tested at day 7: n = 1 and at day 90: n = 2. Cpm: counts per minute; increment: The background reaction (autologous value) was subtracted from the specific reaction toward tetanus toxoid or Candida albicans
Table 2.
Spearman correlation analysis of kidney functiona and cellular in vitro immunity in 20 patients receiving DOTATOC therapy
| Parameter of immune function | Time point (day) | r | p |
|---|---|---|---|
| ConA LTT | 1 | 0.47 | 0.04 |
| 90 | 0.59 | 0.009 | |
| PWM LTT | 1 | 0.45 | <0.05 |
| 90 | 0.61 | 0.008 | |
| OKT3 LTT | 90 | 0.70 | 0.001 |
| PPD LTT | 1 | 0.61 | 0.004 |
| 7 | 0.69 | 0.001 | |
| 90 | 0.63 | 0.005 | |
| CAN LTT | 1 | 0.49 | 0.03 |
| 7 | 0.61 | 0.005 | |
| 90 | 0.48 | <0.05 | |
| INV-A LTT | 1 | 0.45 | <0.05 |
| 7 | 0.54 | 0.02 | |
| INV-B LTT | 1 | 0.51 | 0.02 |
| 7 | 0.55 | 0.02 | |
| 90 | 0.61 | 0.008 | |
| Cum. antigen score | 1 | 0.50 | 0.03 |
| 7 | 0.65 | 0.003 | |
| PWM IFN-γ ELISpot | 90 | 0.60 | 0.009 |
| PPD IFN-γ ELISpot | 0 | 0.54 | 0.02 |
| 1 | 0.77 | <0.0001 | |
| 7 | 0.60 | 0.007 | |
| 90 | 0.61 | 0.007 | |
| CAN IFN-γ ELISpot | 7 | 0.59 | 0.008 |
| 90 | 0.51 | 0.03 | |
| ConA IL-10 ELISpot | 0 | 0.50 | 0.02 |
| 1 | 0.51 | 0.03 |
ConA concanavalin A, LTT lymphocyte transformation test, PWM pokeweed mitogen, OKT3 anti-CD3 monoclonal antibody, PPD purified protein derivate (tuberculin), CAN Candida albicans, INV-A influenza virus A, INV-B influenza virus B, Cum. antigen score cumulative antigen score (sum of reactions toward antigens) [18], IFN interferon, IL interleukin
aKidney function was determined by radioisotope nephrography and was measured as glomerular filtration rate
Patients with bone metastases had significantly (p < 0.05) lower cellular immune reactivity toward several microbial antigens (tetanus toxoid, Candida albicans, Herpes simplex virus type 1, Varicella zoster virus and influenza A virus) and toward a set of twelve recall antigens (cumulative antigen score). Figure 3 depicts reactions toward two of these recall antigens, tetanus toxoid and Candida albicans. Differences were observed at all time points; i.e., patients with bone metastases had lower immune function pre- and post-therapy.
A similar result was observed for patients with progressive versus stable disease; reduced T cell responses were detectable in patients with progressive disease pre- and post-therapy. These patients had lower T lymphocyte proliferation after stimulation with microbial antigens. However, differences only reached statistical significance (p < 0.05) for Herpes simplex virus type 1.
Patients with higher versus lower tumor burden had impaired immune function. This was also observed pre- and post-therapy. Summing up reactions toward twelve recall antigens (cumulative antigen score), significant differences between patients with high and low tumor burden could be observed (p < 0.05). Differences for single antigens were all non-significant.
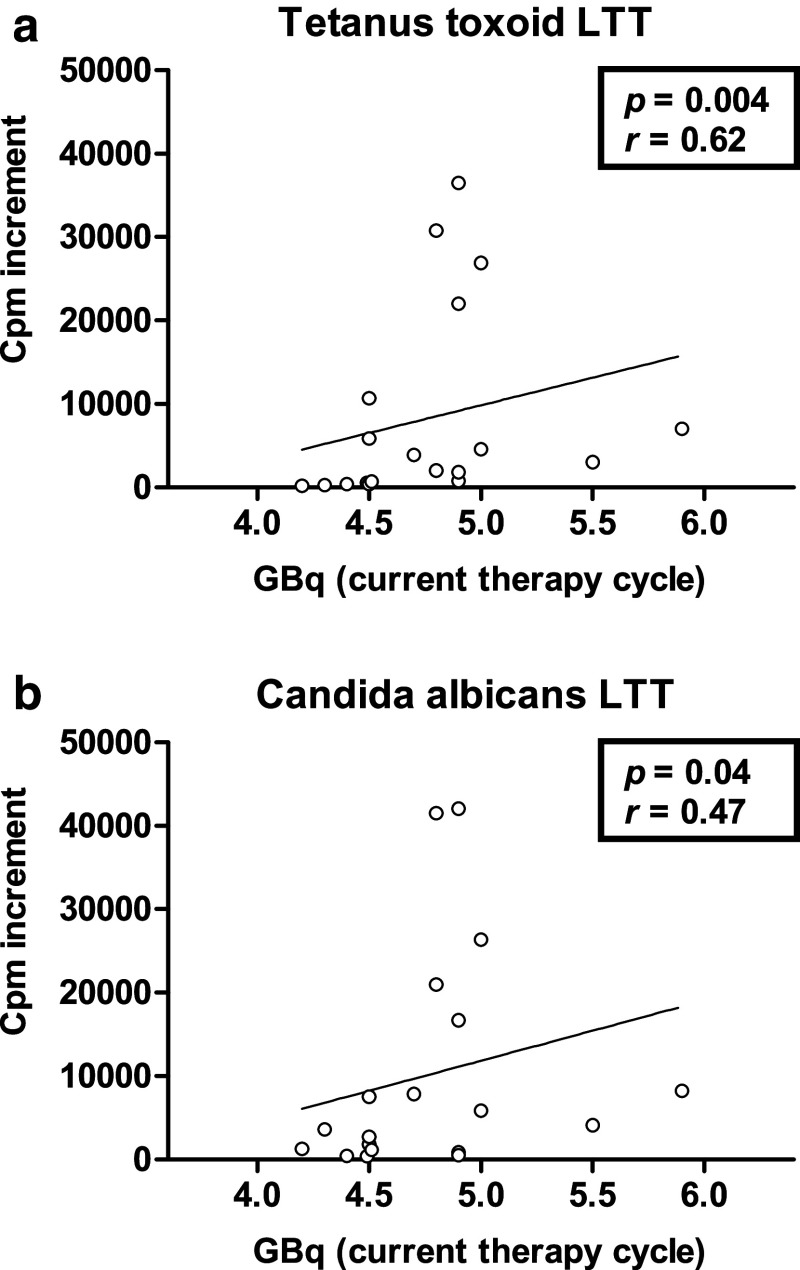
Interestingly, we also observed a significant correlation (p < 0.05) between activity of the current therapy cycle and immune function. T cell proliferation after stimulation with various antigens (tetanus toxoid, Candida albicans, Varicella zoster virus, influenza A and B virus), IFN-γ and IL-10 production were positively (r values between 0.45 and 0.66) correlated with the current activity (Fig. 4).
Fig. 4.
Immune responses toward microbial antigens are dependent on the activity of the current therapy cycle in patients with 90Y DOTATOC therapy. T cell proliferation as determined by lymphocyte transformation test (LTT) at day 7 post-therapy was significantly correlated with the activity of the current therapy cycle. Spearman correlation analysis is shown for immune responses toward tetanus toxoid (a) and Candida albicans (b) (n = 19 each). Cpm: counts per minute; increment: The background reaction (autologous value) was subtracted from the specific reaction toward tetanus toxoid or Candida albicans
As expected, Spearman correlation analysis indicated that cellular in vitro immunity was positively correlated with kidney function; i.e., better kidney function led to better immune function (Table 2). Kidney function was determined by radioisotope nephrography and was measured as glomerular filtration rate.
Finally, gender, age, therapy cycle number, activity of all therapy cycles and primary tumor had no significant influence on immune function.
Summarizing, factors suppressing immune function comprised the presence of bone metastases, progressive disease, higher tumor burden and impaired kidney function.
Discussion
DOTATOC therapy led to a decrease in antimicrobial immune responses in vitro. We could observe a moderate immunosuppression at day 1 and a more pronounced suppression at day 7 after treatment. In contrast, a previous study on radioiodine therapy [7] showed that immune function was increased at day 1 after treatment and returned to baseline at day 7. Notably, we used exactly the same assay conditions for the radioiodine and DOTATOC study, and age and sex distribution were similar in both cohorts. Thus, the underlying disease and treatment appeared as the major differences. A direct comparison between the immunosuppressive effects of iodine-131 in thyroid cancer patients described in an earlier paper [7] and the current results for peptide receptor radionuclide therapy with 90Y DOTATOC is problematic because the biodistribution and kinetics of these radiopharmaceuticals differ substantially. Although 90Y DOTATOC has a substantially faster blood clearance than iodine-131 [19–21], only 60 % of the injected activity is excreted after 24 h and little more after 48 h [21], whereas the whole-body retention of iodine-131 is only 14 % after 48 h [22]. 90Y DOTATOC is retained with half-life >2 days in well-perfused organs such as liver, spleen and kidneys and irradiates circulating cells [20, 21]. Furthermore, the contribution to the dose of circulating immune cells is related to fractional cardiac output, mean transit time and activity concentration, whereas the dose to sessile cells is dominated by the binding and kinetics in the host organ and the circulating blood. The kinetics of the exchange between the pools of sessile and circulating cells is unknown. The biological radiation effects may be further confounded by changes in the biological effectiveness due to the described time-varying dose rates from different sources [23]. The development and validation of a rigorous dosimetric model of these effects are beyond the scope of this study.
At day 90 after treatment, lymphocyte responses were still slightly lower than pre-therapy. This finding is line with the long life span of CD4- and CD8-positive T lymphocytes, i.e., helper T and cytotoxic T lymphocytes. Using mathematical modeling, the median half-life time for naïve CD4- and CD8-positive T lymphocytes (stimulable with mitogens) was estimated as 4.2 and 6.5 years, and the median half-life time for CD4- and CD8-positive memory T lymphocytes (stimulable with mitogens and recall antigens) as 155 and 244 days, respectively [24]. Moreover, following total body irradiation and hematopoietic stem cell transplantation, it takes approximately 2.5 years until T lymphocyte responses recover [25]. Thus, it appears as likely that lymphocytes have not been completely replaced at day 90 after DOTATOC therapy.
Currently, there are no data on the effect of 90Y DOTATOC treatment on immune function. However, a previous study [26] analyzed the short- and long-term effects of another tumor therapy containing 90Y. It was shown that intra-arterial application of 90Y microspheres in patients with hepatocellular carcinoma was associated with profound and prolonged lymphopenia. Lymphocytes (and not granulocytes, platelets or NK cells) were sensitive to treatment with 90Y microspheres. Using 90Y microspheres, predominantly circulating cells are irradiated from 90Y in the liver as opposed to 90Y DOTATOC where the bone marrow is also exposed. Despite the different distribution of the radiopharmaceuticals, similar results on leukocyte subpopulations were observed after 90Y microsphere and 90Y DOTATOC therapy. Our study also showed that the percentage of lymphocytes was reduced after therapy, whereas the percentage of granulocytes tended to increase. Also in line with our data, no fungal or bacterial infections were noted after therapy with 90Y microspheres. Unfortunately, the authors of the 90Y microsphere study [26] did not present data on viral infections. In our study, an increase in viral infection/reactivation was not observed. Whereas fungal and bacterial infections are mainly controlled by granulocytes, viral infection/reactivation is especially controlled by lymphocytes. Presumably, the number of patients in our study was too small to elucidate whether viral control was impaired after 90Y DOTATOC treatment.
In addition to this clinical study, early mouse experiments [27] indicated that intravenous treatment with a mixture of strontium-90 (90Sr) and 90Y [2.5 − 10 µCi (=92.5 − 370 kBq)/mouse] either depressed or increased the response of splenocytes to mitogens within the first week after irradiation. The long-term irradiation led to higher splenocyte reactivity. However, it is hard to compare this study with ours because of different biodistributions. Presumably, the effect in our patients was similar to that of the high-dose irradiations in mice; within the first week, we also observed a decrease in lymphocyte reactivity. But long-term effects post-treatment (32 days in mice and 90 days in man) differed. We could not observe enhanced lymphocyte reactivity. Further, a previous study on hemodialysis patients [28] indicated that extracorporeal irradiation of the blood using a portable 90Sr–90Y source (beta-ray emitter) resulted in selective lymphocytopenia, but not in a definite effect on lymphocyte response, as measured in vitro with lymphocyte transformation tests (n = 6). In this study, low transit doses (100–600 rad = 1–6 Gy) were applied over a long period. Data on lymphocyte function can hardly be compared with that previous study due to the field of irradiation (in vivo vs. extracorporal) and duration and dose of irradiation.
To answer the question whether there was impairment not only in lymphocyte numbers but also in function per cell, the percentage of reduction in number and in function at day 7 versus day 0 was compared. For example, considering lymphocyte proliferation after stimulation with twelve microbial antigens—which is most likely the best test to mimic in vivo immune responses—the reduction in numbers was 27 % and in function 46 % (mean value for all antigens). According to these data, number and function per cell were both reduced. Of note, this is only a rough estimate as PBMCs were used for the functional assays—and not pure lymphocyte preparations. For example, PBMCs contain approximately 14 % CD14+ cells [29] that “dilute” the lymphocytes, indicating that the effect on lymphocyte function may be even stronger than calculated.
The presence of bone metastases could be identified as an important influencing factor of baseline immune function and of radiation effects. Apparently, patients with bone metastases are in a worse clinical condition. This also applies for patients with progressive disease, higher tumor burden and impaired kidney function. Previously, it was described that adverse clinical condition and disease progress were accompanied by decreased immune function [30]. Moreover, the radiopharmaceutical 90Y DOTATOC accumulates in bone metastases which consequently results in higher bone marrow doses. Thus, it is expectable that the lymphocyte function was more severely impaired in patients with bone metastases.
In conclusion, our findings demonstrate for the first time that 90Y DOTATOC therapy in patients with advanced somatostatin receptor-positive tumors led to a significant reduction in cellular antimicrobial T cell responses in vitro. We could observe immunosuppression at days 1 and 7 after therapy; but it was nearly undetectable at day 90. The clinical relevance of that finding and the duration of immunosuppression, however, have to be further defined.
Acknowledgments
This article is a partial fulfillment of requirements for the doctor’s degree at the Medical Faculty, University of Essen, Germany, for Mr. C. Hueben.
Ethical standards
All patients provided written informed consent prior to their inclusion in the study. The study was approved by the institutional review board and carried out in accordance with the Helsinki Declaration of 1964, as revised in 2000.
Conflict of interest
The authors declare that they have no conflict of interest.
Abbreviations
- Cpm
Counts per minute
- DOTATOC
DOTA-d-Phe(1)-Tyr(3)-octreotide
- DTPA
Diethylene-triamine-pentaacetate
- ELISpot
Enzyme-linked immunospot
- 68Ga
Gallium-68
- IFN
Interferon
- IL
Interleukin
- LTT
Lymphocyte transformation test
- OKT3
Anti-CD3 monoclonal antibody
- PBMCs
Peripheral blood mononuclear cells
- PET/CT
Positron emission tomography together with computed tomography
- 90Sr
Strontium-90
- 90Y
Yttrium-90
References
- 1.Perico ME, Chinol M, Nacca A, Luison E, Paganelli G, Canevari S. The humoral immune response to macrocyclic chelating agent DOTA depends on the carrier molecule. J Nucl Med. 2001;42(11):1697–1703. [PubMed] [Google Scholar]
- 2.Harrison A, Walker CA, Parker D, et al. The in vivo release of 90Y from cyclic and acyclic ligand-antibody conjugates. Int J Rad Appl Instrum B. 1991;18(5):469–476. doi: 10.1016/0883-2897(91)90107-V. [DOI] [PubMed] [Google Scholar]
- 3.Kozak RW, Raubitschek A, Mirzadeh S, Brechbiel MW, Junghans RP, Gansow OA, Waldmann TA. Nature of the bifunctional chelating agent used for radioimmunotherapy with yttrium-90 monoclonal antibodies: critical factors in determining in vivo survival and organ toxicity. Cancer Res. 1989;49(10):2639–2644. [PubMed] [Google Scholar]
- 4.Knox SJ, Goris ML, Trisler K, et al. Yttrium-90-labeled anti-CD20 monoclonal antibody therapy of recurrent B-cell lymphoma. Clin Cancer Res. 1996;2(3):457–470. [PubMed] [Google Scholar]
- 5.Otte A, Mueller-Brand J, Dellas S, Nitzsche EU, Herrmann R, Maecke HR. Yttrium-90-labelled somatostatin-analogue for cancer treatment. Lancet. 1998;351(9100):417–418. doi: 10.1016/S0140-6736(05)78355-0. [DOI] [PubMed] [Google Scholar]
- 6.Rufini V, Calcagni ML, Baum RP. Imaging of neuroendocrine tumors. Semin Nucl Med. 2006;36(3):228–247. doi: 10.1053/j.semnuclmed.2006.03.007. [DOI] [PubMed] [Google Scholar]
- 7.Barsegian V, Müller SP, Horn PA, Bockisch A, Lindemann M. Lymphocyte function following radioiodine therapy in patients with thyroid carcinoma. Nuklearmedizin. 2011;50(5):195–203. doi: 10.3413/nukmed-04241108. [DOI] [PubMed] [Google Scholar]
- 8.Anderson RE, Warner NL. Ionizing radiation and the immune response. Adv Immunol. 1976;24:215–335. doi: 10.1016/s0065-2776(08)60331-4. [DOI] [PubMed] [Google Scholar]
- 9.Dehos G, Hinz G, Schwarz E-R. Changes in number and function of the lymphocyte populations as a biological indicator for ionizing radiation. In: Kaul A, Dehos A, Bögl W, Hing G, Kossel F, Schwarz E-R, Stamm A, Stephan G, editors. Biological indicators for radiation dose assessment. München: MMV; 1986. pp. 298–301. [Google Scholar]
- 10.Guedeney G, Grunwald D, Malarbet JL, Doloy MT. Time dependence of chromosomal aberrations induced in human and monkey lymphocytes by acute and fractionated exposure to 60Co. Radiat Res. 1988;116(2):254–262. doi: 10.2307/3577462. [DOI] [PubMed] [Google Scholar]
- 11.Anderson RE, Lefkovits I. Effects of irradiation on the in vitro immune response. Exp Cell Biol. 1980;48(4):255–278. [PubMed] [Google Scholar]
- 12.Belka C, Ottinger H, Kreuzfelder E, Weinmann M, Lindemann M, Lepple-Wienhues A, Budach W, Grosse-Wilde H, Bamberg M. Impact of localized radiotherapy on blood immune cells counts and function in humans. Radiother Oncol. 1999;50(2):199–204. doi: 10.1016/S0167-8140(98)00130-3. [DOI] [PubMed] [Google Scholar]
- 13.Breitz H. Dosimetry in a myeloablative setting. Cancer Biother Radiopharm. 2002;17(1):119–128. doi: 10.1089/10849780252824136. [DOI] [PubMed] [Google Scholar]
- 14.Marincek N, Jorg AC, Brunner P, et al. Somatostatin-based radiotherapy with [90Y-DOTA]-TOC in neuroendocrine tumors: long-term outcome of a phase I dose escalation study. J Transl Med. 2013;11:17. doi: 10.1186/1479-5876-11-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.IRCP . Publication 51. Nonstochastic effects of ionizing radiation. Oxford: Pergamon Press; 1984. [Google Scholar]
- 16.Coleman CN, Blakely WF, Fike JR, et al. Molecular and cellular biology of moderate-dose (1–10 Gy) radiation and potential mechanisms of radiation protection: report of a workshop at Bethesda, Maryland, December 17–18, 2001. Radiat Res. 2003;159(6):812–834. doi: 10.1667/RR3021. [DOI] [PubMed] [Google Scholar]
- 17.Russell CD, Bischoff PG, Kontzen FN, Rowell KL, Yester MV, Lloyd LK, Tauxe WN, Dubovsky EV. Measurement of glomerular filtration rate: single injection plasma clearance method without urine collection. J Nucl Med. 1985;26(11):1243–1247. [PubMed] [Google Scholar]
- 18.Lindemann M, Witzke O, Winterhagen T, et al. T-cell function after interleukin-2 therapy in HIV-infected patients is correlated with serum cortisol concentrations. AIDS. 2004;18(15):2001–2007. doi: 10.1097/00002030-200410210-00004. [DOI] [PubMed] [Google Scholar]
- 19.IRCP (1987) Publication 53 (with Addenda). Radiation dose to patients from radiopharmaceuticals. A report of a Task Group of Committee 2 of the International Commission on Radiological Protection. Ann ICRP 18 (1–4): 1–377 [PubMed]
- 20.IRCP Publication 80: radiation dose to patients from radiopharmaceuticals (addendum 2 to ICRP publication 53) Ann ICRP. 1998;28(3):1–126. doi: 10.1016/S0146-6453(99)00006-8. [DOI] [PubMed] [Google Scholar]
- 21.Förster GJ, Engelbach MJ, Brockmann JJ, Reber HJ, Buchholz HG, Macke HR, Rosch FR, Herzog HR, Bartenstein PR. Preliminary data on biodistribution and dosimetry for therapy planning of somatostatin receptor positive tumours: comparison of (86)Y-DOTATOC and (111)In-DTPA-octreotide. Eur J Nucl Med. 2001;28(12):1743–1750. doi: 10.1007/s002590100628. [DOI] [PubMed] [Google Scholar]
- 22.Hänscheid H, Lassmann M, Luster M, et al. Iodine biokinetics and dosimetry in radioiodine therapy of thyroid cancer: procedures and results of a prospective international controlled study of ablation after rhTSH or hormone withdrawal. J Nucl Med. 2006;47(4):648–654. [PubMed] [Google Scholar]
- 23.Dale RG. Dose-rate effects in targeted radiotherapy. Phys Med Biol. 1996;41(10):1871–1884. doi: 10.1088/0031-9155/41/10/001. [DOI] [PubMed] [Google Scholar]
- 24.Vrisekoop N, den Braber I, de Boer AB, et al. Sparse production but preferential incorporation of recently produced naive T cells in the human peripheral pool. Proc Natl Acad Sci USA. 2008;105(16):6115–6120. doi: 10.1073/pnas.0709713105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lindemann M, Belger P, Ottinger HD, Beelen DW, Grosse-Wilde H. Long-term follow-up of cellular in vitro immunity after allogeneic peripheral blood stem cell versus bone marrow transplantation. Tissue Antigens. 2005;66(5):480. [Google Scholar]
- 26.Carr BI, Metes DM. Peripheral blood lymphocyte depletion after hepatic arterial 90Yttrium microsphere therapy for hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 2012;82(3):1179–1184. doi: 10.1016/j.ijrobp.2010.10.042. [DOI] [PubMed] [Google Scholar]
- 27.Stevenson AF, Daculsi R, Monig H. Haematological studies on 90Sr–90Y-toxicity: II. Femoral CFU-s kinetics and mitogen response of spleen cells. Radiat Environ Biophys. 1982;20(4):275–287. doi: 10.1007/BF01323753. [DOI] [PubMed] [Google Scholar]
- 28.Birkeland SA. The immunosuppressive effect of extracorporeal irradiation of the blood using a portable 90Sr–90Y source and small transit doses. Scand J Immunol. 1976;5(4):323–329. doi: 10.1111/j.1365-3083.1976.tb00286.x. [DOI] [PubMed] [Google Scholar]
- 29.Lindemann M, Rebmann V, Ottinger HD, Schmolke K, Kreuzfelder E, Grosse-Wilde H. rhG-CSF effect on mixed lymphocyte cultures and circulating soluble HLA antigen levels in volunteer stem cell donors. Exp Hematol. 2004;32(11):1103–1109. doi: 10.1016/j.exphem.2004.07.025. [DOI] [PubMed] [Google Scholar]
- 30.Ugurel S, Lindemann M, Schadendorf D, Grosse-Wilde H. Altered surface expression patterns of circulating monocytes in cancer patients: impaired capacity of T-cell stimulation? Cancer Immunol Immunother. 2004;53(11):1051. doi: 10.1007/s00262-004-0565-1. [DOI] [PMC free article] [PubMed] [Google Scholar]