Abstract
Background:
Interleukin-1 (IL-1) signaling has an established role as a cytokine signaling pathway important for progression of abdominal aortic aneurysms (AAAs). While the IL-1β ligand and IL-1R1 have been previously investigated, the role of the IL-1α ligand in AAAs remains unknown. In this study, we sought to examine the role of IL-1α in AAAs using genetic and pharmacologic approaches.
Methods:
Eight-week-old wild-type (WT) or IL-1α knock-out (KO) male and female mice (n = 10–16/group) underwent experimental AAA and were harvested 14 days following surgery to assess AAA size and characteristics. In separate studies, 8-week-old WT mice were treated with an inhibitor to IL-1α during AAA formation and harvested 14 days following surgery. Finally, WT and IL-1α KO mice were administered Anakinra, an IL-R1 inhibitor, during AAA formation to determine the effect of inhibiting IL-1R1 when IL-1α is knocked out.
Results:
Male and female IL-1α KO mice had larger AAAs compared to WT AAAs (male: 153% vs. 89.2%, P = 0.0001; female: 86.6% vs. 63.5%, P = 0.02). IL-1α KO mice had greater elastin breakage (P = 0.01), increased levels of macrophage staining (P = 0.0045), and greater proemetallo proteinase 2 (P = 0.02). Pharmacologic inhibition of WT male mice with an IL-1α neutralizing antibody resulted in larger AAAs (133.1% vs. 77.0%, P < 0.001). Finally, treatment of IL-1α KO male mice with Anakinra decreased AAA formation compared with vehicle control AAAs (Anakinra + IL-1α KO: 47.7% vs. WT: 147.1%; P = 0.0001).
Conclusions:
IL-1α disruption using either genetic or pharmacologic approaches worsens AAAs.
INTRODUCTION
The implementation of national screening programs for abdominal aortic aneurysms (AAAs) in European countries and the United States based on data from four large, randomized trials that reported AAAs in 3.9–7.7% of men over 651–4 made significant strides in identifying and clinically managing AAAs more effectively. Moreover, the increased coverage of nationwide AAA screening has led to more diagnoses of asymptomatic cases, most of which present small AAA lesions (<5.5 or 5 cm for male and female, respectively).5,6 However, recent evidence suggests despite screening, AAAs remain high in the United Kingdom for both men and women with age-standardized death rates of 7.5 per 100,000 and 3.7 per 100,000, respectively, and age-standardized death rates in many European countries have increased slightly since 2012,7 suggesting that AAAs remain a relevant health concern despite recent declines. AAAs remain the 15th leading cause of death annually in the United States.8–11 Elective and emergent AAAs account for more than 15,000 surgical procedures annually, representing a huge burden to healthcare systems.12,13 Aortic rupture remains unpredictable and is difficult to diagnose, as endovascular and open surgical repair remain the mainstay of treatment.12,14 Most problematic, there is currently no medical treatment strategy for slowing the growth rates or inhibiting rupture of this deadly disease nor is the disease identified easily using biomarkers or simple, inexpensive tests.8–11 Recent evidence also suggests that as countries in Africa, Latin America, and Asia, dietary habits have evolved with industrialization, aneurysm prevalence, and rupture rates have increased. 15–18 Patients with AAAs typically have a 5e10 years of monitoring window between diagnosis with imaging and indication for open or endovascular surgical repair.9,10 Patients in this window would benefit from a medical therapy that halts aneurysm growth or decreases rupture risk to increase quality of life.
Aortic aneurysms have historically been characterized by an early elevation of proinflammatory cytokines such as interleukin-1β (IL-1β), monocyte chemoattractant protein-1 (MCP-1), IL-6, tumor necrosis factor-α (TNF-α), interferon-γ (IFN-γ), IL-23, and IL-17. As a result, the pathways of many of these cytokines on AAA formation have been at least initially examined and described in various AAA models. Our laboratory was the first to demonstrate that inhibition of IL-1β and IL-1R1 resulted in significant inhibition of AAAs. We found that elevation of IL-1β was an early event in aortic aneurysm formation and that inhibition of the pathway using the IL-R1 antagonist, Anakinra, resulted in significant attenuation of aneurysm formation.19 In related studies, we found that inhibition of IL-1 signaling also inhibited descending thoracic aortic aneurysm formation20 while other laboratories found inhibition of Kawasaki diseased aneurysms with IL-1 pathway inhibition.21–24 IL-1α has been shown to bind to the IL-1R1 and to activate downstream signaling pathways such as MYD88, IRAK4, and IL-6 in a similar manner to IL-1β signaling.25,26 Numerous studies have demonstrated a role of IL-1 signaling in AAA formation using genetic and pharmacologic inhibition studies19,20,24; however, there is currently little information investigating the role of IL-1α inhibition on AAA progression nor have studies investigated effects of pharmacologic inhibition of IL-1α on AAAs. In the present study, we seek to determine whether genetic and pharmacologic inhibition of IL-1α signaling could inhibit aneurysm progression using murine topical application elastase models of AAA.
METHODS
Animals
Adult male and female C57BL/6J mice (8–12 weeks of age) were obtained from The Jackson Laboratory (Bar Harbor, Maine). Male and female IL-1α, IL-1β, and IL-1R1 mice were kindly provided by Dr. Yoichiro Iwakura.19,27 Mice were housed and maintained at 70°F, 50% humidity, and in 12-hour light-dark cycles per institutional animal protocols. Animal care and use were in accordance with the Guide for the Care and Use of Laboratory Animals. The animal protocol was approved by the University of Virginia Institutional Animal Care and Use Committee (#3634) in compliance with the Office of Laboratory Animal Welfare.
Elastase Treatment Model of Aneurysm Formation
A murine elastase treatment model of AAA formation was used as described by Laser et al.1,2,7 The methods for induction of anesthesia, surgical procedure for AAA formation, and postoperative analgesia have been previously described.28,29 In short, after the abdominal aorta is circumferentially dissected below the left renal vein to the bifurcation and topical application of 5 μL of elastase (Sigma-Aldrich, St. Louis, Missouri; 10.1 mg protein/mL, 19 units/mg protein) to the exposed aortic adventitia for 5 min. After 5 min exposure, the elastase on the aorta is removed with a cotton-tipped applicator and the laparotomy is closed. Video micrometric measurements of aortic diameters were made in situ before and after elastase application at day 0 and before harvesting the aorta on day 14 of the model. Mice from each group were euthanized under anesthesia by overdose and exsanguination. The abdominal aorta, from below the left renal vein to the bifurcation, was dissected. The external aortic adventitia diameter was measured at its maximum diameter and at the intact self-control portion just below the left renal vein using video microscopy with NIS-Elements D.3.10 software attached to the microscope (Nikon SMZ-800; Nikon Instruments, Melville, New York). Aortic dilation percentage was determined using maximal AAA diameter–self-control aortic diameter)/maximal AAA diameter × 100%. A dilation of ≥ 100% was considered to be positive for AAA. The aortas were harvested, and either (1) flash frozen in liquid nitrogen for analyses by real-time polymerase chain reaction or protein extraction or (2) incubated in 4% paraformaldehyde (PFA) overnight for histology or immunohistochemistry.
IL-1α Knock-Out Mouse Studies
C57/B6 male mice (n = 13/group), C57/B6 female mice (n = 11/group), IL-1α knock-out (KO) male mice (n = 16/group), and IL-1α KO female mice (n = 10/group) underwent the topical elastase murine model and were harvested 14 days following AAA induction (Fig. 1A). Mice were harvested and analyzed using video micrometry and then were either snap-frozen in liquid nitrogen or PFA fixed and processed for histological analysis.
Fig. 1.
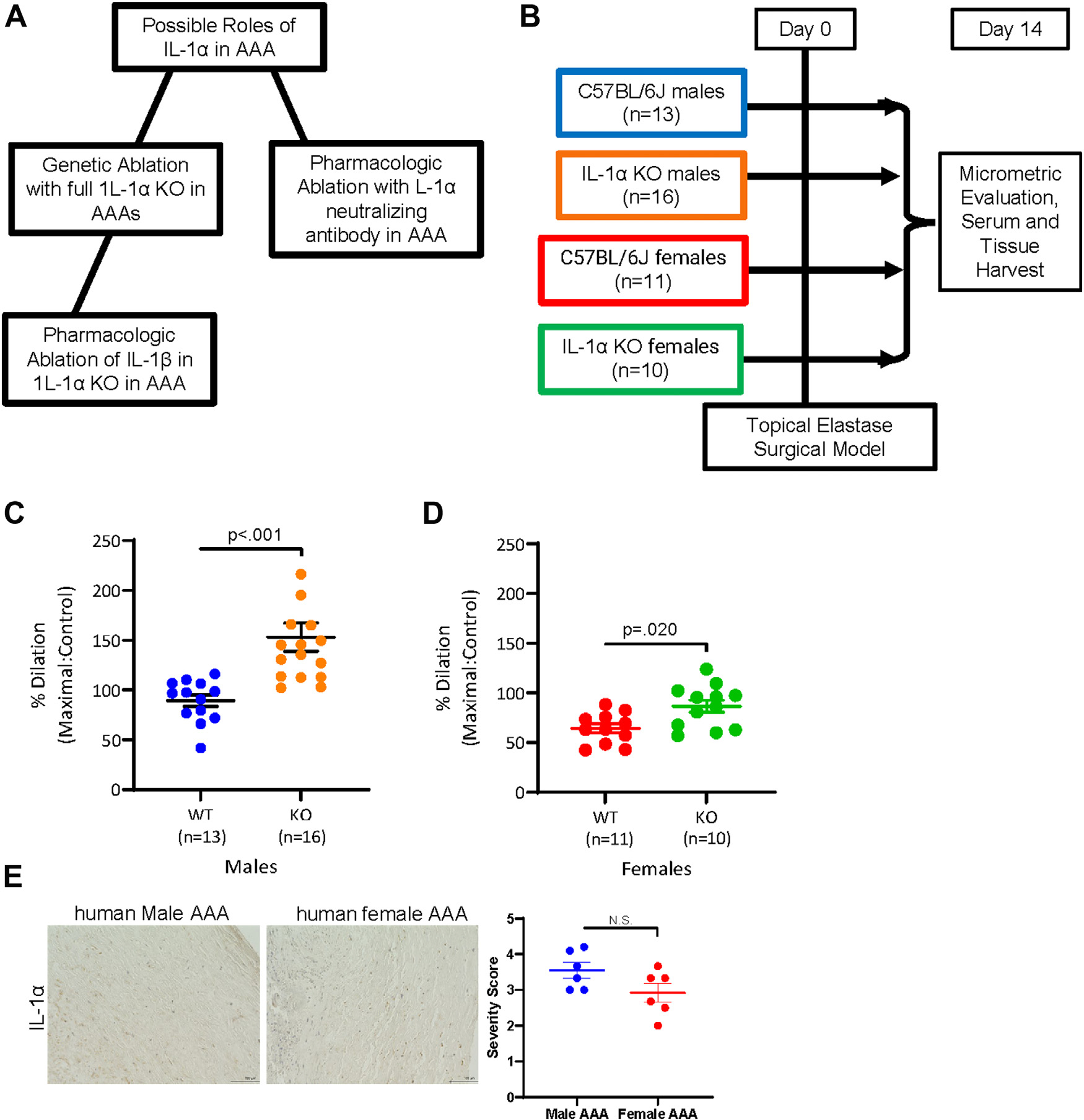
Male and female IL-1α knock-out mice demonstrated augmented AAA formation in comparison to wild-type mice in a topical elastase AAA model. (A) Schematic of the experiment approach used to investigate IL-1α elimination in AAAs. (B) Schematic of the topical elastase surgical model and groups used in the genetic mouse knock-out studies. (C) WT (n = 13/group) and IL-1α (n = 16/group) male mice were harvested 14 days following topical elastase AAA formation and aortic dilation data were measured (P < 0.001; WT: 89.19 ± 5.835% versus IL-1α: 153.0 ± 14.13%). (D) WT (n = 11/group) and IL-1α (n = 10/group) female mice were also harvested 14 days following topical elastase AAA formation and aortic dilation was measured (P = 0.02; WT: 64.36 ± 4.645 versus IL-1α: 86.60 ± 6.138). (E) Male and female human AAA samples (n = 6/group) were embedded, paraffin section and stained for IL-1α, and quantified to determine whether IL-1α is gender dependent in human AAAs.
IL-1α Neutralizing Antibody Studies
C57BL/6J male mice (n = 12/group) underwent the topical elastase murine model and prevention treatment with either a neutralizing antibody to IL-1α (100 mg/kg; BE0243, BioXCell, Lebanon, New Hampshire) or a control antibody (100 mg/kg; BE0091; BioXCell, Lebanon, New Hampshire) and harvested 14 days following AAA induction (Fig. 1A). Mice were harvested and analyzed using video micrometry and then were either snap-frozen in liquid nitrogen or PFA fixed and processed for histological analysis.
Anakinra Treatment of IL-1α Mice
C57BL/6J male mice (n = 11/group) underwent the topical elastase murine model and prevention treatment with either Anakinra (Kineret, 100 mg/kg/day) or a control antibody (100 mg/kg/day) and harvested 14 days following AAA induction (Fig. 1A). IL-1α KO male mice (n = 4/group) also underwent the topical elastase mouse model and prevention treatment with Anakinra (Kineret, 100 mg/kg/day) for 14 days. Mice were harvested and analyzed using video micrometry and then were either snap-frozen in liquid nitrogen or PFA fixed and processed for histological analysis.
Histology
Murine aortas were harvested at sacrifice for histology analysis after undergoing left ventricular puncture and 4% PFA antegrade perfusion at physiologic pressure. Further fixation was achieved by overnight incubation in 4% PFA at 4°C followed by paraffin embedding and sectioning at 5 μm. Day 14–harvested (n = 4–8/group) cross-sections were prepared with Verhoeff van Gieson (VVG) for elastin. Separately for immunohistochemical staining, after microwave antigen retrieval, antibodies were bound and detected using VectaStain Elite Kit (Vector Laboratories Inc., Burlingame, California). Antibodies for immunohistochemical staining were antirat Mac2/LGAL3 for macrophages (1:10,000; Cedarlane Laboratories, Burlington, Canada), antimouse anti-Neutrophil (Ly 6B.2) for neutrophils (1:10,000; AbD Serotec, Oxford, United Kingdom), antirabbit CD31 (1:350; Abcam, Cambridge, Massachusetts), antirabbit S100A4 for fibroblasts (1:100; Cell Signaling, Danvers Massachusetts), antimouse SMαA for smooth muscle α-actin (1:1,000; Abcam, Cambridge, Massachusetts), Ki-67 for proliferation (1:300; Abcam, Cambridge, Massachusetts), cleaved caspase 3 for apoptosis (1:100; Cell Signaling, Danvers Massachusetts), antirabbit IL-1β (1:300; Abcam, Cambridge, Massachusetts), antirabbit IL-1R1 (1:200; Abcam, Cambridge, Massachusetts), antirabbit IL-1α (1:350; Abcam, Cambridge, Massachusetts), and antirabbit IL-6 (1:300, Abcam, Cambridge, Massachusetts). Visualization color development was completed using diaminobenzidine (Dako Corporation, Carpinteria, California) for SMαA, Mac2, antineutrophil, CD-31, S100A4, Ki-67, cleaved caspase 3, IL-1α, IL-1β, IL-1R1, and IL-6.
Images were acquired using AxioCam Software version 4.6 via 10X objectives and an AxioCam MRc camera (Carl Zeiss Inc., Thornwood, New York). Elastin degradation was quantified by counting the number of breaks per vessel and then averaged and graphed. Histology was graded by two blinded reviewers. For elastin grading, 1 corresponded to no degradation, 2 = mild degradation, 3 = moderate degradation, and 4 = severe degradation. For smooth muscle cell (SMC) grading, 1 corresponded to no SMC loss, 2 = mild SMC loss, 3 = moderate SMC loss, and 4 = severe SMC loss.
Gelatin Zymography
Protein was extracted from frozen aortic tissue using phosphate buffer saline and protease inhibitors (Roche Diagnostics, Indianapolis, Indiana) as previously described.30 Gelatin zymography was performed using abdominal aortic tissue samples (n = 4–6/group) to measure pro and active metallo proteinases (MMPs) 2 and 9. Precast zymography gels (10%, Invitrogen, Carlsbad, California) were loaded with 3 mg of aortic protein from each sample. Samples were diluted into 2× tris-glycine sodium dodecyl sulphate sample buffer and separated using electrophoreses under nonreducing conditions. The gels were renatured for 30 min in renaturing buffer (Invitrogen) and incubated for 1 day at 37°C while rocking in developing buffer (Invitrogen, Carlsbad, California). The gels were then stained in Simply Blue Safe Stain (Invitrogen, Carlsbad, California). Optical density levels were quantified using the BioRad Image laboratory software, version 4.0.19
Enzyme-Linked Immunosorbent Assay
An enzyme-linked immunosorbent assay (ELISA) analysis was performed from serum of wild-type (WT) noninjured mice (n = 8/group) versus day 14 AAA mice (n = 11/group) for IL-1β, IL-1α, or IL-1R1 as per the manufacturer’s instructions (BD Biosciences Pharmingen, San Diego, California). Separately, ELISA analysis was performed for IL-1β or IL-1α as per the manufacturer’s instructions (BD Biosciences Pharmingen, San Diego, California) from serum samples isolated from the mouse studies from Figures 1 or 3 (n = 5/group) were used for the IL-1β and, separately, the IL-1α ELISA.
Fig. 3.
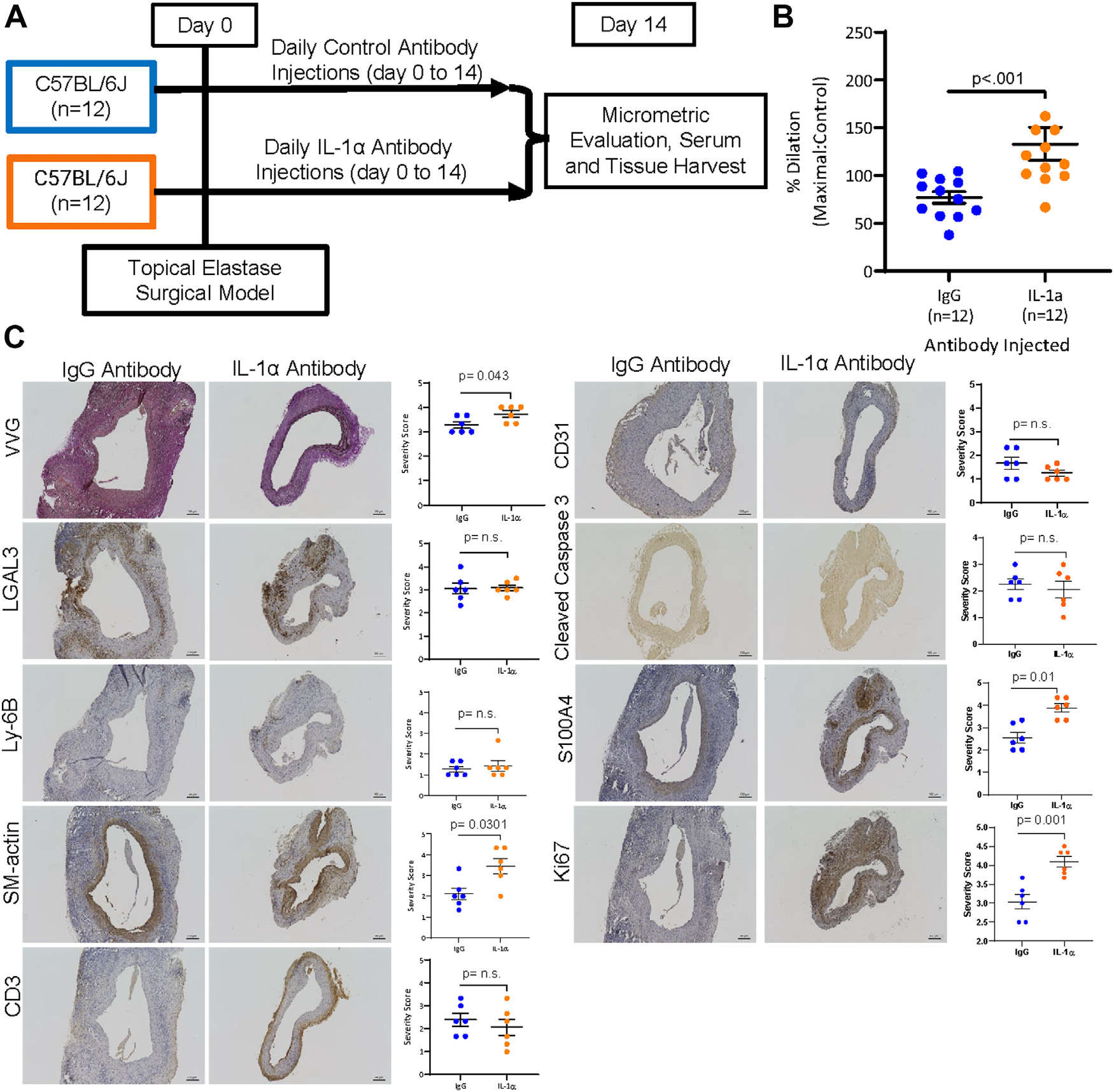
Pharmacologic inhibition of IL-1α in WT male mice demonstrated increased aortic dilation and increased SM-actin and S100A4 expression. (A) Schematic of the topical elastase surgical model and groups used in the IL-1α pharmacologic mouse inhibition studies. (B) WT (n = 12/group) male mice underwent topical elastase application and underwent injections of either an IL-1α antibody or IgG antibody on days 2, 4, 6, 8, 10, and 12 following surgery. Male mice were harvested 14 days following topical elastase AAA formation and aortic dilation data were measured (P < 0.001; IgG antibody: 77.03 ± 6.061% versus IL-1α antibody: 133.1 ± 17.32%). (C) Following harvest, a subset was analyzed for immunohistochemical analysis using VVG, LGAL3, Ly-6B, SM-actin, CD-3, CD31, S100A4, cleaved caspase 3, or Ki67 staining. VVG staining demonstrated a trend toward increased elastin breaks (P = 0.0801, n = 5–6/group), as did SM-actin (P = 0.0301, n = 5–6/group), S100A4 (P = 0.01, n = 5–6/group), and Ki67 (P = 0.01, n = 5–6/group), whereas LGAL3, CD3, Ly-6B, CD31, and cleaved caspase 3 exhibited no change (n = 5–6/group).
Statistical Methods
GraphPad Prism 7 software (GraphPad Software, La Jolla, California) was used for testing statistical analysis. AAA rupture results were analyzed using a Chi-squared test. Statistical analysis of aortic diameters was performed using GraphPad Prism 7 software (GraphPad Software, La Jolla, California). Maximal aortic dilation (%) was calculated as . The internal control was a small segment of normal proximal abdominal aorta just distal to the renal arteries that was above the proximal extent of the AAA. Values are reported as mean ± standard error of the mean. Aortic dilation between groups was compared using Student’s t-test for two sample groups or analysis of variance for three or more sample groups. To compare two groups in other samples, Student’s t-test was used. Paired data were analyzed by paired Student’s t-test. Differences between two or more groups versus a control group were analyzed with one-way analysis of variance plus Bonferroni correction for multiple comparisons. Nonparametric data were analyzed by Mann–Whitney U-test.
RESULTS
IL-1α Knock-Out Mice Have Larger AAAs Compared to Wild-Type Mice
Male and female IL-1α KO mice and WT controls (C57BL/6J controls n = 10–16/group) underwent topical elastase application AAA induction surgery followed by harvest at 14 days. Male IL-1α KO mice exhibited larger AAAs in comparison to WT controls (Fig. 1B; IL-1α: 153.0 ± 14.13% vs. WT: 89.19 ± 5.835%; P < 0.001). Although female mice exhibited smaller AAAs overall as we have previously reported,31 female IL-1α mice also demonstrated larger AAAs compared to C57BL/6J controls (Fig. 1C; IL-1α: 86.60 ± 6.138% versus WT: 64.36 ± 4.645%; P 0.02). Since we saw no gender-based differences in AAAs between male and female IL-1α mice and their controls and because in human staining we did not see gender-dependent expression of IL-1α in male versus female human AAAs, we pursued the remainder of our studies using male mice for our studies.
IL-1α male mice expressed greater elastin breaks (Fig. 2, P = 0.01, n = 5–10/group), increased endothelial cell staining (Fig. 2, P = 0.007, n = 5–10/group), macrophage staining (Fig. 2, P = 0.05, n = 5–10/group), neutrophil (Fig. 2, P = 0.0045, n = 5–10/group), increased cleaved caspase 3 staining (Fig. 2, P = 0.01, n = 5–10/group), and increased SM α-actin (Fig. 2, P = 0.0091, n = 5–10/group). There was a nonsignificant trend toward an increase in adventitial fibroblast staining using the marker S100A4. These immunohistochemical results suggested that IL-1α KO augments AAAs compared to WT mice.
Fig. 2.
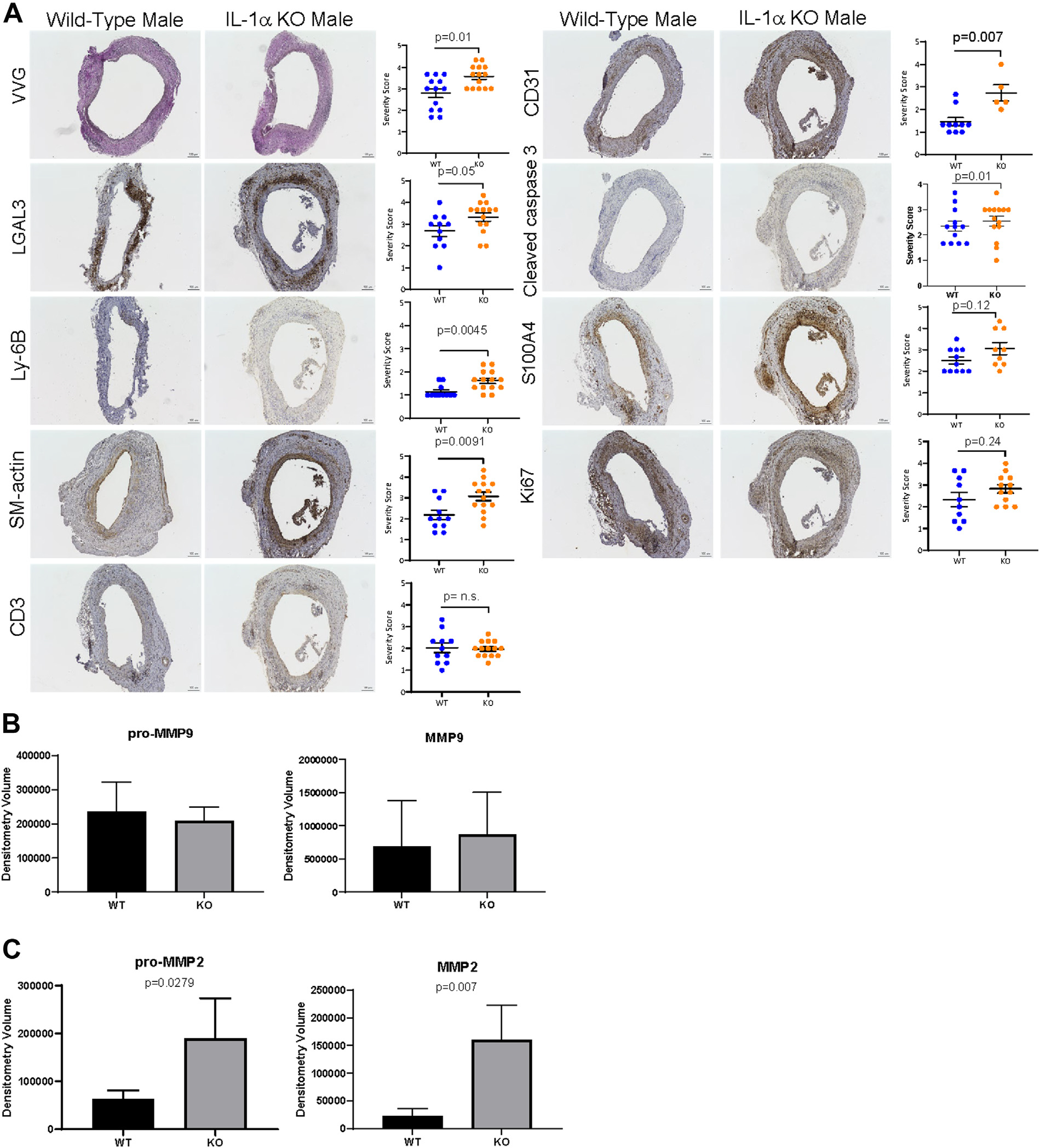
IL-1α knock-out mice demonstrated increased elastin degradation, endothelial cell, and macrophage staining following topical elastase AAA formation. (A) Male WT and IL-1α knock-out mice (n = 5–10/group) were harvested, fixed with 4% paraformaldehyde, paraffin embedded, and stained separately for VVG stain, LGAL3, Ly-6B, SM-actin, CD3, CD31, S100A4, cleaved caspase 3 or Ki67, and counter-stained with hematoxylin. Staining demonstrated increased elastin breaks (P = 0.01, n = 5–10/group), increased macrophage staining (LGAL3; P = 0.05, n = 5–10/group), increased SM-actin staining (SM-actin; P = 0.0091, n = 5–10/group), and increased endothelial cell staining (CD31; P = 0.007, n = 5–10/group) in the IL-1α KO mice. (B) Pro-MMP9 levels in WT and IL-1α knock-out mice were measured using gelatin zymography (n = 4–6/group). MMP9 levels were measured using gelatin zymography (n = 4–6/group). (C) Pro-MMP2 levels were measured using gelatin zymography (65,431 vs. 187,315 densitometry units, P = 0.0279, n = 4–6/group). MMP2 levels were measured in WT and IL-1α knock-out male mice using gelatin zymogram (1,825 vs. 155,721 densitometry units, P = 0.007, n = 4–6/group).
Following immunohistochemical analysis, we next sought to determine matrix MMPs 2 and 9 were altered in IL-1α KO mice versus controls using gelatin zymography. Results demonstrated significantly greater pro and active MMP-2 activity in IL-1α male mice aorta compared to WT control AAA but no change in pro or active MMP-9 levels (Fig. 2: pro-MMP-2: 187,315 vs. 65,431 densitometry units, P = 0.0279, n = 4–6/group and active MMP-2: 155,721 vs. 1,825 densitometry units, P = 0.007, n = 4–6/group).
Prevention Treatment of Wild-Type Mice with IL-1α Neutralizing Antibodies Increased AAAs
Since we were able to demonstrate that IL-1α KO mice exhibit augmented AAAs compared to WT control mice in the topical elastase application mouse model, we next sought to determine whether treatment of WT mice with an IL-1α blocking antibody would also mimic these effects. We treated two groups of male mice (n = 12/group), a control antibody group and an IL-1α neutralizing group beginning day 2 post AAA induction and every other day until harvest at 14 days post AAA formation (Fig. 3A). Interestingly, inhibition of IL-1α in AAAs using a neutralizing antibody also produced augmented AAAs compared to WT controls (Fig. 3B; IL-1α antibody: 133.1 ± 17.32% versus immunoglobulin G [IgG] antibody: 77.03 ± 6.061 %; P < 0.001). We saw a significant difference in AAA dilation between IgG and IL-1α treated WT male mice, when abdominal aortas were analyzed at 14 days following topical elastase; we also saw increased elastin breakage via VVG staining (Fig. 3C; P = 0.0801, n = 5–6/group), increased SM-actin staining (Fig. 3C; P = 0.0301, n = 5–6/group), and increased proliferation (Fig. 3C; P = 0.01, n = 5–6/group).
IL-1β and IL-1R1 Levels are Elevated in IL-1α Knock-Out Mice in AAAs
To examine possible mechanisms behind the augmented AAAs seen in the IL-1α mice versus WT controls, we first measured levels of IL-1α, IL-1β, and IL-1R1 via immunohistochemistry and ELISA in WT mice versus nonsurgical controls. Immunohistochemistry demonstrated increased staining of IL-1α, IL-1β, and IL-1R1 in mice with AAAs harvested 14 days following surgery (Fig. 4A). ELISAs from serum from these mice demonstrated elevated levels of IL-1α (Fig. 4B, P = 0.0002), IL-1β (Fig. 4B, P = 0.0001), and IL-1R1 (Fig. 4B, P = 0.0001) in AAA mice. To further determine a possible mechanism, we then performed immunohistochemical staining in aortas at day 14 after AAA in IL-1α, IL-1β, and IL-1R1 KO male mice versus C57BL/6J controls (Fig. 4C, n = 3–5/group). In the IL-1α KO AAAs, there were elevated levels of IL-1β, IL-1R1, and IL-6 staining over C57BL/6J controls (Fig. 4C). In the IL-1β and IL-1R1 KO mice, we saw minimal levels of staining for IL-1β, IL-1R1, or IL-6 (Fig. 4C).
Fig. 4.
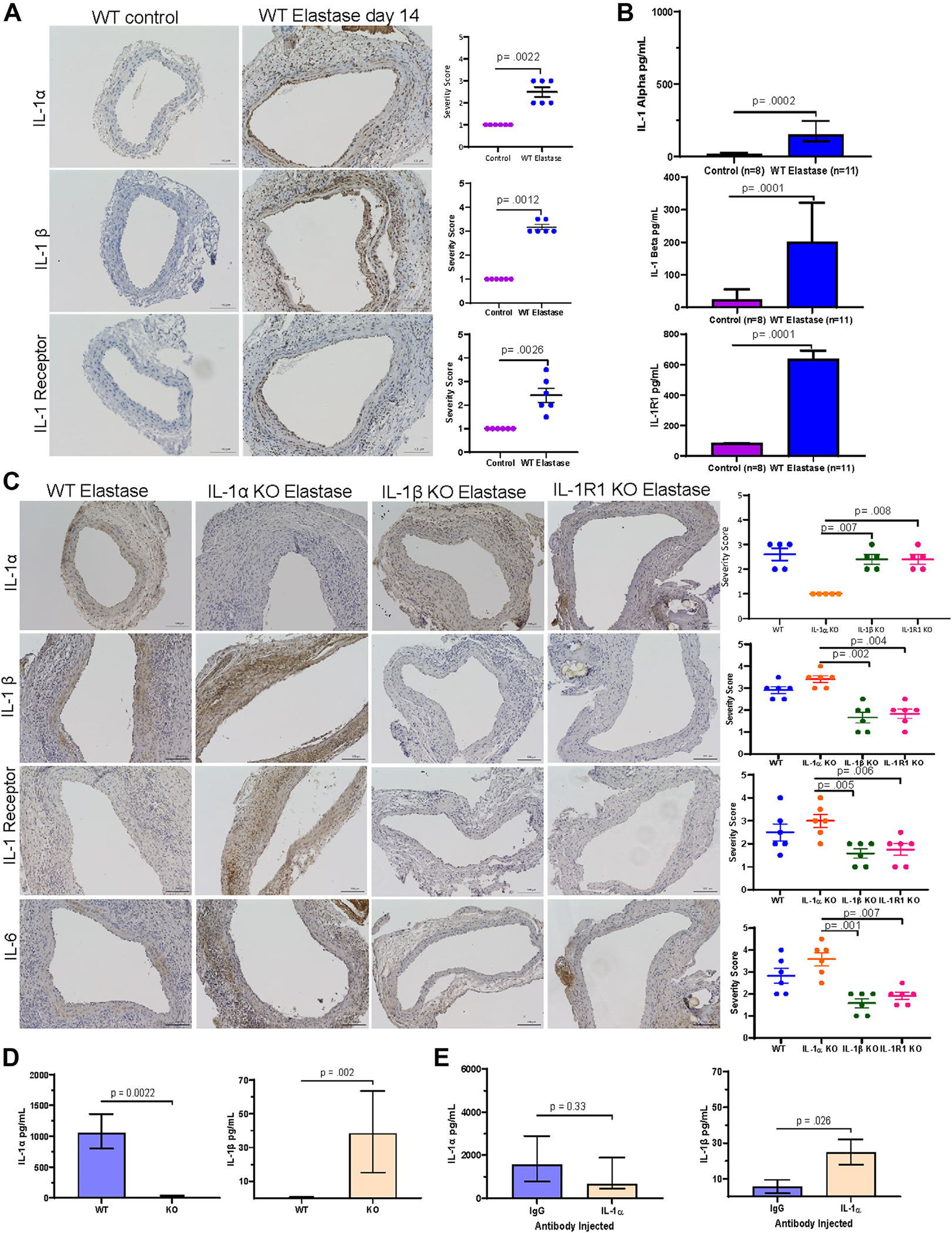
Genetic inhibition of IL-1α in male mice demonstrated increased IL-1b, IL-1R1, and IL-6 staining 14 days following AAA formation. (A) IL-1a, IL-1b, and IL-1R1 were stained via immunohistochemistry in control mice without surgery (n = 6/group) and WT mice that underwent elastase application to the abdominal aorta and harvest 14 days later (n = 6/group). (B) IL-1a, IL-1b, and IL-1R1 were measured via ELISA in control mice without surgery (n = 8/group) and WT AAA mice harvested at 14 days post AAA surgery (n = 11/group). (C) IL-1a, IL-1b, IL-1R1 knock-out, and WT (n = 5/group) male mice underwent elastase AAA surgeries. Male mice were harvested 14 days following topical elastase AAA formation, fixed in PFA and stained for IL-1a, IL-1b, IL-1R1, and IL-6 and results were quantified. (D) Serum was analyzed from WT and IL-1a male mice (n = 5/group) harvested 14 days following AAA formation for IL-1b and IL-1a. (E) Separately, serum was analyzed from WT male mice injected with either IgG or IL-1a antibody (n = 5/group) and harvested 14 days following AAA formation for IL-1b and IL-1a.
To also examine the circulating levels of IL-1α and IL-1β in both the genetic and pharmacologic inhibition studies, we performed protein ELISAs following harvest at day 14 post elastase induction. In the IL-1α KO mice versus WT controls, KO of IL-1α resulted in decreased levels of the IL-1α (Fig. 4D; P = 0.0022; n = 5–6/group) and increased levels of IL-1β (Fig. 4D; P = 0.002; n = 5–6/group). In the IgG versus IL-1α neutralizing experiments, we see a trend toward decreased IL-1α levels (Fig. 4E; P = 0.33, n = 5–6/group) and significantly increased IL-1β levels (Fig. 5B; P = 0.026; n = 5–6/group) in mice treated with a neutralizing antibody to IL-1α.
Fig. 5.
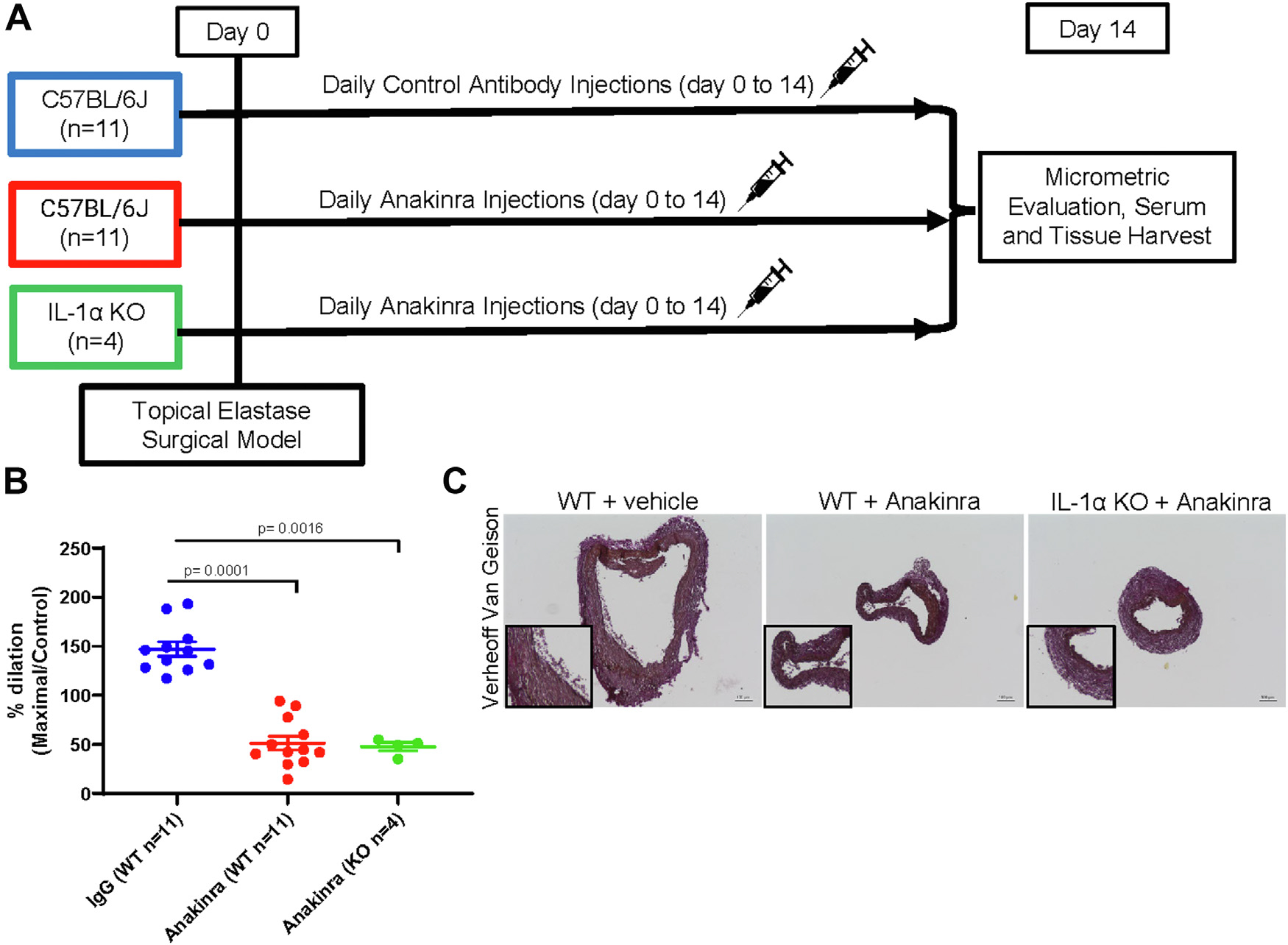
Pharmacologic inhibition of IL-1R1 using Anakinra in IL-1α knock-out mice demonstrates decreased aortic dilation and decreased elastin breaks. (A) Schematic of the topical elastase surgical model and groups used in the WT and IL-1a knock-out pharmacologic mouse inhibition studies. (B) WT (n = 11/group) and IL-1α knock-out (n = 4/group) male mice underwent topical elastase application and underwent injections of either an Anakinra (100 mg/kg) or vehicle control daily (100 mg/kg). Male mice were harvested 14 days following topical elastase AAA formation and aortic dilation data were measured (vehicle antibody: 147.1 ± 24.47 % versus Anakinra: 51.35 ± 24.46% versus IL-1α knock-out plus Anakinra: 47.68 ± 8.628%; P = 0.0001 vehicle versus Anakinra per Student’s t-test; P = 0.0016 vehicle versus IL-1α knock-out plus Anakinra per Student’s t-test). (C) Following harvest, a subset was analyzed for immunohistochemical analysis using VVG staining.
Treatment of IL-1α Knock-Out Mice with Anakinra Abrogates the Augmented AAAs Seen in the IL-1α Knock-Out Mice
We next treated C57BL/6J and IL-1α KO male mice with either Anakinra or a control antibody and harvested mice 14 days following aneurysm induction surgery (Fig. 5A schematic). In IL-1α KO mice, treatment with Anakinra beginning on the day of surgery and continuing daily until harvest at day 14 postsurgery resulted in a significant attenuation of AAA formation and resembled WT mice treatment with Anakinra (Fig. 5B; Anakinra: 51.35 ± 24.46% versus IL-1α KO plus Anakinra: 47.68 ± 8.628% versus vehicle: 147.1 ± 24.47 %; P = 0.001 vehicle treated versus IL-1α KO plus Anakinra). Harvest and histochemical staining for elastin breakage using VVG staining demonstrated AAAs in the IL-1α KO mice treated with Anakinra resembled AAAs from WT mice treated with Anakinra.
DISCUSSION
Using an established murine AAA topical elastase application model,28 our investigations found that male and female IL-1α KO mice have larger AAA size over C57BL/6J control mice. Since women rupture more frequently than males in human disease at smaller sized AAAs, we initially tested to determine whether there were gender-based differences in IL-1α signaling in our murine AAA models and whether this could link back to increased rupture in females with smaller AAAs in humans.32 In this study, there were differences between female and male murine AAA sizes (WT male: 89.19± 5.835% versus WT female: 64.36 ± 4.645) but the trend toward increased AAAs with genetic elimination of IL-1α was demonstrated in both genders and suggests that IL-1α does not exert gender-specific effects in the elastase model of AAAs. We also stained for IL-1α in male and female human AAAs and did not see gender-dependent effects between male and female staining in human AAAs. The greater aneurysm sizes seen in the male mice was also the reason we chose to then focus our analyses on male mice for the remainder of the study because while both genders demonstrated increased AAAs, the male mice had larger AAAs over 100% with genetic inhibition.
Although there are known limitations of animal models compared to human AAA disease, the topical elastase application model is an established standard that has progression and biology consistent with human AAAs.28 Human disease and the topical elastase AAA model exhibit degradation and breakage of elastin and migration, proliferation, and apoptosis of SMCs. MMP-2 and MMP-9 are elevated in human aneurysmal aortic tissue as seen in the topical elastase application model.33–35 Cytokines IL-1β, IL-6, IL-17, IL-23, TNF-α, and IFN-γ are all increased in human AAA disease and are elevated in our topical elastase AAA models.19,28 In our study of murine AAA disease in a topical elastase model developed within our laboratory,28 IL-1α KO mice had increased elastin breaks, increased levels of inflammatory macrophages and neutrophils, increased SM-actin, increased cleaved caspase 3, and increased MMPs that correlate with larger AAAs in previous findings in our laboratory.19 In the neutralizing antibody studies, we see similar results with increased dilation aortic dilation, increased elastin breakage, and increased SM-actin and Ki67 staining. These data suggest that elimination of IL-1α leads to increased inflammation at least in the context of the topical elastase AAA model.
These results also show rather surprisingly that IL-1α and IL-1β could play separate rather than overlapping roles in AAA disease progression and rupture. IL-1α and IL-1β both bind to the IL-1 receptor to activate NF-Kβ signaling through MYD88 signaling cascade. Both IL-1α and IL-1β genes are expressed as precursor genes that are normally cleaved with and activated by external stimuli. The IL-1α precursor gene is expressed in cells, including kidney, liver, lung, endothelial cells, astrocytes, and gastrointestinal epithelium.36 IL-1β is expressed in a limited number of cell types and must be processed from its precursor form to become active in the IL-1 pathway. IL-1β is transcribed by monocytes, macrophages, and dendritic cells subsequent to toll-like receptor activation. The inactive IL-1β precursor needs to be processed by caspase-1 cleavage, which first requires activation by danger-associated molecular patterns. Some controversy exists whether IL-1α and IL-1β act redundantly in the IL-1 signaling pathway or whether there are separate signaling mechanisms for these cytokines. Previous studies in sterile inflammation in IL-1α and IL-1β KO mice suggested that these cytokines exert different effects with IL-1α primarily exerting its effects on neutrophils while IL-1β exerts its effects via macrophages.37,38 Our present study would suggest that IL-1α and IL-1β might also exert different effects in the context of a murine AAA model.
Our laboratory’s previous work has shown that the IL-1 pathway plays a critical, early role in AAA formation.19 IL-1β was seen to be elevated early in murine AAAs and KO of IL-1β or IL-R1 resulted in significant protection from AAA formation.19 These studies also demonstrated that both prevention and treatment of small aneurysms with Anakinra, an IL-R1 neutralizing antibody inhibitor, resulted in attenuated AAAs. Separate but related studies in descending thoracic aortic aneurysms also demonstrated that genetic and pharmacologic inhibition of the IL-1 signaling pathway attenuated aneurysms.20 These studies suggest a negative role of IL-1 signaling in formation and progression of aneurysms as both IL-1β and IL-1R1 inhibition resulted in significant attenuation of AAA formation. Surprisingly, our study demonstrated that IL-1α genetic and pharmacologic inhibition augments AAAs and produces increased inflammation. Our study also demonstrated that the augmentation could be in part related to increased IL-1β, IL-1R1, and IL-6 levels in IL-1α KOmice. These studies suggest that IL-1β and IL-1α are not redundant and could exhibit different effects with respect to AAAs. Febrile brain models have also suggested that IL-1β and IL-1α were not redundant and could exhibit different effects.27 In separate studies, IL-1α but not IL-1β was found to be important for allergen-specific T-cell production.39 These results suggest that in AAAs, IL-1β and IL-1α are not redundant. To further elucidate differences in IL-1 signaling in AAAs, future studies will need to address the possible roles of IL-1β and IL-1α in AAA chronic and thrombotic models leading to rupture40 and to address whether inhibition of the IL-1 pathway could inhibit large AAAs or prevent rupture. It has been hypothesized that IL-1α′s function depends on its subcellular location, much like HMGB1 or IL-33, where under homeostasis, IL-1α function in the cytosol and can mediate normal gene expression from the cytosol or nucleus but upon activation of necrosis, IL-1α can function as an alarmin to promote inflammation.38
Clinically, our ideal goal is to develop a treatment therapy to prolong the time between diagnosis and need for surgery/invasive intervention. Since numerous studies and our own suggest that the IL-1 signaling pathway could be clinically relevant for AAAs, the present study provides additional insight into how the IL-1 pathway could function in AAA disease. These studies also suggest that targeting the IL-1α or IL-1β ligand could produce different effects and could suggest that targeting their common receptor, the IL1R1, might be the preferred target for future pharmacologic inhibition studies or for clinical targeting using Anakinra. These studies suggest that IL-1 signaling in AAAs is more complex than previously believed and could help us understand and better target possible pathways in AAAs.
CONCLUSION
Using a murine model, we investigated the role of IL-1α signaling in AAA formation. In this study, we demonstrated that IL-1α KO male and female mice have augmented AAA formation. In WT mice, pharmacologic prevention studies in male mice also demonstrated increased AAA formation. Finally, treatment of male IL-1α KO mice with Anakinra, an IL-1R1 inhibitor, resulted in attenuated AAA levels that were consistent with Anakinra treatment in WT mice. Future studies will investigate further differences between IL-1β and IL-1α signaling in the context of AAA formation and rupture. These results suggest that IL-1α could play a role in AAA disease progression.
Acknowledgments
We thank Anthony Herring and Cindy Dodson for their knowledge and technical expertise.
Funding:
This work was supported by American Heart Association Scientist Development Grant 14SDG18730000 (M.S.) and R01 HL126668 (G.A.) grants. This project was supported by Award Number T32HL007849 from the National Heart, Lung, and Blood Institute (NHLBI) (J. Dahl, PI: Irving L. Kron, MD). The content is solely the responsibility of the authors and does not necessarily represent the views of the NHLBI.
REFERENCES
- 1.Lindholt JS, Vammen S, Juul S, et al. The validity of ultrasonographic scanning as screening method for abdominal aortic aneurysm. Eur J Vasc Endovasc Surg 1999;17:472–5. [DOI] [PubMed] [Google Scholar]
- 2.Norman PE, Semmens JB, Lawrence-Brown MMD. Long-term relative survival following surgery for abdominal aortic aneurysm: a review. Cardiovasc Surg 2001;9:219–24. [DOI] [PubMed] [Google Scholar]
- 3.Scott RA, Wilson NM, Ashton HA, et al. Influence of screening on the incidence of ruptured abdominal aortic aneurysm: 5-year results of a randomized controlled study. Br J Surg 1995;82:1066–70. [DOI] [PubMed] [Google Scholar]
- 4.Scott RAP, Bridgewater SG, Ashton HA. Randomized clinical trial of screening for abdominal aortic aneurysm in women. Br J Surg 2002;89:283–5. [DOI] [PubMed] [Google Scholar]
- 5.Kontopodis N, Pantidis D, Dedes A, et al. The - not So - Solid 5.5 cm threshold for abdominal aortic aneurysm repair: facts, misinterpretations, and future directions. Front Surg 2016;3:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brodersen J, Hansson A, Johansson M, et al. Consequences of screening in abdominal aortic aneurysm: development and dimensionality of a questionnaire. J Patient Rep Outcomes 2017;2:37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Al-Balah A, Goodall R, Salciccioli JD, et al. Mortality from abdominal aortic aneurysm: trends in European Union 15+ countries from 1990 to 2017. Br J Surg 2020;107:1459–67. [DOI] [PubMed] [Google Scholar]
- 8.Prevention. USCfDCa. Deaths: preliminary data for 2011. Natl Vital Stat Rep 2012;61:1–51. [PubMed] [Google Scholar]
- 9.United States Centers for Disease Control and Prevention OoSaP, and the National Center for Injury Prevention and Control.WISQARS. Leading Causes of Death Reports. 20 leading causes of death: United States, 2007. Available at: http://webappacdcgov/sasweb/ncipc/leadcaus10html. 2007 Accessed July 15, 2021.
- 10.Kent KC, Zwolak RM, Egorova NN, et al. Analysis of risk factors for abdominal aortic aneurysm in a cohort of more than 3 million individuals. J Vasc Surg 2010;52:539–48. [DOI] [PubMed] [Google Scholar]
- 11.Bhak RH, Wininger M, Johnson GR, et al. Factors associated with small abdominal aortic aneurysm expansion rate. JAMA Surg 2014;150:44–50. [DOI] [PubMed] [Google Scholar]
- 12.Kent KC. Clinical practice. Abdominal aortic aneurysms. N Engl J Med 2014;371:2101–8. [DOI] [PubMed] [Google Scholar]
- 13.Upchurch GR Jr, Schaub TA. Abdominal aortic aneurysm. Am Fam Physician 2006;73:1198–204. [PubMed] [Google Scholar]
- 14.Baxter BT, Matsumura J, Curci J, et al. Non-invasive Treatment of Abdominal Aortic Aneurysm Clinical Trial (N-TA(3) CT): design of a phase IIb, placebo-controlled, double-blind, randomized clinical trial of doxycycline for the reduction of growth of small abdominal aortic aneurysm. Contemp Clin Trials 2016;48:91–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ogeng’o JA, Olabu BO, Kilonzi JP. Pattern of aortic aneurysms in an African country. J Thorac Cardiovasc Surg 2010;140:797–800. [DOI] [PubMed] [Google Scholar]
- 16.Sule AZ, Ardil B, Ojo EO. Abdominal aortic aneurysm and the challenges of management in a developing country: a review of three cases. Ann Afr Med 2012;11:176–81. [DOI] [PubMed] [Google Scholar]
- 17.Wang SW, Huang YB, Huang JW, et al. Epidemiology, clinical features, and prescribing patterns of aortic aneurysm in Asian population from 2005 to 2011. Medicine (Baltimore) 2015;94:e1716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Calderón M, Brito V, Alcaraz A, et al. Reparación Endovascular para Aneurisma de Aorta: Revisión Panorámica Sobre su Evidencia en el Mundo y su Aplicación en Latinoamérica. Value Health Reg Issues 2018;17:94–101. [DOI] [PubMed] [Google Scholar]
- 19.Johnston WF, Salmon M, Su G, et al. Genetic and pharmacologic disruption of interleukin-1β signaling inhibits experimental aortic aneurysm formation. Arterioscler Thromb Vasc Biol 2013;33:294–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Johnston WF, Salmon M, Pope NH, et al. Inhibition of interleukin-1β decreases aneurysm formation and progression in a novel model of thoracic aortic aneurysms. Circulation 2014;130:S51–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Carmo M, Colombo L, Bruno A, et al. Alteration of elastin, collagen and their cross-links in abdominal aortic aneurysms. Eur J Vasc Endovascular Surg 2002;23:543–9. [DOI] [PubMed] [Google Scholar]
- 22.Henderson EL, Geng YJ, Sukhova GK, et al. Death of smooth muscle cells and expression of mediators of apoptosis by T lymphocytes in human abdominal aortic aneurysms. Circulation 1999;99:96–104. [DOI] [PubMed] [Google Scholar]
- 23.Middleton RK, Lloyd GM, Bown MJ, et al. The pro-inflammatory and chemotactic cytokine microenvironment of the abdominal aortic aneurysm wall: a protein array study. J Vasc Surg 2007;45:574–80. [DOI] [PubMed] [Google Scholar]
- 24.Wakita D, Kurashima Y, Crother TR, et al. Role of interleukin-1 signaling in a mouse model of Kawasaki disease-associated abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol 2016;36:886–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dinarello CA. A clinical perspective of IL-1β as the gatekeeper of inflammation. Eur J Immunol 2011;41:1203–17. [DOI] [PubMed] [Google Scholar]
- 26.Kaneko N, Kurata M, Yamamoto T, et al. The role of interleukin-1 in general pathology. Inflamm Regen 2019;39:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Horai R, Asano M, Sudo K, et al. Production of mice deficient in genes for interleukin (IL)-1alpha, IL-1beta, IL-1alpha/beta, and IL-1 receptor antagonist shows that IL-1beta is crucial in turpentine-induced fever development and glucocorticoid secretion. J Exp Med 1998;187:1463–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bhamidipati CM, Mehta GS, Lu G, et al. Development of a novel murine model of aortic aneurysms using periadventitial elastase. Surgery 2012;152:238–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Salmon M, Schaheen B, Spinosa M, et al. ZFP148 (Zinc-Finger protein 148) binds cooperatively with NF-1 (Neurofibromin 1) to inhibit smooth muscle marker gene expression during abdominal aortic aneurysm formation. Arterioscler Thromb Vasc Biol 2019;39:73–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Salmon M, Johnston WF, Woo A, et al. KLF4 Regulates abdominal aortic aneurysm morphology and deletion attenuates aneurysm formation. Circulation 2013;128:S163–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ailawadi G, Eliason JL, Roelofs KJ, et al. Gender differences in experimental aortic aneurysm formation. Arterioscler Thromb Vasc Biol 2004;24:2116–22. [DOI] [PubMed] [Google Scholar]
- 32.Hannawa KK, Cho BS, Sinha I, et al. Attenuation of experimental aortic aneurysm formation in P-Selectin Knockout mice. Ann N Y Acad Sci 2006;1085:353–9. [DOI] [PubMed] [Google Scholar]
- 33.Aziz F, Kuivaniemi H. Role of matrix metalloproteinase inhibitors in preventing abdominal aortic aneurysm. Ann Vasc Surg 2007;21:392–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Elmore JR, Keister BF, Franklin DP, et al. Expression of matrix metalloproteinases and TIMPs in human abdominal aortic aneurysms. Ann Vasc Surg 1998;12:221–8. [DOI] [PubMed] [Google Scholar]
- 35.Freestone T, Turner RJ, Coady A, et al. Inflammation and matrix metalloproteinases in the Enlarging abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol 1995;15:1145–51. [DOI] [PubMed] [Google Scholar]
- 36.Garlanda C, Dinarello Charles A, Mantovani A. The interleukin-1 family: back to the future. Immunity 2013;39:1003–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rider P, Carmi Y, Guttman O, et al. IL-1α and IL-1β recruit different myeloid cells and promote different stages of sterile inflammation. J Immunol 2011;187:4835–43. [DOI] [PubMed] [Google Scholar]
- 38.Voronov E, Dotan S, Krelin Y, et al. Unique versus redundant functions of IL-1α and IL-1β in the tumor microenvironment. Front Immunol 2013;4:177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nakae S, Naruse-Nakajima C, Sudo K, et al. IL-1 alpha, but not IL-1 beta, is required for contact-allergen-specific T cell activation during the sensitization phase in contact hypersensitivity. Int Immunol 2001;13:1471–8. [DOI] [PubMed] [Google Scholar]
- 40.Lu G, Su G, Davis JP, et al. A novel chronic advanced stage abdominal aortic aneurysm murine model. J Vasc Surg 2017;66:232–242.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]


