Abstract
Purpose
Pharmacologic DNA hypomethylation holds strong promises in cancer immunotherapy due to its immunomodulatory activity on neoplastic cells. Searching for more efficient DNA hypomethylating agents to be utilized to design novel immunotherapeutic strategies in cancer, we investigated the immunomodulatory properties of the new DNA hypomethylating agent SGI-110, that is resistant to in vivo inactivation by cytidine deaminase.
Experimental design
Cutaneous melanoma, mesothelioma, renal cell carcinoma, and sarcoma cells were treated in vitro with SGI-110. RT-PCR, quantitative RT-PCR, quantitative methylation-specific PCR, and flow cytometric analyses were performed to investigate changes induced by SGI-110 in the constitutive immune profile of cancer cells. The recognition by gp100-specific CTL of gp100-positive melanoma cells, treated or not with SGI-110, was tested by LDH release assays.
Results
SGI-110 induced/up-regulated the expression of investigated cancer/testis antigens (CTA) (i.e., MAGE-A1, MAGE-A2, MAGE-A3, MAGE-A4, MAGE-A10, GAGE 1-2, GAGE 1-6, NY-ESO-1, and SSX 1-5) in all cancer cell lines studied, both at mRNA and at protein levels. Quantitative methylation-specific PCR analyses identified a hypomethylation of MAGE-A1 and NY-ESO-1 promoters in SGI-110-treated neoplastic cells, demonstrating a direct role of pharmacologic DNA demethylation in CTA induction. SGI-110 also up-regulated the expression of HLA class I antigens and of ICAM-1, resulting in an improved recognition of cancer cells by gp100-specific CTL.
Conclusions
Our findings show that SGI-110 is a highly attractive therapeutic agent to comprehensively increase immunogenicity and immune recognition of neoplastic cells, and provide the scientific rationale for its clinical development to design novel chemo-immunotherapeutic approaches in cancer patients.
Electronic supplementary material
The online version of this article (doi:10.1007/s00262-012-1365-7) contains supplementary material, which is available to authorized users.
Keywords: DNA hypomethylating agent, Cancer, Immunotherapy, Epigenetics
Introduction
Upcoming evidences are demonstrating a significant immunomodulatory role of DNA hypomethylating agents (DHA) on cancer cells. In particular, the clinically available DHA 5-aza-2′-deoxycytidine (5-AZA-CdR) significantly improved the immunogenicity and immune recognition of neoplastic cells [1]. As far as tumor-associated antigens (TAA), these studies mainly focused on cancer/testis antigens (CTA) due to their current use in clinical trials, demonstrating that 5-AZA-CdR induced/up-regulated their expression, homogenized their intratumor expression, improved CTA-based recognition of cancer cells by cytotoxic T lymphocytes (CTL), and elicited anti-CTA humoral immune responses in vivo [1–8].
These well-described immunomodulatory properties of 5-AZA-CdR strongly support the clinical application of DHA to design new immunotherapeutic approaches for cancer treatment. Nevertheless, the extremely short half-life, sensitivity to inactivation by cytidine deaminase in vivo and pronounced hemopoietic toxicity of 5-AZA-CdR, may hurdle its effective and safe clinical use in cancer therapies [9]. Based on these notions, short oligonucleotides were recently synthesized and identified as more stable and effective prodrugs for the delivery of inhibitors of DNA methylation [10]. Among these, SGI-110 is a highly promising alternative to 5-AZA-CdR having a higher stability and a better tolerability in cancer patients, thanks to its lower C max as compared to molar equivalent doses of 5-AZA-CdR [11]. SGI-110 is a dinucleotide that consists of 5-AZA-CdR bonds, with a natural phosphodiester linkage, with a deoxyguanosine. It was demonstrated to be effective in retarding tumor growth in human bladder cancer xenografts by both i.p. and s.c. deliveries and to possess epigenetic remodeling activity, being able to restore the expression of p16 in cancer cells, through demethylation of the 5′-end region of the gene [10, 12].
Despite these initial promising data on epigenetic remodeling and anti-tumor properties of SGI-110, no study has investigated its immunomodulatory activity on neoplastic cells. Thus, aiming to identify new tools to improve the efficacy of current cancer immunotherapy approaches, we extensively investigated the immunologic effects of SGI-110 on a panel of neoplastic cells from solid tumors of different histological origin. SGI-110 significantly modulated the constitutive immune profile of cancer cells, resulting in their more efficient functional recognition by TAA-specific CTL.
These newly characterized immunomodulatory properties of SGI-110, combined with its favorable pharmacologic and pharmacokinetic features, identify SGI-110 as a useful agent to implement novel combined epigenetic–immunotherapeutic strategies in cancer patients.
Materials and methods
Cells
Cells were grown in RPMI 1640 (Flow Laboratories, Inc., McLean, VA) supplemented with 10 % heat-inactivated FCS and 2 mM l-glutamine. Cutaneous melanoma cell lines were generated from surgically removed metastatic lesions from melanoma patients, as described previously [13]. Renal cell carcinoma (RCC) cells were obtained as described previously [14]. Mesothelioma cell lines from pleural effusion of mesothelioma patients were obtained and grown as previously described [15]. The human cell lines SK-LMS-1 (leiomyosarcoma) and MG-63 (osteosarcoma) were purchased from American Type Culture Collection (Rockville, MD).
Monoclonal antibodies and reagents
The monoclonal antibodies (mAb) W6/32, against HLA class I, and HMB-45, against gp100, were purchased from AbD Serotec (Oxford, UK). The anti-HLA-A2 mAb BB7.2 and the anti-CD54 (ICAM-1) mAb 84H10 were purchased from Becton–Dickinson (Franklin Lakes, NJ). The anti-MAGE-A1 mAb MA454 was purchased from Santa Cruz Biotechnology (Heidelberg, Germany). The anti-NY-ESO-1 mAb D8.38 was kindly provided by Dr. Spagnoli GC (University of Basel, Switzerland). The FITC-conjugated F(ab’)2 fragments of rabbit anti-mouse immunoglobulins (Ig) were purchased from DAKO (Milan, Italy). ChromePure mouse IgG was purchased from Jackson ImmunoResearch (Suffolk, UK).
The dinucleotide SGI-110 [10] and its 3′-3′-linked isomer (3′-3′-DpG), which was designed to resist the cleavage by natural phosphodiesterases, were kindly provided by Astex Pharmaceuticals, Inc. (Dublin, CA). 5-AZA-CdR was purchased from Sigma Chemical Co.
In vitro treatment for tumor cells with SGI-110
Cells (3–4 × 105) were seeded in a T75 tissue culture flask and treated 24 h later with SGI-110, 5-AZA-CdR, or 3′-3′-DpG, by replacing the medium with fresh one containing 1 μM or 10 μM of SGI-110, 1 μM of 5-AZA-CdR, or 3′-3′-DpG, every 12 h for 2 days (4 pulses) and then with fresh medium without drugs for additional 2 days. Control cultures were treated under similar experimental conditions in the absence of drug.
RT-PCR analysis
Extraction of total RNA and RT-PCR analyses, using oligonucleotide primer sequences and PCR amplification programs specific for MAGE-A1, MAGE-A2, MAGE-A3, MAGE-A4 and MAGE-A10, NY-ESO-1, GAGE 1–2, GAGE 1–6, and SSX 1-5, were performed as described previously [5]. The integrity of RNA and oligo(dT)-synthesized cDNA was assessed by the amplification of all cDNA samples with β-actin-specific primers, as described previously [5]. 10 μl of each RT-PCR sample was run on a 2 % agarose gel and visualized by ethidium bromide staining.
Real-time quantitative RT-PCR analysis
Real-time quantitative RT-PCR analyses were performed as described [16]. Total RNA was digested with RNAse-free DNAse (Roche Diagnostics, Milan, Italy). Synthesis of cDNA was performed on 1 μg of total RNA using M-MLV reverse transcriptase (Invitrogen, Milan, Italy) and random hexamer primers (Promega, Milan, Italy), according to the manufacturer’s instructions. The ABI prism 7000 Sequence Detection System (Applied Biosystems, Milan, Italy) was utilized to perform the measurement of gene expression, as previously described [16]. cDNA standards were obtained by RT-PCR amplification of the specific mRNAs and quantitated by NanoDrop® ND-1000 UV–Vis spectrophotometer. Quantitative RT-PCRs were performed on 20 ng retrotranscribed total RNA in a final volume of 25 μl 1× TaqMan Master Mix (NY-ESO-1, MAGE-A3 and β-actin) or 25 μl 1× SYBR Green Master Mix (MAGE-A1) (Applied Biosystems). The primers and the probes utilized for quantitative RT-PCR assays were the following: NY-ESO-1, forward 5′-TGCTTGAGTTCTACCTCGCCA-3′, reverse 5′-TATGTTGCCGGACACAGTGAA-3′ and probe 5′-AGGATGCCCCACCGCTTCCC-3′; MAGE-A3, forward 5′-TGTCGTCGGAAATTGGCAGTAT-3′, reverse 5′-CAAAGACCAGCTGCAAGGAACT-3′ and probe 5′-TCTTTCCTGTGATCTTC-3′; β-actin, forward 5′-CGAGCGCGGCTACAGCTT-3′, reverse 5′-CCTTAATGTCACGCACGATT-3′ and probe 5′-ACCACCACGGCCGAGCGG-3′; MAGE-A1, forward 5′-AACCTGACCCAGGCTCTGT-3′, reverse 5′-CAGGCAGATCTTCTCCTTGG-3′. The copy number of NY-ESO-1, MAGE-A3, MAGE-A1, and β-actin molecules was established in each sample by extrapolation of the standard curve. The number of NY-ESO-1, MAGE-A3, and MAGE-A1 cDNA molecules in each sample was normalized to the number of 03B2-actin cDNA molecules.
Serologic assays
Indirect immunofluorescence analysis was done as described [17]. A sample was classified as positive when more than 10 % of cells were stained with the appropriate mAb. The intracellular expression of gp100, MAGE-A1, and NY-ESO-1 was evaluated on paraformaldehyde-fixed cells, permeabilized with the Leucoperm kit (AbD Serotec) following the manufacturer’s instructions.
Quantitative methylation-specific PCR (qMSP) analyses
Genomic DNA was extracted from untreated and SGI-110-treated neoplastic cells by proteinase K treatment, followed by standard phenol/chloroform extraction and ethanol precipitation [18]. Bisulfite conversion was carried out on 500 ng genomic DNA using EZ DNA Methylation-Gold™ Kit (Zymo Research, Orange, CA), according to the manufacturer’s protocol. Primers for the analysis of the methylation status of MAGE-A1 and NY-ESO-1, designed using the free online software MethPrimer [19], were as follows: MAGE-A1 methylated, forward 5′-TAGGTTTTTTCGGTCGAATTTTAC-3′, reverse 5′-CGAATATAACGCCACTAACTTACG-3′; MAGE-A1 unmethylated, forward 5′-GTTAGGTTTTTTTGGTTGAATTTTAT-3′, reverse 5′-CCCAAATATAACACCACTAACTTACA-3′; NY-ESO-1 methylated, forward 5′-AGGATTTCGGGTTGTTTATTTTAC-3′, reverse 5′-ACCTTCGACCTACATAACTCCG-3′; NY-ESO-1 unmethylated, forward 5′-GAGGATTTTGGGTTGTTTATTTTAT-3′, reverse 5′-CTAACCTTCAACCTACATAACTCCA-3′. SYBR green qMSP reactions were performed on 2 μl of bisulfite-modified genomic DNA in a final volume of 25 μl 1× Power SYBR green mastermix (Applied Biosystems) at 95 °C for 10 min, followed by 45 cycles of 15 s at 95 °C and 1 min at 60 °C, using methylated- or unmethylated-specific primer pairs. Real-time measurement of fluorescence intensity was made utilizing the ABI PRISM 7000 Sequence Detection System (Applied Biosystems). The copy number of methylated or unmethylated sequences, for each target gene, was established in each sample by extrapolation from the standard curves. The percentage of methylation was calculated as the ratio between methylated molecules and the sum of methylated and unmethylated molecules.
Cytotoxicity assay
Cytolytic activity of HLA-A2-restricted gp100-specific CTL was tested against Mel 275 melanoma cells untreated or treated with 1 μM SGI-110, using the Colorimetric Cytotoxicity Assay Kit (Oxford Biomedical Research, Rochester Hills, MI) that quantitatively measures LDH release. Cells were used at effector/target (E/T) ratios of 25/1, 12/1, 6/1, and 3/1. The percentage of specific lysis was determined following the manufacturer’s instruction.
The blocking effect of HLA class I antigens and ICAM-1 was studied on SGI-110-treated Mel 275 melanoma cells pre-incubated with 20 μg/ml of the anti-HLA class I mAb W6/32 or the anti-ICAM-1 mAb 84H10, for 30 min at 37 °C. Then, cells were washed and used as targets for HLA-A2-restricted gp100-specific CTL in LDH release assay at E/T ratio of 25/1.
Statistical analysis
Data analyzed by Student’s paired t test with p < 0.05 were considered statistically significant.
Results
Induction of CTA expression in cancer cells by SGI-110
To evaluate global effects of SGI-110 on the constitutive expression of CTA in malignant cells of different histological origin, RT-PCR analyses were performed on in vitro cultured melanoma, sarcoma, renal cell carcinoma, and mesothelioma cell lines, untreated or treated with SGI-110 1 μM. Exposure to SGI-110 induced the expression of investigated CTA (i.e., MAGE-A1, MAGE-A2, MAGE-A3, MAGE-A4 and MAGE-A10, GAGE 1–2, GAGE 1–6, NY-ESO-1, and SSX 1–5) in CTA-negative cancer cells (Table 1). A similar pattern of CTA expression was induced following the exposure of cancer cells to a higher concentration of SGI-110 (10 μM, data not shown), demonstrating that the 1 μM dose is sufficient to achieve the maximal effectiveness on CTA induction. Moreover, no difference in the pattern of CTA induction was observed by comparing the effects of SGI-110 to those of equimolar concentration of 5-AZA-CdR (Table 1). To evaluate the mechanism of action underlying the modulatory activity of SGI-110 on CTA profile, similar treatments were performed using the SGI-110-related compound 3′-3′-DpG, which cannot deliver 5-AZA-CdR, due its 3′-3′ bond that is resistant to the cleavage by natural phosphodiesterases. No changes in the constitutive expression of CTA were observed following treatment for all neoplastic cells investigated (data not shown), demonstrating that the immunomodulatory activity of SGI-110 is exerted exclusively through the delivery of 5-AZA-CdR.
Table 1.
RT-PCR analysis of CTA expressed in malignant cells of different histotype following SGI-110 and 5-AZA-CdR treatment
| Histotype | Cells | Treatment | MAGE-A1 | MAGE-A2 | MAGE-A3 | MAGE-A4 | MAGE-A10 | GAGE 1-2 | GAGE 1-6 | NY-ESO-1 | SSX 1-5 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Melanoma | Mel 275 | Ctrl | −a | + | + | − | + | − | − | − | − |
| SGI-110 | + | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | + | + | + | − | + | + | + | ||
| Mel 313 | Ctrl | − | − | − | − | − | − | + | − | − | |
| SGI-110 | + | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | − | − | − | − | + | + | + | ||
| Mel 611 | Ctrl | + | + | + | − | + | + | + | + | + | |
| SGI-110 | + | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | + | + | + | + | + | + | + | ||
| Mel 684 | Ctrl | + | + | + | − | + | − | + | − | + | |
| SGI-110 | + | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | + | + | + | − | + | + | + | ||
| Mel 195 | Ctrl | + | − | − | − | − | − | − | − | − | |
| SGI-110 | + | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | + | − | + | + | + | + | + | ||
| Sarcoma | MG-63 | Ctrl | − | − | − | − | − | − | − | − | − |
| SGI-110 | − | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | − | + | + | + | + | + | + | + | + | ||
| SK-LMS-1 | Ctrl | − | − | − | − | − | − | + | − | + | |
| SGI-110 | + | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | + | + | + | + | + | + | + | ||
| Renal cell carcinoma | MZ-1257 | Ctrl | − | − | − | − | − | − | − | − | − |
| SGI-110 | + | + | − | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | − | + | + | + | + | + | + | ||
| LE-9104 | Ctrl | − | − | − | − | − | − | − | − | − | |
| SGI-110 | + | + | − | + | − | + | + | + | + | ||
| 5-AZA-CdR | + | + | − | + | − | + | + | + | + | ||
| Mesothelioma | MES-1 | Ctrl | − | − | − | − | − | − | − | − | − |
| SGI-110 | + | + | − | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | − | + | + | + | + | + | + | ||
| MPP-89 | Ctrl | + | + | + | − | − | − | − | − | − | |
| SGI-110 | + | + | + | + | + | + | + | + | + | ||
| 5-AZA-CdR | + | + | + | + | + | + | + | + | + |
RT-PCR analyses were performed on total RNA extracted from human cancer cell lines either untreated (ctrl) or treated with SGI-110 1 μM or 5-AZA-CdR 1 μM, every 12 h for 2 days (4 pulses)
aRT-PCR products: − CTA-negative; + CTA-positive
Quantitative modulation of the constitutive expression of CTA in cancer cells by SGI-110
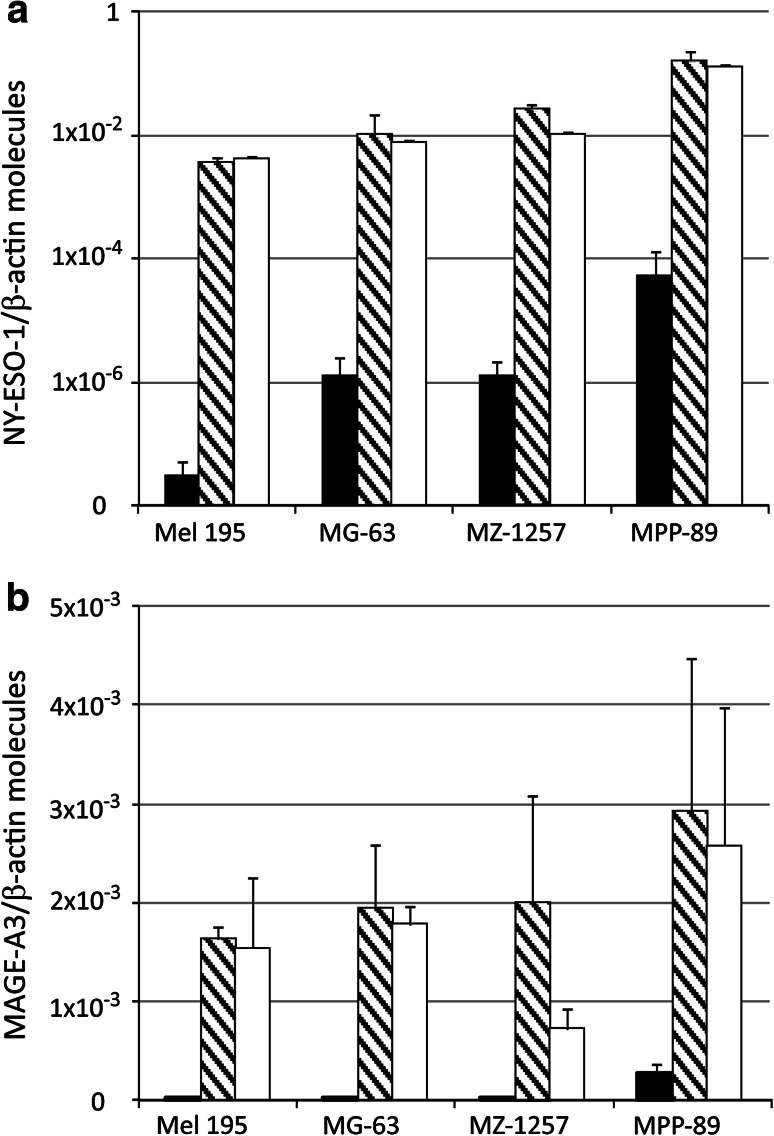
To quantify the effects of SGI-110 on the constitutive levels of CTA expressed by neoplastic cells, real-time quantitative RT-PCR analysis was performed in melanoma, sarcoma, renal cell carcinoma, and mesothelioma cell lines, untreated and treated with SGI-110 1 μM. We specifically focused on the CTA MAGE-A3 and NY-ESO-1, which represent the most attractive targets for cancer immunotherapy due to their ability to elicit spontaneous humoral and cellular immune responses in cancer patients [20]. Figure 1 shows that SGI-110 administration induced and/or strongly up-regulated the constitutive levels of MAGE-A3- and NY-ESO-1-specific mRNA expression in neoplastic cells of all histotypes investigated. No significant difference was observed on quantitative expression of the CTA following increase in SGI-110 dose to 10 μM (data not shown). Moreover, a similar pattern of CTA induction/up-regulation was observed by comparing the effects of SGI-110 to those of equimolar concentration of 5-AZA-CdR (Fig. 1).
Fig. 1.
Quantitative RT-PCR analysis of CTA expression in cells from different solid malignancies treated with SGI-110. Total RNA was extracted from human melanoma (Mel 195), sarcoma (MG-63), renal cell carcinoma (MZ-1257), and mesothelioma (MPP-89) cell lines, either untreated (black) or treated with 1 μM SGI-110 (striped) or 1 μM 5-AZA-CdR (white) every 12 h for 2 days (4 pulses). TaqMan quantitative RT-PCRs were performed on retrotranscribed total RNA, utilizing NY-ESO-1- (a), MAGE-A3- (b), and β-actin-specific primers. CTA expression was normalized to the expression of the β-actin gene. Values are reported as CTA molecules/β-actin molecules, on a logarithmic (a) or linear (b) scale. Data shown are relative to a single tumor cell line that is representative for each investigated histotype
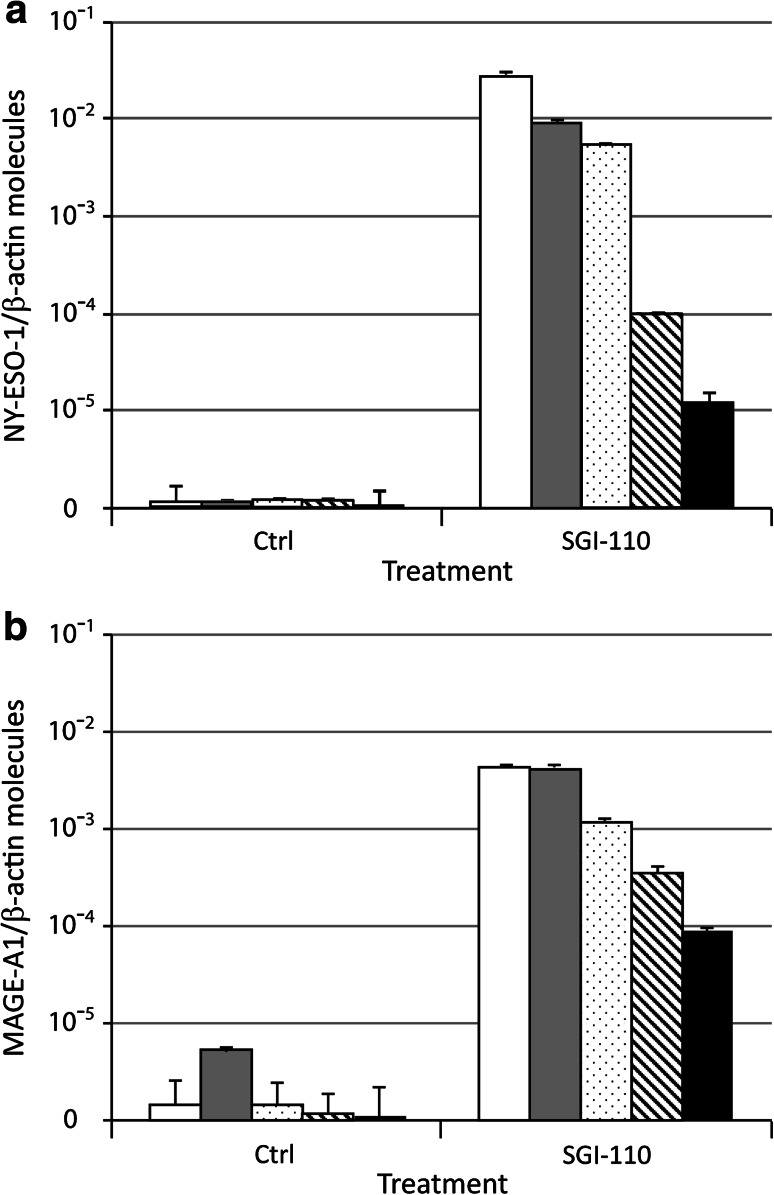
Time course analysis of Mel 275 melanoma cells cultured in vitro for different times after the end of treatment demonstrated that the expression of CTA (i.e., NY-ESO-1 and MAGE-A1) induced by SGI-110 was persistent (Fig. 2). In fact, although it progressively decreased with time, it was still detectable after 80 days of culture in SGI-110-free medium (Fig. 2).
Fig. 2.
Time dependency of CTA expression induced in cancer cells by SGI-110. Total RNA was extracted from Mel 275 melanoma cells untreated (ctrl) or treated (SGI-110) with 1 μM SGI-110 every 12 h for 2 days (4 pulses) and further cultured for 0 (white columns), 15 (gray columns), 45 (dotted columns), 65 (striped columns), and 80 (black columns) days after the end of treatment. Quantitative RT-PCRs were performed on retrotranscribed total RNA, utilizing NY-ESO-1- (a), MAGE-A1- (b), and β-actin-specific primers. CTA expression was normalized to the expression of the β-actin gene. Values are reported as CTA molecules/β-actin molecules, on a logarithmic scale
No significant changes were induced by SGI-110 in the constitutive mRNA levels of other investigated TAA (i.e., tyrosinase and gp100) in different investigated cancer cells (data not shown).
Modulation of the immune phenotype of cancer cells by SGI-110
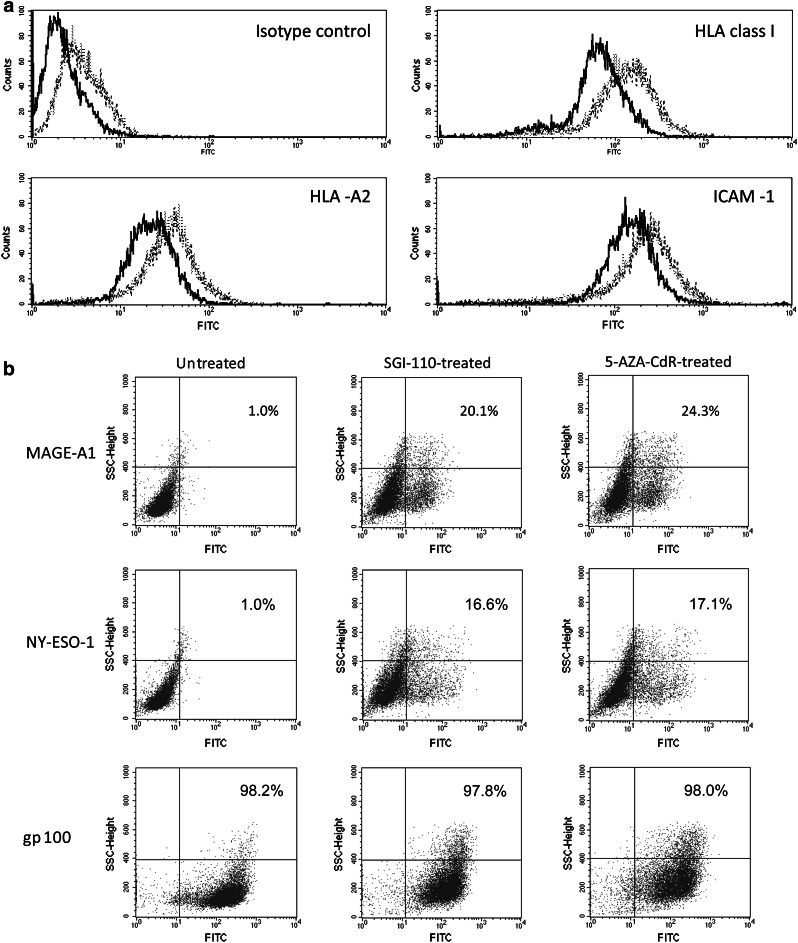
Exposure to SGI-110 significantly (p < 0.05) up-regulated the constitutive levels of expression of HLA class I antigens, HLA-A2 allospecificity, and of the co-stimulatory molecule ICAM-1, on Mel 275 melanoma cells. Results obtained from three independent experiments showed that mean values of mean fluorescence intensity ± SD, for Mel 275 melanoma cells untreated or treated with SGI-110 1 μM, were 84.7 ± 16.4 and 214.3 ± 71.8 for HLA class I, 20.3 ± 4 and 43.7 ± 8.8 for HLA-A2, 216 ± 57.5 and 327 ± 83.4 for ICAM-1, respectively. Representative data are shown in Fig. 3a. The statistically significant (p ≤ 0.05) difference, between untreated and SGI-110-treated cells, remained detectable when values of mean fluorescence intensity for each investigated antigen were normalized to the values of the relative isotype control (data not shown). Moreover, similar immunomodulatory effects induced by SGI-110 were observed in melanoma cells treated with equimolar concentration of 5-AZA-CdR (Fig. 3a). Of note, the immunomodulatory activity of SGI-110 affected cancer cells irrespectively of tumor histotype. Along this line, exposure to SGI-110 up-regulated the constitutive values of mean fluorescence intensity of HLA class I antigens (from 130 to 460), HLA-A2 (from 68.3 to 176), and ICAM-1 (from 63 to 163.3) in Mel 531 melanoma cells, and HLA class I antigens (from 1,270 to 1,517), HLA-A2 (from 422 to 693), and ICAM-1 (from 332 to 581) in MZ-1257 renal cell carcinoma cells (Supplementary Figure 1S). No significant changes were observed in the up-regulated expression of immune molecules following increase in SGI-110 dose to 10 μM (data not shown). Interestingly, no modulation of HLA class I antigens, HLA-A2 allospecificity, and ICAM-1 was observed following treatment for neoplastic cells with 3′-3′-DpG (data not shown), further demonstrating that the immunomodulatory activity of SGI-110 is sustained by the delivery of 5-AZA-CdR.
Fig. 3.
Indirect immunofluorescence analysis of the antigenic profile of SGI-110-treated Mel 275 melanoma cells. a Mel 275 melanoma cells untreated (solid line) and treated with 1 μM SGI-110 (dotted line) or with 1 μM 5-AZA-CdR (dashed line) were sequentially incubated with the isotype-matched mouse Ig, the anti-HLA class I mAb W6/32, the anti-HLA-A2 mAb BB7.2 or the anti-ICAM-1 mAb 84H10, and with FITC-conjugated F(ab′)2 fragments of rabbit anti-mouse Ig. Cells were then analyzed by flow cytometry. b Paraformaldehyde-fixed and permeabilized Mel 275 melanoma cells, either untreated or treated with 1 μM SGI-110, were sequentially incubated with the anti-MAGE-A1 mAb MA454, the anti-NY-ESO-1 mAb D8.38, or the anti-gp100 mAb HMB-45, and with FITC-conjugated F(ab′)2 fragments of rabbit anti-mouse Ig, followed by flow cytometry. The reported numbers represent the percentage of TAA-positive cells in upper and lower right quadrants. Data are representative of three independent experiments
Intracytoplasmic flow cytometric analyses confirmed, at protein level, that treatment with SGI-110 1 μM induced and/or up-regulated the expression of CTA, as evidenced by the generation of MAGE-A1- and NY-ESO-1-positive cells in SGI-110-treated Mel 275 and Mel 531 melanoma cells and MZ-1257 renal cell carcinoma cells, as compared to untreated ones (Fig. 3b and Supplementary Figure 1S). On the other hand, in vitro exposure to SGI-110 did not significantly affect the constitutive expression of the TAA gp100 in all investigated cancer cells (Fig. 3b and Supplementary Figure 1S).
Methylation status of CTA promoters in neoplastic cells treated with SGI-110
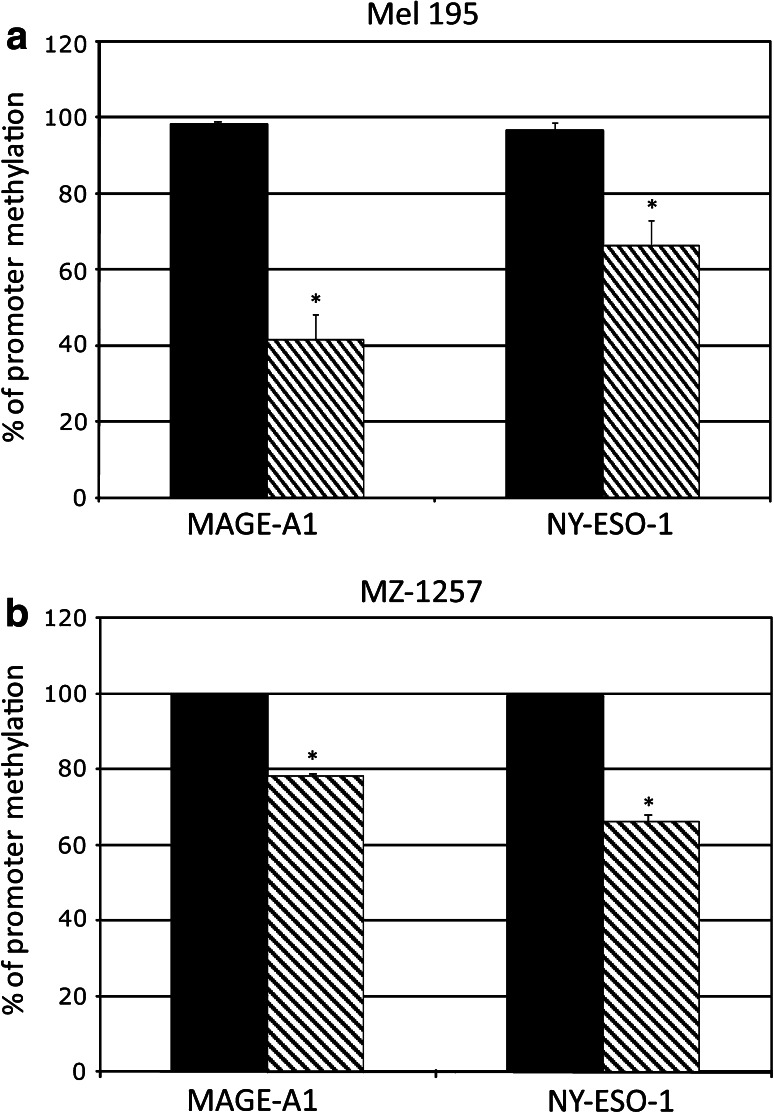
The methylation of CpG dinucleotides located in the promoter regions of MAGE-A1 and NY-ESO-1 was evaluated in selected investigated cancer cells to analyze whether hypomethylation of CTA promoter is the molecular mechanism sustaining the induced/up-regulated expression of CTA observed following treatment with SGI-110 (Fig. 4). DNA methylation analysis was done on the MAGE-A1-weakly positive and NY-ESO-1-negative Mel 195 melanoma cells and on the MAGE-A1- and NY-ESO-1-negative MZ-1257 cancer cells. Results showed that treatment with SGI-110 induced a significant (p < 0.01) reduction in the constitutive methylation levels of CTA promoters in investigated cancer cells (Fig. 4). Mean values of the percentage of demethylation induced by SGI-110 in MAGE-A1 and NY-ESO-1 promoters were 57 and 30 %, in Mel 195, and 22 and 33 % in MZ-1257 RCC cells, respectively (Fig. 4). The kinetic of hypomethylation induced by SGI-110 treatment in cancer cells was evaluated by the analysis of the methylation status of NY-ESO-1 promoter, in DNA extracted from Mel 195 cells at different time points during the treatment (Supplementary Figure 2S a). Results showed that the highest hypomethylating effect of SGI-110 was observed 2 days by the end of the 4 pulses treatment (Supplementary Figure 2S a). The hypomethylation of NY-ESO-1 promoter correlated with the levels of NY-ESO-1-specific mRNA expression induced by SGI-110 in Mel 195 cells, tested at the same investigated time points, throughout the treatment (Supplementary Figure 2S b).
Fig. 4.
qMSP analysis of the methylation status of CTA promoter in solid malignancies treated with SGI-110. Genomic DNA was extracted from Mel 195 (a) and MZ-1257 (b) cells, untreated (black columns) or treated (striped columns) with 1 μM SGI-110 and treated with sodium bisulfite. Real-time qMSP analyses of MAGE-A1 and NY-ESO-1 promoters were performed on bisulfite-modified genomic DNA using methylated- or unmethylated-specific primer pairs. Data are reported as percentage of methylation that was defined as the ratio between methylated molecules and the sum of methylated and unmethylated molecules
Susceptibility of SGI-110-treated melanoma cells to lysis by HLA-A2-restricted gp100-specific CTL
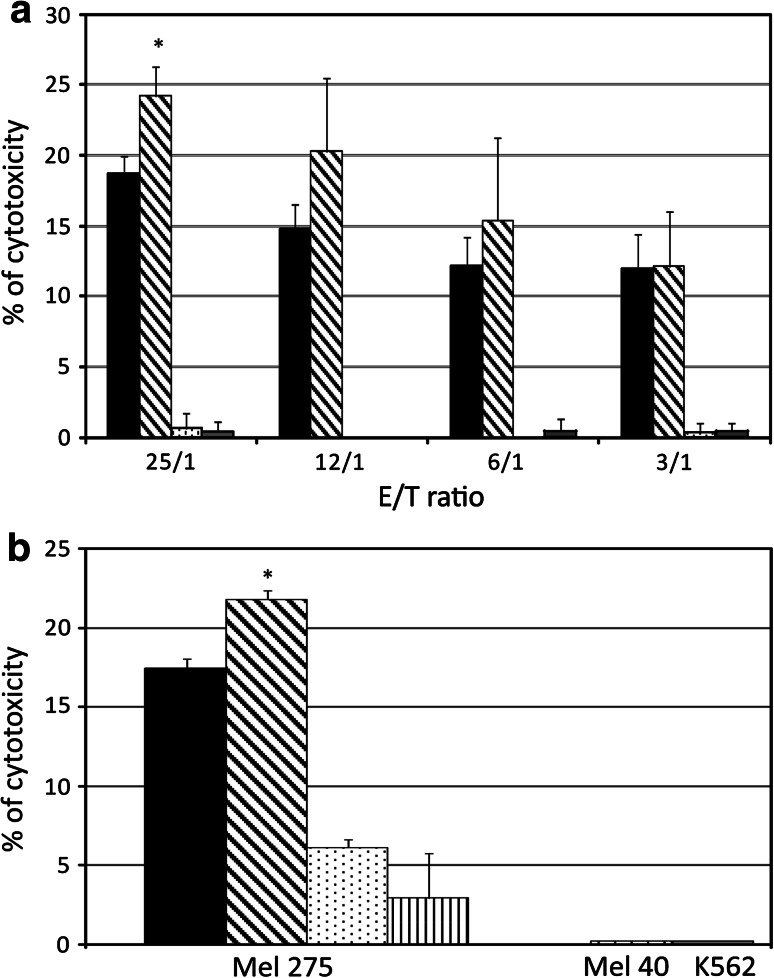
To evaluate whether modulation of the immune profile induced by SGI-110 was effective in increasing immune recognition of cancer cells, cytotoxicity of HLA-A2-restricted gp100-specific CTL against HLA-A2- and gp100-positive Mel 275 melanoma cells, untreated or treated with SGI-110, and HLA-A2-negative and gp100-positive Mel 40 melanoma cells were assessed at different E/T ratios. As evidenced in Fig. 5a, the results of three independent experiments showed that mean levels of gp100-specific CTL-mediated lysis were significantly (p < 0.05) higher for SGI-110-treated Mel 275 cells as compared to untreated cells at E/T ratio 25/1. This higher recognition of SGI-110-treated cells was also observed for lower E/T ratios, though the low lytic activity of the HLA-A2-restricted gp100-specific CTL bulk utilized did not allow to highlight a statistically significant difference (Fig. 5a). On the other hand, the HLA-A2-negative Mel 40 cells were resistant to the lysis by CTL at all the E/T ratios investigated (Fig. 5a). Furthermore, the absence of cytotoxicity observed when K562 cells were used as targets indicates the absence of natural killer activity in the CTL culture used for functional assays (Fig. 5a).
Fig. 5.
Susceptibility of SGI-110-treated melanoma cells to lysis by gp100-specific CTL. a Cytolytic activity of HLA-A2-restricted gp100-specific CTL was tested by LDH release assay against Mel 275 melanoma cells, untreated (black columns) or treated with 1 μM SGI-110 (striped columns), at effector/target (E/T) ratios of 25/1, 12/1, 6/1, and 3/1. Mel 40 melanoma cells (dashed columns) and K562 leukemia cells (gray columns) were used as control target cells. b Untreated (black columns) or SGI-110-treated (striped columns) Mel 275 melanoma cells, Mel 40 melanoma cells (dashed columns), and K562 leukemia cells (gray columns) were used as targets for HLA-A2-restricted gp100-specific CTL in LDH release assay at E/T ratio of 25/1. A fraction of SGI-110-treated Mel 275 cells has been pre-incubated with 20 μg/ml of anti-HLA class I antigens mAbW6/32 (dotted columns) or anti-ICAM-1 mAb 84H10 (vertical striped columns)
The increased susceptibility of SGI-110-treated Mel 275 melanoma cells to CTL lysis was suppressed when the assay was performed in the presence of anti-HLA class I mAb W6/32 or the anti-ICAM-1 mAb 84H10 at E/T ratio of 25/1 (Fig. 5b).
Similarly, the functional role of phenotypic changes induced by SGI-110 was further sustained by the higher susceptibility to gp100-specific CTL-mediated lysis observed in additional investigated cancer cells (Supplementary Figure 3S), directly related to a positive modulation of their immune profile (Supplementary Figure 1S and data not shown).
Discussion
This study identified and characterized the immunomodulatory potential of the newly synthesized DHA, SGI-110. In detail, we demonstrated the ability of SGI-110 to induce and/or to up-regulate the levels of expression of different immune-related antigens on human cancer cells, regardless of their histotype, improving their immunogenic potential and their immune recognition. This immunomodulatory activity of SGI-110 is particularly promising from a therapeutic point of view. Indeed, the global reversion of CTA-negative phenotype, induced by SGI-110 in all investigated histotypes, will permit targeting of neoplastic cells by (multi-)CTA-specific immune response. Thus, tumor immunomodulation by SGI-110 administration could become a pharmacologic approach allowing vaccination against multiple therapeutic targets, conferring biologic eligibility for CTA-directed immunotherapy to virtually all cancer patients. Similarly, treatment with SGI-110 could render tumors more susceptible to vaccination-stimulated CTA-specific immune responses, as it enhances the constitutively low levels of CTA expressed in cancer cells.
These proposed therapeutic applications of SGI-110 are strengthened by its long-lasting effects on CTA profile of tumor cells, as shown by the de novo expression of NY-ESO-1 that remained detectable 80 days after the end of treatment (Fig. 2). This finding is consistent with the inheritance of DHA-modified CTA profiles that we recently observed throughout the replication of cancer cells [2] and supports a prolonged immunological targeting of SGI-110-modified neoplastic cells.
An additional key finding of this study is the broad immunomodulatory activity of SGI-110 on cancer cells, regardless to their histological origin. Indeed, changes in CTA profile were invariably induced by SGI-110 in all investigated cancer cell lines, without significant differences between distinct histotypes analyzed.
A more global immunomodulatory potential of SGI-110 derives from its ability to up-regulate HLA class I antigens that play a crucial role in the presentation of TAA peptides to CTL [21] and in the susceptibility of tumor cells to CTL-mediated lysis, which correlates with levels of specific peptide HLA class I expression [22]. In this context, the up-regulation of HLA class I antigens, induced by SGI-110, could increase the number of molecules available for binding and presentation of antigenic peptides, accounting for the observed improved susceptibility of SGI-110-treated cancer cells to the cytotoxic activity of HLA-A2-restricted gp100-specific CTL. A further contribution to the increased recognition of SGI-110-treated cancer cells is given by the up-regulated expression of ICAM-1, a co-stimulatory ligand that binds to lymphocyte function-associated antigen-1, promoting the activation of T cells [23]. These assertions are sustained by the reversibility of the increased recognition of SGI-110-treated cells by gp100-specific CTL, following blockade of HLA class I antigens and ICAM-1 molecules with specific mAb. These additional findings clearly show that the immunomodulatory activity of SGI-110 is efficacious in improving CTL recognition of different therapeutic TAA, independently from their expression being regulated by DNA methylation. Along this line, further support to the feasibility and safety of the clinical use of DHA as immunomodulators comes from our most recent transcriptomic studies, in a syngeneic mouse cancer model, demonstrating that variations in gene expression profile, induced by in vivo administration of DHA, were mainly associated with immune processes and preferentially directed to tumor tissues, leaving essentially unaffected normal ones [24].
Overall, our present study widens the characterization of the biologic properties of SGI-110, demonstrating that its immunomodulatory activities are an important additional therapeutic feature of this compound that holds strong promises for its clinical application in cancer immunotherapies. This latter issue will receive further support from the ongoing first-in-human phase 1–2 clinical trial, which is moving SGI-110 into the cancer clinic to test its biological activity, safety, and efficacy in myelodysplastic syndromes and acute myeloid leukemia patients (ClinicalTrials.gov identifier: NCT01261312).
Altogether, these evidences strongly suggest that SGI-110 is an attractive therapeutic agent to comprehensively increase immunogenicity and immune recognition of cancer cells and provides the scientific rationale to translate the use of this novel DHA into clinical development of cancer immunotherapy, especially in combination with upcoming immunotherapeutic approaches.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
This work was supported in part by the grants from Associazione Italiana per la Ricerca sul Cancro (IG 11746 to MM and MFAG 9195 to LS), Fondazione Monte dei Paschi di Siena (SC), Harry J. Lloyd Charitable Trust (MM), Istituto Superiore di Sanità (MM), Istituto Toscano Tumori (MM), and Regione Toscana “Regional Health Research Program 2009″ (MM).
Conflict of interest
The authors declare no potential conflict of interest.
References
- 1.Sigalotti L, Fratta E, Coral S, et al. Epigenetic drugs as pleiotropic agents in cancer treatment: biomolecular aspects and clinical applications. J Cell Physiol. 2007;212:330–344. doi: 10.1002/jcp.21066. [DOI] [PubMed] [Google Scholar]
- 2.Fratta E, Sigalotti L, Colizzi F, et al. Epigenetically regulated clonal heritability of CTA expression profiles in human melanoma. J Cell Physiol. 2010;223:352–358. doi: 10.1002/jcp.22040. [DOI] [PubMed] [Google Scholar]
- 3.Fonsatti E, Nicolay HJ, Sigalotti L, et al. Functional up-regulation of human leukocyte antigen class I antigens expression by 5-aza-2′-deoxycytidine in cutaneous melanoma: immunotherapeutic implications. Clin Cancer Res. 2007;13:3333–3338. doi: 10.1158/1078-0432.CCR-06-3091. [DOI] [PubMed] [Google Scholar]
- 4.Coral S, Sigalotti L, Colizzi F, et al. Phenotypic and functional changes of human melanoma xenografts induced by DNA hypomethylation: immunotherapeutic implications. J Cell Physiol. 2006;207:58–66. doi: 10.1002/jcp.20540. [DOI] [PubMed] [Google Scholar]
- 5.Sigalotti L, Fratta E, Coral S, et al. Intratumor heterogeneity of cancer/testis antigens expression in human cutaneous melanoma is methylation-regulated and functionally reverted by 5-aza-2′-deoxycytidine. Cancer Res. 2004;64:9167–9171. doi: 10.1158/0008-5472.CAN-04-1442. [DOI] [PubMed] [Google Scholar]
- 6.Schrump DS, Fischette MR, Nguyen DM, et al. Phase I study of decitabine-mediated gene expression in patients with cancers involving the lungs, esophagus, or pleura. Clin Cancer Res. 2006;12:5777–5785. doi: 10.1158/1078-0432.CCR-06-0669. [DOI] [PubMed] [Google Scholar]
- 7.Weiser TS, Guo ZS, Ohnmacht GA, et al. Sequential 5-Aza-2 deoxycytidine-depsipeptide FR901228 treatment induces apoptosis preferentially in cancer cells and facilitates their recognition by cytolytic T lymphocytes specific for NY-ESO-1. J Immunother. 2001;24:151–161. doi: 10.1097/00002371-200103000-00010. [DOI] [PubMed] [Google Scholar]
- 8.Weiser TS, Ohnmacht GA, Guo ZS, Fischette MR, Chen GA, Hong JA, Nguyen DM, Schrump DS. Induction of MAGE-3 expression in lung and esophageal cancer cells. Ann Thorac Surg. 2001;71:295–301. doi: 10.1016/S0003-4975(00)02421-8. [DOI] [PubMed] [Google Scholar]
- 9.Momparler RL. Molecular, cellular and animal pharmacology of 5-aza-2′-deoxycytidine. Pharmacol Ther. 1985;30:287–299. doi: 10.1016/0163-7258(85)90053-1. [DOI] [PubMed] [Google Scholar]
- 10.Yoo CB, Jeong S, Egger G, Liang G, Phiasivongsa P, Tang C, Redkar S, Jones PA. Delivery of 5-aza-2′-deoxycytidine to cells using oligodeoxynucleotides. Cancer Res. 2007;67:6400–6408. doi: 10.1158/0008-5472.CAN-07-0251. [DOI] [PubMed] [Google Scholar]
- 11.Issa JP, Roboz G, Rizzieri D, Faderl S, O’Connell C, Stock W, Tibes R, Griffiths E, Yee K, Chung W, Choy G, Oganesian A, Taverna P, Azab M, Kantarjian H (2012) Abstract LB-214: Interim results from a randomized Phase 1-2 first-in-human (FIH) study of PK/PD guided escalating doses of SGI-110, a novel subcutaneous (SQ) second generation hypomethylating agent (HMA) in relapsed/refractory MDS and AML. Cancer Res 72
- 12.Chuang JC, Warner SL, Vollmer D, Vankayalapati H, Redkar S, Bearss DJ, Qiu X, Yoo CB, Jones PA. S110, a 5-Aza-2′-deoxycytidine-containing dinucleotide, is an effective DNA methylation inhibitor in vivo and can reduce tumor growth. Mol Cancer Ther. 2010;9:1443–1450. doi: 10.1158/1535-7163.MCT-09-1048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Altomonte M, Gloghini A, Bertola G, Gasparollo A, Carbone A, Ferrone S, Maio M. Differential expression of cell adhesion molecules CD54/CD11a and CD58/CD2 by human melanoma cells and functional role in their interaction with cytotoxic cells. Cancer Res. 1993;53:3343–3348. [PubMed] [Google Scholar]
- 14.Coral S, Sigalotti L, Altomonte M, et al. 5-aza-2′-deoxycytidine-induced expression of functional cancer testis antigens in human renal cell carcinoma: immunotherapeutic implications. Clin Cancer Res. 2002;8:2690–2695. [PubMed] [Google Scholar]
- 15.Mutti L, Valle MT, Balbi B, Orengo AM, Lazzaro A, Alciato P, Gatti E, Betta PG, Pozzi E. Primary human mesothelioma cells express class II MHC, ICAM-1 and B7–2 and can present recall antigens to autologous blood lymphocytes. Int J Cancer. 1998;78:740–749. doi: 10.1002/(SICI)1097-0215(19981209)78:6<740::AID-IJC12>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 16.Calabro L, Fonsatti E, Altomonte M, Pezzani L, Colizzi F, Nanni P, Gattei V, Sigalotti L, Maio M. Methylation-regulated expression of cancer testis antigens in primary effusion lymphoma: immunotherapeutic implications. J Cell Physiol. 2005;202:474–477. doi: 10.1002/jcp.20133. [DOI] [PubMed] [Google Scholar]
- 17.Coral S, Sigalotti L, Gasparollo A, Cattarossi I, Visintin A, Cattelan A, Altomonte M, Maio M. Prolonged upregulation of the expression of HLA class I antigens and costimulatory molecules on melanoma cells treated with 5-aza-2′-deoxycytidine (5-AZA-CdR) J Immunother. 1999;22:16–24. doi: 10.1097/00002371-199901000-00003. [DOI] [PubMed] [Google Scholar]
- 18.Ausubel FM, Brent R, Kingston RE, et al. Current protocols in molecular biology. New York: Wiley; 1998. [Google Scholar]
- 19.Li LC, Dahiya R. MethPrimer: designing primers for methylation PCRs. Bioinformatics. 2002;18:1427–1431. doi: 10.1093/bioinformatics/18.11.1427. [DOI] [PubMed] [Google Scholar]
- 20.Caballero OL, Chen YT. Cancer/testis (CT) antigens: potential targets for immunotherapy. Cancer Sci. 2009;100:2014–2021. doi: 10.1111/j.1349-7006.2009.01303.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Aptsiauri N, Cabrera T, Garcia-Lora A, Lopez-Nevot MA, Ruiz-Cabello F, Garrido F. MHC class I antigens and immune surveillance in transformed cells. Int Rev Cytol. 2007;256:139–189. doi: 10.1016/S0074-7696(07)56005-5. [DOI] [PubMed] [Google Scholar]
- 22.Weidanz JA, Nguyen T, Woodburn T, Neethling FA, Chiriva-Internati M, Hildebrand WH, Lustgarten J. Levels of specific peptide-HLA class I complex predicts tumor cell susceptibility to CTL killing. J Immunol. 2006;177:5088–5097. doi: 10.4049/jimmunol.177.8.5088. [DOI] [PubMed] [Google Scholar]
- 23.Lebedeva T, Dustin ML, Sykulev Y. ICAM-1 co-stimulates target cells to facilitate antigen presentation. Curr Opin Immunol. 2005;17:251–258. doi: 10.1016/j.coi.2005.04.008. [DOI] [PubMed] [Google Scholar]
- 24.Coral S, Covre A, Jmg Nicolay H, et al. Epigenetic remodelling of gene expression profiles of neoplastic and normal tissues: immunotherapeutic implications. Br J Cancer. 2012 doi: 10.1038/bjc.2012.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.