Abstract
Glioblastomas are primary intracranial tumors for which there is no cure. Patients receiving standard of care, chemotherapy and irradiation, survive approximately 15 months prompting studies of alternative therapies including vaccination. In a pilot study, a vaccine consisting of Lucite diffusion chambers containing irradiated autologous tumor cells pre-treated with an antisense oligodeoxynucleotide (AS-ODN) directed against the insulin-like growth factor type 1 receptor was found to elicit positive clinical responses in 8/12 patients when implanted in the rectus sheath for 24 h. Our preliminary observations supported an immune response, and we have since reopened a second Phase 1 trial to assess this possibility among other exploratory objectives. The current study makes use of a murine glioma model and samples from glioblastoma patients in this second Phase 1 trial to investigate this novel therapeutic intervention more thoroughly. Implantation of the chamber-based vaccine protected mice from tumor challenge, and we posit this occurred through the release of immunostimulatory AS-ODN and antigen-bearing exosomes. Exosomes secreted by glioblastoma cultures are immunogenic, eliciting and binding antibodies present in the sera of immunized mice. Similarly, exosomes released by human glioblastoma cells bear antigens recognized by the sera of 6/12 patients with recurrent glioblastomas. These results suggest that the release of AS-ODN together with selective release of exosomes from glioblastoma cells implanted in chambers may drive the therapeutic effect seen in the pilot vaccine trial.
Keywords: Antisense, Exosome, Antibody, Diffusion chamber, GL261, Glioblastoma
Introduction
Astrocytic gliomas including Glioblastoma multiforme (GBM), the most common and devastating primary brain tumor in adults, are defined by certain pathological hallmarks including necrosis and vascular proliferation. Although conventional treatments, such as surgery, radiation, and chemotherapy have seen much progress over the past two decades, median survival remains 15 months for GBM patients. Poor prognosis and side effects associated with current regimens have led many investigators to novel therapies including immunotherapy. We conducted a pilot study in which autologous tumor cells were treated with an antisense deoxyoligonucleotide (AS-ODN) to the insulin-like growth factor receptor type 1 (IGF-1R) ex vivo, encapsulated in a Lucite chamber with 0.1-μM pores and implanted into the rectus sheath for 24 h [1]. We observed objective clinical and radiographic responses in 8/12 patients that were consistent with the induction of anti-tumor immunity [1]. Consequently, a follow-up trial has been initiated based on this paradigm but with the addition of a novel IGF-1R/AS-ODN to the chamber (Clinical trial # NCT01550523.) The mechanisms underlying the apparent immunogenicity of this chamber-based vaccine are not fully understood. IGF-1R is a cell surface receptor, involved in a pathway that facilitates cell proliferation [2] and survival [3], that is commonly overexpressed by tumor cells. Downregulation of this molecule with AS-ODN is believed to induce apoptosis in treated cells which may cause the release of immunogens [4]. The AS-ODN used in this study also contains CpG and other immunostimulatory motifs [5–10].
Based on the 0.1-μM pore size of the chambers, we recognized that the size of immunomodulatory agents must be small. While cell components released by dying cells could provide antigens capable of leaking from the chambers, another possibility is that IGF-1R/AS-ODN-treated tumor cells do not die, but release antigenic vesicles. Exosomes, small unilamellar vesicles (50–100 nM), released following fusion of the multivesicular compartment [11] with the plasma membrane via a mechanism resembling exocytosis [12] and the somewhat larger microvesicles (100-200 nm) that bud directly from the cell surface [13] are both candidates. Several methods have been utilized to characterize vesicles including immunophenotyping [14, 15] and bead-based flow cytometry assays [16, 17]. Exosomes can be defined by the presence of proteins found in the late vesicular compartment [18–20] including several tetraspanins (CD9, CD63, and CD81). Secreted by a wide variety of normal cells [21–24], exosomes are thought to be involved in intercellular communication [25] and membrane exchange [26]. However, exosomes are also secreted by multiple types of tumor cells including GBM [18, 26–28], and the literature is conflicted concerning whether these are naturally immunogenic [15, 18, 29–33] or tolerogenic/immunosuppressive [28, 34, 35]. The current study is directed at establishing whether the release of immunostimulatory AS-ODN and exosomes from IGF-1R/AS-ODN-treated GBM tumor cells may explain the therapeutic effects seen in the pilot clinical trial.
Methods
Cell lines and reagents
Human glioblastoma cell line, U118 [American Type Culture Collection (ATCC) HTB-15], was maintained in DMEM supplemented with 10 % fetal bovine serum (FBS) and gentamicin (all from Invitrogen). Primary patient tumor cell lines were generated as described previously [36]. Briefly, fresh tumor tissue was mechanically disrupted and trypsinized to obtain single cell suspensions. To obviate any concerns of immune activation from exogenous antigens derived from FBS, human glioblastoma tumor cells were cultured on Biocoat plates (BD Bioscience) in Neurobasal medium supplemented with penicillin/streptomycin, amphotericin B, B27 supplement, and N2 supplement (All from Invitrogen.) Immediately before use, basic fibroblast growth factor (B-FGF, 50 ng/ml) and epidermal growth factor (EGF, 50 ng/ml, both from R&D Systems) were added to the culture medium. Human peripheral blood mononuclear cells (PBMC) were purified from normal donor buffy coats using Ficoll–Hypaque centrifugation. All human tissue was collected according to a protocol approved by the Thomas Jefferson University Institutional Review Board.
Murine glioma cell line GL261 (Glioma 261, NCI Tumor Repository) and lymph node cell cultures were maintained in RPMI supplemented with 10 % FBS and gentamicin (all from Invitrogen). Exosome-free FBS was used in all experiments and obtained by centrifuging FBS overnight (100,000xg).
Flow cytometry size calibration
Small particle gates were established by analyzing sized polystyrene microspheres (Invitrogen) on a FACSCaliber or LSRII cytometer (both from BD Bioscience).
Vesicle enrichment
Glioma cells were cultured for 3 days in complete medium. Supernatants were centrifuged (4 °C) at 1,200xg (20 min) to remove debris. Supernatants were further centrifuged at 20,000xg (30 min) to yield a population of larger vesicles. A final centrifugation at 100,000xg (60 min) generated a population of smaller vesicles. Vesicle pellets were resuspended in PBS for further analysis, immunization, or plating.
Chamber diffusion assay
Glioma cells (3 × 106 cells) were incubated with fluorescent dyes to label lipid membranes (0.2 mg/ml 3,3′-dioctadecyloxacarbocyanine perchlorate; DiO, Invitrogen) for 30 min at 37 °C. Large and small vesicle preparations isolated from cells labeled with fluorescent dyes or FITC-tagged NOBEL AS-ODN (IDT) were encapsulated in vaccine chambers and placed in six-well plates containing phosphate buffered saline (PBS). Chambers were removed, and the extracapsular fluid (PBS) was assayed for fluorescence using a plate reader (Synergy H1, Biotek). In some experiments, unlabeled exosomes, DiO-labeled exosomes or microvesicles, or the extracapsular PBS from chamber wells were analyzed on a LSR II flow cytometer (BD Biosciences).
Flow cytometric phenotyping
Small vesicle preparations were aliquoted into 96 well, V-bottom plates (20 μl/well). To each well, mAb specific for human CD11c (B-ly6), CD14 (M5E2), CD25 (M-A251), CD63 (H5C6), CD71 (M-A712), CD81 (JS-81), CD83 (HB15e, all from BD Bioscience) were added as phycoerythrin conjugates. Vesicle/antibody suspensions were incubated for 40 min at 4 °C, transferred to flow cytometry tubes and analyzed immediately by flow cytometry (FACSCaliber or LSRII, BD Bioscience). Post-collection analysis was performed with FlowJo software (Tree Star Inc).
Electron microscopy
Small vesicle preparations were fixed with 2.5 % glutaraldehyde and 2.0 % paraformaldehyde in a 0.1 M cacodylate buffer, pH 7.4, for 1 h at 4 °C, washed, and post-fixed in 2 % osmium tetroxide, subsequent to dehydration in a graded ethanol series. Samples were embedded in Poly/Bed 812 (Polysciences Inc,), sectioned, stained with bismuth subnitrate, and examined with a JEOL 1010 electron microscope fitted with a Hamamatsu digital camera system and AMT Advantage image capturing software.
AS-ODN uptake and stimulation experiments
Human PBMC were incubated with FITC-tagged NOBEL AS-ODN (107 cells/1 μg AS-ODN/ml, IDT) for 30 min at 37 °C or unlabeled AS-ODN (107 cells/100 μg AS-ODN/ml, Girindus) for 24 h at 37 °C. Samples were aliquoted into 96 well, V-bottom plates (100 μl/well). To each well, fluorescently conjugated mAb specific for human CD11c (B-ly6), CD14 (M5E2), CD16 (3G8), CD20 (2H7), CD56 (B159), CD80 (L307.4), CD83 (HB15e), CD86 (FUN-1), CD123 (9F5, all from BD Bioscience) were incubated for 40 min at 4 °C and analyzed immediately by flow cytometry (EasyCyte 8HT, Millipore). Post-collection analysis was performed with FlowJo software (Tree Star Inc).
Mice, immunization, and treatment
Male, 8–10 week old C57BL/6 mice were purchased from Taconic. GL261 cells were harvested and resuspended in PBS-containing NOBEL AS-ODN (107 cells/40 mg AS-ODN/ml). The mixture of cells and AS-ODN (100 μl), or PBS were loaded into Lucite diffusion chambers capped with 0.1-μM Millipore filters (vaccine chambers) [1, 4, 37] and implanted into a subcutaneous pocket in left flank of anesthetized mice. Chambers were removed after 24 h.
In other experiments, exosomes were harvested from untreated or NOBEL AS-ODN-treated GL261 cells and quantified using a Bradford–Lowry protein quantitation assay (BioRad). Exosomes were diluted in PBS and subcutaneously injected into the left flank of anesthetized mice.
Wild-type GL261 (106 cells) were subcutaneously injected into the right flank, 1 week following exosome injections or 2 weeks following chamber explantation. Mice were monitored every 3–4 days for tumor growth and killed once tumor volumes reached 1 cm3. Tumors were photographed (Nikon Coolpix digital camera) and stored at −80 °C. Tumor sections were cut, stained with hematoxylin and eosin, and imaged with a Leica DM6000 microscope and DFC295 color camera. The Thomas Jefferson University Institutional Animal Care and Use Committee approved all murine experiments.
Ex vivo cytokine induction and analysis
Draining lymph nodes (DLN) were collected from C57BL/6 mice either 3 days following 3 weekly injections of GL261-derived exosomes or 21 days following a single injection of exosomes (2 μg). DLN were dissected with scalpels, digested with a mixture of collagenase (1 mg/ml)/DNase (0.02 mg/ml, both from Sigma-Aldrich) for 30 min, and isolated cells cultured ex vivo in the presence or absence of exosomes for 3 days. Brefeldin A (BFA, BD Bioscience) was added to cultures during the last 18 h to allow cytokine accumulation in cells. Cultured cells were collected in 96-well, V-bottom plates and stained with fluorescently conjugated mAb specific for mouse CD3 (UCHT1), CD4 (RPA-T4), and CD8 (RPA-T8, all from BD Bioscience) for 40 min at 4 °C, washed and incubated with BD Cytofix/Cytoperm™, and stained with fluorescently conjugated mAb specific for mouse interleukin-2 (IL2, MQ1-17H12), IL4 (8D4-8), IL5 (TRFK5), IL10 (JES3-19F1), IL13 (JES 10-5A2), or Ifn-γ (B27, all from BD Bioscience) in the presence of BD Perm/Wash™ buffer. Cells were then analyzed immediately by flow cytometry (EasyCyte 8HT, Millipore). Post-collection analysis was performed with FlowJo software (Tree Star Inc).
CELISA
Blood samples were collected in heparinized capillary tubes from the orbital plexus while mice were under anesthesia. Tubes were centrifuged at 300xg (20 min), and plasma was collected, frozen, and stored at −80 °C.
Round-bottom, 96-well plates were seeded with GL261 (36 k/well). Plasma samples were thawed, diluted with PBS (1:40), added to plates and incubated for 1 h at 4 °C with intermittent shaking. Bound antibody was detected using anti-mouse IgG-alkaline phosphatase and p-Nitrophenyl phosphate. Absorbance (405 nm) was measured at 1 h using a plate reader (Synergy H1, Biotek).
Exosomes ELISA
Blood from recurrent glioblastoma patients was collected in heparinized tubes before or during surgery. Tubes were centrifuged at 300xg (20 min), and plasma was collected, frozen, and stored at −80 °C.
Immulon 4HBX 96-well plates (Dynex) were coated with exosomes generated from primary human tumor cell culture supernatants (24 h at 4 °C). Plasma samples were thawed, diluted with PBS (1:10), added to plates, and incubated overnight at 4 °C. Bound antibody was detected using fluorescent antibodies specific for human IgM (APC) and IgG (PE, both from BD Bioscience). Fluorescence was measured using a plate reader (Synergy H1, Biotek).
Statistical analysis
All statistical analysis was performed using JMP software (SAS Institute). Student’s t test was used to assess relationships between tumor incidence groups, murine antibody binding and ex vivo/in vitro cytokine analyses. Log-rank and Wilcoxon tests were used to determine significance between Kaplan–Meier groups. Kruskal–Wallis one-way ANOVA followed by Tukey’s post-test analysis were used to analyze group differences within exosome-chamber diffusion and glioblastoma patient antibody binding experiments.
Results
Flow cytometric analysis of small particles
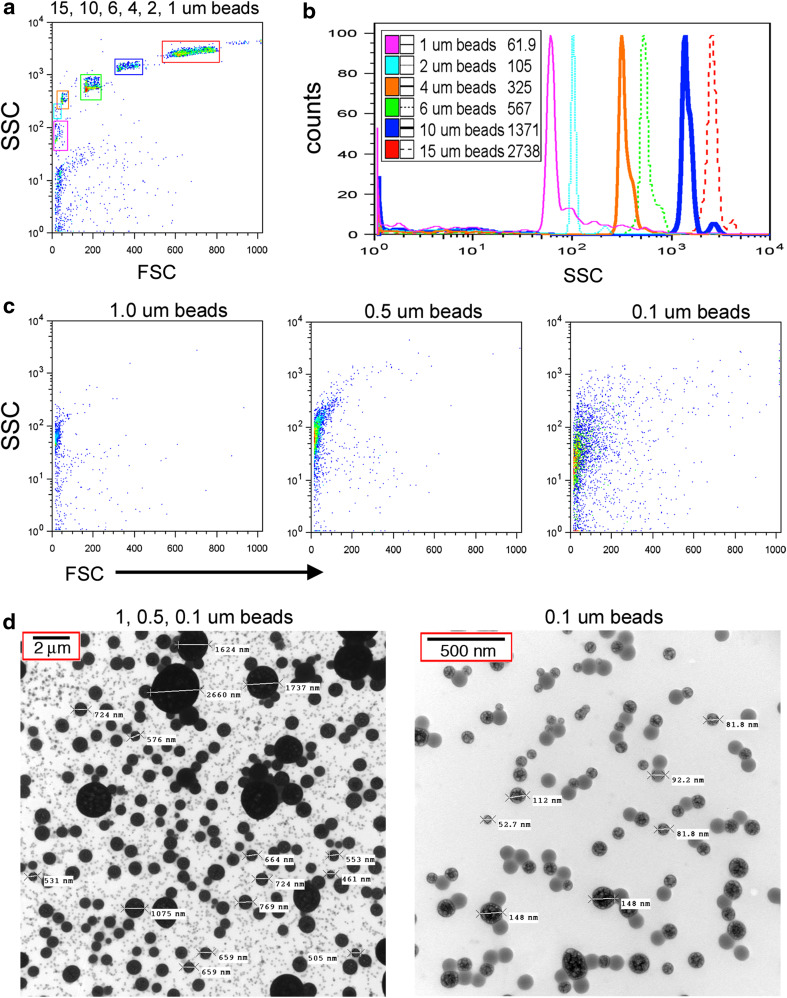
Transient implantation of GBM patients with IGF-1R/AS-ODN treated autologous tumor cells encapsulated in a diffusion chamber has shown therapeutic potential [1] through mechanisms that are not entirely understood but thought to involve the induction of an immune response. Immunogenic material must be able to cross the 0.1-μM pores of the chamber membrane. As shown in Fig. 1, we established flow cytometry parameters to investigate the nature of any small particles that may be released from chambers. Initially, mixtures containing commercial calibration beads were used to define profiles (15–1 μM) of six distinct populations (Fig. 1a) readily distinguished by side-scatter (Fig. 1b). Beads more likely to represent the size of particles released from chambers (1.0, 0.5, or 0.1 μM) also differ by side-scatter with somewhat less resolution in scatter as particle size decreases (Fig. 1c). To confirm the size of the calibration beads, we collected electron microscopy images. The largest beads (1 μm) are seen as black spheres with a measured diameter of 1–2 μM (Fig. 1d, left panel). Similarly, the 0.5-μM beads are dark gray in color with a measured diameter of 0.4–0.8 μM (Fig. 1d, left panel). The 0.1-μm beads appear as light gray specks (Fig. 1d, left panel) with measurements of 0.05–0.15 μM particles (Fig. 1d, right panel).
Fig. 1.
Analysis of small particles by flow cytometry and electron microscopy. a Forward scatter (FSC) and side scatter (SSC) analysis of large calibration beads (1–15 μm in diameter). b SSC histograms of gated bead populations. Median intensities are listed in the legend. c FSC and SSC analysis of small calibration beads (0.1–1 μm in diameter). d Electron microscopy images of small calibration beads (0.1–1 μm in diameter). Scale bars are located in the upper left corner of the images
Vaccine chambers release small particles in a time-dependent manner
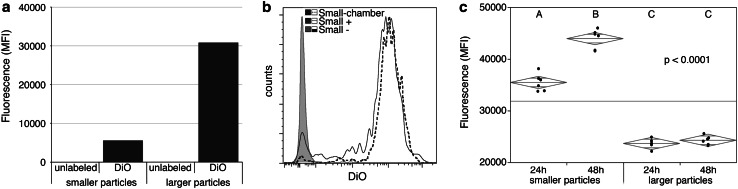
To assess particles released by the glioblastoma cells that are encapsulated in our vaccine paradigm, we added a fluorescent lipophilic dye to 3-day-old cell cultures which would label the cell membranes and any membrane-based vesicles present in the supernatant. We then assayed the culture supernatant using our novel, small particle flow cytometry detection methods. First, particles were crudely separated into two different size ranges based on centrifugation. As expected, smaller particles were fluorescent, but at lower levels than larger particles (Fig. 2a). More extensive analysis of smaller particles revealed that they exhibit a 3-log increase in fluorescence over that of unlabeled particles (Small-) and readily diffuse from the chambers (Small-chamber). Figure 2c shows that over time, these chambers release significant amounts of fluorescence (p < 0.0001) only when seeded with smaller particles recovered from cultured cells.
Fig. 2.
Vaccine chambers release small particles. Membranous particles in glioma culture supernatants were labeled with lipophilic dye (DiO). Particles were separated based on size using ultracentrifugation. a, b Unencapsulated particles were analyzed by flow cytometry. a Median fluorescence intensity (MFI) and b representative histograms of unlabeled (−) or DiO-labeled particles (+) are shown. b, c DiO-labeled particles were encapsulated in chambers and incubated in wells containing PBS. Extracapsular fluid was analyzed by flow cytometry (b, Small-chamber) or plate reader (c). MFI at 24 and 48 h is displayed with group median (diamond center line) and 95 % confidence intervals (diamond points). Statistical significance was assessed by ANOVA and Tukey’s post hoc analysis (letter groupings are statistically different, p < 0.0001)
Small particles produced by glioma cells characterize phenotypically as exosomes
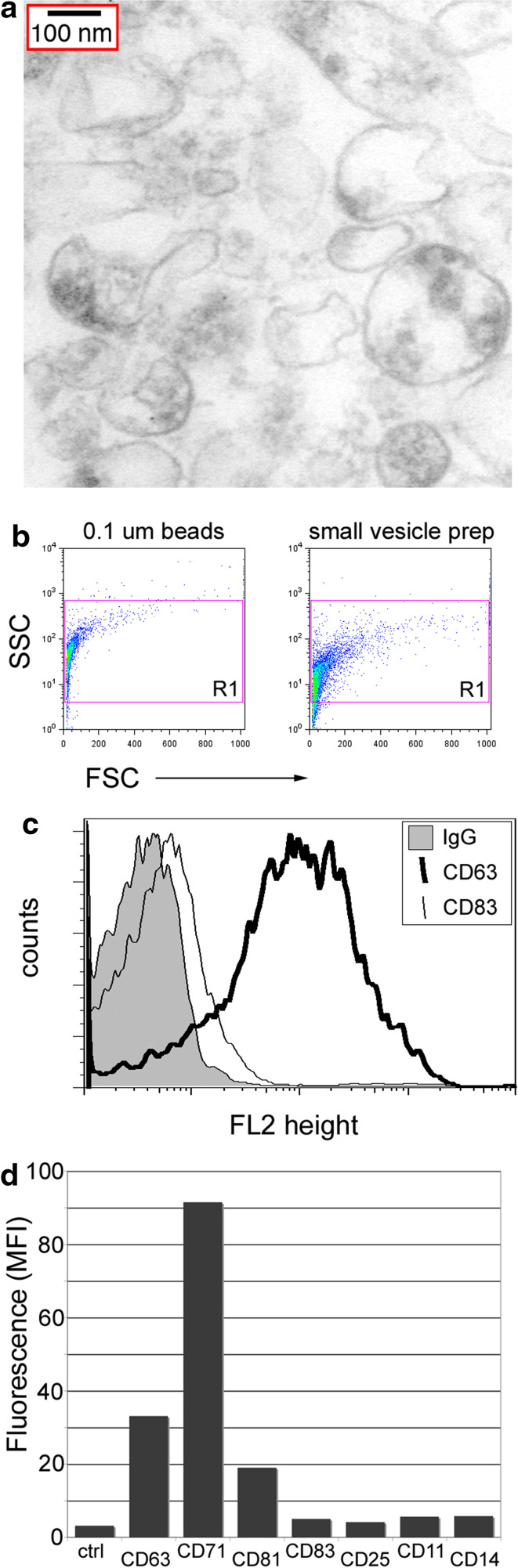
Electron microscopy analysis of small vesicles released from glioblastoma cells revealed vesicles of 0.05–0.2 μM in size (Fig. 3a). A small particle flow cytometry gate based on the scatter profiles of 0.1-μm beads (Fig. 3b, left panel), includes a majority of the particles in small vesicle preparations from cultured glioblastoma cells (Fig. 3b, right panel). These small vesicles bind antibodies specific for the endosomal antigens CD63, CD71, and low levels of CD81, but not antibodies specific for irrelevant cell surface antigens including CD83 (Fig. 3c, d) and are therefore defined as exosomes.
Fig. 3.
Small, glioma-derived particles are exosomes. a Electron microscopy image of small particle preparation is shown with a 100-nm scale bar. b–d Small particles were stained with monoclonal antibodies and analyzed by flow cytometry. b Scatter profiles of 0.1-μm beads (left panel) and small particles (right panel) were used to define analysis gate, R1. c Representative histogram plots for classical exosome antigen (CD63) or control antigen (IgG and CD83) fluorescence in small particles. d Summary plot containing median fluorescent intensities for endosomal and cell surface marker antibody labeling of small glioma particles
Vaccine chambers release AS-ODN that activates lymphoid-derived cells
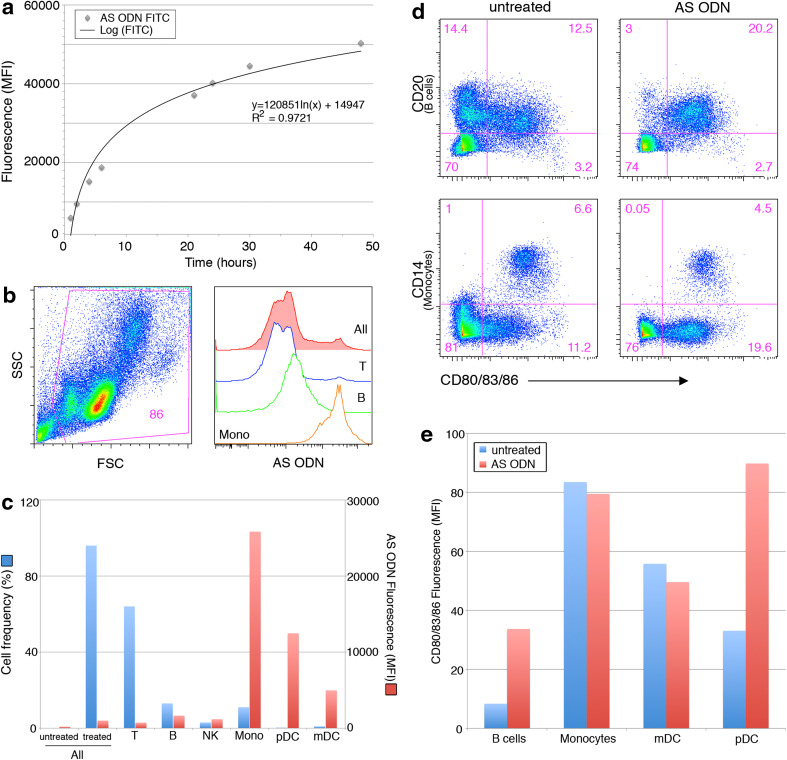
In addition to exosomes, our vaccine chamber also contains NOBEL, an AS-ODN which was originally used to modulate IGF-1R expression and tumor cell viability [4]. However, due to the presence of several potentially immunomodulatory motifs [6–10], NOBEL may impact immune functions such as antigen presentation. This AS-ODN is expected to freely pass through a 0.1-μm pore. This was confirmed by analysis of extracapsular fluid from cultures of chambers containing fluorescently tagged AS-ODN. Levels of fluorescence in the extracapsular fluid increased logarithmically over 48 h (Fig. 4a, R 2 = 0.9721). To determine if NOBEL released from the chamber may contribute to the therapeutic paradigm, we performed in vitro AS-ODN uptake experiments with PBMC and assayed for activation of immune cell subsets. The highest uptake of AS-ODN occurred in endocytic antigen presenting cells: monocytes, dendritic cells (DC), and B cells (Fig. 4b, c). Negligible uptake was observed in T cells or NK cells (Fig. 4b, c). AS-ODN-treated plasmacytoid dendritic cells (pDC), and B cells, increased expression of costimulatory molecules important in T cell activation (CD80, 83, and 86). Despite observing the highest levels of AS-ODN uptake, expression levels of CD80, 83, and 86 were unaltered in monocytes and myeloid dendritic cells (mDC, Fig. 4d, e).
Fig. 4.
Antigen-presenting cells take up AS-ODN released from chambers and express increased costimulatory molecules. a FITC-conjugated AS-ODN was encapsulated in diffusion chambers and incubated in wells containing PBS. Median fluorescence intensity of extracapsular fluid was analyzed over 48 h on a plate reader. A best-fit line, corresponding equation, and R 2 value are included in the chart. b–e PBMC were incubated in the absence (untreated) or presence of FITC-conjugated AS-ODN for 30 min (b, c) or unlabeled AS-ODN for 24 h (d, e). Cell subsets were defined using lineage-specific antibodies. b Viable cells were identified using scatter gating. Representative histograms show increasing AS-ODN uptake by T cells, B cells, and monocytes. c Summary data of cell frequency (left Y-axis) and median fluorescence intensity (right Y-axis) of all major PBMC subsets following AS-ODN incubation. d Representative dot plots and e summary chart document expression levels of CD80/83/86 in PBMC before and after AS-ODN treatment
Implantation of chambers containing mouse GL261 glioma cells and IGF-1R/AS-ODN is immunogenic in mice through the release of exosomes and AS-ODN
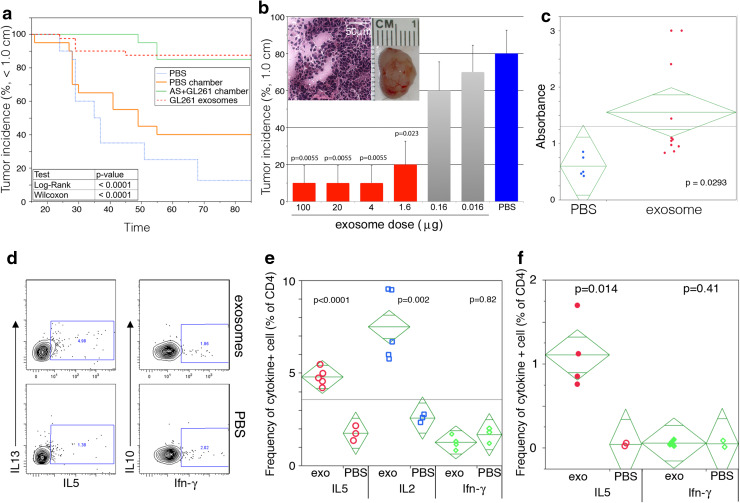
Immunization of mice by implantation with chambers containing GL261 mixed with IGF-1R/AS-ODN (AS + GL261 chamber) reduced their susceptibility to GL261 cells subsequently injected into the flank (Fig. 5a, p < 0.0001 as determined by log-rank and Wilcoxon tests). While 65 % of mice that received PBS-containing chambers grew tumors upon GL261 challenge, only 15 % receiving AS + GL261 chamber grew tumors and their growth was delayed by approximately 30 days over that seen in controls (Fig. 5a). To determine if the protective effect may be due to exosomes released from the chambers, exosomes were prepared from GL261 cells and different doses administered subcutaneously to the flanks of congenic C57BL/6 mice 1 week before subcutaneous implantation of GL261 cells. At exosome doses >1.6 μg, the vast majority of the mice failed to grow tumors (Fig. 5a, p < 0.0001). However, by a dose of 0.16, no reduction in tumor incidence was apparent (Fig. 5b). Gross pathological and histological examination revealed that by 32 days post-GL261 cell inoculation the tumors from untreated animals generally consist of large (1 cm3), dense accumulations of cells (Fig. 5b inset). To confirm that the anti-tumor response is likely to be a consequence of immune mechanisms elicited by exosomes, mice were injected with 2 μg of exosomes and sera and lymph node cells assessed for GL261-specific immunity. Exosome inoculation resulted in the rapid induction of an immune response to GL261 cells, as measured by serum antibodies (Fig. 5c, p = 0.0293), which was associated with evidence of enhanced T cell activity. Firstly, lymph node CD4+ T cells from mice that received exosomes expressed more IL2 and IL5 (p < 0.0001 and p = 0.002, respectively) ex vivo than similar cells from PBS-inoculated animals (Fig. 5d–e). More importantly, lymph node CD4+ T cells from the animals that received exosomes expressed higher levels of IL5 (p = 0.014) when restimulated in vitro with the exosomes (Fig. 5f). Elevated production of Ifn-γ, IL10, or IL13 was not observed (Fig. 5d–f).
Fig. 5.
GL261-derived exosomes are highly immunogenic. C57BL/6 mice were implanted with: a PBS (n = 20), exosomes (n = 40), or vaccine chambers containing either AS + GL261 cells (n = 20) or PBS (n = 20); b varying concentrations of exosomes (n = 10 per concentration, 100–0 μg). Mice were challenged with 106 GL261 cells 1–2 weeks later, tumor growth was monitored for 85 days, and mice euthanized if tumors reached 1 cm in diameter. a The incidence of mice with tumors <1.0 cm is presented as a percentage in Kaplan–Meier curves with statistical significance being determined by the log-rank test (p < 0.0001) and Wilcoxon rank-sum test (p < 0.0001). b Tumor (1 cm) incidence is plotted as a percentage ± SD with statistical significance assessed by the Student’s t test (p = 0.023–0.0055). Inset contains images of an excised tumor and a representative H&E stained section(40× with 50 μM scale bar). c Levels of GL261-specific IgG antibodies in sera from PBS or exosome immunized mice. Statistical significance was assessed using a one-tailed Student’s t test (p = 0.0293). Cytokine production was assessed by intracellular flow cytometry in DLN cultures from mice given: d–e weekly exosome (exo) or PBS injections (3×); f single injections of exosomes (exo) or PBS and restimulated in vitro with exosomes. Cytokine production in CD4+ gated T cells is shown by contour plots in (d) and by the frequency of cytokine producing CD4+ T cells in (e, f) where statistical significance was assessed by Student’s t test. The background proportions of cytokine producing cells in (f) were not statistically different between the groups of mice, averaging 0.48 ± 0.28 %, and were subtracted from test values
Exosomes from primary glioma cell lines are detected by antibodies in the plasma of recurrent glioblastoma patients
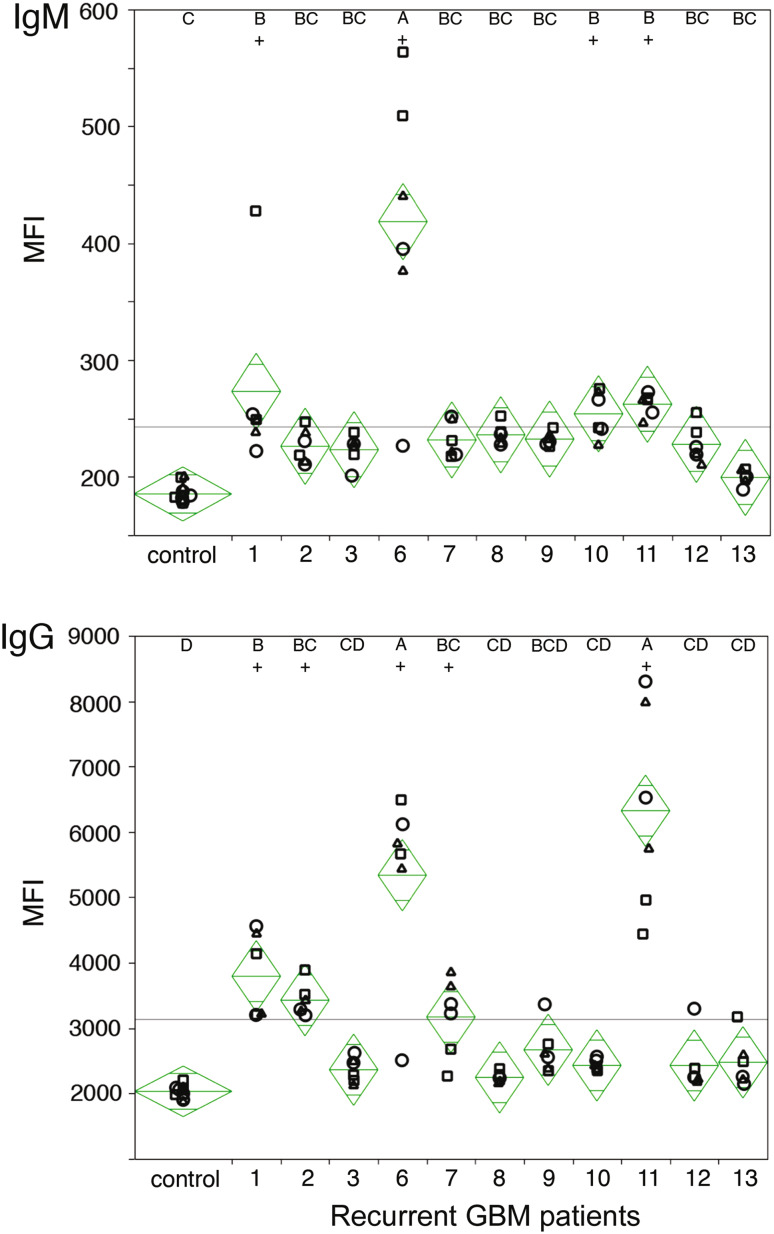
It is conceivable that due to tumor growth or surgical intervention that GBM patients may become sensitized to tumor antigens and that at least some of these may be borne by exosomes. To test this possibility, we coated 96-well plates with exosomes derived from three different primary glioblastoma cell lines and assessed binding of patient plasma antibodies. Antibody binding was measured using fluorescently conjugated secondary antibodies specific for human IgG or IgM. Regardless of the exosome source, we observed a statistically significant increase (p < 0.0001) in median fluorescence intensity (MFI) indicating the presence of exosome-binding antibodies. Four out of eleven patients exhibited glioblastoma exosome-specific IgM antibodies (Fig. 6, top panel ‘+’; patients 1, 6, 10, and 11), while five out of eleven patients possessed IgG antibodies (Fig. 6, bottom panel ‘+’; patients 1, 2, 6, 7, and 11). These data reflect that gliomas are associated with an array of common antigens recognized by host patients.
Fig. 6.
Antibodies from glioblastoma patient plasma bind exosomes. Exosomes, isolated from three primary patient tumor cell lines (circle, rectangle, triangle), were coated onto 96-well plates. Binding of antibodies in recurrent glioblastoma patient plasma collected before surgery were detected with fluorescently conjugated secondary antibodies specific for IgM (top) and IgG isotypes (bottom). Median fluorescence intensity (MFI) is displayed with sample median (diamond center line) and 95 % confidence intervals (diamond points). Statistical significance was assessed by ANOVA and Tukey’s post hoc analysis (letter groupings, p < 0.0001)
Discussion
We show here that the chambers, which act as carriers in our vaccination paradigm, release two components that impact the immune system. The included glioma cells release small antigenic particles that are characterized, using a novel flow cytometry-based approach, as exosomes. Scatter characteristics readily defined particles released by glioma cells in culture that fall within the range of 0.1-μM beads, consistent with the established size of exosomes [11, 14]. When a fluorescent lipophilic dye was included in the cell cultures, the elaborated exosomes also fluoresced, and the inclusion of a fluorescent parameter improved their detection by flow cytometry. Glioma-derived exosomes were found to express CD63, CD71, and CD81, endosomal antigens that define exosomes. No evidence of non-specific or Fc-mediated antibody binding was detected. While larger particles produced by glioma cells can be labeled with DiO, these do not diffuse across the chamber membrane.
To confirm that exosomes produced by glioma cells are immunogenic, we performed proof of principle studies using the mouse GL261 flank model. A single chamber containing GL261 cells and NOBEL AS-ODN, implanted in autologous mice overnight, protects 85 % of the animals from tumor growth following implantation of GL261 cells 14 days later. Administration of different doses of GL261 exosomes revealed protective immunity that was elicited by as little as 1.6 μg. The protection was associated with the production of antibodies that bound GL261 cells in vitro and evidence of T cell reactivity. Lymph node CD4+ T cells from mice recently inoculated with exosomes, but not PBS, produced IL2 and IL5 ex vivo, and CD4+ T cells isolated from the lymph nodes of mice given exosomes 21 days previously responded by expressing IL5 when restimulated with exosomes in vitro. IL5 is a member of the common β-chain family of cytokines, shared with GM-CSF and IL3 [38], is secreted by T helper type 2 (Th2) cells [39] and acts primarily on B cells [40] to promote antibody production [41]. While it has often been considered that exosomes are immunomodulatory [43] or immunosuppressive [28, 42], our results are consistent with previous studies indicating that at least some exosomes may be immunostimulatory. In mouse models, SMA560 astrocytoma-derived exosomes have been found to elicit antibodies and protect against the growth of SMA560 cells in the flank but not CNS [18], while a tumor-specific antibody response elicited by vaccination with GL261 lysate/CpG ODN and boosted with Fc-OX40L has been implicated in protection against GL261 tumor formation following implantation in the flank and CNS [43].
NOBEL AS-ODN has potentially immunogenic structural characteristics and rapidly crosses the chamber membrane. Using fluorescently labeled AS-ODN, we found that uptake over a 30-min period was highest in monocytes. Uptake by myeloid DC was also substantial being approximately 50 % of that of monocytes and nearly threefold higher than plasmacytoid DC. AS-ODN uptake by B cells was also detected at low levels, but not observed in T and NK cells. To examine the effects of AS-ODN on cell activation, we added NOBEL to cultures of PBMC. Overnight incubation with NOBEL AS-ODN had a significant impact on the expression of activation markers CD80, 83, and 86 in B cells and plasmacytoid DC. No effect on the expression of these markers by monocytes and myeloid DC was seen, possibly because these cells constitutively expressed the molecules in culture.
Exosome-elicited antibodies are a component of the immune response to GL261 in mice. Moreover, human glioblastoma cells spontaneously release exosomes, at least in culture. Together with the fact that surgical intervention is likely to release tumor antigens, this led us to speculate that exosome-reactive antibodies may be present in the circulation of some patients following resection of a glioblastoma. To test this possibility, we examined sera from patients returning to the clinic for treatment of recurrent glioblastoma for the capacity to bind exosomes prepared from three distinct primary glioblastoma cultures derived from unrelated subjects. Four of the eleven subjects showed statistically significant IgM antibody binding to all three of the exosome preparations. Three of these subjects and two others had significant levels of IgG antibodies reactive for all three exosome preparations. This confirms that a cohort of subjects with glioblastoma have been sensitized to their tumor antigens either spontaneously or as a consequence of surgery. Notably, antibody activity was shared for all three exosome preparations suggesting that there is a common antigen or antigens involved.
In conclusion, we have used new approaches to identify immunogenic glioma exosomes that are likely to provide the antigen source for our chamber-based vaccine. We have established that the included AS-ODN is released from the chambers, is immunostimulatory, and therefore likely to enhance presentation of these antigens. Finally, we have shown that exosomes prepared from glioblastoma cells from unrelated patients can be used in an ELISA format to detect antibodies in sera from a cohort of glioblastoma patients. These results strongly suggest that some patients can mount an immune response to antigens that are shared by different glioblastomas and may benefit from our vaccination strategy.
Acknowledgments
Financial Support: The Albert Stevens Foundation (David W. Andrews).
Conflict of interest
The authors declare that they have no conflict of interest.
Abbreviations
- AS-ODN
Antisense oligodeoxynucleotide
- B-FGF
Basic fibroblast growth factor
- DiO
3,3′-dioctadecyloxacarbocyanine perchlorate
- DLN
Draining lymph nodes
- EGF
Epidermal growth factor
- FBS
Fetal bovine serum
- GBM
Glioblastoma
- GM-CSF
Granulocyte macrophage colony stimulating factor
- IGF-1R
Insulin-like growth factor type 1 receptor PBS
- IL
Interleukin
- MFI
Median fluorescence intensity
- mDC
Myeloid dendritic cell
- PBMC
Peripheral blood mononuclear cells
- PBS
Phosphate buffered saline
- pDC
Plasmacytoid dendritic cell
- Th2
T helper type 2
References
- 1.Andrews DW, Resnicoff M, Flanders AE, et al. Results of a pilot study involving the use of an antisense oligodeoxynucleotide directed against the insulin-like growth factor type I receptor in malignant astrocytomas. J Clin Oncol. 2001;19:2189–2200. doi: 10.1200/JCO.2001.19.8.2189. [DOI] [PubMed] [Google Scholar]
- 2.Baserga R, Reiss K, Alder H, Pietrzkowski Z, Surmacz E. Inhibition of cell cycle progression by antisense oligodeoxynucleotides. Ann N Y Acad Sci. 1992;660:64–69. doi: 10.1111/j.1749-6632.1992.tb21058.x. [DOI] [PubMed] [Google Scholar]
- 3.Hernandez-Sanchez C, Blakesley V, Kalebic T, Helman L, LeRoith D. The role of the tyrosine kinase domain of the insulin-like growth factor-I receptor in intracellular signaling, cellular proliferation, and tumorigenesis. J Biol Chem. 1995;270:29176–29181. doi: 10.1074/jbc.270.49.29176. [DOI] [PubMed] [Google Scholar]
- 4.Resnicoff M, Abraham D, Yutanawiboonchai W, et al. The insulin-like growth factor I receptor protects tumor cells from apoptosis in vivo. Cancer Res. 1995;55:2463–2469. [PubMed] [Google Scholar]
- 5.Bauer S, Kirschning CJ, Hacker H, et al. Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci U S A. 2001;98:9237–9242. doi: 10.1073/pnas.161293498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Iho S, Yamamoto T, Takahashi T, Yamamoto S. Oligodeoxynucleotides containing palindrome sequences with internal 5′-CpG-3′ act directly on human NK and activated T cells to induce IFN-gamma production in vitro. J Immunol. 1999;163:3642–3652. [PubMed] [Google Scholar]
- 7.Kobayashi N, Hong C, Klinman DM, Shirota H. Oligodeoxynucleotides expressing polyguanosine motifs promote antitumor activity through the upregulation of IL-2. J Immunol. 2013;190:1882–1889. doi: 10.4049/jimmunol.1201063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krieg AM, Yi AK, Matson S, et al. CpG motifs in bacterial DNA trigger direct B-cell activation. Nature. 1995;374:546–549. doi: 10.1038/374546a0. [DOI] [PubMed] [Google Scholar]
- 9.Pasare C, Medzhitov R. Toll pathway-dependent blockade of CD4+ CD25+ T cell-mediated suppression by dendritic cells. Science. 2003;299:1033–1036. doi: 10.1126/science.1078231. [DOI] [PubMed] [Google Scholar]
- 10.Stein CA, Subasinghe C, Shinozuka K, Cohen JS. Physicochemical properties of phosphorothioate oligodeoxynucleotides. Nucleic Acids Res. 1988;16:3209–3221. doi: 10.1093/nar/16.8.3209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002;2:569–579. doi: 10.1038/nri855. [DOI] [PubMed] [Google Scholar]
- 12.Johnstone RM, Adam M, Hammond JR, Orr L, Turbide C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes) J Biol Chem. 1987;262:9412–9420. [PubMed] [Google Scholar]
- 13.Théry C, Boussac M, Véron P, et al. Proteomic analysis of dendritic cell-derived exosomes: a secreted subcellular compartment distinct from apoptotic vesicles. J Immunol. 2001;166:7309–7318. doi: 10.4049/jimmunol.166.12.7309. [DOI] [PubMed] [Google Scholar]
- 14.Raposo G, Nijman HW, Stoorvogel W, et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996;183:1161–1172. doi: 10.1084/jem.183.3.1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wolfers J, Lozier A, Raposo G, et al. Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat Med. 2001;7:297–303. doi: 10.1038/85438. [DOI] [PubMed] [Google Scholar]
- 16.Clayton A, Court J, Navabi H, et al. Analysis of antigen presenting cell derived exosomes, based on immuno-magnetic isolation and flow cytometry. J Immunol Methods. 2001;247:163–174. doi: 10.1016/S0022-1759(00)00321-5. [DOI] [PubMed] [Google Scholar]
- 17.Caby MP, Lankar D, Vincendeau-Scherrer C, Raposo G, Bonnerot C. Exosomal-like vesicles are present in human blood plasma. Int Immunol. 2005;17:879–887. doi: 10.1093/intimm/dxh267. [DOI] [PubMed] [Google Scholar]
- 18.Graner MW, Alzate O, Dechkovskaia AM, et al. Proteomic and immunologic analyses of brain tumor exosomes. FASEB J. 2009;23:1541–1557. doi: 10.1096/fj.08-122184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stoorvogel W, Kleijmeer MJ, Geuze HJ, Raposo G. The biogenesis and functions of exosomes. Traffic. 2002;3:321–330. doi: 10.1034/j.1600-0854.2002.30502.x. [DOI] [PubMed] [Google Scholar]
- 20.Théry C, Amigorena S, Raposo G, Clayton A (2006) Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol. Chapter 3: Unit 3.22. doi: 10.1002/0471143030.cb0322s30 [DOI] [PubMed]
- 21.Faure J, Lachenal G, Court M, et al. Exosomes are released by cultured cortical neurones. Mol Cell Neurosci. 2006;31:642–648. doi: 10.1016/j.mcn.2005.12.003. [DOI] [PubMed] [Google Scholar]
- 22.Kramer-Albers EM, Bretz N, Tenzer S, Winterstein C. Oligodendrocytes secrete exosomes containing major myelin and stress-protective proteins: trophic support for axons? Proteomics Clin Appl. 2007;1:1446–1461. doi: 10.1002/prca.200700522. [DOI] [PubMed] [Google Scholar]
- 23.Potolicchio I, Carven GJ, Xu X, et al. Proteomic analysis of microglia-derived exosomes: metabolic role of the aminopeptidase CD13 in neuropeptide catabolism. J Immunol. 2005;175:2237–2243. doi: 10.4049/jimmunol.175.4.2237. [DOI] [PubMed] [Google Scholar]
- 24.Zitvogel L, Regnault A, Lozier A, et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med. 1998;4:594–600. doi: 10.1038/nm0598-594. [DOI] [PubMed] [Google Scholar]
- 25.Février B, Raposo G. Exosomes: endosomal-derived vesicles shipping extracellular messages. Curr Opin Cell Biol. 2004;16:415–421. doi: 10.1016/j.ceb.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 26.Al-Nedawi K, Meehan B, Micallef J, et al. Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat Cell Biol. 2008;10:619–624. doi: 10.1038/ncb1725. [DOI] [PubMed] [Google Scholar]
- 27.Skog J, Wurdinger T, van Rijn S, et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol. 2008;10:1470–1476. doi: 10.1038/ncb1800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wieckowski E, Whiteside TL. Human tumor-derived vs dendritic cell-derived exosomes have distinct biologic roles and molecular profiles. Immunol Res. 2006;36:247–254. doi: 10.1385/IR:36:1:247. [DOI] [PubMed] [Google Scholar]
- 29.Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4:499–511. doi: 10.1038/nri1391. [DOI] [PubMed] [Google Scholar]
- 30.Andre F, Schartz NE, Movassagh M, et al. Malignant effusions and immunogenic tumour-derived exosomes. Lancet. 2002;360:295–305. doi: 10.1016/S0140-6736(02)09552-1. [DOI] [PubMed] [Google Scholar]
- 31.Chaput N, Schartz NE, André F, et al. Exosomes as potent cell-free peptide-based vaccine. II. Exosomes in CpG adjuvants efficiently prime naive Tc1 lymphocytes leading to tumor rejection. J Immunol. 2004;172:2137–2146. doi: 10.4049/jimmunol.172.4.2137. [DOI] [PubMed] [Google Scholar]
- 32.Creagh EM, O’Neill LA. TLRs, NLRs and RLRs: a trinity of pathogen sensors that co-operate in innate immunity. Trends Immunol. 2006;27:352–357. doi: 10.1016/j.it.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 33.Mignot G, Roux S, Thery C, Ségura E, Zitvogel L. Prospects for exosomes in immunotherapy of cancer. J Cell Mol Med. 2006;10:376–388. doi: 10.1111/j.1582-4934.2006.tb00406.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Morelli AE. The immune regulatory effect of apoptotic cells and exosomes on dendritic cells: its impact on transplantation. Am J Transplant. 2006;6:254–261. doi: 10.1111/j.1600-6143.2005.01197.x. [DOI] [PubMed] [Google Scholar]
- 35.Peche H, Renaudin K, Beriou G, Merieau E, Amigorena S, Cuturi MC. Induction of tolerance by exosomes and short-term immunosuppression in a fully MHC-mismatched rat cardiac allograft model. Am J Transpl. 2006;6:1541–1550. doi: 10.1111/j.1600-6143.2006.01344.x. [DOI] [PubMed] [Google Scholar]
- 36.Tang J, Flomenberg P, Harshyne L, Kenyon L, Andrews DW. Glioblastoma patients exhibit circulating tumor-specific CD8+ T cells. Clin Cancer Res. 2005;11:5292–5299. doi: 10.1158/1078-0432.CCR-05-0545. [DOI] [PubMed] [Google Scholar]
- 37.Abraham D, Rotman HL, Haberstroh HF, et al. Strongyloides stercoralis: protective immunity to third-stage larvae inBALB/cByJ mice. Exp Parasitol. 1995;80:297–307. doi: 10.1006/expr.1995.1036. [DOI] [PubMed] [Google Scholar]
- 38.Tavernier J, Tuypens T, Verhee A, et al. Identification of receptor-binding domains on human interleukin 5 and design of an interleukin 5-derived receptor antagonist. Proc Natl Acad Sci USA. 1995;92:5194–5198. doi: 10.1073/pnas.92.11.5194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lee GR, Fields PE, Griffin TJ, Flavell RA. Regulation of the Th2 cytokine locus by a locus control region. Immunity. 2003;19:145–153. doi: 10.1016/S1074-7613(03)00179-1. [DOI] [PubMed] [Google Scholar]
- 40.Rolink AG, Thalmann P, Kikuchi Y, Erdei A. Characterization of the interleukin 5-reactive splenic B cell population. Eur J Immunol. 1990;20:1949–1956. doi: 10.1002/eji.1830200912. [DOI] [PubMed] [Google Scholar]
- 41.McHeyzer-Williams MG. Combinations of interleukins 2, 4 and 5 regulate the secretion of murine immunoglobulin isotypes. Eur J Immunol. 1989;19:2025–2030. doi: 10.1002/eji.1830191109. [DOI] [PubMed] [Google Scholar]
- 42.Valenti R, Huber V, Iero M, Filipazzi P, Parmiani G, Rivoltini L. Tumor-released microvesicles as vehicles of immunosuppression. Cancer Res. 2007;67:2912–2915. doi: 10.1158/0008-5472.CAN-07-0520. [DOI] [PubMed] [Google Scholar]
- 43.Murphy KA, Erickson JR, Johnson CS, et al. CD8+ T cell-independent tumor regression induced by Fc-OX40L and therapeutic vaccination in a mouse model of glioma. J Immunol. 2014;192:224–233. doi: 10.4049/jimmunol.1301633. [DOI] [PMC free article] [PubMed] [Google Scholar]