Abstract
Purpose
To test the hypothesis that decrease in DNA methylation will increase the expression of cancer-testis antigens (CTA) and class I major histocompatibility complex (MHC)-encoded molecules by ovarian cancer cells, and thus increase the ability of these cells to be recognized by antigen-reactive CD8+ T cells.
Methods
Human ovarian cancer cell lines were cultured in the presence or absence of varying concentrations of the DNA demethylating agent 5-aza-2′-deoxycytidine (DAC) for 3–7 days. The expression levels of 12 CTA genes were measured using the polymerase chain reaction. The protein expression levels of class I MHC molecules and MAGE-A1 were measured by flow cytometry. T cell reactivity was determined using interferon-γ ELISpot analysis.
Results
DAC treatment of ovarian cancer cell lines increased the expression of 11 of 12 CTA genes tested including MAGE-A1, MAGE-A3, MAGE-A4, MAGE-A6, MAGE-A10, MAGE-A12, NY-ESO-1, TAG-1, TAG-2a, TAG-2b, and TAG-2c. In contrast, DAC treatment decreased the already low expression of the MAGE-A2 gene by ovarian cancer cells, a finding not previously observed in cancers of any histological type. DAC treatment increases the expression of class I MHC molecules by the cells. These effects were time-dependent over a 7-day interval, and were dose-dependent up to 1–3 μM for CTA and up to 10 μM for class I MHC molecules. Each cell line tested had a unique pattern of gene upregulation after exposure to DAC. The enhanced expression levels increased the recognition of 2 of 3 antigens recognized by antigen-reactive CD8+ T cells.
Conclusions
These results demonstrate the potential utility of combining DAC therapy with vaccine therapy in an attempt to induce the expression of antigens targeted by the vaccine, but they also demonstrate that care must be taken to target inducible antigens.
Electronic supplementary material
The online version of this article (doi:10.1007/s00262-008-0582-6) contains supplementary material, which is available to authorized users.
Keywords: Ovarian cancer, Class I MHC molecules, Cancer-testis antigens, DNA methylation, 5-Aza-2′-deoxycytidine
Introduction
Surgery and chemotherapy are the mainstays of ovarian cancer therapy but their use only results in a 5-year survival rate of less than 45% [44]. An additional treatment approach under investigation involves the immunization of patients with cancer antigen-derived peptides with the goal being to stimulate antigen-specific cytotoxic T lymphocytes (CTL) that will recognize and kill the tumor [25, 41, 42].
Cancer-testis antigens (CTA) represent a promising group of antigens whose inclusion in an ovarian cancer vaccine may prove efficacious. CTAs have been defined on the basis of their expression in a variable proportion of tumors of many different histological types, expression in the testis, and limited or no expression in other normal tissue [5, 31, 46, 61]. A potential drawback to the use of CTAs in vaccines is that CTAs are expressed in only a subset of all tumor samples studied, with ovarian cancer being considered a moderate expressor in comparison to other cancers [4, 46]. Additionally, CTA expression is frequently heterogeneous within a tumor line or sample [15, 26–28]. Because of these limitations, some ovarian cancer cells may escape immune recognition even if a vaccine based on CTAs is capable of inducing a strong, therapeutic CTL response. The ability to induce high levels of CTA expression in all ovarian cancer cells would overcome these limitations.
Treatment of melanoma cell lines with the demethylating agent 5-aza-2′-deoxycytidine (DAC) was first shown to upregulate the expression of the CTA MAGE-A1 [56]. This observation has since been extended to additional CTAs including BAGE [34], CAGE [7], GAGE1-2 [34, 51], GAGE-1-6 [8, 34, 51], MAGE-A2 [8, 51], MAGE-A3 [8, 21, 34, 51, 58], MAGE-A4 [8, 51], MAGE-A10, and NY-ESO-1 [8, 51, 57]. The enhancement of CTA expression by DNA demethylating agents has also been observed in cells derived from breast cancer [21], colon cancer [30], esophageal cancer [58], gastric cancer [7, 34], lung cancer [58], mesothelioma [51], renal cell cancer [8], and thoracic cancer [57]. Only a single such study has been conducted with ovarian cancer in which microarray analysis demonstrated that DAC treatment of the OVCAR-3 cell line upregulated a variety of CTAs including: CTAG-1A and -1B; MAGE-A1, -A3, -A4, -A6, -A11, -A12, and -B2; and SPAN-X [37]. Upregulation of MAGE-A3 in DAC-treated OVCAR3 was further confirmed by PCR analysis [37]. Absent or low expression of CTAs is associated with promoter region hypermethylation, while high or upregulated expression of CTAs is associated with promoter region hypomethylation [7, 12, 13, 30, 51, 52]. The functional significance of the upregulation of NY-ESO-1 has been demonstrated by the increased recognition of the DAC-treated cells by antigen-specific CTL [8, 57].
The treatment of melanoma [10, 19, 20, 49] and esophageal squamous cell cancers [39] with DAC has also been shown to upregulate the expression of class I major histocompatibility complex (MHC)-encoded molecules. Like the expression of CTAs, the upregulation occurs through the demethylation of the class I MHC gene promoter regions [39]. Promoter methylation primarily regulates class I MHC expression between low and high levels [10, 19, 20], and with the exception of a single cell line [49], generally does not appear to be responsible for the lack of expression of class I MHC molecules [20]. The increased level of class I MHC expression on melanoma cells was functionally significant as it increased the recognition of the cells by antigen-specific CTL [19].
DAC also upregulates the expression of CTAs and class I MHC molecules in vivo. In a xenograft model, BALB/c nu/nu mice were engrafted with human melanoma cells and then treated with DAC [9]. After 1 week, the tumors had either de novo or upregulated expression of ten different CTAs tested, as well as upregulated expression of the class I MHC molecules HLA-A1 and HLA-A2. DAC treatment also induced expression of the murine CTA P1A in tumors of five different histological origins when grown in syngeneic mice [22]. It was further shown that the induced P1A was processed and presented by H-2Ld, and recognized by P1A-specific, H-2Ld-restricted CTL. Importantly, lung metastases were most effectively controlled when tumor bearing mice were pre-treated with DAC and then treated with the adoptive transfer of antigen-specific CTL, while the adoptive transfer of the CTL in the absence of DAC treatment was ineffective.
The effect of DAC on CTA and class I MHC molecule expression has also been assessed in cancer patients. When myelodysplastic syndrome or acute myeloid leukemia patients were treated with DAC over 3 days, MAGE-A1, SSX, and NY-ESO-1 were expressed de novo between 15 and 30 days following treatment [50]. Likewise, when lung, esophageal, and pleural mesothelioma patients were treated with DAC for 3 days and tumor biopsies obtained 1-day following treatment, a third of the samples demonstrated upregulation of NY-ESO-1 or MAGE-A3 [47].
Although CTAs represent a potentially rich source of antigens for inclusion in vaccines designed to treat ovarian cancer, the utility of the individual antigens is limited by their lack of expression in all ovarian cancers [4, 46]. Likewise, targeting of vaccine-induced CTL to ovarian cancer requires that the tumor cells express class I MHC molecules, but the tumors frequently have low levels of class I MHC expression [4]. To overcome these limitations, we sought to determine if the treatment of ovarian cancer cells with DAC would induce or upregulate both CTAs and class I MHC molecule expression.
Materials and methods
Cell culture medium
RPMI-1640 supplemented with 2 mM l-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin was further supplemented with 5% fetal bovine serum (RPMI-5FBS), 10% FBS (RPMI-10FBS), or 10% human AB-serum (RPMI-10HS).
Cell lines
The ovarian cancer lines CAOV-3 [3], CAOV-4 [29], COV413 [32], ES-2 [33], OV-90 [43], OVCAR-3 [23], SK-OV-3 [17], SW626 [18], TOV-21G [43], TOV-112D [43], and TTB-6 [4] were maintained in RPMI-10FBS. C1R-A2 and C1R-A3 were maintained in RPMI-5FBS supplemented with 300 μg/ml G418.
DAC treatment
Cells were counted and plated in new T75 flasks 1-day prior to the treatment. On the day of treatment the media was removed and replaced with media containing the appropriate concentration of DAC (Sigma-Aldrich, St. Louis, MO) that had been freshly dissolved at 1 mM in Dulbecco’s phosphate-buffered saline. After incubation with DAC for 3–7 days, the cells were harvested, counted, and then prepared for RNA extraction or flow cytometry.
Flow cytometry
Monoclonal antibodies (mAb) CR11-351 (anti-HLA-A2,A68,A69; γ1) [45], GAP-A3 (anti-HLA-A3; γ2a) [1], ME1-1.2 (anti-HLA-B7,B27; γ1) [16], and W6/32 (anti-HLA-A, B, C; γ2a) [40] were produced in our laboratory from the corresponding hybridoma and were used at a final concentration of 10 μg/ml. mAb 3F257 (anti-MAGE-A1; γ2a) (United States Biological, Swampscott, MA) was used at a final concentration of 10 μg/ml.
Class I MHC-specific mAbs were added to 2 × 105 cells and incubated for 60 min on ice. The cells were washed twice, 50 μl of a 1:50 dilution of sheep anti-mouse IgG-FITC (ICN, Irvine, CA) was then added, and the cells were incubated an additional 60 min on ice. The cells were then washed once, fixed with 0.5% paraformaldehyde in PBS, and analyzed on a FACSCalibur instrument (BD Biosciences, San Jose, CA). Binding of the 3F257 antibody was evaluated on cells that had been fixed and permeabilized according to the Cytofix/Cytoperm kit instructions (BD Biosciences). Both the primary and secondary antibodies were diluted in perm/wash buffer.
RNA and cDNA preparation
Total RNA was prepared from 2 to 5 × 106 cells using the RNeasy Mini kit (Qiagen) as per the kit instructions. RNA was quantified by absorbance at 260 nm. Total RNA was converted to cDNA using the SuperScript First-Strand Synthesis System (Invitrogen, Carlsbad, CA).
Polymerase chain reaction (PCR)
Primer sequences were as previously published for glyceraldehyde-3-phosphate dehydrogenase (GAPDH) [36], MAGE-A1 [2], MAGE-A2 [14], MAGE-A3 [11], MAGE-A4 [11], MAGE-A6 [11], MAGE-A10 [11], MAGE-A12 [11], NY-ESO-1 [55], TAG-1 [24], TAG-2a [24], TAG-2b [24], and TAG-2c [24]. Each of the primer sequences, annealing temperatures, and predicted product sizes has previously been summarized [4].
PCR was performed on 250 ng of cDNA using Platinum Taq polymerase (Invitrogen). The PCR mixes were heated to 94°C for 2 min, 30 and 40 cycles of amplification were performed (1 cycle = 30 s denaturation at 94°C, 30 s annealing at the temperature given in Ref. [4], 60 s extension at 72°C), and a final extension completed at 72°C for 5 min. Relative expression levels were determined by visualizing DNA bands on ethidium bromide-stained 1.5% agarose gels and are reported on a scale of 0–8. Level 0 represents the inability to visualize a band at 40 cycles of PCR. Levels 1–4 correspond to progressively brighter bands at 40 cycles of PCR analysis, with 1 being weakly visible and 4 being easily visible. Levels 5–8 correspond to progressively brighter bands at 30 cycles of PCR analysis, with 5 being weakly visible and 8 being easily visible.
ELISpot analysis
CD8+ T lymphocyte lines were generated from patients vaccinated with a mixture of peptides as described [6, 53, 60]. Those selected for use in this study were specific for: (a) the HLA-A2-restricted, MAGE-A10-derived peptide GLYDGMEHL (GLY), (b) the HLA-A3-restricted, MAGE-A1-derived peptide SLFRAVITK (SLF), and (c) the HLA-A3-restricted, NY-ESO-1-derived peptide ASGPGGGAPR (ASG). The peptide SLYNTVATL (GAG) derived from the gag protein of HIV-1 was used as an irrelevant peptide control [6].
Lymphocytes were stimulated and tested by ELISpot analyses as previously described [6]. Briefly, the lymphocytes were incubated with 40 μg/ml peptide for 2 h, washed, resuspended in RPMI-10HS containing 20 U/ml IL-2, and then incubated for 14 days. The cells were then plated in quadruplicate at 25,000 and 75,000 cells per well of interferon-γ coated MultiScreenHTS IP plates (Millipore, Billerica, MA). An equal number of peptide-pulsed antigen presenting cells were added to each well and the plates were incubated for 18–20 h. The plates were then washed and biotin-labeled interferon-γ added to each well. Spots were developed using streptavidin conjugated alkaline phosphatase (BD Biosciences, San Jose, CA) and 4-chloro-3-indolyl phosphatesubstrate-Toluidine salt (Pierce, Rockford, IL). Spots were counted using an automated plate reader (Bioreader 4000, Biosys, The Colony, TX).
Human subjects research approval
This research was approved by the University of Virginia Human Investigation Committee in accordance with an assurance filed with and approved by the Department of Health and Human Services.
Results
Effect of the treatment of ovarian cancer cell lines with 1 μM DAC for 3 days on CTA gene expression and class I MHC protein expression
Based on the fact that most studies designed to investigate the effect of DAC treatment on the expression of CTA genes and class I MHC proteins have treated cells for 2–4 days with 1–2 μM DAC [10, 12, 34, 56, 59], we treated 11 ovarian cancer cell lines with 1 μM DAC for 3 days. CTA gene expression was assessed by performing 30 and 40 cycles of PCR analysis. PCR with primers for the housekeeping gene GAPDH was first used to ensure that amplifiable cDNA had been obtained from each cell line. The results of this analysis demonstrated that all of the ovarian cancer cell lines had comparable levels of GAPDH gene expression (data not shown). PCR amplifications were then performed for 12 CTA genes (Table 1, Supplementary Fig. 1). DAC treatment resulted in higher levels of expression of at least 1 CTA gene in ten of the 11 cell lines. Most cell lines had increased levels of expression of 1–5 CTA genes, while one cell line had increased levels of expression of seven CTA genes. The expression levels of the MAGE-A1 and NY-ESO-1 genes were most frequently enhanced while the MAGE-A2, MAGE-A6, and MAGE-A12 genes were not enhanced in any of the cell lines tested. The expression of the remaining CTA genes was shown to increase in 1–3 cell lines each.
Table 1.
The effect of treating ovarian cancer cell lines with DAC on CTA gene expression
| DAC treatment | No. of cell lines with an increase/No. of cell lines where an increase could be measured | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Conditions | MAGE-A1 | MAGE-A2 | MAGE-A3 | MAGE-A4 | MAGE-A6 | MAGE-A10 | MAGE-A12 | NY-ESO-1 | TAG-1 | TAG-2a | TAG-2b | TAG-2C |
| 1 μM, 3 daya | 8/11 | 0/11 | 1/9 | 2/11 | 0/11 | 1/10 | 0/9 | 9/11 | 2/10 | 3/11 | 2/11 | 3/11 |
| 10 μM, 7 dayb | 9/9 | 0/11 | 9/9 | 11/11 | 7/11 | 9/9 | 8/9 | 10/10 | 8/9 | 5/9 | 8/11 | 4/11 |
| DAC treatment | No. of CTA with an increase/No. of CTA where an increase could be measured | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Conditions | CAOV-3 | CAOV-4 | COV413 | ES-2 | OV-90 | OVCAR-3 | SK-OV-3 | SW626 | TOV-21G | TOV-112D | TTB-6 | |
| 1 μM, 3 daya | 2/12 | 3/12 | 7/12 | 5/10 | 0/10 | 3/10 | 1/12 | 2/12 | 2/12 | 5/12 | 1/12 | |
| 10 μM, 7 dayb | 9/12 | 11/12 | 10/12 | 7/9 | 1/7 | 6/9 | 10/12 | 9/12 | 10/12 | 9/10 | 6/10 | |
Eleven ovarian cancer cell lines were incubated in the presence or absence of DAC under the indicated treatment conditions. The expression levels of the 12 indicated CTA genes were determined by PCR as indicated in the “Materials and methods”
aTwo independent experiments were performed for this treatment condition. An increase in expression following DAC treatment was defined as an increase of two or more units on the relative expression scale in each of the two experiments. When a particular CTA/cell line pair could not show an increase because the untreated cells in at least one experiment had a relative expression level of 7 or 8 on a scale of 8, these data points were excluded from the denominator
bThree independent experiments were performed for this treatment condition. An increase in expression following DAC treatment was defined as an increase in expression in each individual experiment and an increase of two or more units on the relative expression scale in at least two of the experiments. When a particular CTA/cell line pair could not show an increase because the untreated cells in at least one experiment had a relative expression level of 7 or 8 on a scale of 8, these data points were excluded from the denominator
Class I MHC protein expression in the untreated and DAC-treated cells was assessed by flow cytometry using mAb W6/32, which recognizes an epitope common to all class I MHC molecules, and mAbs to the individual class I MHC molecules HLA-A2 (mAb CR11-351), HLA-A3 (mAb GAP-A3), and HLA-B7 (mAb ME1-1.2). The treatment resulted in a reproducible, but small increase in HLA-A2 and total class I MHC expression in OV-90 (Supplementary Fig. 2). Conversely, the treatment of SK-OV-3 with DAC did not reproducibly increase HLA-A2, HLA-A3, or total class I MHC expression (Supplementary Fig. 2). The results obtained with OV-90 and SK-OV-3 were representative of those obtained with the remaining nine ovarian cancer cell lines (data not shown).
Effect of varying the length of treatment time of ovarian cancer cell lines with 1 μM DAC on CTA gene expression and class I MHC protein expression
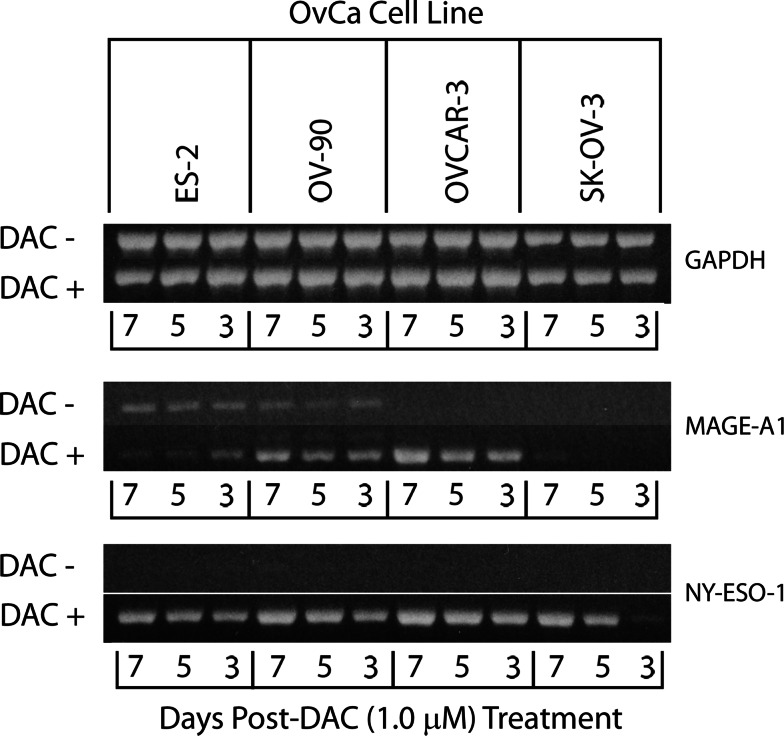
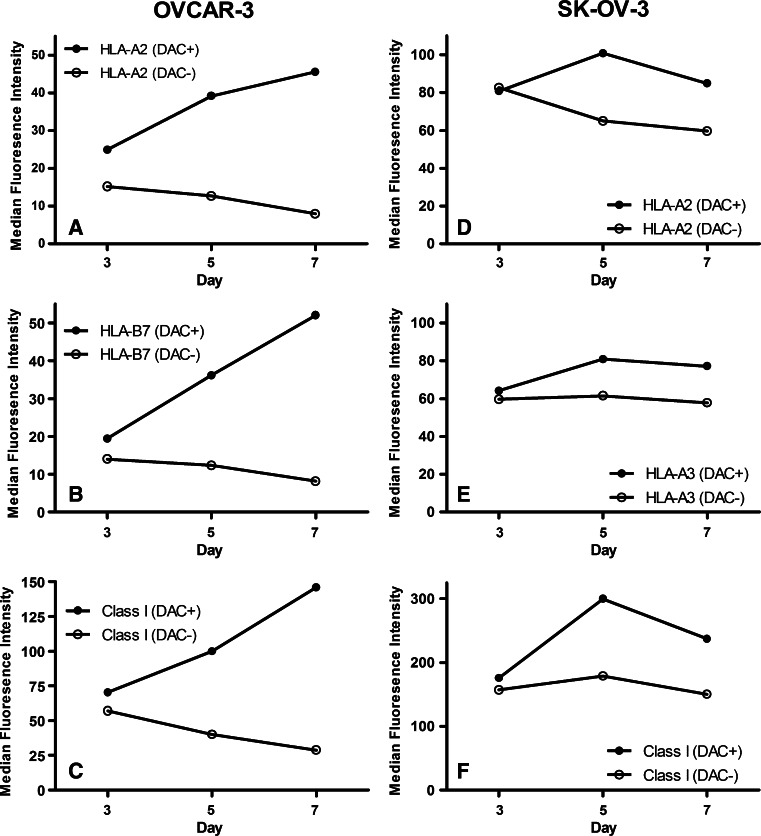
The effect of varying the length of treatment time with DAC was studied by comparing CTA gene and class I MHC protein expression levels in selected cell lines (ES-2, OV-90, OVCAR-3, and SK-OV-3) treated with 1 μM DAC for 3, 5, and 7 days. GAPDH gene expression in the four cell lines was not affected by any length of treatment (Fig. 1). MAGE-A1 gene expression was highest at day 7 for three of the four cell lines, and NY-ESO-1 gene expression was highest at day seven for all four cell lines. Similar results were observed when class I MHC molecule expression was measured by flow cytometry (Fig. 2 and data not shown). The highest levels of expression of HLA-A2, HLA-B7, and overall class I MHC molecule expression on OVCAR-3 were observed on day 7 following treatment. For SK-OV-3, expression of HLA-A2, HLA-A3, and overall class I MHC expression was increased to similar levels following 5 and 7 days of treatment and in all cases exceeded that observed following 3 days of treatment. Consequently, subsequent experiments were conducted with 7 days of DAC treatment.
Fig. 1.
Time course analysis of the effect of treating ovarian cancer cell lines with 1 μM DAC for 3, 5, or 7 days on CTA gene expression. The indicated ovarian cancer cell lines were incubated for 3, 5, or 7 days in the presence or absence of 1 μM DAC, harvested, and cDNA prepared. CTA gene expression was determined by PCR as indicated in the “Materials and methods”. Bands were visualized on ethidium bromide stained 1.5% agarose gels. The results from one of two to three independent experiments are shown
Fig. 2.
Time course analysis of the effect of treating ovarian cancer cell lines with 1 μM DAC for 3, 5, or 7 days on class I MHC protein expression. The indicated ovarian cancer cell lines were incubated for 3, 5, or 7 days in the presence or absence of 1 μM DAC and were then harvested. Class I MHC protein expression levels were determined by flow cytometry using mAb CR11-351 to detect HLA-A2 (a, d), mAb GAP-A3 to detect HLA-A3 (e), mAb ME1-1.2 to detect HLA-B7 (b), and mAb W6/32 (c, f) to detect all class I MHC molecules. The results from one of three independent experiments are shown
Effect of treating ovarian cancer cell lines with varying concentrations of DAC on CTA gene expression and class I MHC protein expression
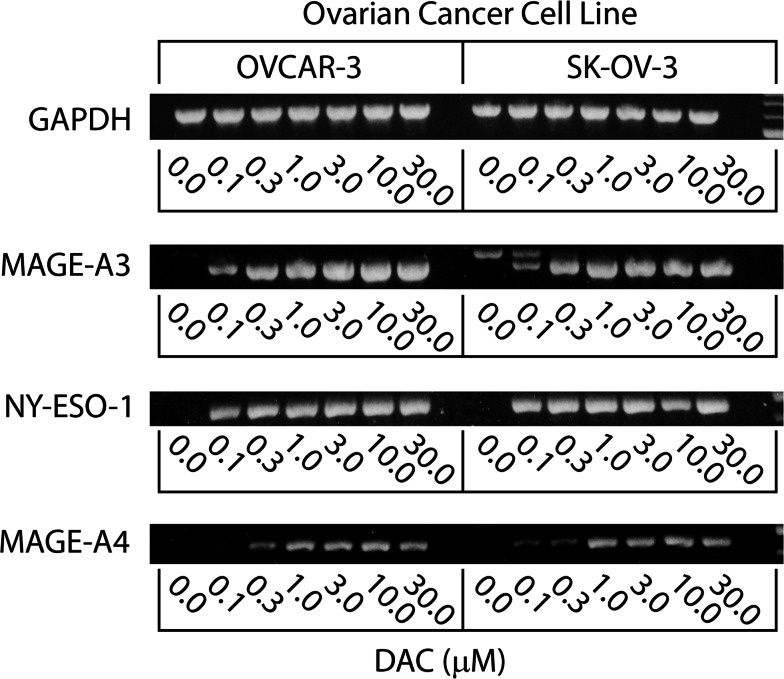
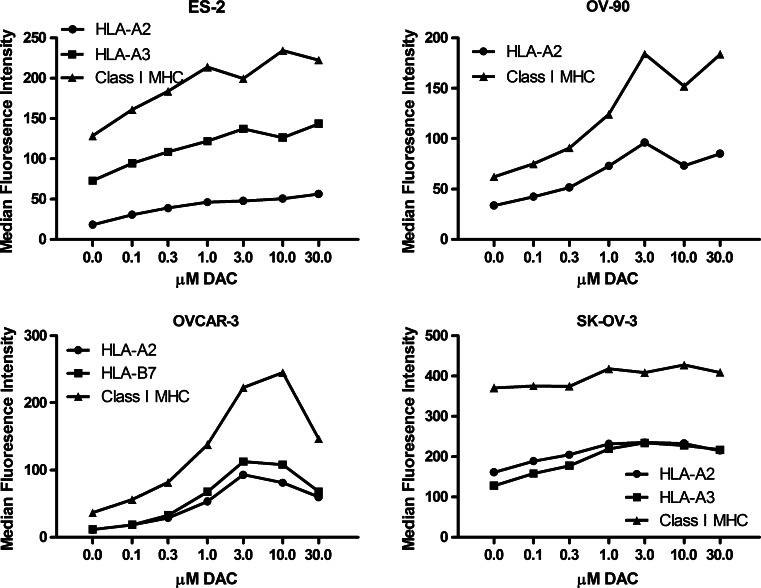
The optimal concentration of DAC needed to enhance the expression of CTA genes and class I MHC proteins was next determined. Ovarian cancer cell lines were untreated, or treated with varying concentrations of DAC (0.1–30 μM) for 7 days. PCR analysis of OVCAR-3 and SK-OV-3 showed that the treatment did not affect GAPDH gene expression (Fig. 3). Conversely, the expression of CTA genes was enhanced by increasing concentrations of DAC, with maximal expression requiring a minimal concentration of 1–3 μM. Maximal expression of class I MHC molecules required a higher concentration of DAC, with 10 μM appearing to be optimal (Fig. 4). Based on the results of dose response and time course experiments, 7 days of incubation with 10 μM DAC was adopted as the standard treatment to enhance CTA gene expression and class I MHC protein expression.
Fig. 3.
Dose response analysis of the effect of treating ovarian cancer cell lines with 0.1–30 μM DAC for 7 days on CTA gene expression. The indicated ovarian cancer cell lines were incubated for 7 days in the presence or absence of 0.1–30 μM DAC, harvested, and cDNA prepared. CTA gene expression was determined by PCR as indicated in the “Materials and methods”. Bands were visualized on ethidium bromide stained 1.5% agarose gels. The results from one of two independent experiments are shown
Fig. 4.
Dose response analysis of the effect of treating ovarian cancer cell lines with 0.1–30 μM DAC for 7 days on class I MHC protein expression. The indicated ovarian cancer cell lines were incubated for 7 days in the presence or absence of 0.1–30 μM DAC and were then harvested. Class I MHC protein expression levels were determined by flow cytometry using mAb CR11-351 to detect HLA-A2, mAb GAP-A3 to detect HLA-A3, mAb ME1-1.2 to detect HLA-B7, and mAb W6/32 to detect all class I MHC molecules. The results from one of two independent experiments are shown
Effect of treating ovarian cancer cell lines with 10 μM DAC for 7 days on CTA gene expression, MAGE-A1 protein expression, and class I MHC protein expression
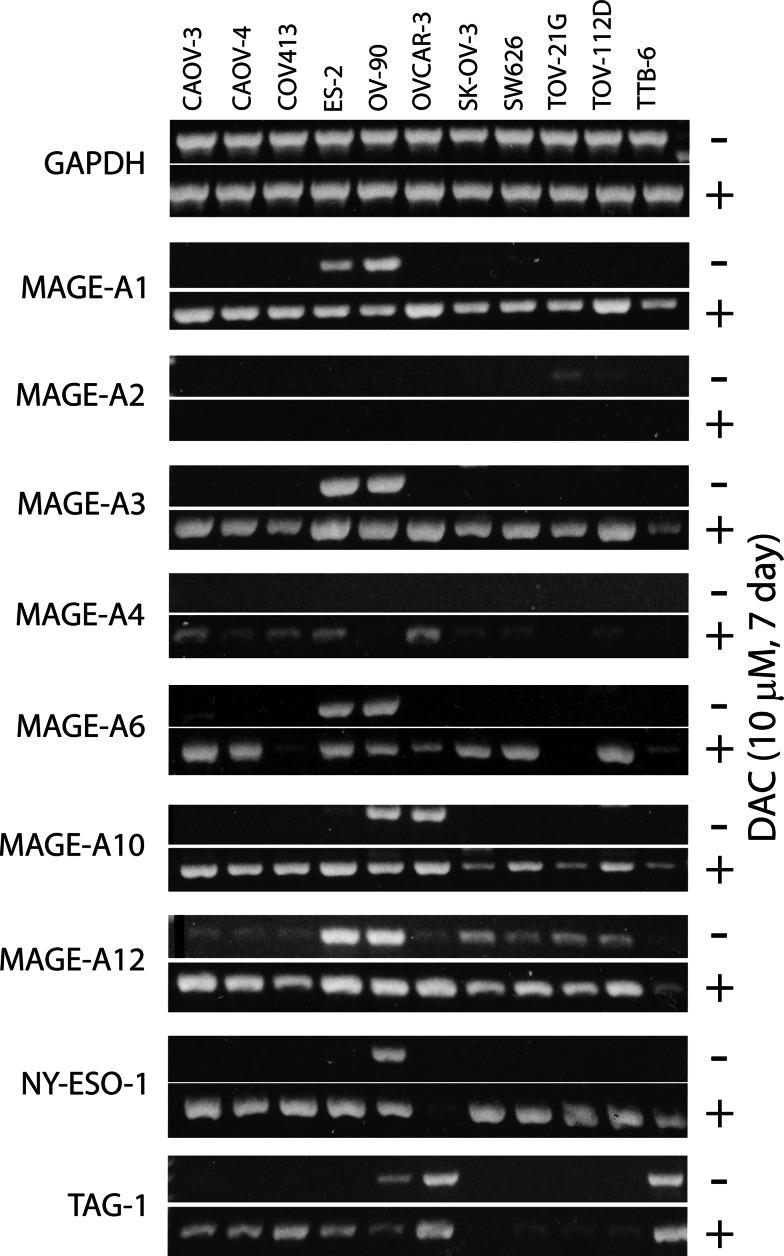
Having determined the optimal concentration and incubation period for DAC treatment, we next treated 11 ovarian cancer cell lines with 10 μM DAC for 7 days. CTA gene expression was determined with both 30 and 40 cycles of PCR analysis. Results of a single experiment analyzed with 30 cycles of PCR demonstrate several points (Fig. 5). First, CTA genes are not readily detectable in most untreated cell lines when tested with 30 cycles of PCR amplification. ES-2 and OV-90 are exceptions in that many of the CTA genes can be detected in these cell lines without prior exposure to DAC. Second, treatment with DAC can be very effective in upregulating the expression of some CTA genes (MAGE-A1, MAGE-A3, MAGE-A10, NY-ESO-1) while it has little (MAGE-A4) to no (MAGE-A2) effect on the expression of other CTA genes when measured at 30 cycles of PCR. Third, the increased expression of a particular CTA gene in one cell line does not predict that it will be increased in all cell lines (MAGE-A6, NY-ESO-1). Fourth, the increased expression of a particular CTA gene in one cell line is not predictive of other CTA genes being increased in the same cell line.
Fig. 5.
The effect of treating ovarian cancer cell lines with 10 μM DAC for 7 days on CTA gene expression. Eleven ovarian cancer cell lines were incubated for 7 days in the presence (+) or absence (−) of 10 μM DAC, harvested, and cDNA prepared. CTA-specific PCR was then performed and the amplification products visualized on ethidium bromide stained 1.5% agarose gels. The 30-cycle results from one of three independent experiments are shown
The sensitivity of this analysis was increased by also conducting the CTA gene expression measurements with 40 cycles of PCR amplification. The combined results of 30 and 40 cycles of PCR analysis are presented in Table 1 and Supplementary Fig. 3. The number of CTA genes with enhanced expression ranged from 1 to 11 per cell line, with most cell lines having enhanced expression of 6 to 11 CTA genes. Correspondingly, the expression of nine of the 12 CTA genes was enhanced in at least seven different ovarian cell lines. Expression of MAGE-A1, MAGE-A3, MAGE-A4, MAGE-A10, and NY-ESO-1 was increased in every evaluable cell line. Only MAGE-A2 did not increase in any cell line, and in fact, tended to decrease from low to no expression following treatment (Supplementary Fig. 3).
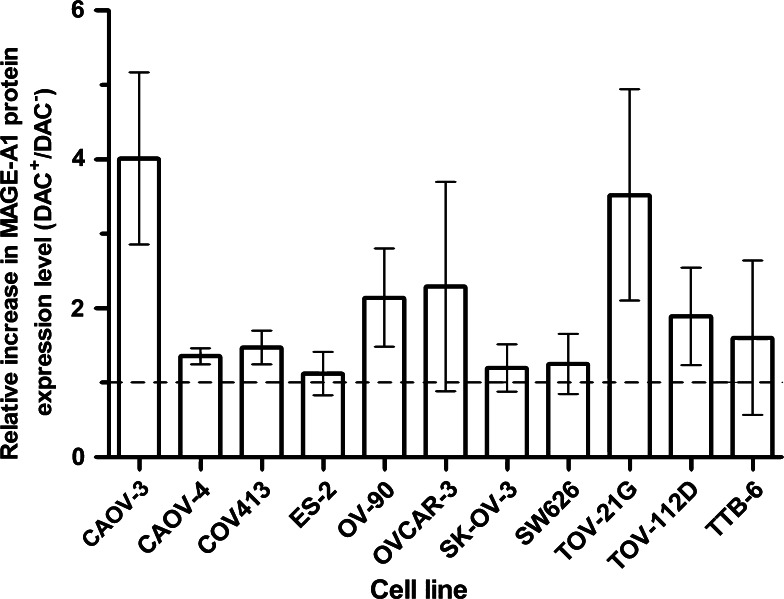
The expression of the MAGE-A1 protein was measured to determine if increased CTA gene expression levels also lead to an increase in the expression of the corresponding protein. Although many lines showed a large increase in the expression of the MAGE-A1 gene when treated with 10 μM DAC for 7 days (Table 1, Supplementary Fig. 3), the cell lines did not show a correspondingly large increase in protein expression (Fig. 6). MAGE-A1 protein expression increased greater than threefold in CAOV-3 and TOV-21G, and greater than 1.9-fold in OV-90, OVCAR-3, and TOV-112D.
Fig. 6.
The effect of treating ovarian cancer cell lines with 10 μM DAC for 7 days on MAGE-A1 protein expression. Eleven ovarian cancer cell lines were incubated for 7 days in the presence or absence of 10 μM DAC and were then harvested. The cells were fixed and permeabilized and then MAGE-A1 expression levels determined by flow cytometry using mAb 3F257. The data are presented as mean ± SEM of two independent experiments and are the ratio of the fluorescence intensity of DAC-treated cells to the fluorescence intensity of the untreated cells. The dashed line represents a 1:1 ratio of the fluorescence intensities
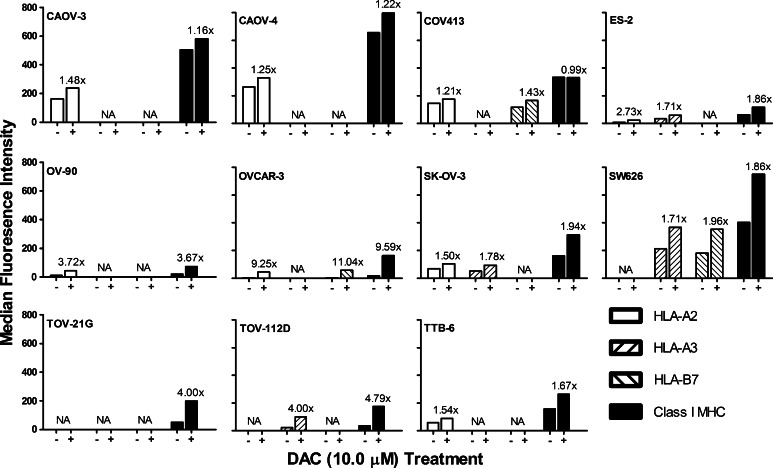
Class I MHC protein expression was also assessed following treatment of the 11 ovarian cancer cell lines with 10 μM DAC for 7 days (Fig. 7). Overall, two patterns of expression emerged. When class I MHC molecules are already expressed at high levels (ex. CAOV-3 and CAOV-4) there was no to only a modest enhancement of expression. When protein expression was low, increases in expression levels ranged from about two- to tenfold; however, despite the high relative increase in expression, the overall protein expression levels remained below that found in a cell lines naturally expressing high levels of class I MHC molecules.
Fig. 7.
The effect of treating ovarian cancer cell lines with 10 μM DAC for 7 days on class I MHC protein expression. Eleven ovarian cancer cell lines were incubated for 7 days in the presence (+) or absence (−) of 10 μM DAC and were then harvested. Class I MHC protein expression levels were determined by flow cytometry using mAb CR11-351 to detect HLA-A2, mAb GAP-A3 to detect HLA-A3, mAb ME1-1.2 to detect HLA-B7, and mAb W6/32 to detect all class I MHC molecules. The data represent one of two independent experiments
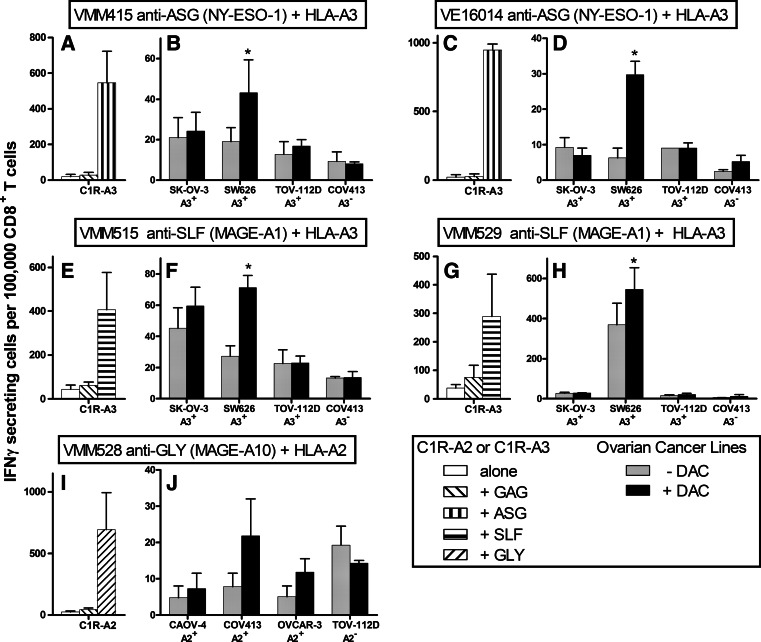
Recognition of DAC-treated (10 μM for 7 days) ovarian cancer cells by antigen-reactive CD8+ T lymphocytes
To determine if enhanced expression of CTA and class I MHC molecules leads to increased recognition of the treated cells by T lymphocytes, antigen-reactive CD8+ T lymphocytes that recognize peptide antigens derived from the MAGE-A1, MAGE-A10, and NY-ESO-1 proteins were tested in an interferon-γ ELISpot assay for their ability to recognize DAC-treated and untreated cells. CD8+ T lymphocytes that recognize the NY-ESO-1-derived peptide ASG in association with HLA-A3 (Fig. 8a, c) were tested against SK-OV-3, SW626, and TOV-112D (all NY-ESO-1+, HLA-A3+) as potential positive stimulator cells and COV413 (NY-ESO-1+, HLA-A3-) as a control cell line (Fig. 8b, d). DAC treatment of SW626, but not the remaining cell lines, led to an increase in T cell responses to those tumor cells. CD8+ T lymphocytes that recognize the MAGE-A1-derived peptide SLF in association with HLA-A3 (Fig. 8e, g) were tested against the same cell lines, all of which also express MAGE-A1+ (Fig. 8f, h). Treatment of SW626 with DAC was again shown to lead to an increase in the ability of the cell line to trigger interferon-γ release by the antigen-specific CD8+ T lymphocytes. Finally, CD8+ T lymphocytes that recognize the MAGE-A10-derived peptide GLY in association with HLA-A2 (Fig. 8i) were tested for interferon-γ release against CAOV-4, COV413, and OVCAR-3 (all MAGE-A10+, HLA-A2+) as potential stimulators and TOV-112D (MAGE-A10+, HLA-A2−) as a control cell line (Fig. 8j). Treatment of these cell lines with DAC did not significantly change their ability to stimulate antigen-specific CD8+ T lymphocytes.
Fig. 8.
The effect of treating ovarian cancer cell lines with 10 μM DAC for 7 days on recognition by antigen-specific CD8+ T lymphocytes. The indicated ovarian cancer cell lines were incubated for 7 days in the presence or absence of 10 μM DAC and were then harvested. The cells were then used as stimulator cells in an interferon-γ ELISpot assay. CD8+ T lymphocytes reactive to the NY-ESO-1-dervied peptide ASG presented in association with HLA-A3 (a–d), for the MAGE-A1-derived peptide SLF presented in association with HLA-A3 (e–h), and the MAGE-A10-derived peptide GLY presented in association with HLA-A2 (i, j) were used. C1R-A2 and C1R-A3 cells pulsed with the relevant peptide served as a positive control, while C1R-A2 and C1R-A3, either unpulsed or pulsed with a control peptide (GAG), served as negative controls. Each panel of ovarian cancer cells used as stimulators included three lines that were HLA-matched for the relevant class I MHC molecule and one line that was HLA-unmatched for the relevant class I MHC molecule. The data are presented as the mean ± SEM for two independent experiments
Discussion
The results presented here clearly indicate that the treatment of ovarian cancer cells with DAC can lead to an enhancement in expression of CTA genes (Table 1, Supplementary Fig. 3). In comparison to other studies in which 3–4 days of treatment with 1–2 μM DAC was routinely used to upregulate CTA gene expression [10, 12, 34, 56, 59], ovarian cancer cell lines required 7 days of treatment with DAC at 1–3 μM to achieve the highest levels of CTA gene expression (Figs. 1, 3). The need for a lengthier treatment time may reflect the longer doubling times that ovarian cell lines have in comparison to melanoma cell lines (unpublished observations). Because DAC acts by inhibiting methylation of a newly synthesized DNA strand, it would be expected that the rate at which demethylation will occur would be a function of the replicative rate of the cells.
It is important to note, however, that DAC treatment is not an universal panacea for upregulating CTA gene expression. MAGE-A2 gene expression, for example, does not increase, but rather decreases in expression following DAC treatment (Supplementary Fig. 3). This result stands in contrast to the results obtained with CTA genes such as MAGE-A1, MAGE-A3, MAGE-A10, and NY-ESO-1, which when ordinarily expressed at low levels undergo a dramatic upregulation following DAC treatment. It is also evident that there is heterogeneity in the response to DAC treatment, both with respect to how frequently the expression of a particular CTA gene is upregulated in different cell lines, and with respect to how many CTA genes are upregulated in a particular cell line. Such heterogeneous response patterns of CTA gene expression are not unique to ovarian cancer cell lines, but have been demonstrated in other cancers [8, 12, 51, 52]. These findings likely reflect the multiple control mechanisms that can affect the expression of any particular gene, as well as the fact that promoter demethylation will have little functional consequence if the gene in question is no longer functional due to a genetic defect.
It is not increased CTA gene expression per se that leads to increased recognition of tumor cells by CTA-specific T cells, but rather it is increased peptide production which can occur through a combination of increased protein production and/or increased protein degradation. There are few CTA-specific antibodies available to measure protein expression levels, although a number of antibodies have been developed which recognize MAGE-A proteins. Although most of the antibodies with specificity for MAGE-A proteins recognize multiple members of the MAGE-A family, the mAb 3F257 is MAGE-A1 specific (USB technical data sheet). mAb 3F257 was used to ask if MAGE-A1 protein expression increased in parallel with CTA gene expression following DAC treatment. Although MAGE-A1 protein expression increased by 1.9- to 4.0-fold in five of the 11 cell lines tested (Fig. 6), the magnitude of the increase is relatively small in comparison to the corresponding increase in gene expression (Table 1, Supplementary Fig. 3). This would suggest that other factors limit the translation of MAGE-A1 mRNA, such that there is no linear relationship between mRNA and protein expression. Alternatively, increased levels of the protein may be associated with an increased rate of degradation of the protein.
The treatment of ovarian cancer cell lines with DAC leads to an increase in the levels of class I MHC protein expression (Fig. 7), similar in magnitude to that observed when melanoma cell lines are treated with DAC [10, 19, 49]. When cell lines already express class I MHC molecules at intermediate to high levels, the amount of increase if any, is less than twofold (e.g., CAOV3, CAOV4, and SW626). This is in accord with the fact that when gene expression levels are already high it is likely that the promoter region of the gene already exists in an unmethylated state and treatment with DAC would not be expected to have a large effect on expression. When cell lines naturally express low levels of class I MHC molecules, treatment with DAC can increase expression by four- to tenfold (see OVCAR-3, TOV-21G, and TOV-112D). In some poorly expressing cell lines such as ES-2 and SK-OV-3, however, class I MHC protein expression is increased by less than twofold. That the expression levels in these cell lines are not increased to the high levels naturally expressed in other cell lines likely reflects the fact that the low expression levels are only partially regulated, or are not regulated at all, by a methylation dependent mechanism. One or more genetic defects, either in the class I MHC genes themselves or in one of the ancillary genes (β2-microglobulin, proteasomes, TAP, etc.) required for class I MHC protein expression could also account for the low expression levels of the class I MHC proteins [48] .
From an immunological perspective the importance of CTAs is that they are recognized as antigens by both antibodies and T cells, with the latter requiring that the CTA first be processed into peptides, which then bind to class I MHC molecules for presentation to CD8+ T cells. From this perspective, a change in CTA and/or class I MHC molecule expression in response to DAC treatment is functionally significant if it alters the ability of the treated cell line to be recognized by antigen-specific CD8+ T cells. As measured by interferon-γ release in ELISpot assays, treatment of SW626 with DAC resulted in increased recognition by NY-ESO-1/HLA-A3-reactive and MAGE-A1/HLA-A3-reactive T cells. A similar increase was not observed with cell lines SK-OV-3 and TOV-112D. These lines are similar in that all of them express intermediate basal levels of the MAGE-A1 gene and high, DAC-stimulated levels of the gene. Conversely, the basal level of expression of HLA-A3 by SW626 cells exceeds the DAC-stimulated levels of HLA-A3 expressed by the cell lines SK-OV-3 and TOV-112D. Thus, the increase in CTA expression may not manifest itself in increased recognition by CD8+ T cells unless there is a certain minimum number of class I MHC molecules available to present the antigen. Similar experiments with CD8+ T cells specific for a MAGE-A10-derived peptide seen in association with HLA-A2 did not result in a statistically significant increase in recognition, although the cell line COV413 had a trend toward increased recognition. It is difficult to compare these results to published results as different methods have been used to measure the T cell response including total interferon-γ release [49, 57], cytotoxicity measured by 51Cr-release [8, 19, 56], and interferon-γ ELISpot [19]. The most comparable study using interferon-γ ELISpot [19] showed a doubling in the number of spots obtained, a result that is similar in magnitude to that observed here.
There are two potential limitations to the clinical use of DNA methylation inhibitors for the purposes of increasing CTA and class I MHC molecule expression. First, promoter methylation is one of many mechanisms that regulates the gene expression and methylation status does not always correlate with gene expression. For example, there is no correlation between methylation status and HLA-G expression in ovarian cancer tumors and normal ovarian epithelial cells [38], and the degree of methylation status of the MAGE-A1 gene does not show a strong correlation with gene expression [54]. Likewise, demethylation of the promoter region of class I MHC genes will not overcome the lack of expression due to defects in the genes coding for class I MHC molecules, β2-microglobulin, proteasomes, and TAP [48]. Thus, this approach can be useful in driving a high expression level of CTA and class I MHC molecules on some, but not all tumor cells. Second, DNA methylation inhibitors are not specific for tumor cells but have the potential to act on all cycling cells in the body. Consistent with this, in vitro DAC treatment has been shown to upregulate CTA expression of normal cells in some [12, 35], but not all [56] studies. The de novo expression of CTA by normal tissues raises the risk that CTA-specific T cells will interact with normal cells as well as tumors. Accordingly, pre-clinical studies will need to assess the degree to which epigenetic therapies increase CTA expression by normal cells, whether different dosing strategies can be used to p`entially enhance CTA expression by tumors and not normal tissue, and to determine if induced CTA expression levels on normal tissue are functionally significant from the perspective of recognition by T lymphocytes.
The results presented here demonstrate that the treatment of ovarian cancer cells with DAC can lead to a large increase in the expression of multiple CTA genes, a modest increase in the expression of class I MHC proteins, and enhanced recognition of the treated cells by antigen-specific CD8+ T cells. Thus, there is a rational basis for treating ovarian cancer with combined DAC therapy and vaccine therapy designed to stimulate a T cell-mediated immune response.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
This work was supported by grant W81XWH-05-1-0012 from the United States Department of Defense to K. T. Hogan. The authors wish to thank Dr. Craig L. Slingluff Jr. for reviewing the manuscript and the members of the UVA Human Immune Therapy Center core laboratory for performing the ELISpot analyses.
Abbreviations
- CTA
Cancer-testis antigen
- CTL
Cytotoxic T lymphocyte
- DAC
5-Aza-2′-deoxycytidine
- DNMT
DNA methyltransferase
- GAPDH
Glyceraldehyde-3-phosphate dehydrogenase
- mAb
Monoclonal antibody
- MHC
Major histocompatibility complex
- PCR
Polymerase chain reaction
References
- 1.Berger AE, Davis JE, Cresswell P. Monoclonal antibody to HLA-A3. Hybridoma. 1982;1:87–90. doi: 10.1089/hyb.1.1982.1.87. [DOI] [PubMed] [Google Scholar]
- 2.Brasseur F, Marchand M, Vanwijck R, Herin M, Lethe B, Chomez P, Boon T. Human gene MAGE-1, which codes for a tumor-rejection antigen, is expressed by some breast tumors. Int J Cancer. 1992;52:839–841. doi: 10.1002/ijc.2910520528. [DOI] [PubMed] [Google Scholar]
- 3.Buick RN, Pullano R, Trent JM. Comparative properties of five human ovarian adenocarcinoma cell lines. Cancer Res. 1985;45:3668–3676. [PubMed] [Google Scholar]
- 4.Carr TM, Adair SJ, Fink MJ, Hogan KT. Immunological profiling of a panel of ovarian cancer cell lines. Cancer Immunol Immunother. 2008;57:31–42. doi: 10.1007/s00262-007-0347-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen YT, Old LJ. Cancer-testis antigens: targets for cancer immunotherapy. Cancer J Sci Am. 1999;5:16–17. doi: 10.1111/j.1349-7006.1999.tb00660.x. [DOI] [PubMed] [Google Scholar]
- 6.Chianese-Bullock KA, Pressley J, Garbee C, Hibbitts S, Murphy C, Yamshchikov G, Petroni GR, Bissonette EA, Neese PY, Grosh WW, Merrill P, Fink R, Woodson EM, Wiernasz CJ, Patterson JW, Slingluff CL., Jr MAGE-A1-, MAGE-A10-, and gp100-derived peptides are immunogenic when combined with granulocyte-macrophage colony-stimulating factor and Montanide ISA-51 adjuvant and administered as part of a multipeptide vaccine for melanoma. J Immunol. 2005;174:3080–3086. doi: 10.4049/jimmunol.174.5.3080. [DOI] [PubMed] [Google Scholar]
- 7.Cho B, Lee H, Jeong S, Bang YJ, Lee HJ, Hwang KS, Kim HY, Lee YS, Kang GH, Jeoung DI. Promoter hypomethylation of a novel cancer/testis antigen gene cage is correlated with its aberrant expression and is seen in premalignant stage of gastric carcinoma. Biochem Biophys Res Commun. 2003;307:52–63. doi: 10.1016/S0006-291X(03)01121-5. [DOI] [PubMed] [Google Scholar]
- 8.Coral S, Sigalotti L, Altomonte M, Engelsberg A, Colizzi F, Cattarossi I, Maraskovsky E, Jager E, Seliger B, Maio M. 5-aza-2′-deoxycytidine-induced expression of functional cancer testis antigens in human renal cell carcinoma: Immunotherapeutic implications. Clin Cancer Res. 2002;8:2690–2695. [PubMed] [Google Scholar]
- 9.Coral S, Sigalotti L, Colizzi F, Spessotto A, Nardi G, Cortini E, Pezzani L, Fratta E, Fonsatti E, Di Giacomo AM, Nicotra MR, Natali PG, Altomonte M, Maio M. Phenotypic and functional changes of human melanoma xenografts induced by DNA hypomethylation: Immunotherapeutic implications. J Cell Physiol. 2006;207:58–66. doi: 10.1002/jcp.20540. [DOI] [PubMed] [Google Scholar]
- 10.Coral S, Sigalotti L, Gasparollo A, Cattarossi I, Visintin A, Cattelan A, Altomonte M, Maio M. Prolonged upregulation of the expression of HLA class I antigens and costimulatory molecules on melanoma cells treated with 5-aza-2′-deoxycytidine (5-aza-cdr) J Immunother. 1999;22:16–24. doi: 10.1097/00002371-199901000-00003. [DOI] [PubMed] [Google Scholar]
- 11.De Plaen E, Arden K, Traversari C, Gaforio JJ, Szikora JP, De Smet C, Brasseur F, van der Bruggen P, Lethe B, Lurquin C, Chomez P, De Backer O, Boon T, Arden K, Cavenee W, Brasseur R. Structure, chromosomal localization, and expression of 12 genes of the MAGE family. Immunogenetics. 1994;40:360–369. doi: 10.1007/BF01246677. [DOI] [PubMed] [Google Scholar]
- 12.De Smet C, De Backer O, Faraoni I, Lurquin C, Brasseur F, Boon T. The activation of human gene MAGE-1 in tumor cells is correlated with genome-wide demethylation. Proc Natl Acad Sci U S A. 1996;93:7149–7153. doi: 10.1073/pnas.93.14.7149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.De Smet C, Lurquin C, Lethe B, Martelange V, Boon T. DNA methylation is the primary silencing mechanism for a set of germ line- and tumor-specific genes with a CpG-rich promoter. Mol Cell Biol. 1999;19:7327–7335. doi: 10.1128/mcb.19.11.7327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.De Smet C, Lurquin C, Van der Bruggen P, De Plaen E, Brasseur F, Boon T. Sequence and expression pattern of the human MAGE2 gene. Immunogenetics. 1994;39:121–129. doi: 10.1007/BF00188615. [DOI] [PubMed] [Google Scholar]
- 15.dos Santos NR, Torensma R, De Vries TJ, Schreurs MWJ, de Bruijn DRH, Kater-Baats E, Ruiter DJ, Adema GJ, van Muijen GNP, van Kessel AG. Heterogeneous expression of the SSX cancer/testis antigens in human melanoma lesions and cell lines. Cancer Res. 2000;60:1654–1662. [PubMed] [Google Scholar]
- 16.Ellis SA, Taylor C, McMichael A. Recognition of HLA-B27 and related antigen by a monoclonal antibody. Hum Immunol. 1982;5:49–59. doi: 10.1016/0198-8859(82)90030-1. [DOI] [PubMed] [Google Scholar]
- 17.Fogh J, Tremple G. New human tumor cell lines. In: Fogh J, editor. Human tumor cell lines in vitro. New York: Plenum Press; 1975. pp. 115–141. [Google Scholar]
- 18.Fogh J, Wright WC, Loveless JD. Absence of HeLa cell contamination in 169 cell lines derived from human tumors. J Natl Cancer Inst. 1977;58:209–214. doi: 10.1093/jnci/58.2.209. [DOI] [PubMed] [Google Scholar]
- 19.Fonsatti E, Nicolay HJM, Sigalotti L, Calabro L, Pezzani L, Colizzi F, Altomonte M, Guidoboni M, Marincola FM, Maio M. Functional up-regulation of human leukocyte antigen class I antigens expression by 5-aza-2′-deoxycytidine in cutaneous melanoma: Immunotherapeutic implications. Clin Cancer Res. 2007;13:3333–3338. doi: 10.1158/1078-0432.CCR-06-3091. [DOI] [PubMed] [Google Scholar]
- 20.Fonsatti E, Sigalotti L, Coral S, Colizzi F, Altomonte M, Maio M. Methylation-regulated expression of HLA class I antigens in melanoma. Int J Cancer. 2003;105:430–431. doi: 10.1002/ijc.11077. [DOI] [PubMed] [Google Scholar]
- 21.Fujie T, Mori M, Ueo H, Sugimachi K, Akiyoshi T. Expression of MAGE and BAGE genes in Japanese breast cancers. Ann Oncol. 1997;8:369–372. doi: 10.1023/A:1008255630202. [DOI] [PubMed] [Google Scholar]
- 22.Guo ZS, Hong JA, Irvine KR, Chen GA, Spiess PJ, Liu Y, Zeng G, Wunderlich JR, Nguyen DM, Restifo NP, Schrump DS. De novo induction of a cancer/testis antigen by 5-aza-2′-deoxycytidine augments adoptive immunotherapy in a murine tumor model. Cancer Res. 2006;66:1105–1113. doi: 10.1158/0008-5472.CAN-05-3020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hamilton TC, Young RC, McKoy WM, Grotzinger KR, Green JA, Chu EW, Whang-Peng J, Rogan AM, Green WR, Ozols RF. Characterization of a human ovarian carcinoma cell line (NIH:OVCAR-3) with androgen and estrogen receptors. Cancer Res. 1983;43:5379–5389. [PubMed] [Google Scholar]
- 24.Hogan KT, Coppola MA, Gatlin CL, Thompson LW, Shabanowitz J, Hunt DF, Engelhard VH, Ross MM, Slingluff CL. Identification of novel and widely expressed cancer/testis gene isoforms that elicit spontaneous cytotoxic T lymphocyte reactivity to melanoma. Cancer Res. 2004;64:1157–1163. doi: 10.1158/0008-5472.CAN-03-2209. [DOI] [PubMed] [Google Scholar]
- 25.Jager D, Jager E, Knuth A. Immune responses to tumour antigens: Implications for antigen specific immunotherapy of cancer. J Clin Pathol. 2001;54:669–674. doi: 10.1136/jcp.54.9.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jungbluth AA, Chen YT, Stockert E, Busam KJ, Kolb D, Iversen K, Coplan K, Williamson B, Altorki N, Old LJ. Immunohistochemical analysis of NY-ESO-1 antigen expression in normal and malignant human tissues.[erratum appears in Int. J Cancer 2002 February. 2001;20(6):878–60. doi: 10.1002/ijc.1282. [DOI] [PubMed] [Google Scholar]
- 27.Jungbluth AA, Stockert E, Chen YT, Kolb D, Iversen K, Coplan K, Williamson B, Altorki N, Busam KJ, Old LJ. Monoclonal antibody MA454 reveals a heterogeneous expression pattern of MAGE-1 antigen in formalin-fixed paraffin embedded lung tumours. Br J Cancer. 2000;83:493–497. doi: 10.1054/bjoc.2000.1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Juretic A, Spagnoli GC, Schultz-Thater E, Sarcevic B. Cancer/testis tumour-associated antigens: immunohistochemical detection with monoclonal antibodies. Lancet Oncol. 2003;4:104–109. doi: 10.1016/S1470-2045(03)00982-3. [DOI] [PubMed] [Google Scholar]
- 29.Karlan BY, Amin W, Band V, Zurawski VR, Littlefield BA. Plasminogen activator secretion by established lines of human ovarian carcinoma cells in vitro. Gynecol Oncol. 1988;31:103–112. doi: 10.1016/0090-8258(88)90276-4. [DOI] [PubMed] [Google Scholar]
- 30.Karpf AR, Lasek AW, Ririe TO, Hanks AN, Grossman D, Jones DA. Limited gene activation in tumor and normal epithelial cells treated with the DNA methyltransferase inhibitor 5-aza-2′-deoxycytidine. Mol Pharmacol. 2004;65:18–27. doi: 10.1124/mol.65.1.18. [DOI] [PubMed] [Google Scholar]
- 31.Kirkin AF, Dzhandzhugazyan KN, Zeuthen J. Cancer/testis antigens: structural and immunobiological properties. Cancer Invest. 2002;20:222–236. doi: 10.1081/CNV-120001150. [DOI] [PubMed] [Google Scholar]
- 32.Kuppen PJK, Schuitemaker H, van’t Veer LJ, de Bruijn EA, van Oosterom AT, Schrier PI. Cis-diamminedichloroplatinum(II)-resistant sublines derived from two human ovarian tumor cell lines. Cancer Res. 1988;48:3355–3359. [PubMed] [Google Scholar]
- 33.Lau DHM, Lewis AD, Ehsan MN, Sikic BI. Multifactorial mechanisms associated with broad cross-resistance of ovarian carcinoma cells selected by cyanomorpholino doxorubicin. Cancer Res. 1991;51:5181–5187. [PubMed] [Google Scholar]
- 34.Li J, Yang Y, Fujie F, Baba K, Ueo H, Mori M, Akiyoshi T. Expression of BAGE, GAGE, and MAGE genes in human gastric carcinoma. Clin Cancer Res. 1996;2:1619–1625. [PubMed] [Google Scholar]
- 35.Lurquin C, De Smet C, Brasseur F, Muscatelli F, Martelange V, De Plaen E, Brasseur R, Monaco AP, Boon T. Two members of the human mageb gene family located in xp21.3 are expressed in tumors of various histological origins. Genomics. 1997;46:397–408. doi: 10.1006/geno.1997.5052. [DOI] [PubMed] [Google Scholar]
- 36.Maier JA, Voulalas P, Roeder D, Maciag T. Extension of the life-span of human endothelial cells by an interleukin-1 alpha antisense oligomer. Science. 1990;249:1570–1574. doi: 10.1126/science.2218499. [DOI] [PubMed] [Google Scholar]
- 37.Menendez L, Walker D, Matyunina LV, Dickerson EB, Bowen NJ, Polavarapu N, Benigno BB, McDonald JF. Identification of candidate methylation-responsive genes in ovarian cancer. Mol Cancer. 2007;6:10. doi: 10.1186/1476-4598-6-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Menendez L, Walker LD, Matyunina LV, Totten KA, Benigno BB, McDonald JF. Epigenetic changes within the promoter region of the HLA-G gene in ovarian tumors. Mol Cancer. 2008;7:43. doi: 10.1186/1476-4598-7-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nie Y, Yang G, Song Y, Zhao X, So C, Liao J, Wang LD, Yang CS. DNA hypermethylation is a mechanism for loss of expression of the HLA class I genes in human esophageal squamous cell carcinomas. Carcinogenesis. 2001;22:1615–1623. doi: 10.1093/carcin/22.10.1615. [DOI] [PubMed] [Google Scholar]
- 40.Parham P, Barnstable CJ, Bodmer WF. Use of a monoclonal antibody (W6/32) in structural studies of HLA-A, b, c antigens. J Immunol. 1979;123:342–349. [PubMed] [Google Scholar]
- 41.Parmiani G, Castelli C, Dalerba P, Mortarini R, Rivoltini L, Marincola FM, Anichini A. Cancer immunotherapy with peptide-based vaccines: what have we achieved? Where are we going? J Natl Cancer Inst. 2002;94:805–818. doi: 10.1093/jnci/94.11.805. [DOI] [PubMed] [Google Scholar]
- 42.Platsoucas CD, Fincke JE, Pappas J, Jung WJ, Heckel M, Schwarting R, Magira E, Monos D, Freedman RS. Immune responses to human tumors: development of tumor vaccines. Anticancer Res. 2003;23:1969–1996. [PubMed] [Google Scholar]
- 43.Provencher DM, Lounis H, Champoux L, Tetrault M, Manderson EN, Wang JC, Eydoux P, Savoie R, Tonin PN, Mes-Masson AM. Characterization of four novel epithelial ovarian cancer cell lines. In Vitro Cell Dev Biol Anim. 2000;36:357–361. doi: 10.1290/1071-2690(2000)036<0357:COFNEO>2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 44.Ries LAG, Melbert D, Krapcho M, Marriotto A, Miller BA, Feuer EJ, Clegg L, Horner MJ, Howlader N, Eisner MP, Reichman M, Edwards BK. Seer cancer statistics review, 1975–2004. Bethesda: Natl Cancer Inst; 2007. [Google Scholar]
- 45.Russo C, Ng AK, Pellegrino MA, Ferrone S. The monoclonal antibody CR11-351 discriminates HLA-A2 variants identified by T cells. Immunogenetics. 1983;18:23–35. doi: 10.1007/BF00401353. [DOI] [PubMed] [Google Scholar]
- 46.Scanlan MJ, Simpson AJ, Old LJ. The cancer/testis genes: review, standardization, and commentary. Cancer Immun. 2004;4:1. [PubMed] [Google Scholar]
- 47.Schrump DS, Fischette MR, Nguyen DM, Zhao M, Li X, Kunst TF, Hancox A, Hong JA, Chen GA, Pishchik V, Figg WD, Murgo AJ, Steinberg SM. Phase I study of decitabine-mediated gene expression in patients with cancers involving the lungs, esophagus, or pleura. Clin Cancer Res. 2006;12:5777–5785. doi: 10.1158/1078-0432.CCR-06-0669. [DOI] [PubMed] [Google Scholar]
- 48.Seliger B, Cabrera T, Garrido F, Ferrone S. HLA class I antigen abnormalities and immune escape by malignant cells. Semin Cancer Biol. 2002;12:3–13. doi: 10.1006/scbi.2001.0404. [DOI] [PubMed] [Google Scholar]
- 49.Serrano A, Tanzarella S, Lionello I, Mendez R, Traversari C, Ruiz-Cabello F, Garrido F. Expression of HLA class I antigens and restoration of antigen-specific CTL response in melanoma cells following 5-aza-2′-deoxycytidine treatment. Int J Cancer. 2001;94:243–251. doi: 10.1002/ijc.1452. [DOI] [PubMed] [Google Scholar]
- 50.Sigalotti L, Altomonte M, Colizzi F, Degan M, Rupolo M, Zagonel V, Pinto A, Gattei V, Maio M, Lubbert M, Wijermans PW, Jones PA, Hellstrom-Lindberg E. 5-aza-2′-deoxycytidine (decitabine) treatment of hematopoietic malignancies: A multimechanism therapeutic approach? Blood. 2003;101:4644–4646. doi: 10.1182/blood-2002-11-3458. [DOI] [PubMed] [Google Scholar]
- 51.Sigalotti L, Coral S, Altomonte M, Natali L, Gaudino G, Cacciotti P, Libener R, Colizzi F, Vianale G, Martini F, Tognon M, Jungbluth A, Cebon J, Maraskovsky E, Mutti L, Maio M. Cancer testis antigens expression in mesothelioma: role of DNA methylation and bioimmunotherapeutic implications. Br J Cancer. 2002;86:979–982. doi: 10.1038/sj.bjc.6600174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sigalotti L, Fratta E, Coral S, Tanzarella S, Danielli R, Colizzi F, Fonsatti E, Traversari C, Altomonte M, Maio M. Intratumor heterogeneity of cancer/testis antigens expression in human cutaneous melanoma is methylation-regulated and functionally reverted by 5-aza-2′-deoxycytidine. Cancer Res. 2004;64:9167–9171. doi: 10.1158/0008-5472.CAN-04-1442. [DOI] [PubMed] [Google Scholar]
- 53.Slingluff CL, Jr, Petroni GR, Chianese-Bullock KA, Smolkin ME, Hibbitts S, Murphy C, Johansen N, Grosh WW, Yamshchikov GV, Neese PY, Patterson JW, Fink R, Rehm PK. Immunologic and clinical outcomes of a randomized phase II trial of two multipeptide vaccines for melanoma in the adjuvant setting. Clin Cancer Res. 2007;13:6386–6395. doi: 10.1158/1078-0432.CCR-07-0486. [DOI] [PubMed] [Google Scholar]
- 54.Suyama T, Ohashi H, Nagai H, Hatano S, Asano H, Murate T, Saito H, Kinoshita T. The MAGE-A2 gene expression is not determined solely by methylation status of the promoter region in hematological malignancies. Leuk Res. 2002;26:1113–1118. doi: 10.1016/S0145-2126(02)00048-6. [DOI] [PubMed] [Google Scholar]
- 55.Wang RF, Johnston SL, Zeng G, Topalian SL, Schwartzentruber DJ, Rosenberg SA. A breast and melanoma-shared tumor antigen: T cell responses to antigenic peptides translated from different open reading frames. J Immunol. 1998;161:3598–3606. [PubMed] [Google Scholar]
- 56.Weber J, Salgaller M, Samid D, Johnson B, Herlyn M, Lassam N, Treisman J, Rosenberg SA. Expression of the MAGE-1 tumor antigen is up-regulated by the demethylating agent 5-aza-2′-deoxycytidine. Cancer Res. 1994;54:1766–1771. [PubMed] [Google Scholar]
- 57.Weiser TS, Guo ZS, Ohnmacht GA, Parkhurst ML, Tong-On P, Marincola FM, Fischette MR, Yu X, Chen GA, Hong JA, Stewart JH, Nguyen DM, Rosenberg SA, Schrump DS. Sequential 5-aza-2′ deoxycytidine-depsipeptide FR901228 treatment induces apoptosis preferentially in cancer cells and facilitates their recognition by cytolytic T lymphocytes specific for NY-ESO-1. J Immunother. 2001;24:151–161. doi: 10.1097/00002371-200103000-00010. [DOI] [PubMed] [Google Scholar]
- 58.Weiser TS, Ohnmacht GA, Guo ZS, Fischette MR, Chen GA, Hong JA, Nguyen DM, Schrump DS. Induction of MAGE-3 expression in lung and esophageal cancer cells. Ann Thorac Surg. 2001;71:295–302. doi: 10.1016/S0003-4975(00)02421-8. [DOI] [PubMed] [Google Scholar]
- 59.Wischnewski F, Pantel K, Schwarzenbach H. Promoter demethylation and histone acetylation mediate gene expression of MAGE-A1, -A2, -A3, and -A12 in human cancer cells. Mol Cancer Res. 2006;4:339–349. doi: 10.1158/1541-7786.MCR-05-0229. [DOI] [PubMed] [Google Scholar]
- 60.Yamshchikov GV, Barnd DL, Eastham S, Galavotti H, Patterson JW, Deacon DH, Teates D, Neese P, Grosh WW, Petroni G, Engelhard VH, Slingluff CL., Jr Evaluation of peptide vaccine immunogenicity in draining lymph nodes and peripheral blood of melanoma patients. Int J Cancer. 2001;92:703–711. doi: 10.1002/1097-0215(20010601)92:5<703::AID-IJC1250>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 61.Zendman AJ, Ruiter DJ, Van Muijen GN. Cancer/testis-associated genes: identification, expression profile, and putative function. J Cell Physiol. 2003;194:272–288. doi: 10.1002/jcp.10215. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.