Abstract
Adoptive therapy of cancer has been mostly tested in advanced cancer patients using tumor-infiltrating lymphocytes (TIL). Following discouraging results likely due to poor tumor-specificity of TIL and/or high tumor burden, recent studies reiterate the enormous potential of this therapy, particularly in melanoma. We had performed a phase II/III randomised trial on 88 stage III melanoma patients, who received autologous TIL plus IL-2 or IL-2 alone, after complete tumour resection. We reported previously clinical and immunological results supporting the ability of tumor reactive TIL infusion to prevent further development of the melanoma disease and to increase overall survival of patients bearing only one tumor invaded lymph node. The absence of correlation between overall and disease-free survival and the amount of infused tumor-specific TIL suggested that therapeutic efficiency might depend on other parameters such as antigen specificity, function or persistence of TIL. Here we studied the recognition of a panel of 38 shared tumor-associated antigens (TAA) by TIL infused to the patients included in this assay, in order to determine if treatment outcome could correlate with particular antigen specificities of infused TIL. Results show that the infusion of Melan-A/MART-1 reactive TIL appears to be associated with a longer relapse-free survival for HLA-A2 patients. These results further support the relevance of Melan-A/MART-1 antigen as a prime target for immunotherapy protocols in melanoma.
Keywords: T cells, Melan-A, Immunotherapy, TIL, Melanoma
Introduction
The efficacy of adoptive T-cell therapy of tumors was first shown in transplanted animal tumor models, using tumor infiltrating lymphocytes (TIL), or specifically sensitized T lymphocytes [12, 24, 29] and was confirmed recently in animal tumor systems that better model human malignancies [11, 14]. In humans, evidence for the efficacy of T cell adoptive therapy has been obtained for hematopoietic cancers, through targeting viral antigens [15, 30]. However, in solid tumors, heterogeneous clinical results have been reported, that might reflect heterogeneity of infused TIL populations, in terms of activation status and frequency of tumor-specific T cells obtained by different culture methods and/or from different tumor samples. This could also be explained by the clinical status of treated patients. Indeed, TIL were initially used to treat bulky disease, and tumor burden may contribute to a significant tumor-derived suppressive environment.
The interest of passive immunotherapy in the treatment of solid tumors has been recently reinforced by clinical trials in malignant melanoma using polyclonal lymphocytes [7, 8, 23] or tumor antigen-specific T cell clones [32, 33]. Significant tumor regression rate [8, 32, 33], vitiligo induction [8], increased relapse-free survival [7], migration of tumor-specific lymphocytes to metastatic sites and lymph nodes have been reported [23, 33]. Although these results hold promise, many questions remain to be thoroughly investigated, such as the antigens recognized by transferred T cells. A previous study addressing this question in melanoma patients treated by TIL therapy, suggested a correlation between objective tumor regressions and the presence of gp100-specific T cells among infused TIL [19]. Nonetheless, recent studies of adoptive transfer of Melan-A-specific lymphocytes strongly support the interest of this antigen for melanoma therapy [8, 32, 33].
We recently showed the efficiency of tumor reactive TIL infusion to prevent the relapse of stage III melanoma patients who had only one ILN, after complete tumor resection [7, 20]. Since this efficiency was not correlated with the amount of melanoma reactive TIL [20], we asked here whether particular antigen specificities of TIL could correlate with therapeutic efficiency. We used COS cells expressing a panel of 38 shared melanoma antigens upon transfection, to check the presence of T cells specific for these antigens, and we analyzed the distribution of antigen responses according to the clinical status of treated patients, i.e. relapse-free (RF) patients and patients who relapsed (R).
Materials and methods
Melanoma patients
Patients under study had been included in an adoptive immunotherapy protocol performed between 1994 and 1998. The trial was designed and conducted in accordance with the Declaration of Helsinki. The therapy protocol was approved by the Institutional Ethics Committee and registered with the regulatory state authority, and all patients gave written informed consent before enrolling in the study. This protocol was a randomized study designed to compare the efficiency of TIL + IL-2 versus IL-2 injections to patients suffering from stage III malignant melanoma (i.e. lymph node metastases as described by the American Joint Committee on Cancer), without distant metastases. Eighty-eight patients were randomly included in this trial, 44 patients were treated by TIL/IL2 and 44 by IL2 only. For all the patients, the treatment was performed within 6–10 weeks after metastatic lymph node resection. Patients included in the arm “ TIL + IL-2 ” received two intravenous infusions of autologous ex-vivo expanded TIL, on day 1 of the treatment, and 1 month later. Each TIL infusion was followed by one daily subcutaneous injection of IL-2 (6 millions I.U., Chiron), from day 1 to 5 and from day 8 to 12, each month, for 2 months. Here we analyzed the antigen specificities recognized by TIL infused to those patients, 7 years post-treatment.
An additional group of five HLA-A2 patients, treated with TIL and IL-2, was also analyzed in the present study. They have been enrolled in a clinical open study performed between December 2001 and April 2005. This study concerned 19 stage III patients bearing only one tumor invaded lymph node, due to the good clinical benefit observed for this subgroup of patients in the previous study. The TIL infused to these five HLA-A2 patients were analyzed for the presence of Melan-A/A2-specific T cells, using tetramers.
Production of melanoma TIL
TIL lines were produced in Good Manufacturing Practice conditions in the Unit of Cellular and Genetic Therapy (CHRU, Nantes, France) according to a procedure described previously [16, 27]. Briefly, short-term cultured TIL were isolated by culturing cryopreserved fragments of stage III metastatic lymph nodes into 12-well tissue culture plates with X-Vivo 15 serum-free medium (Bio*Whittaker, Walkersville, MD, USA) containing 150 U/ml rIL2 (Eurocetus, Rueil-Malmaison, France) and glutamine (1 nM) (Bio*Whittaker) for 10–14 days. Therapeutic ex-vivo expanded TIL were derived as follows: 1.8 × 106 short-term cultured TIL were plated at 300 viable lymphocytes/well with irradiated feeder cells (allogeneic PBL and B-EBV cells) into U-bottom microplates in 200 μl of rIL-2 medium. PHA-P (Difco, Detroit, MI, USA) was added on day 0 (15 μg/ml). Ten days later, lymphocytes were recovered from the culture plates, adjusted to 1 × 106 cells/ml in r-IL2 containing medium and transferred into culture trays for an additional 10 days. The final TIL harvest was performed by centrifuging, washing and suspending the TIL in 4% human serum albumin (LFB, Les Ulis, France). A second TIL expansion was performed, within one month from the first one, starting from 1.8 × 106 cryopreserved short-term cultured TIL. Aliquots of therapeutic TIL were cryopreserved, to study their antigen specificity. These cryopreserved TIL were then expanded following the same procedure during a 14-days period, before testing.
Transfection of COS-7 cells
The COS cells were transfected with the appropriate HLA class I alleles cDNAs (corresponding to each patient’s alleles) and with the cDNA coding for the 38 TAA listed in Table 1. The antigens put together 19 shared tumor specific antigens - MAGE-A1, A2, A3, A4, A6, A12, GAGE-1, 2, 3, 4, 5, 6, LAGE-1a, LAGE-1b, LAGE-2/NY-ESO-1, NA88-A, NA17-A (GnT-V), BAGE (for review see [26]) and NA134-A [10]; for six melanocytic differentiation antigens -Melan-A/MART-1, Tyrosinase, gp100, gp75; MC1R, TRP-2; for eight overexpessed antigens - RAGE-1, 2, 3, 4, p53, PRAME, Her-2 neu, EphA2; and for five mutated and other antigen types-N-RAS (wild type and mutated), hTERT, Survivin, Hsp70 [26].
Table 1.
Antigen coding cDNAs transfected into COS cells
| Shared tumor specific antigens | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| MAGE-1 | MAGE-2 | MAGE-3 | MAGE-4 | MAGE-6 | MAGE-12 | GAGE-1 | GAGE-2 | GAGE-3 | GAGE-4 | GAGE-5 | GAGE-6 |
| 13 | 14 | 15 | 16 | 17 | 18 | 19 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Lage-1a | Lage-1b | Lage-2 | NA88-A | NA17-A | NA134-A | BAGE |
| Melanocytic differentiation antigens | Mutated/other antigens | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | 30 | |
| Melan-A | gp100 | Tyrosin. | MC1R | TRP-2 | Gp75 | ras WT. | ras muta | HSP70 | HTERT | survivin | |
| Overexpressed | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 31 | 32 | 33 | 34 | 35 | 36 | 37 | 38 | ||||
| RAGE−1 | RAGE−2 | RAGE−3 | RAGE−4 | P53 | PRAME | Her2-neu | EphA2 | ||||
aMix of four plasmids at an equimolar ratio containing mutations in the codon 61 of Nras: Q61R, Q61L, Q61K, and Q61P
Many of the HLA class I cDNAs were kindly provided by T. Boon (LICR, Brussels, Belgium) or by E. Houssaint (Unit 601 INSERM, Nantes, France). Some were cloned in our laboratory. cDNAs coding for cancer-germline and melanocytic differentiation antigens were also provided by T. Boon. NA17-A NA88-A, NA-134-A and N-RAS (bearing four different mutations at position 61) cDNAs were cloned in our laboratory [10, 13, 22, 25]. Survivin, telomerase, Hsp70, Her-2 neu and EphA2 cDNAs were a gift from K. Kosmatopoulos (Unit 484 INSERM, Villejuif, France). MC1R cDNA was a gift from R. Kissling (Karolinska institute, Stockholm, Sweeden). COS7 cells transfection was performed by the DEAE-dextran-chloroquine method [4, 5]. In brief, 16.5 × 103 COS-7 cells were co-transfected with 100 ng of plasmid coding for an HLA and 100 ng of plasmid coding for a MAA. Transfected COS cells were then used to stimulate TIL populations.
T cell stimulation assay
TIL (5 × 103–5 × 104) were added to transfected COS-7 cells 48 h after transfection. Culture supernatants were harvested 6 h later and tested for TNF content. TNF-α determination was done by a biological assay measuring the cytotoxicity of culture supernatant on the highly sensitive WEHI 164 clone 13 in a MTT colorimetric assay, as previously described [6, 9].
Construction of HLA-A*0201/peptide tetramers
HLA-A0201/peptide α3-mutated monomers were generated as previously described [3]. Recombinant proteins were produced as inclusion bodies in Escherichia coli XA90F’LacQ1, dissolved in 8 M urea, and refolded with 50 μg/ml of Melan-A A27L peptide (ELAGIGILTV), purchased from Epytop (Nîmes, France). Tetramerisation was performed as previously described [3]. Briefly, HLA monomers were biotinylated for 4 h at 30°C with 6 μg/ml BirA (Immunotech), purified on a monoQ column (Pharmacia, St Quentin en Yvelines, France) and tetramerised with PE-labeled streptavidin (Sigma, St Louis, MO, USA) at a molar ratio 4/0.8.
Tetramer labeling
To minimize non specific staining, HLA-A2-Melan-A tetramer was titered and used at the lowest concentration that showed a clearly distinguishable positive population in Melan-A-specific CTL generated as described above. TIL were coincubated for 1 h at 4°C in the dark with Melan-A tetramer (10 μg/ml) and CD8-FITC mAb (5 μg/ml). After washing in PBS-0.1%BSA, cells were resuspended in PBS and analyzed on a FACScan. Compensation was checked before each acquisition. Events (105 among CD8 positive T cells) were collected using a FACScan set on a maximal flow rate of 1,000 events/s.
IFN-γ production assay
A total of 1 × 105 TIL were stimulated by 3 × 105 stimulator cells (unpulsed T2 cells or T2 pulsed with 5 μM of the natural Melan-A26–35 peptide) in 200 μl of RPMI 1640—10% FCS in the presence of brefeldin A, 10 μg/ml (Sigma, St Louis MO, USA) in round-bottom 96-well plates. The cultures were incubated for 6 h at 37°C in 5% CO2 humidified atmosphere. For intracytoplasmic cytokine staining, cells were then fixed 10 min at room temperature in a solution of PBS 4% paraformaldehyde (Sigma), washed and stored at 4°C until labeling. Fixed stimulated TIL were stained for IFN-γ production using the method described by Jung et al. [17]. After staining, cells were resuspended in PBS and 1.5 × 104 events were analyzed on a FACScan flow cytometer using Cell Quest software (Beckton Dickinson, Grenoble, France). Percent of lymphocytes producing IFN-γ in response to the natural Melan-A peptide were deduced after subtracting the percent obtained on unpulsed T2 cells.
Results
TAA recognized by CD8 TIL infused to patients
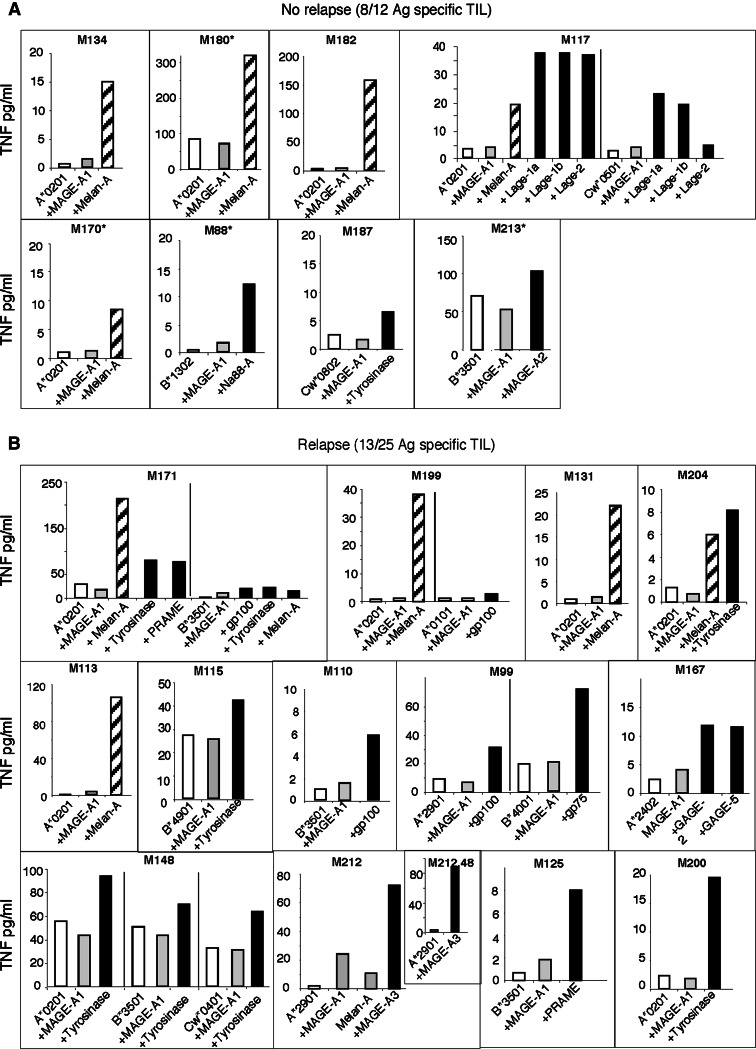
We analyzed retrospectively the ability of TIL populationis that had been infused to 37 of the 44 patients included in the clinical assay described above [7]. TIL populations infused to seven patients could not be analyzed due to the absence of available frozen material. To this end, TIL were stimulated by COS7 cells cotransfected with each HLA class I patient’s cDNA together with each cDNA of the TAA panel shown in Table 1. TNF secretion by stimulated TIL was considered significant when obtained in two separate experiments performed at two TIL/transfected COS cell ratios. Responses to at least one TAA were detected among the TIL infused to 8/12 (67%) patients who did not relapsed (RF-patients) (Fig. 1a; Table 2), and among those infused to 13/25 (52%) patients who relapsed (R-patients) (Fig. 1b; Table 3). Therefore, the frequencies of TIL containing antigen-specific T cells were similar in both groups of patients.
Fig. 1.
Recognition of tumor-associated antigens by TIL populations infused to RF-patients (a) and to R-patients (b). COS cells were transfected with a cDNA coding for an HLA molecule and a cDNA coding for an antigen. After 48 h incubation, COS cells were co-cultured 6 h at 37°C with TIL populations. Thereafter, TNF-α secreted in the supernatant was measured. Open square TNF produced by TIL populations in response to COS cells transfected with the HLA molecule alone. Shaded square responses to COS cells transfected with the HLA and a control antigen. Black shaded square responses to COS cells transfected with a recognized HLA/antigen association. Square with diagonal TNF produced by TIL populations in response to COS cells transfected with the HLA-A*0201 and Melan-A/MART-1. Asterisk patients bearing only one invaded lymph node. Inset in M212 panel illustrates a specific response to MAGE-A3/A29 of a T cell clone derived from this TIL population
Table 2.
HLA/Antigen complexes recognized by tumor-infiltrating lymphocytes (TIL) infused to RF-patients (n = 12)
| Patient | HLA-A | HLA-B | HLA-C | HLA/antigen recognized |
|---|---|---|---|---|
| Positive TIL populations (n = 8)a | ||||
| M88c | A*0201–0301 | B*1302–4002 | Cw*0602–0202 | B*1302/NA88-A |
| M117 | A*0201–2402 | B*4402 | Cw*0501–0202 | A*0201/Melan-A/lage |
| Cw*0501/lage | ||||
| M134 | A*0201 | B*0801 | Cw*0701 | A*0201/Melan-A |
| 170c | A*0201–0301 | B*1402–4402 | Cw*0501–0802 | A*0201/Melan-A |
| 180c | A*0201–0101 | B*0702 | Cw*0602–0702 | A*0201/Melan-A |
| M182 | A*0201 | B*1801–4402 | Cw*1203–0501 | A*0201/Melan-A |
| M187 | A*0301 | B*1402–3503 | Cw*0401–0802 | Cw*0802/tyrosinase |
| M213c | A*0101–0301 | B*3501 | Cw*0303–0401 | B*3501/MAGE-A2 |
| Negative TIL populations (n = 4)b | ||||
| M174c | A*2401–6801 | B*1401–4402 | Cw*0501–0802 | – |
| M197c | A*0301 | B*1801–0702 | Cw*0702–1203 | – |
| M177c | A*2402–2902 | B*0702 | Cw*0102–0802 | – |
| M215c | A*2301–6801 | B*5801 | Cw*0202–0701 | – |
aTIL populations that recognized at least one of the tested antigens in one HLA context
bNo recognition of any HLA/antigen combination tested
cMelanoma patients with only one invaded lymph node at treatment
Table 3.
HLA/Antigen complexes recognized by TIL infused to R-patients (n = 25)
| Patient | HLA-A | HLA-B | HLA-C | HLA/Antigen recognized |
|---|---|---|---|---|
| Positive TIL populations (n = 13)a | ||||
| M99 | A*2901 | B*4001–4403 | Cw*0304–1601 | A*2901/gp100 |
| B*4001/gp75 | ||||
| M110 | A*0301–1101 | B*3501 | Cw*0401–0602 | B*3501/gp100 |
| M113 | A*0201 | B*0702–1302 | Cw*0602–0702 | A*0201/Melan-A |
| M115 | A*0201–2901 | B*4901–4402 | Cw*0501–0701 | B*4901/tyrosinase |
| M125 | A*0201–1101 | B*2705–3501 | Cw*0702–0401 | B*3501/PRAME |
| M131 | A*0201–1101 | B*3501 | Cw*0602–0202 | A*0201/Melan-A |
| M148 | A*0201–1101 | B*3501–5101 | Cw*0401–1502 | A*0201/tyrosinase |
| B*3501/tyrosinase | ||||
| Cw*0401/tyrosinase | ||||
| M167 | A*2402 | B*4001 | Cw*0304 | A*2402/GAGE |
| M171 | A*0101–0201 | B*0801–3501 | Cw*0401–0701 | A*0201/Melan-A/PRAME/tyr. |
| B*3501/Melan-A/tyr./gp100 | ||||
| M199 | A*0201–0101 | B*0801 | Cw*0701–1402 | A*0201/Melan-A |
| A*0101/gp100 | ||||
| M200 | A*0201–0101 | B*1801 | Cw*0701 | A*0201/tyrosinase |
| M204 | A*0201 | B*5101–5501 | Cw*0303–1502 | A*0201/Melan-A/tyrosinase |
| M212 | A*2901–3201 | B*0702 | Cw*0602–0702 | A*2901/MAGE-A3 |
| Negative TIL populations (n = 12)b | ||||
| M100 | A*0201–3201 | B*4402 | Cw*0401–0501 | – |
| M126 | A*0301 | B*3501 | Cw*0401–0602 | – |
| M132 | A*0201–2402 | B*4402 | Cw*0303–0501 | – |
| M144 | A*0101–2401 | B*0801–1402 | nd | – |
| M145 | A*0201–2901 | B*0702–4402 | Cw*0102–0501 | – |
| M149 | A*0201 | B*3508–0702 | Cw*0702–0602 | – |
| M153 | A*0201–2901 | B*0801 | Cw*0701–1402 | – |
| M164 | A*6802 | B*1801–1402 | Cw*0501–0802 | – |
| M192 | A*0201–2902 | B*4501–5101 | Cw*0602–1402 | – |
| M193 | A*2501–3301 | B*1402–1801 | Cw*0802–1203 | – |
| M196c | A*0201–1101 | B*4403 | Cw*1601–0304 | – |
| M209c | A*1101 | B*1801–3501 | Cw*0403–0501 | – |
aThe TIL populations that recognized at least one of the tested antigens in one HLA context
bNo recognition of any HLA/antigen combination tested
cMelanoma patients with only one invaded lymph node at treatment
TNF responses of TIL infused to both groups of patients are shown in Fig. 1. Of note, many of these responses were detected in new HLA contexts, showing the existence of undescribed MAA-derived epitopes (encircled HLA contexts in Fig. 2). This was the case for the responses to gp100 in the HLA-A*0101 and A*2901 contexts, to GAGE in the HLA-A*2402 context, to gp75 in the HLA-B*4001 context and to Lage in the HLA-Cw*0501 context. This was also the case for Tyrosinase, recognized in the HLA- B*4901, Cw*0401 and Cw*0802 contexts, by three different TIL populations.
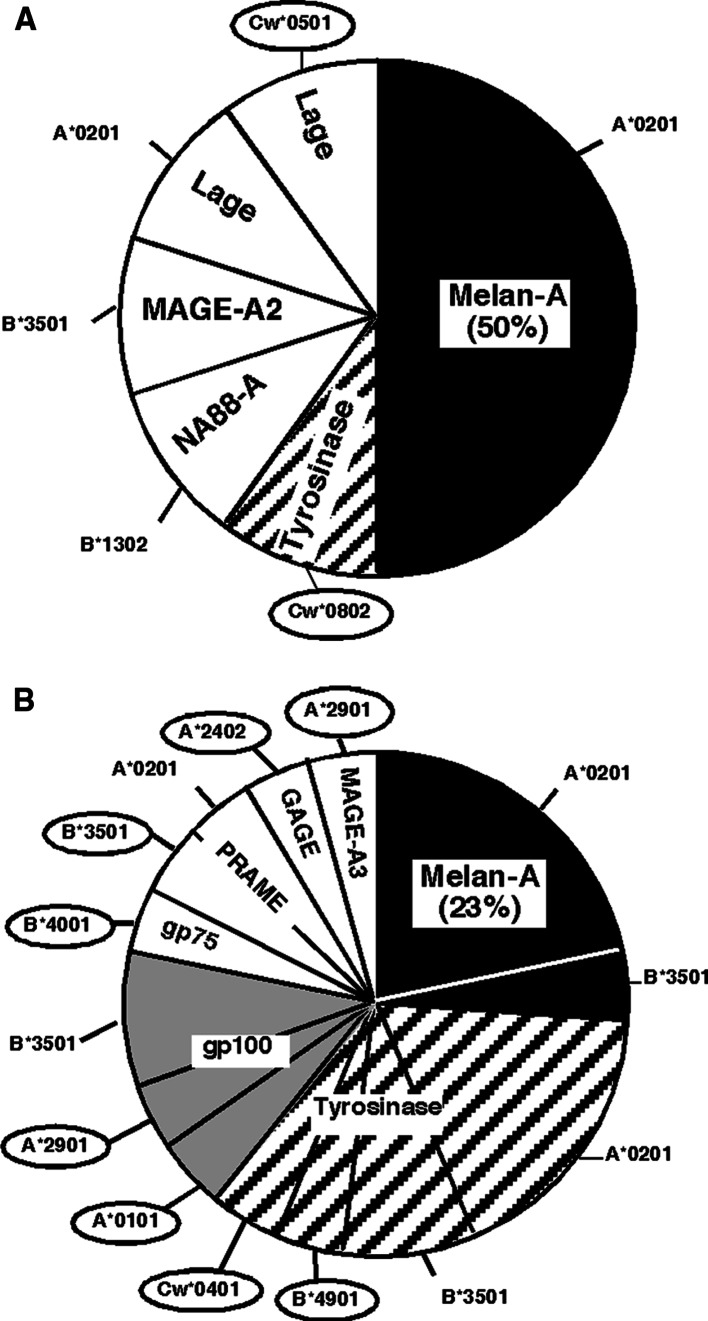
Fig. 2.
Distribution of antigens recognized by 8/12 TIL infused to RF-patients (a) and 13/25 R-patients (b). Encircled HLA contexts illustrated undescribed HLA/Antigen associations
The significance of some weak TNF responses was addressed by deriving T cell clones of identical specificity. This was achieved and reported previously for B*3501 restricted responses of M171 TIL population to gp100, Melan-A and Tyrosinase, which allowed to identify the new corresponding epitopes [2]. We also derived T cell clones specific for A*0101/gp100 and A*2402/GAGE-2, respectively from TIL M199 and M167 [1]. The response of a T cell clone specific for A*2901/MAGE-3 (M212.48) is shown in Fig. 1. These clonal responses establish the specificity of the weak TNF response of polyclonal TIL.
Melan-A/A2-specific TIL infusion and clinical outcome
The distribution of antigens recognized by TIL was then analyzed according to the clinical status of treated patients (Fig. 2). For TIL infused to RF-patients, 60% of the responses were directed against melanocytic differentiation antigens, with a prevalence of responses against Melan–A/MART-1. The remaining 40% of responses were directed against four shared tumor-specific antigens. For TIL infused to R-patients, a higher proportion of TIL responses were directed against differentiation antigens (83). However, Melan-A/A2-specific TIL responses were less frequent in R- than in RF-patients (respectively 23 and 50% of all responses).
As far as HLA distribution could act upon the frequencies of recognized antigens, we then analyzed both groups for HLA distribution. As shown in Tables 2 and 3, frequencies of HLA allele expression were similar between R- and RF-patients. For example, the HLA-A*0201 molecule (in bold in these tables) was expressed by 50 and 64% of RF- and R-patients, respectively. Nonetheless, these two groups of patients clearly differed by the frequency of HLA-A2-restricted responses (in bold in the tables). Indeed, these responses were detected in 5/6 (83%) HLA-A2 TIL populations infused to RF-patients, and in only 7/16 (44%) of those infused to R-patients (Tables 2, 3), suggesting that HLA-A2 specificities of infused TIL could have influence the patient’s clinical outcome. We therefore focused our study on the antigen specificities of TIL infused to the 22 HLA-A2 patients, according to clinical outcome.
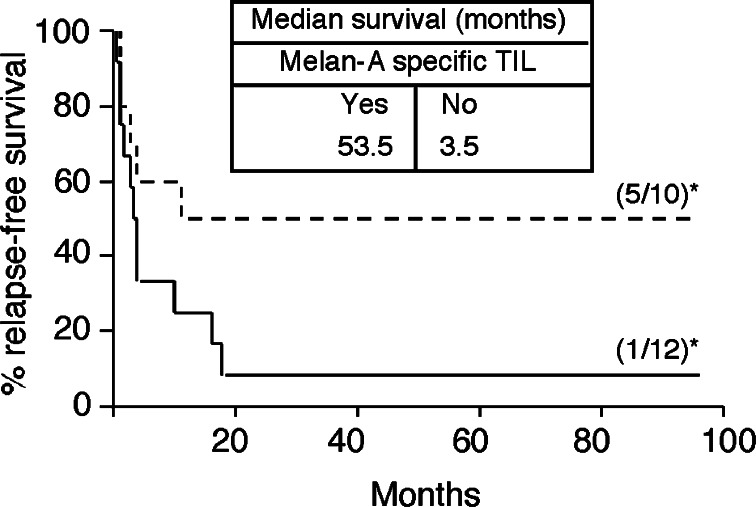
As shown in Tables 2 and 3, 10/22 HLA-A2 patients had been treated with Melan-A-specific TIL and 12/22 did not receive such cells. Among the ten patients treated by Melan-A-specific TIL, five did not relapse, whereas all the patients but one (Table 2), who did not receive such lymphocytes, relapsed. Figure 3 illustrates the duration of relapse-free survival of the two groups of patients. This analysis shows that relapse-free survival was longer for the group of patients that received Melan-A specific TIL, compared with patients who did not receive such cells, median survivals being respectively of 53.5 and 3.5 months. Nonetheless, as far as the number of analyzed HLA-A2 patients was rather low, Logrank statistical analysis of these results remained of low significance (P = 0.06).
Fig. 3.
Relapse-free interval in 22 HLA-A2 melanoma patients treated with IL-2 and TIL containing (dashed line) or not (solid line) Melan-A specific lymphocytes. Asterisk number of RF-patients/total number of patients
In order to increase the number of HLA-A2 patients analyzed, we performed the same analysis on TIL populations infused to patients included in an open study (see Materials and methods). Among the 19 patients that had been included in this study, 5 were HLA-A*0201 and had been treated between October 2002 and April 2005. Four of them received TIL containing Melan-A/A2-specific lymphocytes, and two have not relapsed, whereas the other patient who did not receive such lymphocytes relapsed.
Therefore, we analyzed the clinical responses according to the absence or presence of Melan-A-specific lymphocytes among infused TIL for all the HLA-A2 patients (22 from the first protocol and the 5 additional patients) (Table 4). Taken together, these results support a significant correlation between Melan-A-specific TIL infusion and relapse prevention (two-sided Fisher exact test P = 0.032).
Table 4.
Clinical status of 27 HLA-A2 patients according to the presence of Melan-A-specific lymphocytes among infused TIL
| R-patients | RF-patients | ||
|---|---|---|---|
| Melan-A specific TIL | 7 | 7 | n = 14 |
| No Melan-A specific TIL | 12 | 1 | n = 13 |
| n = 19 | n = 8 |
Amounts of Melan-A-specific lymphocytes infused to HLA-A2 patients
We wondered whether there could be a correlation between the number or the avidity of infused Melan-A-specific TIL and the clinical status of the patients. Therefore, the fraction of Melan-A-specific lymphocytes was quantified using a HLA-A2 tetramer folded with the Melan-A A27L analogue [31], and their ability to produce IFN-γ in response to the natural Melan-A decapeptide was tested in all the HLA-A2 TIL populations (n = 27).
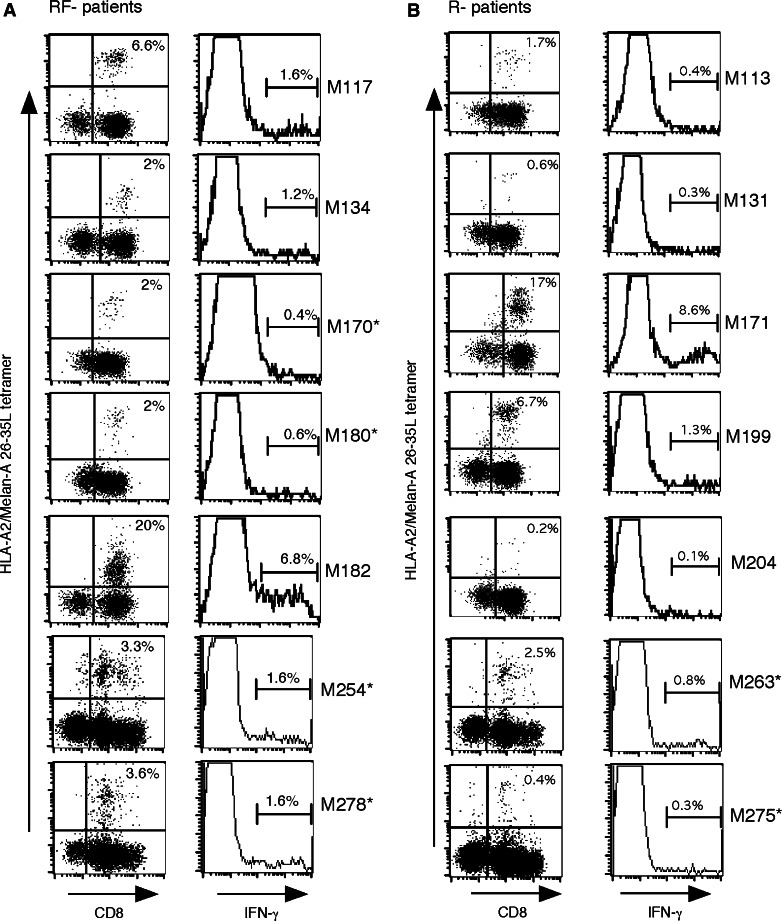
Tetramer positive cells were detected in each TIL population for which a response against Melan-A antigen had been evidenced by the COS transfection method. The fraction of tetramer positive cells ranged between 2 and 20% of TIL infused to relapse-free patients (Fig. 4a, left panel), while it ranged between 0.2 and 17% of TIL infused to patients who have relapsed (Fig. 4b, left panel). As shown by IFN-γ production, only a fraction of tetramer-labeled lymphocytes were reactive against the natural decapeptide, ranging from 0.4 to 6.8% for relapse-free patients (Fig. 4a, right panel) and to 0.1–8.6% for patients who relapsed (Fig. 4b, left panel). The amount of tetramer-labeled TIL and TIL exhibiting a specific IFN-γ response to peptide-pulsed T2 cells, infused to HLA-A2 patients, was therefore estimated from these percentages and from the total amount of infused TIL (Table 5). It appears that 14 patients had been injected with Melan-A-specific TIL ranging from 37 millions to 3.7 billions among them 28 millions–1.9 billions were reactive against the natural decapeptide. However, as shown in Table 5, there was no correlation between the clinical outcome and the number of tetramer-labeled TIL nor the number of Melan-A26–35 reactive TIL infused. Indeed, among the six patients who received the highest number of Melan-A-specific TIL, two relapsed and four did not. Thus, length of relapse-free survival of TIL-treated patients correlated with the infusion of Melan-A-specific lymphocytes, but the minimal number of these cells required for such an effect could not been established.
Fig. 4.
HLA-A2 TIL labeled with the A2/Melan-A26–35L tetramer a RF-patients and b R-patients. TIL were coincubated for 1 h at 4°C in the dark with Melan–A tetramer (10 μg/ml) and CD8-FITC mAb (5 μg/ml). Compensation was checked before each acquisition. Events (105 among CD8 positive T cells) were collected using a FACScan set on a maximal flow rate of 1,000 events/s. Asterisk melanoma patients bearing only one invaded lymph node. Patients M254 to M278 have been enrolled in the open study
Table 5.
Amount of Melan-A-specific TIL infused to patients
| Patients | Total TIL (10−9)b | Melan-A specific TIL (10−6)c | Melan-A26–35 reactive lymphocytes (10−6)d |
|---|---|---|---|
| Melan-A/A2 specific TIL (n = 14) | |||
| RF-patients | |||
| M117 | 33.4 | 2,200 | 534 |
| M182 | 3.7 | 740 | 252 |
| M278 a | 13.2 | 475 | 211 |
| M134 | 22.5 | 450 | 270 |
| M170a | 9.5 | 190 | 38 |
| M180a | 9 | 180 | 54 |
| M254 a | 3.3 | 109 | 53 |
| R-patients | |||
| M171 | 22 | 3,740 | 1,892 |
| M199 | 8.3 | 556 | 108 |
| M113 | 21.2 | 360 | 85 |
| M263 a | 12.6 | 315 | 101 |
| M131 | 15.5 | 93 | 46 |
| M204 | 26 | 52 | 26 |
| M275a | 9.2 | 37 | 28 |
aMelanoma patients bearing only one invaded lymph node
bTotal amount of TIL infused to patients
cEstimated amount of Melan-A specific TIL infused (detected by tetramer labeling). Patients indicated in italics have been patients enrolled in the open study
dEstimated amount of infused Melan-A26–35 reactive lymphocytes (detected by IFN-γ intracellular labeling in response to peptide pulsed-T2 cells)
Discussion
The infusion of autologous tumor-reactive lymphocytes to melanoma patients in an adjuvant setting had been designed to protect stage III melanoma patients against tumor recurrence. Results published previously showed that infusion of TIL containing melanoma-reactive lymphocytes prevented the relapse and increased overall survival for a significant fraction of melanoma patients, bearing only one ILN [7, 20]. As far as this therapeutic efficiency was correlated with the infusion, but not with the amount, of lymphocytes specific for autologous melanoma cells, we investigated now whether it might correlate with the nature and/or the diversity of tumor-associated antigens recognized by these TIL. To this end, TIL populations were tested in hindsight, for their ability to recognize a panel of 38 shared TAA. Although nonexhaustive, this panel put together a significant number of the TAA most frequently targeted in immunotherapy protocols, i.e. tumor-specific antigens such as NA17-A, MAGE and NY-ESO-1 and melanocytic differentiation antigens (Melan-A/MART-1, Tyrosinase, gp100...). It also included overexpressed tumor antigens, ras mutated antigens (expressed by 20% melanoma tumors) and antigens commonly associated with such as telomerase, Hsp70 and survivin.
Data showed that more than half (21/37) TIL populations contained lymphocytes specific for at least one of these antigens. Most responses occurred in already described HLA contexts, but a significant proportion of infused TIL recognized undescribed HLA/antigen associations, i.e. unknown melanoma epitopes. These unknown epitopes represented 30% of all antigen specificities recognized by TIL. A similar observation had been reported in a previous study [1], and these two studies clearly show that a great number of epitopes deriving from known TAA remain to be identified. Therefore, the present results support the notion that melanoma tumors frequently trigger the expansion of TAA-specific lymphocytes, justifying the development of immunotherapy in melanoma patients.
The number of TIL populations containing TAA-specific lymphocytes infused to R and RF-patients was not significantly different, respectively 13/25 (52%) and 8/12 (67%). Overall, this shows that the mere presence of lymphocytes specific for any TAA was not sufficient to influence the clinical outcome of patients enrolled in this study. Although the diversity of TAA-specific responses analyzed here is much higher than these analyzed in similar studies, usually restricted to few HLA contexts and some selected antigens [18, 19], this approach remains limited by the variability of expression of these TAA by melanomas, and by the expression of appropriate HLA contexts restricting their recognition. Furthermore, it has to be stressed that efficient immune response could be triggered by tumor-specific lymphocytes directed against unknown shared tumor antigens or individual mutated neoantigens, not tested in this study [21]. As an example, among the ten TIL populations derived from patients bearing only one invaded lymph node, only four exhibited antigen specific responses (Tables 2, 3). Nevertheless, three out of the six negative populations have been tested in a previous study against the autologous melanoma cell line and contained melanoma reactive TIL [20] suggesting the recognition of unknown antigens, potentially private antigens.
Nonetheless, and despite the absence of patient’s selection on the basis of HLA expression, we observed dissimilar frequencies of responses against COS cells expressing Melan-A/A2 in TIL infused to RF-patients and R-patients (respectively, 50% and 23% of all responses). Since this difference was not due to a higher representation of HLA-A2 patients in the RF-group (50 and 64% of RF and R-patients, respectively), this suggested that the infusion of Melan-A/A2-specific TIL could have been important to prevent the relapse of melanoma patients.
To check this hypothesis, we restricted our comparison to HLA-A2-treated patients. Ten out of 22 HLA-A2 patients had received TIL reactive to Melan-A transfected COS cells. Half of these patients (5/10) remained relapse free whereas only 1/12 patients who did not receive such cells remained relapse free (Tables 2, 3). Median survivals of the two groups of patients also appeared clearly different (53.5 vs 3.5 months, respectively for patients who received or not Melan-A/A2-specific TIL) (Fig. 3), but statistical analysis of these data remained of low significance (two-side P value = 0.06). We therefore extended this analyzis to a group of five HLA-A2 patients treated more recently with the same clinical design in an open study. Together, data from both studies showed that 14/27 patients have been treated with TIL-containing Melan-A/A2-specific TIL and that 7/14 remained relapse free, whereas only 1/13 patients who did not receive such TIL did not relapsed. These results support a significant correlation (P = 0.032) between the infusion of Melan-A-specific TIL and relapse prevention (Table 4).
Responses to transfected COS cells indicated the presence of TNF secreting Melan-A-specific lymphocytes, but did not provide any information about their frequency among infused TIL. Therefore, the last part of our study addressed this question using tetramer labeling of HLA-A2 TIL populations and IFN-γ intracellular labeling in response to the natural decapeptide. Indeed, as the tetramer used was folded with a Melan-A peptide analogue designed to bind with an improved affinity to the HLA-A2 [31], we also quantified the responses against a naturally expressed Melan-A epitope, to validate the relevance of these results. Supporting the significance of the COS study, each population reactive against transfected COS cells contained a significant fraction of lymphocytes labeled with the tetramer, among which a more restricted population was able to produce IFN-γ in response to the Melan-A26–35 peptide. Therefore, from the fraction of tetramer-labeled TIL, the fraction of IFN-γ producing TIL and the total number of TIL transferred to patients [27], we estimated respectively the amount of tetramer labeled TIL and of Melan-A26–35 reactive TIL infused to HLA-A2 patients. As stated in last part of the results, there was no correlation between these amounts and the clinical outcome. This suggests that other factors not analyzed here, such as the persistence of Melan-A-specific TIL in vivo, or their ability to initiate an anti-tumor response and to induce epitope or repertoire spreading might be critical for the clinical efficiency. Indeed, an extensive comparison of tumor reactive TIL adoptively transferred to responder and non responder metastatic melanoma patients suggested that the property best correlated with tumor regression was the persistence of these cells following transfer [28]. Otherwise, we published recently the immune follow-up of melanoma patients who received infusion of Melan-A specific clones, and our results suggested that an anti-tumor response initiated by transferred cells can be evidenced by measuring quantitative or qualitative variations in Melan-A-specific repertoire [32]. These parameters will be evaluated in a future multicentric phase III protocol, using TIL infusion in an adjuvant setting to melanoma patients.
The adoptive transfer of highly enriched, or clonal, Melan-A/A2 specific T cells (derived from TIL or PBMC) had been used previously in clinical studies performed on metastatic melanoma patients. Although these studies were not designed to establish the clinical benefit of this transfer, clinical responses, ranging from tumor stabilizations to complete regressions, have been reported [8, 23, 32, 33]. Nonetheless, the overall response rate observed remained low. This could be ascribed to the heterogeneous functional and survival capacities of transferred cells as suggested by some groups but also to the high tumor burden. Therefore, improving the clinical efficacy of these treatment might be achieved by two major ways: (1) the selection of Melan-A-specific T cells with improved properties such as long-term survival, associated with telomer length and CD28 expression [34], and high tumor reactivity (2) the use of these treatments for patients at an earlier stage of the disease.
Therefore, this present study suggests that the adoptive transfer of even low amounts of Melan-A specific lymphocytes could be an efficient treatment for HLA-A2 melanoma patients at an early stage of the disease. These encouraging clinical results have given rise to the development of a multicentric phase III protocol, which will allow us to check the relevance of these informations.
Acknowledgments
This work was supported by grants from the “Ligue Nationale contre le Cancer”, the “INSERM” and by a grant from ENACT proposal number 503306. We thank Dr. Rivain-Sebille for her help in the statistical analysis of the data.
Contributor Information
Francine Jotereau, Email: jotereau@nantes.inserm.fr.
Nathalie Labarrière, Phone: +33-2-40084720, FAX: +33-2-40356697, Email: nlabar@nantes.inserm.fr.
References
- 1.Benlalam H, Labarriere N, Linard B, Derre L, Diez E, Pandolfino MC, Bonneville M, Jotereau F. Comprehensive analysis of the frequency of recognition of melanoma-associated antigen (MAA) by CD8 melanoma infiltrating lymphocytes (TIL): implications for immunotherapy. Eur J Immunol. 2001;31:2007–2015. doi: 10.1002/1521-4141(200107)31:7<2007::AID-IMMU2007>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 2.Benlalam H, Linard B, Guilloux Y, Moreau-Aubry A, Derre L, Diez E, Dreno B, Jotereau F, Labarriere N. Identification of five new HLA-B*3501-restricted epitopes derived from common melanoma-associated antigens, spontaneously recognized by tumor-infiltrating lymphocytes. J Immunol. 2003;171:6283–6289. doi: 10.4049/jimmunol.171.11.6283. [DOI] [PubMed] [Google Scholar]
- 3.Bodinier M, Peyrat MA, Tournay C, Davodeau F, Romagne F, Bonneville M, Lang F. Efficient detection and immunomagnetic sorting of specific T cells using multimers of MHC class I and peptide with reduced CD8 binding. Nat Med. 2000;6:707–710. doi: 10.1038/76292. [DOI] [PubMed] [Google Scholar]
- 4.Brichard V, Van Pel A, Wolfel T, Wolfel C, De Plaen E, Lethe B, Coulie P, Boon T. The tyrosinase gene codes for an antigen recognized by autologous cytolytic T lymphocytes on HLA-A2 melanomas. J Exp Med. 1993;178:489–495. doi: 10.1084/jem.178.2.489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Coulie PG, Brichard V, Van Pel A, Wolfel T, Schneider J, Traversari C, Mattei S, De Plaen E, Lurquin C, Szikora JP, Renauld JC, Boon T. A new gene coding for a differentiation antigen recognized by autologous cytolytic T lymphocytes on HLA-A2 melanomas. J Exp Med. 1994;180:35–42. doi: 10.1084/jem.180.1.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.De Plaen E, Lurquin C, Lethe B, van der Bruggen P, Brichard V, Renauld JC, Coulie P, Van Pel A, Boon T. Identification of genes coding for tumor antigens recognized by cytolytic T lymphocytes. Methods. 1997;12:125–142. doi: 10.1006/meth.1997.0462. [DOI] [PubMed] [Google Scholar]
- 7.Dreno B, Nguyen JM, Khammari A, Pandolfino MC, Tessier MH, Bercegeay S, Cassidanius A, Lemarre P, Billaudel S, Labarriere N, Jotereau F. Randomized trial of adoptive transfer of melanoma tumor-infiltrating lymphocytes as adjuvant therapy for stage III melanoma. Cancer Immunol Immunother. 2002;51:539–546. doi: 10.1007/s00262-002-0315-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dudley ME, Wunderlich JR, Robbins PF, Yang JC, Hwu P, Schwartzentruber DJ, Topalian SL, Sherry R, Restifo NP, Hubicki AM, Robinson MR, Raffeld M, Duray P, Seipp CA, Rogers-Freezer L, Morton KE, Mavroukakis SA, White DE, Rosenberg SA. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science. 2002;298:850–854. doi: 10.1126/science.1076514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Espevik T, Nissen-Meyer J. A highly sensitive cell line, WEHI 164 clone 13, for measuring cytotoxic factor/tumor necrosis factor from human monocytes. J Immunol Methods. 1986;95:99–105. doi: 10.1016/0022-1759(86)90322-4. [DOI] [PubMed] [Google Scholar]
- 10.Godefroy E, Moreau-Aubry A, Diez E, Dreno B, Jotereau F, Guilloux Y. alpha v beta3-dependent cross-presentation of matrix metalloproteinase-2 by melanoma cells gives rise to a new tumor antigen. J Exp Med. 2005;202:61–72. doi: 10.1084/jem.20042138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Granziero L, Krajewski S, Farness P, Yuan L, Courtney MK, Jackson MR, Peterson PA, Vitiello A. Adoptive immunotherapy prevents prostate cancer in a transgenic animal model. Eur J Immunol. 1999;29:1127–1138. doi: 10.1002/(SICI)1521-4141(199904)29:04<1127::AID-IMMU1127>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 12.Greenberg PD. Adoptive T cell therapy of tumors: mechanisms operative in the recognition and elimination of tumor cells. Adv Immunol. 1991;49:281–355. doi: 10.1016/S0065-2776(08)60778-6. [DOI] [PubMed] [Google Scholar]
- 13.Guilloux Y, Lucas S, Brichard VG, Van Pel A, Viret C, De Plaen E, Brasseur F, Lethe B, Jotereau F, Boon T. A peptide recognized by human cytolytic T lymphocytes on HLA-A2 melanomas is encoded by an intron sequence of the N-acetylglucosaminyltransferase V gene. J Exp Med. 1996;183:1173–1183. doi: 10.1084/jem.183.3.1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hanson HL, Donermeyer DL, Ikeda H, White JM, Shankaran V, Old LJ, Shiku H, Schreiber RD, Allen PM. Eradication of established tumors by CD8+ T cell adoptive immunotherapy. Immunity. 2000;13:265–276. doi: 10.1016/S1074-7613(00)00026-1. [DOI] [PubMed] [Google Scholar]
- 15.Heslop HE, Ng CY, Li C, Smith CA, Loftin SK, Krance RA, Brenner MK, Rooney CM. Long-term restoration of immunity against Epstein-Barr virus infection by adoptive transfer of gene-modified virus-specific T lymphocytes. Nat Med. 1996;2:551–555. doi: 10.1038/nm0596-551. [DOI] [PubMed] [Google Scholar]
- 16.Jotereau F, Pandolfino MC, Boudart D, Diez E, Dreno B, Douillard JY, Muller JY, LeMevel B. High-fold expansion of human cytotoxic T-lymphocytes specific for autologous melanoma cells for use in immunotherapy. J Immunother. 1991;10:405–411. doi: 10.1097/00002371-199112000-00003. [DOI] [PubMed] [Google Scholar]
- 17.Jung T, Schauer U, Heusser C, Neumann C, Rieger C. Detection of intracellular cytokines by flow cytometry. J Immunol Methods. 1993;159:197–207. doi: 10.1016/0022-1759(93)90158-4. [DOI] [PubMed] [Google Scholar]
- 18.Kawakami Y, Dang N, Wang X, Tupesis J, Robbins PF, Wang RF, Wunderlich JR, Yannelli JR, Rosenberg SA. Recognition of shared melanoma antigens in association with major HLA-A alleles by tumor infiltrating T lymphocytes from 123 patients with melanoma. J Immunother. 2000;23:17–27. doi: 10.1097/00002371-200001000-00004. [DOI] [PubMed] [Google Scholar]
- 19.Kawakami Y, Eliyahu S, Jennings C, Sakaguchi K, Kang X, Southwood S, Robbins PF, Sette A, Appella E, Rosenberg SA. Recognition of multiple epitopes in the human melanoma antigen gp100 by tumor-infiltrating T lymphocytes associated with in vivo tumor regression. J Immunol. 1995;154:3961–3968. [PubMed] [Google Scholar]
- 20.Labarriere N, Pandolfino MC, Gervois N, Khammari A, Tessier MH, Dreno B, Jotereau F. Therapeutic efficacy of melanoma-reactive TIL injected in stage III melanoma patients. Cancer Immunol Immunother. 2002;51:532–538. doi: 10.1007/s00262-002-0313-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lennerz V, Fatho M, Gentilini C, Frye RA, Lifke A, Ferel D, Wolfel C, Huber C, Wolfel T. The response of autologous T cells to a human melanoma is dominated by mutated neoantigens. Proc Natl Acad Sci USA. 2005;102:6013–6018. doi: 10.1073/pnas.0500090102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Linard B, Bezieau S, Benlalam H, Labarriere N, Guilloux Y, Diez E, Jotereau F. A ras-mutated peptide targeted by CTL infiltrating a human melanoma lesion. J Immunol. 2002;168:4802–4808. doi: 10.4049/jimmunol.168.9.4802. [DOI] [PubMed] [Google Scholar]
- 23.Meidenbauer N, Marienhagen J, Laumer M, Vogl S, Heymann J, Andreesen R, Mackensen A. Survival and tumor localization of adoptively transferred Melan-A-specific T cells in melanoma patients. J Immunol. 2003;170:2161–2169. doi: 10.4049/jimmunol.170.4.2161. [DOI] [PubMed] [Google Scholar]
- 24.Mitsuma S, Yoshizawa H, Ito K, Moriyama H, Wakabayashi M, Chou T, Arakawa M, Shu S. Adoptive immunotherapy mediated by anti-TCR/IL-2-activated tumour-draining lymph node cells. Immunology. 1994;83:45–51. [PMC free article] [PubMed] [Google Scholar]
- 25.Moreau-Aubry A, Le Guiner S, Labarriere N, Gesnel MC, Jotereau F, Breathnach R. A processed pseudogene codes for a new antigen recognized by a CD8(+) T cell clone on melanoma. J Exp Med. 2000;191:1617–1624. doi: 10.1084/jem.191.9.1617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Novellino L, Castelli C, Parmiani G. A listing of human tumor antigens recognized by T cells: March 2004 update. Cancer Immunol Immunother. 2005;54:187–207. doi: 10.1007/s00262-004-0560-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pandolfino MC, Labarriere N, Tessier MH, Cassidanius A, Bercegeay S, Lemarre P, Dehaut F, Dreno B, Jotereau F. High-scale expansion of melanoma-reactive TIL by a polyclonal stimulus: predictability and relation with disease advancement. Cancer Immunol Immunother. 2001;50:134–140. doi: 10.1007/PL00006683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Robbins PF, Dudley ME, Wunderlich J, El-Gamil M, Li YF, Zhou J, Huang J, Powell DJ, Jr, Rosenberg SA. Cutting edge: persistence of transferred lymphocyte clonotypes correlates with cancer regression in patients receiving cell transfer therapy. J Immunol. 2004;173:7125–7130. doi: 10.4049/jimmunol.173.12.7125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rosenberg SA, Spiess P, Lafreniere R. A new approach to the adoptive immunotherapy of cancer with tumor-infiltrating lymphocytes. Science. 1986;233:1318–1321. doi: 10.1126/science.3489291. [DOI] [PubMed] [Google Scholar]
- 30.Roskrow MA, Suzuki N, Gan Y, Sixbey JW, Ng CY, Kimbrough S, Hudson M, Brenner MK, Heslop HE, Rooney CM. Epstein-Barr virus (EBV)-specific cytotoxic T lymphocytes for the treatment of patients with EBV-positive relapsed Hodgkin’s disease. Blood. 1998;91:2925–2934. [PubMed] [Google Scholar]
- 31.Valmori D, Fonteneau JF, Lizana CM, Gervois N, Lienard D, Rimoldi D, Jongeneel V, Jotereau F, Cerottini JC, Romero P. Enhanced generation of specific tumor-reactive CTL in vitro by selected Melan-A/MART-1 immunodominant peptide analogues. J Immunol. 1998;160:1750–1758. [PubMed] [Google Scholar]
- 32.Vignard V, Lemercier B, Lim A, Pandolfino MC, Guilloux Y, Khammari A, Rabu C, Echasserieau K, Lang F, Gougeon ML, Dreno B, Jotereau F, Labarriere N. Adoptive transfer of tumor-reactive Melan-A-specific CTL clones in melanoma patients is followed by increased frequencies of additional Melan-A-specific T cells. J Immunol. 2005;175:4797–4805. doi: 10.4049/jimmunol.175.7.4797. [DOI] [PubMed] [Google Scholar]
- 33.Yee C, Thompson JA, Byrd D, Riddell SR, Roche P, Celis E, Greenberg PD. Adoptive T cell therapy using antigen-specific CD8+ T cell clones for the treatment of patients with metastatic melanoma: in vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci USA. 2002;99:16168–16173. doi: 10.1073/pnas.242600099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhou J, Shen X, Huang J, Hodes RJ, Rosenberg SA, Robbins PF. Telomere length of transferred lymphocytes correlates with in vivo persistence and tumor regression in melanoma patients receiving cell transfer therapy. J Immunol. 2005;175:7046–7052. doi: 10.4049/jimmunol.175.10.7046. [DOI] [PMC free article] [PubMed] [Google Scholar]