Abstract
Anti-tumor vaccines capable of activating both CD4 and CD8 T cells are preferred for long lasting T cell responses. Induction of a tumor-specific T-cell response can be induced by tumor vaccines that target innate immunity. The ensuing T-cell response depends on efficient antigen presentation from phagocytosed cargo in the antigen presenting cell and is augmented by the presence of Toll-like receptor (TLR) ligands within the cargo. Biodegradable polymers are useful for vaccine delivery in that they are phagocytosed by antigen presenting cells (APCs) and could potentially be loaded with both the antigen and immune stimulatory TLR agents. This study was undertaken to evaluate the effect of poly lactic-co-glycolic acid (PLGA) polymer particles loaded with antigenic tumor lysate and immune stimulatory CpG oligonucleotides on induction of tumor specific immunity in a mouse model of melanoma. We found that after delivery, these immune stimulatory antigen loaded particles (ISAPs) efficiently activated APCs and were incorporated into lysosomal compartments of macrophages and dendritic cells. ISAP vaccination resulted in remarkable T cell proliferation, but only modestly suppressed tumor growth of established melanoma. Due to this discordant effect on tumor immunity we evaluated the role of regulatory T cells (Treg) and found that ISAP vaccination or tumor growth alone induced prolific expansion of tumor specific Treg. When the Treg compartment was suppressed with anti-CD25 antibody, ISAP vaccination induced complete antigen-specific immunity in a prophylactic model. ISAP vaccination is a novel tumor vaccine strategy that is designed to co-load the antigen with a TLR agonist enabling efficient Ag presentation. Targeting of T-reg expansion during vaccination may be necessary for inducing effective tumor-specific immunity.
Keywords: Immune stimulatory DNA, CpG oligodeoxynucleotide, Poly lactic co-glycolic acid polymers, PLGA, Immunotherapy, Tumor lysate, Melanoma, Tumor vaccine
Introduction
Antigen presentation by innate immune cells, most notably macrophages and dendritic cells (DC), is critical for induction of tumor-specific T cells in the form of an adaptive immune response [1]. Macrophages and DCs are dependent on specific signals that enable their transformation into potent immunostimulatory antigen presenting cells (APCs) [2]. Immune stimulatory danger signals can be exploited to shape an adaptive tumor specific immune response through stimulation of the Toll-like receptor (TLR). The TLR gene family is an evolutionarily conserved receptor system essential for host defense against infectious pathogens [3].
Vertebrates have evolved innate immune defense mechanisms that recognize and respond to microbial molecules through these TLRs. Included in this pattern recognition system are unmethylated CpG dinucleotides in particular sequence contexts that are common to bacterial DNA, but are underrepresented (“CpG suppression”) and methylated in vertebrate DNA [4]. Specifically, TLR9 is essential for recognition of CpG DNA, and both human and mouse DCs express TLR9. CpG DNA activate protein kinase cascades through TLR9, up-regulating co-stimulatory molecules and inducing Th1-polarizing cytokines [5]. These cytokine-priming events make CpG ODN particularly useful adjuvants for vaccine therapy.
We previously showed that the combination of CpG ODN and GM-CSF more potently induced immunity in a therapeutic tumor vaccine strategy than either immune stimulant alone [6]. This synergy was mediated through the APC in which GM-CSF enhanced antigen up-take and CpG induced activation and migration of the APCs. In these previous studies the TLR agonist was admixed with irradiated whole tumor cells and GM-CSF for vaccination. It was recently shown that the efficiency of presenting antigens from phagocytosed cargo is dependent on the presence of TLR ligands within the cargo [7]. Biodegradable polymers based on the poly alpha hydroxy acids are ideal for use in controlled drug delivery of which the poly lactic-co-glycolic acid (PLGA) polymers are the most commonly used [8]. These polymers have a broad range of pharmaceutical and biomedical applications based on their versatile degradation kinetics, lack of toxicity and biocompatibility. Degradable particles between 1 and 10 µm in size are preferentially phagocytosed by APCs and macrophages [9]. This enhanced uptake should negate the need for GM-CSF in a tumor vaccine strategy, but more importantly will present the antigen and the TLR ligand within the same phagocytosed microparticle. In the present study, we designed microparticles that we termed immune stimulatory antigen loaded particles (ISAPs) for the purpose of tumor vaccination. These particles were fabricated from PLGA and co-loaded with antigen in the form of irradiated tumor lysate and immune stimulatory CpG ODN.
Regulatory T-cells (T-reg) are defined by their expression of CD4, CD25 and FoxP3 and are considered immuno-suppressive in their innate ability to inhibit both CD4 and CD8 T-cells. T-reg may inhibit anti-tumor T cell responses and could be a major obstacle to effective tumor vaccination and immunotherapy. However, there is significant controversy as to whether TLR ligands enhance the effect of T-regs or render them ineffective. The effect of our TLR based tumor vaccine on T-reg was also evaluated in this study.
We found that ISAPs were efficiently taken up by APCs, localizing to the lysosome and inducing remarkable T-cell proliferation without the need for GM-CSF synergy. Vaccination with the ISAPs modestly suppressed tumor growth in an established tumor model in which we found that the tumor itself and the ISAP vaccine induced prolific T-reg (CD4+; CD25+; FoxP3+) expansion. Broad targeting of Tregs with low dose cyclophosphamide (CY) or anti-CD25 antibody suppressed the number of Treg cells. The anti-CD25 antibody ensured prophylactic immunity with the ISAP vaccination, but did not effect tumor growth in an established tumor model. ISAP vaccination appears to be an effective and novel tumor vaccine approach by virtue of its macrophage/APC targeting, lysosome compartmentalization and vigorous induction of T-cell proliferation. However, specific targeting of T-reg cells during tumor vaccination is needed to ensure protective immunity.
Materials and methods
Mice and murine cell lines
Female C57BL/6 mice were purchased at 6–8 weeks of age from Harlan Teklad (Indianapolis, IN, USA). On arrival, the animals were acclimated for 1 week prior to tumor or vaccine challenge. B16 (F1) (American Type Culture Collection, Manassas, NJ, USA) is a murine melanoma derived from C57BL/6 mice and was cultured in RPMI1640 containing 10% fetal calf serum, 0.5% penicillin/streptomycin, 10% l-glutamine, 10% nonessential amino acid and 10% pyruvate. RAW 264.7 cells (American Type Culture Collection) are a murine macrophage cell line and were tested in vitro for up-take of microparticles and stained for activation markers.
Anesthetic agents and animal care
Mice were anesthetized using Halothane inhalation (Halocarbon Lab., NJ, USA) during inoculation of tumor cells or vaccination. All animals were housed under standard conditions in accordance with our institution’s animal care and use committee, which follows the US Public Health Service’s guide for the care and use of animals.
Tumor model and vaccination
WT B16 (F1) cells (5 × 105 cells) were injected subcutaneously into the hind leg of syngeneic C57BL/6 mice for both therapeutic or prophylactic models. Vaccination was administered intra-peritoneally with either a whole tumor cell vaccine or ISAPs. Tumor cells for vaccination were irradiated with 20 Gy from a cesium source. The timing and components of the vaccine protocols varied with the experiments and are specifically described for each study in the results section. Tumor growth and mouse survival were monitored and compared between groups.
Reagents and antibodies
CpG ODN 1826 with a nuclease resistant phosphorothioate backbone and no detectable endotoxin was purchased from Coley Pharmaceutical Group (Wellesley, MA, USA). The sequence of CpG 1826 is: 5′-TCCATGACGTTCCTGACGTT-3′. CFSE (5(6)-Carboxyfluorescein diacetate N-succinimidyl ester) was purchased from Sigma–Aldrich (St Louis, MO, USA). All monoclonal antibodies; including PE-CD11C, PE-CD11b, FITC-CD86, FITC-CD80, FITC-CD40, FITC-CD54 and FITC-MHCII, FoxP3, CD4, CD8 and CD25 were purchased from either BD Pharmingen (SanDiego, CA, USA) or e-Bioscience (San Diego, CA, USA). The anti-CD25 antibody (clone PC61) used for in vivo studies was obtained from BD PharMingen. A single does of 500 µg was administered intra-peritoneally a day prior to mouse vaccination.
Tumor lysate preparation
Tumor cells were suspended in PBS. The cell suspension was frozen in liquid nitrogen for 1.5 min and then thawed in a 37°C water bath for 4 min. The freeze–thaw cycle was repeated three times in rapid succession; cells were irradiated at 10,000 cGy and stored in liquid nitrogen for later use. To determine concentration of lysate for reproducible loading in PLGA microparticles, the tumor lysate concentration was determined using the BCA assay (Pierce, Rockford, IL, USA).
Fabrication of immune stimulatory antigen loaded particles (ISAPs)
Preparation of microparticles loaded with CpG and tumor lysate: PLGA microparticles were prepared using a double emulsion solvent evaporation methodology. Briefly, 200 mg of 75:25 PLGA (MW = 56,500) was dissolved in 2 ml dichloromethane (DCM). A solution with tumor lysate at a concentration of 100 μg/μl and CpG ODN at 50 μg/μl was prepared in 1% (w/v) PVA (87–89% hydrolyzed, Avg MW = 13,000–23,000; Sigma) in deionized water. Using a microtip probe sonicator set at level 2 (Sonic Dismembrator Model 100, Fisher Scientific, Pittsburgh, PA, USA), 100 μl of the lysate/oligonucleotide solution containing 10 mg tumor lysate and 5 mg CpG was mixed with the PLGA/DCM solution for 20 s (10 s pulses) to form the first emulsion. This emulsion was then rapidly added to 100 ml of 1% (w/v) PVA in deionized water with stirring at 6,400 rpm. The mixture was stirred overnight during which time the DCM solvent evaporated. The microparticles were then washed six times with 50 ml of sterile filtered distilled/deionized water and then lyophilized (Labconco freezone 4.5, Kansas City, MI, USA). PLGA microparticles loaded with Rhodamine 123 were prepared using a single emulsion evaporation technique. Briefly, 200 mg of 75:25 PLGA (MW = 56,500) and 10 mg Rhodamine 123 (Sigma) was dissolved in 2 ml dichloromethane (DCM). This was then rapidly added to 100 ml of 1% (w/v) PVA in deionized water and stirred at 6,400 rpm. The rhodamine labeled particles were washed and collected as described above.
Characterization of ISAPs
Microparticle size and surface morphology analysis
Microparticle size analysis was conducted using the Zetasizer Nano ZS (Malvern, Southborough, MA, USA). Microparticle morphology was assessed by scanning electron microscopy (SEM, Hitachi S-4000). Briefly, air-dried microparticles were placed on adhesive carbon tabs and mounted on SEM specimen stubs. The specimen stubs were coated with approximately 5 nm of gold by ion beam evaporation prior to examination in the SEM operated at 5 kV accelerating voltage.
CpG ODN and tumor lysate loading estimation
Tumor lysate loading in PLGA microparticles was determined by dissolving 200 mg of lyophilized microparticles in 2 ml of acetonitrile. The suspension of dissolved polymer and precipitated tumor lysate was centrifuged at 6,000g for 7 min and the polymer containing supernatant was then discarded. The pellet consisting of the precipitated lysate was redispersed in 0.1 N NaOH. The protein content of the neutralized alkaline solution was estimated using a microBCA protein assay kit (Pierce, Rockford, IL, USA). Empty microparticles in acetonitrile were spiked with a known amount of a antigenic tumor lysate. Following the procedure, recovery of the extracted tumor lysate was found to be complete. Microparticles containing CpG ODN were also dissolved in acetonitrile and the CpG ODN was extracted in TE buffer (10 mM, pH 8.3). The amount of CpG ODN was determined based on absorbance at 260 nm. Empty microparticles in acetonitrile were spiked with a known amount of CpG ODN. Following the procedure, recovery of the extracted CpG ODN was found to be complete.
In vitro release of CpG ODN and tumor lysate
PLGA microparticles containing tumor lysate were suspended in phosphate buffered saline (PBS, 10 ml, 50 mM, pH 7.4) for 2 months. The suspension was gently shaken in a water bath at 37°C. At various time intervals, the supernatant was removed after centrifugation and replaced with fresh medium. Tumor lysate released into the supernatant was quantified using the micro-BCA protein assay. CpG ODN containing microparticles were incubated in TE buffer and the released fraction estimated spectrophotometrically at 260 nm.
Spleen cell preparation
Spleens were removed, pooled and cultured in RPMI-1640 medium containing 10% fetal calf serum. Pooled spleens were then made into single cell suspension with sterile slides. The red blood cells were lysed with ACK solution. The cells were then washed and re-suspended in medium.
Primay macrophage harvest and culture
Murine bone marrow derived macrophages (BMMφ) were established as previously described [10]. Briefly, bone marrow was flushed from the femurs and tibias of mice, using a 23 gauge needle, and collected by centrifugation. Bone marrow cells were suspended in Dulbecco’s Modification of Eagle’s Medium (DMEM, Mediatech) supplemented with 10% HI-FCS, 2 mM glutamine, 100 μg/mL streptomycin and 100 U/ml penicillin G. Cells were then plated in petri dishes, and supplemented with 20% conditioned media from the supernatants of m-CSF (macrophage colony stimulating factor) secreting L-929 fibroblasts. Cells were incubated at 37°C with 5% CO2.
Flow cytometry analysis
To obtain appropriate lymphocyte samples, mice were killed and splenectomized for fresh splenocytes. Where designated, splenocytes were cultured as described above. Splenocytes were incubated with unlabeled primary rabbit mAb of interest for 1 h at 4°C. (Markers of interest include: CD4, CD8, Fox P3 for T-cells and CD11b, CD11c, CD80, CD40, CD54 and MHCII for APCs. All antibodies were purchased from BD Biosciences. Data were analyzed using CellQuestPro software and samples were analyzed using a FACScalibur™ flow cytometer. Quadrant markers were set according to isotype control antibodies.
ELISA
Levels of murine secreted IL-12p40 and TNF protein concentrations in cell supernatants were determined by sandwich ELISA according to protocols provided by eBioscience (San Diego, CA, USA), and utilizing the following antibody pairs: IL-12p40, C15.6 and C17.8; TNF, 1F3F3D4 and XT3/XT22.
Enrichment of CD11c + Splenocytes
Freshly isolated splenocytes were washed with buffer (PBS; 0.5% BSA) and incubated with mouse immunoglobulin to block Fc receptor. The cells were then incubated with either CD11c MicroBeads (Miltenyi Biotec, Auburn, CA, USA) for 15 min at 6–12°C. Cells were then washed, re-suspended and separated with autoMACS (Miltenyi Biotec).
Intracellular staining and flow cytometry analysis
Fresh or cultured cells were labeled with fluorochrome-conjugated mAbs specific for different surface markers for 20 min at 4°C. After binding and washing, cells were fixed with formalin in PBS for flow cytometry analysis. For intracellular staining, cells were first stained with surface markers, and then fixed and permeabilized with fixation and permeabilization buffers, respectively. Cells were then stained with PE-conjugated IFN-γ for 20 min at room temperature in the dark. After washing away excessive antibodies with permeabilization buffer, cells were resuspended in staining buffer for flow cytometry analysis. Data was analyzed using CellQuestPro software. Quadrant markers were set according to isotype control antibodies.
T-cell CFSE proliferation assay
Splenocytes (1 × 107/ml in PBS) were incubated with CFSE (5(6)-Carboxy fluorescein diacetate N-succinimidyl ester) at a concentration of 2 µM at room temperature for 10 min. Stained cells were cultured in 12-well tissue culture plates with or without Ag of choice. After 7 days of incubation, cells were harvested and stained with anti-CD4 and anti-CD8 mAb, and then subjected to flow cytometry analysis.
Results
PLGA particles loaded with tumor lysate and CpG ODN demonstrate burst followed by sustained release profiles of antigen and CpG
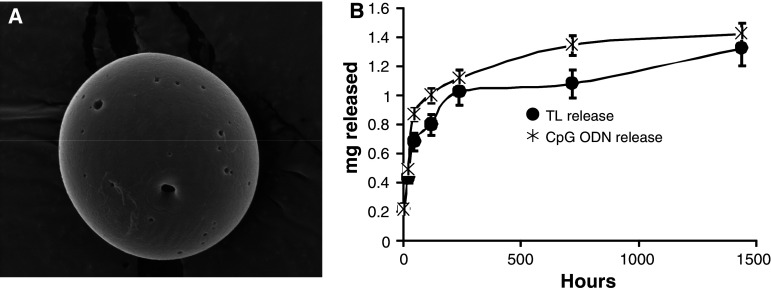
Scanning electron microscopy analysis showed that particles had a smooth morphology and spherical shape (Fig. 1a). Microparticles were an average diameter of 2.4 μm ± 0.43. Entrapment efficiencies of tumor lysate ranged from 17 to 24% with an average loading of 11 μg/mg of microparticles. For CpG ODN, these values were 31–33%, which corresponded to an average loading of 8.3 μg/mg of microparticles. Release profiles of CpG and tumor lysate were measured using UV spectrophotometry at 260/280 nm and the micro-BCA protein assay, respectively. Release profiles of both CpG and tumor lysate showed a burst release profile followed by a more sustained release. (Fig. 1b) We have termed these particles loaded with both immune stimulatory sequences and antigen—Immune Stimulatory Antigen-loaded Particles or ISAPs.
Fig. 1.
Microparticle preparation and release profiles: a Microparticles were prepared from poly lactic and glycolic acid (PLGA) using the water-in-oil-in-water double emulsion solvent evaporation method. Scanning electron microscopy analysis showed that particles had a smooth morphology and spherical shape. b Release profiles of CpG and tumor lysaste were measured using UV spectrophotometry at 260/280 nm and the micro-BCA protein assay, respectively. Release profiles of both CpG and tumor lysate showed a burst followed by a more sustained release profile
ISAPs localize to the lysosomes and induce activation and cytokine secretion in RAW cells and primary macrophages in vitro
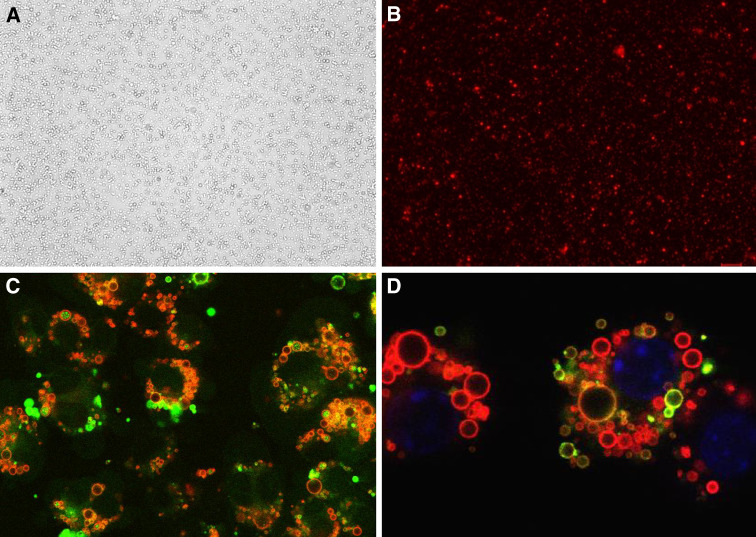
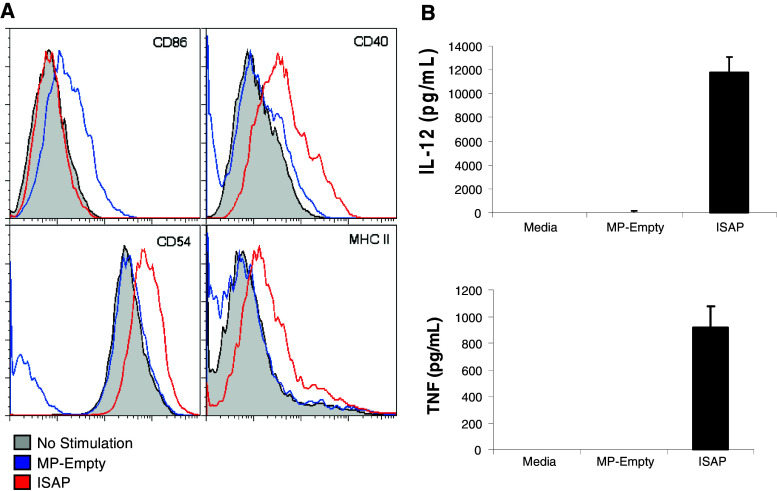
Lysosomes are essential for processing the antigens through the major histocompatability II (MHCII) pathway for helper CD4 T cell activation. We therefore sought to define the localization of the phagocytosed particles in vitro as a means to understanding the uptake and processing of the ISAPs. RAW264.7 cells were treated with microparicles loaded with a model antigen-ovalbumin covalently attached to flouroscein and subsequently counterstained with fluorescent dyes specifically targeting the nucleus and lysosome. Confocal fluorescent microscopy revealed clear localization of the microparticles within the lysosomes of the macrophages (Fig. 2a–d). We treated RAW264.7 mouse macrophage cells with various concentrations of rhodamine loaded particles to test their ability to engulf the particles in vitro. A dose dependent increase in rhodamine uptake by the RAW cells was observed with increased concentration of ISAPs (Data not shown). Similarly, surface markers of activation on RAW cells or primary macrophages (CD40, CD54 and MHCII) were up-regulated following ISAP treatment suggesting effective immune stimulation by the particles. (Fig. 3a, b). ISAP ingestion by primary mouse macrophages induced robust secretion of Th1 cytokines IL-12 and TNF-α, but not IL-10, while empty microparticles or microparticles loaded with lysate only (not shown), failed to induce stimulation.
Fig. 2.
Light microscopy of a, empty microparticles and fluorescent microscopy of b microparticles loaded with rhodamine. Confocal fluorescent microscopy of RAW264.7 cells treated with microparticles loaded with ovalbumin bound to flouroscein (green) and subsequently counterstained with fluorescent dyes specifically targeting, c the lysosome (red) and d nucleus (blue) and lysosome (red). Confocal microscopy revealed localization of the microparticles within the lysosomes of the macrophages (color available only in online publication)
Fig. 3.
Macrophage stimulation by ISAPs. Primary mouse bone-marrow derived macrophages were treated with various components of the microparticles (MP) and their effects on a surface marker expression and b secretion of IL-12 p40 and TNF-α were compared. These studies demonstrate the potent immuno-stimulatory effect of ISAPs on primary macrophages. The immuno-suppressive cytokine IL-10 was not detected in supernatants from any of the groups (data not shown). MP empty microparticle only, MP-CpG MP containing CpG only, MP-B16 MP containing B16 tumor lysate only and ISAP: MP containing CpG and B16 tumor lysate. Studies were performed in triplicate and repeated three times with similar results
Microparticles administered intra-peritoneally are taken up by DC’s and migrate to the spleen in vivo
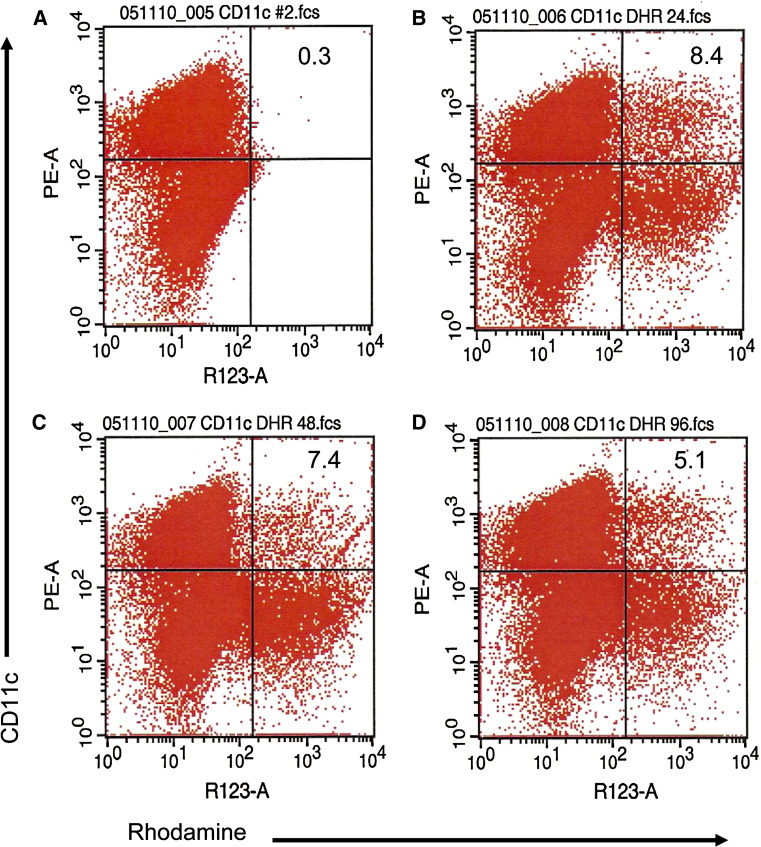
As described above, ISAPs were readily phagocytosed by macrophages in vitro, thus we proceeded to investigate whether ISAPs were also effectively taken up by APCs in vivo. For this purpose, PLGA microparticles (MP) alone or loaded with Rhodamine were administered IP into mice. Mice were sacrificed at 1, 4 and 7 days following their administration and splenocytes were harvested for flow cytometry. Figure 4 shows that rhodamine containing MP were taken up by CD11c + DC (right upper quadrant) and transported to the spleen. The MP were slowly cleared, but were present in the spleen for at least 7 days following administration. The right lower quadrant represents CD11c-cells (macrophages) that also took up the rhodamine positive MP (Fig. 4a–d). These observations suggest that ISAPs are taken up by APCs after IP vaccination and migrate to the spleen.
Fig. 4.
Uptake of microparticles by splenic DCs. Empty PLGA microparticles (MP) or MP loaded with Rhodamine were administered IP into mice. Mice were sacrificed at 1, 3 and 7 days following their administration and splenocytes were harvested for flow cytometry and counter stained for CD11c. a Control empty microparticles, b 1 day, c 3 days and d 4 days following administration of rhodamine loaded MPs. The experiments show that IP administered MPs are taken up and found in splenic DCs following administration for at least 4 days with the highest concentration on day 1. Percentages noted are % of CD11c + (DC) splenocytes. The right lower quadrant probably represents CD11c-macrophages (and possible but unlikely B-cells) that also took up the rhodamine positive MPs. Although B-cells also phagocytose particles, the particle size of the ISAP is predictably too large for B-cells to ingest
ISAP vaccination induces remarkable expansion and proliferation of tumor specific T-cell:
To determine whether the ISAPs identified in splenic APCs are processed for adaptive immunity, splenocytes from mice vaccinated with empty microparticles or ISAPs were harvested 10 days after vaccination and subjected to flow cytometry analysis to determine the percentage of CD4 or CD8 cells present. The CD4 and CD8 cell numbers were remarkably increased as a percentage of total splenocytes following ISAP vaccination (0.62–2.56% for CD4+ cells and 0.11–0.22% for CD8+ cells). Furthermore, the relative numbers increased substantially following co-culture with irradiated whole tumor cells as antigen (9.21–18.47% for CD4+ cells and 0.83–11.6% for CD8+ cells) or tumor lysate (0.56–6.93% for CD4+ cells and 0.05–1.43% for CD8+ cells). (Empty microparticle vs. ISAP vaccinated splenocytes).
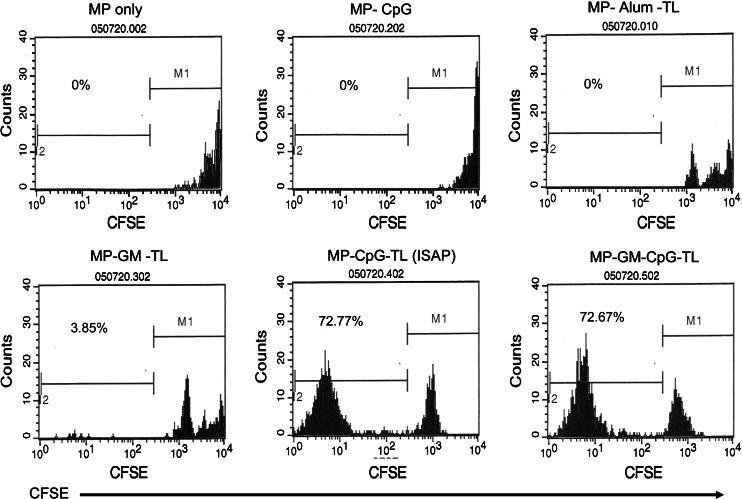
To evaluate proliferation, splenocytes harvested from naïve mice, mice vaccinated with microparticles only or mice vaccinated with various components of the immune stimulatory antigen loaded particles were re-stimulated with tumor lysate and incubated with CFSE. After 5 and 7 day incubations, cells were harvested and stained with anti-CD4 and anti-CD8 mAb, and then subjected to flow cytometry analysis. Proliferating cells are defined by the dilutional effect of CFSE staining in the T-cells. We found that mouse vaccination with the ISAP microparticles induced significant proliferation of CD8 T-cells when both CpG and tumor lysate (TL) were loaded (Fig. 5). Seventy-three percent of the CD8 T-cells proliferated in culture as determined by this assay. It is of interest to note that co-loading the tumor antigens in the form of lysate with the CpG ODN in the microparticles is critical for the induction of T-cell proliferation, while either one alone was inadequate. The addition of GM-CSF does not appear essential for induction of T-cell proliferation, underscoring the concept of microparticles being efficiently taken up by APC without GM-CSF priming (Fig. 5). Similar findings were noted when the CD4+ T cell compartment was gated, but the relative intensity of proliferation was not as great as that of CD8 T-cells.
Fig. 5.
T-cell proliferation following ISAP vaccination. Splenocytes harvested from naïve mice, mice vaccinated with microparticles only or mice vaccinated with various components of the immune stimulatory antigen loaded particles were re-stimulated with tumor lysate and incubated with CFSE. After 7 days of incubation, cells were harvested and stained with anti-CD8 Ab, and then subjected to flow cytometry analysis. Proliferating cells are defined by the dilutional effect of CFSE staining in the T-cells. ISAP vaccination induced significant proliferation of CD8 T-cells when both CpG and tumor lysate (TL) were loaded. (MP empty microparticle only, MP-CpG MP containing CpG only, MP-Alum-TL MP containing alum and B16 tumor lysate, MP-GM-TL MP containing GM-CSF and B16 tumor lysate, MP-CpG-TL (ISAP) MP containing CpG and B16 tumor lysate and MP-GM-CpG-TL MP containing GM-CSF plus CpG plus tumor lysate
ISAPs induce an appropriate cytokine response
FACSarray was used to simultaneously measure cytokine responses following ISAP vaccination from harvested splenocytes. Splenocytes from various groups of vaccinated mice were cultured alone or with tumor lysate and secreted cytokines were measured in supernatants. IFN-γ (Th-1) was appropriately secreted by the vaccinated splenocytes whereas inhibitory cytokines like IL-10 or other Th-2 type cytokine responses like IL-5 were not secreted by the splenocytes following re-stimulation with tumor lysate. Furthermore, the combination of CpG and GM-CSF in the vaccine had no synergistic effect in IFN-γ cytokine secretion over CpG alone.
ISAP vaccination failed to cure mice with established disease
The above data show vigorous induction of CD4 and CD8 tumor specific T-cell expansion following ISAP vaccination, thus we tested this approach in the therapy of established melanoma. Studies in an established stringent melanoma model show that vaccination with ISAPs loaded with tumor lysate and CpG (0.162 ± 0.03 cm3) appeared to suppress tumor growth at day 15 following challenge (P = 0.053 by analysis of variance), when compared to control (0.71 ± 0.37 cm3) or either vaccination with irradiated autologous tumor cells admixed with CpG ODN (0.62 ± 0.34 cm3) or microparticles loaded with CpG only and admixed with autologous irradiated tumor cells (0.68 ± 0.22 cm3). Although tumor growth was inhibited, none of the mice experienced long-term survival and all died of disease or were sacrificed when tumor was greater than 2 cm. Despite the vaccines ability to induce remarkable and robust T-cell expansion, it paradoxically failed to induce comparable tumor immunity, thus we investigated whether induction of Treg cells could partly explain this lack of effect in the established disease model.
Tumor growth alone or ISAP vaccination both independently induce tumor specific expansion of the Treg compartment ex vivo when cultured with antigen
To evaluate the role or Treg in the lack of vaccine efficacy, mice were inoculated with B16 melanoma (1 × 106) and when tumors were either 9 or 18 mm in a single dimension, mice were sacrificed and spleens were harvested. The compartment of CD4 + CD25 + Foxp3 + Treg cells was determined by flow cytometry both immediately and following 5 days of splenocyte culture with or without irradiated tumor cells. The percentage of Treg cells of total splenocytes or of CD4 + gated cells at harvest were similar or decreased in mice bearing large tumors, but following culture the Treg compartment proliferated expansively and increased as the tumor size grew bigger (Fig. 6a). The Treg expansion observed was tumor specific as it only occurred when the splenocytes were cultured with the B16 tumor antigen and not with other unrelated tumor antigens (Fig. 6b).
Fig. 6.
Tumor specific expansion of Treg cells: mice were inoculated with B16 melanoma (1 × 106) and when tumors were either 9 or 18 mm in a single dimension, mice were sacrificed and spleens were harvested. The compartment of CD4 + CD25 + Foxp3 + Treg cells was determined by flow cytometry following 5 days of splenocyte culture with irradiated tumor cells. The percentage of Treg cells of CD4+ gated cells or total splenocytes (in parentheses) proliferated expansively following culture and increased as the tumor size grew larger. (Fig. 6a) In another experiment splenocytes were harvested from a mouse bearing a large tumor and cultured with various lysate antigens. The Treg expansion observed was tumor specific as it only occurred when the splenocytes were cultured with the B16 tumor antigen (lysate) and not with other unrelated tumor antigens (Neuro-2a lysate, murine neuroblastoma) (b)
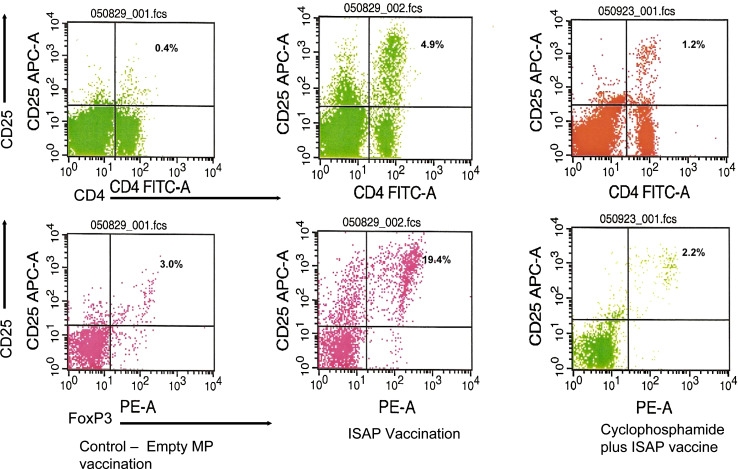
Similarly, ISAP vaccination also proved to be a strong stimulus of Treg expansion. Following vaccination, the number of T-reg harvested in the spleen were similar to baseline at day 6 and 9 post vaccination (Table 1). However, ex vivo culture with antigen allowed for expansion of the T-reg compartment. When empty microparticles were used for vaccination, only 3% of CD4+ T-cells expressed the markers of phenotypic T-reg following 5 days of culture with irradiated tumor cells. However, following vaccination with the ISAPs, 19.4% of the CD4+ T-cells were also positive for the regulatory markers CD25 and FoxP3 (Fig. 7). In a similar experimental design that assessed T-cell proliferation with the CFSE assay, only 2.1% of T-regs proliferated following empty particle vaccination in culture whereas 40% of T-regs proliferated after ISAP vaccination. When ISAP vaccinated splenocytes were co-cultured with parental melanoma tumor cells, 92.6% of Treg proliferated following 5 days of co-culture. Interestingly, tumor co-culture with splenocytes vaccinated with empty microparticles resulted in about 27% of T-reg proliferation suggesting that tumor alone induces T-reg proliferation, but remarkably less than that observed in vaccinated splenocytes. This profound expansion of T-reg induced by vaccination and tumor may partially explain the lack of protective immunity of the ISAP vaccine despite its potent induction of T-cell expansion.
Table 1.
Splenocytes were harvested in control (unvaccinated) mice and mice vaccinated with ISAPs at 6 and 9 days post vaccination
| Control 1 | Control 2 | 6 days post vaccine | 9 days post vaccine | |
|---|---|---|---|---|
| % of CD4 + CD25 + FoxP3 + cells in total splenocytes | 3.31 | 2.51 | 3.01 | 2.98 |
| % of CD4 + CD25 + FoxP3 + cells in CD4 + gated cells | 14.91 | 17.14 | 16.33 | 20.42 |
The percentage of Treg cells as a percent of total splenocytes and of total CD4 + gated cells are shown. There did not appear to be any discrete difference in the percent of Treg present. This led to ex vivo splenocyte culture and analysis of Treg expansion with antigen stimulation. (See Figs. 6 and 7) Similar findings were noted in mice bearing large tumors
Fig. 7.
ISAP induced expansion of T-reg cells: mice were vaccinated twice, 1 week apart with empty MP or MP containing CpG and TL (ISAP) and then sacrificed 1 week later for splenocyte harvest. The compartment of CD4 + CD25 + Foxp3 + Treg cells was determined by flow cytometry following 5 days of splenocyte culture with irradiated tumor cells. The upper panel shows CD4 + CD25 + cells as a percent of splenocytes, whereas the lower panel shows the FoxP3 + CD25 + cells as a percent of CD4 + cells. When empty microparticles were used for vaccination, only 3% of CD4 + T-cells expressed the markers of phenotypic T-reg. However, following vaccination with the ISAPs, 19.4% of the CD4 + T-cells were positive for the T-reg markers. Cyclophosphamide treatment appeared to suppress the ISAP induced compartment of T-reg in which only 2.2% of CD4 + cells subsequently expressed FoxP3 and CD25
ISAP vaccination induces protection against melanoma in a prophylactic model when the T-reg compartment is suppressed, but this suppression did not effect the growth in pre-established tumor
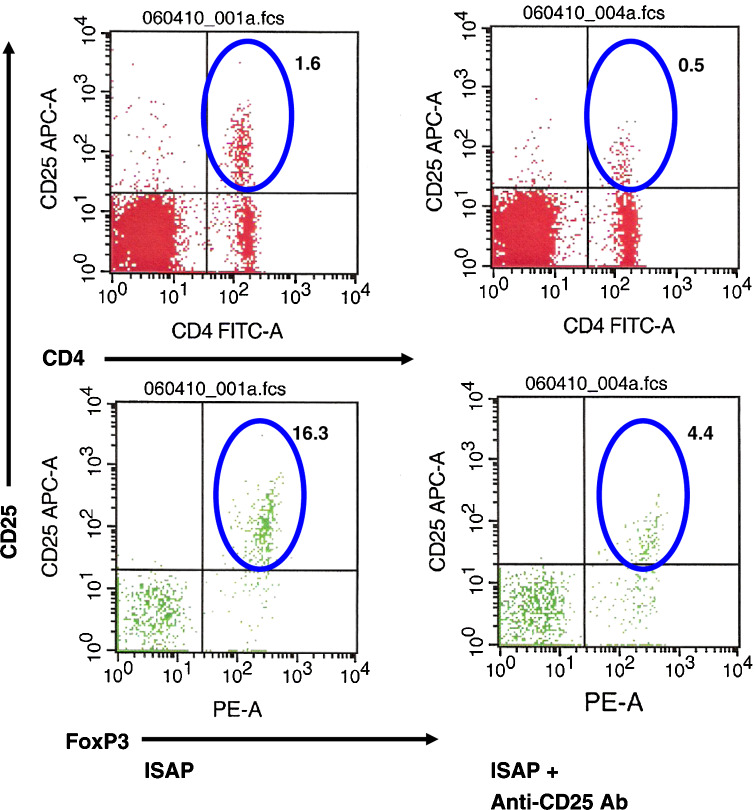
In an attempt to suppress the Treg compartment, low dose cyclophosphamide CY (100 mg/kg) or anti-CD25 antibody (500 µg) was administered IP in conjunction with the vaccine. Both CY and anti-CD25 Ab suppressed the expansion of Treg cells isolated from the spleen (Figs. 7, 8, respectively).
Fig. 8.
CD25 antibody effectively suppressed the T-reg compartment. Anti-CD25 antibody was administered 24 h prior to vaccination with ISAP. One week later, splenocytes were harvested from the mice receiving either ISAP alone or ISAP plus anti-CD25 Ab. Splenocytes were cultured with irradiated tumor cells for 5 days following which flow cytometry was performed to define the T-reg compartment. The upper panel shows CD4 + CD25 + cells as a percent of splenocytes, whereas the lower panel shows the FoxP3 + CD25 + cells as a percent of CD4+ cells. Anti CD25 effectively suppressed the CD4 + CD25 + and the FoxP3 + CD25 + compartment following ISAP vaccination
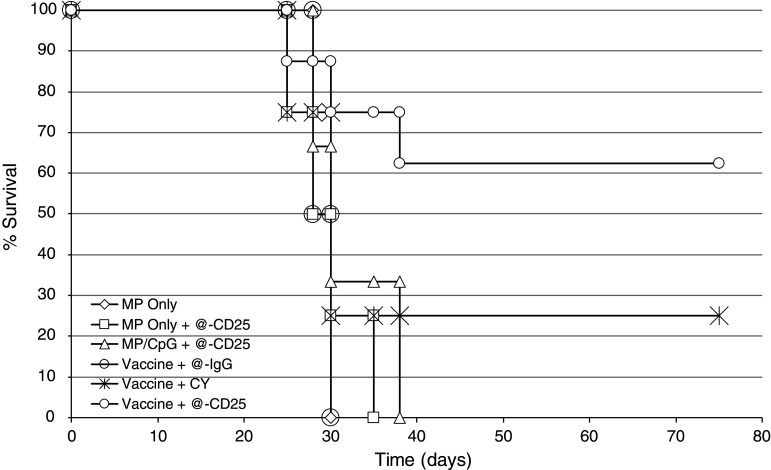
In order to evaluate the role of Treg during tumor vaccination, we studied a prophylactic model in which we vaccinated mice twice, 1 week apart intra-peritoneally with various preparations of ISAPs and then 7 days later challenged mice with high dose live B16 melanoma (5 × 105). Remarkably, mice were protected from developing tumor only when tumor lysate and CpG ODN were co-loaded into the microparticles and anti-CD25 antibody was used to suppress the T-reg compartment. Low dose CY was more protective than vaccine alone, but failed to induce protection to the same level observed for the anti-CD25 Ab. Complete protection was achieved in 75% of mice for 6 weeks and in 63% of mice indefinitely when the anti-CD25 Ab was combined with ISAP vaccination (Fig. 9).
Fig. 9.
Prophylactic tumor model. Mice were vaccinated twice, 1 week apart and then a week later with various preparations of MPs and then 7 days later challenged with high dose live B16 melanoma (5 × 105). Remarkably, mice were protected from developing tumor only when tumor lysate and CpG ODN were co-loaded into the microparticles (ISAPs) and anti-CD25 antibody (@-CD25) was used to suppress the T-reg compartment. Low dose cyclophosphamide (CY) was more protective than vaccine alone, but failed to induce protection to the same level observed for the anti-CD25 Ab. Complete protection was achieved in 75% of mice for 6 weeks and in 63% of mice indefinitely when the anti-CD25 Ab was combined with ISAP vaccination (n = 8)
The efficacy of Treg suppression in a prophylactic model, encouraged us to test T-reg suppression with the anti-CD25 Ab in a therapeutic model. Mice with pre-established tumor were vaccinated at three and ten days following tumor inoculation (5 × 105) in the presence of the anti-CD 25 Ab (500 µg) administered 24 h prior to each vaccination intra-peritoneally. We found no inhibition of tumor growth compared to contol mice or mice receiving either ISAP or the anti-CD25 Ab alone.
Discussion
Antigen presenting cells continuously sample the tissue microenvironment and phagocytose both host apoptotic cells and microbes [11]. Evidence suggests that tolerance to peripheral self-antigens occurs when apoptotic cell are sampled, while immunity against the invading organism is believed to prevail when microbes are encountered [12]. The outcome of microbial antigen presentation is dependent on the activation status of the APC, in which TLR agonists within the microbe activate the APC for induction of immunity [7]. CpG ODN based on bacterial DNA sequences activate TLR9 to stimulate both macrophages and dendritic cells in mice, thus priming these cells for immune induction [4]. TLR-inducible expression of co-stimulatory signals is one mechanism used to differentiate self from non-self [13, 14]. The mechanisms of distinguishing between self antigens and microbial antigens was uncertain until recently, when it was elegantly shown that the efficiency of presenting antigens from phagocytosed cargo is dependent on the presence of TLR ligands within the cargo [7]. We exploited this finding in our vaccine strategy by co-loading the antigen and the TLR agonist into a microparticle platform for delivery to the APC.
Poly(lactide-co-glycolide) PLGA microparticles are an excellent vehicle for vaccine strategies, in that an appropriate sized particle (1–10 µm) is readily phagocytosed by antigen presenting cells like dendritic cells and macrophages [9, 15]. The PLGA matrix employed in the particles have been safely used in humans for nearly three decades in the form of suture material. In our study, PLGA did not appear to be immunogenic in that it failed to elicit any responses when the empty particles were used for vaccination in vitro and in vivo, thus negating the concern of developing immunity against the vehicle as may be the case with viral based particulate systems. Copolymers can be controlled for specific applications with varying degradation kinetics [16, 17].
Microparticle vaccines could mimic pathogens that are recognized, phagocytosed and processed by professional APCs. In so doing, it is feasible to load the microparticle with the antigens of choice and TLR agonists most suitable for inducing immunity. In the current studies we used tumor lysate from whole tumor cells as the antigen source. Whole tumor cell antigens should be effective for vaccination, as they include not only a complete complement of the tumor cell antigens, but also resistant mutations or tumor cell variants. It is possible that B-cells could also phagocytose the beads [18], but this is unlikely. Mouse B cells are unable to ingest beads of 1 micron in diameter or greater whereas the ISAPs have an average diameter of 2.4 µm +0.43.
Regulatory T cells (Treg) inhibit anti-tumor T cell responses and may be a major obstacle to effective tumor vaccination and immunotherapy [19]. It is proposed the murine DC activated through TLR9 may become resistant to the immune suppressive effects of Treg in vitro [20, 21]. On the other hand however, TLR9 ligands [or ligands for TLR2 or TLR4) increase the proliferation of murine Treg cultured in the presence of APC and anti-CD3 [22] and TLR9 activation of purified human pDC enhances the generation of Treg, suppressing naïve T cell responses [23]. In the current study we found marked expansion of the Treg compartment following ISAP (TLR9) vaccination. This was only evident when splenocytes were cultured with the specific tumor antigen suggesting tumor specific Treg expansion, whereas baseline levels of Treg in the spleen were not remarkably different from controls. A similar Treg effect was noted in our studies following inoculation and growth of tumors in the mice. It is also of interest to note that CD4 expansion and proliferation only occurred when the Ag and TLR agonist (CpG) were co-delivered. If either was delivered alone to the APC, neither CD4 nor CD8 expansion was noted.
The lack of significant suppression of tumor growth despite the prolific expansion of T cells following ISAP vaccination would suggest that Tregs play an important role in suppressing the anti-tumor effects of the vaccine. This phenomenon was clearly shown when Tregs were suppressed by the anti-CD25 antibody in which remarkable enhancement of tumor specific immunity was observed. Collectively, these studies indicate that systemic TLR9 activation alone in this model does not overcome Treg in vivo, and may in fact increase Treg activity, perhaps as a feedback mechanism to avoid deleterious immune activation or autoimmunity. In an established tumor model we were unable to demonstrate any benefit to vaccine efficacy with anti-CD25 Ab suppression. The anti-CD25 Ab indiscriminately neutralizes CD25 bearing cells that besides Treg will also include tumor specific activated T-cells. In the established model it may be necessary to target the Treg specifically without impacting activated cytotoxic T-cells to enhance vaccine efficacy. Together with the extensive evidence that Treg contribute to the immune suppression of cancer patients [19, 24], our findings imply that the efficacy of ISAP vaccination for cancer therapy may be significantly enhanced by the prior depletion or inactivation of Treg.
The tumor itself elicits many immuno-suppressive mechanisms in the host, thus making vaccination in the face of bulky established tumor inefficient [25–29]. In the well established melanoma or neuroblastoma mouse models, vaccination alone can delay tumor growth with few long-term survivors, but has little value in curing most animals with disease. Due to the multiple immuno-suppressive mechanisms in play and the lack of a specific Treg inhibitor, we were unable to significantly impact tumor growth with vaccination in the established melanoma model. It is correctly argued that the prophylactic mouse tumor model is inadequate as a clinical correlate, thus the value of testing our strategy in a prophylactic model should be questioned. In the mouse neuroblastoma model we have found that mice can be cured with vaccination following tumor resection that otherwise ensures 100% mortality due to tumor recurrence after resection alone [30]. Unlike this “minimal residual disease” mouse neuroblastoma model; however, resection of inoculated melanoma tumor results in cure of the mice without any further treatment (unpublished observations). Thus, vaccination following resection in this model would be of no value in trying to understand the role of Treg. However, in an attempt to define the role of Treg using our ISAP vaccine strategy we studied a prophylactic melanoma model for the reasons outlined above. We believe that our results show an essential role for suppressing the Treg compartment during vaccination with ISAPs. Anti-tumor vaccines capable of activating both CD4 and CD8 T cells are preferred for long lasting T cell responses. It is shown that by enhancing uptake of antigen via the endosomal pathway, the desirable CD4 and CD8 T cell responses are induced, but Treg are also activated [25]. Although our ISAP vaccine is remarkably effective for inducing desirable T cell responses, the Treg response previously reported through the endosomal pathway is considerable. Treg suppression could thus be critical for therapeutic vaccine protocols. It seems such protocols would be most successful in preventing recurrence of disease following initial ablation with either standard treatment like chemotherapy, radiation therapy and/or surgery.
Footnotes
Supported in part by grants from NIH R21 CA100652-01, the American Cancer Society IRG-77-004-28 and Michael C. Sandler.
References
- 1.Steinman RM. Linking innate to adaptive immunity through dendritic cells. Novartis Found Symp. 2006;279:101–109. [PubMed] [Google Scholar]
- 2.Steinman RM, Hemmi H. Dendritic cells: translating innate to adaptive immunity. Curr Top Microbiol Immunol. 2006;311:17–58. doi: 10.1007/3-540-32636-7_2. [DOI] [PubMed] [Google Scholar]
- 3.Hemmi H, Takeuchi O, Kawai T, Kaisho T, Sato S, Sanjo H, Matsumoto M, Hoshino K, Wagner H, Takeda K, Akira SA. Toll-like receptor recognizes bacterial DNA. Nature. 2000;408:740–745. doi: 10.1038/35047123. [DOI] [PubMed] [Google Scholar]
- 4.Krieg AM. Therapeutic potential of Toll-like receptor 9 activation. Nat Rev Drug Discov. 2006;5:471–484. doi: 10.1038/nrd2059. [DOI] [PubMed] [Google Scholar]
- 5.Krieg AM, Efler SM, Wittpoth M, Al Adhami MJ, Davis HL. Induction of systemic TH1-like innate immunity in normal volunteers following subcutaneous but not intravenous administration of CPG 7909, a synthetic B-class CpG oligodeoxynucleotide TLR9 agonist. J Immunother. 2004;27:460–471. doi: 10.1097/00002371-200411000-00006. [DOI] [PubMed] [Google Scholar]
- 6.Sandler AD, Chihara H, Kobayashi G, Zhu X, Miller MA, Scott DL, Krieg AM. CpG oligonucleotides enhance the tumor antigen-specific immune response of a granulocyte macrophage colony-stimulating factor-based vaccine strategy in neuroblastoma. Cancer Res. 2003;63:394–399. [PubMed] [Google Scholar]
- 7.Blander JM, Medzhitov R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature. 2006;440:808–812. doi: 10.1038/nature04596. [DOI] [PubMed] [Google Scholar]
- 8.Waeckerle-Men Y, Allmen EU, Gander B, Scandella E, Schlosser E, Schmidtke G, Merkle HP, Groettrup M. Encapsulation of proteins and peptides into biodegradable poly(d, l-lactide-co-glycolide) microspheres prolongs and enhances antigen presentation by human dendritic cells. Vaccine. 2006;24:1847–1857. doi: 10.1016/j.vaccine.2005.10.032. [DOI] [PubMed] [Google Scholar]
- 9.Tabata Y, et al. Phagocytosis of polymer microspheres by macrophages. Adv Polymer Sci. 1990;94:107–141. doi: 10.1007/BFb0043062. [DOI] [Google Scholar]
- 10.Johnson CR, Kitz D, Little JR. A method for the derivation and continuous propagation of cloned murine bone marrow macrophages. J Immunol Methods. 1983;65:319–332. doi: 10.1016/0022-1759(83)90127-8. [DOI] [PubMed] [Google Scholar]
- 11.Greenberg S, Grinstein S. Phagocytosis and innate immunity. Curr Opin Immunol. 2002;14:136–145. doi: 10.1016/S0952-7915(01)00309-0. [DOI] [PubMed] [Google Scholar]
- 12.Ravichandran KS. “Recruitment signals” from apoptotic cells: invitation to a quiet meal. Cell. 2003;113:817–820. doi: 10.1016/S0092-8674(03)00471-9. [DOI] [PubMed] [Google Scholar]
- 13.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 14.Iwasaki A, Medzhitov R. Toll-like receptor control of the adaptive immune responses. Nat Immunol. 2004;5:987–995. doi: 10.1038/ni1112. [DOI] [PubMed] [Google Scholar]
- 15.Manzel L, Macfarlane DE. Lack of immune stimulation by immobilized CpG-oligodeoxynucleotide. Antisense Nucleic Acid Drug Dev. 1999;9:459–464. doi: 10.1089/oli.1.1999.9.459. [DOI] [PubMed] [Google Scholar]
- 16.Waeckerle-Men Y, Groettrup M. PLGA microspheres for improved antigen delivery to dendritic cells as cellular vaccines. Adv Drug Deliv Rev. 2005;57:475–482. doi: 10.1016/j.addr.2004.09.007. [DOI] [PubMed] [Google Scholar]
- 17.Tamber H, et al. Formulation aspects of biodegradable polymeric microspheres for antigen delivery. Adv Drug Delivery Rev. 2005;57:357–376. doi: 10.1016/j.addr.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 18.Li J, Barreda DR, Zhang YA, Boshra H, Gelman AE, Lapatra S, Tort L, Sunyer JOB. B lymphocytes from early vertebrates have potent phagocytic and microbicidal abilities. Nat Immunol. 2006;7:1116–1124. doi: 10.1038/ni1389. [DOI] [PubMed] [Google Scholar]
- 19.Zou W. Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol. 2006;6:295–307. doi: 10.1038/nri1806. [DOI] [PubMed] [Google Scholar]
- 20.Pasare C. a. M., R Toll pathway-dependent blockade of CD4 + CD25 + T cell-mediated suppression by dendritic cells. Science. 2003;299:1033–1036. doi: 10.1126/science.1078231. [DOI] [PubMed] [Google Scholar]
- 21.Kubo T, Hatton RD, Oliver J, Liu X, Elson CO, Weaver CT. Regulatory T cell suppression and anergy are differentially regulated by proinflammatory cytokines produced by TLR-activated dendritic cells. J Immunol. 2004;173:7249–7258. doi: 10.4049/jimmunol.173.12.7249. [DOI] [PubMed] [Google Scholar]
- 22.Sutmuller RP, den Brok MH, Kramer M, Bennink EJ, Toonen LW, Kullberg BJ, Joosten LA, Akira S, Netea MG, Adema GJ. Toll-like receptor 2 controls expansion and function of regulatory T cells. J Clin Invest. 2006;116:485–494. doi: 10.1172/JCI25439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Moseman EA, Liang X, Dawson AJ, Panoskaltsis-Mortari A, Krieg AM, Liu YJ, Blazar BR, Chen W. Human plasmacytoid dendritic cells activated by CpG oligodeoxynucleotides induce the generation of CD4 + CD25 + regulatory T cells. J Immunol. 2004;173:4433–4442. doi: 10.4049/jimmunol.173.7.4433. [DOI] [PubMed] [Google Scholar]
- 24.Emens LA, Reilly RT, Jaffee EM. Augmenting the potency of breast cancer vaccines: combined modality immunotherapy. Breast Dis. 2004;20:13–24. doi: 10.3233/bd-2004-20103. [DOI] [PubMed] [Google Scholar]
- 25.Adler AJ. Mechanisms of T cell tolerance and suppression in cancer mediated by tumor-associated antigens and hormones. Curr Cancer Drug Targets. 2007;7:3–14. doi: 10.2174/156800907780006931. [DOI] [PubMed] [Google Scholar]
- 26.Hou DY, Muller AJ, Sharma MD, DuHadaway J, Banerjee T, Johnson M, Mellor AL, Prendergast GC, Munn DH. Inhibition of indoleamine 2, 3-dioxygenase in dendritic cells by stereoisomers of 1-methyl-tryptophan correlates with antitumor responses. Cancer Res. 2007;67:792–801. doi: 10.1158/0008-5472.CAN-06-2925. [DOI] [PubMed] [Google Scholar]
- 27.Johnson BF, Clay TM, Hobeika AC, Lyerly HK, Morse MA. Vascular endothelial growth factor and immunosuppression in cancer: current knowledge and potential for new therapy. Expert Opin Biol Ther. 2007;7:449–460. doi: 10.1517/14712598.7.4.449. [DOI] [PubMed] [Google Scholar]
- 28.Valenti R, Huber V, Iero M, Filipazzi P, Parmiani G, Rivoltini L. Tumor-released microvesicles as vehicles of immunosuppression. Cancer Res. 2007;67:2912–2915. doi: 10.1158/0008-5472.CAN-07-0520. [DOI] [PubMed] [Google Scholar]
- 29.Wang HY, Wang RF. Regulatory T cells and cancer. Curr Opin Immunol. 2007;19:217–223. doi: 10.1016/j.coi.2007.02.004. [DOI] [PubMed] [Google Scholar]
- 30.Ohashi K, Kobayashi G, Fang S, Zhu X, Antonia SJ, Krieg AM, Sandler AD. Surgical excision combined with autologous whole tumor cell vaccination is an effective therapy for murine neuroblastoma. J Pediatr Surg. 2006;41:1361–1368. doi: 10.1016/j.jpedsurg.2006.04.034. [DOI] [PubMed] [Google Scholar]