Abstract
The history of immunizing animals with fetal tissues to generate an antitumor response dates back a century ago. Subsequent reports supported the idea that vaccination with embryonic materials could generate cancer-specific immunity and protect animals from transplantable and chemically induced tumors. In our study, we found C57 BL/6 mice vaccinated with embryonic stem cells (ESCs) received obvious antitumor immunity, which protected them from the formation and development of lung cancer. Furthermore, we investigated the antitumor effects of administration of ESCs in mice with minor and/or heavy tumor load. The tumor growth was monitored, the proliferation of lymphocytes and secretion of cytokines were examined, and finally the tissue sections were approached by immunohistochemical and apoptosis staining. The results suggested that mice injected with ESCs received obvious tumor inhibition and retardation due to significant lymphocyte proliferation and cytokine secretion, which help to rebuild the host’s immunity against cancer to some extent and comprise the main part of antitumor immunity. Moreover, mice with minor tumor load received stronger antitumor effect compared with mice with heavy tumor load, may be due to relatively intact immune system. Thus, besides their function as prophylactic vaccines, administration of ESCs could be a potential treatment for cancer, which obviously prevent and control the proliferation and development of malignant tumors.
Keywords: Embryonic stem cells, Cancer stem cells, Antitumor immunity, Immunotherapy, Cancer vaccine
Introduction
In the previous few years, a new theory called Cancer Stem Cells (CSCs) model was proposed to explain that a few tumor cells would have the properties to initiate and propagate tumors [1]. A series of studies have been accumulating evidence supporting this model and people have identified CSCs in breast [2], brain [3], pancreas [4], colon [5], prostate [6], mesenchymal [7], and neck and head tumors [8]. It was found that CSCs and normal stem cells including embryonic stem cells (ESCs) have some properties in common, such as self-renewal, multilineage differentiation, heterogeneity, similar signal transduction pathways [9–12], especially they share similar cell surface markers and antigens not presented by adult tissues [13–15], so immune response against ESCs would cross-react with cancer cells especially CSCs [16]. All these properties make ESCs vaccine against cancer feasible and reasonable.
Almost 100 years have passed since the practice of immunizing animals with embryonic materials to generate specific antitumor immunity. It was found that this kind of vaccination could efficiently prevent the formation and development of transplantable tumors [17]. The antitumor effect was later investigated on a series of chemically induced tumors such as cancer of the skin, liver, and gastrointestinal tract [18–20]. Moreover, recent research proposed a possibility of human pluripotent stem cells vaccination to generate a broad spectrum of antitumor immunity in mice [16]. In our study, we carried out a similar study and found that mice vaccinated with ESCs obtained a strong and stable antitumor response to protect them from cancer.
However, the use of fetal materials to induce tumor-specific immunity has always acted as prophylactic vaccines and preventive measures. What would happen if they already had a tumor load before the administration of ESCs, or could ESCs induce antitumor effects and control the development and proliferation of tumor masses in tumor-bearing mice? To answer such questions, mice suffering from minor and/or heavy tumor load were injected with ESCs, then the proliferation of lymphocytes, secretion of cytokines, immunohistochemical changes and cell apoptosis in tumor sections were detected. As we expected, significant antitumor responses and enhanced tumor rejection in these mice were found, and more importantly, the immunosuppression caused by cancer was modified to a certain extent without obvious evidence of autoimmune diseases.
Materials and methods
Mice
Wild type female C57 BL/6 mice, approximately 4–6 weeks old on arrival, were obtained from the Institute of Laboratory Animal Science, Chinese Academy of Medical Science. Mice were housed in laminar air-flow cabinets under special pathogen-free conditions using standard guidelines and used at 6–8 weeks of age. All animal experiment protocols were approved by the Ethics Committee of Shandong University (Animals certificaton NO. is SCXK (Lu) 2003-0003).
Cell lines
The C57 BL/6 Mouse Embryonic Stem Cells were obtained from Cyagen Bioscience Inc., cultured on plates pre-coated with gelatin solution and γ-ray irradiated C57 BL/6 Mouse Embryonic Fibroblasts (Cyagen Bioscience Inc.) as feeder cells in Mouse Embryonic Stem Cell Growth Medium (Cyagen Bioscience Inc.). At the time of administration, ESCs were in passage 8–15 approximately and pre-irradiated (15 Gy). Lewis Lung Carcinoma cells [21] (LLCs) were purchased from the Cell Bank of Chinese Academy of Medical Science and cultured in Dulbecco’s Modified Eagle’s Medium (DMEM, Hyclone) supplemented with 10% fetal bovine serum (FBS, Hyclone), and about in passage 10–15 when inoculated.
Reagents and antibodies
Most reagents used in cell culture were obtained from Hyclone Biochemical Products Co., Ltd unless specifically indicated. The antibodies used in flow cytometry were purchased from eBioscience (San Diego, CA, USA) and antibodies used in immunohistochemistry from BioWorld Technology Ltd (Barrie, ON, Canada) and Biosynthesis Biotechnology Co., Ltd (Beijing, China). The ELISA kits were obtained from Jingmei Biotech Co., Ltd (Beijing, China) and the one step TUNEL apoptosis assay kit [22] and reagents used in TUNEL analysis was purchased from Beyotime Biotechnology (Haimen, China).
Experimental protocols
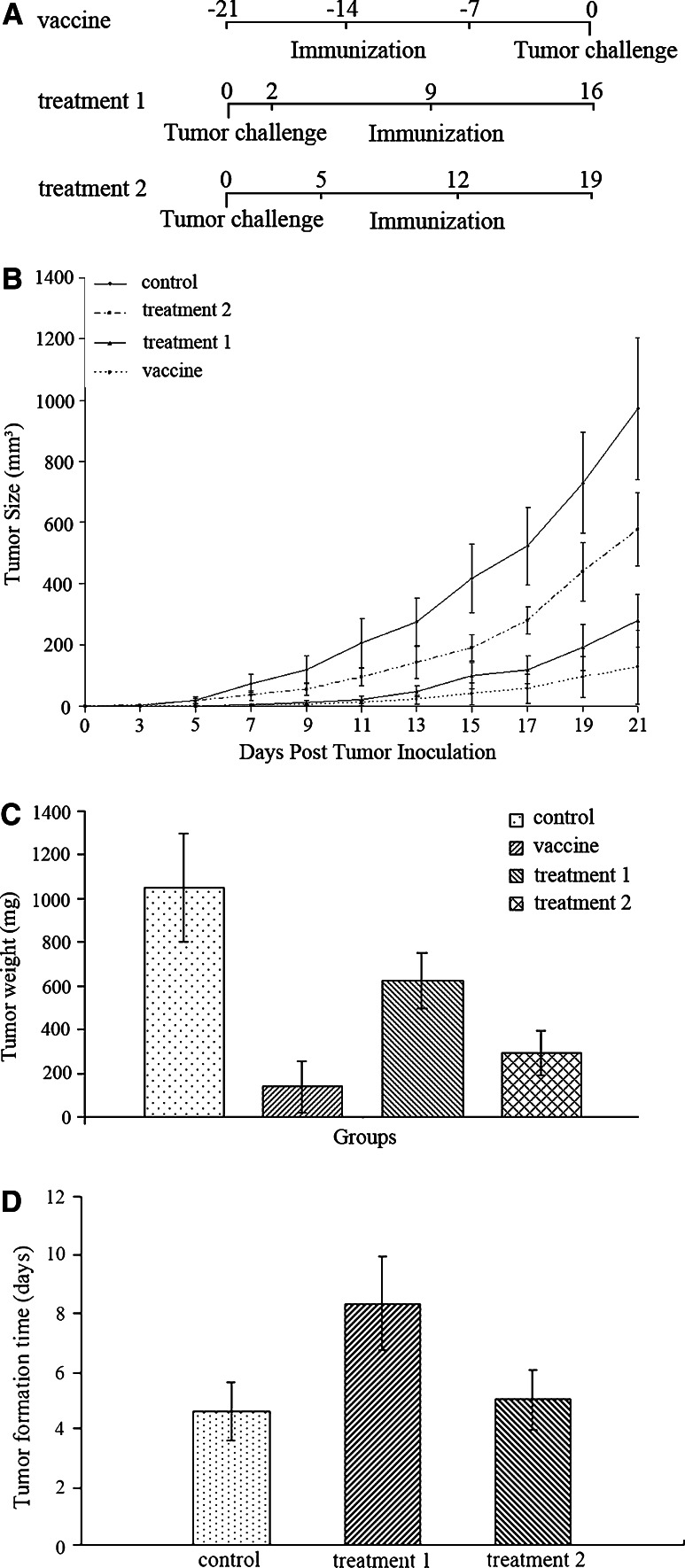
Forty female C57 BL/6 mice were randomly divided into four groups: the vaccine group (n = 10) received subcutaneous vaccination with pre-irradiated ESCs three times at a 1-week interval, and 1 week after the third time of vaccination, LLCs were inoculated subcutaneously (all the number of cells involved was 1 × 106); the treatment group 1 (n = 10) received three times of ESCs treatment (at a 1-week interval) 48 h after inoculation of LLCs, when there were no touchable tumor masses developed; the treatment group 2 (n = 10) received three times of ESCs treatment (at a 1-week interval) 5 days after inoculation of LLCs, when there were macroscopic tumor masses developed subcutaneously; the control group (n = 10) received inoculation of LLCs without vaccination or treatment (Fig. 1a). Tumor size was measured in longitudinal (L) and transverse (W) diameters by digital calipers every other day since tumor cells inoculation and tumor volumes were calculated according to the formula V = L × W 2/2 [23]. The endpoint for this study was set to be one diameter of tumors ≥15 mm when all mice were killed under anesthesia and tumors were excised and weighed. During the experiment, all mice were monitored for some general health indicators: general behavior, feeding and defecation, body weight, neuromuscular tone, appearance of fur, etc.
Fig. 1.
Antitumor immunity induced by administration of ESCs. a Scheme of ESCs immunization and tumor inoculation. Mice in the vaccine group received subcutaneous 1 × 106 pre-irradiated ESCs vaccination three times at a 1-week interval, and 1 week after the third time of vaccination, 1 × 106 LLCs were inoculated subcutaneously. Mice in the treatment groups 1 and 2 also received subcutaneous 1 × 106 pre-irradiated ESCs immunization three times at a 1-week interval, at 2 and 5 days after the tumor challenge, respectively. b After the inoculation of 1 × 106 LLCs, tumor size of all groups was measured using a digital caliper and tumor volumes were calculated. The results indicated statistical difference in tumor size between the vaccine and control group from day 5 on (p < 0.05, and p < 0.01 from day 7 on), between the treatment groups and control group (p < 0.05) from day 7 on, and between the treatment groups 1 and 2 from day 9 on. c At the end of the experiment, all mice were killed under anesthesia and tumors were excised and weighed. The results of tumor weight suggested that there was significant difference between the vaccine and control group (p < 0.01), between the treatment groups and control group (p < 0.05), and between the treatment groups 1 and 2 (p < 0.05). d Tumor formation time of the control group and treatment groups. The difference in tumor formation time between the control group and treatment group 1 was highly significant statistically (p < 0.01). Error bars denote SD
Flow cytometry
Three or four days after the third time of administration of ESCs, citrate-anticoagulated blood was acquired from venous sinus of eye orbit [24] from mice of all groups (in the control group on the same day as the treatment groups). The CD3/4/8- and CD19- positive lymphocytes were stained and analyzed on EPICS XL flow cytometer (Beckman Coulter Corp.), and the results were processed by WinMDI software (Scripps Institute).
ELISA
Fresh blood from all groups of mice was obtained at the same time and in the same way as previously described, and serum was collected. The serum levels of interleukin-4, interleukin-2, and interferon γ were tested by ELISA kits in accordance with the instructions and analyzed on a microplate reader (Wellscan MK3, Labsystems Dragon).
Histology and immunohistochemistry
When the mice were killed, the primary tumors and spleens were excised and fixed in 4% formaldehyde. The specimens were embedded in paraplast and 4-μm thick sections were cut and stained with hematoxylin and eosin by standard methods.
Tissue sections were deparaffinized and rehydrated through a graded alcohol series. Endogenous peroxidase activity was quenched by incubation with 3% hydrogen peroxide for 15 min, after 15-min rises in three changes of PBS, sections were blocked with normal goat serum to suppress nonspecific background staining. The rabbit anti-mouse CD4 and CD8 antibodies (diluted, 1:100) were applied to the sections at 4°C overnight. Then the sections were incubated with biotinylated goat anti-rabbit IgG for 30 min at room temperature and processed according to the SP kit protocol, and finally examined under a light microscope and camera system.
TUNEL
The tumor sections were deparaffinized and rehydrated through a graded alcohol series, incubated with protease K for 25 min at room temperature for antigen retrieval, and then operated according to the TUNEL apoptosis detection kit. The FITC-labeled TUNEL-positive cells were imaged under a fluorescent microscope using 488-nm excitation and 530-nm emission. An image analyser (Image Pro Plus 6.0, Media Cybernetics) was used to score all the staining sections of immunohistochemistry and TUNEL, and results were present in mean optical density (MOD = integrated optical density/area).
Statistical analysis
Statistical significance between groups was determined by ANOVA test and Student–Newman–Keuls multiple comparisons test after confirming the equality of variance with the Levene test using SPSS statistical software (version 17.0). Most data were present as mean ± SD and a p value less than 0.05 was considered statistically significant.
Results
Inhibition of tumor formation in vaccine group
In our study, we chose a well-established Lewis Lung Carcinoma model in C57 BL/6 mice [25], and 1 × 106 cancer cells, given subcutaneously were enough to induce progressive local growth of the tumor masses and eventual death due to distal lung metastasis and cachexia. After the administration of LLCs, the kinetics of tumor growth was then closely monitored (Fig. 1b, c). As we expected, tumor formation rates of mice in the vaccine group were only 40%, and no more mice suffered from tumor formation during the experiment, significantly different from that of the control group (100%, p < 0.05). Moreover, vaccination resulted in a dramatic retardation of tumor growth with statistically significant differences in average tumor size compared with the control group from day 5 on (p < 0.05, and p < 0.01 from day 7 on).
Inhibition of tumor growth in treatment groups
We injected mice in the treatment groups 1 and 2 with ESCs subcutaneously at 2 and 5 days after the inoculation of LLCs, respectively. 1 week later we repeated the administration and the third time of administration was carried out at another 1-week interval (Fig. 1a). The tumor formation time was closely detected (Fig. 1d) and tumor size was measured every other day till the endpoint (Fig. 1b). The results suggested that mice in two treatment groups received obvious antitumor immunity and rejected tumor masses from proliferation and development compared with the control group. Furthermore, mice in the treatment group 1 obtained stronger antitumor response, which protected mice from malignant cancer progression more effectively compared with mice in the treatment group 2 from day 9 on.
Proliferation of lymphocytes
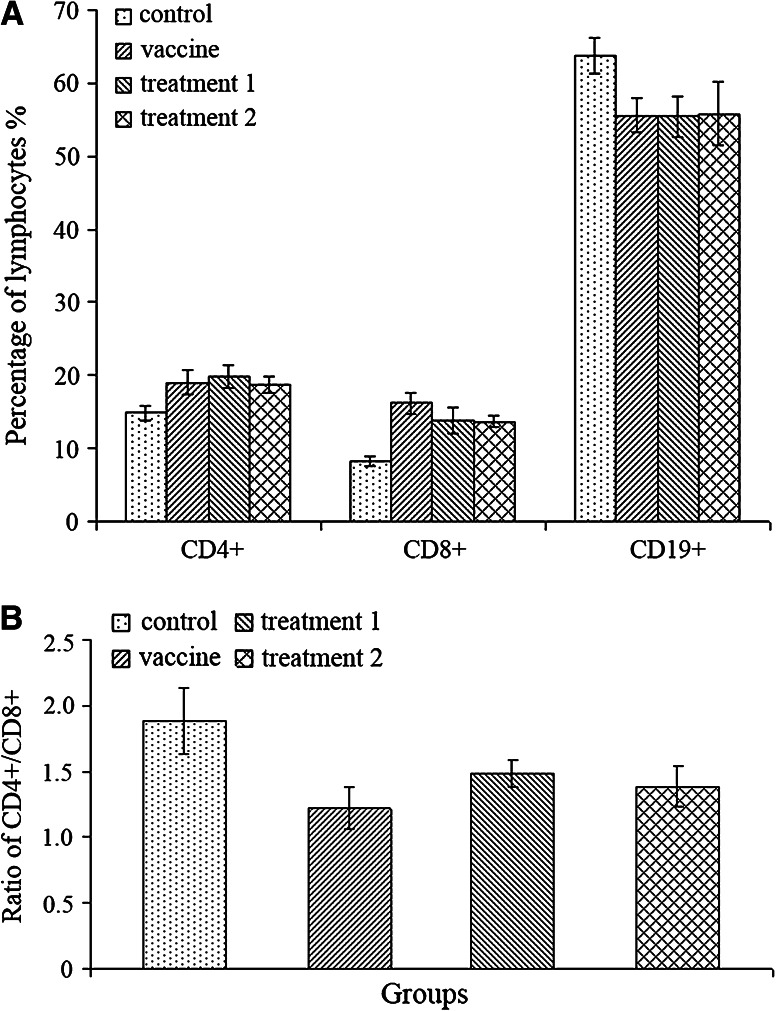
In our study, we examined the proliferation of both T and B lymphocytes in peripheral circulation of all groups of mice, especially we were interested in the changes induced by ESCs inoculation (Fig. 2a). We found statistically significant proliferation of both CD4+ and CD8+ T lymphocytes in the vaccine and treatment groups compared with the control group (p < 0.05), moreover, the ratio of CD4+/CD8+ lymphocytes become smaller (p < 0.05, Fig. 2b), which indicated that there were more CD8+ lymphocytes (cytotoxic T lymphocytes) generated. However, there was an obvious decrease of CD19+ B lymphocytes in these groups of mice (p < 0.05). But there was neither any significant difference between the vaccine and treatment groups in the proliferation level of lymphocytes, nor in the CD4+/CD8+ ratio.
Fig. 2.
The classification of lymphocytes in circulation. a Administration of ESCs induced significant proliferation of CD4+ and CD8+ lymphocytes in peripheral circulation of mice from the vaccine and treatment groups compared with the control group (p < 0.05), however, there was an obvious decrease of CD19+ lymphocytes (p < 0.05). No significant difference was detected among the vaccine and treatment groups. b The ratio of CD4+ and CD8+ lymphocytes of the vaccine and treatment groups become smaller (p < 0.05), so there were more CD8+ lymphocytes (cytotoxic T lymphocytes) generated compared with CD4+ lymphocytes
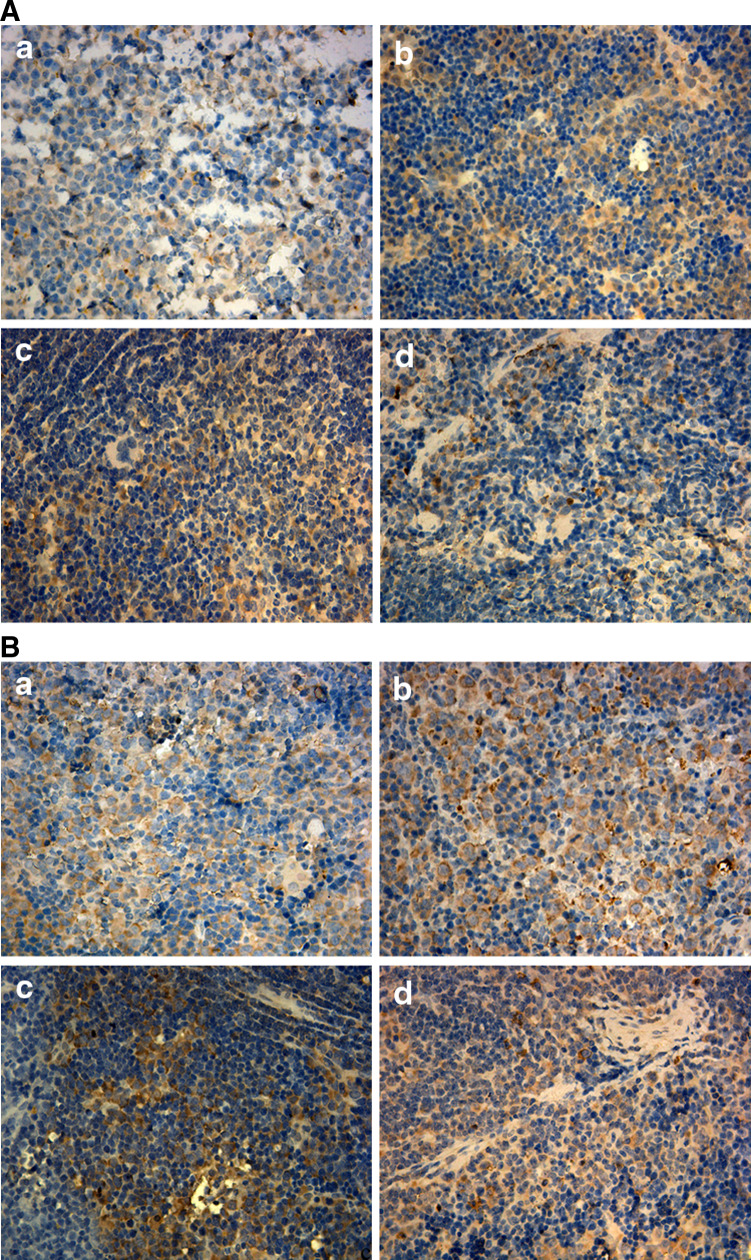
When the mice were killed, we detected the proliferation of CD4+ and CD8+ lymphocytes in spleens by immunohistochemistry (Fig. 3); the results indicated significant difference in the vaccine and treatment groups compared with the control group (p < 0.05), and this was in accordance with the results of cytoflowmetry.
Fig. 3.
The proliferation of lymphocytes in spleens of mice from all groups (a = control group, b = vaccine group, c = treatment group 1, d = treatment group 2). a The immunohistochemistry staining of CD4+ lymphocytes. The MOD (a = 0.14 ± 0.029, b = 0.25 ± 0.048, c = 0.19 ± 0.038, d = 0.23 ± 0.028) indicated significant difference of the vaccine and treatment groups compared with the control group (p < 0.05). b The immunohistochemistry staining of CD8+ lymphocytes. There was significant difference of MOD (a = 0.14 ± 0.032, b = 0.27 ± 0.044, c = 0.20 ± 0.047, d = 0.19 ± 0.034) of the vaccine and treatment groups compared with the control group (p < 0.05)
Cytokines
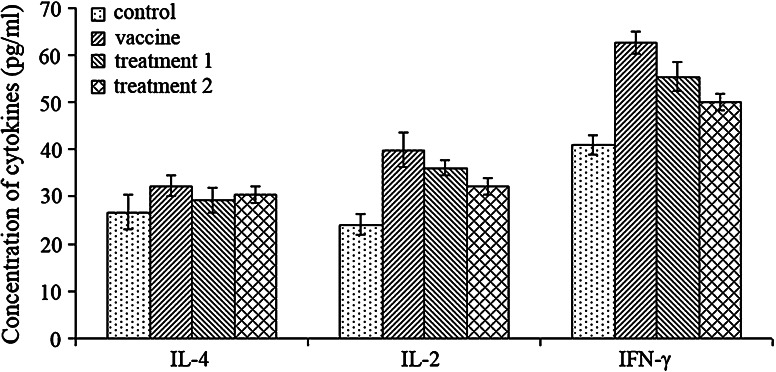
To further study the cellular immune mechanism mediating the antitumor immunity, we analyzed the cytokines produced by lymphocytes in serum. The concentration of interleukin-4, interleukin-2, and interferon γ was detected by ELISA (Fig. 4), and the results suggested that mice in both the vaccine and treatment groups had significantly increased expression of interleukin-2 and interferon γ (p < 0.05), but no obvious change in the serum level of interleukin-4. Furthermore, mice in the treatment group 1 seemed to have higher expression in interleukin-2 (p = 0.102) and interferon γ (p < 0.05) compared with mice in the treatment group 2.
Fig. 4.
Concentration of cytokines in serum of mice from all groups. Mice in both the vaccine and treatment groups had significantly increased expression of interleukin-2 and interferon γ (p < 0.05) compared to the control group. Moreover, mice with minor tumor load had increased expression in interleukin-2 (p = 0.102) and interferon γ (p < 0.05) compared with mice with heavy tumor load. No significant difference was found in concentration of IL-4 of all groups
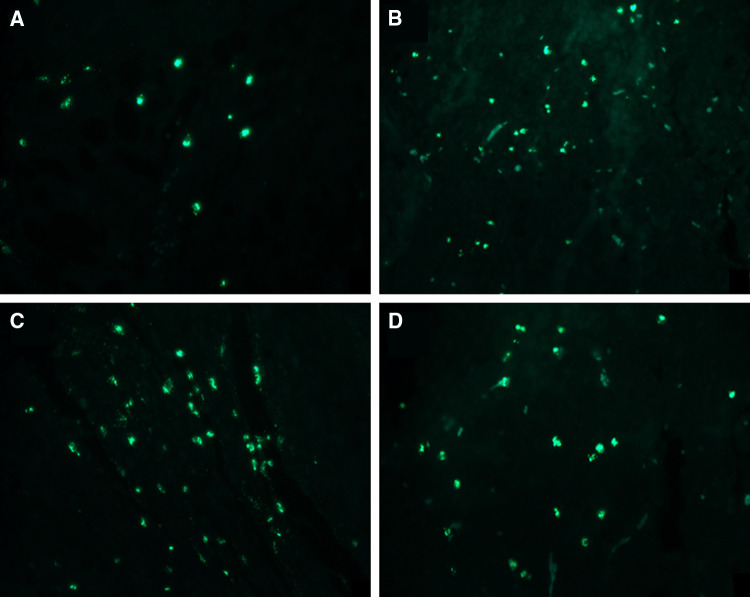
TUNEL
When the mice were killed, we detected the cell apoptosis in the tumor tissues by the TUNEL kit (Fig. 5). We found that apoptosis of tumor cells in the vaccine and treatment groups were statistically different from that of the control group (p < 0.05). However, no significant difference was found between the vaccine and treatment groups, or between the treatment groups 1 and 2.
Fig. 5.
The cell apoptosis of tumor sections from all groups (a = control group, b = vaccine group, c = treatment group 1, d = treatment group 2). The MOD suggested that mice in the vaccine and treatment groups had more cell apoptosis compared with the control group (p < 0.05, a = 0.035 ± 0.0041, b = 0.051 ± 0.0052, c = 0.057 ± 0.0046, d = 0.050 ± 0.0040)
Discussion
It was a long time since embryonic materials has been used as vaccines to prevent the formation and development of tumors in animal experiments, and the original theory basis was thought to be the biological similarities between embryos and tumors [17]. Since the identification of ESCs and CSCs, especially the discovery of antigenic and biological similarities between them, there has been accumulative evidence supporting vaccination with ESCs to generate an antitumor immunity [16]. In our study, we discovered yet another novel application of ESCs, i.e. administration of ESCs in mice with minor and/or heavy tumor load could also generate effective antitumor effect and protect mice from tumor proliferation and/or further development. Furthermore, the antitumor immunity was stronger in mice with minor tumor load, which may be due to more lymphocytes generated and antitumor cytokines secreted compared with mice with heavy tumor load.
The goal of cancer immunotherapy is to develop modalities that specifically target tumor cells, thereby avoiding harmful side effects on normal tissues [26]. The identification of CSCs makes it a topic of considerable interest to targeting and eradicating the stem-cell compartment of a tumor, which means stable or long-lasting remission, and even a cure of cancer [27–29]. However, the function of immunotherapy depends on the host’s immune cells and an intact immune system [30]. But in a tumor loading state, animals and/or patients often suffer from immune dysfunction and apoptosis of antitumor effector cells [31]. Recent research revealed that the reason of immune system failure to eliminate tumors was mainly because appropriate lymphocytes were not activated to a threshold required for tumor rejection [32]. Due to the altered expression of proteins involved in antigen processing and presentation, tumor cells were able to avoid recognition by immune system, and insufficient activation of antitumor immunity lead to poor protective responses [33, 34]. Because of the antigenic similarities between tumor cells and ESCs, immune response against ESCs was expected to cross-react with tumor cells especially CSCs in vivo and antitumor immunity was expected to be rebuilt in tumor-bearing animals and/or patients.
Recently, it has been shown that expression of MHC class I molecules on ESCs is sufficient to induce recognition and rejection by cytotoxic T lymphocytes [35]. The immunogenicity of ESCs has been extensively covered in several reviews [36–38], which share the same perspective that ESCs and their derivatives could generate cellular immune response and active CD4+ and CD8+ lymphocytes in vivo. In our study, we detected the CD3/4/8 (T lymphocyte marker) and CD19 (B lymphocyte marker) positive lymphocytes in both peripheral circulation and spleens of mice. As we expected, there was significant proliferation of T lymphocytes after the administration of ESCs in tumor loading mice (in the treatment groups) as well as healthy mice (in the vaccine group, untreated when received administration of ESCs), especially the proliferation of CD8+ lymphocytes (cytotoxic T lymphocytes), which constitutes the main part of antitumor effector cells [39, 40]. The proliferation of lymphocytes make it possible to eliminate and/or reject tumors in tumor loading mice of the treatment groups and protect animals from tumor formation and development in mice of the vaccine group.
Besides the decrease in quantities of lymphocytes, the cytokines secreted by CD4+ lymphocytes in a Th1/Th2 shift state is another abnormalism of lymphocyte homeostasis caused by cancer [41, 42], which means an increase in Th2 cytokines (interleukin-4, 10 etc.) while a decrease in Th1 cytokines (interleukin-2, interferon γ etc.). So, we investigated the concentration of interleukin-4, interleukin-2, and interferon γ in serum of all groups of mice. We found that administration of ESCs stimulated the secretion of Th1 cytokines, which comprised an important part of antitumor immunity. Furthermore, the statistical difference between the treatment groups 1 and 2 suggested that administration of ESCs in earlier tumor stages, when immune suppression and dysfunction was in a relatively low degree, could stimulate the immune system more effectively to generate effector lymphocytes and cytokines. Both the increase of lymphocytes and normalization of cytokine secretion help to reverse the existing immune dysfunction and rebuild the host’s antitumor immunity.
We also examined the tumor sections for the presence of CD4+ and CD8+ lymphocyte. In all groups including the control group, infiltration with CD4+ and CD8+ cells were observed. Furthermore, cell apoptosis of tumors was examined and the results seemed to correlate significantly with the proliferation of lymphocytes and rejection of tumors. That means the administration of ESCs protects mice of the vaccine and treatment groups from cancer effectively. Meanwhile, we found that mice with tumor load at different stages could all receive antitumor effect, and earlier stage means better immune response and stronger antitumor immunity.
In a further study, we found that administration of ESCs from BALB/c could also generate similar antitumor immunity against cancer in tumor loading mice, with obvious tumor suppression and retardation. We think this may be due to the similar cell-surface markers between ESCs from different species [43], which generate similar immune response in vivo. We have not observed obvious clinical signs of autoimmune diseases in the ESCs-injected mice such as irritability, weight changes, skin rash, arthritis, etc. All immunized mice were generally healthy. However, it is still difficult to illustrate a specific mechanism responsible for the tumor rejection, as it is known to be a complex one involving the coordinated roles of CD4+, CD8+ T lymphocyte, antibody-mediated cytotoxity, NK cells, phagocytic cells, and other soluble factors such as nitric oxide and cytokines [16]. Some issues are still necessary to be explored for further experiments, such as whether the immune response would be against endogenous stem cells, whether there would be specific antibody against lung cancer during the immune response, and whether this kind of immunization is cancer specific, etc.
Although more cancer cell lines are needed to be examined and multiple experiments are required to be carried out in order to make a general statement, our study does raise a possibility that administration of ESCs could effectively reverse the immune dysfunction caused by cancer and induce antitumor immunity in tumor-loading mice of different stages, which effectively prevent and/or delay the occurrence of tumors and suppress their growth when tumor masses already exist. Moreover, administration of ESCs in mice with minor tumor load received more satisfied treatment effects compared with mice with advanced tumors, which may be due to relatively intact immune system. However, it suggests a novel application of ESCs and therapeutic strategy for cancer.
Acknowledgments
This study was supported by the National High-Tech Research and Development Programme of China (Programme 863) (Grant Number 2007AA021802). We would like to thank Professor Liu of Department of Pathology for her assistance in the processing of tissue sections.
Conflict of interest
None.
Footnotes
W. Dong and J. Du contributed equally to this work.
References
- 1.Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- 2.Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA. 2003;100:3983–3988. doi: 10.1073/pnas.0530291100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD, Dirks PB. Identification of human brain tumour initiating cells. Nature. 2004;432:396–401. doi: 10.1038/nature03128. [DOI] [PubMed] [Google Scholar]
- 4.Li C, Heidt DG, Dalerba P, Burant CF, Zhang L, Adsay V, Wicha M, Clarke MF, Simeone DM. Identification of pancreatic cancer stem cells. Cancer Res. 2007;67:1030–1037. doi: 10.1158/0008-5472.CAN-06-2030. [DOI] [PubMed] [Google Scholar]
- 5.O’Brien CA, Pollett A, Gallinger S, Dick JE. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 2007;445:106–110. doi: 10.1038/nature05372. [DOI] [PubMed] [Google Scholar]
- 6.Wang S, Garcia AJ, Wu M, Lawson DA, Witte ON, Wu H. Pten deletion leads to the expansion of a prostatic stem/progenitor cell subpopulation and tumor initiation. Proc Natl Acad Sci USA. 2006;103:1480–1485. doi: 10.1073/pnas.0510652103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gibbs CP, Kukekov VG, Reith JD, Tchigrinova O, Suslov ON, Scott EW, Ghivizzani SC, Ignatova TN, Steindler DA. Stem-like cells in bone sarcomas: implications for tumorigenesis. Neoplasia. 2005;7:967–976. doi: 10.1593/neo.05394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Prince ME, Sivanandan R, Kaczorowski A, Wolf GT, Kaplan MJ, Dalerba P, Weissman IL, Clarke MF, Ailles LE. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc Natl Acad Sci USA. 2007;104:973–978. doi: 10.1073/pnas.0610117104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pardal R, Clarke MF, Morrison SJ. Applying the principles of stem-cell biology to cancer. Nat Rev Cancer. 2003;3:895–902. doi: 10.1038/nrc1232. [DOI] [PubMed] [Google Scholar]
- 10.Cabanillas R, Llorente JL. The Stem Cell Network model: clinical implications in cancer. Eur Arch Otorhinolaryngol. 2009;266:161–170. doi: 10.1007/s00405-008-0809-3. [DOI] [PubMed] [Google Scholar]
- 11.Reya T, Clevers H. Wnt signalling in stem cells and cancer. Nature. 2005;434:843–850. doi: 10.1038/nature03319. [DOI] [PubMed] [Google Scholar]
- 12.Li L, Neaves WB. Normal stem cells and cancer stem cells: the niche matters. Cancer Res. 2006;66:4553–4557. doi: 10.1158/0008-5472.CAN-05-3986. [DOI] [PubMed] [Google Scholar]
- 13.Huang EH, Heidt DG, Li CW, Simeone DM. Cancer stem cells: a new paradigm for understanding tumor progression and therapeutic resistance. Surgery. 2007;141:415–419. doi: 10.1016/j.surg.2006.12.015. [DOI] [PubMed] [Google Scholar]
- 14.Okamoto OK. Cancer stem cell genomics: the quest for early markers of malignant progression. Expert Rev Mol Diagn. 2009;9:545–554. doi: 10.1586/erm.09.40. [DOI] [PubMed] [Google Scholar]
- 15.Rubio D, Garcia-Castro J, Martin MC, de la Fuente R, Cigudosa JC, Lloyd AC, Bernad A. Spontaneous human adult stem cell transformation. Cancer Res. 2005;65:3035–3039. doi: 10.1158/0008-5472.CAN-04-4194. [DOI] [PubMed] [Google Scholar]
- 16.Li Y, Zeng H, Xu RH, Liu B, Li Z. Vaccination with human pluripotent stem cells generates a broad spectrum of immunological and clinical responses against colon cancer. Stem Cells. 2009;27:3103–3111. doi: 10.1002/stem.234. [DOI] [PubMed] [Google Scholar]
- 17.Brewer BG, Mitchell RA, Harandi A, Eaton JW. Embryonic vaccines against cancer: an early history. Exp Mol Pathol. 2009;86:192–197. doi: 10.1016/j.yexmp.2008.12.002. [DOI] [PubMed] [Google Scholar]
- 18.LeMevel BP, Wells SA., Jr Foetal antigens cross-reactive with tumour-specific transplantation antigens. Nat New Biol. 1973;244:183–184. doi: 10.1038/244183a0. [DOI] [PubMed] [Google Scholar]
- 19.Sikora K, Stern P, Lennox E. Immunoprotection by embryonal carcinoma cells for methylcholanthrene-induced murine sarcomas. Nature. 1977;269:813–815. doi: 10.1038/269813a0. [DOI] [PubMed] [Google Scholar]
- 20.Coggin JH, Jr, Adkinson L, Anderson NG. Fetal antigens shared as transplantation rejection antigens on chemically induced mouse and hamster sarcomas. Cancer Res. 1980;40:1568–1573. [PubMed] [Google Scholar]
- 21.Bertram JS, Janik P. Establishment of a cloned line of Lewis Lung Carcinoma cells adapted to cell culture. Cancer Lett. 1980;11:63–73. doi: 10.1016/0304-3835(80)90130-5. [DOI] [PubMed] [Google Scholar]
- 22.Wang Z, Tang X, Li Y, Leu C, Guo L, Zheng X, Zhu D. 20-Hydroxyeicosatetraenoic acid inhibits the apoptotic responses in pulmonary artery smooth muscle cells. Eur J Pharmacol. 2008;588:9–17. doi: 10.1016/j.ejphar.2008.03.045. [DOI] [PubMed] [Google Scholar]
- 23.Rosato A, Zoso A, Dalla Santa S, Milan G, Del Bianco P, De Salvo GL, Zanovello P. Predicting tumor outcome following cancer vaccination by monitoring quantitative and qualitative CD8+ T cell parameters. J Immunol. 2006;176:1999–2006. doi: 10.4049/jimmunol.176.3.1999. [DOI] [PubMed] [Google Scholar]
- 24.van Herck H, Baumans V, Brandt CJ, Hesp AP, Sturkenboom JH, van Lith HA, van Tintelen G, Beynen AC. Orbital sinus blood sampling in rats as performed by different animal technicians: the influence of technique and expertise. Lab Anim. 1998;32:377–386. doi: 10.1258/002367798780599794. [DOI] [PubMed] [Google Scholar]
- 25.Mayo JG. Biologic characterization of the subcutaneously implanted Lewis lung tumor. Cancer Chemother Rep. 1972;23:325–330. [PubMed] [Google Scholar]
- 26.Jaffee EM (2006) Immunotherapy of Cancer. Ann NY Acad Sci 67–72 [DOI] [PubMed]
- 27.Al-Hajj M, Becker MW, Wicha M, Weissman I, Clarke MF. Therapeutic implications of cancer stem cells. Curr Opin Genet Dev. 2004;14:43–47. doi: 10.1016/j.gde.2003.11.007. [DOI] [PubMed] [Google Scholar]
- 28.Jordan CT. Targeting the most critical cells: approaching leukemia therapy as a problem in stem cell biology. Nat Clin Pract Oncol. 2005;2:224–225. doi: 10.1038/ncponc0164. [DOI] [PubMed] [Google Scholar]
- 29.Copland M, Fraser AR, Harrison SJ, Holyoake TL. Targeting the silent minority: emerging immunotherapeutic strategies for eradication of malignant stem cells in chronic myeloid leukaemia. Cancer Immunol Immunother. 2005;54:297–306. doi: 10.1007/s00262-004-0573-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hirschowitz EA, Yannelli JR. Immunotherapy for lung cancer. Proc Am Thorac Soc. 2009;6:224–232. doi: 10.1513/pats.200806-048LC. [DOI] [PubMed] [Google Scholar]
- 31.Whiteside TL. Immune suppression in cancer: effects on immune cells, mechanisms and future therapeutic intervention. Semin Cancer Biol. 2006;16:3–15. doi: 10.1016/j.semcancer.2005.07.008. [DOI] [PubMed] [Google Scholar]
- 32.Sioud M. Does our current understanding of immune tolerance, autoimmunity, and immunosuppressive mechanisms facilitate the design of efficient cancer vaccines? Scand J Immunol. 2009;70:516–525. doi: 10.1111/j.1365-3083.2009.02326.x. [DOI] [PubMed] [Google Scholar]
- 33.Xiong G, Husseiny MI, Song L, Erdreich-Epstein A, Shackleford GM, Seeger RC, Jackel D, Hensel M, Metelitsa LS. Novel cancer vaccine based on genes of Salmonella pathogenicity island 2. Int J Cancer 126:2622–2634 [DOI] [PMC free article] [PubMed]
- 34.Johnsen A, France J, Sy MS, Harding CV. Down-regulation of the transporter for antigen presentation, proteasome subunits, and class I major histocompatibility complex in tumor cell lines. Cancer Res. 1998;58:3660–3667. [PubMed] [Google Scholar]
- 35.Drukker M, Katchman H, Katz G, Even-Tov Friedman S, Shezen E, Hornstein E, Mandelboim O, Reisner Y, Benvenisty N. Human embryonic stem cells and their differentiated derivatives are less susceptible to immune rejection than adult cells. Stem Cells. 2006;24:221–229. doi: 10.1634/stemcells.2005-0188. [DOI] [PubMed] [Google Scholar]
- 36.Drukker M, Benvenisty N. The immunogenicity of human embryonic stem-derived cells. Trends Biotechnol. 2004;22:136–141. doi: 10.1016/j.tibtech.2004.01.003. [DOI] [PubMed] [Google Scholar]
- 37.Fairchild PJ, Cartland S, Nolan KF, Waldmann H. Embryonic stem cells and the challenge of transplantation tolerance. Trends Immunol. 2004;25:465–470. doi: 10.1016/j.it.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 38.Boyd AS, Higashi Y, Wood KJ. Transplanting stem cells: potential targets for immune attack. Modulating the immune response against embryonic stem cell transplantation. Adv Drug Deliv Rev. 2005;57:1944–1969. doi: 10.1016/j.addr.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 39.Begley J, Ribas A. Targeted therapies to improve tumor immunotherapy. Clin Cancer Res. 2008;14:4385–4391. doi: 10.1158/1078-0432.CCR-07-4804. [DOI] [PubMed] [Google Scholar]
- 40.Rosenberg SA, Yang JC, Restifo NP. Cancer immunotherapy: moving beyond current vaccines. Nat Med. 2004;10:909–915. doi: 10.1038/nm1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ikeda H, Chamoto K, Tsuji T, Suzuki Y, Wakita D, Takeshima T, Nishimura T. The critical role of type-1 innate and acquired immunity in tumor immunotherapy. Cancer Sci. 2004;95:697–703. doi: 10.1111/j.1349-7006.2004.tb03248.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Shurin MR, Lu L, Kalinski P, Stewart-Akers AM, Lotze MT. Th1/Th2 balance in cancer, transplantation and pregnancy. Springer Semin Immunopathol. 1999;21:339–359. doi: 10.1007/BF00812261. [DOI] [PubMed] [Google Scholar]
- 43.Koestenbauer S, Zech NH, Juch H, Vanderzwalmen P, Schoonjans L, Dohr G. Embryonic stem cells: similarities and differences between human and murine embryonic stem cells. Am J Reprod Immunol. 2006;55:169–180. doi: 10.1111/j.1600-0897.2005.00354.x. [DOI] [PubMed] [Google Scholar]