Abstract
Natural killer (NK) cells represent a promising cell type to utilize for effective adoptive immunotherapy. However, little is known about the important cytolytic molecules and signaling pathways used by NK cells in the adoptive transfer setting. To address this issue, we developed a novel mouse model to investigate the trafficking and mechanism of action of these cells. We demonstrate that methylcholanthrene-induced RKIK sarcoma cells were susceptible to NK cell-mediated lysis in vitro and in vivo following adoptive transfer of NK cells in C57BL/6 RAG-2−/−γc−/− mice. Cytotoxic molecules perforin, granzymes B and M as well as the death ligand TRAIL and pro-inflammatory cytokine IFN-γ were found to be important in the anti-tumor effect mediated by adoptively transferred NK cells. Importantly, we demonstrate that adoptively transferred NK cells could traffic to the tumor site and persisted in vivo which correlated with the anti-tumor effect observed. Overall, the results of this study have important implications for enhancing NK cell-based immunotherapies.
Keywords: Adoptive immunotherapy, MCA-induced sarcoma, Perforin, TRAIL, Granzymes, Natural killer cells
Introduction
Natural killer (NK) cells are lymphocytes that comprise part of the innate immune system [1]. NK cells express activatory and inhibitory receptors that recognize ligands including cellular stress ligands and MHC class I molecules [2]. These receptors provide a balance of signals leading to either activation or inhibition of the NK cell. These recognition receptors allow NK cells to recognize malignant and pathogen-infected tissue with a degree of specificity to perform an important role in the body’s first line of defense.
Activating NK cell receptors, such as NKG2D, recognize cellular stress ligands that can be induced in conditions of viral infection or malignant transformation, through the heat shock and DNA damage response pathways [3–5]. Upon activation, NK cells can proliferate and secrete cytokines, such as IFN-γ, TNF-α and GM-CSF, which can induce inflammation and stimulate an endogenous immune response. Lysis of target cells by NK cells has been shown to occur through different death pathways, including granule exocytosis involving perforin, as well as through death receptors, such as TRAIL [6, 7]. NK cells are capable of a rapid cytotoxic response following recognition of tumor cells and have been shown to eradicate grafted tumors in vivo and control the growth of carcinogen-induced tumors in mouse models [8, 9]. Combined with the knowledge that NK cells are important in the adaptive–innate crosstalk, NK cells are an attractive option for exploitation in the development of immunotherapy [10].
Immunotherapeutic strategies to maximize NK cell anti-tumor activity have included the use of cytokines aimed at activating NK cells for the treatment of cancer [11]. Other approaches have included the adoptive transfer of autologous, ex vivo, IL-2 lymphokine-activated killer cells (LAK) [12]. In general, these approaches have resulted in only moderate success in restricted numbers of patients and in mouse models [13, 14]. The utilization of NK cells in allogeneic hematopoietic stem cell transplantation has shown more promise and has resulted in improved outcomes for leukemia patients [15, 16]. With improved methodologies for the ex vivo expansion of primary human NK cells, further utilization of adoptively transferred NK cells for anti-cancer immunotherapy is now a viable option [17–20]. Nevertheless, despite the fact that adoptively transferred NK cells can mediate anti-tumor effects, there has been little insight into the trafficking and mechanism of action of transferred NK cells, mainly due to lack of a robust mouse model. Understanding the important molecules and signaling pathways employed by adoptively transferred NK cells could reveal novel strategies for optimizing the use of these cells for anti-cancer immunotherapy.
In this study we have developed a mouse model involving NK cell-mediated inhibition of tumor growth. This involved the use of a C57BL/6 RAG2−/−γc−/− mouse model which lacked endogenous lymphocytes including NK cells. Importantly, this model allowed us to focus entirely on the mechanistic contribution from adoptively transferred NK cells to tumor inhibition. Two methylcholanthrene (MCA)-induced RKIK sarcoma cell lines were found to be sensitive to NK cell-mediated lysis in vitro and the growth of RKIK tumor cells was significantly inhibited in vivo following adoptive transfer of IL-2-activated NK cells. We utilized this tumor model to investigate the mechanism of action of adoptively transferred NK cells. We explored the important molecules in the in vivo anti-tumor response mediated by adoptively transferred NK cells and assessed the ability of adoptively transferred NK cells to persist and traffic to the tumor site. This study provides important information for effectively utilizing adoptively transferred NK cells for cancer immunotherapy.
Materials and methods
Cell lines
The C57BL/6 murine T cell lymphoma cell line RMA-S was maintained in complete DMEM medium containing 10% (v/v) FCS, 2 mM l-glutamine (Gibco, Grand Island, NY, USA), 100 U/ml penicillin and 100 μg/ml streptomycin (Sigma-Aldrich, Castle Hill, NSW, Australia) [21]. The MCA-induced sarcoma cell lines RKIK-4654 and RKIK-4862 were isolated from C57BL/6 RAG-2−/−γc−/− mice and were a kind gift from Professor Robert Schreiber (University of Washington, St Louis, MO, USA). The mouse Moloney leukemia virus-induced lymphoma cell line YAC-1 was purchased from the American Tissue Culture Collection (ATCC, Manassas, VA, USA). YAC-1 and RKIK cell lines were maintained in RPMI 1640 Medium (Gibco), with 10% FCS (v/v), 2 mM l-glutamine, 0.1 mM non-essential amino acids (Gibco), 1 mM sodium pyruvate (Gibco), 100 U/ml penicillin, 100 μg/ml streptomycin and 5 × 10−5 M 2-mercamercaptoethanol (2ME).
Mice
C57BL/6 wildtype (WT) and C57BL/6-ptprc a mice were purchased from the Walter and Eliza Hall Institute of Medical Research (WEHI, Parkville, VIC, Australia). C57BL/6 perforin-deficient (pfp−/−), C57BL/6 interferon-γ-deficient (IFN-γ−/−), C57BL/6 TRAIL-deficient (TRAIL−/−), C57BL/6 Granzyme A-deficient (GzmA−/−), Granzyme B-deficient (GzmB−/−) and Granzyme M-deficient (GzmM−/−), C57BL/6 gld mice [Fas Ligand (FasL) mutant] and C57BL/6 RAG-2−/−γc−/− mice were bred in house, at the Peter MacCallum Cancer Centre (PMCC). All mice were bred and housed in specific pathogen-free conditions at the PMCC and mice 6–14 weeks of age were used in all experiments, according to Animal Experimentation Ethics Committee guidelines.
Isolation of NK cells
Spleens dissected from C57BL/6 or C57BL/6 mutant mice were crushed into hypotonic lysis buffer. Following washing with PBS, the cells were filtered to generate a single cell suspension. NK cells were then isolated using an NK cell isolation beading kit (Miltenyi Biotech, Bergisch Gladbach, Germany) and separated using an autoMACS (Miltenyi Biotech). The NK cells were cultured in RPMI 1640 medium containing 10% (v/v) FCS, 2 mM l-glutamine, 5 × 10−5 mM 2ME, 100 U/ml penicillin, 100 μg/ml streptomycin, 2 mM Hepes (Sigma-Aldrich) and 1,000 IU/ml recombinant human IL-2 (rhIL-2) (Biological Resources Branch Preclinical Repository, National Cancer Institute, Frederick, MD) for 5 days before being used in experiments.
Flow cytometry
Expression of NK cell markers including activating and inhibitory receptors on NK cells was determined using flow cytometry. Antibodies were used according to manufacturer’s instructions and clone numbers for each antibody are shown in brackets. This included FITC-conjugated antibodies specific for NK1.1 (PK136), NKG2A/C/E (20d5) (both from eBioscience, San Diego, CA, USA), 2B4/CD244 (2B4), CD16/32 (93), Ly49D (4ES/E1) and CD69 (H1.2F3) (all from BD Pharmingen, San Jose, CA, USA). Other reagents used in this study were PE-conjugated antibodies specific for DX5 (DX5) (BD Pharmingen), CD94 (18d3), NKG2D (CX5) and TRAIL (N2B2) (all from eBioscience). Biotinylated antibodies specific for CD11b (M1/70), KLRG1 (2F1) and CD27 (LG.7F9), followed by staining with streptavidin conjugated to perCPCy5.5 for detection were used (all from eBioscience), and an APCCy7-conjugated antibody specific for B220 (RA3-6B2) (BD Pharmingen). Expression of NK cell ligands on RKIK tumor cells was detected using PE-conjugated antibodies specific for DR5 (MD5.1) (eBioscience), H-2Kb (AF6.88.5) (BD Pharmingen) and pan RAE-1 (186107) (RnD systems, Minneapolis, MN, USA), and a PE-conjugated NKG2D tetramer (a kind gift from Professor David Raulet, University of California Berkeley, CA, USA).
Cytotoxicity
The ability of NK cells isolated from C56BL/6 WT or C57BL/6 mutant mice to specifically lyse tumor targets was assessed in a 51Chromium (51Cr) release assay as described previously [22]. Briefly, tumor targets were radio-labeled with 51Cr and then co-incubated with NK cells at various effector:target (E:T) ratios. Each E:T ratio was set up in triplicate wells of a 96-well U-bottom plate, in a total volume of 200 μl of supplemented RPMI. Following 4 h incubation, the supernatant was harvested and the percentage of specific release of 51Cr into the supernatant was then calculated as performed previously [22].
Adoptive cell transfer
Groups of 5–10 C57BL/6 RAG-2−/−γc−/− mice were injected subcutaneously (s.c.) with 4 × 105 RKIK-4862 tumor cells. Mice were then treated on days 0 and 1 with 3 × 106 (per injection) of 5 day IL-2-activated NK cells injected intravenously (i.v.) derived from WT or mutant mice. To block NKG2D interactions, 250 μg of monoclonal antibody specific for NKG2D (C7) or isotype control antibody (UC8.1B9) was injected intraperitoneally on days 0, 1 and 3 followed by subsequent twice weekly injections, as described previously [23]. To investigate the trafficking and persistence of adoptively transferred NK cells in vivo, 3 × 106 donor NK cells from congenic C57BL/6-ptprc a (CD45.1+) mice were transferred into RKIK-4862 tumor-bearing C57BL/6 RAG-2−/−γc−/− recipient mice (CD45.2+) on days 0 and 1. Three mice from each group were killed on days 3, 6, 9, 15 and 20 and spleen, liver, lungs and peripheral blood from cardiac puncture were collected. Dissected spleens and livers were crushed into hypotonic lysis buffer and filtered to create a single cell suspension. Lung tissue was minced with scalpel blades followed by dissociation by shaking for 90 min at 37°C in 10 ml RPMI (Gibco) with 100 μg/ml hyaluronidase and 0.03 g collagenase IV (both from Worthington Biochemical Corporation, Lakewood, New Jersey, USA). Hypotonic lysis buffer was used to lyse red blood cells from the cardiac punctures. Single cell suspensions were then stained with FITC-conjugated antibody specific for CD45.1 (clone A20, eBiosciences). Flow cytometry was performed to determine the percentage of CD45.1+ cells in the organs. Enumeration of CD45.1+ cells in these samples was calculated using CaliBRITE beads (BD scientific) as per the manufacturer’s instructions. To determine the presence of NK cells in the tumor, 3 × 106 donor NK cells from congenic C57BL/6-ptprc a mice were transferred into RKIK-4862 tumor-bearing C57BL/6 RAG-2−/−γc−/− recipient mice on days 0 and 1. At day 17, the mice were killed and tumor was collected and minced with scalpel blades followed by dissociation by shaking for 90 min at 37°C in 10 ml RPMI (Gibco) with 100 μg/ml hyaluronidase and 0.03 g Collagenase IV (Worthington). Single cell suspensions were then stained with FITC-conjugated antibody specific for CD45.1. Flow cytometry was performed to determine the percentage of CD45.1+ cells in the tumor.
Statistical analysis
The Mann–Whitney test was used for statistical analysis, using GraphPad prism software. Two tailed P values (p2) less than 0.05 were considered significant.
Results
Phenotypic characterization of RKIK tumor cells and IL-2-activated NK effector cells
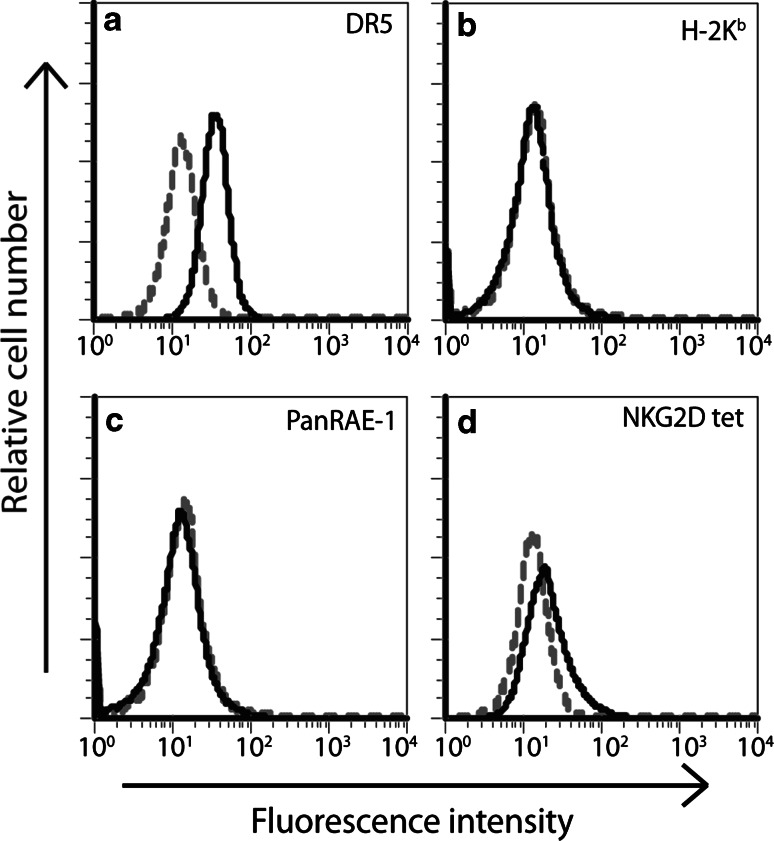
We utilized two MCA-induced sarcoma cell lines that were isolated from C57BL/6 RAG-2−/−γc−/− mice, RKIK-4862 and RKIK-4654. The phenotype of these RKIK tumor lines was determined by flow cytometry. We demonstrated that the RKIK-4862 cell line expressed the TRAIL ligand, DR5 (Fig. 1a) but does not express MHC class I (Fig. 1b). Interestingly, we could not detect expression of RAE-1, a NKG2D ligand, on RKIK-4862 cells following staining with a panRAE-1 antibody (Fig. 1c). However, low NKG2D tetramer staining was detected (Fig. 1d), suggesting expression of an alternative NKG2D ligand. A similar phenotype was observed for the RKIK-4654 cell line (data not shown). Given that MHC class I could not be detected on these cells, the results suggested that these RKIK cell lines may be good targets for NK cell-mediated cytolysis.
Fig. 1.
Phenotypic characterization of the MCA-induced sarcoma cell line, RKIK-4862. Expression of TRAIL receptor DR5 (a), MHC Class I H2Kb (b), RAE1 molecules (c) and NKG2D ligands (d) was determined by staining with specific PE-conjugated antibodies or PE-conjugated NKG2D tetramer (solid lines). Isotype control-stained cells (dotted lines) were used as a control. Data shown are representative of three independent experiments
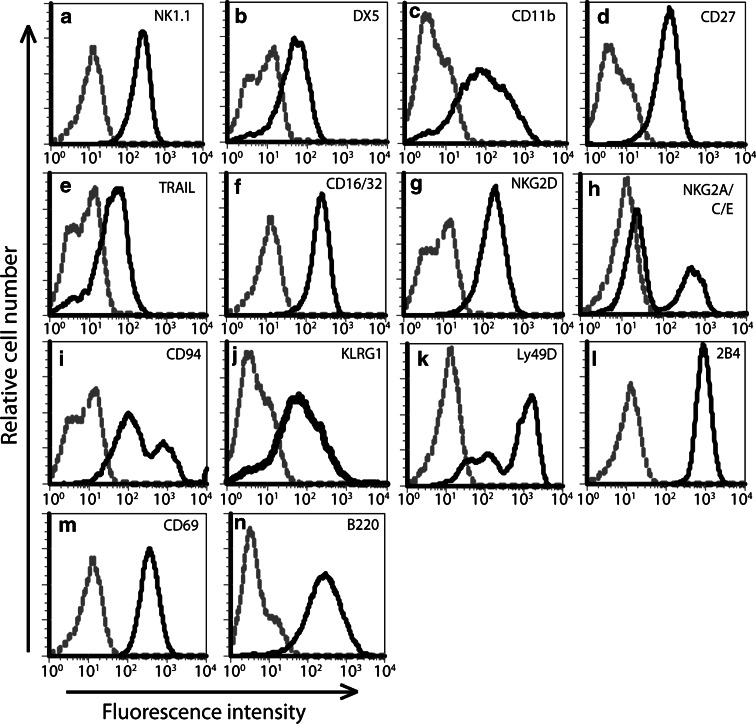
We next characterized the phenotype of NK cells cultured for 5 days in 1,000 IU/ml rhIL-2. In three independent experiments, the population was found to be entirely NK1.1 positive (Fig. 2a), and expressed the NK cell-associated markers DX5, CD11b and CD27 (Fig. 2b–d). In addition, these cells expressed the death ligand TRAIL (Fig. 2e), NK cell activating receptors CD16/32 and NKG2D (Fig. 2f, g) as well as both NKG2A/C/E and CD94 (Fig. 2h, i), a family of receptors that are either activatory (NKG2C/E) or inhibitory (NKG2A). Expression of receptors KLRG1, Ly49D and 2B4 (Fig. 2j–l) and activation markers CD69 and B220 (Fig. 2m, n) were also observed. These results demonstrated that our cell isolation and culture conditions could reproducibly produce a pure and highly activated population of NK cells.
Fig. 2.
Phenotypic characterization of IL-2-activated murine primary NK cells. The surface expression of various NK cell markers (a) NK1.1 (b) DX5 and (c) CD11b, activatory receptors (d) CD27, (e) TRAIL, (f) CD16/32, (g) NKG2D, activatory and inhibitory receptors (h NKG2A/C/E, i CD94, j KLRG1, k Ly49D, l 2B4), as well as activation status markers (m) CD69 and (n) B220, was analyzed by flow cytometry following staining with specific antibodies (solid line). Isotype control-stained NK cells were used as a control. Data shown are representative of three independent experiments
RKIK tumor cells are sensitive to NK cell-mediated lysis in vitro and in vivo
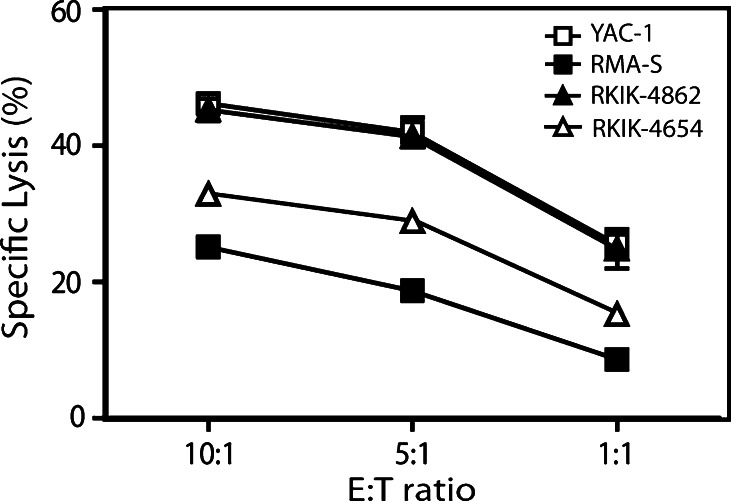
The ability of 5-day, IL-2-activated NK cells to lyse RKIK cells was compared to the lysis of two well-characterized NK cell-sensitive cell lines, YAC-1 and RMA-S, in a 51Cr release assay. We found that NK cells could effectively mediate lysis of both RKIK tumor cell lines although the sensitivity of RKIK-4862 to NK cell lysis was higher than for RKIK-4654 tumor cells (Fig. 3). Although the phenotype of the RKIK cell lines appeared similar, it was possible that expression of other ligands for activating and/or inhibitory NK cell receptors on these two lines may have accounted for differences in NK cell sensitivity observed. The NK cell-mediated lysis of RKIK-4862 sarcoma cells was demonstrated to be equivalent to that observed for YAC-1 cells and lysis of both RKIK cell lines was higher than that observed for RMA-S (Fig. 3). Further in vivo characterization was undertaken utilizing the RKIK-4862 cell line given its greater sensitivity to NK cell-mediated lysis.
Fig. 3.
In vitro sensitivity of RKIK tumor cells to NK cell-mediated lysis. Lysis of RKIK-4862 and RKIK-4654 tumor cells by IL-2-activated NK cells was compared to lysis of YAC-1 and RMA-S tumor cell lines using a 51Cr release assay. Lysis of RKIK-4862 sarcoma cells (closed triangles) by NK cells was equivalent to the killing of YAC-1 lymphoma cells (open squares), and higher than that observed for the RKIK-4654 sarcoma cell line (open triangles) and RMA-S tumor cells (closed squares). Data shown are average of triplicate wells ± SEM and are representative of two independent experiments
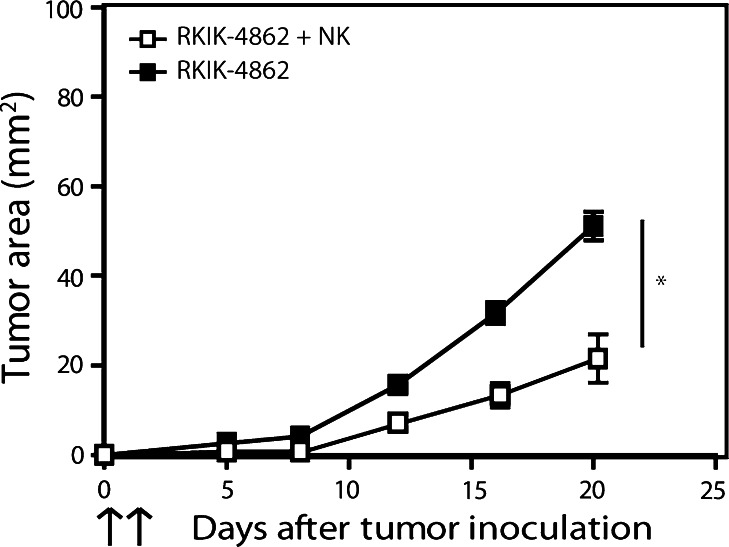
In the next experiment, we tested the capacity of IL-2-activated NK cells to inhibit RKIK tumor growth following adoptive transfer. C57BL/6 RAG-2−/−γc−/− mice bearing subcutaneous RKIK-4862 tumor were treated systemically with 3 × 106 IL-2-activated NK cells. Significant inhibition of tumor growth was observed following adoptive transfer of NK cells compared to untreated mice (Fig. 4). These results demonstrate that utilization of the RKIK tumor model is appropriate to determine NK cell-mediated anti-tumor function both in vitro and in vivo.
Fig. 4.
Adoptively transferred NK cells can inhibit the growth of RKIK-4862 tumor in vivo. The ability of adoptively transferred NK cells to inhibit the growth of RKIK-4862 tumor cells was determined by s.c. injection of tumor cells followed by intravenous injection of 3 × 106 IL-2-activated NK cells (days 0 and 1). Adoptively transferred NK cells were able to significantly inhibit the growth of s.c. RKIK-4862 tumor (open squares) compared to untreated tumor (closed squares, *p2 < 0.05). Results shown are average tumor area ± SEM from 16 and 12 mice for untreated and treated mice, respectively, from two independent experiments
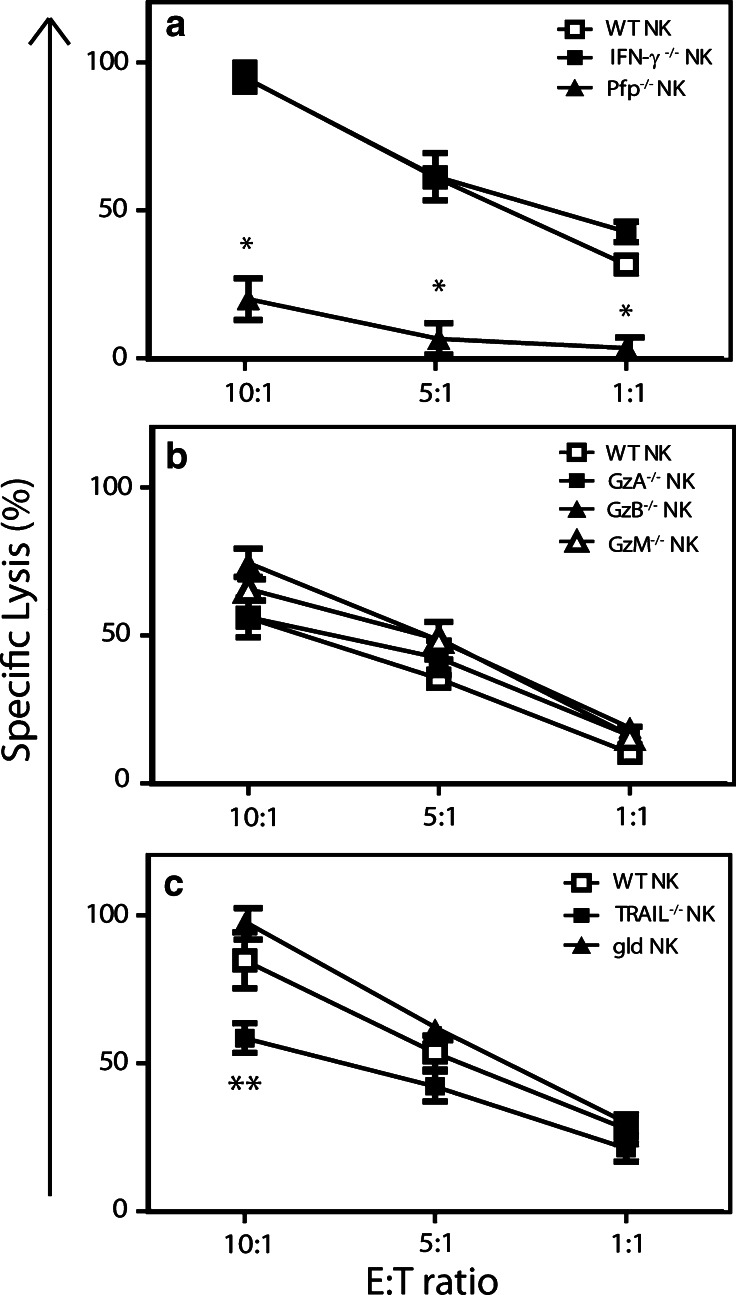
Perforin is required for the NK cell-mediated lysis of RKIK tumor cells in vitro
Previous studies have shown that NK cells can mediate lysis of target cells using the granule exocytosis pathway involving perforin and granzymes, or death receptors including FasL and TRAIL, although this has not been determined utilizing adoptively transferred NK cells [24–26]. To determine which cytolytic pathways and molecules were important in the anti-tumor effects mediated by adoptively transferred NK cells, we compared the ability of NK cells derived from either C56BL/6 wildtype (WT) mice or various C57BL/6 gene-deficient mice to lyse RKIK-4862 target cells. Importantly, the NK cells derived from both WT and gene-targeted mice had a similar phenotype and viability following IL-2 activation (data not shown).
To determine whether perforin was important in NK cell-mediated lysis of RKIK-4862 cells, we first compared NK cells from WT and perforin-deficient (pfp−/−) mice. Cytotoxicity of RKIK-4862 cells by pfp−/− NK cells was significantly reduced compared with WT NK cells (Fig. 5a). Interestingly, equivalent lysis of RKIK-4862 tumor cells by NK cells derived from WT and IFN-γ-deficient mice was observed suggesting that IFN-γ secretion by NK cells was not important for their cytolytic activity in vitro (Fig. 5a). Other molecules important in the granule exocytosis pathway include granzymes, which are serine proteases that promote the apoptosis of target cells [27]. Therefore, we investigated whether granzymes (Gzm) A, B and M are important in the NK cell-mediated lysis of RKIK-4862 cells in vitro. We did this by comparing the lytic capacity of NK cells from GzmA-, GzmB- and GzmM-deficient mice with WT NK cells. In this experiment we found that NK cells from WT and each Gzm knockout mice could equivalently lyse RKIK-4862 tumor cells (Fig. 5b). Expression of death ligands on NK cells such as TRAIL and FasL has been demonstrated to be important in the control of some tumors [26, 28]. The expression of TRAIL on IL-2-activated NK cells and TRAIL receptor, DR5, on RKIK-4862 tumor cells raised the possibility that this pathway may be important in our model. To investigate whether TRAIL and/or FasL are important in the lysis of RKIK-4862 cells, we compared the lytic capacity of NK cells isolated from WT and TRAIL-deficient (TRAIL−/−) or gld mice, which express mutant FasL [29]. There was no significant difference in the lysis of RKIK-4862 mediated by NK cells from gld or TRAIL−/− mice compared to WT NK cells although there was a trend toward reduced killing of RKIK-4862 cells by TRAIL−/− NK cells at the highest effector:target ratio tested (Fig. 5c). Overall, these in vitro results demonstrate that perforin is a critical molecule required for NK cell-mediated lysis of RKIK-4862 sarcoma cells.
Fig. 5.
A critical role for perforin in the NK cell-mediated lysis of RKIK-4862 in vitro. The role of cytolytic molecules perforin, IFN-γ, granzymes (Gzm) A, B and M and death ligands TRAIL and FasL, in the NK cell-mediated lysis of RKIK-4862 cells was determined by isolating NK cells from mice deficient in these molecules. Lysis of RKIK-4862 tumor cells was determined using a 51Cr release assay. a WT NK (open squares) and IFN-γ−/− NK cells (closed squares) were able to lyse RKIK-4862 at similar levels. In contrast, pfp−/− NK cells (closed triangles) were unable to lyse these cells. b WT NK cells (open squares) and GzmA−/− (closed squares), GzmB−/− (closed triangles) or GzmM−/− (open triangles) NK cells were able to equivalently lyse RKIK-4862 tumor cells. c WT NK cells (open squares), gld NK cells (closed triangles) and TRAIL−/− NK cells (closed squares) were able to similarly lyse RKIK-4862 tumor cells. Results shown are average specific lysis ± SEM from triplicate wells and are representative of three independent experiments, *p2 < 0.05 and **p2 > 0.05
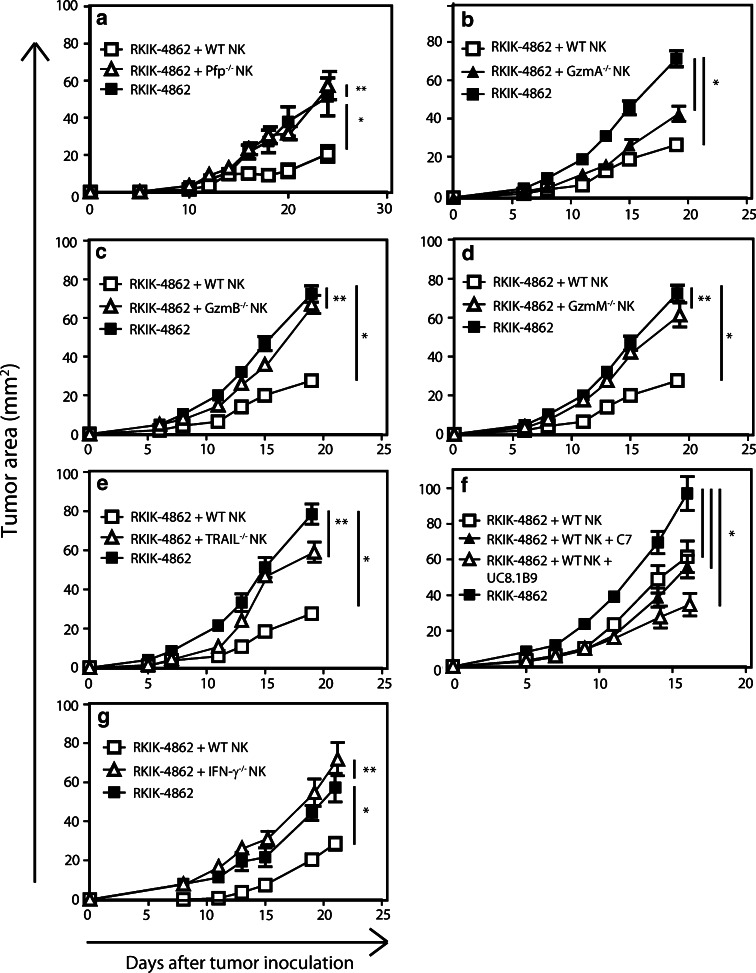
Cytolytic effector molecules perforin, TRAIL and granzymes B and M are important for the anti-tumor response mediated by adoptively transferred NK cells
Given that in vitro assays cannot closely mimic important cellular interactions in vivo, we next explored whether the cytotoxic molecules tested in vitro were playing a role in the NK cell-mediated rejection of tumor in vivo. In these experiments, we compared the ability of adoptively transferred NK cells derived from C57BL/6 WT or various C57BL/6 gene-targeted mice to inhibit RKIK-4862 tumor growth. We found that NK cells derived from pfp−/− mice were unable to inhibit the growth of s.c. tumor compared to WT NK cells (Fig. 6a). This directly correlated with our in vitro results and demonstrated that perforin is important in the anti-tumor response mediated by adoptively transferred NK cells. To determine whether granzymes play a role in the NK cell-mediated inhibition of RKIK-4862 tumor growth, we compared the anti-tumor function of adoptively transferred NK cells from GzmA−/−, GzmB−/− or GzmM−/− mice with NK cells from WT mice. There was no significant difference between NK cells from GzmA−/− mice and WT NK cells to inhibit the growth of RKIK-4862 tumor (Fig. 6b). Interestingly, adoptively transferred NK cells from GzmB−/− and GzmM−/− mice were unable to inhibit the growth of RKIK-4862 tumor compared to WT NK cells (Fig. 6c, d). Our results suggested that GzmB and GzmM play an important role in anti-tumor function of adoptively transferred NK cells. We next investigated whether the TRAIL ligand was playing a role in the NK cell-mediated control of RKIK-4862 cells in vivo. In this experiment, TRAIL−/− NK cells were unable to inhibit RKIK-4862 tumor growth compared to WT NK cells suggesting an important role for the TRAIL death pathway (Fig. 6e). In further experiments we explored whether other receptor–ligand interactions may be important in the anti-tumor response mediated by adoptively transferred NK cells. Given that activated NK cells expressed high levels of NKG2D and there was some positive NKG2D tetramer staining on RKIK-4862 tumor cells, we next investigated whether this pathway may be involved. In this experiment we adoptively transferred WT NK cells followed by injection of monoclonal antibody C7, which specifically blocks the NKG2D receptor or an isotype control antibody (UC8.1B9). Interestingly, blocking NKG2D did not significantly affect the ability of WT NK cells to inhibit the growth of tumor as inhibition of RKIK-4862 tumor growth was similar to mice treated with isotype control antibody (Fig. 6f). Overall, the results in our RKIK-4862 tumor model demonstrated that the granule exocytosis pathway involving perforin and Gzms B and M and the TRAIL death pathway play important roles in the anti-tumor response mediated by adoptively transferred NK cells.
Fig. 6.
Cytolytic effector molecules important for inhibition of RKIK-4862 tumor growth by adoptively transferred NK cells. To investigate the cytolytic effector molecules important for the inhibition of RKIK-4862 tumor growth, C57BL/6 RAG-2−/−γc−/− recipient mice bearing tumor were injected with NK cells derived from pfp−/−, IFN-γ−/−, TRAIL−/−, GzmA−/−, GzmB−/−, GzmM−/− or WT mice on days 0 and 1. To investigate the role of the NKG2D pathway, mice were treated with WT NK cells and intraperitoneal injections of the NKG2D blocking antibody C7 or isotype control antibody UC8.1B9. In all experiments adoptive transfer of WT NK cell-treated mice (open squares) significantly inhibited RKIK-4862 tumor growth compared to untreated mice (closed squares). a Adoptive transfer of pfp−/− NK cells (open triangles) did not inhibit RKIK-4862 tumor growth compared to untreated mice (closed squares). b Adoptive transfer of GzmA−/− NK cells (closed triangles) and WT NK cells (open squares) both led to significant inhibition of RKIK-4862 tumor growth compared to untreated mice (closed squares). c Adoptive transfer of GzmB−/− NK cells (open triangles) did not inhibit RKIK-4862 tumor growth compared to untreated mice (closed squares). d Adoptive transfer of GzmM−/− NK cells (open triangles) did not inhibit RKIK-4862 tumor growth compared to untreated mice (closed squares). e Adoptive transfer of TRAIL−/− NK cells (open triangles) did not inhibit RKIK-4862 tumor growth compared to untreated mice (closed squares). f Adoptive transfer of WT NK cells with NKG2D blocking antibody C7 (closed triangles) or isotype control antibody UC8.1B9 (open triangles) led to significant inhibition of RKIK-4862 tumor growth compared to untreated mice (closed squares). g Adoptive transfer of IFN-γ−/− NK cells (open triangles) did not inhibit RKIK-4862 tumor growth compared to untreated mice (closed squares). Data shown are average tumor area ± SEM from 5 to 15 mice from two independent experiments, **p2 > 0.05 and *p2 < 0.05
A critical role for IFN-γ in the anti-tumor effect mediated by adoptively transferred NK cells
Previous studies in vitro have demonstrated an important role for IFN-γ secreted by activated human NK cells for their activity against tumor cells [30]. Although we found no effect on lysis of RKIK-4862 tumor cells by IFN-γ−/− NK cells compared to WT NK cells, IFN-γ has been reported to play an important role in the anti-tumor immune response in vivo [31, 32]. To determine the importance of this pro-inflammatory cytokine in vivo against RKIK-4862 tumor cells, we adoptively transferred NK cells isolated from WT and IFN-γ−/− mice. We found that adoptive transfer of IFN-γ−/− NK cells could not mediate inhibition of RKIK-4862 tumor growth compared to WT NK cells (Fig. 6g). This experiment demonstrated the importance of IFN-γ secreted by adoptively transferred NK cells in the inhibition of RKIK-4862 tumor growth in vivo.
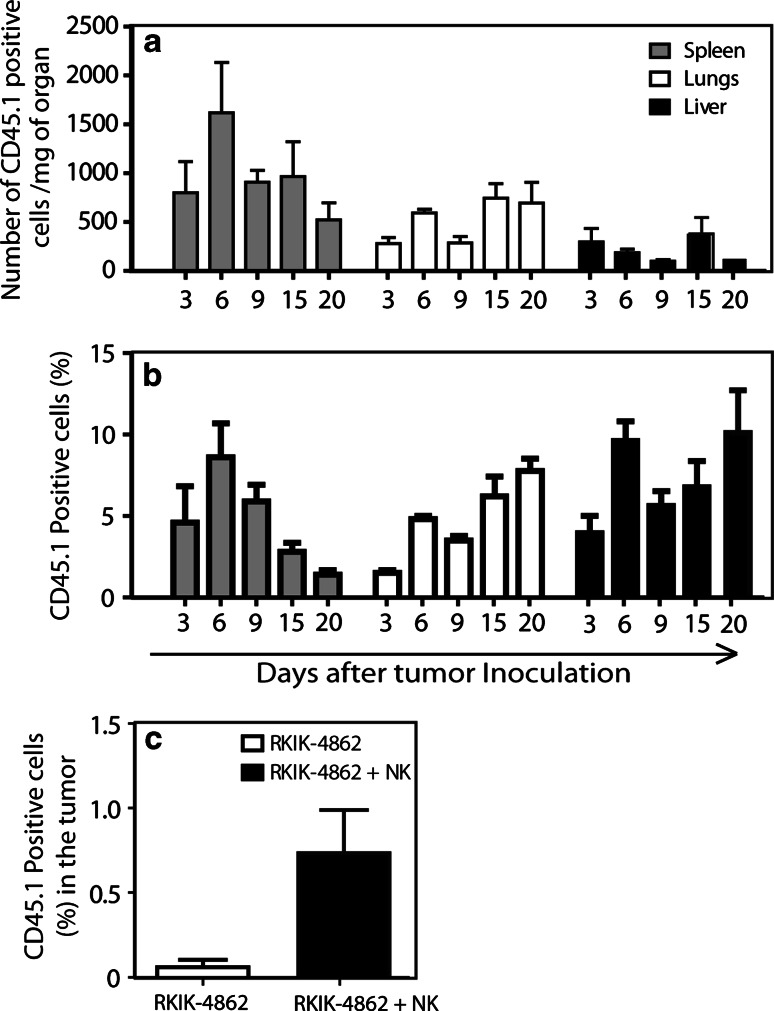
Adoptively transferred NK cells traffic to the tumor site and persist in vivo
It has been previously shown for adoptively transferred T cells that successful anti-tumor responses correlate with their trafficking to the tumor site and persistence in vivo [33, 34]. To determine whether this was the case for adoptively transferred NK cells in this specialized model, congenic (CD45.1+) NK cells were transferred into RKIK-4862 tumor-bearing mice and flow cytometry was utilized to determine the presence of NK cells in the peripheral blood, lungs, liver, spleen and tumor of treated mice. In this experiment up to 5% of cells in the peripheral blood were CD45.1+ and could be detected for at least 20 days post-transfer. These CD45.1+ cells were not detected in untreated RKIK-4862 tumor-bearing mice (Fig. 7a, b). Transferred CD45.1+ NK cells could also be found in the spleen, lungs and liver of treated mice at least 20 days post-transfer (Fig. 8a, b). No CD45.1+ cells were found in these various tissues in non-treated tumor-bearing mice (data not shown). Importantly, we demonstrated that adoptively transferred NK cells could traffic to the tumor site as CD45.1+ cells could be detected in the tumor mass 17 days post-transfer, but were not found in untreated tumor-bearing mice (Fig. 8c). These results strongly suggest that anti-tumor responses mediated by adoptively transferred NK cells correlate with their trafficking to the tumor site and persistence in vivo.
Fig. 7.
Adoptively transferred NK cells persist in the peripheral blood of recipient mice. The persistence of adoptively transferred NK cells was investigated by transfer of congenic NK cells from C57BL/6-ptprc a mice (CD45.1+) into RKIK-4862 tumor-bearing C57BL/6 RAG-2−/−γc−/− mice (CD45.2+). Flow cytometry was used to detect CD45.1+ NK cells in the peripheral blood of NK cell-treated mice (closed bars) or non-treated mice (open bars). Adoptively transferred NK cells persist at detectable levels for at least 20 days post-transfer with respect to a the percentage of CD45.1+ circulating cells and b the number of CD45.1+ circulating cells per microliter of blood. Data shown are average ± SEM of six mice per time point, from two independent experiments
Fig. 8.
Adoptively transferred NK cells can be detected in the liver, lungs, spleen and tumor of recipient mice. The presence of adoptively transferred NK cells in the spleens (gray bars), lungs (white bars) and livers (black bars) of mice receiving RKIK-4862 tumor and adoptively transferred, congenic CD45.1+ NK cells was investigated by flow cytometry. Adoptively transferred NK cells persist at detectable levels for at least 20 days post-transfer with respect to a the percentage of CD45.1+ cells in each organ and b the number of CD45.1+ circulating cells per milligram of organ. Data shown are average ± SEM from six mice from two independent experiments. c The presence of adoptively transferred NK cells in the tumor mass was investigated by flow cytometry to detect CD45.1+ NK cells following mechanical and enzymatic dissociation of the tumors from mice receiving RKIK-4862 tumor and adoptively transferred, congenic CD45.1+ NK cells (black bars) or RKIK-4862 alone (white bars) at day 17, post-transfer. Adoptively transferred NK cells could be detected in the tumor at this time point. p2 < 0.01 (NK cell-treated vs. non-treated groups). Data shown are average ± SEM from five mice per group
Discussion
Adoptive cell transfer therapy is a rapidly developing field of immunotherapy. Indeed, transfer of tumor-infiltrating lymphocytes has shown some remarkable responses in patients with advanced melanoma [35, 36]. Nevertheless, problems with deriving sufficient numbers of these cells from patients, downregulation of MHC class I ligands and co-stimulatory ligands on tumor cells, combined with the lack of persistence of transferred cells have precluded broad utilization of these cells for the treatment of cancer patients. The transfer of other immune subsets, such as NK cells which do not require prior sensitization to respond to target cells and can potently induce a cytolytic response, represents a good alternate or auxiliary cell type for immunotherapy. The use of NK cells in the allogeneic hematopoietic stem cell transplant setting has resulted in improved survival outcomes for leukemia patients, and highlights the promise of these cells for cancer treatment. However, little is known about the important effector molecules and signaling pathways used by adoptively transferred NK cells in mediating anti-tumor responses. This knowledge may serve to help optimize the use of these cells for immunotherapy and help in the selection of tumor-types suitable for targeting by adoptively transferred NK cells. To investigate this, we developed a novel mouse model where adoptively transferred NK cells could mediate significant tumor growth inhibition. We demonstrated that a MCA-induced sarcoma cell line, RKIK-4862, that expressed TRAIL receptor, DR5, and no MHC class I, was sensitive to NK cell-mediated lysis both in vitro and in vivo following adoptive transfer of activated NK cells. The development of this novel model allowed us to investigate the behavior of adoptively transferred NK cells. Cytolytic effector molecules important for anti-tumor effects mediated by adoptively transferred NK cells in vivo included perforin, IFN-γ, TRAIL and granzymes B and M. In contrast, NKG2D and granzyme A were found not to play a critical role in the NK cell-mediated response to RKIK tumor cells. The fact that only perforin was found to be important for NK cell-mediated cytotoxicity against RKIK-4862 tumor cells in vitro, highlighted the importance of our mouse model to investigate potential effector molecules and pathways involved in the in vivo anti-tumor response mediated by adoptively transferred NK cells. In this study we demonstrated that transferred NK cells trafficked to the tumor site and persisted in the lungs, liver, spleen and peripheral blood of recipient mice, which correlated with the anti-tumor response observed.
The adoptive transfer of TRAIL−/− NK cells identified a role for TRAIL in the NK cell-mediated inhibition of RKIK-4862 tumor growth. This correlated with expression of TRAIL on IL-2-activated NK effector cells and DR5 expression on RKIK-4862 tumor cells. However, given that a lack of TRAIL did not totally abolish inhibition of tumor growth mediated by transferred NK cells, it remained a possibility that other receptor–ligand interactions were also playing an important role. Although NKG2D interaction with cellular stress ligands has been shown to be important for NK cell-mediated anti-tumor rejection in some models, we found in this study that blocking the NKG2D receptor had no impact on the ability of adoptively transferred NK cells to inhibit the growth of RKIK-4862 tumor. Our data suggested that other receptors may be playing a role such as DNAX-accessory molecule-1 (DNAM-1) or natural cytotoxicity receptors (NCRs) which have recently been implicated in NK cell-mediated anti-tumor immunity [37]. It would be interesting in future studies to determine whether RKIK-4862 tumor cells express any of the ligands for these different receptors, and what specific NK cell-mediated response results from the stimulation of these receptors.
Our mouse model revealed that perforin is critical for both in vitro NK cell-mediated lysis of RKIK-4862 tumor cells and inhibition of RKIK-4862 tumor growth in vivo by adoptively transferred NK cells. This finding is in agreement with previous studies that have reported perforin-dependent control of carcinogen-induced tumor cells as well as the control of metastases and syngeneic tumors by NK cells [6, 38, 39]. This pore-forming protein has been shown to allow access of granzymes to the target cell leading to death of the target cell, although the exact mechanism of action of perforin is unclear [40–42]. In addition to perforin, we demonstrated the importance of granzymes M and B in the inhibition of RKIK-4862 tumor growth in vivo mediated by adoptively transferred NK cells. Granzyme B has been shown to induce caspase-independent or caspase-dependent target cell death and may also act on Bcl-2 family proteins to induce mitochondrial-dependent cell death [43–45]. Granzyme M, on the other hand, has been reported to induce caspase- and mitochondrial-independent cell death [46]. Although these granzymes can induce target cell death using different pathways, our results suggest a role for both caspase/mitochondrial-independent and caspase/mitochondrial-dependent-induced apoptosis in the inhibition of RKIK-4862 tumor growth mediated by adoptively transferred NK cells. In contrast, granzyme A, which induces caspase-independent cell death and DNA nicks [47], was shown not to play an important role in the inhibition of RKIK-4862 tumor growth. This is consistent with previous reports, which demonstrate that granzyme A-deficient NK cells retain their cytolytic function and suggest that granzyme A is not a strong promoter of NK cell-mediated cytolysis of target cells [48, 49]. Interestingly, the reduced anti-tumor effects observed with granzyme M and B knockout NK cells in vivo was in contrast to the results of the in vitro cytotoxicity assay where WT NK cells and NK cells from granzyme gene-targeted mice could similarly kill RKIK-4862 tumor cells. Although the reason for this discrepancy is unclear, these results suggest that granzymes mediate other important functions other than cell killing. It has been previously reported that granzyme B can induce anoikis in anchorage-dependent cell lines and can facilitate lymphocyte trafficking by cleaving extracellular matrix proteins [50]. In addition, granzymes have also been reported to play a role in inflammatory responses and can induce secretion of proinflammatory cytokines from immune cells such as monocytes and macrophages [51]. Thus, it is possible that granzymes (particularly B and M) may be playing additional roles other than cell killing that contribute to the anti-tumor response by NK cells seen in our in vivo model that were not observed in the in vitro setting.
Previous studies have demonstrated that IFN-γ secreted from NK cells can inhibit the proliferation of tumor cells in vitro [30]. We extend these findings by demonstrating the necessity of IFN-γ in the inhibition of RKIK-4862 tumor growth in vivo following adoptive transfer of activated NK cells. The exact role that IFN-γ secreted by adoptively transferred NK cells is playing in our model is unclear and is complicated by the fact that IFN-γ mediates anti-tumor effects using both direct and indirect mechanisms [31]. Possible mechanisms of action of IFN-γ in our model include increased activation of macrophages and NK cells, promoting adhesion of leukocytes to allow extravasation. It would be interesting in future studies to determine the exact mechanism by which adoptively transferred NK cell-secreted IFN-γ mediates anti-tumor effects in our model.
The lack of persistence of adoptively transferred T cells has frequently been identified as one reason for the failure of adoptive immunotherapy using these cells. It has been demonstrated that enhanced patient responses are associated with increased persistence of transferred T cells [33, 34, 52]. In our study, we demonstrated that adoptively transferred NK cells persisted and could be detected at the tumor site during the course of the experiment. It must be noted that the C57BL/6 RAG2−/−γc−/− mouse model used in this study was primarily chosen to determine the contribution of adoptively transferred NK cells to tumor inhibition in the absence of endogenous lymphocytes but this model does also have relevance to patients treated with lymphodepletion regimens. However, in future experiments, it will be important to also test this therapy in an immunocompetent setting perhaps combined with lymphodepletion to ensure that adoptively transferred NK cells do not cause any autoimmune pathology. If NK cell-mediated anti-tumor activity is reduced in such models, it may be necessary to further stimulate the cells by treating the mice with cytokines such as IL-2 or IL-15. These cytokines have been previously demonstrated to enhance proliferation, function and survival of NK cells [53–55].
In this study, transferred NK cells were predominantly CD27 positive and previous work has demonstrated enhanced functional activities of CD27hi NK cells compared to CD27lo NK cells [56]. However, for clinical translation using human NK cells, it would be interesting to compare the function of different human NK cell subsets. Human NK cells can be divided into two subsets according to CD56 expression. CD56dim NK cells have been reported to have a higher cytolytic capacity and lower cytokine secretion ability compared to CD56bright NK cells, which have high cytokine secretion abilities and low cytolytic capacity [57, 58]. This knowledge may enable matching patient tumor with required NK cell function which may lead to increased efficacy of this treatment. Recent studies have demonstrated new methods for expanding NK cells ex vivo for adoptive NK cell transfer, involving co-culture with stimulator cells and cytokines [59, 60]. This significantly increases the feasibility of this approach for the treatment of patients. Increased knowledge of the important mechanisms required for a NK cell-mediated activity against tumors may also help in the selection of adjuvants that may further enhance desired NK cell function. For example, Lundqvist et al. [61], demonstrated that combining adoptive NK cell transfer with the proteosome inhibitor Bortezomib enhanced the survival of tumor-bearing mice, compared to adoptive NK cell transfer alone. Other adjuvants to improve adoptive NK cell therapy include treatment with immune agonists, such as Toll-like receptor (TLR) agonists. Roda et al. [62], reported that the TLR-9 agonist, CPG, activated NK cells both directly and indirectly. Understanding the underlying mechanisms of NK cell function combined with genetic modification of these cells with tumor-specific recognition receptors may also lead to improved anti-tumor effects. Indeed, we and others have recently shown that genetic modification of NK cells with a chimeric receptor specific for a tumor-associated antigen led to increased anti-tumor effects [19, 22, 63]. Overall, the results of this study have important implications for the use of adoptively transferred NK cells for cancer immunotherapy.
Acknowledgments
We would like to acknowledge the assistance of the Peter MacCallum Cancer Centre Experimental Animal Facility technicians for animal care, in particular Michelle Stirling and Leanne McNiff for maintenance of the gene-targeted mice used in this study. We would also like to acknowledge Nicole McLaughlin for generating some of the antibodies used in these experiments. This work was funded by project grants from the National Health and Medical Research Council (NHMRC), Cancer Council of Victoria and the Susan Komen Breast Cancer Foundation. M.H. Kershaw and P.K. Darcy were supported by a NHMRC Senior Research Fellowship and Career Development Award, respectively. M.J. Smyth was supported by a NHMRC Senior Principal Research Fellowship. N.M. Haynes was supported by a NHMRC CJ Martin Fellowship.
Footnotes
M. H. Kershaw and P. K. Darcy contributed equally as senior authors.
References
- 1.Yokoyama WM, Kim S, French AR. The dynamic life of natural killer cells. Annu Rev Immunol. 2004;22:405–429. doi: 10.1146/annurev.immunol.22.012703.104711. [DOI] [PubMed] [Google Scholar]
- 2.Lanier LL. NK cell recognition. Annu Rev Immunol. 2005;23:225–274. doi: 10.1146/annurev.immunol.23.021704.115526. [DOI] [PubMed] [Google Scholar]
- 3.Cavanaugh VJ, Raulet DH, Campbell AE. Upregulation of CD94/NKG2A receptors and Qa-1b ligand during murine cytomegalovirus infection of salivary glands. J Gen Virol. 2007;88:1440–1445. doi: 10.1099/vir.0.82444-0. [DOI] [PubMed] [Google Scholar]
- 4.Gasser S, Orsulic S, Brown EJ, Raulet DH. The DNA damage pathway regulates innate immune system ligands of the NKG2D receptor. Nature. 2005;436:1186–1190. doi: 10.1038/nature03884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Raulet DH, Guerra N. Oncogenic stress sensed by the immune system: role of natural killer cell receptors. Nat Rev Immunol. 2009;9:568–580. doi: 10.1038/nri2604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Smyth MJ, Thia KY, Cretney E, et al. Perforin is a major contributor to NK cell control of tumor metastasis. J Immunol. 1999;162:6658–6662. [PubMed] [Google Scholar]
- 7.Seki N, Hayakawa Y, Brooks AD, et al. Tumor necrosis factor-related apoptosis-inducing ligand-mediated apoptosis is an important endogenous mechanism for resistance to liver metastases in murine renal cancer. Cancer Res. 2003;63:207–213. [PubMed] [Google Scholar]
- 8.Wu J, Lanier LL. Natural killer cells and cancer. Adv Cancer Res. 2003;90:127–156. doi: 10.1016/S0065-230X(03)90004-2. [DOI] [PubMed] [Google Scholar]
- 9.Smyth MJ, Crowe NY, Godfrey DI. NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int Immunol. 2001;13:459–463. doi: 10.1093/intimm/13.4.459. [DOI] [PubMed] [Google Scholar]
- 10.Degli-Esposti MA, Smyth MJ. Close encounters of different kinds: dendritic cells and NK cells take centre stage. Nat Rev Immunol. 2005;5:112–124. doi: 10.1038/nri1549. [DOI] [PubMed] [Google Scholar]
- 11.Smyth MJ, Cretney E, Kershaw MH, Hayakawa Y. Cytokines in cancer immunity and immunotherapy. Immunol Rev. 2004;202:275–293. doi: 10.1111/j.0105-2896.2004.00199.x. [DOI] [PubMed] [Google Scholar]
- 12.Rosenberg S. Lymphokine-activated killer cells: a new approach to immunotherapy of cancer. J Natl Cancer Inst. 1985;75:595–603. [PubMed] [Google Scholar]
- 13.Bordignon C, Carlo-Stella C, Colombo MP, et al. Cell therapy: achievements and perspectives. Haematologica. 1999;84:1110–1149. [PubMed] [Google Scholar]
- 14.Morecki S, Yacovlev E, Gelfand Y, Vilensky A, Slavin S. Allogeneic versus syngeneic killer splenocytes as effector cells for the induction of graft-versus-tumor effect. Biol Blood Marrow Transplant. 2004;10:40–48. doi: 10.1016/j.bbmt.2003.09.013. [DOI] [PubMed] [Google Scholar]
- 15.Kawase T, Matsuo K, Kashiwase K, et al. HLA mismatch combinations associated with decreased risk of relapse: implications for the molecular mechanism. Blood. 2009;113:2851–2858. doi: 10.1182/blood-2008-08-171934. [DOI] [PubMed] [Google Scholar]
- 16.Ruggeri L, Capanni M, Urbani E, et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science. 2002;295:2097–2100. doi: 10.1126/science.1068440. [DOI] [PubMed] [Google Scholar]
- 17.Guven H, Gilljam M, Chambers BJ, et al. Expansion of natural killer (NK) and natural killer-like T (NKT)-cell populations derived from patients with B-chronic lymphocytic leukemia (B-CLL): a potential source for cellular immunotherapy. Leukemia. 2003;17:1973–1980. doi: 10.1038/sj.leu.2403083. [DOI] [PubMed] [Google Scholar]
- 18.Alici E, Sutlu T, Bjorkstrand B, et al. Autologous antitumor activity by NK cells expanded from myeloma patients using GMP-compliant components. Blood. 2008;111:3155–3162. doi: 10.1182/blood-2007-09-110312. [DOI] [PubMed] [Google Scholar]
- 19.Imai C, Iwamoto S, Campana D. Genetic modification of primary natural killer cells overcomes inhibitory signals and induces specific killing of leukemic cells. Blood. 2005;106:376–383. doi: 10.1182/blood-2004-12-4797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cho D, Campana D. Expansion and activation of natural killer cells for cancer immunotherapy. Korean J Lab Med. 2009;29:89–96. doi: 10.3343/kjlm.2009.29.2.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Karre K, Ljunggren HG, Piontek G, Kiessling R. Selective rejection of H-2-deficient lymphoma variants suggests alternative immune defence strategy. Nature. 1986;319:675–678. doi: 10.1038/319675a0. [DOI] [PubMed] [Google Scholar]
- 22.Pegram HJ, Jackson JT, Smyth MJ, Kershaw MH, Darcy PK. Adoptive transfer of gene-modified primary NK cells can specifically inhibit tumor progression in vivo. J Immunol. 2008;181:3449–3455. doi: 10.4049/jimmunol.181.5.3449. [DOI] [PubMed] [Google Scholar]
- 23.Abdool K, Cretney E, Brooks AD, et al. NK cells use NKG2D to recognize a mouse renal cancer (Renca), yet require intercellular adhesion molecule-1 expression on the tumor cells for optimal perforin-dependent effector function. J Immunol. 2006;177:2575–2583. doi: 10.4049/jimmunol.177.4.2575. [DOI] [PubMed] [Google Scholar]
- 24.Takeda K, Smyth MJ, Cretney E, et al. Critical role for tumor necrosis factor-related apoptosis-inducing ligand in immune surveillance against tumor development. J Exp Med. 2002;195:161–169. doi: 10.1084/jem.20011171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Trapani JA, Smyth MJ. Functional significance of the perforin/granzyme cell death pathway. Nat Rev Immunol. 2002;2:735–747. doi: 10.1038/nri911. [DOI] [PubMed] [Google Scholar]
- 26.Screpanti V, Wallin RP, Ljunggren HG, Grandien A. A central role for death receptor-mediated apoptosis in the rejection of tumors by NK cells. J Immunol. 2001;167:2068–2073. doi: 10.4049/jimmunol.167.4.2068. [DOI] [PubMed] [Google Scholar]
- 27.Trapani JA. Granzymes: a family of lymphocyte granule serine proteases. Genome Biol. 2001;2:3014. doi: 10.1186/gb-2001-2-12-reviews3014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cretney E, Takeda K, Yagita H, Glaccum M, Peschon JJ, Smyth MJ. Increased susceptibility to tumor initiation and metastasis in TNF-related apoptosis-inducing ligand-deficient mice. J Immunol. 2002;168:1356–1361. doi: 10.4049/jimmunol.168.3.1356. [DOI] [PubMed] [Google Scholar]
- 29.Nagata S, Suda T. Fas and Fas ligand: lpr and gld mutations. Immunol Today. 1995;16:39–43. doi: 10.1016/0167-5699(95)80069-7. [DOI] [PubMed] [Google Scholar]
- 30.Rabinowich H, Vitolo D, Altarac S, Herberman RB, Whiteside TL. Role of cytokines in the adoptive immunotherapy of an experimental model of human head and neck cancer by human IL-2-activated natural killer cells. J Immunol. 1992;149:340–349. [PubMed] [Google Scholar]
- 31.Schroder K, Hertzog PJ, Ravasi T, Hume DA. Interferon-gamma: an overview of signals, mechanisms and functions. J Leukoc Biol. 2004;75:163–189. doi: 10.1189/jlb.0603252. [DOI] [PubMed] [Google Scholar]
- 32.Smyth MJ, Swann J, Cretney E, Zerafa N, Yokoyama WM, Hayakawa Y. NKG2D function protects the host from tumor initiation. J Exp Med. 2005;202:583–588. doi: 10.1084/jem.20050994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Huang J, Khong HT, Dudley ME, et al. Survival, persistence, and progressive differentiation of adoptively transferred tumor-reactive T cells associated with tumor regression. J Immunother. 2005;28:258–267. doi: 10.1097/01.cji.0000158855.92792.7a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhou J, Dudley ME, Rosenberg SA, Robbins PF. Persistence of multiple tumor-specific T-cell clones is associated with complete tumor regression in a melanoma patient receiving adoptive cell transfer therapy. J Immunother. 2005;28:53–62. doi: 10.1097/00002371-200501000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dudley ME, Wunderlich JR, Robbins PF, et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science. 2002;298:850–854. doi: 10.1126/science.1076514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Besser MJ, Shapira-Frommer R, Treves AJ, et al. Minimally cultured or selected autologous tumor-infiltrating lymphocytes after a lympho-depleting chemotherapy regimen in metastatic melanoma patients. J Immunother. 2009;32:415–423. doi: 10.1097/CJI.0b013e31819c8bda. [DOI] [PubMed] [Google Scholar]
- 37.Lakshmikanth T, Burke S, Ali TH, et al. NCRs and DNAM-1 mediate NK cell recognition and lysis of human and mouse melanoma cell lines in vitro and in vivo. J Clin Invest. 2009;119:1251–1263. doi: 10.1172/JCI36022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.van den Broek MF, Kagi D, Zinkernagel RM, Hengartner H. Perforin dependence of natural killer cell-mediated tumor control in vivo. Eur J Immunol. 1995;25:3514–3516. doi: 10.1002/eji.1830251246. [DOI] [PubMed] [Google Scholar]
- 39.Street SE, Cretney E, Smyth MJ. Perforin and interferon-gamma activities independently control tumor initiation, growth, and metastasis. Blood. 2001;97:192–197. doi: 10.1182/blood.V97.1.192. [DOI] [PubMed] [Google Scholar]
- 40.Bolitho P, Voskoboinik I, Trapani JA, Smyth MJ. Apoptosis induced by the lymphocyte effector molecule perforin. Curr Opin Immunol. 2007;19:339–347. doi: 10.1016/j.coi.2007.04.007. [DOI] [PubMed] [Google Scholar]
- 41.Keefe D, Shi L, Feske S, et al. Perforin triggers a plasma membrane-repair response that facilitates CTL induction of apoptosis. Immunity. 2005;23:249–262. doi: 10.1016/j.immuni.2005.08.001. [DOI] [PubMed] [Google Scholar]
- 42.Metkar SS, Wang B, Aguilar-Santelises M, et al. Cytotoxic cell granule-mediated apoptosis: perforin delivers granzyme B-serglycin complexes into target cells without plasma membrane pore formation. Immunity. 2002;16:417–428. doi: 10.1016/S1074-7613(02)00286-8. [DOI] [PubMed] [Google Scholar]
- 43.Sarin A, Williams MS, Alexander-Miller MA, Berzofsky JA, Zacharchuk CM, Henkart PA. Target cell lysis by CTL granule exocytosis is independent of ICE/Ced-3 family proteases. Immunity. 1997;6:209–215. doi: 10.1016/S1074-7613(00)80427-6. [DOI] [PubMed] [Google Scholar]
- 44.Darmon AJ, Nicholson DW, Bleackley RC. Activation of the apoptotic protease CPP32 by cytotoxic T-cell-derived granzyme B. Nature. 1995;377:446–448. doi: 10.1038/377446a0. [DOI] [PubMed] [Google Scholar]
- 45.Waterhouse NJ, Sedelies KA, Trapani JA. Role of Bid-induced mitochondrial outer membrane permeabilization in granzyme B-induced apoptosis. Immunol Cell Biol. 2006;84:72–78. doi: 10.1111/j.1440-1711.2005.01416.x. [DOI] [PubMed] [Google Scholar]
- 46.Kelly JM, Waterhouse NJ, Cretney E, et al. Granzyme M mediates a novel form of perforin-dependent cell death. J Biol Chem. 2004;279:22236–22242. doi: 10.1074/jbc.M401670200. [DOI] [PubMed] [Google Scholar]
- 47.Beresford PJ, Xia Z, Greenberg AH, Lieberman J. Granzyme A loading induces rapid cytolysis and a novel form of DNA damage independently of caspase activation. Immunity. 1999;10:585–594. doi: 10.1016/S1074-7613(00)80058-8. [DOI] [PubMed] [Google Scholar]
- 48.Ebnet K, Hausmann M, Lehmann-Grube F, et al. Granzyme A-deficient mice retain potent cell-mediated cytotoxicity. EMBO J. 1995;14:4230–4239. doi: 10.1002/j.1460-2075.1995.tb00097.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Trapani JA, Bird PI. A renaissance in understanding the multiple and diverse functions of granzymes? Immunity. 2008;29:665–667. doi: 10.1016/j.immuni.2008.10.002. [DOI] [PubMed] [Google Scholar]
- 50.Buzza MS, Zamurs L, Sun J, et al. Extracellular matrix remodeling by human granzyme B via cleavage of vitronectin, fibronectin, and laminin. J Biol Chem. 2005;280:23549–23558. doi: 10.1074/jbc.M412001200. [DOI] [PubMed] [Google Scholar]
- 51.Metkar SS, Menaa C, Pardo J, et al. Human and mouse granzyme A induce a proinflammatory cytokine response. Immunity. 2008;29:720–733. doi: 10.1016/j.immuni.2008.08.014. [DOI] [PubMed] [Google Scholar]
- 52.Robbins PF, Dudley ME, Wunderlich J, et al. Cutting edge: persistence of transferred lymphocyte clonotypes correlates with cancer regression in patients receiving cell transfer therapy. J Immunol. 2004;173:7125–7130. doi: 10.4049/jimmunol.173.12.7125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cooper MA, Bush JE, Fehniger TA, et al. In vivo evidence for a dependence on interleukin 15 for survival of natural killer cells. Blood. 2002;100:3633–3638. doi: 10.1182/blood-2001-12-0293. [DOI] [PubMed] [Google Scholar]
- 54.Prlic M, Blazar BR, Farrar MA, Jameson SC. In vivo survival and homeostatic proliferation of natural killer cells. J Exp Med. 2003;197:967–976. doi: 10.1084/jem.20021847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Becknell B, Caligiuri MA. Interleukin-2, interleukin-15, and their roles in human natural killer cells. Adv Immunol. 2005;86:209–239. doi: 10.1016/S0065-2776(04)86006-1. [DOI] [PubMed] [Google Scholar]
- 56.Hayakawa Y, Smyth MJ. CD27 dissects mature NK cells into two subsets with distinct responsiveness and migratory capacity. J Immunol. 2006;176:1517–1524. doi: 10.4049/jimmunol.176.3.1517. [DOI] [PubMed] [Google Scholar]
- 57.Cooper MA, Fehniger TA, Caligiuri MA. The biology of human natural killer-cell subsets. Trends Immunol. 2001;22:633–640. doi: 10.1016/S1471-4906(01)02060-9. [DOI] [PubMed] [Google Scholar]
- 58.Colucci F, Caligiuri MA, Di Santo JP. What does it take to make a natural killer? Nat Rev Immunol. 2003;3:413–425. doi: 10.1038/nri1088. [DOI] [PubMed] [Google Scholar]
- 59.Klingemann HG, Martinson J. Ex vivo expansion of natural killer cells for clinical applications. Cytotherapy. 2004;6:15–22. doi: 10.1080/14653240310004548. [DOI] [PubMed] [Google Scholar]
- 60.Fujisaki H, Kakuda H, Shimasaki N, et al. Expansion of highly cytotoxic human natural killer cells for cancer cell therapy. Cancer Res. 2009;69:4010–4017. doi: 10.1158/0008-5472.CAN-08-3712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lundqvist A, Yokoyama H, Smith A, Berg M, Childs R. Bortezomib treatment and regulatory T-cell depletion enhance the antitumor effects of adoptively infused NK cells. Blood. 2009;113:6120–6127. doi: 10.1182/blood-2008-11-190421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Roda JM, Parihar R, Carson WE., 3rd CpG-containing oligodeoxynucleotides act through TLR9 to enhance the NK cell cytokine response to antibody-coated tumor cells. J Immunol. 2005;175:1619–1627. doi: 10.4049/jimmunol.175.3.1619. [DOI] [PubMed] [Google Scholar]
- 63.Muller T, Uherek C, Maki G, et al. Expression of a CD20-specific chimeric antigen receptor enhances cytotoxic activity of NK cells and overcomes NK-resistance of lymphoma and leukemia cells. Cancer Immunol Immunother. 2008;57:411–423. doi: 10.1007/s00262-007-0383-3. [DOI] [PMC free article] [PubMed] [Google Scholar]