Abstract
In this study, novel nanomagnetic catalysts, namely Fe2O3-MgO@choline formate (Ch. F.) and Fe2O3-MgO@choline cyanide (Ch. CN), were synthesized through immobilizing choline-based ion liquids to magnetic support via a simple and cost-effective methodology. FT-IR, TGA, FE-SEM, VSM, EDS, BET, and XRD techniques were employed to assess and characterize these organic–inorganic compounds. Following the successful preparation of nanoparticles, the catalysts were utilized in Knoevenagel and benzoin condensations. Fe2O3-MgO@Ch.F. exhibited exceptional activity in Knoevenagel condensation under solvent-free conditions at room temperature, achieving high yields (91–98%) in a short timeframe. Similarly, Fe2O3-MgO@Ch.CN demonstrated remarkable activity in benzoin condensation under environmentally friendly solvent conditions, yielding higher isolated yields (76–88%). Furthermore, these magnetically recyclable multifunctional catalysts displayed the ability to be reused up to five times without a significant loss in efficiency. Additionally, the heterogeneity of this nanocatalyst was investigated using the hot filtration technique. The findings indicated that the reaction primarily occurs via a heterogeneous pathway.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13065-024-01176-5.
Keywords: Choline formate, Choline cyanide, Multifunctional catalysts, Nanomagnetic heterogeneous catalyst, Knoevenagel and Benzoin condensations
Introduction
In recent years, significant emphasis has been placed on developing techniques to heterogenize homogeneous catalysts while maintaining their active sites. This approach aims to amalgamate the advantages of selective, homogeneous, reactive catalysis with the recyclability and facile removal of catalysts from the reaction solution [1–9]. The coupling of homogeneous catalysts with inorganic solids is a widely utilized method for achieving heterogeneous synthesis [1, 8, 10]. Numerous homogeneous catalysts for Knoevenagel and benzoin reactions have been explored, with these condensations being recognized for their ability to form carbon–carbon bonds [11–17]. However, homogeneous systems are associated with several drawbacks, including the high cost of catalysts, the challenge of catalyst recovery, catalyst decomposition under basic pH conditions, elevated reaction temperatures, product isolation difficulties, and the use of carcinogenic and environmentally harmful solvents, leading to substantial waste generation [18–20]. To address these challenges and preserve the catalytic active sites inherent in homogeneous counterparts, there is a growing focus on heterogenizing homogeneous catalysts [21]. This research direction has garnered significant attention in both industrial and academic sectors. Choline chloride-based ionic liquids, such as choline azide, choline hydroxide, choline cyanide, and Choline amide serve multifaceted roles in organic processes by acting as safe, cost-effective, and efficient reactants, solvents, and homogeneous catalysts [22–28]. On the other hand, magnetic supports possess various advantageous properties, including low toxicity, cost-effectiveness, extensive surface area, Lewis acid activity, facile production, surface functionalization, rapid dispersion in processes, high conductivity, and efficient recoverability through external magnets [22, 29–36]. In the context of multifunctional catalysts for heterogeneous modification applications, it is imperative to investigate the activity of coordinated cholines on the magnetic surface. Considering the magnetic attraction and potential for agglomeration, incorporating coating stabilizers becomes necessary [37, 38]. The MgO shell acts as a shield for magnetic nanoparticles, preventing further oxidation and aggregation of the magnetic core. Notably, MgO stands out as a superior support compared to alternatives due to possessing Lewis acidic and Lewis basic sites, enabling stabilization of reaction intermediates during catalysis [39–42]. The integration of choline-based ionic liquid as a homogeneous catalyst with iron-magnesium oxide as a super-magnetic support facilitates the synthesis of highly active, multifunctional, and recyclable catalysts. These versatile catalysts are not only cost-effective and environmentally friendly but also hold significant appeal across a wide spectrum of applications. Fe2O3@MgO effectively stabilizes choline-based ionic liquids as magnetic support in diverse applications. This study began with the novel and remarkably simple synthesis of choline formate, employing an inexpensive protocol. Subsequently, attention was directed towards investigating multifunctional heterogeneous catalysts, utilizing coordinated choline cyanides and choline formate on magnetic support. Essentially, an organo-catalyst is stabilized on the magnetic surface, exhibiting Lewis acid and basic characteristics [43, 44]. Initial research focused on the conditions and spectra surrounding the synthesis of choline formate and choline cyanide, followed by coordinating Fe2O3–MgO as catalysts in Knoevenagel and benzoin condensation reactions.
Results and discussion
Catalyst characterization
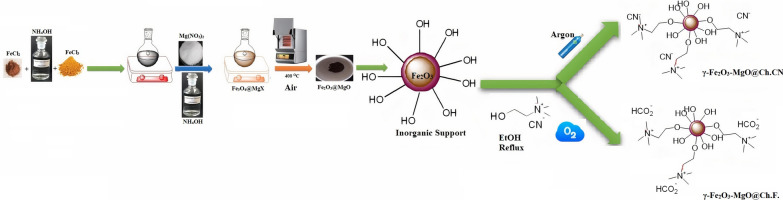
Figure 1 depicts the synthesis pathway of γ-Fe2O3-MgO@Ch.F. and γ-Fe2O3-MgO@Ch.CN. γ-Fe2O3 is initially produced as per the literature [41, 44–47]. To fulfill this objective, Fe3O4 is subjected to a reaction with ammonium hydroxide (27 wt.%) and magnesium nitrate at 70 °C for 12 h. The magnetic powder is subsequently separated and subjected to heating in a furnace for 4 h at 400 °C to yield γ-Fe2O3-MgO. Moreover, Ch.F. and Ch.CN are synthesized, and their magnetic surfaces are stirred at 25 °C, followed by refluxing in EtOH for 12 h at 80 °C to produce γ-Fe2O3-MgO@Ch. F. and γ-Fe2O3-MgO@Ch.CN, respectively. Finally, the catalysts synthesized are characterized through TGA, FTIR, FE-SEM, VSM, EDS, BET, and XRD measurements.
Fig. 1.
Schematic diagram of catalysts preparation. The source of this diagram is taken from https://www.nature.com/articles/s41598-023-44881-2/figures/3. The software tools employed to create this diagram were Chemdraw and Paint. The figure was designed by authors
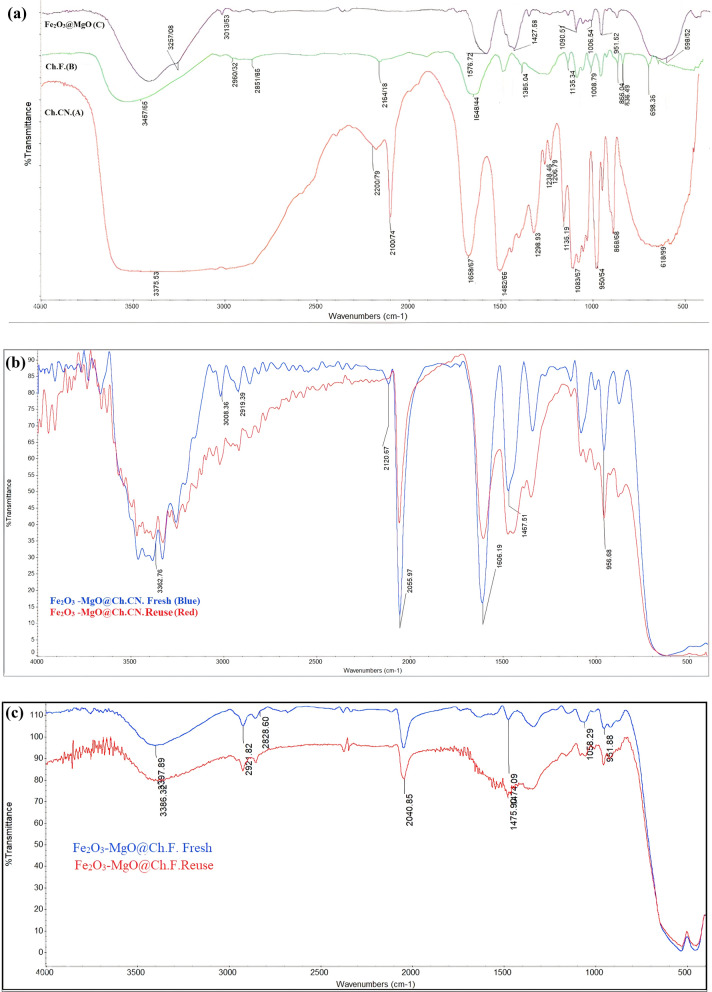
Figure 2 shows the FT-IR spectrum of Ch.CN, Ch.F., and γ-Fe2O3-MgO. In Fig. 2A, the FT-IR spectrum of Ch.CN reveals bands at 3375, 2200, 2100, 1482, 1350, and 1083, potentially attributed to OH stretching (3375 cm−1), C-H bending (1482cm−1), and C–O stretching (1083cm−1). Notably, the FT-IR spectrum of Ch.CN exhibits two distinct bands, 2200cm−1 (C≡N) and 2100cm−1 (N–C), while the N–C peak intensities at Ch.F. are weak (Fig. 2A). Mass spectroscopy and NMR (Additional file 1: Figs. S52–S57) were employed to investigate these proposed structures, with confirmation from sedimentary tests. The FT-IR spectrum of fresh and reused catalysts reveals the Fe–O stretching vibration attributed to the iron oxide in the spinel form, observed around 569 to 584 cm−1 (Fig. 2A–C) [48]. In γ-Fe2O3-MgO@Ch.CN, the FT-IR peak of Ch.CN (2200 and 2100 cm−1) shifts to 2120 and 2055 cm−1, respectively (Fig. 2A and B). Additionally, the FT-IR peak of Ch.F. (2164 cm−1) undergoes a shift to 2040 cm−1 (Fig. 2A and C). This shift is attributed to the surface chelation of organic groups to γ-Fe2O3-MgO.
Fig. 2.
A FT-IR spectrum of preparation of Ch.CN (A) Ch.F. (B) γ-Fe2O3@MgO (C). B FT-IR spectrum of preparation of γ-Fe2O3-MgO@Ch.CN Fresh and Reuse. C FT-IR spectrum of preparation of γ-Fe2O3-MgO@Ch.F. Fresh and Reuse
The TGA is employed for the analysis of catalyst composition and heat resistance (Fig. 3). In the cases of γ-Fe2O3-MgO@Ch.F. and γ-Fe2O3-MgO@Ch.CN, the TGA curve indicates a minor weight loss of approximately 1–2% at 100 °C, attributed to physically adsorbed water. The decomposition of organic compounds from γ-Fe2O3-MgO @Ch. F. and γ-Fe2O3-MgO@Ch.CN leads to a 10–12% weight loss in the temperature range of 200–400 °C. Ultimately, the residual weight of 77% corresponds to Fe2O3@MgO. Thermal analysis demonstrates that the catalysts exhibit thermal stability up to 200 °C (Fig. 3). Notably, the boiling points of Ch.F. and Ch.CN are 195.38 °C and 190.8 °C, respectively, confirming the non-sensitivity and non-energetic nature of these choline salts. DSC is employed to assess the safety of these innovative compounds in relation to chemical reactions and phase transitions as a function of temperature (Additional file 1: Figs. S1 and S2). Furthermore, no exothermic peak is observed in these experiments, with degradation occurring progressively at approximately 300 °C.
Fig. 3.
TGA of γ-Fe2O3-MgO@Ch.F. and γ-Fe2O3-MgO@Ch.CN
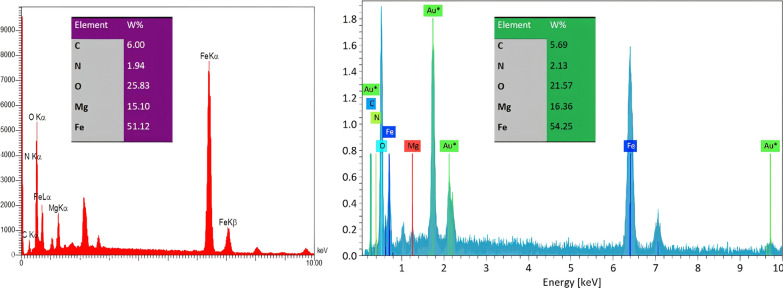
FE-SEM is employed to investigate the surface morphology of fresh catalysts (Fig. 4). The images reveal the formation of spherical particles with an average size ranging from 32 to 43 nm. EDS analysis is conducted to identify the elements present in the catalysts (Fig. 5). In the case of Fe2O3-MgO@Ch.X (X: F,CN), the EDS pattern confirms the presence of iron, magnesium, and oxygen, providing credible evidence of the modification of Fe2O3 by MgO. Alongside these elements, prominent peaks for carbon indicate the successful loading of choline onto Fe2O3@MgO. The Au peaks observed can be attributed to the sample holder [49].
Fig. 4.

SEM images of γ-Fe2O3-MgO (A), γ- Fe2O3-MgO@Ch.F. (B) γ-Fe2O3-MgO@Ch.CN (C)
Fig. 5.
EDS results of Fe2O3-MgO@Ch.F.(Red) and Fe2O3-MgO@Ch.CN(Blue)
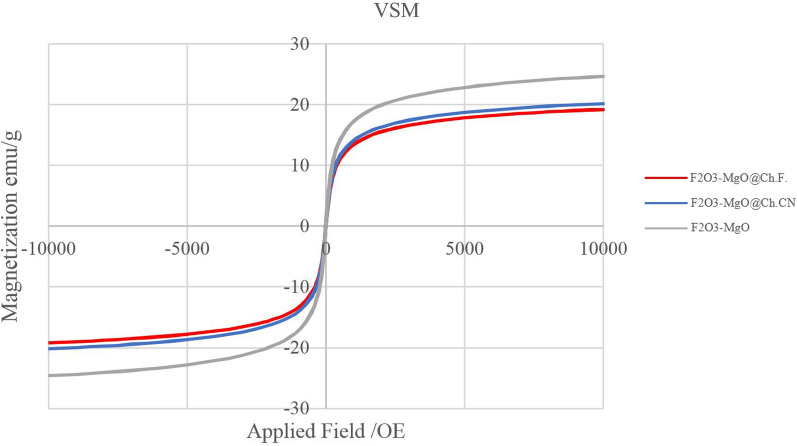
The magnetic hysteresis loop readings for γ-Fe2O3-MgO@Ch.F., γ-Fe2O3-MgO @Ch.CN and γ-Fe2O3-MgO are 19, 20, and 25 emu·g−1, respectively, indicating saturation magnetization. The observed decrease in saturation magnetization (MS value) may be attributed to the conversion of Fe3O4 into γ-Fe2O3 during heating and the coating of MgO on iron oxide [50]. The introduction of Ch.F. and Ch.CN onto the γ-Fe2O3-MgO surface further diminishes the saturation magnetization value (Fig. 6). The effortless attraction of the nanoparticles to an external magnet further showcased their strong magnetization.
Fig. 6.
VSM curve of γ-Fe2O3-MgO(grey), γ-Fe2O3-MgO@Ch.F.(Red) and γ-Fe2O3-MgO@Ch.CN(Blue)
The XRD patterns of γ-Fe2O3-MgO@Ch.F. and γ-Fe2O3-MgO@Ch.CN are examined within the 10°–80° range to discern their crystalline structures. The XRD patterns of γ-Fe2O3-MgO@ Ch. F. (red line) and γ-Fe2O3-MgO@Ch. CN (grey line) (Fig. 7) exhibit minimal variations. Notably, the diffraction peaks at 2Ɵ = 62.7, 62.2, 57.2, 53.8, 42.9, 36.8, 35.6, and 30 in Fig. 7, are consistent with the standard structure of γ-Fe2O3, as per JCPDS card No. 39-1346. Additionally, peaks at approximately 2Ɵ = 62.2, 50.34, and 18.1 are attributed to MgO (JCPDS 4-829) (Fig. 7) [46].
Fig. 7.
XRD pattern of γ-Fe2O3-MgO@Ch.F.(Red) and γ-Fe2O3-MgO@Ch.CN(grey)
The specific surface area of the nanocatalysts were determined using the Brunauer–Emmett–Teller (BET) technique (Additional file 1: Fig. S6) [51]. The Fe2O3-MgO@Ch.CN and Fe2O3-MgO@ch.F. have BET surface areas of 27.05 and 26.74 m2/g, respectively. Incorporating Ch.F. and Ch.CN onto the Fe2O3-MgO surface leads to a decrease in surface area in comparison to Fe2O3-MgO [47].
The catalytic activity
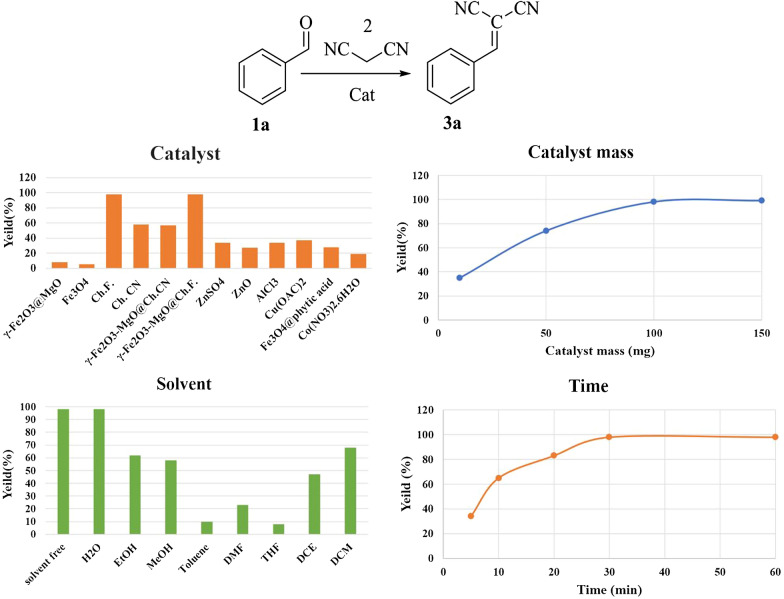
The catalytic activity of the synthesized and characterized catalysts was evaluated in the Knoevenagel condensation using benzaldehyde 1a and malononitrile 2 as a model reaction in room temperature. Various parameters, including catalyst, solvent, catalyst loading, and reaction time, were investigated [52]. Initially, to identify the primary catalytic center of γ-Fe2O3-MgO@Ch.X (X: F/CN), a model reaction was conducted in a solvent-free environment for 30 min, employing γ-Fe2O3-MgO, Ch.F., and Ch.CN as catalysts at room temperature. The results indicated the essential role of an organocatalyst (Ch.F. or Ch.CN) for optimal reaction efficiency. Figure 8 illustrates that the presence of γ-Fe2O3-MgO@Ch.F. as a heterogeneous catalyst and Ch.F. as a homogeneous catalyst resulted in higher efficiency (98%) in the model reaction. Further comparison with various Lewis acids in solvent-free and room temperature conditions revealed weak to moderate yields, with γ-Fe2O3-MgO@Ch.F. emerging as the preferred reusable catalyst with superior activity and the highest yield (98%) for future exploration. Exploring different solvents, such as EtOH, H2O, MeOH, DCM, DMF, THF, DCE, and toluene, with 100 mg γ-Fe2O3-MgO@Ch.F. revealed yields of 62, 98, 58, 68, 23, 8, and 47%, respectively. The solvent-free condition, due to its environmental friendliness and higher efficiency (yield 98%), was determined as the optimal choice. Adjusting the catalyst mass and reaction time showed that using 100 mg of γ-Fe2O3-MgO@Ch.F. and a reaction time of 30 min provided optimal conditions for higher efficiency of the target product (Fig. 8). Therefore, γ-Fe2O3-MgO@Ch.F., serving as an eco-friendly, reusable, and separable magnetic catalyst demonstrated superior efficiency under solvent-free conditions at room temperature for a short duration. Upon establishing optimal conditions, the scope of the reaction was broadened to include various aromatic and heteroaromatic aldehydes and ketones in both heterogeneous and homogeneous catalysis settings. Table 1 reveals that homogeneous systems often yield non-reusable excellent yields, while γ-Fe2O3-MgO@Ch.F. consistently produces a reusable, higher output. Notably, several substituted heterocyclic ketones and aldehydes could be efficiently reacted with malononitrile to yield benzylidene malononitrile with high isolated yields (91–98%).
Fig. 8.
Optimization of Knoevenagel condensation
Table 1.
Screening of the derivatives Knoevenagel condensation
| Entry | Structure | Yield(%)* γ-Fe2O3-MgO@Ch.F | Yield (%)* Ch.F |
M.P. (°C) | References |
|---|---|---|---|---|---|
| 3a | 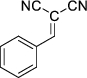 |
98% | 98% | 82–83 | [53] |
| 3b | 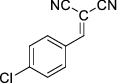 |
97% | 98% | 160–162 | [53] |
| 3c | 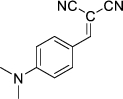 |
97% | 99% | 182 | [54] |
| 3d |  |
96% | 98% | 131–134 | [53] |
| 3e | 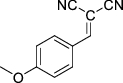 |
97% | 99% | 110–112 | [53] |
| 3f |  |
99% | 99% | 100–101 | [55] |
| 3g | 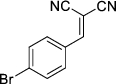 |
98% | 99% | 165–166 | [56] |
| 3h | 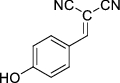 |
95% | 98% | 183–184 | [55] |
| 3i | 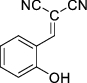 |
95% | 98% | 73–74 | [57] |
| 3j | 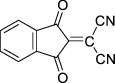 |
91% | 93% | 281 | [58] |
| 3k | 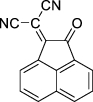 |
92% | 95% | 244–246 | [59] |
| 3l | 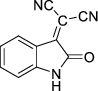 |
94% | 97% | 215–217 | [60] |
| 3m | 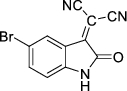 |
93% | 95% | 228–230 | [60] |
| 3n | 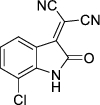 |
93% | 96% | 245–246 | [60] |
*Isolated yield, Benzaldehyde (2.5 mmol), malononitrile (2.5 mmol), Cat: Ch.F.:(100 mg (0.67mmol) or γ-Fe2O3-MgO@Ch.F.:100mg: 0.067mmol)
Encouraged by our success, we sought a new reaction to evaluate chemo-selectivity and explore choline cyanide tendencies, leading us to benzoin condensation. The model reaction involved the reaction of benzaldehyde 1a with itself under room temperature (R.T.) conditions for 1.5 h. Catalysts included Fe3O4, Fe2O3-MgO, γ-Fe2O3-MgO@Ch.F., Ch.F., γ-Fe2O3-MgO@Ch.CN, and Ch.CN and some NaCN situations were also explored. Consequently, γ-Fe2O3-MgO@Ch.CN emerged as the preferred reusable catalyst with superior activity (yield 88%) for further investigation (Fig. 9). Additional experiments at different temperatures revealed 25 °C as the optimal reaction temperature. Figure 9 illustrates that the rise in temperature has resulted in a decline in reaction efficiency. This decline can be attributed to the partial conversion of choline cyanide to choline formate. EtOH, H2O, MeOH, THF, toluene, and dioxane were investigated as solvents to assess their impact on yield and reaction time. Notably, the performance of γ-Fe2O3-MgO@Ch.CN improved in the presence of EtOH. As depicted in Fig. 9, employing 100 mg of catalyst and a reaction time of 1.5 h proved to be optimal conditions for achieving higher isolated efficiency (88%) of the target product at room temperature. Therefore, in benzoin condensation, γ-Fe2O3-MgO@Ch.F. serving as an eco-friendly, reusable, and detachable magnetic catalyst, demonstrated superior isolated efficiency under EtOH as a green solvent at room temperature for a shorter duration.
Fig. 9.
Optimization of benzoin condensation
To assess the breadth of the chemo-selective reaction, various benzyl aldehydes and heteroaromatic compounds with both electron-withdrawing and electron-donating functions were screened under optimum reaction conditions, yielding high isolated efficiency (76–88%) (Table 2). Further details on the proposed methodology can be found in the accompanying material.
Table 2.
Screening of the derivatives Benzoin condensation
| Label | Product | Melting point | References | Yield* Ch.CN (%) |
Yield* Cat (%) |
|---|---|---|---|---|---|
| 4a |  |
132–135 ℃ | [61] | 90% | 88% |
| 4b |  |
65–67 ℃ | [61] | 84% | 82% |
| 4c |  |
94–96 ℃ | [61] | 83% | 80% |
| 4d |  |
76–77 ℃ | [62] | 81% | 79% |
| 4e |  |
109–111 ℃ | [61] | 80% | 80% |
| 4f |  |
134–135 ℃ | [63] | 81% | 76% |
| 4g |  |
107–108 ℃ | [64] | 81% | 78% |
| 4h |  |
239 ℃ | [65] | 82% | 77% |
| 4i |  |
87–89 ℃ | [61] | 83% | 79% |
*Isolated yield Benzaldehyde (1 mmol), Cat:choline cyanide (1 mmol:0.13 g) or: Fe2O3-MgO@Ch.CN:100mg:(0.092mmol)
Recyclability γ-Fe2O3-MgO@Ch.F. and Fe2O3-MgO@Ch.CN
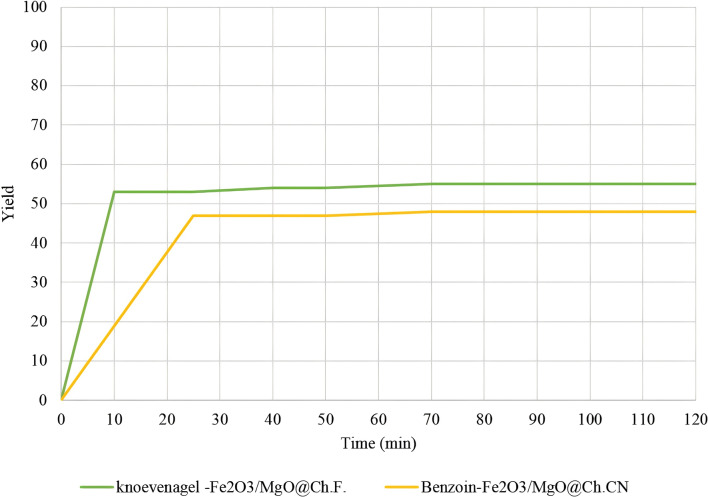
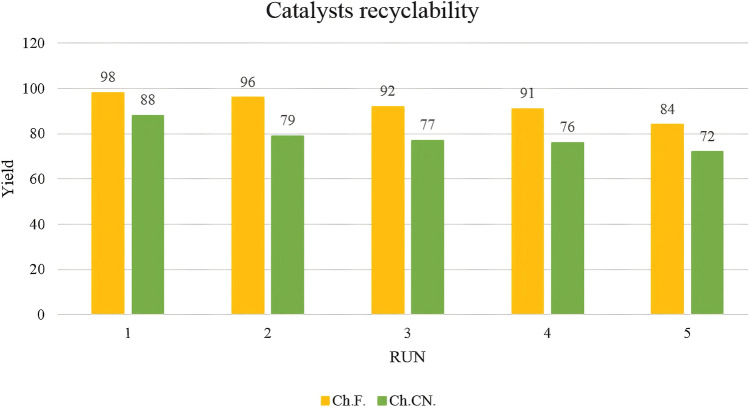
Hot filtration tests were conducted under optimal conditions to assess the leaching of Ch.F. from the heterogeneous catalysts during malononitrile and benzaldehyde reactions. Similarly, γ-Fe2O3-MgO@Ch.CN hot filtration tests were performed under ideal circumstances for benzoin condensation. In these tests, tubes were filled with EtOH after 10 and 25 min, and the catalysts were separated using an external magnet. Subsequently, the catalysts were isolated, and the reactions were terminated. No discernible improvement was observed after 45 min of stirring, as confirmed by GC analysis (Fig. 10). The results suggested that the response predominantly takes place through a heterogeneous pathway. The recyclability of the heterogeneous catalysts was then investigated in model reactions. Both Choline formate and cyanide heterogeneous catalysts could be regenerated for at least five cycles (Fig. 11). Following five runs, the structure of the reused catalysts was examined using FT-IR (Fig. 2B and C), XRD, FE-SEM, and VSM (Additional file 1: Figs. S3–S5). The structure of the reused γ-Fe2O3-MgO @Ch.F. and γ-Fe2O3-MgO@Ch.CN remained comparable to that of the fresh catalyst in all analyses.
Fig. 10.
Hot filtration Test
Fig. 11.
Recyclability of γ-Fe2O3-MgO@Ch.F. and γ-Fe2O3-MgO@Ch.CN
Table 3 provides a comparative analysis of the performance of two heterogeneous catalysts employing the solvent-free condensation approach, in contrast to methodologies reported in the literature. The results highlight the remarkable efficiency of γ-Fe2O3-MgO@Ch.F. and γ-Fe2O3-MgO@Ch.CN in catalyzing condensation reactions. Notably, these catalysts address certain drawbacks associated with previously reported methods. In comparison to many earlier catalysts, the presented catalysts offer distinct advantages. They prevent the need for homogeneous catalysts, which are challenging to separate from the reaction mixture, more costly, involve hazardous organic solvents, and necessitate extended reaction times. In contrast, the protocol outlined in this study offers several benefits, including reusability, cost-effectiveness, simple preparation using readily available materials, easy separation through an external magnetic field, and the ability to achieve good to high yields within a short reaction time. Furthermore, these catalysts operate under mild and environmentally friendly conditions, establishing their superiority over previously reported counterparts. Scheme 1 illustrated a graphical depiction of the condensation reactions aided by the synthesized nanocatalyst.
Table 3.
Compares the efficiency of various methods for solvent-free condensation
| Entry | Time | Mol of cat | cat | Temperature | Yield | TON | TOF | Type of condensation | Reference |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 h | 0.01 mol | Ammonium bicarbonate | 90 °C | 91% | 1.365 | 0.068 | K.C | [66] |
| 2 | 1 h | 0.005 mol | (1-((4-chlorophenyl) amino)-1- oxopropan-2-aminium perchlorate) | 25 °C | 97% | 0.0194 | 0.0194 | K.C | [67] |
| 3 | 3 h | 0.000006 mol | Zn(pcp)(aip). (H2O) | 25 °C | 99% | 32.9 | 10.96 | K.C | [68] |
| 4 | 1 h | 0.2 mol | 1-alkyl-2,3 dimethylimida olium salts (NHC) and DBU(0.2) | 80 oC | 72% | 0.0036 | 0.0036 | B.C | [69] |
| 5 | 12 h | 0.5 mol | Imidazolium salts(NHC)/Cs2CO3 | 30 oC | 95% | 0.00475 | 0.0004 | B.C | [70] |
| 6 | 5 min | 0.02 mol | BMICl/NaOMe | 150 °C/microwave | 97% | 0.53 | 13.2 | B.C | [71] |
| 8 | 30 min | 0.067 mmol | γ-Fe2O3-MgO@Ch.F | 25 °C | 98% | 36.56 | 73.13 | K.C | This work |
| 9 | 1.5 h | 0.092 mmol | γ-Fe2O3-MgO@Ch.CN | 25 °C | 88% | 4.78 | 3.18 | B.C | This work |
K.C.: Knoevenagel condensation; B.C.: Benzoin condensation
Scheme 1.
Graphical abstract of condensation reactions using the synthesized nanocatalyst. The source of this diagram is taken from https://encrypted-tbn1.gstatic.com/images?q=tbn:ANd9GcT2p2AUmPXG_97tGU3HEWCr2pLuNGOZ7qSuHqu_ulo_kR632V75. The software tool employed to create this diagram was Chemdraw. The Scheme was designed by authors
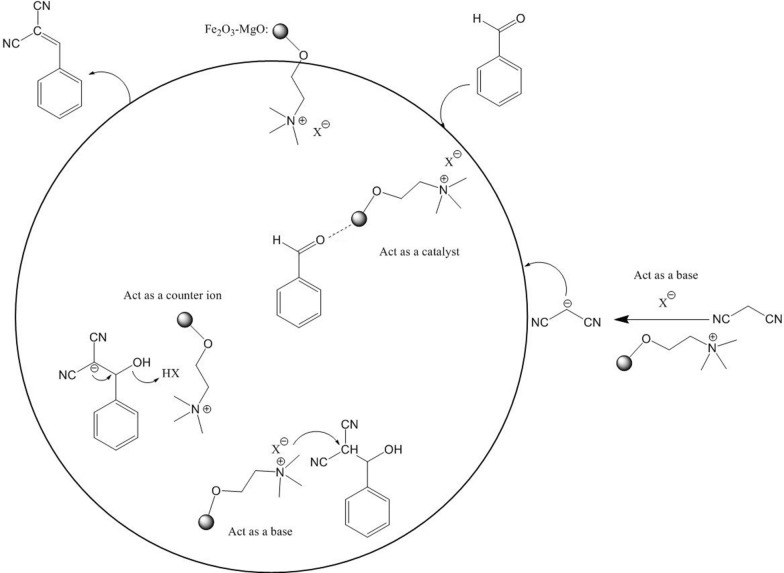
Schemes 2 and 3 depicts the proposed mechanisms for the Knoevenagel and benzoin condensations of aromatic aldehydes catalyzed by Fe2O3-MgO@cholin X (X:CN, F.) nanoparticles, respectively. In the Knoevenagel reaction, the nanocatalyst activates the aromatic aldehyde, initiating a nucleophilic attack by the methylene group of malononitrile. This sequence leads to the formation of a carbon–carbon bond, followed by the dehydration of the intermediate to yield the Knoevenagel product [72]. For the benzoin condensation, the product is synthesized through a classical mechanism [73]. The activated aromatic aldehyde undergoes nucleophilic addition with a cyanide ion, followed by the addition of a carbanion to another molecule of aldehyde. The elimination of the cyanide ion then produces benzoin as the final product. In both reactions, the nanomagnetic support with Lewis and basic sites activates the aromatic aldehydes and stabilizes the carbanion intermediates. The choline cation forms an ion pair with the organic moiety carrying a negative charge. The immobilization of the choline base ion liquid onto the magnetic support enhances the catalyst's reusability, making it an attractive and efficient option for these types of reactions.
Scheme 2.
Proposed reaction mechanism for Knoevenagel condensation
Scheme 3.
Proposed mechanism for benzoin condensation
Conclusions
This study has successfully developed and synthesized novel super-paramagnetic multifunctional nanocatalysts. The process involved a straightforward procedure using inexpensive and readily available materials, focusing on the immobilization of homogeneous organocatalysts on magnetic support. Characterization of these nanoparticles was conducted through various techniques, including FT-IR, TGA, FE-SEM, VSM, EDS, BET, and XRD. The optimization of reaction conditions in benzoin condensations resulted in a remarkable yield of 91–98% under solvent-free and room-temperature Knoevenagel reaction conditions. Additionally, a yield of 76–88% was achieved in EtOH at 25 °C for 1.5 h in benzoin condensation, using 100 mg of the catalyst. The key advantages of this protocol encompass reusability, easy separation facilitated by an external magnetic field, and high yield compared to similar homogeneous catalysts. Results from hot filtration tests, VSM, XRD, and SEM investigations indicate that these catalysts can be effectively reused up to five times without significant loss in efficiency. Moreover, this methodology has demonstrated effectiveness across a broad spectrum of aromatic and heteroaromatic aldehydes in both Knoevenagel and benzoin condensations.
Experimental
General
The chemicals were purchased from Merck, Aldrich, or Fluka without further purification. A BRUKERDRX-4F00AVANCE Advance spectrometer was used to record the NMR spectra. Electrothermal 9100 apparatus was used to measure melting points uncorrected. Nicolet IR100 instrument recorded IR spectra over a range of 400–4000 cm−1 with spectroscopic grade KBr. Vibrating magnetometers/alternating gradient force magnetometers (MD Co., Iran, www.mdk-magnetic.com) were used for the magnetic measurement experiments. Diffraction pattern of the sample was determined using a Philips X‐Pert 1710 diffraction meter. A spectrum of energy-dispersive X-rays (EDX) and field emission scanning electron microscopy (FESEM). Images were recorded on Tescan MIRA3 FE-SEM. A BET analysis was conducted to ascertain the specific surface area of the composite that was prepared, utilizing the Micromeritics Instrument Corporation/TriStar II device. TGA measurements were performed on the Simultaneous Thermal Analyzer (STA 504) (www.tainstruments.com). The Mettler Toledo DSC 1 analyzer was used to carry out differential scanning calorimetry tests on choline salts.
Preparation of choline cyanide
In dry-methanol (500 mL), choline chloride (1 mol) and sodium cyanide (1 mol) were refluxed for 6 h under inert condition. By ion exchange, Choline cyanide was obtained by evaporating the methanol solution under reduced pressure and filtering the sodium salts (NaCl, extra NaCN). An orange-liquid was formed.
Preparation of choline formate
In 500 mL methanol, choline chloride (1 mol) and sodium cyanide (1 mol) were refluxed for 6 h under air condition. By ion exchange, Choline formate was obtained by evaporating the methanol solution under reduced pressure and filtering the sodium salts (NaCl, extra NaCN). A red-liquid was formed.
Preparation of Fe2O3@MgO
In order to prepare Fe3O4 nanoparticles, 100 mL of 10 mmol FeCl3·6H2O in 5 mmol FeCl2·4H2O in aqueous solutions were heated to 85 °C. A drop-wise addition of ammonia (20 mL 27 weight %) was then added under stirring to reach a pH of 10–11. Following 1 h of stirring at room temperature, the black dispersion was heated to reflux for 1 h. Using an external magnet, the brown precipitate was separated. The effluent solution pH was neutralized by washing with deionized water several times. Additionally, ethanol was used to wash and suspend the particles. Magnetite nanoparticle suspensions were sonicated with an excess of water (1:20) and magnesium nitrate or magnesium chloride (5 mmol). Under sonication for 1 h, the reaction mixture was heated, then stirred for 12 h at 70 °C under stirring. The magnet was used to separate the particles, and the ethanol was used to wash them. Afterwards, the powder was calcined at 400 °C for 4 h in air to yield γ-Fe2O3@MgO.
Preparation of Fe2O3-MgO@Ch.X
0.5 g Fe2O3-MgO, dry EtOH (4 mL), and choline formate (under air) or choline cyanide (under argon) (5 mmol) were stirred at 25 °C for 15 min. It was then stirred at 80 °C for 12 h. Burnt brown–red catalyst was separated by potent magnet decantation. After washing with EtOH and acetone, the catalyst was dried at 50 °C for 6 h.
Solvent-free Knoevenagel condensation procedure
A mixture of catalyst (100 mg), aldehyde (2.5 mmol), malononitrile (2.5 mmol), were stirred at 25 °C. After completion of the reaction (TLC), the magnetic catalyst was separated by an external magnet. The reaction solution was diluted with ethyl acetate (2 × 2 mL). The organic layer was separated and then concentrated under reduced pressure. The pure product was obtained by recrystallization with ethyl acetate: n-Hexane.
The general procedure for benzoin condensation
An EtOH solution was stirred with catalyst (100 mg), and aldehydes (1 mmol) at 25 °C. After the reaction (TLC), ethyl acetate was used to extract the product (2 × 2 mL). Separated organic layers were concentrated under reduced pressure. A pure product was obtained by n-hexane and ethyl acetate as solvent: anti solvent.
Supplementary Information
Additional file 1: Supplementary Material 1.
Acknowledgements
We gratefully acknowledge the chemistry faculty at Tarbiat Modares University for supporting this work.
Author contributions
EK, FD, AM and AH designed the research project, carried out the experiments and drafted the manuscript, edited the manuscript. Additionally, EK, FD, AM and AH discussed the results and commented on the manuscript. AH leaded the work.
Funding
Not applicable.
Availability of data and materials
The data that supports the findings of this study are available in the supplementary material of this article. Also, All of Crude data are available at: https://zenodo.org/record/7753834#.ZBiganZBxPY (10.5281/zenodo.7753834).
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Abu-Reziq R, Alper H, Wang D, Post ML. Metal supported on dendronized magnetic nanoparticles: highly selective hydroformylation catalysts. J Am Chem Soc. 2006;128(15):5279–5282. doi: 10.1021/ja060140u. [DOI] [PubMed] [Google Scholar]
- 2.Asadi B, Mohammadpoor-Baltork I, Tangestaninejad S, Moghadam M, Mirkhani V, Landarani-Isfahani A. Synthesis and characterization of Bi (III) immobilized on triazine dendrimer-stabilized magnetic nanoparticles: a reusable catalyst for the synthesis of aminonaphthoquinones and bis-aminonaphthoquinones. New J Chem. 2016;40(7):6171–6184. doi: 10.1039/C5NJ03050A. [DOI] [Google Scholar]
- 3.Biffis A, Zecca M, Basato M. Palladium metal catalysts in Heck C–C coupling reactions. J Mol Catal A: Chem. 2001;173(1–2):249–274. doi: 10.1016/S1381-1169(01)00153-4. [DOI] [Google Scholar]
- 4.Choplin A, Quignard F. From supported homogeneous catalysts to heterogeneous molecular catalysts. Coord Chem Rev. 1998;178:1679–1702. doi: 10.1016/S0010-8545(98)00062-9. [DOI] [Google Scholar]
- 5.Chen J, Alper H. A novel water-soluble rhodium− poly (enolate-co-vinyl alcohol-co-vinyl acetate) catalyst for the hydroformylation of olefins. J Am Chem Soc. 1997;119(5):893–895. doi: 10.1021/ja9623875. [DOI] [Google Scholar]
- 6.Ajjou AN, Alper H. A new, efficient, and in some cases highly regioselective water-soluble polymer rhodium catalyst for olefin hydroformylation. J Am Chem Soc. 1998;120(7):1466–1468. doi: 10.1021/ja973048u. [DOI] [Google Scholar]
- 7.Wang D, Wang B, Ding Y, Wu H, Wu P. A novel acid–base bifunctional catalyst (ZSM-5@ Mg 3 Si 4 O 9 (OH) 4) with core/shell hierarchical structure and superior activities in tandem reactions. Chem Commun. 2016;52(87):12817–12820. doi: 10.1039/C6CC06779D. [DOI] [PubMed] [Google Scholar]
- 8.Maleki B, Taheri F, Tayebee R, Adibian F. Dendrimer-functionalized magnetic graphene oxide for knoevenagel condensation. Org Prep Proced Int. 2021;53(3):284–290. doi: 10.1080/00304948.2021.1875799. [DOI] [Google Scholar]
- 9.Saito S, Yamamoto H. Design of acid− base catalysis for the asymmetric direct aldol reaction. Acc Chem Res. 2004;37(8):570–579. doi: 10.1021/ar030064p. [DOI] [PubMed] [Google Scholar]
- 10.Song CE, Lee S-G. Supported chiral catalysts on inorganic materials. Chem Rev. 2002;102(10):3495–3524. doi: 10.1021/cr0103625. [DOI] [PubMed] [Google Scholar]
- 11.Elhamifar D, Kazempoor S, Karimi B. Amine-functionalized ionic liquid-based mesoporous organosilica as a highly efficient nanocatalyst for the Knoevenagel condensation. Catal Sci Technol. 2016;6(12):4318–4326. doi: 10.1039/C5CY01666E. [DOI] [Google Scholar]
- 12.Dumbre DK, Mozammel T, Selvakannan P, Hamid SBA, Choudhary VR, Bhargava SK. Thermally decomposed mesoporous Nickel Iron hydrotalcite: An active solid-base catalyst for solvent-free Knoevenagel condensation. J Colloid Interface Sci. 2015;441:52–58. doi: 10.1016/j.jcis.2014.11.018. [DOI] [PubMed] [Google Scholar]
- 13.Parida KM, Rath D. Amine functionalized MCM-41: An active and reusable catalyst for Knoevenagel condensation reaction. J Mol Catal A: Chem. 2009;310(1–2):93–100. [Google Scholar]
- 14.Varadwaj GBB, Rana S, Parida K. Amine functionalized K10 montmorillonite: a solid acid–base catalyst for the Knoevenagel condensation reaction. Dalton Trans. 2013;42(14):5122–5129. doi: 10.1039/c3dt32495h. [DOI] [PubMed] [Google Scholar]
- 15.Lin Y, Lei X, Yang Q, Yuan J, Ding Q, Xu J, et al. N-Heterocyclic carbene catalyzed one-pot synthesis of 2, 3-diarylquinoxalines. Synthesis. 2012;44:2699–2706. doi: 10.1055/s-0032-1316687. [DOI] [Google Scholar]
- 16.Enders D, Balensiefer T. Nucleophilic carbenes in asymmetric organocatalysis. Acc Chem Res. 2004;37(8):534–541. doi: 10.1021/ar030050j. [DOI] [PubMed] [Google Scholar]
- 17.Hachisu Y, Bode JW, Suzuki K. Thiazolium Ylide-Catalyzed intramolecular aldehyde - ketone benzoin-forming reactions: substrate scope. Adv Synth Catal. 2004;346(9–10):1097–1100. doi: 10.1002/adsc.200404092. [DOI] [Google Scholar]
- 18.Appaturi JN, Ratti R, Phoon BL, Batagarawa SM, Din IU, Selvaraj M, et al. A review of the recent progress on heterogeneous catalysts for Knoevenagel condensation. Dalton Trans. 2021;50(13):4445–4469. doi: 10.1039/D1DT00456E. [DOI] [PubMed] [Google Scholar]
- 19.Kiasat AR, Badri R, Sayyahi S. Polymer supported cyanide as an efficient catalyst in benzoin condensation: an efficient route to α-hydroxy carbonyl compounds. Bull Korean Chem Soc. 2009;30(5):1164–1166. doi: 10.5012/bkcs.2009.30.5.1164. [DOI] [Google Scholar]
- 20.Ying A, Wang L, Qiu F, Hu H, Yang J. Magnetic nanoparticle supported amine: an efficient and environmental benign catalyst for versatile Knoevenagel condensation under ultrasound irradiation. C R Chim. 2015;18(2):223–232. doi: 10.1016/j.crci.2014.05.012. [DOI] [Google Scholar]
- 21.Dangolani SK, Panahi F, Nourisefat M, Khalafi-Nezhad A. 4-Dialkylaminopyridine modified magnetic nanoparticles: as an efficient nano-organocatalyst for one-pot synthesis of 2-amino-4 H-chromene-3-carbonitrile derivatives in water. RSC Adv. 2016;6(95):92316–92324. doi: 10.1039/C6RA18078G. [DOI] [Google Scholar]
- 22.Dolatkhah Z, Javanshir S, Bazgir A, Mohammadkhani A. Magnetic Isinglass a nano-bio support for copper immobilization: Cu–IG@ Fe3O4 a heterogeneous catalyst for triazoles synthesis. ChemistrySelect. 2018;3(19):5486–5493. doi: 10.1002/slct.201800501. [DOI] [Google Scholar]
- 23.Lu AH, Salabas EL, Schüth F. Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angew Chem Int Ed. 2007;46(8):1222–1244. doi: 10.1002/anie.200602866. [DOI] [PubMed] [Google Scholar]
- 24.Vajekar SN, Shankarling GS. Choline hydroxide promoted sustainable one-pot three-component synthesis of 1H-pyrazolo [1, 2-a] pyridazine-2-carbonitriles under solvent-free conditions. Synth Commun. 2020;50(8):1147–1158. doi: 10.1080/00397911.2020.1720736. [DOI] [Google Scholar]
- 25.Mehraban JA, Azizi K, Jalali MS, Heydari A. Choline azide: new reagent and ionic liquid in catalyst-free and solvent-free synthesis of 5-substituted-1H-tetrazoles: a triple function reagent. ChemistrySelect. 2018;3(1):116–121. doi: 10.1002/slct.201702427. [DOI] [Google Scholar]
- 26.Shahiri-Haghayegh M, Azizi N, Heidarzadeh F. Greener and additive-free ring opening of epoxides by all-in-one choline systems. ChemistrySelect. 2020;5(46):14538–14542. doi: 10.1002/slct.202002815. [DOI] [Google Scholar]
- 27.Wilkes JS. A short history of ionic liquids—from molten salts to neoteric solvents. Green Chem. 2002;4(2):73–80. doi: 10.1039/b110838g. [DOI] [Google Scholar]
- 28.Kamali E, Mohammadkhani A, Pazoki F, Heydari A. Solvent-free choline derivative synthesis as a powerful organic synthesis medium. ChemistrySelect. 2023;8(10):e202204642. doi: 10.1002/slct.202204642. [DOI] [Google Scholar]
- 29.Khoshnevisan K, Vakhshiteh F, Barkhi M, Baharifar H, Poor-Akbar E, Zari N, et al. Immobilization of cellulase enzyme onto magnetic nanoparticles: applications and recent advances. Mol Catal. 2017;442:66–73. doi: 10.1016/j.mcat.2017.09.006. [DOI] [Google Scholar]
- 30.Jia H, Zhu G, Wang P. Catalytic behaviors of enzymes attached to nanoparticles: the effect of particle mobility. Biotechnol Bioeng. 2003;84(4):406–414. doi: 10.1002/bit.10781. [DOI] [PubMed] [Google Scholar]
- 31.Esfandiary N, Bagheri S, Heydari A. Magnetic γ-Fe2O3@ Cu-LDH intercalated with palladium cysteine: an efficient dual nano catalyst in tandem CN coupling and cyclization progress of synthesis quinolines. Appl Clay Sci. 2020;198:105841. doi: 10.1016/j.clay.2020.105841. [DOI] [Google Scholar]
- 32.Ghasemzadeh MA, Abdollahi-Basir MH. Fe3O4@ SiO2-NH2 nanocomposite as a robust and effective catalyst for the one-pot synthesis of polysubstituted dihydropyridines. Acta Chim Slov. 2016;63(3):627–637. doi: 10.17344/acsi.2016.2386. [DOI] [PubMed] [Google Scholar]
- 33.Ghorbani-Vaghei R, Sarmast N. Green synthesis of new pyrimido [4, 5-d] pyrimidine derivatives using 7-aminonaphthalene-1, 3-disulfonic acid-functionalized magnetic Fe3O4@ SiO2 nanoparticles as catalyst. Appl Organomet Chem. 2018;32(2):e4003. doi: 10.1002/aoc.4003. [DOI] [Google Scholar]
- 34.Ghasemzadeh MA. Synthesis and characterization of Fe3O4@ SiO2 NPs as an effective catalyst for the synthesis of tetrahydrobenzo [a] xanthen-11-ones. Acta Chim Slov. 2015;62(4):977–985. doi: 10.17344/acsi.2015.1501. [DOI] [PubMed] [Google Scholar]
- 35.Shylesh S, Schünemann V, Thiel WR. Magnetically separable nanocatalysts: bridges between homogeneous and heterogeneous catalysis. Angew Chem Int Ed. 2010;49(20):3428–3459. doi: 10.1002/anie.200905684. [DOI] [PubMed] [Google Scholar]
- 36.Rostamizadeh S, Tahershamsi L, Zekri N. An efficient, one-pot synthesis of pyrido [2, 3-d: 6, 5-d′] dipyrimidines using SBA-15-supported sulfonic acid nanocatalyst under solvent-free conditions. J Iran Chem Soc. 2015;12:1381–1389. doi: 10.1007/s13738-015-0604-1. [DOI] [Google Scholar]
- 37.Esmaeilpour M, Javidi J, Dodeji FN, Zahmatkesh S. Solvent-free, sonochemical, one-pot, four-component synthesis of 2 H-indazolo [2, 1-b] phthalazine-triones and 1 H-pyrazolo [1, 2-b] phthalazine-diones catalyzed by Fe 3 O 4@ SiO 2-imid-PMA n magnetic nanoparticles. Res Chem Intermed. 2021;47:2629–2652. doi: 10.1007/s11164-016-2462-6. [DOI] [Google Scholar]
- 38.Ghasemzadeh MA, Azimi-Nasrabad M. Nano-Fe3O4 -encapsulated silica particles bearing sulfonic acid groups as a magnetically separable catalyst for the green and efficient synthesis of 14-aryl-14H-dibenzo[a, i]xanthene-8,13-dione derivatives. Res Chem Intermed. 2016;42(2):1057–1069. doi: 10.1007/s11164-015-2073-7. [DOI] [Google Scholar]
- 39.Kumar JA, Amarnath DJ, Jabasingh SA, Kumar PS, Anand KV, Narendrakumar G, et al. One pot Green Synthesis of Nano magnesium oxide-carbon composite: Preparation, characterization and application towards anthracene adsorption. J Clean Prod. 2019;237:117691. doi: 10.1016/j.jclepro.2019.117691. [DOI] [Google Scholar]
- 40.Kaboudin B, Kazemi F, Habibi F. MgO-coated-Fe3O4 nanoparticles as a magnetically recoverable and reusable catalyst for the synthesis of 1-hydroxyphosphonates. J Iran Chem Soc. 2015;12(3):469–475. doi: 10.1007/s13738-014-0504-9. [DOI] [Google Scholar]
- 41.De Matteis L, Custardoy L, Fernández-Pacheco R, Magén C, de la Fuente JM, Marquina C, et al. Ultrathin MgO coating of superparamagnetic magnetite nanoparticles by combined coprecipitation and sol–gel synthesis. Chem Mater. 2012;24(3):451–456. doi: 10.1021/cm202306z. [DOI] [Google Scholar]
- 42.Kaboudin B, Kazemi F, Habibi F. Fe3O4@ MgO nanoparticles as an efficient recyclable catalyst for the synthesis of phosphoroamidates via the Atherton-Todd reaction. Tetrahedron Lett. 2015;56(46):6364–6367. doi: 10.1016/j.tetlet.2015.09.129. [DOI] [Google Scholar]
- 43.Tavakol H, Keshavarzipour F. Preparation of choline chloride–urea deep eutectic solvent-modified magnetic nanoparticles for synthesis of various 2-amino-4H-pyran derivatives in water solution. Appl Organomet Chem. 2017;31(11):e3811. doi: 10.1002/aoc.3811. [DOI] [Google Scholar]
- 44.Mohammadkhani A, Heydari A. Nano-magnetic-iron Oxides@ choline acetate as a heterogeneous catalyst for the synthesis of 1, 2, 3-triazoles. Catal Lett. 2021;152:1–14. [Google Scholar]
- 45.Pazoki F, Salamatmanesh A, Bagheri S, Heydari A. Synthesis and characterization of Copper (I)-cysteine complex supported on magnetic layered double hydroxide as an efficient and recyclable catalyst system for click chemistry using choline azide as reagent and reaction medium. Catal Lett. 2020;150:1186–1195. doi: 10.1007/s10562-019-03011-2. [DOI] [Google Scholar]
- 46.Zhang Y, Xia C. Magnetic hydroxyapatite-encapsulated γ-Fe2O3 nanoparticles functionalized with basic ionic liquids for aqueous Knoevenagel condensation. Appl Catal A. 2009;366(1):141–147. doi: 10.1016/j.apcata.2009.06.041. [DOI] [Google Scholar]
- 47.Azhari A, Sh MS, Golestanifard F, Saberi A. Phase evolution in Fe2O3/MgO nanocomposite prepared via a simple precipitation method. Mater Chem Phys. 2010;124(1):658–663. doi: 10.1016/j.matchemphys.2010.07.030. [DOI] [Google Scholar]
- 48.Arsalani N, Fattahi H, Nazarpoor M. Synthesis and characterization of PVP-functionalized superparamagnetic Fe3O4 nanoparticles as an MRI contrast agent. Express Polym Lett. 2010;4(6):329–338. doi: 10.3144/expresspolymlett.2010.42. [DOI] [Google Scholar]
- 49.Torres-Rivero K, Bastos-Arrieta J, Fiol N, Florido A. Metal and metal oxide nanoparticles: an integrated perspective of the green synthesis methods by natural products and waste valorization: applications and challenges. Compr Anal Chem. 2021;94:433–469. [Google Scholar]
- 50.Bhosale MA, Ummineni D, Sasaki T, Nishio-Hamane D, Bhanage BM. Magnetically separable γ-Fe2O3 nanoparticles: an efficient catalyst for acylation of alcohols, phenols, and amines using sonication energy under solvent free condition. J Mol Catal A: Chem. 2015;404:8–17. doi: 10.1016/j.molcata.2015.04.002. [DOI] [Google Scholar]
- 51.Brunauer S, Emmett PH, Teller E. Adsorption of gases in multimolecular layers. J Am Chem Soc. 1938;60(2):309–319. doi: 10.1021/ja01269a023. [DOI] [Google Scholar]
- 52.Kumar JA, Amarnath DJ, Kumar PS, Kaushik CS, Varghese ME, Saravanan A. Mass transfer and thermodynamic analysis on the removal of naphthalene from aqueous solution using oleic acid modified palm shell activated carbon. Desalin Water Treat. 2018;106:238–250. doi: 10.5004/dwt.2018.22066. [DOI] [Google Scholar]
- 53.Yang H, Dong H, Zhang T, Zhang Q, Zhang G, Wang P, et al. Calcined dolomite: an efficient and recyclable catalyst for synthesis of α, β-unsaturated carbonyl compounds. Catal Lett. 2019;149:778–787. doi: 10.1007/s10562-018-2632-9. [DOI] [Google Scholar]
- 54.Klikar M, Kityk I, Kulwas D, Mikysek T, Pytela O, Bureš F. Multipodal arrangement of push–pull chromophores: a fundamental parameter affecting their electronic and optical properties. New J Chem. 2017;41(4):1459–1472. doi: 10.1039/C6NJ02994A. [DOI] [Google Scholar]
- 55.Zhang J, Jiang T, Han B, Zhu A, Ma X. Knoevenagel condensation catalyzed by 1, 1, 3, 3-tetramethylguanidium lactate. Synth Commun. 2006;36(22):3305–3317. doi: 10.1080/00397910600941190. [DOI] [Google Scholar]
- 56.Jimenez DE, Ferreira IM, Birolli WG, Fonseca LP, Porto AL. Synthesis and biocatalytic ene-reduction of Knoevenagel condensation compounds by the marine-derived fungus Penicillium citrinum CBMAI 1186. Tetrahedron. 2016;72(46):7317–7322. doi: 10.1016/j.tet.2016.02.014. [DOI] [Google Scholar]
- 57.Taduri AK, Devi BR. Alum-Cs2CO3 as a new recyclable solid base catalyst for the efficient syntheses of arylidenemalononitriles, esters and arylcinnamic acids in water. Asian J Chem. 2014;26(7):1938–1942. doi: 10.14233/ajchem.2014.15569. [DOI] [Google Scholar]
- 58.Junek H, Sterk H. Synthesen mit nitrilen, 19. Mitt.: die partielle retro-michael-addition von tetracyanäthylen an indandion-1, 3. Tetrahedron Lett. 1968;9(40):4309–4310. doi: 10.1016/S0040-4039(00)76415-X. [DOI] [Google Scholar]
- 59.Dandia A, Jain AK, Bhati DS. Direct construction of novel dispiro heterocycles through 1, 3-dipolar cycloaddition of azomethine ylides. Tetrahedron Lett. 2011;52(41):5333–5337. doi: 10.1016/j.tetlet.2011.08.014. [DOI] [Google Scholar]
- 60.Ahadi S, Yasaei Z, Bazgir A. A clean and one-pot synthesis of spiroindoline-pyranopyrazoles. J Heterocycl Chem. 2010;47(5):1090–1094. doi: 10.1002/jhet.437. [DOI] [Google Scholar]
- 61.Shen G, Liu H, Chen J, He Z, Zhou Y, Wang L, et al. Zinc salt-catalyzed reduction of α-aryl imino esters, diketones and phenylacetylenes with water as hydrogen source. Org Biomol Chem. 2021;19(16):3601–3610. doi: 10.1039/D1OB00155H. [DOI] [PubMed] [Google Scholar]
- 62.Demir AS, Şeşenoglu Ö, Eren E, Hosrik B, Pohl M, Janzen E, et al. Enantioselective synthesis of α-hydroxy ketones via benzaldehyde lyase-catalyzed C− C bond formation reaction. Adv Synth Catal. 2002;344(1):96–103. doi: 10.1002/1615-4169(200201)344:1<96::AID-ADSC96>3.0.CO;2-Z. [DOI] [Google Scholar]
- 63.Hartman W, Dickey J. The preparation of Furil. J Am Chem Soc. 1933;55(3):1228–1229. doi: 10.1021/ja01330a063. [DOI] [Google Scholar]
- 64.Fragnelli MC, Hoyos P, Romano D, Gandolfi R, Alcántara AR, Molinari F. Enantioselective reduction and deracemisation using the non-conventional yeast Pichia glucozyma in water/organic solvent biphasic systems: preparation of (S)-1, 2-diaryl-2-hydroxyethanones (benzoins) Tetrahedron. 2012;68(2):523–528. doi: 10.1016/j.tet.2011.11.014. [DOI] [Google Scholar]
- 65.Le ZG, Ni K, Guo LT, Xie ZB. The condensation reaction of 4-nitrobenzaldehyde and rhodanine catalyzed by papain. Adv Mater Res. 2014;830:111–114. doi: 10.4028/www.scientific.net/AMR.830.111. [DOI] [Google Scholar]
- 66.van Schijndel J, Molendijk D, Spakman H, Knaven E, Canalle LA, Meuldijk J. Mechanistic considerations and characterization of ammonia-based catalytic active intermediates of the green Knoevenagel reaction of various benzaldehydes. Green Chem Lett Rev. 2019;12(3):323–331. doi: 10.1080/17518253.2019.1643931. [DOI] [Google Scholar]
- 67.Javle BR, Kinage AK. Chiral amino-acid-amide based ionic liquids as a stereoselective organocatalyst in asymmetric transfer hydrogenation of acetophenone at room-temperature. ChemistrySelect. 2018;3(9):2623–2625. doi: 10.1002/slct.201800240. [DOI] [Google Scholar]
- 68.Ma L, Wang X, Deng D, Luo F, Ji B, Zhang J. Five porous zinc (II) coordination polymers functionalized with amide groups: cooperative and size-selective catalysis. J Mater Chem A. 2015;3(40):20210–20217. doi: 10.1039/C5TA06248A. [DOI] [Google Scholar]
- 69.Aupoix A, Pégot B, Vo-Thanh G. Synthesis of imidazolium and pyridinium-based ionic liquids and application of 1-alkyl-3-methylimidazolium salts as pre-catalysts for the benzoin condensation using solvent-free and microwave activation. Tetrahedron. 2010;66(6):1352–1356. doi: 10.1016/j.tet.2009.11.110. [DOI] [Google Scholar]
- 70.Ema T, Nanjo Y, Shiratori S, Terao Y, Kimura R. Solvent-free benzoin and Stetter reactions with a small amount of NHC catalyst in the liquid or semisolid state. Org Lett. 2016;18(21):5764–5767. doi: 10.1021/acs.orglett.6b03115. [DOI] [PubMed] [Google Scholar]
- 71.Estager J, Lévêque J-M, Turgis R, Draye M. Solventless and swift benzoin condensation catalyzed by 1-alkyl-3-methylimidazolium ionic liquids under microwave irradiation. J Mol Catal A: Chem. 2006;256(1–2):261–264. doi: 10.1016/j.molcata.2006.04.055. [DOI] [Google Scholar]
- 72.Gilanizadeh M, Zeynizadeh B. Binary copper and iron oxides immobilized on silica-layered magnetite as a new reusable heterogeneous nanostructure catalyst for the Knoevenagel condensation in water. Res Chem Intermed. 2018;44:6053–6070. doi: 10.1007/s11164-018-3475-0. [DOI] [Google Scholar]
- 73.Yadav GD, Kadam AA. Atom-efficient benzoin condensation in liquid–liquid system using quaternary ammonium salts: pseudo-phase transfer catalysis. Org Process Res Dev. 2012;16(5):755–763. doi: 10.1021/op300027j. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Supplementary Material 1.
Data Availability Statement
The data that supports the findings of this study are available in the supplementary material of this article. Also, All of Crude data are available at: https://zenodo.org/record/7753834#.ZBiganZBxPY (10.5281/zenodo.7753834).