Abstract
In the present study, IL-12 gene-transduced B78-H1 melanoma cells (B78/IL-12) were used in combination with IL-15 to treat melanoma-bearing mice. Genetically modified B78/IL-12 cells, when injected subcutaneously, induced strong activation of antitumour mechanisms resulting in complete loss of tumourigenicity. In a therapeutic model, intratumoural injection of irradiated B78/IL-12 cells significantly delayed tumour growth and led to the regression of melanoma in one case. Similarly, consecutive daily injections of IL-15 markedly inhibited tumour progression with occasional curative effects. When used in combination, vaccination with B78/IL-12 cells and treatment with IL-15 caused eradication of established tumours in all treated mice. The combined treatment with B78/IL-12 cells and IL-15 activated not only a local response against tumour, but also induced systemic antitumour immunity that led to a delay or inhibition of tumour development at a distant site. In vitro studies demonstrated that when used together, B78/IL-12 cells and IL-15 induced a shift from a type Th2 to a type Th1 response. Activation of the antitumour immune response in double-treated mice resulted, in part, from stimulation of IFN-γ production and was accompanied by the development of cytotoxic effectors in the spleen. As shown in a macrophage tumouricidal assay, macrophages could also play a role in the antitumour effects. The results confirmed that vaccination with IL-12 gene-modified tumour cells is superior to the treatment with unmodified tumour cell vaccine and, additionally, showed that IL-15 is an excellent candidate for adjuvant therapy, inducing synergistically not only a delay of tumour growth but also its complete eradication.
Keywords: IL-12, IL-15, Cancer gene therapy, Melanoma
Introduction
Interleukin 12 (IL-12), produced by macrophages [44] and dendritic cells [13, 39], has been found in experimental animal models to be one of the most effective cytokines in cancer therapy. IL-12 promotes Th1-type immune responses and has been shown to enhance cytotoxic activity of natural killer (NK) cells, T cells, lymphokine activated killer cells, and macrophages [4, 31, 45]. Through induction of chemokines, IL-12 exerts also anti-angiogenic effects decreasing blood supply to tumours [47].
The antitumour activity of recombinant IL-12 has been demonstrated in numerous experimental models in mice [4, 9, 19, 21]. However, systemic administration of recombinant IL-12 in clinical trials was associated with severe toxicity [3, 36]. Therefore, new approaches to IL-12 delivery have been explored. One way of avoiding IL-12-induced toxicity is to introduce the IL-12 gene into tumour cells, which then secrete IL-12 continuously at the tumour site. IL-12 gene therapy enabling local IL-12 release at the tumour site has proved to be less toxic than therapy with recombinant IL-12 [36]. In gene therapy experiments IL-12 has been shown to be more effective in antitumour activity as compared with other cytokines used in the same schedule as IL-12 [37]. Hence, IL-12 gene therapy may be a very promising means to treat cancer. Gene therapy with IL-12 is now entering human trials [14, 15, 42] due to encouraging results in preclinical studies [12, 38, 43]. No major toxicity has been observed in initial phase I study [42]. However, some date suggest that effectiveness of this approach could be effectively enhanced by other treatment modalities.
Antitumour efficacy of gene therapy with IL-12 has already been successfully potentiated when combined with some immunotherapeutic approaches. Antitumour effects of IL-12 gene therapy have been increased by combination with costimulatory molecules [11, 28] and cytokines, both interleukins [1, 6, 31, 43] and chemokines [33]. Cytokines in these studies have been directly applied as purified proteins [31] or produced by genetically engineered cells [1, 6, 33, 43]. In one of our previous studies we observed potentiated antitumour effects with IL-12 used in combination with IL-15 [20], and these results were subsequently confirmed by other authors [17]. Therefore, we decided to examine now the therapeutic efficacy of combination therapy based on application of a tumour vaccine, consisting of IL-12 gene-transduced tumour cells, supplemented with systemic administration of IL-15.
Materials and methods
Mice
(C57BL/6xDBA/2)F1 mice, hereafter called B6D2F1, 8 to 12 weeks of age, were used in the experiments. Breeding pairs were obtained from the Inbred Mice Breeding Center of the Institute of Immunology and Experimental Medicine, Wrocław, Poland. Mice were bred in a local animal facility and were kept in conventional conditions with full access to food and water during experiments.
Tumour cells
The B78-H1 melanoma, a weakly immunogenic amelanotic subclone of the murine B16 melanoma cell line, was kindly provided by Dr. L. H. Graf, Chicago, Ill. Tumour cells were cultured in Dulbecco’s MEM supplemented with 10% heat-inactivated FCS, antibiotics, 2-mercaptoethanol (50 μM) and L-glutamine (2 mM) (all from Gibco BRL-Life Technologies Ltd, Paisley, UK) (DMEM culture medium). Cells were maintained in 25 cm2 tissue flasks (Nunc, Roskilde, Denmark) at 37°C in a humidified atmosphere of 5% CO2 and were passaged 2–3 times weekly.
Construction of a retroviral vector carrying the murine IL-12 gene and generation of IL-12-expressing B78-H1 cells
Murine IL-12 gene was excised from pEM12 plasmid by digestion with XhoI and NotI enzymes. The insert was cloned into Samenv retroviral vector into dephosphorylated XhoI and NotI sites. Packaging cells PA317 (American Type Culture Collection, CRL 9078) were electroporated with Samenv-IL-12 vector cDNA suspended in PBS. Subsequently, the cells were plated and cultured in culture medium [DMEM + 10% FCS + gentamicin (0.08 mg/ml)]. Two days following the transfection, the cells were exposed to selection medium containing geneticin (G418, Life Technologies Inc.) (1 mg/ml). Clones of cells resistant to the antibiotic were collected and screened for production of recombinant retroviruses. For transduction purposes, culture medium collected after 24 h of incubation from Samenv-IL-12-packaging cell culture (90% of confluency) was used for transduction of B78-H1. Shortly, B78-H1 cells were incubated in the infectious medium supplemented with polybrene (4 μg/ml) for 24 h. Subsequently, the procedure was repeated, and the cells were incubated in infectious medium for another 24 h. After this time the infectious medium was removed and cells were incubated in selection medium (as described above). Tumour cells resistant to the selection were collected and were further cloned by limiting dilution technique. From 12 clones of B78-H1 cells transduced with IL-12 gene, the clone “9” that produced the highest amount of IL-12 [272 pg/1×106 cells/24 h, as determined by ELISA (Biosource International, Inc., Camarillo, Calif.)] was denoted B78/IL-12 and used in the experiments described below.
IL-15
Recombinant human IL-15 (specific activity 4.96×105 U/μg protein) was kindly provided by Immunex Corp., Seattle, Wash. This cytokine was diluted with 0.1% bovine serum albumin (BSA; Sigma Chemicals, St. Louis, Mo.) in PBS for in vivo experiments.
In vivo experiments
B78-H1 melanoma cells were injected into the footpad of the right hind limb at the dose of 2×105 cells in 20 μl PBS. On day 3 after tumour cell inoculation, mice were treated intratumourally with 1×106 viable IL-12-secreting B78/IL-12 cells (irradiated, 4,500 rads) or, as a control, irradiated melanoma cells containing empty vector (B78/e.v.). IL-15 was given at daily doses of 2 μg in 20 μl 0.1% BSA-PBS on days 6–12. Each injection of a vaccine or IL-15 was paralleled by i.t. injections of diluent into mice from control groups. Local tumour growth was determined by measuring the diameter of the footpad (anterior-posterior dimension) with callipers every 3–4 days, starting from day 7 following inoculation of tumour cells. Tumour size was calculated according to the formula: tumour diameter = (diameter of footpad with growing tumour) − (diameter of non-treated contralateral footpad). Independently, in all experiments, two-dimensional measurements of the footpad (anterior-posterior + side-to-side measure) were performed to evaluate the tumour growth. No substantial difference between the one-dimensional and two-dimensional methods of evaluation of the tumour size was found. Mice that experienced complete tumour regression were rechallenged with 1×105 live nontransduced B78-H1 cells 2 months after the primary tumour inoculation.
ELISPOT assay
To determine antigen-specific antitumour response in tumour-draining lymph nodes (LN), ELISPOT assay was performed. One day after the last therapeutic injection of IL-15 (day 13 of the experiment; for details, see in vivo experiments) 96-well MaxiSorp flat-bottom NuncImmuno microtiter plates (Nunc) were coated with anti-mouse IFN-γ monoclonal antibodies (mAbs) and with anti-mouse IL-4 mAbs (both from Pharmingen/Becton Dickinson, San Diego, Calif.) and incubated for 24 h at 4°C. Plates were washed four times with PBST (1% Tween 20 in PBS) and blocked for 2 h with 0.5% BSA (Sigma, St. Louis, Ill.) in culture medium. After washing five times with PBS, 1×103 target B78-H1 cells suspended in 0.1 ml warm culture medium were added to the wells. Two h later, 1×105/0.1 ml effector LN cells were added to the wells, giving E:T ratio 100:1. Plates were incubated for either 24 h or 48 h at 37°C, 5% CO2, washed five times with PBST and biotinylated secondary antibodies were added to wells (anti-IFN-γ or anti-IL-4, both from Pharmingen/Becton Dickinson). Two h later plates were washed with PBST, and 50 μl of streptavidin-alkaline phosphatase (Gibco BRL) solution were added to the wells. Fifteen min later plates were washed and color reaction was developed using alkaline phosphatase substrate kit III (Vector Laboratories Inc., Burlingame, Calif.). Treatment groups were coded, and two independent investigators counted dots. Cell activity was estimated as dots according to the formula: dots = (experimental dots formation − effector cells spontaneous dots formation − target cells spontaneous dots formation).
Assay for macrophage cytostatic/cytotoxic activity
B6D2F1 mice were treated i.p. with irradiated B78/IL-12 cells (1×106; day 0) and IL-15 (2 µg, daily, days 3–9), alone or in combination. On day 10, peritoneal exudate cells were collected by lavage of the peritoneal cavity with chilled Dulbecco’s phosphate-buffered saline (PBS; Gibco BRL) containing 10 U/ml heparin. After centrifugation, cells were plated in 92×21-mm plastic Petri dishes (Nunc) in culture medium, incubated for 1.5 h at 37°C in 5% CO2, and washed gently three times with warm RPMI-1640 to remove non-adherent cells. Adherent cells with morphological characteristics of macrophages were harvested by forced rinsing with cold Dulbecco’s PBS, using a 50-ml syringe and 20-gauge needle and, additionally, remaining cells were scraped by a rubber policeman. Following one washing, macrophages were resuspended in culture medium, counted and dispensed into wells of a 96-well flat-bottomed microtitre plate at a concentration of 2.5×104 cells in 0.1 ml per well. Then, 5×103 B78-H1 melanoma cells in 0.1 ml DMEM culture medium were added to each well (within 2 h), giving the E:T ratio 5:1. Cultures were incubated for 3 days. The cytostatic/cytotoxic effects of macrophages on melanoma cells were tested in a standard MTT assay, where cells were incubated with MTT for the last 4 h of a 3-day culture. Antitumour effects of macrophages from B78/IL-12 vaccine- and/or IL-15-treated mice, expressed as relative viability of tumour cells (% of control cultures with B78-H1 cells and no macrophages) was calculated according to the formula: relative viability = [(Aexp − Amin)/Amax)] × 100%, where: Aexp = absorbance in experimental macrophage cultures with B78-H1 melanoma cells; Amin = absorbance of macrophages alone; Amax = absorbance of control cultures containing B78-H1 melanoma cells alone. The means and standard errors of the mean were determined for quadruplicate samples.
In vitro cytotoxicity assay
A standard 6-h 51Cr release assay was used to assess the cytotoxic activity of in vitro restimulated splenocytes. Briefly, splenocytes from mice treated with irradiated B78/IL-12 melanoma cells and IL-15 (for details, see in vivo experiments) were isolated as described elsewhere [20], depleted of red blood cells by Tris-ammonium chloride, and resuspended to 1×106 cells/ml in RPMI-1640 culture medium. The cells were plated in 24-well plates and restimulated with 1×105 irradiated B78-H1 melanoma cells ±10 units/ml IL-2 for 3 days. Then 0.5 ml of supernatant was removed from each well followed by replacement with 2×104 51Cr-labeled B78-H1 cells suspended in 0.5 ml culture medium. After a 6-h incubation, aliquots of 100 μl supernatant were collected, and the radioactivity of 51Cr released from target cells was measured in a gamma counter (Wallac, Gaithersburg, Md.). The percentage of specific lysis was calculated according to the formula: [(a−b)/(c−b)]×100, where a is the percentage of 51Cr released from target cells incubated with splenocytes, b is the percentage of 51Cr released spontaneously from target cells incubated alone and c is maximal 51Cr release from target cells incubated with 1% Triton X-100. Calculations were based on triplicate samples.
Determination of IFN-γ in culture supernatants
Splenocytes from mice treated with irradiated B78/IL-12 cells and/or IL-15 (the same treatment protocol as in “In vivo experiments”), isolated 1 day after the last injection of IL-15, were incubated in 24-well plates (1×106 cells in 1 ml) in the presence of IL-12 (1 ng/ml) or irradiated B78-H1 cells (1×105 cells/ml) ±10 units/ml IL-2. Amounts of IFN-γ in supernatants from 1- or 3-day cultures were measured using ELISA kit purchased from R&D Systems Europe Ltd. (Abingdon, UK) (DuoSet ELISA development system).
Flow cytometric analysis
The phenotype of lymphocytes from tumour-draining lymph nodes (LN) was determined by flow cytometry (FACSCalibur, Becton Dickinson, Mountain View, Calif.). Mice were treated with B78/IL-12 cells and/or IL-15 as described in “In vivo experiments.” LNs were removed 1 day after the last injection of IL-15, pooled (4–6 mice per group), and the single-cell suspension was stained with fluorescein isothiocyanate (FITC)- or phycoerythrin (PE)-coupled monoclonal antibodies (mAbs). The following mAbs, all from PharMingen/Becton Dickinson, were used for direct staining of cells: RM4-5(PE) (rat anti-mouse CD4) 53-6.7(PE) (rat anti-mouse CD8a), DX5(PE) (rat anti-mouse anti-CD49b, pan-NK cells), 1D3(PE) (rat anti-mouse CD19) and 145-2C11(FITC) (hamster anti-mouse CD3e). Isotype controls included: A19-3(FITC) (hamster isotype control) (PharMingen/Becton Dickinson) and LO-DNP-16(FITC) (rat isotype control) (Serotec, Kidlington, Oxford, UK). Samples were analysed using CellQuest software (Becton Dickinson). Results shown are representative of two independent experiments.
Statistical analysis
Differences between samples in tests in vitro and differences in tumour diameter in experiments in vivo were analysed for significance by Student’s t test (two-tailed). For the difference in complete tumour regressions, groups were analysed by Wilcoxon’s test. Additionally, to determine the difference in tumour-take rate between groups the χ2 test was used.
Results
Tumourigenicity of B78-H1 cells transduced with the gene for IL-12 (B78/IL-12)
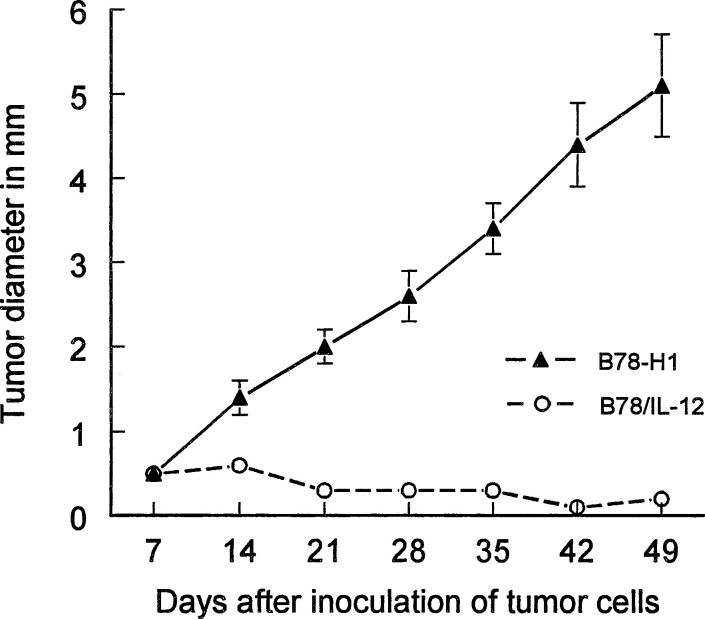
To assess the effect of IL-12 secretion on tumour development, B78/IL-12 melanoma cells (secreting 272 pg/1×106 cells/24 h) were inoculated into the footpad of the right hind limb and examined for tumour formation. As shown in Fig. 1, B78/IL-12 melanoma cells were completely rejected in all mice. In some of these mice long-lasting antitumour systemic immunity developed since two of eight mice were able to reject a rechallenge with live B78-H1 cells (1×105 cells, 2.5 times LD100 dose) performed into the contralateral footpad 2 months after the primary tumour inoculation.
Fig. 1.
In vivo growth of IL-12 gene-transduced B78-H1 melanoma cells (B78/IL-12). 5×105 B78/IL-12 melanoma cells or non-transduced parental B78-H1 cells were injected into the footpad of the right hind limb of B6D2F1 mice
Effects of local treatment with irradiated B78/IL-12 melanoma cells in combination with IL-15 on tumour growth
To evaluate whether vaccination with IL-12 gene-modified B78-H1 cells and administration of IL-15 have therapeutic activity, B78-H1 melanoma-bearing mice were treated i.t. with 1×106 live irradiated IL-12-secreting B78/IL-12 cells (on day 3 after inoculation of melanoma) either alone or with IL-15 (2 μg for 7 consecutive days, starting from day 6). As shown in Fig. 2A, tumour progressed more slowly both in vaccine-treated mice and in mice injected with IL-15, in comparison with control animals (P<0.01, on day 49). However, the strongest therapeutic effect was observed in mice treated with B78/IL-12 cell vaccine in combination with IL-15; all tumours regressed in this group (P<0.01 in comparison with B78/IL-12 vaccine-treated or IL-15-treated mice, and P<0.001 in comparison with controls; Fig. 2B). In one of seven cured mice from the double-treated group, no tumour growth was observed after a rechallenge.
Fig. 2.
Therapeutic effects of B78/IL-12 vaccine in combination with IL-15. Mice were injected into the footpad of the right hind limb with 2×105 B78-H1 melanoma cells. On day 3, mice were treated intratumourally (i.t.) with 1×106 irradiated live B78/IL-12 cells (or control B78/e.v. cells) followed by seven consecutive daily i.t. injections of IL-15 (2 μg per dose, from day 6 to 12). (A) Growth kinetics and (B) percentage of tumour-free mice vaccinated and/or treated with IL-15. Groups contained seven mice
To assess the advantage of IL-12 gene-modified B78-H1 cells over wild-type tumour cells, used as vaccine alone or in combination with IL-15, mice with B78-H1 melanoma were treated with irradiated B78-H1 cell containing empty vector (B78/e.v.) and IL-15 in the same treatment schedule. This treatment induced retardation of the tumour growth in mice treated with B78/e.v. cells + IL-15 in comparison with control mice (P<0.001, on day 49), but not when compared with the group of mice treated with IL-15 alone (Fig. 2A). However, combination of the vaccine containing B78/IL-12 cells + IL-15 was more effective than the treatment based on B78/e.v. cells + IL-15 (P<0.05, on day 49).
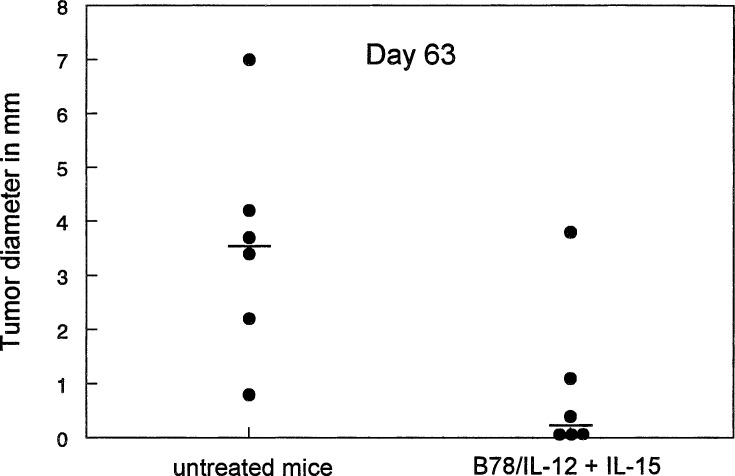
To study if local injections of B78/IL-12 vaccine in combination with IL-15 are able to stimulate effective systemic antitumour response and influence the growth of established tumour at the distant site, mice injected into the footpad of the left hind limb with B78-H1 melanoma cells (day 0) were treated 3 days later into the contralateral footpad with 1×106 irradiated B78/IL-12 vaccine followed by seven consecutive injections of IL-15 (2 μg per dose, at the site of vaccination, starting from day 6). As shown in Fig. 3, all control mice developed tumours within 2 months after inoculation of B78-H1 melanoma cells, while at the same time tumours appeared in only 50% of mice treated with B78/IL-12 + IL-15 (P<0.05).
Fig. 3.
Systemic therapeutic effect of B78/IL-12 vaccine in combination with IL-15 against B78-H1 melanoma. Mice were injected into the footpad of the left hind limb with 1×105 B78-H1 melanoma cells. On day 3, mice were injected into the contralateral footpad with a vaccine containing 1×106 irradiated live B78/IL-12 cells and from days 6 to 12 with 2 μg IL-15. Horizontal bars indicate the median value for each group
Effects of vaccination with B78/IL-12 melanoma cells and IL-15 on cell populations in tumour-draining lymph nodes
Macroscopically, marked enlargement of lymph nodes in B78/IL-12 vaccine- and/or IL-15-treated mice was observed, reflecting expansion of different lymphoid cell populations. The highest cellularity (about a five-fold increase of the cell number in comparison with control mice) was observed in mice treated with B78/IL-12 vaccine and IL-15 together and, as shown in Fig. 4, CD3+CD8+ population predominated among the cells in this group. Of note, NK cell number was also raised in this group (about a nine-fold increase) as well as in the group of mice treated with IL-15 alone (about a ten-fold increase). In the treated mice, stimulation of CD3+CD4+ T cells was also noticeable, especially in mice injected with combination of B78/IL-12 and IL-15 (a five-fold increase) (Fig. 4). The similar phenomenon of CD3+CD8+ and NK cell expansion, albeit to smaller extent, was found in the spleen (data not presented).
Fig. 4.
FACS analysis of lymph node cells from mice treated with B78/IL-12 cells in combination with IL-15. Mice inoculated with 2×105 B78-H1 melanoma cells (day 0) were treated with 1×106 live irradiated B78/IL-12 cells on day 3 and/or 2 μg IL-15, injected daily for 7 consecutive days (starting from day 6). On day 13 lymph nodes were isolated, lymphocytes were stained with anti-CD3, anti-CD4, anti-CD8, anti-NK or anti-CD19 mAbs and analyzed using FACSCalibur flow microfluorocytometer. Groups contained four to six mice
Effects of vaccination with B78/IL-12 cells in combination with IL-15 on specific antitumour response
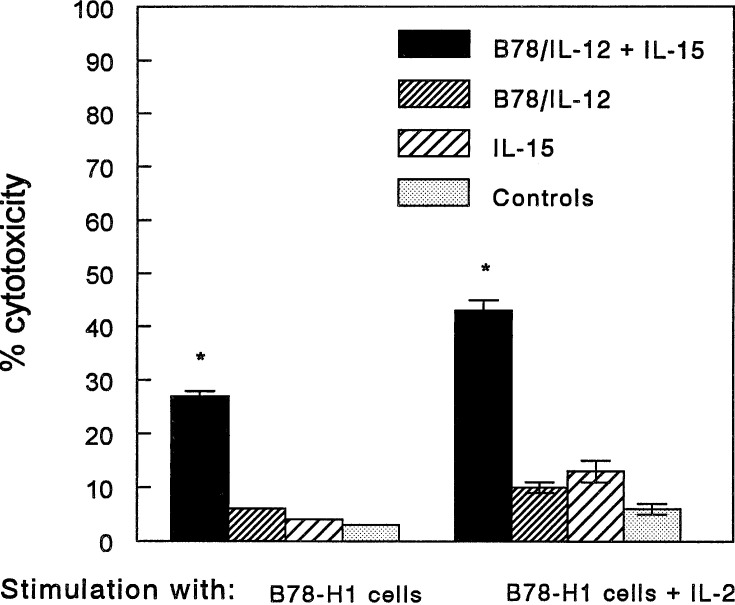
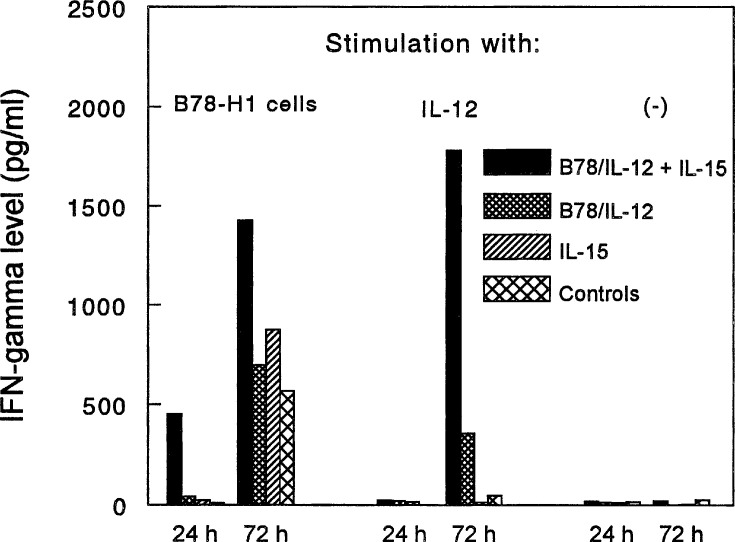
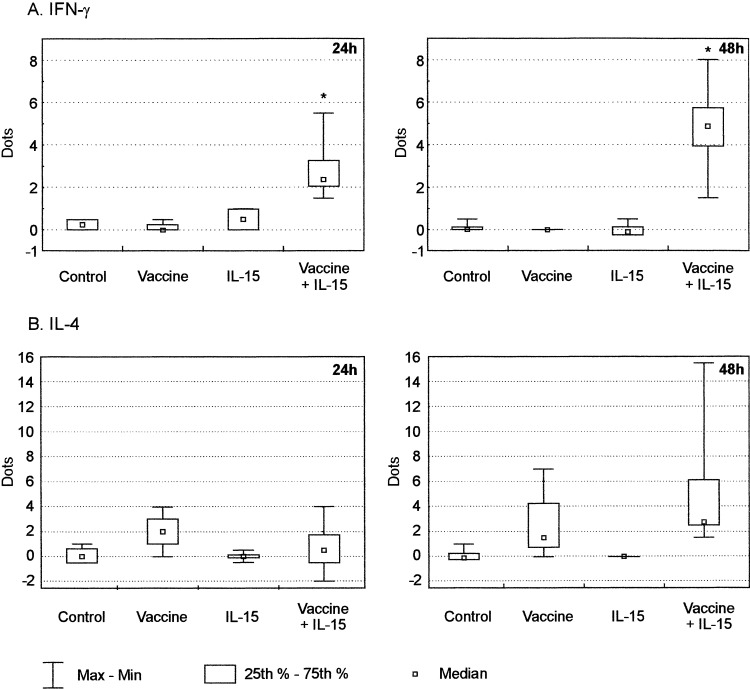
To examine the role of specific immune mechanisms in the antitumour effects of the combined treatment with B78/IL-12 vaccine and IL-15, splenocytes from the treated mice were cultured in vitro and their cytotoxic activity or ability to produce IFN-γ was assessed in functional assays. As shown in Fig. 5, splenocytes from the double-treated mice, restimulated in vitro with irradiated melanoma cells, exerted a strong cytotoxic effect against parental B78-H1 melanoma cells, additionally augmented in cultures supplemented with IL-2. This effect was confirmed in a direct microscopic inspection of restimulated cultures. As shown in Fig. 6, irradiated B78-H1 cells, which were used as stimulatory cells, were almost completely killed in 2-day cultures by splenocytes from mice treated with B78/IL-12 vaccine + IL-15, while in all other groups, including the control, many cells were still alive at that time. Incubation of spleen cells in the presence of the B78-H1 cells stimulated IFN-γ production strongly (Fig. 7). Levels of this cytokine were especially increased in cultures of splenocytes from mice treated with both B78/IL-12 cells and IL-15 and resulted probably from activation of both NK and Th1 cells. The extremely high level of IFN-γ in cultures of splenocytes stimulated with IL-12 supports the role of NK cells in this phenomenon (Fig. 7). In turn, the role of Th1 cells in secretion of IFN-γ was confirmed in ELISPOT assay. As shown in Fig. 8, combination of B78/IL-12 cells and IL-15 induced Th1-type response: restimulation with B78-H1 cells in the assay led to the increase in number of IFN-γ- but not IL-4-producing cells in cultures of splenocytes from the double-treated mice. The number of both populations in either vaccine- or IL-15-treated mice was not different from the control.
Fig. 5.
Cytotoxic effect of splenocytes from mice treated with B78/IL-12 vaccine and/or IL-15 against B78-H1 cells. Details of treatment (see Materials and methods). Splenocytes were incubated for 3 days in the presence of irradiated stimulatory B78-H1 cells with or without IL-2 (10 units/ml). Cytotoxic effect was measured against 51Cr-labelled B78-H1 melanoma cells (E:T =50:1); 6 h incubation time. *P<0.001, in comparison with all other groups (IL-15 alone, vaccine alone and control)
Fig. 6.
Cytotoxic effect of splenocytes from mice treated with B78/IL-12 vaccine and/or IL-15 during stimulation with irradiated B78-H1 melanoma cells. Photographs show 2-day cultures of irradiated B78-H1 cells admixed with splenocytes from mice treated with B78/IL-12 cells + IL-15 (A), B78/IL-12 cells alone (B), IL-15 alone (C) or diluent (D). Details of treatment (see Materials and methods). Note the strong cytotoxic effect against stimulatory cells in culture of splenocytes from double-treated mice—almost no live tumour cells are present in this culture, in comparison with other cultures (arrows). Large dark aggregations indicate accumulation of effector cells and apoptotic tumour cells
Fig. 7.
IFN-γ production in cultures of splenocytes from mice treated with B78/IL-12 vaccine and/or IL-15. Details of treatment (see Materials and methods). Splenocytes were stimulated with either irradiated B78-H1 cells or IL-12 (1 ng/ml), and the amount of IFN-γ in supernatants from 1- or 3-day cultures was measured by ELISA
Fig. 8.
Treatment with B78/IL-12 vaccine and IL-15 stimulates cells secreting IFN-γ. Mice were injected with B78-H1 melanoma cells and treated with B78/IL-12 vaccine and/or IL-15. Details of treatment (see Materials and methods). One day after the last injection of IL-15, lymphocytes from tumour-draining lymph nodes were isolated and restimulated with B78-H1 cells for 24 or 48 h. Frequencies of cells secreting IFN-γ (A) or IL-4 (B) are expressed as dot numbers per well. *P<0.05, in comparison with all other groups (IL-15 alone, vaccine alone and control)
Effects of vaccination with B78/IL-12 cells in combination with IL-15 on tumouridcidal activity of macrophages
As shown in Fig. 7, vaccination with B78/IL-12 cells in combination with IL-15 strongly stimulated production of IFN-γ. Since this cytokine, in turn, potently stimulates macrophages and induces their tumouricidal activity, we decided to examine whether the combined treatment of mice with B78/IL-12 vaccine and IL-15 could produce a similar effects in vivo. The data presented in Table 1 show that, indeed, macrophages could contribute to the antitumour effects in mice treated with B78/IL-12 cells + IL-15. Ex vivo peritoneal macrophages from double-treated mice inhibited proliferation of B78-H1 melanoma cells in cultures more effectively than those from either B78/IL-12- or IL-15-treated mice (34% vs. 81% viability, and 34% vs. 46% viability, P<0.001 and P<0.05, respectively). This effect was probably, at least in part, mediated by nitric oxide (NO) since the highest concentration of NO was detected in cultures of macrophages from mice treated with a vaccine and IL-15 (data not presented).
Table 1.
Antitumour activity of peritoneal macrophages from B78/IL-12 cell- and/or IL-15-treated mice against B78-H1 melanoma cells. Mice were treated i.p. with irradiated B78/IL-12 cells (1×106; day 0) and IL-15 (2 µg, daily, days 3–9), alone or in combination. On day 10, peritoneal macrophages were dispensed into wells of a 96-well flat-bottomed microtitre, and B78-H1 melanoma cells were added to each well within 2 h (final E:T ratio =5:1). After a 3-day incubation, cytostatic/cytotoxic effect of macrophages on melanoma cells was tested in a standard MTT assay (see Materials and methods)
| Viability of B78-H1 melanoma cells in cultures with macrophages | |
|---|---|
| (% of control cultures; mean ± SD) | |
| Mice treated with: | |
| B78/IL-12 + IL-15 | 34±3* |
| B78/IL-12 | 81±3 |
| IL-15 | 46±6 |
| Diluent | 77±3 |
| Control B78-H1 culture | 100±6 |
*Significantly different in comparison with IL-15-treated mice (P<0.05) and mice treated with B78/IL-12 or diluent (P<0.001)
Discussion
The present studies provide the evidence that, using IL-15 as an adjuvant, it is possible to enhance greatly the antitumour effect of the vaccine consisting of IL-12 gene-transduced tumour cells.
Since its discovery, IL-12 has been recognized as a very promising cytokine, with the potential for application in tumour therapy [4, 45]. However, systemic administration of IL-12 in humans was associated with unacceptablely severe toxicity [3, 10] and prompted investigators to explore an IL-12-based gene therapy approach as an alternative. The antitumour potential of IL-12 gene-modified tumour cell vaccines has been reported in a number of different murine models [7, 29, 31, 32, 43], and clinical trials based on IL-12-releasing tumour and normal cells have been started [15, 42].
The available data show that several mechanisms contribute to the overall therapeutic effects of IL-12 gene-engineered vaccines. Firstly, IL-12 promotes MHC-restricted antitumour response. As shown by Nanni et al. [32], dendritic cells accumulate at the site of inoculation of IL-12-releasing melanoma cells and, as demonstrated by many authors and what was confirmed in our studies (Fig. 8), stimulation of the immune response in the presence of IL-12 induces Th1 cell development. IL-12 also augments the development of CD8+ T cells [30]. Secondly, IL-12 is a strong activator of NK cells and, in fact, was originally called natural killer cell stimulatory factor [18]. Finally, by augmenting production of antiangiogenic chemokines, IL-12 can inhibit development of the tumour via nonimmune mechanisms [47].
Despite many spectacular results in animal models, vaccine-based immunotherapeutical approaches (including immunization with tumour cells engineered to express cytokines) are still at the stage of clinical trials in humans, and from numerous vaccines that have been clinically tested, none but one (Melacine in Canada) have been registered to date [2] (see also http://cancernet.nci.nih.gov). Most studies concern melanoma, which has been recognized as the most immunogenic tumour. In some clinical trials a paradoxical phenomenon was observed: the induction of tumour-reactive cells following immunization did not strictly correlate with the objective antitumour response [40, 41]. This observation suggests that, apart from specific antitumour immune effectors, non-MHC-restricted mechanisms can play an important role in the overall antitumour reactions induced by vaccines, and were the main rationale for combining IL-15 with B78/IL-12 vaccine in our studies. We were also encouraged by our previous results in which we observed a synergistic antitumour effect of IL-12 and IL-15 against B16F10 melanoma [20].
IL-15 was identified based on its ability to stimulate proliferation of the IL-2-dependent CTLL-2 cell line, and shares many biological actions with IL-2. However, IL-15 differs from IL-2 in several important aspects [27]. First of all, IL-15 is a much more important physiological inducer of NK cell development and activation than IL-2 [24], and as we showed in our earlier studies, it is the main contributor to the increased NK activity following prolonged administration of IL-12 and IL-15 [20]. Next, IL-15 is an important factor in the differentiation of NKT cells [34]. This cytokine also participates in the development of the optimal adaptive T cell-mediated response. On the one hand, IL-15 (but not IL-2) plays a dominant role in initiating T cell divisions in vivo [23] and is critical in early activation of antigen-presenting cells [35]. On the other hand, IL-15 is recognized as a cytokine necessary for expansion and development of memory and cytotoxic CD8+ T cells [16, 26]. Presumably, at least some of the above-mentioned mechanisms could be responsible for the development of cytotoxic effectors against B78-H1 cells in mice treated with B78/IL-12 cells and IL-15 (Figs. 5, 6).
As shown in Fig. 7, splenocytes from double-treated mice released large amounts of IFN-γ, both in 1- and 3-day cultures. The synergistic effect of IL-12 and IL-15 on IFN-γ production has been well documented [6, 20], and as we discussed elsewhere, could result from reciprocal upregulation of IL-12 and IL-15 receptors by both cytokines [20]. Probably, the increased level of IFN-γ was responsible for augmented tumouricidal activity of macrophages in mice treated with B78/IL-12 vaccine and IL-15 (Table 1) and could contribute in such a way to the synergistic antitumour effects of the combined treatment in our model (Figs. 2, 3). The importance of macrophages (as well as granulocytes) in the antitumour effect of IL-12 and IL-15 was also supported by Di Carlo et al. [6]. In a model of NK-depleted nude mice grafted with human N592 lung cancer cells engineered to secrete IL-12 and IL-15, they found dense infiltrations of macrophages and granulocytes and strong expression of inducible nitric oxide synthase at the site of rejected tumour cells. The important role of macrophages as effector cells in IL-12-induced tumour rejection was also stressed by Tsung et al. [46].
Apart from stimulation of IFN-γ production, the combined treatment with IL-12 and IL-15 acts synergistically on other mechanisms that could be significant to the overall antitumour effect of this combination. Using a model of double-transduced TS/A murine adenocarcinoma cells (with genes for IL-12 and IL-15 in IFN-γ-knockout mice), Comes et al. [5] reported an IFN-γ-independent synergistic antitumour effect of IL-12 and IL-15 mediated by CD8+ T cells and involving secondary-secreted chemokines such as MIP-2 and MCP-1 as well as cytokines TNF-α and GM-CSF.
Encouraging results with IL-12 gene therapy in animal models of neoplasia prompted several groups of investigators to test the efficacy of this approach in cancer patients. Development of DTH reactivity against autologous melanoma cells and reductions of the tumour size were observed in some patients treated with either IL-12 gene-modified melanoma cells [42] or IL-12 gene-transduced fibroblasts [15]. However, many phase III clinical studies with other antitumour vaccines have demonstrated that the combination of vaccines with agents of proved adjuvantic properties is the condition sine qua non of effective antitumour response [22] (see also http://www.cancer.gov/serch/clinical_trials). Because IL-15 has pleiotropic activities bridging the innate and adaptive immune mechanisms and plays a critical role in NK cell development [8, 25, 48], the properties that remarkably enhanced efficacy of IL-12-based vaccine in our model (Figs. 2, 3), we think that the use of this cytokine as an adjuvant in other cancer vaccine approaches is worth further exploration.
Acknowledgements
This work was supported by the Foundation for the development of Diagnostics and Therapy, the Foundation for Polish Science, and Grant 1 M19/W2 from the Medical University, Warsaw, Poland. Ahmad Jalili is a recipient of the Postgraduate School of Molecular Medicine fellowship.
References
- 1.Addison Gene Ther. 1998;5:1400. doi: 10.1038/sj.gt.3300731. [DOI] [PubMed] [Google Scholar]
- 2.Berd Expert Opin Biol Ther. 2002;2:335. doi: 10.1517/14712598.2.3.335. [DOI] [PubMed] [Google Scholar]
- 3.Cohen Science. 1995;270:908. [Google Scholar]
- 4.Colombo Cytokine Growth Factor Rev. 2002;13:155. doi: 10.1016/S1359-6101(01)00032-6. [DOI] [PubMed] [Google Scholar]
- 5.Comes Eur J Immunol. 2002;32:1914. doi: 10.1002/1521-4141(200207)32:7<1914::AID-IMMU1914>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 6.Di J Immunol. 2000;165:3111. doi: 10.4049/jimmunol.165.6.3111. [DOI] [PubMed] [Google Scholar]
- 7.Dunussi-Joannopoulos Blood. 1999;94:4263. [PubMed] [Google Scholar]
- 8.Fehniger TA, Cooper MA, Caligiuri MA. Interleukin-2 and interleukin-15: immunotherapy for cancer. Cytokine Growth Factor Rev. 2002;13:169. doi: 10.1016/s1359-6101(01)00021-1. [DOI] [PubMed] [Google Scholar]
- 9.Golab Int J Mol Medicine. 1999;3:537. doi: 10.3892/ijmm.3.5.537. [DOI] [PubMed] [Google Scholar]
- 10.Gollob J Immunother. 2001;24:91. doi: 10.1097/00002371-200101000-00011. [DOI] [PubMed] [Google Scholar]
- 11.Hallez Int J Cancer. 1999;81:428. doi: 10.1002/(SICI)1097-0215(19990505)81:3<428::AID-IJC17>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 12.Hanna Cancer Gene Ther. 2001;8:151. doi: 10.1038/sj.cgt.7700287. [DOI] [PubMed] [Google Scholar]
- 13.Hochrein J Immunol. 2001;166:5448. doi: 10.4049/jimmunol.166.9.5448. [DOI] [PubMed] [Google Scholar]
- 14.Jenks Natl Cancer Inst. 1996;88:756. [Google Scholar]
- 15.Kang Human Gene Ther. 2001;12:671. doi: 10.1089/104303401300057388. [DOI] [PubMed] [Google Scholar]
- 16.Kennedy J Exp Med. 2000;191:771. doi: 10.1084/jem.191.5.771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kimura Cancer Immunol Immunother. 2000;49:71. doi: 10.1007/s002620050604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kobayashi J Exp Med. 1989;170:827. [Google Scholar]
- 19.Lasek Cancer Immunol Immunother. 1997;45:100. doi: 10.1007/s002620050408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lasek Eur Cytokine Network. 1999;10:345. [PubMed] [Google Scholar]
- 21.Lasek Cancer Gene Ther. 2000;7:1581. doi: 10.1038/sj.cgt.7700263. [DOI] [PubMed] [Google Scholar]
- 22.Le Curr Opin Oncol. 2002;14:641. doi: 10.1097/00001622-200211000-00009. [DOI] [PubMed] [Google Scholar]
- 23.Li Nature Med. 2001;7:114. doi: 10.1038/83253. [DOI] [PubMed] [Google Scholar]
- 24.Liu Immunol Today. 2000;21:113. doi: 10.1016/S0167-5699(99)01581-9. [DOI] [PubMed] [Google Scholar]
- 25.Lodolce Cytokine Growth Factor Rev. 2002;13:429. doi: 10.1016/S1359-6101(02)00029-1. [DOI] [PubMed] [Google Scholar]
- 26.Lu Clin Cancer Res. 2002;8:3877. [PubMed] [Google Scholar]
- 27.Ma J Exp Med. 2000;191:753. doi: 10.1084/jem.191.5.753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Martinet J Natl Cancer Inst. 2000;92:931. doi: 10.1093/jnci/92.11.931. [DOI] [PubMed] [Google Scholar]
- 29.Melero Trends Immunol. 2001;22:113. doi: 10.1016/s1471-4906(00)01824-x. [DOI] [PubMed] [Google Scholar]
- 30.Mortarini Cancer Res. 2000;60:3559. [PubMed] [Google Scholar]
- 31.Nagai Cancer Investig. 2000;18:206. [Google Scholar]
- 32.Nanni Cancer Res. 1998;58:1225. [PubMed] [Google Scholar]
- 33.Narvaiza J Immunol. 2000;164:3112. doi: 10.4049/jimmunol.164.6.3112. [DOI] [PubMed] [Google Scholar]
- 34.Ohteki J Immunol. 1997;159:5931. [PubMed] [Google Scholar]
- 35.Ohteki Nature Immunol. 2001;2:1138. doi: 10.1038/ni729. [DOI] [PubMed] [Google Scholar]
- 36.Rakhmilevich Gene Ther. 1997;8:1303. doi: 10.1089/hum.1997.8.11-1303. [DOI] [PubMed] [Google Scholar]
- 37.Rakhmilevich J Immunother. 1999;22:135. [Google Scholar]
- 38.Rakhmilevich Cancer Gene Ther. 2000;7:826. doi: 10.1038/sj.cgt.7700176. [DOI] [PubMed] [Google Scholar]
- 39.Reise J Exp Med. 1997;186:1819. doi: 10.1084/jem.186.11.1819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rivoltini Immunol Rev. 2002;188:97. doi: 10.1034/j.1600-065X.2002.18809.x. [DOI] [PubMed] [Google Scholar]
- 41.Rosenberg Nature Med. 1998;4:321. [Google Scholar]
- 42.Sun Gene Ther. 1998;5:481. doi: 10.1038/sj.gt.3300619. [DOI] [PubMed] [Google Scholar]
- 43.Tanaka Cancer Gene Ther. 2000;7:1481. doi: 10.1038/sj.cgt.7700251. [DOI] [PubMed] [Google Scholar]
- 44.Trinchieri Adv Immunol. 1998;70:83. doi: 10.1016/s0065-2776(08)60387-9. [DOI] [PubMed] [Google Scholar]
- 45.Trinchieri Nature Rev Immunology. 2003;3:133. doi: 10.1038/nri1001. [DOI] [PubMed] [Google Scholar]
- 46.Tsung Cancer Res. 2002;62:5069. [PubMed] [Google Scholar]
- 47.Voest J Natl Cancer Inst. 1995;87:581. doi: 10.1093/jnci/87.8.581. [DOI] [PubMed] [Google Scholar]
- 48.Waldman Immunity. 2001;14:105. [Google Scholar]