Abstract
Classification criteria have been developed for rheumatoid arthritis (RA) and other rheumatic diseases in order to gather a homogeneous patient population for clinical studies and facilitate the timely implementation of therapeutic measures. Although classification criteria are not intended to be used for diagnosis, they are frequently used to support the diagnostic process in clinical practice, including clinical decision-making. The 2010 American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (EULAR) classification criteria for RA are capable of identifying the majority of symptomatic patients with RA already in the earliest stages of the disease who are not yet showing radiographic changes. These patients will also profit from the early implementation of therapy with disease-modifying antirheumatic drugs (DMARDs). However, the risk of misclassification is higher as compared with the former 1987 ACR criteria, which were considerably less sensitive to the recognition of patients with early RA. Of note, the presence of rheumatoid factors (RFs) and anticitrullinated protein antibodies (ACPAs) has been attributed equal weight in the 2010 ACR/EULAR criteria and may contribute up to 50% of the score needed for being classified as RA. However, while ACPAs have been proven to be the most specific serological markers of RA, the specificity of RF is moderate, especially at lower titres. This may lead to the misclassification of RF-positive patients and, consequently, the unjustified implementation of DMARD therapy. Therefore, issues arise on how comprehensive the criteria should be and whether they should be updated and adapted to findings from the past two decades that might increase both their specificity and sensitivity.
Keywords: Anti-Citrullinated Protein Antibodies; Rheumatoid Factor; Arthritis, Rheumatoid; Methotrexate
KEY MESSAGES.
The current American College of Rheumatology/European Alliance of Associations for Rheumatology 2010 rheumatoid arthritis classification criteria provide the same scoring weight to positive rheumatoid factor (RF) and anticitrullinated protein antibodies (ACPA) results.
However, the specificity of RF in the absence of ACPA is low, whereas the specificity of ACPA in the absence of RF is high.
ACPA/RF dual positivity is considerably more specific than ACPA single positivity and patients with arthralgia with dual positivity have a higher risk of progressing towards RA than those solely positive for ACPA.
Changing weight factors according to the function of serological grades improves the specificity of the RA classification criteria without affecting sensitivity.
The added value of other antimodified protein antibodies to the increased sensitivity and/or specificity of the criteria remains a topic of debate.
Introduction
Rheumatoid arthritis (RA) is a highly inflammatory, debilitating autoimmune disease with complicated multisystem involvement that is difficult to diagnose in its early stages when the characteristic disease features are not yet apparent.1 The process of diagnosis is complex and requires the synthesis of many data points, typically beyond a simple algorithm-based set of criteria.2 3 Hence, it is not possible to clearly define diagnostic criteria for RA (or other systemic autoimmune diseases); however, recommendations and guidelines for the management and treatment of RA have been developed.4 5 In contrast, classification criteria are used as a standardised means for including a well-defined set of patients in clinical studies to ensure better comparability across studies. Importantly, classification criteria are not intended to be used for diagnosis.6 Nonetheless, they may have some impact on diagnosis and clinical decision-making. Therefore, improving both the specificity and sensitivity of classification criteria across different rheumatic diseases remains a challenge and, in that context, especially the increasing importance of autoantibody testing is being discussed.7
RA is classified according to the American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (EULAR) 2010 criteria,8 which, apart from clinical items (joint counts), encompass laboratory parameters including inflammation markers (C reactive protein and erythrocyte sedimentation rate) and, most importantly, the hallmark autoantibodies of RA, rheumatoid factor (RF) and anticitrullinated protein/peptide antibodies (ACPAs). The maximum score of the criteria is 10, with a score ≥6 allowing the classification as RA. Among the laboratory parameters, an elevated inflammation marker contributes one point, the presence of RF or ACPA contributes two points if detectable (ie, low positive) and three points if present at levels >3 times the upper limit of normal as defined by the reference range of the assay. Consequently, laboratory items may account for up to two-thirds of the scores needed to classify definite RA and are thus given a higher weight than in any other set of classification criteria for rheumatic autoimmune diseases.9
From this viewpoint, we are discussing the performance of current criteria and their potential refinement according to insights obtained over the past years, including differences between ACPA and RF contribution as well as the emergence of novel antibodies that might be considered for inclusion to increase the sensitivity and specificity of the criteria.
Sensitivity and specificity of the 2010 ACR/EULAR classification criteria for RA
The 2010 ACR/EULAR classification criteria for RA were created to replace the 1987 revised criteria of the ACR10 because those were considered outdated for several reasons. First and most importantly, the presence of radiographic changes, in general, a more late-stage manifestation of RA, was one of the seven criteria. Therefore, it was the primary goal of the new criteria to classify patients in the earliest stages of disease before radiographic changes are observed, allowing them to be included in clinical studies on early RA. In addition, the criteria should enable a more timely start of therapy with disease-modifying antirheumatic drugs (DMARDs) in individuals at high risk for developing RA or related chronic inflammatory joint diseases. Thus, the ACR/EULAR consortium argued that attention should be focused on the important need for earlier diagnosis and the institution of effective disease-suppressing therapy.8 Moreover, ACPA had to be taken into consideration because it was unknown in 1987 and appeared to be more specific for RA than RF, which was the only serological marker in the 1987 revised ACR criteria. Despite the lower specificity of RF, the ACR/EULAR 2010 classification criteria attribute the same scoring weight to positive RF and ACPA results, which is partially explained by the fact that ACPA assays were then not yet as widely used as they are nowadays. Meanwhile, RF and ACPA are commonly determined in the majority of routine laboratories, and numerous studies have confirmed that ACPA has a significantly higher specificity and likelihood ratio (LR) for RA than RF.11 Moreover, the classification criteria do not consider a difference in scoring weight for RF/ACPA dual positivity, which has proven to be significantly more specific compared with single positivity.12–14In line with this, it has become increasingly evident that patients with arthralgia showing dual positivity have a higher risk of progressing towards RA than patients solely positive for ACPA or RF.15–17
The ACR/EULAR 2010 RA classification criteria turned out to be more sensitive than the 1987 revised ACR criteria and to facilitate early identification of patients with RA immediately after the onset of symptoms, that is, synovitis in at least one small or two large joints. This allows not only the inclusion of patients with early arthritis in clinical studies but also a more timely start of therapy with disease-modifying antirheumatic drugs (DMARDs), depending on the healthcare setting. The higher sensitivity has been demonstrated in several studies, which, however, also revealed decreased specificity.18–21 The lower specificity partially relates to RF and ACPA positivity, as was documented in a study in which the criteria were applied to a cohort of patients with very early inflammatory arthritis.20 Among misclassified patients (non-RA with a score ≥6), 20.5% were RF and/or ACPA positive; of note, the majority of these patients were RF positive, whereas ACPA was less frequently detected. Furthermore, dual RF/ACPA positivity was largely confined to patients with RA. Hence, at the seventh International Congress on Controversies in Rheumatology and Autoimmunity, a discussion was initiated to improve the specificity of the criteria attributed to the ACPA-based and RF-based serological signatures by changing the weight factors of the serological grades.22
Increasing specificity by refining the weights of marker antibodies
To substantiate the arguments in favour of improving the specificity of the criteria by taking the different specificities of ACPA and RF into account, we reanalysed the data from an early arthritis cohort that had previously been used for validation of the 2010 ACR/EULAR criteria.20 The Stop Arthritis Very Early (SAVE) multicentre trial cohort included 325 patients with early arthritis, of whom, after a 1-year follow-up period, 132 patients were diagnosed with RA and 193 patients had other rheumatic diseases.23 A detailed analysis of the serological data revealed very high specificity of RF/ACPA dual positivity, which showed an LR of 21.6 for the development of RA, with only three patients with non-RA showing dual positivity (table 1). Remarkably, this was not dependent on titres because, even at low titres, dual positivity proved highly specific. ACPA single positivity was observed in 9/132 patients with RA and 3/193 patients with non-RA (all undifferentiated arthritis) and was, therefore, quite specific for RA. In contrast, single RF positivity did not discriminate RA from non-RA, not even when present in high titers (table 1). Therefore, giving the same weight to RF and ACPA is no longer justified, also taking into consideration that ACPA is nowadays routinely determined together with RF all over the world, which had not yet been the case in 2010.
Table 1.
Serology results of the Stop Arthritis Very Early early arthritis cohort20 22 23
| RA | Non-RA | LR | Non-RA diagnoses | |
| n | 132 | 193 | ||
| RF low, ACPA− | 2.3% | 4.1% | 0.6 | 4 UDA, 1 SpA, 1 ReA, 1 remission |
| RF high, ACPA− | 2.3% | 2.1% | 1.1 | 2 UDA, 1 Sjögren, 1 viral infection |
| ACPA low, RF− | 1.5% | 0.0 | n.a. | – |
| ACPA high, RF− | 4.5% | 1.6% | 3.3 | 3 UDA |
| ACPA+ and RF+ | 45.4% | 2.1% | 21.6 | 2 UDA, 1 remission |
| ACPA− and RF− | 44.0% | 90.2% | 0.49 |
LR, likelihood ratio; RA, rheumatoid arthritis; ReA, reactive arthritis; SpA, spondyloarthropathy; UDA, undifferentiated arthritis.
Suggested modification of the 2010 ACR/EULAR classification criteria for RA
These data are in accordance with published literature11–16 and confirm that (1) a weak positive RF result in the absence of ACPA is not specific for RA; (2) a high positive RF result in the absence of ACPA is indicative but not specific for RA; (3) a positive ACPA result in the absence of RF is quite specific for RA and (4) the co-occurrence of ACPA and RF is highly specific for RA and significantly more specific than ACPA by itself.
On the basis of these findings and of data from a recent study in which different scoring systems were compared in other early arthritis cohorts (Van Hoovels et al., manuscript in preparation), we propose that different weights should be given to the serological markers, considering the antibody type (RF vs ACPA), the antibody level and the combined positivity. Thus, a suggestion for refined weights of serological scores for RA classification would be as follows: ‘0’ for RF/ACPA negativity as well as for low positive RF; ‘1’ for high RF; ‘2’ for low ACPA; ‘3’ for high ACPA and ‘4’ for ACPA/RF dual positivity (independent of the titre). Therefore, we suggest modifying the ACR/EULAR 2010 criteria accordingly (table 2).
Table 2.
Suggestion for refined American College of Rheumatology/European Alliance of Associations for Rheumatology classification criteria
| Domain | Category | Point score |
| A | Joint involvement | 1–5 |
| B | Serology | 0–4 |
| RF and ACPA negative | 0 | |
| RF low, ACPA negative | 0 | |
| RF high, ACPA negative | 1 | |
| ACPA low, RF negative | 2 | |
| ACPA high, RF negative | 3 | |
| ACPA and RF positive (>cut-off) | 4 | |
| C | Acute-phase reactants | 0–1 |
| D | Duration of symptoms | 0–1 |
| Maximum score | 11 | |
| Score for classification as RA | 6 |
ACPA, anticitrullinated protein antibodies ; RA, rheumatoid arthiritis; RF, rheumatoid factor.
Applying the refined score to the SAVE cohort reduced misclassification of patients with seropositive non-RA from 79% (ACR/EULAR scoring) to 47.4% (refined scoring) without affecting diagnostic sensitivity for seropositive RA, which was identical for both classification scores (figure 1). This reduction is statistically significant and certainly not negligible, particularly when considering the impact of classification criteria on the more complex diagnostic process and, even more importantly, the implementation of DMARD therapy. This was already discussed by the consortium that developed the criteria, which aimed to ‘develop uniform criteria for discriminating, from a population of individuals presenting with undifferentiated synovitis, the subgroup with the highest probability of persistent or erosive RA who would therefore benefit from early DMARD intervention’ before a definite diagnosis could be established.8 In line with this, a recently published large observational study showed that DMARD therapy employed in patients with undifferentiated arthritis not fulfilling the ACR/EULAR criteria did not have similar positive long-term benefits as in RA.24
Figure 1.
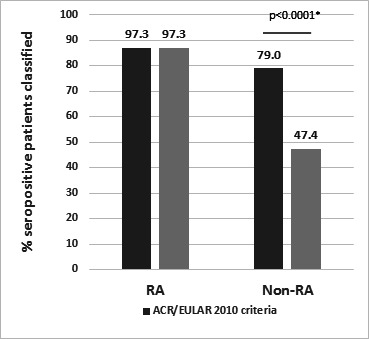
Classification of patients with seropositive RA and non-RA in the Stop Arthritis Very Early cohort according to the established 2010 ACR/EULAR classification criteria and refined criteria. Application of the refined serological scoring system improved specificity significantly (79% vs 47.4% misclassified patients with non-RA, p<0.0001), whereas sensitivity was not affected (97.3% correctly classified patients with RA). *χ2 for proportions (MedCalc Software, Ostend, Belgium). RA, rheumatoid arthritis.
The refined scoring system by itself would theoretically allow the classification of patients without joint involvement (patients with a serological score of ‘4’, elevated acute-phase reactants and disease duration >6 weeks); however, the target population should remain the same, which requires that clinical apparent synovitis (swollen joints) without better explanation8 remains a prerequisite for classification as RA according to the proposed refined criteria.
Outlook
Classification of at-risk patients
For patients at high risk of developing RA, that is, patients with arthralgia suggestive of progression to RA, discrete classification criteria would be required in which the antibody status plays a pivotal role.15–17 Indeed, already more than 10 years ago, a risk calculation form was suggested in which single IgM-RF positivity contributed 0 points, low single ACPA positivity 2 points, high single ACPA positivity 3 points and RF and ACPA double positivity 4 points, with 5–6 points indicating intermediate risk and ≥7 points high risk.15 Criteria for patients at risk of progressing to RA will become essential for inclusion in arthritis prevention trials.24 These intervention trials are now being considered as a novel strategy aimed at preventing or at least delaying the development of clinical RA.25 26
Seronegative patients: are they really all seronegative?
Taking into account that serostatus is highly weighted in the 2010 ACR/EULAR criteria, it is not surprising that seronegative patients take longer to be classified (and diagnosed) and also often show higher disease activity at diagnosis. It is estimated that among seronegative patients, 20%–30% show features of autoimmunity that are not covered by currently used assays in routine diagnostics. Therefore, the added diagnostic value of other antimodified protein antibodies, such as antibodies to carbamylated or acetylated peptides, is being discussed.27–31 However, until now, no striking evidence has been provided that such antibodies would increase the sensitivity of the criteria because their specificity is low when ACPA or RF are absent. Thus, a small gain in sensitivity would come at the expense of specificity and make the criteria more complicated. Furthermore, the inclusion of IgA isotypes of RF and ACPA has been suggested but is hampered by the fact that IgA isotypes are almost invariably co-occurring with IgM RF or IgG ACPA, even though the dual occurrence of IgM and IgA RF (in the absence of ACPA) appears to be quite specific for RA.13 14
Nevertheless, it needs to be emphasised that the presence of multiple reactivities (including IgA isotypes) appears to be a feature highly characteristic of RA12 13 30 31 while the presence of a single autoreactivity is generally not very helpful for diagnostics (with the notable exception of ACPA). Therefore, assays allowing concomitant detection of a larger number of autoantibodies might indeed increase the sensitivity and specificity of RA serodiagnostics and, moreover, allow further stratification of patients into clinically distinct subgroups. Thus, including multiplex results in the classification criteria might become an option in the future.
Acknowledgments
We wish to thank Professor Zoltan Szekanecz and Professor Annmaria Iagnocco for stimulating discussions and Professor Klaus Machold for providing clinical data for the SAVE cohort and Professor Annette van der Helm-van Mil for sharing data on the Leiden Early Arthritis Cohort.
Footnotes
@Stiddyo
Contributors: All authors contributed to writing the manuscript and sharing their ideas and opinions on classification criteria for rheumatoid arthritis.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
Not applicable.
References
- 1.Smolen JS, Aletaha D, Barton A, et al. Rheumatoid arthritis. Nat Rev Dis Primers 2018;4:18001. 10.1038/nrdp.2018.1 [DOI] [PubMed] [Google Scholar]
- 2.Aletaha D, Smolen JS. Diagnosis and management of rheumatoid arthritis: a review. JAMA 2018;320:1360–72. 10.1001/jama.2018.13103 [DOI] [PubMed] [Google Scholar]
- 3.Baker JF. Diagnosis and differential diagnosis of RA. UpToDate; 2024. Available: www.update.com/content [Google Scholar]
- 4.Fraenkel I, Bathon JM, England BR, et al. ACR guideline for the treatment of RA. Arthritis Care Res 2021;73:924–39. 10.1002/acr.24596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nagy G, Roodenrijs NMT, Welsing PMJ, et al. EULAR points to consider for the management of difficult-to-treat rheumatoid arthritis. Ann Rheum Dis 2022;81:20–33. 10.1136/annrheumdis-2021-220973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aggarwal R, Ringold S, Khanna D, et al. Distinctions between diagnostic and classification criteria. Arthritis Care Res (Hoboken) 2015;67:891–7. 10.1002/acr.22583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Damoiseaux J. Autoantibodies in the criteria of autoimmune diseases:is it sufficient to know that the test is positive. J Transl Autoimmun 2022;5:100144. 10.1016/j.jtauto.2022.100144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aletaha D, Neogi T, Silman AJ, et al. Rheumatoid arthritis classification criteria: an American college of rheumatology/European League against rheumatism collaborative initiative. Ann Rheum Dis 2010;69:1580–8. 10.1136/ard.2010.138461 [DOI] [PubMed] [Google Scholar]
- 9.Van Hoovels L, Studenic P, Sieghart D, et al. Impact of autoimmune serology test results on RA classification and diagnosis. J Transl Autoimmun 2022;5:100142. 10.1016/j.jtauto.2022.100142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Arnett FC, Edworthy SM, Bloch DA, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988;31:315–24. 10.1002/art.1780310302 [DOI] [PubMed] [Google Scholar]
- 11.Van Hoovels L, Vander Cruyssen B, Sieghart D, et al. A multicenter study to improve clinical interpretation of rheumatoid factor and anti-citrullinated protein/peptide antibodies test results. RMD Open 2022;8:e002099. 10.1136/rmdopen-2021-002099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Verheul MK, Böhringer S, van Delft MAM, et al. Triple positivity for anti-citrullinated protein autoantibodies, rheumatoid factor, and anti-carbamylated protein antibodies conferring high specificity for rheumatoid arthritis: implications for very early identification of at-risk individuals. Arthritis Rheumatol 2018;70:1721–31. 10.1002/art.40562 [DOI] [PubMed] [Google Scholar]
- 13.Sieghart D, Platzer A, Studenic P, et al. Determination of autoantibody isotypes increases the sensitivity of serodiagnostics in rheumatoid arthritis. Front Immunol 2018;9:876. 10.3389/fimmu.2018.00876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Van Hoovels L, Vander Cruyssen B, Sieghart D, et al. IgA rheumatoid factor in rheumatoid arthritis. Clin Chem Lab Med 2022;60:1617–26. 10.1515/cclm-2022-0244 [DOI] [PubMed] [Google Scholar]
- 15.van de Stadt LA, Witte BI, Bos WH, et al. A prediction rule for seropositive arthritis in seropositive arthralgia patients. Ann Rheum Dis 2013;72:1920–6. 10.1136/annrheumdis-2012-202127 [DOI] [PubMed] [Google Scholar]
- 16.ten Brinck RM, van Steenbergen HW, van Delft MAM, et al. The risk of individual autoantibodies, autoantibody combinations and levels for arthritis development in clinically suspect arthralgia. Rheumatology 2017;56:2145–53. 10.1093/rheumatology/kex340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.van Boheemen L, van Schaardenburg D. Predicting rheumatoid arthritis in at-risk individuals. Clin Ther 2019;41:1286–98. 10.1016/j.clinthera.2019.04.017 [DOI] [PubMed] [Google Scholar]
- 18.van der Linden MPM, Knevel R, Huizinga TWJ, et al. Classification of rheumatoid arthritis: comparison of the 1987 American College of Rheumatology criteria and the 2010 American College of Rheumatology/European League against rheumatism criteria. Arthritis Rheum 2011;63:37–42. 10.1002/art.30100 [DOI] [PubMed] [Google Scholar]
- 19.Cader MZ, Filer A, Hazlehurst J, et al. Performance of the 2010 ACR/EULAR criteria for rheumatoid arthritis: comparison with 1987 ACR criteria in a very early synovitis cohort. Ann Rheum Dis 2011;70:949–55. 10.1136/ard.2010.143560 [DOI] [PubMed] [Google Scholar]
- 20.Biliavska I, Stamm TA, Martinez-Avila J, et al. Application of the 2010 ACR/EULAR classification criteria in patients with very early inflammatory arthritis: analysis of sensitivity, specificity and predictive values in the SAVE study cohort. Ann Rheum Dis 2013;72:1335–41. 10.1136/annrheumdis-2012-201909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Radner H, Neogi T, Smolen JS, et al. Performance of the 2010 ACR/EULAR classification criteria for rheumatoid arthritis: a systematic literature review. Ann Rheum Dis 2014;73:114–23. 10.1136/annrheumdis-2013-203284 [DOI] [PubMed] [Google Scholar]
- 22.Steiner G, Van Hoovels L, Csige D, et al. Should ACR/EULAR criteria be revised changing the RF and ACPA scores. Autoimmun Rev 2024;23:103421. 10.1016/j.autrev.2023.103421 [DOI] [PubMed] [Google Scholar]
- 23.Machold KP, Landewé R, Smolen JS, et al. The stop arthritis very early (SAVE) trial, an international Multicentre, randomised, double-blind, placebo-controlled trial on glucocorticoids in very early arthritis. Ann Rheum Dis 2010;69:495–502. 10.1136/ard.2009.122473 [DOI] [PubMed] [Google Scholar]
- 24.Verstappen M, Matthijssen XME, van der Helm-van Mil AHM. Undifferentiated arthritis: a changing population who did not benefit from enhanced disease-modifying anti-rheumatic drug strategies-results from a 25 year longitudinal inception cohort. Rheumatology (Oxford) 2022;61:3212–22. 10.1093/rheumatology/keab880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mankia K, Siddle HJ, Kerschbaumer A, et al. EULAR points to consider for conducting clinical trials and observational studies in individuals at risk of rheumatoid arthritis. Ann Rheum Dis 2021;80:1286–98. 10.1136/annrheumdis-2021-220884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Van Steenbergen HW, Cope AP, van der Helm-van Mil AHM. RA prevention in arthralgia: fantasy or reality? Nat Rev Rheum 2023;19:767–77. 10.1038/s41584-023-01035-y [DOI] [PubMed] [Google Scholar]
- 27.De Stefano L, D’Onofrio B, Gandolfo S, et al. Seronegative rheumatoid arthritis: one year in review 2023. Clin Exp Rheumatol 2023;41:554–64. 10.55563/clinexprheumatol/go7g26 [DOI] [PubMed] [Google Scholar]
- 28.Reed E, Hedström AK, Hansson M, et al. Presence of autoantibodies in ‘seronegative’ rheumatoid arthritis associates with classical risk factors and high disease activity. Arthritis Res Ther 2020;22:170. 10.1186/s13075-020-02191-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shi J, van Steenbergen HW, van Nies JAB, et al. The specificity of anti-carbamylated protein antibodies for rheumatoid arthritis in a setting of early arthritis. Arthritis Res Ther 2015;17:339. 10.1186/s13075-015-0860-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Studenic P, Alunno A, Sieghart D, et al. Presence of anti-acetylated peptide antibodies (AAPA) in inflammatory arthritis and other rheumatic diseases suggests Discriminative diagnostic capacity towards early rheumatoid arthritis. Ther Adv Musculoskelet Dis 2021;13:1759720X211022533. 10.1177/1759720X211022533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rodriguez-Martínez L, Bang H, Regueiro C, et al. Improved classification of rheumatoid arthritis with a score including anti-acetylated ornithine antibodies. Sci Rep 2020;10:19263. 10.1038/s41598-020-73919-y [DOI] [PMC free article] [PubMed] [Google Scholar]


