Abstract
Purpose of review:
In this review, we explore the investigational applications of optical coherence tomography (OCT) in retinopathy of prematurity (ROP), the insights they have delivered thus far, and key milestones for its integration into the standard of care.
Recent findings:
While OCT has been widely integrated into clinical management of common retinal diseases, its use in pediatric contexts has been undermined by limitations in ergonomics, image acquisition time, and field of view. Recently, investigational handheld OCT devices have been reported with advancements including ultra-widefield view, non-contact use, and high-speed image capture permitting real-time en face visualization. These developments are compelling for OCT as a more objective alternative with reduced neonatal stress compared to indirect ophthalmoscopy and/or fundus photography as a means of classifying and monitoring ROP.
Summary:
OCT may become a viable modality in management of ROP. Ongoing innovation surrounding handheld devices should aim to optimize patient comfort and image resolution in the retinal periphery. Future clinical investigations may seek to objectively characterize features of peripheral stage and explore novel biomarkers of disease activity.
Keywords: retinopathy of prematurity, optical coherence tomography, biomarker
Introduction
Retinopathy of prematurity (ROP) is a leading cause of childhood blindness. While nearly always avoidable with prompt diagnosis and treatment, preventable blindness due to ROP occurs in approximately 20,000–50,000 infants annually due to missed, delayed, or incorrect diagnoses.[1–6] The current standard of care for monitoring involves weekly or bi-weekly dilated binocular indirect ophthalmoscopy with scleral depression at the bedside, or review of ultra-widefield digital fundus imaging via telemedicine. Since 1984, the International Classification of Retinopathy of Prematurity (ICROP) has established guidelines for interpreting clinical findings from these exams to classify disease according to features including zone and stage of retinopathy, and presence of plus disease.[7] Despite longstanding acceptance of these recommendations, classification of ROP remains highly subjective and prone to interobserver variability, undermining standardization of management paradigms across providers and institutions.[8-12**] This observation has driven exploration of alternative approaches for ROP diagnosis. Of these, optical coherence tomography (OCT) has shown promise as a means of observing pathology and identifying novel biomarkers of disease with higher resolution and objectivity than ophthalmoscopic exam alone.[13-15*] While OCT has been adopted as standard of care in other leading causes of blindness including age-related macular degeneration and diabetic retinopathy, it has yet to be broadly implemented into clinical practice in ROP after over a decade of investigation into its utility within this disease model.[16] In this review, we will explore the use of OCT in ROP with regard to its clinical advantages, the insights it has delivered thus far, and the potential pathways to its adoption into the standard of care.
The Advantages of OCT in ROP Diagnosis
The current standard of care for in-person screening and monitoring of ROP remains bedside ophthalmoscopy in the neonatal intensive care unit (NICU), which traditionally involves topical anesthesia, pharmacologic pupillary dilation, insertion of an eyelid speculum and examination with the bright light of binocular indirect ophthalmoscopy aided by scleral depression.[17] By this means, ROP zone, stage and plus disease are subjectively assessed. An accepted alternative is the fundus photography-based telemedicine approach, which still requires the topical anesthesia, pupillary dilation, eyelid speculum and a contact based camera. These approaches are imperfect for two reasons: 1) the ROP examination is stressful, adding medical risks to fragile neonates, 2) both ophthalmoscopic examination and telemedicine interpretation are limited by subjectivity and inter-observer diagnostic variability. OCT-aided visualization has been shown to offer a viable alternative to overcome these challenges.
The ROP examination adds medical risks to fragile neonates
While necessary to identify potential signs of vision-threatening conditions at the bedside, ophthalmoscopic exams are physiologically stressful for neonates, producing changes in heart rate, and apnea during and after examination.[18, 19] Handheld WDFI devices used in bedside and telemedicine exams for ROP require similar preparation including bright light exposure which is physiologically stimulating.[20, 21] In addition to stress during the exam, cycloplegic medication for pharmacologic dilation may cause systemic complications like impaired gut motility, require time for dilation, and incur cost of additional medication.[22] Handheld retinal imaging devices capable of noncontact photography may obviate the need for topical anesthesia, but they have lower field of view.[23**, 24]
The use of OCT in monitoring of pediatric vitreoretinal disease has been historically challenging, particularly in awake NICU patients.[25] Recently, handheld spectral-domain OCT devices can provide high-resolution en face and cross-sectional retinal and optic nerve images in supine NICU infants that are inherently unable to position adequately into tabletop devices.[15*, 16, 26–30] The use of non-visible, infrared light in these devices eliminates the stress added by the bright, visible light of binocular indirect ophthalmoscopy. Further, some handheld devices have been designed to capture images without direct contact with the cornea or use of an eyelid speculum.[16, 23**] Using noncontact handheld OCT, Mangalesh et al. demonstrated lower behavioral and cardiovascular markers of stress during bedside ROP exam compared to binocular indirect ophthalmoscopy, despite longer examination time.[23**] Speed of acquisition is another challenge being addressed to optimize comfort in awake patients and quality in the setting of photographer hand tremor and subject motion during exams. Thus, the introduction of handheld OCT pediatric bedside exams has accelerated imaging speeds from tens of kilohertz (kH) to more than one megahertz (MHz), reducing examination time to less than that of conventional binocular indirect ophthalmoscopy, and even making OCT angiography (OCTA) possible.[31] Innovation in handheld OCT devices has introduced a modality with potential to reduce the duration and physiologic stress of conventional bedside exams. Looking forward, development of nonmydriatic image acquisition as an additional feature could eliminate the need for pharmacologic dilation and reduce physiologic risks of these topical medications.
ROP grading is subjective and prone to interobserver variability
The framework for current treatment guidelines in ROP is derived from the following classification schema provided by ICROP in 1984: the zone (location of border of non-vascularized retina in relation to pre-determined borders), stage (degree of vascular abnormality at the avascular border on a 0-5 scale), and plus disease (degree of arterial tortuosity and venous dilation in the posterior retina according to original standard photographs).[32, 33] In 2005, ICROP was updated with the introduction an intermediate classification of “pre-plus” disease, introducing the concept that plus disease manifests on a spectrum, as opposed to a binary (i.e. “plus” or “no plus”) distribution.[32] In its most recent update in 2021, the new ICROP formally acknowledged the manifestation of plus disease along a continuous spectrum.[7] While these increasingly nuanced recommendations for classification more accurately reflect the profile and natural history of disease, inter-observer diagnosis remains discordant. Clinicians have been shown to disagree on all ICROP classifications of zone, stage and plus with both binocular indirect ophthalmoscopy and widefield digital fundus imaging.[8, 9, 34–36] Additionally, clinicians disagree on the relative relevance of various clinical features to classification and prognosis.[37, 38] This observed disagreement undermines consistency in diagnostic standards and may lead to delayed or missed diagnosis of treatment-requiring disease.
OCT’s ability to capture ultra-widefield high resolution images of the retina in cross section could provide a solution to the subjectivity of clinical examinations in two ways: 1) quantification of established components of disease classification, and 2) identification of new biomarkers of disease not observable by ophthalmoscopy.
Quantification of novel components of disease classification
While plus disease has been the first disease trait to be formally recognized by ICROP to manifest along a continuous spectrum, we posit that all 3 components of the ICROP classification (zone, stage, and plus) reflect continuous findings that are grouped into ordinal categories based on the clinical examination. “Zone 1” ROP has a worse prognosis (higher risk of disease progression requiring treatment, and retinal detachment), and the ICROP recently recommended further subdividing zone II into posterior and anterior zone II for the same reason. However, clinician determination of zone is subjective and inconsistent, and fundamentally reflects a clinical variable (area of avascular retina) that is inherently continuous. Recently, a handheld ultra-widefield OCT device capable of real time, en face visualization of an up to 140° field of view was demonstrated to provide higher contrast of the vascular-avascular border than binocular indirect ophthalmoscopy or widefield digital fundus imaging, including visualization to the ora serrata without scleral depression (Figure 1).[14*, 15*, 39*] Longitudinal characterization of the vascular-avascular border with such a device could facilitate measurement of the area of vascularized retina, and permit objective assessment of this feature as a continuous variable.
Figure 1:
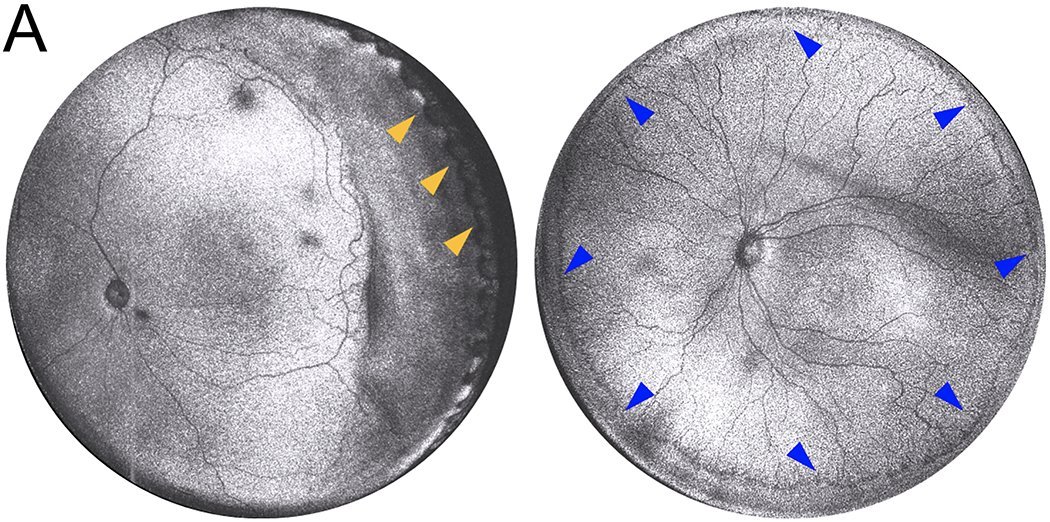
Representative ultra-widefield en face ROP fundus images obtained via prototype handheld OCT developed at Oregon Health & Science University [14, 65]. (A) Simultaneous visualization of the posterior pole and ora serrata is possible without scleral depression (yellow arrowheads). (B) Subtle features of stage at the vascular-avascular border are visible in 360 degrees.
In addition to en face assessment of zone, OCT B-scans permit the examiner to assess three-dimensional relationships to inform classification of stage.[40] The ability to reconstruct the topographic anatomy of the retina reveals fine details like popcorn neovascularization and the continuity of stage progression as extraretinal fibrovascular proliferation coalesces into characteristic stage 3 appearance.[15*] This modality not only allows examiners to monitor known features of staging with finer detail but could also reveal new biomarkers for diagnosis and progression. In a longitudinal study, Nguyen et al. found a strong association between OCT-measured retinal thickness measurements at the vascular-avascular junction and ordinal disease classification in patients with ROP, indicating that OCT-based quantification of peripheral stage in ROP may have future implications for disease classification and longitudinal monitoring as an objective and quantitative biomarker.[14*] With features enabling ultra-widefield visualization and quantification of three-dimensional changes, OCT could offer more precise and continuous monitoring of stage across the length of the vascular-avascular junction than “maximum stage”, as is the current convention. In advanced ROP, OCT has been used to differentiate retinal detachment from retinoschisis, which are often difficult to distinguish by indirect ophthalmoscopy alone. Chen et al. also found handheld OCT to be useful in determining Stages 4A from 4B, which each differ greatly in visual prognosis.[41]
Vascular changes in moderate to severe ROP present along the spectrum of plus disease, and are related to the zone, stage, and extent of disease.[7**, 40, 42] Ultra-widefield OCT would be capable of visualizing all of these relationships simultaneously, non-invasively, and in three dimensions. With this imaging modality, longitudinal observation and quantification of these metrics could be utilized through an infant’s NICU course, improving clinical care and offering researchers the novel opportunity of monitoring vascular growth patterns in vivo. En face imaging could also be utilized to generate AI-derived predictions of disease severity, as has been demonstrated previously with fundus photography.[42]
Prototype handheld OCTA devices have also been developed for pediatric use, recently described to be capable of high-speed, high-resolution, and ultra-widefield image capture.[26, 29, 31, 43] Ni et al. recently reported a prototype handheld OCTA device capable of visualization of peripheral retinal vasculature and neovascularization. This would conceivably provide greater resolution for measurement of area of vascularization and localizing neovascularization, as well as nonperfusion within the vascularized retina. Used in combination with peripheral OCT B-scans, these modalities would also enable more precise ROP staging by localizing neovascularization either confined beneath or broken through the ILM.[44] Conversely, observation of avascular regions may be informative in cases of spontaneous regression or longitudinal follow-up, and serve as a potential biomarker for risk of vision threatening events like retinal detachment in the future.[45]
Identification of novel biomarkers of disease
In addition to finer detail and quantification of known clinical findings that contribute to ordinal ICROP classifications, OCT may also reveal new biomarkers for prognosticating and monitoring disease. A myriad of potential markers have already been identified in the literature.[13] Vitreous opacities have been increasingly studied for their association with ROP and correlation with severity. Vitreous material shadowing was first described by Lee et al. using handheld OCT, and later described by Zepeda et al. as dense, band-like opacities either in contact with, or hovering parallel to the retinal surface.[46–48] Legocki et al. demonstrated that, when in contact with the retinal surface, these bands are associated with a diagnosis of plus disease.[47] Separately, Scoville et al. observed punctate hyperreflective vitreous opacities in newborns, noting them to appear with higher density in premature infants. Investigations by Legocki et al. and Scoville et al. have drawn associations between presence of these opacities and ROP severity according to zone, stage, plus disease, and Type I ROP.[47, 49]
Cystoid macular edema (CME) is a feature directly observable through OCT, and commonly seen in premature infants undergoing ROP screening.[50, 51] With some exceptions, many studies have shown prevalence of CME to increase with stage, and degree of CME to correlate with stage, presence of plus disease, response to laser treatment, and a host of systemic factors.[52–55] Other investigations have implicated CME in reduced visual acuity as early as 3 months of age.
Objectively measuring choroidal and retinal features in cross section has begun to elucidate these complex relationships between choroidal structure, prematurity, and ROP. Choroidal thinning has been reported in preterm infants and associated with higher ROP stage, plus disease, lower gestational age, and lower birth weight.[54, 56, 57] This may in part be explained by the association between choroidal and retinal development.[58] Indicators of retinal immaturity including shallow foveal pit, persistence of foveal inner retinal layers, attenuated ellipsoid zones, and retinal layer thinning have been extensively described in neonates.[51, 59–61] Additionally, vertical optic nerve cup diameter and vertical optic nerve diameter have been found to be larger, and may be associated with CNS pathology and future cognitive development in preterm infants.[62]
Alternative biomarkers for plus disease have additionally been explored using OCT. Maldonado et al. calculate a vascular abnormality (VASO) score through presence of one of the following OCT-derived features: retinal vessel elevation, scalloped retinal layers, hyporeflective vessels and retinal spaces.[63] The VASO score was found to be higher in patients with clinical plus disease and demonstrated the effects of vascular dilation and tortuosity on perivascular retinal tissue.
Pathway to adoption
Since its commercial introduction in 1996, OCT has become adopted as standard of care for diagnosis and monitoring of numerous disease models in ophthalmology, and an estimated 30 million ophthalmic OCT studies are captured annually worldwide.[64] However, the widespread acceptance of OCT as a diagnostic tool has been minimally adopted in neonatal contexts. The translation of OCT to clinical use in ROP would not only be beneficial, but also possible if three criteria are fulfilled: technological feasibility, clinical utility and industry support.
Technological Feasibility
Historically, the speed and positioning limitations of tabletop OCT and OCTA devices have prohibited widespread use in the NICU. However, innovations in handheld OCT devices have made bedside NICU exams ergonomically feasible and increasingly capable of capturing scans of awake neonates. The ideal combination of features for OCT-based ROP screening remains an open question. The development of non-contact probes may reduce infection risk, at the expense of lower field of view. Contact-probes may provide UWF visualization and may work without pupillary dilation. Alternatively, contact-based probes with either disposable or cleanable lenses may provide the best combination of features and risk.
Clinical Utility
A key driver in the clinical adoption of OCT was the simultaneous introduction of pharmacologic treatment for macular disease which OCT could directly monitor. Establishing a similar link between diagnostics and therapy in the context of ROP is necessary may facilitate more rapid clinical uptake of this technology. If the next generation of handheld OCT device(s) can more objectively monitor progression and regression of the stage of ROP with less stress, a compelling case could be made for replacing the ophthalmoscopic examination. Simultaneously, investigation of alternative, OCT-derived biomarkers of disease is ongoing. While it remains to be seen whether these novel biomarkers may supplant known markers of disease, OCT lends itself to deeper innovation for solving this question. Tools for quantification and AI-based image acquisition may soon be implemented into these devices to enable validation of objective biomarkers of disease, both conventional and novel.
Industry Support
While technological advances and published research draw an increasingly compelling argument for the implementation of OCT in clinical management of ROP, buy-in from device manufacturers is essential for widespread distribution of NICU-compatible devices. There is now a network of innovation in OCT with products including handheld, home-use, and intraoperative devices, possibly lowering the threshold for device manufacturers to adopt new iterations to their line. Currently, innovation in handheld devices for ROP examination has been reported by few groups with investigational models touting such features as ultra-widefield views and non-contact image capture.[26, 29, 65*] Once optimized for clinical use, these prototypes may be brought to market by means of either a new company formed by the developers, or through licensing to a larger OCT device manufacturer. Published research demonstrating the clinical indications and technical capabilities of such devices for ROP screening is key for manufacturers to consider adding them to their product line, especially if devices can be shown to be easier to use than widefield digital fundus imaging and without the need for pupillary dilation.
The growing demand for a product that can deliver more efficient and objective means of ROP screening is evident. Advancements in neonatal care and higher preterm infant survival rates have paradoxically elevated ROP burden, heralding what may be considered a modern ROP epidemic.[66, 67] In a recent study, Bhatnagar et al. estimated an 86% increased incidence in ROP between 2003 and 2019, disproportionately affecting newborns from lower-income households and of minority demographics.[68**] To meet this growing demand, pilot tele-screening initiatives such as the Stanford University Network for Diagnosis of Retinopathy of Prematurity (SUNDROP) program have been established to remotely diagnose treatment-requiring ROP from fundus photographs obtained by trained nurses and interpreted at a single location.[17, 69–71] Implementation of OCT as an alternative to color fundus photography in a growing landscape of telescreening programs could broaden its acceptance as a diagnostic tool not only for ROP, but also for other conditions. The ongoing Newborn Eye Screen Test Study and other reports of universal newborn eye screening protocols have explored the incidence of ocular pathology in otherwise healthy infants, reporting abnormal findings in up to 23% of cases.[72–77] As conditions in which early recognition and intervention can improve visual outcomes are identified, OCT may be increasingly utilized as both a screening and diagnostic tool.
Conclusion
OCT may offer a promising solution to the subjectivity and physiologic stress associated with current methods of ROP screening. Research with investigational OCT devices has validated both non-contact and Ultra-widefield approaches, demonstrating potential for feasible use in the NICU setting. Peripheral visibility, high resolution, and cross-sectional tissue analysis could objectively measure known pathologic features and identify novel biomarkers of disease. In time, OCT may become a preferred modality in the diagnosis and monitoring of ROP.
Key Points.
Retinopathy of prematurity is traditionally diagnosed and monitored by clinical examination, which has been shown to be highly subjective and prone to inter-observer variation.
Though widely adopted as a standard of care in adult retinal conditions, use of optical coherence tomography (OCT) in neonatal contexts has historically been limited due to technical limitations including positioning difficulties and speed of image capture.
UWF-OCT could be a promising alternative to bedside indirect ophthalmoscopy for its potential to reduce infant stress during examinations, reduce examination times and objectively quantify pathology.
Recent technical advancements including ultra-widefield visualization and non-contact, high-speed image acquisition in investigational OCT devices have enabled visualization of ROP stage and revealed potential novel biomarkers for disease activity and prognostication.
Continued refinement of technologic advancements, demonstration of clinical utility in published research and partnership with industry for manufacturing NICU-compatible OCT devices are key checkpoints in the translation of OCT to clinical management of ROP.
Acknowledgments
This work was supported by the following grants from the National Institutes of Health (Bethesda, MD): R01 EY019474, R01 EY031331, R21 EY031883, and P30 EY010572
2. Financial support and sponsorship:
This work was supported by grants R01 EY019474, R01 EY031331, R21 EY031883, and P30 EY010572, from the National Institutes of Health (Bethesda, MD), the Malcolm M. Marquis, MD Endowed Fund for Innovation, and by unrestricted departmental funding and a Career Development Award (JPC) from Research to Prevent Blindness (New York, NY). BKY is supported by a Knights Templar Eye Foundation Career Starter Grant. AMH is supported by funding from the Heed Ophthalmic Foundation. The funding organizations had no role in the design or conduct of this research.
3. Conflicts of interest:
JPC receives research support from Genentech (San Francisco, CA). The i-ROP DL system has been licensed to Siloam Vision by Oregon Health & Science University, Massachusetts General Hospital, Northeastern University and the University of Illinois Chicago, which may result in royalties to JPC in the future. JPC was a consultant to Boston AI Lab (Boston, MA). JPC is an equity owner of Siloam Vision.
References
- 1.Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol. 2003;121(12):1684–94. [DOI] [PubMed] [Google Scholar]
- 2.Multicenter trial of cryotherapy for retinopathy of prematurity. Preliminary results. Cryotherapy for Retinopathy of Prematurity Cooperative Group. Arch Ophthalmol. 1988;106(4):471–9. [DOI] [PubMed] [Google Scholar]
- 3.Fleck BW, Williams C, Juszczak E, et al. An international comparison of retinopathy of prematurity grading performance within the Benefits of Oxygen Saturation Targeting II trials. Eye (Lond). 2018;32(1):74–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gilbert C Retinopathy of prematurity: a global perspective of the epidemics, population of babies at risk and implications for control. Early Hum Dev. 2008;84(2):77–82. [DOI] [PubMed] [Google Scholar]
- 5.Mintz-Hittner HA, Kennedy KA, Chuang AZ. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Engl J Med. 2011;364(7):603–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Reynolds JD, Dobson V, Quinn GE, et al. Evidence-based screening criteria for retinopathy of prematurity: natural history data from the CRYO-ROP and LIGHT-ROP studies. Arch Ophthalmol. 2002;120(11):1470–6. [DOI] [PubMed] [Google Scholar]
- 7.Chiang MF, Quinn GE, Fielder AR, et al. International Classification of Retinopathy of Prematurity, Third Edition. Ophthalmology. 2021;128(10):e51–e68. [DOI] [PMC free article] [PubMed] [Google Scholar]; **The most recent ICROP update, in which the concern about subjectivity in current disease classification and manifestation of plus disease on a spectrum of vascular abnormality are addressed.
- 8.Campbell JP, Kalpathy-Cramer J, Erdogmus D, et al. Plus Disease in Retinopathy of Prematurity: A Continuous Spectrum of Vascular Abnormality as a Basis of Diagnostic Variability. Ophthalmology. 2016;123(11):2338–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Campbell JP, Ryan MC, Lore E, et al. Diagnostic Discrepancies in Retinopathy of Prematurity Classification. Ophthalmology. 2016;123(8):1795–801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cole ED, Park SH, Kim SJ, et al. Variability in Plus Disease Diagnosis using Single and Serial Images. Ophthalmol Retina. 2022;6(12):1122–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Moleta C, Campbell JP, Kalpathy-Cramer J, et al. Plus Disease in Retinopathy of Prematurity: Diagnostic Trends in 2016 Versus 2007. Am J Ophthalmol. 2017;176:70–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nguyen TP, Young BK, Coyner A, et al. Discrepancies in Diagnosis of Treatment-Requiring Retinopathy of Prematurity. Ophthalmol Retina. 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]; **This was a multicenter cohort study evaluating differences in diagnosis of treatment-requiring ROP between clinician examiners and masked graders. Most infants who received treatment in the cohort were treated despite not being diagnosed as having type 1 disease according to masked graders. Additionally, the study noted a tendency in treating physicians to diagnose disease as more severe, particularly with regard for classification of stage and plus.
- 13.Kubsad D, Ohan MA, Wu JG, Cabrera MT. Vitreoretinal biomarkers of retinopathy of prematurity using handheld optical coherence tomography: a review. Front Pediatr. 2023;11:1191174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nguyen TP, Ni S, Ostmo S, et al. Association of Optical Coherence Tomography-Measured Fibrovascular Ridge Thickness and Clinical Disease Stage in Retinopathy of Prematurity. JAMA Ophthalmol. 2022;140(11):1121–7. [DOI] [PMC free article] [PubMed] [Google Scholar]; *The authors use widefield OCT to measure and establish vascular-avascular junction thickness as a novel quantitative biomarker for peripheral stage in ROP, which may refine diagnosis and longitudinal monitoring in the future.
- 15.Scruggs BA, Ni S, Nguyen TP, et al. Peripheral OCT Assisted by Scleral Depression in Retinopathy of Prematurity. Ophthalmol Sci. 2022;2(1). [DOI] [PMC free article] [PubMed] [Google Scholar]; *The authors demonstrate the feasibility of visualizing the retinal periphery with widefield OCT aided by scleral depression for the detection and monitoring of ROP disease. Here, the clnical value of OCT for ROP screenig is shown as a means of detecting progressive stage, extent of disease and retinal detachment.
- 16.Maldonado RS, Izatt JA, Sarin N, et al. Optimizing hand-held spectral domain optical coherence tomography imaging for neonates, infants, and children. Invest Ophthalmol Vis Sci. 2010;51(5):2678–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fierson WM. Screening Examination of Premature Infants for Retinopathy of Prematurity. Pediatrics. 2018;142(6). [DOI] [PubMed] [Google Scholar]
- 18.Rush R, Rush S, Ighani F, et al. The effects of comfort care on the pain response in preterm infants undergoing screening for retinopathy of prematurity. Retina. 2005;25(1):59–62. [DOI] [PubMed] [Google Scholar]
- 19.Tan JBC, Dunbar J, Hopper A, et al. Differential effects of the retinopathy of prematurity exam on the physiology of premature infants. J Perinatol. 2019;39(5):708–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dhaliwal CA, Wright E, McIntosh N, et al. Pain in neonates during screening for retinopathy of prematurity using binocular indirect ophthalmoscopy and wide-field digital retinal imaging: a randomised comparison. Arch Dis Child Fetal Neonatal Ed. 2010;95(2):F146–8. [DOI] [PubMed] [Google Scholar]
- 21.Moral-Pumarega MT, Caserío-Carbonero S, De-La-Cruz-Bértolo J, et al. Pain and stress assessment after retinopathy of prematurity screening examination: indirect ophthalmoscopy versus digital retinal imaging. BMC Pediatr. 2012;12:132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Obata S, Imamura T, Kakinoki M, et al. Systemic adverse events after screening of retinopathy of prematurity with mydriatic. PLoS One. 2021;16(9):e0256878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mangalesh S, Sarin N, McGeehan B, et al. Preterm Infant Stress During Handheld Optical Coherence Tomography vs Binocular Indirect Ophthalmoscopy Examination for Retinopathy of Prematurity. JAMA Ophthalmol. 2021;139(5):567–74. [DOI] [PMC free article] [PubMed] [Google Scholar]; **Indicators of neonatal pain including cry score, facial expression heart rate and respiratory rate are utilized to compare preterm infant stress during bedside examination with OCT and bilateral indirect ophthalmoscopy. Their findings are suggestive of OCT as a less stressful means of examination.
- 24.Prakalapakorn SG, Stinnett SS, Freedman SF, et al. Non-contact retinal imaging compared to indirect ophthalmoscopy for retinopathy of prematurity screening: infant safety profile. J Perinatol. 2018;38(9):1266–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wakabayashi T, Patel SN, Campbell JP, et al. Advances in retinopathy of prematurity imaging. Saudi J Ophthalmol. 2022;36(3):243–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Campbell JP, Nudleman E, Yang J, et al. Handheld Optical Coherence Tomography Angiography and Ultra-Wide-Field Optical Coherence Tomography in Retinopathy of Prematurity. JAMA Ophthalmol. 2017;135(9):977–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Scott AW, Farsiu S, Enyedi LB, et al. Imaging the infant retina with a hand-held spectral-domain optical coherence tomography device. Am J Ophthalmol. 2009;147(2):364–73.e2. [DOI] [PubMed] [Google Scholar]
- 28.Legocki AT, Lee AY, Ding L, et al. Multivariate Models to Diagnose Early Referral-Warranted Retinopathy of Prematurity With Handheld Optical Coherence Tomography. Transl Vis Sci Technol. 2023;12(5):26. [DOI] [PMC free article] [PubMed] [Google Scholar]; *A generalized linear mixed model composed of previously described OCT biomarkers was found to be both highly sensitive and specific for early referral-warranted ROP. This work supports OCT as an alternative, better tolerated means of early ROP screening.
- 29.Song S, Zhou K, Xu JJ, et al. Development of a clinical prototype of a miniature hand-held optical coherence tomography probe for prematurity and pediatric ophthalmic imaging. Biomed Opt Express. 2019;10(5):2383–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yang J, Liu L, Campbell JP, et al. Handheld optical coherence tomography angiography. Biomed Opt Express. 2017;8(4):2287–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ni S, Wei X, Ng R, et al. High-speed and widefield handheld swept-source OCT angiography with a VCSEL light source. Biomed Opt Express. 2021;12(6):3553–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol. 2005;123(7):991–9. [DOI] [PubMed] [Google Scholar]
- 33.An international classification of retinopathy of prematurity. The Committee for the Classification of Retinopathy of Prematurity. Arch Ophthalmol. 1984;102(8):1130–4. [DOI] [PubMed] [Google Scholar]
- 34.Chiang MF, Jiang L, Gelman R, et al. Interexpert agreement of plus disease diagnosis in retinopathy of prematurity. Arch Ophthalmol. 2007;125(7):875–80. [DOI] [PubMed] [Google Scholar]
- 35.Chiang MF, Thyparampil PJ, Rabinowitz D. Interexpert agreement in the identification of macular location in infants at risk for retinopathy of prematurity. Archives of ophthalmology. 2010;128(9):1153–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gschließer A, Stifter E, Neumayer T, et al. Inter-expert and intra-expert agreement on the diagnosis and treatment of retinopathy of prematurity. Am J Ophthalmol. 2015;160(3):553–60.e3. [DOI] [PubMed] [Google Scholar]
- 37.Hewing NJ, Kaufman DR, Chan RV, Chiang MF. Plus disease in retinopathy of prematurity: qualitative analysis of diagnostic process by experts. JAMA Ophthalmol. 2013;131(8):1026–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kim SJ, Campbell JP, Kalpathy-Cramer J, et al. Accuracy and Reliability of Eye-Based vs Quadrant-Based Diagnosis of Plus Disease in Retinopathy of Prematurity. JAMA Ophthalmol. 2018;136(6):648–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nguyen TP, Ni S, Khan S, et al. Advantages of Widefield Optical Coherence Tomography in the Diagnosis of Retinopathy of Prematurity. Front Pediatr. 2021;9:797684. [DOI] [PMC free article] [PubMed] [Google Scholar]; *This observational study provides an overview of OCT as a superior tool for disease assessment tool in ROP due to its potential for objective disease categorization, minimally-invasive image acquisition, three dimensional visualization of the vitreoretinal interface and dye-free profiling of normal and pathologic vascular development.
- 40.Gupta K, Campbell JP, Taylor S, et al. A Quantitative Severity Scale for Retinopathy of Prematurity Using Deep Learning to Monitor Disease Regression After Treatment. JAMA Ophthalmol. 2019;137(9):1029–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen X, Prakalapakorn SG, Freedman SF, et al. Differentiating Retinal Detachment and Retinoschisis Using Handheld Optical Coherence Tomography in Stage 4 Retinopathy of Prematurity. JAMA Ophthalmol. 2020;138(1):81–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Campbell JP, Kim SJ, Brown JM, et al. Evaluation of a Deep Learning-Derived Quantitative Retinopathy of Prematurity Severity Scale. Ophthalmology. 2021;128(7):1070–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Viehland C, Chen X, Tran-Viet D, et al. Ergonomic handheld OCT angiography probe optimized for pediatric and supine imaging. Biomed Opt Express. 2019;10(5):2623–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chen X, Mangalesh S, Dandridge A, et al. Spectral-Domain OCT Findings of Retinal Vascular-Avascular Junction in Infants with Retinopathy of Prematurity. Ophthalmol Retina. 2018;2(9):963–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hanif AM, Gensure RH, Scruggs BA, et al. Prevalence of persistent avascular retina in untreated children with a history of retinopathy of prematurity screening. Journal of American Association for Pediatric Ophthalmology and Strabismus. 2022;26(1):29–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lee AC, Maldonado RS, Sarin N, et al. Macular features from spectral-domain optical coherence tomography as an adjunct to indirect ophthalmoscopy in retinopathy of prematurity. Retina. 2011;31(8):1470–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Legocki AT, Zepeda EM, Gillette TB, et al. Vitreous Findings by Handheld Spectral-Domain OCT Correlate with Retinopathy of Prematurity Severity. Ophthalmol Retina. 2020;4(10):1008–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zepeda EM, Shariff A, Gillette TB, et al. Vitreous Bands Identified by Handheld Spectral-Domain Optical Coherence Tomography Among Premature Infants. JAMA Ophthalmol. 2018;136(7):753–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Scoville NM, Legocki AT, Touch P, et al. Vitreous opacities in infants born full-term and preterm by handheld swept-source optical coherence tomography. J aapos. 2022;26(1):20.e1–.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Vinekar A, Avadhani K, Sivakumar M, et al. Understanding clinically undetected macular changes in early retinopathy of prematurity on spectral domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2011;52(8):5183–8. [DOI] [PubMed] [Google Scholar]
- 51.Dubis AM, Subramaniam CD, Godara P, et al. Subclinical macular findings in infants screened for retinopathy of prematurity with spectral-domain optical coherence tomography. Ophthalmology. 2013;120(8):1665–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Maldonado RS, O’Connell R, Ascher SB, et al. Spectral-domain optical coherence tomographic assessment of severity of cystoid macular edema in retinopathy of prematurity. Arch Ophthalmol. 2012;130(5):569–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Maldonado RS, Toth CA. Optical coherence tomography in retinopathy of prematurity: looking beyond the vessels. Clin Perinatol. 2013;40(2):271–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mangalesh S, McGeehan B, Tai V, et al. Macular OCT Characteristics at 36 Weeks’ Postmenstrual Age in Infants Examined for Retinopathy of Prematurity. Ophthalmol Retina. 2021;5(6):580–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Erol MK, Ozdemir O, Turgut Coban D, et al. Macular findings obtained by spectral domain optical coherence tomography in retinopathy of prematurity. J Ophthalmol. 2014;2014:468653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Erol MK, Coban DT, Ozdemir O, et al. CHOROIDAL THICKNESS IN INFANTS WITH RETINOPATHY OF PREMATURITY. Retina. 2016;36(6):1191–8. [DOI] [PubMed] [Google Scholar]
- 57.Michalak SM, Mangalesh S, Shen LL, et al. Systemic Factors Associated with a Thinner Choroid in Preterm Infants. Ophthalmol Sci. 2021;1(2):100032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Huang LC, Zhou H, Legocki AT, et al. Choroidal Thickness by Handheld Swept-Source Optical Coherence Tomography in Term Newborns. Transl Vis Sci Technol. 2021;10(2):27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Maldonado RS, O’Connell RV, Sarin N, et al. Dynamics of human foveal development after premature birth. Ophthalmology. 2011;118(12):2315–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lawson SE, Tam EK, Zheng Y, et al. Semi-Automated Analysis of Foveal Maturity in Premature and Full-Term Infants Using Handheld Swept-Source Optical Coherence Tomography. Transl Vis Sci Technol. 2023;12(3):5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.O’Sullivan ML, Ying GS, Mangalesh S, et al. Foveal Differentiation and Inner Retinal Displacement Are Arrested in Extremely Premature Infants. Invest Ophthalmol Vis Sci. 2021;62(2):25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Tong AY, El-Dairi M, Maldonado RS, et al. Evaluation of optic nerve development in preterm and term infants using handheld spectral-domain optical coherence tomography. Ophthalmology. 2014;121(9):1818–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Maldonado RS, Yuan E, Tran-Viet D, et al. Three-dimensional assessment of vascular and perivascular characteristics in subjects with retinopathy of prematurity. Ophthalmology. 2014;121(6):1289–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Swanson E, Huang D. Ophthalmic OCT reaches $1 billion per year. Retinal Physician. 2011;8(4):58–9. [Google Scholar]
- 65.Ni S, Nguyen TP, Ng R, et al. 105° field of view non-contact handheld swept-source optical coherence tomography. Opt Lett. 2021;46(23):5878–81. [DOI] [PMC free article] [PubMed] [Google Scholar]; *Report of a handheld, noncontact widefield OCT protytpe device achiving the widest field of view among all portable retinal OCT imaging research protoypes and commercial systems.
- 66.Ludwig CA, Chen TA, Hernandez-Boussard T, et al. The Epidemiology of Retinopathy of Prematurity in the United States. Ophthalmic Surg Lasers Imaging Retina. 2017;48(7):553–62. [DOI] [PubMed] [Google Scholar]
- 67.Tasman W Retinopathy of prematurity: do we still have a problem?: the Charles L. Schepens lecture. Arch Ophthalmol. 2011;129(8):1083–6. [DOI] [PubMed] [Google Scholar]
- 68.Bhatnagar A, Skrehot HC, Bhatt A, et al. Epidemiology of Retinopathy of Prematurity in the US From 2003 to 2019. JAMA Ophthalmology. 2023;141(5):479–85. [DOI] [PMC free article] [PubMed] [Google Scholar]; **Retrospective database cohort study demonstrating that incidence of ROP in the United States increased from 2003 to 2019. Incidence of ROP was found to be proportionally higher amongst infants of black and hispanic race, and of low-income background.
- 69.Murakami Y, Jain A, Silva RA, et al. Stanford University Network for Diagnosis of Retinopathy of Prematurity (SUNDROP): 12-month experience with telemedicine screening. British Journal of Ophthalmology. 2008;92(11):1456. [DOI] [PubMed] [Google Scholar]
- 70.Photographic Screening For Retinopathy Of Prematurity Cooperative G. THE PHOTOGRAPHIC SCREENING FOR RETINOPATHY OF PREMATURITY STUDY (PHOTO-ROP): Primary Outcomes. RETINA. 2008;28(3). [DOI] [PubMed] [Google Scholar]
- 71.Chiang MF, Melia M, Buffenn AN, et al. Detection of clinically significant retinopathy of prematurity using wide-angle digital retinal photography: a report by the American Academy of Ophthalmology. Ophthalmology. 2012;119(6):1272–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Fei P, Liu Z, He L, et al. Early detection of ocular abnormalities in a Chinese multicentre neonatal eye screening programme—1-year result. Acta Ophthalmologica. 2021;99(3):e415–e22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sitorus RS, Pambudy IM, Rohsiswatmo R, et al. Retinal abnormalities in universal eye screening of healthy, full-term newborn infants in Jakarta. The incidence and its risk factors: a pilot study. Int J Retina Vitreous. 2021;7(1):67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Li-Hong L Universal Eye Screening in Healthy Neonates Retina Today2013 [Available from: https://retinatoday.com/articles/2013-mar/universal-eye-screening-in-healthy-neonates.
- 75.Vinekar A, Govindaraj I, Jayadev C, et al. Universal ocular screening of 1021 term infants using wide-field digital imaging in a single public hospital in I ndia–a pilot study. Acta ophthalmologica. 2015;93(5):e372–e6. [DOI] [PubMed] [Google Scholar]
- 76.Li L-H, Li N, Zhao J-Y, et al. Findings of perinatal ocular examination performed on 3573, healthy full-term newborns. British Journal of Ophthalmology. 2013;97(5):588–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Chee RI, Chan RVP. Universal newborn eye screening: an effective strategy to improve ocular health? Eye. 2018;32(1):50–2. [DOI] [PMC free article] [PubMed] [Google Scholar]


