Abstract
Introduction
Tetrahydrocannabinol (THC) is often prescribed for ambulatory palliative patients to improve sleep quality and appetite and to reduce anxiety, stress, and pain. However, it is not known if THC has also an effect on the mortality of these patients.
Method
The objective was the impact of THC on mortality of ambulatory palliative patients. For this purpose, data from the palliative treatment documentation from 5 ambulatory palliative care teams in Brandenburg, Germany were used for this analysis. Survival time was calculated for 3 groups of patients: (1) without THC; (2) with THC in a low dosage (≤4.7 mg per day); and (3) THC in higher doses (≥4.7 mg per day). The analysis was done for 2 cohorts of patients. Cohort 1: all patients with a survival time of at least 7 days after inclusion in specialized ambulatory palliative care (SAPC) and cohort 2: a subgroup of patients with a survival time between 7 and 100 days. Kaplan-Meier curves were created, and multivariate analysis was done to investigate the impact of THC on mortality.
Results
A total of 9,419 patients with a survival time of at least 7 days after inclusion in SAPC were included in the analysis (cohort 1). 7,085 among them had a survival time between 7 and 100 days (cohort 2). In both cohorts, survival time was significantly prolonged by THC, but only when the daily THC dose was above the median of 4.7 mg. Survival time was 15 days longer in cohort 2 (40 vs. 25 days), when more than 4.7 mg THC were prescribed per day.
Conclusion
Use of THC is associated with a significant increase in survival time in ambulatory palliative patients which survive longer than 7 days the initiation of THC prescription and which use of THC >4.7 mg/day.
Keywords: Medical cannabis, Palliative care, Survival, Tetrahydrocannabinol
Introduction
The use of cannabis-based medicine (CBM) by trained palliative care teams (PCTs) is still very limited in Germany although the German Act Amending Narcotic Drugs Provisions and Other Related Provisions of 2017 was explicitly targeted at improving care to palliative patients [1]. However, many physicians and nurses working in palliative care observe the positive effects of tetrahydrocannabinol (THC) in their daily practice, including the reduction of morbidity and drug adverse events, as well as an improvement in the quality of life. Schleider et al. [2] performed an analysis of a group of cancer patients in Israel and reported that CBM therapy was well-tolerated and was an effective and safe option to help patients cope with malignancy-related symptoms. Macari et al. [3] carried out a survey of a community hematology-oncology population in Michigan and reported a reduction in pain in 81% of patients, an increase in appetite in 77% and a reduction of anxiety in 73% of patients. Wendelmuth et al. [4] reported substantial therapeutic benefits of CBM for the treatment of geriatric pain and for palliative patients with tolerable adverse events in outpatient care in Germany.
The improvement of the symptoms mentioned above led to improved quality of life in that phase of the disease and might also prolong survival time in the palliative care period. However, to our knowledge, there are no data on the impact of CBM on mortality in ambulatory palliative patients. Thus, this study was undertaken to investigate the impact of THC as one of the most relevant CBM substances on survival time in a group of ambulatory palliative patients in Brandenburg.
Materials and Methods
Setting and Data Collection
In Germany, there is a special system for organizing ambulatory palliative care. In the first step, it is general practitioners who usually care for palliative patients. As the disease progresses along with severe symptoms, patients can be included in so-called specialized ambulatory palliative care (SAPC). In SAPC, these patients are cared for by dedicated teams of physicians and nurses with additional qualifications in palliative medicine. There are two requirements that it be administered by the treating physician and that the costs of the care first be approved by the patient’s health insurance provider. Once the provider has approved the treatment, SAPC can begin. The day of the first visit by a physician of a PCT is the starting point for calculating survival time for all palliative patients included in the study.
In addition, a companion survey, required by law, must be carried out before CBM can be used in palliative care. PalliDOC® is one of the documentation systems that fulfill this legal requirement. It includes patients’ demographic data, the start and end of SAPC, diagnoses, symptoms, and the dosage of CBM, e.g., THC. In Brandenburg, many PCTs use PalliDOC®. PCTs in Brandenburg that use PalliDOC® were invited to participate in this retrospective analysis. Five teams agreed and provided data retrospectively for SAPC patients who died between April 1, 2017, and September 30, 2021.
Statistical Analysis
Patient data from the five centers were combined and patients were stratified in three groups according to survival time (length of therapy from start in SAPC to death):
Survival time <7 days
Survival time >7 (cohort 1)
Survival time >7 days but not >100 days (cohort 2).
THC therapy is not indicated for some patients because of the severity of their illness. It usually takes about 1 week before THC treatment can be started and see the first result of therapy as a result of German regulations concerning the prescription of CBM to SAPC patients. Hence, the first group was excluded from further analysis.
Other patients are in SAPC longer than expected. They are, therefore, not true palliative patients from the viewpoint of SAPC regulations. Thus, the two cohorts were analyzed separately: first, the group of all patients with a survival time of 7 days or more (cohort 1) and second, the subgroup of palliative patients whose survival time was between 7 and 100 days (cohort 2), which perhaps best characterizes palliative care status.
In addition to the simple question of whether a THC dose was administered (yes/no), we calculated categories of THC treatment depending on amount of a patient’s daily dose in cohort 2: (1) no THC administered, (2) <median THC dose per day, (3) ≥ median THC dose per day. In our cohort 1, the medium daily THC dose was 4.7 mg. In order to create two groups with an equal size for statistical analysis, this dosage number was used as cut-off parameter.
The THC dosage usually started at a very low dose. After a short observation period, a decision was made whether to increase the dosage. This procedure was repeated until sufficient therapeutic effects (e.g., less anxiety) and/or tolerability was achieved. Only the last registered dose value in the documentation system was taken into account for this stratification.
In the descriptive analysis, we calculated either number and percent, or median and interquartile range, depending on the distribution of the parameters. Differences were tested using χ2 test or Wilcoxon rank-sum test. We calculated Kaplan-Meier survival curves and tested the differences with the log rank (Mantel-Cox) test in order to visualize the survival times of the THC treatment groups.
In the multivariable analysis, Cox-proportional hazard models were calculated to analyze the independent effect of a THC dose on survival time. Our models were extended to models that incorporate cluster-specific random effects to account for outcomes within-cluster homogeneity in outcomes. These random effects denote increased or decreased hazard for distinct classes (e.g., clusters such as PCTs) [5].
We calculated a crude model, with the CBM treatment parameter only, a model adjusted by age and gender, and finally a model that included all significant parameters where symptom parameters of patients were also considered. p < 0.05 was considered for significance. All analyses were exploratory in nature and were performed on SPSS version 29 (IBM SPSS statistics, Somer, NY, USA) and SAS 9.4 (SAS Institute, Cary, NC, USA).
Medical doctors in Germany are required by the German “Act Amending Narcotic Drugs Provisions and other related Provisions” of 2017 to document treatment in a companion survey. Since all data used for this publication were collected in accordance with this act in the legally required companion survey, ethical approval or informed consent were not required. Only anonymized data were used for this analysis.
Results
A total of 11,883 patients were documented by the five PCTs. After the exclusion of patients with a survival time of fewer than 7 days in SAPC, 9,419 patients remained (cohort 1). Cohort 2 comprised 7,085 patients with a survival time between 7 and 100 days after inclusion in SAPC. Table 1 shows the patient characteristics of the 3 patient groups based on survival time after inclusion in SAPC. The median survival time in cohort 2 was 25 days.
Table 1.
Overall patient (N = 11,883) characteristics stratified according to survival time (time of SAPC therapy from start to death)
| Parameter | Category | Survival time (from SAPC start to death) | p value | ||
|---|---|---|---|---|---|
| <7 days, N (%)/Med (IQR) | 7–100 days, N (%)/Med (IQR) | >100 days, N (%)/Med (IQR) | |||
| Patients | 2,464 (100) | 7,085 (100) | 2,334 (100) | ||
| Center | A | 466 (18.9) | 1,265 (17.9) | 491 (21) | <0.001 |
| B | 390 (15.8) | 914 (12.9) | 308 (13.2) | ||
| C | 983 (39.9) | 3,263 (46.1) | 1,087 (46.6) | ||
| D | 598 (24.3) | 1,548 (21.8) | 425 (18.2) | ||
| E | 27 (1.1) | 95 (1.3) | 23 (1) | ||
| Age, years | Med (IQR) | 80 (72–87) | 77 (66–83) | 74 (63–81) | <0.001 |
| Age group | <56 years | 129 (5.2) | 668 (9.4) | 287 (12.3) | <0.001 |
| >75 years | 1,629 (66.1) | 3,842 (54.2) | 1,073 (46) | ||
| 56–65 years | 254 (10.3) | 989 (14) | 438 (18.8) | ||
| 66–75 years | 452 (18.3) | 1,586 (22.4) | 536 (23) | ||
| Gender | Female | 1,199 (48.7) | 3,332 (47) | 1,133 (48.5) | 0.240 |
| Male | 1,265 (51.3) | 3,753 (53) | 1,201 (51.5) | ||
| Leading symptoms | Pain | 1,487 (60.3) | 4,518 (63.8) | 1,422 (60.9) | 0.002 |
| Urology | 214 (8.7) | 714 (10.1) | 293 (12.6) | <0.001 | |
| Cardiorespiratory | 1,017 (41.3) | 2,439 (34.4) | 769 (32.9) | <0.001 | |
| Gastrointestinal | 630 (25.6) | 2,576 (36.4) | 825 (35.3) | <0.001 | |
| Wound | 169 (6.9) | 525 (7.4) | 195 (8.4) | 0.135 | |
| Neuropsychiatric | 1,289 (52.3) | 3,463 (48.9) | 1,104 (47.3) | 0.001 | |
| Other severe symptoms | 1,793 (72.8) | 4,817 (68) | 1,565 (67.1) | <0.001 | |
| Survival time, days | Med (IQR) | 3 (2–5) | 25 (13–49) | 209 (139–369) | <0.001 |
| THC | No | 2,446 (99.3) | 6,875 (97) | 2,234 (95.7) | <0.001 |
| Yes | 18 (0.7) | 210 (3) | 100 (4.3) | ||
| THC dose, mg/day | Med (IQR) | 2.4 (2.4–4.8) | 4.7 (2.4–7.2) | 6.8 (3.2–9.6) | <0.001 |
| THC dose | ≤4.7 mg/day | 13 (72.2) | 105 (50) | 32 (32) | <0.001 |
| >4.7 mg/day | 5 (27.8) | 105 (50) | 68 (68) | ||
Med, median; IQR, interquartile range; N, number; THC, tetrahydrocannabinol.
310 patients (3.3%) in cohort 1 received THC with a dosage >6.8 mg/day – median THC. In cohort 2,210 patients (3.0%) received a median daily THC dose of 4.7 mg; hence, we stratified the data according to a daily dose of less than or equal to 4.7 mg and more than 4.7 mg. Tables 2 and 3 show the patient characteristics of cohort 1 and 2 based on whether they received THC therapy (yes/no) and the dosage (less than or equal to/more than 4.7 mg/day).
Table 2.
Patients (N = 9,419) with survival time (time of SAPC therapy from start to death) of at least 7 days stratified according to daily treatment with tetrahydrocannabinol (THC) (Cohort 1)
| Parameter | Category | THC | p value* | THC dose | p value** | ||
|---|---|---|---|---|---|---|---|
| no, N (%)/Med (IQR) | yes, N (%)/Med (IQR) | ≤4.7 mg/day, N (%)/Med (IQR) | >4.7 mg/day, N (%)/Med (IQR) | ||||
| Patients | 9,109 (100) | 310 (100) | 137 (100) | 173 (100) | |||
| Center | A | 1,731 (19) | 25 (8.1) | <0.001 | 7 (5.1) | 18 (10.4) | <0.001 |
| B | 1,030 (11.3) | 192 (61.9) | 94 (68.6) | 98 (56.6) | |||
| C | 4,334 (47.6) | 16 (5.2) | 5 (3.6) | 11 (6.4) | |||
| D | 1,911 (21%) | 62 (20) | 25 (18.2) | 37 (21.4) | |||
| E | 103 (1.1) | 15 (4.8) | 6 (4.4) | 9 (5.2) | |||
| Age, years | Med (IQR) | 76 (66–83) | 71 (62–81) | <0.001 | 74 (64–81) | 71 (61–81) | <0.001 |
| Age group | <56 years | 912 (10) | 43 (13.9) | <0.001 | 15 (10.9) | 28 (16.2) | <0.001 |
| >75 years | 4,787 (52.6) | 128 (41.3) | 64 (46.7) | 64 (37) | |||
| 56–65 years | 1,357 (14.9) | 70 (22.6) | 27 (19.7) | 43 (24.9) | |||
| 66–75 years | 2,053 (22.5) | 69 (22.3) | 31 (22.6) | 38 (22) | |||
| Gender | Female | 4,327 (47.5) | 138 (44.5) | 0.300 | 68 (49.6) | 70 (40.5) | 0.161 |
| Male | 4,782 (52.5) | 172 (55.5) | 69 (50.4) | 103 (59.5) | |||
| Leading symptoms | Pain | 5,781 (63.5) | 159 (51.3) | <0.001 | 71 (51.8) | 88 (50.9) | <0.001 |
| Urology | 988 (10.8) | 19 (6.1) | 0.008 | 10 (7.3) | 9 (5.2) | 0.025 | |
| Cardiorespiratory | 3,132 (34.4) | 76 (24.5) | <0.001 | 32 (23.4) | 44 (25.4) | <0.001 | |
| Gastrointestinal | 3,301 (36.2) | 100 (32.3) | 0.151 | 40 (29.2) | 60 (34.7) | 0.217 | |
| Wound | 713 (7.8) | 7 (2.3) | <0.001 | 1 (0.7) | 6 (3.5) | <0.001 | |
| Neuropsychiatric | 4,462 (49) | 105 (33.9) | <0.001 | 55 (40.1) | 50 (28.9) | <0.001 | |
| Other severe symptoms | 6,180 (67.8) | 202 (65.2) | 0.320 | 96 (70.1) | 106 (61.3) | 0.157 | |
| THC dose, mg/day (cont.) | Med (IQR) | 4.8 (2.4–8) | 2.4 (2.4–3.2) | 7.5 (4.8–12) | <0.001 | ||
| Survival time, days (cont.) | Med (IQR) | 38 (16–98) | 55 (25–132) | <0.001 | 35 (16–93) | 74 (33–176) | <0.001 |
| Survival time (cat.) | 7–100 days | 6,875 (75.5) | 210 (67.7) | 0.002 | 105 (76.6) | 105 (60.7) | <0.001 |
| >100 days | 2,234 (24.5) | 100 (32.3) | 0.002 | 32 (23.4) | 68 (39.3) | ||
Med, median; IQR, interquartile range; N, number; THC, tetrahydrocannabinol.
*p value compared THC yes with no.
**Compared no THC with THC dose ≤4.7 and >4.7 group.
Table 3.
Patients (N = 7,085) with survival time from 7 to 100 days stratified according to daily treatment with tetrahydrocannabinol (THC) (cohort 2)
| Parameter | Category | THC | p value* | THC dose | p value** | ||
|---|---|---|---|---|---|---|---|
| no, N (%)/Med (IQR) | yes, N (%)/Med (IQR) | ≤4.7 mg/day, N (%)/Med (IQR) | >4.7 mg/day, N (%)/Med (IQR) | ||||
| Patients | 6,875 (100) | 210 (100) | 105 (100) | 105 (100) | |||
| Center | A | 1,252 (18.2) | 13 (6.2) | <0.001 | 2 (1.9) | 11 (10.5) | <0.001 |
| B | 782 (11.4) | 132 (62.9) | 79 (75.2) | 53 (50.5) | |||
| C | 3,252 (47.3) | 11 (5.2) | 3 (2.9) | 8 (7.6) | |||
| D | 1,505 (21.9) | 43 (20.5) | 17 (16.2) | 26 (24.8) | |||
| E | 84 (1.2) | 11 (5.2) | 4 (3.8) | 7 (6.7) | |||
| Age, years | Med (IQR) | 77 (66–83) | 72 (62–81) | 0.002 | 76 (64–81) | 71 (62–81) | 0.006 |
| Age group | <56 years | 643 (9.4) | 25 (11.9) | 0.007 | 11 (10.5) | 14 (13.3) | 0.029 |
| 56–65 years | 945 (13.7) | 44 (21) | 53 (50.5) | 43 (41) | |||
| 66–75 years | 1,541 (22.4) | 45 (21.4) | 20 (19) | 24 (22.9) | |||
| >75 years | 3,746 (54.5) | 96 (45.7) | 21 (20) | 24 (22.9) | |||
| Gender | Female | 3,238 (47.1) | 94 (44.8) | 0.504 | 51 (48.6) | 43 (41) | 0.434 |
| Male | 3,637 (52.9) | 116 (55.2) | 54 (51.4) | 62 (59) | |||
| Leading symptom | Pain | 4,414 (64.2) | 104 (49.5) | <0.001 | 55 (52.4) | 49 (46.7) | <0.001 |
| Urology | 701 (10.2) | 13 (6.2) | 0.057 | 8 (7.6) | 5 (4.8) | 0.130 | |
| Cardiorespiratory | 2,391 (34.8) | 48 (22.9) | <0.001 | 20 (19) | 28 (26.7) | <0.001 | |
| Gastrointestinal | 2,506 (36.5) | 70 (33.3) | 0.355 | 29 (27.6) | 41 (39) | 0.148 | |
| Wound | 519 (7.5) | 6 (2.9) | 0.011 | 0 (0) | 6 (5.7) | 0.011 | |
| Neuropsychiatric | 3,390 (49.3) | 73 (34.8) | <0.001 | 44 (41.9) | 29 (27.6) | <0.001 | |
| Other severe symptoms | 4,680 (68.1) | 137 (65.2) | 0.386 | 75 (71.4) | 62 (59) | 0.108 | |
| THC dose, mg/day | Med (IQR) | 4.7 (2.4–7.2) | <0.001 | 2.4 (2.4–3.2) | 7.2 (4.8–12) | <0.001 | |
| Survival time,a days | Med (IQR) | 25 (13–49) | 33 (18–56) | <0.001 | 25 (14–47) | 40 (25–66) | <0.001 |
Med, median; IQR, interquartile range; N, number; THC, tetrahydrocannabinol.
aSurvival time, time of therapy from start to death.
*p value compared THC yes with no.
**Compared no THC with THC dose ≤4.7 and >4.7 group.
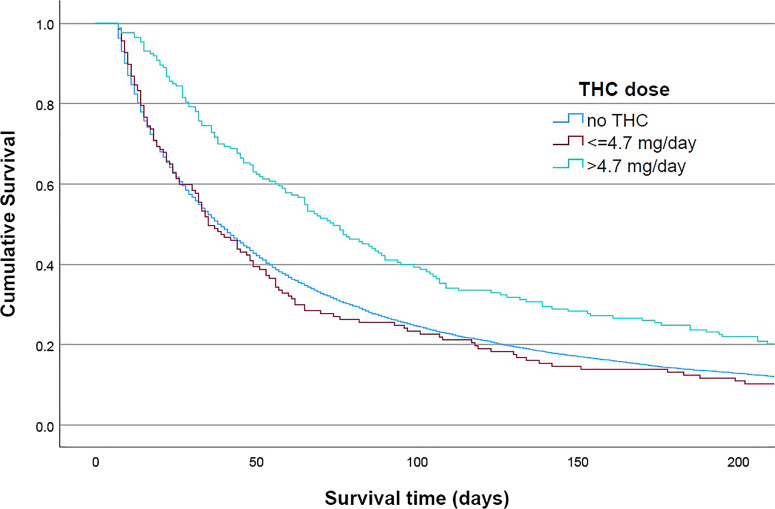
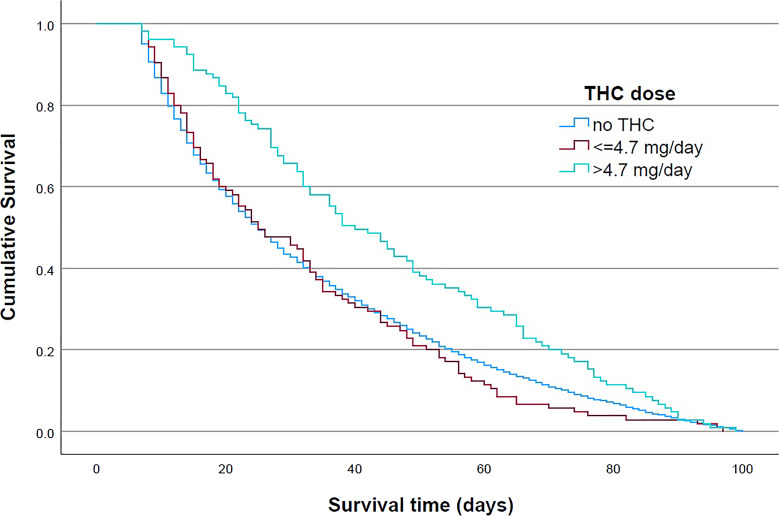
Irrespective of the THC dosage, the therapy with CBM was associated with prolonged median survival after initiation of SAPC treatment-from 38 to 55 days in cohort 1 and from 25 to 33 days in cohort 2. When a higher THC dose was given, one of more than 4.7 mg, the median survival time increased to 74 days in cohort 1 and 40 days in cohort 2. The prolongation of survival time in cohort 2 compared to patients who were not treated with THC was 15 days. Figures 1 and 2 show the results of Kaplan-Meier statistics for cohorts 1 and 2, respectively. THC had an impact on mortality in both groups, but only when a dose higher than 4.7 mg per day was administered.
Fig. 1.
Kaplan-Meier curve by tetrahydrocannabinol (THC) dose for patients with survival time ≥7 days. Patients with survival time ≥7 days: log rank (Mantel-Cox) p < 0.001, test of equality of survival distributions for the different levels of THC doses: no THC/≤4.7 mg per day/>4.7 mg per day.
Fig. 2.
Kaplan-Meier curve by daily tetrahydrocannabinol (THC) dose for patients with survival time 7–100 days. Patients with survival time 7–100 days: log rank (Mantel-Cox) p < 0.001, test of equality of survival distributions for the different levels of THC doses: no THC/≤4.7 mg per day/>4.7 mg per day.
The results of the multivariable analysis for both cohorts can be seen in Table 4. Regarding cohort 1, this analysis confirmed the significant effect of a higher dose of THC on survival time as opposed to no THC with a hazard ratio of 0.69 (95 CI: 0.59–0.81) in the adjusted model, whereas a THC dose of less than 4.7 mg/day did not show any effect.
Table 4.
Results of multivariable analysis with Cox-proportional hazard regression (both cohorts)
| Parameter | Category | Hazard ratio | 95% CI hazard ratio | p value |
|---|---|---|---|---|
| Cohort 1: patients survival time ≥7 days | ||||
| Crude model | ||||
| THC dose | ≤4.7 mg/day versus no THC | 1.06 | 0.9–1.26 | 0.486 |
| >4.7 mg/day versus no THC | 0.67 | 0.58–0.79 | <0.0001 | |
| Adjusted model | ||||
| THC dose | ≤4.7 mg/day versus no THC | 1.08 | 0.91–1.28 | 0.372 |
| >4.7 mg/day versus no THC | 0.69 | 0.59–0.81 | <0.0001 | |
| Age | per year | 1.01 | 1.01–1.01 | <0.0001 |
| Gender | female versus male | 0.95 | 0.91–0.99 | 0.011 |
| Model with significant parameters only | ||||
| THC dose | ≤4.7 mg/day versus no THC | 1.08 | 0.91–1.28 | 0.395 |
| >4.7 mg/day versus no THC | 0.68 | 0.59–0.8 | <0.0001 | |
| Age | per year | 1.01 | 1.01–1.01 | <0.0001 |
| Gender | female versus male | 0.94 | 0.9–0.98 | 0.003 |
| Symptom | pain | 1.11 | 1.06–1.16 | <0.0001 |
| urology | 0.87 | 0.81–0.93 | <0.0001 | |
| wound | 0.93 | 0.86–1 | 0.047 | |
| Cohort 2: Patients survival time 7–100 days | ||||
| Crude model | ||||
| THC dose | ≤4.7 mg/day versus no THC | 1.02 | 0.84–1.24 | 0.863 |
| >4.7 mg/day versus no THC | 0.68 | 0.56–0.82 | <0.0001 | |
| Adjusted model | ||||
| THC dose | ≤4.7 mg/day versus no THC | 1.05 | 0.86–1.27 | 0.644 |
| >4.7 mg/day versus no THC | 0.70 | 0.57–0.85 | <0.001 | |
| Age | per year | 1.01 | 1.01–1.01 | <0.0001 |
| Gender | female versus male | 0.99 | 0.94–1.04 | 0.622 |
| Model with significant parameters only | ||||
| THC dose | ≤4.7 mg/day versus no THC | 1.04 | 0.85–1.26 | 0.707 |
| >4.7 mg/day versus no THC | 0.70 | 0.58–0.85 | <0.001 | |
| Age | per year | 1.01 | 1.01–1.01 | <0.0001 |
| Symptom | other severe symptoms | 1.09 | 1.04–1.15 | 0.001 |
CI, confidence interval; THC, tetrahydrocannabinol.
*Complex symptom.
In addition, female patients had a longer survival time than male patients (hazard ratio 0.05, 95% CI: 0.91–0.99), but this effect was only significant in cohort 1. Survival time decreased with increasing age. When leading symptoms were included in the model, THC use was irrelevant to the abovementioned results.
Patients with urological and wound symptoms had significantly longer survival times. Patients whose primary symptom was pain had shorter survival time. The findings of the adjusted model for cohort 2 are very similar to cohort 1. But in cohort 2, gender was not relevant in terms of endpoint survival time and patients with other severe symptoms had a higher hazard ratio.
Discussion and Conclusion
The data of the study show an significant impact of THC on survival in ambulatory palliative care patients which survive the prescription by at least 7 days and use more than 4.7 mg/day. Median survival time was prolonged by 15 days in what is probably the most suitable patient group (cohort 2) – from survival time of 25 days without THC therapy to 40 days with a daily THC dose higher than 4.7 mg per day. This prolongation by more than 2 weeks can be considered substantial. In addition to mere survival, patients with THC become more mentally and physically active, which is also in accordance with the pharmacological effects of THC and other cannabinoids [6–9]. The increased activity and improved quality of life might enable the patients to renew social contact with relatives and friends and to settle essential affairs before dying.
This impact on survival could not be found in the group of patients with a THC dose less than 4.7 mg per day, and this demonstrates a dose-response effect of THC on survival. This low dose corresponds to 6 drops of 0.8 mg each of THC, or less than one tablet with 5 mg per day. The median dose in the group with a higher dose was 7.2 mg/day, but this is still substantially lower than 15 mg/day, which is the average dosage for non-palliative patients with chronic pain or other indications [10–12].
Moreover, the German Society for Palliative Medicine recommends between 5 and 160 mg THC per day to increase appetite [13]. For pain therapy, a daily dosage of up to 30 mg THC is recommended [14].
Low-dose THC therapy is relevant in the context of multiple comorbidities and polymedication as in palliative care and it is, therefore, important to realize that low-dose THC improves the syndrome of psycho-vegetative stabilization or quality of life [4]. A possible explanation for the lack of an effect on the group of patients with less than 4.7 mg per day may rest on our observation that even patients with very low doses of THC become mentally clearer (e.g., because of saving opioids) [4] and develop the ability to let go and die.
Last but not least, we explicitly do not want to discuss any anti-cancer effects of THC and CBM, respectively. THC was used in the terminal phase with short survival times in which anti-cancer effects – if at all – are most unlikely to be effective. The issue of anti-cancer effects is extensively discussed elsewhere [15].
This study has several limitations:
The study had a retrospective design and used real world data from the legally required companion survey. However, using the date of the first visit by the SAPC physician after inclusion of the patient in SAPC as is the starting point for the calculation of survival time makes it possible to compare patients with and without THC.
Second, this study included a large number of palliative patients, but not all of them were considered for analysis. The exclusion of patients with less than 7 days in SAPC from cohorts 1 and 2 seems justifiable because of the time necessary to provide the patient with THC and to establish a possible therapeutical effect. By excluding patients with more than 100 days in SAPC in cohort 2, patients without a severe palliative status were excluded from the study. Thus, cohort 2 better reflects the real world of ambulatory palliative patients.
Third, this analysis based on the highest documented dose of THC given to each patient. We cannot exclude the possibility that between inclusion in SAPC and the last record made even higher doses were given. In addition, we do not document any interruptions in THC administration. Finally, we cannot exclude the possibility that patients received further CBM at some later point. Such treatment is, however, very rare in Brandenburg.
Fourth, there might be differences in how THC was used in the five centers. This point was addressed by taking into consideration a cluster effect in the statistical analysis. Fifth, this study was not designed to consider the effect of THC on other endpoints such as pain, appetite, anxiety, sleeping quality, and did not record side effects in the three groups.
We cannot exclude that patients with an overall better condition were more likely to receive THC and we were unable to consider this point in our analysis. This is the reason to perform further more rigorous studies investigating this subject. Prospective studies that consider other endpoints, including mortality and side effects, should be carried out in the future. From the available data, we can conclude that current prescription practices deprive patients of days of life. Thus, in view of its significant prolongation of patient survival time, THC therapy should be included as part of the first-line therapy for ambulatory palliative patients.
Acknowledgments
We thank all members of the participating SAPC teams for providing their data for analysis:
Dr. Hartmut Parthe, SAPV – Team Priegnitz.
Dr. Michael Schmidt, PCT-Oderfluß/Frankfurth/Oder.
Dipl.-Med. Astrid Tributh, PCT Potsdam und Umland GbR.
Dr. Steffen Wolf, PCT Spreewald, Cottbus.
In addition, we thank Ralf Bimberg from StatConsult IT-Service GmbH who provided the software for analyzing data from “PalliDOC®” and Ruwen Böhm who did a first preliminary data analysis.
Statement of Ethics
Written informed consent from participants was not required in accordance with national guidelines. This retrospective review of patient data did not require ethical approval in accordance with local/national guidelines.
Conflict of Interest Statement
T.H.: fees for lectures and travel expenses from cannabis distributing and/or producing companies such as Avextra, Demecan, Spectrum, Sanity-Group and Stada; fees for lectures from the Deutschen Gesellschaft für Schmerzmedizin and the Bundesverband der pharmazeutischen Cannabinoidunternehmen (BPC); T.H. is member of Advisory Boards of Avextra and Sanity-Group. K.G.: fees for lectures and travel expenses from cannabis distributing and/or producing companies such as Demecan, Sanity-Group and Stada; K.G. is member of Advisory Boards of Sanity-Group. A.G.: fees for lectures from cannabis distributing and/or producing company Stada. F.S.: no conflict of interest.
Funding Sources
This study had no external funding.
Author Contributions
K.G. and A.G. developed the initial idea of the study and wrote the draft manuscript. F.S. analyzed the data from PalliDOC® and prepared the figures. T.H. evaluated and discussed the results. All authors read and approved the final manuscript.
Funding Statement
This study had no external funding.
Data Availability Statement
The research data associated with this paper are blinded patient data. These data are not available due to ethical reasons. Further inquiries can be directed to the corresponding author.
References
- 1. Van Mechelen W, Aertgeerts B, De Ceulaer K, Thoonsen B, Vermandere M, Warmenhoven F, et al. Defining the palliative care patient: a systematic review. Palliat Med. 2013;27(3):197–208. [DOI] [PubMed] [Google Scholar]
- 2. Bar-Lev Schleider L, Mechoulam R, Lederman V, Hilou M, Lencovsky O, Betzalel O, et al. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med. 2018;49:37–43. [DOI] [PubMed] [Google Scholar]
- 3. Macari DM, Gbadamosi B, Jaiyesimi I, Gaikazian S. Medical cannabis in cancer patients: a survey of a community hematology oncology population. Am J Clin Oncol. 2020;43(9):636–9. [DOI] [PubMed] [Google Scholar]
- 4. Wendelmuth C, Wirz S, Torontali M, Gastmeier A, Gastmeier K. Dronabinol bei geriatrischen Schmerz- und Palliativpatienten: Eine retrospektive Auswertung der ambulanten kassenärztlichen Therapie. 2019. Schmerz [Internet]. 31 [zitiert 17. September 2019]; Verfügbar unter: http://link.springer.com/10.1007/s00482-019-00408-1. [Google Scholar]
- 5. Austin PC. A tutorial on multilevel survival analysis: methods, models and applications. Int Stat Rev. 2017;85(2):185–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Krcevski-Skvarc N, Wells C, Häuser W. Availability and approval of cannabis-based medicines for chronic pain management and palliative/supportive care in Europe: a survey of the status in the chapters of the European Pain Federation. Eur J Pain. 2018;22(3):440–54. [DOI] [PubMed] [Google Scholar]
- 7. Fitzcharles MA, Baerwald C, Ablin J, Häuser W. Efficacy, tolerability and safety of cannabinoids in chronic pain associated with rheumatic diseases (fibromyalgia syndrome, back pain, osteoarthritis, rheumatoid arthritis): a systematic review of randomized controlled trials. Februar. 2016;30(1):47–61. [DOI] [PubMed] [Google Scholar]
- 8. Fitzcharles MA, Eisenberg E. Medical cannabis: a forward vision for the clinician. Eur J Pain. 2018;22(3):485–91. [DOI] [PubMed] [Google Scholar]
- 9. Fitzcharles MA, Ste-Marie PA, Clauw DJ, Jamal S, Karsh J, LeClercq S, et al. Rheumatologists lack confidence in their knowledge of cannabinoids pertaining to the management of rheumatic complaints. BMC Musculoskelet Disord. 2014;15(1):258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Begleiterhebung BfArMAbschlussbericht. [Internet]. 2022 Juli [zitiert 17. Juli 2022]. Verfügbar unter Available from: https://www.bfarm.de/SharedDocs/Downloads/DE/Bundesopiumstelle/Cannabis/Abschlussbericht_Begleiterhebung.html.
- 11. Ueberall MA, Horlemann J, Schuermann N, Kalaba M, Ware MA. Effectiveness and tolerability of dronabinol use in patients with chronic pain: a retrospective analysis of 12-week open-label real-world data provided by the German pain e-registry. Pain Med. 2022;23(8):1409–22, [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Böttge-Wolpers C, Bialas P, Gottschling S, Juckenhöfel S, Konietzke D, Madlinger A, et al. [Benefits and harms of cannabis-based medicines from the viewpoint of patients with chronic pain and their physicians : a cohort study in three pain centers of the German federal state Saarland]. Schmerz. 2023;20. [DOI] [PubMed] [Google Scholar]
- 13. Erweiterte AWMF. 2020./001OL.S3-Leitlinie Palliativmedizin für Patienten mit nicht -heilbaren Krebserkrankung, Langversion 2.2. - Sept. 2020 AWMF-Registernr128.
- 14. Petzke F, Enax-Krumova EK, Häuser W. Wirksamkeit, Verträglichkeit und Sicherheit von Cannabinoiden bei neuropathischen Schmerzsyndromen: Eine systematische Übersichtsarbeit von randomisierten, kontrollierten Studien. Schmerz. Schmerz. 2016;30(1):62–88. [DOI] [PubMed] [Google Scholar]
- 15. Hinz B, Ramer R. Cannabinoids as anticancer drugs: current status of preclinical research. Br J Cancer. 2022;127(1):1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The research data associated with this paper are blinded patient data. These data are not available due to ethical reasons. Further inquiries can be directed to the corresponding author.