
Keywords: acyl-CoA synthetase long-chain family member 4, ferroptosis, glutathione peroxidase 4, heme oxygenase-1, inflammation, iron, lipid peroxidation, metformin, neuroprotection, spinal cord injury
Abstract
Previous studies have reported upregulation of heme oxygenase-1 in different central nervous system injury models. Heme oxygenase-1 plays a critical anti-inflammatory role and is essential for regulating cellular redox homeostasis. Metformin is a classic drug used to treat type 2 diabetes that can inhibit ferroptosis. Previous studies have shown that, when used to treat cardiovascular and digestive system diseases, metformin can also upregulate heme oxygenase-1 expression. Therefore, we hypothesized that heme oxygenase-1 plays a significant role in mediating the beneficial effects of metformin on neuronal ferroptosis after spinal cord injury. To test this, we first performed a bioinformatics analysis based on the GEO database and found that heme oxygenase-1 was upregulated in the lesion of rats with spinal cord injury. Next, we confirmed this finding in a rat model of T9 spinal cord compression injury that exhibited spinal cord nerve cell ferroptosis. Continuous intraperitoneal injection of metformin for 14 days was found to both upregulate heme oxygenase-1 expression and reduce neuronal ferroptosis in rats with spinal cord injury. Subsequently, we used a lentivirus vector to knock down heme oxygenase-1 expression in the spinal cord, and found that this significantly reduced the effect of metformin on ferroptosis after spinal cord injury. Taken together, these findings suggest that metformin inhibits neuronal ferroptosis after spinal cord injury, and that this effect is partially dependent on upregulation of heme oxygenase-1.
Introduction
Studies have shown that patients with traumatic spinal cord injury (TSCI) often die from acute respiratory failure or a combination of other severe injuries (Bach, 2012; Xie et al., 2022; Yu et al., 2022). In addition, TSCI survivors often exhibit varying degrees of concomitant motor dysfunction. Despite surviving the acute phase of injury, many patients with tetraplegia or paraplegia continue to face various complications, including pulmonary infections, spasticity, autonomic dysreflexia, hypertension, venous thromboembolism, and pain (Adegeest et al., 2022). These secondary health conditions are common long-term complications of TSCI that negatively affect quality of life. Studies have shown that various forms of cell death play a role in histopathologic deterioration after TSCI (Shi et al., 2021). Ferroptosis is a novel type of non-necrotic regulatory cell death regulated by various molecular mechanisms (Stockwell et al., 2017) that aggravates the deteriorating microenvironment following spinal cord injury (SCI; Li et al., 2022). Both invasive and non-invasive interventions can effectively promote functional recovery in experimental animal models of TSCI by inhibiting ferroptosis (Ge et al., 2021, 2022; Ying et al., 2023).
Several recent studies have reported that the ferroptosis-related gene (FRG) heme oxygenase-1 (HMOX1) is significantly upregulated after SCI (Kang et al., 2022; Dong et al., 2023; Li et al., 2023). HMOX1, an inducible enzyme, is known to have anti-inflammatory, antioxidant, and neuroprotective properties. The prevailing factors contributing to ferroptosis induction and progression include marked down-regulation of glutathione peroxidase 4 (GPX4) and upregulation of acyl-coenzyme A synthetase long-chain family member 4 (ACSL4) (Wei et al., 2020; Liu et al., 2022a). In contrast, the role or overall effect of HMOX1 in regulating ferroptosis is controversial. Some studies have suggested that HMOX1 promotes ferroptosis, as it can cause iron accumulation by catalyzing heme degradation to iron, carbon monoxide, and biliverdin (Fang et al., 2019; Fernández-Mendívil et al., 2021; Zeng et al., 2021). In contrast, other studies have shown that HMOX1 inhibits ferroptosis, thereby preventing disease development and progression, by alleviating oxidative stress injury and inflammation (Ma et al., 2020; Cai et al., 2022; Meng et al., 2022; Ryter, 2022). In addition, many reports have suggested that HMOX1 upregulation has beneficial effects on the injured spinal cord (Yamauchi et al., 2004; Vargas et al., 2005; Kanno et al., 2009; Lin et al., 2017, 2021; Wang et al., 2017). However, a recent study suggested that HMOX1 upregulation after SCI aggravates oxidative stress by increasing ferrous iron release, which leads to the accumulation of reactive oxygen species (ROS) (Dong et al., 2023). Nevertheless, other reports suggest that HMOX1 upregulation in the injured spinal cord inhibits ferroptosis (Zhou et al., 2020; Ge et al., 2021). Thus, animal experiments based on bioinformatics analysis results are needed to determine the effect of HMOX1 on ferroptosis after SCI.
Previous studies have shown that metformin (Met), a widely used hypoglycemic drug and classic adenosine 5′-monophosphate-activated protein kinase activator, has anti-ferroptotic effects (Lee et al., 2020; Ma et al., 2021; Yan et al., 2022; Zhao et al., 2023). In addition, Met has been shown to have a neuroprotective effect in many central nervous system diseases (Leech et al., 2019; Dziedzic et al., 2020; Cao et al., 2022; Du et al., 2022; Ning et al., 2022). A recent meta-analysis concluded that treatment with Met results in significant locomotor function recovery in rats with SCI (Chen et al., 2021a). We previously reported that Met promotes functional recovery after SCI by inhibiting ferroptosis (Wang et al., 2022b). Other groups have shown that Met effectively treats cardiovascular and digestive diseases by promoting HMOX1 upregulation (Sauvé et al., 2010; Wu et al., 2018; Yan et al., 2019; Patel et al., 2021). In this study, we explored the correlation between Met administration and HMOX1 expression after SCI and the mechanism by which Met modifies the injury microenvironment by regulating HMOX1 expression. In addition, we investigated the anti-ferroptotic effect of HMOX1 after SCI and the effect of HMOX1 knockdown on Met-mediated inhibition of ferroptosis.
Methods
Data acquisition and gene expression analysis
All SCI data were obtained from the GEO database (https://www.ncbi.nlm.nih.gov/gds/). Ferroptosis markers and regulators were downloaded from the FerrDb V2 database (http://www.zhounan.org/ferrdb/current/; Zhou et al., 2023). Additional Table 1 shows detailed information on all datasets. In brief, the degree of normalization among samples was detected by box plot analysis. Differences in clusters between the sample groups were analyzed by principal component analysis. Analysis of differential expression between the two groups was performed using the “limma” package in R software (https://www.r-project.org/; version 4.0.1, R Foundation, Auckland, New Zealand). The results were visualized by volcano plot with |logFC| > 1 and adjusted P < 0.05.
Additional Table 1.
The detailed information of all datasets
| Dataset | Animal | Model | Group | Status | Country |
|---|---|---|---|---|---|
| GSE45006 | Wistar rats, ♀ | The aneurysm clip impact-compression model; T7 level | Sham, 1 dpi, 3 dpi, 7 dpi, 14 dpi, 56 dpi (days post injury); 4 rep. | Public on November 15, 2013 | Canada |
| GSE2599 | Fischer 344 rats, ♀ | Contusion injury; T8 level | Sham, 35 dpi; 3 rep. | Public on May 03, 2005 | USA |
| GSE42828 | C57BL/6J mice, ♂ | Contusion injury; T9 level | WT: Sham, 1 dpi, 3 dpi, 7 dpi; 4-5 rep. | Public on December 07, 2015 | USA |
| GSE93561 | C57BL/6 mice, ♀ | Contusion injury; T9 level | Sham, 9 dpi; 6 rep. | Public on January 13,2017 | Japan |
| GSE92657 | C57BL/6 mice, ♀ | Transection injury; T7/8 level | Sham, 9 hpi; 3 rep. | Public on October 01,2017 | Germany |
♀: Female; ♂: male; dpi: day(s) post injury; hpi: hours post injury; rep: repetitions.
Enrichment analysis and protein-protein interaction network construction
Using the Gene Ontology database (http://geneontology.org/) (Thomas et al., 2022) and the Kyoto Encyclopedia of Genes and Genomes database (https://www.genome.jp/kegg/) (Kanehisa and Goto, 2000), enrichment analysis was performed for 17 ferroptosis-related differentially expressed genes (DEGs) to investigate their role in SCI. The Gene Ontology annotation analysis included biological process, molecular function, and cellular component. Functional and pathway enrichment analysis was performed using the clusterProfiler package in R software. To detect protein-protein interactions, network analysis was performed using the online STRING database (https://cn.string-db.org/) with the default settings (Szklarczyk et al., 2019).
Animals
All animal care and experimental protocols were carried out in accordance with the Animal Management guidelines of the Ministry of Health of the People's Republic of China. This study was approved by the Medical Ethics Committee of the Animal Experimental Center of Nanchang University on March 10, 2021. Because men predominate patients with TSCI (Wang et al., 2013, 2022a), only male rats were used in this study. In total, 220 specific pathogen-free male Sprague-Dawley rats (weight 220–250 g, age 8 weeks) were purchased from the Experimental Animal Center of Jiangxi University of Chinese Medicine (Nanchang, Jiangxi, China; license No. SCXK (Gan) 2018-0003). The rats were randomly grouped and housed in standardized cages (five rats per cage) for at least 7 days before surgery, in a controlled environment at 22–24°C, 50% relative humidity, and a 12/12-hour light/dark cycle.
SCI model and Met intervention
As shown in Figure 1, this study involved three animal experiments (Parts 1–3). In Part 1, to identify ferroptotic characteristics at different time points after SCI, the rats were randomly assigned to sham and SCI groups. Three rats were included in each group for transmission electron microscopy (TEM), five rats were included in each group for western blotting, and six rats were included in each group for Prussian blue staining, Fluoro-Jade B (FJB), and immunohistochemistry. Anesthesia was induced by inhalation of 5% isoflurane (RWD, Shenzhen, China) and maintained with 1.5% isoflurane. The sham group underwent laminectomy at the T9 level without injury to the spinal cord, whereas the SCI group underwent laminectomy followed by compression injury at the T9 level delivered by application of an aneurysm clip (No. FT220T; Am Aesculap-Platz, Tuttlingen, Germany) for 10 seconds. Subsequently, the spinal cord was removed at a predetermined time point. In Part 2, rats were randomly assigned to sham, SCI, and Met groups to characterize the anti-ferroptotic effect of Met after SCI. The sham group underwent laminectomy only; the SCI group underwent laminectomy and compression SCI followed by intraperitoneal injection of about 1 mL of normal saline 30 minutes after injury and then once a day until the rats were sacrificed (up to 14 days after injury); and the Met group underwent laminectomy, compression SCI, and intraperitoneal injection of 200 mg/kg Met (Selleck, Houston, TX, USA) 30 minutes after SCI and then once a day until the rats were sacrificed (up to 14 days after SCI). Six rats were included in each group for immunofluorescence staining and PCR detection, and five rats were included in each group for western blotting. In Part 3, rats were randomly assigned to normal control (NC) and knockdown (KD) groups to determine the effect of target gene knockdown on ferroptosis and the anti-ferroptotic capacity of Met. Animals in the sham, SCI, and Met subgroups were treated as described above. Three rats were included in each subgroup for spinal cord sectioning, and five rats were included in each subgroup for western blotting, immunofluorescence staining, and FJB. After each surgical procedure, the skin and muscles were carefully stitched together. As a precaution against infection, penicillin (Dingjian, Sichuan, China) was injected subcutaneously for 3 days at a dosage of 2.5 mg/kg daily. The bladder was evacuated manually three times a day until autonomous urinary function was regained, which generally took 10 to 14 days. The in vivo experimental process is shown in Figure 1.
Figure 1.
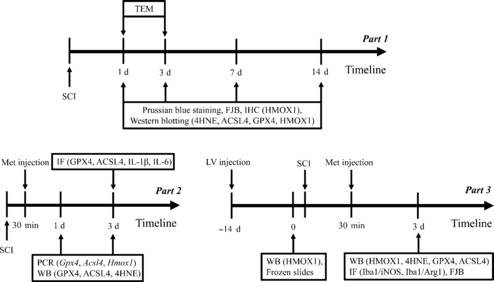
Experimental flowchart for the in vivo experiments.
4HNE: 4-Hydroxynonenal; ACSL4: acyl-coenzyme A synthetase long-chain family member 4; Arg1: arginase 1; GPX4: glutathione peroxidase 4; HMOX1: heme oxygenase-1; IF: immunofluorescence; IHC: immunohistochemistry; Iba1: ionized calcium binding adaptor molecule 1; IL-1β: interleukin 1β; IL-6: interleukin 6; iNOS: inducible nitric oxide synthase; LV: lentivirus; PCR: polymerase phain peaction; SCI: spinal cord injury; TEM: transmission electron microscopy; WB: western blotting.
Cell culture
The PC-12 cell line was purchased from Otwo Biotechnology Co., Ltd. (Shenzhen, Guangdong, China, Cat# HTX1783, RRID: CVCL_0481). This is a rat-derived cell line that was used as alternative to neurons for the in vitro experiments. Briefly, cells were cultured in high-glucose Dulbecco's modified Eagle's medium (Gibco, Beijing, China) supplemented with fetal bovine serum (Cat# 10100147C, Gibco, Grand Island, NY, USA), 100 U/mL penicillin G, and 0.1 mg/mL streptomycin in a 5% CO2 and 95% air incubator at 37°C.
Construction of stable cell line and in vivo transfection
Lentivirus (LVs) containing Hmox1-shRNA (NM_012580) were constructed and synthesized by Genechem Co., Ltd. (Shanghai, China). The LV sequences and titer information are shown in Additional Table 2. In addition, an LV-GFP-Hmox1-shRNA green fluorescent protein (GFP)-tagged LV was constructed to evaluate transfection efficiency. In brief, stable HMOX1 knockdown cell line were generated by transfecting cells with one of the two LVs. HMOX1 expression at the mRNA and protein level in the resulting two stable cell lines was evaluated to determine transfection effectiveness. To generate an in vivo model of HMOX1 knockdown, rats were randomly assigned to the NC and KD groups and injected with one of the two LVs, using the same injection site, technique, and dosage. In addition, all procedures were performed by the same investigator. Each rat was subjected to full anesthesia with isoflurane, after which the T9 segment was fully exposed and 10 μL of LV containing the target gene (shRNA) was injected into the spinal cord using a micro-syringe (outer diameter/inner diameter of the needle: 0.16/0.06 mm, 207434; RWD, Shenzhen, China). No hind limb motor function loss was observed when the rats had recovered from the anesthesia. Fourteen days after injection with the LVs, the rats were euthanized by inhaling excessive isoflurane, and the range of LV infection in tissue slices was assessed by evaluating green fluorescence. Knockdown was validated at the protein level.
Additional Table 2.
Sequences and titer information of lentiviruses
| Accession | Target Seq | CDS | GC% | Titer (TU/mL) | |
|---|---|---|---|---|---|
| LV-Hmox 1 -RNAi(a) | NM 012580 | GGTGTCCAGGGAAGGCTTTAA | 117...986 | 52.38 | 1E+9 |
| LV-Hmox1-RNAi(b) | NM 012580 | GCTCAACATTGAGCTGTTTGA | 117...986 | 42.86 | 8E+8 |
| LV-Hmoxl-RNAi(c) | NM 012580 | GCACATCCGTGCAGAGAATTC | 117.986 | 52.38 | 1E+9 |
CDS: Coding sequence.
Frozen section preparation
Anesthesia was induced by inhalation of 5% isoflurane and maintained with 1.5% isoflurane; then, the right atrial appendage was excised, and the rats were perfused with 250–300 mL of 4°C normal saline. Once the effluent liquid ran clear, the whole spinal cord was harvested and dried with gauze. The spinal cord tissue was then snap-frozen in liquid nitrogen for 15 seconds, embedded in optimal cutting temperature compound, cut into large pieces, then sliced into 8- to 10-μm-thick sections.
Prussian blue staining
Iron deposition was detected by Prussian blue staining (Perls' staining) as described previously, with minor modifications (Li et al., 2017; Xie et al., 2019). For spinal cord tissues, the region of interest was cut into 3-μm-thick coronal sections. Then, the sections were placed into xylene, absolute ethanol, and 75% alcohol, followed by washing with tap water and rinsing three times with distilled water. Next, the sections were placed in a 1:1 mixture of 2% potassium ferrohydride and 2% hydrochloric acid and then washed twice with distilled water. Next, the sections were stained with diaminobenzidine (Servicebio, Wuhan, China) for 5–10 minutes. The staining solution was removed, and the stained sections were rinsed once with 0.01 M phosphate-buffered saline, followed by three washes with distilled water. Next, the degree of color development was visualized under an optical microscope (Axio Lab. A1 + ERC5S, ZEISS, Jena, Germany). Sections were counterstained with hematoxylin (Servicebio) for 1 minute, followed by washing with tap water. The sections were differentiated with a hydrochloric acid aqueous solution followed by washing with tap water. Next, the sections were immersed in an aqueous ammonia solution and washed with tap water. Then, they were soaked in absolute ethanol and sealed with neutral balsam. The sections were viewed and photographed under a microscope (Axio Lab. A1 + ERC5S).
FJB staining
For FJB staining, spinal cord tissue sections were incubated in two changes of xylene, dehydrated in two changes of pure ethanol, and dehydrated by passaging through 85% and 75% ethanol. After washing in distilled water, the sections were air-dried. Freshly prepared 50% glacial acetic acid was used as the solvent for the 1:400 FJB working solution (Servicebio). The sections were incubated with dilute FJB solution at 4°C overnight in a humid box. After washing three times with distilled water on a shaker, the sections were incubated with 4′,6-diamidino-2-phenylindole solution (Servicebio) at about 25°C and stored in the dark. They were then rinsed with distilled water, dried with a hair dryer, cleared with xylene, and mounted with resin mounting medium. Representative images were captured using an inverted fluorescence microscope (Axio Observer + Axiocam 208, ZEISS).
Western blotting
Western blotting was performed to detect levels of 4-hydroxynonenal (4HNE), GPX4, ACSL4, and HMOX1 in spinal cord tissues and HMOX1 in PC12 cells. The primary antibodies used were rabbit polyclonal antibody against 4HNE (1:1000, Abcam, Cambridge, UK, Cat# ab46545, RRID: AB_722490), rabbit monoclonal antibody against ACSL4 (1:10,000, Abcam, Cat# ab155282, RRID: AB_2714020), rabbit monoclonal antibody against GPX4 (1:10,000, Abcam, Cat# ab125066, RRID: AB_10973901), rabbit monoclonal antibody against HMOX1 (1:2000, Abcam, Cat# ab189491, RRID: AB_1267209), and rabbit polyclonal antibody against β-actin (1:10,000, Affinity, Cincinnati, OH, USA, Cat# AF7018, RRID: AB_2839420). The membranes were incubated with the primary antibodies overnight (12 hours) at 4°C. Goat anti-rabbit IgG(H+L) (1:2000, Immunoway, Plano, TX, USA, Cat# RS0002, RRID: AB_2938759) was used as a secondary antibody for 1-hour incubation at about 25°C. Then, the proteins were subjected to SDS-PAGE then transferred to a nitrocellulose filter membrane at 200 mA constant current for 120–150 minutes in an ice bath. An enhanced chemiluminescence system (ChemiDoc XRS+ System; Bio-Rad, Hercules, CA, USA) was used to visualize the target protein bands. The relative protein levels were normalized to optical density of β-actin and were quantified using ImageJ (v2021.8.0, National Institutes of Health, Bethesda, MD, USA; Schneider et al., 2012).
TEM
For TEM, spinal cord tissues were rapidly fixed in phosphate-buffered glutaraldehyde (2.5%), followed by osmium tetroxide (1%). Then, they were dehydrated in graded ethanol solutions (50–70–80–90–95–100–100%) and acetone (100%) before embedding in epoxy resin. After resin penetration and embedding, the samples were incubated at 65°C for more than 48 hours in an oven to polymerize. The resin blocks were cut into 60- to 80-nm-thick slices using an ultramicrotome (Leica UC7; Leica, Wetzlar, Germany). Finally, the mitochondrial ultrastructure was examined, and images were captured, by TEM (HT7700; Hitachi, Tokyo, Japan).
Immunohistochemistry
For immunohistochemistry, spinal cord slices mounted on slides were immersed in sodium citrate antigen retrieval solution (pH 6.0), deparaffinized, rehydrated, and maintained at a sub-boiling temperature. Then, they were immersed in 3% H2O2 and incubated at room temperature in the dark to block the activity of endogenous peroxidase. The slides were then incubated with primary rabbit monoclonal antibody against HMOX1 (1:20,000, Abcam, Cat# ab189491, RRID: AB_1267209) at 4°C overnight in a humid box. Next, the slides were incubated at about 25°C for 50 minutes with a secondary antibody (1:500, goat anti-rabbit IgG(H+L), RS0002, Immunoway, RRID: AB_2938759) labeled with horseradish peroxidase. After developing with diaminobenzidine and counterstaining the nucleus with hematoxylin staining solution, the sections were embedded in resin mounting medium. As described previously (Li et al., 2020), HMOX1 expression in the spinal cord was quantified by two pathologists blinded to the group assignments. The immunostaining intensity was scored as negative = 0, weak = 1, moderate = 2, or strong = 3. The percentage of positively stained cells was scored as follows: < 25% = 1, 25–50% = 2, 50–75% = 3, and > 75% = 4. The overall score was then calculated by multiplying the intensity score by the proportion score (score = intensity × percentage of positively stained cells). Images were captured using an optical microscope (Axio Lab. A1 + ERC5S).
Quantitative polymerase chain reaction
For quantitative polymerase chain reaction (PCR), the expression levels of FRGs including glutathione peroxidase 4 (Gpx4), acyl-coenzyme A synthetase long-chain family member 4 (Acsl4), and heme oxygenase-1 (Hmox1) were analyzed in injured rats and sham controls 3 days after SCI. Briefly, total RNA was isolated from spinal cord samples using Trizol reagent (R401-01; Vazyme, Nanjing, China). The extracted RNA was quantified using a NanoDrop One (Thermo Fisher Scientific, MA, USA). cDNA was synthesized using a Hifair 1st Strand cDNA Synthesis SuperMix Kit (11141ES60; Yeasen, Shanghai, China). mRNA expression was detected with CFX96 Touch (Bio-Rad) using Hieff qPCR SYBR Green Master Mix (11201ES08; Yeasen). Gene expression was normalized to dehydrogenase (GAPDH). The specific primers are listed in Additional Table 3.
Additional Table 3.
Primer sequences used in PCR analysis
| Gene | Primer sequence (5'-3') |
|---|---|
| Hmoxl | Forward primer: GCTTTAAGCTGGTGATGG Reverse primer: CTGGTGTGTAAGGGATGG |
| Gpx4 | Forward primer: GCCGAGTGTGGTTTACGA Reverse primer: CTTGGGCTGGACTTTCAT |
| Acsl4 | Forward primer: ACTTTGATACCAGGGCTTGA Reverse primer: GGCTCTCTGAAATGCACAC |
| GAPDH | Forward primer: GCGAGATCCCGCTAACATCA Reverse primer: CTCGTGGTTCACACCCATCA |
Acsl4: acyl-coenzyme A synthetase long-chain family member 4; Gpx4: glutathione peroxidase 4; Hmoxl : heme oxygenase-1.
Immunofluorescence staining
For immunofluorescence staining, spinal cord sections were deparaffinized by sequential incubation in xylene, anhydrous ethanol, 85% ethanol, and 75% ethanol. After washing in distilled water, they were microwaved in citric acid antigen retrieval buffer (pH 6.0) for antigen retrieval. Next, the sections were placed in hydrogen peroxide and bovine serum albumin, and then the primary antibody was added, and the sections were incubated at 4°C overnight in a humid box. The primary antibodies used for immunofluorescence were rabbit monoclonal antibody against GPX4 (1:500, Abcam, Cat# ab125066, RRID: AB_10973901), rabbit monoclonal antibody against ACSL4 (1:500, Abcam, Cat# ab155282, RRID: AB_2714020), rabbit polyclonal antibody against interleukin-1β (1:200, Affinity, Cat# AF5103, RRID: AB_2837589), rabbit polyclonal antibody against interleukin-6 (1:200, Affinity, Cat# DF6087, RRID: AB_2838055), rabbit polyclonal antibody against neuronal nuclei (1:2000, Servicebio, Cat# GB11138, RRID: AB_2868432), rabbit monoclonal antibody against ionized calcium binding adaptor molecule 1 (Iba1; 1:3000, Abcam, Cat# ab178846, RRID: AB_263685), rabbit monoclonal antibody against inducible nitric oxide synthase (iNOS, 1:300, Abcam, Cat# ab178945, RRID: AB_2861417), and rabbit polyclonal antibody against arginase 1 (Arg1, 1:300, GeneTex, San-Antonio, TX, USA, Cat# GTX109242, RRID: AB_2036264). The secondary antibodies used for immunofluorescence were Cy3-conjugated goat anti-rabbit IgG (H+L) (1:300, Servicebio, Cat# GB21303, RRID: AB_2861435) and Alexa Fluor 488-conjugated goat anti-rabbit IgG (H+L) (1:400, Servicebio, Cat# GB25303, RRID: AB_2910224). Next, the cell nuclei were counterstained with 4′,6-diamidino-2-phenylindole, followed by autofluorescence quenching and mounting with anti-fluorescence quenching mounting tablets. The fresh tissues were embedded and in optimal cutting temperature compound (G6059-110ML; Servicebio, Wuhan, China) and sliced into 8- to 10-μM-thick sections once the compound turned white and hard. Then, a coverslip was placed on top of the tissue slice and sealed. Representative images were captured using the fluorescence microscope (Axio Observer + Axiocam 208). During image analysis, three people who were unaware of the grouping counted protein-positive neurons or microglia and averaged the results. The main inclusion criterion is that the cells have distinct red (neurons or microglia), green (GPX4, ACSL4, IL-1β, or IL-6), and blue (DAPI) fluorescence at the same time.
Statistical analysis
No sample size calculation was performed to determine the small sample size for this study; however, our sample sizes are similar to those used in previous studies (Chen et al., 2022; Li et al., 2023). The evaluator was blinded to the group assignments, and no rats were excluded from the analyses. All statistical analyses were performed using SPSS software (version 25.0, IBM Corp., Armonk, NY, USA), R software, and GraphPad Prism (version 8.02, Graphpad, San Diego, CA, USA, http://www.graphpad.com). All data are expressed as the mean ± standard error of the mean (SEM). Data distribution was assessed using the Shapiro-Wilk normality test. Differences between the two groups were analyzed using the independent samples t test. Differences between multiple groups were analyzed using one-way or two-way analysis of variance with Bonferroni or Tukey's post hoc test. P < 0.05 was considered statistically significant.
Results
Bioinformatics analysis reveals significant Hmox1 upregulation in rats after SCI
To identify differentially expressed FRGs in the injured spinal cord microenvironment in rats at different time points after SCI, two eligible datasets (GSE45006 and GSE2599) were downloaded from the GEO database, and differential expression analyses were performed. Preliminary analysis of the GSE45006 dataset identified 808 DEGs in the SCI group at five different time points (1, 3, 7, 14, and 56 days post-injury (dpi)) (Figure 2A). Further analysis identified 17 differentially expressed FRGs, including Hmox1, Cybb, Atf3, Capg, Lrrfip1, Gls2, Tlr4, Plin2, Gch1, Cd44, Ano6, Sat1, Ripk1, Chmp6, Tgfbr1, Gria3, and Stat3 (Figure 2B). The GSE2599 dataset only contained gene expression data from 35 days after SCI. Only seven differentially expressed FRGs (Hmox1, Cp, Tgfbr1, Srebf1, Gch1, Ifng, and Got1) were identified (Figure 2C). Comparison of these two datasets reveled three shared DEGs (Hmox1, Tgfbr1, and Gch1) (Figure 2D). The differential expression of these genes was analyzed using volcano plots (Figure 2E–J). Hmox1 was upregulated at each time point assessed (1, 3, 7, 14, 35, and 56 dpi). Analysis of three other datasets (GSE92657, GSE42828, and GSE93561) showed that Hmox1 is also upregulated in mice after SCI (Additional Figure 1 (2.9MB, tif) ).
Figure 2.
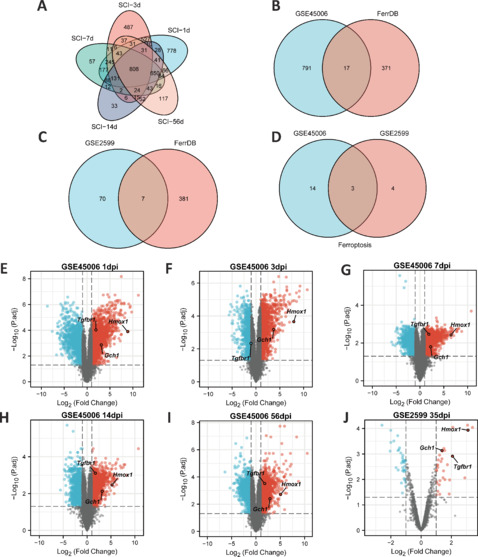
Hmox1 is significantly upregulated in a rat model of SCI.
(A) Common DEGs at five time points in the GSE45006 dataset. (B) Ferroptosis-related DEGs (from the FerrDb V2 database (http://www.zhounan.org/ferrdb/current/)) in the GSE45006 dataset. (C) Ferroptosis-related DEGs in the GSE2599 dataset. (D) Common ferroptosis-related DEGs in the GSE45006 and GSE2599 datasets. (E–I) Visualization of DEGs at different time points in the GSE45006 dataset, including three common ferroptosis-related DEGs. (J) Visualization of DEGs in the GSE2599 dataset, containing three common ferroptosis-related DEGs. |logFC| > 1, adjusted P value < 0.05. DEGs: Differentially expressed genes; dpi: day(s) post injury; FC: fold change; Gch1: GTP cyclohydrolase 1 gene; Hmox1: heme oxygenase-1; SCI: spinal cord injury; Tgfbr1: transforming growth factor beta receptor 1.
Bioinformatics analysis reveals dynamic changes in the expression of inflammatory factors and FRGs after SCI
Further analyses focused on the GSE45006 dataset. Gene Ontology and Kyoto Encyclopedia of Genes and Genomes enrichment analyses were performed to determine the function of the 17 DEGs. The results confirmed an association between ferroptosis and SCI (Figure 3A and B). Next, the DEGs were uploaded to the STRING database to construct a protein-protein interaction network using the default settings (Figure 3C), which showed that HMOX1 may play a critical role in cellular ferroptosis after SCI. Several classical indicators of inflammation were selected and analyzed to investigate dynamic changes in the expression level of genes associated with the inflammatory response and FRGs after SCI. The results showed that, after normalization to the sham group, interleukin 1β (Il-1β) and interleukin 6 (Il-6) expression levels increased in the SCI group on day 1, peaked on day 3, and returned to baseline levels on day 7 (Figure 3D and E). Interleukin 10 (Il-10) expression decreased continuously for 7 days and fluctuated around baseline levels for 49 days, remaining at roughly normal levels throughout the follow-up period (Figure 3F). Although the fold change (FC) value was not significant, expression of Gpx4, a key protein involved in ferroptosis, continued to decrease after SCI (Figure 3G). Slc7a11 expression fluctuated around baseline, with an overall increasing trend, whereas Slc3a2 remained upregulated (Figure 3H and I). Acsl4 increased on day 1, peaked on day 3, and returned to baseline on day 14 (Figure 3J). Lpcat3 was upregulated at several time points, although this change was not significant on day 3 (Figure 3K). Hmox1 was upregulated throughout the follow-up period (Figure 3L).
Figure 3.
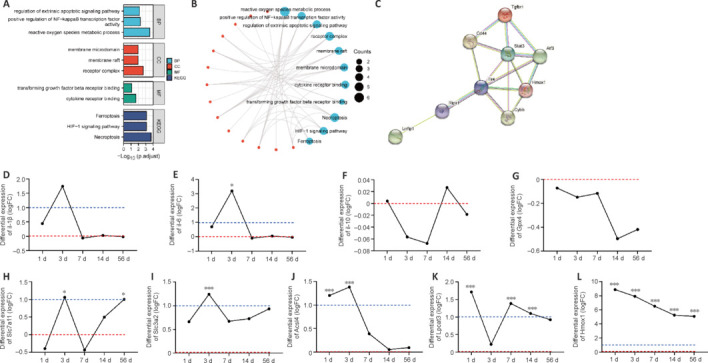
Dynamic changes in the expression of inflammatory cytokine genes and ferroptosis-related genes (FRGs) after SCI.
(A, B) Functional annotation and pathway enrichment analysis of 17 ferroptosis-related DEGs from the GSE45006 dataset. Red represents molecules, and blue represents GO and KEGG items. (C) Protein-protein interaction network of 17 ferroptosis-related DEGs. (D–L) Dynamic changes in nine FRGs of interest after SCI. Red line: baseline value, indicating that the expression level of this gene at this time point was 1 time that of the sham group (20). Blue line: |logFC| = 1, indicating that the expression level of this gene at this time point was 2 time that of the sham group (21). Adjusted *P < 0.05, adjusted ***P < 0.001 (independent samples t test). Acsl4: Acyl-coenzyme A synthetase long-chain family member 4; Atf3: activating transcription factor 3; BP: biological brocess; CC: cell component; CD44: cluster of differentiation 44; Cybb: cytochrome b-245 beta chain; DEGs: differentially expressed genes; FRG: ferroptosis-related gene; Gpx4: glutathione peroxidase 4; HIF-1: hypoxia inducible factor-1; Hmox1: heme oxygenase-1; Il-10: interleukin 10; Il-1β: interleukin 1β; Il-6: interleukin 6; KEGG: Kyoto Encyclopedia of Genes and Genomes; Lpcat3: lysophosphatidylcholine acyltransferase 3; Lrrfip1: lrr binding flii interacting protein 1; MF: molecular function; Ripk1: receptor-interacting protein kinase 1; SCI: spinal cord injury; Slc3a2: solute carrier family 3 member 2; Slc7a11: solute carrier family 7 member 11; Stat3: signal transducer and activator of transcription 3; Tgfbr1: transforming growth factor beta receptor 1; Tlr4: Toll-like receptor 4.
SCI-induced lipid peroxidation and iron deposition aggravate ferroptosis
The schematic image shown in Figure 4A depicts the morphological changes observed in the spinal cord on days 1 and 7 after SCI. The representative images were selected from the approximate area of the anterior horn of the spinal cord within the region of interest (Figure 4B). We detected iron deposition starting on day 1 after SCI, and iron deposition levels were significantly elevated a week later (Figure 4C and D). Unlike iron deposition, which appeared later, neuron degeneration was readily apparent within the first 3 days after SCI and did not increase significantly thereafter (Figure 4E and F). Western blotting showed that 4HNE and ACSL4 were upregulated in the first 3 days and returned to near-normal levels on day 7 (Figure 4G and H). GPX4 expression decreased slowly over the follow-up period, while HMOX1 remained upregulated (Figure 4I and J). During the initial 3-day period, we did not observe any mitochondria with typical characteristics associated with ferroptotic cell death; instead, we identified damaged mitochondria showing reduced or absent cristae (Figure 4K). Owing to the inflammatory response, lipid peroxidation and neuronal cell death were more pronounced in the first 3 days after SCI than at later time points, suggesting that this is the appropriate time frame for assessing the role of ferroptosis in SCI.
Figure 4.
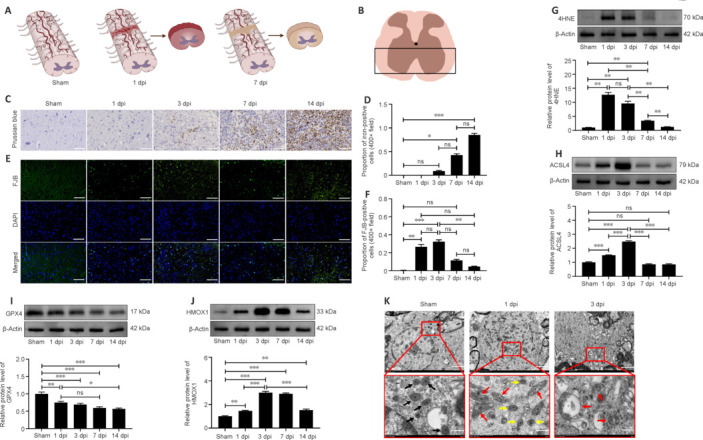
SCI-induced lipid peroxidation and iron deposition aggravate ferroptosis.
(A) Schematic diagrams of the sham, 1 dpi, and 7 dpi groups and the main region of interest (ROI). (B) Representative area of interest in the ROI. (C, D) Representative images of Prussian blue staining and quantification of iron-positive cells. Iron deposition was not detected until 3 days after SCI and was significantly elevated on days 7 and 14 compared with the sham group (n = 6). Scale bars: 50 μm. (E, F) Representative images of Fluoro-Jade B (FJB) staining and quantification of FJB-positive neurons. The number of degenerating neurons was significantly increased on day 3, but not on days 7 and 14, after SCI compared with the sham group (n = 6). Scale bars: 50 μm. (G–J) Relative protein expression levels of 4HNE, ACSL4, GPX4, and HMOX1, normalized to the sham group (n = 5). (K) Mitochondrial ultrastructure was examined by transmission electron microscopy (n = 3). Scale bars: 2 μm (upper panel), 500 nm (lower panel). Black arrows: normal mitochondria, red arrows: damaged mitochondria, yellow arrows: smaller mitochondria (that did not exhibit shrunken morphology). The data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001 (one-way analysis of variance with Bonferroni post hoc test). 4HNE: 4-Hydroxynonenal; ACSL4: acyl-coenzyme A synthetase long-chain family member 4; dpi: day(s) post injury; GPX4: glutathione peroxidase 4; HMOX1: heme oxygenase-1; hpi: hours post-injury; ns: not significant; SCI: spinal cord injury.
Met attenuates SCI-induced ferroptosis in vivo
Based on the results of bioinformatics and western blotting analyses, we assessed HMOX1 expression in the gray matter and white matter of the spinal cord at different time points after SCI. The immunohistochemistry results showed that HMOX1 was uniformly and consistently upregulated in the SCI group compared with the sham group (Figure 5A–D). Studies have suggested that GPX4 downregulation and ACSL4 upregulation induce and promote the progression of ferroptosis (Chen et al., 2021b; Liu et al., 2022a). Here, we found that SCI induced a significant decrease in Gpx4 expression on day 3, rather than on day 1, after SCI. In addition, Gpx4 expression was significantly upregulated within the first 3 days after Met treatment (Figure 5E). ACSL4 and HMOX1 were upregulated after SCI, while Met treatment downregulated ACSL4 and upregulated HMOX1 (Figure 5F–I). Met treatment also downregulated the trauma-induced 4HNE expression, further demonstrating that Met effectively regulates lipid peroxidation levels after SCI (Figure 5J). To determine the extent of ferroptotic neuronal cell death at the tissue level, we performed double immunofluorescence labeling of GPX4, ACSL4, IL-1β, and IL-6 with neuronal marker and found that, compared with the SCI group, neurons from rats in the Met treatment group exhibited upregulated GPX4 expression and downregulated ACSL4, IL-1β, and IL-6 expression (Figure 6A–H). This indicated that Met treatment reduced both ferroptosis and inflammation in neurons after SCI. In conclusion, these findings show that Met inhibit ferroptosis at the tissue and neuronal levels.
Figure 5.
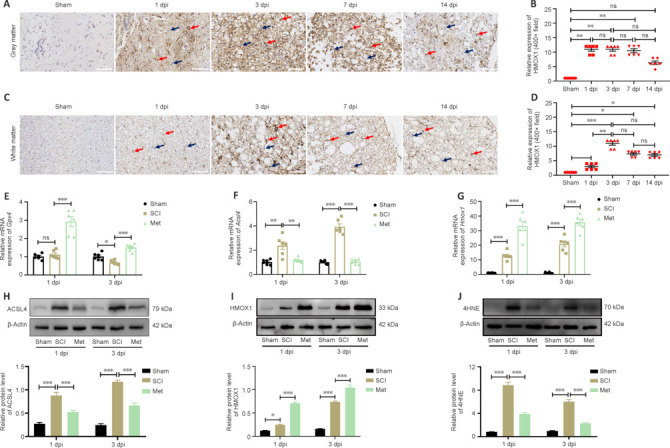
Met treatment increases HMOX1 expression after SCI.
(A, B) Representative images of immunohistochemistry (IHC) staining and quantification of HMOX1 expression in the gray matter. HMOX1 expression was significantly upregulated in the first 7 days after injury in the SCI group compared with the sham group (n = 6). Scale bars: 50 μm. (C, D) Representative images of IHC staining and quantification of HMOX1 expression in the white matter. HMOX1 expression was significantly upregulated in the first 14 days after injury in the SCI group compared with the sham group. HMOX1 expression was highest on day 3 after SCI compared with the Sham group (n = 6). Red arrows: positively stained cells, black arrows: non-stained cells. Scale bars: 50 μm. (E–G) Relative mRNA levels of Gpx4, Acsl4, and Hmox1 were determined by PCR (n = 6). (H–J) Relative protein expression levels of ACSL4, HMOX1, and 4HNE in the indicated groups (n = 5). Expression levels shown in E–G were normalized to those in the sham group. The data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001 (one-way analysis of variance with Bonferroni post hoc test (B, D) or two-way analysis of variance with Tukey's post hoc test (E–J)). 4HNE: 4-Hydroxynonenal; ACSL4: acyl-coenzyme A synthetase long-chain family member 4; dpi: day(s) post injury; GPX4: glutathione peroxidase 4; HMOX1: heme oxygenase-1; Met: metformin; ns: not significant; SCI: spinal cord injury.
Figure 6.
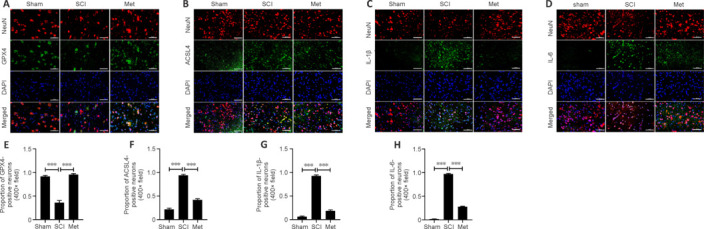
Met attenuates neuronal cell ferroptosis and neuroinflammation after SCI.
(A–D) Representative IF staining images showing GPX4+ and ACSL4+ neurons, and quantitative analysis of the results. Ferroptosis in the SCI group was increased compared with the sham group, and this effect was dramatically reversed by Met treatment (n = 5). (E–H) Representative IF staining images showing IL-1β+ and IL-6+ neurons, and quantitative analysis of the results. Neuroinflammation in the SCI group was increased compared with the sham group, and this effect was dramatically reversed by Met treatment (n = 5). Red indicates Cy3-positive neurons, and green indicates cells that stained positively for GPX4, ACSL4, IL-1β, or IL-6. Scale bars: 50 μm. The data are expressed as the mean ± SEM. ***P < 0.001 (one-way analysis of variance with Bonferroni post hoc test). ACSL4: Acyl-coenzyme A synthetase long-chain family member 4; GPX4: glutathione peroxidase 4; IF: immunofluorescence; IL-1β: interleukin 1β; IL-6: interleukin 6; Met: metformin; SCI: spinal cord injury.
HMOX1 knockdown partially reverses the neuroprotective effects of Met after SCI
To minimize the use of laboratory animals, we first used the PC-12 cell line to evaluate the effectiveness of the LV vector. HMOX1 expression was reduced in all transfected PC-12 cells compared with NC cells, indicating the effectiveness of the knockdown construct (Additional Figure 2 (3.4MB, tif) A–C). LV-Hmox1-RNAi(c) was selected for use in the animal experiments owing to its superior knockdown effect. On day 14 after LV injection, rat spinal cord tissues were harvested and prepared for western blotting. The western blot results demonstrated the effectiveness of LV-mediated knockdown in downregulating HMOX1 expression in vivo (Additional Figure 2 (3.4MB, tif) D–F). To assess the role of HMOX1 in ferroptosis and the anti-ferroptotic effects of Met, rats were injected with LV 14 days prior to compression injury, and spinal cord tissues were collected on day 3 after SCI. HMOX1 expression was then evaluated in all six subgroups: the NC group (sham, SCI, and Met subgroups) and the KD group (sham, SCI, and Met subgroups). In the NC group, HMOX1 expression was significantly upregulated after SCI, and it was further increased after Met treatment. LV-mediated knockdown of HMOX1 expression was stable and long-lasting. Neither SCI nor Met treatment was effective in reversing this effect (Figure 7A). In both the NC and KD groups, 4HNE was upregulated after SCI, but it was downregulated after Met treatment. In the SCI subgroups (SCI subgroup of the KD group vs. SCI subgroup of the NC group), 4HNE expression was not significantly increased after HMOX1 knockdown. In the Met subgroups (Met subgroup of the KD group vs. Met subgroup of the NC group), however, 4HNE expression was significantly upregulated after HMOX1 knockdown (Figure 7B). Similarly, compared with the sham group, ACSL4 levels were upregulated after SCI in both the NC and the KD group. However, Met treatment effectively reduced ACSL4 expression levels in the NC group compared with the KD group. In the SCI and Met subgroups, ACSL4 expression increased significantlyafter HMOX1 knockdown (Figure 7C). Finally, compared with sham group, we found that GPX4 levels were significantly downregulated after SCI in both the NC and KD groups. However, Met treatment effectively increased GPX4 expression levels in the NC group compared with the KD group. In addition, in both the SCI and Met subgroups, GPX4 expression was significantly downregulated after HMOX1 knockdown (Figure 7D). These results show that HMOX1 has at least some anti-ferroptotic effect after SCI, and that the anti-ferroptotic effect of Met depends partly on HMOX1 upregulation.
Figure 7.
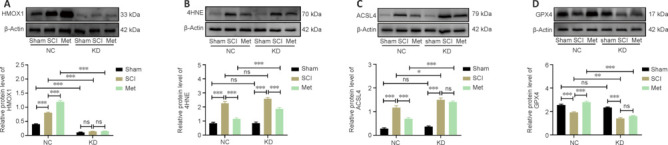
HMOX1 knockdown partially reverses the anti-ferroptotic effects of Met after SCI.
(A–D) Representative western blot showing HMOX1, 4HNE, ACSL4, and GPX4 expression levels in the indicated groups at 3 dpi (n = 5). Expression levels were normalized to the β-actin expression level in the same lane. The data are expressed as the mean ± standard error of the mean. *P < 0.05, **P < 0.01, ***P < 0.001 (two-way analysis of variance with Tukey's post hoc test). 4HNE: 4-Hydroxynonenal; ACSL4: acyl-coenzyme A synthetase long-chain family member 4; dpi: day(s) post injury; GPX4: glutathione peroxidase 4; HMOX1: heme oxygenase-1; KD: knockdown; Met: metformin; NC: normal control; ns: not significant; SCI: spinal cord injury.
After TSCI, activated M1 microglia release nitric oxide (NO), causing oxidative stress and a neurotoxic cascade in neurons. It is thought that Arg1 blocks uncoupling of nitric oxide synthase (NOS) in M2 microglia (Ma et al., 2017). HMOX1 expression in macrophages is induced by stress and promotes their differentiation into an M2 phenotype, with associated anti-inflammatory activities (Campbell et al., 2021). In both the NC and KD groups, the proportion of Iba1+/iNOS+ microglia/macrophages was significantly upregulated after SCI, and significantly downregulated after Met therapy. In the SCI and Met subgroups (SCI subgroup of the KD group vs. SCI subgroup of the NC group, Met subgroup of the KD group vs. Met subgroup of the NC group), the proportion of Iba1+/iNOS+ microglia/macrophages was significantly upregulated after HMOX1 knockdown. This indicates that HMOX1 upregulation and Met treatment prevented overactivation of M1-type microglia in SCI rats (Figure 8A and B). Similarly, in both the NC and KD groups, the ratio of Iba1+/Arg1+ microglia/macrophages was significantly increased after SCI, and Met treatment further increased this ratio. However, in the SCI and Met subgroups, the ratio of Iba1+/Arg1+ microglia/macrophages was significantly decreased after HMOX1 knockdown. This indicates that HMOX1 upregulation and Met treatment promoted activation of M2-type microglia cells in SCI rats (Figure 8C and D). These findings suggest that both HMOX1 upregulation and Met treatment after SCI can promote the polarization of M1 microglia/macrophages towards an M2 phenotype, and that the effects of Met may not be dependent on HMOX1 upregulation.
Figure 8.
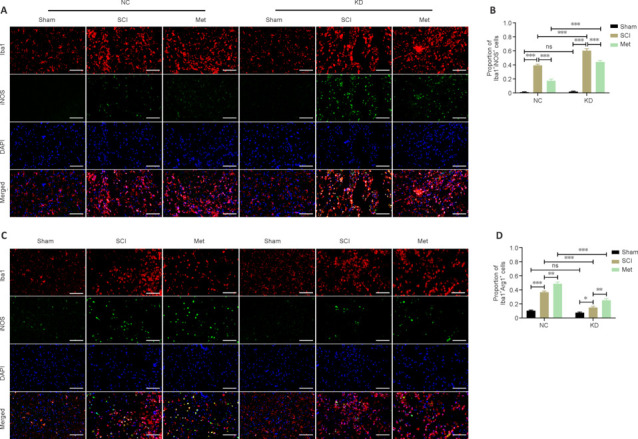
Met promotes the polarization of M1-type microglia/macrophages towards an M2 phenotype in an HMOX1-independent manner.
(A, B) Representative IF staining images and the proportion of iNOS+-stained microglia/macrophages in the indicated groups at 3 dpi. In both the NC and KD groups, the ratio of M1-type to M2-type microglia in the SCI group was increased compared with that in the sham group, and this effect was dramatically reversed by Met treatment. After HMOX1 knockdown, the ratio in the SCI and Met subgroups of the KD group was higher than that in the NC group (n = 5). Scale bars: 50 μm. (C, D) Representative IF staining images and the proportion of Arg1+-stained microglia/macrophages in the indicated groups at 3 dpi. In both the NC and KD groups, the ratio of M2-type to M1-type microglia in the SCI group was increased compared with that in the sham group, and this ratio was further increased by Met treatment. After HMOX1 knockdown, the ratio in the SCI and Met subgroups of the KD group was lower than that in the NC group (n = 5). Red indicates Iba1 (stained with Cy3), and green indicates iNOS or Arg1 (stained with Alexa Fluor 488). Scale bars: 50 μm. *P < 0.05, **P < 0.01, ***P < 0.001 (two-way analysis of variance with Tukey's post hoc test). Arg1: Arginase 1; dpi: day(s) post injury; IF: immunofluorescence; iNOS: inducible nitric oxide synthase; KD: knockdown; Met: metformin; NC: normal control; ns: not significant; SCI: spinal cord injury.
Finally, we examined the effect of HMOX1 knockdown on neuronal damage. As depicted in Figure 9, the proportion of FJB-positive neurons increased significantly following SCI in the NC and KD groups. In the NC group, Met treatment noticeably reduced neuronal damage, but this effect was not pronounced in the KD group. In the SCI and Met subgroup, HMOX1 knockdown led to a considerable increase in neuronal damage. These findings indicate that HMOX1 upregulation following SCI helps attenuate nerve damage, and that the effectiveness of Met treatment partly relies on HMOX1 upregulation.
Figure 9.
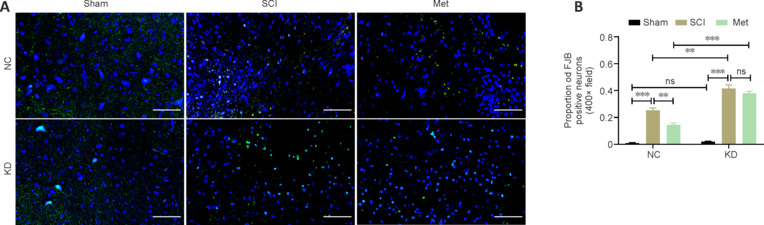
Met exerts a neuroprotective effect in an HMOX1-dependent manner.
(A, B) Representative FJB staining images and the proportion of FJB+ neurons at 3 dpi. In both the NC and KD groups, the ratio of FJB+ neurons to unstained neurons in the SCI group was increased compared with that in the sham group. This effect was dramatically reversed by Met treatment in the NC group but not in the KD group. After HMOX1 knockdown, the ratio in the SCI subgroup and the Met subgroup of the KD group was higher than that in the NC group (n = 5). Scale bars: 50 μm. **P < 0.01, ***P < 0.001 (two-way analysis of variance with Tukey's post hoc test). dpi: Day(s) post injury; FJB: Fluoro-Jade B; KD: knockdown; Met: metformin; NC: normal control; ns: not significant; SCI: spinal cord injury.
Discussion
In our previous study, we demonstrated that Met has anti-ferroptotic effects after SCI (Wang et al., 2022b). This neuroprotective effect was ascribed to the antioxidant mechanism mediated by the nuclear factor erythroid2-related factor 2 (Nrf2) signaling pathway. In further studies, we found that HMOX1, a downstream target gene of the Nrf2 signaling pathway, is stably expressed at high levels in the early and late period of SCI. HMOX1 is generally believed to play a protective role in various diseases (Puentes-Pardo et al., 2020; Ryter, 2021, 2022). However, it is considered to be harmful to tissues owing to its promotion of heme degradation, leading to excess iron production (Fang et al., 2019; Zeng et al., 2021). In the present study, we found that it plays anti-ferroptotic roles in TSCI than it does in other diseases. Direct mechanical trauma and subsequent lipid peroxidation, as well as inflammation, are the main drivers of nerve cell death (Hu et al., 2021; Dong and Yong, 2022). Here we found that cell death occurred primarily within 1 week after SCI, as confirmed by FJB staining. Mechanical trauma was also the main cause of erythrocyte rupture and iron deposition, which was significantly elevated 1 week after SCI. Therefore, there does not appear to be a significant relationship between the death of nerve cells in the early stage of injury and iron deposition. However, we cannot completely rule out the possibility that free iron does not contribute to nerve damage. Seven days after SCI, oxidative stress and inflammation had returned to normal levels. GPX4 expression levels also leveled off, and at this stage, high levels of HMOX1 seemed even less likely to contribute to neuronal cell ferroptosis.
Apoptosis has been considered to be the main form of cell death that occurs after SCI. Given that enhanced oxidative stress, lipid peroxide accumulation, and inflammation were detected in the post-injury microenvironment, it is likely that other types of cell death are also involved. The earliest report on a possible association between ferroptosis and TSCI was published in 2019 (Zhang et al., 2019), and the results from our current study support this association. However, the 2019 study showed that 4HNE expression remained elevated on the 14th day, and mitochondria with ferroptotic morphology were detected at multiple time points after. In contrast, we only detected high 4HNE expression levels 7 days after SCI, with a peak on day 3, which is basically consistent with the conclusions from several previous studies (Xiong et al., 2007; Carrico et al., 2009; Ge et al., 2021; Liu et al., 2022b). Unlike previous reports, we did not observe any mitochondria with typical ferroptotic features on days 1 and 3 after SCI. This was confirmed by morphological analysis showing no marked iron deposition in tissues within 24 hours, or indeed up to day 3, while significant iron deposition was observed on the 7th day after SCI. This differs from previous studies, which reported that significantly increased iron content can be detected at 1, 4, and 24 hours after SCI, presumably because of massive erythrocyte stasis induced by trauma in the acute stage of SCI. This discrepancy may be attributable to the use of different animal models and detection methods.
Inflammation in the microenvironment is an important factor associated with worse outcomes after SCI. Inflammatory cytokines have been reported to participate in and promote ferroptosis (Bin et al., 2021; Han et al., 2021; Yao et al., 2021). IL-1β can induce the accumulation of ROS and lipid ROS in chondrocytes and significantly alter the expression of ferroptosis-related proteins. Ferrostatin-1, a specific inhibitor of ferroptosis, can reduce IL-1β-induced cytotoxicity and the accumulation of ROS and lipid ROS, as well as modulate the expression of ferroptosis-related proteins and promote activation of the Nrf2 antioxidant system (Yao et al., 2021). Disc degeneration can cause low back pain, often accompanied by severe inflammation. Studies have shown that the inflammatory cytokine IL-6 can aggravate intervertebral disc degeneration by inducing chondrocyte ferroptosis (Bin et al., 2021). In addition, IL-6 has been implicated in the pathogenesis of asthma, owing to its ability to promote reactive oxygen-dependent lipid peroxidation and disrupt iron homeostasis, which promotes ferroptosis in bronchial epithelial cells (Han et al., 2021). In the present study, expression levels of the inflammatory factors IL-1β and IL-6 were significantly increased in the injured spinal cord on the 3rd day after SCI. Met treatment significantly reduced the overall levels of inflammatory factors, alleviated inflammation-induced damage to neurons, and inhibited ferroptosis in the injury microenvironment.
As mentioned earlier, HMOX1 plays dual roles in some diseases (Choi and Kim, 2022; Yang et al., 2022). Its overall role in ferroptosis after SCI is currently controversial. In the current study, we explored HMOX1 expression after SCI using different methods. We demonstrated that its expression is significantly increased over a long period of time after SCI. In addition, we found that Met treatment promoted motor function recovery in a rat model of SCI by inhibiting ferroptosis. The anti-ferroptotic effect of Met treatment was mediated by the increase in HMOX1 levels. In further tests, we knocked down HMOX1 expression in the spinal cord of rats. In our study, all rats received 10 μL LV by injection into the spinal cord at the T9 level after laminectomy. Once satisfactory outcomes were obtained, we divided the injected rats into two main groups and six subgroups, some of which were subjected to SCI and some of which were also treated with Met. On day 3 after SCI, spinal cord tissues were collected for western blotting and immunofluorescence analysis. It demonstrated that the anti-ferroptotic effect of Met is partly mediated by increased HMOX1 expression by western blotting analysis. It also implies that HMOX1 has a polarized anti-inflammatory effect in the early phase of SCI and promotes the conversion of M1-polarized Iba1+/iNOS+ microglia/macrophages to M2-polarized Iba1+/Arg1+ microglia/macrophages by immunofluorescence analysis. Moreover, our results suggest that the anti-inflammatory effect of Met was not reversed by HMOX1 knockdown. This demonstrates that, even when HMOX1 is knocked down, Met treatment effectively reduced the proportion of M1-polarized Iba1+/iNOS+ microglia/macrophages and increased the proportion of M2-polarized Iba1+/Arg1+ microglia/macrophages. Finally, we demonstrated that HMOX1 upregulation following SCI plays a vital role in combating nerve damage, and that the effectiveness of Met treatment is partially dependent on HMOX1 upregulation.
We did not screen for drug targets or for relevant signaling pathways. In addition, we did not perform behavioral experiments or explore the molecular mechanism underlying the effects of HMOX1 knockdown in vitro. Taken together, the findings from our study suggest that Met inhibits ferroptosis after SCI, and that this effect is partly dependent on high levels of HMOX1 expression. Met exerted an anti-inflammatory effect that mitigated further damage after SCI through HMOX1-independent pathways. In addition, high levels of HMOX1 expression played a neuroprotective role after SCI by reducing inflammation, alleviating oxidative stress, and inhibiting ferroptosis. Therefore, HMOX1 is a promising therapeutic target for inhibiting nerve cell ferroptosis after SCI.
Additional files:
Additional Table 1: The detailed information of all datasets.
Additional Table 2: Sequences and titer information of lentiviruses.
Additional Table 3: Primer sequences used in PCR analysis.
Additional Figure 1 (2.9MB, tif) : Hmox1 is significantly upregulated in SCI rat models.
Hmox1 is significantly upregulated in SCI rat models.
(A) Visualization of DEGs at 9 hpi in the GSE92657 dataset. (B-D) Visualization of DEGs at 1, 3, and 7 dpi in the GSE42828 dataset. (E) Visualization of DEGs at 9 dpi in the GSE93561 dataset. |logFC| > 1, adjusted P value < 0.05. DEGs: differentially expressed genes; dpi: day(s) post injury; Hmox1: heme oxygenase-1; hpi: hours post injury; SCI: spinal cord injury.
Additional Figure 2 (3.4MB, tif) : Lentivirus-mediated knockdown reduced HMOX1 expression both in vitro and in vivo.
Lentivirus-mediated knockdown reduced HMOX1 expression both in vitro and in vivo.
(A) Hmox1 mRNA expression levels in PC-12 cells after transfection (n = 6 independent experiments). (B, C) HMOX1 protein expression levels in PC-12 cells after transfection (n = 5 independent experiments). (D, E) HMOX1 protein expression levels in rats in the sham group 14 days after injection (n = 5). (F) Distribution of green fluorescent protein (GFP) expression illustrating the extent of lentiviral infection in frozen spinal cord sections 14 days after injection (n = 3). Green fluorescence indicates tissue transfected with LV. Scale bars: 3 mm. The data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001 (one-way analysis of variance with Bonferroni post hoc test (A, C) or independent-samples t-test (E)). HMOX1: heme oxygenase-1; KD: knockdown; NC: normal control; ns: not significant.
Additional file 1: Open peer review reports 1 and 2 (94.8KB, pdf) .
Footnotes
Conflicts of interest: The authors declare that they have no conflicts of interest.
Data availability statement: All data generated or analyzed during this study are included in this published article and its Additional files.
Open peer reviewers: George M. Smith, Temple University, USA; Omar Tawakol, Illinois Institute of Technology, USA.
C-Editor: Zhao M; S-Editors: Yu J, Li CH; L-Editors: Crow E, Song LP; T-Editor: Jia Y
References
- Adegeest CY, van Gent JAN, Stolwijk-Swüste JM, Post MWM, Vandertop WP, Öner FC, Peul WC, Wengel PVT. Influence of severity and level of injury on the occurrence of complications during the subacute and chronic stage of traumatic spinal cord injury: a systematic review. J Neurosurg Spine. 2022;36:632–652. doi: 10.3171/2021.7.SPINE21537. [DOI] [PubMed] [Google Scholar]
- Bach JR. Noninvasive respiratory management of high level spinal cord injury. J Spinal Cord Med. 2012;35:72–80. doi: 10.1179/2045772311Y.0000000051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bin S, Xin L, Lin Z, Jinhua Z, Rui G, Xiang Z. Targeting miR-10a-5p/IL-6R axis for reducing IL-6-induced cartilage cell ferroptosis. Exp Mol Pathol. 2021;118:104570. doi: 10.1016/j.yexmp.2020.104570. [DOI] [PubMed] [Google Scholar]
- Cai X, Hua S, Deng J, Du Z, Zhang D, Liu Z, Khan NU, Zhou M, Chen Z. Astaxanthin activated the Nrf2/HO-1 pathway to enhance autophagy and inhibit ferroptosis, ameliorating acetaminophen-induced liver injury. ACS Appl Mater Interfaces. 2022;14:42887–42903. doi: 10.1021/acsami.2c10506. [DOI] [PubMed] [Google Scholar]
- Campbell NK, Fitzgerald HK, Dunne A. Regulation of inflammation by the antioxidant haem oxygenase 1. Nat Rev Immunol. 2021;21:411–425. doi: 10.1038/s41577-020-00491-x. [DOI] [PubMed] [Google Scholar]
- Cao G, Gong T, Du Y, Wang Y, Ge T, Liu J. Mechanism of metformin regulation in central nervous system: Progression and future perspectives. Biomed Pharmacother. 2022;156:113686. doi: 10.1016/j.biopha.2022.113686. [DOI] [PubMed] [Google Scholar]
- Carrico KM, Vaishnav R, Hall ED. Temporal and spatial dynamics of peroxynitrite-induced oxidative damage after spinal cord contusion injury. J Neurotrauma. 2009;26:1369–1378. doi: 10.1089/neu.2008-0870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Q, Xie D, Yao Q, Yang L. Effect of metformin on locomotor function recovery in rat spinal cord injury model: a meta-analysis. Oxid Med Cell Longev. 2021a;2021:1948003. doi: 10.1155/2021/1948003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Li J, Kang R, Klionsky DJ, Tang D. Ferroptosis: machinery and regulation. Autophagy. 2021b;17:2054–2081. doi: 10.1080/15548627.2020.1810918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen YX, Zuliyaer T, Liu B, Guo S, Yang DG, Gao F, Yu Y, Yang ML, Du LJ, Li JJ. Sodium selenite promotes neurological function recovery after spinal cord injury by inhibiting ferroptosis. Neural Regen Res. 2022;17:2702–2709. doi: 10.4103/1673-5374.339491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi YK, Kim YM. Beneficial and detrimental roles of heme oxygenase-1 in the neurovascular system. Int J Mol Sci. 2022;23:7041. doi: 10.3390/ijms23137041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong H, Zhang C, Shi D, Xiao X, Chen X, Zeng Y, Li X, Xie R. Ferroptosis related genes participate in the pathogenesis of spinal cord injury via HIF-1 signaling pathway. Brain Res Bull. 2023;192:192–202. doi: 10.1016/j.brainresbull.2022.11.010. [DOI] [PubMed] [Google Scholar]
- Dong Y, Yong VW. Oxidized phospholipids as novel mediators of neurodegeneration. Trends Neurosci. 2022;45:419–429. doi: 10.1016/j.tins.2022.03.002. [DOI] [PubMed] [Google Scholar]
- Du MR, Gao QY, Liu CL, Bai LY, Li T, Wei FL. Exploring the pharmacological potential of metformin for neurodegenerative diseases. Front Aging Neurosci. 2022;14:838173. doi: 10.3389/fnagi.2022.838173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dziedzic A, Saluk-Bijak J, Miller E, Bijak M. Metformin as a potential agent in the treatment of multiple sclerosis. Int J Mol Sci. 2020;21:5957. doi: 10.3390/ijms21175957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang X, Wang H, Han D, Xie E, Yang X, Wei J, Gu S, Gao F, Zhu N, Yin X, Cheng Q, Zhang P, Dai W, Chen J, Yang F, Yang HT, Linkermann A, Gu W, Min J, Wang F. Ferroptosis as a target for protection against cardiomyopathy. Proc Natl Acad Sci U S A. 2019;116:2672–2680. doi: 10.1073/pnas.1821022116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernández-Mendívil C, Luengo E, Trigo-Alonso P, García-Magro N, Negredo P, López MG. Protective role of microglial HO-1 blockade in aging: Implication of iron metabolism. Redox Biol. 2021;38:101789. doi: 10.1016/j.redox.2020.101789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge H, Xue X, Xian J, Yuan L, Wang L, Zou Y, Zhong J, Jiang Z, Shi J, Chen T, Su H, Feng H, Hu S. Ferrostatin-1 alleviates white matter injury via decreasing ferroptosis following spinal cord injury. Mol Neurobiol. 2022;59:161–176. doi: 10.1007/s12035-021-02571-y. [DOI] [PubMed] [Google Scholar]
- Ge MH, Tian H, Mao L, Li DY, Lin JQ, Hu HS, Huang SC, Zhang CJ, Mei XF. Zinc attenuates ferroptosis and promotes functional recovery in contusion spinal cord injury by activating Nrf2/GPX4 defense pathway. CNS Neurosci Ther. 2021;27:1023–1040. doi: 10.1111/cns.13657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han F, Li S, Yang Y, Bai Z. Interleukin-6 promotes ferroptosis in bronchial epithelial cells by inducing reactive oxygen species-dependent lipid peroxidation and disrupting iron homeostasis. Bioengineered. 2021;12:5279–5288. doi: 10.1080/21655979.2021.1964158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X, Xu Y, Xu H, Jin C, Zhang H, Su H, Li Y, Zhou K, Ni W. Progress in understanding ferroptosis and its targeting for therapeutic benefits in traumatic brain and spinal cord injuries. Front Cell Dev Biol. 2021;9:705786. doi: 10.3389/fcell.2021.705786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanehisa M, Goto S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang Y, Li Q, Zhu R, Li S, Xu X, Shi X, Yin Z. Identification of ferroptotic genes in spinal cord injury at different time points: bioinformatics and experimental validation. Mol Neurobiol. 2022;59:5766–5784. doi: 10.1007/s12035-022-02935-y. [DOI] [PubMed] [Google Scholar]
- Kanno H, Ozawa H, Dohi Y, Sekiguchi A, Igarashi K, Itoi E. Genetic ablation of transcription repressor Bach1 reduces neural tissue damage and improves locomotor function after spinal cord injury in mice. J Neurotrauma. 2009;26:31–39. doi: 10.1089/neu.2008.0667. [DOI] [PubMed] [Google Scholar]
- Lee H, Zandkarimi F, Zhang Y, Meena JK, Kim J, Zhuang L, Tyagi S, Ma L, Westbrook TF, Steinberg GR, Nakada D, Stockwell BR, Gan B. Energy-stress-mediated AMPK activation inhibits ferroptosis. Nat Cell Biol. 2020;22:225–234. doi: 10.1038/s41556-020-0461-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leech T, Chattipakorn N, Chattipakorn SC. The beneficial roles of metformin on the brain with cerebral ischaemia/reperfusion injury. Pharmacol Res. 2019;146:104261. doi: 10.1016/j.phrs.2019.104261. [DOI] [PubMed] [Google Scholar]
- Li F, Wang H, Chen H, Guo J, Dang X, Ru Y, Wang H. Mechanism of ferroptosis and its role in spinal cord injury. Front Neurol. 2022;13:926780. doi: 10.3389/fneur.2022.926780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li JZ, Fan BY, Sun T, Wang XX, Li JJ, Zhang JP, Gu GJ, Shen WY, Liu DR, Wei ZJ, Feng SQ. Bioinformatics analysis of ferroptosis in spinal cord injury. Neural Regen Res. 2023;18:626–633. doi: 10.4103/1673-5374.350209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q, Han X, Lan X, Gao Y, Wan J, Durham F, Cheng T, Yang J, Wang Z, Jiang C, Ying M, Koehler RC, Stockwell BR, Wang J. Inhibition of neuronal ferroptosis protects hemorrhagic brain. JCI Insight. 2017;2:e90777. doi: 10.1172/jci.insight.90777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Wang Q, Peng S, Yao K, Chen J, Tao Y, Gao Z, Wang F, Li H, Cai W, Lai Y, Li K, Chen X, Huang H. The metastatic promoter DEPDC1B induces epithelial-mesenchymal transition and promotes prostate cancer cell proliferation via Rac1-PAK1 signaling. Clin Transl Med. 2020;10:e191. doi: 10.1002/ctm2.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin W, Wang S, Yang Z, Lin J, Ke Q, Lan W, Shi J, Wu S, Cai B. Heme oxygenase-1 inhibits neuronal apoptosis in spinal cord injury through down-regulation of Cdc42-MLK3-MKK7-JNK3 axis. J Neurotrauma. 2017;34:695–706. doi: 10.1089/neu.2016.4608. [DOI] [PubMed] [Google Scholar]
- Lin W, Chen W, Liu K, Ma P, Qiu P, Zheng C, Zhang X, Tan P, Xi X, He X. Mitigation of microglia-mediated acute neuroinflammation and tissue damage by heme oxygenase 1 in a rat spinal cord injury model. Neuroscience. 2021;457:27–40. doi: 10.1016/j.neuroscience.2020.08.001. [DOI] [PubMed] [Google Scholar]
- Liu J, Kang R, Tang D. Signaling pathways and defense mechanisms of ferroptosis. FEBS J. 2022a;289:7038–7050. doi: 10.1111/febs.16059. [DOI] [PubMed] [Google Scholar]
- Liu NK, Deng LX, Wang M, Lu QB, Wang C, Wu X, Wu W, Wang Y, Qu W, Han Q, Xia Y, Ravenscraft B, Li JL, You SW, Wipf P, Han X, Xu XM. Restoring mitochondrial cardiolipin homeostasis reduces cell death and promotes recovery after spinal cord injury. Cell Death Dis. 2022b;13:1058. doi: 10.1038/s41419-022-05369-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma H, Wang X, Zhang W, Li H, Zhao W, Sun J, Yang M. Melatonin suppresses ferroptosis induced by high glucose via activation of the Nrf2/HO-1 signaling pathway in type 2 diabetic osteoporosis. Oxid Med Cell Longev. 2020;2020:9067610. doi: 10.1155/2020/9067610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma WQ, Sun XJ, Zhu Y, Liu NF. Metformin attenuates hyperlipidaemia-associated vascular calcification through anti-ferroptotic effects. Free Radic Biol Med. 2021;165:229–242. doi: 10.1016/j.freeradbiomed.2021.01.033. [DOI] [PubMed] [Google Scholar]
- Ma Y, Wang J, Wang Y, Yang GY. The biphasic function of microglia in ischemic stroke. Prog Neurobiol. 2017;157:247–272. doi: 10.1016/j.pneurobio.2016.01.005. [DOI] [PubMed] [Google Scholar]
- Meng H, Wu J, Shen L, Chen G, Jin L, Yan M, Wan H, He Y. Microwave assisted extraction, characterization of a polysaccharide from Salvia miltiorrhiza Bunge and its antioxidant effects via ferroptosis-mediated activation of the Nrf2/HO-1 pathway. Int J Biol Macromol. 2022;215:398–412. doi: 10.1016/j.ijbiomac.2022.06.064. [DOI] [PubMed] [Google Scholar]
- Ning P, Luo A, Mu X, Xu Y, Li T. Exploring the dual character of metformin in Alzheimer's disease. Neuropharmacology. 2022;207:108966. doi: 10.1016/j.neuropharm.2022.108966. [DOI] [PubMed] [Google Scholar]
- Patel F, Parwani K, Rao P, Patel D, Rawal R, Mandal P. Prophylactic treatment of probiotic and metformin mitigates ethanol-induced intestinal barrier injury: in vitro, in vivo, and in silico approaches. Mediators Inflamm. 2021;2021:5245197. doi: 10.1155/2021/5245197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puentes-Pardo JD, Moreno-SanJuan S, Carazo Á, León J. Heme oxygenase-1 in gastrointestinal tract health and disease. Antioxidants (Basel) 2020;9:1214. doi: 10.3390/antiox9121214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryter SW. Heme oxgenase-1, a cardinal modulator of regulated cell death and inflammation. Cells. 2021;10:515. doi: 10.3390/cells10030515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryter SW. Heme oxygenase-1: an anti-inflammatory effector in cardiovascular, lung, and related metabolic disorders. Antioxidants (Basel) 2022;11:555. doi: 10.3390/antiox11030555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauvé M, Ban K, Momen MA, Zhou YQ, Henkelman RM, Husain M, Drucker DJ. Genetic deletion or pharmacological inhibition of dipeptidyl peptidase-4 improves cardiovascular outcomes after myocardial infarction in mice. Diabetes. 2010;59:1063–1073. doi: 10.2337/db09-0955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9:671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Z, Yuan S, Shi L, Li J, Ning G, Kong X, Feng S. Programmed cell death in spinal cord injury pathogenesis and therapy. Cell Prolif. 2021;54:e12992. doi: 10.1111/cpr.12992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascón S, Hatzios SK, Kagan VE, Noel K, Jiang X, Linkermann A, Murphy ME, Overholtzer M, Oyagi A, Pagnussat GC, Park J, Ran Q, Rosenfeld CS, et al. Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell. 2017;171:273–285. doi: 10.1016/j.cell.2017.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork P, Jensen LJ, Mering CV. STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019;47:D607–D613. doi: 10.1093/nar/gky1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas PD, Ebert D, Muruganujan A, Mushayahama T, Albou LP, Mi H. PANTHER: making genome-scale phylogenetics accessible to all. Protein Sci. 2022;31:8–22. doi: 10.1002/pro.4218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vargas MR, Pehar M, Cassina P, Martínez-Palma L, Thompson JA, Beckman JS, Barbeito L. Fibroblast growth factor-1 induces heme oxygenase-1 via nuclear factor erythroid 2-related factor 2 (Nrf2) in spinal cord astrocytes: consequences for motor neuron survival. J Biol Chem. 2005;280:25571–25579. doi: 10.1074/jbc.M501920200. [DOI] [PubMed] [Google Scholar]
- Wang H, Xiang Q, Li C, Zhou Y. Epidemiology of traumatic cervical spinal fractures and risk factors for traumatic cervical spinal cord injury in China. J Spinal Disord Tech. 2013;26:E306–313. doi: 10.1097/BSD.0b013e3182886db9. [DOI] [PubMed] [Google Scholar]
- Wang S, Zhang T, Yang Z, Lin J, Cai B, Ke Q, Lan W, Shi J, Wu S, Lin W. Heme oxygenase-1 protects spinal cord neurons from hydrogen peroxide-induced apoptosis via suppression of Cdc42/MLK3/MKK7/JNK3 signaling. Apoptosis. 2017;22:449–462. doi: 10.1007/s10495-016-1329-z. [DOI] [PubMed] [Google Scholar]
- Wang Z, Zhou W, Li M. Epidemiological characteristics of 1,806 patients with traumatic spinal cord injury: A retrospective study. Front Surg. 2022a;9:988853. doi: 10.3389/fsurg.2022.988853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z, Wu Z, Xie Z, Zhou W, Li M. Metformin attenuates ferroptosis and promotes functional recovery of spinal cord injury. World Neurosurg. 2022b;167:e929–e939. doi: 10.1016/j.wneu.2022.08.121. [DOI] [PubMed] [Google Scholar]
- Wei X, Yi X, Zhu XH, Jiang DS. Posttranslational modifications in ferroptosis. Oxid Med Cell Longev. 2020;2020:8832043. doi: 10.1155/2020/8832043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu W, Wang S, Liu Q, Shan T, Wang Y. Metformin protects against LPS-induced intestinal barrier dysfunction by activating AMPK pathway. Mol Pharm. 2018;15:3272–3284. doi: 10.1021/acs.molpharmaceut.8b00332. [DOI] [PubMed] [Google Scholar]
- Xie BS, Wang YQ, Lin Y, Mao Q, Feng JF, Gao GY, Jiang JY. Inhibition of ferroptosis attenuates tissue damage and improves long-term outcomes after traumatic brain injury in mice. CNS Neurosci Ther. 2019;25:465–475. doi: 10.1111/cns.13069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie Y, Wang Y, Zhou Y, Liu M, Li S, Bao Y, Jiang W, Tang S, Li F, Xue H, Li L, Gong X, Liu Y, Wang W, Li T. A nomogram for predicting acute respiratory failure after cervical traumatic spinal cord injury based on admission clinical findings. Neurocrit Care. 2022;36:421–433. doi: 10.1007/s12028-021-01302-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong Y, Rabchevsky AG, Hall ED. Role of peroxynitrite in secondary oxidative damage after spinal cord injury. J Neurochem. 2007;100:639–649. doi: 10.1111/j.1471-4159.2006.04312.x. [DOI] [PubMed] [Google Scholar]
- Yamauchi T, Lin Y, Sharp FR, Noble-Haeusslein LJ. Hemin induces heme oxygenase-1 in spinal cord vasculature and attenuates barrier disruption and neutrophil infiltration in the injured murine spinal cord. J Neurotrauma. 2004;21:1017–1030. doi: 10.1089/0897715041651042. [DOI] [PubMed] [Google Scholar]
- Yan J, Feng G, Ma L, Chen Z, Jin Q. Metformin alleviates osteoarthritis in mice by inhibiting chondrocyte ferroptosis and improving subchondral osteosclerosis and angiogenesis. J Orthop Surg Res. 2022;17:333. doi: 10.1186/s13018-022-03225-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Y, Jun C, Lu Y, Jiangmei S. Combination of metformin and luteolin synergistically protects carbon tetrachloride-induced hepatotoxicity: Mechanism involves antioxidant, anti-inflammatory, antiapoptotic, and Nrf2/HO-1 signaling pathway. Biofactors. 2019;45:598–606. doi: 10.1002/biof.1521. [DOI] [PubMed] [Google Scholar]
- Yang S, Ouyang J, Lu Y, Harypursat V, Chen Y. A dual role of heme oxygenase-1 in tuberculosis. Front Immunol. 2022;13:842858. doi: 10.3389/fimmu.2022.842858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao X, Sun K, Yu S, Luo J, Guo J, Lin J, Wang G, Guo Z, Ye Y, Guo F. Chondrocyte ferroptosis contribute to the progression of osteoarthritis. J Orthop Translat. 2021;27:33–43. doi: 10.1016/j.jot.2020.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ying Y, Huang Z, Tu Y, Wu Q, Li Z, Zhang Y, Yu H, Zeng A, Huang H, Ye J, Ying W, Chen M, Feng Z, Xiang Z, Ye Q, Zhu S, Wang Z. A shear-thinning, ROS-scavenging hydrogel combined with dental pulp stem cells promotes spinal cord repair by inhibiting ferroptosis. Bioact Mater. 2023;22:274–290. doi: 10.1016/j.bioactmat.2022.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu WK, Chen YC, Chen WC, Yi-Fong Su V, Yang KY, Kou YR. Influencing factors for tracheostomy in patients with acute traumatic C3-C5 spinal cord injury and acute respiratory failure. J Chin Med Assoc. 2022;85:167–174. doi: 10.1097/JCMA.0000000000000656. [DOI] [PubMed] [Google Scholar]
- Zeng L, Zhou J, Wang X, Zhang Y, Wang M, Su P. Cadmium attenuates testosterone synthesis by promoting ferroptosis and blocking autophagosome-lysosome fusion. Free Radic Biol Med. 2021;176:176–188. doi: 10.1016/j.freeradbiomed.2021.09.028. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Sun C, Zhao C, Hao J, Zhang Y, Fan B, Li B, Duan H, Liu C, Kong X, Wu P, Yao X, Feng S. Ferroptosis inhibitor SRS 16-86 attenuates ferroptosis and promotes functional recovery in contusion spinal cord injury. Brain Res. 20191706:48–57. doi: 10.1016/j.brainres.2018.10.023. [DOI] [PubMed] [Google Scholar]
- Zhao JY, Sheng XL, Li CJ, Qin T, He RD, Dai GY, Cao Y, Lu HB, Duan CY, Hu JZ. Metformin promotes angiogenesis and functional recovery in aged mice after spinal cord injury by adenosine monophosphate-activated protein kinase/endothelial nitric oxide synthase pathway. Neural Regen Res. 2023;18:1553–1562. doi: 10.4103/1673-5374.360245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou H, Yin C, Zhang Z, Tang H, Shen W, Zha X, Gao M, Sun J, Xu X, Chen Q. Proanthocyanidin promotes functional recovery of spinal cord injury via inhibiting ferroptosis. J Chem Neuroanat. 2020;107:101807. doi: 10.1016/j.jchemneu.2020.101807. [DOI] [PubMed] [Google Scholar]
- Zhou N, Yuan X, Du Q, Zhang Z, Shi X, Bao J, Ning Y, Peng L. FerrDb V2: update of the manually curated database of ferroptosis regulators and ferroptosis-disease associations. Nucleic Acids Res. 2023;51:D571–582. doi: 10.1093/nar/gkac935. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Hmox1 is significantly upregulated in SCI rat models.
(A) Visualization of DEGs at 9 hpi in the GSE92657 dataset. (B-D) Visualization of DEGs at 1, 3, and 7 dpi in the GSE42828 dataset. (E) Visualization of DEGs at 9 dpi in the GSE93561 dataset. |logFC| > 1, adjusted P value < 0.05. DEGs: differentially expressed genes; dpi: day(s) post injury; Hmox1: heme oxygenase-1; hpi: hours post injury; SCI: spinal cord injury.
Lentivirus-mediated knockdown reduced HMOX1 expression both in vitro and in vivo.
(A) Hmox1 mRNA expression levels in PC-12 cells after transfection (n = 6 independent experiments). (B, C) HMOX1 protein expression levels in PC-12 cells after transfection (n = 5 independent experiments). (D, E) HMOX1 protein expression levels in rats in the sham group 14 days after injection (n = 5). (F) Distribution of green fluorescent protein (GFP) expression illustrating the extent of lentiviral infection in frozen spinal cord sections 14 days after injection (n = 3). Green fluorescence indicates tissue transfected with LV. Scale bars: 3 mm. The data are expressed as the mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001 (one-way analysis of variance with Bonferroni post hoc test (A, C) or independent-samples t-test (E)). HMOX1: heme oxygenase-1; KD: knockdown; NC: normal control; ns: not significant.


