Abstract
Previous studies have demonstrated a bidirectional relationship between inflammation and depression. Activation of the nucleotide-binding oligomerization domain, leucine-rich repeat, and NLR family pyrin domain-containing 3 (NLRP3) inflammasomes is closely related to the pathogenesis of various neurological diseases. In patients with major depressive disorder, NLRP3 inflammasome levels are significantly elevated. Understanding the role that NLRP3 inflammasome-mediated neuroinflammation plays in the pathogenesis of depression may be beneficial for future therapeutic strategies. In this review, we aimed to elucidate the mechanisms that lead to the activation of the NLRP3 inflammasome in depression as well as to provide insight into therapeutic strategies that target the NLRP3 inflammasome. Moreover, we outlined various therapeutic strategies that target the NLRP3 inflammasome, including NLRP3 inflammatory pathway inhibitors, natural compounds, and other therapeutic compounds that have been shown to be effective in treating depression. Additionally, we summarized the application of NLRP3 inflammasome inhibitors in clinical trials related to depression. Currently, there is a scarcity of clinical trials dedicated to investigating the applications of NLRP3 inflammasome inhibitors in depression treatment. The modulation of NLRP3 inflammasomes in microglia holds promise for the management of depression. Further investigations are necessary to ascertain the efficacy and safety of these therapeutic approaches as potential novel antidepressant treatments.
Keywords: depression, microglia, neuroinflammation, NLRP3 inflammasome
Introduction
Depression is the most prevalent mental disorder globally, which affects thoughts, mood, and behavior. It can cause persistent sadness, loss of interest, sleep disturbances, loss of appetite, fatigue, difficulty concentrating, feelings of worthlessness or guilt, and thoughts of suicide or self-harm. Depression can also cause physical symptoms such as headaches and stomache aches. Moreover, depression can present a significant social burden, as it can affect a person's ability to function at work, perform daily tasks, and maintain healthy relationships, leading to social isolation, withdrawal, and decreased productivity, which lead to increased healthcare costs and decreased quality of life (Ferrari et al., 2013). In light of the high prevalence and serious consequences of depression, effective diagnostic and treatments are urgently needed (McCarron et al., 2021). In clinical practice, there are two main types of antidepressants that are currently available: selective serotonin uptake inhibitors and tricyclics. These drugs, however, have some disadvantages, such as adverse effects, risk of dependency, variable treatment responses, safety concerns, tolerability issues, withdrawal symptoms, and therapeutic lag (Agius and Bonnici, 2017; Rakesh et al., 2017; Moncrieff, 2019). Therefore, it is essential to explore and develop new targets and effective therapies for depression.
There is a strong connection between depression and elevated levels of inflammation. Chronic inflammation is defined as a persistent state of inflammation that lasts for an extended period of time. This sustained inflammatory response has been implicated in the pathogenesis and progression of depression, and depression has also been associated with increased levels of inflammation (Beurel et al., 2020). Studies have found that individuals with depression often show increased blood levels of tumor necrosis factor-alpha (TNF-α), interleukin (IL)-6, and C-reactive protein (CRP) (Dantzer et al., 2008; Felger and Treadway, 2017). Conversely, people with chronic inflammation, such as those with chronic infections or autoimmune diseases, are more likely to develop depression and anxiety symptoms (Rosenblat et al., 2014; Brites and Fernandes, 2015). People living with human immunodeficiency virus infection have an elevated susceptibility to depression, demonstrating a link between depression and a communicable inflammatory condition. In a study examining biomarkers, monokine release induced by interferon-γ and TNF-α in blood plasma as well as macrophage inflammatory protein 1-alpha and IL-6 in cerebrospinal fluid were identified as potential mediators in the association between human immunodeficiency virus infection and depression symptoms (Mudra Rakshasa-Loots et al., 2023). Similarly, in non-communicable inflammatory conditions such as asthma bronchiale, a bidirectional relationship with depression has been observed. Imbalances in cytokine levels, including TNF-α, IL-6, and IL-4, have been implicated in the onset and remission of both asthma and depression (Jiang et al., 2014). These studies suggested that there is a bidirectional relationship between inflammation and depression.
The role of CRP in depression remains a topic of debate and conflicting findings. While some studies have reported a positive correlation between elevated CRP levels and the severity of depression symptoms, as well as poorer treatment responses (Howren et al., 2009; Haapakoski et al., 2015; Pitharouli et al., 2021; Orsolini et al., 2022), others have failed to find any significant association between CRP levels in people with depression compared with healthy controls (Jeenger et al., 2018; Shahyad et al., 2023). A recent study investigating high-sensitivity CRP test levels in patients with major depressive disorder (MDD), bipolar disorder in a depressive state, and healthy individuals found no significant associations (Shahyad et al., 2023). It is worth noting that various factors, such as age, body mass index, low-grade inflammation, and resting heart rate, as indicators of physical fitness, may influence serum CRP levels (Ma et al., 2010). Consequently, further research is needed to elucidate the exact role of CRP in MDD.
During neuroinflammation, microglia, the resident immune cells of the central nervous system (CNS), including the brain and spinal cord, are involved (Wies Mancini et al., 2023). Neuronal damage and dysfunction can be caused by activated microglia, which produce nitric oxide, reactive oxygen species (ROS), and proinflammatory cytokines. Research has demonstrated that microglia are crucial to depression etiology (Deng et al., 2020; Jia et al., 2021). Positron emission tomography (PET) studies have shown evidence of microglial activation in depression-related brain regions (Setiawan et al., 2015). Many studies have indicated that neuroinflammation mediated by microglia contributes to the pathophysiology of depression (Crupi and Cuzzocrea, 2016; Jeon and Kim, 2016; Benedetti et al., 2020; Troubat et al., 2021), paving the way for developing novel therapeutic strategies for this debilitating disorder.
Inflammasomes are intracellular protein complexes that are critical regulators of inflammatory responses in the brain and other tissues. The inflammasome is an essential component of neuroinflammation. Inflammasomes are found in neurons, microglia, and astrocytes in the brain and respond to pathogen-associated molecular patterns (PAMPs) and danger-associated molecular patterns (DAMPs). Inflammasomes comprise the pattern recognition receptors, precursor procaspase-1, and adaptor apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) protein. Inflammasome activation results in caspase-1 maturation and the production of IL-18 and IL-1β (Leemans et al., 2011). There are several classes of inflammasomes in brain cells. The most well-known are nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing 3 (NLRP3), also called nucleotide-binding oligomerization domain-, leucine-rich repeat-, and pyrin domain-containing protein 3 (NLRP3), pyrin, and absent in melanoma 2. Among the various inflammasomes, NLRP3 has been best characterized in brain cells.
Multiple neurological disorders are associated with NLRP3 inflammasome activation in microglia (Feng et al., 2021; Ravichandran and Heneka, 2021; Cui et al., 2023; Liu et al., 2023; Possemato et al., 2023), including Parkinson's disease, Alzheimer's disease, traumatic brain injury, and multiple sclerosis. In Alzheimer's disease, microglial NLRP3 inflammasome activation may contribute to the release of proinflammatory cytokines, leading to neuroinflammation and neuronal damage. Microglial NLRP3 inflammasome activation is a key mediator of amyloid β-induced neurotoxicity in Alzheimer's disease (Liang et al., 2022; Jha et al., 2023). Likewise, in Parkinson's disease, microglial NLRP3 inflammasome activation may promote the release of proinflammatory cytokines, contributing to dopaminergic neuronal loss and neuroinflammation. NLRP3 inflammasome activation in microglia exacerbates neuronal inflammation-induced cell death and neurodegeneration in Parkinson's disease (Han and Le, 2023; Soraci et al., 2023). In multiple sclerosis, microglial NLRP3 inflammasome activation may contribute to demyelination and axonal damage by promoting the release of proinflammatory cytokines in the CNS (Cui et al., 2022; Malhotra et al., 2023). Furthermore, microglial NLRP3 inflammasome activation was observed in the acute phase of traumatic brain injury, which triggers neuroinflammation and the production of proinflammatory cytokines, leading to progressive neuronal damage and cognitive decline (Chakraborty et al., 2023). These findings revealed that NLRP3 inflammasome activation is closely related to the pathogenesis of various neurological disorders. Developing selective inhibitors of NLRP3 inflammasomes may represent an effective therapeutic approach for treating these CNS diseases.
Evidence has demonstrated that the blood of individuals diagnosed with MDD exhibited significant increases in mRNA levels of NLRP3 and caspase-1 genes compared with those in healthy controls (Taene et al., 2020). Thus, the NLRP3 inflammasome may also contribute to the etiology of depression (Alcocer-Gómez and Cordero, 2014). Depression is a complex neuropsychiatric disorder that involves dysregulation of neurobiological pathways, including immune and inflammatory responses. Although accumulating evidence has suggested pathogenic effects of the NLRP3 inflammasome in many brain disorders, its relationship with depression remains not well understood. Therefore, understanding the mechanisms underpinning NLRP3 inflammasome-mediated neuroinflammation in depression is essential for developing future therapeutic strategies.
Further investigations are needed to elucidate the dynamic interplay between NLRP3 inflammasome-mediated neuroinflammation and depression and identify potential therapeutic targets for improving depression therapy. This review aimed to elucidate the mechanisms that lead to activation of NLRP3 inflammasomes in depression as well as to provide insight into therapeutic strategies that target NLRP3 inflammasomes. Furthermore, we outlined various therapeutic strategies, including NLRP3 inflammatory pathway inhibitors, natural compounds, other therapeutic compounds, and non-drug therapies, which have been shown to be effective in treating depression. These approaches offer potential avenues for the development of effective treatments for depression by interfering with the NLRP3 inflammasome-mediated inflammatory response. In addition, the topic of NLRP3 inflammasome inhibitors for depression in clinical trials is also addressed.
Search Strategy
The search strategy utilized the PubMed database to retrieve articles related to the topic. The search was conducted using the following terms: “NLRP3”and “depression” or “depressive” in titles and abstracts. The search was limited to articles published between 2010 and 2023, including research papers, review articles, clinical trials, and meta-analyses. A total of 353 results were obtained. The search focused primarily on articles published within the past 5 years. The search results were further screened for relevance to the topic. Non-relevant articles were excluded from the analysis.
NLRP3 Inflammasome Formation and Activation
Inflammasomes are composed of ASC, pattern recognition receptors, and procaspase-1. Intracellular pattern recognition receptors can be divided into three types: tripartite motif-containing protein pyrin/tripartite motif-containing 20, absent in melanoma-like receptors, and nucleotide-binding domain and leucine-rich repeat-containing receptors (NLRs). They detect cytosolic DAMPs and PAMPs as immunological sensors (Dubois et al., 2016). The caspase recruitment domain of procaspase-1 connects to the pyrin domain of NLRs, melanoma-like receptors, or pyrins through the ASC (Voet et al., 2019). There are a number of NLR proteins in the NLR family, including NLRP3, NLR family caspase recruitment domain-containing 4, NLRP1, NLRP2, NLRP6, NLRP7, and NLRP12 (Guan and Han, 2020). A multimeric protein complex called the NLRP3 inflammasome is formed when NLRP3 is activated, which regulates innate immunity. There are many stimuli that activate the NLRP3 inflammasome, including PAMPs, DAMPs, and environmental toxins (Elliott and Sutterwala, 2015; Jo et al., 2016). While the activation of the NLRP3 inflammasome plays a key role in host defense, the sustained or dysregulated activation of this complex can lead to inflammation and tissue damage (Elliott and Sutterwala, 2015; Jo et al., 2016). The pathogenesis of various inflammatory and autoimmune disorders is associated with this, including depression.
There are two distinct signaling pathways that result in NLRP3 inflammasome activation: the canonical and noncanonical pathways (Xiang et al., 2020). Both canonical and noncanonical pathways play a pivotal role in the NLRP3 inflammasome activation and the subsequent regulation of inflammation and immune responses. Gram-negative bacteria or lipopolysaccharides (LPS) activate the noncanonical signal pathway, mainly by activating caspase-11, which is mediated by caspase-4, caspase-5, and caspase-8, while caspase-1 is a key player in the canonical signaling pathway (Lamkanfi and Dixit, 2012, 2014). There are two steps to the canonical signaling pathway: priming and protein complex assembly (Paik et al., 2021). Signals from TNF receptors and Toll-like receptors (TLRs) activate the priming process. The nuclear factor-kappa B (NF-κB)-dependent pathway is then activated and promotes the transcription and expression of NLRP3 as well as pro-IL-18 and pro-IL-1β (McKee and Coll, 2020). The protein complex assembly process is stimulated by DAMPs and PAMPs, such as extracellular and cellular events. Extracellular events include the release of pore-forming toxins and adenosine triphosphate (ATP). Cellular events include ion flux (Ca2+ influx and K+/Cl– efflux), the production of ROS, cathepsin B from lysosomal damage, and mitochondrial impairment (Cullen et al., 2015; Kelley et al., 2019; Swanson et al., 2019). Afterward, the inflammasome complex leads to the maturation of caspase-1 and then cleaves the proinflammatory cytokines IL-1β and IL-18 into mature molecules. Mature caspase-1 also converts the pyroptotic substrate gasdermin-D (GSDMD) into its C-terminus and N-terminus (N-GSDMD) (Bai et al., 2020; Rashidi et al., 2020). Finally, the N-GSDMD oligomerizes to form the plasma membrane pores, then promotes the release of mature IL-18 and IL-1β and mediates the programmed cell death known as pyroptosis. The sustained and dysregulated NLRP3 inflammasome activation and subsequent pyroptosis lead to inflammation and tissue damage.
Neuroinflammation Mediated by NLRP3 Inflammasomes in Depression Pathogenesis
A clinical study found elevated caspase-1 and NLRP3 gene expression and elevated IL-18 and IL-1β levels in the blood of non-treated patients with MDD. Meanwhile, the tricyclic antidepressant amitriptyline reduced these alterations (Alcocer-Gómez et al., 2014). According to studies using animal models, chronic unpredictable mild stress (CUMS) obviously elevated IL-1β levels and induced the activation of the prefrontal cortex NLRP3 inflammasome of depressed rats (Pan et al., 2014). The elevated NLRP3 protein was co-located with ionized calcium-binding adaptor molecule 1, which suggested the microglial NLRP3 inflammasome activation contributed to the depressive-like behavior of rats (Pan et al., 2014). Both preclinical and clinical evidence have shown increased NLRP3 complex assembly and secretion of IL-18 and IL-1β in rodent models and patients with MDD (Kaufmann et al., 2017). Thus, NLRP3-mediated neuroinflammation in microglia plays a critical role in depression. As shown in Figure 1, there are two processes in the canonical signaling pathway activating the NLRP3 inflammasome: priming and protein complex assembly.
Figure 1.
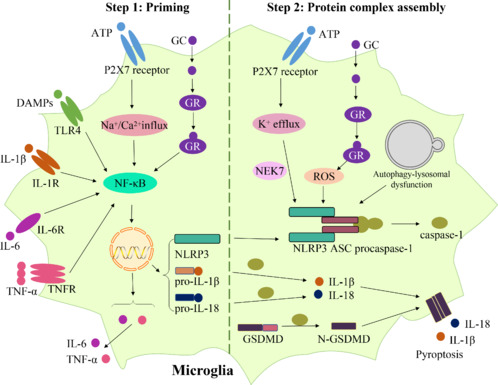
Mechanisms underlying NLRP3 inflammasome-mediated neuroinflammation in depression.
In the priming process, the GR, P2X7 receptor, TLR4, IL-1R, IL-6R, and TNFR can recognize their corresponding ligands, inducing the NF-κB pathway activation, increased production of NLRP3, pro-IL-18, and pro-IL-1β, and increased expression of IL-6 and TNF-α in microglia. In the protein complex assembly process, GC can bind to GR and then promotes ROS formation. ATP binds to the P2X7 receptor and then induces K+ efflux. ROS production, K+ efflux, as well as autophagy-lysosomal dysfunction, can promote NLRP3 inflammasome activation. Afterward, caspase-1 is activated by NLRP3 inflammasome, resulting in cytokine and GSDMD cleavage, leading to plasma membrane pore formation that mediates pyroptosis and the release of the mature IL-18 and IL-1β. Created with Microsoft PowerPoint. ASC: Apoptosis-associated speck-like protein containing a caspase recruitment domain; ATP: adenosine triphosphate; DAMPs: danger-associated molecular patterns; GC: glucocorticoid; GR: glucocorticoid receptor; GSDMD: gasdermin D; IL-18: interleukin-18; IL-1R: interleukin-1 receptor; IL-1β: interleukin-1beta; IL-6: interleukin-6; IL-6R: interleukin-6 receptor; NEK7: NIMA-related kinase 7; NF-κB: nuclear factor-kappa B; N-GSDMD: N-terminus of GSDMD; NLRP3: nucleotide-binding oligomerization domain-, leucine-rich repeat-, and pyrin domain-containing protein 3; P2X7: purinergic receptor P2X7; ROS: reactive oxygen species; TLR4: Toll-like receptor 4; TNF-α: tumor necrosis factor-alpha; TNFR: tumor necrosis factor receptor.
Priming process
A major risk factor for depression is stress. Glucocorticoids (GCs) are a class of steroid hormones that are naturally produced by the body and are involved in regulating stress responses. However, high doses or prolonged use of synthetic GCs have been found to be associated with depression in some individuals. It has been shown that chronic restraint stress causes increased corticosterone levels in serum and depression-like behaviors in rats. Corticosterone binds to the glucocorticoid receptor (GR), inducing increased Iba-1, NF-κB, NLRP3, cleaved caspase-1, IL-18, and IL-1β contents in the hippocampus of rats. Findings have suggested that the activation of the GR-NF-κB-NLRP3 pathway mediates chronic stress-induced depression-like behaviors and microglial neuroinflammation in rats (Feng et al., 2019). TLR4 has also been shown to mediate neuroinflammation. TLR4 acts as a signaling molecule upstream of the activation of the NLRP3 inflammasome and can promote the secretion of proinflammatory cytokines (Fu et al., 2019). The NF-κB pathway appears to be vital in regulating neuroinflammation in depression. The activated NF-κB then induces the expression of NLRP3, pro-IL-18, and pro-IL-1β levels subsequently (McKee and Coll, 2020).
Moreover, the extracellular release of ATP occurs in neurons and astrocytes in response to excessive glutamate induced by psychosocial stress (Ribeiro et al., 2019; Wang et al., 2020a; von Muecke-Heim et al., 2021). It activates the purinergic receptor P2X7 on microglia and causes Na+ and Ca2+ influx, resulting in the upregulation of NF-κB, inducing increased production of pro-IL-18 and pro-IL-1β and increased expression of IL-6 and TNF-α (von Muecke-Heim et al., 2021). Activation of the ATP-P2X7 receptor can stimulate the secretion of proinflammatory cytokines, and genetic or pharmacological inhibition of the ATP-P2X7 receptor is able to alleviate depression-like behavior in animal models (Bartlett et al., 2014), suggesting that the ATP-P2X7 receptor is involved in the development of depression. Then, IL-6, IL-1β, and TNF-α bind to their receptors, the IL-6 receptor, IL-1 receptor, and TNF receptor, respectively, activating NF-κB, triggering and aggravating neuroinflammation (von Muecke-Heim et al., 2021). Overall, during the priming process, various inflammatory stimuli activate the NF-κB pathway and trigger the expression of proinflammatory cytokines and NLRP3.
Protein complex assembly process
GC may induce brain oxidative stress, resulting in the production of ROS. Several cellular components, including lipids, proteins, and DNA, can be damaged by this. As previously mentioned, chronic restraint stress causes an increase in the GC level, which binds to the GR and then promotes ROS formation in the hippocampus of rats (Feng et al., 2019). ROS production can promote the NLRP3 inflammasome activation, also called protein complex assembly (Martinon, 2010; Harijith et al., 2014). The NLRP3 inflammasome can be activated by ROS via stimulating the production of mitochondrial ROS that can result in mitochondrial dysfunction and cytoplasmic release of mitochondrial DNA (Heid et al., 2013). The NLRP3 inflammasome can then be activated when mitochondrial DNA binds to it.
Besides, it has also been documented that the P2X7 receptor mediates the NLRP3 inflammasome activation in depression progression (Ribeiro et al., 2019; Wang et al., 2020a; von Muecke-Heim et al., 2021). Our previous study indicated that hippocampal extracellular ATP in CUMS mice is significantly enhanced, and the microglial P2X7 receptor and subsequent NLRP3 inflammasome activation contributed to depressive-like behaviors (Yue et al., 2017). The P2X7 receptors open when extracellular ATP binds to them, allowing for K+ to pass through and leading to K+ efflux. K+ concentrations in the cells are reduced as a result of K+ efflux. There is evidence that K+ efflux is a key cellular event that triggers NLRP3 inflammasome activation (Muñoz-Planillo et al., 2013). This pathway is NIMA-related kinase 7 dependent (He et al., 2016). Thus, the ATP-P2X7 receptor-K+ efflux signaling pathway and its downstream mediator NIMA-related kinase 7-NLRP3 inflammasome complex contribute to depression pathogenesis.
Moreover, the autophagic process is also related to NLRP3 inflammasome activation. Autophagy is a process of breaking down and recycling damaged components of cells, such as organelles and proteins. It maintains cellular homeostasis. The lysosome is a cellular organelle that is involved in the degradation of cellular waste and macromolecules that are sequestered during autophagy. Autophagy-lysosomal pathway dysfunction may slow the degradation of the NLRP3 inflammasome, contributing to depression-like behavior in CUMS mice (Li et al., 2022). Thus, in the protein complex assembly process, the NLRP3 activation results in the formation of the inflammasome complex, resulting in the secretion of proinflammatory cytokines such as IL-18 and IL-1β.
In summary, in the priming process, the GR, P2X7 receptor, TLR4, IL-1 receptor, IL-6 receptor, and TNF receptor can recognize their corresponding ligands, inducing activation of the NF-κB pathway, increased production of NLRP3, pro-IL-18, and pro-IL-1β, and increased expression of IL-6 and TNF-α in microglia. In the protein complex assembly process, GC can bind to GR and then promote ROS formation. ATP binds to the P2X7 receptor and then induces K+ efflux. ROS production, K+ efflux, as well as autophagy-lysosomal dysfunction, can promote NLRP3 inflammasome activation. Afterward, caspase-1 is activated by the NLRP3 inflammasome, resulting in cytokine and GSDMD cleavage, leading to plasma membrane pore formation that mediates pyroptosis and the release of mature IL-18 and IL-1β. IL-18 and IL-1β play significant roles in depression. The persistent release of IL-18 and IL-1β can lead to neuronal damage and impairment of neural circuitry, resulting in symptoms of depression. These processes are illustrated in Figure 1.
Treatment Approaches for Depression Based on the NLRP3 Inflammasome
There are multiple therapeutic strategies that target the NLRP3 inflammasome in depression informed by the etiological role of NLRP3 inflammasome-mediated neuroinflammation. These strategies include inhibitors of the NLRP3 inflammatory pathway, natural compounds, other therapeutic compounds, and non-drug therapies. These approaches have demonstrated efficacy in targeting the NLRP3 inflammasome to treat depression.
Inhibitors targeting the NLRP3 inflammasome pathway
CY-09 is a small molecule inhibitor of the NLRP3 inflammasome that has shown effects in several disease models characterized by NLRP3-mediated inflammation, including arthritis, gout, and Alzheimer's disease. One study suggested that CY-09 may also have antidepressant effects by modulating the NLRP3 inflammasome pathway in the brain (Wang et al., 2022b). In a preclinical study, a thorough biochemical analysis revealed that the administration of LPS resulted in a significant upregulation of NLRP3, ASC, caspase-1, IL-1β, IL-6, and TNF-α protein levels in the mouse hippocampus, while the administration of CY-09 demonstrated a preventive effect on these alterations (Wang et al., 2022b). Another study indicated that MCC950, an NLRP3 inflammasome inhibitor, could prevent depression-like behaviors in a mouse model of depression induced by chronic social isolation by decreasing NLRP3, IL-1β, and IL-18 protein levels in the hippocampus (Li et al., 2021b). Likewise, beta-hydroxybutyrate (BHB), a key ketone body, acts as an inhibitor of the NLRP3 inflammasome and exhibits antidepressant properties by reducing proinflammatory cytokine levels in the prefrontal cortex of rats (Kajitani et al., 2020). Goldberg et al. (2017) discovered that a ketogenic diet could elevate BHB levels, which then deactivate the neutrophil NLRP3 inflammasome, providing relief from gout flares induced by uric acid crystals. Their investigation revealed that BHB inhibits the NLRP3 inflammasome in neutrophils by diminishing priming and assembly processes (Goldberg et al., 2017). Boccella et al. (2019) treated neuropathic mice with dimethyl fumarate and BHB, resulting in an upregulation of hydroxyl carboxylic acid receptor type 2, the endogenous receptor for BHB, in the sciatic nerve and dorsal root ganglia. They identified a significant role of hydroxyl carboxylic acid receptor type 2 in modulating neuropathic pain plasticity (Boccella et al., 2019). These studies collectively highlighted the potential beneficial effects of BHB in regulating inflammatory responses and alleviating neuropathic pain.
Further, Cli-095, a TLR4 inhibitor, exerted antidepressant effects via the suppression of the TLR4–NF-κB–NLRP3 pathway in CUMS mice (Fu et al., 2019). BAY 117082, an inhibitor of NF-κB, decreased levels of NF-κB, IL-18, IL-1β, NLRP3, ASC, and cleaved caspase-1 (Feng et al., 2019). Additionally, both Brilliant Blue G and A438079, P2X7 receptor inhibitors, alleviated depression-like behaviors by inhibiting the ATP–P2X7 receptor–NLRP3 signaling pathway in CUMS rats (Yue et al., 2017). Moreover, studies have revealed that a specific inhibitor of caspase-1, VX-765, could decrease serum and hippocampal IL-1β levels and GSDMD-dependent pyroptosis, and alleviate behavioral dysfunctions in depressive rats induced by monosodium glutamate (Yang et al., 2020), in CUMS-induced depressive mice (Zhang et al., 2015), or ovariectomy-induced depressive mice (Xu et al., 2016). Figure 2 displays the structures of inhibitors that target the NLRP3 inflammasome pathway, which has been identified as a potential therapeutic target for depression treatment. Table 1 summarizes the pharmacological activities of these compounds.
Figure 2.
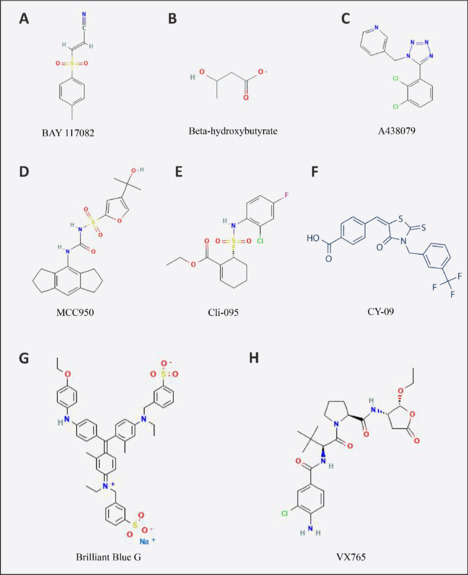
The structures of inhibitors that target the NLRP3 inflammasome pathway for the treatment of depression.
The structures were obtained from https://pubchem.ncbi.nlm.nih.gov/ (Kim et al., 2023). (A) BAY 117082; (B) beta-hydroxybutyrate; (C) A438079; (D) MCC950; (E) Cli-095; (F) CY-09; (G) Brilliant Blue G; (H) VX765.
Table 1.
The pharmacological activities of drugs targeting the NLRP3 inflammasome pathway as potential therapeutic treatments for depression
| Classification | Compound | Cell line/animal model | Drug administration | Mechanism | Analyzed region | Reference |
|---|---|---|---|---|---|---|
| NLRP3 inflammasome inhibitor | CY-09 | LPS-induced male ICR mouse model | CY-09 (10 mg/kg) was administered intraperitoneally daily for 3 d | CY-09 inhibited the NLRP3/ASC/cytokine signaling pathway and microglial activation | Hippocampus | Wang et al., 2022b |
| MCC950 | Chronic social isolation-induced male C57BL/6J mouse model | MCC950 (5 mg/kg) was administered intraperitoneally once a day, beginning 6 wk after social isolation | MCC950 remarkably decreased NLRP3, IL-1β, and IL-18 levels | Hippocampus | Li et al., 2021b | |
| BHB | CUMS-induced male Sprague-Dawley rat model | BHB (80 mg/mL, two pumps per rat) was microinjected into the prefrontal cortex for 21 d | BHB down-regulated the proinflammatory cytokines levels | Prefrontal cortex | Kajitani et al., 2020 | |
| TLR4 inhibitor | Cli-095 | CUMS-induced male ICR mouse model | Mice were administered with Cli-095 (3 mg/kg) via gastric irrigation daily for 4 wk | Cli-095 inhibited the TLR4-NF-κB-NLRP3 signaling pathway | Prefrontal cortex and hippocampus | Fu et al., 2019 |
| NF-κB inhibitor | BAY 117082 | Dexamethasone-induced HAPI cell model | BAY 117082 (10 μM) was administered 30 min before dexamethasone | BAY 117082 decreased the NF-κB, IL-1β, IL-18, NLRP3, ASC, and cleaved caspase-1 contents | NA | Feng et al., 2019 |
| P2X7 receptor inhibitor | Brilliant Blue G | CUMS-induced male Sprague-Dawley rat model | Brilliant Blue G (1 pM) was delivered into the hippocampus of rats for 21 d | Brilliant Blue G inhibited the ATP-P2X7 receptor-NLRP3 signaling pathway | Hippocampus | Yue et al., 2017 |
| A438079 | CUMS-induced male Sprague-Dawley rat model | A438079 (1.75 nM) was delivered into the hippocampus of rats for 21 d | A438079 inhibited the ATP-P2X7 receptor-NLRP3 signaling pathway | Hippocampus | Yue et al., 2017 | |
| Caspase-1 inhibitor | VX-765 | Newborn Sprague-Dawley rats were subcutaneously administered monosodium glutamate (4 mg/kg/d). Only male rats were used 2 mon later | VX-765 (200 mg/kg/d) was administrated intraperitoneally in model rats for 4 wk | VX-765 inhibited GSDMD-dependent pyroptosis | Hippocampus | Yang et al., 2020 |
| CUMS-induced male BALB/c mouse model | VX-765 (50 mg/kg) was injected intraperitoneally daily for 4 wk | VX-765 down-regulated IL-1β contents in serum and hippocampus | Serum and hippocampus | Zhang et al., 2015 | ||
| Ovariectomy-induced female C57BL/6J mouse model | VX-765 (25, 50 mg/kg/d) was injected subcutaneously for 4 wk | VX-765 down-regulated the IL-1β and IL-18 contents | Hippocampus | Xu et al., 2016 |
ASC: Apoptosis-associated speck-like protein containing a caspase recruitment domain; ATP: adenosine triphosphate; BHB: beta-hydroxybutyrate; CUMS: chronic unpredictable mild stress; GSDMD: gasdermin D; ICR: Institute of Cancer Research; IL-18: interleukin-18; IL-1β: interleukin-1beta; NA: not applicable; NF-κB: nuclear factor-kappa B; NLRP3: nucleotide-binding oligomerization domain-, leucine-rich repeat-, and pyrin domain-containing protein 3; P2X7: purinergic receptor P2X 7; TLR4: Toll-like receptor 4.
Natural compounds that target NLRP3 inflammasome
The use of natural compounds derived from plants has gained attention as potential alternatives or adjuvants to antidepressant compounds owing to the fewer side effects and better tolerability. Numerous studies have investigated the salutary effects of natural compounds derived from plants on depression. Findings have demonstrated that numerous natural compounds are capable of alleviating depressive symptoms in rodents by inhibiting NLRP3 inflammasome activation, such as antcin K (Han et al., 2023), allicin (Gao et al., 2019), apigenin (Li et al., 2016), astragaloside IV (Song et al., 2018a), baicalin (Liu and Liu, 2017; Zhang et al., 2018a), berberine (Qin et al., 2023), betaine (Zhang et al., 2022), catalpol (Wang et al., 2021), curcumin (Zhang et al., 2019a), crocin (Zhang et al., 2018b), dihydromyricetin (Wei et al., 2022), ginsenoside Rg1 (Zhang et al., 2019b), ginsenosides Rb1 (Jiang et al., 2022), gastrodin (Ye et al., 2018), hesperidin (Xie et al., 2020), hypericin (Zhai et al., 2022), isoliquiritin (Li et al., 2021d), Lonicera japonica polysaccharide (Liu et al., 2019), liquiritin (Liu et al., 2022), 4-methylesculetin (Choudhary et al., 2023), oridonin (Li et al., 2021a), perilla aldehyde (Song et al., 2018b), patchouli alcohol (He et al., 2023), salvianolic acid B (Huang et al., 2019), senegenin (Li et al., 2017), salidroside (Chai et al., 2022), and trans-cinnamaldehyde (Wang et al., 2020b).
Specifically, in a study by Li et al. (2021d), isoliquiritin, a compound found in licorice root, was found to possess the ability to inhibit NLRP3-mediated pyroptosis. The study observed reduced serum levels of miRNA-27a in patients with depression, and a similar reduction was also noted in animal models of depression. The introduction of a mimic of miRNA-27a significantly decreased the expression of the spleen tyrosine kinase (SYK) gene. Isoliquiritin demonstrated effective alleviation of depression symptoms and anxiety induced by LPS or chronic social defeat stress. Isoliquiritin restored levels of miRNA-27a mRNA and SYK protein, phosphorylation of NF-κB, NF-κB, NLRP3, cleaved caspase-1, IL-1β, N-GSDMD, IL-6, and TNF-α in the hippocampus. Additionally, isoliquiritin improved neuronal survival and morphology while reducing neuronal cell death associated with pyroptosis. It also provided protection to microglia against activation of the NLRP3 inflammasome. These findings indicated that isoliquiritin exhibits potent antidepressant properties, which are mediated through the miRNA-27a/SYK/NF-κB axis in reducing pyroptosis via the NLRP3 cascade (Li et al., 2021d).
One study by Wang et al. (2021) investigated the neuroprotective effects of catalpol, a compound found in Rehmannia glutinosa, in mice subjected to CUMS by examining the involvement of the NLRP3 inflammasome and neuroinflammation. The behavioral impairments induced by CUMS could be reversed in the absence of NLRP3. Additionally, similar to the NLRP3 inflammasome, the expression of IL-1β, TNF-α, and inducible nitric oxide synthase was increased after CUMS. The results demonstrated that catalpol exerted anti-inflammatory effects on CUMS mice and inhibited microglial polarization toward the M1 phenotype. The study provided evidence suggesting that catalpol exhibited antidepressant effects on CUMS mice potentially through the regulation of NLRP3 and neuroinflammation mediated by oxidative stress (Wang et al., 2021).
A study by Song et al. (2018a) explored the effects of Astragaloside IV, a compound derived from Astragalus membranaceus, on depression and neuroinflammation in mice. The researchers induced depression-like behavior in mice using repeated restraint stress or LPS and found that administration of Astragaloside IV significantly reduced these behaviors. Astragaloside IV also decreased the levels of inflammatory markers TNF-α and IL-1β, increased the expression of PPARgamma and phosphorylation of GSK3beta, and reduced the activation of NF-κB and the NLRP3 inflammasome in the hippocampus. These findings suggested that Astragaloside IV has potential as a treatment for depression by reducing neuroinflammation (Song et al., 2018a). These studies were well-designed and provided reliable results. Therefore, natural compounds derived from plants may represent promising alternatives or adjuvants to conventional antidepressants due to their ability to ameliorate depressive behaviors via inhibition of NLRP3 inflammasome activation.
Other therapeutic compounds that target the NLRP3 inflammasome
Other compounds have also been shown to indirectly inhibit NLRP3 inflammasome-mediated neuroinflammation in depression. For instance, melatonin, a hormone that has a role in sleep, inhibited the NLRP3 inflammasome activation and pyroptosis induced by LPS and ATP (Arioz et al., 2019). Moreover, metformin, a drug used to treat diabetes mellitus type 2, alleviated CUMS-induced depression-like behavior via the inhibition of NLRP3 inflammasome activation in the hippocampus and peripheral macrophages in mice (Du and Bu, 2020).
Fingolimod, an analog of sphingosine, not only protected neurons from damage and oxidative stress but also improved depression-like behavior and cognitive function in rats exposed to CUMS. It achieved this by inhibiting the activation of NF-κB and preventing the assembly of NLRP3 inflammasome by downregulating NLRP3, ACS, and caspase-1. Fingolimod inhibited the markers associated with proinflammatory microglial M1 polarization, such as inducible nitric oxide synthase and CD16, while promoting markers associated with anti-inflammatory M2 polarization, such as Arg-1 and CD206. Consequently, it reduced levels of the proinflammatory cytokines IL-1β, TNF-α, and IL-6 and increased levels of the anti-inflammatory cytokine IL-10 in the hippocampus, shielding hippocampal neurons from stress-induced damage and alleviating depressive symptoms by inhibiting neuroinflammation (Guo et al., 2020).
Simvastatin, a statin drug primarily used to lower cholesterol levels, has been found to possess neuroprotective properties. One study demonstrated that simvastatin effectively reduced depression-like behavior in rats that had undergone ovariectomy. It achieved this by inhibiting the activation of microglia in the hippocampus, suppressing the expression of P2X7 receptors, TLR2, and TLR4, and inhibiting the activation of the NLRP3 inflammasome. As a result, the levels of proinflammatory mediators IL-1β and IL-18 were reduced. Simvastatin significantly increased the expression of ERalpha and ERbeta in the hippocampus, without affecting uterine weight or ERalpha expression, suggesting that it could be a safer alternative for managing postmenopausal depression without causing hyperplastic effects on the uterus (Menze et al., 2021).
Tert-butylhydroquinone, a synthetic derivative of hydroquinone, is an antioxidant known for its anti-inflammatory properties. In one study, tert-butylhydroquinone treatment was found to significantly alleviate depression-like behaviors induced by LPS in mice. Tert-butylhydroquinone treatment suppressed the expression of NLRP3 protein, caspase-1, IL-1β, and IL-18, all of which are induced by LPS. Tert-butylhydroquinone treatment also inhibited the activation of microglia triggered by LPS. These findings suggested that tert-butylhydroquinone effectively improves LPS-induced inflammation-related depression-like behavior by modulating the NLRP3 signaling pathway and reducing neuroinflammation (Bian et al., 2022). Additionally, the other compounds, including resatorvid (Luo et al., 2020) and rapamycin (Aghaie et al., 2021) could also alleviate depression-like behavior by suppressing the microglial activation of the NLRP3 inflammasome in rodents. Thus, inflammatory responses are regulated by the NLRP3 inflammasome, which plays a major role in depression pathogenesis.
Non-drug therapies regulating the NLRP3 inflammasome pathway
Non-drug therapies targeting the NLRP3 inflammasome pathway have emerged as promising therapeutic alternatives for treating depression, such as physical exercise, acupuncture, electroacupuncture, and environmental enrichment. Physical exercise has been shown to have antidepressant effects, and evidence has suggested that exercise can modulate NLRP3 inflammasome activation and attenuate neuroinflammation in an ovariectomy-induced animal model of depression (Wang et al., 2016). The study findings revealed that ovariectomy in mice led to depression-like behavior and elevated levels of IL-1β and IL-18 in the hippocampus. However, exercise intervention improved depression-like behavior and reduced the levels of IL-1β and IL-18 in the hippocampus. Hippocampal IL-1β and IL-18 levels were found to be closely related to depression-like behavior in ovariectomized mice. Additionally, the levels of NLRP3, cleaved caspase-1 P10, and CD11b in the hippocampus were higher in ovariectomized mice compared to a control group. Exercise was effective in lowering the levels of NLRP3, cleaved caspase-1 P10, and CD11b in ovariectomized mice. These results suggested that exercise-mediated improvement in depression-like behavior is associated with the suppression of NLRP3 inflammasome activation in the hippocampus of this ovariectomy model (Wang et al., 2016).
Traditionally, acupuncture is a technique in traditional Chinese medicine that involves inserting thin needles into the skin at various points on the body. Acupuncture is used as an important complementary therapy for the treatment of depression. Recent research has suggested that acupuncture may exert antidepressant effects by regulating prefrontal cortex NLRP3 inflammasome-mediated neuroinflammation of chronic stress-induced depressive rats (Li et al., 2021c). The results demonstrated that acupuncture treatment effectively reversed depression-like behaviors in stressed rats. In comparison to control rats, CUMS rats exhibited a significant increase in the protein expression of NLRP3, ASC, and caspase-1, as well as the mRNA and protein expressions of IL-1β and IL-18 in the prefrontal cortex. Acupuncture treatment notably reduced the levels of NLRP3 inflammasome components and inflammatory cytokines. Additionally, acupuncture treatment decreased the number of terminal deoxynucleotidyl transferase dUTP nick end labeling-positive cells, indicating a reduction in apoptosis. These findings suggested that acupuncture possesses antidepressant-like effects, potentially mediated through the inhibition of NLRP3 inflammasome activation and apoptosis in the prefrontal cortex (Li et al., 2021c).
Electroacupuncture is a form of acupuncture that involves the use of small electrical currents applied to the needles inserted in acupuncture points. This technique enhances the effectiveness of traditional acupuncture by increasing the stimulation intensity and duration and modulating the frequency, leading to improved therapeutic benefits. Electroacupuncture can also prevent depressive-like behaviors by regulating hippocampal NLRP3 inflammasomes in chronic mild stress-induced depressive mice (Wang et al., 2022a). It was observed that chronic mild stress led to an increase in depression-like behaviors, which were subsequently reversed after electroacupuncture treatment. Additionally, chronic mild stress resulted in the nuclear entry of NF-κB (as indicated by the nuclear/cytoplasmic ratio of NF-κB) and increased protein levels of phosphorylated NF-κB in the hippocampus. Furthermore, NLRP3, IL-6, IL-1β, IL-18, and TNF-α levels in the hippocampus were elevated due to chronic mild stress. However, these effects were reversed by electroacupuncture, suggesting that electroacupuncture effectively prevents depression-like behaviors by inhibiting the NF-κB/NLRP3 inflammatory pathway (Wang et al., 2022a). Both electroacupuncture and acupuncture have been shown to be safe and effective treatment modalities (Zhang et al., 2023).
In addition, environmental enrichment, encompassing various forms of sensory, cognitive, and social stimulation, has been shown to have antidepressant effects and increase hippocampal neuroplasticity. Recent evidence has suggested that environmental enrichment can mitigate NLRP3 inflammasome activation and improve neuroinflammation in animal models of depression (Xu et al., 2022). CUMS led to the development of depression-like behaviors and cognitive impairment in rats. In CUMS rats, there was a decrease in the expression of autophagy-related proteins, specifically microtubule-associated protein 1 light chain 3 alpha and beclin-1, while there was an increase in inflammation activation markers including microglia numbers, NLRP3 inflammasomes, and proinflammatory cytokines such as IL-1β, IL-6, and TNF-α. However, after receiving environmental enrichment, these changes were effectively reversed (Xu et al., 2022). Therefore, non-drug therapies, such as physical exercise, acupuncture, electroacupuncture, and environmental enrichment, can provide alternative approaches for the treatment of depression by regulating the NLRP3 inflammasome pathway.
NLRP3 Inflammasome Inhibitors in Clinical Trials
There are several NLRP3 inflammasome inhibitors that are currently in clinical trials for various diseases. One study indicated that MCC950 could be utilized for the treatment of inflammatory diseases (Coll et al., 2015). Subsequently, the studies of NLRP3 inhibitors progressed into clinical testing stages, where three representative companies emerged. (1) Following the acquisition by Roche (Basel, Switzerland), with MCC950 as the basis, Inflazome Company developed three compounds with distinct properties for tissue distribution (Schwaid and Spencer, 2021). A CNS-penetrant NLRP3 inhibitor called Inzomelid was developed for the treatment of amyotrophic lateral sclerosis, Parkinson's disease, Alzheimer's disease, and cryopyrin-associated periodic syndrome. Specifically designed to treat cryopyrin-associated periodic syndrome, the NLRP3 inhibitor Somalix has peripherally restricted action. Compounds that are gut-restricted are also being developed. (2) IFM Tre company, after being acquired by Novartis, IFM-2747, which is different from MCC950 and is tailored for treating Crohn's disease, nonalcoholic fatty liver disease, coronary artery disease, and gout (Schwaid and Spencer, 2021). (3) As the most advanced NLRP3 inhibitor, a simple β-sulfonyl nitrile called dapansutrile was created by Olatec Company for the treatment of acute gout (Klück et al., 2020). These inhibitors are currently being evaluated in clinical trials, and it is expected that their safety and efficacy will be further assessed in larger patient populations. If successful, these inhibitors may provide a new therapeutic approach for treating many inflammatory and autoimmune disorders.
Although there is increasing evidence that NLRP3 inflammasomes contributes to depression pathogenesis, as of now, there is a limited availability of clinical trials specifically exploring the use of NLRP3 inflammasome inhibitors for the treatment of depression. However, many preclinical studies have suggested that targeting NLRP3 inflammasomes may have therapeutic potential for depression. Based on these preclinical studies, targeting NLRP3 inflammasomes may be a promising therapeutic strategy for depression. However, the use of NLRP3 inflammasome inhibitors presents some challenges. While the NLRP3 inflammasome is essential in the body's defense against various infections, inhibiting it can disrupt the host's response. For instance, studies have demonstrated that inhibiting the NLRP3 inflammasome interferes with the body's response to fungal (Kasper et al., 2018), Streptococcus pneumoniae (Witzenrath et al., 2011), and influenza A infection (Tate et al., 2016). Additionally, as depression is a CNS disorder, it is crucial to increase the blood–brain barrier penetration of NLRP3 inhibitors. To achieve this, the inhibitor molecules can be modified by activating carrier transporters (glucose transporter 1, L-type amino acid transporter), decreasing efflux (breast cancer resistance protein, multidrug resistance 1), and enhancing diffusion (controlling multiple parameters, reducing pKa, enhancing rigidity, reducing topological polar surface area, reducing hydrogen bond donor capacity, increasing lipophilicity) as evidenced by a recent study (Xiong et al., 2021). These strategies can serve as a reference for future CNS drug development using small molecules. Lastly, the activation mechanisms of NLRP3 are complex, and posing an additional challenge for researchers, so further study is needed to clarify the vital role of NLRP3 inflammasomes in the pathophysiology of depression and to evaluate the safety and efficacy of NLRP3 inhibitors in human clinical trials for this indication.
It is indeed interesting to note that a clinical study (NCT05792540) is currently being conducted to evaluate the potential efficacy of dapagliflozin and atorvastatin in patients with MDD. The study is currently in the recruiting phase. In this study, the drugs being investigated are fluoxetine (20 mg), dapagliflozin (10 mg tablets), and atorvastatin (80 mg). The primary endpoint of the study is changes in Hamilton Depression Rating Scale scores, a commonly used scale for assessing the severity of depression. The secondary endpoint involves evaluating changes in serum biomarkers such as nuclear factor erythroid 2-related factor 2, which is associated with oxidative stress and inflammation. Dapagliflozin, a sodium-glucose cotransporter 2 inhibitor used for treating diabetes mellitus type 2, and atorvastatin, a statin medication used for managing high cholesterol levels, have been studied for their potential effects on NLRP3 inflammasome activation. Previous research has suggested that dapagliflozin may have anti-inflammatory effects by inhibiting NLRP3 inflammasome activation (Muhammad et al., 2021). Similarly, studies have indicated that atorvastatin may be able to suppress NLRP3 inflammasome activation, leading to a reduction in proinflammatory cytokine production (Liu et al., 2021; Chen et al., 2022). These findings suggested that both dapagliflozin and atorvastatin may possess anti-inflammatory properties related to modulating the NLRP3 inflammasome pathway. An ongoing clinical study in Egypt aims to further investigate the potential efficacy of dapagliflozin and atorvastatin in patients with MDD, including their effects on NLRP3 inflammasome activation and associated inflammatory pathways. It will be interesting to see the results of this study and how these medications may contribute to the management of depression.
Conclusion
Depression is a prevalent mental disorder characterized by alterations in mood. Though the mechanisms behind the pathogenesis of depression are not well understood, there have been a number of hypotheses put forward, such as the monoamine hypothesis (Wong and Licinio, 2001), neurotrophic hypothesis (Duman, 2005), inflammatory hypothesis (Miller and Raison, 2016), stress-diathesis model (Kendler and Gardner, 1998), hypothalamic–pituitary-adrenal axis dysfunction hypothesis (Pariante and Lightman, 2008), circadian rhythm hypothesis (McClung, 2007), and glutamatergic hypothesis (Sanacora et al., 2012). These mechanisms are not mutually exclusive but may interact with each other. Among these, the inflammatory hypothesis has emerged as a compelling avenue of investigation in recent years. This suggests that depression is linked to inflammation in the brain and systemic inflammation (Miller and Raison, 2016). Growing evidence indicates that chronic immune system activation and neuroinflammation are related to the pathogenesis of depression. The NLRP3 inflammasome acts as a vital mediator of inflammation and innate immunity and is believed to play a critical role in the progression and development of depression (Xia et al., 2023).
Due to the current prominence of research on the NLRP3 inflammasome in depression, several recent reviews (Kaufmann et al., 2017; Kouba et al., 2022; Wan et al., 2022; Roy et al., 2023; Xia et al., 2023) have been published on this topic. This review aligns closely with these previous reviews in elucidating the underlying mechanisms and clarifying the therapeutic effects of NLRP3 inflammasome inhibitors, natural compounds, and other compounds. However, this review also describes non-drug therapies such as physical exercise, acupuncture, electroacupuncture, and environmental enrichment, as well as the progress and challenges of clinical trials involving NLRP3 inflammasome inhibitors, distinguishing it from the existing literature.
It should be noted that the relationship between NLRP3 inflammasome activation and depression appears to be complex and multifaceted. On the one hand, studies have suggested that NLRP3 inflammasome activation may contribute to the development or worsening of depressive symptoms (Kouba et al., 2022). Specifically, excessive or prolonged NLRP3 inflammasome activation can lead to the release of proinflammatory cytokines, such as IL-1β and IL-18, which contribute to neuroinflammation and have been associated with depressive symptoms (Kouba et al., 2022). On the other hand, depression itself may induce NLRP3 inflammasome activation through various mechanisms. For example, chronic psychological stress and depression-related alterations in the hypothalamic-pituitary-adrenal axis and cortisol levels may be responsible for activation of NLRP3 inflammasomes (Hassamal, 2023; Xia et al., 2023). Furthermore, NLRP3 inflammasome activation can exacerbate depression by triggering neuroinflammation and inducing pyroptosis (Wan et al., 2022). More research is needed to fully understand the causal relationship and the underlying mechanisms involved.
Several studies have indicated that age and sex differences exist in NLRP3 inflammasome activation within the context of depression. Aging has been associated with increased NLRP3 inflammasome activation, which may contribute to the chronic inflammation observed in older individuals (Andina et al., 2023). Age-related depression has also been linked to elevated levels of proinflammatory cytokines, such as IL-1β, which is involved in the alternative NLRP3 inflammasome pathway within the brain (Godbout et al., 2008). Consequently, older age may be associated with higher levels of NLRP3 inflammasome components and an augmented release of proinflammatory cytokines, potentially exacerbating depression symptoms. Moreover, evidence suggests that women exhibit greater NLRP3 inflammasome activation in the context of depression compared to men. One study found higher levels of IL-1β in the peripheral blood of treatment-naïve young women with MDD when compared to their male counterparts (Kim et al., 2021). Another study identified a common functional IL-18 haplotype associated with increased proinflammatory responses, which conferred susceptibility to stress-related depression and anxiety primarily in women through effects on threat-related amygdala function (Swartz et al., 2017). Therefore, these sex differences may contribute to the higher prevalence of depression observed in women. Considering the age and sex differences in NLRP3 inflammasome activation in depression is crucial for preclinical research as it aids in understanding the underlying mechanisms of inflammation-related depressive disorders and enables the development of tailored interventions. By developing targeted therapies that account for age and sex-specific factors, more effective treatment strategies for depression can be developed.
Selecting an appropriate animal model of depression is crucial for specific experimental purposes. Several commonly used models include CUMS, chronic social defeat stress, LPS administration, and ovariectomy-induced models. The CUMS model involves subjecting animals to a variety of unpredictable and mild stressors over an extended period (Hu et al., 2017; Antoniuk et al., 2019; Markov and Novosadova, 2022). This mimics the chronic and unpredictable stress experienced by humans. It is suitable for simulating chronic stress-related depression and can induce various behavioral, neurochemical, and neuroendocrine alterations associated with depression-like symptoms. The chronic social defeat stress model entails repeated social defeat experiences where an animal is exposed to aggression from a conspecific (Huang et al., 2016; Kim et al., 2017; Harris et al., 2018). This model is particularly useful for studying depression associated with social stressors and models the social hierarchy disruptions observed in some human depressive disorders. LPS is a component of gram-negative bacteria that, when administered, induces an immune response and triggers inflammation by activating proinflammatory cytokines (Lin and Wang, 2014; Rodrigues et al., 2018; Yin et al., 2023). This model is ideal for investigating inflammation-induced depression and allows researchers to explore the role of neuroinflammation in depressive-like behaviors. Ovariectomy involves surgically removing the ovaries in female animals, leading to a decline in hormone levels, particularly estrogen and progesterone (Li et al., 2014; Schoenrock et al., 2016; Puga-Olguín et al., 2019). Ovariectomy is mainly used to investigate depression associated with hormonal changes, such as postpartum depression or menopause-related depression. In summary, LPS administration is suitable for investigating NLRP3 inflammasome-mediated neuroinflammation in depression through controlled induction of inflammation. However, it specifically focuses on inflammation-related depression and may not reflect the entire spectrum of depressive disorders. Therefore, when selecting an animal model of depression, researchers should consider the specific research objectives and purposes.
Biomarkers are essential in disease diagnosis, prognosis, and treatment. In the context of understanding NLRP3 biology and developing drugs targeting the NLRP3 inflammasome for depression, the identification of a suitable PET tracer is valuable. A recent study has reported the discovery of such a PET tracer for NLRP3 (Xu et al., 2021). The study involved a medicinal chemistry campaign focused on developing sulfonamide-based NLRP3 inhibitors, which led to the creation of an analog, referred to as compound 1. This analog incorporated a methoxy substituent that could be labeled with carbon-11. PET/computed tomography imaging studies were conducted using [11C]1, revealing rapid penetration through the blood-brain barrier, moderate brain uptake, and preventable uptake in the brain. These findings suggest that [11C]1 has the potential to serve as a useful tool for imaging NLRP3 inflammasomes in living brains (Xu et al., 2021). Since NLRP3 inflammasome activation may not be the primary cause of depression in every case, identifying biomarkers that can predict treatment response to NLRP3 inhibitors is crucial for optimizing patient selection and improving treatment outcomes.
Ketamine has garnered significant interest in recent years due to its ability to rapidly and effectively alleviate symptoms of depression. Notably, one study revealed that ketamine induces a pro-resilient phenotype in response to depression-like behavior triggered by LPS or TNF-α administration. This effect has been linked to the suppression of the NLRP3 inflammasome-mediated signaling pathway (Camargo et al., 2021). Besides, fluoxetine, a commonly prescribed antidepressant, has been found to possess potential anti-inflammatory activity. An investigation has demonstrated that fluoxetine effectively inhibits the activation of the NLRP3 inflammasome, subsequent caspase-1 cleavage, and secretion of IL-1β in both peripheral macrophages and central microglia. These findings suggested that fluoxetine exerts its antidepressant effects in part through the suppression of peripheral and central NLRP3 inflammasome activation (Du et al., 2016). Therefore, both ketamine and fluoxetine exhibit potential clinical utility in the treatment of depression, specifically in cases where depression is associated with NLRP3 inflammasome-driven inflammatory conditions.
Although there is growing evidence of the involvement of NLRP3 inflammasomes in depression, there is a limited number of clinical trials investigating the use of NLRP3 inflammasome inhibitors as a targeted treatment for depression. While NLRP3 inhibitors have shown potential as a treatment for depression, there are several considerations and challenges that need to be addressed. In addition to the previously mentioned roles of NLRP3 in innate immune response and the blood-brain barrier penetration ability of NLRP3 inhibitors, it is important to assess if NLRP3 inhibitors are effective for individuals with depression by identifying NLRP3 inflammasome-related biomarkers. Therefore, further research is needed to comprehensively understand the safety, efficacy, limitations, and drawbacks associated with the use of NLRP3 inhibitors as a therapeutic option for depression.
In conclusion, the activation of microglial NLRP3 inflammasome is considered to be a critical factor in the pathogenesis of depression. The activation process of the NLRP3 inflammasome involves two distinct steps: priming and assembly, which exhibit a complex mechanism requiring further investigation. Targeting NLRP3 inflammasomes has the potential to offer novel therapeutic avenues for managing depression. NLRP3 inflammasome-targeted therapeutic strategies encompass the use of agents such as NLRP3 inflammatory pathway inhibitors, natural products, other therapeutic compounds, and non-drug therapies. It is necessary to conduct further research to explore the effectiveness and safety of the therapeutic strategies as potential antidepressant treatments.
Funding Statement
Funding: This work was supported by Health Commission of Pudong New Area Health and Family Planning Scientific Research Project, No. PW2020E-4 (to GL); Siming Youth Fund Project of Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, No. SGKJ-202119 (to RH); Medical Innovation Research Special Project of 2021 “Science and Technology Innovation Action Plan” of Shanghai, No. 21Y21920200 (to GL); Shanghai Rising-Star Program and Shanghai Sailing Program, No. 23YF1418200 (to QH); Shanghai Municipal Health Commission Foundation grant, No. 20234Y0294 (to QH); Hundred Teacher Talent Program of Shanghai University of Medicine and Health Sciences, No. A1-2601-23-311007-21 (to QH); the Scientific and Technological Innovation Program of Higher Education Institution in Shanxi, No. 2021L350 (to XC); the Fundamental Research Program of Shanxi Province, No. 20210302124194 (to XC).
Footnotes
Conflicts of interest: The authors declare no conflict of interest.
Data availability statement: Not applicable.
C-Editor: Zhao M; S-Editors: Yu J, Li CH; L-Editors: Yu J, Song LP; T-Editor: Jia Y
References
- Aghaie F, Moradifar F, Hosseini A. Rapamycin attenuates depression and anxiety-like behaviors through modulation of the NLRP3 pathway in pentylenetetrazole-kindled male Wistar rats. Fundam Clin Pharmacol. 2021;35:1045–1054. doi: 10.1111/fcp.12689. [DOI] [PubMed] [Google Scholar]
- Agius M, Bonnici H. Antidepressants in use in clinical practice. Psychiatria Danubina. 2017;29:667–671. [PubMed] [Google Scholar]
- Alcocer-Gómez E, Cordero MD. NLRP3 inflammasome: a new target in major depressive disorder. CNS Neurosci Ther. 2014;20:294–295. doi: 10.1111/cns.12230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alcocer-Gómez E, de Miguel M, Casas-Barquero N, Núñez-Vasco J, Sánchez-Alcazar JA, Fernández-Rodríguez A, Cordero MD. NLRP3 inflammasome is activated in mononuclear blood cells from patients with major depressive disorder. Brain Behav Immun. 2014;36:111–117. doi: 10.1016/j.bbi.2013.10.017. [DOI] [PubMed] [Google Scholar]
- Andina N, de Meuron L, Schnegg-Kaufmann AS, Sarangdhar MA, Ansermet C, Bombaci G, Batta K, Keller N, Porret NA, Angelillo-Scherrer A, Bonadies N, Allam R. Increased inflammasome activation is associated with aging and chronic myelomonocytic leukemia disease severity. J Immunol. 2023;210:580–589. doi: 10.4049/jimmunol.2200412. [DOI] [PubMed] [Google Scholar]
- Antoniuk S, Bijata M, Ponimaskin E, Wlodarczyk J. Chronic unpredictable mild stress for modeling depression in rodents: Meta-analysis of model reliability. Neurosci Biobehav Rev. 2019;99:101–116. doi: 10.1016/j.neubiorev.2018.12.002. [DOI] [PubMed] [Google Scholar]
- Arioz BI, Tastan B, Tarakcioglu E, Tufekci KU, Olcum M, Ersoy N, Bagriyanik A, Genc K, Genc S. Melatonin attenuates LPS-induced acute depressive-like behaviors and microglial NLRP3 inflammasome activation through the SIRT1/Nrf2 pathway. Front Immunol. 2019;10:1511. doi: 10.3389/fimmu.2019.01511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai B, Yang Y, Wang Q, Li M, Tian C, Liu Y, Aung LHH, Li PF, Yu T, Chu XM. NLRP3 inflammasome in endothelial dysfunction. Cell Death Dis. 2020;11:776. doi: 10.1038/s41419-020-02985-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartlett R, Stokes L, Sluyter R. The P2X7 receptor channel: recent developments and the use of P2X7 antagonists in models of disease. Pharmacol Rev. 2014;66:638–675. doi: 10.1124/pr.113.008003. [DOI] [PubMed] [Google Scholar]
- Benedetti F, Aggio V, Pratesi ML, Greco G, Furlan R. Neuroinflammation in bipolar depression. Front Psychiatry. 2020;11:71. doi: 10.3389/fpsyt.2020.00071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beurel E, Toups M, Nemeroff CB. The bidirectional relationship of depression and inflammation: double trouble. Neuron. 2020;107:234–256. doi: 10.1016/j.neuron.2020.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bian H, Yan F, Li W, Tu W, Ji X. Tert-butylhydroquinone prevents neuroinflammation and relieves depression via regulation of NLRP3 signaling in mice. Int Immunopharmacol. 2022;107:108723. doi: 10.1016/j.intimp.2022.108723. [DOI] [PubMed] [Google Scholar]
- Boccella S, Guida F, De Logu F, De Gregorio D, Mazzitelli M, Belardo C, Iannotta M, Serra N, Nassini R, de Novellis V, Geppetti P, Maione S, Luongo L. Ketones and pain: unexplored role of hydroxyl carboxylic acid receptor type 2 in the pathophysiology of neuropathic pain. FASEB J. 2019;33:1062–1073. doi: 10.1096/fj.201801033R. [DOI] [PubMed] [Google Scholar]
- Brites D, Fernandes A. Neuroinflammation and depression: microglia activation, extracellular microvesicles and microRNA dysregulation. Front Cell Neurosci. 2015;9:476. doi: 10.3389/fncel.2015.00476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camargo A, Dalmagro AP, Wolin IAV, Kaster MP, Rodrigues ALS. The resilient phenotype elicited by ketamine against inflammatory stressors-induced depressive-like behavior is associated with NLRP3-driven signaling pathway. J Psychiatr Res. 2021;144:118–128. doi: 10.1016/j.jpsychires.2021.09.057. [DOI] [PubMed] [Google Scholar]
- Chai Y, Cai Y, Fu Y, Wang Y, Zhang Y, Zhang X, Zhu L, Miao M, Yan T. Salidroside ameliorates depression by suppressing NLRP3-mediated pyroptosis via P2X7/NF-κB/NLRP3 signaling pathway. Front Pharmacol. 2022;13:812362. doi: 10.3389/fphar.2022.812362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakraborty R, Tabassum H, Parvez S. NLRP3 inflammasome in traumatic brain injury: Its implication in the disease pathophysiology and potential as a therapeutic target. Life Sci. 2023;314:121352. doi: 10.1016/j.lfs.2022.121352. [DOI] [PubMed] [Google Scholar]
- Chen D, Sui L, Chen C, Liu S, Sun X, Guan J. Atorvastatin suppresses NLRP3 inflammasome activation in intracerebral hemorrhage via TLR4- and MyD88-dependent pathways. Aging (Albany NY) 2022;14:462–476. doi: 10.18632/aging.203824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choudhary K, Prasad SR, Lokhande KB, Murti K, Singh S, Ravichandiran V, Kumar N. 4-Methylesculetin ameliorates LPS-induced depression-like behavior through the inhibition of NLRP3 inflammasome. Front Pharmacol. 2023;14:1120508. doi: 10.3389/fphar.2023.1120508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coll RC, Robertson AA, Chae JJ, Higgins SC, Muñoz-Planillo R, Inserra MC, Vetter I, Dungan LS, Monks BG, Stutz A, Croker DE, Butler MS, Haneklaus M, Sutton CE, Núñez G, Latz E, Kastner DL, Mills KH, Masters SL, Schroder K, et al. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat Med. 2015;21:248–255. doi: 10.1038/nm.3806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crupi R, Cuzzocrea S. Neuroinflammation and immunity: a new pharmacological target in depression. CNS Neurol Disord Drug Targets. 2016;15:464–476. doi: 10.2174/1871527315666160321105339. [DOI] [PubMed] [Google Scholar]
- Cui Y, Yu H, Bu Z, Wen L, Yan L, Feng J. Focus on the role of the NLRP3 inflammasome in multiple sclerosis: pathogenesis, diagnosis, and therapeutics. Front Mol Neurosci. 2022;15:894298. doi: 10.3389/fnmol.2022.894298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui ZQ, Hu XY, Yang T, Guan JW, Gu Y, Li HY, Zhang HY, Xiao QH, Sun XH. TMEM16F may be a new therapeutic target for Alzheimer's disease. Neural Regen Res. 2023;18:643–651. doi: 10.4103/1673-5374.350211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cullen SP, Kearney CJ, Clancy DM, Martin SJ. Diverse activators of the NLRP3 inflammasome promote IL-1β secretion by triggering necrosis. Cell Rep. 2015;11:1535–1548. doi: 10.1016/j.celrep.2015.05.003. [DOI] [PubMed] [Google Scholar]
- Dantzer R, O'Connor JC, Freund GG, Johnson RW, Kelley KW. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci. 2008;9:46–56. doi: 10.1038/nrn2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng SL, Chen JG, Wang F. Microglia: a central player in depression. Curr Med Sci. 2020;40:391–400. doi: 10.1007/s11596-020-2193-1. [DOI] [PubMed] [Google Scholar]
- Du RH, Tan J, Sun XY, Lu M, Ding JH, Hu G. Fluoxetine inhibits NLRP3 inflammasome activation: implication in depression. Int J Neuropsychopharmacol. 2016;19:pyw037. doi: 10.1093/ijnp/pyw037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du RW, Bu WG. Metformin improves depressive-like symptoms in mice via inhibition of peripheral and central NF-κB-NLRP3 inflammation activation. Exp Brain Res. 2020;238:2549–2556. doi: 10.1007/s00221-020-05911-x. [DOI] [PubMed] [Google Scholar]
- Dubois H, Wullaert A, Lamkanfi M. General strategies in inflammasome biology. Curr Top Microbiol Immunol. 2016;397:1–22. doi: 10.1007/978-3-319-41171-2_1. [DOI] [PubMed] [Google Scholar]
- Duman RS. Neurotrophic factors and regulation of mood: role of exercise, diet and metabolism. Neurobiol Aging 26 Suppl. 2005;1:88–93. doi: 10.1016/j.neurobiolaging.2005.08.018. [DOI] [PubMed] [Google Scholar]
- Elliott EI, Sutterwala FS. Initiation and perpetuation of NLRP3 inflammasome activation and assembly. Immunol Rev. 2015;265:35–52. doi: 10.1111/imr.12286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Felger JC, Treadway MT. Inflammation effects on motivation and motor activity: role of dopamine. Neuropsychopharmacology. 2017;42:216–241. doi: 10.1038/npp.2016.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng X, Zhao Y, Yang T, Song M, Wang C, Yao Y, Fan H. Glucocorticoid-driven NLRP3 inflammasome activation in hippocampal microglia mediates chronic stress-induced depressive-like behaviors. Front Mol Neurosci. 2019;12:210. doi: 10.3389/fnmol.2019.00210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng YS, Tan ZX, Wu LY, Dong F, Zhang F. The involvement of NLRP3 inflammasome in the treatment of neurodegenerative diseases. Biomed Pharmacother. 2021;138:111428. doi: 10.1016/j.biopha.2021.111428. [DOI] [PubMed] [Google Scholar]
- Ferrari AJ, Charlson FJ, Norman RE, Patten SB, Freedman G, Murray CJ, Vos T, Whiteford HA. Burden of depressive disorders by country, sex, age, and year: findings from the global burden of disease study 2010. PLoS Med. 2013;10:e1001547. doi: 10.1371/journal.pmed.1001547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu S, Wang J, Hao C, Dang H, Jiang S. Tetramethylpyrazine ameliorates depression by inhibiting TLR4-NLRP3 inflammasome signal pathway in mice. Psychopharmacology (Berl) 2019;236:2173–2185. doi: 10.1007/s00213-019-05210-6. [DOI] [PubMed] [Google Scholar]
- Gao W, Wang W, Liu G, Zhang J, Yang J, Deng Z. Allicin attenuated chronic social defeat stress induced depressive-like behaviors through suppression of NLRP3 inflammasome. Metab Brain Dis. 2019;34:319–329. doi: 10.1007/s11011-018-0342-z. [DOI] [PubMed] [Google Scholar]
- Godbout JP, Moreau M, Lestage J, Chen J, Sparkman NL, O'Connor J, Castanon N, Kelley KW, Dantzer R, Johnson RW. Aging exacerbates depressive-like behavior in mice in response to activation of the peripheral innate immune system. Neuropsychopharmacology. 2008;33:2341–2351. doi: 10.1038/sj.npp.1301649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberg EL, Asher JL, Molony RD, Shaw AC, Zeiss CJ, Wang C, Morozova-Roche LA, Herzog RI, Iwasaki A, Dixit VD. β-Hydroxybutyrate deactivates neutrophil NLRP3 inflammasome to relieve gout flares. Cell Rep. 2017;18:2077–2087. doi: 10.1016/j.celrep.2017.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan Y, Han F. Key mechanisms and potential targets of the NLRP3 inflammasome in neurodegenerative diseases. Front Integr Neurosci. 2020;14:37. doi: 10.3389/fnint.2020.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Y, Gan X, Zhou H, Zhou H, Pu S, Long X, Ren C, Feng T, Tang H. Fingolimod suppressed the chronic unpredictable mild stress-induced depressive-like behaviors via affecting microglial and NLRP3 inflammasome activation. Life Sci. 2020;263:118582. doi: 10.1016/j.lfs.2020.118582. [DOI] [PubMed] [Google Scholar]
- Haapakoski R, Mathieu J, Ebmeier KP, Alenius H, Kivimäki M. Cumulative meta-analysis of interleukins 6 and 1β, tumour necrosis factor α and C-reactive protein in patients with major depressive disorder. Brain Behav Immun. 2015;49:206–215. doi: 10.1016/j.bbi.2015.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han C, Pei H, Shen H, Zhai L, Yang Y, Li W, Wang J. Antcin K targets NLRP3 to suppress neuroinflammation and improve the neurological behaviors of mice with depression. Int Immunopharmacol. 2023;117:109908. doi: 10.1016/j.intimp.2023.109908. [DOI] [PubMed] [Google Scholar]
- Han QQ, Le W. NLRP3 inflammasome-mediated neuroinflammation and related mitochondrial impairment in Parkinson's disease. Neurosci Bull. 2023;39:832–844. doi: 10.1007/s12264-023-01023-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harijith A, Ebenezer DL, Natarajan V. Reactive oxygen species at the crossroads of inflammasome and inflammation. Front Physiol. 2014;5:352. doi: 10.3389/fphys.2014.00352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris AZ, Atsak P, Bretton ZH, Holt ES, Alam R, Morton MP, Abbas AI, Leonardo ED, Bolkan SS, Hen R, Gordon JA. A novel method for chronic social defeat stress in female mice. Neuropsychopharmacology. 2018;43:1276–1283. doi: 10.1038/npp.2017.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hassamal S. Chronic stress, neuroinflammation, and depression: an overview of pathophysiological mechanisms and emerging anti-inflammatories. Front Psychiatry. 2023;14:1130989. doi: 10.3389/fpsyt.2023.1130989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He H, Xie X, Zhang J, Mo L, Kang X, Zhang Y, Wang L, Hu N, Xie L, Peng C, You Z. Patchouli alcohol ameliorates depression-like behaviors through inhibiting NLRP3-mediated neuroinflammation in male stress-exposed mice. J Affect Disord. 2023;326:120–131. doi: 10.1016/j.jad.2023.01.065. [DOI] [PubMed] [Google Scholar]
- He Y, Zeng MY, Yang D, Motro B, Núñez G. NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux. Nature. 2016;530:354–357. doi: 10.1038/nature16959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heid ME, Keyel PA, Kamga C, Shiva S, Watkins SC, Salter RD. Mitochondrial reactive oxygen species induces NLRP3-dependent lysosomal damage and inflammasome activation. J Immunol. 2013;191:5230–5238. doi: 10.4049/jimmunol.1301490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howren MB, Lamkin DM, Suls J. Associations of depression with C-reactive protein, IL-1, and IL-6: a meta-analysis. Psychosom Med. 2009;71:171–186. doi: 10.1097/PSY.0b013e3181907c1b. [DOI] [PubMed] [Google Scholar]
- Hu C, Luo Y, Wang H, Kuang S, Liang G, Yang Y, Mai S, Yang J. Re-evaluation of the interrelationships among the behavioral tests in rats exposed to chronic unpredictable mild stress. PLoS One. 2017;12:e0185129. doi: 10.1371/journal.pone.0185129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang GB, Zhao T, Gao XL, Zhang HX, Xu YM, Li H, Lv LX. Effect of chronic social defeat stress on behaviors and dopamine receptor in adult mice. Prog Neuropsychopharmacol Biol Psychiatry. 2016;66:73–79. doi: 10.1016/j.pnpbp.2015.12.002. [DOI] [PubMed] [Google Scholar]
- Huang Q, Ye X, Wang L, Pan J. Salvianolic acid B abolished chronic mild stress-induced depression through suppressing oxidative stress and neuro-inflammation via regulating NLRP3 inflammasome activation. J Food Biochem. 2019;43:e12742. doi: 10.1111/jfbc.12742. [DOI] [PubMed] [Google Scholar]
- Jeenger J, Singroha V, Sharma M, Mathur DM. C-reactive protein, brain-derived neurotrophic factor, interleukin-2, and stressful life events in drug-naive first-episode and recurrent depression: a cross-sectional study. Indian J Psychiatry. 2018;60:334–339. doi: 10.4103/psychiatry.IndianJPsychiatry_169_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeon SW, Kim YK. Neuroinflammation and cytokine abnormality in major depression: cause or consequence in that illness? World J Psychiatry. 2016;6:283–293. doi: 10.5498/wjp.v6.i3.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jha D, Bakker E, Kumar R. Mechanistic and therapeutic role of NLRP3 inflammasome in the pathogenesis of Alzheimer's disease. J Neurochem. 2023 doi: 10.1111/jnc.15788. doi: 10.1111/jnc.15788. [DOI] [PubMed] [Google Scholar]
- Jia X, Gao Z, Hu H. Microglia in depression: current perspectives. Sci China Life Sci. 2021;64:911–925. doi: 10.1007/s11427-020-1815-6. [DOI] [PubMed] [Google Scholar]
- Jiang M, Qin P, Yang X. Comorbidity between depression and asthma via immune-inflammatory pathways: a meta-analysis. J Affect Disord. 2014;166:22–29. doi: 10.1016/j.jad.2014.04.027. [DOI] [PubMed] [Google Scholar]
- Jiang N, Zhang Y, Yao C, Huang H, Wang Q, Huang S, He Q, Liu X. Ginsenosides Rb1 attenuates chronic social defeat stress-induced depressive behavior via regulation of SIRT1-NLRP3/Nrf2 pathways. Front Nutr. 2022;9:868833. doi: 10.3389/fnut.2022.868833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jo EK, Kim JK, Shin DM, Sasakawa C. Molecular mechanisms regulating NLRP3 inflammasome activation. Cell Mol Immunol. 2016;13:148–159. doi: 10.1038/cmi.2015.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajitani N, Iwata M, Miura A, Tsunetomi K, Yamanashi T, Matsuo R, Nishiguchi T, Fukuda S, Nagata M, Shibushita M, Yamauchi T, Pu S, Shirayama Y, Watanabe K, Kaneko K. Prefrontal cortex infusion of beta-hydroxybutyrate, an endogenous NLRP3 inflammasome inhibitor, produces antidepressant-like effects in a rodent model of depression. Neuropsychopharmacol Rep. 2020;40:157–165. doi: 10.1002/npr2.12099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasper L, König A, Koenig PA, Gresnigt MS, Westman J, Drummond RA, Lionakis MS, Groß O, Ruland J, Naglik JR, Hube B. The fungal peptide toxin Candidalysin activates the NLRP3 inflammasome and causes cytolysis in mononuclear phagocytes. Nat Commun. 2018;9:4260. doi: 10.1038/s41467-018-06607-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufmann FN, Costa AP, Ghisleni G, Diaz AP, Rodrigues ALS, Peluffo H, Kaster MP. NLRP3 inflammasome-driven pathways in depression: Clinical and preclinical findings. Brain Behav Immun. 2017;64:367–383. doi: 10.1016/j.bbi.2017.03.002. [DOI] [PubMed] [Google Scholar]
- Kelley N, Jeltema D, Duan Y, He Y. The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci. 2019;20:3328. doi: 10.3390/ijms20133328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kendler KS, Gardner CO., Jr Twin studies of adult psychiatric and substance dependence disorders: are they biased by differences in the environmental experiences of monozygotic and dizygotic twins in childhood and adolescence? Psychol Med. 1998;28:625–633. doi: 10.1017/s0033291798006643. [DOI] [PubMed] [Google Scholar]
- Kim HD, Call T, Carotenuto S, Johnson R, Ferguson D. Testing depression in mice: a chronic social defeat stress model. Bio Protoc. 2017;7:e2203. doi: 10.21769/BioProtoc.2203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J, Kim JH, Chang KA. Sex difference in peripheral inflammatory biomarkers in drug-naïve patients with major depression in young adulthood. Biomedicines. 2021;9:708. doi: 10.3390/biomedicines9070708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S, Chen J, Cheng T, Gindulyte A, He J, He S, Li Q, Shoemaker BA, Thiessen PA, Yu B, Zaslavsky L, Zhang J, Bolton EE. PubChem 2023 update. Nucleic Acids Res. 2023;51:D1373–D1380. doi: 10.1093/nar/gkac956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klück V, Jansen T, Janssen M, Comarniceanu A, Efdé M, Tengesdal IW, Schraa K, Cleophas MCP, Scribner CL, Skouras DB, Marchetti C, Dinarello CA, Joosten LAB. Dapansutrile, an oral selective NLRP3 inflammasome inhibitor, for treatment of gout flares: an open-label, dose-adaptive, proof-of-concept, phase 2a trial. The Lancet Rheumatology. 2020;2:e270–e280. doi: 10.1016/s2665-9913(20)30065-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kouba BR, Gil-Mohapel J, AL SR. NLRP3 inflammasome: from pathophysiology to therapeutic target in major depressive disorder. Int J Mol Sci. 2022;24:133. doi: 10.3390/ijms24010133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamkanfi M, Dixit VM. Inflammasomes and their roles in health and disease. Annu Rev Cell Dev Biol. 2012;28:137–161. doi: 10.1146/annurev-cellbio-101011-155745. [DOI] [PubMed] [Google Scholar]
- Lamkanfi M, Dixit VM. Mechanisms and functions of inflammasomes. Cell. 2014;157:1013–1022. doi: 10.1016/j.cell.2014.04.007. [DOI] [PubMed] [Google Scholar]
- Leemans JC, Cassel SL, Sutterwala FS. Sensing damage by the NLRP3 inflammasome. Immunol Rev. 2011;243:152–162. doi: 10.1111/j.1600-065X.2011.01043.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li C, Zhu Y, Wu Y, Fu M, Wu Y, Wu Y, Qiu Y, Zhang H, Ding M. Oridonin alleviates LPS-induced depression by inhibiting NLRP3 inflammasome via activation of autophagy. Front Med (Lausanne) 2021a;8:813047. doi: 10.3389/fmed.2021.813047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Lin S, Qin T, Li H, Ma Z, Ma S. Senegenin exerts anti-depression effect in mice induced by chronic un-predictable mild stress via inhibition of NF-κB regulating NLRP3 signal pathway. Int Immunopharmacol. 2017;53:24–32. doi: 10.1016/j.intimp.2017.10.001. [DOI] [PubMed] [Google Scholar]
- Li LH, Wang ZC, Yu J, Zhang YQ. Ovariectomy results in variable changes in nociception, mood and depression in adult female rats. PLoS One. 2014;9:e94312. doi: 10.1371/journal.pone.0094312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li MM, Wang X, Chen XD, Yang HL, Xu HS, Zhou P, Gao R, Zhang N, Wang J, Jiang L, Liu N. Lysosomal dysfunction is associated with NLRP3 inflammasome activation in chronic unpredictable mild stress-induced depressive mice. Behav Brain Res. 2022;432:113987. doi: 10.1016/j.bbr.2022.113987. [DOI] [PubMed] [Google Scholar]
- Li R, Wang X, Qin T, Qu R, Ma S. Apigenin ameliorates chronic mild stress-induced depressive behavior by inhibiting interleukin-1β production and NLRP3 inflammasome activation in the rat brain. Behav Brain Res. 2016;296:318–325. doi: 10.1016/j.bbr.2015.09.031. [DOI] [PubMed] [Google Scholar]
- Li W, Niu L, Liu Z, Xu X, Shi M, Zhang Y, Deng Y, He J, Xu Y, Wan W, Sun Q, Zhong X, Cao W. Inhibition of the NLRP3 inflammasome with MCC950 prevents chronic social isolation-induced depression-like behavior in male mice. Neurosci Lett. 2021b;765:136290. doi: 10.1016/j.neulet.2021.136290. [DOI] [PubMed] [Google Scholar]
- Li X, Wang H, Li C, Wu J, Lu J, Guo JY, Tu Y. Acupuncture inhibits NLRP3 inflammasome activation in the prefrontal cortex of a chronic stress rat model of depression. Anat Rec (Hoboken) 2021c;304:2470–2479. doi: 10.1002/ar.24778. [DOI] [PubMed] [Google Scholar]
- Li Y, Song W, Tong Y, Zhang X, Zhao J, Gao X, Yong J, Wang H. Isoliquiritin ameliorates depression by suppressing NLRP3-mediated pyroptosis via miRNA-27a/SYK/NF-κB axis. J Neuroinflammation. 2021d;18:1. doi: 10.1186/s12974-020-02040-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang T, Zhang Y, Wu S, Chen Q, Wang L. The role of NLRP3 inflammasome in Alzheimer's disease and potential therapeutic targets. Front Pharmacol. 2022;13:845185. doi: 10.3389/fphar.2022.845185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YL, Wang S. Prenatal lipopolysaccharide exposure increases depression-like behaviors and reduces hippocampal neurogenesis in adult rats. Behav Brain Res. 2014;259:24–34. doi: 10.1016/j.bbr.2013.10.034. [DOI] [PubMed] [Google Scholar]
- Liu C, Yuan D, Zhang C, Tao Y, Meng Y, Jin M, Song W, Wang B, Wei L. Liquiritin alleviates depression-like behavior in CUMS mice by inhibiting oxidative stress and NLRP3 inflammasome in hippocampus. Evid Based Complement Alternat Med. 2022;2022:7558825. doi: 10.1155/2022/7558825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu P, Bai X, Zhang T, Zhou L, Li J, Zhang L. The protective effect of Lonicera japonica polysaccharide on mice with depression by inhibiting NLRP3 inflammasome. Ann Transl Med. 2019;7:811. doi: 10.21037/atm.2019.12.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu P, Gao Q, Guan L, Hu Y, Jiang J, Gao T, Sheng W, Xue X, Qiao H, Li T. Atorvastatin attenuates surgery-induced BBB disruption and cognitive impairment partly by suppressing NF-κB pathway and NLRP3 inflammasome activation in aged mice. Acta Biochim Biophys Sin (Shanghai) 2021;53:528–537. doi: 10.1093/abbs/gmab022. [DOI] [PubMed] [Google Scholar]
- Liu X, Liu C. Baicalin ameliorates chronic unpredictable mild stress-induced depressive behavior: Involving the inhibition of NLRP3 inflammasome activation in rat prefrontal cortex. Int Immunopharmacol. 2017;48:30–34. doi: 10.1016/j.intimp.2017.04.019. [DOI] [PubMed] [Google Scholar]
- Liu XL, Sun DD, Zheng MT, Li XT, Niu HH, Zhang L, Zhou ZW, Rong HT, Wang Y, Wang JW, Yang GL, Liu X, Chen FL, Zhou Y, Zhang S, Zhang JN. Maraviroc promotes recovery from traumatic brain injury in mice by suppression of neuroinflammation and activation of neurotoxic reactive astrocytes. Neural Regen Res. 2023;18:141–149. doi: 10.4103/1673-5374.344829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo W, Han Y, Meng P, Yang Q, Zhao H, Ling J, Wang Y. Resatorvid relieves breast cancer complicated with depression by inactivating hippocampal microglia through TLR4/NF-κB/NLRP3 signaling pathway. Cancer Manag Res. 2020;12:13003–13014. doi: 10.2147/CMAR.S279800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Y, Chiriboga DE, Pagoto SL, Rosal MC, Li W, Merriam PA, Hébert JR, Whited MC, Ockene IS. Association between depression and C-reactive protein. Cardiol Res Pract. 2010;2011:286509. doi: 10.4061/2011/286509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malhotra S, Hurtado-Navarro L, Pappolla A, Villar LMM, Río J, Montalban X, Pelegrin P, Comabella M. Increased NLRP3 inflammasome activation and pyroptosis in patients with multiple sclerosis with fingolimod treatment failure. Neurol Neuroimmunol Neuroinflamm. 2023;10:e200100. doi: 10.1212/NXI.0000000000200100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markov DD, Novosadova EV. Chronic unpredictable mild stress model of depression: possible sources of poor reproducibility and latent variables. Biology (Basel) 2022;11:1621. doi: 10.3390/biology11111621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinon F. Signaling by ROS drives inflammasome activation. Eur J Immunol. 2010;40:616–619. doi: 10.1002/eji.200940168. [DOI] [PubMed] [Google Scholar]
- McCarron RM, Shapiro B, Rawles J, Luo J. Depression. Ann Intern Med. 2021;174:ITC65–ITC80. doi: 10.7326/AITC202105180. [DOI] [PubMed] [Google Scholar]
- McClung CA. Circadian genes, rhythms and the biology of mood disorders. Pharmacol Ther. 2007;114:222–232. doi: 10.1016/j.pharmthera.2007.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKee CM, Coll RC. NLRP3 inflammasome priming: a riddle wrapped in a mystery inside an enigma. J Leukoc Biol. 2020;108:937–952. doi: 10.1002/JLB.3MR0720-513R. [DOI] [PubMed] [Google Scholar]
- Menze ET, Ezzat H, Shawky S, Sami M, Selim EH, Ahmed S, Maged N, Nadeem N, Eldash S, Michel HE. Simvastatin mitigates depressive-like behavior in ovariectomized rats: Possible role of NLRP3 inflammasome and estrogen receptors' modulation. Int Immunopharmacol. 2021;95:107582. doi: 10.1016/j.intimp.2021.107582. [DOI] [PubMed] [Google Scholar]
- Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. 2016;16:22–34. doi: 10.1038/nri.2015.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moncrieff J. Persistent adverse effects of antidepressants. Epidemiol Psychiatr Sci. 2019;29:e56. doi: 10.1017/S2045796019000520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mudra Rakshasa-Loots A, Bakewell N, Sharp DJ, Gisslén M, Zetterberg H, Alagaratnam J, Wit F, Kootstra NA, Winston A, Reiss P, Sabin CA, Vera JH. Biomarkers of central and peripheral inflammation mediate the association between HIV and depressive symptoms. Transl Psychiatry. 2023;13:190. doi: 10.1038/s41398-023-02489-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muhammad RN, Ahmed LA, Abdul Salam RM, Ahmed KA, Attia AS. Crosstalk among NLRP3 inflammasome, ET(B)R signaling, and miRNAs in stress-induced depression-like behavior: a modulatory role for SGLT2 inhibitors. Neurotherapeutics. 2021;18:2664–2681. doi: 10.1007/s13311-021-01140-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muñoz-Planillo R, Kuffa P, Martínez-Colón G, Smith BL, Rajendiran TM, Núñez G. K⁺ efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter. Immunity. 2013;38:1142–1153. doi: 10.1016/j.immuni.2013.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orsolini L, Pompili S, Tempia Valenta S, Salvi V, Volpe U. C-reactive protein as a biomarker for major depressive disorder? Int J Mol Sci. 2022;23:1616. doi: 10.3390/ijms23031616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paik S, Kim JK, Silwal P, Sasakawa C, Jo EK. An update on the regulatory mechanisms of NLRP3 inflammasome activation. Cell Mol Immunol. 2021;18:1141–1160. doi: 10.1038/s41423-021-00670-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan Y, Chen XY, Zhang QY, Kong LD. Microglial NLRP3 inflammasome activation mediates IL-1β-related inflammation in prefrontal cortex of depressive rats. Brain Behav Immun. 2014;41:90–100. doi: 10.1016/j.bbi.2014.04.007. [DOI] [PubMed] [Google Scholar]
- Pariante CM, Lightman SL. The HPA axis in major depression: classical theories and new developments. Trends Neurosci. 2008;31:464–468. doi: 10.1016/j.tins.2008.06.006. [DOI] [PubMed] [Google Scholar]
- Pitharouli MC, Hagenaars SP, Glanville KP, Coleman JRI, Hotopf M, Lewis CM, Pariante CM. Elevated C-reactive protein in patients with depression, independent of genetic, health, and psychosocial factors: results from the UK Biobank. Am J Psychiatry. 2021;178:522–529. doi: 10.1176/appi.ajp.2020.20060947. [DOI] [PubMed] [Google Scholar]
- Possemato E, La Barbera L, Nobili A, Krashia P, D'Amelio M. The role of dopamine in NLRP3 inflammasome inhibition: Implications for neurodegenerative diseases. Ageing Res Rev. 2023;87:101907. doi: 10.1016/j.arr.2023.101907. [DOI] [PubMed] [Google Scholar]
- Puga-Olguín A, Rodríguez-Landa JF, Rovirosa-Hernández MJ, Germán-Ponciano LJ, Caba M, Meza E, Guillén-Ruiz G, Olmos-Vázquez OJ. Long-term ovariectomy increases anxiety- and despair-like behaviors associated with lower Fos immunoreactivity in the lateral septal nucleus in rats. Behav Brain Res. 2019;360:185–195. doi: 10.1016/j.bbr.2018.12.017. [DOI] [PubMed] [Google Scholar]
- Qin Z, Shi DD, Li W, Cheng D, Zhang YD, Zhang S, Tsoi B, Zhao J, Wang Z, Zhang ZJ. Berberine ameliorates depression-like behaviors in mice via inhibiting NLRP3 inflammasome-mediated neuroinflammation and preventing neuroplasticity disruption. J Neuroinflammation. 2023;20:54. doi: 10.1186/s12974-023-02744-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rakesh G, Pae CU, Masand PS. Beyond serotonin: newer antidepressants in the future. Expert Rev Neurother. 2017;17:777–790. doi: 10.1080/14737175.2017.1341310. [DOI] [PubMed] [Google Scholar]
- Rashidi M, Wicks IP, Vince JE. Inflammasomes and cell death: common pathways in microparticle diseases. Trends Mol Med. 2020;26:1003–1020. doi: 10.1016/j.molmed.2020.06.005. [DOI] [PubMed] [Google Scholar]
- Ravichandran KA, Heneka MT. Inflammasome activation in neurodegenerative diseases. Essays Biochem. 2021;65:885–904. doi: 10.1042/EBC20210021. [DOI] [PubMed] [Google Scholar]
- Ribeiro DE, Roncalho AL, Glaser T, Ulrich H, Wegener G, Joca S. P2X7 receptor signaling in stress and depression. Int J Mol Sci. 2019;20:2778. doi: 10.3390/ijms20112778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodrigues FTS, de Souza MRM, Lima CNC, da Silva FER, Costa D, Dos Santos CC, Miyajima F, de Sousa FCF, Vasconcelos SMM, Barichello T, Quevedo J, Maes M, de Lucena DF, Macedo D. Major depression model induced by repeated and intermittent lipopolysaccharide administration: long-lasting behavioral, neuroimmune and neuroprogressive alterations. J Psychiatr Res. 2018;107:57–67. doi: 10.1016/j.jpsychires.2018.10.003. [DOI] [PubMed] [Google Scholar]
- Rosenblat JD, Cha DS, Mansur RB, McIntyre RS. Inflamed moods: a review of the interactions between inflammation and mood disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2014;53:23–34. doi: 10.1016/j.pnpbp.2014.01.013. [DOI] [PubMed] [Google Scholar]
- Roy S, Arif Ansari M, Choudhary K, Singh S. NLRP3 inflammasome in depression: a review. Int Immunopharmacol. 2023;117:109916. doi: 10.1016/j.intimp.2023.109916. [DOI] [PubMed] [Google Scholar]
- Sanacora G, Treccani G, Popoli M. Towards a glutamate hypothesis of depression: an emerging frontier of neuropsychopharmacology for mood disorders. Neuropharmacology. 2012;62:63–77. doi: 10.1016/j.neuropharm.2011.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoenrock SA, Oreper D, Young N, Ervin RB, Bogue MA, Valdar W, Tarantino LM. Ovariectomy results in inbred strain-specific increases in anxiety-like behavior in mice. Physiol Behav. 2016;167:404–412. doi: 10.1016/j.physbeh.2016.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwaid AG, Spencer KB. Strategies for targeting the NLRP3 inflammasome in the clinical and preclinical space. J Med Chem. 2021;64:101–122. doi: 10.1021/acs.jmedchem.0c01307. [DOI] [PubMed] [Google Scholar]
- Setiawan E, Wilson AA, Mizrahi R, Rusjan PM, Miler L, Rajkowska G, Suridjan I, Kennedy JL, Rekkas PV, Houle S, Meyer JH. Role of translocator protein density, a marker of neuroinflammation, in the brain during major depressive episodes. JAMA Psychiatry. 2015;72:268–275. doi: 10.1001/jamapsychiatry.2014.2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahyad S, Kheirabadi GR, Jahromi GP, Massaly M. Brain-derived neurotrophic factor and high sensitive C-reactive protein in bipolar depression and unipolar depression: the practical usage as a discriminatory tool. Clin Psychopharmacol Neurosci. 2023;21:108–117. doi: 10.9758/cpn.2023.21.1.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song MT, Ruan J, Zhang RY, Deng J, Ma ZQ, Ma SP. Astragaloside IV ameliorates neuroinflammation-induced depressive-like behaviors in mice via the PPARγ/NF-κB/NLRP3 inflammasome axis. Acta Pharmacol Sin. 2018a;39:1559–1570. doi: 10.1038/aps.2017.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song Y, Sun R, Ji Z, Li X, Fu Q, Ma S. Perilla aldehyde attenuates CUMS-induced depressive-like behaviors via regulating TXNIP/TRX/NLRP3 pathway in rats. Life Sci. 2018b;206:117–124. doi: 10.1016/j.lfs.2018.05.038. [DOI] [PubMed] [Google Scholar]
- Soraci L, Gambuzza ME, Biscetti L, Laganà P, Lo Russo C, Buda A, Barresi G, Corsonello A, Lattanzio F, Lorello G, Filippelli G, Marino S. Toll-like receptors and NLRP3 inflammasome-dependent pathways in Parkinson's disease: mechanisms and therapeutic implications. J Neurol. 2023;270:1346–1360. doi: 10.1007/s00415-022-11491-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanson KV, Deng M, Ting JP. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol. 2019;19:477–489. doi: 10.1038/s41577-019-0165-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swartz JR, Prather AA, Di Iorio CR, Bogdan R, Hariri AR. A functional interleukin-18 haplotype predicts depression and anxiety through increased threat-related amygdala reactivity in women but not men. Neuropsychopharmacology. 2017;42:419–426. doi: 10.1038/npp.2016.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taene A, Khalili-Tanha G, Esmaeili A, Mobasheri L, Kooshkaki O, Jafari S, Shokouhifar A, Sarab GA. The association of major depressive disorder with activation of NLRP3 inflammasome, lipid peroxidation, and total antioxidant capacity. J Mol Neurosci. 2020;70:65–70. doi: 10.1007/s12031-019-01401-0. [DOI] [PubMed] [Google Scholar]
- Tate MD, Ong JDH, Dowling JK, McAuley JL, Robertson AB, Latz E, Drummond GR, Cooper MA, Hertzog PJ, Mansell A. Reassessing the role of the NLRP3 inflammasome during pathogenic influenza A virus infection via temporal inhibition. Sci Rep. 2016;6:27912. doi: 10.1038/srep27912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Troubat R, Barone P, Leman S, Desmidt T, Cressant A, Atanasova B, Brizard B, El Hage W, Surget A, Belzung C, Camus V. Neuroinflammation and depression: a review. Eur J Neurosci. 2021;53:151–171. doi: 10.1111/ejn.14720. [DOI] [PubMed] [Google Scholar]
- Voet S, Srinivasan S, Lamkanfi M, van Loo G. Inflammasomes in neuroinflammatory and neurodegenerative diseases. EMBO Mol Med. 2019;11:e10248. doi: 10.15252/emmm.201810248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Muecke-Heim IA, Ries C, Urbina L, Deussing JM. P2X7R antagonists in chronic stress-based depression models: a review. Eur Arch Psychiatry Clin Neurosci. 2021;271:1343–1358. doi: 10.1007/s00406-021-01306-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan T, Li X, Fu M, Gao X, Li P, Guo W. NLRP3-dependent pyroptosis: a candidate therapeutic target for depression. Front Cell Neurosci. 2022;16:863426. doi: 10.3389/fncel.2022.863426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, Wang H, Gao H, Zhang H, Zhang H, Wang Q, Sun Z. P2X7 receptor mediates NLRP3 inflammasome activation in depression and diabetes. Cell Biosci. 2020a;10:28. doi: 10.1186/s13578-020-00388-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M, Yan S, Zhou Y, Xie P. Trans-Cinnamaldehyde reverses depressive-like behaviors in chronic unpredictable mild stress rats by inhibiting NF-κB/NLRP3 inflammasome pathway. Evid Based Complement Alternat Med. 2020b;2020:4572185. doi: 10.1155/2020/4572185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Bi H, Huang H, Wang Y, Gong L, Qi N, Li D, Jin X, Xu T, Shi B. Electroacupuncture prevents the depression-like behavior by inhibiting the NF-κB/NLRP3 inflammatory pathway in hippocampus of mice subjected to chronic mild stress. Neuropsychobiology. 2022a;81:237–245. doi: 10.1159/000521185. [DOI] [PubMed] [Google Scholar]
- Wang Y, Xu Y, Sheng H, Ni X, Lu J. Exercise amelioration of depression-like behavior in OVX mice is associated with suppression of NLRP3 inflammasome activation in hippocampus. Behav Brain Res. 2016;307:18–24. doi: 10.1016/j.bbr.2016.03.044. [DOI] [PubMed] [Google Scholar]
- Wang Y, Liu YJ, Zhang MM, Zhou H, Gao YH, Cheng WJ, Ye ZW, Yuan ZY, Xu GH, Li CF, Yi LT. CY-09 alleviates the depression-like behaviors via inhibiting NLRP3 inflammasome-mediated neuroinflammation in lipopolysaccharide-induced mice. ACS Chem Neurosci. 2022b;13:3291–3302. doi: 10.1021/acschemneuro.2c00348. [DOI] [PubMed] [Google Scholar]
- Wang YL, Wu HR, Zhang SS, Xiao HL, Yu J, Ma YY, Zhang YD, Liu Q. Catalpol ameliorates depressive-like behaviors in CUMS mice via oxidative stress-mediated NLRP3 inflammasome and neuroinflammation. Transl Psychiatry. 2021;11:353. doi: 10.1038/s41398-021-01468-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei Y, Hu Y, Qi K, Li Y, Chen J, Wang R. Dihydromyricetin improves LPS-induced sickness and depressive-like behaviors in mice by inhibiting the TLR4/Akt/HIF1a/NLRP3 pathway. Behav Brain Res. 2022;423:113775. doi: 10.1016/j.bbr.2022.113775. [DOI] [PubMed] [Google Scholar]
- Wies Mancini VSB, Di Pietro AA, Pasquini LA. Microglia depletion as a therapeutic strategy: friend or foe in multiple sclerosis models? Neural Regen Res. 2023;18:267–272. doi: 10.4103/1673-5374.346538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witzenrath M, Pache F, Lorenz D, Koppe U, Gutbier B, Tabeling C, Reppe K, Meixenberger K, Dorhoi A, Ma J, Holmes A, Trendelenburg G, Heimesaat MM, Bereswill S, van der Linden M, Tschopp J, Mitchell TJ, Suttorp N, Opitz B. The NLRP3 inflammasome is differentially activated by pneumolysin variants and contributes to host defense in pneumococcal pneumonia. J Immunol. 2011;187:434–440. doi: 10.4049/jimmunol.1003143. [DOI] [PubMed] [Google Scholar]
- Wong ML, Licinio J. Research and treatment approaches to depression. Nat Rev Neurosci. 2001;2:343–351. doi: 10.1038/35072566. [DOI] [PubMed] [Google Scholar]
- Xia CY, Guo YX, Lian WW, Yan Y, Ma BZ, Cheng YC, Xu JK, He J, Zhang WK. The NLRP3 inflammasome in depression: Potential mechanisms and therapies. Pharmacol Res. 2023;187:106625. doi: 10.1016/j.phrs.2022.106625. [DOI] [PubMed] [Google Scholar]
- Xiang H, Zhu F, Xu Z, Xiong J. Role of inflammasomes in kidney diseases via both canonical and non-canonical pathways. Front Cell Dev Biol. 2020;8:106. doi: 10.3389/fcell.2020.00106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie L, Gu Z, Liu H, Jia B, Wang Y, Cao M, Song R, Zhang Z, Bian Y. The anti-depressive effects of hesperidin and the relative mechanisms based on the NLRP3 inflammatory signaling pathway. Front Pharmacol. 2020;11:1251. doi: 10.3389/fphar.2020.01251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong B, Wang Y, Chen Y, Xing S, Liao Q, Chen Y, Li Q, Li W, Sun H. Strategies for structural modification of small molecules to improve blood-brain barrier penetration: a recent perspective. J Med Chem. 2021;64:13152–13173. doi: 10.1021/acs.jmedchem.1c00910. [DOI] [PubMed] [Google Scholar]
- Xu L, Sun H, Qu C, Shen J, Qu C, Song H, Li T, Zheng J, Zhang J. The environmental enrichment ameliorates chronic unpredictable mild stress-induced depressive-like behaviors and cognitive decline by inducing autophagy-mediated inflammation inhibition. Brain Res Bull. 2022;187:98–110. doi: 10.1016/j.brainresbull.2022.07.001. [DOI] [PubMed] [Google Scholar]
- Xu Y, Sheng H, Bao Q, Wang Y, Lu J, Ni X. NLRP3 inflammasome activation mediates estrogen deficiency-induced depression- and anxiety-like behavior and hippocampal inflammation in mice. Brain Behav Immun. 2016;56:175–186. doi: 10.1016/j.bbi.2016.02.022. [DOI] [PubMed] [Google Scholar]
- Xu Y, Xu Y, Blevins H, Lan Y, Liu Y, Yuan G, Striar R, Zagaroli JS, Tocci DR, Langan AG, Zhang C, Zhang S, Wang C. Discovery of carbon-11 labeled sulfonamide derivative: a PET tracer for imaging brain NLRP3 inflammasome. Bioorg Med Chem Lett. 2021;34:127777. doi: 10.1016/j.bmcl.2021.127777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang F, Zhu W, Cai X, Zhang W, Yu Z, Li X, Zhang J, Cai M, Xiang J, Cai D. Minocycline alleviates NLRP3 inflammasome-dependent pyroptosis in monosodium glutamate-induced depressive rats. Biochem Biophys Res Commun. 2020;526:553–559. doi: 10.1016/j.bbrc.2020.02.149. [DOI] [PubMed] [Google Scholar]
- Ye T, Meng X, Wang R, Zhang C, He S, Sun G, Sun X. Gastrodin alleviates cognitive dysfunction and depressive-like behaviors by inhibiting ER stress and NLRP3 inflammasome activation in db/db mice. Int J Mol Sci. 2018;19:3977. doi: 10.3390/ijms19123977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin R, Zhang K, Li Y, Tang Z, Zheng R, Ma Y, Chen Z, Lei N, Xiong L, Guo P, Li G, Xie Y. Lipopolysaccharide-induced depression-like model in mice: meta-analysis and systematic evaluation. Front Immunol. 2023;14:1181973. doi: 10.3389/fimmu.2023.1181973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yue N, Huang H, Zhu X, Han Q, Wang Y, Li B, Liu Q, Wu G, Zhang Y, Yu J. Activation of P2X7 receptor and NLRP3 inflammasome assembly in hippocampal glial cells mediates chronic stress-induced depressive-like behaviors. J Neuroinflammation. 2017;14:102. doi: 10.1186/s12974-017-0865-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai X, Chen Y, Han X, Zhu Y, Li X, Zhang Y, Lu Y. The protective effect of hypericin on postpartum depression rat model by inhibiting the NLRP3 inflammasome activation and regulating glucocorticoid metabolism. Int Immunopharmacol. 2022;105:108560. doi: 10.1016/j.intimp.2022.108560. [DOI] [PubMed] [Google Scholar]
- Zhang CY, Zeng MJ, Zhou LP, Li YQ, Zhao F, Shang ZY, Deng XY, Ma ZQ, Fu Q, Ma SP, Qu R. Baicalin exerts neuroprotective effects via inhibiting activation of GSK3β/NF-κB/NLRP3 signal pathway in a rat model of depression. Int Immunopharmacol. 2018a;64:175–182. doi: 10.1016/j.intimp.2018.09.001. [DOI] [PubMed] [Google Scholar]
- Zhang HM, Luo D, Chen R, Wang SH, Zhao YJ, Li JX, Zhou MF, Yu ZM, Zhang JL, Liang FX. Research progress on acupuncture treatment in central nervous system diseases based on NLRP3 inflammasome in animal models. Front Neurosci. 2023;17:1118508. doi: 10.3389/fnins.2023.1118508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Previn R, Lu L, Liao RF, Jin Y, Wang RK. Crocin, a natural product attenuates lipopolysaccharide-induced anxiety and depressive-like behaviors through suppressing NF-kB and NLRP3 signaling pathway. Brain Res Bull. 2018b;142:352–359. doi: 10.1016/j.brainresbull.2018.08.021. [DOI] [PubMed] [Google Scholar]
- Zhang M, Wang XL, Shi H, Meng LQ, Quan HF, Yan L, Yang HF, Peng XD. Betaine inhibits NLRP3 inflammasome hyperactivation and regulates microglial M1/M2 phenotypic differentiation, thereby attenuating lipopolysaccharide-induced depression-like behavior. J Immunol Res. 2022;2022:9313436. doi: 10.1155/2022/9313436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang WY, Guo YJ, Han WX, Yang MQ, Wen LP, Wang KY, Jiang P. Curcumin relieves depressive-like behaviors via inhibition of the NLRP3 inflammasome and kynurenine pathway in rats suffering from chronic unpredictable mild stress. Int Immunopharmacol. 2019a;67:138–144. doi: 10.1016/j.intimp.2018.12.012. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Liu L, Liu YZ, Shen XL, Wu TY, Zhang T, Wang W, Wang YX, Jiang CL. NLRP3 inflammasome mediates chronic mild stress-induced depression in mice via neuroinflammation. Int J Neuropsychopharmacol. 2015;18:pyv006. doi: 10.1093/ijnp/pyv006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang YQ, Wang XB, Xue RR, Gao XX, Li W. Ginsenoside Rg1 attenuates chronic unpredictable mild stress-induced depressive-like effect via regulating NF-κB/NLRP3 pathway in rats. Neuroreport. 2019b;30:893–900. doi: 10.1097/WNR.0000000000001302. [DOI] [PubMed] [Google Scholar]


