Abstract
The mimivirus 1.2 Mb genome was shown to be organized into a nucleocapsid-like genomic fiber encased in the nucleoid compartment inside the icosahedral capsid. The genomic fiber protein shell is composed of a mixture of two GMC-oxidoreductase paralogs, one of them being the main component of the glycosylated layer of fibrils at the surface of the virion. In this study, we determined the effect of the deletion of each of the corresponding genes on the genomic fiber and the layer of surface fibrils. First, we deleted the GMC-oxidoreductase, the most abundant in the genomic fiber, and determined its structure and composition in the mutant. As expected, it was composed of the second GMC-oxidoreductase and contained 5- and 6-start helices similar to the wild-type fiber. This result led us to propose a model explaining their coexistence. Then we deleted the GMC-oxidoreductase, the most abundant in the layer of fibrils, to analyze its protein composition in the mutant. Second, we showed that the fitness of single mutants and the double mutant were not decreased compared with the wild-type viruses under laboratory conditions. Third, we determined that deleting the GMC-oxidoreductase genes did not impact the glycosylation or the glycan composition of the layer of surface fibrils, despite modifying their protein composition. Because the glycosylation machinery and glycan composition of members of different clades are different, we expanded the analysis of the protein composition of the layer of fibrils to members of the B and C clades and showed that it was different among the three clades and even among isolates within the same clade. Taken together, the results obtained on two distinct central processes (genome packaging and virion coating) illustrate an unexpected functional redundancy in members of the family Mimiviridae, suggesting this may be the major evolutionary force behind their giant genomes.
Keywords: Mimivirus, giant virus, genomic fiber, cryo-EM, helical reconstruction, layer of fibrils, glycosylation, MS-based proteomics
Functional redundancy preserves the genomic fiber of mimivirus and the layer of fibril formation.
Introduction
Mimivirus is the inaugural member of the family Mimiviridae, part of the Nucleocytoviricota phylum encompassing large and giant DNA viruses infecting eukaryotes (Koonin et al. 2020). Members of the family Mimiviridae infecting amoeba have dsDNA genomes up to 1.5 Mb encoding more than 1000 proteins, including a complete glycosylation machinery (Raoult et al. 2004, Abergel et al. 2007, Abrahão et al. 2018, Notaro et al. 2021, 2022, Speciale et al. 2022). Mimivirus virion penetrate the cell through phagocytosis, and the acidic vacuole mediates opening of the stargate structure at one vertex of its icosahedral capsid (Zauberman et al. 2008, Schrad et al. 2020). The internal membrane unwraps and fuses with the phagosome membrane, allowing transfer of the nucleoid compartment into the host cytoplasm, while empty capsids remain in the vacuole (Raoult et al. 2004, Zauberman et al. 2008, Claverie and Abergel 2010). The infectious cycle occurs in the cytoplasm where a large viral factory is developed (Raoult et al. 2004, Suzan-Monti et al. 2007, Claverie and Abergel 2010, Mutsafi et al. 2010). At the late stage of the cycle, neo-synthesized virions bud at the periphery of the viral factory where they are filled with the genome. Finally, a glycosylated layer of fibrils composed of proteins and two large polysaccharides synthesized by the virally encoded machinery is added to the capsids (Kuznetsov et al. 2013, Notaro et al. 2021, de Aquino et al. 2023). As a result, the 750-nm diameter virions resemble Russian dolls made of the external layer of reticulated glycosylated fibrils (referred to as the “layer of fibrils”) decorating the surface of the icosahedral capsids. Underneath the capsid shell, the nucleoid compartment encases the 1.2 Mb dsDNA genome organized into a 30-nm large nucleocapsid-like structure (referred to as the “genomic fiber”). The genomic fiber is made of a protein shell internally lined by the folded DNA and a central channel that can accommodate large proteins such as the viral RNA polymerase (Villalta et al. 2022). Three independent genomic fiber structures have been determined by cryo-electron microscopy (cryo-EM): two compact 5- and 6-start DNA-containing helices, and a 5-start relaxed helix, without DNA (Villalta et al. 2022). Unexpectedly, the protein shell was found to be composed of two glucose–methanol–choline (GMC) oxidoreductases sharing 69% sequence identity, with a ratio of 5 between qu_946 and qu_143 according to the protein composition of the purified genomic fiber (Villalta et al. 2022). The resolution of the reconstructions (3.7 and 4.4 Å) prevented us from determining whether each helix contained a single paralog or a mixture of both. Interestingly, one of the two GMC-oxidoreductases, qu_143 (R135 in mimivirus prototype), was known to compose the external layer of fibrils at the surface of mimivirus capsids and it was hypothesized that it was the major target for glycosylation (Klose et al. 1993, Boyer et al. 2011, Notaro et al. 2021, Speciale et al. 2022). However, despite its involvement in both the mimivirus genomic fiber inside the nucleoid, and the layer of fibrils at the periphery of the icosahedral capsid, GMC-oxidoreductase homologs are absent in the laboratory-evolved mimivirus M4 strain (Boyer et al. 2011). M4 also lacks the glycosylation machinery, described for members of different clades of the subfamily Megamimivirinae and responsible for synthesizing and branching the polysaccharides on the capsids (Notaro et al. 2021, 2022). In the present study, we used in-house developed tools (Philippe et al. 2024) to delete mimivirus GMC-oxidoreductase genes. We then assessed the fitness cost associated with these deletions and investigated their impact on the formation of the genomic fiber and the protein and glycan composition of the layer of fibrils. Cryo-EM was used to determine the structure of the KO_946 genomic fiber made of qu_143, the less abundant in the wild-type (wt) genomic fiber. Nuclear magnetic resonance (NMR) and gas chromatography mass spectrometry (GC-MS) were used to analyze the compositions in glycans and their structures for each mutant and to compare them with the wt layer of fibrils. We used mass spectrometry (MS)-based proteomics to analyze, for each of the three mutants, the protein composition of their layer of fibrils and extended the study to members belonging to B and C clades (moumouviruses and megaviruses) known to glycosylate their layer of fibrils with different glycans using clade-specific glycosylation machineries (Notaro et al. 2022, Speciale et al. 2022). While confirming the non-essentiality of the two GMC-oxidoreductases, our results document the unexpected resilience of mimivirus to the deletion of these two genes through the use of alternative proteins to compensate their disruptions.
Materials and methods
Cloning of DNA constructs
A detailed protocol for gene manipulation of giant viruses and their host is provided in Philippe et al. (2024).
Gene knock-out vectors
The plasmid for gene knock-out was generated by sequential cloning of the 3’ UTR of mg_18 (megavirus chiliensis), the promoter of mg_741 (megavirus chiliensis) and a nourseothricin N-acetyl transferase (NAT) or a neomycin (NEO) selection cassette (Fig. 1A). Each cloning step was performed using the Phusion Taq polymerase (ThermoFisher) and InFusion (Takara). Finally, 500-bp homology arms were introduced at the 5’ and 3’ end of the cassette to induce homologous recombination with the viral DNA (Philippe et al. 2024). Before transfection, plasmids were digested with EcoRI and NotI. All primers are shown in Fig. S1.
Figure 1.
Mimivirus mutants’ generation and phenotypic characterization. (A) Schematic representation of the vector and strategy utilized for deletion of qu_143 (KO_qu143) and qu_946 (KO_qu946) in mimivirus reunion strain; goi: gene of interest. Selection cassette was introduced by homologous recombination and recombinant viruses were generated, as shown in (B). Primer annealing sites are also shown and the sequence of the primers is included in Table S1. (B) Graphic depicting the strategy for the selection of recombinant viruses. Viral infection was performed 1-h post-transfection; Ntc: Nourseothricin; P, passage. (C) Growth competition assays revealed no significant defects in the lytic cycle of deletion strains. The competition was also performed in the presence of Nourseothricin, which allows the outcompetition of the recombinant strains due to the expression of a Nourseothricin selection cassette. Measurements were performed by qPCR of an endogenous locus (present in wt and recombinant strains) and the Nourseothricin selection cassette (only present in recombinant viruses).
Establishment of viral lines
Gene knock-out
Gene knockout strategy was performed as previously described for pandoravirus (Bisio et al. 2023). Briefly, 1.5×105 Acanthamoeba castellanii cells were transfected with 6 µg of linearized plasmid using Polyfect (QIAGEN) in phosphate saline buffer (PBS). One hour after transfection, PBS was replaced with PPYG, and cells were infected with 1.5 × 107 mimivirus reunion particles for 1 h with sequential washes to remove extracellular virions. Next, 24 h after infection, the new generation of viruses (P0) was collected and used to infect new cells. An aliquot of P0 viruses was utilized for genotyping to confirm the integration of the selection cassette. Primers used for genotyping are shown in Table S1. A new infection was allowed to proceed for 1 h, then washed to remove extracellular virions and nourseothricin was added to the media. Viral growth was allowed to proceed for 24 h. This procedure was repeated one more time before removing the nourseothricin selection to allow viruses to expand more rapidly. Once the viral infection was visible, the selection procedure was repeated one more time (Fig. 1B). Viruses produced after this new round of selection were used for genotyping and cloning (Philippe et al. 2024). Double knockout of the GMC-oxidoreductases was obtaining by using a clonal population of qu_143 knockout viruses as parental strain. The locus of qu_946 was replaced by a neomycin resistance cassette. The transfection and selection of recombinant viruses’ procedure was performed identical to the process to generate single knockout, but replacing nourseothricin by geneticin.
Cloning and genotyping
Next, 150 000 A. castellanii cells were seeded on 6-well plates with 2 mL of PPYG. After adhesion, viruses were added to the well at a multiplicity of infection (MOI) of 1. One-hour post-infection, the well was washed five times with 1 mL of PPYG, and cells were recovered by well scraping. Amoebas were then diluted until obtaining a suspension of 1 amoeba/µL. Then 1 µL of such suspension was added in each well of a 96-well plate containing 1000 uninfected A. castellanii cells and 200 µL of PPYG. Wells were later monitored for cell death and 100 µL collected for genotyping (Philippe et al. 2024). Genotyping was performed using Terra PCR Direct Polymerase Mix (Takara) following the manufacturer's specifications. Primers used for genotyping are shown in Table S1.
Competition assay and quantitative PCR analysis
Equal infectious particles of wild-type and recombinant mimivirus reunion were mixed and used to infect A. castellanii at an approximate MOI of 0.8. Viruses were allowed to grow overnight in the presence or absence of nourseothricin. Subsequent viral progenies were used to infect new A. castellanii cells in reiterative passages. A fraction of each passage was collected for genomic DNA extraction.
Viral genomes were purified using Wizard genomic DNA purification kit (PROMEGA). To determine the amplification kinetic, the fluorescence of the EvaGreen dye incorporated into the PCR product was measured at the end of each cycle using SoFast EvaGreen Supermix 2× kit (Bio-Rad, France). A standard curve using the gDNA of purified viruses was performed in parallel with each experiment. For each point, a technical triplicate was performed. Quantitative real-time PCR (qRT-PCR) analyses were performed on a CFX96 Real-Time System (Bio-Rad) (Fig. 1C).
Genome sequencing and assembly of mutants’ genomes
Genomic DNA was extracted from 1010 virus particles using PureLink TM Genomic DNA mini kit (Invitrogen) according to the manufacturer's protocol. Clones of individual mutants and wt were sequenced on an illumina platform (Novogen). For mimivirus wt, we obtained 4 819 885 150 nt paired-end reads. For KO_qu946, KO_qu946 and 2KO, we obtained 4 662 744, 4 572 873 and 5 068 030 150 nt paired-end reads, respectively. Genomes were assembled using spades v. 3.13, with the option “careful” and we obtained five contigs for the wt, four for KO_qu946 and seven for KO_qu143 and 2KO. The wt sequence was consistent with the original mimivirus reunion genome sequence (GI MW004169) and for the clone KO_qu946 the gene was interrupted from position 1 151 358 to 1 152 985 relative to wt genome (position 302 to 1741 in the corresponding qu_946 gene) and for the clone KO_qu143 the gene was interrupted from position 165 934 to 167 572 relative to wt genome (position 306 to 1741 in the corresponding qu_143 gene). The region is replaced in both cases by the nourseothricin cassette sequence (1637 nt). For the 2KO, the gene qu_143 is interrupted from positions 306 to 1741 (165 934 to 167 572 on the wt genome) and replaced by the nourseothricin cassette sequence (1637 nt), and gene qu_946 is interrupted from positions 302 to 1206 (1 151 551 to 1 153 410 on the genome) and replaced by the geneticin cassette sequence (1859 nt). To confirm the deletion of each mutant, the reads were mapped on the wt genome resulting in homogeneous coverage along the genome, except for the qu_143 and qu_946 central positions, which are covered. In addition, we used the central part of the GMC-oxidoreductase genes (deleted in mutants) as blast queries against the reads of each mutant genome, which also confirmed the absence of the central region.
Extraction and purification of the qu_946 and qu_143 mutants' genomic fiber
The genomic fiber of the mimivirus reunion single mutants of qu_946 (KO_qu946) and qu_143 (KO_qu143) were extracted as described in Villalta et al. (2022) for the wt virus. The genomic fiber was extracted from 12 mL of purified single deletion mutant virions at 2 × 1010 particles/mL, split into 12×1 mL samples processed in parallel. Trypsin (Sigma T8003) in 40 mM Tris-HCl pH 7.5 buffer was added at a final concentration of 50 µg/mL and the virus-enzyme mix was incubated for 2 h at 30°C in a heating dry block (Grant Bio PCH-1). DTT was then added at a final concentration of 10 mM and incubated at 30°C for 16 h. Finally, 0.001% Triton X-100 was added to the mix and incubated for 4 h at 30°C. Each tube was centrifuged at 4000 x g for 5 min to pellet the opened capsids. The supernatant was recovered and concentrated by centrifugation at 15 000 x g for 4 h at 4°C. Most of the supernatant was discarded, leaving 12x ∼200 µL of concentrated broken pieces of genomic fiber that were pooled and layered on top of ultracentrifuge tubes of 4 mL (polypropylene centrifuge tubes, Beckman Coulter) containing a discontinuous cesium chloride gradient (1.4, 1.3, 1.2 g/cm3 in 40 mM Tris-HCl pH 7.5 buffer). The gradients were centrifuged at 200 000 x g for 16 h at 4°C. Because no visible band was observed, successive 0.5 mL fractions were recovered from the bottom of the tube. Each fraction was dialyzed using 20 kDa Slide-A-Lyzers (ThermoFisher) against 40 mM Tris-HCl pH 7.5 to remove the CsCl. These fractions were further concentrated by centrifugation at 15 000 x g, at 4°C for 4 h, and most of the supernatant was removed, leaving ∼100 µL of sample at the bottom of each tube. At each step of the extraction procedure, the sample was imaged by negative staining transmission electron microscopy (NS-TEM) to assess the integrity of the genomic fiber. Each fraction of the gradient was finally controlled by NS-TEM.
Negative staining TEM
The 300-mesh ultra-thin carbon-coated copper grids (Electron Microscopy Sciences) were prepared for negative staining by adsorbing 4–7 µL of the sample for 3 min, followed by blotting excess liquid and staining for 2 min in 2% uranyl acetate to image the genomic fiber. For fibrils and mutant virions, staining was performed with a drop of 1% uranyl followed by blotting after 10–15 s, and a drop of uranyl acetate coupled with methylcellulose (2% and 0.2%, respectively) was added twice and left for 5–10 s before blotting.
The grids were imaged either on an FEI Tecnai G2 microscope operated at 200 keV and equipped with an Olympus Veleta 2k camera (IBDM microscopy platform, Marseille, France); an FEI Tecnai G2 microscope operated at 200 keV and equipped with a Gatan OneView camera (IMM, microscopy platform, France) (Fig. 2).
Figure 2.
Micrograph of negative stained genomic fiber of (A) wild-type mimivirus, (B) KO_qu143 mutant, (C) KO_qu946 mutant and (D) 2KO double mutant. Scale bars 100 nm.
Single-particle analysis by cryo-EM
Sample preparation
For the KO_qu946 mutant, 3 µL of the purified sample was applied to glow-discharged Quantifoil R 2/1 Cu grids, blotted for 2 s using a Vitrobot Mk IV (Thermo Scientific) and applying the following parameters: 4°C, 100% humidity and blotting force 0 then plunged frozen in liquid ethane/propane cooled to liquid nitrogen temperature.
Data acquisition
Grids were imaged using a Titan Krios (Thermo Scientific) microscope operated at 300 keV and equipped with a K3 direct electron detector and a GIF BioQuantum energy filter (Gatan). Next, 2224 movie frames were collected using EPU software (Thermo Scientific) at a nominal magnification of 81 000x with a pixel size of 1.0859 Å and a defocus range of −0.6 to −2.8 µm. Micrographs were acquired using EPU (Thermo Scientific) with 2.3 s exposure time, fractionated into 40 frames and 18.25 e−/pixel/s (total fluence of 35.597 e−/Ų).
2D classification and clustering of 2D classes
All movie frames were aligned using MotionCor2 (Zheng et al. 2017) and used for contrast transfer function (CTF) estimation with CTFFIND-4.1 (Rohou and Grigorieff 2015). Helical segments of the purified genomic fiber, manually picked with Relion 3.1.0, were extracted with 400-pixel box sizes (decimated to 100 pixels) using a rise of 7.93 Å and a tube diameter of 300 Å. Particles were subjected to reference-free 2D classification in Relion 3.1.0 (Scheres 2012, He and Scheres 2017). We then performed additional cluster analysis of the initial 2D classes provided by Relion to aim for more homogeneous clusters (Villalta et al. 2022), eventually corresponding to different states (Figs 3 and 4).
Figure 3.
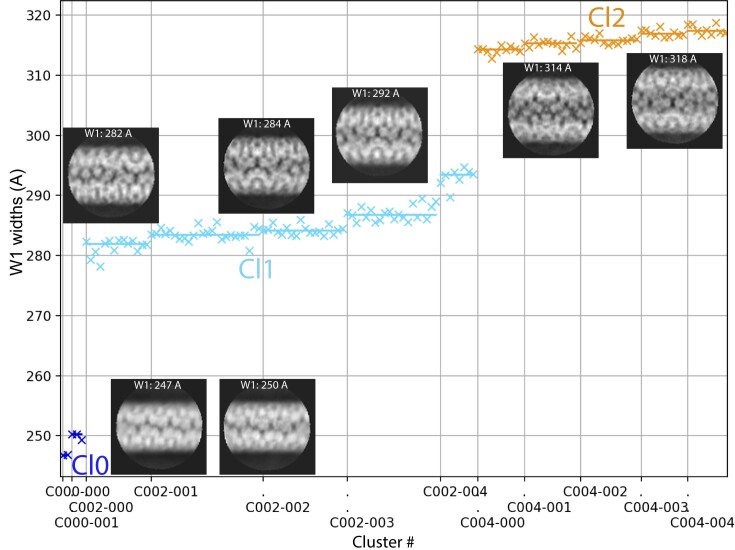
Clustering of the 2D classes obtained with qu_143 genomic fiber. Automatic sorting of the 2D classes using the fiber width W1 and pairwise correlations of the 2D classes resulted in two main clusters (5-start Cl1 in cyan; 6-start Cl2 in orange) and a smaller cluster (Cl0 in dark blue). Each cross corresponds to a 2D class and its associated W1. Representative 2D classes are displayed with their respective W1.
Figure 4.
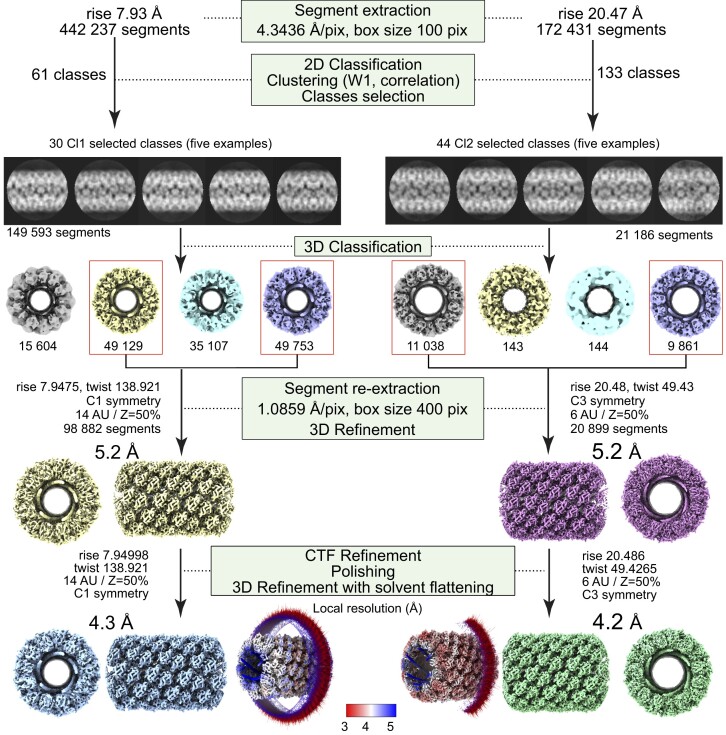
Workflow of the 5- and 6-start helices reconstruction processes. Segment extraction was performed with a box size of 400 pixels (pix) binned (box size 100 pix, 4.3436 Å/pix). The distance between consecutive boxes was equal to the axial rise calculated by indexation of the power spectrum. After clustering, 2D classes were selected for Cl1 and Cl2 and 3D classification was carried out using the selected segments, helical symmetry parameters from the power spectrum indexation and a 300 Å or 330 Å featureless cylinder as 3D reference for Cl1 and Cl2, respectively. 3D refinement of the two boxed 3D classes was achieved using one low pass filtered 3D class as reference on the unbinned segments. A first 3D refinement was performed with solvent flattening followed by CTF refinement and polishing of the selected segments. A last 3D refinement was achieved with solvent flattening. The EM maps colored by local resolution from 5 Å (blue) to 3 Å (red) with Euler angle distribution of the particles used for the 3D reconstruction are presented.
Identification of candidate helical parameters
Fourier transform analysis methods were used to confirm the helical parameters were the same as in wt (Diaz et al. 2010, Sachse 2015, Coudray et al. 2016, Villalta et al. 2022) for the Cl1 (Cl1a in wt) and Cl2.
Cryo-EM data processing and 3D reconstruction
After determining the helical parameters, segments were extracted with a box size of 400 pixels (decimated to 100 pixels) using the proper rises for the Cl1 (7.93 Å, cylinder 300 Å, 442 237 segments) and the Cl2 (20.47 Å, cylinder 330 Å, 172 431 segments). A dedicated 2D classification protocol was performed independently on each extraction. For the Cl2, one round of 50 expectation-maximization (E-M) iterations was sufficient to produce 133 homogeneous 2D classes submitted to cluster analysis and 44 2D classes were selected (21 186 segments). For the Cl1 extraction, three iterative 2D classification/selection rounds were performed (25, 50 and 100 E-M iterations) producing 61 classes, from which 30 (149 593 segments) were finally selected based on the cluster analysis.
Values of the helical parameters, rise and twist (Cl1: 7.9475 Å, 138.921°; Cl2: 20.48 Å, 49.43°), were then used for Relion 3D classification (Scheres 2012, He and Scheres 2017), with a +/−0.5 units freedom search range, using a featureless cylinder as initial reference (diameter of 300 Å and C1 symmetry for Cl1 and 330 Å and C3 symmetry for Cl2). In real space, the helical symmetry was searched using 50% of the central part of the box for both structures. The number of asymmetrical units in each segment box was set to one for Cl1 and to six for Cl2. The entire helical reconstruction and protein shell dimensions were obtained using an in-house developed program.
The superimposable 3D classes (same helical parameters, same helix orientation) were then selected, reducing the dataset to 98 882 segments for the 5-start helix (Cl1) and 20 899 segments for the 6-start helix (Cl2). After re-extraction of the selected segments without scaling, a first unmasked 3D refinement was performed with a rescaled 3D classification output low pass filtered to 15 Å as reference, followed by a 3D refinement step using solvent flattened, Fourier shell correlation (FSCs) and CTF refinement using the standard procedure described in Relion. To further improve the resolution of the maps, Bayesian polishing was applied using 10 000 segments for training and default values for polishing. A last round of 3D refinement with solvent flattening was applied to the previous refined map using the polished particles. At that point the maps were sufficiently resolved (Cl1: 4.3 Å, Cl2: 4.2; FSC threshold 0.5) to identify secondary structure elements (with visible cylinders corresponding to the helices) and were used to compute local resolution.
Structures refinement
The best resolution Cl2 map was used to fit the qu_143 dimeric structure (PDB 7YX3) using UCSF ChimeraX 1.5 (Pettersen et al. 2004). Each monomer was then rigid-body fitted independently into the map. The entire protein was then inspected within coot 0.9.7 (Emsley et al. 2010) to fix local inconsistencies and was further refined against the map using the real-space refinement program in PHENIX 1.20.1 (Liebschner et al. 2019). The protein was submitted to five cycles of rigid body refinement (with each chain defined) followed by twice 10 cycles of refinement with the default PHENIX options. The resulting structure was manually corrected within coot. The resulting protein model was submitted to the same steps refinement in PHENIX. This final model was then fitted into the Cl1 map, inspected with coot and refined using five cycles of rigid body refinement and simulated annealing, followed by twice 10 cycles of refinement with the default PHENIX options. Validations were performed with PHENIX using the comprehensive validation program (Table S3). RMSDs between different structures (monomers and dimers, Table S4) was computed using the align procedure in Pymol suite (The PyMOL Molecular Graphics System, Version 3.0 Schrödinger, LLC. as recommended by the authors: https://pymol.org/support.html) (PyMOL).
Extraction and purification of the mutants’ fibrils
To analyze the glycan composition and polysaccharides structures of the three mutants and wt mimivirus reunion strain, we applied an already described protocol (Notaro et al. 2021). Briefly, 4 × 1011 viral particles were centrifuged at 14 000 g for 10 min, the supernatant was discarded and the pellet was re-suspended in 10 ml of 50 mM DTT. Fibril extraction was performed at 100°C under stirring. After 2 h, the tube was centrifuged at 14 000 g for 15 min, at 4°C, and the fibrils recovered with the supernatant. The fibrils were then dried and purified on Biogel P10, followed by subsequent NMR analysis of each mutant and wt.
We also developed a softer defibrillation protocol to recover the fibrils without contaminating them with proteins released by damaged virions in order to analyze the fibrils' protein composition by MS-based proteomics. Purified virions (1.5 × 1010) were incubated in Eppendorf tubes in 500-µL 40 mM Tris-HCl pH 7.5 buffer, 500 mM DTT for 2 h at 30°C. Tubes were then centrifuged at 14 000 g for 10 min. The supernatants containing the fibrils were recovered and concentrated on vivaspin® 3 KDa (Sartorius, VS04T91) at 3000 g. The pellet was washed twice with 40 mM Tris-HCl pH 7.5 buffer and centrifuged at 14 000 g for 10 min and finally resuspended in the same buffer. Intact virions, pellets and fibrils were imaged by NS-TEM to assess the integrity of the defibrillated virions in the pellet and the presence of fibrils in the supernatant. For the Nqu143-GFP mutants, defibrillated virions were also observed by fluorescence microscopy, which confirmed the absence of fluorescence due to the removal of the GFP together with the layer of fibrils.
Sugar composition of viral particles of Mimivirus reunion wt and mutants
Monosaccharide composition analyses as acetylated methyl glycoside (AMG) were performed on the intact viral particles (1.25 × 1010, ∼250 µl) of mimivirus reunion wt and mutants, following the procedure reported by De Castro et al. (2010). The obtained AMG was analyzed via gas chromatography-mass spectrometry (GC-MS) on an Agilent instrument (GC instrument Agilent 6850 coupled to MS Agilent 5973) equipped with a SPB-5 capillary column (Supelco, 30 m × 0.25 i.d., flow rate, 0.8 mL min–1) and He as the carrier gas. The identification of the monosaccharides derivatized as AMG was obtained by studying the fragmentation pattern corresponding to each peak of the chromatogram and by comparison with suitable standards.
Purification and 1H NMR analysis of the fibrils
The fibrils of mimivirus reunion wt and mutants, extracted as reported above, were purified to remove the DTT used for the extraction procedure.
Briefly, the glycoproteins (protein/s carrying the polysaccharides) were precipitated with cold acetone at 80%, at 4°C, for 16 h, twice. The supernatant containing DTT and salts was discarded, while the precipitate was dissolved in water and freeze-dried. Then the precipitate was purified by size exclusion chromatography (Biogel P10, flow: 12 ml/h) to completely remove the DTT. The eluted fractions were checked by 1H NMR, revealing that the glycan-containing material was eluted at one-third of the column volume (full spectra are shown in Fig. 5B).
Figure 5.
Compositional and NMR analysis of the fibrils of mimivirus reunion strain wt and mutants. (A) GC-MS chromatogram profiles of the sugars composing the fibrils of wt (a), KO_qu946 (b), KO_qu143 (c) and 2KO (d). (B) Comparison of the 1H NMR spectra of mimivirus reunion strain wt and related mutants with that of mimivirus prototype strain. The anomeric signals related to poly_1 (C and D units) are in black, while those of poly_2 are in red. (C) Structures of mimivirus polysaccharides as reported in Notaro et al. (2021).
The 1H NMR measurements were carried out on a 600-MHz Bruker instrument, equipped with a CryoProbe™ at 310 K. The intensity of the solvent signal was reduced by measuring a mono-dimensional DOSY spectrum, setting δ and Δ to 2.4 and 100 ms, respectively, and the variable gradient to 50% of its maximum power. Spectra were processed and analyzed using the Bruker TopSpin 4.0.9 program.
In the 1H NMR spectra, the anomeric region (Speciale et al. 2022) (5.5–4.4 ppm) perfectly overlapped with that of the previously studied mimivirus strain (Notaro et al. 2021) (Fig. 5B, Fig. S7), confirming that the different sugars were assembled the same way as in the reference mimivirus to produce the same two polysaccharides (Fig. 5C). The signal at 4.80 ppm is the result of the overlap of two anomeric protons, one of a 3-α-Rha (labeled C) and one of a 3-β-GlcNAc (D) modified with pyruvic acid at the hydroxyl functions 4 and 6. These two residues are the building blocks of the repeating unit of polysaccharide 1 (poly_1, Fig. 5C) (Notaro et al. 2021). The other anomeric signals, labeled with the capital letters A, A’ (2,3-α- L-Rha), B (3-β-D-GlcNAc), E (2OMe-β-D-VioNAc) and E’(β-D-Vio4NAc), are part of the polysaccharide 2 (poly_2) that presents a backbone with a disaccharide repeating unit of 2)-α-L-Rha-(1→3)-β-D-GlcNAc-(1→, with the rhamnose residue further substituted with a viosamine methylated at position 2 (E) or not methylated (E'unit), thus taking the labels A and A’, respectively (Fig. 5C) (Notaro et al. 2021).
Mass spectrometry-based proteomic analyses
Proteins were solubilized with Laemmli buffer (four volumes of sample with one volume of Laemmli 5X—125 mM Tris-HCl pH 6.8, 10% SDS, 20% glycerol, 25% β-mercaptoethanol and traces of bromophenol blue) and heated for 10 min at 95°C. The extracted proteins were stacked in the top of an SDS-PAGE gel (4–12% NuPAGE, Life Technologies), stained with Coomassie blue R-250 (Bio-Rad) before in-gel digestion using modified trypsin (Promega, sequencing grade) as previously described (Casabona et al. 2013). The resulting peptides were analyzed by online nanoliquid chromatography coupled to tandem MS (UltiMate 3000 RSLCnano and Q-Exactive Plus or Q-Exactive HF, Thermo Scientific). Peptides were sampled on a 300 µm x 5 mm PepMap C18 precolumn and separated on a 75 µm x 250 mm C18 column (Reprosil-Pur 120 C18-AQ, 1.9 µm, Dr. Maisch, except for KO_qu143, KO_qu946 and Nqu143-GFP mutant samples separated on Aurora, 1.7 µm, IonOpticks) using a 140-min gradient (except for fibrils from Nqu143-GFP mutant, for which a 60-min gradient was used). MS and MS/MS data were acquired using Xcalibur 4.0 (Thermo Scientific). Peptides and proteins were identified using Mascot (version 2.8.0, Matrix Science) through concomitant searches against a homemade A. castellanii protein sequence database, homemade virus-specific protein sequence databases, and a homemade database containing the sequences of classical contaminant proteins found in proteomic analyses (human keratins, trypsin…). Trypsin/P was chosen as the enzyme and two missed cleavages were allowed. Precursor and fragment mass error tolerances were set at 10 and 20 ppm, respectively. Peptide modifications allowed during the search were: Carbamidomethyl (C, fixed), Acetyl (Protein N-term, variable) and Oxidation (M, variable). Proline software version 2.2.0 (Bouyssié et al. 2020) was used for the compilation, grouping and filtering of the results: conservation of rank 1 peptides, peptide length ≥ 6 amino acids, peptide-spectrum-match identification false discovery rate < 1% (Couté et al. 2020) and a minimum of one specific peptide per identified protein group. Proline was then used to perform a MS1-based quantification of the identified protein groups. Intensity-based absolute quantification (iBAQ) (Schwanhäusser et al. 2011) values were calculated for each protein group from the MS intensities of razor and specific peptides (Table S2). The relative abundance of individual proteins in virions and fibrils was calculated as the ratio of the individual protein iBAQ values to the sum of the iBAQ values of all proteins in each sample. The relative enrichment of individual proteins between virions and fibrils was calculated as the ratio of their relative abundances in each fraction (Villalta et al. 2022).
Results
Neither of the two GMC-oxidoreductases is essential
We used our recently developed protocol (Philippe et al. 2024) combining homologous recombination with the introduction of a NAT selection cassette to delete each of the two genes encoding the GMC-oxidoreductases (qu_946 and qu_143). We selected recombinant viruses that were cloned to obtain homogeneous populations (Fig. 1B) (Philippe et al. 2024). Each mutant was easily produced and genotyped to confirm the mutation (Fig. S1). Using a second resistance gene (NEO selection cassette) we were able to delete both genes (Fig. S1), demonstrating that the two GMC-oxidoreductases were not essential. The absence of additional mutations in every mutant was confirmed by genome sequencing.
Mutants’ fitness
To assess whether a fitness cost was associated with the mutations, we performed competition assays against wt mimivirus reunion strain by measuring the abundance of each mutant over several cycles in the presence and absence of selection. In contrast to the wt, each mutant presents a common nourseothricin resistance gene. The double deletion mutant (2KO) encodes for an additional geneticin resistance gene. As a result, in the presence of nourseothricin, each mutant ratio increased, with the disappearance of the wt virus after five passages (Fig. 1C). These data allowed competition assays to be validated as an effective tool to assess the fitness of recombinant viruses. In the absence of selection, the relative abundance of the mutants compared with wt remained ~0.5 over five passages, supporting the absence of a fitness cost, even for the double deletion mutant (Fig. 1c).
Composition of the genomic fiber of single mutants
To determine the composition of the genomic fiber of each single mutant we extracted and purified their genomic fiber. MS-based proteomics confirmed that the most abundant protein was the remaining GMC-oxidoreductase. NS-TEM highlighted surprising differences between the two structures (Fig. 2), despite the fact that the two proteins (qu_946 and qu_143) share 69% sequence identity (81% similarity). Specifically, the genomic fiber extracted from the KO_qu143 mutant and made of qu_946 (the most abundant in the wild type genomic fiber) was mostly in the unwound state (Fig. 2B), while the one made of qu_143 (KO_qu946) resulted in very long and stable helices that did not unwind (Fig. 2c). Thus, the use of both GMC-oxidoreductases in the wt genomic fiber could contribute to a fine tuning of its biophysical properties, with an intermediate state between the wt and each deletion mutant (Fig. 2A). In the case of the double mutant, the protocols for capsid opening to extract the genomic fiber of wt or single mutants did not work properly. An optimized protocol allowed the extraction of a possible thinner genomic fiber, but in poor yield, preventing its purification and compositional characterization (Fig. 2D).
Cryo-EM single particle analysis of the qu_143 genomic fiber
To determine the contribution of each GMC-oxidoreductase to the wt genomic fiber we performed cryo-EM single-particle analysis on the most stable genomic fiber composed by qu_143 (mutant KO_qu946). As for the wt, the 2D classification revealed an heterogeneity of the sample, and the 2D classes were sorted by applying our already described clustering protocol (Villalta et al. 2022). The two main clusters corresponding in width to the compact (Cl1a) and (Cl2) structures of the wt genomic fiber were respectively named Cl1 and Cl2 (Fig. 3).
For each main cluster, we confirmed that the helical symmetry parameters were the same as the wt genomic fiber and proceeded to structure determination and refinement (Fig. 4). For the less populated smaller cluster (Cl0, ∼25 nm), absent from the wt genomic fiber 2D classes, we failed to identify its helical parameters due to the lower number of segments and the resulting lower resolution. After 3D refinement, we obtained a 4.3 Å resolution helical ∼29 nm diameter structure (FSC threshold 0.5, masked) for Cl1. This structure corresponds to the same 5-start left-handed helix as the wt (Cl1a), made of an ∼8-nm thick proteinaceous external shell with five dsDNA strands lining the interior of the shell and an ∼9-nm wide central channel (Fig. 4, Fig. S2). The 4.2 Å resolution Cl2 map obtained after 3D refinement (Fig. 4, Fig. S2) corresponds to the same ∼32-nm diameter 6-start left-handed helix as the wt, with six dsDNA strands lining the external shell and an ∼12-nm wide inner channel (Fig. 4).
Because the helical parameters between the wt Cl1a and the mutant Cl1 are the same, we used the qu_143 dimeric structure refined in the Cl1a focus refined map for refinement into the mutant maps (Materials and Methods section and Table S3). The density that can be attributed to the FAD cofactor was present in both maps (Fig. S3) and the models of Cl1 and Cl2 dimers are superimposable with a core RMSD of 0.467 Å based on Cα atoms (Table S4). As the qu_143 helices are more stable than the wt (composed of both GMC-oxidoreductases), the relaxed Cl3 cluster was never observed with this mutant.
Model explaining the co-existence of 5- and 6-start helices
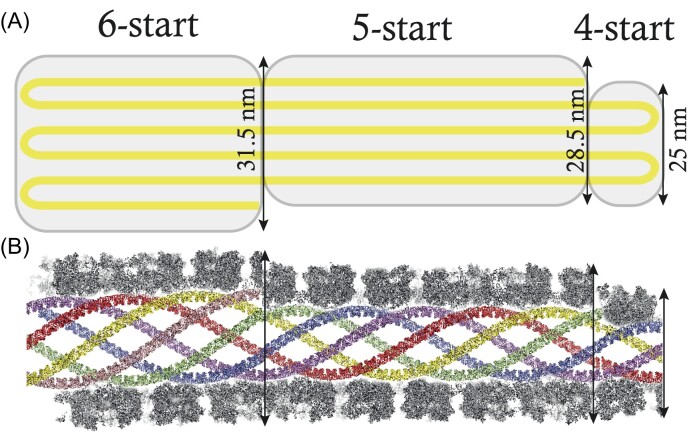
As for the wt, the genomic fiber of KO_qu946 is composed of a mixture of five and six strands of DNA, despite the presence of a single GMC-oxidoreductase in the shell. We estimated the ratio of 5-start and 6-start from the clustering (Fig. 3) and can now propose a model that reconciles the co-occurrence of the two structures. In this model, the whole genome would be folded into six parallel strands, five longer than the sixth one. The helix would then be formed initially as a 6-start helix until the sixth strand ends and, from that point, becomes a 5-start helix (Fig. 6). According to this model, assuming the length of the genomic fiber is limited by the size of mimivirus nucleoid compartment, we can estimate that the maximum genome length would be ∼1.4 Mb for a full 6-start helix with an 8-nm thick protein shell. We hypothesize that the last cluster (Cl0, Fig. 3) could correspond to a 4-start with an additional shorter DNA strand.
Figure 6.
Model explaining the transition from a 6- to a 5-start helix. (A) Flat models of the transition from a 6- to a 5-start involving a decrease of the helix diameter by ∼3 nm. The small cluster could thus correspond to a 25-nm diameter 4-start helix. (B) Model of the different helices. A longitudinal section of the GMC-oxidoreductase shell is represented around the central channel and each helix was positioned to produce a certain continuity of the DNA strands.
The N-terminal cys-pro-rich domain is an addressing domain to the virion surface
The sequence of the cys-pro-rich N-ter domain of the GMC-oxidoreductases is not covered by proteomic analysis of the purified genomic fiber but is covered by peptides in the purified fibrils forming the external layer at the surface of the capsids (Villalta et al. 2022). To assess whether this cys-pro-rich N-ter domain could be a structural signature used to address proteins to the layer of fibrils, we replaced the second GMC-oxidoreductase (qu_143) in the genome of the mutant KO_qu946 by the sequence of the GFP in fusion with the sequence of the qu_143 N-terminal domain (Nqu143-GFP). We then analyzed the resulting virions by MS-based proteomics (Table 1 and Table S2) and fluorescence microscopy. Purified Nqu143-GFP virions showed a strong fluorescence supporting the incorporation of the chimeric protein into the viral particles (Fig. 7). Moreover, after defibrillation of the virions, the GFP fluorescence was lost. In addition, Nqu143-GFP was identified in purified virions (ranked 97th in terms of relative abundance, Table S2) and was found enriched 12 times in the fraction containing the external fibrils (ranked 20th, Table 1 and Table S2) with peptides covering the N-terminal domain (Fig. S5). Taken together, these data indicate that the N-terminal cys-pro-rich domain of the GMC oxidoreductase is sufficient to direct the proteins to the layer of fibrils at the surface of mimivirus particles.
Table 1.
Rank of the most abundant proteins in the purified fibrils of members of clade A, B and C.
Figure 7.
Fluorescent virion (A) images and (B) quantification in infected cells overexpressing GFP (Vc2) or in cells infected by the mutant Nqu143-GFP. Scale bar 5 µm. (CTVF: corrected total virion fluorescence).
Composition of the purified external fibrils
Protein composition of the layer of fibrils in additional members of the family Mimiviridae
The members of the Megavirinae subfamily (Gallot-Lavallée et al. 2017) currently encompass five clades, the mimiviruses (A clade), moumouviruses (B clade), megaviruses (C clade), tupanviruses (D clade) (Abrahão et al. 2018) and cotonvirus japonicus (E clade) (Takahashi et al. 2021). As of today, all members of the Megavirinae are characterized by a layer of fibrils that differs in thickness and lengths among the clades and, except for clade A (Boyer et al. 2011, Villalta et al. 2022), their protein composition is unknown. While the cys-pro-rich N-ter domain of the GMC-oxidoreductases is conserved in all members of the A clade, it is absent in the orthologous proteins in members of the B and C clades. Moreover, homologs of the two GMC-oxidoreductases are pseudogenized in tupanviruses, suggesting that different proteins should compose their fibrils (Fig. S6). We thus conducted a systematic proteomic analysis of the purified fibrils of different members of the three clades, in addition to the mutants. For the double mutant (2KO) we identified a group of proteins as the most abundant in the fibrils fraction (qu_734, qu_757, qu_384 and qu_482, Table 1 and Table S2). These proteins were also highly ranked in the Nqu143-GFP double mutant fibrils and the orthologous protein of qu_734 was identified as the most abundant protein in the laboratory-evolved M4 mutant fibrils (696-L688, Table 1 and Table S2). This mutant does not encode a glycosylation machinery and is not glycosylated (Boyer et al. 2011, Notaro et al. 2021), thus its capsid lacks the large reticulated layer of fibrils decorating the mimivirus capsid (Fig. S8f). Interestingly, the qu_734 protein also possesses a cys-pro-rich N-ter domain that is conserved in all clades (Fig. S6). It was not possible to determine if one of these proteins was also the building block composing the 2KO genomic fiber, given the difficulty in opening the 2KO capsids. Thus we concluded that the change in protein composition of the fibrils led to capsids with different stability properties. While, as for the wt, the most abundant protein in KO_qu946 remains qu_143, the lack of qu143 in KO_qu143 does not lead to its replacement by the second GMC-oxidoreductase. Instead, it is replaced by a group of proteins, with qu_465 (predicted as a thioredoxin domain containing protein) as the most abundant, followed by qu_757, the second most abundant in the wt fibrils (Table 1 and Table S2).
The proteomic analysis of fibrils purified from two isolates of the B clade, moumouvirus australiensis and maliensis, showed similar protein compositions, but with slight differences in their relative abundances (Table 1 and Table S2). The moumouvirus maliensis protein mm_751, ranked first in its fibrils, is absent from A clade fibrils and is in the top three in the fibrils of members of the B and C clades. Cystein and proline amino acids are present in the N-ter domain of mm_751, but less abundant than in the GMC-oxidoreductases. For ma_195 (qu_738 in mimivirus reunion, Table 1 and Table S2), ranked first in the fibrils of moumouvirus australiensis, the N-ter domain is not cys-pro-rich. The best ranked in the fibrils of members of the C clade are the same as for members of the B clade. The first ranked in megavirus chilensis is mg749 (qu_657 in mimivirus reunion), which presents a cys-pro-rich N-ter domain, while the first ranked in megavirus vitis, mvi_646 (qu_600 in mimivirus reunion), lacks a cys-pro-rich N-ter domain. Yet, for each virus in each clade, there is at least one protein with a cys-pro-rich N-terminal domain among the most abundant in the fibrils.
Glycan composition of the fibrils in mimivirus mutants
We recently established that the fibrils were coated with glycans (Notaro et al. 2022, Speciale et al. 2022) and demonstrated that for mimivirus, the prototype of the A clade, they were made of two distinct large molecular weight polysaccharides (Notaro et al. 2021). Because the cluster of 12 genes responsible for the biosynthesis of the polysaccharides is conserved in the A clade (Notaro et al. 2022), we hypothesized that the fibrils of the mimivirus reunion strain would have the same composition as mimivirus. To assess whether the knockout of the most abundant protein composing the fibrils could also affect the branching of these polysaccharides and their composition, we analyzed the sugar content of the viral particles of all three mimivirus reunion mutants (KO_qu946, KO_qu143 and 2KO) together with the wt strain (as reported in Notaro et al. 2021, 2022). The chemical characterization revealed for each of them the presence of sugars (Fig. 5A), with rhamnose (Rha), viosamine (Vio), 2-OMe-Vio, glucose (Glc) and glucosamine (GlcN) (Fig. 5A), confirming that mimivirus reunion strain and all mutants had the same glycan composition as mimivirus prototype (Notaro et al. 2022). We then analyzed the fibrils of the mutants and wt by 1H NMR spectroscopy (Speciale et al. 2022) and compared them with the mimivirus prototype, which confirmed that the different sugars were assembled the same way as in the reference mimivirus to produce the same two polysaccharides (Fig. 5, Fig. S7) (Notaro et al. 2021). NS-TEM images of the virions of the different mutants were obtained after methylcellulose staining to assess whether the mutations changed the layer of fibrils appearance, which would suggest that the change in protein impacted their level of glycosylation (Hacker et al. 2016). All mutants’ virions showed the same cross-linked outer layer, suggesting that despite their differences in protein composition, their glycosylation was not affected (Fig. S8).
It was hypothesized that mimivirus R135 GMC oxidoreductase (qu_143 in mimivirus reunion) was the major target for glycosylation in the surface fibrils of mimivirus (Boyer et al. 2011, Notaro et al. 2021). The results presented here indicate that the mutations, including the deletion of the two GMC-oxidoreductase genes, did not affect the surface glycosylation. These data suggest that the GMC-oxidoreductases are not the main target for glycosylation. Alternatively, it is also possible that the glycosylation machinery was able to use the qu_734 protein as support for the two polysaccharides. As expected, no glycans were identified in the laboratory-evolved M4 mutant layer of fibrils.
Discussion
Genome packaging is essential for the propagation of viruses. For instance, the packaging ATPase of poxviruses (Cassetti et al. 1998) or the histones of marseilleviruses (Liu et al. 2021, Valencia-Sánchez et al. 2021) are essential for the productive infection by their virions. In addition, proteins at the surface of viral capsids usually play a central role in interacting with the host cell surface and the initial steps of infection (Sobhy 2017). The two processes, genome encapsidation and viral entry, are thus essential. As GMC-oxidoreductases compose both the genomic fiber and the surface fibrils (Villalta et al. 2022), we could have expected them to be essential. However, these two enzymes are absent in the laboratory-evolved mimivirus M4 strain that also lost the glycosylation machinery (Boyer et al. 2011). Our recent implementation of genetics tools for cytoplasmic giant viruses provided the first opportunity to directly assess the impact of the GMC-oxidoreductases deletions in mimivirus reunion strain (Philippe et al. 2024). Given the homology between the two enzymes, the deletion of the most abundant one in the wt genomic fiber induced its replacement by the second one, with no apparent cost for the virus, under laboratory conditions. Yet, the single mutant genomic fibers were significantly different compared with the wt (Fig. 2), indicating a certain degree of functional specialization for each GMC-oxidoreductase. While the wt genomic fiber was highly heterogeneous, with fully relaxed helices having lost DNA (Villalta et al. 2022), the KO_qu946 genomic fiber appeared more stable, with no occurrence of fully relaxed structures. By contrast, the deletion of qu_143 seemed to increase the genomic fiber instability, suggesting a stabilizing role for this second GMC-oxidoreductase (Fig. 2). The cryo-EM analysis of the KO_qu946 genomic fiber structure, as for the wt, produced the same two 5- and 6-start helices, but composed of a single GMC-oxidoreductase. This led us to propose a model to explain the co-existence of both structures. In this model, the genome is folded into six strands before assembly into a 6-start helix. The first and the last strand can be shorter than the other strands, leading to a 5-start helix with five DNA strands and a 4-start helix with four DNA strands. Finally, the deletion of both GMC-oxidoreductases with no significant loss of viral fitness demonstrated that they were not essential.
The qu_143 GMC oxidoreductase is five times less abundant than qu_946 in the genomic fiber but it is ranked first in the layer of fibrils and is three times more abundant than the next one, qu_757. It was previously predicted as the main target for glycosylation (Klose et al. 1993, Boyer et al. 2011, Notaro et al. 2021), while qu_946 is only ranked 13th in the fibrils (Table S2). MS-based proteomics revealed that the cys-pro-rich N-ter domain of the GMC-oxidoreductase is present in the fibrils and cleaved in the genomic fiber (Villalta et al. 2022), but the protease and precise cleavage site remains to be identified. Interestingly, an additional mutant, in which this cys-pro-rich N-ter domain was fused to GFP, produced virions in which the GFP was identified at the surface of the virions and is enriched in the surface layer of fibrils. Further studies will be needed to elucidate the mechanism behind the recognition of this domain and its addressing to the fibrils. In addition, the virions of all mutants were decorated by long, reticulated fibrils, as evidenced by TEM (Fig. S8). Except for the KO_qu946 mutant, MS-based proteomic analysis of the fibrils revealed that they were composed of other proteins, some of which with a cys-pro-rich N-ter domain (Fig. S6). Finally, the glycan analysis of the GMC-oxidoreductases single and double mutants confirmed that their fibrils were still glycosylated by the same two polysaccharides as the wt virions.
In a previous study, it was reported that the glycosylation machinery was clade-specific and produced different glycans (Notaro et al. 2022). In the present study, the analysis of the purified fibrils of members of the B and C clades revealed they were also composed of different proteins, inter- and even intra-clade, and that B and C clade virions’ fibrils present a protein composition closer to each other than that of the A clade.
The glycosylated layer of fibrils bears glycans echoing bacterial ones, recognized by the amoeba that feeds on bacteria (Rodrigues et al. 2015, Notaro et al. 2022). They thus appear to be key for productive infection. In a given environment, the variable composition of the fibrils could be used to favor the engulfment of a given virion by a given amoeba, compared with other virions and even bacteria. The protein content of the layer of fibrils appears to have been optimized in a given clade, but their complex composition suggests that they can be made from a large set of diverse proteins. Having a flexible toolbox for building the external layer of fibrils would reflect the need to constantly adjust the capsid composition to outcompete other parasites and secure infection. We can thus hypothesize that in the population of virions resulting from a single infection, this composition might also be variable, helping out the virus to ensure the productive infection of at least one of the possible hosts present in that environment by at least one virion.
The question whether it is always the same protein that makes both the layer of fibril and the genomic fiber, even when other proteins than the GMC-oxidoreductases are used, remains unanswered. Yet the most abundant protein composing the external layer of fibrils of the 2KO and M4 corresponds to the 222 amino-acids qu_734 protein, which, according to the high confidence alphaFold (Jumper et al. 2021) prediction, could be almost twice as small (∼4.5 nm) than the GMC-oxidoreductase. From the previous study, the central part of the genomic fiber has to correspond to a central channel of at least 9 nm to accommodate proteins such as the RNA polymerase (Villalta et al. 2022). There is an additional 4 nm for the DNA lining the protein shell, and the height of the GMC-oxidoreductase is 8 nm, which makes the helical shell 16 nm, leading to an ∼29-nm helix. The protein making the shell is therefore defining the final dimension of the genomic fiber. For the 2KO, if this is the same protein that makes the external layer of fibrils and the shell of the genomic fiber, we expect a genomic fiber of 22 nm, compatible with the thinner structure observed in Fig. 2D (∼20–25 nm).
We have previously proposed an increased genome redundancy as a contributing factor to the appearance of viral gigantism (Bisio et al. 2023). The data presented here validate such a premise and extend these predictions outside of the Pandoraviridae. Overall, our results reveal the resilience of mimivirus, with redundant solutions securing essential functions such as infectiousness and genome packaging. Functional redundancy, well documented in the cellular world as a way to preserve essential function such as cell division (Erickson and Osawa 2010), and until now a hallmark of the optimized microorganisms, may thus also be at work in the viral world.
Supplementary Material
Acknowledgements
We thank Jean-Michel Claverie for his comments on the manuscript and discussions all along the project and Elsa Garcin for editing the revised manuscript. The data were collected at the Cryo-EM Swedish National Facility funded by the Knut and Alice Wallenberg, Family Erling Persson and Kempe Foundations, SciLifeLab, Stockholm University, and Umeå University under the expert assistance of Dr. Matthieu Coinçon. The preliminary electron microscopy experiments were performed on the PiCSL-FBI core facility (Aïcha Aouane, IBDM, AMU-Marseille), a member of the France-BioImaging national research infrastructure, sample freezing conditions and cryo-EM preliminary acquisitions were performed at the AFMB microscopy platform and on the IMM imaging platform (Dr. Artemis Kosta). We thank the PACA Bioinfo platform for computing support. The authors thank Dr. Matthias Fischer and Dr. Jônatas Santos Abrahão for their constructive comments during the peer review process.
Contributor Information
Jean-Marie Alempic, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Hugo Bisio, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Alejandro Villalta, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Sébastien Santini, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Audrey Lartigue, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Alain Schmitt, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Claire Bugnot, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Anna Notaro, Department of Agricultural Sciences, University of Naples Federico II, Via Università 100, 80055 Portici, Italy.
Lucid Belmudes, Univ. Grenoble Alpes, CEA, INSERM, UA13 BGE, CNRS, CEA, FR2048, 38000 Grenoble, France.
Annie Adrait, Univ. Grenoble Alpes, CEA, INSERM, UA13 BGE, CNRS, CEA, FR2048, 38000 Grenoble, France.
Olivier Poirot, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Denis Ptchelkine, Aix–Marseille University, Centre National de la Recherche Scientifique, Architecture et Fonction des Macromolécules Biologiques, Unité Mixte de Recherche 7257 (IM2B), 13288 Marseille Cedex 9, France.
Cristina De Castro, Department of Agricultural Sciences, University of Naples Federico II, Via Università 100, 80055 Portici, Italy.
Yohann Couté, Univ. Grenoble Alpes, CEA, INSERM, UA13 BGE, CNRS, CEA, FR2048, 38000 Grenoble, France.
Chantal Abergel, Aix–Marseille University, Centre National de la Recherche Scientifique, Information Génomique & Structurale (IGS), Unité Mixte de Recherche 7256 (Institut de Microbiologie de la Méditerranée, FR3479, IM2B, IOM), 13288 Marseille Cedex 9, France.
Author contributions
C.A. conceived and designed the research; J-M.A., H.B., A.V., S.S., A.L., A.S., C.B. A.N., L.B., A.A., O.P. C.D.C., Y.C. and C.A. performed research; D.P. granted access to microscopy platform and helped optimizing sample freezing with A.V.; J-M. A and A.L. developed methodologies for genomic fiber and fibrils extraction and purification; H.B. developed the genetic tools and analyzed mutants; C.B. and A.L. built the Nqu143-GFP mutant and analyzed the data; A.V. collected CryoEM data; A.V., S.S., A.S. and C.A. analyzed CryoEM data; Y.C., L.B. and A.A. performed MS-based proteomics and analyzed the data; A.N. and C.D.C. performed GC-MS and NMR analyses and analyzed the data; H.B., A.V., A.S., S.S. A.N., C.D.C., Y.C. and C.A. wrote the manuscript.
Competing interests
The authors declare that they have no competing interests.
Funding
This project has received funding from the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (grant agreement No 832 601). Proteomic experiments were partly supported by ProFI (ANR-10-INBS-08-01) and GRAL, a program from the Chemistry Biology Health (CBH) Graduate School of University Grenoble Alpes (ANR-17-EURE-0003). France-BioImaging national research infrastructure (ANR-10-INBS-04). CDC gratefully acknowledges STARPLUS 2020 (project no. 21‐UNINA‐EPIG‐042) from the University of Napoli for financial support.
Data Availability
Genome sequences of the mutants of mimivirus reunion have been deposited to NCBI (accession numbers: KO_qu_946: OQ700912; KO_qu143: OQ700913, 2KO: OQ700914). 3D reconstruction maps and the corresponding PDB have been deposited to EMDB (Deposition number Cl1: 8ORS, EMD-17131; Cl2: 8ORH, EMD-17125). The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE (Perez-Riverol et al. 2022) partner repository with the dataset identifier PXD041298. AlphaFold predictions were performed using HPC/AI resources from GENCI-IDRIS (Grant 2022-AD011013526).
References
- Abergel C, Rudinger-Thirion J, Giegé R et al. Virus-encoded aminoacyl-tRNA synthetases: structural and functional characterization of mimivirus TyrRS and MetRS. J Virol. 2007;81:12406–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abrahão J, Silva L, Silva LS et al. Tailed giant Tupanvirus possesses the most complete translational apparatus of the known virosphere. Nat Commun. 2018;9:749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisio H, Legendre M, Giry C et al. Evolution of giant pandoravirus revealed by CRISPR/Cas9. Nat Commun. 2023;14:428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouyssié D, Hesse AM, Mouton-Barbosa E et al. Proline: an efficient and user-friendly software suite for large-scale proteomics. Bioinforma Oxf Engl. 2020;36:3148–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyer M, Azza S, Barrassi L et al. Mimivirus shows dramatic genome reduction after intraamoebal culture. P Natl Acad Sci USA. 2011;108:10296–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casabona MG, Vandenbrouck Y, Attree I et al. Proteomic characterization of Pseudomonas aeruginosa PAO1 inner membrane. Proteomics. 2013;13:2419–23. [DOI] [PubMed] [Google Scholar]
- Cassetti MC, Merchlinsky M, Wolffe EJ et al. DNA packaging mutant: repression of the vaccinia virus a32 gene results in noninfectious, DNA-deficient, spherical, enveloped particles. J Virol. 1998;72:5769–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Claverie J-M, Abergel C. Mimivirus: the emerging paradox of quasi-autonomous viruses. Trends Genet. 2010;26:431–7. [DOI] [PubMed] [Google Scholar]
- Coudray N, Lasala R, Zhang Z et al. Deducing the symmetry of helical assemblies: applications to membrane proteins. J Struct Biol. 2016;195:167–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Couté Y, Bruley C, Burger T. Beyond target-decoy competition: stable validation of peptide and protein identifications in mass spectrometry-based discovery proteomics. Anal Chem. 2020;92:14898–906. [DOI] [PubMed] [Google Scholar]
- de Aquino ILM, Serafim MSM, Machado TB et al. Diversity of surface fibril patterns in mimivirus isolates. J Virol. 2023;97:e0182422. 10.1128/jvi.01824-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Castro C, Parrilli M, Holst O et al. Microbe-associated molecular patterns in innate immunity: extraction and chemical analysis of gram-negative bacterial lipopolysaccharides. Methods Enzymol. 2010;480:89–115. 10.1016/S0076-6879(10)80005-9. [DOI] [PubMed] [Google Scholar]
- Diaz R, Rice WJ, Stokes DL. Fourier-Bessel reconstruction of helical assemblies. Methods Enzymol. 2010;482:131–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emsley P, Lohkamp B, Scott WG et al. Features and development of Coot. Acta Crystallogr D Biol Crystallogr. 2010;66:486–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson HP, Osawa M. Cell division without FtsZ–a variety of redundant mechanisms. Mol Microbiol. 2010;78:267–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallot-Lavallée L, Blanc G, Claverie J-M. Comparative genomics of Chrysochromulina ericina virus and other microalga-infecting large DNA viruses highlights their intricate evolutionary relationship with the established Mimiviridae family. J Virol. 2017;91:e00230–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hacker C, Asadi J, Pliotas C et al. Nanoparticle suspensions enclosed in methylcellulose: a new approach for quantifying nanoparticles in transmission electron microscopy. Sci Rep. 2016;6:25275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He S, Scheres SHW. Helical reconstruction in RELION. J Struct Biol. 2017;198:163–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jumper J, Evans R, Pritzel A et al. Highly accurate protein structure prediction with AlphaFold. Nature. 2021;596:583–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klose T, Herbst DA, Zhu H et al. A Mimivirus enzyme that participates in viral entry. Struct Lond Engl 1993. 2015;23:1058–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koonin EV, Dolja VV, Krupovic M et al. Global organization and proposed megataxonomy of the virus world. Microbiol Mol Biol Rev. 2020;84:e00061–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuznetsov YG, Klose T, Rossmann M et al. Morphogenesis of mimivirus and its viral factories: an atomic force microscopy study of infected cells. J Virol. 2013;87:11200–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liebschner D, Afonine PV, Baker ML et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr D Struct Biol. 2019;75:861–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Bisio H, Toner CM et al. Virus-encoded histone doublets are essential and form nucleosome-like structures. Cell. 2021;184:4237–4250.e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mutsafi Y, Zauberman N, Sabanay I et al. Vaccinia-like cytoplasmic replication of the giant Mimivirus. P Natl Acad Sci USA. 2010;107:5978–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Notaro A, Couté Y, Belmudes L et al. Expanding the occurrence of polysaccharides to the viral world: the case of Mimivirus. Angew Chem Int Ed. 2021;60:19897–904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Notaro A, Poirot O, Garcin ED et al. Giant viruses of the Megavirinae subfamily possess biosynthetic pathways to produce rare bacterial-like sugars in a clade-specific manner. microLife. 2022;3:uqac002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perez-Riverol Y, Bai J, Bandla C et al. The PRIDE database resources in 2022: a hub for mass spectrometry-based proteomics evidences. Nucleic Acids Res. 2022;50:D543–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettersen EF, Goddard TD, Huang CC et al. UCSF chimera-A visualization system for exploratory research and analysis. J Comput Chem. 2004;25:1605–12. [DOI] [PubMed] [Google Scholar]
- Philippe N, Shukla A, Abergel C et al. Genetic manipulation of giant viruses and their host, Acanthamoeba castellanii. Nat Protoc. 2024;19:3–29. https:/doi.org/10.1038/s41596-023-00910-y. [DOI] [PubMed] [Google Scholar]
- Raoult D, Audic S, Robert C et al. The 1.2-megabase genome sequence of Mimivirus. Science. 2004;306:1344–50. [DOI] [PubMed] [Google Scholar]
- Rodrigues RAL, dos Santos Silva LK, Dornas FP et al. Mimivirus fibrils are important for viral attachment to the microbial world by a diverse glycoside interaction repertoire. J Virol. 2015;89:11812–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohou A, Grigorieff N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J Struct Biol. 2015;192:216–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachse C. Single-particle based helical reconstruction—how to make the most of real and Fourier space. AIMS Biophys. 2015;2:219–44. [Google Scholar]
- Scheres SHW. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J Struct Biol. 2012;180:519–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schrad JR, Abrahão JS, Cortines JR et al. Structural and proteomic characterization of the initiation of giant virus infection. Cell. 2020;181:1046–1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwanhäusser B, Busse D, Li N et al. Global quantification of mammalian gene expression control. Nature. 2011;473:337–42. [DOI] [PubMed] [Google Scholar]
- Sobhy H. A comparative review of viral entry and attachment during large and giant dsDNA virus infections. Arch Virol. 2017;162:3567–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speciale I, Notaro A, Abergel C et al. The astounding world of glycans from giant viruses. Chem Rev. 2022;122:15717–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speciale I, Notaro A, Garcia-Vello P et al. Liquid-state NMR spectroscopy for complex carbohydrate structural analysis: a hitchhiker's guide. Carbohydr Polym. 2022;277:118885. [DOI] [PubMed] [Google Scholar]
- Suzan-Monti M, Scola BL, Barrassi L et al. Ultrastructural characterization of the giant volcano-like virus factory of acanthamoeba polyphaga Mimivirus. PLoS One. 2007;2:e328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi H, Fukaya S, Song C et al. Morphological and taxonomic properties of the newly isolated Cotonvirus japonicus, a new lineage of the subfamily Megavirinae. J Virol. 2021;95:e0091921. https:/doi.org/10.1128/JVI.00919-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valencia-Sánchez MI, Abini-Agbomson S, Wang M et al. The structure of a virus-encoded nucleosome. Nat Struct Mol Biol. 2021;28:413–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villalta A, Schmitt A, Estrozi LF et al. The giant mimivirus 1.2 Mb genome is elegantly organized into a 30-nm diameter helical protein shield. eLife. 2022;11:e77607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zauberman N, Mutsafi Y, Halevy DB et al. Distinct DNA exit and packaging portals in the virus Acanthamoeba polyphaga mimivirus. PLoS Biol. 2008;6:e114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng SQ, Palovcak E, Armache JP et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat Methods. 2017;14:331–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Genome sequences of the mutants of mimivirus reunion have been deposited to NCBI (accession numbers: KO_qu_946: OQ700912; KO_qu143: OQ700913, 2KO: OQ700914). 3D reconstruction maps and the corresponding PDB have been deposited to EMDB (Deposition number Cl1: 8ORS, EMD-17131; Cl2: 8ORH, EMD-17125). The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE (Perez-Riverol et al. 2022) partner repository with the dataset identifier PXD041298. AlphaFold predictions were performed using HPC/AI resources from GENCI-IDRIS (Grant 2022-AD011013526).