Abstract
OBJECTIVE
To describe the natural history of diabetes in Indians.
RESEARCH DESIGN AND METHODS
Data are from participants older than 20 years in the Centre for Cardiometabolic Risk Reduction in South Asia longitudinal study. Glycemic states were defined per American Diabetes Association criteria. Markov models were used to estimate annual transition probabilities and sojourn time through states.
RESULTS
Among 2,714 diabetes-free participants, 641 had isolated impaired fasting glucose (iIFG), and 341 had impaired glucose tolerance (IGT). The annual transition to diabetes for those with IGT was 13.9% (95% CI 12.0, 15.9) versus 8.6% (7.3, 9.8) for iIFG. In the normoglycemia ↔ iIFG → diabetes model, mean sojourn time in normoglycemia was 40.3 (34.6, 48.2) years, and sojourn time in iIFG was 9.7 (8.4, 11.4) years. For the normoglycemia ↔ IGT → diabetes model, mean sojourn time in normoglycemia was 34.5 (29.5, 40.8) years, and sojourn time in IGT was 6.1 (5.3, 7.1) years.
CONCLUSIONS
Individuals reside in normoglycemia for 35–40 years; however, progression from prediabetes to diabetes is rapid.
Graphical Abstract
Introduction
Indian people are at heightened risk of type 2 diabetes (1,2). However, it is unclear how Indians transition through the natural history of diabetes (e.g., from normoglycemia to prediabetes [impaired fasting glucose [IFG] or impaired glucose tolerance [IGT]), reversal to normoglycemia, and from prediabetes to diabetes), and how long people reside in each state (sojourn time).
Research Design and Methods
We used data from the Chennai site of the longitudinal Centre for Cardiometabolic Risk Reduction in South Asia (CARRS) study (2010–2012) (3,4), up to the fourth follow-up (2016–2017) and implemented a continuous-time Markov model (5).
Inputs
Glycemic states (normoglycemia, IGT, iIFG, diabetes) were defined according to American Diabetes Association criteria, and distributions and characteristics of each glycemic state and the rates of progression from one stage to the next were obtained from the CARRS study (3,4) (details of definitions and cohort are provided in the Supplementary Material) (6).
Among 5,961 participants with glucose measurements at baseline, 3,475 were free of diabetes. Of these, 2,714 participants had had a complete oral glucose tolerance test and at least one follow-up assessment before 2017 to estimate changes in glycemia. The characteristics of the 2,714 participants included in the final analyses (Supplementary Fig. 3) were similar to those of the overall sample (Supplementary Table 1). Because only 187 people had combined IFG and IGT, they were classified as having IGT.
Multistate Analysis
We used multistate Markov models (7,8) to calculate annual transition probabilities for each state specified in Supplementary Fig. 1. The mean sojourn time was also calculated. For every participant, the outcome of interest was iIFG, IGT, or diabetes. Time was estimated from date of interview to the time of outcome diagnosis, last date of visit, or death, whichever came first. We fitted two models: 1) normoglycemia to iIFG and regression to normoglycemia or progression to diabetes (i.e., normoglycemia ↔ iIFG → diabetes); and 2) normoglycemia to IGT and regression to normoglycemia or progression to diabetes (normoglycemia ↔ IGT → diabetes). In the base case analysis, we assumed bidirectional change in states, allowing regression from prediabetes to normoglycemia (Supplementary Fig. 1). In a sensitivity analysis, we examined unidirectional progression, which assumes people cannot move back from prediabetes to normoglycemia (Supplementary Fig. 2). We performed stratified analyses by age (≤40 years vs. >40 years), sex, and BMI (<23 kg/m2 vs. ≥23 kg/m2) (9) to estimate the annual transition probabilities for each set of models. The data were analyzed using the msm package in R software, version 3.2.4, and Stata 16.0/MP.
Results
As shown in Table 1, iIFG was nearly twice as frequent as IGT, and iIFG was more frequent in women (71.5%), and IGT in men (53.1%). Those with normoglycemia, followed by iIFG, and then IGT, had the lowest mean age (normoglycemia vs. iIFG vs. IGT: 37.6 vs. 40.9 vs. 43.7 years, respectively), weight (61.6 vs. 65.5 vs. 66.3 kg, respectively), BMI (25.0 vs. 27.2 vs. 27.0 kg/m2, respectively), waist circumference (81.6 vs. 85.7 vs. 88.1 cm, respectively), triglyceride levels (107.0 vs. 124.0 vs. 137.0 mg/dL, respectively), and total cholesterol level (180.4 vs. 187.4 vs. 189.9 mg/dL, respectively). The insulin levels at 0, 30, 120 min (iIFG: 7.7, 48.2, and 40.0 vs. IGT: 8.5, 53.1, and 59.6, respectively) and HOMA-β (iIFG vs IGT: 69.3 vs. 91.5 μIU/mL/mmol/L, respectively) were lower in those with iIFG compared with participants with IGT, thus indicating iIFG is a more insulin-deficient state of prediabetes.
Table 1.
Characteristics of participants in normoglycemia, iIFG, and IGT
| Characteristic | Normoglycemia* (n = 2,205) | iIFG† (n = 641) | IGT‡ (n = 341) |
|---|---|---|---|
| Age (years), mean (SD) | 37.6 (10.8)§ | 40.9 (10.3)¶ | 43.7 (12.5)⊥ |
| Age (years), median (IQR) | 36.0 (30.0, 44.0)§ | 40.0 (33.0, 48.0)¶ | 42.0 (35.0, 51.0)⊥ |
| Sex, n (%) | |||
| Male | 924 (41.9)§ | 183 (28.5)¶ | 160 (46.9)⊥ |
| Female | 1,281 (58.1) | 458 (71.5) | 181 (53.1) |
| Family history of diabetes§, n (%) | 745 (33.8)§ | 237 (37.0) | 139 (40.8) |
| Weight (kg), mean (SD) | 61.6 (12.0)§ | 65.5 (12.2)¶ | 66.3 (12.0) |
| Height (cm), mean (SD) | 157.1 (8.9)§ | 155.1 (8.4)¶ | 156.9 (9.0) |
| BMI (kg/m2), mean (SD) | 25.0 (4.7)§ | 27.2 (4.6)¶ | 27.0 (4.7) |
| Waist circumference (cm), mean (SD) | |||
| Overall | 81.6 (11.1)§ | 85.7 (10.4)¶ | 88.1 (10.6)⊥ |
| Male | 84.8 (10.7)§ | 89.5 (10.3)¶ | 90.9 (10.8) |
| Female | 79.5 (10.8)§ | 84.2 (10.1)¶ | 85.7 (9.8) |
| Total cholesterol (mg/dL), mean (SD) | 180.4 (36.3)§ | 187.4 (37.1)¶ | 189.9 (34.3) |
| Triglycerides (mg/dL), median (IQR) | 107.0 (78.0, 150.0)§ | 124.0 (89.0, 171.0)¶ | 137.0 (100.0, 186.0)⊥ |
| LDL cholesterol (mg/dL), mean (SD) | 111.7 (28.9)§ | 118.2 (31.6)¶ | 117.5 (27.9) |
| HDL cholesterol (mg/dL), mean (SD) | |||
| Overall | 41.1 (9.5) | 40.1 (8.2)¶ | 40.3 (10.4) |
| Male | 39.6 (10.6) | 37.8 (8.3) | 39.7 (12.0) |
| Female | 42.1 (8.5) | 41.1 (7.9) | 40.8 (10.4) |
| FPG (mg/dL), mean (SD); median (IQR) | 90.0 (7.1)§; 90.0 (85.0, 95.0)§ | 102.7 (7.9)¶; 103.0 (100.0, 107.0)¶ | 98.3 (10.0)⊥; 98.0 (91.0, 106.0)⊥ |
| Glucose level at 30 min (mg/dL), mean (SD); median (IQR) | 140.7 (28.2)§; 140.0 (119.0, 160.0)§ | 164.0 (29.5)¶; 165.0 (144.0, 185.0)¶ | 174.4 (30.0)⊥; 176.0 (155.0, 193.0)⊥ |
| Glucose level at 120 min (mg/dL), mean (SD); median (IQR) | 94.7 (20.9)§; 94.0 (81.0, 107.0)§ | 107.1 (22.7)¶; 106.0 (93.0, 122.0)¶ | 138.6 (29.1)⊥; 143.0 (116.0, 159.0)⊥ |
| HbA1c (%), mean (SD); median (IQR) | 5.6 (0.4)§; 5.6 (5.3, 5.8)§ | 5.8 (0.4)¶; 5.8 (5.6, 6.1)¶ | 5.9 (0.4); 5.9 (5.6, 6.1) |
| HbA1c (mmol/mol), mean (SD); median (IQR) | 38 (0.4)§; 38 (34, 40)§ | 40 (0.4)¶; 40 (38, 43)¶ | 41 (0.4); 41 (38, 43) |
| Insulin, fasting (pmol/L), median (IQR) | 6.4 (4.4, 9.4) | 7.7 (5.5, 10.8)¶ | 8.5 (6.7, 11.2) |
| Insulin level at 30 min (pmol/L), median (IQR) | 46.6 (32.2, 72.8) | 48.2 (33.7, 71.5) | 53.1 (35.6, 76.2) |
| Insulin level at 120 min (pmol/L), median (IQR) | 31.4 (22.4, 51.0)§ | 40.0 (26.3, 65.0)¶ | 59.6 (37.8, 98.6) |
| HOMA-IR** (μIU/mL*mmol/L), median (IQR) | 1.4 (1.0, 2.1)§ | 2.0 (1.4, 2.8)¶ | 2.1 (1.6, 2.8) |
| HOMA-ↆ (μIU/mL/mmol/L), median (IQR) | 89.0 (61.2, 130.9) | 69.3 (48.9, 101.1)¶ | 91.5 (66.2, 129.9)⊥ |
| DIo‡‡, median (IQR) | 0.1 (0.1, 0.3)§ | 0.1 (0.1, 0.2)¶ | 0.1 (0.0, 0.1)⊥ |
| Insulinogenic Index§§, median (IQR) | 0.9 (0.5, 1.8)§ | 0.7 (0.4, 1.3)¶ | 0.6 (0.4, 1.0)⊥ |
DIo, Oral Disposition Index; FPG, fasting plasma glucose; IQR, interquartile range; IR, insulin resistance.
Normoglycemia defined as FPG <5.6 mmol/L (100 mg/dL) and 2-hour postload glucose (2h-PG) <7.8 mmol/L (140 mg/dL) and no medication.
IFG defined as FPG between 5.6 and 6.9 mmol/L (100–125 mg/dL) and 2h-PG <7.8 mmol/L (140 mg/dL) and no medication.
IGT defined as FPG <7.0 mmol/L (126 mg/dL) and 2h-PG between 7.8–11.0 mmol/L (140–199 mg/dL) and no medication.
Significant P value for normoglycemia versus IGT.
Significant P value for normoglycemia versus iIFG.
Significant P value for iIFG versus IGT.
Diabetes defined as FPG ≥7.0 mmol/L (126 mg/dL) or 2-h PG ≥11.1 mmol/L (200 mg/dL) or HbA1c ≥6.5% (48 mmol/mol) or on medication.
HOMA-IR = fasting insulin (μU/L) × fasting glucose (nmol/L)/22.5 or (I0(μIU/mL) × G0 (mmol/L)/22.5).
HOMA-B = (20 × insulin)/(glucose − 3.5) or (20 × I0(μIU/mL)/G0 (mmol/L) − 3.5).
DIo = (ΔI0 − 30/ΔG0 − 30) × (1/fasting insulin).
Insulinogenic Index = (ΔI0–30/ΔG0–30) where ΔI0–30 = insulin at 30 min minus fasting insulin; ΔG0–30 = glucose at 30 min minus FPG.
Markov Model Transition Probabilities
Normoglycemia ↔ iIFG → Diabetes Pathway Model
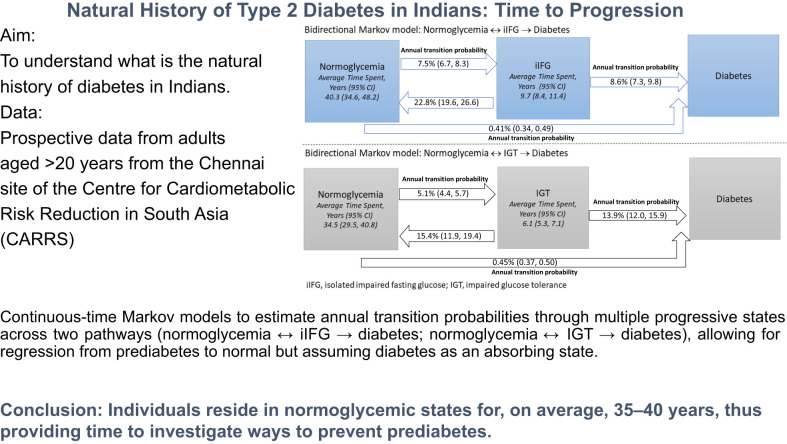
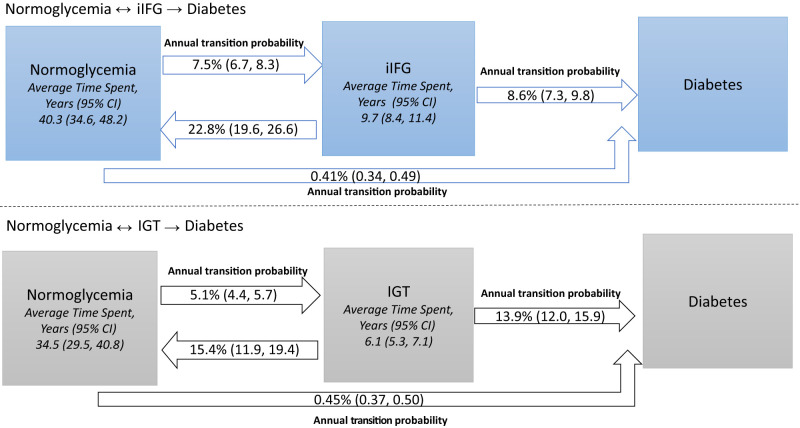
The estimated mean annual probability of remaining in normoglycemia and in iIFG were 92.1% (95% CI 91.2, 92.9) and 68.6% (65.2, 71.8), respectively (Table 2). The annual probability of conversion from normoglycemia to iIFG was 7.5% (6.7, 8.3) and from iIFG to normoglycemia was 22.8% (19.6, 26.6). Thus, it is about three times more likely for iIFG to revert to normoglycemia than it is for normoglycemia to progress to iIFG. The annual probability of conversion from iIFG to diabetes was 8.6% (5.7, 11.9). The estimated mean sojourn times were 40.3 (34.6, 48.2) years and 9.7 (8.4, 11.4) years in normoglycemia and iIFG, respectively (Fig. 1).
Table 2.
Normoglycemia↔ iIFG →diabetes multistate Markov model annual probability of transition across states (overall and stratified by age, sex, and BMI)
| n | Annual transition probabilities, % (95% CI) | |
|---|---|---|
| Overall | ||
| Normoglycemia → normoglycemia | 1,937 | 92.1 (91.2, 92.9) |
| Normoglycemia → iIFG | 276 | 7.5 (6.7, 8.3) |
| Normoglycemia → diabetes | 64 | 0.41 (0.34, 0.49) |
| iIFG → iIFG | 193 | 68.6 (65.2, 71.8) |
| iIFG → normoglycemia | 207 | 22.8 (19.6, 26.6) |
| iIFG → diabetes | 78 | 8.6 (7.3, 9.8) |
| Age ≤40 years | ||
| Normoglycemia → normoglycemia | 1,362 | 93.2 (92.1, 94.2) |
| Normoglycemia → iIFG | 164 | 6.4 (5.5, 7.4) |
| Normoglycemia → diabetes | 37 | 0.35 (0.27, 0.45) |
| iIFG → iIFG | 98 | 67.5 (62.7, 71.6) |
| iIFG → normoglycemia | 121 | 24.1 (19.9, 29.5) |
| iIFG → diabetes | 42 | 8.4 (6.8, 10.0) |
| Age >40 years | ||
| Normoglycemia → normoglycemia | 575 | 89.5 (87.4, 91.4) |
| Normoglycemia → iIFG | 112 | 9.9 (8.1, 11.9) |
| Normoglycemia → diabetes | 27 | 0.56 (0.41, 0.73) |
| iIFG → iIFG | 95 | 69.7 (64.4, 74.2) |
| iIFG → normoglycemia | 86 | 21.6 (17.0, 27.1) |
| iIFG → diabetes | 36 | 8.7 (6.7, 10.6) |
| Male participants | ||
| Normoglycemia → normoglycemia | 769 | 92.7 (91.2, 93.9) |
| Normoglycemia → iIFG | 87 | 6.8 (5.6, 8.1) |
| Normoglycemia → diabetes | 40 | 0.52 (0.38, 0.72) |
| iIFG → iIFG | 49 | 64.6 (56.9, 70.4) |
| iIFG → normoglycemia | 61 | 23.9 (17.9, 31.6) |
| iIFG → diabetes | 20 | 11.4 (9.0, 13.9) |
| Female participants | ||
| Normoglycemia → normoglycemia | 1,168 | 91.7 (90.6, 92.9) |
| Normoglycemia → iIFG | 189 | 7.9 (6.8, 9.0) |
| Normoglycemia → diabetes | 24 | 0.37 (0.28, 0.46) |
| iIFG → iIFG | 144 | 70.4 (66.5, 73.6) |
| iIFG → normoglycemia | 146 | 22.4 (18.9, 26.3) |
| iIFG → diabetes | 58 | 7.2 (5.9, 8.6) |
| BMI <23 kg/m2 | ||
| Normoglycemia → normoglycemia | 655 | 94.2 (92.4, 95.6) |
| Normoglycemia → iIFG | 59 | 5.5 (4.1, 7.1) |
| Normoglycemia → diabetes | 16 | 0.34 (0.21, 0.54) |
| iIFG → iIFG | 18 | 59.1 (45.6, 68.6) |
| iIFG → normoglycemia | 43 | 32.0 (23.1, 46.9) |
| iIFG → diabetes | 11 | 8.8 (5.7, 11.9) |
| BMI ≥23 kg/m2 | ||
| Normoglycemia → normoglycemia | 1,099 | 90.5 (89.2, 91.8) |
| Normoglycemia → iIFG | 194 | 8.9 (7.7, 10.2) |
| Normoglycemia → Diabetes | 38 | 0.48 (0.38, 0.59) |
| iIFG → iIFG | 158 | 69.6 (65.7, 72.9) |
| iIFG → normoglycemia | 153 | 22.1 (18.6, 26.3) |
| iIFG → diabetes | 63 | 8.3 (6.9, 9.8) |
Figure 1.
Multistate Markov models. The annual probability of remaining in the same state or transitioning to the next state.
The annual probability of transition for normoglycemia to iIFG was greater for those age >40 (9.9% [95% CI 8.1, 11.9]) years compared with those age ≤40 (6.4% [5.5, 7.4]) years (Table 2). The annual probability of transition from iIFG to diabetes was higher in female participants than male participants (female: 7.9% [6.8, 9.0]; male: 6.8% [5.6, 8.1]). The annual probability of transition from normoglycemia to iIFG was greater for those with BMI ≥23 kg/m2 (8.9% [7.7, 10.2]) compared with BMI <23 kg/m2 (5.5% [4.1, 7.1]).
Normoglycemia ↔ IGT → Diabetes Pathway Model
The estimated annual probability of remaining in normoglycemia and IGT were 94.5% (95% CI 93.8, 95.2) and 70.1% (67.1, 73.8), respectively (Table 3). The annual probability of transition from normoglycemia to IGT was 5.1% (4.4, 5.7) and from IGT to normoglycemia was 15.4% (11.9, 19.4). Thus, it is about three times more likely for IGT to revert to normoglycemia than it is for normoglycemia to progress to IGT. The annual probability of transition from IGT to diabetes was 13.9% (12.0, 15.9). The estimated mean sojourn times were 34.5 (29.5, 40.8) years and 6.1 (5.3, 7.1) years for normoglycemia and IGT, respectively (Fig. 1).
Table 3.
Normoglycemia ↔ IGT→ diabetes multistate Markov model annual probability of transition across states (overall and stratified by age, sex, and BMI)
| n | Annual transition probability, % (95% CI) | |
|---|---|---|
| Overall | ||
| Normoglycemia → normoglycemia | 1,894 | 94.5 (93.8, 95.2) |
| Normoglycemia → IGT | 193 | 5.1 (4.4, 5.7) |
| Normoglycemia → diabetes | 60 | 0.45 (0.37, 0.50) |
| IGT → IGT | 67 | 70.1 (67.1, 73.8) |
| IGT → normoglycemia | 71 | 15.4 (11.9, 19.4) |
| IGT → diabetes | 66 | 13.9(12.0, 15.9) |
| Age ≤40 years | ||
| Normoglycemia → normoglycemia | 1,333 | 95.7 (94.9, 96.4) |
| Normoglycemia → IGT | 106 | 3.9 (3.3, 4.7) |
| Normoglycemia → diabetes | 34 | 0.36 (0.28, 0.47) |
| IGT → IGT | 27 | 71.6 (66.4, 76.2) |
| IGT → normoglycemia | 29 | 13.5 (8.9, 19.2) |
| IGT → diabetes | 33 | 14.9 (11.9, 17.9) |
| Age >40 years | ||
| Normoglycemia → normoglycemia | 561 | 91.7 (90.1, 93.2) |
| Normoglycemia → IGT | 87 | 7.6 (6.2, 9.1) |
| Normoglycemia → diabetes | 26 | 0.64 (0.47, 0.82) |
| IGT → IGT | 40 | 69.4 (63.9, 74.5) |
| IGT → normoglycemia | 42 | 17.6 (12.6, 23.7) |
| IGT → diabetes | 33 | 12.9 (10.3, 15.6) |
| Male participants | ||
| Normoglycemia → normoglycemia | 762 | 92.6 (91.3, 93.8) |
| Normoglycemia → IGT | 100 | 6.8 (5.7, 8.0) |
| Normoglycemia → diabetes | 39 | 0.59 (0.46, 0.74) |
| IGT → IGT | 43 | 69.2 (63.7, 74.1) |
| IGT → normoglycemia | 43 | 17.4 (12.4, 23.3) |
| IGT → diabetes | 30 | 13.4 (10.8, 16.3) |
| Female participants | ||
| Normoglycemia → normoglycemia | 1,132 | 95.7 (94.9, 96.7) |
| Normoglycemia → IGT | 93 | 3.9 (3.1, 4.6) |
| Normoglycemia → diabetes | 21 | 0.35 (0.26, 0.43) |
| IGT → IGT | 24 | 71.9 (66.9, 76.3) |
| IGT → normoglycemia | 28 | 13.6 (9.1, 19.0) |
| IGT → diabetes | 36 | 14.4 (11.5, 17.3) |
| BMI <23 kg/m2 | ||
| Normoglycemia → normoglycemia | 647 | 96.2 (95.1, 97.2) |
| Normoglycemia → IGT | 41 | 3.5 (2.6, 4.5) |
| Normoglycemia → diabetes | 15 | 0.27 (0.16, 0.41) |
| IGT → IGT | 12 | 69.4 (57.1, 78.4) |
| IGT → normoglycemia | 14 | 18.6 (9.6, 32.4) |
| IGT → diabetes | 6 | 12.1 (7.5, 16.8) |
| BMI ≥23 kg/m2 | ||
| Normoglycemia → normoglycemia | 1,062 | 93.3 (92.3, 94.3) |
| Normoglycemia → IGT | 135 | 6.2 (5.2, 7.1) |
| Normoglycemia → Diabetes | 35 | 0.51 (0.41, 0.63) |
| IGT → IGT | 51 | 71.0 (66.6, 75.0) |
| IGT → normoglycemia | 53 | 15.9 (11.4, 20.7) |
| IGT → diabetes | 49 | 13.1 (10.8, 15.5) |
The annual probability of transition from normoglycemia to IGT was higher among those aged >40 years (7.6% [95% CI 6.2, 9.1]) as compared with those aged ≤40 years (3.9% [3.3, 4.7]) (Table 3). The annual transition probability from normoglycemia to IGT was higher in men (6.8% [5.7, 8.0]) than women (3.9% [3.1, 4.6]). The annual probability of transition from normoglycemia to IGT was higher among those with BMI ≥23 kg/m2 (6.2% [5.2, 7.1]) as compared with those with BMI <23 kg/m2 (3.5% [2.6, 4.5]).
Sensitivity Analysis (Unidirectional Models)
In a sensitivity analysis, the probabilities of transition based on unidirectional models (i.e., not allowing regression from prediabetes to normoglycemia) were similar to those of base case bidirectional models (Supplementary Tables 1–4).
Conclusions
In an urban Indian population aged ≥20 years, progression to diabetes is rapid once an individual has prediabetes. On average, people reside 35–40 years in normoglycemic states, and only 9.7 years in iIFG or 6.1 years in IGT before advancing to diabetes (assuming bidirectional transition from normoglycemia to prediabetes). Prediabetes represents a fragile state, with a nearly three times likelihood of either iIFG or IGT reverting to normoglycemia than normoglycemia progressing to prediabetes. However, at the onset of prediabetes, and after accounting for reversibility, the rate of progression from prediabetes to diabetes was rapid, and those with IGT progressed to diabetes faster (13.9% per annum) than those with iIFG (8.6% per annum).
Similar to our findings, several studies have reported a high incidence of diabetes and prediabetes in Indians (1,10–12). In addition, we found a higher rate of conversion from normoglycemia to iIFG than to IGT, suggesting reduced insulin secretion (lower HOMA of β-cell function [HOMA-β]) as an early defect (13). However, among those with prediabetes, those with IGT had a more rapid conversion to diabetes, suggesting poorer insulin sensitivity as a key factor at later stages in those already susceptible, and iIFG and IGT being potentially different phenotypes with differences in pathophysiology (13–15). Similar to previous reports (16), we also found that iIFG is the more frequent (almost two-thirds) prediabetes manifestation in Indians, iIFG is the common phenotype in women, and IGT the more common phenotype in men. Although lifestyle interventions are effective in reducing the incidence of diabetes among adults with IGT (17,18), these interventions seem not effective in individuals with iIFG (19).
The strengths of our study include data from a representative sample, high response and retention rates, multiple time points of follow-up, and objective measures of glycemia derived from three-step oral glucose tolerance tests. To our knowledge, no previous study of diabetes in Indians has estimated time to progression or time spent in each glycemic state. Given the fragility of the prediabetes state, we conservatively assumed bidirectional transition from normoglycemia to iIFG or IGT (in separate models), but we also performed sensitivity analyses to explore the effect of unidirectional transitions and other assumptions. The results for transition probabilities across states were robust regardless of assumption of bidirectional or unidirectional progression, but the estimates of time in each state were substantially longer under assumption of bidirectionality. Last, we performed stratified analyses by age, sex, and BMI. Our study has some limitations, including that data are from one city; however, diabetes incidence across urban India is quite similar, and the sample reflects the age and sociodemographic distribution of populations of cities in India.
In conclusion, we found a high rate of conversion from normoglycemia to IFG or IGT in Indians, and once an individual has prediabetes, the conversion to diabetes is rapid. On the hopeful side, people at risk for diabetes reside in normoglycemic states for an average of 35–40 years, and those transitioning through the more frequent iIFG stage reside there for an average of 9.7 years (as opposed to 6.1 years in IGT). These findings suggest the need to test interventions to prevent the occurrence of prediabetes.
This article contains supplementary material online at https://doi.org/10.2337/figshare.25213064.
Article Information
Acknowledgments. The authors thank the staff and participants of the CARRS for their important contributions.
Funding. The CARRS Study was funded in part by the National Heart, Lung, and Blood Institute (NHLBI), National Institutes of Health (NIH), Department of Health and Human Services (contract HHSN268200900026C and grant PO1HL154996); and the United Health Group (Minneapolis, MN). K.M.V.N., M.K.A., U.P.G., and S.A.P. were funded in part by the National Institute of Diabetes and Digestive and Kidney Diseases, NIH (grant P30DK111024). K.M.V.N. was funded in part by the Worksite Lifestyle Program for Reducing Diabetes and Cardiovascular Risk in India project funded by the NHLBI (grant R01HL125442). S.A.P. was funded in part by the NHLBI (grant 5U01HL138635-02). S.A.P., K.M.V.N., M.K.A., N.T., and D.P. were supported in part by the NIH (grant 5U01HL138635) under the Hypertension Outcomes for T4 Research Within Lower Middle-Income Countries program. D.K. has been supported by the NIH’s Fogarty International Center for the Public Health Leader course (grant D43TW009135).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. K.M.V.N. wrote the first draft of the manuscript and reviewed and edited the manuscript. D.K. and H.H.C. researched the data and performed the formal analysis for the article. D.M., R.M.A., and M.K.A. reviewed and edited the manuscript. U.P.G., L.R.S., and S.A.P. contributed to methods and reviewed and edited the manuscript. D.P., N.T., and V.M. contributed to discussion and reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript. K.M.V.N. and D.P. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Handling Editors. The journal editors responsible for overseeing the review of the manuscript were Steven E. Kahn and Namratha R. Kandula.
Funding Statement
The CARRS Study was funded in part by the National Heart, Lung, and Blood Institute (NHLBI), National Institutes of Health (NIH), Department of Health and Human Services (contract HHSN268200900026C and grant PO1HL154996); and the United Health Group (Minneapolis, MN). K.M.V.N., M.K.A., U.P.G., and S.A.P. were funded in part by the National Institute of Diabetes and Digestive and Kidney Diseases, NIH (grant P30DK111024). K.M.V.N. was funded in part by the Worksite Lifestyle Program for Reducing Diabetes and Cardiovascular Risk in India project funded by the NHLBI (grant R01HL125442). S.A.P. was funded in part by the NHLBI (grant 5U01HL138635-02). S.A.P., K.M.V.N., M.K.A., N.T., and D.P. were supported in part by the NIH (grant 5U01HL138635) under the Hypertension Outcomes for T4 Research Within Lower Middle-Income Countries program. D.K. has been supported by the NIH’s Fogarty International Center for the Public Health Leader course (grant D43TW009135).
References
- 1. Chiu M, Austin PC, Manuel DG, Shah BR, Tu JV. Deriving ethnic-specific BMI cutoff points for assessing diabetes risk. Diabetes Care 2011;34:1741–1748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Oza-Frank R, Narayan KMV. Overweight and diabetes prevalence among US immigrants. Am J Public Health 2010;100:661–668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Nair M, Ali MK, Ajay VS, et al. CARRS surveillance study: design and methods to assess burdens from multiple perspectives. BMC Public Health 2012;12:701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Kondal D, Patel SA, Ali MK, et al. Cohort profile: the Center for cArdiometabolic Risk Reduction in South Asia (CARRS). Int J Epidemiol 2022;51:e358–e371 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Marshall G, Jones RH. Multi-state models and diabetic retinopathy. Stat Med 1995;14:1975–1983 [DOI] [PubMed] [Google Scholar]
- 6. American Diabetes Association . 2. Classification and diagnosis of diabetes: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018;41(Suppl. 1):S13–S27 [DOI] [PubMed] [Google Scholar]
- 7. Kalbfleisch JD, Lawless JF. The analysis of panel data under a Markov assumption. J Am Stat Assoc 1985;80:863–871 [Google Scholar]
- 8. Kay R. A Markov model for analysing cancer markers and disease states in survival studies. Biometrics 1986;42:855–865 [PubMed] [Google Scholar]
- 9. World Health Organization Expert Consultation . Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004;363:157–163 [DOI] [PubMed] [Google Scholar]
- 10. Mohan V, Deepa M, Anjana R, Lanthorn H, Deepa R. Incidence of diabetes and pre-diabetes in a selected urban South Indian population (CUPS-19). J Assoc Physicians India 2008;56:152–157 [PubMed] [Google Scholar]
- 11. Gujral UP, Narayan KMV, Kandula NR, Liu K, Kanaya AM. Incidence of diabetes and prediabetes and predictors of glycemic change among South Asians in the USA: the MASALA study. BMJ Open Diabetes Res Care 2020;8:e001063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Fazli GS, Moineddin R, Bierman AS, Booth GL. Ethnic differences in prediabetes incidence among immigrants to Canada: a population-based cohort study. BMC Med 2019;17:100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Abdul-Ghani MA, Tripathy D, DeFronzo RA. Contributions of β-cell dysfunction and insulin resistance to the pathogenesis of impaired glucose tolerance and impaired fasting glucose. Diabetes Care 2006;29:1130–1139 [DOI] [PubMed] [Google Scholar]
- 14. Faerch K, Borch-Johnsen K, Holst JJ, Vaag A. Pathophysiology and aetiology of impaired fasting glycaemia and impaired glucose tolerance: does it matter for prevention and treatment of type 2 diabetes? Diabetologia 2009;52:1714–1723 [DOI] [PubMed] [Google Scholar]
- 15. Nathan DM, Davidson MB, DeFronzo RA, et al. ; American Diabetes Association . Impaired fasting glucose and impaired glucose tolerance: implications for care. Diabetes Care 2007;30:753–759 [DOI] [PubMed] [Google Scholar]
- 16. Anjana RM, Unnikrishnan R, Deepa M, et al. ; ICMR-INDIAB Collaborative Study Group . Metabolic non-communicable disease health report of India: the ICMR-INDIAB national cross-sectional study (ICMR-INDIAB-17). Lancet Diabetes Endocrinol 2023;11:474–489 [DOI] [PubMed] [Google Scholar]
- 17. Weber MB, Ranjani H, Staimez LR, et al. The stepwise approach to diabetes prevention: results from the D-CLIP randomized controlled trial. Diabetes Care 2016;39:1760–1767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ramachandran A, Snehalatha C, Mary S, Mukesh B, Bhaskar AD; Indian Diabetes Prevention Programme (IDPP) . The Indian Diabetes Prevention Programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1). Diabetologia 2006;49:289–297 [DOI] [PubMed] [Google Scholar]
- 19. Sathish T, Khunti K, Narayan KMV, et al. Effect of conventional lifestyle interventions on type 2 diabetes incidence by glucose-defined prediabetes phenotype: an individual participant data meta-analysis of randomized controlled trials. Diabetes Care 2023;46:1903–1907 [DOI] [PMC free article] [PubMed] [Google Scholar]