Abstract
Background
While immunotherapy has been highly successful for the treatment of some cancers, for others, the immune response to tumor antigens is weak leading to treatment failure. The resistance of tumors to checkpoint inhibitor therapy may be caused by T cell exhaustion resulting from checkpoint activation.
Methods
In this study, lentiviral vectors that expressed T cell epitopes of an experimentally introduced tumor antigen, ovalbumin, or the endogenous tumor antigen, Trp1 were developed. The vectors coexpressed CD40 ligand (CD40L), which served to mature the dendritic cells (DCs), and a soluble programmed cell death protein 1 (PD-1) microbody to prevent checkpoint activation. Vaccination of mice bearing B16.OVA melanomas with vector-transduced DCs induced the proliferation and activation of functional, antigen-specific, cytolytic CD8 T cells.
Results
Vaccination induced the expansion of CD8 T cells that infiltrated the tumors to suppress tumor growth. Vector-encoded CD40L and PD-1 microbody increased the extent of tumor growth suppression. Adoptive transfer demonstrated that the effect was mediated by CD8 T cells. Direct injection of the vector, without the need for ex vivo transduction of DCs, was also effective.
Conclusions
This study suggests that therapeutic vaccination that induces tumor antigen-specific CD8 T cells coupled with a vector-expressed checkpoint inhibitor can be an effective means to suppress the growth of tumors that are resistant to conventional immunotherapy.
Keywords: Dendritic, Vaccine, Tumor infiltrating lymphocyte - TIL, Immune Checkpoint Inhibitor, T cell
WHAT IS ALREADY KNOWN ON THIS TOPIC
Cancer can be controlled by T cells that recognize tumor antigens. Checkpoint immunotherapy increases the effectiveness of T cells in controlling cancer by restoring function to exhausted T cells. In a significant fraction of patients, immunotherapy is not successful, most likely because there are not a sufficient number of T cells that target tumor antigens. Dendritic cells present tumor antigen epitopes to T cells on major histocompatibility complex class I but may not activate a sufficient number of antitumor T cells. Therapeutic dendritic cell vaccines that present tumor antigen to T cells have seen some clinical success. Lentiviral vectors provide a means to achieve long-term expression of transgenes by virtue of their ability to integrate into the host cell chromosome.
WHAT THIS STUDY ADDS
In this study, we transduced dendritic cells with lentiviral vectors that express tumor antigen epitope together with CD40 ligand, a protein that activates antigen presentation pathways of dendritic cells and a protein that acts as a checkpoint inhibitor. The vaccination, either by ex vivo dendritic cell transduction or direct injection of the viral vector, greatly slowed the growth of melanoma in the mice. The vaccination increased the number of CD8 T cells that infiltrated the tumor microenvironment and that were cytotoxic to tumor cells.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
Lentiviral vectors similar to these that express human tumor antigens could serve as therapeutic vaccines that would stimulate rare T cell clones to control solid tumor growth. The immunization could be combined with checkpoint antibody injection or might not require immunotherapy by virtue of the vector-encoded checkpoint inhibitor. Lentiviral vectors have proven safe in chimeric antigen receptor T cell therapies that involve ex vivo transduction of patient cells.
Introduction
Cancer immunotherapy has seen a major advance with the development of checkpoint inhibitors for the treatment of several types of cancer including melanoma, non-small cell lung cancer, bladder cancer, and other solid tumors.1–4 While checkpoint immunotherapy is successful in many patients, the success rate remains 30%–50% for melanoma, non-small cell lung cancer, and bladder cancer.5 Several factors are thought to influence the effectiveness of immunotherapy. A high mutational burden in the tumor is associated with better responses due to the expression of neoantigens that leads to the induction of antitumor CD8 T cells.6 7 In addition, the tumor microenvironment can be immunosuppressive as a result of T regulatory cells that dampen the functionality of infiltrating T cells8 9 and myeloid suppressor cells that similarly negatively regulate antiviral tumor T cell activity.10 11
One approach to increase the success rate of immunotherapy is that of therapeutic vaccination. The combination of therapeutic vaccination with checkpoint inhibitors may synergize to stimulate antigen-specific T cells with enhanced T cell function,12 13 resulting in stronger immune responses. In this approach, vaccination serves to enhance the T cell response to tumor antigens and thereby synergize with checkpoint inhibitors. Dendritic cell (DC) vaccines use patient-derived DCs pulsed with synthetic peptide epitope or exposed to tumor-associated antigens and reinfused to activate tumor-specific T cells.14–16 Alternatively, DCs can be transduced with a tumor antigen-expressing viral vector, an approach that can result in longer-term antigen expression and in which antigen is expressed endogenously, resulting in more efficient antigen processing and presentation on major histocompatibility complex (MHC) class I without the need for cross-presentation.17–19
Lentiviral vectors have several features that are advantageous for therapeutic vaccination. They integrate into transcriptionally active sites in chromosomal DNA resulting in long-term stable expression20–22; they do not encode any viral proteins and, as opposed to other viral vectors, such as adenoviral vectors, are generally not subject to pre-existing neutralizing antibody in humans.23 The vectors do not specifically target DCs, but the efficiency with which they transduce myeloid cells can be improved by incorporation of the lentiviral accessory protein Vpx into the virion which counteracts the inhibitory activity of the myeloid restriction factor SAM and HD domain containing deoxynucleoside triphosphate triphosphohydrolase 1 (SAMHD1).24–31 Vpx-containing lentiviral vectors transduced DCs with a 2-log increase in titer.24 32 While Vpx effectively counteracts human SAMHD1, it fails to counteract murine SAMHD1 and, as a result, does not increase the lentiviral vector transduction frequency of mouse DCs.33 For mouse studies, SAMHD1 knockout mice (SAMHD1 KO) can be used to increase transduction efficiencies similar to what is achieved using Vpx-containing vectors on human DCs.34
We previously reported on a lentiviral vector-based DC vaccine against HIV-1, lymphocytic choriomeningitis virus (LCMV), and SARS-CoV-2 that prevented infection in mice and reduced virus loads in acute and chronically infected mice.34 35 The vector encoded a single CD8 T cell epitope and coexpressed CD40 ligand (CD40L) that served to mature and activate the DCs.24 32 Immunization of mice with transduced DCs or by direct injection of the viral vector into mice induced a strong CD8 T cell response that protected mice from infection. In chronically infected mice, the introduction of a PD-1 microbody consisting of the PD-1 ectodomain fused to an IgG1 CH3 region into the vector enhanced antiviral cytotoxic T lymphocyte (CTL) activity and reversed T cell exhaustion.36 The microbody protein was secreted as a dimer that bound programmed death-ligand 1 (PD-L1) on the DCs, preventing the interaction of PD-L1 on the DCs with PD-1 on T cells,32 thereby preventing checkpoint activation and T cell exhaustion. LCMV epitopes also strongly induced CTL response and prevented and cured LCMV infection in mouse model.34 Direct injection resulted in long-term stable gene expression.37
In this study, we tested whether a similar lentiviral vector-based vaccine encoding tumor antigen epitopes could be used to immunize against subsequent introduction of melanoma or colon carcinoma tumors either by the injection of transduced DCs or by direct injection of the vector. The therapy synergized with the injection of an anti-PD-L1 monoclonal antibody. Alternatively, the checkpoint inhibitor could be encoded by the lentiviral vaccine vector in the form of a PD-1 microbody, obviating the need to treat with a high dose of monoclonal antibody. The results support the effectiveness of therapeutic lentiviral vector-based DCs that both expand antitumor CD8 T cells and prevent checkpoint activation.
Results
Immunization with lentiviral vector-transduced DCs suppresses tumor growth in preclinical tumor models
DCs orchestrate the T cell response to pathogens and neoantigen-expressing tumors. To harness the antigen-presenting function of DCs, we constructed lentiviral vectors that coexpressed an MHC class I restricted T cell epitope and the immunostimulatory protein CD40L. Lentiviral vectors are advantageous because they stably express transgenes in DCs and are compatible with insert sizes capable of expressing multiple open reading frames.32 34 37 They also provide a means to coexpress cytokines that further potentiate the cellular immune response. Vectors were constructed that expressed the MHC class I epitope ovalbumin (OVA)257-264 (OVA1) and MHC class II epitope OVA323-339 (OVA2), either separately or in combination, or the tyrosinase-related protein 1 (TRP1455-463) tumor antigen, a self-protein that is overexpressed on tumors.38 The peptide epitopes were expressed as fusions to CD40L, a potent inducer of DC maturation and activation, separated by a P2A self-cleaving epitope to ensure equimolar expression of the proteins. Additional vectors were constructed that coexpressed a PD-1 microbody. The microbody is a secreted form of the extracellular domain of PD-1 fused to the CH3 domain of a human IgG1 immunoglobulin heavy chain.32 The microbody binds PD-L1 on responding T cells, inhibiting checkpoint activation as they interact with antigen-presenting cells (APCs). Control vectors expressed CD40L alone or CD40L and the PD-1 microbody (figure 1A).
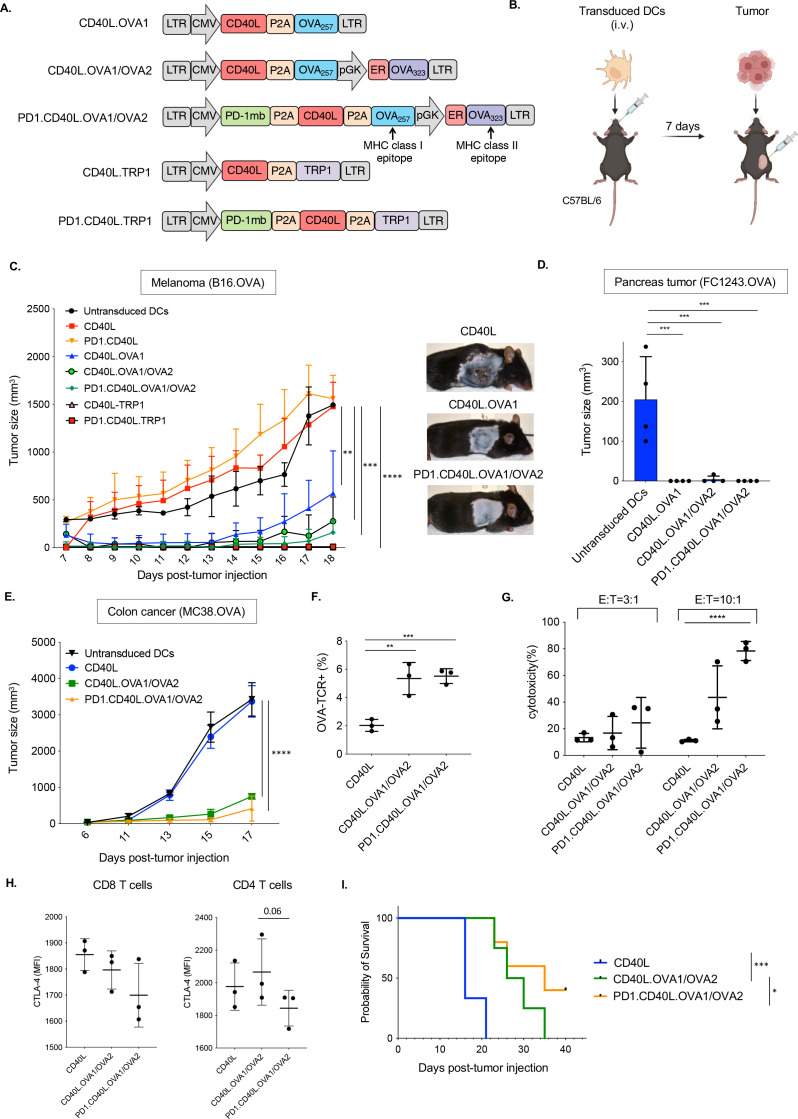
Figure 1.
Immunization with lentiviral vector-transduced DC vaccine vectors slows the growth of solid tumors. (A) The structure of lentiviral vaccine vectors is diagrammed. The vectors express CD40L, OVA class I and class II restricted T cell epitopes OVA257-264 (OVA1) and OVA323-339 (OVA2), and TRP1 and the PD-1 microbody (PD-1mb). (B) DCs were transduced with each lentivirus and then injected intravenously into C57BL/6 mice (1×106 cells). One week later, the mice were inoculated subcutaneously with 2.5×105 B16.OVA cells. (C) The tumor size was measured over 18 days (n=4). (D) DCs were transduced with vaccine vectors and injected intravenously. One week later, the mice were inoculated orthotopically with 1×106 FC1242.OVA tumor cells. Tumor size was measured at 21 days post-DC injection (n=4). (E) Transduced DCs were injected intravenously. After 7 days, the mice were inoculated with 1×106 MC38.OVA colon carcinoma cells. Tumor sizes were measured over 17 days (n=3). (F) Mice were immunized with transduced DCs. After 7 days, the splenocytes were analyzed by flow cytometry with OVA-specific class I tetramers CD8+T cells, gating on CD8+cells. (G) The cytolytic activity of splenocytes from DC-immunized mice against MC38.OVA target cells was measured in an in vitro cytolysis assay. The MC38.OVA target cells were stained with CFSE and incubated for 24 hours with splenocyte effectors at ratios of 1:3 and 1:10. The percentage of lysed cells was then determined by staining with viability dye and analysis by flow cytometry. (H) Survival of the mice following immunization and MC38.OVA implantation is shown (n=5). **p≤0.01, ***p≤0.001, ****p≤0.0001. DCs, dendritic cells ; CD40L, CD40 ligand; OVA, ovalbumin; TRP1, tyrosinase-related protein 1; PD-1, programmed cell death 1; CFSE, carboxyfluorescein succinimidyl ester.
To test the ability of the vectors to induce a protective immune response, C57BL/6 mice were immunized with vector-transduced DCs and 7 days later injected with B16.OVA melanoma cells (figure 1B). In mice injected with CD40L or PD-1.CD40L control vector-transduced DCs, tumors grew as rapidly as in mock-immunized mice (figure 1C). In contrast, in mice injected with DCs transduced with OVA or TRP1 peptide epitope-expressing vectors, melanoma growth was greatly reduced. The CD40L.OVA1 vector was only partially effective at limiting tumor growth. The addition of the OVA2 epitope increased its effectiveness as did the addition of the PD-1 microbody. The TRP1-expressing vectors, with or without the PD-1 microbody, appeared to fully prevent tumor growth. The OVA vectors, with and without the OVA2 epitope or PD-1 microbody, were also effective at preventing tumor growth in the orthotopic pancreatic cancer model with FC1243.OVA cells (figure 1D) and in the MC38.OVA colon cancer model (figure 1E).
17 days post-tumor inoculation, the antigen-specific CD8 T cell response induced by the DC immunization was determined by analyzing the number of OVA-specific CD8 T cells using an MHC class I OVA tetramers. In mice immunized with control CD40L vector-transduced DCs, about 2% of the CD8 T cells were tetramer positive. Immunization with CD40L.OVA1/OVA2 vector-transduced DCs increased the number of tetramer+CD8 T cells from 3% to 6% (figure 1F). The addition of the PD-1 microbody showed a similar number of antigen-specific CD8 T cells. The cytolytic activity of the CD8 T cells was determined by an in vitro assay in which splenocytes from the immunized mice were incubated with carboxyfluorescein succinimidyl ester (CFSE)-labeled MC38.OVA target cells at E:T ratios of 3:1 and 10:1. The number of lysed target cells was determined after overnight incubation by flow cytometry. The results showed that the CD8 T cells from the immunized mice killed the targets (figure 1G). Cytolytic activity of the CD8 T cells from mice immunized with the PD-1 microbody vector was significantly increased compared with those from mice immunized with vectors without the PD-1 microbody. PD-1.CD40L.OVA1/OVA2 DCs slightly decreased exhaustion marker expression level (CTLA-4) both in CD8+ and CD4+T cell (figure 1H). The analysis of survival rates showed that mice vaccinated with CD40L vector died 16–21 days post-tumor inoculation, while the mice vaccinated with CD40L.OVA1/OVA2 DCs died 35 days post-tumor inoculation, and 40% of mice vaccinated with PD.1.CD40L.OVA1/OVA2 DCs survived (figure 1I). To examine the CD40L effect in antitumor activity, lentiviral vectors expressing CD40L or a mutated inactive CD40L (mutCD40L) were generated (online supplemental figure 1A). Mice were immunized with transduced DCs, and B16.OVA cells were inoculated in mice 7 days postimmunization (online supplemental figure 1B). Immunization with the mutCD40L resulted in tumors that were more than twice the mass of those immunized with active CD40L-expressing vector (online supplemental figure 1C). In addition, mutCD40L resulted in a small decrease in interferon gamma (IFNγ)+ CD8 T cells (online supplemental figure 1D). These data demonstrate the importance of CD40L in the induction of antitumor activity in vivo.
jitc-2023-008761supp001.pdf (1.2MB, pdf)
Vaccination with lentiviral vector-transduced DCs suppresses the growth of established tumors regardless of the route of administration
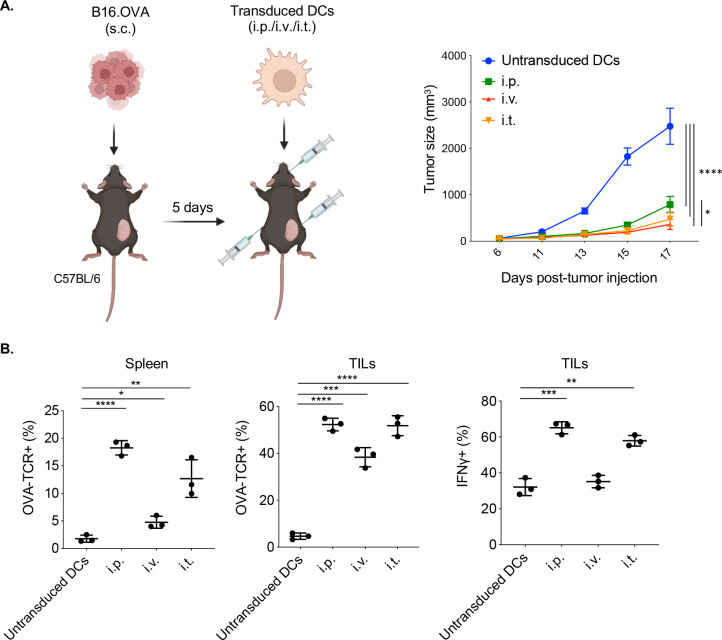
A comparison of the routes of DC immunization showed that intraperitoneal, intravenous, and intrathecal injection were similarly effective in suppressing tumor growth (figure 2A). Intraperitoneal injection of CD40L.OVA1/OVA2 vector-transduced DCs resulted in the largest increase in antigen-specific CD8 T cells in the spleen (18%), and intravenous injection, the smallest (4%) (figure 2B, left). The analysis of the TILs showed an enrichment of antigen-specific CD8 T cells in the tumor, reaching about 50% with a similar ratio among the three routes of DC introduction (figure 2B, middle). IFNγ levels in the antigen-specific CD8+ tumor-infiltrating lymphocytes (TILs) followed a similar pattern, with intraperitoneal injection resulting in the highest levels of IFNγ and perforin and intravenous, the lowest (figure 2B, right).
Figure 2.
Comparison of tumor growth suppression and T cell responses by intraperitoneal, intravenous, and intrathecal injection of transduced DCs. (A) Mice were injected with B16.OVA colon carcinoma cells and after 5 days injected intraperitoneally, intravenously, and intrathecally with CD40L.OVA1/OVA2 lentiviral vector-transduced DCs. Tumor sizes were measured over 17 days (n=3). (B) OVA tetramer+CD8+ T cells in the spleen and tumors (TILs) of the vaccinated mice were quantified by flow cytometry. Intracellular IFNγ and perforin levels in the antigen-specific (TCR+) CD8+TILs of the vaccinated mice were quantified by flow cytometry. *p≤0.05, **p≤0.01, ***p≤0.001, ****p≤0.0001. DCs, dendritic cells; TILs, tumor-infiltrating lymphocytes.
Vaccination with lentiviral vector-transduced DCs suppresses the growth of established tumors
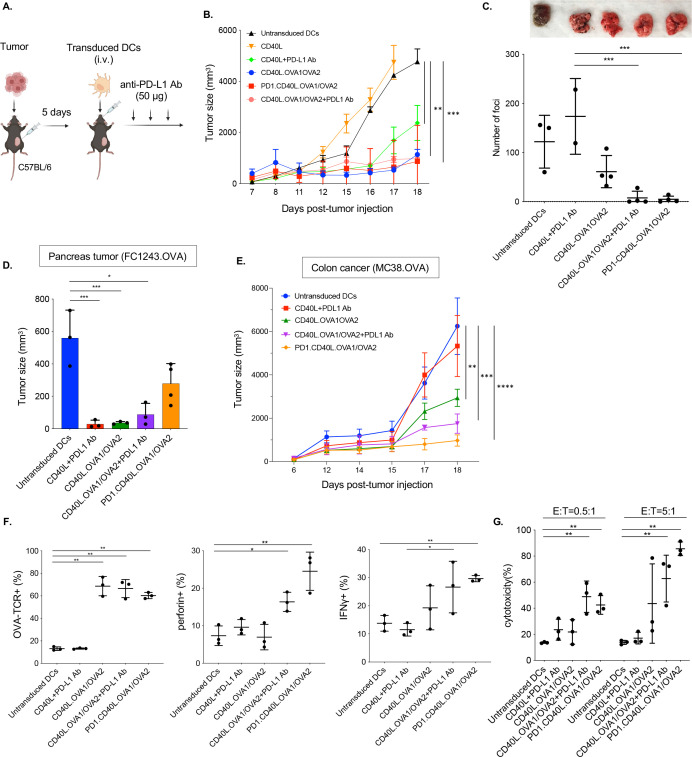
To determine whether the DC immunization would slow the growth of established tumors, mice were injected first with B16.OVA melanoma cells and then immunized by intravenous injection of DCs transduced with CD40L.OVA1/OVA2 or PD-1.CD40L.OVA1/OVA2 vector (figure 3A). The results showed that both vectors suppressed tumor growth (figure 3B). Intravenous injection of B16.OVA cells results in metastatic disease that is difficult to treat. To determine whether DC immunization could treat metastatic tumors, intravenously, and 5 days later, the mice were immunized with transduced DCs. To determine whether the antitumor effect could be enhanced by checkpoint inhibition, another group of mice was immunized and treated with three injections of anti-PD-L1 antibody. At 25 days posttumor initiation, there were numerous foci in the lungs of mice immunized with mock-transduced or control vector-transduced DCs. Immunization with the CD40L.OVA1/OVA2 vector decreased the number of foci by about twofold (figure 3C). In contrast, immunization with the vector encoding the PD-1 microbody eliminated visible metastases. Supplementation of the vaccination with an injection of anti-PD-L1 antibody similarly resulted in the absence of visible foci. The effect was not simply caused by the checkpoint inhibitor as control immunization with empty vector-transduced DCs and anti-PD-L1 antibody injection did not decrease the number of foci.
Figure 3.
Lentiviral vector-based DC vaccine-induced CTL activity and cured established tumors. (A) The experimental scheme is diagrammed. Mice were injected subcutaneously or intravenously with 2.5×105 B16.OVA cells and, after 5 days, injected intravenously with transduced DCs, with or without three injections every 3 days of 50 µg anti-PD-L1 antibody (n=4). (B) The tumor size was measured over 18 days. (C) Lung foci were counted 30 days after intravenous injection (n=4). One mouse (untransduced DCs) and two mice (CD40L+PD-L1 Ab) died before analysis, and data were not shown in the graph. Photographs of the lungs are shown above. (D) Mice were injected orthotopically with FC1242.OVA cells and then with transduced DCs. After 21 days, the size of tumors in the pancreas was measured (n=3–4). (E) Mice were injected with MC38.OVA and then immunized with transduced DCs by intravenous injection. Tumor size was measured over 18 days (n=3). (F) TILs were isolated from the immunized mice at day 18. The number of CD8+TILs was quantified by flow cytometry. The proportion of OVA tetramer+, IFNγ+, and perforin+CD8+ TILs were determined. (G) The cytotoxic activity of the CD8+T cells against MC38.OVA targets was determined. MC38.OVA target cells were stained with CFSE and incubated with TILs of immunized mice. After 24 hours, the cells were stained with viability dye, and the percentage of lysed cells was determined by flow cytometry. *p≤0.05, **p≤0.01, ***p≤0.001, ****p≤0.0001. DC, dendritic cell; TILs, tumor-infiltrating lymphocytes; OVA, ovalbumin; TCR+, T cell receptor; CTL, cytotoxic T lymphocytes; OVA, ovalbumin; PD-L1, programmed death-ligand 1; CFSE, carboxyfluorescein succinimidyl ester.
To test the effectiveness of vaccination against other tumor types, mice with orthotopic FC1243.OVA pancreatic tumors or MC38.OVA colon cancer cells were vaccinated with transduced DCs with or without anti-PD-L1 antibody injections. In mice immunized with CD40L.OVA1/OVA2 with or without anti-PD-L1 antibody, tumor growth was greatly reduced. Pancreatic tumors were partially suppressed by immunization with the PD-1.CD40L.OVA1/OVA2 vector (figure 3D). In addition, vaccination with the CD40L.OVA1/OVA2 partially suppressed the growth of the colon cancer tumors. In mice with colon cancer, tumor suppression was enhanced by subsequent injection of anti-PD-L1 antibody or by the inclusion of the PD-1 microbody in the vaccine vector (figure 3E).
The immune response stimulated by DC vaccination was analyzed in the TILs of the immunized mice. The immunizations increased the number of antigen-specific CD8 T cells from 12% to 60%–70% in control DC-immunized mice, regardless of whether a checkpoint inhibitor was included (figure 3F, left). Immunization with the CD40L.OVA1/OVA2 vector did not increase perforin expression in the antigen-specific CD8 T cells, but the addition of anti-PD-L1 antibody or the use of the vector encoding the PD-1 microbody increased the number of perforin-expressing cells by threefold (figure 3F, middle). Similar results were found for IFNγ with the vector encoding the PD-1 microbody showing the most consistent increase (figure 3F, right). Quantification of cytolytic activity of the TILs against MC38.OVA target cells showed results consistent with perforin/IFNγ expression. The addition of anti-PD-L1 antibody increased the cytolytic activity of cells immunized with the OVA1/OVA2 vector, and the cytolytic activity was greatest in the TILs of mice immunized with the PD-1 microbody-expressing vector (figure 3G).
Vaccination by direct injection of lentiviral vector
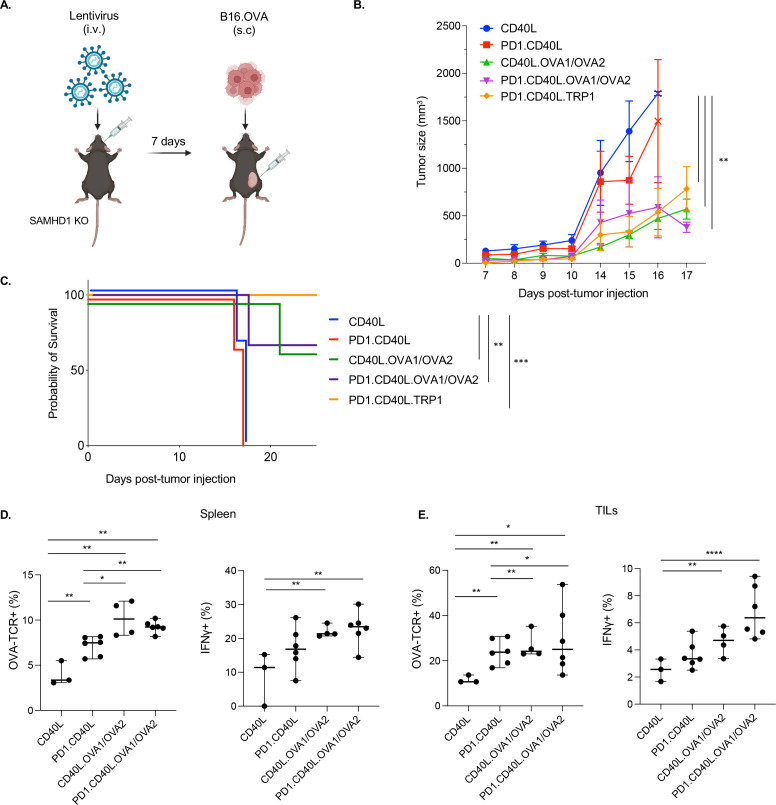
In the experiments described above, mice were vaccinated using ex vivo transduced DCs, a strategy that targets the vector to DCs. Direct injection of the vector, if effective, would simplify the procedure. Whether this route would induce a robust immune response is unclear as the vesicular stomatitis virus-glycoprotein (VSV-G) pseudotyped vector has a broad tropism and might not result in the transduction of sufficient numbers of antigen-presenting myeloid cells. To test the effectiveness of direct vector injection, mice were injected intravenously with the CD40L.OVA1/OVA2 vectors and, after 7 days, inoculated by subcutaneous injection of B16.OVA melanoma cells (figure 4A). The measurement of tumor size over the next 17 days showed that immunization with the control PD-1.CD40L vector had a modest effect on tumor growth, while the CD40L.OVA1/OVA2 and PD-1.CD40L.OVA1/OVA2 vectors strongly suppressed tumor growth. The suppression was slightly enhanced for the PD-1.CD40L.TRP1 vector, although the difference was not statistically significant (figure 4B). The analysis of survival rates following immunization showed that mice vaccinated with the control CD40L and PD-1.CD40L vectors died 17–18 days post-tumor inoculation, while 60% of mice vaccinated with the CD40L.OVA1/OVA2 epitope-expressing vectors and 100% of mice vaccinated with the PD.1.CD40L.TRP1 vector survived (figure 4C).
Figure 4.
Effective vaccination by direct injection of lentiviral vector. (A) The experimental protocol is diagrammed. (B) SAMHD1 knockout mice were injected intravenously with 3×106 I.U. of CD40L, PD-1.CD40L, CD40L.OVA1/OVA2, PD-1.CD40L.OVA1/OVA2, or PD-1.CD40L.TRP1 vectors (n=4–6) and, after 7 days, injected subcutaneously with 2.5×105 B16.OVA melanoma cells. Tumors were measured over 12 days. (C) Survival of the mice following immunization and tumor implantation is shown (n=4). (D) The fraction of antigen-specific CD8 T cells and the fraction of IFNγ+ CD8 T cells in the spleen were determined by flow cytometry (n=4–6). (E) The fraction of antigen-specific CD8 TILs and the fraction of IFNγ+ CD8 TILs were determined by flow cytometry. *p≤0.05, **p≤0.01, ****p≤0.001, ****p≤0.0001. IFNγ, interferon gamma; TILs, tumor-infiltrating lymphocytes; I.U., infectious unit.
To understand the basis of the protective immune response raised by direct vector injection, we analyzed the number and functionality of antigen-specific CD8 T cells that had infiltrated the tumors. In the spleen, vaccination with the PD-1 vector caused a twofold increase in antigen-specific CD8+T cells, and this was further increased by vaccination with the CD40L.OVA1/OVA2 and PD-1.CD40L.OVA1/OVA2 vectors (figure 4D). Within the tumor, the proportion of antigen-specific CD8 T cells was about twofold higher, suggesting an enrichment of antigen-specific CD8 T cells, presumably due to homing or expansion of the cells (figure 4E).
To test the durability of the immune response elicited by direct injection of lentiviral vector, wild-type and SAMHD1 KO mice were vaccinated with CD40L.TRP1 or PD-1.CD40L.TRP1 lentiviral vector and then challenged 7 or 30 days later by inoculation with B16 tumor cells (online supplemental figure 2A). The SAMHD1 KO removes SAMHD1 from the myeloid cells, potentially increasing the number of transduced DCs and increasing vaccine effectiveness. The results showed that in mice challenged 7 days postvaccination, both vectors were effective in wild-type and SAMHD1 KO mice, with a slight advantage of the PD-1 microbody-expressing vector. In mice challenged 30 days postvaccination, tumor suppression in the wild-type mice had lost some of its strength but remained strong in the SAMHD1 KO for the PD-1 microbody-expressing vector (online supplemental figure 2B). This result suggests that the immune response is more durable in the absence of SAMHD1, arguing for the benefit in human clinical application of Vpx-containing vectors.
Checkpoint inhibitor synergizes with injected lentiviral vector to decrease CD8 T cell exhaustion and suppress metastatic disease
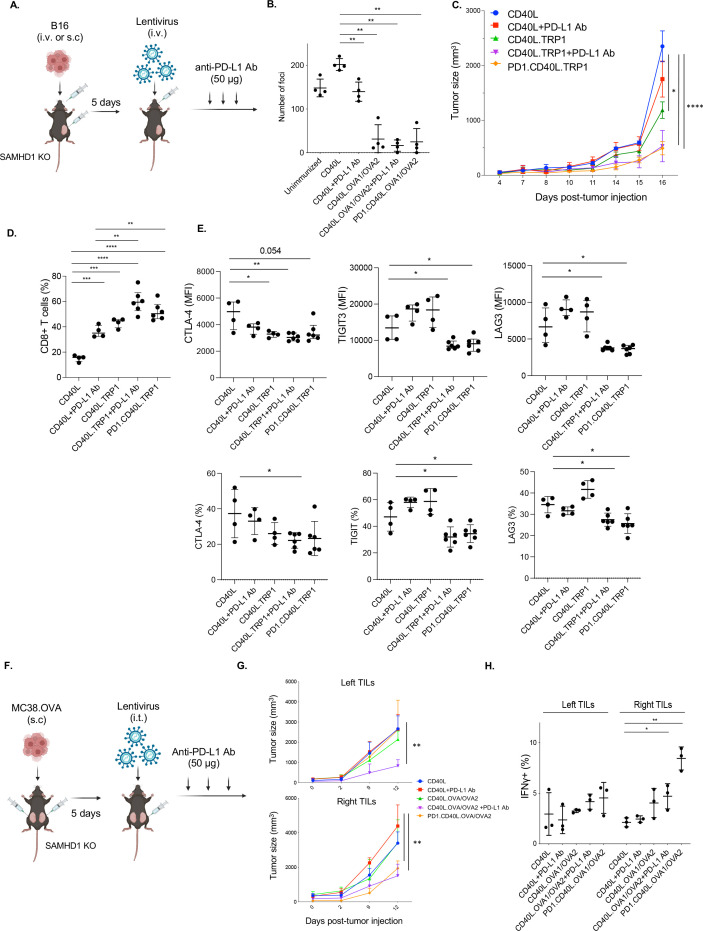
Vaccination by direct vector injection was effective in suppressing the growth of B16.OVA metastasis. Metastases formed following intravenous injection of the tumor cells were decreased to nearly undetectable numbers on vaccination, both with and without anti-PD-L1 or vector-encoded PD-1 microbody checkpoint inhibitors (figure 5A,B). To determine whether checkpoint inhibition would synergize with vaccination by direct injection, mice were injected in the right flank with B16 melanoma cells and, after 5 days, injected intravenously with CD40L.TRP1 or PD-1.CD40L.TRP1 lentiviral vector (figure 5A). Mice immunized with the CD40L.TRP1 vector were supplemented with anti-PD-L1 antibody postimmunization. The vaccinations suppressed tumor growth (figure 5C). Anti-PD-L1 and vector-encoded PD-1 microbody did not further enhance the growth suppression; however, both checkpoint inhibitors increased the number of CD8+TILs (figure 5D). Anti-PD-L1 decreased CTLA4 levels in the CD8 T cells. Moreover, vaccination together with anti-PD-L1 antibody or vector-encoded PD-1 microbody caused significant decreases in CD8 T cell TIGIT3 and LAG3 expression levels (figure 5D).
Figure 5.
Lentiviral vaccine and checkpoint inhibitor combination therapy boosts CTL activity. (A) As diagrammed, SAMHD1 KO mice were injected intravenously or subcutaneously with B16.OVA melanoma cells (n=4–6) and, 5 days later, injected intravenously with 3×106 I.U. of lentiviral vectors encoding CD40L, CD40L.TRP1, or PD-1.CD40L.TRP1. In additional groups, the vaccinated mice with CD40L and CD40L.TRP1 were treated three times every other day with anti-PD-L1 antibody starting 5 days postvaccination. (B) The effect of vaccination on the number of metastatic lung foci was determined. SAMHD1 KO mice were injected with B16.OVA intravenously (n=4) and, after 4 days, injected intravenously with lentiviral vaccine vector. An additional set of CD40L and CD40L/OVA1/OVA2 vector-vaccinated mice were treated with anti-PD-L1 antibody (50 µg) injected every 3 days. After 25 days, metastatic foci in the lungs were counted. (C) Tumor size in tumor-bearing mice (subcutaneous injection) was measured beginning on the day of vaccination. (D) The fraction of CD8 T cells in the spleen was determined by flow cytometry (n=4–6). (E) Mean fluorescence intensity (MFI: top) and the percentage (bottom) of exhaustion markers CTLA-4, TIGIT3, and LAG3 on the CD8+T cells were analyzed by flow cytometry. (F) As diagrammed, mice were injected subcutaneously with MC38.OVA on the left and right flanks. The tumor on the right side was then injected with the vaccine lentiviral vector followed by three injections of anti-PD-L1 (n=3). (G) Tumor size on the right and left sides was measured over 12 days starting from the day of vaccination. (H) The number of IFNγ+ CD8+ T cells was analyzed by flow cytometry. *p≤0.05, **p≤0.01, ***p≤0.001, ****p≤0.0001. KO, knockout; IFNγ, interferon gamma; CTL, cytotoxic T lymphocytes; OVA, ovalbumin; PD-L1, programmed death-ligand 1; TRP1, tyrosinase-related protein 1; CTLA4, cytotoxic T-lymphocyte-associated protein 4; TIGIT3, T-cell immunoreceptor with immunoglobulin and immunoreceptor tyrosine-based inhibitory motif domains 3; LAG3, lymphocyte activation gene 3; I.U., infectious unit.
Tumor growth suppression at a distance
Direct injection of vaccine vector into the tumor might result in immunity localized to the site of injection that does not become systemic. To test this possibility, tumors were established in mice on the left and right dorsal flanks. The tumor that appeared on the left flank was then injected with lentiviral vaccine vector CD40L.OVA1/OVA2, PD-1.CD40L.OVA1/OVA2, or CD40L.OVA1/OVA2 followed by intraperitoneal injection of anti-PD-L1 antibody (figure 5E). The results showed that the OVA1/OVA2 vector alone was not suppressive on either side, but that when anti-PD-L1 antibody was added to the vaccination, tumor growth was suppressed on both the right and left sides, indicating that the immune response triggered on the right side could be transferred to the left side. In contrast, the PD-1.CD40L.OVA1/OVA2 vector suppressed tumor growth only on the right side where it had been injected (figure 5F). The analysis of IFNγ levels confirmed that the increase in IFNγ+ CD8 T cells was specific to the right side (figure 5G). Thus, when the checkpoint inhibitor is present only locally at the site of T cell activation, the protection is localized to that site; however, when the checkpoint inhibitor is present systemically, as in the case of anti-PD-L1 antibody injection, the protection is more widespread. These findings suggest that checkpoint blockade needs to occur when the T cell becomes activated and to remain present over time to prevent T cell exhaustion as the T cell migrates to another site in the body.
Preferential transduction of DCs and myeloid cells by direct injection
To determine which cell types are transduced by direct injection, tumor-bearing mice were injected intraperitoneally, intravenously, and intrathecally with GFP-expressing lentiviral vector, and after 5 days, splenocytes and TILs were analyzed by flow cytometry with antibodies that distinguished total leukocytes (CD45+), CD4 T cells (CD3+/CD4+), CD8 T cells (CD3+/CD8+), B cells (CD3-/CD19+), NK cells (CD3-/CD49b+), monocytes (CD115-/CD11b+), and DCs (CD11c+/MHCII+). By intraperitoneal and intravenous injection, the majority of cells transduced were DCs. Smaller numbers of monocytes and B cells were also transduced (online supplemental figure 3). By intrathecal injection, the largest number of transduced cells was the tumor cells themselves, with the other cell types present in small numbers.
Tumor suppression is mediated by CD8 T cells
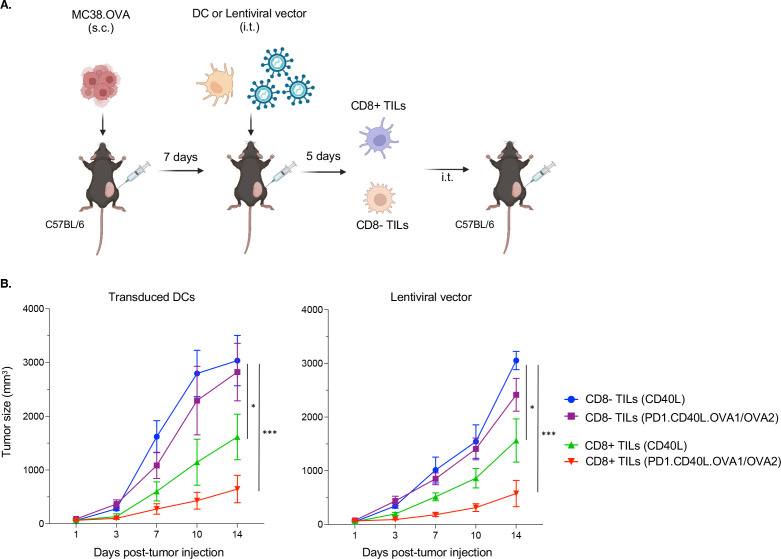
To determine whether CD8 T cells were sufficient to suppress tumor growth, we tested purified CD8+TILs from mice vaccinated by transduced DCs or direct injection by adoptive transfer. The mice were immunized with control CD40L or PD-1.CD40L.OVA1/OVA2 vectors. After 1 week, TILs were isolated and separated on magnetic beads into CD8+ and CD8− populations (figure 6A). The populations were then injected separately intrathecally into tumor-bearing recipients. The results showed that the CD8+ but not the CD8− population suppressed tumor growth (figure 6B).
Figure 6.
Tumor suppression is mediated by CD8+T cells. (A) MC38.OVA tumor-bearing mice were immunized with transduced DCs or by direct injection with lentiviral vector (CD40L or PD-1.CD40L.OVA1/OVA2). Seven days postimmunization, TILs were separated into CD8+ and CD8− populations on magnetic beads and injected intrathecally into tumor-bearing mice. The CD8 population was 95% pure, while the CD8− population was 99% pure. (B) After adoptive transfer of CD8+ and CD8− TILs, tumor size (n=5) was monitored. *p≤0.05, ***p≤0.001. DCs, dendritic cells; TILs, tumor-infiltrating lymphocytes.
Discussion
The results presented here demonstrate the effectiveness of a checkpoint inhibitor-encoding lentiviral vector in suppressing solid tumor growth in preclinical mouse models of melanoma and colon cancer. The delivery of the vaccine by lentiviral vector transduction provided extended expression in DCs that expanded the number of antitumor T cells and prevented checkpoint-driven T cell exhaustion. The vaccination was effective using ex vivo transduced DCs or by direct injection of lentiviral vector and could be used prophylactically, before tumor initiation, or therapeutically, post-tumor initiation. Both routes induced the proliferation of epitope-specific CD8 T cells that expressed markers indicative of their functionality and were cytolytic for the tumor cells. Adoptive transfer showed that vaccine-induced CD8 T cells were sufficient to suppress tumor growth. The T cell response to vaccination was enhanced in combination with PD-1 blockade, either through injection of anti-PD-L1 antibody or through vector-encoded a PD-1 microbody.
Treatment with anti-PD-L1 antibody or with a vector-encoding PD-1 microbody enhanced the effectiveness of vaccination. The vector-encoded checkpoint inhibitor appeared to have a greater effect on tumor growth, an effect that might be attributable to the localization of the checkpoint inhibitor to the APC, at the site of T cell activation. When tumors were initiated on both flanks and the vaccine was administeredintrathecally on the right, the PD-1 microbody-expressing vector suppressed tumor growth on the right but not the left flank, supporting the conclusion that its effect is localized to the site of T cell activation. The vector-encoded inhibitor simplifies the treatment protocol by omitting the multiple injections of large amounts of potentially inflammatory recombinant monoclonal antibody. While checkpoint inhibitor-encoding lentiviral vector therapy could also have unwanted inflammatory consequences, evidence of generalized inflammation was not detected. In mice treated with CD40L or PD-1 microbody-expressing vectors, blood leukocyte subpopulations were not altered, there was no non-specific T cell activation, and proinflammatory cytokine levels measured by flow cytometry were not increased (data not shown).
Overall, transduced DCs induced a more effective immune response than direct lentiviral vector injection. Transduction of DCs focuses the vaccine vector on DCs as opposed to direct injection in which the VSV-pseudotyped vector transduces several cell types in the body. Transduced DCs are also advantageous because their half-life is on the order of 7 days, limiting the possibility of adverse consequences that could result from long-term expression of potentially inflammatory cytokines and antigens. On the other hand, the long-term expression that occurs by direct injection of lentiviral vectors could have the benefit of providing continued immune stimulation of antigen-specific T cells that would lead to sustained tumor growth suppression. Although we did not elucidate the main source of antitumor activity, we previously demonstrated that DCs were the main targets of the vectors when directly injected, both in SAMHD1 KO and wild-type mice. B cells and monocytes were also targeted but to a much lesser extent.39 This specificity is at least partially due to the permissivity of myeloid cells to lentiviral vector transduction, while resting T cells and other cell types are resistant. In a previous study using lentiviral vectors expressing LCMV peptide epitopes, we found that the protective effect was mediated by CD8+T cells.39 While direct injection of lentiviral vectors is not yet approved for clinical use, lentiviral vectors are in use clinically in chimeric antigen receptor T cell approaches in which T cells are transduced ex vivo; adverse consequences resulting from insertional mutagenesis or clonal proliferation have not been reported to our knowledge.
The treatment of melanoma, lung, bladder, and renal cell cancers with checkpoint inhibitor antibodies against CTLA-4, PD-1, and PD-L1 restores CTL function and causes tumor remission in some patients. The timing of PD-1/PD-L1 blockage is critical for the T cell response.39–41 While anti-PD-L1 antibody treatment during the DC-mediated antigen-specific naïve T cells may have a negative effect, inhibition of the pathway after the T cells differentiate into effectors has a positive effect.42 43 PD-L1 blockade initiated shortly after vaccination in mice proved synergistic in strengthening the vaccine-induced antitumor immune response, suggesting that the timing of administration led to an enhanced effector memory response that overcame CTL exhaustion.
The efficiency of lentiviral vector transduction of DCs is limited by SAMHD1 which imposes a block to reverse transcription in myeloid cells. The block can be alleviated by the use of vectors that contain the lentiviral accessory protein Vpx which facilitates the degradation of SAMHD1 in the newly transduced cell.24–31 Vpx does not induce the degradation of SAMHD1, but the potential usefulness of Vpx-containing vectors in humans was modeled here using SAMHD1 KO mice.34 The DCs of the mice are transduced 2–5-fold more efficiently than wild type.34 The immune response generated in the SAMHD1 KO mice was more durable as shown by better tumor growth suppression when tumor initiation was delayed 30 days postvaccination, a result supporting the use of Vpx-containing vectors in clinical applications.
The findings presented here suggest that the success rate of checkpoint inhibitor therapy can be increased in combination with therapeutic vaccination. The combined approach could stimulate rare T cell clones to respond to “cold” tumors that lack a proinflammatory immune response. The effectiveness of this approach will increase with the identification of additional tumor antigens and with improvements in MHC I-restricted peptide epitope prediction algorithms, opening the possibility of personalized vaccines for the treatment of malignancies.
Materials and methods
Study approval
Animal research was done under a protocol approved by the NYU Animal Research Committee (#170304) and was in compliance with federal, state, and local guidelines.
Plasmids
To construct pLenti.CD40L-OVA257-264 (OVA1), CD40L was joined to P2A and the codon-optimized OVA257 peptide encoding the SIINFEKL epitope44 45 by overlap extension PCR. The amplicon was cleaved with Xba-I and Sal-I and ligated to cleaved pLenti.CMV.GFP.puro. To construct pLenti.CD40L.OVA257-264 (OVA1)/OVA323-339 (OVA2), an amplicon encoding in-frame to codon-optimized DNA sequence encoding the peptide epitope OVA2 (323ISQAVHAAHAEINEAGR339)46 flanked by Pst-I and Xho-I sites was generated. The amplicon was then ligated 3’ to the phosphoglycerate kinase 1 promoter in pLenti.CD40L.OVA1 or to pLenti.PD-1mb-CD40L. To construct pLenti.CD40L.TRP1, CD40L was joined to P2A and the codon-optimized peptide epitope mTRP1 (455TAPDNLGYM463) by overlap extension PCR. The amplicon was cleaved with Xba-I and Sal-I and ligated to cleaved pLenti.CMV.GFP.puro. To generate pLenti.cOVA.puro, cOVA was amplified by PCR from pCDH-cOVA (Addgene) using primers containing a 5’-XbaI and 3’-SalI site. The amplicon was digested with Xba-I and Sal-I and ligated to similarly cleaved pLenti.CMV.GFP.puro (Addgene). To generate pLenti.PD-1.CD40L.OVA1/OVA2, an amplicon was constructed expressing a codon-optimized PD-1 signal peptide fused to the PD-1mb sequence with a 5’-XbaI site and 3’−8(His)-tag and P2A motif. An amplicon containing the PD-1mb fused to CD40L was generated by overlap extension PCR, cleaved with Xba-I and Sal-I and ligated to cleaved pLenti.CMV.GFP.puro.
Cells
293T and B16 cells were cultured in DMEM/10% fetal bovine serum (FBS) at 37°C in 5% CO2. B16.OVA cells were cultured with the addition of 0.4 mg/mL G418. MC38 and MC38.OVA cells were cultured in Roswell Park Memorial Institute (RPMI)/10% FBS. To produce MC38.OVA cells, MC38 cells were transduced with pLenti.cOVA.puro lentiviral vector followed by selection in 1 µg/mL puromycin. The preparation of BMDC has been previously described.34 37
Lentiviral vector preparation
Lentiviral vector stocks were prepared by calcium phosphate cotransfection of 293 T cells with lentiviral vector plasmids pMDL, pcVSV-G, and pcRev at a ratio of 28:20:7:5 as previously described.32 34 37 Virus-containing supernatant was harvested 2 days post-transfection and concentrated by ultracentrifugation for 90 min at 30 000 rpm. The virus was resuspended in 1/10 vol PBS and titered on 293 T cells by flow cytometry.
Antibodies and flow cytometry
Fc receptors on splenocytes were blocked by treating 30 min at 4°C with anti-CD16/CD32 mAb. The cells were then stained with fixable viability dye eFluor 450 (eBioscience) followed by an antibody. Antibodies used in cell surface staining were Alexa 700-anti-CD3 (BD biosciences), PerCP Cy5.5 anti-CD8a, APC-Cy7 anti-CD4 and APC H-2b OVA tetramer (NIH tetramer core), APC CD11c, PerCP Cy5.5-anti-CD11b, PE-Cy7-anti-CD19, APC-Cy7-anti-I-A/I-E (MHC II), PE-Cy7 anti-CD107a (BioLegend), and BV421. For intracellular staining, cells were fixed 10 min in 4% paraformaldehyde, permeabilized with PBS/0.1% saponin, and then stained with the antibody. Antibodies for intracellular staining were PE-anti-IFNγ, APC-Cy7-anti-TNFa, PE-anti-perforin, and AF647-anti-granzyme B antibody (BioLegend). The cells were analyzed by flow cytometry on a BD Biosciences LSR-II (Franklin Lakes, NJ), and the data were analyzed with FlowJo software.
Prophylactic and therapeutic vaccination
For prophylactic vaccination, C57BL/6 or SAMHD1 KO mice were injected intraperitoneally or intravenously with 2.5×106 I.U. lentiviral vector. Seven days postvaccination, the mice were injected with 2.5×105 B16.OVA or MC38.OVA cells in Matrigel Basement Membrane Matrix (Corning). For therapeutic vaccination, mice were injected with 2.5×105 B16.OVA or MC38.OVA cells in Matrigel Basement Membrane Matrix. One week postinjection, 3×106 I.U. lentiviral vector was injected intraperitoneally, intravenously, or intrathecally. Where indicated, the mice were injected three times, every third day, with 50 µg anti-PD-L1 antibody (BioTek Instruments). For lung metastasis, mice were injected intravenously with 2×104 cells of B16.OVA. 5 days postinjection; the mice were injected with 2.5×106 IU lentiviral vector. Twenty-five days post-tumor injection, lung foci were counted visually.
In vitro cytolysis assay
CD8 T cell cytotoxic activity was quantified in a flow-cytometry-based assay. MC38 and MC38.OVA target cells were stained with for 20 min with 5 µM CellTrace CFSE dye (Invitrogen) at 37°C. The reaction was stopped by incubation for 5 min at 37°C in RPMI/10% FBS. CFSE-labeled target cells (1×104) were placed in a 96well plate. After a 6-hour incubation, effector cells from purified TIL populations (0.5–10×104) were added. After 24 hours, the cells were stained for 30 min at 4°C with Fixable Viability Dye eFluor 450 (eBioscience), and the number of killed cells was analyzed by flow cytometry on a BD Biosciences LSR-II. The data were analyzed with FlowJo software.
Adoptive transfer
C57BL/6 mice were injected with 2.5×105 MC38.OVA cells in Matrigel Basement Membrane Matrix. One week postinjection, the mice were immunized with 3×106 I.U. of intrathecal injected lentiviral vector. Five days postimmunization, the tumors were harvested, and the TILs were separated into CD8+ and CD8− populations on CD8 Microbeads UltraPure (Miltenyi Biotec). The cells (1×106) were injected intrathecally into tumor-bearing mice, and tumor sizes were measured over 14 days.
Statistics
Experiment diagrams are generated by BioRender. Experiments were done two or three times, and representative results are shown. Statistical significance was determined by two-tailed, unpaired t test. Statistical significance was calculated with GraphPad Prism 7 7.0e. Significance was based on two-sided testing and attributed to p<0.05. CIs are shown as the mean±SD or SEM (*p≤0.05, **p≤0.01, ***p≤0.001, ****p≤0.0001).
Acknowledgments
We thank George Miller for B16, B16.OVA, and MC38 tumor cell lines and Adam Mor for pCDH.mPD-L1.
Footnotes
TT and TDN contributed equally.
Contributors: TT, TDN, and NRL designed the experiments. TT, TDN, and RL carried out the experiments. NRL directed the study. TT, TDN, and NRL wrote the manuscript. All authors have read and approved the final manuscript. NRL acts as the guarantor and takes full responsibility for the work and the conduct of the study, has access to the data, and controlled the decision to publish.
Funding: The study was funded by the NIH (grants DA046100, AI122390 and AI120898).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. The data used in this study are available upon request from the lead contact.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
Not applicable.
References
- 1. Shiravand Y, Khodadadi F, Kashani SMA, et al. Immune checkpoint inhibitors in cancer therapy. Curr Oncol 2022;29:3044–60. 10.3390/curroncol29050247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science 2018;359:1350–5. 10.1126/science.aar4060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Huang AC, Zappasodi R. A decade of checkpoint blockade immunotherapy in melanoma: understanding the molecular basis for immune sensitivity and resistance. Nat Immunol 2022;23:660–70. 10.1038/s41590-022-01141-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Topalian SL, Drake CG, Pardoll DM. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell 2015;27:450–61. 10.1016/j.ccell.2015.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Walsh RJ, Sundar R, Lim JSJ. Immune checkpoint inhibitor combinations-current and emerging strategies. Br J Cancer 2023;128:1415–7. 10.1038/s41416-023-02181-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Yarchoan M, Johnson BA, Lutz ER, et al. Targeting neoantigens to augment antitumour immunity. Nat Rev Cancer 2017;17:209–22. 10.1038/nrc.2016.154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Maleki Vareki S. High and low mutational burden tumors versus immunologically hot and cold tumors and response to immune checkpoint inhibitors. J Immunother Cancer 2018;6:157. 10.1186/s40425-018-0479-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Chaudhary B, Elkord E. Regulatory T cells in the tumor microenvironment and cancer progression: role and therapeutic targeting. Vaccines (Basel) 2016;4:28. 10.3390/vaccines4030028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Nishikawa H, Koyama S. Mechanisms of regulatory T cell infiltration in tumors: implications for innovative immune precision therapies. J Immunother Cancer 2021;9:e002591. 10.1136/jitc-2021-002591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Krishnamoorthy M, Gerhardt L, Maleki Vareki S. Immunosuppressive effects of myeloid-derived suppressor cells in cancer and immunotherapy. Cells 2021;10:1170. 10.3390/cells10051170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Glover A, Zhang Z, Shannon-Lowe C. Deciphering the roles of myeloid derived suppressor cells in viral oncogenesis. Front Immunol 2023;14:1161848. 10.3389/fimmu.2023.1161848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Mougel A, Terme M, Tanchot C. Therapeutic cancer vaccine and combinations with antiangiogenic therapies and immune checkpoint blockade. Front Immunol 2019;10:467. 10.3389/fimmu.2019.00467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zhao J, Chen Y, Ding Z-Y, et al. Safety and efficacy of therapeutic cancer vaccines alone or in combination with immune checkpoint inhibitors in cancer treatment. Front Pharmacol 2019;10:1184. 10.3389/fphar.2019.01184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Markov O, Oshchepkova A, Mironova N. Immunotherapy based on dendritic cell-targeted/-derived extracellular vesicles-a novel strategy for enhancement of the anti-tumor immune response. Front Pharmacol 2019;10:1152. 10.3389/fphar.2019.01152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Sprooten J, Ceusters J, Coosemans A, et al. Trial watch: dendritic cell vaccination for cancer immunotherapy. Oncoimmunology 2019;8:e1638212. 10.1080/2162402X.2019.1638212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Santos PM, Butterfield LH. Dendritic cell-based cancer vaccines. J Immunol 2018;200:443–9. 10.4049/jimmunol.1701024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Sabado RL, Bhardwaj N. Directing dendritic cell immunotherapy towards successful cancer treatment. Immunotherapy 2010;2:37–56. 10.2217/imt.09.43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Goyvaerts C, Breckpot K. The journey of in vivo virus engineered dendritic cells from bench to bedside: a bumpy road. Front Immunol 2018;9:2052. 10.3389/fimmu.2018.02052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jenne L, Schuler G, Steinkasserer A. Viral vectors for dendritic cell-based immunotherapy. Trends Immunol 2001;22:102–7. 10.1016/s1471-4906(00)01813-5 [DOI] [PubMed] [Google Scholar]
- 20. Moiani A, Paleari Y, Sartori D, et al. Lentiviral vector integration in the human genome induces alternative splicing and generates aberrant transcripts. J Clin Invest 2012;122:1653–66. 10.1172/JCI61852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Mitchell RS, Beitzel BF, Schroder ARW, et al. Retroviral DNA integration: ASLV, HIV, and MLV show distinct target site preferences. PLoS Biol 2004;2:e234. 10.1371/journal.pbio.0020234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Schröder ARW, Shinn P, Chen H, et al. HIV-1 integration in the human genome favors active genes and local Hotspots. Cell 2002;110:521–9. 10.1016/s0092-8674(02)00864-4 [DOI] [PubMed] [Google Scholar]
- 23. Nemirov K, Bourgine M, Anna F, et al. Lentiviral vectors as a vaccine platform against infectious diseases. Pharmaceutics 2023;15:846. 10.3390/pharmaceutics15030846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Norton TD, Miller EA, Bhardwaj N, et al. Vpx-containing dendritic cell vaccine induces CTLs and reactivates latent HIV-1 in vitro. Gene Ther 2015;22:227–36. 10.1038/gt.2014.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hofmann H, Logue EC, Bloch N, et al. The Vpx lentiviral accessory protein targets SAMHD1 for degradation in the nucleus. J Virol 2012;86:12552–60. 10.1128/JVI.01657-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Baldauf H-M, Stegmann L, Schwarz S-M, et al. Vpx overcomes a SAMHD1-independent block to HIV reverse transcription that is specific to resting Cd4 T cells. Proc Natl Acad Sci U S A 2017;114:2729–34. 10.1073/pnas.1613635114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Goldstone DC, Ennis-Adeniran V, Hedden JJ, et al. HIV-1 restriction factor SAMHD1 is a deoxynucleoside triphosphate triphosphohydrolase. Nature 2011;480:379–82. 10.1038/nature10623 [DOI] [PubMed] [Google Scholar]
- 28. Hrecka K, Hao C, Gierszewska M, et al. Vpx relieves inhibition of HIV-1 infection of Macrophages mediated by the SAMHD1 protein. Nature 2011;474:658–61. 10.1038/nature10195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Laguette N, Sobhian B, Casartelli N, et al. SAMHD1 is the dendritic- and myeloid-cell-specific HIV-1 restriction factor counteracted by Vpx. Nature 2011;474:654–7. 10.1038/nature10117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lahouassa H, Daddacha W, Hofmann H, et al. SAMHD1 restricts the replication of human immunodeficiency virus type 1 by depleting the intracellular pool of deoxynucleoside triphosphates. Nat Immunol 2012;13:223–8. 10.1038/ni.2236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Bobadilla S, Sunseri N, Landau NR. Efficient transduction of myeloid cells by an HIV-1-derived lentiviral vector that packages the Vpx accessory protein. Gene Ther 2013;20:514–20. 10.1038/gt.2012.61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Norton TD, Zhen A, Tada T, et al. Lentiviral vector-based dendritic cell vaccine suppresses HIV replication in humanized mice. Mol Ther 2019;27:960–73. 10.1016/j.ymthe.2019.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Jáuregui P, Logue EC, Schultz ML, et al. Degradation of SAMHD1 by Vpx is independent of uncoating. J Virol 2015;89:5701–13. 10.1128/JVI.03575-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Norton TD, Tada T, Leibowitz R, et al. Lentiviral-vector-based dendritic cell vaccine synergizes with checkpoint blockade to clear chronic viral infection. Mol Ther 2020;28:1795–805. 10.1016/j.ymthe.2020.05.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Tada T, Peng J-Y, Dcosta BM, et al. Single-EPITOPE T cell-based vaccine protects against SARS-CoV-2 infection in a preclinical animal model. JCI Insight 2023;8:e167306. 10.1172/jci.insight.167306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Maute RL, Gordon SR, Mayer AT, et al. Engineering high-affinity PD-1 variants for Optimized Immunotherapy and Immuno-PET imaging. Proc Natl Acad Sci USA 2015;112:E6506–14. 10.1073/pnas.1519623112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Tada T, Norton TD, Leibowitz R, et al. Directly injected lentiviral vector-based T cell vaccine protects mice against acute and chronic viral infection. JCI Insight 2022;7:e161598. 10.1172/jci.insight.161598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Dougan SK, Dougan M, Kim J, et al. Transnuclear TRP1-specific Cd8 T cells with high or low affinity TCRs show equivalent antitumor activity. Cancer Immunol Res 2013;1:99–111. 10.1158/2326-6066.CIR-13-0047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Peng Q, Qiu X, Zhang Z, et al. PD-L1 on dendritic cells attenuates T cell activation and regulates response to immune checkpoint blockade. Nat Commun 2020;11:4835. 10.1038/s41467-020-18570-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Sun J-Y, Zhang D, Wu S, et al. Resistance to PD-1/PD-L1 blockade cancer immunotherapy: mechanisms, predictive factors, and future perspectives. Biomark Res 2020;8:35. 10.1186/s40364-020-00212-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Xu-Monette ZY, Zhang M, Li J, et al. PD-1/PD-L1 blockade: have we found the key to unleash the antitumor immune response?. Front Immunol 2017;8:1597. 10.3389/fimmu.2017.01597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Rowe JH, Johanns TM, Ertelt JM, et al. PDL-1 blockade Impedes T cell expansion and protective immunity primed by attenuated listeria monocytogenes. J Immunol 2008;180:7553–7. 10.4049/jimmunol.180.11.7553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Garcia-Bates TM, Palma ML, Shen C, et al. Contrasting roles of the PD-1 signaling pathway in dendritic cell-mediated induction and regulation of HIV-1-specific Effector T cell functions. J Virol 2019;93:e02035-18. 10.1128/JVI.02035-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Dersh D, Yewdell JW, Wei J. A SIINFEKL-based system to measure MHC class I antigen presentation efficiency and kinetics. Methods Mol Biol 2019;1988:109–22. 10.1007/978-1-4939-9450-2_9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Pritchard GH, Cross EW, Strobel M, et al. Spontaneous partial loss of the OT-I transgene. Nat Immunol 2016;17:471. 10.1038/ni.3411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Rasmussen IB, Lunde E, Michaelsen TE, et al. The principle of delivery of T cell epitopes to antigen-presenting cells applied to peptides from influenza virus, ovalbumin, and hen egg lysozyme: implications for peptide vaccination. Proc Natl Acad Sci U S A 2001;98:10296–301. 10.1073/pnas.181336898 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2023-008761supp001.pdf (1.2MB, pdf)
Data Availability Statement
Data are available upon reasonable request. The data used in this study are available upon request from the lead contact.