Abstract
Colchicine, an anti-inflammatory alkaloid, has assumed an important role in the management of cardiovascular inflammation nearly 3,500 years after its first medicinal use in ancient Egypt. Primarily used in extremely high-doses for the treatment of acute gout flares during the 20th century, research in the early 21st century demonstrated that low-dose colchicine effectively treats acute gout attacks, lowers the risk of recurrent pericarditis, and can even add to secondary prevention of major adverse cardiovascular events. As the first FDA-approved targeted anti-inflammatory cardiovascular therapy, colchicine currently has a unique role in the management of atherosclerotic cardiovascular disease. The safe use of colchicine requires careful monitoring for drug-drug interactions, changes in kidney and liver function, and counseling regarding gastrointestinal upset. Future research should elucidate the mechanisms anti-inflammatory effects of colchicine relevant to atherosclerosis, the potential role of colchicine in primary prevention, in other cardiometabolic conditions, colchicine’s safety in cardiovascular patients, and opportunities for individualizing colchicine therapy using clinical and molecular diagnostics.
Keywords: colchicine, atherosclerotic cardiovascular disease, inflammation
Graphical Abstract

Introduction
Colchicine has long seen use as an anti-inflammatory treatment for gout and Familial Mediterranean Fever.1,2 Beginning in the 20th century, randomized clinical trials established low-dose colchicine as an effective treatment for pericarditis and atherosclerotic cardiovascular disease, two conditions driven by the NLRP3 inflammasome and interleukin-1.3–5 The mechanisms through which colchicine exerts its beneficial anti-inflammatory and cardiovascular effects remain an area of vigorous investigation.6 When prescribing colchicine, clinicians must consider drug-drug interactions, impaired kidney and liver function, and adverse effect monitoring.7
This Brief Review follows colchicine’s journey from an herbal medicine used to treat joint swelling and pain through its contemporary use in cardiovascular disease. We discuss colchicine’s effects on immune cells, highlight landmark clinical trials, and consider the practical aspects of colchicine use.
A Brief History of Colchicine
Hartung published an authoritative review of colchicine’s history based upon careful examination of several dozen historical documents.8 The earliest medical use of colchicine possibly dates to 1,500 BCE based on writings that describe the use of a plant likely containing colchicine for the treatment of pain and swelling (Figure 1). In the 550s CE, Alexander of Tralles, a physician in the Byzantine Empire, described the use of hermodactyl, a plant resembling the Autumn crocus, for the treatment of gout as well as the botanical’s adverse gastrointestinal effects. Baron Anton Stork provided an early description of the use of colchicine for pericarditis in the 18th century. Around the same time, Nicolas Husson developed the first commercial colchicine preparation: “Eau Medicinale”.
Figure 1. Notable historical moments related to colchicine use since 1500 BCE.


The figure displays approximate geographical locations of historical mentions of colchicine according to Hartung’s comprehensive history on the subject (Hartung EF. Arthritis Rheum. 1961;4:18–26.). Present-day names of the locations are used. All dates are approximate. Recruitment locations for landmark ASCVD clinical trials are indicated as blue circles. Map image downloaded from: https://www.vecteezy.com/free-vector/world-map-outline. ASCVD = atherosclerotic cardiovascular disease; BCE = before common era; CE = common era
Case reports and clinical trials established colchicine’s efficacy in Familial Mediterranean Fever, an inherited auto-inflammatory disorder, in the late 20th century (Figure 1).2,9–14 These studies then inspired testing of colchicine for pericarditis, which demonstrated that this agent reduced the risk of recurrent pericarditis by up to 50%.15 Colchicine remains a mainstay treatment for acute gout attacks, although the traditional strategy of administration until limited by gastrointestinal tolerance has given way to a regimen of 1.8 mg loading dose and 0.6 mg once or twice daily.1
Treatment of Cardiovascular Disease with Colchicine
Coronary Artery Disease
The NLRP3 inflammasome activates the inflammatory cytokines interleukin-1 and interleukin-18 in response to molecular danger signals.16 Both interleukin-1 and interleukin-18 contribute to the genesis and progression of coronary atherosclerosis.17 The Canakinumab Antiinflammatory Thrombosis Outcome Study (CANTOS) validated the role of inflammation and interleukin-1 in atherosclerosis by demonstrating that blocking interleukin-1β with a monoclonal antibody reduced major adverse cardiovascular events without altering blood lipid levels.18
Colchicine lowers high-sensitivity C-reactive protein (hsCRP) and may decrease coronary artery plaque volume.19,20 In an open-label, randomized clinical trial of colchicine versus no colchicine (LoDoCo), colchicine lowered the risk of the composite of acute coronary syndrome, out-of-hospital cardiac arrest, or non-cardioembolic ischemic stroke by 67% in people with stable coronary artery disease.21
More definitive evidence of the benefits of colchicine on atherosclerotic cardiovascular disease events comes from the LoDoCo2 (Low-Dose Colchicine-2) and COLCOT (Colchicine Cardiovascular Outcomes Trial) studies.4,5 LoDoCo2 and COLCOT each compared the effects of adding either low-dose colchicine (0.5 mg once daily) or placebo to background guideline-directed medical therapy, including antiplatelet and statin therapy, on major adverse cardiovascular events in approximately 5,000 participants per trial over 2-years of follow-up. COLCOT enrolled individuals within 30 days of a myocardial infarction while LoDoCo2 enrolled individuals with chronic coronary artery disease (at least 6 months following an acute coronary syndrome [ACS]). In both studies, low-dose colchicine lowered the risk of major adverse cardiovascular events by more than 30% compared to placebo (Figure 2).
Figure 2. Effects of colchicine on major adverse cardiovascular events.

The LoDoCo2 and COLCOT studies demonstrated the efficacy of colchicine in lowering the risk of major adverse cardiovascular events in people with stable coronary artery disease (LoDoCo2) and recent myocardial infarction (COLCOT). The primary endpoints were the composite of cardiovascular death, myocardial infarction, ischemic stroke, or ischemia-driven revascularization in LoDoCo2 and the composite of cardiovascular death, myocardial infarction, stroke, urgent hospitalization for angina leading to revascularization, and resuscitated cardiac arrest in COLCOT. The secondary endpoints shown are the composite of cardiovascular death, myocardial infarction and stroke for LoDoCo2 and the composite of cardiovascular death, myocardial infarction, stroke and resuscitated cardiac arrest in COLCOT (resuscitated cardiac arrest occurred in 0.2% of COLCOT participants and did not contribute meaningfully to the endpoint). CI = confidence interval; COLCOT = Colchicine Cardiovascular Outcomes Trial; HR = hazard ratio; LoDoCo2 = Low-Dose Colchicine-2
In LoDoCo2, the benefit derived from a reduction in myocardial infarction whereas in COLCOT the benefit derived from reductions in stroke and revascularization. The differential effects of colchicine on revascularization and myocardial infarction in LoDoCo2 and COLCOT mirror the effects of statins, which also primarily lower the risk of revascularization when initiated soon after an acute coronary syndrome, and the effects of colchicine in the Colchicine in Patients with Acute Coronary Syndrome (COPS) trial (discussed below), in which colchicine demonstrated more favorable effects on revascularization than on recurrent myocardial infarction.22,23 In LoDoCo2, colchicine lowered the risk of major adverse cardiovascular events in people with or without a history of ACS, although 85% of participants did have a prior ACS. Adding colchicine to statin, antiplatelet and modern revascularization therapy did not lower the risk of all-cause death or cardiovascular death in LoDoCo2 or COLCOT. Similarly, clinical trials of adding ezetimibe, a PCSK9 inhibitor, bempedoic acid, or canakinumab (anti-interleukin-1β antibody) to contemporary medical therapy lowered the risk of major adverse cardiovascular events, but not cardiovascular or all-cause death over the study timespan.18,24–27
Two clinical trials have evaluated the effects of colchicine, started urgently after an acute coronary syndrome, on major adverse cardiovascular events. Lack of sufficient power and other issues limited interpretation of these studies. COPS randomized 795 individuals with an ACS to colchicine 0.5 mg twice daily for 30 days followed by 0.5 mg once daily for 11 months or placebo.28 The study design intended an enrollment of 1,009 participants to provide 80% power to detect a 53% reduction in the primary composite outcome of all-cause death, acute coronary syndrome, stroke or urgent revascularization assuming a control event rate of 7.2%. The steering committee stopped the trial early due to slow enrollment. Numerically fewer primary composite events occurred in the colchicine arm at 365 days (24 [6%] vs. 38 [10%]; hazard ratio [95% confidence interval]: 0.65 [0.38–1.09]). Colchicine did lower the risk of a post-hoc composite outcome of cardiovascular death, acute coronary syndrome, stroke, or urgent revascularization (similar to the outcome in COLCOT and LoDoCo2) (5% vs. 10%; hazard ratio [95% confidence interval]: 0.51 [0.29–0.89]). In 2-year follow-up, the risk of the primary outcome was significantly lower in the colchicine arm (32 [8%] vs. 54 [14%]; log-rank P-value = 0.02).29 All-cause mortality was higher in the colchicine arm, a finding that we discuss below in greater detail.
The second colchicine ACS trial randomized 361 people with an acute coronary syndrome to colchicine 0.5 mg daily or placebo for 6 months and used a different primary composite outcome (all-cause death, hospital admission due to typical chest pain [acute myocardial infarction or unstable angina], non-cardioembolic stroke, urgent revascularization or decompensated heart failure) that was not adjudicated by an independent committee.30 In this trial, colchicine significantly lowered the risk of the primary composite outcome, driven primarily by a reduction in unstable angina, as only 13 myocardial infarction events occurred in both arms.
Other Cardiovascular Conditions
Colchicine may have beneficial effects on infarct size and ischemia-reperfusion injury when given before revascularization in ACS, in people with diabetes undergoing percutaneous coronary intervention with a bare-metal stent and in people undergoing coronary artery bypass grafting surgery.31–36 One clinical trial did not demonstrate decreased infarct size with colchicine in ACS using cardiac magnetic resonance imaging, but colchicine showed no effect on neutrophil count at 24-hours and only a trend towards lower hsCRP levels at 24-hours.37
Several trials have evaluated the effect of colchicine on the risk of post-pericardiotomy syndrome38–42 and atrial fibrillation43–45 after cardiac surgery and atrial fibrillation ablation. The role of colchicine after cardiac procedures remains under investigation. In people with acute cerebrovascular accident, colchicine starting 24 hours after symptom onset may lower indices of inflammation.46
Low-dose colchicine 0.5 mg daily had no effect on severely elevated albuminuria or major adverse kidney events in 160 people with type 2 diabetes and moderately elevated albuminuria.47 In a trial of 279 individuals with stable heart failure and reduced ejection fraction, colchicine 0.5 mg twice daily did not achieve the primary endpoint of a one-grade improvement in New York Heart Association functional class at 6-months, but did associate with greater reductions in left ventricular end-diastolic and end-systolic diameters.48 In the colchicine arm on-treatment hsCRP levels of 5.5 mg/L suggests the need for more potent anti-inflammatory effects in this setting.48
Mechanisms of Colchicine’s Anti-Inflammatory Effects
Colchicine binds tubulin, which interferes with microtubule-dependent cellular processes in rapidly dividing cells.49 Colchicine decreases interleukin-1β secretion by blocking the colocalization of the NLRP3 inflammasome components ASC and NLRP3 (but not the NLRC4 or AIM2 inflammasomes).50,51 These experiments suggest that colchicine can inhibit the systemic inflammatory response related to molecular patterns that can trigger NLRP3 while leaving other host defense mechanisms intact.
In people with non-ST segment-elevation ACS, colchicine pre-treatment before cardiac catheterization significantly lowered the transcoronary serum concentrations of interleukin-1β, interleukin-18 and interleukin-6 compared to no colchicine.52 Similarly, colchicine lowered the levels of interleukin-1β secretion from ex vivo stimulated monocytes derived from people with an ACS.53 In a substudy of participants in the LoDoCo2 trial, colchicine significantly lowered NLRP3 protein levels in extracellular vesicles.54
Colchicine reduced neutrophil extracellular trap production from neutrophils derived from people with an ACS.55 In people with Familial Mediterranean Fever, colchicine decreased neutrophil chemotaxis.56 Colchicine also may decrease neutrophil superoxide production.57 Pharmacoproteomic studies suggest that colchicine alters circulating levels of several immune-related proteins, including many involved in neutrophil degranulation that associate with cardiovascular risk.58–60 Somatic mutations in genes such as TET2 that accumulate with age enhance inflammasome activity and associate with a higher risk of cardiovascular disease.61 Colchicine decreased atherosclerosis in mice with Tet2-mutant clonal hematopoiesis.62
Colchicine may possess broad effects on innate and adaptive immune cells. In a study of 35 people with obesity, colchicine 0.6 mg twice daily over 3 months significantly lowered circulating levels of classical monocytes and CD8+ cytotoxic T cells.63 Colchicine also blunted the increase in natural killer cells and CD4+ effector cells observed in the placebo arm of that same study.63
One experimental study suggests that colchicine exerts its anti-inflammatory effects in leukocytes indirectly through hepatic GDF-15 secretion.6 Colchicine’s gastrointestinal effects have been attributed to tubulin-dependent mitotic arrest, induction of apoptosis, and altered intestinal permeability that resolves upon treatment discontinuation.64–66 Contrary to animal studies, colchicine appears to have no effect on the gut microbiome in people with metabolic syndrome.65,67 Targeting the downstream effects of colchicine may provide safer anti-inflammatory therapies.
Practical Considerations for the Use of Colchicine in Cardiovascular Disease
Patient Selection
Clinicians have considerable flexibility regarding the timing of colchicine initiation. Studies support colchicine initiation during hospitalization for ACS, a strategy that has worked well for the implementation of guideline-directed medical therapy for heart failure.29,30,68,69 Conversely, a LoDoCo2 post-hoc analysis found that colchicine significantly lowered the risk of major adverse cardiovascular events even in participants whose most recent ACS occurred between 2 to 7 years before randomization or more than 7 years before randomization.70
Notably, none of the landmark colchicine cardiovascular clinical trials required an elevated hsCRP level for enrollment and the US FDA approved indication does not mention hsCRP testing. Clinicians could consider hsCRP testing to maximize the benefit-risk ratio in routine practice as the JUPITER trial demonstrated that primary prevention statin therapy among people with elevated high-sensitivity C-reactive protein (hsCRP) lowers the risk of major adverse cardiovascular events.71 Since COLCOT and LoDoCo2 enrolled participants already on background statin therapy, colchicine should be an adjunct to LDL cholesterol lowering. Altogether, the evidence from multiple clinical trials indicates that all individuals with coronary artery disease should be considered a potential candidate for colchicine therapy (Figure 3).
Figure 3. Proposed algorithm for use of low-dose colchicine in secondary atherosclerosis prevention.
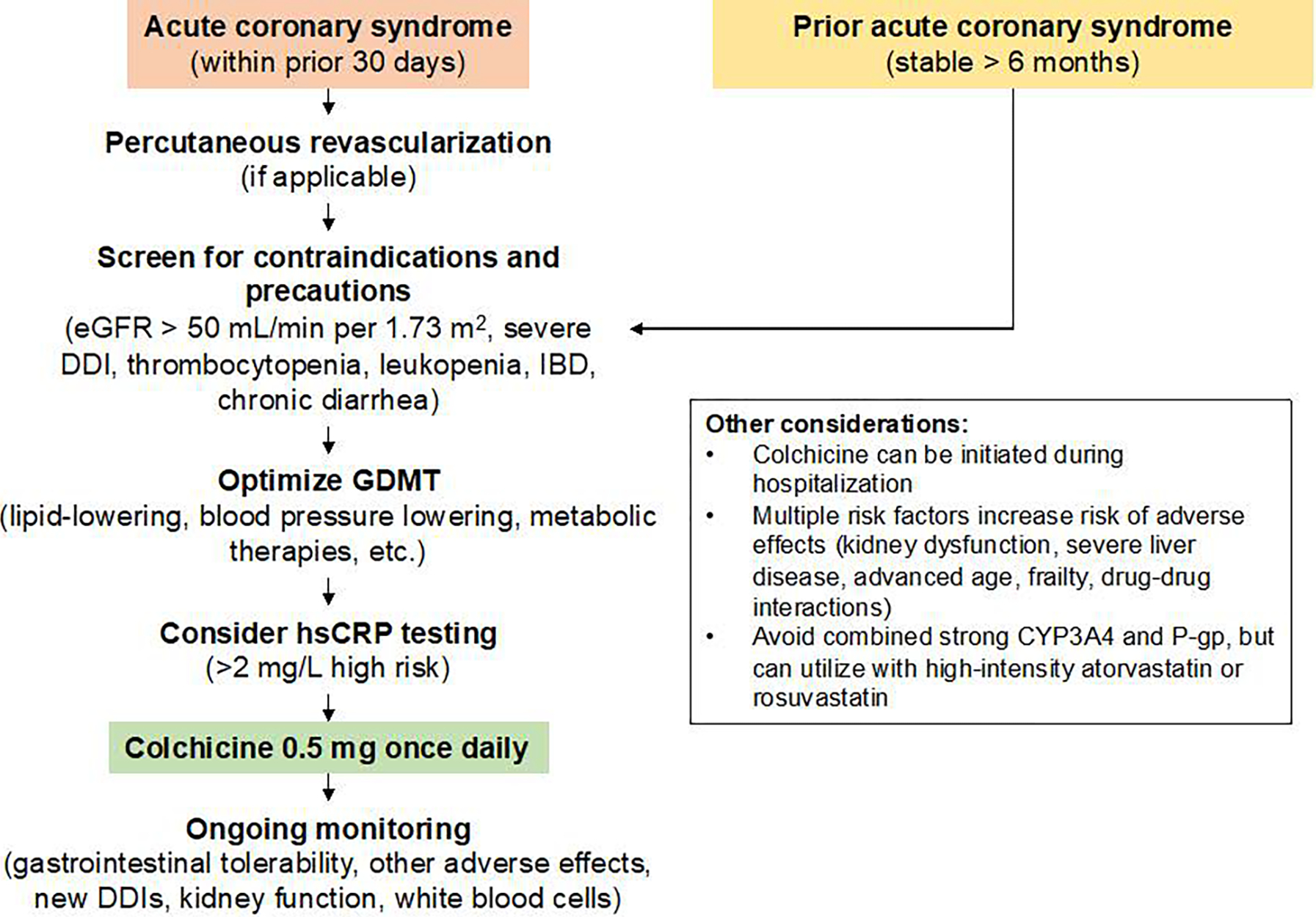
This figure summarizes an approach for the use of low-dose colchicine in secondary atherosclerosis prevention. Low-dose colchicine can be initiated in people with an acute coronary syndrome during hospitalization or within the next 30 days as well as in people with stable coronary artery disease whose acute coronary syndrome occurred more than 6 months ago. Individuals should be screened for potential contraindications and precautions prior to colchicine initiation, and have optimal guideline-directed medical therapy. Measuring high-sensitivity C-reactive protein (hsCRP) can provide evidence of residual inflammatory risk, although the landmark colchicine clinical trials did not require a specific hsCRP level for inclusion. CYP = cytochrome P450; DDI = drug-drug interaction; eGFR = estimated glomerular filtration rate; hsCRP = high-sensitivity C-reactive protein; IBD = inflammatory bowel disease; P-gp = P-glycoprotein
Adverse Effect Profile
A systematic review and meta-analysis of all colchicine clinical trials regardless of indication published prior to 2020 (n studies=35; participants = 4,225 on colchicine [2,366 from COLCOT] and 3,956 on placebo [2,379 from COLCOT]) found a 1.74-times higher risk of any gastrointestinal event with colchicine (18% vs. 13%).72 Lower doses of colchicine appear as effective in resolving the gout flare and better tolerated1 and colchicine-related diarrhea is self-limiting and resolves within 3 days in many cases.45
The aforementioned landmark clinical trials also provide the largest randomized safety data for low-dose colchicine. In LoDoCo2, 437 participants (7%) dropped out during the open-label run-in phase due to gastrointestinal upset and in COLCOT nausea and flatulence occurred more frequently with colchicine than placebo in COLCOT. Yet diarrhea was not more common with colchicine than placebo in COLCOT, serious gastrointestinal events were not different between colchicine and placebo in either COLCOT or LoDoCo2 and, most important, treatment discontinuation did not differ between colchicine and placebo in either COLCOT or LoDoCo2. Nor did more serious and rarer colchicine adverse effects, such as leukopenia, thrombocytopenia, or neuropathy, differ between colchicine and placebo in COLCOT or LoDoCo2. These data indicate that low-dose colchicine possesses a tolerable gastrointestinal profile in most people with cardiovascular disease.
Two unexpected signals for a higher risk of mortality emerged from the LoDoCo2 trial (non-cardiovascular mortality hazard ratio [95% confidence interval]: 1.51 [0.99–2.31]) and the 1-year outcomes of the COPS trial (all-cause death occurred in 8 colchicine participants compared to 1 placebo participant).5,28 In COPS, 3 of the 5 non-cardiovascular deaths in the 1-year report occurred off-treatment and one occurred due to cancer (not a known risk of colchicine therapy) (Table S1). The open-label 2-year follow-up of COPS, which included 13 deaths, showed no difference in the risk of all-cause or non-cardiovascular death between colchicine and placebo.29 Furthermore, the risks of cardiovascular death or all-cause death were not significantly different between colchicine and placebo in COLCOT (all-cause death hazard ratio [95% confidence interval]: 0.98 [0.64–1.49]).4 A systematic review and meta-analysis of colchicine use in people with cirrhosis found no significant difference in the risk of all-cause mortality between colchicine and control.73 A non-randomized study of people with gout reported a lower risk of all-cause mortality with colchicine use.74
Pneumonia occurred more frequently in colchicine-treated participants in COLCOT (0.9% vs. 0.4%; P=0.03). Conversely, neither a systematic review and meta-analysis nor LoDoCo2 found a difference in infection outcomes between colchicine and placebo.72 These signals warrant further investigation.
Drug-Drug Interactions
Colchicine is metabolized by enteral and hepatic cytochrome P450 3A4 (CYP3A4) and P-glycoprotein, a xenobiotic extruder found in the intestine, biliary tract, kidney, and mononuclear immune cells (but not neutrophils). The management of a drug-drug interaction requires consideration of the relationship between drug exposure and clinical outcomes, the duration of concomitant use, the availability of monitoring parameters, and the clinical indication for the interacting drugs (Figure 4).
Figure 4. Proposed management of statin and non-statin drug-drug interactions with colchicine.

Colchicine interacts with many commonly used cardiovascular therapies, including statins. Colchicine can be used concomitantly with rosuvastatin, pitavastatin, or pravastatin without enhanced monitoring or dose adjustments. Concomitant use of colchicine with other statins requires enhanced monitoring for muscle-related adverse effects. The management of other drug-drug interactions with colchicine should consider the severity and duration of the interaction, the consequences of withholding colchicine therapy, other risk factors for colchicine adverse effects and the availability of alternative therapies or enhanced monitoring.
COLCOT and LoDoCo2 indicated the general safety of combining statins with colchicine. Yet, case reports have described serious colchicine toxicity in people taking a statin with colchicine due to competitive inhibition of CYP3A4 and P-gp. Myalgia and myotoxic effects occurred more commonly among colchicine-treated participants (21% vs. 19%; P < 0.05) in LoDoCo2 participants enrolled in The Netherlands (muscle-related symptoms were not assessed in COLCOT or LoDoCo2 participants in Australia). In 1,775 LoDoCo2 participants with available end-of-study blood samples, colchicine was associated with higher creatine kinase levels (123 vs. 110 U/L for colchicine vs. placebo; P < 0.01), but the mean levels remains within the normal range, creatine kinase levels greater than 5-times the upper-limit-of-normal were rare (0.7% vs. 0.2% for colchicine vs. placebo) and there were no instances of levels greater than 10-times the upper-limit-of-normal. Many candidates for colchicine therapy will require concomitant treatment with a high-intensity statin. Rosuvastatin (neither a substrate nor inhibitor of CYP3A4 and P-gp) can be used with colchicine without enhanced monitoring or dose adjustment.7,75 Atorvastatin, a weak inhibitor of CYP3A4 and a P-gp substrate, can be used as an alternative to rosuvastatin. Atorvastatin 40 mg daily (a weak CYP3A4 and P-gp inhibitor) increased the total exposure of a single colchicine 0.6 mg dose (as measured by area-under-the-concentration-time curve) by 24% (90% confidence interval: 110%–141%) in healthy volunteers, slightly above the US FDA threshold for clinical significance.76,77 Non-fibrate therapy for triglyceride lowering should be preferred for concomitant use with colchicine.78
Other cardiovascular medications that interact with colchicine include amiodarone, dronedarone, quinidine, and propafenone.7,75 Concomitant use of these anti-arrhythmic agents with colchicine should involve collaborative monitoring with an electrophysiologist. Concomitant use of diltiazem and verapamil with colchicine should be avoided when possible in favor of other rate control agents like beta-blockers and more effective blood pressure-lowering agents.
Other Considerations
LoDoCo2 excluded people with eGFR less than 50 mL/min per 1.73 m2, although the US FDA-approved indication for low-dose colchicine (which was a modification of the existing approval for colchicine in gout rather than a new indication review) allows for use with eGFR as low as 30 mL/min per 1.73 m2.79 Advanced age, in the absence of reduced kidney function, does not significantly augment colchicine exposure.80 Due to its metabolism by hepatic CYP3A4, colchicine should be avoided in people with Child-Pugh Class C liver disease.
In the United States, colchicine has been available as 0.6 mg doses, whereas the rest of the world uses 0.5 mg. Since the 0.5 mg doses proved effective in large-scale cardiovascular clinical trials, the 0.5 mg dose should receive preference when available and affordable. If patients cannot access the 0.5 mg dose, judicious use of 0.6 mg furnishes an acceptable alternative to forgoing treatment with the only available anti-inflammatory therapy for atherosclerotic cardiovascular disease. Clinical observations of people with colchicine toxicity have led to estimations that the therapeutic plasma concentration lies between 0.5 and 3 ng/mL.7,81,82 Pharmacokinetic modeling studies suggest that both the 0.5 mg and 0.6 mg doses provide therapeutic plasma concentrations, but the 0.5 mg dose should be used in people with kidney disease and those with concomitant drug-drug interactions.83 Important limitations to the use of plasma colchicine concentrations for therapeutic drug monitoring include the observation that colchicine concentrations within leukocytes exceed those of plasma by several orders of magnitude and become saturated at plasma levels well below the proposed therapeutic range (Figure 5).84,85 Additionally, people with Familial Mediterranean Fever seem to tolerate daily colchicine doses up to 3 mg daily and plasma concentrations well above 3 ng/mL (Figure S1).86
Figure 5. Non-linear relationship between colchicine plasma concentrations and circulating leukocyte concentrations.

Colchicine concentrations in plasma (total bound and unbound colchicine) and in granulocytes and mononuclear cells was measured in people with Familial Mediterranean Fever who were taking colchicine 0.5 to 2.0 mg daily. Annotations indicate the approximate plasma concentration at which leukocyte concentrations plateau, estimated maximal concentrations (Cmax) observed in people according to kidney function and the estimated safe upper limit plasma concentration. Granulocyte and mononuclear cell concentrations were extrapolated visually across the higher end of the plasma concentration range. Figure was based on data from Br J Clin Pharmacol. 1994;38(1):87–9. and Am J Med. 2022;135(1):32–38. Cmax = maximal concentration
Conclusions
Colchicine exerts pleiotropic anti-inflammatory effects on assembly of the NLRP3 inflammasome and various neutrophil functions. Based on multiple, large randomized controlled trials, individuals with coronary artery disease and preserved kidney function may benefit from low-dose colchicine therapy. When prescribing colchicine, clinicians should be cognizant of gastrointestinal adverse effects, drug-drug interactions and changes in kidney and liver function. Colchicine provides an example of the long history of natural product to cardiovascular drug development (digoxin, reserpine, gliflozins). While the use of colchicine in cardiovascular disease requires much further study, landmark trials have pointed the way for its clinical adoption as an approved targeted anti-inflammatory agent in the management of atherosclerotic risk.
Supplementary Material
Table 1.
Design and participant characteristics of the COLCOT and LoDoCo2 studies CAC = coronary artery calcium; eGFR = estimated glomerular filtration rate
| COLCOT | LoDoCo2 | |||
|---|---|---|---|---|
| Design features | ||||
| Design | Randomized, double-blind, placebo-controlled, event-driven clinical trial | Randomized, double-blind, placebo-controlled, event-driven clinical trial (1 month open-label run-in period) | ||
| Key inclusion criteria | • Myocardial infarction within the prior 30 days • Use of guideline-directed medical therapy |
• Coronary artery disease by invasive or non-invasive angiography or CAC score > 400 Agatston units • Clinically stable for 6 months |
||
| Key exclusion criteria | • Serum creatinine greater than 2-times the upper limit of normal • Inflammatory bowel disease • Progressive neuromuscular disease • Leukopenia or thrombocytopenia • Symptomatic heart failure |
• Serum creatinine greater than 1.7 mg/dL (150 umol/L) or eGFR less than 50 mL/min per 1.73 m2 • Symptomatic heart failure • Peripheral neuritis, myositis or myosensitivity to statins |
||
| Colchicine regimen | 0.5 mg once daily | 0.5 mg once daily | ||
| Run-in period | NA | 6,528 entered run-in period • 611 stopped due to side effects (9%) • 395 stopped for other reasons (6%) • 5,522 randomized |
||
| Participant characteristics | ||||
| Colchicine | Placebo | Colchicine | Placebo | |
| Number of participants | 2,366 | 2,379 | 2,762 | 2,760 |
| Age, years | 61 | 61 | 66 | 66 |
| Women, % | 20% | 18% | 17% | 14% |
| White, % | 73% | 72% | NR | NR |
| Current smoking, % | 30% | 30% | 12% | 12% |
| Hypertension, % | 50% | 52% | 51% | 50% |
| Diabetes | 20% | 21% | 23% | 24% |
| Mean time to randomization from index MI, days | 13 | 13 | NA | NA |
| Most recent ACS within 24 months | NA | NA | 27% | 26% |
| Statin | 99% | 99% | 94% | 94% |
| Antiplatelet agent | 98% | 98% | 90% | 90% |
| Median follow-up duration, months | 23 | 29 | ||
| Study treatment discontinuation, % | 18% | 19% | 11% | 11% |
Highlights.
Randomized clinical trials established low-dose colchicine as an effective treatment for pericarditis and atherosclerotic cardiovascular disease
The mechanisms through which colchicine exerts its beneficial anti-inflammatory and cardiovascular effects remain an area of vigorous investigation
When prescribing colchicine, clinicians must consider drug-drug interactions, impaired kidney and liver function, and adverse effect monitoring
While the use of colchicine in cardiovascular disease requires much further study, landmark trials have pointed the way for its clinical adoption as an approved targeted anti-inflammatory agent in the management of atherosclerotic risk
Sources of Funding:
LFB is supported by NIH/NHLBI K23HL150311, an ASN/KidneyCure Carl W. Gottschalk Research Scholar Grant and the BWH Khoury Innovation Fund. Dr. Libby receives funding support from the National Heart, Lung, and Blood Institute (1R01HL134892 and 1R01HL163099-01), the RRM Charitable Fund and the Simard Fund.
Footnotes
Disclosures: LFB has received consulting fees from Kiniksa Pharmaceuticals and speakers fees from Boehringer Ingelheim Pharmaceuticals, Inc. and Eli Lilly and Company through ASHP Advantage. Dr. Libby is an unpaid consultant to, or involved in clinical trials for Amgen, AstraZeneca, Baim Institute, Beren Therapeutics, Esperion Therapeutics, Genentech, Kancera, Kowa Pharmaceuticals, Medimmune, Merck, Moderna, Novo Nordisk, Novartis, Pfizer, and Sanofi-Regeneron. Dr. Libby is a member of the scientific advisory board for Amgen, Caristo Diagnostics, Cartesian Therapeutics, CSL Behring, DalCor Pharmaceuticals, Dewpoint Therapeutics, Eulicid Bioimaging, Kancera, Kowa Pharmaceuticals, Olatec Therapeutics, Medimmune, Novartis, PlaqueTec, TenSixteen Bio, Soley Thereapeutics, and XBiotech, Inc. Dr. Libby’s laboratory has received research funding in the last 2 years from Novartis, Novo Nordisk and Genentech. Dr. Libby is on the Board of Directors of XBiotech, Inc. Dr. Libby has a financial interest in Xbiotech, a company developing therapeutic human antibodies, in TenSixteen Bio, a company targeting somatic mosaicism and clonal hematopoiesis of indeterminate potential (CHIP) to discover and develop novel therapeutics to treat age-related diseases, and in Soley Therapeutics, a biotechnology company that is combining artificial intelligence with molecular and cellular response detection for discovering and developing new drugs, currently focusing on cancer therapeutics. Dr. Libby’s interests were reviewed and are managed by Brigham and Women’s Hospital and Mass General Brigham in accordance with their conflict-of-interest policies.
References
- 1.Terkeltaub RA, Furst DE, Bennett K, Kook KA, Crockett RS, Davis MW. High versus low dosing of oral colchicine for early acute gout flare: Twenty-four-hour outcome of the first multicenter, randomized, double-blind, placebo-controlled, parallel-group, dose-comparison colchicine study. Arthritis Rheum. 2010;62:1060–1068. [DOI] [PubMed] [Google Scholar]
- 2.Dinarello CA, Wolff SM, Goldfinger SE, Dale DC, Alling DW. Colchicine therapy for familial mediterranean fever. A double-blind trial. N Engl J Med. 1974;291:934–937. [DOI] [PubMed] [Google Scholar]
- 3.Chiabrando JG, Bonaventura A, Vecchié A, Wohlford GF, Mauro AG, Jordan JH, Grizzard JD, Montecucco F, Berrocal DH, Brucato A, Imazio M, Abbate A. Management of Acute and Recurrent Pericarditis: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:76–92. [DOI] [PubMed] [Google Scholar]
- 4.Tardif J-C, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP, Pinto FJ, Ibrahim R, Gamra H, Kiwan GS, Berry C, López-Sendón J, Ostadal P, Koenig W, Angoulvant D, Grégoire JC, Lavoie M-A, Dubé M-P, Rhainds D, Provencher M, Blondeau L, Orfanos A, L’Allier PL, Guertin M-C, Roubille F. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N Engl J Med. 2019;381:2497–2505. [DOI] [PubMed] [Google Scholar]
- 5.Nidorf SM, Fiolet ATL, Mosterd A, Eikelboom JW, Schut A, Opstal TSJ, The SHK, Xu X-F, Ireland MA, Lenderink T, Latchem D, Hoogslag P, Jerzewski A, Nierop P, Whelan A, Hendriks R, Swart H, Schaap J, Kuijper AFM, van Hessen MWJ, Saklani P, Tan I, Thompson AG, Morton A, Judkins C, Bax WA, Dirksen M, Alings M, Hankey GJ, Budgeon CA, Tijssen JGP, Cornel JH, Thompson PL. Colchicine in Patients with Chronic Coronary Disease. N Engl J Med. 2020; [DOI] [PubMed] [Google Scholar]
- 6.Weng JH, Koch PD, Luan HH, Tu HC, Shimada K, Ngan I, Ventura R, Jiang R, Mitchison TJ. Colchicine acts selectively in the liver to induce hepatokines that inhibit myeloid cell activation. Nat Metab. 2021;3:513–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Robinson PC, Terkeltaub R, Pillinger MH, Shah B, Karalis V, Karatza E, Liew D, Imazio M, Cornel JH, Thompson PL, Nidorf M. Consensus Statement Regarding the Efficacy and Safety of Long-Term Low-Dose Colchicine in Gout and Cardiovascular Disease. Am J Med. 2022;135:32–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hartung EF. History of the use of colchicum and related medicaments in gout; with suggestions for further research. Ann Rheum Dis. 1954;13:190–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mamou H [Amylogenic periodic disease]. Sem Hopitaux Organe Fonde Par Assoc Enseign Med Hopitaux Paris. 1955;31:388–391. [PubMed] [Google Scholar]
- 10.Sohar E, Gafni J, Pras M, Heller H. Familial Mediterranean fever. A survey of 470 cases and review of the literature. Am J Med. 1967;43:227–253. [DOI] [PubMed] [Google Scholar]
- 11.Schwabe AD, Peters RS. Familial Mediterranean Fever in Armenians. Analysis of 100 cases. Medicine (Baltimore). 1974;53:453–462. [DOI] [PubMed] [Google Scholar]
- 12.Goldstein RC, Schwabe AD. Prophylactic colchicine therapy in familial Mediterranean fever. A controlled, double-blind study. Ann Intern Med. 1974;81:792–794. [DOI] [PubMed] [Google Scholar]
- 13.Zemer D, Revach M, Pras M, Modan B, Schor S, Sohar E, Gafni J. A controlled trial of colchicine in preventing attacks of familial mediterranean fever. N Engl J Med. 1974;291:932–934. [DOI] [PubMed] [Google Scholar]
- 14.Wright DG, Wolff SM, Fauci AS, Alling DW. Efficacy of intermittent colchicine therapy in familial Mediterranean fever. Ann Intern Med. 1977;86:162–165. [DOI] [PubMed] [Google Scholar]
- 15.Imazio M, Gaita F, LeWinter M. Evaluation and treatment of pericarditis: A systematic review. JAMA - J Am Med Assoc. 2015;314:1498–1506. [DOI] [PubMed] [Google Scholar]
- 16.Chan AH, Schroder K. Inflammasome signaling and regulation of interleukin-1 family cytokines. J Exp Med. 2019;217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Toldo S, Abbate A. The role of the NLRP3 inflammasome and pyroptosis in cardiovascular diseases. Nat Rev Cardiol. 2023; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ, Krum H, Varigos J, Siostrzonek P, Sinnaeve P, Gotcheva N, Yong H, Urina-Triana M, Milicic D, Vettus R, Manolis AJ, Wyss F, Sigurdsson A, Fucili A, Veze I, Petrauskiene B, Salvador L, Klemsdal TO, Medina F, Budaj A, Otasevic P, Lainscak M, Seung KB, Commerford P, Donath M, Hwang JJ, Kultursay H, Bilazarian S, East C, Forgosh L, Harris B, Ligueros M, Bohula E, Charmarthi B, Cheng S, Chou S, Danik J, McMahon G, Maron B, Ning M, Olenchock B, Pande R, Perlstein T, Pradhan A, Rost N, Singhal A, Taqueti V, Wei N, Burris H, Cioffi A, Dalseg AM, Ghosh N, Gralow J, Mayer T, Rugo H, Fowler V, Limaye AP, Cosgrove S, Levine D, Lopes R, Scott J, Hilkert R, Tamesby G, Mickel C, Manning B, Woelcke J, Tan M, Manfreda S, Ponce T, Kam J, Saini R, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377:1119–1131. [DOI] [PubMed] [Google Scholar]
- 19.Nidorf M, Thompson PL. Effect of Colchicine (0.5 mg Twice Daily) on High-Sensitivity C-Reactive Protein Independent of Aspirin and Atorvastatin in Patients With Stable Coronary Artery Disease. Am J Cardiol. 2007;99:805–807. [DOI] [PubMed] [Google Scholar]
- 20.Vaidya K, Arnott C, Martínez GJ, Ng B, McCormack S, Sullivan DR, Celermajer DS, Patel S. Colchicine Therapy and Plaque Stabilization in Patients With Acute Coronary Syndrome. JACC Cardiovasc Imaging. 2018;11:305–316. [DOI] [PubMed] [Google Scholar]
- 21.Nidorf SM, Eikelboom JW, Budgeon CA, Thompson PL. Low-Dose Colchicine for Secondary Prevention of Cardiovascular Disease. J Am Coll Cardiol. 2013;61:404–410. [DOI] [PubMed] [Google Scholar]
- 22.Schwartz GG, Olsson AG, Ezekowitz MD, Ganz P, Oliver MF, Waters D, Zeiher A, Chaitman BR, Leslie S, Stern T, for the Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering (MIRACL) Study Investigators. Effects of Atorvastatin on Early Recurrent Ischemic Events in Acute Coronary SyndromesThe MIRACL Study: A Randomized Controlled Trial. JAMA. 2001;285:1711–1718. [DOI] [PubMed] [Google Scholar]
- 23.Cannon CP, Braunwald E, McCabe CH, Rader DJ, Rouleau JL, Belder R, Joyal SV, Hill KA, Pfeffer MA, Skene AM. Intensive versus Moderate Lipid Lowering with Statins after Acute Coronary Syndromes. N Engl J Med. 2004;350:1495–1504. [DOI] [PubMed] [Google Scholar]
- 24.Cannon CP, Blazing MA, Giugliano RP, McCagg A, White JA, Theroux P, Darius H, Lewis BS, Ophuis TO, Jukema JW, De Ferrari GM, Ruzyllo W, De Lucca P, Im K, Bohula EA, Reist C, Wiviott SD, Tershakovec AM, Musliner TA, Braunwald E, Califf RM, IMPROVE-IT Investigators. Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes. N Engl J Med. 2015;372:2387–2397. [DOI] [PubMed] [Google Scholar]
- 25.Sabatine MS, Giugliano RP, Keech AC, Honarpour N, Wiviott SD, Murphy SA, Kuder JF, Wang H, Liu T, Wasserman SM, Sever PS, Pedersen TR, Fourier Steering Committee, Investigators. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med. 2017;376:1713–1722. [DOI] [PubMed] [Google Scholar]
- 26.Schwartz GG, Steg PG, Szarek M, Bhatt DL, Bittner VA, Diaz R, Edelberg JM, Goodman SG, Hanotin C, Harrington RA, Jukema JW, Lecorps G, Mahaffey KW, Moryusef A, Pordy R, Quintero K, Roe MT, Sasiela WJ, Tamby J-F, Tricoci P, White HD, Zeiher AM, Odyssey Outcomes Committees, Investigators. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N Engl J Med. 2018;379:2097–2107. [DOI] [PubMed] [Google Scholar]
- 27.Nissen SE, Lincoff AM, Brennan D, Ray KK, Mason D, Kastelein JJP, Thompson PD, Libby P, Cho L, Plutzky J, Bays HE, Moriarty PM, Menon V, Grobbee DE, Louie MJ, Chen C-F, Li N, Bloedon L, Robinson P, Horner M, Sasiela WJ, McCluskey J, Davey D, Fajardo-Campos P, Petrovic P, Fedacko J, Zmuda W, Lukyanov Y, Nicholls SJ, CLEAR Outcomes Investigators. Bempedoic Acid and Cardiovascular Outcomes in Statin-Intolerant Patients. N Engl J Med. 2023;388:1353–1364. [DOI] [PubMed] [Google Scholar]
- 28.Tong David C., Quinn Stephen, Nasis Arthur, Hiew Chin, Philip Roberts-Thomson Heath Adams, Sriamareswaran Rumes, Htun Nay M., Wilson William, Stub Dion, William van Gaal Laurie Howes, Collins Nicholas, Yong Andy, Bhindi Ravinay, Whitbourn Robert, Lee Astin, Hengel Chris, Asrress Kaleab, Freeman Melanie, Amerena John, Wilson Andrew, Layland Jamie. Colchicine in Patients With Acute Coronary Syndrome. Circulation. 2020;142:1890–1900. [DOI] [PubMed] [Google Scholar]
- 29.Tong DC, Bloom JE, Quinn S, Nasis A, Hiew C, Roberts-Thomson P, Adams H, Sriamareswaran R, Htun NM, Wilson W, Stub D, van Gaal W, Howes L, Yeap A, Yip B, Wu S, Perera P, Collins N, Yong A, Bhindi R, Whitbourn R, Lee A, Premaratne M, Asrress K, Freeman M, Amerena J, Layland J. Colchicine in Patients With Acute Coronary Syndrome: Two-Year Follow-Up of the Australian COPS Randomized Clinical Trial. Circulation. 2021;144:1584–1586. [DOI] [PubMed] [Google Scholar]
- 30.Akrami M, Izadpanah P, Bazrafshan M, Hatamipour U, Nouraein N, Drissi HB, Manafi A. Effects of colchicine on major adverse cardiac events in next 6-month period after acute coronary syndrome occurrence; a randomized placebo-control trial. BMC Cardiovasc Disord. 2021;21:583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Deftereos S, Giannopoulos G, Angelidis C, Alexopoulos N, Filippatos G, Papoutsidakis N, Sianos G, Goudevenos J, Alexopoulos D, Pyrgakis V, Cleman MW, Manolis AS, Tousoulis D, Lekakis J. Anti-Inflammatory Treatment With Colchicine in Acute Myocardial Infarction. Circulation. 2015;132:1395–1403. [DOI] [PubMed] [Google Scholar]
- 32.Cole J, Htun N, Lew R, Freilich M, Quinn S, Layland J. Colchicine to Prevent Periprocedural Myocardial Injury in Percutaneous Coronary Intervention: The COPE-PCI Pilot Trial. Circ Cardiovasc Interv. 2021;14:e009992. [DOI] [PubMed] [Google Scholar]
- 33.Deftereos S, Giannopoulos G, Raisakis K, Kossyvakis C, Kaoukis A, Panagopoulou V, Driva M, Hahalis G, Pyrgakis V, Alexopoulos D, Manolis AS, Stefanadis C, Cleman MW. Colchicine Treatment for the Prevention of Bare-Metal Stent Restenosis in Diabetic Patients. J Am Coll Cardiol. 2013;61:1679–1685. [DOI] [PubMed] [Google Scholar]
- 34.Giannopoulos G, Angelidis C, Kouritas VK, Dedeilias P, Filippatos G, Cleman MW, Panagopoulou V, Siasos G, Tousoulis D, Lekakis J, Deftereos S. Usefulness of colchicine to reduce perioperative myocardial damage in patients who underwent on-pump coronary artery bypass grafting. Am J Cardiol. 2015;115:1376–1381. [DOI] [PubMed] [Google Scholar]
- 35.Pan T, Jiang C-Y, Zhang H, Han X-K, Zhang H-T, Jiang X-Y, Chen W, Wang K, Fan F-D, Pan J, Zhou Q, Wang C-S, Zhang L, Wang D-J. The low-dose colchicine in patients after non-CABG cardiac surgery: a randomized controlled trial. Crit Care Lond Engl. 2023;27:49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cole J, Htun N, Lew R, Freilich M, Quinn S, Layland J. COlchicine to Prevent PeriprocEdural Myocardial Injury in Percutaneous Coronary Intervention (COPE-PCI): Coronary Microvascular Physiology Pilot Substudy. J Intervent Cardiol. 2022;2022:1098429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mewton N, Croisille P, Gahide G, Rioufol G, Bonnefoy E, Sanchez I, Cung TT, Sportouch C, Angoulvant D, Finet G, André-Fouët X, Derumeaux G, Piot C, Vernhet H, Revel D, Ovize M. Effect of Cyclosporine on Left Ventricular Remodeling After Reperfused Myocardial Infarction. J Am Coll Cardiol. 2010;55:1200–1205. [DOI] [PubMed] [Google Scholar]
- 38.Imazio M, Trinchero R, Brucato A, Rovere ME, Gandino A, Cemin R, Ferrua S, Maestroni S, Zingarelli E, Barosi A, Simon C, Sansone F, Patrini D, Vitali E, Ferrazzi P, Spodick DH, Adler Y, COPPS Investigators. COlchicine for the Prevention of the Post-pericardiotomy Syndrome (COPPS): a multicentre, randomized, double-blind, placebo-controlled trial. Eur Heart J. 2010;31:2749–2754. [DOI] [PubMed] [Google Scholar]
- 39.Imazio M, Brucato A, Ferrazzi P, Pullara A, Adler Y, Barosi A, Caforio AL, Cemin R, Chirillo F, Comoglio C, Cugola D, Cumetti D, Dyrda O, Ferrua S, Finkelstein Y, Flocco R, Gandino A, Hoit B, Innocente F, Maestroni S, Musumeci F, Oh J, Pergolini A, Polizzi V, Ristić A, Simon C, Spodick DH, Tarzia V, Trimboli S, Valenti A, Belli R, Gaita F. Colchicine for prevention of postpericardiotomy syndrome and postoperative atrial fibrillation: The COPPS-2 randomized clinical trial. JAMA - J Am Med Assoc. 2014;312:1016–1023. [DOI] [PubMed] [Google Scholar]
- 40.Meurin P, Lelay-Kubas S, Pierre B, Pereira H, Pavy B, Iliou MC, Bussière JL, Weber H, Beugin JP, Farrokhi T, Bellemain-Appaix A, Briota L, Tabet JY, French Society of Cardiology. Colchicine for postoperative pericardial effusion: a multicentre, double-blind, randomised controlled trial. Heart Br Card Soc. 2015;101:1711–1716. [DOI] [PubMed] [Google Scholar]
- 41.Izadi Amoli A, Bozorgi A, HajHossein Talasaz A, Salehi Omran A, Mortazavi SH, Jalali A, Nasirpour S, Jenab Y. Efficacy of colchicine versus placebo for the treatment of pericardial effusion after open-heart surgery: A randomized, placebo-controlled trial. Am Heart J. 2015;170:1195–1201. [DOI] [PubMed] [Google Scholar]
- 42.Ahmed AS, Miller J, Foreman J, Golden K, Shah A, Field J, Gilge J, Clark B, Joshi S, Nair G, Olson J, Padanilam BJ, Prystowsky E, Patel PJ. Prophylactic Colchicine After Radiofrequency Ablation of Atrial Fibrillation: The PAPERS Study. JACC Clin Electrophysiol. 2023;9:1060–1066. [DOI] [PubMed] [Google Scholar]
- 43.Tabbalat RA, Hamad NM, Alhaddad IA, Hammoudeh A, Akasheh BF, Khader Y. Effect of ColchiciNe on the InciDence of Atrial Fibrillation in Open Heart Surgery Patients: END-AF Trial. Am Heart J. 2016;178:102–107. [DOI] [PubMed] [Google Scholar]
- 44.Tabbalat RA, Alhaddad I, Hammoudeh A, Khader YS, Khalaf HA, Obaidat M, Barakat J. Effect of Low-dose ColchiciNe on the InciDence of Atrial Fibrillation in Open Heart Surgery Patients: END-AF Low Dose Trial. J Int Med Res. 2020;48:300060520939832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Conen D, Ke Wang M, Popova E, Chan MTV, Landoni G, Cata JP, Reimer C, McLean SR, Srinathan SK, Reyes JCT, Grande AM, Tallada AG, Sessler DI, Fleischmann E, Kabon B, Voltolini L, Cruz P, Maziak DE, Gutiérrez-Soriano L, McIntyre WF, Tandon V, Martínez-Téllez E, Guerra-Londono JJ, DuMerton D, Wong RHL, McGuire AL, Kidane B, Roux DP, Shargall Y, Wells JR, Ofori SN, Vincent J, Xu L, Li Z, Eikelboom JW, Jolly SS, Healey JS, Devereaux PJ, COP-AF Investigators. Effect of colchicine on perioperative atrial fibrillation and myocardial injury after non-cardiac surgery in patients undergoing major thoracic surgery (COP-AF): an international randomised trial. Lancet Lond Engl. 2023;S0140-6736(23)01689-6. [DOI] [PubMed] [Google Scholar]
- 46.Yuan B, Meng X, Wang A, Niu S, Xie X, Jing J, Li H, Chang L, Wang Y, Li J. Effect of different doses of colchicine on high sensitivity C-reactive protein in patients with acute minor stroke or transient ischemic attack: A pilot randomized controlled trial. Eur J Pharm Sci. 2022;178:106288. [DOI] [PubMed] [Google Scholar]
- 47.Wang Y, Peng X, Hu J, Luo T, Wang Z, Cheng Q, Mei M, He W, Peng C, Ma L, Gong L, Yang S, Li Q. Low-dose colchicine in type 2 diabetes with microalbuminuria: A double-blind randomized clinical trial. J Diabetes. 2021;13:827–836. [DOI] [PubMed] [Google Scholar]
- 48.Deftereos S, Giannopoulos G, Panagopoulou V, Bouras G, Raisakis K, Kossyvakis C, Karageorgiou S, Papadimitriou C, Vastaki M, Kaoukis A, Angelidis C, Pagoni S, Pyrgakis V, Alexopoulos D, Manolis AS, Stefanadis C, Cleman MW. Anti-Inflammatory Treatment With Colchicine in Stable Chronic Heart Failure. JACC Heart Fail. 2014;2:131–137. [DOI] [PubMed] [Google Scholar]
- 49.Weisenberg RC, Borisy GG, Taylor EW. The colchicine-binding protein of mammalian brain and its relation to microtubules. Biochemistry. 1968;7:4466–4479. [DOI] [PubMed] [Google Scholar]
- 50.Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237–241. [DOI] [PubMed] [Google Scholar]
- 51.Misawa T, Takahama M, Kozaki T, Lee H, Zou J, Saitoh T, Akira S. Microtubule-driven spatial arrangement of mitochondria promotes activation of the NLRP3 inflammasome. Nat Immunol. 2013;14:454–460. [DOI] [PubMed] [Google Scholar]
- 52.Martínez GJ, Robertson S, Barraclough J, Xia Q, Mallat Z, Bursill C, Celermajer DS, Patel S. Colchicine Acutely Suppresses Local Cardiac Production of Inflammatory Cytokines in Patients With an Acute Coronary Syndrome. J Am Heart Assoc. 2015;4:e002128–e002128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Robertson S, Martinez GJ, Payet CA, Barraclough JY, Celermajer DS, Bursill C, Patel S. Colchicine therapy in acute coronary syndrome patients acts on caspase-1 to suppress NLRP3 inflammasome monocyte activation. Clin Sci. 2016;130:1237–1246. [DOI] [PubMed] [Google Scholar]
- 54.Silvis MJM, Fiolet ATL, Opstal TSJ, Dekker M, Suquilanda D, Zivkovic M, Duyvendak M, The SHK, Timmers L, Bax WA, Mosterd A, Cornel JH, de Kleijn DPV. Colchicine reduces extracellular vesicle NLRP3 inflammasome protein levels in chronic coronary disease: A LoDoCo2 biomarker substudy. Atherosclerosis. 2021;334:93–100. [DOI] [PubMed] [Google Scholar]
- 55.Vaidya K, Tucker B, Kurup R, Khandkar C, Pandzic E, Barraclough J, Machet J, Misra A, Kavurma M, Martinez G, Rye KA, Cochran BJ, Patel S. Colchicine Inhibits Neutrophil Extracellular Trap Formation in Patients With Acute Coronary Syndrome After Percutaneous Coronary Intervention. J Am Heart Assoc. 2021;10:e018993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dinarello CA, Chusid MJ, Fauci AS, Gallin JI, Dale DC, Wolff SM. Effect of prophylactic colchicine therapy on leukocyte function in patients with familial mediterranean fever. Arthritis Rheum. 1976;19:618–622. [DOI] [PubMed] [Google Scholar]
- 57.Chia EW, Grainger R, Harper JL. Colchicine suppresses neutrophil superoxide production in a murine model of gouty arthritis: A rationale for use of low-dose colchicine. Br J Pharmacol. 2008;153:1288–1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Opstal TSJ, Hoogeveen RM, Fiolet ATL, Silvis MJM, The SHK, Bax WA, de Kleijn DPV, Mosterd A, Stroes ESG, Cornel JH. Colchicine Attenuates Inflammation Beyond the Inflammasome in Chronic Coronary Artery Disease: A LoDoCo2 Proteomic Substudy. Circulation. 2020;142:1996–1998. [DOI] [PubMed] [Google Scholar]
- 59.Demidowich AP, Levine JA, Apps R, Cheung FK, Chen J, Fantoni G, Patel TP, Yanovski JA. Colchicine’s effects on metabolic and inflammatory molecules in adults with obesity and metabolic syndrome: results from a pilot randomized controlled trial. Int J Obes. 2020;44:1793–1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Buckley Leo F., Dorbala Pranav, Claggett Brian L., Libby Peter, Tang Weihong, Coresh Josef, Ballantyne Christie M., Hoogeveen Ron C., Yu Bing, Shah Amil M. Circulating Neutrophil-Related Proteins Associate with Incident Heart Failure and Cardiac Dysfunction: The ARIC Study. Eur J Heart Fail. 2023;Accepted for publication. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Libby P, Sidlow R, Lin AE, Gupta D, Jones LW, Moslehi J, Zeiher A, Jaiswal S, Schulz C, Blankstein R, Bolton KL, Steensma D, Levine RL, Ebert BL. Clonal Hematopoiesis. J Am Coll Cardiol. 2019;74:567–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zuriaga MA, Yu Z, Matesanz N, Truong B, Asensio-Lopez MC, Uddin MM, Nakao T, Niroula A, Zorita V, Moro R, Ebert BL, Honigberg M, Pacual-Figal D, Natarajan P, Fuster JJ. Colchicine prevents accelerated atherosclerosis development in TET2-mutant clonal hematopoiesis. Eur Heart J. 2023;44:ehad655.3264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Patel TP, Levine JA, Elizondo DM, Arner BE, Jain A, Saxena A, Lopez-Ocasio M, Dagur PK, Famuyiwa O, Gupta S, Sarrafan-Chaharsoughi Z, Biancotto A, McCoy JP, Demidowich AP, Yanovski JA. Immunomodulatory effects of colchicine on peripheral blood mononuclear cell subpopulations in human obesity: Data from a randomized controlled trial. Obes Silver Spring Md. 2023;31:466–478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Iacobuzio-Donahue CA, Lee EL, Abraham SC, Yardley JH, Wu TT. Colchicine toxicity: distinct morphologic findings in gastrointestinal biopsies. Am J Surg Pathol. 2001;25:1067–1073. [DOI] [PubMed] [Google Scholar]
- 65.Shi Y, Li J, Yang P, Niu Z, Wei L, Chen L, Gao L. Colchicine increases intestinal permeability, suppresses inflammatory responses, and alters gut microbiota in mice. Toxicol Lett. 2020;334:66–77. [DOI] [PubMed] [Google Scholar]
- 66.Liu L, Tessier S, Duarte-Chavez R, Marino D, Kaza A, Longo S, Nanda S. Gastrointestinal histopathology of acute colchicine toxicity after lower dose treatment of pericarditis: A case report. Int J Crit Illn Inj Sci. 2022;12:174–176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kisimba CM, Donahue JL, Chivukula KK, Subramanian P, Mistry SD, Wolska A, Remaley AT, Yanovski JA, Demidowich AP. Colchicine effects on the gut microbiome in adults with metabolic syndrome. Biosci Microbiota Food Health. 2023;42:236–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bouabdallaoui N, Tardif J-C, Waters DD, Pinto FJ, Maggioni AP, Diaz R, Berry C, Koenig W, Lopez-Sendon J, Gamra H, Kiwan GS, Blondeau L, Orfanos A, Ibrahim R, Grégoire JC, Dubé M-P, Samuel M, Morel O, Lim P, Bertrand OF, Kouz S, Guertin M-C, L’Allier PL, Roubille F. Time-to-treatment initiation of colchicine and cardiovascular outcomes after myocardial infarction in the Colchicine Cardiovascular Outcomes Trial (COLCOT). Eur Heart J. 2020;41:4092–4099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bhatt AS, Varshney AS, Moscone A, Claggett BL, Miao ZM, Chatur S, Lopes MS, Ostrominski JW, Pabon MA, Unlu O, Wang X, Bernier TD, Buckley LF, Cook B, Eaton R, Fiene J, Kanaan D, Kelly J, Knowles DM, Lupi K, Matta LS, Pimentel LY, Rhoten MN, Malloy R, Ting C, Chhor R, Guerin JR, Schissel SL, Hoa B, Lio CH, Milewski K, Espinosa ME, Liu Z, McHatton R, Cunningham JW, Jering KS, Bertot JH, Kaur G, Ahmad A, Akash M, Davoudi F, Hinrichsen MZ, Rabin DL, Gordan PL, Roberts DJ, Urma D, McElrath EE, Hinchey ED, Choudhry NK, Nekoui M, Solomon SD, Adler DS, Vaduganathan M. Virtual Care Team Guided Management of Patients With Heart Failure During Hospitalization. J Am Coll Cardiol. 2023;81:1680–1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Opstal TSJ, Fiolet ATL, van Broekhoven A, Mosterd A, Eikelboom JW, Nidorf SM, Thompson PL, Duyvendak M, van Eck JWM, van Beek EA, den Hartog F, Budgeon CA, Bax WA, Tijssen JGP, El Messaoudi S, Cornel JH, LoDoCo2 Trial Investigators. Colchicine in Patients With Chronic Coronary Disease in Relation to Prior Acute Coronary Syndrome. J Am Coll Cardiol. 2021;78:859–866. [DOI] [PubMed] [Google Scholar]
- 71.Ridker PM, Danielson E, Fonseca FAH, Genest J, Gotto AM, Kastelein JJP, Koenig W, Libby P, Lorenzatti AJ, MacFadyen JG, Nordestgaard BG, Shepherd J, Willerson JT, Glynn RJ, JUPITER Study Group. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359:2195–2207. [DOI] [PubMed] [Google Scholar]
- 72.Stewart S, Yang KCK, Atkins K, Dalbeth N, Robinson PC. Adverse events during oral colchicine use: a systematic review and meta-analysis of randomised controlled trials. Arthritis Res Ther. 2020;22:28–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rambaldi A, Gluud C. Colchicine for alcoholic and non-alcoholic liver fibrosis and cirrhosis. Cochrane Database Syst Rev. 2005;2005:CD002148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Solomon DH, Liu C-C, Kuo IH, Zak A, Kim SC. Effects of colchicine on risk of cardiovascular events and mortality among patients with gout: a cohort study using electronic medical records linked with Medicare claims. Ann Rheum Dis. 2016;75:1674–1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wiggins BS, Saseen JJ, Page RL, Reed BN, Sneed K, Kostis JB, Lanfear D, Virani S, Morris PB. Recommendations for Management of Clinically Significant Drug-Drug Interactions With Statins and Select Agents Used in Patients With Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation. 2016;134. [DOI] [PubMed] [Google Scholar]
- 76.Davis MW, Wason S. Effect of steady-state atorvastatin on the pharmacokinetics of a single dose of colchicine in healthy adults under fasted conditions. Clin Drug Investig. 2014;34:259–67. [DOI] [PubMed] [Google Scholar]
- 77.Research C for DE and. Clinical Drug Interaction Studies — Cytochrome P450 Enzyme- and Transporter-Mediated Drug Interactions Guidance for Industry [Internet]. 2021. [cited 2023 Nov 13];Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/clinical-drug-interaction-studies-cytochrome-p450-enzyme-and-transporter-mediated-drug-interactions
- 78.Hansten PD, Tan MS, Horn JR, Gomez-Lumbreras A, Villa-Zapata L, Boyce RD, Subbian V, Romero A, Gephart S, Malone DC. Colchicine Drug Interaction Errors and Misunderstandings: Recommendations for Improved Evidence-Based Management. Drug Saf. 2023;46:223–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wason S, Mount D, Faulkner R. Single-Dose, Open-Label Study of the Differences in Pharmacokinetics of Colchicine in Subjects with Renal Impairment, Including End-Stage Renal Disease. Clin Drug Investig. 2014;34:845–855. [DOI] [PubMed] [Google Scholar]
- 80.Wason S, Faulkner RD, Davis MW. Are dosing adjustments required for colchicine in the elderly compared with younger patients? Adv Ther. 2012;29:551–561. [DOI] [PubMed] [Google Scholar]
- 81.Molad Y Update on colchicine and its mechanism of action. Curr Rheumatol Rep. 2002;4:252–256. [DOI] [PubMed] [Google Scholar]
- 82.Niel E, Scherrmann J-M. Colchicine today. Joint Bone Spine. 2006;73:672–678. [DOI] [PubMed] [Google Scholar]
- 83.Karatza E, Ismailos G, Karalis V. Colchicine for the treatment of COVID-19 patients: efficacy, safety, and model informed dosage regimens. Xenobiotica Fate Foreign Compd Biol Syst. 2021;51:643–656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chappey ON, Niel E, Wautier J-LL, Hung PP, Dervichian M, Cattan D, Scherrmann JM. Colchicine disposition in human leukocytes after single and multiple oral administration. Clin Pharmacol Ther. 1993;54:360–367. [DOI] [PubMed] [Google Scholar]
- 85.Chappey O, Niel E, Dervichian M, Wautier JL, Scherrmann JM, Cattan D. Colchicine concentration in leukocytes of patients with familial Mediterranean fever. Br J Clin Pharmacol. 1994;38:87–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Katz EZ, Ehrenfeld M, Levy M, Eliakim M. Plasma colchicine concentration in patients with recurrent polyserositis (familial Mediterranean fever) on long-term prophylaxis. Arthritis Rheum. 1982;25:227–231. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


