Abstract
Erectile dysfunction (ED) is a common clinical condition that mainly affects men aged over 40 years. Various causes contribute to the progression of ED, including pelvic nerve injury, diabetes, metabolic syndrome, age, Peyronie’s disease, smoking, and psychological disorders. Current treatments for ED are limited to symptom relief and do not address the root cause. Stem cells, with their powerful ability to proliferate and differentiate, are a promising approach for the treatment of male ED and are gradually gaining widespread attention. Current uses for treating ED have been studied primarily in experimental animals, with most studies observing improvements in erectile quality as well as improvements in erectile tissue. However, research on stem cell therapy for human ED is still limited. This article summarizes the recent literature on basic stem cell research on ED, including cavernous nerve injury, aging, diabetes, and sclerosing penile disease, and describes mechanisms of action and therapeutic effects of various stem cell therapies in experimental animals. Stem cells are also believed to interact with host tissue in a paracrine manner, and improved function can be supported through both implantation and paracrine factors. To date, stem cells have shown some preliminary promising results in animal and human models of ED.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40259-024-00650-9.
Key Points
| Erectile dysfunction (ED) is the persistent inability of the penis to achieve and/or maintain an erection sufficient for satisfactory sexual intercourse, seriously jeopardizing the patient’s quality of life and sense of well-being. |
| Stem cell therapy is a promising new area of treatment for ED compared with traditional treatments. |
| Animal model studies and clinical trials revealed the potential use of various sources of stem cells in ED treatment. |
Introduction
The high incidence and rising prevalence of erectile dysfunction (ED) worldwide is an important sign of a condition that seriously affects men’s physical and mental health, and adversely affects the quality of life of couples [1]. Research has found that ED is closely associated with various factors such as age, smoking, diabetes, hypertension, dyslipidemia, obesity, and psychological states [2]. Studies have also shown that accidental injury to penile cavernous nerve tissue due to surgery, chemical side effects, traumatic injury to genital tissue structures, underlying physiological dysfunction of the endocrine system, and fibrosis of penile vascular smooth muscle tissue are important clinical causes inducing the development of ED in men [3].
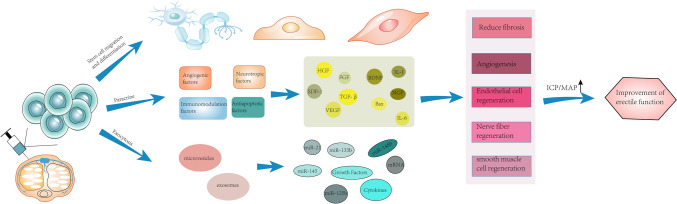
Treatment of ED has had mixed results and currently focuses on symptom relief rather than a permanent cure. Guidelines from the American Urological Association suggest that consultation with a mental health professional may be an adjunctive treatment and has been shown to improve adherence to treatment plans and enhance the effectiveness of other treatments in patients with ED [4]. Various therapeutic strategies exist to improve erectile function, such as oral medications, urethral suppositories, intracorporeal injections, vacuum erection devices, penile implants, and stem cell therapy. In recent years, as theoretical research on regenerative medicine has continued to advance, stem cells are considered to be a promising approach in the treatment of male erectile dysfunction, gradually attracting widespread attention from researchers in related fields. Stem cells play an important role in the treatment of different causes of ED not only by maximizing the replacement of damaged or apoptotic penile tissue cells, but also by secreting factors that repair damaged penile tissue (Fig. 1). Phosphodiesterase type 5 (PDE5) inhibitors are drugs that inhibit phosphodiesterase activity. They increase the intracellular concentration of cyclic guanosine monophosphate (cGMP) by inhibiting the activity of cGMP-degrading PDE5, which leads to relaxation of the smooth muscle and increased blood flow to the intracavernous arteries of the penis, resulting in an erection. Most international clinical guidelines, for example, the European Association of Urology Guidelines, consider PDE5 inhibitors to have good efficacy and safety as first-line treatment for ED [5]. In addition, PDE5 inhibitors provide only temporary relief of symptoms and rarely address the underlying etiology of ED in the patient [6, 7]. Invasive treatments, including vacuum constriction devices, intraurethral and extracorporeal injections, and surgical implantation of penile prostheses, have been shown to be effective for many men. However, these invasive treatments lack long-term efficacy and remain generally unsatisfactory.
Fig. 1.
Stem cells injected into the corpus cavernosum may act in three ways: 1. Stem cells migrate directly to the site of injury and differentiate into neuronal cells, endothelial cells, and smooth muscle cells; 2. stem cells may produce paracrine angiogenic factors, neurostimulatory factors, immunomodulatory factors, and anti-apoptotic factors; 3. stem cells may play a role in cytotoxicity by releasing microvesicles and exosomes.
In view of the above-noted research background, this article reviews recent advances in basic research and clinical applications of stem cells for the treatment of ED of different etiologies, highlighting the importance of stem cell research in the field of male science and suggesting potential therapeutic options suitable for future research.
Stem Cells in the Treatment of ED Resulting from Cavernous Nerve Injury
ED is a common complication after radical prostatectomy and major pelvic surgery. The development of nerve-preserving surgical techniques and the use of robot-assisted laparoscopic techniques still cannot completely prevent postoperative ED [8]. Statistically, the prevalence of ED after radical prostatectomy can be as high as 90% [9]. In clinical practice, oral PDE5 inhibitors are the first-line treatment for patients with ED caused by cavernous nerve injury (CNI). However, the treatment outcomes are poor compared with those in the general ED population [10]. Several studies have now demonstrated the ability of stem cell therapy to improve nerve injury ED and reduce patients’ dependence on PDE5 inhibitors [11], with therapeutic effects varying by stem cell source (Supplementary Table 1).
First, adipose-derived stem cells (ADSCs) are abundant and readily available in adipose tissue; second, ADSCs are derived from autologous cells, which are more helpful in reducing complications. ADSCs are more readily induced and stably expressed by genetic factors in the initial state. Intracavernosal injection of ADSCs was performed in rats with CNI, confirming that ADSCs prevented neuronal cell apoptosis [12]. The recovery of erectile function and the regeneration of cavernous nerves are neuromodulated by the in vitro induction of ADSC differentiated nerve cells injected into the cavernous nerve injury site [13]. In vitro pretreatment of ADSCs accelerates the recovery of cavernous structure and function in vivo after transplantation [14]. Other researchers transplanted muscle-derived stem cells (MDSCs) into the cavernous body of rats to improve erectile function by repairing and replacing cavernous nerves [15, 16]. They also induced pluripotent stem cells to improve erectile dysfunction caused by cavernous nerve injury through paracrine secretion [17]. Acquisition of bone-marrow-derived stem cells and adipose-derived stem cells requires invasive procedures and carries the risk of infection, yet placental tissue is rich in stem cells and is often discarded as medical waste. Studies have found that embryonic stem cells may replace existing damaged nerve cells or stimulate host factors to stimulate nerve growth to promote regeneration. Gu et al. [18] reported improved erectile function after applying embryonic neural stem cell therapy in a pelvic neurovascular injury rat model. The apoptosis of embryonic stem cells was monitored in real time by labeling the cells and observing the recovery of endothelial cells, smooth muscle cells and nerve fibers. That study had the longest reported follow-up, but the optimal dose for stem cell application and the distribution of trophic factors have not been studied. Ethical issues remain a limiting factor for the clinical application of human embryonic stem cell therapy. Little is known about the therapeutic effects of human gingiva-derived mesenchymal stem cells (hGMSCs) in CNI rats. Wu et al. [19] injected hGMSCs around the bilateral major pelvic ganglia (MPG) in a CNI rat model and evaluated their efficacy. The results showed that hGMSCs deflected macrophage polarity from M1 to M2 antiinflammatory phenotype on the one hand, and on the other hand enhanced smooth muscle and vascular endothelial content and restored neuronal nitric oxide synthase (nNOS) expression, among other functions. This study confirmed that hGMSCs significantly improved CNI-related ED, which provides new clues for preclinical applications.
As research continues, scholars have suggested that in a rat model of cavernous nerve injury, the mechanism of mesenchymal stem cells (MSCs) action may be due to soluble factors released during paracrine secretion and activation of receptor regeneration in tissue cells. Even if stem cells do not differentiate directly into replacement cells in damaged tissues, paracrine effects promote tissue repair by producing anti-apoptotic factors and angiogenic factors. Thus, MSCs do not need to be transplanted into host tissues to produce structural and functional effects [20]. Exosomes are microvesicles derived from various stem cells and are rich in DNA, mRNA, microRNA, and proteins that can be transported to recipient cells and are involved in the regulation of pathophysiological processes in many diseases. Given these benefits, exosomes are now widely used in experimental animal research. Li et al. [16] applied exosomes of ADSCs and bone marrow mesenchymal stem cells (BMSCs) to a rat model of ED with bilateral cavernous nerve injury, aiming to explore the potential therapeutic effects. Research has shown that both ADSC-Exo and BMSC-Exo attenuate pathological changes such as endothelial cells, smooth muscle cells, and collagen in bilateral CNI-related ED rats, thereby improving ED. In the future, ADSC-Exo and BMSC-Exo may be a potential alternative to PDE5 inhibitors for the treatment of postoperative radical prostatectomy.
Later, stem cell therapy was also gradually combined with other therapies to improve ED, combining nanotechnology with stem cells to more effectively retain ADSCs in the corpus cavernosum. This approach offers better results in improving erectile function than conventional stem cell therapy, while reducing the dose of stem cell injections reduces the risk of tumor formation [21, 22]. Cheng et al. [23] further combined genetic recombination, nanotechnology, and stem cell therapy, and transfected the Neuregulin-1 gene into rat-derived ADSCs with superparamagnetic iron oxide nanoparticles to further enhance the therapeutic effect of ADSCs on ED. This approach provides a new strategy for the treatment of EDs by ADSCs. Shao et al. [24] designed an erythropoietin (EPO) multifunctional hydrogel equipped with ADSCs applied in a rat ED model. This method effectively enhanced the proliferation and paracrine activity of ADSC therapy, ensuring the aggregation of ADSC at the injury site, inhibiting apoptosis in a timely and effective manner, and promoting the repair of denervated penile tissue and nerves. Liang et al. [25] established polydopamine nanoparticles that incorporated poly (ethylene glycol)-poly(ε-caprolactone-co-lactide) (PDNPs-PELA) temperature-sensitive hydrogel to prepare ADSC exosomes for precise injection in the white membranes of ED rats under the guidance of photoacoustic imaging. That process effectively inhibited apoptosis of cavernous sinus smooth muscle cells, promoted recovery of vascular endothelial cells, and accelerated the recovery of erectile function to overcome some of the limitations of stem cell therapy, such as nutritional deficiencies and oxygen supply to transplanted stem cells. Jung et al. [26] used a stem cell/oxygen-releasing hollow microparticles (HPs) hybrid system to prolong BMSCs survival and further improve cyclic guanosine monophosphate levels and NOS levels, thus improving erectile function in a rat model of bilateral cavernous nerve injury.
Stem Cells in the ED of Diabetes and Metabolic Syndrome
A high prevalence of ED has been reported in patients with diabetes mellitus (DM), reaching about 67.4% [27]. As a result, DM is considered a major risk factor for the development of sexual dysfunction in men, which is three times the risk of healthy men developing ED. The main effects of DM on erectile function are endothelial cell dysfunction and smooth muscle structure and dysfunction, nitrogenergic neuropathy, and endocrine abnormalities [28]. Other factors such as psychological factors and diabetes-related infection may also influence the development of ED in men with diabetes [29]. Insulin-resistance-induced vascular and erectile tissue damage is a hallmark of ED in DM [34]. The regenerative ability of stem cells may allow them to repair cavernous nerves, endothelial cells, and smooth muscle cells that have been damaged by DM. Currently, therapy with stem cells has been considered a promising approach for the treatment of diabetic ED (DMED), and most related studies have observed improvements in erectile tissue as well as erection quality in experimental animals (Supplementary Table 2).
However, the application of stem cell therapy alone has a short maintenance time in vivo, and Wang et al. [30] improved this short maintenance time by combining probucol and BMSCs. In recent years, non-coding RNAs (lncRNAs) have gradually become a hot spot for ED research. Long-stranded lncRNAs are reported to be involved in the biological functions of BMSCs. Researchers [31, 32] have identified the lncRNA maternally expressed gene 3 (MEG3), a bone marrow-associated lncRNA that inhibits the proliferation and migration of endothelial cells. MEG3 was found to be involved in endothelial cell differentiation of BMSCs through the regulation of Recombinant Forkhead Box Protein M1/vascular endothelial growth factor (FOXM1/VEGF). Metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) contains a binding site for miR-206 that can target the 3′-UTR of VEGFA. The interaction between miR-206 and MALAT1 or VEGFA was explored subsequently, finding that MALAT1 promotes the differentiation of BMSCs into endothelial cells by regulating the miR-206/VEGFA axis [31]. These results have provided new insights into the role of BMSCs in restoring endothelial cell function and improving DMED.
ADSCs have attracted much attention as promising tools for the treatment of DMED and are very similar to BMSCs in terms of differentiation and therapeutic potential, since they are able to express and secrete a wide range of growth factors and cytokines. The role of ADSCs in the treatment of DMED appears to be an indirect mechanism, that is, ADSCs improve the extracellular environment and local tissue function in the treated area through paracrine effects, rather than through direct conversion of ADSCs to local cell types [33]. Stem cells in DM mitigate tissue damage and promote tissue repair not only by paracrine secretion of many trophic factors, cytokines, and signaling molecules, but also by the secretion of a large number of exosomes [34]. Chen et al. [35] first reported that exosomes improve erectile function in rats with DM-related ED after isolating exosomes from the supernatant of cultured ADSCs and observing their efficacy in improving DM-related ED in rats. Thus, exosomes may possibly be used as a new therapeutic strategy and as an alternative to ADSCs, thus avoiding the adverse consequences of cell therapy. Although extracellular vesicles have generated widespread interest in the field of medicine and biology, their low yield is the main factor hindering the use of extracellular vesicle treatment in clinical applications. Some studies have combined gene therapy with stem cell therapy to improve ED treatment outcomes. Liu et al. [36] found that erectile dysfunction was closely associated with impaired endothelial function and disruption of the vascular endothelial growth factor (VEGF) signaling pathway. VEGF-expressing ADSCs were used to enhance endothelial function and increase smooth muscle content to promote the recovery of erectile function. ADSCs modified by the VEGF gene showed a greater therapeutic effect than unmodified ADSCs in improving erectile function in DMED rats. However, considering the risk of integrating exogenous genes into the host genome, transgenic technology remains limited in clinical applications. Endothelial progenitor cells (EPCs) play an important role in the regeneration of injured blood vessels in vivo and have the ability to generate mature endothelial cells in vitro and in vivo [37]. Yang et al. [38] found that paracrine stromal cell-derived factor-1 (SDF-1) from ADSCs enhanced the value-added differentiation of EPCs, and enhanced the expression of cavernous smooth muscle and nNOS. SDF-1 also had a synergistic effect on the repair of endothelial function by combined transplantation of ADSCs and EPCs, which had a significantly better therapeutic effect than treatment with ADSCs or EPCs alone, providing new therapeutic opportunities for patients with type 2 diabetes. Some scholars have recently suggested that diabetes impairs the regenerative capacity of MSCs, and by comparing the erectile function of diabetic rats with that of non-diabetic rats, they found that diabetes is not a limiting factor for autologous ADSCs [39]. Luo et al. [40] transfected short hairpin nucleotide-binding oligomerization domain (NOD)-, leucine-rich repeat (LRR)-, and pyrin domain-containing protein 3 (shNLRP3) lentivirus into ADSCs to detect the levels of apoptosis and reactive oxygen species (ROS) in each group under high glucose conditions and thus evaluate the effect of ADSCs on erectile function. That study found that shNLRP3 lentivirus (LV-shNLRP3) enhanced ADSCs to improve DMED by protecting the paracrine function of ADSCs in hyperglycemic state through anti-apoptotic and anti-ROS deposition mechanisms, reducing cavernous endothelial dysfunction and smooth muscle cell injury. They also found that knockdown or overexpression of certain genes in stem cells such as NRIP1 and HIF 1α increased the response of stem cells in a hyperglycemic hypoxic environment.
Most studies have focused on bone marrow or adipose tissue-derived MSCs for use in diabetic ED [38, 41, 42]. However, some scholars have argued that urine-derived stem cells (USCs) have the advantages of being obtained noninvasively and at low cost, characteristics that suggest strong potential in treating endothelial function in diabetes-related ED [17, 43]. Ouyang et al. [40] demonstrated that USCs or basic fibroblast growth factor (bFGF) gene-modified USCs improved the expression of functional endothelial proteins and repaired intracorporeal structures, which in turn improved erectile function in DMED models. It is known that the regenerative effect of stem cells can be achieved through their paracrine action and differentiation. USCs can improve DMED by producing many pro-angiogenic growth factors through paracrine action and have endothelial differentiation potential. Galhom et al. [44] showed that in a rat model of DMED, the use of USCs to produce cytoactive molecules via paracrine action repaired the cavernous structure and improve copulatory function in rats. Comparing the effects of USCs or their lysates (usc-l) on improving cavernous function in rats with DMED, the USCs lysates avoided the problems of poor differentiation and low viability of USCs after transplantation, and appeared to be a more valuable new treatment for DMED [44]. Zhang et al. [45] suggested that the repair of cavernous endothelial dysfunction by USCs acts by regulating autophagic activity. Impaired autophagy was found to be associated with cavernous endothelial dysfunction and ED in rats with diabetes-related ED. USCs improve cavernous endothelial dysfunction by enhancing the autophagic activity of cavernous endothelial cells. Stem-cell-based preclinical studies highlight the improvement in erectile function in animals with diabetes-related ED after stem cell treatment and explain the underlying mechanisms (Fig 2).
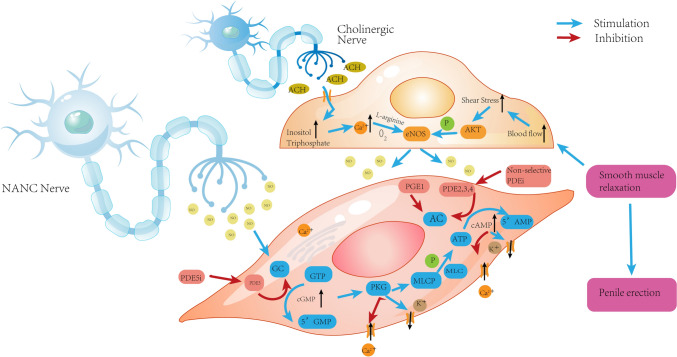
Fig. 2.
The microscopic mechanism of penis erection. GC guanylate cyclase, PKG protein kinase G, MLC myosin light chain, AC adenylate cyclase, PGE1 prostagtandin E1, NANC nonadrenergic noncholinergic, GTP guanosine triphosphate, cGMP cyclic guanosine monophosphate, NO nitric oxide, ATP adenosine triphosphate, AMP adenosine monophosphate, Ach acetylcholine, eNOS endothelial nitric oxide synthase, AKT protein kinase B, PDE5i phosphodiesterase 5 inhibitors, PDE5 phosphodiesterase 5, MLCP myosin light chain phosphatase
Human umbilical cord mesenchymal stem cells (hUC-MSCs) are shown to be a safe and effective treatment for DM by reducing diabetic complications through antiinflammatory, antifibrotic, pro-angiogenic, and immunomodulatory modalities on clinical treatment [46]. Compared with other stem cells, hUC-MSCs are more capable of self-renewal, easier to obtain, and have less immune rejection [47]. Wang et al. [48] evaluated the value of hUC-MSCs in the treatment of DMED. Reducing cavernous fibrosis by inhibiting Toll-like receptor 4 activation and increasing VEGF and endothelial nitric oxide synthase (eNOS) expression improved erectile function in diabetic rats. Mukti et al. [49] showed for the first time that hUC-MSCs improved the expression of transforming growth factor-beta (TGF-β), α-smooth muscle actin (α-SMA), and collagen in diabetic rats with ED and had a good protective effect on inflammatory response and structural improvement in rats with DMED. Feng et al. [50] aimed to verify whether iron sagging was associated with the progression of DMED. After labeling hUC-MSCs, only a very small number were found to be colonized in the corpus cavernosum, along with expansion of smooth muscle in the penis, a decrease in collagen, and a significant decrease in oxidative stress levels in the penis as well as tissue iron content. In addition, hUC-MSCs restored the normal morphology of mitochondria within the high glucose-altered corpus cavernosum smooth muscle cells (CCSMCs), demonstrating that hUC-MSCs restore erectile function by attenuating diabetes-induced iron prolapse in CCSMCs. These results suggest a new idea for hUC-MSCs as a therapeutic strategy for DMED.
In addition to the above, several studies have explored the combination of stem cells with conventional therapies. Shan et al. [51] combined BMSCs and low-energy shock-wave therapy (LESWT) to treat a DMED rat model. By increasing SDF-1, LESWT was able to promote BMSC recruitment and induce vascular growth in the diabetic cavernosa, establish a microenvironment beneficial to stem cell survival, improve the survival rate of BMSCs, and more effectively improve erectile function in diabetic rats. Liu et al. [52] found that low-intensity pulsed ultrasound (LIPUS) enhanced the proliferation and secretion of ADSCs. Those researchers combined LIPUS with ADSCs to improve erectile function in DMED rats and assessed the effects of LIPUS on the proliferation and secretion of ADSCs in vitro, and also predicted the mechanisms involved regarding LIPUS-mediated ADSCs. LIPUS was observed to promote the proliferation of ADSCs and enhance the secretion of cytokines such as SDF-1, fibroblast growth factor 2 (FGF2), and VEGF. LIPUS also enhanced the secretion of ADSC-VEGF by stimulating the Piezo-extracellular regulated protein kinases (ERK) pathway, thereby improving erectile function.
Hyperlipidemia occurs as a result of disorders of lipid metabolism in the body, leading to serum lipid concentration beyond normal levels. Hyperlipidemia is another metabolic factor that contributes to ED [53]. Studies have previously shown a linear relationship between the degree of hyperlipidemia and the risk of developing ED [54]. Hyperlipidemia causes ED through neuronal and endothelial dysfunction, leading to reduced cavernous NO levels, which are noted for their key role in the development of ED in men [55–57]. That study [54]showed that each 1 mmol/L increase in total cholesterol was associated with a 32% increase in ED risk, while each 1 mmol/L increase in high-density lipoprotein (HDL) was associated with a significant decrease in ED risk. Huang et al. [55] found that ADSC ameliorates hyperlipidemia-related adverse effects by differentiating into endothelial cells and secreting vasotropic and neurotrophic factors in a rat model.
Stem Cells in Aging-Associated ED
Erectile dysfunction is a common condition in older men, and as men’s life expectancy increases, the number of those desiring to regain sexual function is also likely to grow. The prevalence of ED increases with age, from 30.7% in men aged 57–64 years to 43.3% in men aged 75–85 years, with a linear increase in severity (P < 0.001) [58]. Drug therapy is a common treatment method, but the results are not satisfactory. Therefore, more effective strategies for erectile failure associated with older adult males are needed. Little is known about the underlying mechanisms of ED in older men, although some experts have suggested that it may be associated with smooth muscle and collagen content [59]. Accordingly, increasing the amount of smooth muscle and reducing collagen deposition is essential to improving age-associated ED. Aziz et al. [60] evaluated the effect of bone marrow mesenchymal stem cells on erection in aged rats. BMSCs were found to be a feasible treatment for aging-induced ED by measuring intracavernosal pressure, assessing cyclic guanosine monophosphate (cGMP) levels, and examining histopathology. Yang et al. [61] applied ADSCs to aged ED rats and found that ADSCs significantly improved erectile function in aged rats and that these cells also secreted large amounts of insulin-like growth factor-1 (IGF-1), bFGF, and VEGF in vitro by paracrine action and increased the concentrations of IGF-1, bFGF, and VEGF in penile tissue, increasing cavernous smooth muscle and endothelial content to further improve erectile function in aged rats. The authors therefore concluded that ADSCs improve aging-related ED function by secreting IGF-1, bFGF, and VEGF. As research continues, the potential role of non-coding RNAs composed of lncRNAs and microRNAs has become a hot topic in the progress of ED [62]. Liu et al. [63] evaluated the therapeutic efficiency of BMSCs overexpressing microRNA-145 (miR-145) in aged ED rats. They found that BMSCs overexpressing miR-145 increased the amount of smooth muscle by directly targeting Kruppel-like factor 4 (KLF4) and decreasing the expression of collagen 1 and matrix metallopeptidase 2. These changes resulted in a significantly enhanced erectile response in aged rats.
Stem Cells in the ED of Peyronie’s Disease and Corpus Cavernous Injury
Peyronie’s disease (PD) is a connective tissue disorder characterized by the presence of fibrous scar formation in the tunica albuginea, resulting in curvature deformity of the penis, penile pain on erection, and ED. The prevalence of this disease in the population is estimated to be as high as 7.1% [64]. Currently, the main treatments for PD include oral medications, ultrasound therapy, local injection of drugs into plaque lesions, and surgical correction; there is no gold standard for treating this disease [65].
Currently, studies involving rat models have been conducted to determine the effect of stem cell therapy on PD. Castiglione et al. [66] first investigated the application of stem cells in an animal model of PD and found that stem cells prevented cavernous fibrosis and elastic tissue formation in rats by local injection of ADSCs into the active phase of the PD rat model. Gokce et al. [67] found that the injection of ADSCs in a rat model both prevented and treated PD-related ED, and reported for the first time that the antifibrotic mechanism of ADSCs in animal models of PD supported further investigation of ADSCs as a new option for PD treatment. The surface marker characteristics were similar between BMSCs and ADSCs, but BMSCs showed stronger cartilage formation than ADSCs. BMSCs may be a more effective treatment for PD, but the precise mechanism has not been reported. Wang et al. [68] observed the effect of delivering BMSCs by spinal injection in a PD rat model during the active and chronic phases and analyzed the mechanism to prevent penile fibrosis in PD rats. The antifibrotic effect of treatment with BMSCs in the active phase of the PD rat model was found to be superior to that in the chronic phase. The antifibrotic mechanism of BMSCs was associated with increased mothers-against-decapentaplegic homolog 7 (SMAD7) expression. Some studies have reported that exosomes from stem cells significantly alleviate cardiovascular, pulmonary, and hepatic fibrosis [69–71], but there are no reports on the application of exosomes from stem cells for the prevention of fibrosis in PD. Yang et al. [72] injected exosomes of human USCs into PD rats, which significantly improved leukocyte fibrosis and enhanced erectile function by inhibiting the differentiation of fibroblasts to myofibroblasts and enhancing matrix protease activity in leukocytes. This mechanism provided a new therapeutic measure for the clinical application of USC-exo in the treatment of PD. Levy et al. [73] first used placental stroma-derived mesenchymal stem cells (pm-MSCs) to assess the feasibility and efficacy of treating human PD. At 6-week, 3-month, and 6-month follow-up intervals, they found a significant increase in peak systolic flow velocity in the penis after pm-MSC injections and the complete disappearance of seven penile plaques at the 3-month follow-up. Their research has also shown that pm-MSCs may be effective as a non-surgical treatment for patients with PD. Further studies will require observational follow-up in a larger group of patients.
Corpus cavernous injury is the main underlying cause of organic injury-related ED [74]. The corpus cavernosum tissue is mainly dependent on the blood supply from the cavernous sinus to maintain normal erectile function, so the microvascular system is an integral part of cavernous injury ED [75]. Corpus cavernous injury is a common cause of organic ED and requires safe and effective treatment. The cellular origin of the repaired tissue after penile injury remains unclear and controversial. One possible source is stem cells of endogenous origin in the injured tissue, or stem cells that migrate from nearby tissues to the site of tissue injury. Exogenous stem cells are another ideal cell source for repairing penile injury. Song et al. [76] investigated the feasibility of neural crest stem cells (NCSCs) for repairing penile corpus cavernosum injury and transplanted human neural stem crest cells HNC10. K10 (one of the immortalized human NCSC lines) immortalized the NCSC cell line into adult rat penile corpus cavernosum, and the transplanted NCSCs were able to differentiate into endothelial and smooth muscle cells in response to signals provided by the local microenvironment. Zou et al. [75] transfected miR-126 into MDSCs to establish miR126-MDSCs, which were then injected into rats with cavernous lesions. That study found that miRNA-126 MDSCs promoted revascularization and increased smooth muscle actin (SMA) content by directly targeting insulin receptor substrate 1 and Kruppel-like factor 10 (KLF10), which in turn improved erectile function in rats. MSC-Exos have great potential as a new cell-free therapeutic approach in ED treatment. Liu et al. [77] explored the mechanism of MSC-Exos treatment in an ED model of arterial injury in rats. In contrast to stem cell therapy, MSC-Exos provide a basis for clinical treatment of ED caused by severe arterial injury by promoting cavernous sinus endothelial formation, reducing tissue oxidative stress injury, and increasing nitric oxide synthase and smooth muscle content in the cavernous body. An et al. [78] combined mutated MDSCs with 3D printing technology to construct a hydrogel scaffold expressing hypoxia inducible factor-1α (HIF-1α); this was then transplanted into a rabbit model of cavernous injury, resulting in significant secretion of angiogenic factors by MDSCs under both hypoxic and aerobic conditions, promoting new angiogenesis and cavernous tissue repair and successfully restoring erectile and ejaculatory function. That was the first report of a 3D-printed bioengineered scaffold that successfully repairs erectile and ejaculatory function and restores reproductive capacity in rabbits, providing a theoretical basis for clinical application.
Stem Cell Applications in Human Clinical Trials
Stem cell transplantation has significantly advanced the treatment of animal models of ED, but there are fewer human clinical trials of stem cell treatment of ED. Significant differences are shown between animal and human models, with human trials focusing on the safety and efficacy of stem cell therapy for ED (Table 1). In most studies, the primary endpoints have been safety and tolerability of the treatment rather than impact on erectile function. In addition, these studies were difficult to standardize, which can only be done through further confirmatory studies using stem cells from different sources, leading to a broad safety hypothesis. Demour et al. [79] first reported the treatment of patients with DMED after two consecutive intracavitary autologous BMSC injections to observe patient tolerability, safety, and efficacy. That study found a significant increase in penile stiffness after a single transplant of BMSCs, but administration of a single dose and different multiple doses were not sufficient to restore penile mating function. Bahk et al. [80] injected hUC-MSCs into type 2 diabetic ED patients via cavernous injection to observe the effect of stem cell therapy. All patients had DMED and had not previously responded to drug therapy. After a single injection of allogeneic stem cells, some patients were found to have morning erections in the first month, and after 11 months of follow-up, patients were found to have improved ED and blood glucose levels simultaneously, but the exact mechanism was not elucidated and the safety of intracavernosal stem cell injection was not reported. Levy et al. [81] applied placental stem cells to patients with ED with 6-week, 3-month, and 6-month follow-ups and measured peak systolic velocity, end-diastolic flow rate, and penile length stretch, width, and erectile function, demonstrating that placental stem cells are useful for ED treatment. However, the sample size was too small and the authors recommended further studies. Protogerou et al. [82] introduced a new approach in the treatment of DMED by comparing the effects of autologous adipose stem cell therapy with those of platelet lysate. After 3 months of follow-up, patients treated with only injectable platelet lysate appeared to have comparable treatment outcomes to those treated with resuspended platelet lysate from ADMSCs, and no conclusion was made regarding which method was more effective.
Table 1.
Summary of the application of stem cells in clinical trials
| Author (year) | Patient number | Patient model | Mean age (years) | Stem cell type | Transplant method | Follow-up time | Number | Assessment | Side effects |
|---|---|---|---|---|---|---|---|---|---|
| Bahk et al. [80] (2010) | 7 | DMED | 69.5 | hUC-MSCs | ICI | 11 months | 1.5 × 107 | IIEF-5, blood glucose, hormonal levels, morning erection | No serious adverse events were reported. |
| Levy et al. [73] (2015) | 5 | PD | 51.2 | PM-MSCs | ICI | 6 weeks, 3 months, 6 months | 1 mL | Plaque volume, penile curvature, erectile function status, CDDU, IIEF, stretched penile length, penile girth | No serious adverse events were reported. |
| Levy et al. [81] (2016) | 8 | organic ED | – | PM-MSCs | ICI | 6 weeks, 3 months, 6 months | 1 mL | Plaque volume, penile curvature, erectile function status, CDDU, IIEF, stretched penile length, penile girth | No serious adverse events were reported. |
| Yiou et al. [83] (2016) | 12 | CNIED | 63.6 ± 4.2 | BM-MNCs | ICI | 1, 3, 6, and 12 months and last follow-up | 2 × 107, 2 × 108, 1 × 109, 2 × 109 | IIEF-15 EHS Penile length, CDDU | No serious adverse events were reported. |
| Yiou et al. [84] (2017) | 6 | CNIED | 59.9 ± 3.8 | BM-MNCs | ICI | 1, 3, and 6 months | 1 × 109 | IIEF-15 EHS Penile length, CDDU | No serious adverse events were reported. |
| Haahr et al. [85] (2016) | 17 | CNIED | 63.0 (9.0) | ADRCs | ICI | 1, 3, 6, and 12 months | 8.4–37.2 million | IIEF-5, EHS | No serious adverse events were reported; two men had experienced transient redness and swelling at the injection sites. One had a scrotal and penile hematoma. |
| Haahr et al. [86] (2018) | 21 | CNIED | 60.2 | ADRCs | ICI | 1, 3, 6, and 12 months | 2.21 × 107 | IIEF-5, EHS | No serious adverse events occurred. |
| Ory et al. [87] (2020) | 21 | CNIED | 61 | ADRCs | ICI | 1, 3, 6, and 12 months | 2.21 × 107 | IIEF-5, EHS | No serious adverse events were reported. Eight men experienced transient redness and swelling at the injection sites, and three reported reaction in the penile area. Five men stated minor abdominal hematomas. One man had an abdominal hematoma, which led to scrotal and penile hematomas. Eight patients reported light abdominal discomfort after liposuction, and four reported sensitive abdominal skin, but only 1 patient needed analgesic drugs in the days following liposuction. |
| Protogerou et al. [82] (2019) | 8 | CNIED, PD and DMED | – | ADSCs | ICI | 1 month and 3 months | 2 × 105 | IIEF-5, PSV, EDV | No severe adverse reactions were observed in any patient except minor pain at the site of injection. |
| Al Demour et al. [79] (2018) | 4 | DMED | 54.75 | BMSCs | ICI | At 24 h after first and second intracavernous injections, and at 1, 3, 6, 12, and 24 months after the second intracavernous injection | 30 × 106 | IIEF-15, EHS | No serious adverse events were reported. |
| You et al. [88] (2021) | 10 | CNIED and DMED | 62.0 ± 13.0 | BMSCs | ICI | 1, 3, 6, 9, and 12 months | 3 × 107 | IIEF, SEP 2 and 3, GAQ, CDDU | No serious adverse events were reported. One patient with post-prostatectomy ED experienced two TEAEs (pyrexia and back pain), and two patients with DM-associated ED experienced a total of five TEAEs (one case each of viral upper respiratory tract infection, prostatitis, and pruritus and two cases of hyperglycemia). All TEAEs were considered not to be related to autologous BMSC therapy. |
| Mirzaei et al. [89] (2021) | 20 | DMED | 63.8 ± 7.4 and 65.6 ± 5.1 | MSCs | ICI | 1 month and 3 months | 50–60 ×106 | IIEF-5, CDDU, FBS, HbA1c, morning erections | No serious adverse events were reported. |
| Al Demour et al. [90] (2021) | 22 | DMED | 59.18 ± 8.45 | Wharton’s jelly-derived mesenchymal stem cells | ICI | Immediately, 24 h, and 1, 3, 6, and 12 months | 20 × 106 | IIEF-5, EHS, CDDU | No serious adverse events were reported. |
| Koga et al. [91] (2022) | 38 | CNIED and DMED | 56 | Dental pulp stem cells | ICI | – | 1 × 107 | IIEF-5 | No serious adverse events were reported. |
hUC-MSCs, human umbilical cord mesenchymal stem cells, PD Peyronie’s disease, BM-MNCs bone marrow mononuclear cells, PM-MSCs placental matrix–derived mesenchymal stem cells, EHS erection hardness score, IIEF international index for erectile function, CDDU color duplex Doppler ultrasound, ADRCs adipose-derived regenerative cells, GAQ global assessment question, SEP sexual encounter profile, FBS fasting blood sugar, HbA1C glycosylated hemoglobin, type A1C TEAEs treatment-emergent adverse events
The efficacy of PDE-5 inhibitors (e.g., sildenafil, vardenafil, taldalafil, avanafil) in patients with ED with cavernous nerve denervation due to pelvic surgery and DM has not been satisfactory. You et al. [88] selected ten patients (five with DM-related ED and five with post-prostatectomy ED) who were unable to have sex despite long-term treatment with PDE5 inhibitors for a phase I clinical trial to assess the safety and potential efficacy of autologous BMSC treatment in patients with ED. The potential efficacy of autologous BMSC treatment for ED was assessed using international index of erectile function (IIEF) scores and participants’ satisfaction with intercourse. Both the safety and potential efficacy of the autologous BMSCs were demonstrated in patients with ED but the results still need to be confirmed with a phase II clinical trial. Haahr et al. [85] used freshly isolated autologous ADSCs to treat patients with ED after radical prostatectomy; the patients regained erectile function within 3 months and this effect lasted for 6 months, allowing the authors to conclude that freshly isolated ADSCs are a safe and well-tolerated treatment for patients. Another study by Haahr et al. [86] investigated the safety and effectiveness of ADSCs in treating patients with ED after radical prostatectomy. A total of 21 patients received autologous ADSCs injected into the corpus cavernosum and were followed for 1 year, assessing and recording adverse events using international index of erectile function questionnaire-5 (IIEF-5) scores and erection hardness grading scale (EHGS) scores. Results showed that 8 of the 21 participants regained adequate erectile function during the 12-month observation period. These eight patients had poor erectile assistance prior to stem cell treatment. After treatment, three men were able to complete sexual intercourse without erectile assistance. Six participants had ED prior to radical prostatectomy, and these six patients did not regain erectile function after receiving stem cell therapy. Levy et al. [81] studied the feasibility and efficacy of transplanting placental stromal MSCs in men with ED, concluding that it was a feasible treatment for ED. Koga et al. [91] first reported the efficacy of mastodontic dental pulp stem cells in patients with ED, finding that regeneration of damaged vascular tissue in the penile corpus cavernosum and restoration of erectile function at the cellular level was achieved by injecting conditioned medium of human shed mastodont stem cells (SHED-CM) into the penis of 38 patients with ED. According to their findings, patients with ED caused by mild-to-moderate vascular disease are considered suitable candidates for SHED-CM treatment. Protogerou et al. [92] performed stem cell therapy combined with platelet lysate plasma (PLP) in five patients with ED to improve erectile function, and erectile function was improved in all but one patient. However, the authors concluded that stem cell therapy was not a radical treatment, and that it was too early to promote this therapy, which should continue to be explored.
Human clinical trials have shown the promise of stem cell therapy as a restorative therapy for ED. However, despite the promise, data on the use of stem cell therapy for ED in humans remain very limited. As clinics offering stem cell therapy for ED expand, further research is needed on the safety, efficacy, and standardization of stem cell therapy. In addition, long-term follow-up studies are necessary to evaluate stem cell therapy.
Conclusions
Stem cell therapy is potentially safe and effective in the treatment of ED and remains a promising area of research. Significant results have been achieved through numerous preclinical studies, with stem cells improving erectile function by differentiating into neural cells, smooth muscle cells, and endothelial cells on the one hand, and enhancing erectile function by promoting angiogenesis and antifibrosis and anti-apoptosis through paracrine effects on the other hand.
However, stem cells still have many unresolved challenges in the treatment of ED. The relevant mechanisms in humans have not yet been fully elucidated, and the future direction of cell therapy still requires large-scale human studies. Meanwhile, many issues must still be resolved to achieve the safety, efficacy, and specific mechanisms of action of stem cells in treatment. Previous studies have reported that MSC can penetrate into prostate cancer cells, including prostate tissue to promote tumorigenesis and progression [93]. It is also meaningful to explore the origin of stem cells. Current research has found that both autologous and exogenous stem cells can improve erectile function, but the choice is worth exploring. Autologous stem cells are easy to obtain and safe, and can overcome the antigenicity problem of cell transplantation. Recently, it was found that allogeneic transplantation of ADSC secreted cytokines to regulate T cell and natural killer cells, thus avoiding immune rejection [12]. Autologous transplantation is not indicated in elderly patients and those with cancer [94]. These unresolved issues may include determining the optimal treatment protocol to address the immunogenicity and heterogeneity of stem cells, and evaluating the high cost and low efficiency of their application. Other issues may include small sample sizes, lack of blinding, and so on. Secondly, the types of stem cells, injectable doses, duration of treatment, and endpoints for evaluation of therapeutic effects have not yet been determined, coupled with ethical, legal, and social issues affecting the widespread use of stem cell therapy.
We believe that in the near future, more phase II and phase III clinical trials will be conducted to generate a high level of evidence, further clarify the specific mechanism of action, reduce the drawbacks of high cost and inefficiency, and fully prepare for the translation of stem cell therapy for ED from preclinical studies to clinical application.
Supplementary Information
Below is the link to the electronic supplementary material.
Declarations
Funding
No funding was received for the preparation of this manuscript.
Conflict of interest
Wei Wang, Ying Liu, Zuo-bin Zhu, Kun Pang, Jing-kai Wang, Jun Gu, Zhen-bei Li, Jian Wang, Zhen-duo Shi, and Cong-hui Han have no conflicts of interest that are directly relevant to the content of this article.
Ethics approval
No ethics approval was required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
All data represented are available in the public domain.
Code availability
Not applicable.
Author contributions
WW wrote the manuscript and designed the figures; WW, YL, ZB, KP, JKW, and ZS edited the manuscript; and ZL, JG, JW, and CH provided administration support. All authors read and approved the final manuscript.
Contributor Information
Zhen-duo Shi, Email: bujiniao2008@qq.com.
Cong-hui Han, Email: hanchdoctor@st.btbu.edu.cn.
References
- 1.Korneyev IA, et al. Prevalence and risk factors for erectile dysfunction and lower urinary tract symptoms in Russian Federation men: analysis from a national population-based multicenter study. Int J Impot Res. 2016;28(2):74–79. doi: 10.1038/ijir.2016.8. [DOI] [PubMed] [Google Scholar]
- 2.Irwin GM. Erectile dysfunction. Prim Care. 2019;46(2):249–255. doi: 10.1016/j.pop.2019.02.006. [DOI] [PubMed] [Google Scholar]
- 3.Mangir N, Turkeri L. Stem cell therapies in post-prostatectomy erectile dysfunction: a critical review. Can J Urol. 2017;24(1):8609–8619. [PubMed] [Google Scholar]
- 4.Burnett AL, et al. Erectile dysfunction: AUA guideline. J Urol. 2018;200(3):633–641. doi: 10.1016/j.juro.2018.05.004. [DOI] [PubMed] [Google Scholar]
- 5.Salonia A, et al. European association of urology guidelines on sexual and reproductive health-2021 update: male sexual dysfunction. Eur Urol. 2021;80(3):333–357. doi: 10.1016/j.eururo.2021.06.007. [DOI] [PubMed] [Google Scholar]
- 6.Alwaal A, et al. Stem cell treatment of erectile dysfunction. Adv Drug Deliv Rev. 2015;82–83:137–144. doi: 10.1016/j.addr.2014.11.012. [DOI] [PubMed] [Google Scholar]
- 7.Yafi FA, et al. Erectile dysfunction. Nat Rev Dis Primers. 2016;2:16003. doi: 10.1038/nrdp.2016.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chung DY, et al. Neutralizing antibody to proNGF rescues erectile function by regulating the expression of neurotrophic and angiogenic factors in a mouse model of cavernous nerve injury. Andrology. 2021;9(1):329–341. doi: 10.1111/andr.12873. [DOI] [PubMed] [Google Scholar]
- 9.Bratu O, et al. Erectile dysfunction post-radical prostatectomy—a challenge for both patient and physician. J Med Life. 2017;10(1):13–18. [PMC free article] [PubMed] [Google Scholar]
- 10.Martinez-Jabaloyas JM, et al. Prognostic factors for response to sildenafil in patients with erectile dysfunction. Eur Urol. 2001;40(6):641–646. doi: 10.1159/000049850. [DOI] [PubMed] [Google Scholar]
- 11.Asker H, et al. An update on the current status and future prospects of erectile dysfunction following radical prostatectomy. Prostate. 2022;82(12):1135–1161. doi: 10.1002/pros.24366. [DOI] [PubMed] [Google Scholar]
- 12.Albersen M, et al. Injections of adipose tissue-derived stem cells and stem cell lysate improve recovery of erectile function in a rat model of cavernous nerve injury. J Sex Med. 2010;7(10):3331–3340. doi: 10.1111/j.1743-6109.2010.01875.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ying CC, et al. Neural-like cells from adipose-derived stem cells for cavernous nerve injury in rats. Neural Regen Res. 2019;14(6):1085–1090. doi: 10.4103/1673-5374.250630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yang J, et al. Preconditioning of adipose-derived stem cells by phosphodiesterase-5 inhibition enhances therapeutic efficacy against diabetes-induced erectile dysfunction. Andrology. 2020;8(1):231–240. doi: 10.1111/andr.12661. [DOI] [PubMed] [Google Scholar]
- 15.Woo JC, et al. Transplantation of muscle-derived stem cells into the corpus cavernosum restores erectile function in a rat model of cavernous nerve injury. Korean J Urol. 2011;52(5):359–363. doi: 10.4111/kju.2011.52.5.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li M, et al. Exosomes derived from mesenchymal stem cells exert therapeutic effect in a rat model of cavernous nerves injury. Andrology. 2018;6(6):927–935. doi: 10.1111/andr.12519. [DOI] [PubMed] [Google Scholar]
- 17.Zhuang J, et al. Extracellular vesicles from human urine-derived stem cells merged in hyaluronic acid ameliorate erectile dysfunction in type 2 diabetic rats by glans administration. Andrology. 2022;10(8):1673–1686. doi: 10.1111/andr.13293. [DOI] [PubMed] [Google Scholar]
- 18.Gu X, et al. Dynamic changes in erectile function and histological architecture after intracorporal injection of human placental stem cells in a pelvic neurovascular injury rat model. J Sex Med. 2020;17(3):400–411. doi: 10.1016/j.jsxm.2019.12.002. [DOI] [PubMed] [Google Scholar]
- 19.Wu J, et al. Transplantation of human gingiva-derived mesenchymal stem cells ameliorates neurotic erectile dysfunction in a rat model. Front Bioeng Biotechnol. 2021;9:630076. doi: 10.3389/fbioe.2021.630076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Matz EL, et al. Stem cell therapy for erectile dysfunction. Sex Med Rev. 2019;7(2):321–328. doi: 10.1016/j.sxmr.2017.12.008. [DOI] [PubMed] [Google Scholar]
- 21.Wu H, et al. Nanotechnology-assisted adipose-derived stem cell (ADSC) therapy for erectile dysfunction of cavernous nerve injury: in vivo cell tracking, optimized injection dosage, and functional evaluation. Asian J Androl. 2018;20(5):442–447. doi: 10.4103/aja.aja_48_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lin H, et al. Nanoparticle improved stem cell therapy for erectile dysfunction in a rat model of cavernous nerve injury. J Urol. 2016;195(3):788–795. doi: 10.1016/j.juro.2015.10.129. [DOI] [PubMed] [Google Scholar]
- 23.Cheng J, et al. A new strategy for stem cells therapy for erectile dysfunction: adipose-derived stem cells transfect Neuregulin-1 gene through superparamagnetic iron oxide nanoparticles. Investig Clin Urol. 2022;63(3):359–367. doi: 10.4111/icu.20220016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shao J, et al. An EPO-loaded multifunctional hydrogel synergizing with adipose-derived stem cells restores neurogenic erectile function via enhancing nerve regeneration and penile rehabilitation. Bioeng Transl Med. 2022;7(3):e10319. doi: 10.1002/btm2.10319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liang L, et al. Photoacoustic image-guided corpus cavernosum intratunical injection of adipose stem cell-derived exosomes loaded polydopamine thermosensitive hydrogel for erectile dysfunction treatment. Bioact Mater. 2022;9:147–156. doi: 10.1016/j.bioactmat.2021.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jung AR, et al. Stem cell/oxygen-releasing microparticle enhances erectile function in a cavernous nerve injury model. Tissue Eng Part A. 2021;27(1–2):50–62. doi: 10.1089/ten.tea.2019.0240. [DOI] [PubMed] [Google Scholar]
- 27.Anwar Z, et al. Erectile dysfunction: an underestimated presentation in patients with diabetes mellitus. Indian J Psychol Med. 2017;39(5):600–604. doi: 10.4103/0253-7176.217015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Albersen M, Weyne E, Bivalacqua TJ. Stem cell therapy for erectile dysfunction: progress and future directions. Sex Med Rev. 2013;1(1):50–64. doi: 10.1002/smrj.5. [DOI] [PubMed] [Google Scholar]
- 29.Pakpahan C, et al. Stem cell therapy and diabetic erectile dysfunction: a critical review. World J Stem Cells. 2021;13(10):1549–1563. doi: 10.4252/wjsc.v13.i10.1549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang H, et al. Probucol enhances the therapeutic efficiency of mesenchymal stem cells in the treatment of erectile dysfunction in diabetic rats by prolonging their survival time via Nrf2 pathway. Stem Cell Res Ther. 2020;11(1):302. doi: 10.1186/s13287-020-01788-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sun X, et al. Down-regulation of lncRNA MEG3 promotes endothelial differentiation of bone marrow derived mesenchymal stem cells in repairing erectile dysfunction. Life Sci. 2018;208:246–252. doi: 10.1016/j.lfs.2018.07.024. [DOI] [PubMed] [Google Scholar]
- 32.Sun X, Luo L, Li J. LncRNA MALAT1 facilitates BM-MSCs differentiation into endothelial cells via targeting miR-206/VEGFA axis. Cell Cycle. 2020;19(22):3018–3028. doi: 10.1080/15384101.2020.1829799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Garcia MM, et al. Treatment of erectile dysfunction in the obese type 2 diabetic ZDF rat with adipose tissue-derived stem cells. J Sex Med. 2010;7(1 Pt 1):89–98. doi: 10.1111/j.1743-6109.2009.01541.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bonafede R, et al. Exosome derived from murine adipose-derived stromal cells: neuroprotective effect on in vitro model of amyotrophic lateral sclerosis. Exp Cell Res. 2016;340(1):150–158. doi: 10.1016/j.yexcr.2015.12.009. [DOI] [PubMed] [Google Scholar]
- 35.Chen F, et al. Adipose-derived stem cell-derived exosomes ameliorate erectile dysfunction in a rat model of type 2 diabetes. J Sex Med. 2017;14(9):1084–1094. doi: 10.1016/j.jsxm.2017.07.005. [DOI] [PubMed] [Google Scholar]
- 36.Liu G, et al. Correction of diabetic erectile dysfunction with adipose derived stem cells modified with the vascular endothelial growth factor gene in a rodent diabetic model. PLoS One. 2013;8(8):e72790. doi: 10.1371/journal.pone.0072790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wils J, Favre J, Bellien J. Modulating putative endothelial progenitor cells for the treatment of endothelial dysfunction and cardiovascular complications in diabetes. Pharmacol Ther. 2017;170:98–115. doi: 10.1016/j.pharmthera.2016.10.014. [DOI] [PubMed] [Google Scholar]
- 38.Yang Q, et al. Combined transplantation of adipose tissue-derived stem cells and endothelial progenitor cells improve diabetic erectile dysfunction in a rat model. Stem Cells Int. 2020;2020:2154053. doi: 10.1155/2020/2154053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Quaade ML, et al. Adipose-derived stem cells from type 2 diabetic rats retain positive effects in a rat model of erectile dysfunction. Int J Mol Sci. 2022;23(3):1692. doi: 10.3390/ijms23031692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Luo C, et al. NLRP3 downregulation enhances engraftment and functionality of adipose-derived stem cells to alleviate erectile dysfunction in diabetic rats. Front Endocrinol (Lausanne) 2022;13:913296. doi: 10.3389/fendo.2022.913296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen S, et al. Comparison of the therapeutic effects of adipose-derived and bone marrow mesenchymal stem cells on erectile dysfunction in diabetic rats. Int J Mol Med. 2019;44(3):1006–1014. doi: 10.3892/ijmm.2019.4254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sun X, et al. B cell lymphoma-2-modified bone marrow-derived mesenchymal stem cells transplantation for the treatment of diabetes mellitus-induced erectile dysfunction in a rat model. Urol Int. 2017;98(3):358–366. doi: 10.1159/000452253. [DOI] [PubMed] [Google Scholar]
- 43.Ouyang B, et al. Extracellular vesicles from human urine-derived stem cells ameliorate erectile dysfunction in a diabetic rat model by delivering proangiogenic microRNA. Sex Med. 2019;7(2):241–250. doi: 10.1016/j.esxm.2019.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Galhom RA, et al. Urine-derived stem cells versus their lysate in ameliorating erectile dysfunction in a rat model of type 2 diabetes. Front Physiol. 2022;13:854949. doi: 10.3389/fphys.2022.854949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zhang C, et al. Transplantation of human urine-derived stem cells ameliorates erectile function and cavernosal endothelial function by promoting autophagy of corpus cavernosal endothelial cells in diabetic erectile dysfunction rats. Stem Cells Int. 2019;2019:2168709. doi: 10.1155/2019/2168709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Xie Q, et al. What is the impact of human umbilical cord mesenchymal stem cell transplantation on clinical treatment? Stem Cell Res Ther. 2020;11(1):519. doi: 10.1186/s13287-020-02011-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cengiz T, et al. Intracavernous injection of human umbilical cord blood mononuclear cells improves erectile dysfunction in streptozotocin-induced diabetic rats. J Sex Med. 2017;14(1):50–58. doi: 10.1016/j.jsxm.2016.11.314. [DOI] [PubMed] [Google Scholar]
- 48.Wang S, et al. A study of diabetes-induced erectile dysfunction treated with human umbilical cord mesenchymal stem cells. Andrologia. 2022;54(7):e14440. doi: 10.1111/and.14440. [DOI] [PubMed] [Google Scholar]
- 49.Mukti AI, et al. Umbilical cord-derived mesenchymal stem cells improve TGF-beta, alpha-SMA and collagen on erectile dysfunction in streptozotocin-induced diabetic rats. Med Arch. 2022;76(1):4–11. doi: 10.5455/medarh.2022.76.4-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Feng H, et al. Human umbilical cord mesenchymal stem cells ameliorate erectile dysfunction in rats with diabetes mellitus through the attenuation of ferroptosis. Stem Cell Res Ther. 2022;13(1):450. doi: 10.1186/s13287-022-03147-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shan HT, et al. Combination of low-energy shock-wave therapy and bone marrow mesenchymal stem cell transplantation to improve the erectile function of diabetic rats. Asian J Androl. 2017;19(1):26–33. doi: 10.4103/1008-682X.184271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu S, et al. Low-intensity pulsed ultrasound enhanced adipose-derived stem cell-mediated angiogenesis in the treatment of diabetic erectile dysfunction through the Piezo-ERK-VEGF Axis. Stem Cells Int. 2022;2022:6202842. doi: 10.1155/2022/6202842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Virag R, Bouilly P, Frydman D. Is impotence an arterial disorder? A study of arterial risk factors in 440 impotent men. Lancet. 1985;1(8422):181–184. doi: 10.1016/S0140-6736(85)92023-9. [DOI] [PubMed] [Google Scholar]
- 54.Wei M, et al. Total cholesterol and high density lipoprotein cholesterol as important predictors of erectile dysfunction. Am J Epidemiol. 1994;140(10):930–937. doi: 10.1093/oxfordjournals.aje.a117181. [DOI] [PubMed] [Google Scholar]
- 55.Huang YC, et al. The effect of intracavernous injection of adipose tissue-derived stem cells on hyperlipidemia-associated erectile dysfunction in a rat model. J Sex Med. 2010;7(4 Pt 1):1391–1400. doi: 10.1111/j.1743-6109.2009.01697.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bocchio M, et al. Endothelial cell activation in men with erectile dysfunction without cardiovascular risk factors and overt vascular damage. J Urol. 2004;171(4):1601–1604. doi: 10.1097/01.ju.0000116325.06572.85. [DOI] [PubMed] [Google Scholar]
- 57.Yetik-Anacak G, Catravas JD. Nitric oxide and the endothelium: history and impact on cardiovascular disease. Vascul Pharmacol. 2006;45(5):268–276. doi: 10.1016/j.vph.2006.08.002. [DOI] [PubMed] [Google Scholar]
- 58.Shamloul R, Ghanem H. Erectile dysfunction. Lancet. 2013;381(9861):153–165. doi: 10.1016/S0140-6736(12)60520-0. [DOI] [PubMed] [Google Scholar]
- 59.Ferrer JE, Velez JD, Herrera AM. Age-related morphological changes in smooth muscle and collagen content in human corpus cavernosum. J Sex Med. 2010;7(8):2723–2728. doi: 10.1111/j.1743-6109.2009.01508.x. [DOI] [PubMed] [Google Scholar]
- 60.Abdel AM, et al. Effect of mesenchymal stem cell penile transplantation on erectile signaling of aged rats. Andrologia. 2010;42(3):187–192. doi: 10.1111/j.1439-0272.2009.00977.x. [DOI] [PubMed] [Google Scholar]
- 61.Yang J, et al. Adipose-derived stem cells improve erectile function partially through the secretion of IGF-1, bFGF, and VEGF in aged rats. Andrology. 2018;6(3):498–509. doi: 10.1111/andr.12483. [DOI] [PubMed] [Google Scholar]
- 62.Pan L, et al. Long non-coding RNA expression profiling in aging rats with erectile dysfunction. Cell Physiol Biochem. 2015;37(4):1513–1526. doi: 10.1159/000438519. [DOI] [PubMed] [Google Scholar]
- 63.Liu Q, et al. MicroRNA-145 engineered bone marrow-derived mesenchymal stem cells alleviated erectile dysfunction in aged rats. Stem Cell Res Ther. 2019;10(1):398. doi: 10.1186/s13287-019-1509-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Garaffa G, et al. Understanding the course of Peyronie's disease. Int J Clin Pract. 2013;67(8):781–788. doi: 10.1111/ijcp.12129. [DOI] [PubMed] [Google Scholar]
- 65.Cowper MG, et al. Penile stretching as a treatment for Peyronie's disease: a review. Sex Med Rev. 2019;7(3):508–515. doi: 10.1016/j.sxmr.2018.11.002. [DOI] [PubMed] [Google Scholar]
- 66.Castiglione F, et al. Intratunical injection of human adipose tissue-derived stem cells prevents fibrosis and is associated with improved erectile function in a rat model of Peyronie's disease. Eur Urol. 2013;63(3):551–560. doi: 10.1016/j.eururo.2012.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gokce A, et al. Adipose tissue-derived stem cell therapy for prevention and treatment of erectile dysfunction in a rat model of Peyronie's disease. Andrology. 2014;2(2):244–251. doi: 10.1111/j.2047-2927.2013.00181.x. [DOI] [PubMed] [Google Scholar]
- 68.Wang W, et al. Intratunical injection of rat-derived bone marrow mesenchymal stem cells prevents fibrosis and is associated with increased Smad7 expression in a rat model of Peyronie's disease. Stem Cell Res Ther. 2022;13(1):390. doi: 10.1186/s13287-022-03090-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Moghaddam AS, et al. Cardioprotective microRNAs: Lessons from stem cell-derived exosomal microRNAs to treat cardiovascular disease. Atherosclerosis. 2019;285:1–9. doi: 10.1016/j.atherosclerosis.2019.03.016. [DOI] [PubMed] [Google Scholar]
- 70.Rong X, et al. Human bone marrow mesenchymal stem cells-derived exosomes alleviate liver fibrosis through the Wnt/beta-catenin pathway. Stem Cell Res Ther. 2019;10(1):98. doi: 10.1186/s13287-019-1204-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sun L, et al. Exosomal miRNA Let-7 from menstrual blood-derived endometrial stem cells alleviates pulmonary fibrosis through regulating mitochondrial DNA damage. Oxid Med Cell Longev. 2019;2019:4506303. doi: 10.1155/2019/4506303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yang Q, et al. Intratunical injection of human urine-derived stem cells derived exosomes prevents fibrosis and improves erectile function in a rat model of Peyronie's disease. Andrologia. 2020;52(11):e13831. doi: 10.1111/and.13831. [DOI] [PubMed] [Google Scholar]
- 73.Levy JA, et al. Effects of stem cell treatment in human patients with Peyronie disease. J Am Osteopath Assoc. 2015;115(10):e8–13. doi: 10.7556/jaoa.2015.124. [DOI] [PubMed] [Google Scholar]
- 74.Jiann BP, Su CC, Tsai JY. Is female sexual function related to the male partners' erectile function? J Sex Med. 2013;10(2):420–429. doi: 10.1111/j.1743-6109.2012.03007.x. [DOI] [PubMed] [Google Scholar]
- 75.Zou Z, et al. MicroRNA-126 engineered muscle-derived stem cells attenuates cavernosa injury-induced erectile dysfunction in rats. Aging (Albany NY) 2021;13(10):14399–14415. doi: 10.18632/aging.203057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Song YS, et al. Human neural crest stem cells transplanted in rat penile corpus cavernosum to repair erectil e dysfunction. BJU Int. 2008;102(2):220–224. doi: 10.1111/j.1464-410X.2008.07469.x. [DOI] [PubMed] [Google Scholar]
- 77.Liu Y, et al. Mesenchymal stem cell-derived exosomes ameliorate erection by reducing oxidative stress damage of corpus cavernosum in a rat model of artery injury. J Cell Mol Med. 2019;23(11):7462–7473. doi: 10.1111/jcmm.14615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.An G, et al. Functional reconstruction of injured corpus cavernosa using 3D-printed hydrogel scaffolds seeded with HIF-1alpha-expressing stem cells. Nat Commun. 2020;11(1):2687. doi: 10.1038/s41467-020-16192-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Al DS, et al. Safety and potential therapeutic effect of two intracavernous autologous bone marrow derived mesenchymal stem cells injections in diabetic patients with erectile dysfunction: an open label phase i clinical trial. Urol Int. 2018;101(3):358–365. doi: 10.1159/000492120. [DOI] [PubMed] [Google Scholar]
- 80.Bahk JY, et al. Treatment of diabetic impotence with umbilical cord blood stem cell intracavernosal transplant: preliminary report of 7 cases. Exp Clin Transplant. 2010;8(2):150–160. [PubMed] [Google Scholar]
- 81.Levy JA, et al. Determining the feasibility of managing erectile dysfunction in humans with placental-derived stem cells. J Am Osteopath Assoc. 2016;116(1):e1–5. doi: 10.7556/jaoa.2016.007. [DOI] [PubMed] [Google Scholar]
- 82.Protogerou V, et al. Administration of adipose derived mesenchymal stem cells and platelet lysate in erectile dysfunction: a single center pilot study. Bioengineering (Basel) 2019;6(1):21. doi: 10.3390/bioengineering6010021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yiou R, et al. Safety of intracavernous bone marrow-mononuclear cells for postradical prostatectomy erectile dysfunction: an open dose-escalation pilot study. Eur Urol. 2016;69(6):988–991. doi: 10.1016/j.eururo.2015.09.026. [DOI] [PubMed] [Google Scholar]
- 84.Yiou R, et al. Intracavernous injections of bone marrow mononucleated cells for postradical prostatectomy erectile dysfunction: final results of the INSTIN clinical trial. Eur Urol Focus. 2017;3(6):643–645. doi: 10.1016/j.euf.2017.06.009. [DOI] [PubMed] [Google Scholar]
- 85.Haahr MK, et al. Safety and potential effect of a single intracavernous injection of autologous adipose-derived regenerative cells in patients with erectile dysfunction following radical prostatectomy: an open-label phase i clinical trial. EBioMedicine. 2016;5:204–210. doi: 10.1016/j.ebiom.2016.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Haahr MK, et al. A 12-month follow-up after a single intracavernous injection of autologous adipose-derived regenerative cells in patients with erectile dysfunction following radical prostatectomy: an open-label phase i clinical trial. Urology. 2018;121:203.e6–203.e13. doi: 10.1016/j.urology.2018.06.018. [DOI] [PubMed] [Google Scholar]
- 87.Ory J, et al. The effect of transendocardial stem cell injection on erectile function in men with cardiomyopathy: results from the TRIDENT, POSEIDON, and TAC-HFT trials. J Sex Med. 2020;17(4):695–701. doi: 10.1016/j.jsxm.2020.01.003. [DOI] [PubMed] [Google Scholar]
- 88.You D, et al. Safety of autologous bone marrow-derived mesenchymal stem cells in erectile dysfunction: an open-label phase 1 clinical trial. Cytotherapy. 2021;23(10):931–938. doi: 10.1016/j.jcyt.2021.06.001. [DOI] [PubMed] [Google Scholar]
- 89.Mirzaei M, et al. The effect of intracavernosal injection of stem cell in the treatment of erectile dysfunction in diabetic patients: a randomized single-blinded clinical trial. Urol J. 2021;18(6):675–681. doi: 10.22037/uj.v18i.6503. [DOI] [PubMed] [Google Scholar]
- 90.Al DS, et al. Safety and efficacy of 2 intracavernous injections of allogeneic Wharton's jelly-derived mesenchymal stem cells in diabetic patients with erectile dysfunction: phase 1/2 clinical trial. Urol Int. 2021;105(11–12):935–943. doi: 10.1159/000517364. [DOI] [PubMed] [Google Scholar]
- 91.Koga S, Horiguchi Y. Efficacy of a cultured conditioned medium of exfoliated deciduous dental pulp stem cells in erectile dysfunction patients. J Cell Mol Med. 2022;26(1):195–201. doi: 10.1111/jcmm.17072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Protogerou V, et al. The combined use of stem cells and platelet lysate plasma for the treatment of erectile dysfunction: a pilot study-6 months results. Medicines (Basel) 2020;7(3):14. doi: 10.3390/medicines7030014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Brennen WN, et al. Mesenchymal stem cell infiltration during neoplastic transformation of the human prostate. Oncotarget. 2017;8(29):46710–46727. doi: 10.18632/oncotarget.17362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Mohr A, Zwacka R. The future of mesenchymal stem cell-based therapeutic approaches for cancer—from cells to ghosts. Cancer Lett. 2018;414:239–249. doi: 10.1016/j.canlet.2017.11.025. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data represented are available in the public domain.