Abstract
The African swine fever virus gene A179L has been shown to be a functional member of the ced9/bcl-2 family of apoptosis inhibitors in mammalian cell lines. In this work we have expressed the A179L gene product (p21) under the control of the baculovirus polyhedrin promoter using a baculovirus system. Expression of the A179L gene neither altered the baculovirus replication phenotype nor delayed the shutoff of cellular protein synthesis, but it extended the survival of the infected insect cells to very late times postinfection. The increase in cell survival rates correlated with a marked apoptosis reduction after baculovirus infection. Interestingly, prevention of apoptosis was observed when recombinant baculovirus infections were carried out in monolayer cell cultures but not when cells were infected in suspension, suggesting a cell anchorage dependence for p21 function in insect cells. Cell survival was enhanced under optimal conditions of cell attachment and cell-to-cell contact as provided by extracellular matrix components or poly-d-lysine. Since it was observed that cytoskeleton organization varied depending on culture conditions of insect cells (grown in monolayer versus grown in suspension), these results suggested that A179L might regulate apoptosis in insect cells only when the cytoskeletal support of intracellular signaling is maintained upon cell adhesion. Thus, cell shape and cytoskeleton status might allow variations in intracellular transduction of signals related to cell survival in virus-infected cells.
African swine fever virus (ASFV), the causative agent for an important disease of swine, is a large double-stranded DNA virus that replicates not only in members of the Suidae family but also in soft ticks of the Ornithodoros genus (24, 31, 41). ASFV infects a variety of cells of the mononuclear phagocytic system and produces a characteristic apoptotic cell death in infected swine macrophages and bystander uninfected lymphocytes (33–35). Virus-induced apoptosis in target cells is produced late during infection, suggesting the existence of viral genes that prevent early apoptosis to support the productive infection. An ASFV gene, 5HL (A179L in Ba71V virus), with sequence similarity to the ced9/bcl-2 gene family has been described (28). This gene encodes a protein of approximately 21 kDa (p21) that is synthesized in infected cells at both early and late times postinfection (28). The similarity of this gene to bcl-2 has pointed to its possible role in apoptosis inhibition during ASFV infection. We have recently demonstrated that expression of the A179L gene by a recombinant vaccinia virus inhibits apoptosis mediated by the interferon-induced double-stranded RNA-activated protein kinase in HeLa and BSC-40 cells (5). Also, the A179L product is able to suppress apoptotic cell death in the FL5.12 mouse prolymphocytic cell line and in the K562 human myeloid leukemia cell line (1, 37). Thus, the ability of the A179L gene to suppress apoptosis in mammalian cells has been clearly shown. However, the function in insect cells of the bcl-2 gene, the prototype of a death-regulator gene family, remains controversial (3, 8, 11), and the function of A179L during ASFV infection of nonmammalian cells is still unknown. Other ASFV genes potentially involved in the regulation of intracellular apoptosis pathways are A238L and 23NL, which have sequence similarity to the IκB factor (32) and the ICP34.5 gene of herpes simplex virus I (14, 40), respectively. The existence of an ASFV gene (A224L) with similarity to the iap family of apoptosis inhibitors suggested that A179L and A224L could function as host range genes (9). Nevertheless, there are no current data supporting the function of A224L as an apoptosis inhibitor, and studies conducted to analyze its role during ASFV infection demonstrated that this gene is dispensable for ASFV growth in swine macrophages (29).
Therefore, the study of the apoptosis regulatory functions of the A179L gene in different cell lines and under different culture conditions is important to an understanding of the role of this gene during ASFV infection in its different hosts. To examine these functions, we have expressed the A179L gene in Spodoptera frugiperda (Sf9) cells using a baculovirus system. We demonstrated that this gene is functional and prevents virus-induced apoptosis in these cells only when intracellular signaling upon cell adhesion is maintained.
Expression of the A179L gene product in Sf9 cells.
The construction of recombinant baculovirus expressing the A179L gene product was carried out as previously described (21, 30). Briefly, DNA amplification of the A179L gene from the ASFV isolate E70 was carried out by PCR with ampliTaq DNA polymerase (Perkin-Elmer Cetus) with the primers (i) 5′-AAATATAGGGATCCGCTATGGAGGG (5′ primer) and (ii) 5′-CCGCGTGGATCCTATATCAAATTGC (3′ primer). Both primers contain the recognition sequence for the BamHI restriction enzyme. The PCR product was digested with BamHI and cloned into the BamHI site of the baculovirus transfer vector pBacPAK8 (Clontech) under the control of the polyhedrin promoter. The cloned gene was sequenced by the dideoxynucleotide chain terminator method by using specific primers to check possible sequence changes introduced by PCR amplification of the gene. Then, Sf9 cells were cotransfected with the transfer recombinant vector (pBacPAK-A179L) and the purified, noninfectious BacPAK6 DNA (Bsu36I digested), which contains the β-galactosidase gene under the control of the polyhedrin promoter. Isolation of recombinant baculovirus was achieved by negative selection due to replacement of the β-galactosidase gene by the newly introduced recombinant gene. The selected virus was further purified after three successive plaque assays in Sf9 cells. The recombinant baculovirus expressing the A179L gene was denominated BVA179L. Another recombinant baculovirus bearing the A179L gene cloned in the opposite orientation (antisense, BVA179L-AS) was constructed by similar methods.
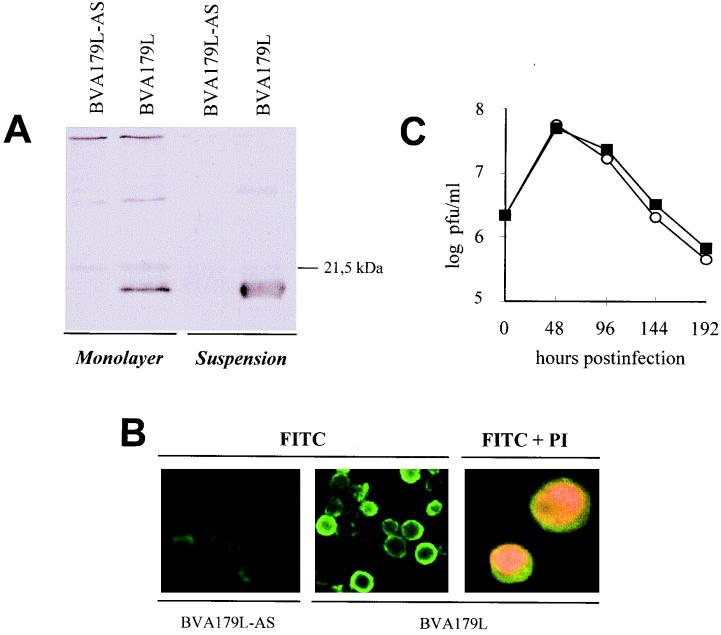
Expression of the A179L gene by the recombinant baculovirus BVA179L was analyzed by Western blotting at 72 h postinfection (p.i.) in cells from suspension and monolayer cultures (Fig. 1A). Cell extracts with similar numbers of infected cells from both cultures were lysed, electrophoresed in sodium dodecyl sulfate (SDS)–15% polyacrylamide gels, transferred to nitrocellulose filters (Bio-Rad), and then incubated with a polyclonal antiserum raised against the product of the ASFV gene 5HL expressed in Escherichia coli cells (28). The serum reacted with a 21-kDa polypeptide (p21) but failed to react with any protein product of this electrophoretic mobility in mock-infected cells (data not shown) or in cells infected with the recombinant baculovirus BVA179L-AS (Fig. 1A).
FIG. 1.
Expression of the ASFV A179L gene by using a baculovirus system. (A) Western blot analysis of infected Sf9 cell extracts. Cell lysates from Sf9 cells synchronously infected either with the recombinant baculovirus BVA179L or with BVA179L-AS reacted at 72 h p.i. with a specific antiserum raised against the A179L gene product expressed in E. coli cells. (B) Immunofluorescence of BVA179L- or BVA179L-AS-infected insect cells in suspension, analyzed at 72 h p.i. by using the same serum against A179L stained with fluorescein isothiocyanate (FITC). Green cytoplasmic fluorescence was detected only in cells infected with BVA179L. The cell nucleus was contrasted with propidium iodide (PI) (right). (C) Growth curves of BVA179L and BVA179L-AS recombinant baculoviruses in Sf9 monolayer cell cultures. Extracellular virus obtained from culture supernatants at the indicated time points was titrated in a conventional plaque assay. Squares, BVA179L-AS; circles, BVA179L.
Indirect immunofluorescence of fixed and permeabilized BVA179L- or BVA179L-AS-infected Sf9 cells, using the same anti-p21 serum, revealed predominantly cytoplasmic staining for p21 (Fig. 1B, center), which contrasted with the nuclear localization of propidium iodide, a DNA-intercalating agent (Fig. 1B, right). This distribution of p21 was similar in cells grown in suspension or in adherent growth conditions.
The effect of p21 expression on recombinant baculovirus growth was also investigated (Fig. 1C). Cells were infected (multiplicity of infection [MOI], 2), supernatants were collected at different times postinfection for titration, and fresh medium was added to the cultures. Infection with BVA179L or BVA179L-AS yielded similar virus titers, suggesting that overexpression of the A179L gene did not alter the baculovirus replication phenotype in Sf9 cells. Maximum viral titers were reached at 48 h p.i. with both recombinant viruses (Fig. 1C). As expected, virus yields dropped drastically after this time point, because the experiment measured virus release to the extracellular medium rather than virus accumulation. This result suggested the occurrence of a strong shutoff of protein synthesis due to baculovirus infection at late time points.
In order to confirm this fact, long-term synthesis of baculovirus-induced proteins in the presence of the apoptosis inhibitor p21 was analyzed by metabolic pulse labeling of infected cells at different times postinfection. Sf9 cells (105) were mock infected or infected at a MOI of 10 in 96-well culture plates. At 22, 46, 70, and 94 h p.i. the medium was replaced with Grace’s medium lacking methionine (Gibco) and maintained for 1 h prior to the addition of fresh methionine-deficient medium containing 200 μCi of [35S]methionine/ml (>1,000 Ci/mmol). At the selected time points cell pellets were harvested, lysed in SDS buffer, and analyzed by autoradiography after SDS-polyacrylamide gel electrophoresis. A strong shutoff of protein synthesis was observed starting from 48 h p.i. in monolayer cultures infected with BVA179L, BVA179L-AS, or a baculovirus expressing β-galactosidase (data not shown). At 96 h p.i. no newly synthesized cellular or viral-induced proteins were detected in any infected culture.
Functionality of the A179L gene in insect cells.
To determine the possible role of the A179L gene in prevention of baculovirus-induced apoptosis, viability of cell cultures infected at MOI of 2, 10 and 100 was determined at various times postinfection by trypan blue exclusion by counting 1 × 103 to 1.4 × 103 cells in five independent fields each (Fig. 2). High MOI were used to assure a synchronized infection with 99% infected cells. Viral infections on monolayer cell cultures were carried out in multiwell plastic dishes (Nunc). For the infection of suspension cultures, 250-ml flasks in a rotary shaker, with constant stirring at 80 rpm, were used. Both monolayer and suspension cultures were maintained in Grace’s insect medium (Gibco-BRL) supplemented with 10% fetal bovine serum. Cells were inoculated with extracellular, budded virus, and viral titers were determined by plaque assay on 106 cells seeded onto 35-mm dishes and overlaid with a mixture of 0.7% agarose (Sigma) in 10% fetal bovine serum–Grace’s medium. Infections with a control baculovirus expressing the reporter gene β-galactosidase at a MOI of 10 or higher yielded more than 95% cells expressing the reporter gene in both monolayer and suspension (data not shown). With all recombinants, all cells showed a clear cytopathic effect characteristic of baculovirus productive infection.
FIG. 2.
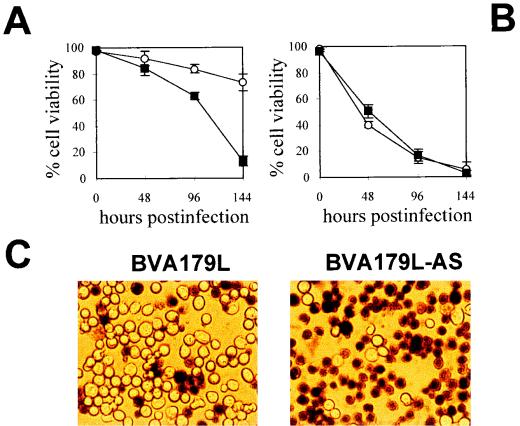
Effect of A179L gene expression on the survival of baculovirus-infected Sf9 cells. Viability of insect cells grown in monolayers (A) or in suspension (B) and infected with BVA179L-AS (squares) or BVA179L (circles) at a MOI of 100 was determined by trypan blue exclusion at different times postinfection. Means and standard errors were calculated from three independent experiments. (C) A representative field of trypan blue-stained monolayer cultures of BVA179L- or BVA179L-AS-infected Sf9 cells at 144 h p.i. Original magnification, ×200.
The viability levels of baculovirus-infected cells correlated with the postinfection time. Differences in cell viability were consistently found after 48 h p.i., when viral yields reached a maximum and protein shutoff was more evident. In monolayer cultures (Fig. 2A), fewer than 20% of the BVA179L-AS-infected cells survived to infection at 144 h p.i. (Fig. 2A). In contrast, cells infected with the baculovirus expressing the A179L gene presented only a slight decrease in viability at this time point (Fig. 2A). A representative field of cells infected with recombinant baculoviruses stained with trypan blue at 144 h p.i. is shown in Fig. 2C. Thus, the expression of p21 at late times postinfection increased the survival of baculovirus-infected Sf9 cells cultured in monolayer.
However, in suspension, viability experiments carried out with cells infected with either BVA179L or BVA179L-AS did not result in demonstrable differences in survival rates (Fig. 2B). High proportions of dead cells (about 50%) were found at 48 h p.i. in spite of p21 expression. These discrepancies found between monolayer and suspension cultures suggested that the effect of p21 expression on Sf9 viability could be related to the lack of cell attachment to a substrate in suspended cultures.
We then investigated whether the increased viability found in BVA179L-infected cells in monolayer cultures was due to apoptosis inhibition. Since both viruses expressed identical levels of baculovirus p35 at early times (11), infection-induced apoptosis should be similar in both cases, thus minimizing the differences in survival rates before 48 h p.i. Beyond this time point, when p35 is no longer functional (8, 11), differences between BVA179L and BVA179L-AS, not expressing p21, might become evident. As programmed cell death is relatively highly conserved during evolution and inhibitors of apoptosis are functionally interchangeable among distant species, it might be reasonable to suggest a function for A179L in insect cells similar to that displayed in mammalian cells. However, the bcl-2 homolog gene function in insect cells still remains controversial. Alnemri et al. (3) found that overexpression of human bcl-2 increased survival of baculovirus-infected Sf9 cells by prevention of apoptosis. Since the gene encoding p21 is a bcl-2 homolog (1, 5, 37), it seems likely that both genes act in similar apoptosis pathways. Nevertheless, it was reported that expression of bcl-2 or the adenovirus gene E1B-19K did not rescue the wild-type phenotypes of baculoviruses lacking the p35 gene (8, 11), so it was postulated that early apoptosis induction prevented by p35 expression could be mediated by bcl-2-independent mechanisms in Sf9 cells. Baculovirus infection could then trigger two different apoptosis mechanisms, an early apoptosis blocked by p35 but not by bcl-2 or its homologs, such as p21, and a late apoptosis induction in which p21/bcl-2 might be functional.
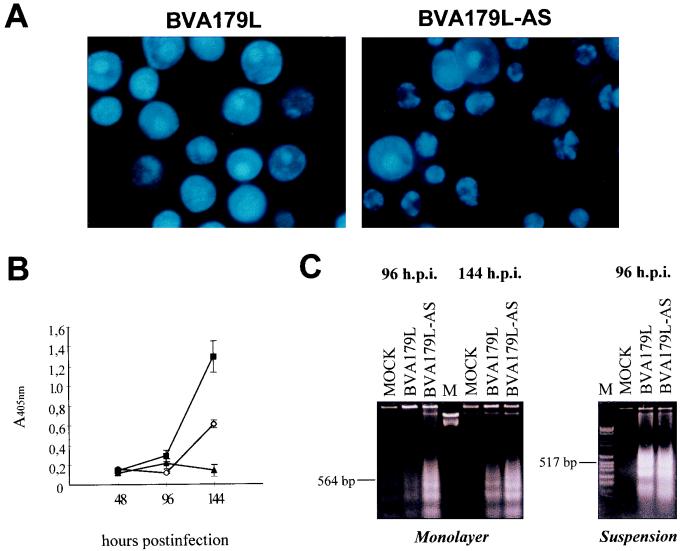
The chromatin fragmentation of baculovirus-infected cells by different methods was then analyzed (Fig. 3A to C). First we carried out a comparative Hoechst 33258 staining of infected cells in monolayer at different hours postinfection with BVA179L or BVA179L-AS viruses (Fig. 3A). Cells were methanol fixed for 10 min before incubation with 10 μg of Hoechst dye per ml in phosphate-buffered saline for 30 min at room temperature. BVA179L-AS-virus-infected cells exhibited a chromatin fragmentation pattern characteristic of apoptosis (Fig. 3A, right) which increased in a time-dependent fashion (data not shown). In contrast, at 144 h p.i. Sf9 cells infected with the BVA179L virus presented very few figures of apoptosis (Fig. 3A, left). This result indicated a correlation between cell viability and occurrence of chromatin fragmentation, which was confirmed by DNA laddering analysis (Fig. 3C) in 1.6% agarose gels (5). These experiments confirmed the lack of apoptosis prevention by p21 under nonadherent cell culture growth conditions (Fig. 3C, right).
FIG. 3.
DNA fragmentation in infected Sf9 cells (MOI of 10). (A) Hoechst 33258 staining of BVA179L- or BVA179L-AS-infected Sf9 cells grown in plates at 144 h p.i. Original magnification, ×400. (B) ELISA quantitation of the DNA linked to histone proteins in the cytoplasmic fraction of apoptotic Sf9 cells infected in monolayer at different time points. Squares: BVA179L-AS-infected cells; circles, BVA179L-infected cells; triangles, mock-infected cells. Means and standard errors were calculated from three independent experiments. (C) Agarose gel electrophoresis of the internucleosomal DNA laddering detected in infected or mock-infected Sf9 cells in monolayer cultures at 96 and 144 h p.i. (left) or in suspension cultures at 96 h p.i. (right). M, molecular size markers.
Quantitation of histone-associated DNA fragments released to the cytoplasm was carried out by a specific enzyme-linked immunosorbent assay (ELISA) (Boehringer Mannheim) (5). The results obtained after this analysis clearly indicated that expression of the ASFV gene A179L prevented the onset of apoptosis induced by baculovirus infection of cells cultured in monolayer (Fig. 3B). In contrast, at 144 h p.i. BVA179L-AS-virus-infected cells (Fig. 3B) yielded higher apoptosis rates than cells expressing p21.
The above results strongly suggest that the antiapoptotic function of p21 is dependent on cell attachment, because in suspended cells, A179L expression was unable to prevent baculovirus-induced apoptosis. Cell attachment is important for many cell functions. In fact, most types of normal cells require extracellular matrix attachment to respond to growth factor stimulation and other signals controlling cell proliferation or survival. When detached from their matrix, some cells undergo apoptosis (7, 15, 19, 26).
Effect of cell attachment on p21 function.
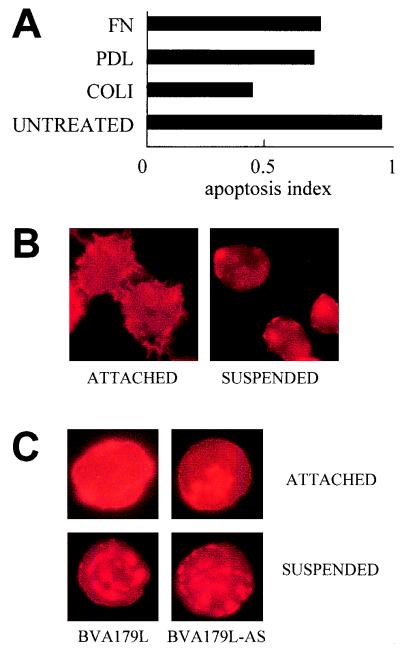
In an attempt to confirm if the activity of p21 was dependent on cell attachment, we performed experiments by infecting Sf9 cells with recombinant baculoviruses under conditions that improved cell adhesion to the culture surface (Fig. 4A). Sf9 cells were cultured on extracellular matrix components, such as rat collagen type I and human fibronectin, or on poly-d-lysine-coated glass plates (Falcon). Then, cells were infected with BVA179L or BVA179L-AS viruses at a MOI of 10. Figure 4A shows the apoptosis indices (AIs) of those cultures measured by anti-histone quantitative ELISA (inverse correlation). In BVA179L-infected cultures, apoptosis rate reduction was detected with all substrates that facilitated cell attachment by any pathway. Either extracellular matrix components (collagen type I and fibronectin) that support integrin-mediated cell adhesion or a substrate that mediates adhesion by nonspecific interactions (poly-d-lysine) enhanced apoptosis protection with respect to apoptosis of cells grown on uncoated nonadherent glass surfaces (AI of ≤1). Interestingly, infections performed on cells on a collagen type I matrix yielded more differences in apoptosis inhibition by p21 (AI of ≤0.5) with respect to apoptosis of cells on untreated surfaces (Fig. 4A). This could be related to the fact that Sf9 cells grown on this type of surface also had increased cell-to-cell interactions (not shown).
FIG. 4.
(A) Apoptosis induction of Sf9 cells (expressing p21) expressed as the ratio to that in BVA179L-AS-infected cells grown on different substrates to improve adhesiveness, measured by ELISA. The AI is given by the quotient (BVA179L-infected cells/BVA179L-AS-infected cells) of mean absorbance values at 405 nm from two replicate experiments at 96 h p.i. FN, human fibronectin; PDL, poly-d-lysine; COLI, rat tail collagen type I; UNTREATED, BVA179L-infected cells. (B) Actin cytoskeleton organization in adherent insect cells (left). Cells cultured in suspended growth conditions presented actin focalization (right). Cellular F-actin was stained with phalloidin-tetramethyl rhodamine isothiocyanate. (C) Characteristic individual infected Sf9 cells cultured in monolayer (upper) or in suspension (lower). Actin clumped in coarse fragments showed intense red staining. Original magnification, ×600 (panels B and C).
Cell attachment to underlying extracellular matrix is mediated by specialized membrane proteins called integrins, which interact with determined extracellular matrix components (6, 22). Integrin-mediated cell adhesion initiates a cascade of events that allow the transduction of survival signals that can block programmed cell death (13, 25). Induction of this survival pathway includes the upregulation of antiapoptotic proteins such as bcl-2 family members (16, 27, 42, 43). Also, cell-to-cell interaction inversely correlates with apoptosis associated with bcl-2 protein expression (4); in our results, aggregation of Sf9 cells plated on collagen I matrix resulted in enhanced survival. Apoptosis inhibition by p21 was also obtained with a nonspecific adhesive, such as poly-d-lysine, a synthetic compound altering surface charge, that increases cell adhesion not mediated by integrins. Our results indicate that cell attachment alone is sufficient to allow for the antiapoptotic activity of p21 in Sf9 cells infected by baculovirus. In fact, recent findings focus on cell shape changes and cytoskeleton integrity as supportive of rescue from apoptosis (10, 38).
Cytoskeleton organization in insect cells during infection with baculovirus in presence of p21.
Since conditions of cell anchorage may modify cytoskeleton organization, we analyzed the actin cytoskeleton of insect cells cultured under adherent and suspended growth conditions (Fig. 4). Sf9 cells were grown either directly on 96-well multiwell plates or in spinner flasks. Cells from suspension cultures were removed and allowed to sediment on glass slides. In both cases, cells were fixed with 4% paraformaldehyde, permeabilized in 0.1% Triton X-100 in phosphate-buffered saline, and stained for 30 min with 1:300 phalloidin-tetramethyl rhodamine isothiocyanate (Sigma), a marker for F-actin. Uninfected Sf9 cells attached to a surface displayed morphology different from that of cells that were grown in suspension. A profuse and fine surface microvillar network present in attached cells (Fig. 4B, left) was lacking in suspension. In contrast, suspended cells showed marked focalization of actin staining, indicating the occurrence of cytoskeleton reorganization (Fig. 4B, right). Cells in suspension infected with either BVA179L or BVA179L-AS baculoviruses showed irregular clumping of actin (Fig. 4C, lower). Dense actin staining was found concentrated in coarse fragments, and such elements were found in the cultures in proportions similar to that in the nonviable cell fraction (Fig. 2A and B). However, this pattern of actin clumping was not found in attached cells infected with BVA179L (Fig. 4C, upper left). Moreover, in monolayer, the overall intensity of actin staining decreased in a time-dependent manner in cells infected with BVA179L-AS, but it was maintained longer in infected cells expressing p21 (not shown). This result suggests that the expression of p21 in BVA179L-infected cells could contribute to the preservation of the cellular actin cytoskeleton at late postinfection times.
Cell adhesion to underlying extracellular matrix is mediated by sites of tight adhesion, called focal adhesions, that develop in cells in culture (12). Focal adhesions provide a structural link between the actin cytoskeleton and the extracellular matrix and are regions of signal transduction related to gene expression, growth control, and cell survival. It was recently suggested that cell attachment to matrix or integrin binding per se is not sufficient for maintaining cell viability and that cells need to undergo some minimal degree of shape changes to survive (10, 36, 38). It was recently reported that suspended endothelial cells acquired rounded shape, presented cytoskeleton disorganization, and underwent apoptosis (36). In contrast, when cells were grown on fibronectin or vitronectin, they became flattened, showed actin microfilament organization, and retained viability (36). Interleukin 4, which is able to activate neutrophil cytoskeletal rearrangements, produces a delay of apoptosis (20). Also, epithelial cells cultured on extracellular matrix components or laminin had a more well-developed actin cytoskeleton than cells cultured on noncoated dishes, which underwent apoptosis (2). The organization of the actin cytoskeleton in Sf9 cells grown attached to a surface was quite different from that displayed by cells grown in suspension. Consequently, actin organization and cell shape changes might provide the conditions for p21 protective function.
The in vivo relevance of changes in cell anchorage has been mainly focused on to date, either in the leukocyte movement out of vessels along endothelial cells in inflammation (23, 39) or in the loss of adherence of transformed cells in metastasis production (17, 18). Our findings suggest a role for cell shape and cytoskeleton status in viral diseases as well. Variations in those conditions in determined tissues or cell types in viral infections might explain differences in intracellular transduction of signals related to cell growth and survival. The cell anchorage dependence demonstrated by the ASFV bcl-2 homolog could have important consequences in the infection among the different cell compartments in vivo. Nevertheless, the physiological relevance of the biological effects of a protein overexpressed to the levels reported here should be an object of future studies. Based on these findings, it should be further analyzed if the absence of function of A179L apoptosis inhibitor in nonattached infected cells, such as circulating cells, might favor an early apoptosis induction and death of those cells. In fact, during in vivo infection with ASFV, only small percentages of infected monocytes are detected in peripheral blood (33). In contrast, tissue-fixed macrophages attached to extracellular matrix could be more prone to support the function of the bcl-2 viral homolog in vivo, leading to apoptosis inhibition in these cells, and could constitute a viral reservoir during persistent infection.
In conclusion, the above-presented data demonstrate that the product of the ASFV A179L gene, p21, is functional in insect cells and prevents late apoptosis after baculovirus infection. p21 increases the viability of infected cells in the context of a strong shutoff of protein synthesis and without modifying the baculovirus infection cycle. This suggests that p21 probably inhibits, in a way similar to human bcl-2, a highly conserved component of the apoptosis execution program.
Acknowledgments
This work was supported by Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria (INIA) grant SC97-066 and Programa Sectorial de Promoción General del Conocimiento PB96-0105.
REFERENCES
- 1.Afonso C L, Neilan J G, Kutish G F, Rock D L. An African swine fever virus Bcl-2 homolog, 5-HL, suppresses apoptotic cell death. J Virol. 1996;70:4858–4863. doi: 10.1128/jvi.70.7.4858-4863.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Aharoni D, Meiri I, Atzmon R, Vlodavsky I, Amsterdam A. Differential effect of components of the extracellular matrix on differentiation and apoptosis. Curr Biol. 1997;7:43–51. doi: 10.1016/s0960-9822(06)00026-1. [DOI] [PubMed] [Google Scholar]
- 3.Alnemri E S, Robertson N M, Fernandes T F, Croce C M, Litwack G. Overexpressed full-length human Bcl-2 extends the survival of baculovirus-infected Sf9 insect cells. Proc Natl Acad Sci USA. 1992;89:7295–7299. doi: 10.1073/pnas.89.16.7295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aoshiba K, Rennard S I, Spurzem J M. Cell-matrix and cell-cell interactions modulate apoptosis of bronchial epithelial cells. Am J Physiol. 1997;272:L28–L37. doi: 10.1152/ajplung.1997.272.1.L28. [DOI] [PubMed] [Google Scholar]
- 5.Brun A, Rivas C, Esteban M, Escribano J M, Alonso C. African swine fever virus gene A179L, a viral homologue of Bcl-2, protects cells from programmed cell death. Virology. 1996;225:227–230. doi: 10.1006/viro.1996.0592. [DOI] [PubMed] [Google Scholar]
- 6.Burridge K, Chrzanowska-Wodnicka M. Focal adhesions, contractility and signaling. Annu Rev Cell Dev Biol. 1996;12:463–519. doi: 10.1146/annurev.cellbio.12.1.463. [DOI] [PubMed] [Google Scholar]
- 7.Cardone M H, Salvesen G S, Widmann C, Johnson G, Frisch S M. The regulation of anoikis: MEKK-1 activation requires cleavage by caspases. Cell. 1997;90:315–323. doi: 10.1016/s0092-8674(00)80339-6. [DOI] [PubMed] [Google Scholar]
- 8.Cartier J L, Hershberger P A, Friesen P D. Suppression of apoptosis in insect cells stably transfected with baculovirus p35: dominant interference by N-terminal sequences p351-76. J Virol. 1994;68:7728–7737. doi: 10.1128/jvi.68.12.7728-7737.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chacón M R, Almazán F, Nogal M L, Viñuela E, Rodríguez J F. The African swine fever virus IAP homolog is a late structural polypeptide. Virology. 1996;214:670–674. doi: 10.1006/viro.1995.0083. [DOI] [PubMed] [Google Scholar]
- 10.Chen C S, Mrksich M, Huang S, Whitesides G M, Ingber D E. Geometric control of cell life and death. Science. 1997;276:1425–1428. doi: 10.1126/science.276.5317.1425. [DOI] [PubMed] [Google Scholar]
- 11.Clem R J, Miller L K. Control of programmed cell death by the baculovirus genes p35 and iap. Mol Cell Biol. 1994;14:5212–5222. doi: 10.1128/mcb.14.8.5212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Craig S W, Johnson R P. Assembly of focal adhesions: progress, paradigms and portents. Curr Opin Cell Biol. 1996;8:74–85. doi: 10.1016/s0955-0674(96)80051-2. [DOI] [PubMed] [Google Scholar]
- 13.Day M L, Foster R G, Day K C, Zhao X, Humphrey P, Swanson P, Postigo A A, Zhang S H, Dean D C. Cell anchorage regulates apoptosis through the retinoblastoma tumor suppressor/E2F pathway. J Biol Chem. 1997;272:8125–8128. doi: 10.1074/jbc.272.13.8125. [DOI] [PubMed] [Google Scholar]
- 14.Dixon L, Baylis S, Vydelingum S, Twigg S, Hammond J, Hingcamp C, Bristow C, Wilkinson P, Smith G. African swine fever virus genome content and variability. Arch Virol. 1993;7:185–199. doi: 10.1007/978-3-7091-9300-6_15. [DOI] [PubMed] [Google Scholar]
- 15.Frisch S M, Francis H. Disruption of epithelial cell-matrix interactions induces apoptosis. J Cell Biol. 1994;124:619–626. doi: 10.1083/jcb.124.4.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Frisch S M, Vuori K, Kelaita D, Sicks S. A role for Jun-N-terminal kinase in anoikis; suppression by bcl-2 and crmA. J Cell Biol. 1996;135:1377–1382. doi: 10.1083/jcb.135.5.1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Frisch S M, Vuori K, Ruoslahti E, Chang-Hui P Y. Control of adhesion-dependent cell survival by focal adhesion kinase. J Cell Biol. 1996;134:793–799. doi: 10.1083/jcb.134.3.793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Giancotti F, Ruoslahti E. Elevated levels of fibronectin receptor suppress the transformed phenotype of CHO cells. Cell. 1990;60:849–859. doi: 10.1016/0092-8674(90)90098-y. [DOI] [PubMed] [Google Scholar]
- 19.Ginis I, Faller D V. Protection from apoptosis in human neutrophils is determined by the surface of adhesion. Am J Physiol. 1997;272:C295–C309. doi: 10.1152/ajpcell.1997.272.1.C295. [DOI] [PubMed] [Google Scholar]
- 20.Girard D, Paquin R, Beaulieu A D. Responsiveness of human neutrophils to interleukin-4: induction of cytoskeletal rearrangements, de novo protein synthesis and delay of apoptosis. Biochem J. 1997;325:147–153. doi: 10.1042/bj3250147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gómez-Puertas P, Rodríguez F, Oviedo J M, Ramiro-Ibáñez F, Ruiz-Gonzalvo F, Alonso C, Escribano J M. Neutralizing antibodies to different proteins of African swine fever virus inhibit both virus attachment and internalization. J Virol. 1996;70:5689–5694. doi: 10.1128/jvi.70.8.5689-5694.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hynes R O. Integrins: versatility, modulation and signaling in cell adhesion. Cell. 1992;69:11–25. doi: 10.1016/0092-8674(92)90115-s. [DOI] [PubMed] [Google Scholar]
- 23.Iwaki K, Ohashi K, Ikeda M, Tsujioka K, Kajiya F, Kurimoto M. Decrease in the amount of focal adhesion kinase (p125FAK) in interleukin-1β-stimulated human umbilical vein endothelial cells by binding of human monocytic cell lines. J Biol Chem. 1997;272:20665–20670. doi: 10.1074/jbc.272.33.20665. [DOI] [PubMed] [Google Scholar]
- 24.Kleiboeker S B, Burrage T G, Scoles G A, Fish D, Rock D L. African swine fever virus infection in the argasid host, Ornithodoros porcinus porcinus. J Virol. 1998;72:1711–1724. doi: 10.1128/jvi.72.3.1711-1724.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kwaja A, Rodriguez-Viciana P, Wennstrom S, Warne P H, Downward J. Matrix adhesion and Ras transformation both activate a phosphoinositide 3′OH kinase and protein kinase B/Akt cellular survival pathway. EMBO J. 1997;16:2783–2793. doi: 10.1093/emboj/16.10.2783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meredith J E, Fazeli B, Schwartz M A. The extracellular matrix as a cell survival factor. Mol Biol Cell. 1993;4:953–961. doi: 10.1091/mbc.4.9.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Merlo G R, Cella N, Hynes N E. Apoptosis is accompanied by changes in Bcl-2 and Bax expression, induced by loss of attachment, and inhibited by specific extracellular matrix proteins in mammary epithelial cells. Cell Growth Differ. 1997;8:251–260. [PubMed] [Google Scholar]
- 28.Neilan J G, Lu Z, Alfonso C L, Kutish G F, Sussman M D, Rock D L. An African swine fever virus gene with similarity to the proto-oncogene bcl-2 and the Epstein-Barr virus gene BHRF1. J Virol. 1993;67:4391–4394. doi: 10.1128/jvi.67.7.4391-4394.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Neilan J G, Kutish G F, Zsak L, Burrage T G, Borca M V, Carrillo C, Rock D L. A BIR motif containing gene of ASFV, 4CL, is non-essential for growth in vitro and viral virulence. Virology. 1997;230:252–264. doi: 10.1006/viro.1997.8481. [DOI] [PubMed] [Google Scholar]
- 30.Oviedo J M, Rodriguez F, Gómez-Puertas P, Brun A, Gómez N, Alonso C, Escribano J M. High level expression of the major antigenic African swine fever virus proteins p54 and p30 in baculovirus and their potential use as diagnostic reagents. J Virol Methods. 1997;64:27–35. doi: 10.1016/s0166-0934(96)02140-4. [DOI] [PubMed] [Google Scholar]
- 31.Plowright W, Parker J, Pierce M. African swine fever virus in ticks (Ornitodoros moubata, Murray) collected from animal burrows in Tanzania. Nature. 1969;221:1071–1073. doi: 10.1038/2211071a0. [DOI] [PubMed] [Google Scholar]
- 32.Powell P P, Dixon L K, Parkhouse R M E. An IκB homolog encoded by African swine fever virus provides a novel mechanism for downregulation of proinflammatory cytokine responses in host macrophages. J Virol. 1996;70:8527–8533. doi: 10.1128/jvi.70.12.8527-8533.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ramiro-Ibáñez F, Escribano J M, Alonso C. Application of a monoclonal antibody recognizing protein p30 to detect African swine fever virus-infected cells in peripheral blood. J Virol Methods. 1995;55:339–345. doi: 10.1016/0166-0934(95)00071-1. [DOI] [PubMed] [Google Scholar]
- 34.Ramiro-Ibáñez F, Ortega A, Brun A, Escribano J M, Alonso C. Apoptosis: a mechanism of cell killing and lymphoid organ impairment during acute African swine fever virus infection. J Gen Virol. 1996;77:2209–2219. doi: 10.1099/0022-1317-77-9-2209. [DOI] [PubMed] [Google Scholar]
- 35.Ramiro-Ibáñez F, Ortega A, Ruiz-Gonzalvo F, Escribano J M, Alonso C. Modulation of immune cell populations and activation markers in the pathogenesis of African swine fever virus infection. Virus Res. 1997;47:31–40. doi: 10.1016/s0168-1702(96)01403-7. [DOI] [PubMed] [Google Scholar]
- 36.Re F, Zanetti A, Sironi M, Polentarutti N, Lanfrancone L, Dejana E, Colotta F. Inhibition of anchorage-dependent cell spreading triggers apoptosis in cultured human endothelial cells. J Cell Biol. 1994;127:537–546. doi: 10.1083/jcb.127.2.537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Revilla Y, Cebrian A, Baixeras E, Martínez C, Viñuela E, Salas M L. Inhibition of apoptosis by the African swine fever virus Bcl-2 homologue: role of the BH1 domain. Virology. 1997;228:400–404. doi: 10.1006/viro.1996.8395. [DOI] [PubMed] [Google Scholar]
- 38.Ruoslahti E. Stretching is good for a cell. Science. 1997;276:1345–1346. doi: 10.1126/science.276.5317.1345. [DOI] [PubMed] [Google Scholar]
- 39.Steeber D A, Engel P, Miller A S, Sheetz M P, Tedder T F. Ligation of L-selectin through conserved regions within the lectin domain activates signal transduction pathways and integrin function in human, mouse and rat leukocytes. J Immunol. 1997;159:952–963. [PubMed] [Google Scholar]
- 40.Sussman M, Lu Z, Kutish G, Afonso C, Roberts P, Rock D. Identification of an African swine fever virus gene with similarity to a myeloid differentiation primary response gene and a neurovirulence-associated gene of herpes simplex virus. J Virol. 1992;66:5586–5589. doi: 10.1128/jvi.66.9.5586-5589.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wilkinson P J. African swine fever virus. In: Pensaert M B, editor. Virus infections of porcines. Amsterdam, The Netherlands: Elsevier Science Publishers B.V.; 1989. pp. 17–37. [Google Scholar]
- 42.Yang C, Li S W, Helminen H J, Khillan J S, Bao Y, Procop D J. Apoptosis of chondocytes in transgenic mice lacking collagen II. Exp Cell Res. 1997;235:370–373. doi: 10.1006/excr.1997.3692. [DOI] [PubMed] [Google Scholar]
- 43.Zhang Z, Vuori K, Reed J C, Ruoslahti E. The α5β1 integrin supports survival of cells on fibronectin and up-regulates bcl-2 expression. Proc Natl Acad Sci USA. 1995;92:6161–6165. doi: 10.1073/pnas.92.13.6161. [DOI] [PMC free article] [PubMed] [Google Scholar]