Abstract
The vif gene of human immunodeficiency virus type 1 (HIV-1) encodes a basic Mr 23,000 protein that is necessary for production of infectious virions by nonpermissive cells (human lymphocytes and macrophages) but not by permissive cells such as HeLa-CD4. It had been proposed that permissive cells may contain an unidentified factor that functions like the viral Vif protein. To test this hypothesis, we produced pseudotyped wild-type and vif-deleted HIV gpt virions (which contain the HIV-1 genome with the bacterial mycophenolic acid resistance gene gpt in place of the viral env gene) in permissive cells, and we used them to generate nonpermissive H9 leukemic T cells that express these proviruses. We then fused these H9 cells with permissive HeLa cells that express the HIV-1 envelope glycoprotein gp120-gp41, and we asked whether the heterokaryons would release infectious HIV gpt virions. The results clearly showed that the vif-deleted virions released by the heterokaryons were noninfectious whereas the wild-type virions were highly infectious. This strongly suggests that nonpermissive cells, the natural targets of HIV-1, contain a potent endogenous inhibitor of HIV-1 replication that is overcome by Vif.
The vif gene of human immunodeficiency virus type 1 (HIV-1) encodes a highly basic Mr 23,000 protein that collapses intermediate filaments (14), locates in both cytosolic and nuclear sites (11, 14), is present in small amounts in virions (16), and is highly conserved among lentiviruses (20). Vif protein is not essential for HIV-1 replication in permissive cells such as HeLa-CD4 or in semipermissive cells such as SupT1 (9, 24). However, it is necessary for production of infectious virions by cells that are natural targets for infection, including CD4-positive T lymphocytes and macrophages, and by the H9 line of leukemic T cells (2, 9, 28). Nonpermissive cells can be infected by vif mutant virus made in permissive cells, but the result is production of only weakly infectious virions that cannot spread in cultures. Recent studies have shown that the defectiveness of vif mutant virus made in nonpermissive cells is not caused by gross abnormalities in protein composition (4, 8) and cannot be overcome by assaying the virus in permissive cells or in nonpermissive cells that contain Vif protein (7). The defective virus enters target cells and begins reverse transcription, but the resulting proviral DNA is prematurely degraded (3, 7, 28). Thus, the defectiveness of vif mutant HIV-1 conditionally depends on the cellular source of the virus rather than on the cells used to analyze infectivity.
The above results strongly suggest that the Vif protein performs a critical function in cells that produce HIV-1 virions. However, little is known about this critical Vif function. It has been proposed that permissive cells may contain a cellular protein that functions like Vif, thereby enabling active replication of vif mutant HIV-1 (26, 27). Alternatively, nonpermissive cells might contain an inhibitor of HIV-1 replication that is counteracted by Vif. These distinct hypotheses have not previously been investigated.
We have addressed this issue by analyzing infectivities of wild-type and vif mutant virions produced by heterokaryons formed by fusion of nonpermissive H9 leukemic T cells with fully permissive HeLa cells. Presumably, if permissive cells contain a protein that functions like Vif, the vif mutant virions released from the heterokaryons would be infectious. Alternatively, if nonpermissive cells contain an inhibitor that is counteracted by Vif, the vif mutant virions made by the heterokaryons would be inactive.
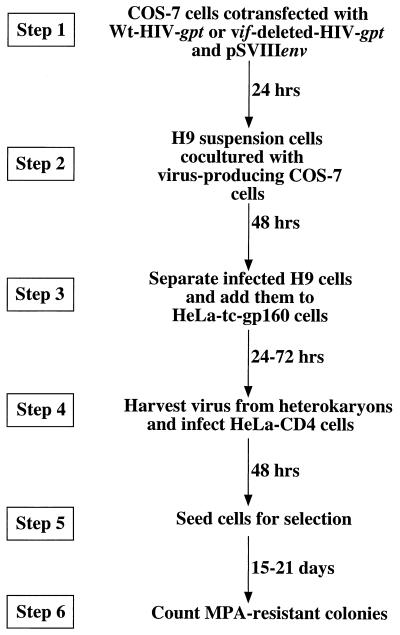
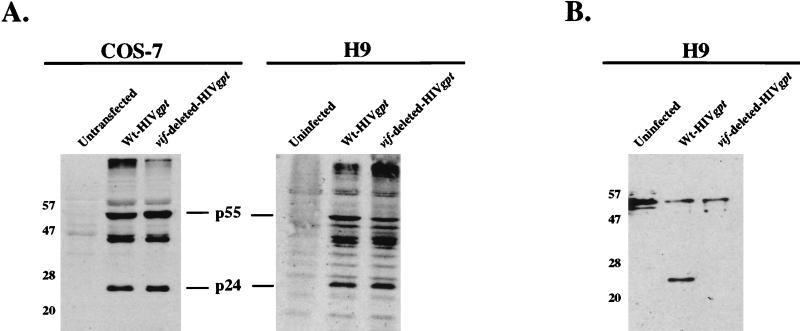
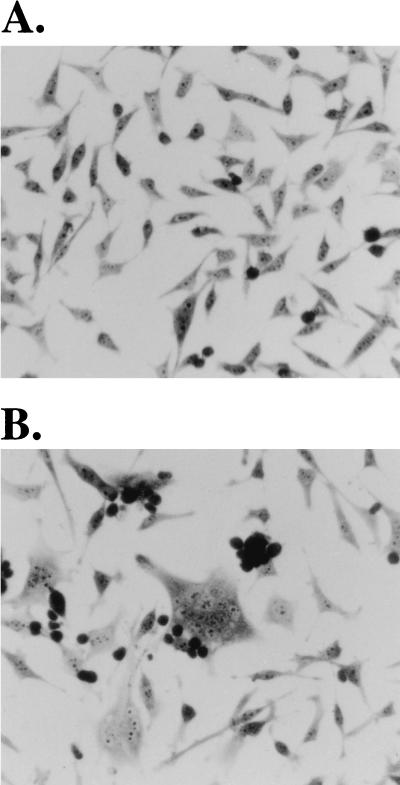
A schematic outline of this genetic complementation experiment is shown in Fig. 1. The strategy involves production of wild-type and vif-deleted HIV gpt virions. pHIV-gpt encodes the HIV-1 provirus HXBII, with the bacterial gpt gene for mycophenolic acid resistance substituted for the viral env gene (21). This plasmid was modified to construct pHIV-vif-deleted-gpt by digestion with SalI and NdeI restriction endonucleases (New England Biolabs, Beverly, Mass.), blunting with the Klenow fragment of DNA polymerase I (Life Technologies, Grand Island, N.Y.), and incubation with T4 DNA ligase (Life Technologies) to allow self ligation. This deleted a large portion of the vif gene and also removed a portion of the vpr gene. However, since the nonessential accessory genes vpr, vpu, and nef are all defective in the HXBII molecular clone of HIV-1 (with vpr having a mutation in the initiation codon) (19), this deletion did not have any effect on virus production (see below). Step 1 involved cotransfection of fully permissive COS-7 cells with pSVIII-env, encoding HXBII envelope, plus either pHIV-gpt or pHIV-vif-deleted-gpt by the DEAE-dextran with chloroquine transfection protocol (1). COS-7 cells were propagated as described previously (22). As shown by Western blotting, the transfected COS-7 cells expressed HIV-1 Gag proteins to equal extents, indicating that deletion of vif did not inhibit expression of the HIV gpt provirus (see Fig. 2). Moreover, these transfected COS-7 cells released equal titers of the wild-type and vif-deleted HIV gpt viruses. For example, in a representative experiment when 4 ml of cell-free media from wild-type and vif-deleted HIV gpt-transfected COS-7 cells was used to infect HeLa-CD4 cells, we obtained titers of 3,250 and 3,050 colonies per 100-mm-diameter culture dish for wild-type and vif-deleted HIV gpt, respectively. Therefore, COS-7 cells are fully permissive. In step 2, the virus-producing monolayers of adherent COS-7 cells were cocultured with a suspension of nonpermissive H9 T cells; 48 h later, the newly infected H9 cells were removed into fresh cultures. The resulting suspensions of H9 cells appeared morphologically homogeneous and did not produce any infectious HIV gpt virions, indicating that they were not significantly contaminated with the virus-producing COS-7 cells. Other control studies substantiated this conclusion (results not shown). As illustrated by Western blotting (Fig. 2A), the H9 cells that had been infected with these wild-type and vif-deleted HIV gpt virions reproducibly expressed similar amounts of the HIV-1 p55 and p24gag core proteins. This suggested that the infections in step 2 occurred with similar efficiencies and that processing of the core p55gag polyproteins in nonpermissive H9 cells was not significantly altered by the vif deletion. As also shown by Western blotting (Fig. 2B), the Mr 23,000 Vif protein was present in H9 cells that had been infected with wild-type HIV gpt virus but was absent from cells that had been infected with the vif-deleted HIV gpt virus. In step 3, the infected H9 cells were mixed with a clone of tetracycline-inducible HeLa-gp160 cells that expressed the HIV-1 IIIB env gene. These cells were generated by transfecting HeLa-tTA (12) cells with pUHD10-3(gp160/rev) by the calcium phosphate transfection method (1). pUHD10-3(gp160/rev) was constructed by cloning an XbaI 2.9-kbp gp160/rev fragment from pGCneo/gp160-rev (21a) into the same site of the tetracycline-responsive plasmid pUHD10-3. Figure 3 presents evidence that our clone of HeLa-tc-gp160 cells expressed abundant gp120-gp41 Env glycoproteins when they were induced by removal of tetracycline from the medium. We also used the methods described above for COS-7 cells and confirmed that this clone of HeLa-tc-gp160 cells was fully permissive for HIV-1 production. Moreover, the induced cells formed abundant syncytia when they were cocultured in step 3 with the infected H9 cells (Fig. 4). Staining of the rinsed monolayers after this coculturing of HeLa-tc-gp160 cells with the suspension of CD4-positive H9 HIV gpt or H9 vif-deleted HIV gpt cells showed equal numbers of syncytia. Thus, in one experiment these two monolayers contained 29% ± 3% and 27% ± 4% of the total nuclei in syncytia, respectively. Because uninfected H9 cells could also form syncytia with induced HeLa-tc-gp160 cells, we isolated stable populations of H9 HIV gpt and H9 vif-deleted HIV gpt cells by selection with mycophenolic acid. In control experiments, we found that these selected cells and the control uninfected H9 cells all formed syncytia with HeLa-tc-gp160 cells to equal extents. Moreover, the H9 cells that stably expressed the HIV gpt proviruses contained the same quantities of cell surface CD4 as the uninfected H9 cells, as determined by binding a monoclonal antibody to CD4 followed by 125I-protein A (13). Consequently, there was no effect of vif expression on cellular quantities of CD4 or on the ability of H9 cells to fuse with the induced HeLa-tc-gp160 cells. Because vpu and nef genes can down-modulate CD4 expression (5), their absence from the HXBII-derived pHIV-gpt provirus (19) was presumably a positive factor in enhancing syncytium formation in step 3. It is noteworthy that many of the syncytia contained giant nuclei or nuclei of widely divergent sizes. It is believed that these are generated after heterokaryons proceed through aberrant mitoses and the nuclear membranes then reform around groups of intermixed chromosomes (18). This morphological feature suggests that the nuclear contents within the syncytia had substantially intermixed by 24 to 48 h after initiation of step 3.
FIG. 1.
Schematic diagram of the vif complementation assay. In step 1, COS-7 cells at 80% confluency in a 100-mm culture dish were cotransfected with 5 μg of wild-type (Wt) pHIV-gpt or pHIVvif-deleted-gpt and 5 μg of pSVIIIenv. In step 2, suspensions of nonpermissive H9 leukemic T cells were added to the virus-producing COS-7 monolayers for 48 h. In step 3, the H9 cells were separated from the monolayer and cocultured with induced HeLa-tc-gp160 cells. This resulted in spontaneous cell fusion to produce heterokaryons, and virus was harvested from the culture media after 24, 48, and 72 h. In step 4, virus was filtered (0.45-μm-pore-size filters) and then used to infect HeLa-CD4 (H1-J clone) cells. In step 5, infected HeLa-CD4 cells were seeded in a 100-mm culture dish and selected with medium containing mycophenolic acid (MPA) (40 μg/ml). Mycophenolic acid-resistant colonies were fixed, stained, and counted after 15 to 21 days of selection (22).
FIG. 2.
Western immunoblot analyses of viral protein production in transfected COS-7 and infected H9 cells. H9 cells were cocultured with transfected virus-producing COS-7 cells as described in the legend to Fig. 1. At 72 h posttransfection of COS-7 cells (5 × 106 cells per 100-mm culture dish) and 48 h postinfection of H9 cells (1 × 106 cells), extracts of control and transfected or infected cells were obtained by washing the cells in phosphate-buffered saline (Life Technologies), followed by cell lysis in sodium dodecyl sulfate-polyacrylamide gel electrophoresis sample buffer (62.5 mM Tris-HCl [pH 6.8], 10% glycerol, 2% sodium dodecyl sulfate, 0.1% bromophenol blue, 10% 2-mercaptoethanol). The samples were then boiled, and equal amounts were loaded onto 10% polyacrylamide gels in the presence of 0.1% sodium dodecyl sulfate and subjected to electrophoresis. The proteins were then electrotransferred to nitrocellulose membranes and used for immunoblotting (21). Viral proteins were detected by incubating the membranes with HIV-IG antiserum (A) (obtained through the National Institutes of Health [NIH] AIDS Research and Reference Reagent Program, donated by Alfred Prince) or with HIV-1HXB2 Vif antiserum (B) (obtained through the NIH AIDS Research and Reference Reagent Program, donated by Dana Gabuzda) at a 1:1,000 dilution in 5% milk–0.1% Tween 20–Tris-buffered saline (Bio-Rad Laboratories, Hercules, Calif.), followed by protein A-conjugated horseradish peroxidase (HRP) at a 1:10,000 dilution (Bio-Rad). Antibody binding was then detected with a phototope-HRP Western blot detection kit (New England Biolabs). Mrs are indicated on the left, in thousands. Wt, wild type.
FIG. 3.
Western immunoblot analysis showing induced expression of gp160 upon removal of tetracycline (TC) from HeLa-tc-gp160 cells. HeLa-tc-gp160 cells were seeded in a 25-cm2 tissue culture flask and grown in the absence or presence of tetracycline (0.5 μg/ml; Sigma). Total cell extracts were collected 48 h later. Protein concentrations were measured by the Bio-Rad Bradford assay, and 20 μg of protein was analyzed by electrophoresis and immunoblotting, as described in the legend to Fig. 2. The blot was developed with a 1:1,000 dilution of sheep anti-gp120 antiserum (obtained through the NIH AIDS Research and Reference Reagent Program, donated by Michael Phelan), followed by protein G-conjugated horseradish peroxidase at a 1:5,000 dilution (Bio-Rad) and detection as described in the legend to Fig. 2.
FIG. 4.
Formation of syncytia by coculturing of HeLa-tc-gp160 and infected H9-42 (vif-deleted HIV gpt) cells. (A) HeLa-tc-gp160 cells grown in the presence of tetracycline do not synthesize gp160 or fuse with H9-42 cells. HeLa-tc-gp160 cells (3 × 105) were seeded in a 25-cm2 tissue culture flask and grown for 2 days in medium containing 0.5 μg of tetracycline (Sigma) per ml before H9 cells (4 × 106) were added. The cocultures were then grown for 48 h. Cells were observed under light microscopy after removal of the medium and unabsorbed cells, rinsing of adherent cells with phosphate-buffered saline (Life Technologies), fixing of cells with cold 100% methanol, and staining of cells with 0.1% toluidene blue in 30% ethanol. (B) H9 cells infected with vif-deleted HIV gpt were added to HeLa-tc-gp160 cells that were grown in the absence of tetracycline. Heterokaryons were allowed to form for 48 h. The cells were then fixed and stained as described for panel A. The small round darkly stained cells in both panels are H9 cells that adhered to the monolayers and were not removed by rinsing. Indistinguishable results were obtained with H9 cells that expressed wild-type HIV gpt (see text). Magnification, ×800.
In steps 4 through 6, virus produced by the heterokaryons was used to infect HeLa-CD4 cells (clone H1-J) (13) that were pretreated with Polybrene (8 μg/ml; Sigma, St. Louis, Mo.) for 30 min at 37°C. The infected cells were seeded 48 h later in the presence of 40 μg of mycophenolic acid per ml, and the resistant colonies were fixed and stained 15 to 21 days later. We have obtained identical results in nine independent experiments, including the four representative studies summarized in Table 1. In all cases, the heterokaryons that contained the wild-type HIV gpt provirus produced much more infectious virus than the heterokaryons that contained the vif-deleted HIV gpt provirus. Indeed, the latter titers were zero in several experiments, and the average titers of wild-type virus in our nine independent experiments were approximately 20 times higher than the average titers of vif-deleted virus. This basic result was obtained regardless of whether the infectious virions were harvested at 24, 48, or 72 h after mixing of the HIV gpt-expressing H9 cells with the HeLa-tc-gp160 cells.
TABLE 1.
Differences between the infectivities of wild-type and vif-deleted HIV gpt virions produced from heterokaryons assayed on HeLa-CD4 cells
| Expta | Time of virus harvest (h)b | HIV gpt infection (no. of colonies/dish)c
|
|
|---|---|---|---|
| Wild type | vif deleted | ||
| 1 | 24 | 20 | 0 |
| 48 | 13 | 0 | |
| 72 | 4 | 0 | |
| 2 | 24 | 36 | 7 |
| 48 | 10 | 0 | |
| 72 | 2 | 0 | |
| 3 | 24 | 41 | 7 |
| 48 | 16 | 3 | |
| 72 | 10 | 3 | |
| 4 | 48 | 110 | 0 |
| 48 | 39 | 0 | |
| 48 | 34 | 0 | |
In experiments 1, 2, and 3, H9-42 cells that expressed high levels of CD4 were used for coculturing with HeLa-tc-gp160 cells. H9-42 cells were made by transducing H9 cells with pSFF-CD4 retroviral vectors, as described previously (13), and were generously donated by Emily J. Platt.
Heterokaryons were formed by coculturing H9 cells with HeLa-tc-gp160 cells as described in the legend to Fig. 1. Virus harvested at 24, 48, and 72 h after coculturing of cells was used to infect HeLa-CD4 cells.
HeLa-CD4 cells were infected with wild-type or vif-deleted HIV gpt virus produced from heterokaryons. Infected cells were selected with medium containing mycophenolic acid, and selected colonies were counted 15 to 21 days later.
We also attempted to perform this genetic complementation study in the reverse manner by expressing the HIV gpt provirus in HeLa-CD4 cells and the HXB2 env gene in H9 cells. Unfortunately, the H9 cells could be transfected only inefficiently, and we were unable to obtain significant numbers of syncytia in step 3 or significant amounts of rescued infectious virus in step 4. Even if more efficient transfection of H9 cells could be achieved, it is likely that the resulting cells with gp120-gp41 complexes would fuse not only with HeLa-CD4 cells but also with other CD4-positive H9 cells in cultures. Apoptotic changes within the transfected H9 cultures would also have been expected (17). For these reasons, we concluded that the simple inverse protocol was not technically feasible.
The simplest interpretation of these results is that nonpermissive H9 cells contain a potent inhibitor of infectious HIV-1 production and that this inhibitor is counteracted by the virus-encoded Vif protein. The alternative hypothesis, that permissive HeLa cells contain a permissivity factor that replaces the requirement for Vif, appears to be inconsistent with our results. Even if this putative factor were slowly acting or were incapable of repairing aberrant HIV-1 components that had been synthesized in H9 cells prior to heterokaryon formation, we would have expected to see a relatively enhanced titer of vif-deleted HIV gpt virus by 48 to 72 h after initiation of heterokaryon formation. However, the heterokaryons that contained the vif-deleted HIV gpt provirus did not slowly begin to produce infectious virus. Alternatively, it is conceivable that a putative permissivity factor of HeLa cells might become inactivated in heterokaryons. However, this seems very unlikely, because its function was not observed even at the initial stages of heterokaryon formation (Table 1).
Although the heterokaryons that express wild-type HIV gpt release many more infectious virions than the heterokaryons that express vif-deleted HIV gpt, the titers in these experiments were also quite low (Table 1). Indeed, these titers were generally only approximately 1 to 2% as high as the titers released in step 1 by the initially transfected COS-7 cells. This quantitative difference is reasonable in part because COS-7 cells express transfected plasmids that have the simian virus 40 origin very efficiently. Moreover, only small fractions of the H9 nuclei appeared to enter the heterokaryons in step 3, and the latter may produce virions inefficiently. In addition, the H9 and HeLa-tc-gp160 cells aggregated in the cocultures, and many aggregates lifted into the culture medium. It has been reported that CD4-positive lymphoid cells undergo degenerative changes including apoptosis in cultures with cells that express HIV-1 envelope glycoprotein (17). These morphological changes occurred equally in the cocultures that contained the wild-type and vif-deleted HIV gpt proviruses. For the same reasons, however, even trace amounts of H9 cell contamination with virus or transfected COS-7 cells in step 3 might distort the eventual results. Because COS-7 cells are fully permissive, as documented above, this factor would have contributed equally to the titers of the wild-type and vif-deleted viruses and could not have reproducibly given the results that we observed.
Based on these considerations, we conclude that nonpermissive human T lymphocytes contain an endogenous and potent inhibitor of HIV-1 production that is overcome by the virus-encoded Vif protein, and we infer that this inhibitor may also occur in other nonpermissive cells, including macrophages. It is ironic that this inhibitor is apparently present only in these natural targets for HIV-1 infection and that it is absent in other human cells. Thus, we propose that CD4-positive T lymphocytes and macrophages may have an endogenous intracellular capacity to block HIV-1 replication and to cure the disease but that this potent inhibitor is held in check by Vif. This conclusion is consistent with a recent report that the Vif proteins of HIV-1 and simian immunodeficiency virus from African green monkeys may be active only in CD4-positive T lymphocytes from the respective species (25). Moreover, these Vif proteins act in a cell-specific rather than in a virus-specific manner. These results imply that Vif neutralizes the inhibitory activity of a cellular factor that occurs in lymphocytes, in agreement with our results. Several viral proteins function to thwart the host immune system (6, 10, 23), to block apoptosis, or to extend cellular lifespans (15), but we are unaware of another viral protein that blocks an intracellular inhibitor of infectious virus production. Further work will be needed to determine whether this inhibitor damages the virions during assembly in producer cells or whether it is incorporated into the virions to block their infectivity in target cells. We are currently attempting to identify an endogenous inhibitor in nonpermissive human cells. The implication that Vif counteracts a potent inhibitor of HIV-1 replication suggests that it would be an exceptionally promising target for drug development.
Acknowledgments
This research was supported by NIH grant CA67358. N.M. is partially supported by an NIH predoctoral fellowship in molecular hematology and oncology (T32HL07781).
We thank Hermann Bujard (Universitat Heidelberg, Heidelberg, Germany) for the generous gift of pUHD10-3. We are especially grateful to our colleague Emily Platt for generously donating the H9-42 cells that were used in several experiments, and for providing advice for the construction of vif-deleted pHIV-gpt, and to Susan Kozak, Shawn Kuhmann, Ali Nouri, David Johnson, and Chetankumar Tailor for encouragement and helpful suggestions.
REFERENCES
- 1.Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. Current protocols in molecular biology. New York, N.Y: John Wiley & Sons, Inc.; 1994. [Google Scholar]
- 2.Blanc D, Patience C, Schulz T F, Weiss R, Spire B. Transcomplementation of VIF− HIV-1 mutants in CEM cells suggests that VIF affects late steps of the viral life cycle. Virology. 1993;193:186–192. doi: 10.1006/viro.1993.1114. [DOI] [PubMed] [Google Scholar]
- 3.Borman A M, Quillent C, Charneau P, Dauguet C, Clavel F. Human immunodeficiency virus type 1 Vif− mutant particles from restrictive cells: role of Vif in correct particle assembly and infectivity. J Virol. 1995;69:2058–2067. doi: 10.1128/jvi.69.4.2058-2067.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bouyac M, Rey F, Nascimbeni M, Courcoul M, Sire J, Blanc D, Clavel F, Vigne R, Spire B. Phenotypically Vif− human immunodeficiency virus type 1 is produced by chronically infected restrictive cells. J Virol. 1997;71:2473–2477. doi: 10.1128/jvi.71.3.2473-2477.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen B K, Gandhi R T, Baltimore D. CD4 down-modulation during infection of human T cells with human immunodeficiency virus type 1 involves independent activities of vpu, env, and nef. J Virol. 1996;70:6044–6053. doi: 10.1128/jvi.70.9.6044-6053.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Collins K L, Chen B K, Kalams S A, Walker B D, Baltimore D. HIV-1 Nef protein protects infected primary cells against killing by cytotoxic T lymphocytes. Nature. 1998;391:397–401. doi: 10.1038/34929. [DOI] [PubMed] [Google Scholar]
- 7.Courcoul M, Patience C, Rey F, Blanc D, Harmache A, Sire J, Vigne R, Spire B. Peripheral blood mononuclear cells produce normal amounts of defective Vif− human immunodeficiency virus type 1 particles which are restricted for the preretrotranscription steps. J Virol. 1995;69:2068–2074. doi: 10.1128/jvi.69.4.2068-2074.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fouchier R A, Simon J H, Jaffe A B, Malim M H. Human immunodeficiency virus type 1 Vif does not influence expression or virion incorporation of gag-, pol-, and env-encoded proteins. J Virol. 1996;70:8263–8269. doi: 10.1128/jvi.70.12.8263-8269.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gabuzda D H, Lawrence K, Langhoff E, Terwilliger E, Dorfman T, Haseltine W A, Sodroski J. Role of vif in replication of human immunodeficiency virus type 1 in CD4+ T lymphocytes. J Virol. 1992;66:6489–6495. doi: 10.1128/jvi.66.11.6489-6495.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Goldsmith K, Chen W, Johnson D C, Hendricks R L. Infected cell protein (ICP)47 enhances herpes simplex virus neurovirulence by blocking the CD8+ T cell response. J Exp Med. 1998;187:341–348. doi: 10.1084/jem.187.3.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Goncalves J, Jallepalli P, Gabuzda D H. Subcellular localization of the Vif protein of human immunodeficiency virus type 1. J Virol. 1994;68:704–712. doi: 10.1128/jvi.68.2.704-712.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gossen M, Freundlieb S, Bender G, Muller G, Hillen W, Bujard H. Transcriptional activation by tetracyclines in mammalian cells. Science. 1995;268:1766–1769. doi: 10.1126/science.7792603. [DOI] [PubMed] [Google Scholar]
- 13.Kabat D, Kozak S L, Wehrly K, Chesebro B. Differences in CD4 dependence for infectivity of laboratory-adapted and primary patient isolates of human immunodeficiency virus type 1. J Virol. 1994;68:2570–2577. doi: 10.1128/jvi.68.4.2570-2577.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Karczewski M K, Strebel K. Cytoskeleton association and virion incorporation of the human immunodeficiency virus type 1 Vif protein. J Virol. 1996;70:494–507. doi: 10.1128/jvi.70.1.494-507.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Klingelhutz A J, Foster S A, McDougall J K. Telomerase activation by the E6 gene product of human papillomavirus type 16. Nature. 1996;380:79–82. doi: 10.1038/380079a0. [DOI] [PubMed] [Google Scholar]
- 16.Liu H, Wu X, Newman M, Shaw G M, Hahn B H, Kappes J C. The Vif protein of human and simian immunodeficiency viruses is packaged into virions and associates with viral core structures. J Virol. 1995;69:7630–7638. doi: 10.1128/jvi.69.12.7630-7638.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Maldarelli F, Sato H, Berthold E, Orenstein J, Martin M A. Rapid induction of apoptosis by cell-to-cell transmission of human immunodeficiency virus type 1. J Virol. 1995;69:6457–6465. doi: 10.1128/jvi.69.10.6457-6465.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Manandhar G, Khodiakov A L, Onishchenko G E. The characteristics of the course of mitosis in polykaryons formed during cell fusion. Tsitologiya. 1991;33:39–47. [PubMed] [Google Scholar]
- 19.Mustafa F, Robinson H L. Context-dependent role of human immunodeficiency virus type 1 auxiliary genes in the establishment of chronic virus producers. J Virol. 1993;67:6909–6915. doi: 10.1128/jvi.67.11.6909-6915.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Oberste M S, Gonda M A. Conservation of amino-acid sequence motifs in lentivirus Vif proteins. Virus Genes. 1992;6:95–102. doi: 10.1007/BF01703760. [DOI] [PubMed] [Google Scholar]
- 21.Page K A, Landau N R, Littman D R. Construction and use of a human immunodeficiency virus vector for analysis of virus infectivity. J Virol. 1990;64:5270–5276. doi: 10.1128/jvi.64.11.5270-5276.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21a.Platt, E., and D. Kabat. Unpublished data.
- 22.Platt E J, Madani N, Kozak S L, Kabat D. Infectious properties of human immunodeficiency virus type 1 mutants with distinct affinities for the CD4 receptor. J Virol. 1997;71:883–890. doi: 10.1128/jvi.71.2.883-890.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ploegh H L. Viral strategies of immune evasion. Science. 1998;280:248–253. doi: 10.1126/science.280.5361.248. [DOI] [PubMed] [Google Scholar]
- 24.Sakai H, Shibata R, Sakuragi J, Sakuragi S, Kawamura M, Adachi A. Cell-dependent requirement of human immunodeficiency virus type 1 Vif protein for maturation of virus particles. J Virol. 1993;67:1663–1666. doi: 10.1128/jvi.67.3.1663-1666.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Simon J H, Miller D L, Fouchier R A, Soares M A, Peden K W, Malim M H. The regulation of primate immunodeficiency virus infectivity by Vif is cell species restricted: a role for Vif in determining virus host range and cross-species transmission. EMBO J. 1998;17:1259–1267. doi: 10.1093/emboj/17.5.1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Trono D. HIV accessory proteins: leading roles for the supporting cast. Cell. 1995;82:189–192. doi: 10.1016/0092-8674(95)90306-2. [DOI] [PubMed] [Google Scholar]
- 27.Volsky D J, Potash M J, Simm M, Sova P, Ma X Y, Chao W, Shahabuddin M. The human immunodeficiency virus type 1 vif gene: the road from an accessory to an essential role in human immunodeficiency virus type 1 replication. Curr Top Microbiol Immunol. 1995;193:157–168. doi: 10.1007/978-3-642-78929-8_9. [DOI] [PubMed] [Google Scholar]
- 28.von Schwedler U, Song J, Aiken C, Trono D. Vif is crucial for human immunodeficiency virus type 1 proviral DNA synthesis in infected cells. J Virol. 1993;67:4945–4955. doi: 10.1128/jvi.67.8.4945-4955.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]