Abstract
Background:
The correlation between systolic blood pressure (SBP) and mortality in hypertensive patients with different phenotypes of heart failure (HF) has not been adequately studied, and optimal blood pressure control targets remain controversial. To explore the link between SBP and prognosis in all or three ejection fraction (EF) phenotypes of HF patients with hypertension.
Methods:
We analyzed 1279 HF patients complicated by hypertension in a retrospective cohort. The SBP <130 mmHg group included 383 patients, and the SBP ≥130 mmHg group included 896 patients. The major end point was all-cause mortality.
Results:
Of the 1279 study patients, with a median age of 66.0 ± 12.0 years, 45.3% were female. The proportions of the three subtypes of heart failure complicated with hypertension (HFrEF, HEmrEF, and HFpEF) were 26.8%, 29.3%, and 43.9%, respectively. During the 1-year follow-up, 223 patients experienced all-cause death, and 133 experienced cardiovascular death. Restricted cubic splines showed that the risk of all-cause and cardiovascular death increased gradually as the SBP level decreased in patients with HFrEF and HFmrEF. Furthermore, the multivariate Cox proportional hazards model revealed that SBP <130 mmHg was also associated with an increased risk of all-cause death [hazard ratio (HR) 2.53, 95% confidence interval (CI) 1.23–5.20, P = 0.011] and cardiovascular death (HR 1.91, 95% CI 1.01–3.63, P = 0.047) in HFrEF patients. A trend toward increased risk was observed among HFmrEF patients, but it was not statistically significant. This trend was not observed in HFpEF patients.
Conclusion:
In HFrEF patients, SBP <130 mmHg was associated with an increased risk of all-cause and cardiovascular mortality. A trend toward increased risk was observed among HFmrEF patients, but not among HFpEF patients.
Keywords: heart failure, heart failure with mid-range or mildly reduced ejection fraction, heart failure with preserved ejection fraction, heart failure with reduced ejection fraction, mortality, systolic blood pressure
INTRODUCTION
Despite recent advances in heart failure (HF) care, it remains a major cause of mortality and disability in modern society due to population expansion, aging, and the rising frequency of comorbidities [1–6]. The Framingham Heart Study's 20-year follow-up data revealed that hypertension is the most prevalent attributable risk factor for HF [7–9]. In different age and gender groups, a higher BP correlated with a higher risk of heart failure [8,10]. Paradoxically, lower systolic blood pressure (SBP) levels may be connected with a higher risk of death in heart failure patients [11–16]. A study of the KorAHF registry showed that in heart failure patients, SBP <130 mmHg was associated with an increased risk of all-cause mortality [14]. However, the association between SBP level and mortality in persons with heart failure and hypertension remains unknown.
Heart failure has traditionally been classified into three phenotypes based on the assessment of the left ventricular ejection fraction (LVEF): HF with reduced ejection fraction (HFrEF), HF with mid-range or mildly reduced ejection fraction (HFmrEF), and HF with preserved ejection fraction (HFpEF) [17–19]. The correlation between SBP and mortality in hypertensive patients with different phenotypes of HF has not been adequately studied, and optimal blood pressure control targets remain controversial.
Northeast China has more hypertensive patients than other regions due to the high-salt diet and cold climate [20]. However, few studies on SBP levels in patients with HF and hypertension have been conducted in Northeast China [21–23]. Therefore, our study is based on patients registered in the Heart Failure Center of the Second Affiliated Hospital of Harbin Medical University (HMUSAH) to evaluate the association between SBP and prognosis in all or three types of HF patients with hypertension.
METHODS
Data source
The Heart Failure Center of the HMUSAH was established in May 2018 and is in northeast China. It is one of the first heart failure centers and the first demonstration unit of the Heart Failure Center in China. The heart failure center oversees screening and registering heart failure patients at our hospital, supervising diagnosis and treatment plans, establishing paper and electronic files, ensuring that patients are followed up regularly, and sending baseline and follow-up information about heart failure patients to the China Heart Failure Center website (https://www.chinahfc.org/).
Study participants
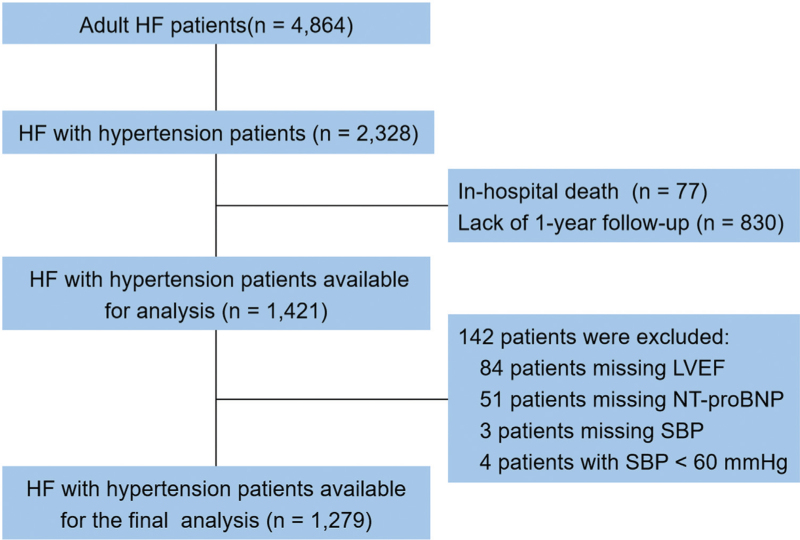
This is a retrospective observational study. From May 2018 to December 2019, 4864 heart failure patients were admitted for the first time to our hospital's heart failure center. Of these patients, 2328 had hypertension. After excluding patients who died before discharge and lacked 1-year follow-up information and then further excluding patients who lacked LVEF, N-terminal pro-B-type natriuretic peptide (NT-proBNP), SBP, and SBP <60 mmHg. The final analysis included 1279 patients with heart failure and hypertension. Figure 1 depicts the complete data integration process. The diagnosis of HF was based on the China Heart Failure Guidelines recommendation, which includes dyspnea, fatigue, or decreased activity tolerance; signs of fluid retention (pulmonary congestion and peripheral edema); echocardiography showing cardiac structure and/or abnormal function; and NT-proBNP >125 ng/l [24]. Office SBP ≥140 mmHg and/or diastolic blood pressure (DBP) ≥90 mmHg in the absence of antihypertensive medicines, self-reported physician-diagnosed hypertension, or current use of antihypertensive drugs were considered hypertension.
FIGURE 1.
Flow chart of study participants. Patients were from the heart failure center of the Second Affiliated Hospital of Harbin Medical University from May 2018 to December 2019. HF, heart failure; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal pro brain natriuretic peptide; SBP, systolic blood pressure.
Study outcomes
The major end point was all-cause mortality. Secondary end points were cardiovascular mortality, which was defined as death from heart failure, myocardial infarction, malignant arrhythmia, cardiac rupture, cardiogenic shock, and sudden death. We tracked endpoint events via phone or outpatient follow-up at 1 week, 1, 3 and 12 months after patients were discharged from the hospital. The events were reported by the patients or their close family members, and no decisions were made.
Data collection and study variables
Blood pressure was measured according to the recommendations of European Society of Hypertension guidelines [25]. Resting BP measurements were performed with the patients in the sitting position on at least two separate days at least 1 week apart. Caffeine (tea, coffee, carbonated beverages), eating, heavy physical activities, and smoking were prohibited for 30 min before measuring BP. The blood pressure measurement procedure was described to participants who rested for at least 5 min on a chair in the sitting position with arms supported. BP was measured 3 times while in the sitting position; first left arm, then right arm, then again left arm-the arithmetic mean was used for analyses.
The clinical research coordinator was responsible for collecting the basic information of the patients, including demographics, comorbidities, blood pressure, heart rate, NYHA class, echocardiographic indicators, laboratory test indicators at admission, current and previous cardiac surgery, and oral medication at discharge. Detailed indicators are recorded in Table 1. All clinical research coordinators were formally trained.
TABLE 1.
Baseline patient characteristics by admission systolic blood pressure
| SBP <130 mmHg | SBP ≥130 mmHg | ||
| Characteristic | (n = 383) | (n = 896) | P value |
| Age (years)a | 67 ± 11 | 66 ± 13 | 0.292 |
| Female (%)a | 163 (42.6) | 417 (46.5) | 0.212 |
| Smoker/former smoker (%)a | 145 (37.9) | 295 (32.9) | 0.102 |
| DBP (mmHg)a | 75.4 ± 12.48 | 93.57 ± 17.03 | <0.001 |
| Heart rate (bpm)a | 88 ± 23 | 89 ± 22 | 0.151 |
| Ultrasonic cardiogram | |||
| LVEF (%)a | 45.8 ± 11.9 | 48.2 ± 11.2 | 0.002 |
| LA (mm)a | 41.7 ± 7.2 | 41.9 ± 6.3 | 0.354 |
| LVED (mm)a | 53.8 ± 9.8 | 53.2 ± 8.5 | 0.717 |
| LVES (mm)a | 39.7 ± 11.9 | 38.3 ± 10.5 | 0.138 |
| IVS (mm)a | 10.8 ± 1.9 | 11.5 ± 2.2 | <0.001 |
| LVPW (mm)a | 10.4 ± 1.59 | 11.1 ± 1.8 | <0.001 |
| NYHA class (%)a | 0.569 | ||
| I | 1 (0.3) | 2 (0.2) | |
| II | 75 (19.6) | 187 (20.9) | |
| III | 170 (44.4) | 357 (39.8) | |
| IV | 137 (35.8) | 350 (39.1) | |
| Comorbidities (%) | |||
| CHD/MIa | 317 (82.8) | 730 (81.5) | 0.638 |
| Diabetes mellitusa | 144 (37.6) | 338 (37.7) | 0.966 |
| Cerebrovascular diseasea | 117 (30.5) | 231 (25.8) | 0.092 |
| Atrial fibrillationa | 94 (24.5) | 236 (26.3) | 0.547 |
| Valvular diseasea | 13 (3.4) | 30 (3.3) | 0.967 |
| Renal insufficiency | 62 (16.2) | 177 (19.8) | 0.155 |
| Laboratory | |||
| NT-proBNP (pg/ml)a | 3166 (1358–6709) | 3387 (1481–8526) | 0.130 |
| Hemoglobin (g/l)a | 131.7 ± 21.9 | 132.0 ± 23.8 | 0.923 |
| FBG (mmol/l)a | 6.9 ± 3.3 | 6.9 ± 3.1 | 0.970 |
| Cholesterol (mmol/l)a | 3.8 ± 1.1 | 4.2 ± 1.2 | <0.001 |
| Triglyceride (mmol/l)a | 1.35 (1–1.88) | 1.4 (1–1.9) | 0.442 |
| HDL (mmol/l)a | 1.0 ± 0.59 | 1.1 ± 0.3 | 0.011 |
| LDL (mmol/l)a | 2.3 ± 0.8 | 2.6 ± 0.9 | <0.001 |
| eGFR (ml/min/1.73 m2)a | 59.8 ± 22.1 | 58.6 ± 25.6 | 0.666 |
| Creatinine (μmol/l) | 100.5 (82–122.8) | 101 (80–136.5) | 0.588 |
| Sodium (mmol/l)a | 138.8 ± 4.6 | 139.0 ± 4.2 | 0.349 |
| Potassium (mmol/l)a | 4.1 ± 0.6 | 4.1 ± 0.6 | 0.516 |
| Surgical treatment (%) | |||
| PCI/CABGa | 141 (36.8) | 302 (33.7) | 0.314 |
| Medication (%) | |||
| ACEI/ARB/ARNIa | 269 (70.2) | 666 (74.3) | 0.149 |
| β receptor blockera | 246 (64.2) | 559 (62.4) | 0.575 |
| Spironolactonea | 278 (72.6) | 616 (68.8) | 0.193 |
| Diureticsa | 161 (42.0) | 390 (43.5) | 0.666 |
| Calcium channel blockersa | 54 (14.1) | 242 (27) | <0.001 |
| Nitratesa | 172 (44.9) | 386 (43.1) | 0.588 |
| Digoxina | 52 (13.6) | 114 (12.7) | 0.745 |
ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; ARNI, angiotensin receptor neprilysin inhibitor; CABG, coronary artery bypass grafting; CHD, coronary heart disease; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; FBG, fasting blood glucose; HDL, high density lipoprotein; IVS, inter ventricular septum; LA, left atrium; LDL, low density lipoprotein; LVED, left ventricular end-diastolic; LVEF, left ventricular ejection fraction; LVES, left ventricular end-systolic; LVPW, left ventricular posterior wall; MI, myocardial infarction; NT-proBNP, N-terminal pro brain natriuretic peptide; NYHA, New York Heart Association; PCI, percutaneous coronary intervention; SBP, systolic blood pressure.
Variables marked with an asterisk were included in univariate logistic regression.
Statistical analysis
According to admission SBP, patients were separated into two groups: the SBP <130 mmHg group as well as the SBP ≥130 mmHg group. The Wilcoxon rank-sum test was used to compare continuous variables between groups, and data are expressed as the mean ± standard deviation or medians [interquartile ranges (IQR)] as appropriate. The chi-square test was used to compare categorical variable data between groups, and data was expressed in counts (proportions).
Restricted cubic splines (RCS) were employed to assess the nonlinear correlation of SBP with all-cause and cardiovascular death. The Kaplan–Meier method was used to examine the two groups’ cumulative survival rates, and the log-rank test was used to compare group differences. The Poisson distribution was used to estimate the mortality rate as the number of deaths per 100 person-years. Univariate and multivariate Cox proportional hazards models were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for 1-year all-cause and cardiovascular mortality in the SBP <130 mmHg group compared with the SBP ≥130 mmHg group. All variables included in univariate analysis are marked with asterisks in Table 1, and P < 0.05 in univariate analysis are included in the multivariate model (Tables S1–S8, Supplemental Digital Content).
We then performed subgroup analyses based on age, sex, diabetes, estimated glomerular filtration rate (eGFR) level, use of angiotensin-converting enzyme inhibitor/angiotensin receptor blocker/angiotensin receptor-neprilysin inhibitor (ACEI/ARB/ARNI), use of beta-blockers, use of calcium channel blockers, and use of diuretics. We used multiplicative interactions to determine whether the association of SBP with outcome was influenced by these underlying determinants.
Two-tailed P values <0.05 were regarded as statistically significant. The Stata (version 17.1) and R language (R version 4.2.1) were used to conduct the statistical analysis.
RESULTS
Baseline characteristics
Of the 1279 HF patients whose cases were complicated by hypertension included in the final analysis, 45.3% were female, and the average age was 66.0 ± 12.0 years. The <130 mmHg group (n = 383) and the ≥130 mmHg group (n = 896). Table 1 summarizes the patient's baseline characteristics, which include previous disease history, cardiac function classification, cardiac ultrasound results, laboratory test results, and treatments. The two groups had no difference in age, sex, smoking history, or previous combined disease histories. Cardiac ultrasound showed that ejection fraction (EF), intact ventricular septum (IVS), and left ventricular posterior wall (LVPW) in the SBP ≥130 mmHg group were significantly higher than those in the SBP <130 mmHg group. In laboratory examinations, the levels of serum cholesterol, high-density lipoprotein (HDL), and low-density lipoprotein (LDL) were higher in the SBP ≥130 mmHg group than in the SBP <130 mmHg group, and no difference was detected in other results. In terms of drug treatment, the proportion of calcium channel blockers used was higher in the SBP ≥130 mmHg group than in the SBP <130 mmHg group, and other drugs did not differ between the two groups.
SBP and mortality
During the one-year follow-up period, 223 individuals died from all cause, and 133 died from cardiovascular causes. The association between SBP and all-cause or cardiovascular death is visually presented using unadjusted RCS (Figure S1, Supplemental Digital Content). When SBP was <130 mmHg, the risk of all-cause and cardiovascular mortality increased gradually as the SBP level decreased. The all-cause mortality rate was 23.6 (18.9–29.5) per 100 person-years in the SBP <130 mmHg group and 18.3 (15.5–21.5) per 100 person-years in the SBP ≥130 mmHg group. The cardiovascular mortality rate was 14.1 (10.6–18.8) per 100 person-years in the SBP <130 mmHg group and 10.9 (8.8–13.4) per 100 person-years in the SBP ≥130 mmHg group (Table 2).
TABLE 2.
Mortality rate per 100 person-years and 95% CIs according to SBP <130 mmHg and ≥130 mmHg groups
| SBP <130 mmHg | SBP ≥130 mmHg | |
| All-cause death | ||
| Total population | 23.6 (18.9–29.5) | 18.3 (15.5–21.5) |
| HFrEF | 28.4 (19.9–40.5) | 14.5 (10.1–20.8) |
| HFmrEF | 27.5 (18.8–40.3) | 20.6 (15.5–27.3) |
| HFpEF | 16.1 (10.5–24.6) | 18.9 (14.9–23.8) |
| Cardiovascular death | ||
| Total population | 14.1 (10.6–18.8) | 10.9 (8.8–13.4) |
| HFrEF | 18.9 (12.2–29.2) | 10.5 (6.9–16.0) |
| HFmrEF | 18.0 (11.2–28.8) | 14.4 (10.3–20.3) |
| HFpEF | 6.9 (3.6–13.1) | 8.9 (6.3–12.5) |
HFmrEF, heart failure with mid-range ejection fraction; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; SBP, systolic blood pressure.
Figure S2, Supplemental Digital Content shows the Kaplan–Meier curves for cumulative survival during the follow-up period by SBP group. The variables with P < 0.05 in the univariate Cox proportional hazards model of all-cause death and cardiovascular death are shown in Table S1, Supplemental Digital Content and Table S2, Supplemental Digital Content. In the multivariate model that included the above variables, the results showed that compared with the SBP ≥130 mmHg group, the risk of all-cause and cardiovascular mortality in the SBP <130 mmHg group tended to increase (HR 1.29, 95% CI 0.91–1.82, P = 0.147 and HR 1.28, 95% CI 0.87–1.88, P = 0.204, respectively) (Table 3).
TABLE 3.
Hazard ratios (95% CIs) of all-cause death and cardiovascular death according to <130 mmHg vs. SBP ≥130 mmHg
| Univariate analysis | Multivariate analysis | |||
| HR (95% CI) | P value | HR (95% CI) | P value | |
| All-cause death | ||||
| Total population | 1.29 (0.98–1.69) | 0.075 | 1.29 (0.91–1.82) | 0.147 |
| HFrEF | 2.01 (1.20–3.34) | 0.008 | 2.53 (1.23–5.20) | 0.011 |
| HFmrEF | 1.32 (0.82–2.14) | 0.252 | 1.60 (0.85–3.02) | 0.148 |
| HFpEF | 0.86 (0.53–1.40) | 0.534 | 0.97 (0.56–1.66) | 0.902 |
| Cardiovascular death | ||||
| Total population | 1.30 (0.91–1.85) | 0.156 | 1.28 (0.87–1.88) | 0.204 |
| HFrEF | 1.86 (1.01–3.44) | 0.046 | 1.91 (1.01–3.63) | 0.047 |
| HFmrEF | 1.24 (0.69–2.23) | 0.466 | 1.24 (0.60–2.57) | 0.564 |
| HFpEF | 0.78 (0.37–1.63) | 0.508 | 0.99 (0.46–2.14) | 0.983 |
HFmrEF, heart failure with mid-range ejection fraction; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; SBP, systolic blood pressure.
SBP level and mortality across the ejection fraction spectrum
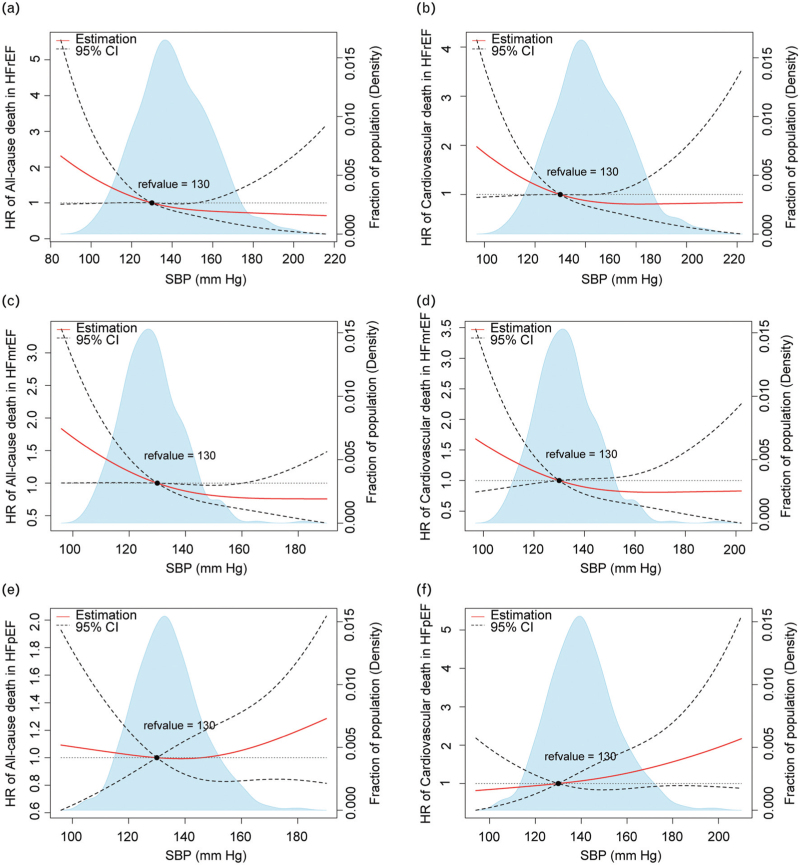
Of the 1279 individuals, the proportions of the three subtypes of heart failure complicated by hypertension (HFrEF, HEmrEF, and HFpEF) were 26.8%, 29.3%, and 43.9%, respectively. In HFrEF and HFmrEF patients, the RCS spline plot showed that when SBP was below 130 mmHg, the risk of all-cause death and cardiovascular death gradually increased as SBP decreased (Fig. 2a–d). However, this trend was not clear in patients with HFpEF (Figure 2e and f). The per 100 person-years grouped by SBP groups in patients with HFrEF, HFmrEF, and HFpEF are shown in Table 2.
FIGURE 2.
Restricted cubic spline fitting for the correlation between SBP levels and mortality across the spectrum of heart failure. HRs were evaluated by setting the SBP value = 130 mmHg as a reference. (a) All-cause mortality in HFrEF. (b) Cardiovascular mortality in HFrEF. (c) All-cause mortality in HFmrEF. (d) Cardiovascular mortality in HFmrEF. (e) All-cause mortality in HFpEF. (f) Cardiovascular mortality in HFpEF. HFmrEF, heart failure with mid-range ejection fraction; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; SBP, systolic blood pressure.
The Kaplan–Meier curves showed that in patients with HFrEF, the risk of all-cause and cardiovascular death was significantly higher in the SBP <130 mmHg group than the SBP ≥130 mmHg group (Fig. 3a and b). After adjusting for the confounding variables listed in Tables S3 and S4, Supplemental Digital Content, the multivariate Cox proportional hazards model revealed that the risks of all-cause death (HR 2.53, 95% CI 1.23–5.20, P = 0.011) and cardiovascular death (HR 1.91, 95% CI 1.01–3.63, P = 0.047) were also significantly higher in the SBP <130 mmHg group (Table 3). In HFmrEF patients, the risk of all-cause and cardiovascular death increased in the SBP < 130 mmHg group, but this increase was not significant (Fig. 3c, d and Table 3). However, this trend was not evident in HFpEF patients (Fig. 3e, f and Table 3).
FIGURE 3.
Kaplan–Meier curve analysis for the risk of mortality according to <130 mmHg vs. SBP ≥130 mmHg across the spectrum of heart failure. (a) All-cause mortality in HFrEF. (b) Cardiovascular mortality in HFrEF. (c) All-cause mortality in HFmrEF. (d) Cardiovascular mortality in HFmrEF. (e) All-cause mortality in HFpEF. (f) Cardiovascular mortality in HFpEF. HFmrEF, heart failure with mid-range ejection fraction; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; SBP, systolic blood pressure.
SBP level and mortality in subgroups
In both the overall patient population and the HFrEF patient population, we discovered a significant interaction between the SBP groups and eGFR for the risk of all-cause and cardiovascular mortality (Table 4). In all patients, the HR of all-cause mortality was 2.03 (95% CI, 1.21–3.41), and cardiovascular mortality was 2.43 (95% CI, 1.39–4.26) for the eGFR ≥ 45 ml/min/1.73 m2 subgroup. In the HFrEF patients, the HR of all-cause mortality was 6.19 (95% CI, 2.10–18.26), and cardiovascular mortality was 6.88 (95% CI, 2.15–21.98) (Table 4). In the eGFR < 45 ml/min/1.73 m2 subgroup, the HR did not differ significantly across SBP groups (Table 4).
TABLE 4.
Subgroup analyses of all-cause death and cardiovascular death by <130 mmHg vs. SBP ≥130 mmHg
| Total population | HFrEF | HFmrEF | HFpEF | |||||
| HR (95% CI) | P for interation | HR (95% CI) | P for interation | HR (95% CI) | P for interation | HR (95% CI) | P for interation | |
| All-cause death | ||||||||
| Age | ||||||||
| <65 | 1.33 (0.64–2.74) | 0.960 | 2.90 (0.64–13.17) | 0.964 | 3.13 (0.74–13.19) | 0.502 | 1.20 (0.35–4.16) | 0.357 |
| ≥65 | 1.23 (0.82–1.84) | 3.03 (1.28–7.16) | 1.34 (0.62–2.91) | 0.87 (0.45–1.68) | ||||
| Gender | ||||||||
| Male | 1.36 (0.84–2.21) | 0.211 | 2.71 (0.99–7.39) | 0.505 | 0.85 (0.33–2.19) | 0.900 | 0.97 (0.42–2.28) | 0.459 |
| Female | 1.23 (0.74–2.07) | 2.43 (0.69–8.52) | 2.27 (0.87–5.93) | 1.05 (0.49–2.27) | ||||
| Diabetes | ||||||||
| No | 1.33 (0.83–2.12) | 0.530 | 3.09 (1.31–7.28) | 0.039 | 1.09 (0.36–3.35) | 0.615 | 1.00 (0.46–2.14) | 0.372 |
| Yes | 1.24 (0.73–2.08) | 2.98 (0.42–21.33) | 2.14 (0.93–4.89) | 0.96 (0.42–2.22) | ||||
| eGFR | ||||||||
| <45 | 0.87 (0.53–1.41) | 0.002 | 1.00 (0.26–3.80) | 0.007 | 1.69 (0.66–4.36) | 0.313 | 0.93 (0.39–2.20) | 0.052 |
| ≥45 | 2.03 (1.21–3.41) | 6.19 (2.10–18.26) | 2.24 (0.80–6.22) | 1.02 (0.48–2.17) | ||||
| ACEI/ARB/ARNI | ||||||||
| No | 0.95 (0.54–1.69) | 0.271 | 0.52 (0.12–2.25) | 0.148 | 1.85 (0.53–6.41) | 0.596 | 0.92 (0.38–2.22) | 0.511 |
| Yes | 1.66 (1.06–2.61) | 3.72 (1.57–8.82) | 1.19 (0.49–2.90) | 1.01 (0.50–2.04) | ||||
| β-Blockers | ||||||||
| No | 1.50 (0.90–2.50) | 0.314 | 2.93 (0.82–10.49) | 0.865 | 3.44 (1.09–10.86) | 0.205 | 1.03 (0.45–2.37) | 0.888 |
| Yes | 1.22 (0.75–1.98) | 2.34 (0.93–5.90) | 0.68 (0.28–1.67) | 0.94 (0.45–1.98) | ||||
| Calcium channel blockers | ||||||||
| No | 1.38 (0.92–2.06) | 0.852 | 2.70 (1.23–5.92) | 0.678 | 2.17 (0.97–4.83) | 0.678 | 0.85 (0.43–1.65) | 0.957 |
| Yes | 1.25 (0.57–2.73) | 673.63 (2.46–184202.80) | 0.80 (0.16–4.01) | 1.75 (0.63–4.84) | ||||
| Diuretics | ||||||||
| No | 1.32 (0.84–2.06) | 0.825 | 1.55 (0.65–3.66) | 0.030 | 2.97 (1.15–7.68) | 0.012 | 1.00 (0.53–1.90) | 0.874 |
| Yes | 1.34 (0.75–2.40) | 8.54 (1.64–44.59) | 0.67 (0.22–2.06) | 1.03 (0.34–3.08) | ||||
| Cardiovascular death | ||||||||
| Age | ||||||||
| <65 | 1.64 (0.78–3.49) | 0.576 | 1.87 (0.56–6.23) | 0.964 | 1.50 (0.28–8.07) | 0.521 | 2.03 (0.46–8.91) | 0.028 |
| ≥65 | 1.28 (0.80–2.03) | 2.02 (0.92–4.43) | 1.17 (0.48–2.85) | 0.58 (0.19–1.71) | ||||
| Gender | ||||||||
| Male | 1.21 (0.75–1.96) | 0.272 | 1.98 (0.88–4.47) | 0.752 | 0.71 (0.25–2.01) | 0.785 | 1.47 (0.54–4.01) | 0.046 |
| Female | 1.23 (0.64–2.35) | 4.13 (1.00–17.09) | 1.61 (0.50–5.20) | 0.66 (0.14–3.02) | ||||
| Diabetes | ||||||||
| No | 1.22 (0.73–2.04) | 0.813 | 2.43 (1.09–5.41) | 0.398 | 0.48 (0.13–1.76) | 0.268 | 1.14 (0.40–3.25) | 0.893 |
| Yes | 1.43 (0.80–2.57) | 2.79 (0.51–15.28) | 2.29 (0.92–5.72) | 0.93 (0.29–3.00) | ||||
| eGFR | ||||||||
| <45 | 0.74 (0.41–1.32) | 0.003 | 0.56 (0.17–1.90) | 0.002 | 1.26 (0.46–3.47) | 0.728 | 0.76 (0.17–3.47) | 0.054 |
| ≥45 | 2.43 (1.39–4.26) | 6.88 (2.15–21.98) | 1.62 (0.52–5.00) | 1.47 (0.55–3.94) | ||||
| ACEI/ARB/ARNI | ||||||||
| No | 1.15 (0.59–2.25) | 0.568 | 0.68 (0.16–2.92) | 0.110 | 3.36 (0.89–12.71) | 0.093 | 0.84 (0.22–3.20) | 0.318 |
| Yes | 1.38 (0.86–2.23) | 3.25 (1.45–7.29) | 0.71 (0.27–1.89) | 1.16 (0.44–3.04) | ||||
| β-Blockers | ||||||||
| No | 1.74 (0.99–3.05) | 0.163 | 2.55 (0.97–6.67) | 0.469 | 5.08 (1.41–18.27) | 0.072 | 1.28 (0.38–4.23) | 0.824 |
| Yes | 0.97 (0.57–1.66) | 1.58 (0.63–3.95) | 0.47 (0.16–1.33) | 0.87 (0.30–2.49) | ||||
| Calcium channel blockers | ||||||||
| No | 1.24 (0.80–1.90) | 0.931 | 1.73 (0.89–3.35) | 0.796 | 1.26 (0.50–3.20) | 0.107 | 1.06 (0.40–2.80) | 0.726 |
| Yes | 1.30 (0.55–3.09) | 306.13 (0–2.0128E126) | 1.98 (0.48–8.17) | 1.07 (0.28–4.09) | ||||
| Diuretics | ||||||||
| No | 1.22 (0.75–1.98) | 0.995 | 1.11 (0.50–2.49) | 0.023 | 3.18 (0.99–10.20) | 0.025 | 1.09 (0.45–2.63) | 0.967 |
| Yes | 1.29 (0.68–2.44) | 9.55 (2.31–39.52) | 0.59 (0.18–1.90) | 0.88 (0.17–4.62) | ||||
ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; ARNI, angiotensin receptor neprilysin inhibitor; eGFR, estimated glomerular filtration rate; SBP, systolic blood pressure.
In addition, for all patients, only the subgroup that used ACEI/ARB/ARNI had a higher risk of all-cause mortality in the SBP <130 mmHg group (Table 4). For HFrEF patients, a SBP <130 mmHg represented a much higher risk of all-cause death for patients aged ≥65, patients without diabetes, patients using ACEI/ARB/ARNI and patients were not using calcium channel blockers. The risk of cardiovascular death was much higher for patients without diabetes and those using ACEI/ARB/ARNI or diuretics (Table 4).
DISCUSSION
Our study examined the relationship between SBP and prognosis in all or three EF phenotypes of heart failure (HF) patients with hypertension. We discovered that when SBP < 130 mmHg, the risk of all-cause and cardiovascular death increased gradually as the SBP level decreased in all patients. Furthermore, SBP <130 mmHg was also associated with an increased risk of all-cause and cardiovascular mortality in HFrEF patients. A trend toward increased risk was observed among HFmrEF patients, but it was not statistically significant. This trend was not observed in HFpEF patients.
In our study, the incidence of cumulative all-cause death and cardiovascular death was higher in the SBP <130 mmHg group. The results of our study on HFrEF complicated by hypertension are similar to those of Cherine Arundel [11]. In his study the all-cause mortality at 30 days and 1 year was significantly higher in the SBP <130 mmHg group than in the SBP ≥130 mmHg group among all matched patients. However, the main difference between his study and ours is that in his study, only 70% of HF cases were complicated by hypertension, while 100% of our selected cases were complicated by hypertension [11]. This difference suggests that the current SBP of patients with HF may have a more significant impact the prognosis than the history of hypertension itself. Sameer Ather [26] obtained results similar to ours. In his study on the relationship between SBP and HFrEF mortality, a U-shaped association was observed between SBP and mortality in patients with mild to moderate left ventricular systolic dysfunction. In patients with severe left ventricular systolic dysfunction, SBP was linearly associated with mortality, and lower SBP was associated with increased mortality [26].
In hypertensive patients with HFpEF, no statistically significant difference was detected between the SBP < 130 mmHg group and the SBP ≥130 mmHg group, which may be associated with the grouping of SBP levels at admission. Similar studies have been reported in HFpEF patients. Yossi Rosman found that if HFpEF patients were divided into four groups according to the BP level at admission, the in-hospital and 1-year mortality of patients with the lowest SBP (<127 mmHg) at admission was significantly higher than that of other groups [15]. Charles Faselis found that despite the lack of difference in the incidence of all-cause death and HF readmission between the SBP ≥ 130 mmHg and SBP <130 mmHg groups in elderly HFpEF patients, SBP <120 mmHg was associated with a higher risk of all-cause death than SBP ≥130 mmHg [27]. Regarding the correlation between BP and HFpEF prognosis, the conclusions differ by study, possibly because HFpEF is a highly heterogeneous disease, and the included populations differ by study. In addition, patients with HFpEF without hypertension were included in the Yossi Rosman study, which may also have contributed to differential results [15].
In hypertensive patients with HFmrEF, the incidence of all-cause death and cardiovascular death was lower in the SBP ≥130 mmHg group than in the SBP <130 mmHg group, but this difference was not statistically significant. To our knowledge, the correlation between the SBP level of hypertensive patients in HFmrEF and mortality has not yet been studied. However, the 2021 ESC guidelines recommended that HFmrEF be renamed from “heart failure with intermediate ejection fraction” to “heart failure with slightly reduced ejection fraction” and that HFmrEF is a disease state closer to HFrEF [18].
Hypertension and diabetes are long-term chronic diseases, and heart failure patients with diabetes or hypertension are more special groups. Therefore, the blood glucose control and blood pressure control of patients with heart failure should be systematic management rather than the traditional single hypoglycemic or hypotensive control treatment.
Our study is a single-center observational study and as such has some inherent limitations. Most of the population was from northeast China, and the applicability of our results to other populations with different genetic backgrounds needs to be further verified. In addition, the compliance with drugs regimens for the treatment of HF, whether the drugs were titrated to the optimal concentration, and whether the patient's own management is appropriate are all important factors affecting the prognosis of patients. We adjusted our analyses for a large number of cardiovascular risk factors but there is likely residual and unmeasured confounding. Finally, the follow-up time is relatively short with an average follow-up of one year at the time of this report and additional follow-up is awaited.
In conclusion, we revealed that in HFrEF patients, SBP <130 mmHg was associated with an increased risk of all-cause and cardiovascular mortality. A trend toward increased risk was observed among HFmrEF patients, but it was not statistically significant. This trend was not observed in HFpEF patients.
ACKNOWLEDGEMENTS
We would like to thank all of the authors for their efforts and support.
Authors contributions statement: Xueyan Lang and Yanxiu Zhang designed the analysis and wrote the first draft of the article. Rong Gao, Bing Zhao and Yilan Li conducted the model parameterization and the statistical analyses. Yao Zhang and Cheng Peng revised the manuscript. All authors read and approved the final manuscript.
Xueyan Lang and Cheng Peng: Xueyan Lang and Cheng Peng are co-first authors who equally contributed to this work.
Data availability statement: Datasets are not accessible to the general public, but they are available upon reasonable request from the corresponding author.
Funding: This work was supported by the National Natural Science Foundation of China (grant 81770255), the Key Laboratory of Myocardial Ischemia, Ministry of Education (grant KF202216 and KF202204).
Conflicts of interest
The authors certify that they have no financial conflicts of interest.
Supplementary Material
Xueyan Lang and Cheng Peng are co- first authors who equally contributed to this work.
Abbreviations: ACEI/ARB/ARNI, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker/angiotensin receptor-neprilysin inhibitor; CIs, confidence intervals; DBP, diastolic blood pressure; EF, ejection fraction; eGFR, estimated glomerular filtration rate; HDL, high-density lipoprotein; HF, heart failure; HFmrEF, HF with mid-range or mildly reduced ejection fraction; HFpEF, HF with preserved ejection fraction; HFrEF, HF with reduced ejection fraction; HMUSAH, The Second Affiliated Hospital of Harbin Medical University; HRs, hazard ratios; IQR, interquartile ranges; LDL, low-density lipoprotein; LVEF, left ventricular ejection fraction; LVPW, left ventricular posterior wall; NT-proBNP, N-terminal pro-B-type natriuretic peptide; RCS, restricted cubic splines; SBP, systolic blood pressure
Revised 7 January, 2024
Supplemental digital content is available for this article.
REFERENCES
- 1.Heidenreich PA, Albert NM, Allen LA, Bluemke DA, Butler J, Fonarow GC, et al. Forecasting the impact of heart failure in the United States: a policy statement from the American Heart Association. Circ Heart Fail 2013; 6:606–619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Virani SS, Alonso A, Aparicio HJ, Benjamin EJ, Bittencourt MS, Callaway CW, et al. Heart Disease and Stroke Statistics-2021 update: a report from the American Heart Association. Circulation 2021; 143:e254–e743. [DOI] [PubMed] [Google Scholar]
- 3.Ni H, Xu J. Recent trends in heart failure-related mortality: United States. NCHS Data Brief 2000. –2014; 2015:1–8. [PubMed] [Google Scholar]
- 4.Agarwal MA, Fonarow GC, Ziaeian B. National trends in heart failure hospitalizations and readmissions from 2010 to 2017. JAMA Cardiol 2021; 6:952–956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Khera R, Kondamudi N, Zhong L, Vaduganathan M, Parker J, Das SR, et al. Temporal trends in heart failure incidence among medicare beneficiaries across risk factor strata, 2011 to 2016. JAMA Network Open 2020; 3:e2022190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tsao CW, Lyass A, Enserro D, Larson MG, Ho JE, Kizer JR, et al. Temporal trends in the incidence of and mortality associated with heart failure with preserved and reduced ejection fraction. JACC Heart Fail 2018; 6:678–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Haider AW, Larson MG, Franklin SS, Levy D, Framingham Heart S. Systolic blood pressure, diastolic blood pressure, and pulse pressure as predictors of risk for congestive heart failure in the Framingham Heart Study. Ann Intern Med 2003; 138:10–16. [DOI] [PubMed] [Google Scholar]
- 8.Mahmood SS, Levy D, Vasan RS, Wang TJ. The Framingham Heart Study and the epidemiology of cardiovascular disease: a historical perspective. Lancet 2014; 383:999–1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Andersson C, Nayor M, Tsao CW, Levy D, Vasan RS. Framingham heart study: JACC Focus Seminar, 1/8. J Am Coll Cardiol 2021; 77:2680–2692. [DOI] [PubMed] [Google Scholar]
- 10.Lee DS, Austin PC, Rouleau JL, Liu PP, Naimark D, Tu JV. Predicting mortality among patients hospitalized for heart failure: derivation and validation of a clinical model. JAMA 2003; 290:2581–2587. [DOI] [PubMed] [Google Scholar]
- 11.Arundel C, Lam PH, Gill GS, Patel S, Panjrath G, Faselis C, et al. Systolic blood pressure and outcomes in patients with heart failure with reduced ejection fraction. J Am Coll Cardiol 2019; 73:3054–3063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tsimploulis A, Lam PH, Arundel C, Singh SN, Morgan CJ, Faselis C, et al. Systolic blood pressure and outcomes in patients with heart failure with preserved ejection fraction. JAMA Cardiol 2018; 3:288–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jackson AM, Benson L, Savarese G, Hage C, Jhund PS, Petrie MC, et al. Apparent treatment-resistant hypertension across the spectrum of heart failure phenotypes in the Swedish HF Registry. JACC Heart Fail 2022; 10:380–392. [DOI] [PubMed] [Google Scholar]
- 14.Lee SE, Lee HY, Cho HJ, Choe WS, Kim H, Choi JO, et al. Reverse J-curve relationship between on-treatment blood pressure and mortality in patients with heart failure. JACC Heart Fail 2017; 5:810–819. [DOI] [PubMed] [Google Scholar]
- 15.Rosman Y, Kopel E, Shlomai G, Goldenberg I, Grossman E. The association between admission systolic blood pressure of heart failure patients with preserved systolic function and mortality outcomes. Eur J Intern Med 2015; 26:807–812. [DOI] [PubMed] [Google Scholar]
- 16.Buiciuc O, Rusinaru D, Lévy F, Peltier M, Slama M, Leborgne L, et al. Low systolic blood pressure at admission predicts long-term mortality in heart failure with preserved ejection fraction. J Cardiac Fail 2011; 17:907–915. [DOI] [PubMed] [Google Scholar]
- 17.Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 2016; 18:891–975. [DOI] [PubMed] [Google Scholar]
- 18.McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Bohm M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2021; 42:3599–3726. [DOI] [PubMed] [Google Scholar]
- 19.Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol 2022; 79:e263–e421. [DOI] [PubMed] [Google Scholar]
- 20.Kelly TN, Gu D, Chen J, Huang JF, Chen JC, Duan X, et al. Hypertension subtype and risk of cardiovascular disease in Chinese adults. Circulation 2008; 118:1558–1566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fonarow GC, Adams KF, Jr, Abraham WT, Yancy CW, Boscardin WJ, Adhere Scientific Advisory Committee SG, et al. Risk stratification for in-hospital mortality in acutely decompensated heart failure: classification and regression tree analysis. JAMA 2005; 293:572–580. [DOI] [PubMed] [Google Scholar]
- 22.Gheorghiade M, Abraham WT, Albert NM, Greenberg BH, O’Connor CM, She L, et al. Systolic blood pressure at admission, clinical characteristics, and outcomes in patients hospitalized with acute heart failure. JAMA 2006; 296:2217–2226. [DOI] [PubMed] [Google Scholar]
- 23.Nunez J, Nunez E, Fonarow GC, Sanchis J, Bodi V, Bertomeu-Gonzalez V, et al. Differential prognostic effect of systolic blood pressure on mortality according to left-ventricular function in patients with acute heart failure. Eur J Heart Fail 2010; 12:38–44. [DOI] [PubMed] [Google Scholar]
- 24.Heart Failure Group of Chinese Society of Cardiology of Chinese Medical Association, Chinese Heart Failure Association of Chinese Medical Doctor Association, Editorial Board of Chinese Journal of Cardiology. Chinese guidelines for the diagnosis and treatment of heart failure 2018. Zhonghua Xin Xue Guan Bing Za Zhi 2018; 46:760–789. [DOI] [PubMed] [Google Scholar]
- 25.Stergiou GS, Palatini P, Parati G, O’Brien E, Januszewicz A, Lurbe E, et al. 2021 European Society of Hypertension practice guidelines for office and out-of-office blood pressure measurement. J Hypertens 2021; 39:1293–1302. [DOI] [PubMed] [Google Scholar]
- 26.Ather S, Chan W, Chillar A, Aguilar D, Pritchett AM, Ramasubbu K, et al. Association of systolic blood pressure with mortality in patients with heart failure with reduced ejection fraction: a complex relationship. Am Heart J 2011; 161:567–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Faselis C, Lam PH, Zile MR, Bhyan P, Tsimploulis A, Arundel C, et al. Systolic blood pressure and outcomes in older patients with HFpEF and hypertension. Am J Med 2021; 134:e252–e263. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.