Abstract
Chloride is a key anion involved in cellular physiology by regulating its homeostasis and rheostatic processes. Changes in cellular Cl− concentration result in differential regulation of cellular functions such as transcription and translation, post-translation modifications, cell cycle and proliferation, cell volume, and pH levels. In intracellular compartments, Cl− modulates the function of lysosomes, mitochondria, endosomes, phagosomes, the nucleus, and the endoplasmic reticulum. In extracellular fluid (ECF), Cl− is present in blood/plasma and interstitial fluid compartments. A reduction in Cl− levels in ECF can result in cell volume contraction. Cl− is the key physiological anion and is a principal compensatory ion for the movement of the major cations such as Na+, K+, and Ca2+. Over the past 25 years, we have increased our understanding of cellular signaling mediated by Cl−, which has helped in understanding the molecular and metabolic changes observed in pathologies with altered Cl− levels. Here, we review the concentration of Cl− in various organs and cellular compartments, ion channels responsible for its transportation, and recent information on its physiological roles.
Keywords: Anion transport, Chloride channels, Chloride ions, Chloride transporters, Ion homeostasis
Introduction
Chloride (Cl−) is the most abundant ion in humans after sodium [1] and accounts for 70% of the total anions in extracellular fluid (ECF) [2]. There are approximately 115 g of Cl− in an average human adult body, making up to 0.15% of the total body weight as a key macromineral [3]. Cl− are vital for maintaining osmotic pressure, muscle movement, and acid-base balance in the body [3]. Cl− homeostasis is generally overlooked but is known to govern several key physiological functions inside and outside the cell [2,4–9]. Along with cations, Cl− is responsible for maintaining ionic homeostasis, osmotic pressure, and acid–base balance. Therefore, disturbances of Cl− levels are indicative of metabolic disorders including hypochloremic metabolic alkalosis and hyperchloremic metabolic acidosis [2,10]. Cl− does not follow the electrochemical equilibrium in most mammalian cells. In several cells, including primary sensory neurons, leukocytes, epithelial, sympathetic ganglion, and muscle cells, intracellular Cl− is maintained above equilibrium levels. The transport of Cl− occurs via channels, exchangers, and co-transporters that utilize chemical as well as electrical gradients [2,11].
Cl− is a component of a daily diet in the form of sodium chloride (NaCl). It is classified as an electrolyte as it carries a negative charge along with its positive counterparts, K+ and Na+. Cl− is mainly found in a diet consisting of seaweed, rye, vegetables such as lettuce, tomatoes, olives, celery, fruits such as apples, melons, berries, and bananas, as well as red meats [12–14]. Most of the Cl− also comes from added salt in several food preparations [14]. The dietary intake levels for Cl− vary with development as shown in Table 1: 0.3 g/day for infants aged 7–11 months, 1.7 g/day for children aged 1–3 years, 2.0 g/day for children aged 4–6 years, 2.6 g/day for children aged 7–10 years, 3.1 g/day for children aged 11–17 years, and 3.1 g/day for adults, including pregnant and lactating women [8]. Cl− deficiency is extremely rare as the average diet is high in NaCl [8]. A loss of Cl− is accompanied by a loss of sodium (Na) ions, observed in patients with prolonged diarrhea, vomiting, or excessive sweating [15,16]. Additionally, diuretics or high blood glucose levels can result in decreased Cl− levels [17]. In contrast, hyperchloremia (above the reference range of 97−107 mmol/L) is caused by an excessive intake of NaCl, severe dehydration, or metabolic abnormalities [3]. Excreted Cl− levels in urine are independent of Cl− intake, making it difficult to evaluate the status of Cl− in the body [17]. There are limited studies where the role of Cl− was evaluated in pathological conditions [2]. Only studies on cardiovascular diseases tend to incorporate a control such as normal Na+ and low Cl− levels to implicate Cl− in determining the outcome and survivability of patients [5,7,18–23].
Table 1. Chloride levels in various human organs.
| Organ | Fetus (mM) | Infant (mM) | Adult (mM) |
|---|---|---|---|
| Skin | 90–96 | 67–72 | 71 |
| Heart | 41 | 45–50 | 45 |
| Liver | 57–62 | 42–55 | 38 |
| Kidney | 60–67 | 61 | 58 |
| Brain | 72 | 66 | 41 |
| Blood | 96–106 | 90–110 | 98–106 |
Cl− is specifically necessary for the formation of hydrochloric acid (HCl) in the stomach, which activates several gastric enzymes involved in the digestion [24]. The concentration of Cl− in the stomach is 150 mM, whereas in the blood it is 98–106 mM [25]. Therefore, Cl− must be secreted in the lumen against the concentration gradient. The membrane potential at the apical surface of the resting cell is −70 mV [24]. This facilitates Cl− secretion against the electrical gradient. In conditions like excessive vomiting, the loss of stomach content results in an abnormal feedback mechanism for acid-mediated secretion of digestive enzymes [24]. Several clinical conditions are related to the decreased concentration of Cl− in the serum, termed hypochloremia (typically below the reference range of 97−106 mmol/L)-, which manifests in metabolic alkalosis [26]. Conversely, high Cl− concentration above the reference range results in hyperchloremia. An excessive loss of bicarbonate tends to cause a proportional increase of Cl− [27] as a result of excessive carbonate loss observed during severe diarrhea [2,26] or the intake of certain medications such as acetazolamide and triamterene.
Cl− is a key ion of the extracellular fluid compartment (ECF), and with a concentration of 155 mM, it makes up 66% of all the ECF anions [27]. In addition to ECF, Cl− is also present in the intracellular spaces, albeit at lower concentrations [27]. The slight concentration difference between two different compartments is due to capillary impermeability to proteins such as albumin [27]. The intracellular Cl− concentration depends on the cell types and function with respect to other ions [4]. On average, the intracellular concentration of Cl− ranges from 5 to 60 mM [28]. Muscle cells have a resting potential of approximately −70 mV and a low Cl− concentration of 3–4 mM [29]. However, cells with high membrane potential, such as erythrocytes, have a higher concentration of Cl− of around 70 mM [30]. This higher concentration is essential in moving Cl− into and out of the cell effectively during the phenomenon of ‘chloride shift’ between the plasma and the red blood cells [30,31].
In this review, we summarize the recent information on the role of Cl− in organ (Figure 1) and cellular (Figure 2) physiology. Although abnormal Cl− levels are indicators of several physiological conditions, the ion channels and transporters that conduct ions remain understudied compared with their cationic counterparts [32].
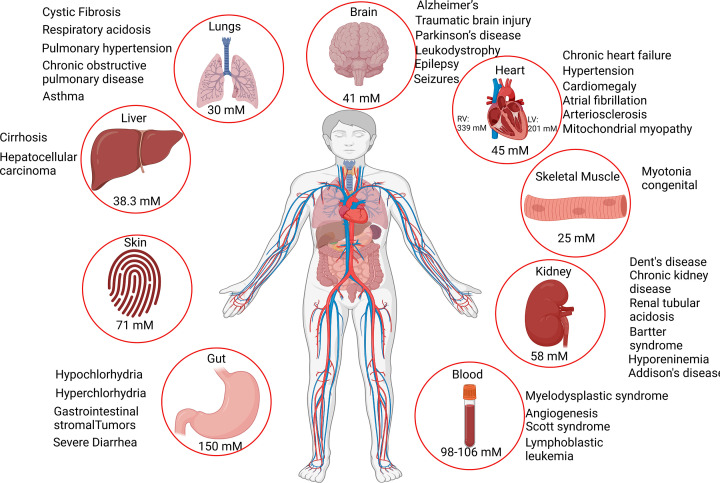
Figure 1. Chloride concentrations in adult human organs.
The Cl− concentration in adult human organs varies in different organ systems. The Cl− concentration in the brain (41 mM), heart (overall 45 mM, and specifically right ventricle 339 mM and left ventricle 201 mM), muscle (25 mM), kidney (58 mM), blood (98–106 mM), gut (150 mM), skin (71 mM), liver (38 mM), and lungs (30 mM). Associated human diseases for various organs are highlighted. All the values were obtained from previous studies [26–28,31,36,40,50,81–83,88,116,156,157,169,195–206]. Images were generated by Biorender.
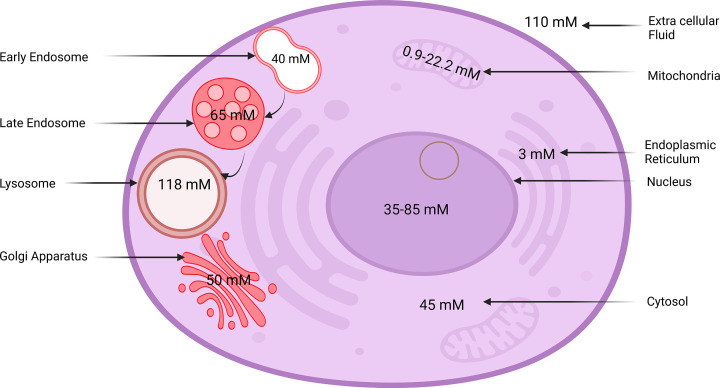
Figure 2. Schematic representation of chloride concentration in the cell organelles.
The Cl− concentration in an extracellular and cellular compartment maintains cellular homeostasis. The extracellular Cl− concentration (110 mM), cytosol (36 mM), early endosome (40 mM), late endosome (65 mM), lysosome (118 mM), mitochondria (0.9–22.2 mM), nucleus (35–85 mM), endoplasmic reticulum (3 mM), and Golgi apparatus (50 mM) [83,133,134,169,175,192,207,208].
Chloride and organ systems
Cl− levels in the body are regulated by kidneys [33]. In the glomerular ultrafiltrate, Cl− is the most prevalent ion after sodium. Most of the Cl− is filtered and reabsorbed in the renal tubules by both active and passive transportation mechanisms [34]. In addition to the kidneys, the intestines also absorb Cl [35]. In this section, we will discuss the role of each organ system and its Cl− levels. During early development and preterm infancy, Cl− levels (Table 1) are influenced by sodium, Cl− intake, and gestational age [36].
Chloride in the kidneys
The excretion of Cl− is mainly done via the kidneys (Figure 1). Approximately 99% of the Cl− filtered through the kidney gets reabsorbed along with Na+ [37]. Therefore, only a small fraction gets excreted [27]. Reabsorption occurs either at the paracellular proximal tubule via Cl− channels and transporters, or at the apical membrane via Cl−/anion exchangers or basolateral via Cl−/carbonate exchanger [38]. In the kidneys, the proximal tubule and the ascending loop of Henle are responsible for reabsorbing the majority of the filtered Cl− in the body [33]. In contrast, the distal tubule and collecting duct absorb a very small amount of Cl− [39]. However, they still play a significant physiological role in maintaining Cl− homeostasis [40]. Proximal convoluted tubule (PCT) absorbs most of the water and 50% of Cl− along with Ca2+, Mg2+, and HPO42−. In basal membranes, the Na+/K+ ATPase generates an electrochemical gradient that facilitates the reabsorption of Cl− by Na+/ Cl− symporters in the apical membrane. While Na+ is actively transported from the basal side of the cell into the interstitial fluid, Cl− and Na+ are pumped into the interstitial fluid by a paracellular route between cells through leaky tight junctions.
In the collecting ducts of the kidneys, vacuolar H+-ATPase and Slc26a11 regulate pH and renal acid–base secretion [41]. Bicarbonate transporters also cause an uptake of NaCl [42]. All the bicarbonate transporters carry HCO3− and/or CO3− along with at least one either Na2+ or Cl− [42]. In the connecting segments and the collecting tubules of the kidneys, aldosterone, a major mineralocorticoid steroid hormone secreted by glomerulosa cells in the adrenal cortex, is another vital component in facilitating the reabsorption of NaCl [43]. Therefore, a deficiency in this hormone would result in hyperkalemic and hyperchloremic acidosis (Figure 1). The key mechanism involves aldosterone by increasing the number of Na and Cl− transporters in the luminal membrane [44,45]. When tubular reabsorption of Cl− is enhanced, it leads to a Na imbalance and extracellular volume expansion, which causes hypertension and hyporeninemia [5]. Kidneys must adapt to metabolic acidosis and acid-base disturbances [46]. Kidneys mainly adapt to these imbalances via Cl− excretion [47]. Kidneys increase acid secretion by enhancing NH4Cl secretion via the apical sodium/hydrogen exchanger (NHE3), which also works in tandem with the Na+/K+/2Cl− cotransporter [48,49]. When there is a prolonged period without sodium excretion, the lack of ion exchange pushes the system to reabsorb bicarbonate and return pH levels to normal [50]. Recently, the outcome of hypochloremeia was evaluated in patients with decompensated cirrhosis. Surprisingly, hypochloremia increases mortality in patient [51].
Chloride in the gut
Cl− in the gut comes from the consumption of table salt as well as foods containing other types of Cl− salts. Most of the Cl− is absorbed from the intestines during digestion [52] (Figure 1). Cl− in the intestinal lumen gets absorbed by three different mechanisms: a passive or paracellular pathway, an electroneutral pathway involving the Na/H and Cl−/carbonate exchange, and a carbonate-dependent Cl− absorption pathway [35].
Hydrochloric acid in gastric juice is composed of Cl− that is secreted into the stomach [53]. Parietal cells located in the middle part of the glands of the fundus-body region of the stomach produce HCl by secreting H+ and Cl− [54]. Hydrochloric acid activates digestive enzymes, controls foodborne microorganisms, limits microorganism growth in the intestine, and facilitates the absorption of several nutrients [53]. At pH below 4.0, gastric juices have an anti-microbial effect [53], which is recognized as a ‘gastric bactericidal barrier’ since 1925 [55]. The H+K+ATPase (the proton pump) in the basolateral and apical membranes of the gut control the secretion of hydrochloric acid into the stomach [56,57]. Moreover, recently identified Cl− channels can also facilitate the secretion of Cl−. Some of these are calcium-activated Cl− channels (CaCC), cystic fibrosis conductance regulator (CFTR), and chloride type-2 (ClC-2) channels [58,59]. Na+/K+ ATPase pumps, potassium channels, and Na+/K+/Cl− transporters move Cl− across basolateral membranes [58–60]. Another major function of Cl− in the gut is facilitating water absorption [59]. Cl− contributes to the osmotic gradient needed to regulate water secretion into the gut [61] (Figure 1). As water cannot be actively secreted, the driving force is the osmotic gradient generated by negative ions like Cl−, as well as carbonate [58]. Na+ participates as the counter ion in the paracellular regions [62].
Chloride in the brain
Cl− in the brain is associated with the regulation of ionic homeostasis and water concentrations [63]. Water accounts for 80% of the total brain, but its transport needs an osmotic gradient by anions [64]. The balance between transporters and Cl− channels in the plasma membrane regulates and maintains the intracellular concentration of Cl− [65]. Neurons and astrocytes express a plenteous set of Cl− channels and transporters belonging to several protein families with unique modes of regulation and activation [65]. Abnormal levels of Cl− are associated with brain disorders, trauma, hypoxic-ischemic encephalopathy, edema, and post-traumatic seizures (Figure 1) [32]. In the brain, the concentration of Cl− levels is low (Figure 1), but in cerebral spinal fluid, the concentration is around 120 mM [66]. There is mounting evidence that disorders of the nervous system are caused by abnormal homeostasis of the intracellular concentration of Cl− [65]. This also causes significant abnormalities in neuronal excitability and neurotransmission.
In the central nervous system, Cl− channels and transporters (Table 2) are essential for the growth and development of neurons, the uptake of neurotransmitters, intracellular pH regulation, cell volume regulation, control of membrane potential, cell proliferation, apoptosis, and, most importantly, the adjustment of [Cl−]i to its equilibrium potential [67]. In neurons and astrocytes, Cl− channels, such as CLIC1 [68–70], are pivotal in regulating ion and water homeostasis as they play a key role in action potential generation and impulse conduction [70]. By regulating the postsynaptic reactions of GABA and glycine neurotransmitters, Cl− plays a critical role in modulating neuronal excitability [71,72]. GABA and glycine receptors are ligand-gated Cl− channels that respond to GABA and glycine neurotransmitters, respectively. When these receptors are activated, they cause an influx or efflux of Cl−, depending on the electrochemical potential of Cl− for the cell. These Cl− fluxes lead to inhibitory and sometimes excitatory responses [72]. GABAergic signals are the primary inhibitory transmitters in the adult brain and are an important part of coordinating the assembly of neuronal circuits in the developing brain [73]. GABA is the primary neurotransmitter active within the developing brain and facilitates the proliferation of neuronal progenitor cells [74]. The dysregulation of GABAergic signaling has been linked to a variety of neurological and neurodevelopmental disorders, including epilepsy, schizophrenia, Down’s syndrome (DS), and autism spectrum disorders [75]. In relapsing remitting multiple sclerosis, elevated Cl− levels of ≥123.2 mmol/L were associated with an increased frequency of relapse as compared with patients with a cerebrospinal fluid Cl− level of <123.2 mmol/L [76]. Cl− in cerebrospinal fluid is a key electrolyte in maintaining the ionic homeostasis of the brain and spinal cord [76]. In fact, for a long period, spinal fluid Cl− levels were associated with tuberculous meningitis [77]. Any variability in Cl− concentration in cerebrospinal fluid could result in neurological conditions such as hydrocephalus, meningitis, and encephalitis.
Table 2. Chloride ion channels and transporters.
| Name | Localization | Pathophysiology | Conductance (pS) | Permeability |
|---|---|---|---|---|
| ClC1 | Plasma membrane | Myotonia congenital | 1–2 | Cl− > Br− > I− |
| ClC2 | Plasma membrane | Leukodystrophy | 2–3 | Cl− > Br− > I− > Cl− (in cell swelling) |
| ClC3 | Plasma membrane and late endosomes | Degeneration of CNS and retina | ∼40 | Cl− > I− |
| ClC4 | Endosomes | Epilepsy | ∼1 | Cl− > I− |
| ClC5 | Endosomes | Dent’s disease and impaired renal endocytosis | NO3− > Cl− > Br− > I− | |
| ClC6 | Late endosomes | Lysosomal storage in neurons | ∼100 (from bilayer recordings) | – |
| ClC7 | Lysosomes | Osteopetrosis, CNS, and retina degeneration | – | |
| ClCKa | Plasma membrane of inner ear and kidney | Diabetes insipidus | – | Cl− > Br− > NO3 > I− |
| ClCKb | Plasma membrane of inner ear and kidney | Bartter’s syndrome | 20–25 (with barttin subunit) | Br− > I− > Cl− |
| CFTR | Plasma membrane | Cystic fibrosis, acute pancreatitis, chronic obstructive pulmonary disease, and the hyper-responsiveness in asthma | ∼10 | Br− ≥ Cl− > I− > F− |
| GABAARs | Plasma membrane | Neurological functions, seizures, hypotonia, and hyperreflexia | ∼28, 18, and 12 | Cl− > HCO3− |
| ORCC | Plasma membrane | Cystic fibrosis | 30–60 | Cl− ≥ Br− > I− |
| TMEM16A; Anoctamin-1; ANO1 | Plasma membrane | Up-regulation in gastrointestinal stromal tumors (GISTs), in breast cancer, and in head and neck squamous cell carcinomas (HNSCCs); up-regulated in asthma | 1–14 | I− > NO3− > Br− > Cl− > F− > CH3SO4 |
| TMEM16B; Anoctamin-2; ANO2 | Plasma membrane | Anxiety modulation | ∼10 | SCN− (14) > I− > NO3− > Br− |
| TMEM16F; Anoctamin-6; ANO6 | Plasma membrane | Mutated in Scott syndrome | 1–3 | I− > Br− > Cl− > F− > aspartate |
| CLIC1 | Cytoplasm, exosomes, plasma membrane, intracellular membrane, mitochondria, and nucleoplasm | Myelodysplastic syndrome and several cancers | 35–50 (from bilayer recordings) with sub states | I− > SCN− ≥ Cl− ≥ NO2− and NO3−≥ Br− ≥ F− (in symmetrical ionic conditions) I− > F− = SCN− > Cl− = NO2− and NO3− = Br− (in asymmetrical ionic conditions) |
| CLIC2 | Cytoplasm, nucleus, and endoplasmic reticulum | X-linked cognitive disability, congestive heart failure, cardiomegaly, erythematosus, seizures, myopia, and atrial fibrillation | 30–40 (from bilayer recordings) | Cl > Choline |
| CLIC3 | Nucleus, exosome, and plasma membrane | Fetal growth restriction, pre-eclampsia, and breast cancer | ∼1–2 nS | – |
| CLIC4 | Cytoplasm, mitochondrial associated membrane (cardiomyocytes), nucleus, exosomes, golgi apparatus, plasma membrane, and intracellular membrane | Several cancers, benign familial infantile seizures, and pulmonary hypertension | 10, 30, and 57 (from bilayer and tip dip recordings) | – |
| CLIC5 | Nucleus, inner mitochondrial membrane (cardiomyocytes), exosomes, Golgi apparatus, plasma membrane, intracellular membrane, and secretory vesicles in renal glomeruli | Renal dysfunction, juvenile myoclonic epilepsy, migraine, macular degeneration, and childhood acute lymphoblastic leukemia | ∼105 (from bilayer recordings) | – |
| CLIC6 | Cytoplasm, exosomes, nucleus, and plasma membrane |
Familial goiter and developmental delay | 1–3 | Cl− > Br− > F− |
| VDAC1 | Plasma membrane and mitochondrial outer membrane | Cystic fibrosis, mitochondrial myopathy, and calcium-induced neurotoxicity | 200–250 | Cl− > K+ > Na+ > glutamate > ATP > acetylcholine > dopamine |
| VDAC2 | Mitochondrial outer membrane | Alzheimer’s, thyroid cancer, temporal lobe epilepsy (TLE), hypoxia, iron deprivation, and adipogenesis | 1–2 nS | Cl− > K+ (from nanodiscs) |
| VDAC3 | Mitochondrial outer membrane | Hepatocellular carcinoma | 3–4 nS | Cl− > K+ (from nanodiscs) |
| IMAC | Mitochondrial inner membrane | Type 2 diabetes, Parkinson’s disease, atherosclerotic heart disease, stroke, Alzheimer’s disease, and cancer | 107–150 | Cl− > SO42−> Pi ≅ 1,2,3-BTC > 1,3,5-BTC |
| VRAC; VSOR; VSOAC | Plasma membrane | Angiogenesis, cancer, ischemic, and apoptosis | 10–90 | I− ≥ Br− > Cl− > F− > taurine > glutamate |
| PAC; ASOR; PAORAC; TMEM206 | Endosomes | Ischemic stroke, cancer, and hypoxia | 40–10 | SCN− > I− > NO3− > Br− > Cl− |
Numerous chloride channels and transporters are highlighted by their localization in the cell, pathophysiology, conductance, and permeability. ClC1, chloride channel 1; ClC2, chloride channel 2; ClC3, chloride channel 3; ClC4, chloride channel 4; ClC5, chloride channel 5; ClC6, chloride channel 6; ClC7, chloride channel 7; CLIC1, chloride intracellular channel 1; CLIC2, chloride intracellular channel 2; CLIC3, chloride intracellular channel 3; CLIC4, chloride intracellular channel 4; CLIC5, chloride intracellular channel 5; CLIC6, chloride intracellular channel 6; ClCKA, kidney-specific chloride channel A; ClCKB, kidney-specific chloride channel B; CFTR, cystic fibrosis transmembrane conductance regulator; GABAARs, γ-aminobutyric acid type A receptors; IMAC, mitochondrial inner membrane anion channel; ORCC, outward rectifying Cl− channel; PAC, proton-activated Cl− channel; PAORAC/ASOR, acid-sensitive outwardly-rectifying anion channel; TMEM16A/ANO1, calcium-activated chloride channel ANO1/TMEM16A; TMEM16B/ANO2, calcium-activated chloride channel ANO2/TMEM16B; TMEM16F/ANO6, calcium-activated chloride channel ANO6/TMEM16F; VDAC1, voltage-dependent anion-selective channel 1; VDAC2, voltage-dependent anion-selective channel 2; VDAC3, voltage-dependent anion-selective channel 3; VRAC, volume-regulated anion channel; VSOR, volume-sensitive outwardly rectifying anion; VSOAC, volume-sensitive organic osmolyte/anion channel [4,67,68,84,98,103,122,123,126,135,136,139,140,144,145,165,168,176–194].
In the brain, Cl− was characterized for regulating the circadian rhythm [78]. Circadian rhythm is regulated by the suprachiasmatic nucleus (SCN), which predominantly comprises of GABAergic neurons. In SCN, GABAergic neurons elicit excitatory responses, which are facilitated by an increase in intracellular Cl− levels [79]. Also, the Cl− levels in cortical pyramidal neurons were found to be associated with the sleep–wake cycle [78]. During the sleep part of the cycle, Cl− levels decrease, but during the wake part of the cycle, the levels increase [78]. The increase in Cl− levels during wakefulness is associated with inhibitory synaptic transmission in the cortex [80]. In sleep-deprived animals, alterations in Cl− levels were found to be sufficient to correct the drop in their cognitive performance levels [80]. The major mechanism in this Cl−mediated sleep–wake regulation is the equilibrium potential for the GABAA receptor [80,81]. Decreasing Cl− to hyperpolarizing equilibrium potential for the GABAAR in animals deprived of sleep was sufficient to restore performance levels [80]. These findings indicate that targeting Cl− regulatory mechanisms could improve therapeutic effects in sleep disorders.
Chloride in the liver
In the liver, there is limited information available on the physiological role of Cl−. Cl− levels in newborns were found to be 55 mM, whereas in adults they were reported to be at 38.3 mM (Figure 1) [82,83]. Surprisingly, in the same tissue, although the cytosolic Cl− levels were found to be higher, these levels still showed a general decrease from 60 mM in newborns to 38 mM in adults [83]. The alteration in levels of Cl− could be attributed to the food or ion intake or to different expressions of ion channels and transporters in adults as compared with newborns. Additionally, mitochondria in the liver cells of newborns had approximately 5 mM of Cl−, approximately 30-fold lower than the cytosolic Cl− levels [83]. However, with age, the Cl− levels do not show as strong of an inverse trend in the mitochondria as observed for cytosolic Cl− levels [83]. Though there is a strong electrochemical gradient between the cytosol and mitochondria for Cl−, the levels indicate a tight regulation, possibly by ion channels and transporters [83].
Hepatocytes have Cl− channels in several intracellular compartments as well as at the plasma membrane [84]. The regulation of intracellular organelle acidification and cell volume depends on these channels [84]. Ca2+-activated Cl− channels have been found in the plasma membranes of hepatocytes [84]. The mitochondrial voltage-dependent anion channel, members of the newly discovered CLIC family of intracellular chloride channels (CLIC-1 and CLIC-4), members of the ClC channel family (ClC-2, ClC-3, ClC-5, and ClC-7), and a newly discovered intracellular channel, MCLC (Mid-1 related chloride channel), are among the Cl− channel molecules that have been demonstrated to be expressed in hepatocytes [11,83,85,86].
There has not been much research done on the significance of Cl− alterations for the prognosis of cirrhosis patients (Figure 1). In critically ill patients with decompensated cirrhosis, two independent studies found hypochloremia to be associated with short-term mortality, but not hyponatremia [87,88]. Interestingly, hypochloremia was found to be a more significant indicator of a patient’s prognosis than hyponatremia [89].
Chloride in the lungs
Cl− levels in the lungs are essential to maintaining membrane excitability, transepithelial transport, and homeostasis of ions as well as water [72]. The Cl− concentration in lung cells is maintained by a plethora of ion channels and transporters [90]. The earliest diagnosis involving Cl− was made for cystic fibrosis transmembrane conductance regulator (CFTR), a condition where the sweat of affected children tastes saltier than normal children [91]. In CFTR patients, there is a notable increase in Cl− levels of sweat to 60 mM as opposed to normal levels of 30 mM (Figure 1) [91]. If the Cl− is not moving in the correct direction, water is unable to hydrate the surface of cells. This causes thick and sticky mucus to cover the cells, resulting in many of the symptoms related to cystic fibrosis. In addition, patients with a chronic cough have been reported to have both reduced pH and Cl− levels [92].
In the lungs, Cl− and water move paracellularly to maintain both electroneutrality and osmotic balance [93]. Passive absorption of Cl− by various pathways is driven in response to the electrical driving force generated by active Na+ absorption. However, transepithelial Cl− transporters are implicated in active alveolar secretion and cardiogenic edema formation [93]. In airways surface the liquid Cl− concentration is approximately 123 mM [94], and in the airway epithelia, the range is from 30 to 50 mM [95]. Furthermore, it was shown that both transepithelial alveolar Cl− and fluid flux can reverse from an absorptive to a secretory mode in lung hydrostatic stress [93]. When Cl− was replaced with iso-osmolar NO3−, it attenuated alveolar fluid clearance [93]. Cl− must follow electroneutrality in lung cells [93]. Failure to maintain electroneutrality limits transepithelial Na+ flux, hence, affecting alveolar fluid clearance [93]. The idea of a significant role for transepithelial Cl− transport in alveolar fluid secretion is further supported by the fact that alveolar fluid secretion is prevented in Cl− free perfused lungs [93].
Cl− channels are highly expressed in the lung in both the lung parenchyma and the pulmonary blood vessels. They can develop pulmonary diseases (Figure 1) because of their compromised function or regulation [90]. The major challenges in the identification of Cl− channels and transporters are weak, non-selective inhibitors or a lack of genetic studies [9]. The major channels and transporters implicated in lung cells are TMEMs [96], cAMP-activated Cl−channels [97], ClC family [98], ligand-gated Cl− channels [99], SLC26 [100], CLIC4 [101,102], and CLIC6 [103].
Chloride in the muscles
Cl− regulates the excitability of muscle cells in skeletal muscles via their movements in and out of cells [32,104–107]. The electrical potential of the cells is stabilized by this flux, preventing abnormal muscle contraction. Although the resting Cl− conductance is not high, Cl− levels increase the excitability of cardiac cells in cardiac muscle, also known as the myocardium [108] (Figure 1).
Various vascular responses involve Cl− currents, indicating the existence Cl− channels such as transmembrane protein 16 (TMEM16)/anoctamin (ANO), bestrophins, voltage-gated Cl− channels (CLCs), cystic fibrosis (CF) transmembrane conductance regulator (CFTR) [109–112]. Vascular smooth muscle cells have been found to harbor all known Cl− channel families, with the exception of the GABA-/glycine-receptor family [109]. It has been proposed that at least one member of the voltage-activated ClC family, ClC-3, is involved in cell proliferation, myogenic constriction, and anti-apoptotic activity in rat vascular smooth muscle cells (VSMCs) [113]. VSMCs also exhibit the transmembrane conductance regulation associated with cystic fibrosis [114].
Myotonia congenita (MC), a genetic neuromuscular channelopathy, affects the skeletal muscle fibers, which are the striated muscles under the control of the somatic nervous system [115]. It is also associated with the abnormal functioning of Cl− channels such as ClC-1 (Figure 1) [32,116,117]. Myotonia, the disease’s hallmark, is defined as a delay or failure of relaxation in contracted skeletal muscle [115]. It causes prolonged rigidity, leading to cramping, stiffness, and muscle hypertrophy [115]. The CLCN1 gene, which codes for voltage-gated chloride (CIC-1) channels in the sarcolemmal membrane, is mutated in MC [116]. Repetitive depolarization and myotonia are caused by abnormal hyperexcitability of skeletal muscle cells due to defective CIC-1 channels [118].
In addition to VSMCs, Cl− channels have also been discovered in cardiac tissues. Levels of Cl− in the serum can determine the survival outcome after cardiac insults such as a heart attack or chronic heart failure [18–21,23]. Pharmacological and genetic approaches have indicated that IAA-94-sensitive Cl− channels such as chloride intracellular channels (CLICs), CLIC1, CLIC2, CLIC4, and CLIC5 are present in the cardiac tissue [119–123]. Blocking or absence of these channels increased myocardial infarction after ischemia and reperfusion injury [119,124–127]. Similarly, voltage-dependent anion channel (VDAC) ablation also results in dilated cardiomyopathy and cell death [128–130]. In skeletal muscle fibers, intracellular Cl− levels have a small potentiating effect on the Ca2+ release, which influences the cellular Ca2+ levels [131]. Pharmacological approaches have also implicated Cl− fluxes in charge compensation in smooth muscle cells [132]. It was further shown that different channels and transporters are involved in smooth and cardiac muscle cells [132].
Chloride in intracellular organelles
Cl− levels in the ECF are 110 mM, but in the cytosol, the levels are as low as 45 mM [133]. With the advent of new nano sensors and technologies, it is possible to quantify the absolute concentration of Cl− concentrations in various cellular compartments [133,134]. The Cl− concentration inside cellular organelles is tightly regulated for their physiological function [65]. The regulation is vital for maintaining ionic homeostasis and water concentrations. Different Cl− concentrations in different cellular compartments are provided in Figure 2.
Chloride ion channels and transporters
Cl− is moved across the cellular membrane through ion channels and transporters [135]. They are activated by pH, Ca2+, voltage, and volume [4]. After being ignored for several decades, Cl− channels and transporters have been discovered through the cloning of VDACs [136–138], ClC family [139,140], GABAA receptors [141–143], and CLIC proteins [144–146], as well as through the identification of mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) [147]. So far, over 53 Cl− transporting proteins have been identified [90]. These ion channels and transporters are associated with several human disorders or disease-like symptoms (Table 2) [148]. The major challenge in the Cl− channel and transport fields is the lack of pharmacological agents that can activate, block, inhibit, or facilitate membrane trafficking of these proteins. A major multidisciplinary effort is required to push for Cl− channels and transporters as drug candidates. Most of the Cl− channels and transporters are listed as potential drug targets that are not extensively studied [149]. Recently, a few Cl− transporters have been identified as targets of FDA-approved drugs [9,11]. For example, diuretics target SLC12 cation-Cl− co-transporters, which are used to reduce volume overload in hypertension and heart failure [150]. Barbiturates and benzodiazepines are known to target GABA-gated Cl− channels, and are commonly used for anxiety disorders, depression, and insomnia [151]. Ivacaftor was approved in 2012, and several correctors approved in 2015 for CFTR were highly specific steps to exclusively target Cl− channels [152]. More importantly, several drug candidates, such as acamprosate, alprazolam, bendroflumethiazide, benthiazide, bumetanide, butabarbital, butalbital, chlorothiazide, chlordiazepoxide, chlorthalidone, clobazam, clonazepam, clorazepic acid, crofelemer, cyclothiazide, desflurane, diazepam, enflurane, estazolam, eszopiclone, ethacrynic acid, ethchlorvynol, etomidate, flumazenil, flurazepam, furosemide, glutethimide, halazepam, halothane, hydrochlorothiazide, hydroflumethaiazide, indapamide, isoflurane, ivermectin, lindane, lorazepam, lubiprostone, lumacaftor, meprobamate, metharbital, methohexital, methoxyflurane, methyclothiazide, methyprylon, metolazone, midazolam, oxazepam, pentobarbital, polythiazide, prazepam, primidone, propofol, quazepam, quinethazone, secobarbital, sevoflurane, talbutal, temazepam, thiamylal, thiopental, tiagabine, topiramate, torsemide, triazolam, trichloromethiazide, triclofos, targeting Cl− channels, and transporters are listed with FDA clinical trial efforts [153]. There are several Cl− channels and transporters characterized as summarized in Table 2.
Perspectives
-
1.
Cl− are major anions in the body, and recent literature suggests that a decrease in Cl− levels in the body can result in detrimental effects [6,9,18,22,50,65,67,76,90,91,115,120,154,155]. A specific mechanism to increase Cl− in organs could improve the survival rate and the health of human beings.
-
2.
Cl− levels vary in different organ systems during development; however, there is no clear information on how these chloride ions are important in development and aging [3,4,9,23,75,94,107,112,120,133,156–161]. Recognition of variability in ion concentration during development and aging will facilitate novel targets for development-related pathological conditions.
-
3.
Cl− levels in organelles and cells are tightly regulated by ion channels and transporters. Identification and regulatory mechanisms of these channels and transporters hold the key to modulating cellular and extra-cellular Cl− levels [4,71,72,83,133,145,146,162–175].
Acknowledgements
The authors would like to thank Dr Raghvendra Baliga for key discussions on Cl in diseases, and Prof. Yamuna Krishnan (University of Chicago) for helpful discussions on various organellar chloride concentrations.
Abbreviations
- ANO
anoctamin
- CF
cystic fibrosis
- CFTR
CF transmembrane conductance regulator
- CLC
Cl− channel
- ECF
extracellular fluid
- NHE
Sodium Hydrogen antiporter 3
- PCT
proximal convoluted tubule
- TMEM16
transmembrane protein 16
- VDAC
voltage-dependent anion channel
- VSMC
vascular smooth muscle cell
Data Availability
Not applicable
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
Funding
S.S. is a recipient of the OSU Presidential Predoctoral Fellowship. V.C.L. is a recipient of an American Heart Association-Research Supplement to Promote Diversity in Science [grant number 23DIVSUP1074277]. S.G.R. is supported by the American Heart Association-Transformational Project Award [grant number 972077]. This work is supported by the National Centre for Advancing Translational Sciences [grant number TR004178] and, in part, by the National Heart, Lung, and Blood Institute [grant numbers HL133050 and HL157453] and the American Heart Association-Transformational Project Award [grant number 965301].
CRediT Author Contribution
Satish K Raut: Writing—original draft. Kulwinder Singh: Writing—original draft. Shridhar Sanghvi: Writing—original draft. Veronica Loyo-Celis: Writing—original draft. Liyah Varghese: Writing—original draft. Ekam R Singh: Writing—original draft. Shubha Gururaja Rao: Resources, Formal analysis, Supervision, Funding acquisition, Writing—original draft, Project administration. Harpreet Singh: Conceptualization, Resources, Formal analysis, Supervision, Funding acquisition, Project administration, Writing—review & editing.
References
- 1.Wang W., Zhang X., Gao Q., Lawas M., Yu L., Cheng X.et al. (2017) A voltage-dependent K(+) channel in the lysosome is required for refilling lysosomal Ca(2+) stores. J. Cell Biol. 216, 1715–1730, Epub 2017/05/05. PubMed Central PMCID: PMCPMC5461029 10.1083/jcb.201612123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Berend K., van Hulsteijn L.H. and Gans R.O. (2012) Chloride: the queen of electrolytes? Eur. J. Int. Med. 23, 203–211 10.1016/j.ejim.2011.11.013 [DOI] [PubMed] [Google Scholar]
- 3.Astapenko D., Navratil P., Pouska J. and Cerny V. (2020) Clinical physiology aspects of chloremia in fluid therapy: a systematic review. Perioper Med. (Lond.) 9, 40, Epub 2020/12/11. PubMed Central PMCID: PMCPMC7727154 10.1186/s13741-020-00171-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gururaja Rao S., Patel N.J. and Singh H. (2020) Intracellular chloride channels: novel biomarkers in diseases. Front. Physiol. 11, 96, Epub 2020/03/03. PubMed Central PMCID: PMCPMC7034325 10.3389/fphys.2020.00096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McCallum L., Lip S. and Padmanabhan S. (2015) The hidden hand of chloride in hypertension. Pflugers Arch. 467, 595–603, PubMed Central PMCID: PMC4325190 10.1007/s00424-015-1690-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Elborn J.S. (2016) Cystic fibrosis. Lancet 388, 2519–2531 10.1016/S0140-6736(16)00576-6 [DOI] [PubMed] [Google Scholar]
- 7.Vaduganathan M., Pallais J.C., Fenves A.Z., Butler J. and Gheorghiade M. (2016) Serum chloride in heart failure: a salty prognosis. Eur. J. Heart Fail. 18, 669–671, Epub 2016/04/29 10.1002/ejhf.546 [DOI] [PubMed] [Google Scholar]
- 8.Efsa Panel on Nutrition NF Food A., Turck D., Castenmiller J., de Henauw S., Hirsch-Ernst K.I.et al. (2019) Dietary reference values for chloride. EFSA J. 17, e05779, Epub 2020/07/07. PubMed Central PMCID: PMCPMC7009052 10.2903/j.efsa.2019.5779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Verkman A.S. and Galietta L.J.V. (2021) Chloride transport modulators as drug candidates. Am. J. Physiol. Cell Physiol. 321, C932–C946, Epub 2021/10/14. PubMed Central PMCID: PMCPMC8714991 10.1152/ajpcell.00334.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Powers F. (1999) The role of chloride in acid-base balance. J. Intraven. Nurs. 22, 286–291, Epub 2000/04/25. PubMed PMID: 10776193 [PubMed] [Google Scholar]
- 11.Verkman A.S. and Galietta L.J. (2009) Chloride channels as drug targets. Nat. Rev. Drug Discovery 8, 153–171, Epub 2009/01/21. PubMed Central PMCID: PMCPMC3601949 10.1038/nrd2780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mason J.B. (2012) Vitamins, trace minerals, and other micronutrients. Goldman's Cecil Medicine 2, 24th ed, 1397–1406 [Google Scholar]
- 13.Mason J.B. and Booth S.L. (2020) Vitamins, trace minerals, and other micronutrients. Goldman-Cecil Medicine, 26th edElsevier [Google Scholar]
- 14.Ayling R.M. and Crook M. (2022) Tietz Textbook of Laboratory Medicine, 7th ed, Saunders [Google Scholar]
- 15.Tello L. and Perez-Freytes R. (2017) Fluid and electrolyte therapy during vomiting and diarrhea. Vet. Clin. North Am. Small Anim. Pract. 47, 505–519, Epub 2016/12/13. PubMed Central PMCID: PMCPMC7185384 10.1016/j.cvsm.2016.09.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yu X.M., Groveman B.R., Fang X.Q. and Lin S.X. (2010) The role of intracellular sodium (Na) in the regulation of calcium (Ca)-mediated signaling and toxicity. Health (Irvine Calif) 2, 8–15, Epub 2011/01/19. PubMed Central PMCID: PMCPMC3020095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zandijk A.J.L., van Norel M.R., Julius F.E.C., Sepehrvand N., Pannu N., McAlister F.A.et al. (2021) Chloride in heart failure: the neglected electrolyte. JACC Heart Fail. 9, 904–915, Epub 2021/12/04 10.1016/j.jchf.2021.07.006 [DOI] [PubMed] [Google Scholar]
- 18.Grodin J.L., Verbrugge F.H., Ellis S.G., Mullens W., Testani J.M. and Tang W.H. (2016) Importance of abnormal chloride homeostasis in stable chronic heart failure. Circ Heart Fail. 9, e002453, Epub 2016/01/02. PubMed Central PMCID: PMCPMC4702267 10.1161/CIRCHEARTFAILURE.115.002453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hanberg J.S., Rao V., Ter Maaten J.M., Laur O., Brisco M.A., Perry Wilson F.et al. (2016) Hypochloremia and diuretic resistance in heart failure: mechanistic insights. Circ Heart Fail. 9, Epub 2016/08/11. PubMed Central PMCID: PMCPMC4988527 10.1161/CIRCHEARTFAILURE.116.003180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ter Maaten J.M., Damman K., Hanberg J.S., Givertz M.M., Metra M., O'Connor C.M.et al. (2016) Hypochloremia, diuretic resistance, and outcome in patients with acute heart failure. Circ Heart Fail. 9, e003109.Epub 2016/08/11 10.1161/CIRCHEARTFAILURE.116.003109 [DOI] [PubMed] [Google Scholar]
- 21.Testani J.M., Hanberg J.S., Arroyo J.P., Brisco M.A., Ter Maaten J.M., Wilson F.P.et al. (2016) Hypochloraemia is strongly and independently associated with mortality in patients with chronic heart failure. Eur. J. Heart Fail. 18, 660–668, Epub 2016/01/15. PubMed Central PMCID: PMCPMC5471359 10.1002/ejhf.477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ellison D.H. and Felker G.M. (2017) Diuretic treatment in heart failure. N. Engl. J. Med. 377, 1964–1975, Epub 2017/11/16. PubMed Central PMCID: PMCPMC5811193 10.1056/NEJMra1703100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cuthbert J.J., Pellicori P., Rigby A., Pan D., Kazmi S., Shah P.et al. (2018) Low serum chloride in patients with chronic heart failure: clinical associations and prognostic significance. Eur. J. Heart Fail. 20, 1426–1435, Epub 2018/06/27 10.1002/ejhf.1247 [DOI] [PubMed] [Google Scholar]
- 24.Smith M.E. and Morton D.G. (2010) The stomach: Basic functions. Digest. Syst. 39–50 10.1016/B978-0-7020-3367-4.00003-7 [DOI] [Google Scholar]
- 25.Kararli T.T. (1995) Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm. Drug Dispos. 16, 351–380, Epub 1995/07/01 10.1002/bdd.2510160502 [DOI] [PubMed] [Google Scholar]
- 26.Pfortmueller C.A., Uehlinger D., von Haehling S. and Schefold J.C. (2018) Serum chloride levels in critical illness-the hidden story. Intensive Care Med. Exp. 6, 10, Epub 2018/04/15. PubMed Central PMCID: PMCPMC5899079 10.1186/s40635-018-0174-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yunos N.M., Bellomo R., Story D. and Kellum J. (2010) Bench-to-bedside review: Chloride in critical illness. Crit. Care 14, 226, Epub 2010/07/29. PubMed Central PMCID: PMCPMC2945073 10.1186/cc9052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Glykys J., Dzhala V., Egawa K., Balena T., Saponjian Y., Kuchibhotla K.V.et al. (2014) Local impermeant anions establish the neuronal chloride concentration. Science 343, 670–675, Epub 2014/02/08. PubMed Central PMCID: PMCPMC4220679 10.1126/science.1245423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yamada J., Okabe A., Toyoda H., Kilb W., Luhmann H.J. and Fukuda A. (2004) Cl- uptake promoting depolarizing GABA actions in immature rat neocortical neurones is mediated by NKCC1. J. Physiol. 557, 829–841, Epub 2004/04/20. PubMed Central PMCID: PMCPMC1665166 10.1113/jphysiol.2004.062471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Westen E.A. and Prange H.D. (2003) A reexamination of the mechanisms underlying the arteriovenous chloride shift. Physiol. Biochem. Zool. 76, 603–614, Epub 2003/12/13 10.1086/380208 [DOI] [PubMed] [Google Scholar]
- 31.Brix O., Thomsen B., Nuutinen M., Hakala A., Pudas J. and Giardina B. (1990) The chloride shift may facilitate oxygen loading and unloading to/from the hemoglobin from the brown bear (Ursus arctos L.). Comp. Biochem. Physiol. B. 95, 865–868, Epub 1990/01/01 10.1016/0305-0491(90)90330-V [DOI] [PubMed] [Google Scholar]
- 32.Puljak L. and Kilic G. (2006) Emerging roles of chloride channels in human diseases. Biochim. Biophys. Acta 1762, 404–413, Epub 2006/02/07 10.1016/j.bbadis.2005.12.008 [DOI] [PubMed] [Google Scholar]
- 33.Nagami G.T. (2016) Hyperchloremia - why and how. Nefrologia 36, 347–353, Epub 2016/06/09 10.1016/j.nefro.2016.04.001 [DOI] [PubMed] [Google Scholar]
- 34.Christov M. and Alper S.L. (2010) Tubular transport: core curriculum 2010. Am. J. Kidney Dis. 56, 1202–1217, Epub 2010/11/03. PubMed Central PMCID: PMCPMC3018279 10.1053/j.ajkd.2010.09.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kiela P.R. and Ghishan F.K. (2016) Physiology of intestinal absorption and secretion. Best Pract. Res. Clin. Gastroenterol. 30, 145–159, Epub 2016/04/19. PubMed Central PMCID: PMCPMC4956471 10.1016/j.bpg.2016.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Iacobelli S., Kermorvant-Duchemin E., Bonsante F., Lapillonne A. and Gouyon J.B. (2012) Chloride balance in preterm infants during the first week of life. Int. J. Pediatr. 2012, 931597, Epub 2012/04/17. PubMed Central PMCID: PMCPMC3312278 10.1155/2012/931597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bichet D.G. and Fujiwara T.M. (2004) Reabsorption of sodium chloride–lessons from the chloride channels. N. Engl. J. Med. 350, 1281–1283, Epub 2004/03/27 10.1056/NEJMp048026 [DOI] [PubMed] [Google Scholar]
- 38.Wieth J.O., Andersen O.S., Brahm J., Bjerrum P.J. and Borders C.L. Jr (1982) Chloride–bicarbonate exchange in red blood cells: physiology of transport and chemical modification of binding sites. Philos. Trans. R. Soc. Lond. B Biol. Sci. 299, 383–399, Epub 1982/12/01 [DOI] [PubMed] [Google Scholar]
- 39.Verlander J.W. (2020) Solute reabsorption. Cunningham's Textbook of Veterinary Physiology. 489–500, 10.1016/B978-0-323-55227-1.00042-9 [DOI] [Google Scholar]
- 40.Morrison G. (1990) Serum Chloride. In Clinical Methods: The History, Physical, and Laboratory Examinations, 3rd ed(Walker H.K., Hall W.D. and Hurst J.W., eds), Boston: [PubMed] [Google Scholar]
- 41.Xu J., Barone S., Li H., Holiday S., Zahedi K. and Soleimani M. (2011) Slc26a11, a chloride transporter, localizes with the vacuolar H(+)-ATPase of A-intercalated cells of the kidney. Kidney Int. 80, 926–937, Epub 2011/07/01 10.1038/ki.2011.196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Romero M.F., Chen A.P., Parker M.D. and Boron W.F. (2013) The SLC4 family of bicarbonate (HCO(3)(-)) transporters. Mol. Aspects Med. 34, 159–182, Epub 2013/03/20. PubMed Central PMCID: PMCPMC3605756 10.1016/j.mam.2012.10.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Leiz J. and Schmidt-Ott K.M. (2019) Claudins in the renal collecting duct. Int. J. Mol. Sci. 21, 221, Epub 2020/01/08. PubMed Central PMCID: PMCPMC6981911 10.3390/ijms21010221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Good D.W. (2007) Nongenomic actions of aldosterone on the renal tubule. Hypertension 49, 728–739, Epub 2007/02/21 10.1161/01.HYP.0000259797.48382.b2 [DOI] [PubMed] [Google Scholar]
- 45.Salyer S.A., Parks J., Barati M.T., Lederer E.D., Clark B.J., Klein J.D.et al. (2013) Aldosterone regulates Na(+), K(+) ATPase activity in human renal proximal tubule cells through mineralocorticoid receptor. Biochim. Biophys. Acta 1833, 2143–2152, Epub 2013/05/21 10.1016/j.bbamcr.2013.05.009 [DOI] [PubMed] [Google Scholar]
- 46.Vincent-Johnson A. and Scialla J.J. (2022) Importance of metabolic acidosis as a health risk in chronic kidney disease. Adv. Chronic Kidney Dis. 29, 329–336, Epub 2022/09/30 10.1053/j.ackd.2022.05.002 [DOI] [PubMed] [Google Scholar]
- 47.Kim H.J. (2021) Metabolic acidosis in chronic kidney disease: pathogenesis, clinical consequences, and treatment. Electrolyte Blood Press. 19, 29–37, Epub 2022/01/11. PubMed Central PMCID: PMCPMC8715222 10.5049/EBP.2021.19.2.29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.de Morais H.A., Bach J.F. and DiBartola S.P. (2008) Metabolic acid-base disorders in the critical care unit. Vet. Clin. North Am. Small Anim. Pract. 38, 559–574, x-xi. Epub 2008/04/12 10.1016/j.cvsm.2008.02.003 [DOI] [PubMed] [Google Scholar]
- 49.Kim H.Y. (2009) Renal handling of ammonium and acid base regulation. Electrolyte Blood Press. 7, 9–13, Epub 2009/06/01. PubMed Central PMCID: PMCPMC3041479 10.5049/EBP.2009.7.1.9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sharma S., Hashmi M.F. and Aggarwal S. (2023) Hyperchloremic acidosis. StatPearls, Treasure Island (FL) ineligible companies, Disclosure: Muhammad Hashmi declares no relevant financial relationships with ineligible companies. Disclosure: Sandeep Aggarwal declares no relevant financial relationships with ineligible companies. [PubMed] [Google Scholar]
- 51.Sumarsono A., Wang J., Xie L., Chiang G.C., Tielleman T., Messiah S.E.et al. (2020) Prognostic value of hypochloremia in critically ill patients with decompensated cirrhosis. Crit. Care Med. 48, e1054–e1061, Epub 2020/09/19 10.1097/CCM.0000000000004620 [DOI] [PubMed] [Google Scholar]
- 52.Bucking C. and Wood C.M. (2006) Gastrointestinal processing of Na+, Cl-, and K+ during digestion: implications for homeostatic balance in freshwater rainbow trout. Am. J. Physiol. Regul. Integr. Comp. Physiol. 291, R1764–R1772, Epub 2006/08/12 10.1152/ajpregu.00224.2006 [DOI] [PubMed] [Google Scholar]
- 53.Martinsen T.C., Fossmark R. and Waldum H.L. (2019) The phylogeny and biological function of gastric juice-microbiological consequences of removing gastric acid. Int. J. Mol. Sci. 20, 6031, Epub 2019/12/05. PubMed Central PMCID: PMCPMC6928904 10.3390/ijms20236031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Heda R., Toro F. and Tombazzi C.R. (2024) Physiology, Pepsin. StatPearls, Treasure Island (FL) ineligible companies, Disclosure: Fadi Toro declares no relevant financial relationships with ineligible companies. Disclosure: Claudio Tombazzi declares no relevant financial relationships with ineligible companies. [PubMed] [Google Scholar]
- 55.Bartle H.J. and Harkins M.J. (1925) The gastric secretion: its bactericidal value to man. Am. J. Med. Sci. 169, 373–388 10.1097/00000441-192503000-00008 [DOI] [Google Scholar]
- 56.Schermer L. (1988) Physiologic and technical variables affecting hemodynamic measurements. Crit. Care Nurse 8, 33–40, 2. Epub 1988/03/01. PubMed PMID: 3077613 10.4037/ccn1988.8.2.33 [DOI] [PubMed] [Google Scholar]
- 57.Engevik A.C., Kaji I. and Goldenring J.R. (2020) The physiology of the gastric parietal cell. Physiol. Rev. 100, 573–602, Epub 2019/11/02. PubMed Central PMCID: PMCPMC7327232 10.1152/physrev.00016.2019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Murek M., Kopic S. and Geibel J. (2010) Evidence for intestinal chloride secretion. Exp. Physiol. 95, 471–478, Epub 2010/03/18 10.1113/expphysiol.2009.049445 [DOI] [PubMed] [Google Scholar]
- 59.Kopic S. and Geibel J.P. (2010) Update on the mechanisms of gastric acid secretion. Curr. Gastroenterol. Rep. 12, 458–464, Epub 2010/09/08 10.1007/s11894-010-0137-9 [DOI] [PubMed] [Google Scholar]
- 60.Barrett K.E. and Keely S.J. (2000) Chloride secretion by the intestinal epithelium: molecular basis and regulatory aspects. Annu. Rev. Physiol. 62, 535–572, Epub 2000/06/09 10.1146/annurev.physiol.62.1.535 [DOI] [PubMed] [Google Scholar]
- 61.Keely S.J. and Barrett K.E. (2022) Intestinal secretory mechanisms and diarrhea. Am. J. Physiol. Gastrointest. Liver Physiol. 322, G405–G420, Epub 2022/02/17. PubMed Central PMCID: PMCPMC8917926 10.1152/ajpgi.00316.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Cairns C.B. (1998) Apoptosis: only the good die young? Acad. Emerg. Med. 5, 948–950, Epub 1998/12/23 10.1111/j.1553-2712.1998.tb02768.x [DOI] [PubMed] [Google Scholar]
- 63.Zhou Z., Zhan J., Cai Q., Xu F., Chai R., Lam K.et al. (2022) The water transport system in astrocytes-aquaporins. Cells 11, 2564, Epub 2022/08/27. PubMed Central PMCID: PMCPMC9406552 10.3390/cells11162564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tait M.J., Saadoun S., Bell B.A. and Papadopoulos M.C. (2008) Water movements in the brain: role of aquaporins. Trends Neurosci. 31, 37–43, Epub 2007/12/07 10.1016/j.tins.2007.11.003 [DOI] [PubMed] [Google Scholar]
- 65.Rahmati N., Hoebeek F.E., Peter S. and De Zeeuw C.I. (2018) Chloride homeostasis in neurons with special emphasis on the olivocerebellar system: differential roles for transporters and channels. Front Cell Neurosci. 12, 101, Epub 2018/05/17. PubMed Central PMCID: PMCPMC5938380 10.3389/fncel.2018.00101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Langer T., Zadek F., Carbonara M., Caccioppola A., Brusatori S., Zoerle T.et al. (2022) Cerebrospinal fluid and arterial acid-base equilibrium of spontaneously breathing patients with aneurismal subarachnoid hemorrhage. Neurocrit Care 37, 102–110, Epub 2022/02/25. PubMed Central PMCID: PMCPMC9283163 10.1007/s12028-022-01450-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Elorza-Vidal X., Gaitan-Penas H. and Estevez R. (2019) Chloride channels in astrocytes: structure, roles in brain homeostasis and implications in disease. Int. J. Mol. Sci. 20, 1034, Epub 2019/03/02. PubMed Central PMCID: PMCPMC6429410 10.3390/ijms20051034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Singh H. and Ashley R.H. (2006) Redox regulation of CLIC1 by cysteine residues associated with the putative channel pore. Biophys. J. 90, 1628–1638, PubMed Central PMCID: PMC1367314 10.1529/biophysj.105.072678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Milton R.H., Abeti R., Averaimo S., DeBiasi S., Vitellaro L., Jiang L.et al. (2008) CLIC1 function is required for beta-amyloid-induced generation of reactive oxygen species by microglia. J. Neurosci. 28, 11488–11499 10.1523/JNEUROSCI.2431-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Skaper S.D. (2011) Ion channels on microglia: therapeutic targets for neuroprotection. CNS Neurol Disord Drug Targets 10, 44–56, Epub 2010/12/15 10.2174/187152711794488638 [DOI] [PubMed] [Google Scholar]
- 71.Marunaka Y. (2023) Physiological roles of chloride ions in bodily and cellular functions. J Physiol Sci. 73, 31, Epub 2023/11/16. PubMed Central PMCID: PMCPMC10717538 10.1186/s12576-023-00889-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Martinez A.H. and Mohiuddin S.S. (2024) Biochemistry, chloride channels. StatPearls, Treasure Island (FL) ineligible companies, Disclosure: Shamim Mohiuddin declares no relevant financial relationships with ineligible companies. [PubMed] [Google Scholar]
- 73.Abruzzo P.M., Panisi C. and Marini M. (2021) The Alteration of Chloride Homeostasis/GABAergic Signaling in Brain Disorders: Could Oxidative Stress Play a Role? Antioxidants (Basel) 10, Epub 2021/08/28. PubMed Central PMCID: PMCPMC8389245 10.3390/antiox10081316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Allen M.J., Sabir S. and Sharma S. (2024) GABA Receptor. StatPearls, Treasure Island (FL) ineligible companies, Disclosure: Sarah Sabir declares no relevant financial relationships with ineligible companies. Disclosure: Sandeep Sharma declares no relevant financial relationships with ineligible companies. [PubMed] [Google Scholar]
- 75.Cherubini E., Di Cristo G. and Avoli M. (2021) Dysregulation of GABAergic signaling in neurodevelomental disorders: targeting cation-chloride co-transporters to re-establish a proper E/I balance. Front Cell Neurosci. 15, 813441, Epub 2022/01/25. PubMed Central PMCID: PMCPMC8766311 10.3389/fncel.2021.813441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fang X., Lu Y., Fu Y., Liu Z., Kermode A.G., Qiu W.et al. (2023) Cerebrospinal fluid chloride is associated with disease activity of relapsing-remitting multiple sclerosis: a retrospective cohort study. Brain Sci. 13, 924, Epub 2023/06/28. PubMed Central PMCID: PMCPMC10296568 10.3390/brainsci13060924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Schoen E.J. (1984) Spinal fluid chloride: a test 40 years past its time. JAMA 251, 37–38, Epub 1984/01/06 10.1001/jama.1984.03340250021008 [DOI] [PubMed] [Google Scholar]
- 78.Lodovichi C. and Ratto G.M. (2023) Control of circadian rhythm on cortical excitability and synaptic plasticity. Front Neural. Circuits 17, 1099598, Epub 2023/04/18. PubMed Central PMCID: PMCPMC10098176 10.3389/fncir.2023.1099598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Salihu S., Meor Azlan N.F., Josiah S.S., Wu Z., Wang Y. and Zhang J. (2021) Role of the cation-chloride-cotransporters in the circadian system. Asian J. Pharm. Sci. 16, 589–597, Epub 2021/12/02. PubMed Central PMCID: PMCPMC8609385 10.1016/j.ajps.2020.10.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Alfonsa H., Burman R.J., Brodersen P.J.N., Newey S.E., Mahfooz K., Yamagata T.et al. (2023) Intracellular chloride regulation mediates local sleep pressure in the cortex. Nat. Neurosci. 26, 64–78, Epub 2022/12/13. PubMed Central PMCID: PMCPMC7614036 10.1038/s41593-022-01214-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sorensen A.T., Ledri M., Melis M., Nikitidou Ledri L., Andersson M. and Kokaia M. (2017) Altered chloride homeostasis decreases the action potential threshold and increases hyperexcitability in hippocampal neurons. eNeuro 4, Epub 2018/01/31. PubMed Central PMCID: PMCPMC5783240 10.1523/ENEURO.0172-17.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Widdowson E.M. and Dickerson J.W. (1960) The effect of growth and function on the chemical composition of soft tissues. Biochem. J. 77, 30–43, Epub 1960/10/01. PubMed Central PMCID: PMCPMC1204895 10.1042/bj0770030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Jahn S.C., Rowland-Faux L., Stacpoole P.W. and James M.O. (2015) Chloride concentrations in human hepatic cytosol and mitochondria are a function of age. Biochem. Biophys. Res. Commun. 459, 463–468, Epub 2015/03/10. PubMed Central PMCID: PMCPMC4380591 10.1016/j.bbrc.2015.02.128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Li X. and Weinman S.A. (2002) Chloride channels and hepatocellular function: prospects for molecular identification. Annu. Rev. Physiol. 64, 609–633, Epub 2002/02/05 10.1146/annurev.physiol.64.090501.145429 [DOI] [PubMed] [Google Scholar]
- 85.Azzi A. and Azzone G.F. (1967) Swelling and shrinkage phenomena in liver mitochondria. VI. Metabolism-independent swelling coupled to ion movement. Biochim. Biophys. Acta 131, 468–478, Epub 1967/05/09 10.1016/0005-2728(67)90006-0 [DOI] [PubMed] [Google Scholar]
- 86.Redhead C., Sullivan S.K., Koseki C., Fujiwara K. and Edwards J.C. (1997) Subcellular distribution and targeting of the intracellular chloride channel p64. Mol. Biol. Cell. 8, 691–704, Epub 1997/04/01. PubMed Central PMCID: PMCPMC276119 10.1091/mbc.8.4.691 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Semmler G., Scheiner B., Balcar L., Paternostro R., Simbrunner B., Pinter M.et al. (2023) Disturbances in sodium and chloride homeostasis predict outcome in stable and critically ill patients with cirrhosis. Aliment. Pharmacol. Ther. 58, 71–79, Epub 2023/04/06 10.1111/apt.17507 [DOI] [PubMed] [Google Scholar]
- 88.Ji Y. and Li L. (2021) Lower serum chloride concentrations are associated with increased risk of mortality in critically ill cirrhotic patients: an analysis of the MIMIC-III database. BMC Gastroenterol. 21, 200, Epub 2021/05/03. PubMed Central PMCID: PMCPMC8088682 10.1186/s12876-021-01797-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kubota K., Sakaguchi Y., Hamano T., Oka T., Yamaguchi S., Shimada K.et al. (2020) Prognostic value of hypochloremia versus hyponatremia among patients with chronic kidney disease-a retrospective cohort study. Nephrol. Dial. Transplant. 35, 987–994, Epub 2018/10/23 10.1093/ndt/gfy299 [DOI] [PubMed] [Google Scholar]
- 90.Sinha M., Zabini D., Guntur D., Nagaraj C., Enyedi P., Olschewski H.et al. (2022) Chloride channels in the lung: Challenges and perspectives for viral infections, pulmonary arterial hypertension, and cystic fibrosis. Pharmacol. Therapeut. 237, 108249, Epub 2022/07/26 10.1016/j.pharmthera.2022.108249 [DOI] [PubMed] [Google Scholar]
- 91.Caudri D., Zitter D., Bronsveld I. and Tiddens H. (2017) Is sweat chloride predictive of severity of cystic fibrosis lung disease assessed by chest computed tomography? Pediatr. Pulmonol. 52, 1135–1141, Epub 2017/06/07 10.1002/ppul.23739 [DOI] [PubMed] [Google Scholar]
- 92.Niimi A., Nguyen L.T., Usmani O., Mann B. and Chung K.F. (2004) Reduced pH and chloride levels in exhaled breath condensate of patients with chronic cough. Thorax 59, 608–612, Epub 2004/06/30. PubMed Central PMCID: PMCPMC1747079 10.1136/thx.2003.012906 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Solymosi E.A., Kaestle-Gembardt S.M., Vadasz I., Wang L., Neye N., Chupin C.J.et al. (2013) Chloride transport-driven alveolar fluid secretion is a major contributor to cardiogenic lung edema. PNAS 110, E2308–E2316, Epub 2013/05/07. PubMed Central PMCID: PMCPMC3690871 10.1073/pnas.1216382110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Song Y., Thiagarajah J. and Verkman A.S. (2003) Sodium and chloride concentrations, pH, and depth of airway surface liquid in distal airways. J. Gen. Physiol. 122, 511–519, Epub 2003/10/15. PubMed Central PMCID: PMCPMC2229580 10.1085/jgp.200308866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Uyekubo S.N., Fischer H., Maminishkis A., Illek B., Miller S.S. and Widdicombe J.H. (1998) cAMP-dependent absorption of chloride across airway epithelium. Am. J. Physiol. 275, L1219–L1227, Epub 1998/12/09 10.1152/ajplung.1998.275.6.L1219 [DOI] [PubMed] [Google Scholar]
- 96.Zhang S., Dai H., Li W., Wang R., Wu H., Shen M.et al. (2021) TMEM116 is required for lung cancer cell motility and metastasis through PDK1 signaling pathway. Cell Death Dis. 12, 1086, Epub 2021/11/19. PubMed Central PMCID: PMCPMC8599864 10.1038/s41419-021-04369-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Anderson M.P. and Welsh M.J. (1991) Calcium and cAMP activate different chloride channels in the apical membrane of normal and cystic fibrosis epithelia. Proc. Natl. Acad. Sci. USA 88, 6003–6007, Epub 1991/07/15. PubMed Central PMCID: PMCPMC52010 10.1073/pnas.88.14.6003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mummery J.L., Killey J. and Linsdell P. (2005) Expression of the chloride channel CLC-K in human airway epithelial cells. Can. J. Physiol. Pharmacol. 83, 1123–1128, Epub 2006/02/08 10.1139/y05-112 [DOI] [PubMed] [Google Scholar]
- 99.Jin N., Guo Y., Sun P., Bell A., Chintagari N.R., Bhaskaran M.et al. (2008) Ionotropic GABA receptor expression in the lung during development. Gene Expr. Patterns 8, 397–403, Epub 2008/06/10. PubMed Central PMCID: PMCPMC2581461 10.1016/j.gep.2008.04.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Nofziger C., Dossena S., Suzuki S., Izuhara K. and Paulmichl M. (2011) Pendrin function in airway epithelia. Cell. Physiol. Biochem. Int. J. Experiment. Cell. Physiol. Biochem. Pharmacol. 28, 571–578, Epub 2011/11/26 10.1159/000335115 [DOI] [PubMed] [Google Scholar]
- 101.Abdul-Salam V.B., Russomanno G., Chien-Nien C., Mahomed A.S., Yates L.A., Wilkins M.R.et al. (2019) CLIC4/Arf6 Pathway. Circ. Res. 124, 52–65, Epub 2018/12/26. PubMed Central PMCID: PMCPMC6325770 10.1161/CIRCRESAHA.118.313705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wojciak-Stothard B., Abdul-Salam V.B., Lao K.H., Tsang H., Irwin D.C., Lisk C.et al. (2014) Aberrant chloride intracellular channel 4 expression contributes to endothelial dysfunction in pulmonary arterial hypertension. Circulation 129, 1770–1780, PubMed Central PMCID: PMC4033409 10.1161/CIRCULATIONAHA.113.006797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Loyo-Celis V., Patel D., Sanghvi S., Kaur K., Ponnalagu D., Zheng Y.et al. (2023) Biophysical characterization of chloride intracellular channel 6 (CLIC6). J. Biol. Chem. 299, 105349, 10.1016/j.jbc.2023.105349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hutter O.F. and Noble D. (1961) Anion conductance of cardiac muscle. J. Physiol. 157, 335–350, Epub 1961/07/01. PubMed Central PMCID: PMCPMC1359956 10.1113/jphysiol.1961.sp006726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Nelson M.T., Conway M.A., Knot H.J. and Brayden J.E. (1997) Chloride channel blockers inhibit myogenic tone in rat cerebral arteries. J. Physiol. 502, 259–264, Epub 1997/07/15. PubMed Central PMCID: PMCPMC1159547 10.1111/j.1469-7793.1997.259bk.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Weber-Schurholz S., Wischmeyer E., Laurien M., Jockusch H., Schurholz T., Landry D.W.et al. (1993) Indanyloxyacetic acid-sensitive chloride channels from outer membranes of skeletal muscle. J. Biol. Chem. 268, 547–551, PubMed PMID: 7678003 10.1016/S0021-9258(18)54186-7 [DOI] [PubMed] [Google Scholar]
- 107.Boedtkjer E., Matchkov V.V., Boedtkjer D.M. and Aalkjaer C. (2016) Negative News: Cl- and HCO3- in the Vascular Wall. Physiology (Bethesda). 31, 370–383 10.1152/physiol.00001.2016 [DOI] [PubMed] [Google Scholar]
- 108.Vaughan-Jones R.D. (1982) Chloride activity and its control in skeletal and cardiac muscle. Philos. Trans. R. Soc. Lond. B Biol. Sci. 299, 537–548, Epub 1982/12/01 10.1098/rstb.1982.0150 [DOI] [PubMed] [Google Scholar]
- 109.Bulley S. and Jaggar J.H. (2014) Cl(-) channels in smooth muscle cells. Pflugers Archiv: Eur. J. Physiol. 466, 861–872, PubMed Central PMCID: PMC3969453 10.1007/s00424-013-1357-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Duan D.Y., Liu L.L., Bozeat N., Huang Z.M., Xiang S.Y., Wang G.L.et al. (2005) Functional role of anion channels in cardiac diseases. Acta Pharmacol. Sin. 26, 265–278 10.1111/j.1745-7254.2005.00061.x [DOI] [PubMed] [Google Scholar]
- 111.Duan D.D. (2013) Phenomics of cardiac chloride channels. Comprehens. Physiol. 3, 667–692, PubMed Central PMCID: PMC3966620 10.1002/cphy.c110014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Duan D., Ye L., Britton F., Horowitz B. and Hume J.R. (2000) A novel anionic inward rectifier in native cardiac myocytes. Circ. Res. 86, E63–E71, PubMed PMID: 10700456 10.1161/01.RES.86.4.e63 [DOI] [PubMed] [Google Scholar]
- 113.Duan D.D. (2011) The ClC-3 chloride channels in cardiovascular disease. Acta Pharmacol. Sin. 32, 675–684, Epub 2011/05/24. PubMed Central PMCID: PMCPMC3417079 10.1038/aps.2011.30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Zeng J.W., Zeng X.L., Li F.Y., Ma M.M., Yuan F., Liu J.et al. (2014) Cystic fibrosis transmembrane conductance regulator (CFTR) prevents apoptosis induced by hydrogen peroxide in basilar artery smooth muscle cells. Apoptosis 19, 1317–1329, Epub 2014/07/08 10.1007/s10495-014-1014-z [DOI] [PubMed] [Google Scholar]
- 115.Bryan E.S. and Alsaleem M. (2024) Myotonia congenita. StatPearls, Treasure Island (FL) ineligible companies, Disclosure: Mahdi Alsaleem declares no relevant financial relationships with ineligible companies. [PubMed] [Google Scholar]
- 116.Stolting G., Fischer M. and Fahlke C. (2014) CLC channel function and dysfunction in health and disease. Front. Physiol. 5, 378, PubMed Central PMCID: PMC4188032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Tang C.Y. and Chen T.Y. (2011) Physiology and pathophysiology of CLC-1: mechanisms of a chloride channel disease, myotonia. J. Biomed. Biotechnol. 2011, 685328, Epub 2011/12/22. PubMed Central PMCID: PMCPMC3237021 10.1155/2011/685328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Cannon S.C. (2015) Channelopathies of skeletal muscle excitability. Comprehens. Physiol. 5, 761–790, Epub 2015/04/17. PubMed Central PMCID: PMCPMC4754081 10.1002/cphy.c140062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Ponnalagu D., Hamilton S., Sanghvi S., Antelo D., Schwieterman N., Hansra I.et al. (2022) CLIC4 localizes to mitochondrial-associated membranes and mediates cardioprotection. Sci. Adv. 8, eabo1244, Epub 2022/10/22. PubMed Central PMCID: PMCPMC9586484 10.1126/sciadv.abo1244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ponnalagu D. and Singh H. (2017) Anion channels of mitochondria. Handb. Exp. Pharmacol. 240, 71–101, Epub 2016/10/27. PubMed Central PMCID: PMCPMC5855116 10.1007/164_2016_39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Ponnalagu D., Hussain A.T., Thanawala R., Meka J., Bednarczyk P., Feng Y.et al. (2019) Chloride channel blocker IAA-94 increases myocardial infarction by reducing calcium retention capacity of the cardiac mitochondria. Life Sci. 235, 116841, Epub 2019/09/09. PubMed Central PMCID: PMCPMC7664129 10.1016/j.lfs.2019.116841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Ponnalagu D., Gururaja Rao S., Farber J., Xin W., Hussain A.T., Shah K.et al. (2016) Molecular identity of cardiac mitochondrial chloride intracellular channel proteins. Mitochondrion 27, 6–14, Epub 2016/01/19 10.1016/j.mito.2016.01.001 [DOI] [PubMed] [Google Scholar]
- 123.Ponnalagu D., Rao S.G., Farber J., Xin W., Hussain A.T., Shah K.et al. (2016) Data supporting characterization of CLIC1, CLIC4, CLIC5 and DmCLIC antibodies and localization of CLICs in endoplasmic reticulum of cardiomyocytes. Data Brief 7, 1038–1044, Epub 2016/04/23. PubMed Central PMCID: PMCPMC4826591 10.1016/j.dib.2016.03.061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Diaz R.J., Losito V.A., Mao G.D., Ford M.K., Backx P.H. and Wilson G.J. (1999) Chloride channel inhibition blocks the protection of ischemic preconditioning and hypo-osmotic stress in rabbit ventricular myocardium. Circ. Res. 84, 763–775, PubMed PMID: 10205144 10.1161/01.RES.84.7.763 [DOI] [PubMed] [Google Scholar]
- 125.Batthish M., Diaz R.J., Zeng H.P., Backx P.H. and Wilson G.J. (2002) Pharmacological preconditioning in rabbit myocardium is blocked by chloride channel inhibition. Cardiovasc. Res. 55, 660–671, PubMed PMID: 12160963 10.1016/S0008-6363(02)00454-6 [DOI] [PubMed] [Google Scholar]
- 126.Li J., Wu X. and Cui T. (2008) Functional characteristics and molecular identification of swelling-activated chloride conductance in adult rabbit heart ventricles. J. Huazhong Univ. Sci. Technol. Med. Sci. 28, 37–41 10.1007/s11596-008-0109-6 [DOI] [PubMed] [Google Scholar]
- 127.Diaz R.J., Fernandes K., Lytvyn Y., Hawrylyshyn K., Harvey K., Hossain T.et al. (2013) Enhanced cell-volume regulation in cyclosporin A cardioprotection. Cardiovasc. Res. 98, 411–419 10.1093/cvr/cvt056 [DOI] [PubMed] [Google Scholar]
- 128.Baines C.P., Kaiser R.A., Sheiko T., Craigen W.J. and Molkentin J.D. (2007) Voltage-dependent anion channels are dispensable for mitochondrial-dependent cell death. Nat. Cell Biol. 9, 550–555, PubMed Central PMCID: PMC2680246 10.1038/ncb1575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Anflous-Pharayra K., Lee N., Armstrong D.L. and Craigen W.J. (2011) VDAC3 has differing mitochondrial functions in two types of striated muscles. Biochim. Biophys. Acta 1807, 150–156, Epub 2010/09/30. PubMed Central PMCID: PMCPMC2998388 10.1016/j.bbabio.2010.09.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Shankar T.S., Ramadurai D.K.A., Steinhorst K., Sommakia S., Badolia R., Thodou Krokidi A.et al. (2021) Cardiac-specific deletion of voltage dependent anion channel 2 leads to dilated cardiomyopathy by altering calcium homeostasis. Nature Communications 12, 4583, Epub 2021/07/30. PubMed Central PMCID: PMCPMC8319341 10.1038/s41467-021-24869-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Coonan J.R. and Lamb G.D. (1998) Effect of chloride on Ca2+ release from the sarcoplasmic reticulum of mechanically skinned skeletal muscle fibres. Pflugers Arch. 435, 720–730, Epub 1998/04/16 10.1007/s004240050574 [DOI] [PubMed] [Google Scholar]
- 132.Pollock N.S., Kargacin M.E. and Kargacin G.J. (1998) Chloride channel blockers inhibit Ca2+ uptake by the smooth muscle sarcoplasmic reticulum. Biophys. J. 75, 1759–1766, Epub 1998/09/24. PubMed Central PMCID: PMCPMC1299847 10.1016/S0006-3495(98)77617-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Chakraborty K., Leung K. and Krishnan Y. (2017) High lumenal chloride in the lysosome is critical for lysosome function. Elife 6, e28862, Epub 2017/07/26. PubMed Central PMCID: PMCPMC5526669 10.7554/eLife.28862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zajac M., Chakraborty K., Saha S., Mahadevan V., Infield D.T., Accardi A.et al. (2020) What biologists want from their chloride reporters - a conversation between chemists and biologists. J. Cell Sci. 133, jcs240390, Epub 2020/01/25 10.1242/jcs.240390 [DOI] [PubMed] [Google Scholar]
- 135.Poroca D.R., Pelis R.M. and Chappe V.M. (2017) ClC channels and transporters: structure, physiological functions, and implications in human chloride channelopathies. Front Pharmacol. 8, 151, Epub 2017/04/08. PubMed Central PMCID: PMCPMC5362633 10.3389/fphar.2017.00151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Schein S.J., Colombini M. and Finkelstein A. (1976) Reconstitution in planar lipid bilayers of a voltage-dependent anion-selective channel obtained from paramecium mitochondria 1. J. Membr. Biol. 30, 99–120 10.1007/BF01869662 [DOI] [PubMed] [Google Scholar]
- 137.Colombini M. (1979) A candidate for the permeability pathway of the outer mitochondrial membrane. Nature 279, 643–645, PubMed PMID: 450112 10.1038/279643a0 [DOI] [PubMed] [Google Scholar]
- 138.Colombini M. and Mannella C.A. (2012) VDAC, the early days. Biochim. Biophys. Acta 1818, 1438–1443, PubMed Central PMCID: PMC3296906 10.1016/j.bbamem.2011.11.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Jentsch T.J. (2015) Discovery of CLC transport proteins: cloning, structure, function and pathophysiology. J. Physiol. 593, 4091–4109, PubMed Central PMCID: PMCPMC4594286 10.1113/JP270043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Miller C. (2015) In the beginning: a personal reminiscence on the origin and legacy of ClC-0, the ‘Torpedo Cl(-) channel’. J. Physiol. 593, 4085–4090, PubMed Central PMCID: PMCPMC4594285 10.1113/jphysiol.2014.286260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Gassmann M. and Bettler B. (2012) Regulation of neuronal GABA(B) receptor functions by subunit composition. Nat. Rev. Neurosci. 13, 380–394, Epub 2012/05/19 10.1038/nrn3249 [DOI] [PubMed] [Google Scholar]
- 142.Motiwala Z., Aduri N.G., Shaye H., Han G.W., Lam J.H., Katritch V.et al. (2022) Structural basis of GABA reuptake inhibition. Nature 606, 820–826, Epub 2022/06/09. PubMed Central PMCID: PMCPMC9394549 10.1038/s41586-022-04814-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Farrant M. and Nusser Z. (2005) Variations on an inhibitory theme: phasic and tonic activation of GABA(A) receptors. Nat. Rev. Neurosci. 6, 215–229, Epub 2005/03/02 10.1038/nrn1625 [DOI] [PubMed] [Google Scholar]
- 144.Singh H. (2010) Two decades with dimorphic Chloride Intracellular Channels (CLICs). FEBS Lett. 584, 2112–2121 10.1016/j.febslet.2010.03.013 [DOI] [PubMed] [Google Scholar]
- 145.Gururaja Rao S., Ponnalagu D., Patel N.J. and Singh H. (2018) Three decades of chloride intracellular channel proteins: from organelle to organ physiology. Curr Protoc Pharmacol. 80, 11 21 1–11 21 17, Epub 2018/07/25. PubMed Central PMCID: PMCPMC6060641 10.1002/cpph.36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Redhead C.R., Edelman A.E., Brown D., Landry D.W. and al-Awqati Q. (1992) A ubiquitous 64-kDa protein is a component of a chloride channel of plasma and intracellular membranes. Proc. Natl. Acad. Sci. USA 89, 3716–3720, PubMed PMID: 1315034; PubMed Central PMCID: PMC525561 10.1073/pnas.89.9.3716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Dawson D.C., Smith S.S. and Mansoura M.K. (1999) CFTR: mechanism of anion conduction. Physiol. Rev. 79, S47–S75, PubMed PMID: 9922376 10.1152/physrev.1999.79.1.S47 [DOI] [PubMed] [Google Scholar]
- 148.Dworakowska B. and Dolowy K. (2000) Ion channels-related diseases. Acta Biochim. Pol. 47, 685–703, Epub 2001/04/20. PubMed PMID: 11310970 10.18388/abp.2000_3989 [DOI] [PubMed] [Google Scholar]
- 149.Sharma K.R., Colvis C.M., Rodgers G.P. and Sheeley D.M. (2023) Illuminating the druggable genome: Pathways to progress. Drug Discov. Today 29, 103805, Epub 2023/10/28 10.1016/j.drudis.2023.103805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Zhang S., Meor Azlan N.F., Josiah S.S., Zhou J., Zhou X., Jie L.et al. (2023) The role of SLC12A family of cation-chloride cotransporters and drug discovery methodologies. J. Pharm. Anal. 13, 1471–1495, Epub 2024/01/15. PubMed Central PMCID: PMCPMC10785268 10.1016/j.jpha.2023.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Delaney A.J. and Sah P. (1999) GABA receptors inhibited by benzodiazepines mediate fast inhibitory transmission in the central amygdala. J. Neurosci. 19, 9698–9704, Epub 1999/11/13. PubMed Central PMCID: PMCPMC6782952 10.1523/JNEUROSCI.19-22-09698.1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Burgener E.B. and Moss R.B. (2018) Cystic fibrosis transmembrane conductance regulator modulators: precision medicine in cystic fibrosis. Curr. Opin. Pediatr. 30, 372–377, Epub 2018/03/15. PubMed Central PMCID: PMCPMC6398332 10.1097/MOP.0000000000000627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Santos R., Ursu O., Gaulton A., Bento A.P., Donadi R.S., Bologa C.G.et al. (2017) A comprehensive map of molecular drug targets. Nat. Rev. Drug Discovery 16, 19–34, Epub 2016/12/03. PubMed Central PMCID: PMCPMC6314433 10.1038/nrd.2016.230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Jiang Y.Y., Hou H.T., Yang Q., Liu X.C. and He G.W. (2017) Chloride channels are involved in the development of atrial fibrillation - a transcriptomic and proteomic study. Sci. Rep. 7, 10215, Epub 2017/09/02. PubMed Central PMCID: PMCPMC5579191 10.1038/s41598-017-10590-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Szabo I. and Szewczyk A. (2023) Mitochondrial Ion Channels. Annu. Rev. Biophys. 52, 229–254, Epub 2023/05/09 10.1146/annurev-biophys-092622-094853 [DOI] [PubMed] [Google Scholar]
- 156.Salomonsson M., Gonzalez E., Westerlund P. and Persson A.E. (1991) Chloride concentration in macula densa and cortical thick ascending limb cells. Kidney Int. Suppl. 32, S51–S54, Epub 1991/06/01. PubMed PMID: 1881050 [PubMed] [Google Scholar]
- 157.Grodin J.L., Testani J.M., Pandey A., Sambandam K., Drazner M.H., Fang J.C.et al. (2018) Perturbations in serum chloride homeostasis in heart failure with preserved ejection fraction: insights from TOPCAT. Eur. J. Heart Fail. 20, 1436–1443, Epub 2018/06/13 10.1002/ejhf.1229 [DOI] [PubMed] [Google Scholar]
- 158.Gronich N., Kumar A., Zhang Y., Efimov I.R. and Soldatov N.M. (2010) Molecular remodeling of ion channels, exchangers and pumps in atrial and ventricular myocytes in ischemic cardiomyopathy. Channels (Austin) 4, 101–107, PubMed PMID: 20090424; PubMed Central PMCID: PMCPMC2891309 10.4161/chan.4.2.10975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Gururaja Rao S., Bednarczyk P., Towheed A., Shah K., Karekar P., Ponnalagu D.et al. (2019) BKCa (Slo) channel regulates mitochondrial function and lifespan in Drosophila melanogaster. Cells 8, 945, Epub 2019/08/24. PubMed Central PMCID: PMCPMC6770356 10.3390/cells8090945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Paul S.M. and Beitel G.J. (2003) Developmental biology. Tubulogenesis CLICs into place. Science 302, 2077–2078 10.1126/science.1093203 [DOI] [PubMed] [Google Scholar]
- 161.Padmakumar V., Masiuk K.E., Luger D., Lee C., Coppola V., Tessarollo L.et al. (2014) Detection of differential fetal and adult expression of chloride intracellular channel 4 (CLIC4) protein by analysis of a green fluorescent protein knock-in mouse line. BMC Dev. Biol. 14, 24, PubMed Central PMCID: PMC4073518 10.1186/1471-213X-14-24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Sorgato M.C., Keller B.U. and Stuhmer W. (1987) Patch-clamping of the inner mitochondrial membrane reveals a voltage-dependent ion channel. Nature 330, 498–500 10.1038/330498a0 [DOI] [PubMed] [Google Scholar]
- 163.Valenzuela S.M., Martin D.K., Por S.B., Robbins J.M., Warton K., Bootcov M.R.et al. (1997) Molecular cloning and expression of a chloride ion channel of cell nuclei. J. Biol. Chem. 272, 12575–12582, PubMed PMID: 9139710 10.1074/jbc.272.19.12575 [DOI] [PubMed] [Google Scholar]
- 164.Suh K.S., Mutoh M., Nagashima K., Fernandez-Salas E., Edwards L.E., Hayes D.D.et al. (2004) The organellular chloride channel protein CLIC4/mtCLIC translocates to the nucleus in response to cellular stress and accelerates apoptosis. J. Biol. Chem. 279, 4632–4641 10.1074/jbc.M311632200 [DOI] [PubMed] [Google Scholar]
- 165.Singh H., Cousin M.A. and Ashley R.H. (2007) Functional reconstitution of mammalian ‘chloride intracellular channels’ CLIC1, CLIC4 and CLIC5 reveals differential regulation by cytoskeletal actin. FEBS J. 274, 6306–6316 10.1111/j.1742-4658.2007.06145.x [DOI] [PubMed] [Google Scholar]
- 166.Suh K.S., Malik M., Shukla A. and Yuspa S.H. (2007) CLIC4, skin homeostasis and cutaneous cancer: surprising connections. Mol. Carcinog. 46, 599–604 10.1002/mc.20324 [DOI] [PubMed] [Google Scholar]
- 167.Leanza L., Biasutto L., Manago A., Gulbins E., Zoratti M. and Szabo I. (2013) Intracellular ion channels and cancer. Front. Physiol. 4, 227, PubMed Central PMCID: PMC3759743 10.3389/fphys.2013.00227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Stauber T. and Jentsch T.J. (2013) Chloride in vesicular trafficking and function. Annu. Rev. Physiol. 75, 453–477 10.1146/annurev-physiol-030212-183702 [DOI] [PubMed] [Google Scholar]
- 169.Saha S., Prakash V., Halder S., Chakraborty K. and Krishnan Y. (2015) A pH-independent DNA nanodevice for quantifying chloride transport in organelles of living cells. Nat. Nanotechnol. 10, 645–651 10.1038/nnano.2015.130 [DOI] [PubMed] [Google Scholar]
- 170.Zhu G., Zhang Y., Xu H. and Jiang C. (1998) Identification of endogenous outward currents in the human embryonic kidney (HEK 293) cell line. J. Neurosci. Methods 81, 73–83, Epub 1998/08/08 10.1016/S0165-0270(98)00019-3 [DOI] [PubMed] [Google Scholar]
- 171.Dulhunty A.F., Pouliquin P., Coggan M., Gage P.W. and Board P.G. (2005) A recently identified member of the glutathione transferase structural family modifies cardiac RyR2 substate activity, coupled gating and activation by Ca2+ and ATP. Biochem. J. 390, 333–343, Epub 2005/05/27. PubMed Central PMCID: PMCPMC1184587 10.1042/BJ20042113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Achilonu I., Fanucchi S., Cross M., Fernandes M. and Dirr H.W. (2012) Role of individual histidines in the pH-dependent global stability of human chloride intracellular channel 1. Biochemistry 51, 995–1004, Epub 2012/01/17 10.1021/bi201541w [DOI] [PubMed] [Google Scholar]
- 173.Seidlmayer L.K., Juettner V.V., Kettlewell S., Pavlov E.V., Blatter L.A. and Dedkova E.N. (2015) Distinct mPTP activation mechanisms in ischaemia-reperfusion: contributions of Ca2+, ROS, pH, and inorganic polyphosphate. Cardiovasc. Res. 106, 237–248, Epub 2015/03/07. PubMed Central PMCID: PMCPMC4415062 10.1093/cvr/cvv097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Wang G. (2016) Chloride flux in phagocytes. Immunol. Rev. 273, 219–231, Epub 2016/08/26 10.1111/imr.12438 [DOI] [PubMed] [Google Scholar]
- 175.Shariati K., Zhang Y., Giubbolini S., Parra R., Liang S., Edwards A.et al. (2022) A superfolder green fluorescent protein-based biosensor allows monitoring of chloride in the endoplasmic reticulum. ACS Sens 7, 2218–2224, Epub 2022/08/12. PubMed Central PMCID: PMCPMC9425558 10.1021/acssensors.2c00626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Decher N., Lang H.J., Nilius B., Bruggemann A., Busch A.E. and Steinmeyer K. (2001) DCPIB is a novel selective blocker of I(Cl,swell) and prevents swelling-induced shortening of guinea-pig atrial action potential duration. Br. J. Pharmacol. 134, 1467–1479, PubMed Central PMCID: PMC1573095 10.1038/sj.bjp.0704413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Lin C.W. and Chen T.Y. (2003) Probing the pore of ClC-0 by substituted cysteine accessibility method using methane thiosulfonate reagents. J. Gen. Physiol. 122, 147–159, Epub 2003/07/30. PubMed Central PMCID: PMCPMC2229544 10.1085/jgp.200308845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Singh H. and Ashley R.H. (2007) CLIC4 (p64H1) and its putative transmembrane domain form poorly selective, redox-regulated ion channels. Mol. Membr. Biol. 24, 41–52 10.1080/09687860600927907 [DOI] [PubMed] [Google Scholar]
- 179.Gururaja Rao S., Ponnalagu D., Sukur S., Singh H., Sanghvi S., Mei Y.et al. (2017) Identification and characterization of a bacterial homolog of chloride intracellular channel (CLIC) protein. Sci. Rep. 7, 8500 10.1038/s41598-017-08742-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Voss F.K., Ullrich F., Munch J., Lazarow K., Lutter D., Mah N.et al. (2014) Identification of LRRC8 heteromers as an essential component of the volume-regulated anion channel VRAC. Science 344, 634–638, Epub 2014/05/03 10.1126/science.1252826 [DOI] [PubMed] [Google Scholar]
- 181.Jentsch T.J. (2016) VRACs and other ion channels and transporters in the regulation of cell volume and beyond. Nat. Rev. Mol. Cell Biol. 17, 293–307 10.1038/nrm.2016.29 [DOI] [PubMed] [Google Scholar]
- 182.Pasantes-Morales H. (2016) Channels and volume changes in the life and death of the cell. Mol. Pharmacol. 90, 358–370, Epub 2016/07/01 10.1124/mol.116.104158 [DOI] [PubMed] [Google Scholar]
- 183.Qiu Z., Dubin A.E., Mathur J., Tu B., Reddy K., Miraglia L.J.et al. (2014) SWELL1, a plasma membrane protein, is an essential component of volume-regulated anion channel. Cell 157, 447–458, Epub 2014/04/15. PubMed Central PMCID: PMCPMC4023864 10.1016/j.cell.2014.03.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Ujwal R., Cascio D., Colletier J.P., Faham S., Zhang J., Toro L.et al. (2008) The crystal structure of mouse VDAC1 at 2.3 A resolution reveals mechanistic insights into metabolite gating. Proc. Natl. Acad. Sci. U.S.A. 105, 17742–17747 10.1073/pnas.0809634105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Hiller S., Garces R.G., Malia T.J., Orekhov V.Y., Colombini M. and Wagner G. (2008) Solution structure of the integral human membrane protein VDAC-1 in detergent micelles. Science 321, 1206–1210, PubMed Central PMCID: PMC2579273 10.1126/science.1161302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Ben-Hail D. and Shoshan-Barmatz V. (2014) Reconstitution of purified VDAC1 into a lipid bilayer and recording of channel conductance. Cold Spring Harbor Protocols 2014, 100–105 10.1101/pdb.prot073148 [DOI] [PubMed] [Google Scholar]
- 187.Banerjee J. and Ghosh S. (2004) Bax increases the pore size of rat brain mitochondrial voltage-dependent anion channel in the presence of tBid. Biochem. Biophys. Res. Commun. 323, 310–314 10.1016/j.bbrc.2004.08.094 [DOI] [PubMed] [Google Scholar]
- 188.Shimizu S., Ide T., Yanagida T. and Tsujimoto Y. (2000) Electrophysiological study of a novel large pore formed by Bax and the voltage-dependent anion channel that is permeable to cytochrome c. J. Biol. Chem. 275, 12321–12325, PubMed PMID: 10766872 10.1074/jbc.275.16.12321 [DOI] [PubMed] [Google Scholar]
- 189.Ullrich F., Blin S., Lazarow K., Daubitz T., von Kries J.P. and Jentsch T.J. (2019) Identification of TMEM206 proteins as pore of PAORAC/ASOR acid-sensitive chloride channels. Elife 8, e49187, Epub 2019/07/19. PubMed Central PMCID: PMCPMC6663466 10.7554/eLife.49187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Averaimo S., Abeti R., Savalli N., Brown L.J., Curmi P.M., Breit S.N.et al. (2013) Point mutations in the transmembrane region of the clic1 ion channel selectively modify its biophysical properties. PLoS ONE 8, e74523, PubMed Central PMCID: PMC3776819 10.1371/journal.pone.0074523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Novarino G., Fabrizi C., Tonini R., Denti M.A., Malchiodi-Albedi F., Lauro G.M.et al. (2004) Involvement of the intracellular ion channel CLIC1 in microglia-mediated beta-amyloid-induced neurotoxicity. J. Neurosci. 24, 5322–5330 10.1523/JNEUROSCI.1170-04.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Kuner T. and Augustine G.J. (2000) A genetically encoded ratiometric indicator for chloride: capturing chloride transients in cultured hippocampal neurons. Neuron 27, 447–459, Epub 2000/10/31 10.1016/S0896-6273(00)00056-8 [DOI] [PubMed] [Google Scholar]
- 193.Whitlock J.M. and Hartzell H.C. (2017) Anoctamins/TMEM16 proteins: chloride channels flirting with lipids and extracellular vesicles. Annu. Rev. Physiol. 79, 119–143, Epub 2016/11/20. PubMed Central PMCID: PMCPMC5556385 10.1146/annurev-physiol-022516-034031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Nguyen D.M., Kwon H.C. and Chen T.Y. (2021) Divalent cation modulation of ion permeation in TMEM16 proteins. Int. J. Mol. Sci. 22, 2209, Epub 2021/03/07. PubMed Central PMCID: PMCPMC7926781 10.3390/ijms22042209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Lipicky R.J. and Bryant S.H. (1966) Sodium, potassium, and chloride fluxes in intercostal muscle from normal goats and goats with hereditary myotonia. J. Gen. Physiol. 50, 89–111, Epub 1966/09/01. PubMed Central PMCID: PMCPMC2225635 10.1085/jgp.50.1.89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Gunn R.B., Wieth J.O. and Tosteson D.C. (1975) Some effects of low pH on chloride exchange in human red blood cells. J. Gen. Physiol. 65, 731–749, Epub 1975/06/01. PubMed Central PMCID: PMCPMC2214889 10.1085/jgp.65.6.731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Sulis Sato S., Artoni P., Landi S., Cozzolino O., Parra R., Pracucci E.et al. (2017) Simultaneous two-photon imaging of intracellular chloride concentration and pH in mouse pyramidal neurons in vivo. PNAS 114, E8770–E8779, Epub 2017/10/05. PubMed Central PMCID: PMCPMC5642681 10.1073/pnas.1702861114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Shaw A.D., Raghunathan K., Peyerl F.W., Munson S.H., Paluszkiewicz S.M. and Schermer C.R. (2014) Association between intravenous chloride load during resuscitation and in-hospital mortality among patients with SIRS. Intensive Care Med. 40, 1897–1905, Epub 2014/10/09. PubMed Central PMCID: PMCPMC4239799 10.1007/s00134-014-3505-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Arosio D., Ricci F., Marchetti L., Gualdani R., Albertazzi L. and Beltram F. (2010) Simultaneous intracellular chloride and pH measurements using a GFP-based sensor. Nat. Methods 7, 516–518, Epub 2010/06/29 10.1038/nmeth.1471 [DOI] [PubMed] [Google Scholar]
- 200.Miyazaki H., Shiozaki A., Niisato N., Ohsawa R., Itoi H., Ueda Y.et al. (2008) Chloride ions control the G1/S cell-cycle checkpoint by regulating the expression of p21 through a p53-independent pathway in human gastric cancer cells. Biochem. Biophys. Res. Commun. 366, 506–512, Epub 2007/12/11 10.1016/j.bbrc.2007.11.144 [DOI] [PubMed] [Google Scholar]
- 201.Shiozaki A., Otsuji E. and Marunaka Y. (2011) Intracellular chloride regulates the G(1)/S cell cycle progression in gastric cancer cells. World J. Gastrointest Oncol. 3, 119–122, Epub 2011/10/19. PubMed Central PMCID: PMCPMC3192220 10.4251/wjgo.v3.i8.119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Kenyon J.L. and Gibbons W.R. (1979) Influence of chloride, potassium, and tetraethylammonium on the early outward current of sheep cardiac Purkinje fibers. J. Gen. Physiol. 73, 117–138, Epub 1979/02/01. PubMed Central PMCID: PMCPMC2215241 10.1085/jgp.73.2.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Heimlich G. and Cidlowski J.A. (2006) Selective role of intracellular chloride in the regulation of the intrinsic but not extrinsic pathway of apoptosis in Jurkat T-cells. J. Biol. Chem. 281, 2232–2241, Epub 2005/11/22 10.1074/jbc.M507367200 [DOI] [PubMed] [Google Scholar]
- 204.Misak A., Grman M., Malekova L., Novotova M., Markova J., Krizanova O.et al. (2013) Mitochondrial chloride channels: electrophysiological characterization and pH induction of channel pore dilation. Eur. Biophys. J. 42, 709–720 10.1007/s00249-013-0920-2 [DOI] [PubMed] [Google Scholar]
- 205.Griffon N., Jeanneteau F., Prieur F., Diaz J. and Sokoloff P. (2003) CLIC6, a member of the intracellular chloride channel family, interacts with dopamine D(2)-like receptors. Brain Res. Mol. Brain Res. 117, 47–57, PubMed PMID: 14499480 10.1016/S0169-328X(03)00283-3 [DOI] [PubMed] [Google Scholar]
- 206.De Bacquer D., De Backer G., De Buyzere M. and Kornitzer M. (1998) Is low serum chloride level a risk factor for cardiovascular mortality? J. Cardiovasc. Risk 5, 177–184, PubMed PMID: 10201555 10.1097/00043798-199806000-00007 [DOI] [PubMed] [Google Scholar]
- 207.Biwersi J. and Verkman A.S. (1991) Cell-permeable fluorescent indicator for cytosolic chloride. Biochemistry 30, 7879–7883, Epub 1991/08/13 10.1021/bi00246a001 [DOI] [PubMed] [Google Scholar]
- 208.Sonawane N.D. and Verkman A.S. (2003) Determinants of [Cl-] in recycling and late endosomes and Golgi complex measured using fluorescent ligands. J. Cell Biol. 160, 1129–1138, Epub 2003/04/02. PubMed Central PMCID: PMCPMC2172765 10.1083/jcb.200211098 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable