Abstract
A single guanosine deletion/insertion (4G/5G) polymorphism in the promoter region of plasminogen activator inhibitor-1 (PAI-1) gene encoding PAI-1 protein has been investigated in deep vein thrombosis (DVT) patients. The association between PAI-1 4G/5G polymorphism and increased risk of DVT has been reported in some studies, while others have reported a lack of association. The present study aimed to investigate if the PAI-1 4G/5G polymorphism is associated with an increased risk of DVT in the Indian population and to assess its association with thrombophilic risk factors. Fifty-two adult patients with a history of chronic or recurrent DVT and 52 healthy adult controls were genotyped for PAI-1 4G/5G polymorphism. Plasma levels of PAI-1 and other thrombophilic risk factors were also measured. PAI-1 4G/5G polymorphism was not significantly associated with an increased risk of DVT. Protein C deficiency was significantly associated with the 4G/4G genotype. Patients with the 4G/4G genotype had significantly reduced PAI-1 levels as compared to the controls. PAI-1 4G/5G polymorphism did not significantly contribute to an increased risk of DVT in the Indian population. However, in the presence of thrombophilic risk factor abnormalities, the risk of DVT is increased in individuals with the 4G/4G genotype in the Indian cohort.
Supplementary Information
The online version contains supplementary material available at 10.1007/s12288-023-01660-3.
Keywords: Deep vein thrombosis, Fibrinolysis, Plasminogen activator inhibitor-1, PAI-1 4G/5G polymorphism, Thrombophilia
Introduction
Plasminogen activator inhibitor (PAI-1), a 52-kD serpin protein is the principal inhibitor of fibrinolysis. By inhibiting tissue plasminogen activator (tPA), it prevents the activation of plasminogen into plasmin, the chief mediator of fibrinolysis. PAI-1 is secreted into plasma by endothelial cells and α-granules of activated platelets. The PAI-1 gene is located on chromosome 7q22 comprising 9 exons and 8 introns.
A single guanosine deletion/insertion PAI-1 4G/5G promoter polymorphism, 675 bp upstream of the transcription start site has been investigated in deep vein thrombosis (DVT) [1–7]. 4G allele activates the PAI-1 gene transcription, leading to increased PAI-1 plasma levels and an increased predisposition to venous thrombosis. Few studies have found a lack of association [8–12]. Thus, the data regarding the association between PAI-1 4G/5G polymorphism and DVT is quite heterogeneous and limited. The present study was undertaken to assess the association between PAI-1 4G/5G polymorphism and the risk of DVT in the Indian population. Additionally, the effect of this polymorphism on plasma PAI-1 antigen levels and its association with thrombophilic risk factors were assessed.
Materials and Methods
Fifty-two adult patients (≥ 18 years of age) with a history of recurrent or chronic lower limb DVT diagnosed on Doppler ultrasound and/or magnetic resonance venography were enrolled in the study after written informed consent from July 2018–December 2020. Fifty-two healthy adult volunteers ≥ 18 years of age served as controls. Blood samples were collected from each case at ≥ 6 weeks of the onset of DVT, that is after the acute phase of thrombosis and ≥ 4 weeks of completion of anticoagulant therapy. The patients with acute thrombosis, on anticoagulation therapy, and lacking complete clinical details were excluded. The study was approved by the Institute Ethics Committee (INT/IEC/2018/002070).
PAI-1 4G/5G Polymorphism
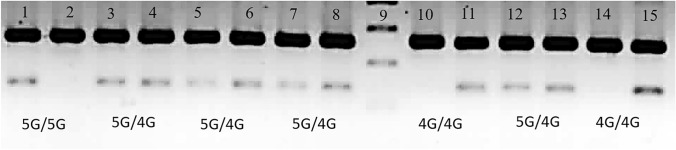
Amplification refractory mutation system-based polymerase chain reaction (ARMS-PCR) was performed on genomic DNA using primer sequences for PAI-1 4G/5G genotyping (Fig. 1).
Fig. 1.
Ethidium bromide-stained 3% agarose gel showing amplified products of PAI-1 4G/5G polymorphism by ARMS PCR. Lanes 1 and 2 display the products of amplification with a primer specific to the 5G allele (138 bp); Lanes 3 and 4, 5 and 6, 7 and 8, 12 and 13, represent 4G/5G genotype (138 bp each); and lanes 10 and 11, 14 and 15 represent 4G allele (138 bp). Lane 9 is the DNA ladder
PAI-1 and tPA Antigen
PAI-1 and tPA antigen assays were performed by kit-based enzyme-linked immunosorbent assay (ELISA) as per the manufacturer’s instructions (Cloud Clone-Corp; Texas, USA for PAI-1 and ASSERACHROM, Diagnostica Stago, France for tPA antigen).
Thrombophilia Work-Up
Protein C (PC), free protein S (PS), anti-thrombin-III (AT), lupus anticoagulant (LAC), anti-cardiolipin antibody (ACA), and anti-β2 glycoprotein 1 antibody (anti-β2GP1) were tested. Factor V Leiden (FVL) was tested by restriction fragment length polymorphism-based polymerase chain reaction (RFLP-PCR).
Results
Male to female ratio in the patient group was 1.08:1 and 2.05:1 in the control group. The median age of the patients was 35.5 years (24–66 years) and that of the controls was 30 years (24–45 years). Left lower limb DVT was present in 53.8% of patients and the right lower limb was affected in 38.5% of patients. Bilateral lower limb DVT was present in 7.7% of patients. DVT was provoked in 38.5% of patients and unprovoked in 61.5% of patients. Recurrent DVT was found in 3 patients. Laboratory-based thrombophilic risk factor abnormalities were found in 18 (34.6%) patients of which inherited risk factor abnormalities were detected in 16 (88.9%) patients, and APL was detected in 2 (11.1%) patients. Within the inherited risk factor abnormalities, FVL 1691G/A (rs6025) (11; 68.8%), PC deficiency (3;18.8%), and AT deficiency (2;12.4%) were detected. None of the patients had free PS deficiency. Of 11 cases, 10 (90%) patients were heterozygous, and 1 (10%) patient was homozygous for FVL 1691G/A (rs6025). One healthy control was heterozygous for FVL 1691G/A (rs 6025). Overall, of 52 cases, FVL 1691G/A (rs6025) was detected in 21.2% of cases, PC deficiency in 5.8%, and AT deficiency in 3.8% of cases.
PAI-1 4G/5G Polymorphism and risk of DVT
The most common genotype in cases and controls was 4G/5G heterozygous (42.3% and 46%, respectively). Risk assessment of PAI-1 4G/5G polymorphism for DVT was performed by calculating the odds ratio with a 95% confidence interval. The polymorphism was not found to be significantly associated with an enhanced risk of DVT in Indian patients (Table 1).
Table 1.
Risk assessment of PAI-1 4G/5G polymorphism for DVT
| Genotype | Cases | Controls | Odds ratio | 95% Confidence Interval | P value |
|---|---|---|---|---|---|
| 4G/5G | 22 | 24 | 0.702 | 0.267–1.845 | 0.471 |
| 4G/4G | 15 | 17 | 0.647 | 0.228–1.835 | 0.413 |
| 5G/5G | 15 | 11 | 1 (reference) | – | – |
Thrombophilic Risk Factors and PAI-1 4G/5G Polymorphism
Of all the thrombophilic risk factors, PC deficiency was significantly associated with the 4G/4G homozygous genotype in the Indian population. No significant association was found between other thrombophilic risk factors and the polymorphism (Table 2). The polymorphism was also not found to be significantly associated with unprovoked and/or idiopathic DVT cases.
Table 2.
Thrombophilic risk factors and PAI-1 4G/5G polymorphism in cases
| Risk factor | 4G/5G | 4G/4G | 5G/5G | P value |
|---|---|---|---|---|
| PC deficiency | ||||
| P | 0 | 3 | 0 | 0.020* |
| A | 22 | 12 | 15 | |
| AT deficiency | ||||
| P | 0 | 2 | 0 | 0.077 |
| A | 22 | 13 | 15 | |
| Factor V Leiden (FVL) 1691G/A (rs6025) | ||||
| P | 6 | 3 | 2 | 0.590 |
| A | 16 | 12 | 13 | |
| aPL¥ | ||||
| P | 2 | 0 | 0 | 0.371 |
| A | 20 | 15 | 13 | |
P: Present; A: Absent; *: significant P value (Chi-square test); ¥: two lupus anticoagulant positive cases were excluded from the analysis due to single-time testing performed (loss of follow-up). Protein S deficiency was not found in any case, hence not shown in the table
PAI-1 and tPA Plasma Antigen Levels and Association with PAI-1 4G/5G Polymorphism
PAI-1 antigen levels were significantly lower in cases than in controls (median of 0.85 ng/ml in cases versus 1.16 ng/ml in controls, p-0.032). Median tPA antigen levels were significantly higher in cases (n = 50, 11.21 ng/ml) than in the controls (n = 30, 8.62 ng/ml, p-0.003). PAI-1 antigen levels were significantly lower in patients with the 4G/4G genotype than in controls. The difference in PAI-1 antigen levels was not statistically significant between cases and controls of 5G/5G and 4G/5G genotype groups each (Supplementary Table 1).
Discussion
A significantly increased risk of DVT in patients with the 4G/4G genotype has been reported [1–5]. Akhter et al. found a positive association between the 4G allele and DVT in an Asian Indian population (n = 110) [6]. Prabhudesai et al. reported that the 4G/4G genotype is associated with an elevated risk of DVT (n = 163, odds ratio = 1.75, 95% CI 0.98–3.02, P = 0.0225), although it was more strongly associated with splanchnic vein thrombosis (SVT) [7].
Few studies have reported no association between this polymorphism and the risk of DVT (Supplementary Table 2) [8–12]. An African-American study comprising 91 cases and 185 controls reported the PAI-1 4G/5G polymorphism does not increase the risk of DVT [8]. In an Italian study by Sciacca et al. including 250 cases of high-grade glioma, 270 controls, and 90 non-neoplastic cases, the PAI-1 and tPA gene polymorphisms did not significantly increase the risk of DVT or pulmonary embolism (PE) [9]. Mansilha et al. reported that PAI-1 4G/5G polymorphism was not associated with recurrent DVT in 87 Portugal patients below 40 years of age [10]. Giurgea et al. concluded that the polymorphism was not associated with the persistence of residual occlusion after acute idiopathic DVT in 43 patients of Austrian origin [11]. Vuckovic et al. found that PAI-1 4G/5G polymorphism does not increase the risk of VT in Serbian Caucasians [12]. Similarly, the present study also reports that PAI-1 4G/5G polymorphism is not significantly associated with an increased risk of DVT found in the Indian population.
Segui et al. reported significantly increased PAI-1 antigen and PAI-1 activity levels in DVT patients of Spanish origin than controls and in patients with 4G/4G genotypes than 4G/5G and 5G/5G genotypes [1]. Sartori et al. reported significantly higher levels of PAI-1 antigen in Italian patients with 4G/4G genotype and symptomatic thrombophilia than controls [2]. In an Italian study by Incalcaterra et al. PAI-1 activity was elevated in the 4G allele carriers [4]. In the present study, we report that PAI-1 antigen levels were significantly reduced in DVT cases than in controls in the Indian population. This could be explained based on significantly higher tPA levels in DVT cases that were tested ≥ 6 weeks of acute onset. PAI-1 inhibits fibrinolysis by binding with tPA forming the PAI-1- tPA complex. Thus, the significant increase in tPA level might be responsible for the increased binding of PAI-1 with tPA and resulting in lower PAI-1 antigen levels.
Segui et al. reported an Odds Ratio of 8.14 (2.81–28.71, 95% CI) for venous thrombosis in Spanish patients carrying the 4G allele combined with genetic thrombophilic defects (13%) as compared to controls (2%) [1]. A Turkish study by Akar et al. investigated 136 DVT patients and 113 controls and concluded that the combined presence of either PAI-1 4G/4G or 4G/5G genotypes and FVL 1691G/A (rs6025) increases the risk of thrombosis (Odds Ratio = 9.8 and 6.9, 95% CI 2.9–32.7 and 1.3–35.8, respectively) [13]. In a Swedish study by Sundquist et al. 177 patients were investigated for PAI-1 4G/5G polymorphism where-in the patients carrying the 4G allele with concomitant FVL 1691G/A (rs6025) were at an increased risk of VTE recurrence than patients without FVL 1691G/A (rs6025). The patients carrying the 5G allele irrespective of FVL 1691G/A (rs6025) had a lower risk of VTE recurrence than patients with the 4G allele without FVL 1691G/A (rs6025) [14]. In our study, the 4G/4G genotype was significantly associated with inherited PC deficiency (p-value: 0.020), implying that the 4G/4G genotype with concomitant thrombophilic risk factor abnormality may accentuate the risk of DVT in the Indian population.
In conclusion, the risk of DVT may be accelerated in patients with the 4G/4G genotype in the Indian population. However, a larger sample size is required to validate the findings.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Author Contributions
SS and NK designed the study, retrieved the data, and drafted the manuscript. SS, MJ, VU, HKS, and MJi performed the laboratory assays. JA, RD, NV performed laboratory data analysis and PM performed clinical data analysis. All authors reviewed, and approved the final manuscript.
Funding
NK received financial assistance for this research from the Postgraduate Institute of Medical Education & Research, Chandigarh (reference no,71/2-Edu-16/80 dated 06/01/18). SS received the Indian Council of Medical Research (ICMR), New Delhi research Grant award, for this study [reference no. 3/2/Jan.2018/PG-Thesis-HRD (41) dated 07/09/2018].
Declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Seguí R, Estellés A, Mira Y, et al. PAI-1 promoter 4G/5G genotype as an additional risk factor for venous thrombosis in subjects with genetic thrombophilic defects. Br J Haematol. 2000;111:122–128. doi: 10.1046/j.1365-2141.2000.02321.x. [DOI] [PubMed] [Google Scholar]
- 2.Sartori MT, Danesin C, Saggiorato The PAI-I gene 4G/5G polymorphism and deep vein thrombosis in patients with inherited Thrombophilia. Clin Appl Thromb Hemost. 2003;9:299–307. doi: 10.1177/107602960300900405. [DOI] [PubMed] [Google Scholar]
- 3.Farajzadeh M, Bargahi N, Zonouzi AP, et al. Polymorphisms in thrombophilic genes are associated with deep venous thromboembolism in an iranian population. Meta Gene. 2014;2:505–513. doi: 10.1016/j.mgene.2014.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Incalcaterra E, Meli F, Muratori I, et al. Residual vein thrombosis and onset of post-thrombotic syndrome: influence of the 4G/5G polymorphism of plasminogen activator inhibitor 1 gene. Thromb Res. 2014;133:371–374. doi: 10.1016/j.thromres.2013.12.032. [DOI] [PubMed] [Google Scholar]
- 5.Hosseini S, Kalantar E, Hosseini MS, et al. Genetic risk factors in patients with deep venous thrombosis, a retrospective case-control study on iranian population. Thromb J. 2015;13:35. doi: 10.1186/s12959-015-0064-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Akhter MS, Biswas A, Ranjan R, et al. Plasminogen activator Inhibitor-1 (PAI-1) gene 4G/5G promoter polymorphism is seen in higher frequency in the indian patients with deep vein thrombosis. Clin Appl Thromb/Hemost. 2010;16:184–188. doi: 10.1177/1076029609333673. [DOI] [PubMed] [Google Scholar]
- 7.Prabhudesai A, Shetty S, Ghosh K, et al. Investigation of plasminogen activator Inhibitor-1 (PAI-1) 4G/5G promoter polymorphism in indian venous thrombosis patients: a case-control study. Eur J Haematol. 2017;99:249–254. doi: 10.1111/ejh.12912. [DOI] [PubMed] [Google Scholar]
- 8.Hooper WC, Lally C, Austin H, et al. The role of the t-PA I/D and PAI-1 4G/5G polymorphisms in african-american adults with a diagnosis of myocardial infarction or venous thromboembolism. Thromb Res. 2000;99:223–230. doi: 10.1016/S0049-3848(00)00236-X. [DOI] [PubMed] [Google Scholar]
- 9.Sciacca FL, Ciusani E, Silvani A, et al. Genetic and plasma markers of venous thromboembolism in patients with high-grade glioma. Clin Cancer Res. 2004;10:1312–1317. doi: 10.1158/1078-0432.CCR-03-0198. [DOI] [PubMed] [Google Scholar]
- 10.Mansilha A, Arau´jo F, Severo M, et al. Genetic polymorphisms and risk of recurrent deep venous thrombosis in Young People: prospective cohort study. Eur J Vasc Endovasc Surg. 2005;30:545–549. doi: 10.1016/j.ejvs.2005.05.038. [DOI] [PubMed] [Google Scholar]
- 11.Giurgea GA, Brunner-Ziegler S, Jilma B, et al. Plasminogen activator inhibitor-1 4G/5G genotype and residual venous occlusion following acute unprovoked deep vein thrombosis of the lower limb: a prospective cohort study. Thromb Res. 2017;153:71–75. doi: 10.1016/j.thromres.2017.03.017. [DOI] [PubMed] [Google Scholar]
- 12.Vuckovic BA, Djeric MJ, Tomic BV, et al. Influence of decreased fibrinolytic activity and plasminogen activator inhibitor-1 4G/5G polymorphism on the risk of venous thrombosis. Blood Coagul Fibrinolysis. 2018;29:19–24. doi: 10.1097/MBC.0000000000000656. [DOI] [PubMed] [Google Scholar]
- 13.Akar N, Yilmaz E, Akar E, et al. Effect of plasminogen activator inhibitor-1 4G/5G polymorphism in turkish deep vein thrombotic patients with and without FV1691 G-A. Thromb Res. 2000;97:227–230. doi: 10.1016/S0049-3848(99)00164-4. [DOI] [PubMed] [Google Scholar]
- 14.Sundquist K, Wang X, Svensson PJ, et al. Plasminogen activator inhibitor-1 4G/5G polymorphism, factor V Leiden, prothrombin mutations and the risk of VTE recurrence. Thromb Haemost. 2015;114:1156–1164. doi: 10.1160/TH15-01-0031. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.