Abstract
Background
Managing systolic blood pressure (SBP), diastolic blood pressure (DBP), and heart rate (HR) is pivotal in acute aortic dissection (AAD) care. However, no prior studies have jointly analyzed the trajectories of these parameters. This research aimed to characterize their joint longitudinal trajectories and investigate the influence on AAD prognosis.
Methods
We included AAD patients from the Medical Information Mart for Intensive Care (MIMIC)-IV database. Using group-based multi-trajectory modeling (GBMTM), we identified combined trajectories of SBP, DBP, and HR within the initial 24 h of intensive care unit (ICU) admission. Cox proportional hazard regression, log-binomial regression, and logistic regression were employed to assess the association between trajectory groups and mortality outcomes.
Results
Data from 337 patients were analyzed. GBMTM identified five combined trajectory groups. Group 1 featured rapidly declining SBP and DBP with high pulse pressure and low HR; Group 2 showed high to moderate SBP with slight rebound and persistently low HR; Group 3 displayed persistently moderate BP and HR; Group 4 was characterized by moderate blood pressure with persistently high HR; and Group 5 had high to moderate SBP with slight rebound, high but gradually declining DBP, and slightly high HR. Group 3 demonstrated a lower risk of mortality, with an adjusted hazard ratio of 0.32 (95 % CI, 0.14–0.74), and the adjusted relative risks for in-hospital, 30-day, and 1-year mortalities were 0.37 (95 % CI, 0.15–0.87), 0.25 (95 % CI, 0.10–0.62), and 0.41 (95 % CI, 0.22–0.79), respectively. The time-independent C-index curve demonstrated that the multi-trajectory groups had higher C-index values than any univariate trajectory groups or admission values of SBP, DBP, and HR.
Conclusions
Utilization of GBMTM can yield data-driven insights to identify distinct subphenotypes in AAD patients. The combined trajectories of SBP, DBP, and HR within 24 h of ICU admission significantly influenced the mortality rate.
Keywords: Acute aortic dissection, Blood pressure, Heart rate, Group-based multi-trajectory modeling, Trajectory, ICU
1. Background
Acute aortic dissection (AAD) is a medical emergency caused by aortic intimal tear, with an in-hospital mortality rate of 13%–25 % [1,2]. Most deaths occur within 48 h of the onset of aortic dissection (AD) when there is no intervention [3]. Guidelines for the management of AAD emphasize the importance of blood pressure (BP) and heart rate (HR) control, and numerous studies have evaluated the impact of BP or HR, measured at a single time point, on patient prognosis [[4], [5], [6], [7]]. However, existing research has certain limitations, including a lack of high-quality evidence for optimal BP and HR control. This lack may conceal the effect of changing BP or HR patterns. Furthermore, previous studies have typically analyzed systolic blood pressure (SBP), diastolic blood pressure (DBP), and HR as separate indicators instead of considering them together as a comprehensive indicator. Given that BP and HR are both regulated by the autonomic nervous system and can be influenced by commonly used medical treatments like sedatives, analgesics, and antihypertensives, it is important to take into account their combined changes.
Group-based trajectory modeling (GBTM) is a descriptive data analysis technique based on finite mixture modelling [8]. It uses maximum likelihood estimation to identify groups of people who follow similar trajectories on a single indicator. Group-based multi-trajectory modeling (GBMTM) identifies potential populations of individuals that simultaneously follow similar trajectories across two or more distinct but related indicators [9,10]. These models are suitable for repeated follow-up measurements of the same individual at multiple time points and can better represent the dynamic trend of variables over time. Some studies have successfully applied this method to trajectories in SBP [11], intracranial pressure [12], body temperature [13], and other indicators in clinical patients. BP and HR are two closely related vital signs that are commonly monitored in critically ill patients. We propose that more useful information for intensive care unit (ICU) care strategies may be provided by multi-trajectory groups of these vital signs. In this study, we aimed to investigate the trajectories of AAD patients in the ICU, based on their SBP, DBP, and HR, to define groups and assess their associations with clinical outcomes.
2. Materials and methods
2.1. Data source and ethics approval
This study utilized data extracted from the Medical Information Mart for Intensive Care IV (MIMIC-IV, V2.0) [14], a comprehensive clinical database containing high-resolution information on patients admitted to the Beth Israel Deaconess Medical Center in Boston, MA, between 2008 and 2019. The Institutional Review Board at the medical center has reviewed the MIMIC database and granted a waiver of informed consent as well as approval for the data sharing initiative. To comply with relevant regulations, the first author (XS) obtained a Collaborative Institutional Training Initiative (CITI) license (certification No. 48810644) and permission to access the MIMIC-IV database.
2.2. Study subjects
International Classification of Diseases (ICD) codes, Ninth and Tenth Revision, were utilized to select patients. Specifically, AAD was defined as the occurrence of the disease with ICD codes I71.0 and 4410 in the first discharge diagnosis position, specifically for the first ICU admission during the first hospitalization in cases of patients with multiple ICU stays. Exclusion criteria were applied to patients younger than 18 years, pregnant patients, and those diagnosed with connective tissue disorders, Marfan syndrome, or traumatic aortic dissection. Additionally, we only included patients who had more than three BP and HR recordings.
2.3. Data collection
Structured query language (SQL) was used to extract the necessary data from the MIMIC-IV database for our analysis. The extracted data included demographic data (age, gender, ethnicity, height, weight), comorbidities (hypertension, diabetes, coronary diseases, chronic kidney disease (CKD), cancer), laboratory values, medication records (e.g., calcium channel blocker (CCB), angiotensin-converting enzyme inhibitor (ACEI), angiotensin receptor blocker (ARB), beta-blocker, analgesic), treatment (endovascular repair, surgical procedure), continuous renal replacement therapy (CRRT), mechanical ventilation, and disease severity scores, including the Sequential Organ Failure Assessment (SOFA), Acute Physiology Score III (APSIII), Oxford Acute Severity of Illness Score (OASIS), and Simplified Acute Physiology Score II (SAPS II). Furthermore, the dissection site was categorized according to ICD codes.
2.4. BP, HR, and mortality
Measurements of SBP, DBP, and HR were taken and recorded within the first 24 h following ICU admission. In instances where these variables were documented multiple times within a 1-h interval, we computed the average value of these records for further analysis in this study. The primary outcome of interest in this study was all-cause mortality. Hospital mortality was defined as death occurring during the hospitalization period. Furthermore, we ascertained 30-day mortality and 1-year mortality based on the duration between the date of ICU admission and the date of death.
2.5. Statistical analysis
In this study, GBMTM was used to analyze the multi-trajectories of SBP, DBP, and HR measures within 24 h of ICU admission, as well as univariate trajectory modeling for each variable separately by GBTM. To ensure sufficient sample sizes in each group, we created models with 2–5 groups and assigned group membership accordingly. We determined the optimal number of groups by considering several parameters, including lower absolute value Bayesian and Akaike information criteria (BIC and AIC), log-likelihood (LL), average posterior probability (AvePP) of groups greater than 0.7, odds of correct classification (OCC) greater than 5, and group size greater than 5 %. After identifying multi-trajectory groups, patient demographics and baseline clinical characteristics were summarized using means and standard deviations for continuous variables, and frequencies and percentages for categorical variables. We compared categorical variables using the chi-square test or Fisher exact test when appropriate. Analysis of variance (ANOVA) or the Kruskal-Wallis test was used to compare continuous variables among multiple groups.
In order to thoroughly examine the connection between trajectory groups and mortality, we employed various statistical models. Firstly, we utilized Cox proportional hazard models to calculate hazard ratios when the assumption of proportional hazards was met. Secondly, we used log-binomial regression or poisson regression models with a robust variance estimator if the binomial model failed to converge, to estimate relative risk (RR). Additionally, we validated our findings by employing a logistic regression model and conducted multiple imputations to address missing data for body mass index (BMI).
In the multivariate analysis, we employed three models to elucidate the influence of trajectory grouping on patient prognoses. In Model 1, we examined the groups without any covariates. In Model 2, we adjusted for age, gender, and race. Finally, in Model 3, we further adjusted for BMI, diabetes, hypertension, SOFA score, AD location, and management. The selection of adjusted covariates was based on their clinical significance, previous publication in the literature, and data available in the database.
By expanding on the criteria for covariate selection, we ensured the robustness of our models and minimized the risk of overfitting by approximating adherence to the principle of 10 events per variable (EPV) in our logistic and Cox regression analyses. Furthermore, guided by literature on the impact of blood pressure on the prognosis of AAD [15,16], we selected demographic factors (age, gender), physiological parameters (BMI), and clinical history (hypertension, diabetes) for inclusion. The AD location was utilized as a proxy for the Stanford classification, and treatment strategies (conservative treatment, thoracic endovascular aortic repair (TEVAR), surgical intervention, hybrid surgery) were incorporated due to their critical role in affecting patient mortality. The SOFA score was also included, informed by univariate analyses that demonstrated its significant prognostic value in reflecting the severity of a patient's condition upon admission. This score encompasses a comprehensive assessment of organ dysfunction, covering respiratory, cardiovascular, hepatic, neurological, renal, and coagulation functions. Finally, we evaluated collinearity among the covariates using tolerance and Variance Inflation Factor (VIF) tests. Significant collinearity was deemed present if the tolerance value fell below 0.1 or if the VIF exceeded 10, ensuring the integrity and reliability of our model adjustments. Given the potential influence of uniformity in the treatment approach for BP and HR on the study's results, variability in treatment approaches, including the use of analgesics and other medications, was accounted for as covariates in the sensitivity analysis.
The time-independent concordance index (C-index) was employed to evaluate the prognostic significance of multi-trajectory groups, univariable trajectory groups, and baseline clinical measurements—specifically, SBP, DBP, and HR—in predicting 1-year mortality outcomes following hospital admission. The analysis was performed using the TRAJ procedure in STATA version 17. Descriptive tables and statistical analyses were conducted in R (version 4.2.1) and SPSS software (version 27.0), and statistical significance was determined at a two-sided p-value of less than 0.05.
3. Results
The study initially identified 643 critically ill patients with AD from the MIMIC-IV database. After applying specific criteria, the study ultimately included 337 patients with AAD in the analysis (Fig. 1). Through GBMTM analyses, the study identified five distinct multi-trajectory groups (Fig. 2). As presented in Appendix Table S1, these groups demonstrated high AvePP (>0.97), the smallest BIC, AIC, and LL values, and a group size exceeding 5 % for all groups. Additionally, the OCC values exceeded 5.0 in all groups, indicating excellent grouping. The longitudinal trajectories within each group exhibited progressive differences. The five multi-trajectory groups were as follows:
Fig. 1.
Flowchart of the study
Abbreviations: AD: aortic dissection; ICU: intensive care unit; AAD: acute aortic dissection.
Fig. 2.
Trajectory groups of 24-h SBP/DBP/HR by group-based multi-trajectory modeling
Abbreviations: SBP: systolic blood pressure; DBP: diastolic blood pressure; HR: heart rate.Different colored lines represent distinct trajectory groups. The percentages in each graph denote the proportion of the total sample represented by each trajectory group.
Group 1 (acutely descending BP with slight rebound, high pulse pressure, and persistently low HR, n = 31, 9.32 %): Comprising 31 individuals or 9.32 % of the total sample, this group initially presented with high SBP. There was a sharp decrease in the SBP to 110 mmHg between the 6th to 7th hour. Despite this, a slight rebound was observed, causing the SBP to rise to 115 mmHg. The DBP paralleled the SBP's trajectory, but the pulse pressure (the difference between SBP and DBP) remained notably high at around 70 mmHg. The HR consistently hovered at a low range of approximately 60–65 beats per minute (bpm) throughout.
Group 2 (high to moderate SBP with slight rebound; medium and gradually declining DBP; persistently low HR, n = 54, 16.50 %): This group represents 54 individuals, making up 16.50 % of the total. They began with a high SBP, which showed a gradual decrease, stabilizing at a moderate level of about 120 mmHg. In sync with this, the DBP in this group also demonstrated a steady decline from approximately 70 to 60 mmHg. Echoing Group 1, the HR in Group 2 remained consistently low at approximately 60–65 bpm.
Group 3 (persistently moderate, n = 118, 34.98 %): This group, constituting the largest proportion of the sample (34.98 %), is marked by a steady SBP of approximately 110 mmHg, showing a slight but not significant decrease within the 4–12 h time frame. The DBP exhibited a minor and consistent decline from 60 to 52 mmHg, whereas the HR was steadily maintained at around 75 bpm.
Group 4 (moderate BP and persistently high HR, n = 78, 22.85 %): This group mirrored the BP trend seen in Group 3; however, the HR remained consistently above 90 bpm, significantly higher than the previous groups.
Group 5 (high to moderate SBP with slight rebound; high but gradually declining DBP; slightly high HR, n = 56, 16.34 %): This group displayed a similar SBP trend as seen in Group 2. However, unlike Group 2, the DBP gradually decreased from above 75 to 65 mmHg, and the HR showed a mild fluctuation around 80 bpm.
The basic characteristics of the 337 patients with AAD based on multi-trajectory groups are shown in Table 1. The median age was higher in Group 1 (78.5 years, IQR 72.9, 82.3) and Group 3 (70.6 years, IQR 58.5, 78.8) compared to the other groups. There was also a slight discrepancy in the distribution of race across the groups. Baseline BP and HR upon ICU admission varied between the trajectory groups, highlighting higher SBP in Groups 1, 2, and 5, and higher HR in Groups 4 and 5. Groups 1, 2, and 5 shared a common characteristic - patients exhibited high volatility in SBP (high, declined, then rebound).
Table 1.
Demographic and clinical characteristics.
| [ALL] N = 337 |
Group1 N = 31 |
Group2 N = 54 |
Group3 N = 118 |
Group4 N = 78 |
Group5 N = 56 |
P.overall | |
|---|---|---|---|---|---|---|---|
| Baseline characteristics | |||||||
| Age | 68.6 [58.1; 78.5] | 78.5 [72.9; 82.3] | 69.0 [61.1; 79.1] | 70.6 [58.5; 78.8] | 65.3 [55.4; 75.1] | 62.7 [55.9; 72.3] | <0.001 |
| Gender(Female) | 196 (58.2 %) | 13 (41.9 %) | 29 (53.7 %) | 65 (55.1 %) | 50 (64.1 %) | 39 (69.6 %) | 0.077 |
| Race | 0.042 | ||||||
| Black | 45 (13.4 %) | 4 (12.9 %) | 11 (20.4 %) | 8 (6.78 %) | 11 (14.1 %) | 11 (19.6 %) | |
| Hispanic | 18 (5.34 %) | 2 (6.45 %) | 2 (3.70 %) | 4 (3.39 %) | 5 (6.41 %) | 5 (8.93 %) | |
| Other/Unknown | 91 (27.0 %) | 4 (12.9 %) | 10 (18.5 %) | 38 (32.2 %) | 28 (35.9 %) | 11 (19.6 %) | |
| White | 183 (54.3 %) | 21 (67.7 %) | 31 (57.4 %) | 68 (57.6 %) | 34 (43.6 %) | 29 (51.8 %) | |
| BMI | 27.0 [23.7; 31.6] | 27.4 [21.8; 30.1] | 25.5 [21.3; 34.9] | 27.1 [23.9; 31.2] | 27.3 [23.8; 32.7] | 26.9 [24.0; 31.4] | 0.628 |
| Location | 0.119 | ||||||
| Abdominal aorta | 41 (12.2 %) | 8 (25.8 %) | 9 (16.7 %) | 13 (11.0 %) | 4 (5.13 %) | 7 (12.5 %) | |
| Thoracic aorta | 224 (66.5 %) | 18 (58.1 %) | 34 (63.0 %) | 75 (63.6 %) | 60 (76.9 %) | 37 (66.1 %) | |
| Thoracoabdominal aorta | 65 (19.3 %) | 4 (12.9 %) | 10 (18.5 %) | 28 (23.7 %) | 14 (17.9 %) | 9 (16.1 %) | |
| Unspecified site | 7 (2.08 %) | 1 (3.23 %) | 1 (1.85 %) | 2 (1.69 %) | 0 (0.00 %) | 3 (5.36 %) | |
| Smoking history | 42 (12.5 %) | 6 (19.4 %) | 12 (22.2 %) | 12 (10.2 %) | 5 (6.41 %) | 7 (12.5 %) | 0.055 |
| Vital signs at the beginning | |||||||
| Heart rate | 79.0 [69.0; 87.0] | 66.0 [61.0; 72.5] | 66.0 [61.0; 74.2] | 79.0 [70.0; 85.0] | 89.5 [80.0; 97.0] | 82.5 [74.8; 89.0] | <0.001 |
| Respiration rate | 16.0 [14.0; 19.0] | 16.0 [14.0; 19.0] | 16.0 [14.0; 20.0] | 16.0 [14.0; 20.0] | 16.0 [14.0; 18.0] | 17.0 [14.0; 19.0] | 0.546 |
| Temperature | 36.7 [36.4; 37.0] | 36.6 [36.1; 36.7] | 36.7 [36.4; 36.9] | 36.7 [36.4; 37.0] | 36.8 [36.3; 37.3] | 36.8 [36.4; 37.0] | 0.041 |
| SBP | 118 (21.8) | 121 (21.2) | 129 (22.1) | 112 (18.8) | 112 (21.9) | 124 (21.9) | <0.001 |
| DBP | 62.0 [54.0; 74.2] | 57.5 [46.2; 64.5] | 73.0 [64.0; 81.0] | 58.5 [53.0; 66.8] | 59.0 [49.8; 70.0] | 74.0 [64.5; 84.0] | <0.001 |
| Co-morbidity | |||||||
| Cancer | 12 (3.56 %) | 3 (9.68 %) | 0 (0.00 %) | 4 (3.39 %) | 2 (2.56 %) | 3 (5.36 %) | 0.163 |
| Chronic kidney disease | 40 (11.9 %) | 6 (19.4 %) | 6 (11.1 %) | 12 (10.2 %) | 5 (6.41 %) | 11 (19.6 %) | 0.110 |
| Diabetes | 46 (13.6 %) | 8 (25.8 %) | 9 (16.7 %) | 12 (10.2 %) | 10 (12.8 %) | 7 (12.5 %) | 0.243 |
| Hypertension | 287 (85.2 %) | 28 (90.3 %) | 48 (88.9 %) | 101 (85.6 %) | 63 (80.8 %) | 47 (83.9 %) | 0.683 |
| Coronary diseases | 82 (24.3 %) | 13 (41.9 %) | 17 (31.5 %) | 26 (22.0 %) | 15 (19.2 %) | 11 (19.6 %) | 0.066 |
| Blood biomarkers | |||||||
| Blood urea nitrogen (mg/dL) | 18.0 [13.0; 23.0] | 21.0 [13.0; 29.5] | 16.5 [12.0; 23.5] | 17.0 [14.0; 22.0] | 18.0 [14.0; 23.8] | 18.0 [14.0; 23.0] | 0.351 |
| Creatinine (mg/dL) | 1.00 [0.80; 1.30] | 1.10 [0.80; 1.35] | 1.00 [0.80; 1.20] | 1.00 [0.80; 1.20] | 1.10 [0.90; 1.40] | 1.10 [0.88; 1.33] | 0.303 |
| Sodium (mEq/L) | 139 [137; 141] | 139 [136; 141] | 139 [137; 141] | 139 [136; 142] | 140 [138; 142] | 139 [137; 141] | 0.238 |
| Potassium (mEq/L) | 4.20 [3.80; 4.60] | 4.20 [3.90; 4.80] | 4.10 [3.90; 4.40] | 4.00 [3.70; 4.40] | 4.30 [3.90; 4.70] | 4.20 [3.80; 4.50] | 0.021 |
| Calcium (mg/dL) | 8.70 [8.30; 9.20] | 8.40 [7.97; 8.70] | 8.80 [8.40; 9.25] | 8.60 [8.15; 9.10] | 8.75 [8.10; 9.30] | 8.90 [8.50; 9.20] | 0.042 |
| Medication | |||||||
| Calcium channel blocker | 144 (42.7 %) | 13 (41.9 %) | 27 (50.0 %) | 56 (47.5 %) | 25 (32.1 %) | 23 (41.1 %) | 0.203 |
| ACEI | 126 (37.4 %) | 15 (48.4 %) | 26 (48.1 %) | 41 (34.7 %) | 27 (34.6 %) | 17 (30.4 %) | 0.194 |
| ARB | 38 (11.3 %) | 4 (12.9 %) | 9 (16.7 %) | 14 (11.9 %) | 3 (3.85 %) | 8 (14.3 %) | 0.106 |
| Beta-blocker | 303 (89.9 %) | 30 (96.8 %) | 48 (88.9 %) | 102 (86.4 %) | 74 (94.9 %) | 49 (87.5 %) | 0.222 |
| Analgesic | 259 (76.9 %) | 22 (71.0 %) | 28 (51.9 %) | 96 (81.4 %) | 73 (93.6 %) | 40 (71.4 %) | <0.001 |
| Management | <0.001 | ||||||
| Medical therapy | 225 (66.8 %) | 26 (83.9 %) | 48 (88.9 %) | 83 (70.3 %) | 32 (41.0 %) | 36 (64.3 %) | |
| Endovascular | 86 (25.5 %) | 2 (6.45 %) | 1 (1.85 %) | 28 (23.7 %) | 42 (53.8 %) | 13 (23.2 %) | |
| Surgery | 25 (7.42 %) | 3 (9.68 %) | 5 (9.26 %) | 6 (5.08 %) | 4 (5.13 %) | 7 (12.5 %) | |
| Hybrid | 1 (0.30 %) | 0 (0.00 %) | 0 (0.00 %) | 1 (0.85 %) | 0 (0.00 %) | 0 (0.00 %) | |
| Received ventilation | 180 (53.4 %) | 12 (38.7 %) | 9 (16.7 %) | 66 (55.9 %) | 65 (83.3 %) | 28 (50.0 %) | <0.001 |
| Received CRRT in ICU | 32 (9.50 %) | 4 (12.9 %) | 1 (1.85 %) | 9 (7.63 %) | 13 (16.7 %) | 5 (8.93 %) | 0.045 |
| Severity of illness score | |||||||
| SOFA | 5.00 [2.00; 10.0] | 4.00 [3.00; 7.00] | 2.00 [1.00; 4.75] | 5.00 [2.25; 10.0] | 9.00 [6.00; 13.0] | 3.00 [2.00; 8.25] | <0.001 |
| APSIII | 43.0 [31.0; 64.0] | 46.0 [37.5; 62.0] | 33.5 [25.0; 46.0] | 42.5 [33.0; 63.2] | 58.0 [42.2; 82.8] | 36.5 [24.8; 53.2] | <0.001 |
| OASIS | 34.0 [27.0; 40.0] | 34.0 [29.0; 36.0] | 28.0 [24.0; 32.8] | 34.5 [28.0; 39.0] | 41.0 [35.0; 46.0] | 29.5 [23.8; 38.2] | <0.001 |
| SAPSII | 37.0 [30.0; 45.0] | 38.0 [33.5; 44.0] | 31.0 [27.0; 36.8] | 38.0 [31.0; 48.0] | 40.5 [31.0; 50.0] | 32.5 [23.0; 42.0] | <0.001 |
| AKI in ICU | 280 (83.1 %) | 27 (87.1 %) | 39 (72.2 %) | 102 (86.4 %) | 73 (93.6 %) | 39 (69.6 %) | 0.001 |
| AKI Stages | 0.483 | ||||||
| 1 | 64 (22.9 %) | 5 (18.5 %) | 11 (28.2 %) | 22 (21.6 %) | 15 (20.5 %) | 11 (28.2 %) | |
| 2 | 125 (44.6 %) | 9 (33.3 %) | 19 (48.7 %) | 51 (50.0 %) | 31 (42.5 %) | 15 (38.5 %) | |
| 3 | 91 (32.5 %) | 13 (48.1 %) | 9 (23.1 %) | 29 (28.4 %) | 27 (37.0 %) | 13 (33.3 %) | |
| Hospital LOS (hour) | 267 (232) | 283 (211) | 216 (197) | 250 (212) | 358 (282) | 215 (209) | 0.001 |
| ICU LOS (hour) | 78.5 [43.9; 160] | 91.9 [34.9; 174] | 53.0 [32.1; 88.6] | 85.4 [47.8; 139] | 141 [56.2; 337] | 66.6 [46.4; 117] | <0.001 |
Qualitative data are presented as absolute frequencies and percentage n (%). Quantitative data are presented as mean (SD) or median (first to third quartile).
Abbreviations: BMI = body mass index; SBP = systolic blood pressure; DBP = diastolic blood pressure; ACEI = angiotensin-converting enzyme inhibitor; ARB = angiotensin receptor blocker; SOFA = sequential organ failure assessment; APS III = acute physiology score III; OASIS = oxford acute severity of illness score; SAPS II = simplified acute physiology score II; ICU = intensive care unit; LOS = length of stay.
Treatment characteristics varied among the 5 groups. A majority of the individuals in Group 1, 2, and 3 underwent medical therapy alone (83.9 %, 88.9 %, and 70.3 % respectively), while individuals in Group 4 received more endovascular repair treatments. Regarding medication usage, our analysis revealed no significant differences in the use of ACEI, ARB, CCB, and beta-blockers among the different groups, indicating a generally uniform approach to managing BP and HR across the groups. However, a notable variation was observed in the usage rates of analgesics between the groups, with particularly higher rates in Group 3 and Group 4. This discrepancy suggests differing pain management strategies among the groups. Furthermore, significant differences emerged between the trajectory groups concerning SOFA, APS III, OASIS, and SAPS II scoring systems. All scores for Group 4 were markedly the highest, followed by those of Group 3, indicating a more critical state of health in Group 4 upon ICU admission.
Notably, the incidence of acute kidney injury (AKI) was more prevalent in Group 4 than in the other groups, implying that Group 4 received more aggressive treatment. For instance, they had a significantly higher rate of receiving mechanical ventilation (83.3 %) and CRRT (16.7 %) compared to the other groups (p < 0.05). This group also exhibited the longest durations for both hospital stay and ICU stay.
3.1. Outcomes
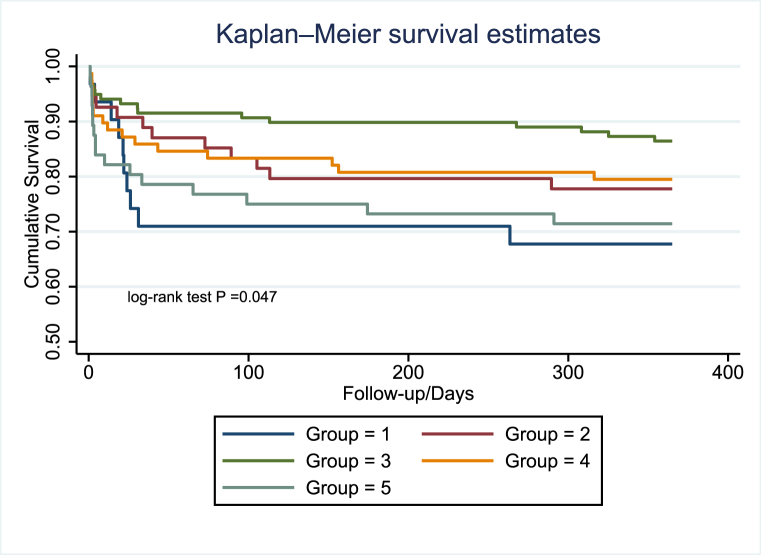
Over the course of a 1-year follow-up period, a total of 70 all-cause deaths were recorded. KM analysis plotted the survival probabilities for each trajectory group, as shown in Fig. 3. Patients in Group 3 demonstrated the lowest mortality rate, while patients in Group 1 exhibited the highest. Notably, Log-rank tests indicated a significant difference in survival probability between groups (p = 0.047).
Fig. 3.
Kaplan–Meier curves with five different trajectories groups.
Appendix Table S2 provides hazard ratios and 95 % confidence intervals (CI) for the association between trajectory groups and mortality. When compared with the reference group (Group 1), the crude hazard ratios (95 % CI) for Group 3 were 0.38 (0.17, 0.83). Even after adjusting for potential confounders, Group 3 maintained its positive association with a reduced risk of mortality. Specifically, in Model 2 and Model 3, the hazard ratio and 95 % CI of mortality in Group 3 were recorded as 0.39 (0.17–0.89) and 0.32 (0.14–0.74), respectively.
RR and 95 % CI estimated using log-binomial regression or poisson regression with robust variance estimator is provided in Table 2. Using Group 1 as the reference, the persistently moderate group (Group 3) showed significantly lower RR of in-hospital mortality, 30-day mortality, and 1-year mortality, with adjusted RR values of 0.37 [95 % CI, 0.15–0.87], 0.25 [95 % CI, 0.10–0.62], and 0.41 [95 % CI, 0.22–0.79], respectively, regardless of whether covariates were included in the model or not. After adjusting for all confounding factors, Group 4 also had lower in-hospital mortality and 30-day mortality than Group 1, with adjusted RR values of 0.42 [95 % CI, 0.18–0.99] and 0.40 [95 % CI, 0.17–0.91], respectively. Similar results were obtained from logistic regression, as shown in Appendix Table S3.
Table 2.
Relative risk (RR) of multi-trajectory groups on in-hospital mortality, 30-day mortality, and 1-year mortality.
| Group1 | Group2 | Group3 | Group4 | Group5 | |
|---|---|---|---|---|---|
| In-hospital mortality | |||||
| Model1 | 1[Reference] | 0.57(0.20–1.63); P = 0.30 | 0.39(0.15–1.02); P = 0.06 | 0.66(0.26–1.67); P = 0.38 | 1.01(0.42–2.48); P = 0.97 |
| Model2 | 1[Reference] | 0.59(0.21–1.64); P = 0.31 | 0.34(0.13–0.87); P = 0.02 | 0.61(0.24–1.53); P = 0.29 | 1.17(0.47–2.89); P = 0.73 |
| Model3 | 1[Reference] | 1.02(0.42–2.46); P = 0.97 | 0.37(0.15–0.87); P = 0.02 | 0.42(0.18–0.99); = 0.05 | 1.64(0.74–3.62); P = 0.22 |
| 30-day mortality | |||||
| Model1 | 1[Reference] | 0.36(013–1.00); P = 0.05 | 0.26(0.11–0.64); P = 0.003 | 0.55(0.24–1.23); P = 0.14 | 0.76(0.34–1.69); P = 0.50 |
| Model2 | 1[Reference] | 0.40(0.14–1.12); P = 0.08 | 0.25(0.10–0.62); P = 0.003 | 0.59(0.27–1.29); P = 0.19 | 1.02(0.46–2.28); P = 0.96 |
| Model3 | 1[Reference] | 0.56(0.21–1.49); P = 0.24 | 0.25(0.10–0.62); P = 0.003 | 0.40(0.17–0.91); P = 0.03 | 1.26(0.60–2.63); P = 0.54 |
| 1-year mortality | |||||
| Model1 | 1[Reference] | 0.69(0.34–1.41); P = 0.31 | 0.42(0.21–0.83); P = 0.01 | 0.64(0.32–1.24); P = 0.19 | 0.89(0.46–1.71); P = 0.72 |
| Model2 | 1[Reference] | 0.75(0.37–1.55); P = 0.44 | 0.41(0.20–0.81); P = 0.01 | 0.68(0.34–1.36); P = 0.28 | 1.11(0.57–2.16); P = 0.76 |
| Model3 | 1[Reference] | 1.05(0.53–2.06); P = 0.90 | 0.41(0.22–0.79); P = 0.01 | 0.55(0.27–1.11); P = 0.10 | 1.30(0.69–2.43); P = 0.42 |
Model 1, no adjustment; Model 2 adjusted for age, gender, and race; Model 3 adjusted for BMI, diabetes, hypertension, SOFA score, AD location, and management based on Model 2.
Results from our sensitivity analyses, which included additional adjustments for the use of analgesics and a comprehensive range of medications (analgesics, ACEI, ARB, CCB, β-Blocker) in Cox regression analyses, were largely consistent with our main findings. Specifically, even after accounting for variability in analgesic use and adjusting for all medications, the prognosis for Group 3 within the multi-trajectory analysis remained most favorable (see Supplementary Table S4 for detailed statistics).
Univariate trajectory groups for SBP, DBP, and HR defined by GBTM separately are presented in the Appendix (Fig. S1). Notably, within these trajectory groups, only the DBP trajectory groups demonstrated a statistically significant correlation with patient prognosis, as evidenced by Appendix Tables S5–S7. The DBP trajectories for the second and third groups exhibited a marked decrease, ranging from 67 to 52 mmHg. This trend was paralleled in the DBP trajectory observed within Group 3 of the multi-trajectory analysis, suggesting these specific groups may be indicative of a more favorable prognosis.
The time-independent C-index curve, presented in Fig. 4, elucidates that the multi-trajectory groups afford a more substantial prognostic contribution compared to any of the univariate trajectory groups or baseline SBP, DBP, and HR measurements obtained at admission.
Fig. 4.
Time-dependent C-index curves for different indicators.
4. Discussion
In this longitudinal study, we identified five distinct multi-trajectory groups, based on SBP, DBP, and HR, that correlated with the prognosis of AAD. Primarily, our results showed that patients in Group 3, characterized by a persistently moderate BP and HR over the 24 h post-ICU admission, demonstrated the lowest risk of all-cause mortality. This included in-hospital mortality, 30-day mortality, and 1-year mortality, independent of factors such as age, gender, SOFA score, and treatment program, among others.
Indeed, while previous studies have explored the association of SBP, DBP, or HR with AAD prognosis, it is noteworthy that our study is the first to jointly explore the trajectories of SBP, DBP, and HR as a unified whole. In comparison with BP and HR at single time points, these trajectories can offer further insights into patients' response to treatment and provide dynamic information.
Our GBMTM analysis may suggest a composite phenotype as an indicator of AAD patients' prognosis. The results of this study propose that maintaining stable control of SBP at around 110 mmHg, DBP at around 55 mmHg, and HR at around 75 bpm could contribute to reducing mortality in critically ill patients with AAD. In our view, this is the most significant finding of our study. Furthermore, the multiple trajectories in Group 3 could serve as a potential target for BP and HR management in patients with AAD within the initial 24 h of ICU admission, thereby providing a valuable reference for physicians.
Current guidelines recommend a target SBP lower than 120 mmHg or 100–120 mmHg [4,17,18]. However, these recommendations are mainly based on early animal experiments and a few single-center retrospective studies (level of evidence: C). Our results are consistent with these recommended threshold values for SBP, but we narrow the threshold further to about 110 mmHg and emphasize the crucial role of stable and steady BP control. Most studies on BP related to AAD have focused on SBP, and many of them are based on admission SBP [7,19,20], whether patients received intensive SBP control [21], and some analyzed SBP variability [22], circadian rhythms [23], and changing patterns [15], taking into account the time factor to some extent. Fewer studies have reported on DBP in aortic dissection. A previous study reported that DBP on admission is associated with 24-month aortic-related adverse events, but an appropriate range of values was not given [24]. In our study, the DBP trajectory of Group 3 trended from about 62 mmHg to 52 mmHg, congruent with the target BP that aims to maintain a systolic pressure below 120 mm Hg or mean pressure below 80 mmHg [1].
The goal of HR control therapy for AAD patients has different standards due to the lack of definite clinical evidence. The Chinese national expert consensus recommends an HR target of 60–80 bpm [17], which differs from the strict target of 60 bpm in the AHA guidelines [18], while some investigators adopt a target of less than 70 bpm [5]. Our study suggests that a stable HR of around 75 achieves better prognosis, falling within the recommended range by the Chinese guidelines. Some retrospective studies with small sample sizes have shown better prognosis for patients with an HR of 70–80 bpm [25,26], although due to the small sample size, a significant conclusion cannot be made. Further prospective studies with large sample sizes are necessary for HR control.
GBMTM has been successfully employed to concurrently estimate the developmental trajectories of other related indicators, such as lipid indices (including low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and triglycerides) [27], as well as identify trajectories based on ultrasound measures of fetal growth (concurrent head circumference, abdominal circumference, and femur length measures) [28]. In the intensive care unit, there are numerous continuous monitoring time series data. We believe that the GBMTM method can be extended to other monitoring indicators of various diseases, providing a comprehensive description of similar correlation indicator trajectories.
In this study, we jointly analyzed three important vital signs (SBP/DBP/HR) as a whole and characterized the patients into five different trajectory groups over time (hours) by GBMTM. The association between grouping and prognosis was robust to various types of adjustment and consistent across different statistical methods. Our sensitivity analyses, which adjusted for the use of analgesics and a comprehensive range of medications, including ACEI, ARB, CCB, and β-Blockers, confirmed the robustness of our primary findings. Notably, the consistently favorable prognosis in Group 3, even under varied treatment regimens, highlights the robustness of our main conclusions against potential confounding factors related to BP and HR management strategies. This consistency suggests that while individual medication choices might vary, the overall treatment effect within our identified trajectory groups remains a significant predictor of outcomes in AAD patients.
Univariate (DBP) trajectory groups defined by GBTM also showed a different risk of death. However, compared with univariate trajectory grouping, multi-trajectory grouping jointly assesses the level and trajectory of SBP, DBP, and HR, providing more comprehensive information for monitoring vital signs. The time-independent C-index curve further substantiated that the multi-trajectory groups had a higher C-index than any of the univariate trajectory groups or baseline measurements of SBP, DBP, and HR. This finding underscores the importance of dynamic monitoring of multiple indicators. The enhanced predictive capability of this multivariate trajectory analysis offers a more nuanced and comprehensive understanding of patient prognosis.
One of the key strengths of our study is the application of GBMTM in cohorts with repeated measurements of SBP, DBP, and HR, which enabled us to delineate common temporal patterns of multiple related parameters. This approach allowed us to view the three measures as a unified entity, thereby identifying the distinct trajectory subgroups that influence the risk of patient mortality. The identified trajectories of this study build upon the results from previous studies that have examined the factors separately at specific time points, and provide comprehensive dynamic information.
However, this study has some limitations. Firstly, being a single-center retrospective study, it may exhibit selection and observation biases, potentially affecting the generalizability of the findings. Secondly, although a wide range of mortality predictors for AAD patients have been identified—including, but not limited to, a history of aortic valve replacement, migrating chest pain, hypotension, shock, tamponade, as well as various preoperative conditions and biochemical markers—our dependence on publicly available databases imposed significant limitations. Specifically, these constraints prevented access to comprehensive data on patient symptoms, signs, and key biomarkers like D-dimer and C-reactive protein, leading to a considerable amount of missing information. Furthermore, the relatively small sample size limited our ability to include a more extensive set of covariates in our analysis [29,30]. To account for these limitations, the covariates included the SOFA score as a comprehensive indicator to indirectly evaluate key functions across several systems, including respiratory, cardiovascular, hepatic, coagulation, renal, and neurological systems. Additionally, we leveraged multiple statistical methodologies to estimate hazard rates, odds ratios (OR), and RR, and we meticulously adjusted for covariates at varying levels in multivariate analyses to enhance the robustness and reliability of our findings. Thirdly, GBTM and other trajectory analysis methods may not always accurately delineate individual trajectories, leading to potential misclassification [31,32]. The groups identified through GBTM are based on probabilistic assignments, which might introduce an unquantifiable level of uncertainty into regression analyses. Furthermore, non-identifiability presents another concern, as GBTM assigns group membership based on models that may lack sufficient generalization, potentially weakening the estimates of relationships between trajectories and outcomes [33]. GBMTM exacerbates these challenges by necessitating substantial data for model training and complicating the interpretation of multiple trajectory groups. These concerns are not exclusive to GBTM but are inherent in latent class modeling. Exploring various trajectory modeling techniques to confirm our results is highly recommended.
Our findings highlight the need for future multi-center, prospective studies with larger sample sizes to validate these results and to further investigate the optimal control strategies for BP and HR in AAD patients in the ICU.
5. Conclusions
Using the GBMTM approach, data-driven observations may help identify subphenotypes of AAD patients. The combined trajectory of SBP, DBP, and HR within 24 h of ICU admission had an impact on in-hospital mortality, 30-day mortality, and 1-year mortality. The findings can contribute to developing better care for AAD patients in ICU.
Ethics approval and consent to participate
The MIMIC IV database has received ethical approval from the Institutional Review Board at the Beth Israel Deaconess Medical Center(Boston, MA, USA), who granted a waiver of informed consent and approved the data sharing initiative. All data in this database are de-identified to remove patient information.
Data availability statement
The MIMIC-IV includes comprehensive and high-quality data of patients admitted to intensive care units (ICUs) at the Beth Israel Deaconess Medical Center (BIDMC) between 2008 and 2019, developed by the computational physiology laboratory of Massachusetts Institute of Technology (MIT) and approved by the institutional review boards of MIT and BIDMC. After passing the “Protecting Human Research Participants” exam on the website of the National Institutes of Health (NIH), the author was approved to extract data from this database.
Funding
This work was supported by 2022 Guangdong Province Science and Technology Special Fund (2022-124-6).
CRediT authorship contribution statement
Xuejun Shen: Conceptualization, Data curation, Methodology, Software, Writing – original draft, Writing – review & editing. Jufang Li: Data curation, Methodology, Writing – original draft. Hongle Yan: Writing – original draft, Data curation, Methodology. Shuyi Zhou: Methodology, Writing – original draft. Shengli Yang: Conceptualization, Formal analysis, Methodology, Resources, Supervision, Writing – review & editing. Weiping Li: Conceptualization, Methodology, Supervision, Validation, Writing – original draft, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
We thank Engineer Zefeng Yu, Shen Chen, and Hua Zhu for their guidance and help in using the database. We also acknowledge Dr. Stanley Lin for revising the manuscript for English language and thank Dr. You Mo for discussions and sharing experiences.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.heliyon.2024.e29934.
Contributor Information
Shengli Yang, Email: slyang@caeshc.com.cn.
Weiping Li, Email: wpli818@126.com.
Abbreviations
- SBP
systolic blood pressure
- DBP
diastolic blood pressure
- HR
heart rate
- AAD
acute aortic dissection
- MIMIC
the Medical Information Mart for Intensive Care
- GBTM
group-based trajectory modelling
- GBMTM
group-based multi-trajectory modeling
- ICD
International Classification of Diseases
- SQL
Structured query language
- CKD
chronic kidney disease
- CRRT
continuous renal replacement therapy
- SOFA
Sequential Organ Failure Assessment
- APS III
Acute Physiology Score III
- OASIS
the Oxford Acute Severity of Illness Score
- SAPS II
Simplified Acute Physiology Score II
- AKI
acute kidney injury
- RR
relative risks
- ICU
intensive care unit
- BMI
body mass index
- TEVAR
thoracic endovascular aortic repair
- OR
odds ratio
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Mussa F.F., Horton J.D., Moridzadeh R., Nicholson J., Trimarchi S., Eagle K.A. Acute aortic dissection and intramural hematoma: a systematic review. JAMA. 2016;316(7):754–763. doi: 10.1001/jama.2016.10026. [DOI] [PubMed] [Google Scholar]
- 2.Evangelista A., Isselbacher E.M., Bossone E., Gleason T.G., Eusanio M.D., Sechtem U., Ehrlich M.P., Trimarchi S., Braverman A.C., Myrmel T., Harris K.M., Hutchinson S., O'Gara P., Suzuki T., Nienaber C.A., Eagle K.A., Investigators I. Insights from the international registry of acute aortic dissection: a 20-year experience of collaborative clinical research. Circulation. 2018;137(17):1846–1860. doi: 10.1161/CIRCULATIONAHA.117.031264. [DOI] [PubMed] [Google Scholar]
- 3.Yu X., Suki B., Zhang Y. Avalanches and power law behavior in aortic dissection propagation. Sci. Adv. 2020;6(21) doi: 10.1126/sciadv.aaz1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Erbel R., Aboyans V., Boileau C., Bossone E., Bartolomeo R.D., Eggebrecht H., Evangelista A., Falk V., Frank H., Gaemperli O., Grabenwoger M., Haverich A., Iung B., Manolis A.J., Meijboom F., Nienaber C.A., Roffi M., Rousseau H., Sechtem U., Sirnes P.A., Allmen R.S., Vrints C.J., EsccfP Guidelines. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC) Eur. Heart J. 2014;35(41):2873–2926. doi: 10.1093/eurheartj/ehu281. [DOI] [PubMed] [Google Scholar]
- 5.MacGillivray T.E., Gleason T.G., Patel H.J., Aldea G.S., Bavaria J.E., Beaver T.M., Chen E.P., Czerny M., Estrera A.L., Firestone S., Fischbein M.P., Hughes G.C., Hui D.S., Kissoon K., Lawton J.S., Pacini D., Reece T.B., Roselli E.E., Stulak J. The society of thoracic surgeons/American association for thoracic surgery clinical practice guidelines on the management of type B aortic dissection. Ann. Thorac. Surg. 2022;113(4):1073–1092. doi: 10.1016/j.athoracsur.2021.11.002. [DOI] [PubMed] [Google Scholar]
- 6.Hiratzka L.F., Bakris G.L., Beckman J.A., Bersin R.M., Carr V.F., Casey D.E., Jr., Eagle K.A., Hermann L.K., Isselbacher E.M., Kazerooni E.A., Kouchoukos N.T., Lytle B.W., Milewicz D.M., Reich D.L., Sen S., Shinn J.A., Svensson L.G., Williams D.M., American College of Cardiology Foundation/American Heart Association Task Force on Practice G, American Association for Thoracic S, American College of R, American Stroke A, Society of Cardiovascular A, Society for Cardiovascular A, Interventions, Society of Interventional R, Society of Thoracic S, Society for Vascular M 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with thoracic aortic disease: a report of the American college of cardiology foundation/American heart association task force on practice guidelines, American association for thoracic surgery, American college of radiology, American stroke association, society of cardiovascular anesthesiologists, society for cardiovascular angiography and interventions, society of interventional radiology, society of thoracic surgeons, and society for vascular medicine. Circulation. 2010;121(13):e266–e369. doi: 10.1161/CIR.0b013e3181d4739e. [DOI] [PubMed] [Google Scholar]
- 7.Bossone E., Gorla R., LaBounty T.M., Suzuki T., Gilon D., Strauss C., Ballotta A., Patel H.J., Evangelista A., Ehrlich M.P., Hutchison S., Kline-Rogers E., Montgomery D.G., Nienaber C.A., Isselbacher E.M., Eagle K.A. Presenting systolic blood pressure and outcomes in patients with acute aortic dissection. J. Am. Coll. Cardiol. 2018;71(13):1432–1440. doi: 10.1016/j.jacc.2018.01.064. [DOI] [PubMed] [Google Scholar]
- 8.Nagin D.S., Odgers C.L. Group-based trajectory modeling in clinical research. Annu. Rev. Clin. Psychol. 2010;6:109–138. doi: 10.1146/annurev.clinpsy.121208.131413. [DOI] [PubMed] [Google Scholar]
- 9.Nagin D.S., Jones B.L., Passos V.L., Tremblay R.E. Group-based multi-trajectory modeling. Stat. Methods Med. Res. 2018;27(7):2015–2023. doi: 10.1177/0962280216673085. [DOI] [PubMed] [Google Scholar]
- 10.Cooney E.E., Reisner S.L., Saleem H.T., Althoff K.N., Beckham S.W., Radix A., Cannon C.M., Schneider J.S., Haw J.S., Rodriguez A.E., Wawrzyniak A.J., Poteat T.C., Mayer K.H., Beyrer C., Wirtz A.L., American Cohort To Study HIVAATWSG Prevention-effective adherence trajectories among transgender women indicated for PrEP in the United States: a prospective cohort study. Ann. Epidemiol. 2022;70:23–31. doi: 10.1016/j.annepidem.2022.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tanaka K., Koga M., Fukuda-Doi M., Qureshi A.I., Yamamoto H., Miwa K., Ihara M., Toyoda K., Investigators A.-T. Temporal trajectory of systolic blood pressure and outcomes in acute intracerebral hemorrhage: ATACH-2 trial cohort. Stroke. 2022;53(6):1854–1862. doi: 10.1161/STROKEAHA.121.037186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang F., Peng C., Peng L., Wang P., Cheng C., Zuo W., Zhao L., Jin Z., Li W. Group-based trajectory modeling of intracranial pressure in patients with acute brain injury: results from multi-center ICUs, 2008-2019. CNS Neurosci. Ther. 2022;28(8):1218–1228. doi: 10.1111/cns.13854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bhavani S.V., Wolfe K.S., Hrusch C.L., Greenberg J.A., Krishack P.A., Lin J., Lecompte-Osorio P., Carey K.A., Kress J.P., Coopersmith C.M., Sperling A.I., Verhoef P.A., Churpek M.M., Patel B.K. Temperature trajectory subphenotypes correlate with immune responses in patients with sepsis. Crit. Care Med. 2020;48(11):1645–1653. doi: 10.1097/CCM.0000000000004610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Johnson A.B.L., Pollard T., Horng S., Celi L.A., Mark R. PhysioNet.; 2022. MIMIC-IV. 2022. [DOI] [Google Scholar]
- 15.Wu Z., Li Y., Qiu P., Liu H., Liu K., Li W., Wang R., Chen T., Lu X. Prognostic impact of blood pressure change patterns on patients with aortic dissection after admission. Front Cardiovasc Med. 2022;9 doi: 10.3389/fcvm.2022.832770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Oliveira M., Rocha W.E.M., Soares J.D., L'Armee V., Martins M.P.G., Rocha A.M., Feitosa A.D.M., Lima R.C., Oliveira P.P.M., Silveira-Filho L.M., Coelho-Filho O.R., Matos-Souza J.R., Petrucci O Jr, Sposito A.C., Nadruz W Jr. Impact of hypertension history and blood pressure at presentation on cardiac remodeling and mortality in aortic dissection. Front Cardiovasc Med. 2021;8 doi: 10.3389/fcvm.2021.803283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Surgeons tCoGVoCAoC Chinese experts' consensus of standardized diagnosis and treatment for aortic dissection. Chinese Journal of Thoracic and Cardiovascular Surgery. 2017;33(11):641–654. [Google Scholar]
- 18.Hiratzka L.F., Bakris G.L., Beckman J.A., Bersin R.M., Carr V.F., Casey D.E., Jr., Eagle K.A., Hermann L.K., Isselbacher E.M., Kazerooni E.A., Kouchoukos N.T., Lytle B.W., Milewicz D.M., Reich D.L., Sen S., Shinn J.A., Svensson L.G., Williams D.M., American College of Cardiology Foundation/American Heart Association Task Force on Practice G, American Association for Thoracic S, American College of R, American Stroke A, Society of Cardiovascular A, Society for Cardiovascular A, Interventions, Society of Interventional R, Society of Thoracic S, Society for Vascular M 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with thoracic aortic disease. A report of the American college of cardiology foundation/American heart association task force on practice guidelines, American association for thoracic surgery, American college of radiology,American stroke association, society of cardiovascular anesthesiologists, society for cardiovascular angiography and interventions, society of interventional radiology, society of thoracic Surgeons,and society for vascular medicine. J. Am. Coll. Cardiol. 2010;55(14):e27–e129. doi: 10.1016/j.jacc.2010.02.015. [DOI] [PubMed] [Google Scholar]
- 19.Yang G., Peng W., Zhou Y., He H., Pan X., Li X., Chai X. Admission systolic blood pressure and in-hospital mortality in acute type A aortic dissection: a retrospective observational study. Front. Med. 2021;8 doi: 10.3389/fmed.2021.542212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tolenaar J.L., Froehlich W., Jonker F.H., Upchurch G.R., Jr., Rampoldi V., Tsai T.T., Bossone E., Evangelista A., O'Gara P., Pape L., Montgomery D., Isselbacher E.M., Nienaber C.A., Eagle K.A., Trimarchi S. Predicting in-hospital mortality in acute type B aortic dissection: evidence from international registry of acute aortic dissection. Circulation. 2014;130(11 Suppl 1):S45–S50. doi: 10.1161/CIRCULATIONAHA.113.007117. [DOI] [PubMed] [Google Scholar]
- 21.Qian S., Ding X., Liu H., He X., Wang S., Du Y., Zhang H., Li H. 2022. Does Intensive Blood Pressure Control Benefit Type B Aortic Dissection Patients Who Undergoing Surgical Repair? Perfusion. [DOI] [PubMed] [Google Scholar]
- 22.Zhang L., Tian W., Feng R., Song C., Zhao Z., Bao J., Liu A., Su D., Zhou J., Jing Z. Prognostic impact of blood pressure variability on aortic dissection patients after endovascular therapy. Medicine (Baltim.) 2015;94(38) doi: 10.1097/MD.0000000000001591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Delsart P., Ledieu G.J., Ramdane N., Sobocinski J.P., Clough R.E., Azzaoui R.O., Mounier-Vehier C., Nienaber C.A., Haulon S. Impact of the management of type B aortic dissection on the long-term blood pressure. Am. J. Cardiol. 2017;120(3):484–488. doi: 10.1016/j.amjcard.2017.04.052. [DOI] [PubMed] [Google Scholar]
- 24.Lu N., Xu T., He Z., Zhu J., Yan J., Hu P., Wang J., Ma X., Tan X. Diastolic, but not systolic, blood pressure at admission is associated with aortic-related adverse events in type B dissection after thoracic endovascular aortic repair. Int. J. Cardiol. 2020;299:257–262. doi: 10.1016/j.ijcard.2019.07.064. [DOI] [PubMed] [Google Scholar]
- 25.Kumar K.U., Zhao Q., Bai X., Li A., Anjali P., Yu H., Zhu W., Zou T., Ma Y., Ma X. Controlled heart rate and blood pressure reduce the life threatening aortic events and increase survival in patients with type B aortic dissection: a single center experience. Int J Cardiol Heart Vasc. 2015;8:73–74. doi: 10.1016/j.ijcha.2015.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhou Y., Luo Q., Guo X., Wang H., Jia Y., Cao L., Wang Y., Yan F., Yu C., Yuan S. Predictive value of heart rate in patients with acute type A aortic dissection: a retrospective cohort study. BMJ Open. 2021;11(11) doi: 10.1136/bmjopen-2020-047221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Koohi F., Khalili D., Mansournia M.A., Hadaegh F., Soori H. Multi-trajectories of lipid indices with incident cardiovascular disease, heart failure, and all-cause mortality: 23 years follow-up of two US cohort studies. J. Transl. Med. 2021;19(1):286. doi: 10.1186/s12967-021-02966-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bommarito P.A., Cantonwine D.E., Stevens D.R., Welch B.M., Davalos A.D., Zhao S., McElrath T.F., Ferguson K.K. An application of group-based trajectory modeling to define fetal growth phenotypes among small-for-gestational-age births in the LIFECODES Fetal Growth Study. Am. J. Obstet. Gynecol. 2023;228(3):334 e331–e334 e321. doi: 10.1016/j.ajog.2022.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vittinghoff E., McCulloch C.E. Relaxing the rule of ten events per variable in logistic and Cox regression. Am. J. Epidemiol. 2007;165(6):710–718. doi: 10.1093/aje/kwk052. [DOI] [PubMed] [Google Scholar]
- 30.Riley R.D., Ensor J., Snell K.I.E., Harrell F.E., Jr., Martin G.P., Reitsma J.B., Moons K.G.M., Collins G., van Smeden M. Calculating the sample size required for developing a clinical prediction model. BMJ. 2020;368 doi: 10.1136/bmj.m441. [DOI] [PubMed] [Google Scholar]
- 31.Mesidor M., Rousseau M.C., O'Loughlin J., Sylvestre M.P. Does group-based trajectory modeling estimate spurious trajectories? BMC Med. Res. Methodol. 2022;22(1):194. doi: 10.1186/s12874-022-01622-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gao Y., Luo J., Zhang Y., Pan C., Ren Y., Zhang J., Tian Y., Shanghai Birth C. Prenatal exposure to per- and polyfluoroalkyl substances and child growth trajectories in the first two years. Environ. Health Perspect. 2022;130(3) doi: 10.1289/EHP9875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Memoli V., Ekanmian G., Lunghi C., Bouhnik A.D., Lauzier S., Guenette L. What methods are used to study the association between medication adherence trajectories, estimated with the group-based trajectory modeling (GBTM) method, and health-related outcomes?-a protocol for a systematic review. Syst. Rev. 2022;11(1):102. doi: 10.1186/s13643-022-01971-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The MIMIC-IV includes comprehensive and high-quality data of patients admitted to intensive care units (ICUs) at the Beth Israel Deaconess Medical Center (BIDMC) between 2008 and 2019, developed by the computational physiology laboratory of Massachusetts Institute of Technology (MIT) and approved by the institutional review boards of MIT and BIDMC. After passing the “Protecting Human Research Participants” exam on the website of the National Institutes of Health (NIH), the author was approved to extract data from this database.