Abstract
Background
Pneumocystis pneumonia is an uncommon precipitant of acute respiratory distress syndrome and is associated with high mortality. Prone positioning ventilation has been proven to reduce mortality in patients with moderate–severe acute respiratory distress syndrome. We investigated the effect of prone positioning on oxygenation and mortality in intubated patients with pneumocystis pneumonia comorbid with moderate–severe acute respiratory distress syndrome.
Methods
In this single-center, retrospective, observational, cohort study, eligible patients were enrolled at West China Hospital of Sichuan University from January 1, 2017, to December 31, 2021. Data on demographics, clinical features, ventilation parameters, arterial blood gas, and outcomes were collected. Patients were assigned to the prone cohort or supine cohort according to whether they received prone positioning ventilation. The main outcome was 28-day mortality.
Findings
A total of 79 patients were included in the study. Sixty-three patients were enrolled in the prone cohort, and 16 patients were enrolled in the supine cohort. The 28-day mortality was 61.9% in the prone cohort and 68.8% in the supine cohort (P = 0.26), and 90-day mortality was 66.7% in the prone cohort and 68.8% in the supine cohort (P = 0.55). Patients in the supine cohort had fewer invasive mechanical ventilation days and more ventilator-free days. The incidence of complications was higher in the prone cohort than in the supine cohort.
Conclusions
In patients with pneumocystis pneumonia and moderate–severe acute respiratory distress syndrome, prone positioning did not decrease 28-day or 90-day mortality.
Trial registration ClinicalTrials.gov number, ChiCTR2200063889. Registered on 20 September 2022, https://www.chictr.org.cn/showproj.html?proj=174886.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40001-024-01868-7.
Keywords: PJP, Prone positioning, Moderate–severe ARDS
Introduction
Pneumocystis pneumonia (PCP) is an opportunistic pulmonary infection that occurs in immunocompromised or HIV-infected individuals or patients with malignancy [1, 2]. Patients with PCP may develop symptoms, such as fever, cough, progressive dyspnea, and hypoxemia [3, 4]. PCP is an uncommon precipitant of acute respiratory distress syndrome (ARDS) [5], but is associated with a high mortality rate of up to 0% to 20% in HIV-infected patients and 30–75% in non-HIV-infected patients [1, 4–6]. However, quality evidence regarding how to ventilate patients with critical PCP is lacking.
The fungus Pneumocystis jirovecii is transmitted through the airborne route [4], and computed tomography (CT) scans of PCP show predominant ground-glass opacification (GGO) with diffuse, bilateral and central locations in the early stage and patchy or focal consolidation in the middle or late stages [7–10], indicating a more homogeneous ventilation distribution between the ventral lung, middle lung and dorsal lung than “typical” ARDS. Prone positioning ventilation (PPV) has been used to treat hypoxemia in patients with severe or moderate ARDS for several years [11]. The principle underlying PPV for remedying hypoxemia involves improving ventilation and alveolar recruitment in the dorsal lung, the so-called “dependent pulmonary units”, thus improving the ventilation/perfusion ratio and reduction in intrapulmonary shunting [12–14]. However, it is unclear whether a patient with PCP can benefit from PPV treatment. To our knowledge, there is no study or case report available referring to the efficacy of prone positioning in PCP patients.
In this retrospective, cohort study, it was hypothesized that PPV would not improve oxygenation or reduce mortality in intubated patients with PCP and moderate–severe ARDS.
Material and methods
Study design and participants
In this single-center, retrospective, observational, cohort study, we recruited eligible patients admitted to the medical intensive care unit (MICU) and center intensive care unit (CICU) of West China Hospital of Sichuan University, a large tertiary teaching hospital, between January 1, 2017, and December 31, 2021.
Patients were eligible to be included in the study if they were between 18–85 years of age, were diagnosed with PCP by etiological examination (fungal smear/cultures or metagenomics next generation sequencing) of sputum or bronchoalveolar lavage fluid, and received invasive mechanical ventilation targeted at moderate–severe ARDS for less than 72 h prior to admission to the ICU.
Exclusion criteria were pregnancy, advanced carcinoma or massive hemorrhage, and invasive mechanical ventilation less than 72 h after admission to the ICU.
Patients who received PPV treatment were enrolled in the prone cohort, and those who received no PPV were enrolled in the supine cohort.
PPV and mechanical ventilation strategies
The PPV procedure conducted in our study was based on the protocol published as part of the PROSEVA trial [15]. The durations mostly occurred for 12 h [11, 16] and were extended to 16–18 h when necessary [15, 17]. Patients were eligible to receive PPV if the PaO2/FiO2 ratio was lower than 200 mmHg (typically lower than 150 mmHg), upon evaluation by their attending physician. Complications leading to immediate termination of PPV included unexpected artificial airway extubation or obstruction, hemoptysis, oxygen saturation less than 85% on pulse oximetry for more than 2 min when the FiO2 was 1.0, cardiac arrest, a heart rate of less than 50 beats or more than 160 beats per minute for more than 30 s, a systolic blood pressure of less than 90 mm Hg for more than 5 min, or any other life-threatening incidents.
Mechanical ventilation was delivered according to the ALVEOLI study [18], with end-inspiratory plateau pressure (Pplat) maintained below 30 cm H2O, and volumes guaranteed between 6–8 mL/kg PBW or 4–6 mL/kg PBW during extracorporeal membrane oxygenation (ECMO) therapy. Permissive hypercapnia was flexibly performed when lung-protective ventilation was impeded, with pH values of 7.30–7.35 and 7.25–7.35 in patients with chronic obstructive pulmonary disease.
Outcome measures
The primary end point was mortality at 28 days. The secondary endpoints were mortality at 90 days, the rate of successful extubation on day 28, the length of invasive mechanical ventilation days and ventilator-free days at 28 days, the length of ICU stay on day 90, the pneumothorax incidence rate, and the tracheotomy rate at 90 days.
Successful extubation was defined as no reintubation for 48 h after extubation. In patients who had undergone tracheotomy, successful weaning from the ventilator was defined as the ability to breathe through the tracheostomy cannula for at least 48 h with oxygen therapy or high-flow oxygen therapy.
Data source
We collected data via the hospital electronic health record system, including age, sex, body mass index, Sequential Organ Failure Assessment (SOFA) score, Simplified Acute Physiology Score II (SAPS II), smoking history, patient origin, coexisting conditions, coexisting germs in the respiratory system, anti-PCP drug before ICU admission, cointerventions, ventilator settings and arterial blood gas (ABG) parameters, laboratory tests and radiological characteristics.
Chronic obstructive pulmonary disease, asthma, and interstitial lung disease were recorded as chronic bronchopulmonary disease, while patients using immuno-suppressive drugs, such as cyclosporin A, protopic, mycophenolate mofetil, azathioprine, and prednisone were considered receiving immuno-suppressive therapies.
For the prone cohort, ventilator settings, ABG values and adverse events were collected for every single PPV session at three time points: 0–2 h before prone positioning (SPV1), 0–2 h after shifting to prone positioning (PPV) and 1–2 h after supervised repositioning (SPV2).
Statistical analysis
Efficacy analyses were performed using the full analysis set, and patients who received at least one PPV session were assigned to the prone cohort. To investigate the effect of PPV on oxygenation, we compared patient PaO2/FiO2 ratios between the SPV1 and PPV time points in the first PPV session and compared the PaO2/FiO2 ratio between cohorts during the first 3 ICU days. We compared the PaO2/FiO2 of the PPV versus SPV1 time points. The effects of prone positioning were classified into three degrees according to the change in the PaO2/FiO2 ratio between PPV and SPV1 as follows: type A represented conditions for which the PaO2/FiO2 ratio increased by over 15%, type B represented conditions for which the PaO2/FiO2 ratio changed between − 15% and + 15%, and type C represented conditions for which the PaO2/FiO2 ratio decreased by over 15%. Adverse events occurring in the first 3 ICU days in both cohorts were compared. We chose patients who received at least 3 consecutive PPV sessions in the prone cohort and all patients from the supine cohort to compare PaO2/FiO2 trends between cohorts, and PaO2/FiO2 values were gathered corresponding to the supine position after PPV. The analysis was performed with repeated-measures ANOVA and the ΔPaO2/FiO2 from D2 to D1 and D3 to D1 were compared by Student’s t test.
Continuous variables are expressed as means with standard deviations (SD) or quartiles (upper quartile Q1, median Q2, lower quartile Q3), according to the distribution, and categorical variables are described as percentages. Continuous variables were compared between groups with Student’s t test or Mann–Whitney U tests, based on the distribution, and categorical variables were compared by Chi-square test or Fisher’s exact test. Patient survival was analyzed by the Kaplan–Meier method and compared between groups with the use of the log-rank test. Factors associated with mortality were identified with Cox proportional hazards regression, and the results were expressed as hazard ratios (HRs) with 95% CI. The proportional hazard assumption was verified using the Schoenfeld test.
Missing data were appended by mean completer or regression completer according to their randomization. For patients who were transferred to other hospitals, we contacted their family members by phone to obtain information on their outcomes.
The statistical analysis was performed using R software (R for Mac, version 4.2.3) and Prism (for Mac, version 9.4.1). All reported P values were two-sided, and a P value of less than 0.05 was considered to indicate statistical significance.
Results
Patient characteristics
From January 1, 2017, to December 31, 2021, a total of 137 intubated patients with PCP were selected, 58 patients were subsequently excluded, and 79 patients were included in the current study (Fig. 1). Patients in the prone cohort received at least one prone positioning session (median 5, IQR 2–7) and underwent continuous PPV with an average duration of 12.6 ± 2.8 h. Table 1 shows the characteristics, cointerventions, ventilator settings and arterial blood gas measurements of the study population. There was no significant difference between the two cohorts. Laboratory tests and typical radiologic characteristics of the study population are provided in Table 2. There were no significant differences between the two cohorts.
Fig. 1.
Study profile. PCP pneumocystis pneumonia, ICU intensive care unit, DNI do not intubate, PPV prone positioning ventilation, SPV supine positioning cohort
Table 1.
Characteristics of the participants at inclusion in the study
| Characteristic | Supine cohort (n = 16) | Prone cohort (n = 63) | P value |
|---|---|---|---|
| Age, years | 52.9 (13.5) | 53.6 (14.5) | 0.861 |
| Male sex, no. (%) | 10 (62.5) | 36 (57.1) | 0.917 |
| Body mass index (kg.m−2), Q2 (Q1, Q3) | 21.3 (20.5, 21.3) | 21.9 (19.8, 23.4) | 0.152 |
| SOFA, Q2 (Q1, Q3) | 8.3 (6.0, 12.0) | 8.0 (5.0, 9.0) | 0.619 |
| SAPS II | 39.4 (10.2) | 37.3(8.1) | 0.439 |
| Smoking history, no. (%) | |||
| Never smoked | 9 (56.3) | 43 (68.3) | 0.504 |
| Ever smoker | 3 (18.8) | 11 (17.5) | |
| Active smoker | 4 (25.0) | 9 (14.3) | |
| Patient origin, no. (%) | |||
| Emergency room | 6 (37.5) | 28 (44.4) | 0.282 |
| Inpatient department | 9 (56.3) | 35 (55.6) | |
| Other | 1 (6.3) | 0 | |
| Coexisting conditions, no. (%) | |||
| Diabetes | 4 (25.0) | 16 (25.4) | 1.000 |
| Chronic bronchopulmonary disease | 4 (25.0) | 10 (15.9) | 0.466 |
| Chronic heart disease | 0 | 3 (4.8) | 1.000 |
| Chronic renal disease | 8 (50.0) | 22(34.9) | 0.387 |
| Solid organ transplant | 4 (25.0) | 9 (14.3) | 0.449 |
| Malignancy | 1 (6.3) | 9 (14.3) | 0.677 |
| Hypogammaglobulinemia | 7 (43.8) | 22 (34.9) | 0.568 |
| Immuno-suppressive therapies for at least 3 months | 10 (62.5) | 33 (52.4) | 0.578 |
| HIV positive | 1 (6.3) | 2 (3.2) | 0.498 |
| Coexisting germ, no. (%) | |||
| Bacteria | 11 (68.8) | 37 (58.7) | 0.572 |
| Other fungal | 6 (37.5) | 17 (37.0) | 0.535 |
| Virus | 12 (75.0) | 41 (65.1) | 0.559 |
| Mycoplasma/chlamydia | 1 (6.3) | 1 (1.6) | 0.366 |
| Anti-PCP drug before ICU admission, no. (%) | 10 (62.5) | 50 (79.4) | 0.194 |
| NIV before intubation, no(%) | 11 (68.8) | 54 (85.7) | 0.144 |
| Co-interventions, n (%) | |||
| Vasopressors | 9 (56.3) | 25 (39.7) | 0.268 |
| Neuromuscular blockers | 8 (50.0) | 36 (57.1) | 0.779 |
| Renal replacement therapy | 4 (25.0) | 21 (33.3) | 0.764 |
| ECMO therapy | 0 | 6 (9.5) | 0.338 |
| Ventilator settings and arterial blood gas measurements at the time of inclusiona | |||
| Vt, ml | 411 (35.0) | 400 (48.0) | 0.128 |
| Vt/per kg of PBW | 6.9 (0.4) | 6.7 (0.9) | 0.749 |
| PEEP, cmH2O, Q2 (Q1, Q3) | 11.0 (10.0, 12.5) | 10.0 (8.5,12.0) | 0.382 |
| FiO2 | 0.8 (0.2) | 0.8 (0.2) | 0.754 |
| PaO2, mmHg, Q2 (Q1, Q3) | 71.5 (61.7, 83.5) | 75.3 (65.4, 102.0) | 0.157 |
| PaCO2, mmHg | 45.3 (15.0) | 47.8 (17.4) | 0.566 |
| pH, Q2 (Q1, Q3) | 7.31 (7.29,7.38) | 7.37 (7.30, 7.42) | 0.247 |
| PaO2/FiO2, mmHg, Q2 (Q1, Q3) | 97.1 (72.7, 115.2) | 110.3(94.1, 130.4) | 0.075 |
| Lactate, mmol/L | 1.9 (0.82) | 1.9 (0.94) | 0.930 |
| Plasma bicarbonate, mmol/L, Q2 (Q1, Q3) | 19.9 (19.2, 24.7) | 23.1(20.2, 27.7) | 0.182 |
Data are mean (SD), n (%), or median (IQR). Student’s t test or Mann–Whitney rank-sum test and Chi-square or Fisher exact test, were used as appropriate
SOFA Sequential Organ Failure Assessment, SAPS II Simplified Acute Physiology Score II, PCP Pneumocystis pneumonia, NIV non-invasive ventilation, ECMO extracorporeal membrane oxygenation, PBW predicted body weight, FiO2 the fraction of inspired oxygen, PaO2 partial pressure of arterial oxygen, PaCO2 partial pressure of arterial carbon dioxide, PEEP positive end-expiratory pressure
aWe excluded 6 patients in prone positioning cohort who received ECMO therapy
Table 2.
Laboratory tests and radiologic characteristics of patients in the study
| Supine cohort (n = 16) | Prone cohort (n = 63) | P value | |
|---|---|---|---|
| Laboratory test | |||
| CD4 count | 83.4 (42.6) | 94.5 (58.1) | 0.395 |
| Albumin, Q2 (Q1, Q3) | 28.1 (23.8, 32.5) | 27.6 (27.6, 27.6) | 0.855 |
| LDH | 757.8 (498.1) | 788.1 (475.0) | 0.828 |
| WBC, Q2 (Q1, Q3) | 8.5 (6.5, 13.3) | 8.2 (4.2, 12.3) | 0.774 |
| Procalcitonin | 17.1 (25.9) | 5.3 (9.6) | 0.093 |
| CRP, Q2 (Q1, Q3) | 133.8 (114.8) | 166.8 (106.5) | 0.309 |
| Radiologic characteristics, n (%) | |||
| Ground glass opacity | 9 (56.3) | 27 (42.9) | 0.405 |
| Lung nodules | 5 (31.2) | 12 (19.0) | 0.316 |
| Interstitial markings | 8 (50.0) | 39 (61.9) | 0.561 |
| Consolidation | 9 (56.3) | 30 (47.6) | 0.736 |
| Focal | 1 | 4 | |
| Non-focal | 8 | 26 | |
LDH lactate dehydrogenase, WBC white blood cell, CRP C-reactive protein
Outcomes
Table 3 shows the outcomes of the patients in the two cohorts. There was no significant difference between the supine cohort and prone cohort in mortality on day 28 (68.8% vs 61.9%, P = 0.26) or day 90 (68.8% vs 66.7%, P = 0.55). Figure 2 shows the Kaplan–Meier plot of both groups on day 90.
Table 3.
Primary and secondary outcomes of patients in the study
| Outcome | Supine cohort (n = 16) | Prone cohort (n = 63) | P value |
|---|---|---|---|
| Mortality at d 28 | 11 (68.8) | 39 (61.9) | 0.260 |
| Mortality at d 90 | 11 (68.8) | 42 (66.7) | 0.550 |
| Successful extubation at day 28 | 5 (31.3) | 11 (17.5) | 0.295 |
| Length of IMV days at day 28, d | 8.6 (3.7) | 16.0 (8.7) | 0.002 |
| Ventilator-free days at day28 | 18.0 (16, 18.5) | 0 (0, 10.5) | 0.001 |
| Length of ICU stay, assessed at day 90-days, Q2 (Q1, Q3) | |||
| Survivors | 12.5 (9.0, 12.0) | 8.1 (5.4, 11.8) | 0.173 |
| Non-survivors | 19.0 (11.6, 39.0) | 11.7 (6.5, 20.5) | 0.057 |
| Pneumothorax | 0 | 11 (17.5%) | 0.108 |
| Tracheotomy of survivor at day 90 | 0 | 11 (17.5%) | 0.108 |
IMV invasive mechanical ventilation
Fig. 2.
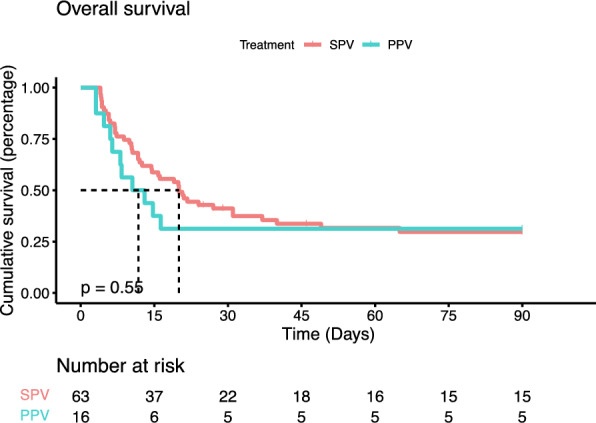
Kaplan–Meier plot of the probability of survival at day 90. PPV prone positioning ventilation cohort, SPV supine positioning ventilation cohort
Patients in the supine cohort had fewer invasive mechanical ventilation days and more ventilator-free days than those in the prone cohort at 28 days. The rate of successful extubation, ICU stay days, incidence of pneumothorax and rate of tracheotomy did not differ significantly between the two groups.
Six patients received ECMO (all in the prone cohort) with 100% mortality on day 90.
Prone positioning ventilation
The PaO2/FiO2 ratio of patients in both cohorts was higher at day 2 and day 3 than at day 1 (Fig. 3), but when comparing ΔPaO2/FiO2, there was no significant difference between the two cohorts on day 2 or day 3 (Table 4).
Fig. 3.

PaO2/FiO2 ratio of patients in both cohort in the first 3 days. PPV prone positioning ventilation, SPV supine positioning ventilation. *P < 0.05 versus D1; NS, no significant difference
Table 4.
Changes of PaO2/FiO2 ratio of patients in both cohorts in the first 3 days
| Time | Prone cohort (n = 38) | Supine cohort (n = 16) | P value of change between cohorts | ||
|---|---|---|---|---|---|
| PaO2/FiO2, mmHg | Change from Day 1 (95% CI) | PaO2/FiO2, mmHg | Change from Day 1(95% CI) | ||
| Day 1 | 102.9 (31.9) | … | 101.3 (26.3) | … | … |
| Day 2 | 127.8 (49.7) | 24.9 (7.7, 42.0) | 166.9 (78.0)* | 65.6 (27.1, 104.0) | 0.053 |
| Day 3 | 136.7 (66.2) | 33.8 (10.8, 56.7) | 140.2 (58.4) | 38.9 (10.9, 66.8) | 0.770 |
For patients in the prone cohort, we only included those who received 3 consecutive sessions of PPV
*P < 0.05 versus the prone cohort
For the first PPV, sessions were terminated earlier for 6 patients (1 for arrhythmia, 3 for low SpO2 and 2 for shock), and 6 were excluded due to receiving ECMO treatment. Of the 51 eligible patient sessions, the PaO2/FiO2 ratio was 105.3 mmHg (SD 28.5) in the SPV1 phase and 115.5 mmHg (IQR 91.5–147.7) in the PPV phase, and there was no significant difference between the SPV1 phase and PPV phase (P = 0.1173). The per-time trajectory of PaO2/FiO2 at the three study time phases is shown in Fig. 4.
Fig. 4.

PaO2/FiO2 trajectory in the first PPV sessions. SPV1, 0–2 h before prone positioning ventilation; PPV, 0–2 h after shifting to prone positioning ventilation; SPV2, 1–2 h after resupervised positioning. Type A, PaO2/FiO2 ratio increased by over 15%; type B, PaO2/FiO2 ratio changed between − 15% and + 15%; type C, PaO2/FiO2 ratio decreased by over 15%. * P < 0.05 versus SPV1
The complications of the first 3 PPV sessions are listed in Table 5. In the first PPV session, SpO2 decreased by ≥ 4% in 29 (46%) patients, which was significantly higher than that of patients in the SPV cohort on day 1 (P = 0.0033).
Table 5.
Adverse events of both cohort in the first 3 sessions of prone positioning or first 3 days
| S1/D1 | S2/D2 | S3/D3 | ||||
|---|---|---|---|---|---|---|
| PPV cohort (n = 63) | SPV cohort (n = 16) | PPV cohort (n = 49) | SPV cohort (n = 16) | PPV cohort (n = 44) | SPV cohort (n = 16) | |
| Arrhythmia, n (%) | 0 | 1 (6.3) | 3 (6.1) | 1 (6.3) | 2 (4.5) | 1 (6.3) |
| Shock, n (%) | 6 (9.5) | 2 (12.5) | 2 (4.1) | 1 (6.3) | 2 (4.5) | 0 |
| SpO2 < 85% for more than 2 min, n (%) | 8 (12.7) | 2 (12.5) | 4 (8.2) | 2 (12.5) | 2 (4.5) | 1 (6.3) |
| SpO2 decrease by ≥ 4%, n (%) | 29 (46.0)* | 1 (6.3) | 13 (26.5) | 2 (12.5) | 4 (9.1) | 0 |
| Airway obstruction, n (%) | 0 | 0 | 0 | 0 | 0 | 0 |
| Unexpected extubation, n (%) | 0 | 0 | 0 | 0 | 0 | 0 |
| Early termination, n (%) | 6 (9.5) | … | 2 (4.1) | … | 1 (2.3) | … |
*P < 0.05 versus the supine cohort
Factors associated with 28-day mortality
Figure 5 shows the results of the Cox proportional hazards regression for factors associated with ICU mortality at 28 days. High levels of blood urea nitrogen (HR, 1.04; 95% CI 1.00–1.08; P = 0.043) and lactate (HR, 2.23; 95% CI 1.59–3.15; P < 0.001) and smoking (HR, 1.77; 95% CI 1.07–2.94; P = 0.027) were associated with higher 28-day mortality, and female sex (HR, 0.27; 95% CI 0.12–0.63; P = 0.002) was associated with lower 28-day mortality. PPV was not associated with the risk of ICU 28-day mortality (HR, 0.55; 95% CI 0.27 to 1.14, P = 0.109). Univariate analysis results of Cox proportional hazards regression was appended as supplementary file 1.
Fig. 5.
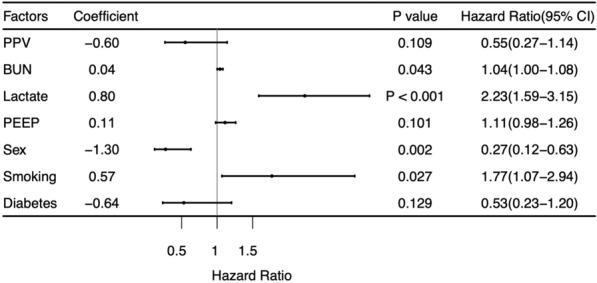
Effect of demographic and disease features on mortality. PPV prone positioning ventilation, BUN blood urea nitrogen, PEEP positive end-expiratory pressure
Discussion
In this observational, retrospective, cohort study, we found that PPV did not improve oxygenation or reduce the mortality of patients with PCP and moderate–severe ARDS. Furthermore, PPV was associated with more invasive mechanical ventilation days and fewer ventilator-free days at 28 days.
To our knowledge, this is the first study examining the effect of PPV on patients with PCP. Our findings are consistent with previous studies showing that patients with ARDS, a heterogeneous syndrome, may show distinct physiological and clinical responses to the same treatment [19–21]. Lung morphology is a promising approach by which ventilation protocols for patients with ARDS can be formulated, and PPV may not be appropriate for every patient as therapies, such as high PEEP and recruitment maneuvers.
Our findings regarding oxygenation evolution during PPV can be explained as follows. First, cases and trials have consistently reported that patients with PCP show GGOs, increased interstitial markings in early stages and patchy consolidation in mid or late stages [7–10], demonstrating a more even distribution of ventilation along the anteroposterior axis than patients with non-PCP ARDS. Previous studies have demonstrated that lung perfusion distribution is similar in the prone and supine positions [22, 23]. Consequently, ventilation of the dorsal lung may change minimally during PPV, eventually resulting in an approximate ventilation/perfusion ratio in the supine position. This finding agrees with a previous study showing that higher PEEP and recruitment maneuvers might be more suitable for patients with nonfocal ARDS, whereas patients with focal ARDS might benefit more from PPV and lower PEEP [24–27].
Second, 58 (73%) patients in our study had coexisting microbes in the lung, leading to nonuniform changes in ventilation in the dorsal lung and ventilation/perfusion ratio that varied across patients resulting in different responses to prone positioning. Third, the LIVE study showed that ventilated patients with nonfocal ARDS in the personalized group that had a high PEEP reached a Pplat of 30 cm H2O and recruitment maneuvers [26]. However, in our study, patients were ventilated according to the ALVEOLI study, and recruitment maneuvers were not performed routinely [17, 28, 29], and the collapsed alveolar group may not have been fully recruited compared to nonfocal ARDS in the personalized group of the LIVE study. Thus, in the first PPV session, which was conducted in the first 24 h after ICU admission, ventilation may redistribute and result in considerable decreases in SpO2.
Furthermore, most patients in our study received NIV before ICU admission but failed, and many patients showed consolidation and were in the late stage of PCP [3, 10]. The mortality remained high despite standard pharmacotherapy, lung-protective ventilation and even with ECMO.
Our study has several limitations. First, the retrospective nature of the study, together with the absence of Pplat measures at each time phase, may have affected the outcomes observed in this study. Second, lung perfusion and ventilation of PCP may not have been assessed accurately, and ventilation/perfusion matching requires further monitoring in the supine position and prone position. Future studies with a prospective, randomized controlled trial design and with larger sample sizes are therefore needed.
In summary, we found that prone positioning did not improve oxygenation or the survival rate of intubated patients with PCP and moderate–severe ARDS. PPV results in more invasive mechanical ventilation days and is associated with a high incidence of adverse events. Our findings do not support the routine use of PPV in these patients (Additional file 1).
Supplementary Information
Additional file 1. Univariate analysis results of Cox proportional hazards regression.
Acknowledgements
We thank Huajing Wan for her advice on the study protocol. We thank the R Core Team, Hadley Wickham, Terry M Therneau, Douglas Bates and their coworkers for their generous sharing of R packages.
Author contributions
Zhen Wang conceived the original idea and designed the study. Min Zhu, He Yu and Fengming Luo revised the study protocol. Zhen Wang recruited patients, and Yuyan Zhou, Yubei Zhou and Faping Wang collected data. Zhen Wang and Yuyan Zhou performed the statistical analysis. Zhen Wang wrote the manuscript and created the figures and tables. Min Zhu and Fengming Luo edited the manuscript. All authors analyzed and discussed the results. All the authors have read and approved the final manuscript.
Funding
This research was supported by the National Key Research and Development Program of China (2021YFF0702000), and grants from the National Natural Science Foundation of China (No. 32070764 (F.L.), Sichuan Province Science, and the Technology Support Program (No 2021YFQ0030).
Availability of data and materials
The datasets used for the current study are available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
This study received approval from the Ethics Committee of the West China Hospital of Sichuan University (REC2022-824). The Ethics Committee of the West China Hospital of Sichuan University waived the requirement for ethical committee approval for this study (Chair: Shaolin Deng, Ref: 2022-824). The study was performed according to the Declaration of Helsinki. The study is reported according to STROBE guidelines [30].
Consent for publication
Not needed.
Competing interests
None of the authors have any conflicts of interest to declare.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Weyant RB, Kabbani D, Doucette K, Lau C, Cervera C. Pneumocystis jirovecii: a review with a focus on prevention and treatment. Expert Opin Pharmacother. 2021;22(12):1579–1592. doi: 10.1080/14656566.2021.1915989. [DOI] [PubMed] [Google Scholar]
- 2.Salzer HJF, Schäfer G, Hoenigl M, Günther G, Hoffmann C, Kalsdorf B, Alanio A, Lange C. Clinical, diagnostic, and treatment disparities between HIV-infected and non-HIV-infected immunocompromised patients with Pneumocystis jirovecii pneumonia. Respiration. 2018;96(1):52–65. doi: 10.1159/000487713. [DOI] [PubMed] [Google Scholar]
- 3.White PL, Backx M, Barnes RA. Diagnosis and management of Pneumocystis jirovecii infection. Expert Rev Anti Infect Ther. 2017;15(5):435–447. doi: 10.1080/14787210.2017.1305887. [DOI] [PubMed] [Google Scholar]
- 4.Kovacs JA, Masur H. Evolving health effects of Pneumocystis: one hundred years of progress in diagnosis and treatment. JAMA. 2009;301(24):2578–2585. doi: 10.1001/jama.2009.880. [DOI] [PubMed] [Google Scholar]
- 5.Meyer NJ, Gattinoni L, Calfee CS. Acute respiratory distress syndrome. Lancet. 2021;398(10300):622–637. doi: 10.1016/S0140-6736(21)00439-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Roux A, Canet E, Valade S, et al. Pneumocystis jirovecii pneumonia in patients with or without AIDS. France Emerg Infect Dis. 2014;20(9):1490–1497. doi: 10.3201/eid2009.131668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kumagai S, Arita M, Koyama T, Kumazawa T, Inoue D, Nakagawa A, Kaji Y, Furuta K, Fukui M, Tomii K, Taguchi Y, Tomioka H, Ishida T. Prognostic significance of crazy paving ground grass opacities in non-HIV Pneumocystis jirovecii pneumonia: an observational cohort study. BMC Pulm Med. 2019;19(1):47. doi: 10.1186/s12890-019-0813-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hsu JM, Hass A, Gingras MA, Chong J, Costiniuk C, Ezer N, Fraser RS, McDonald EG, Lee TC. Radiographic features in investigated for Pneumocystis jirovecii pneumonia: a nested case-control study. BMC Infect Dis. 2020;20(1):492. doi: 10.1186/s12879-020-05217-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hardak E, Brook O, Yigla M. Radiological features of Pneumocystis jirovecii Pneumonia in immunocompromised patients with and without AIDS. Lung. 2010;188(2):159–163. doi: 10.1007/s00408-009-9214-y. [DOI] [PubMed] [Google Scholar]
- 10.Mu XD, Jia P, Gao L, Su L, Zhang C, Wang RG, Wang GF. Relationship between radiological stages and prognoses of Pneumocystis pneumonia in non-AIDS immunocompromised patients. Chin Med J (Engl) 2016;129(17):2020–2025. doi: 10.4103/0366-6999.189068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fan E, Del Sorbo L, Goligher EC, American Thoracic Society, European Society of Intensive Care Medicine, and Society of Critical Care Medicine et al. An Official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: mechanical ventilation in adult patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2017;195(9):1253–1263. doi: 10.1164/rccm.201703-0548ST. [DOI] [PubMed] [Google Scholar]
- 12.Galiatsou E, Kostanti E, Svarna E, Kitsakos A, Koulouras V, Efremidis SC, Nakos G. Prone position augments recruitment and prevents alveolar overinflation in acute lung injury. Am J Respir Crit Care Med. 2006;174(2):187–197. doi: 10.1164/rccm.200506-899OC. [DOI] [PubMed] [Google Scholar]
- 13.Guerin C, Baboi L, Richard JC. Mechanisms of the effects of prone positioning in acute respiratory distress syndrome. Intensive Care Med. 2014;40(11):1634–1642. doi: 10.1007/s00134-014-3500-8. [DOI] [PubMed] [Google Scholar]
- 14.Wang YX, Zhong M, Dong MH, et al. Prone positioning improves ventilation-perfusion matching assessed by electrical impedance tomography in patients with ARDS: a prospective physiological study. Crit Care. 2022;26(1):154. doi: 10.1186/s13054-022-04021-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guérin C, Reignier J, Richard JC, PROSEVA Study Group et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368(23):2159–2168. doi: 10.1056/NEJMoa1214103. [DOI] [PubMed] [Google Scholar]
- 16.Griffiths MJD, McAuley DF, Perkins GD, Barrett N, Blackwood B, Boyle A, et al. Guidelines on the management of acute respiratory distress syndrome. BMJ Open Respir Res. 2019;6(1):e000420. doi: 10.1136/bmjresp-2019-000420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Papazian L, Aubron C, Brochard L, Chiche JD, Combes A, Dreyfuss D, Forel JM, Guérin C, Jaber S, Mekontso-Dessap A, Mercat A, Richard JC, Roux D, Vieillard-Baron A, Faure H. Formal guidelines: management of acute respiratory distress syndrome. Ann Intensive Care. 2019;9(1):69. doi: 10.1186/s13613-019-0540-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Papazian L, Forel JM, Gacouin A, ACURASYS Study Investigators et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363(12):1107–1116. doi: 10.1056/NEJMoa1005372. [DOI] [PubMed] [Google Scholar]
- 19.Calfee CS, Janz DR, Bernard GR, May AK, Kangelaris KN, Matthay MA, Ware LB. Distinct molecular phenotypes of direct vs indirect ARDS in single-center and multicenter studies. Chest. 2015;147(6):1539–1548. doi: 10.1378/chest.14-2454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Calfee CS, Delucchi KL, Sinha P, Matthay MA, Hackett J, Shankar-Hari M, McDowell C, Laffey JG, O'Kane CM, McAuley DF, Irish Critical Care Trials Group Acute respiratory distress syndrome subphenotypes and differential response to simvastatin: secondary analysis of a randomised controlled trial. Lancet Respir Med. 2018;6(9):691–698. doi: 10.1016/S2213-2600(18)30177-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jabaudon M, Blondonnet R, Lutz J, Roszyk L, Bouvier D, Guérin R, Perbet S, Cayot S, Godet T, Blanchon L, Bazin JE, Futier E, Sapin V, Constantin JM. Net alveolar fluid clearance is associated with lung morphology phenotypes in acute respiratory distress syndrome. Anaesth Crit Care Pain Med. 2016;35(2):81–86. doi: 10.1016/j.accpm.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 22.Musch G, Layfield JD, Harris RS, Melo MF, Winkler T, Callahan RJ, Fischman AJ, Venegas JG. Topographical distribution of pulmonary perfusion and ventilation, assessed by PET in supine and prone humans. J Appl Physiol (1985) 2002;93(5):1841–1851. doi: 10.1152/japplphysiol.00223.2002. [DOI] [PubMed] [Google Scholar]
- 23.Scholten EL, Beitler JR, Prisk GK, Malhotra A. Treatment of ARDS with prone positioning. Chest. 2017;151(1):215–224. doi: 10.1016/j.chest.2016.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Puybasset L, Cluzel P, Gusman P, Grenier P, Preteux F, Rouby JJ. Regional distribution of gas and tissue in acute respiratory distress syndrome. I. Consequences for lung morphology. CT Scan ARDS Study Group. Intensive Care Med. 2000;26(7):857–869. doi: 10.1007/s001340051274. [DOI] [PubMed] [Google Scholar]
- 25.Gattinoni L, Caironi P, Cressoni M, Chiumello D, Ranieri VM, Quintel M, Russo S, Patroniti N, Cornejo R, Bugedo G. Lung recruitment in patients with the acute respiratory distress syndrome. N Engl J Med. 2006;354(17):1775–1786. doi: 10.1056/NEJMoa052052. [DOI] [PubMed] [Google Scholar]
- 26.Constantin JM, Jabaudon M, Lefrant JY, AZUREA Network et al. Personalised mechanical ventilation tailored to lung morphology versus low positive end-expiratory pressure for patients with acute respiratory distress syndrome in France (the LIVE study): a multicentre, single-blind, randomised controlled trial. Lancet Respir Med. 2019;7(10):870–880. doi: 10.1016/S2213-2600(19)30138-9. [DOI] [PubMed] [Google Scholar]
- 27.Coppola S, Pozzi T, Gurgitano M, Liguori A, Duka E, Bichi F, Ciabattoni A, Chiumello D. Radiological pattern in ARDS patients: partitioned respiratory mechanics, gas exchange and lung recruitability. Ann Intensive Care. 2021;11(1):78. doi: 10.1186/s13613-021-00870-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cavalcanti AB, Suzumura ÉA, Laranjeira LN, Writing Group for the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART) Investigators et al. Effect of lung recruitment and titrated positive end-expiratory pressure (PEEP) vs low PEEP on mortality in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2017;318(14):1335–1345. doi: 10.1001/jama.2017.14171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Grasselli G, Calfee CS, Camporota L, European Society of Intensive Care Medicine Taskforce on ARDS et al. ESICM guidelines on acute respiratory distress syndrome: definition, phenotyping and respiratory support strategies. Intensive Care Med. 2023;49(7):727–759. doi: 10.1007/s00134-023-07050-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370:1453–1457. doi: 10.1016/S0140-6736(07)61602-X. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Univariate analysis results of Cox proportional hazards regression.
Data Availability Statement
The datasets used for the current study are available from the corresponding author upon reasonable request.



