Abstract
Introduction
Oral chlorhexidine has been widely used for ventilator-associated pneumonia prevention in the critical care setting; however, previous studies and evidence synthesis have generated inconsistent findings. Our study aims to investigate if different concentrations of oral chlorhexidine may be effective in preventing such complication in intensive care unit patients.
Methods
After pre-registration (Open Science Framework: 8CUKF), we conducted a network meta-analysis with the following PICOS: adult patients (age > 18 years old) undergoing invasive mechanical ventilation admitted in ICU (P); any concentration of chlorhexidine used for oral hygiene (I); placebo, sham intervention, usual care, or no intervention (C); rate of VAP (primary outcome), mechanical ventilation length, ICU length of stay (LOS), hospital LOS, mortality (secondary outcomes) (O); randomized controlled trials (S). We used the following database: PubMed, the Cochrane Central Register of Controlled Trials (CENTRAL), Scopus, and EMBASE without any limitation in publication date or language.
Results
Chlorhexidine did not demonstrate any significant advantage over the control group in preventing ventilator-associated pneumonia or reducing mortality, duration of mechanical ventilation, length of stay in the intensive care unit, or overall mortality.
Conclusions
Chlorhexidine oral decontamination does not reduce the rate of ventilator-associated pneumonia in critically ill adult patients and its routine use could not be recommended.
Trial registration
Registration number: Open Science Framework: 8CUKF.
Supplementary Information
The online version contains supplementary material available at 10.1186/s44158-024-00166-2.
Keywords: Critical care, Chlorhexidine, Meta-analysis, Ventilator-associated pneumonia, Mechanical ventilation
Introduction
Hospital-acquired infections contribute to prolonging hospital stays, increasing patients’ morbidity and mortality, and inflating hospitalization costs [1, 2]. Patients admitted to intensive care units (ICUs) face an increased risk of acquiring such an infection that in some studies has been estimated to be around 30% [3].
Lower respiratory system is the most common site of infection in ICU patients [4].
Ventilator-associated pneumonia (VAP) is a distinct form of pneumonia occurring in patients undergoing invasive mechanical ventilation. Micro-organisms access the respiratory system through entry points, such as the endotracheal tube, or via leakage of secretions around the endotracheal cuff [5]. Numerous factors contribute to the development of VAP in critically ill patients, e.g., the aspiration of gastrointestinal microbes, compromised cough reflex, the inability to effectively clear secretions through the pharynx and mouth, and inadequate oral care [5].
The occurrence of VAP is associated with a mortality risk ranging from 1 to 10% [6]. One of the proposed strategies for VAP prophylaxis is the use of oral chlorhexidine washes to prevent the growth and aspiration of bacteria. Being simple and low-cost, the vast majority of ICUs have adopted daily oral care with chlorhexidine in their patients [7]. Despite the robust rationale, the assessment of oral antiseptics use as a preventive strategy for VAP has generated inconsistent findings in prior studies [8–10]. Moreover, a meta-analysis, overall including 16 randomized controlled trials and 3630 patients, did not support the use of chlorhexidine for the prevention of VAP in non-cardiac surgery patients [11]. However, this work did not consider the effect of the different concentrations of oral chlorhexidine employed in the included studies.
The objective of this network meta-analysis is to assess whether different concentrations of oral chlorhexidine may be effective in preventing VAP in ICU patients. Secondary outcomes were duration of invasive mechanical ventilation, ICU length of stay, and hospital length of stay and mortality.
Methods
The protocol for this network meta-analysis has been prospectively registered on Open Science Framework [12], and the Preferred Reporting Items for Systematic reviews and Meta-Analysis (PRISMA) statement guidelines was followed for the reporting of the present manuscript [13].
Eligibility criteria
Studies were considered to be eligible for inclusion using the following PICOS criteria: adult patients (age > 18 years old) undergoing invasive mechanical ventilation admitted in ICU (P); any concentration of chlorhexidine used for oral hygiene (I); placebo, sham intervention, usual care, or no intervention (C); rate of VAP (primary outcome), mechanical ventilation length, ICU length of stay (LOS), hospital LOS, mortality (secondary outcomes) (O); randomized controlled trials (S).
Search strategy
We performed a systematic search of the medical literature for the identification, screening, and inclusion of articles. We did not apply any restriction related to language or year of publication. We queried the following database from inception to May 17, 2023: PubMed, The Cochrane Central Register of Controlled Trials (CENTRAL), Scopus, and EMBASE.
Study selection
Three researchers (CL, AS, CP) independently screened titles and abstracts of the identified papers in order to select relevant manuscripts. Each citation was reviewed in full-text form if considered potentially relevant. All the references of the included literature were examined to retrieve further relevant studies. The search strategy for each database is available as supplementary material. After identifying those studies meeting inclusion criteria, two authors (VB, AB) manually reviewed and assessed each of the included studies.
Quality assessment and certainty of evidence assessment
Risk of bias was assessed independently by two members of the team not previously involved in the study selection phase (ADC, TP). The assessment was performed using the Risk of Bias (RoB) 2 Tool, expressing the overall risk of bias on a three-grade scale (“low risk of bias,” “high risk of bias,” or “some concerns”) [14]. In case of disagreements after discussion among assessors, a third researcher (PN) was consulted.
Statistical methods
Meta-analysis of data was performed using R version 4.1 (R Foundation for Statistical Computing, Vienna, Austria) and the package “netmeta.” The treatment effect for continuous outcomes was measured using mean difference (MD) with 95% confidence interval (CI). For dichotomous outcomes, we expressed the treatment effect as odds ratio (OR) with 95% CI. Availability of evidence, transitivity assumption, intra-network connectivity, and network coherence were considered to assess the feasibility of conducting a network meta-analysis [15]. To rank comparators, we conducted a ranking analysis using the frequentist analogue of the surface under the cumulative ranking curve (SUCRA) [16]. In case of data expressed as median and quartiles, we utilized Hozo’s method [17] to estimate the mean and standard deviation (SD). Additionally, we abstained from applying continuity correction to cases with zero events.
Sensitivity analysis
We decided to perform the following post hoc sensitivity analysis for the primary outcome: (a) excluding postoperative patients, (b) excluding high risk-of-bias studies, (c) excluding all comparators other than placebo/no intervention.
Heterogeneity and publication bias analysis
For assessment of study inconsistency and heterogeneity, the I2 and Tau2 statistics were used. Values of I2 were categorized as follows: low heterogeneity: I2 < 25%, moderate heterogeneity: I2 25 to 50%, or high heterogeneity: I2 > 50%) [18]. A random-effect model was preferred, regardless of heterogeneity. Publication bias was evaluated both by visual inspection of funnel plots or Egger’s test when more than ten studies were available for a specific outcome.
Results
Study characteristics
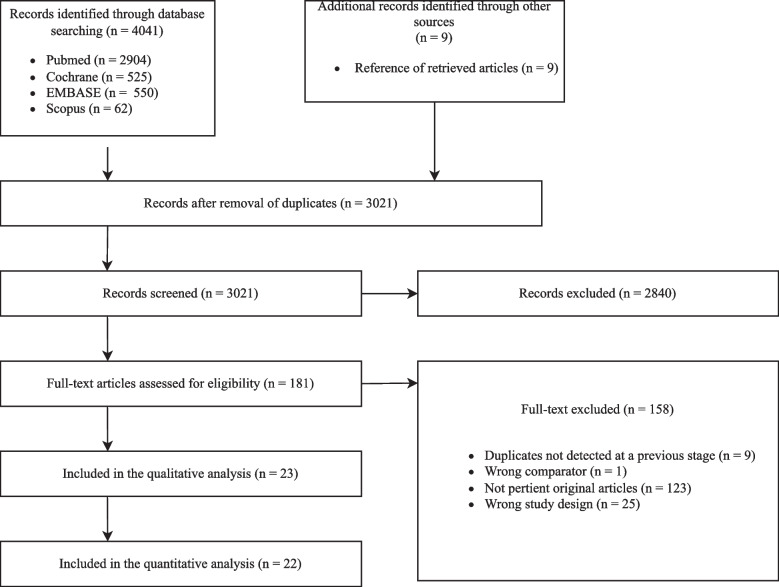
PRISMA flowchart of the included studies is depicted in Fig. 1. We selected 181 articles for full-text assessment. Of these, only 23 articles met our inclusion criteria [8–10, 19–38]. However, one study was excluded, as it presented a mixed intubated/not intubated cohort of patients, and the corresponding author was unable to provide us with the subset of intubated patients only [38]. Therefore, 22 articles (5314 patients) were eventually included for qualitative and quantitative analysis.
Fig. 1.
PRISMA flowchart
Characteristics of the included studies are reported in Table 1. The identified chlorhexidine concentrations were as follows: 0.12% (nine studies) [8, 20, 22, 24–28, 33], 0.2% (eight studies) [9, 10, 21, 23, 29, 30, 36, 37], 2% (six studies) [19, 30–32, 34, 35]. Among these studies, only one performed a direct comparison among chlorhexidine concentrations, i.e., 0.2% vs 2.0% [30].
Table 1.
Study characteristics
| Study | Country | Population | Main outcome | Group 1 | Group 2 | Nt | VAP criteria |
|---|---|---|---|---|---|---|---|
| Meinberg (2012) [19] | Brazil | ICU | VAP | Chlorhexidine 2% | Placebo | 4 | CDC |
| Scannapieco (2009) [20] | USA | ICU | Oral bacterial colonization | Chlorhexidine 0.12% | Placebo | 2 | CPIS or PC |
| Ozczka (2012) [21] | Turkey | ICU | VAP | Chlorhexidine 2% | Placebo | 4 | PC |
| De Riso (1996) [22] | USA | Postsurgical | Nosocomial infections | Chlorhexidine 0.12% | Placebo | 2 | CDC |
| Fourrier (2005) [9] | France | ICU | Nosocomial infections | Chlorhexidine 0.2% | Placebo | ≥ 3 | CDC |
| Segers (2006) [8] | The Netherlands | Postsurgical | Nosocomial infections | Chlorhexidine 0.12% | Placebo | 4 | CDC |
| Panchabhai (2009) [23] | India | ICU | VAP | Chlorhexidine 0.2% | Sham (potassium permanganate) | 2 | CDC |
| Bellissimo-Rodrigues (2009)[24] | Brazil | ICU | Nosocomial respiratory infections | Chlorhexidine 0.12% | Placebo | 3 | CDC |
| Dale (2021) [25] | Canada | ICU | ICU Mortality | Chlorhexidine 0.12% | Usual care | 3 | CDC |
| Dale (2009) [26] | USA | ICU | VAP | Chlorhexidine 0.12% | Usual care | 3 | CPIS |
| Zarinfar (2021) [27] | Iran | ICU | VAP | Chlorhexidine 0.12% | Usual care | 2 | CPIS and PC |
| Jo Grap (2011) [28] | USA | Trauma | VAP | Chlorhexidine 0.12% | Usual care | 1 | CPIS |
| Jahanshir (2022) [29] | Iran | ICU | VAP | Chlorhexidine 0.2% | Sham (clove extract) | 2 | mCPIS |
| Zand (2017) [30] | Iran | ICU | VAP | Chlorhexidine 0.2% | Chlorhexidine | 1 | CPIS |
| Lin (2015) [31] | China | Postsurgical | VAP | Chlorhexidine 0.2% | Normal saline | 3 | CPIS |
| Tantipong (2008) [10] | Thailand | ICU | VAP | Chlorhexidine 2.0% | Normal saline | 4 | CDC |
| Tuon (2016) [32] | Brazil | ICU | VAP | Chlorhexidine 2.0% | Normal saline | 2 | CDC |
| Pobo (2009) [33] | Spain | ICU | VAP | Chlorhexidine 0.12% | Usual care | 3 | CDC |
| Meidani (2018) [34] | Iran | ICU | VAP | Chlorhexidine 0.2% | Placebo | 2 | CDC |
| Koeman (2006) [35] | The Netherlands | ICU | VAP | Chlorhexidine 2% | Placebo | 4 | Clinical decision |
| Fourrier (2000) [36] | France | ICU | Oral bacterial colonization | Chlorhexidine 0.2% | Sham (bicarbonate) | 3 | CDC |
| Berry (2009) [37] | Australia | ICU | Oral bacterial colonization | Chlorhexidine 0.2% | Sham (sterile water) | 2 | Clinical decision |
ICU intensive care unit, CDC Center for Disease Control and Prevention, CPIS clinical pulmonary infection score, PC positive culture, Nt number of times the treatment was applied
Risk of bias assessment
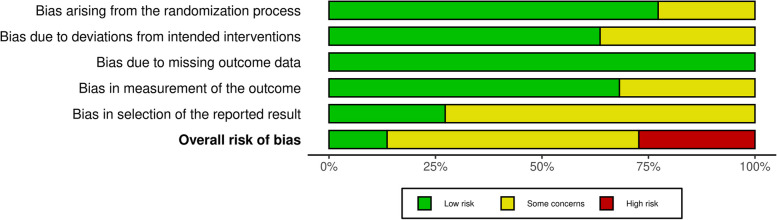
The overall risk of bias assessment is summarized in Fig. 2 and detailed in Additional file 2. Six studies were evaluated to be at high risk of bias, thirteen studies to some concern, while the remaining studies were evaluated to be at low risk of bias.
Fig. 2.
Overall risk of bias assessment
Outcomes
Results for all the outcomes are summarized in Table 2, and the SUCRA analysis is shown in Table 3. Publication bias was detected for no outcomes (Additional file 3).
Table 2.
Study outcomes
| Group | VAP (OR, 95% CI) | p-value | MV length (MD, 95% CI) | p-value | ICU LOS (MD, 95% CI) | p-value | Hospital LOS (MD, 95% CI) | p-value | Mortality (OR, 95% CI) | p-value |
|---|---|---|---|---|---|---|---|---|---|---|
| 0.12% | 0.70 (0.41;1.19) | 0.195 | − 0.25 (− 1.55; 1.04) | 0.699 | 0.46 (− 1.25; 2.17) | 0.597 | − 0.60 (− 0.72; − 0.47) | < 0.001* | 1.02 (0.73–1.42) | 0.896 |
| 0.2% | 0.93 (0.52;1.65) | 0.806 | 0.63 (− 1.02; 2.29) | 0.453 | − 1.51 (− 3.36; 0.34) | 0.110 | − 1.64 (− 5.08; 1.79) | 0.349 | 1.16 (0.78; 1.72) | 0.439 |
| 2.0% | 0.56 (0.28;1.13) | 0.104 | 0.22 (− 1.88; 2.32) | 0.836 | 0.11 (− 2.67; 2.90) | 0.935 | − 1.53 (− 7.25; 4.19) | 0.599 | 0.64 (0.31; 1.34) | 0.241 |
Placebo, sham intervention, usual care, or no intervention are used as reference group
CI confidence intervals, ICU intensive care unit, LOS length of stay, MD mean difference, MV mechanical ventilation, OR odds ratio, VAP ventilator-associated pneumonia
*Statistically significant
Table 3.
Surface under the cumulative ranking curve analysis
| Group | VAP | Mechanical ventilation length | ICU LOS | Hospital LOS | Mortality |
|---|---|---|---|---|---|
| Reference | 0.184 | 0.568 | 0.429 | 0.158 | 0.484 |
| 0.12% | 0.320 | 0.698 | 0.260 | 0.550 | 0.424 |
| 0.2% | 0.654 | 0.270 | 0.903 | 0.687 | 0.196 |
| 2.0% | 0.842 | 0.463 | 0.406 | 0.603 | 0.895 |
Placebo, sham intervention, usual care, or no intervention are used as the reference group
ICU intensive care unit, LOS length of stay, VAP ventilator-associated pneumonia
VAP
Twenty-one studies evaluated VAP in 6626 patients overall (367 patients assigned to the 2.0% concentration, 464 to the 0.2%, 2471 to the 0.12%, and 3324 assigned to the control group). None of the concentrations of chlorhexidine was associated with a statistically significant reduction in VAP, when compared to either non-chlorhexidine comparators or other chlorhexidine concentrations (Table 1). Heterogeneity was high (I2 66.8%, Tau2 0.39). The estimation of direct and indirect evidence is available as supplementary material (Additional file 4), while the graph describing the network among intervention is presented as Additional file 5. There was no evidence of publication bias at the funnel plot (Egger test p-value 0.635).
Mechanical ventilation duration
Twelve studies evaluated the mechanical ventilation duration and randomized 5379 patients (255 to the 2.0% concentration, 188 to the 0.2%, 2191 to the 0.12%, while 2745 were assigned to the control group). The utilization of chlorhexidine at any concentration was not found to reduce mechanical ventilation duration, in comparison with the control group. Heterogeneity was high (I2 56%; Tau2 1.23). No publication was detected at funnel plot (p-value 0.456).
ICU LOS
Ten studies reported results for ICU LOS, randomizing a total of 2040 patients (255 to the 2.0% concentration, 188 to the 0.2%, 594 to the 0.12%, and 1003 to the control group). We did not observe shorter ICU LOS in patients receiving any concentration of chlorhexidine, when compared to the control group. Heterogeneity was low (I2 21.7%, Tau2 1.02), and there were no signs of publication bias (p-value 0.457).
Hospital LOS
Analysis of seven studies, randomizing a total of 1834 patients (155 to the 2.0% concentration, 70 to the 0.2%, 695 to the 0.12%, while 914 to the control group), found only a statistically significant but clinically unimportant reduced LOS in 0.12% patients (− 0.60; CI − 0.72; − 0.47 days). In this analysis, there was low heterogeneity (I2 0%, Tau2 0) without publication bias at the visual inspection of the funnel plot.
Mortality
Fourteen studies reported data for this outcome, enrolling 5978 patients overall (185 in the 2.0% group, 347 in the 0.2% group, 2447 in the 0.12% group, while 2999 patients in the control group). No significant effect was detected for any of the intervention. Low heterogeneity was reported (I2 21.4%, Tau2 0.045) and no publication bias (p-value 0.490).
Sensitivity analysis
Results for the sensitivity analyses are reported in Table 4. Briefly, none of the subgroup analyses was able to determine the superiority of chlorhexidine at any concentration over controls.
Table 4.
Sensitivity analysis for the primary outcome
| Risk of bias | No postoperative patients | Only placebo/usual care | |||||||
|---|---|---|---|---|---|---|---|---|---|
| N (6138) | OR (95% CI) | p-value | N (5467) | OR (95% CI) | p-value | N (6469) | OR (95% CI) | p-value | |
| Control | 3078 | Ref | Ref | 2751 | Ref | Ref | 3250 | Ref | Ref |
| 0.12% | 2590 | 0.70 (0.44; 1.12) | 0.140 | 1932 | 0.81 (0.41; 1.61) | 0.557 | 2590 | 0.70 (0.44; 1.11) | 0.132 |
| 0.2% | 211 | 1.42 (0.71; 2.79) | 0.317 | 464 | 0.94 (0.50; 1.77) | 0.858 | 262 | 0.97 (0.49; 1.91) | 0.935 |
| 2.0% | 259 | 0.57 (0.27; 1.21) | 0.145 | 320 | 0.64 (0.28; 1.47) | 0.293 | 367 | 0.55 (0.30; 1.05) | 0.069 |
| I2 | 62.6 | 70.5 | 56.5 | ||||||
| Tau2 | 0.27 | 0.52 | 0.25 | ||||||
CI confidence interval, N number of patients, OR odds ratio
Discussion
This systematic review and network meta-analysis including 5314 adult patients from 21 RCTs found that no concentration of chlorhexidine was associated with reduced rate of VAP, in comparison either with other chlorhexidine concentrations or with no-chlorhexidine interventions. Moreover, compared to other chlorhexidine concentrations or no-chlorhexidine interventions, no concentration of chlorhexidine improved mechanical ventilation duration, LOS, and mortality.
Since the finding is that no concentration of chlorhexidine is effective in preventing VAP or any of the other investigated outcomes, our network meta-analysis contributes additional evidence to a previous pairwise meta-analysis, published in 2014 [11], already questioning the impact of chlorhexidine on preventing VAP in non-cardiac surgery patients.
Previous meta-analysis [39, 40] showed that certain mode of chlorhexidine delivery (solution but no gel or rinse) or frequency of use (4 times/die) could have an impact on VAP incidence; however, the paucity of mode of deliveries and frequency of administrations when subcategorized for chlorhexidine solutions prevented us to conduct such subgroups analysis. This remains for sure an interesting point for future research.
Moreover, 0.12% chlorhexidine concentration group is more represented (2590 patients), compared to the 0.2% (464 patients) and 2% (367 patients) groups. Therefore, expanding the sample size for the other groups might reveal significant benefits on the rate of VAP. However, higher chlorhexidine concentrations may increase the risk of oral lesions [40], selecting the growth of germs resistant to chlorhexidine [41, 42].
Over the years, there have been significant changes in guidelines regarding the use of chlorhexidine for preventing VAP in ventilated patients.
When examining the guidelines, it is essential to note that the “Zero-VAP” bundle (Spanish guidelines) [43] suggested the standard use of chlorhexidine to prevent VAP, recommending concentrations as high as 2%. However, not all the scientific societies agreed with such a recommendation. In fact, in the same year (2014), the SHEA/IDSA guidelines [44] categorized oral care with chlorhexidine under special approaches instead of basic practices due to potential risks and unclear benefits.
A more recent European guideline [45] does not provide a formal recommendation on the use of chlorhexidine for oral care in mechanically ventilated patients due to a lack of safety data and an unclear balance between the potential reduction in VAP and the potential increase in mortality. The latest update from SHEA [46] does not recommend the use of oral chlorhexidine as it may increase mortality rates. Our study aligns with the most recent guidelines and further strengthened these recommendations.
Our research has some limitations that warrants discussion. First, intransitivity may have arisen from the inclusion of studies published over a 26-year period. Over this period, there could have been substantial modifications to VAP prevention bundles, antimicrobial therapies, and other clinical practices, potentially impacting the research outcomes. Second, the main analysis on primary outcome showed high heterogeneity that was not explained by our subgroup analyses, reducing the overall confidence in our results. The among-studies heterogeneity in protocols for chlorhexidine oral decontamination and antimicrobial stewardship and outcome definitions may explain such finding. Third, we included all non-chlorhexidine interventions, i.e., placebo, sham intervention, usual care, and no intervention, in the same group.
Conclusion
Chlorhexidine oral decontamination does not reduce the rate of VAP in critically ill adult patients in the ICU, and we could not recommend its routine use. Nevertheless, further research is warranted, particularly investigating the potential benefits of chlorhexidine at higher concentrations.
Supplementary Information
Additional file 2. Risk of bias assessment.
Additional file 4. Direct–indirect evidence for the main outcome.
Acknowledgements
None
Abbreviations
- ICU
Intensive care unit
- LOS
Length of stay
- MD
Mean difference
- OR
Odds ratio
- SD
Standard deviation
- SUCRA
Surface under the cumulative ranking curve
- VAP
Ventilator-associated pneumonia
Authors’ contributions
Conceptualization: Alessandro De Cassai; Methodology: Alessandro De Cassai, Tommaso Pettenuzzo; Formal analysis and investigation: Alessandro De Cassai; Writing - original draft preparation:Alessandro De Cassai Tommaso Pettenuzzo; Writing - review and editing: Alessandro De Cassai, Tommaso Pettenuzzo, Paolo Navalesi, Marina Munari, Veronica Busetto, Christian Legnaro, Chiara Pretto, Alessio Rotondi, Nicolò Sella; Funding acquisition: all the authors; Supervision: Paolo Navalesi and Marina Munari
Funding
None.
Availability of data and materials
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Melsen WG, Rovers MM, Groenwold RH, et al. Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis. 2013;13:665–671. doi: 10.1016/S1473-3099(13)70081-1. [DOI] [PubMed] [Google Scholar]
- 2.Emmerson AM, Enstone JE, Griffin M, Kelsey MC, Smyth ET. The Second National Prevalence Survey of infection in hospitals–overview of the results. J Hosp Infect. 1996;32:175–190. doi: 10.1016/s0195-6701(96)90144-1049. [DOI] [PubMed] [Google Scholar]
- 3.Aly NY, Al-Mousa HH, Asar Al, el SM, Nosocomial infections in a medical-surgical intensive care unit. Med Princ Pract. 2008;17:373–377. doi: 10.1159/000141500. [DOI] [PubMed] [Google Scholar]
- 4.Farag AM, Tawfick MM, Abozeed MY, Shaban EA, Abo-Shadi MA. Microbiological profile of ventilator-associated pneumonia among intensive care unit patients in tertiary Egyptian hospitals. J Infect Dev Ctries. 2020;14:153–161. doi: 10.3855/jidc.12012. [DOI] [PubMed] [Google Scholar]
- 5.Wu D, Wu C, Zhang S, Zhong Y. Risk factors of ventilator-associated pneumonia in critically iii patients. Front Pharmacol. 2019;10:482. doi: 10.3389/fphar.2019.00482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Muscedere J, Sinuff T, Heyland DK, et al. The clinical impact and preventability of ventilator-associated conditions in critically ill patients who are mechanically ventilated. Chest. 2013;144:1453–1460. doi: 10.1378/chest.13-0853. [DOI] [PubMed] [Google Scholar]
- 7.Small J, Campbell R, Paton L, Mackay A. A survey of chlorhexidine oral care in Scottish intensive care units. ICMx. 2015;3(Suppl 1):A710. doi: 10.1186/2197-425X-3-S1-A710. [DOI] [Google Scholar]
- 8.Segers P, Speekenbrink RGH, Ubbink DT, Van Ogtrop ML, De Mol BA. Prevention of nosocomial infection in cardiac surgery by decontamination of the nasopharynx and oropharynx with chlorhexidine gluconate: a randomized controlled trial. JAMA. 2006;269:2460–2466. doi: 10.1001/jama.296.20.2460. [DOI] [PubMed] [Google Scholar]
- 9.Fourrier F, Dubois D, Pronnier P, et al. Effect of gingival and dental plaque antiseptic decontamination on nosocomial infections acquired in the intensive care unit: a double-blind placebo-controlled multicenter study. Crit Care Med. 2005;33:1728–1735. doi: 10.1097/01.CCM.0000171537.03493.B0. [DOI] [PubMed] [Google Scholar]
- 10.Tantipong H, Morkchareonpong C, Jaiyindee S, Thamlikitkul, Randomized controlled trial and meta-analysis of oral decontamination with 2% chlorhexidine solution for the prevention of ventilator-associated pneumonia. Infect Control Hosp Epidemiol. 2008;29:131–136. doi: 10.1086/526438. [DOI] [PubMed] [Google Scholar]
- 11.Klompas M, Speck K, Howell MD, Greene LR, Berenholtz SM. Reappraisal of routine oral care with chlorhexidine gluconate for patients receiving mechanical ventilation: systematic review and meta-analysis. JAMA Intern Med. 2014;174:751–761. doi: 10.1001/jamainternmed.2014.359. [DOI] [PubMed] [Google Scholar]
- 12.De Cassai A (2023) Effectiveness of different concentrations of chlorhexidine for prevention of ventilator-associated pneumonia: a network meta-analysis. Open Science Framework. 10.17605/OSF.IO/8CUKF
- 13.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta - analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. doi: 10.1136/bmj.b2700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 15.Chaimani A, Caldwell DM, Li T, Higgins JPT, Salanti G (2022) Chapter 11: Undertaking network meta-analyses. In: Higgins JPT, Thomas J, Chandler J, et al., eds. Cochrane Handbook for Systematic Reviews of Interventions version 6.3 (updated February 2022). Cochrane. Available from www.training.cochrane.org/handbook. Accessed 11 Apr 2023
- 16.Rücker G, Schwarzer G. Ranking treatments in frequentist network meta - analysis works without resampling methods. BMC Med Res Methodol. 2015;15:58. doi: 10.1186/s12874-015-0060-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13. doi: 10.1186/1471-2288-5-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta - analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Meinberg MC, Cheade Mde F, Miranda AL, Fachini MM, Lobo SM. The use of 2% chlorhexidine gel and toothbrushing for oral hygiene of patients receiving mechanical ventilation: effects on ventilator-associated pneumonia. Rev Bras Ter Intensiva. 2012;24:369–374. doi: 10.1590/s0103-507x2012000400013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Scannapieco FA, Yu J, Raghavendran K, et al. A randomized trial of chlorhexidine gluconate on oral bacterial pathogens in mechanically ventilated patients. Crit Care. 2009;13:R117. doi: 10.1186/cc7967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Özçaka Ö, Başoğlu OK, Buduneli N, Taşbakan MS, Bacakoğlu F, Kinane DF. Chlorhexidine decreases the risk of ventilator-associated pneumonia in intensive care unit patients: a randomized clinical trial. J Periodontal Res. 2012;47:584–592. doi: 10.1111/j.1600-0765.2012.01470.x. [DOI] [PubMed] [Google Scholar]
- 22.De Riso AJ, II, Ladowski JS, Dillon TA, Justice JW, Peterson AC, Chlorhexidine gluconate 0.12% oral rinse reduces the incidence of total nosocomial respiratory infection and nonprophylactic systemic antibiotic use in patients undergoing heart surgery. Chest. 1996;109:1556–1561. doi: 10.1378/chest.109.6.1556. [DOI] [PubMed] [Google Scholar]
- 23.Panchabhai TS, Dangayach NS, Krishnan A, Kothari VM, Karnad DR. Oropharyngeal cleansing with 0.2% chlorhexidine for prevention of nosocomial pneumonia in critically ill patients: an open-label randomized trial with 0.01% potassium permanganate as control. Chest. 2009;135:1150–1156. doi: 10.1378/chest.08-1321. [DOI] [PubMed] [Google Scholar]
- 24.Bellissimo-Rodrigues F, Bellissimo-Rodrigues WT, Viana JM, et al. Effectiveness of oral rinse with chlorhexidine in preventing nosocomial respiratory tract infections among intensive care unit patients. Infect Control Hosp Epidemiol. 2009;30:952–958. doi: 10.1086/605722. [DOI] [PubMed] [Google Scholar]
- 25.Dale CM, Rose L, Carbone S, et al. Effect of oral chlorhexidine de-adoption and implementation of an oral care bundle on mortality for mechanically ventilated patients in the intensive care unit (CHORAL): a multi-center stepped wedge cluster-randomized controlled trial. Intensive Care Med. 2021;47:1295–1302. doi: 10.1007/s00134-021-06475-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Munro CL, Grap MJ, Jones DJ, McClish DK, Sessler CN. Chlorhexidine, toothbrushing, and preventing ventilator-associated pneumonia in critically ill adults. Am J Crit Care. 2009;18:428–437. doi: 10.4037/ajcc2009792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zarinfar N, Ghaznavi-Rad E, Mahmoodiyeh B, Reyhani A. Comparison of three interventional approaches to prevent ventilator-associated pneumonia in intensive care units (ICUs): a clinical trial study. Qatar Med J. 2021;2021:21. doi: 10.5339/qmj.2021.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Grap MJ, Munro CL, Hamilton VA, Elswick RK, Jr, Sessler CN, Ward KR. Early, single chlorhexidine application reduces ventilator-associated pneumonia in trauma patients. Heart Lung. 2011;40:e115–e122. doi: 10.1016/j.hrtlng.2011.01.006. [DOI] [PubMed] [Google Scholar]
- 29.Jahanshir M, Nobahar M, Ghorbani R, Malek F. Effect of clove mouthwash on the incidence of ventilator-associated pneumonia in intensive care unit patients: a comparative randomized triple-blind clinical trial. Clin Oral Investig. 2023;27:3589–3600. doi: 10.1007/s00784-023-04972-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zand F, Zahed L, Mansouri P, Dehghanrad F, Bahrani M, Ghorbani M. The effects of oral rinse with 0.2% and 2% chlorhexidine on oropharyngeal colonization and ventilator associated pneumonia in adults' intensive care units. J Crit Care. 2017;40:318–322. doi: 10.1016/j.jcrc.2017.02.029. [DOI] [PubMed] [Google Scholar]
- 31.Lin YJ, Xu L, Huang XZ, et al. Reduced occurrence of ventilator-associated pneumonia after cardiac surgery using preoperative 0.2% chlorhexidine oral rinse: results from a single-centre single-blinded randomized trial. J Hosp Infect. 2015;91:362–366. doi: 10.1016/j.jhin.2015.08.018. [DOI] [PubMed] [Google Scholar]
- 32.Tuon FF, Gavrilko O, Almeida S, et al. Prospective, randomised, controlled study evaluating early modification of oral microbiota following admission to the intensive care unit and oral hygiene with chlorhexidine. J Glob Antimicrob Resist. 2017;8:159–163. doi: 10.1016/j.jgar.2016.12.007. [DOI] [PubMed] [Google Scholar]
- 33.Pobo A, Lisboa T, Rodriguez A, et al. A randomized trial of dental brushing for preventing ventilator-associated pneumonia. Chest. 2009;136:433–439. doi: 10.1378/chest.09-0706. [DOI] [PubMed] [Google Scholar]
- 34.Meidani M, Khorvash F, Abbasi S, Cheshmavar M, Tavakoli H. Oropharyngeal irrigation to prevent ventilator-associated-pneumonia: comparing potassium permangenate with chlorhexidine. Int J Prev Med. 2018;9:93. doi: 10.4103/ijpvm.IJPVM_370_17.PMID:30450176;PMCID:PMC6202778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Koeman M, van der Ven AJ, Hak E, et al. Oral decontamination with chlorhexidine reduces the incidence of ventilator-associated pneumonia. Am J Respir Crit Care Med. 2006;173:1348–1355. doi: 10.1164/rccm.200505-820OC. [DOI] [PubMed] [Google Scholar]
- 36.Fourrier F, Cau-Pottier E, Boutigny H, Roussel-Delvallez M, Jourdain M, Chopin C. Effects of dental plaque antiseptic decontamination on bacterial colonization and nosocomial infections in critically ill patients. Intensive Care Med. 2000;26:1239–1247. doi: 10.1007/s001340000585. [DOI] [PubMed] [Google Scholar]
- 37.Berry AM, Davidson PM, Masters J, Rolls K, Ollerton R. Effects of three approaches to standardized oral hygiene to reduce bacterial colonization and ventilator associated pneumonia in mechanically ventilated patients: a randomised control trial. Int J Nurs Stud. 2011;48:681–688. doi: 10.1016/j.ijnurstu.2010.11.004. [DOI] [PubMed] [Google Scholar]
- 38.Cabov T, Macan D, Husedzinović I, et al. The impact of oral health and 0.2% chlorhexidine oral gel on the prevalence of nosocomial infections in surgical intensive-care patients: a randomized placebo-controlled study. Wien Klin Wochenschr. 2010;122:397–404. doi: 10.1007/s00508-010-1397-y. [DOI] [PubMed] [Google Scholar]
- 39.Lee S, Lighvan NL, McCredie V, et al. Chlorhexidine-related mortality rate in critically ill subjects in intensive care units: a systematic review and meta-analysis. Respir Care. 2019;64:337–349. doi: 10.4187/respcare.06434. [DOI] [PubMed] [Google Scholar]
- 40.Villar CC, Pannuti CM, Nery DM, Morillo CM, Carmona MJ, Romito GA. Effectiveness of Intraoral chlorhexidine protocols in the prevention of ventilator-associated pneumonia: meta-analysis and systematic review. Respir Care. 2016;61:1245–1259. doi: 10.4187/respcare.04610. [DOI] [PubMed] [Google Scholar]
- 41.Plantinga NL, Wittekamp BHJ, Leleu K, et al. Oral mucosal adverse events with chlorhexidine 2% mouthwash in ICU. Intensive Care Med. 2016;42:620–621. doi: 10.1007/s00134-016-4217-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cieplik F, Jakubovics NS, Buchalla W, Maisch T, Hellwig E, Al-Ahmad A. Resistance toward chlorhexidine in oral bacteria - is there cause for concern? Front Microbiol. 2019;10:587. doi: 10.3389/fmicb.2019.00587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Alvarez Lerma F, Sanchez Garcia M, Lorente L, et al. Guidelines for the prevention of ventilator-associated pneumonia and their implementation. The Spanish “Zero-VAP” bundle. Med Intensiva. 2014;38:226–236. doi: 10.1016/j.medin.2013.12.007. [DOI] [PubMed] [Google Scholar]
- 44.Klompas M, Branson R, Eichenwald EC, et al. Strategies to prevent ventilator-associated pneumonia in acute care hospitals: 2014 update. Infect Control Hosp Epidemiol. 2014;35(Suppl 2):S133–154. doi: 10.1017/s0899823x00193894. [DOI] [PubMed] [Google Scholar]
- 45.Torres A, Niederman MS, Chastre J, et al. International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European Respiratory Society (ERS), European Society of Intensive Care Medicine (ESICM), European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and Asociacion Latinoamericana del Torax (ALAT) Eur Respir J. 2017;50(3):1700582. doi: 10.1183/13993003.00582-2017. [DOI] [PubMed] [Google Scholar]
- 46.Klompas M, Branson R, Cawcutt K, et al. Strategies to prevent ventilator-associated pneumonia, ventilator-associated events, and nonventilator hospital-acquired pneumonia in acute-care hospitals: 2022 Update. Infect Control Hosp Epidemiol. 2022;43:687–713. doi: 10.1017/ice.2022.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 2. Risk of bias assessment.
Additional file 4. Direct–indirect evidence for the main outcome.
Data Availability Statement
No datasets were generated or analysed during the current study.