Abstract
The Antenatal Late Preterm Steroids (ALPS) trial was designed to address respiratory morbidity common in infants born late preterm. The study was published published in April, 2016 and, shortly thereafter, changed clinical practice in obstetrics in the United States. The following chapter describes the ALPS trial study design in detail, including the background leading to the trial, the study outcomes, and initial findings of the long-term follow-up study. The ALPS story would not be complete without Elizabeth Thom, PhD, who died before her time. Her brilliance largely contributed to the design of the ALPS trial.
Introduction
In 1994, the National Institute of Child Health and Human Development (NICHD) held a consensus conference to examine the effect of antenatal corticosteroids for fetal lung maturation.1 This 16-member panel inclusive of obstetricians, neonatologists, psychologists, and biostatisticians, among others, was convened to review the data surrounding antenatal corticosteroids for fetal benefit and to develop consensus guidelines based on the evidence. The panel noted that only 12% to 18% of eligible parturients delivering infants under 1500 grams received steroids due to concerns surrounding efficacy and possible complications associated with steroid treatment. As is well known, the group concluded that antenatal corticosteroid therapy was not only indicated in those at risk for preterm delivery but would also substantially decrease neonatal morbidity and mortality, as well as save health care dollars. Antenatal corticosteroids were recommended for pregnancies at risk for preterm delivery from 24-34 weeks gestation. The group acknowledged that there was insufficient data to make a recommendation for or against antenatal steroids at greater than 34 weeks.1 These important events in the history of obstetric care occurred early in my years of training and peaked my interest in the study of fetal lung maturation. Of particular interest to me was why antenatal corticosteroids were not offered to pregnancies resulting in infants born preterm, but after 34 weeks, recognizing that so many of these infants were admitted to the neonatal intensive care unit for respiratory support.
Background
Preterm Birth (PTB) is a major public health problem in the United States and is the leading cause of morbidity and mortality in non-anomalous infants.2-5 Currently, approximately 1 in 10 US women deliver preterm, a rate that is unacceptably high.6 Roughly 75% of preterm births in North America occur in the “late preterm” period between 340 and 366 weeks gestation.7 In 2021, the rate of late preterm birth increased to 7.67% from 7.4% in 2020. This represents a 3.5% increase and is the highest late preterm birth rate since this number was recorded in 2007.8
Prior to 2005, the prevailing thought in obstetrics had been that infants born in the late preterm period may have mild respiratory compromise requiring brief periods of oxygen, but that more severe and costly prematurity-related morbidities were negligible. However, emerging data at the time, now confirmed in multiple publications, showed significant and unrecognized respiratory compromise.9-12 In one of the seminal papers of that epoch, investigators leveraged data from the Safe Labor Consortium to show the effect of decreasing gestational age at birth on respiratory morbidity, using 39-40 weeks as the reference group.13 All respiratory morbidities assessed, including respiratory distress syndrome (RDS), transient tachypnea of the newborn (TTN), pneumonia, respiratory failure, surfactant use, and the need for a ventilator or oscillator, were significantly more common at all gestational weeks from 34 to 38 weeks compared to the reference group.13 In 2005, surrounding newer acknowledgement of the size and unsuspected complexity of this group of neonates, the NICHD convened a workshop entitled “Optimizing Care and Outcome of the Near-Term Pregnancy and the Near-Term Newborn Infant”. At this meeting, the morbidity of births occurring from 340 to 366 weeks was recognized and its impact quantified.10 Gaps in both basic science and clinical knowledge were identified and specific research questions enumerated. One of the most critical questions was whether extending the therapeutic window for administration of antenatal corticosteroids to include the late preterm population would reduce morbidity in this group. The Antenatal Late Preterm Steroids (ALPS) trial was designed to specifically address this knowledge gap.
The ALPS trial
Study Design
The primary research question from the ALPS trial addressed whether infants of pregnant people with an anticipated delivery in the late preterm period, from 34+0 to 36+6 weeks, who had not previously received a course of steroids, would have a decreased need for respiratory support when exposed to antenatal betamethasone versus no betamethasone. To answer this question, a double-blind, randomized, placebo-controlled trial was performed including individuals with singleton pregnancies who were likely to deliver late preterm. A high likelihood for late preterm delivery was defined by the following criteria: preterm prelabor rupture of the membranes, preterm labor with intact membranes with a cervix that was at least 3cm dilated or 75% effaced in the presence of regular contractions, or indicated preterm delivery, where an induction had to be scheduled by 36+5 weeks or an indicated delivery by cesarean had to occur by 36+6 weeks. There were several exclusion criteria, many of which were in place to prevent contamination of the placebo group (prior course of steroids in the pregnancy, candidates for stress dose steroids), to prevent unblinding (pregestational diabetes), or for internal validity (delivery expected within 12 hours, fetal demise, known anomaly, participation in a concurrent interventional trial). Potential participants were also excluded if they had known chorioamnionitis, evidence of non-reassuring fetal status requiring immediate delivery, twin gestation or higher order multiple, or maternal contraindication to betamethasone. Initially, potential participants with gestational diabetes were also excluded due to the potential for unblinding, but after recruitment that was slower than expected in the first 6 months of the study, individuals with gestational diabetes were ultimately included.
After signing informed consent, which also included an additional optional consent for future contact, eligible participants were randomized in a 1:1 ratio to receive either an intramuscular injection of 12 mg of betamethasone or 12 mg of an identical placebo. If the subject was undelivered in 24 hours, a second equivalent dose of either betamethasone or placebo, based on initial treatment assignment, was administered. Participants were not tocolyzed to “complete” steroid administration, such that only one intervention would be studied.
Primary and Secondary Outcomes
The study primary outcome was considered carefully. It was acknowledged that respiratory morbidity included more than just respiratory distress syndrome. Adequate fetal lung fluid is required for normal pulmonary development. Until near term, the fetal lung actively transports chloride across the airway epithelium, resulting in electrical and osmotic gradients that favor the movement of water into the alveolar lumen.14 The last few weeks of gestation are particularly important for the fetal respiratory transition to air breathing. For effective gas exchange to occur, alveolar spaces must be cleared of excess fluid and pulmonary blood flow must increase to match ventilation with perfusion. To prepare for this, a switch from lung fluid secretion into the alveoli to net fluid absorption must take place to allow for rapid fluid clearance at birth.15 Sodium transport via the epithelial sodium channels (ENaC) within the fetal/neonatal lung has emerged as a key event in the clearance of alveolar fluid.16-25 Decreased ENaC activity has been implicated in several disease states, including TTN;26 importantly, corticosteroids have been shown to increase ENaC expression in vitro, in animal models, and in preterm infants.16
Levering these mechanistic data, it was thought that the potential benefit of corticosteroid administration would manifest through fetal lung fluid clearance. Thus, the chosen primary outcome reflected the need for neonatal respiratory support that would be needed due to impeded fluid clearance. Thus, the endpoint chosen was the need for respiratory treatment instead of a specific diagnosis. This endpoint was a common denominator of the steroid-responsive biological processes that initiated the cascade that led to the need for respiratory support. In addition, the endpoint provided insight into the impact of disease on resource utilization, the risk for iatrogenic complications, and the delay in initiation of feeds and maternal bonding. Therefore, after consultation with leading neonatologists around the country, the following outcome composite was developed:
CPAP or humidified high-flow nasal cannula (HHFNC) for ≥ 2 continuous hours in the first 72 hours of life. HHFNC is defined as a flow rate of > 1 Liter/minute
Oxygen requirement of FiO2 ≥ 0.3 by any delivery method for ≥ 4 continuous hours in the first 72 hours of life
Mechanical ventilation in the first 72 hours of life
Extra corporeal membrane oxygenation (ECMO). It was recognized that it is highly unlikely that an infant that is placed on ECMO would not satisfy one of the other components of the primary outcome
Stillbirth or neonatal death less than 24 hours of age. Though expected to be rare, this was included because of the competing risk potential, i.e. a fetus that dies in utero or shortly after birth could be precluded from receiving respiratory support.
The main secondary outcome included all the elements in the primary outcome but extended the need for respiratory support to include the following:
CPAP or HHFNC for ≥ 12 total hours in the first 72 hours of life
Oxygen requirement of FiO2 ≥ 0.3 for ≥ 24 total hours in the first 72 hours of life
Other neonatal outcomes thought to be common in the late preterm period were included in the abstracted secondary outcomes. These included specific respiratory diagnoses (including RDS, TTN, and apnea), as well as other late preterm morbidities (including hypoglycemia, hyperbilirubinemia, and neonatal infectious morbidity). Maternal outcomes included maternal infectious morbidity (chorioamnionitis and postpartum endomyometritis), length of labor, mode of delivery, delivery indications, and time from dose to delivery.
Study Results
The study enrolled 2,831 participants from October 2010 to February 2015. The primary outcome, the composite of need for respiratory support, was lower in the betamethasone group than in the placebo group (11.6% vs. 14.4%; relative risk [RR], 0.80; 95% confidence interval [CI], 0.66 to 0.97; P = 0.02, Figure 1).27 The number needed to treat to prevent one case of the primary outcome was 35. The main secondary outcome, a more severe outcome which extended the need for respiratory support as described above, was also significantly decreased in the betamethasone group. The proportion of infants with this outcome was initially reported as 8.1% (betamethasone) vs. 12.1% (placebo); RR 0.67; 95% CI, 0.53 to 0.84; P<0.001.27 However, an error in the variable used to calculate this outcome was discovered after a reader inquiry, and our updated results were sent to the journal and published as an erratum.28 In fact, there was slightly more benefit: 7.9% in the betamethasone group: versus 12.1% in the placebo group; RR 0.66; 95% CI, 0.52 to 0.82; P<0.001 (Figure 2).28 This changed the number needed to treat for the more severe outcome from 25 to 24. Additionally, the supplementary appendix initially indicated the possibility of a greater benefit to those delivered by cesarean. However, this was also incorrect and updated in the currently available supplement.27,28 None of the subgroup analyses had significant interactions.28
Figure 1. ALPS Primary Outcome, Composite of Need for Respiratory Support.
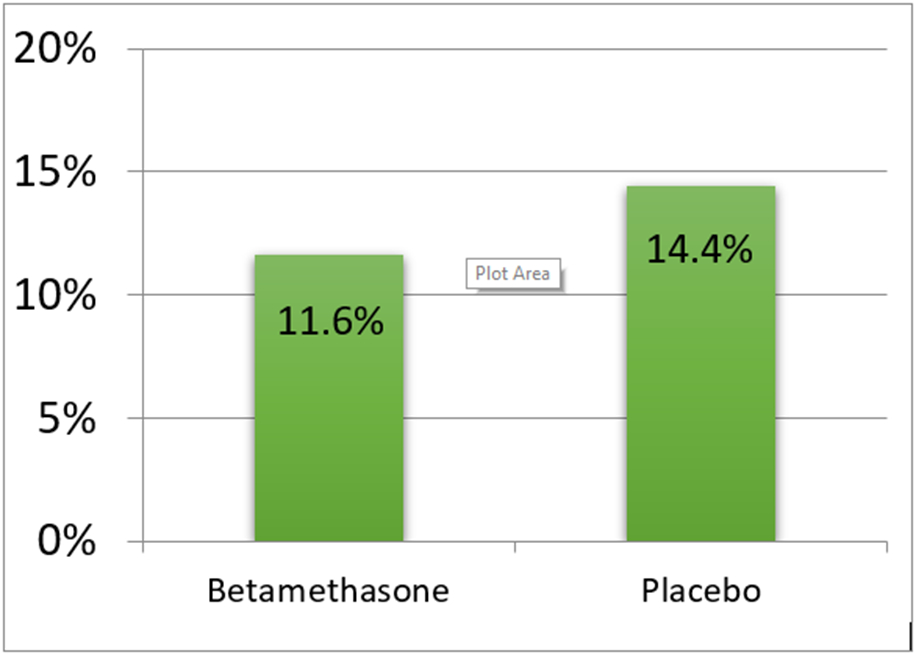
Composite of Need for Respiratory Support, Relative Risk 0.80, 95% CI 0.66-0.97, P=0.02
Figure 2. ALPS Main Secondary Outcome, Severe Respiratory Complication.

Severe Respiratory Morbidity, Relative Risk 0.66; 95% CI, 0.52 to 0.82; P<0.001
There are several secondary outcomes that are worth noting. There were significant decreases in TTN (RR 0.68, 95% CI 0.53 to 0.87), surfactant use (RR 0.59, 95% CI 0.37 to 0.96), and the need for resuscitation at birth (RR 0.78, 95% CI 0.66 to 0.92). Hypoglycemia was more common in infants exposed to steroids antenatally (RR 1.6, 95% CI 1.37 to 1.87). While biologically plausible, with expected maternal glycemia after steroid administration possibly resulting in lower neonatal blood glucose concentrations, this difference in hypoglycemia had not been noted in the initial trial of antenatal corticosteroids by Liggins and Howie29 and subsequently was not studied in other early steroid trials.30-33
While the short-term benefit of antenatal corticosteroids administered from 34 to 36 weeks were clear from this study27 and from the Cochrane review assessing this late preterm epoch,34 the questions raised by the increase in hypoglycemia warranted further investigation. Late preterm infants are known to have a higher incidence of hypoglycemia than those born at term due to immature glucose homeostasis, such that the American Academy of Pediatrics has included these infants, along with infants of diabetic mothers, and small- or large- for gestational age infants, as those who should be screened for hypoglycemia at birth.35 Because persistent or prolonged hypoglycemia has been associated with adverse neurodevelopment,35 understanding the nature and length of time of hypoglycemia became important.
Neonatal Hypoglycemia
To address the question of hypoglycemia, ALPS investigators performed a secondary analysis of the ALPS trial after re-abstracting all available charts.36 Unreviewed charts were those from centers no longer participating in the Maternal-Fetal Medicine Units (MFMU) Network (n=209), those lost to follow up (n=4), and those with missing charts (n=9). In the parent trial, hypoglycemia was coded as a binary variable, yes or no, based on a cut-off of <40 mg/dL. The re-abstraction, rather, captured all listed glucose values in the neonatal chart in order to ascertain the onset, lowest value, and duration of hypoglycemia if present. Expert consensus is that prolonged or persistent hypoglycemia is that which has persisted for ≥ 72 hours after birth,37 so this definition was employed in this analysis.
Of 2,831 individuals in the parent trial, 2,609 (92.2%) participant charts were reviewed. The onset of hypoglycemia occurred in the first 24 hours for the majority of both groups: 374 of 385 (97.1%) in the betamethasone group and 214 of 222 (96.4%) in the placebo group (P=0.63).36 Similarly, the lowest median (interquartile range [IQR]) recorded blood glucose value was the same in both groups: 31mg/dL (24mg/dL to 35 mg/dL) in the betamethasone group and 31mg/dL (25mg/dL to 36 mg/dL) in the placebo group. There was no significant difference in treatment for hypoglycemia, with less than half of the infants in both groups requiring formal treatment [132 of 387 infants with hypoglycemia in the betamethasone group (34.1%) and 73 of 223 infants with hypoglycemia in the placebo group (32.7%), p=0.73]. However, there was a difference in the time to resolution of hypoglycemia. Neonatal hypoglycemia resolved more quickly in the betamethasone group [median, IQR (2.80 hrs, 2.03 to 7.03 hrs)] compared with hypoglycemia in the placebo group (3.74 (2.15 to 15.08)). In fact, the majority of hypoglycemia resolved within 6 hours in the betamethasone group.36 This study showed that hypoglycemia was neither persistent nor prolonged and resolved more rapidly in the group exposed to betamethasone.
Cost Analysis
It has been suggested that former late preterm infants account for the majority of costs related to preterm delivery through childhood due to the sheer magnitude of their numbers in relation to the fewer early preterm births.38,39 Late preterm birth has been associated with higher health care costs when compared to infants born at term, primarily due to longer hospitalizations, which are related in part to respiratory morbidity.40-44 Because of the reduction in respiratory morbidity noted in the ALPS trial,27 we further sought to assess whether this translated to decreased costs by performing a cost analysis.45
The analysis included participants who received at least one dose of the study drug and who were not lost to follow-up and included data from the parent trial on resource utilization, which was collected through hospital discharge on the mother and infant.45 The analysis took the perspective of a third-party payer, in which direct medical costs and associated overhead, accruing to hospitals and medical payers for the care of enrolled subjects and their infants, were included.
Maternal costs included those of betamethasone, as well as of the outpatient visits or inpatient stay required to administer betamethasone. Enrolled participants who received betamethasone during the stay in which they delivered incurred no additional costs for drug administration. No additional cost of treatment was assigned to the placebo group, as they would be considered untreated without need to return for a visit specifically for steroid administration. Prices for maternal interventions were based on Medicaid rates.46,47 Neonatal costs included all direct medical costs for newborn care. For infants admitted to the Neonatal Intensive Care Unit (NICU), comprehensive daily costs were stratified by the acuity of respiratory illness. Nationally-representative cost estimates from the literature were used to estimate costs for infants admitted to the regular newborn nursery.
Employing these assumptions, treatment with betamethasone resulted in a total mean mother-infant dyad cost of $4,681 ± $5,798, which was significantly less than that for mother-infant dyads in the placebo group, $5,379 ± $8,422, p=0.022, 95% confidence interval: $186, $1,257. Nonparametric bootstrapping was employed to test the validity of the model under varied assumptions. Assessment of bootstrap replications showed that late preterm antenatal steroid administration was the dominant modal in 98.8% of samples, thus confirming that steroid administration was the less costly strategy.45
Population-level data
Once the findings of the ALPS trial were disseminated, there was a quick uptake in use of this intervention in the United States.48 One group of investigators sought to evaluate whether this change in practice had resulted in a decrease in late preterm respiratory morbidity49 by performing an interrupted time series analysis using US natality data. They reviewed assisted ventilation use and ventilation use for >6 hours in infants born between 34 and 36 completed weeks of gestation. They selected a 12-month period prior to the first presentation of the ALPS trial, which was presented and simultaneously published online in February, 2016, and a 12-month follow-up period after the obstetric societies had published their recommendations (November, 2016 to October, 2017).49-51 The authors found both an increase in exposure to antenatal corticosteroids and a corresponding decrease in ventilation use after the dissemination period (adjusted incidence rate ratio 0.91, 95% CI 0.85 to 0.89). They did not find a decrease in ventilator use for >6 hours, but this was not an outcome studied in the ALPS trial. They noted a few limitations of their analysis, which are inherent in analyses from birth certificate data, including the inability to get granular data on exposure timing and outcomes. However, they also report that the decrease in the risk of ventilatory support was larger than expected from changes that would be from unknown factors or from chance alone.49
Safety of antenatal corticosteroids
Prior to starting the ALPS trial, safety data on the use of antenatal corticosteroids were reviewed. It was acknowledged that the safety and efficacy of a single course of antenatal corticosteroids in singletons had been well-established, particularly after the 1994 NICHD Consensus Conference.1 Randomized controlled trials had not demonstrated growth restriction in neonates treated with a single course of antenatal steroids.52,53 In addition, no long-term alterations of development have been described following a single course of corticosteroid therapy. Four large trials had monitored children through early childhood.52-55 Follow-up data on physical growth and neurodevelopment was reported at various ages: two years of age in the Helsinki trial 54, three years of age in the U.S. trial 52, six years of age in the Auckland trial 53, and 12 years of age in the Amsterdam trial55. All of these studies have confirmed the absence of an adverse effect of corticosteroids on long-term development. The Helsinki trial also demonstrated that the reduction of neonatal cerebral complications with antenatal glucocorticoids may translate to a lower incidence of cerebral palsy in surviving premature children.
Neurodevelopment after antenatal corticosteroids administered at >34 weeks
After the ALPS trial was underway, but prior to its completion, two important studies were published related to safety and long-term outcomes of antenatal corticosteroids, particularly as it relates to neurodevelopment.56,57 One, named the ASTECS-2 study, was follow up to a study that began in 2005, in which investigators completed a trial of open-label betamethasone compared with usual care in individuals undergoing term scheduled cesarean delivery (the Antenatal Steroid for Term Elective Cesarean Trial (ASTECS)).58 ASTECS-2 was not pre-planned and used questionnaires completed by parents as well as school assessment data.56 At the time of assessment, the children were ages 8-15 years; data were available for 407 children (41% of trial offspring). No differences were observed between the offspring of mothers randomized to antenatal betamethasone compared with the offspring of mothers randomized to usual care in the total difficulties score of the strengths and difficulties questionnaire (SDQ) (mean of 8.03 ± 6.83 compared with mean of 7.85 ± 6.49, respectively) or in any of the subscales. No significant differences were observed between the betamethasone and usual care groups for standard assessment tests, with similar level-4 achievement observed for mathematics, science, and English. The only difference in outcomes between groups was in the school assessment of quartile by ability, a subjective measure, which showed a higher percent of children in the lower quartile of academic ability in the betamethasone group (18%, compared with 9% in the usual care group; p=.03). While the study authors concluded “no adverse effect was seen on health, behaviour and academic achievement of children born following a single course of antenatal betamethasone at term,” and despite the lack of difference in multiple cognitive measures and the risk of a Type 1 error with multiple comparisons, this isolated finding has been used to suggest a signal for harm after steroid exposure at term.59
The second study assessing neurodevelopment in this time period, also published prior to completion of the ALPS trial, was a meta-analysis by Sotiriadis et al., which included randomized and prospective observational studies of individuals delivering preterm and exposed to a single course of steroids.57 While most studies included had steroid exposure up to 34 weeks, this analysis included the initial Auckland cohort from Liggins and Howie, a largely late preterm study29 with long-term follow-up from steroid exposure up to 36 6/7 wks.60 The authors concluded that antenatal corticosteroids decreased the risk of severe disability and increased intact survival.57 Of note, the median age at delivery for the 30-year neurodevelopmental study from the Aukland cohort was 35 0/7 wks (IQR 33 3/7 to 38 0/7).60 IQ testing revealed no differences between groups, and psychiatric diagnoses were also similar. Including data from only a single course of steroids is important, as some reviews have conflated outcomes of single-course with multiple-course administration (>2), a practice that has fallen out of favor in the most of the world due the known relationship of multiple courses with adverse neurodevelopment.50,59,61,62
Since the publication of ALPS, there have been a few cohort studies that have attempted to evaluate long-term outcomes after late preterm exposure to antenatal corticosteroids in the absence of data from follow-up from the ALPS randomized trial, as randomized trial follow-up is the gold standard. Raikkonnen and colleagues evaluated birth records from a Finnish dataset to assess whether there was an association between antenatal corticosteroid treatment and a list of ten ICD-10 billing codes related to childhood mental and behavioral disorders.63 In this study, neither the gestational age of steroid exposure nor the indication for the exposure was known, outcomes were limited to those who were liveborn, and there was no adjustment for multiple comparisons.63 The authors found an association with four of 10 diagnoses in those born at term, and 2 of 10 diagnoses in those born preterm. The authors state that the practice in Finland during the majority of the study period limited steroid administration to no later than 34 completed weeks. Yet, the authors’ conclusions were that late preterm steroids, not evaluated in this study, should be reconsidered. While there are many limitations to the study that should be considered, perhaps the greatest is in the study design, which limits the outcomes to liveborn infants. Antenatal corticosteroids are known to improve neonatal survival, particularly in early preterm infants. Non-exposed early preterm gestations have a higher likelihood of neonatal death, which is a competing outcome for adverse mental or behavioral disorders. If more deaths occurred in the non-exposed group, and if death or mental disability were the study outcome, then the results would be more likely to move towards the null. This statistical phenomenon was elegantly described in a letter to the editor64 from a separate editorial,65 which also called for a reconsideration of late preterm steroids, in part using data from Raikkonen et al. The letter to the editor stated the following: “Restricting analyses to live-born children may lead to collider-stratification bias.66 This kind of selection bias can easily be detected by drawing a directed acyclic graph, which depicts the assumed casual relations between exposure and outcome concerning their common causes or effects.67 An estimated causal relationship means (1) the exposure truly causes the outcome, (2) there is confounding (common causes), (3) a common effect (a collider) is conditioned upon, or (4) the relationship is there by chance.68”64 The author goes on to say, “Because of the possible presence of collider-stratification bias in these studies, one cannot conclude that ACSs have a negative influence on long-term outcomes in term-born children.”64 However, the data by Raikkonen and colleagues63,69 have been included in a meta-analysis that largely includes similar cohort studies, and not surprisingly, similar conclusions have been drawn.70
ALPS Neurodevelopmental Follow-up
The initial proposal for the ALPS study included a 2-year follow-up, but both funding challenges and the uncertain outcome of the parent trial prevented this plan from coming to fruition. Acknowledging the desire to perform follow-up by investigators, participants in the parent trial were consented for the possibility of future contact. Once the intervention was found to be beneficial, investigators were able to secure funding for childhood follow-up at 6 years of age or greater. Though there are ample data on neurodevelopmental follow-up after steroid administration from randomized trials, neurocognitive testing was considered of importance after finding discortant incidences of neonatal hypoglycemia.27 While some have said that the potential risks are overstated,71,72 others have called for data from the ALPS follow up since follow-up from a randomized trial is the most reliable method of follow-up, given it limits confounding bias due to indication.65,73 The ALPS follow-up study completed recruitment in 2022, and results were presented at the Society for Maternal-Fetal Medicine meeting in 2023. While the manuscript is pending publication, the published abstract data can be shared.74 Enrolled offspring from the ALPS trial underwent cognitive testing using the Differential Ability Scales, 2nd Edition (DAS-II) core components of the general conceptual ability (GCA), which include verbal, non-verbal reasoning, and spatial ability. Secondary outcomes included the Gross Motor Function Classification System (GMFCS) level, the Social Responsiveness Scale (SRS) >65, and Child Behavior Checklist (CBCL) scores. The primary outcome, the proportion of GCA scores < 85 (−1 standard deviation), was assessed by the parent-trial treatment assignment, BMZ or placebo, which remained blinded to investigators, caregivers, and research staff. Of the 949 who completed the DAS-II (479 in the betamethasone group and 470 in the placebo), there was no difference in the primary outcome, 17.1% for BMZ and 18.5% for placebo (adjusted RR 0.94; 95% CI 0.73 to 1.22). There were no differences in any of the secondary outcomes.74
Summary and Conclusions
The ALPS trial changed clinical practice in the United States.50,51 The strategy of administering late preterm steroids for those at high risk of delivery conferred a short-term benefit, inclusive of fewer post-natal interventions and less respiratory support, and was a less costly strategy.27,45 Neonatal hypoglycemia, though more common in the betamethasone group, was neither prolonged nor persistent, and resolved more quickly in the steroid group.36 Finally, the ALPS neurodevelopmental follow-up study found no differences in childhood neurodevelopment.74 The ALPS cohort is also being followed for other health outcomes. The data from the generous participants and offspring of the ALPS trial will continue to provide useful data to the scientific community.
References
- 1.Effect of corticosteroids for fetal maturation on perinatal outcomes. NIH Consensus Development Panel on the Effect of Corticosteroids for Fetal Maturation on Perinatal Outcomes. JAMA 1995;273(5):413–8. DOI: 10.1001/jama.1995.03520290065031. [DOI] [PubMed] [Google Scholar]
- 2.Goldenberg RL, Culhane JF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet 2008;371(9606):75–84. DOI: 10.1016/S0140-6736(08)60074-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Manuck TA. Pharmacogenomics of preterm birth prevention and treatment. BJOG : an international journal of obstetrics and gynaecology 2016;123(3):368–75. DOI: 10.1111/1471-0528.13744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Russell RB, Green NS, Steiner CA, et al. Cost of hospitalization for preterm and low birth weight infants in the United States. Pediatrics 2007;120(1):e1–9. DOI: 10.1542/peds.2006-2386. [DOI] [PubMed] [Google Scholar]
- 5.Ananth CV, Friedman AM, Gyamfi-Bannerman C. Epidemiology of moderate preterm, late preterm and early term delivery. Clinics in perinatology 2013;40(4):601–10. DOI: 10.1016/j.clp.2013.07.001. [DOI] [PubMed] [Google Scholar]
- 6.Gyamfi-Bannerman C, Ananth CV. Trends in spontaneous and indicated preterm delivery among singleton gestations in the United States, 2005-2012. Obstetrics and gynecology 2014;124(6):1069–1074. DOI: 10.1097/AOG.0000000000000546. [DOI] [PubMed] [Google Scholar]
- 7.Davidoff MJ, Dias T, Damus K, et al. Changes in the gestational age distribution among U.S. singleton births: impact on rates of late preterm birth, 1992 to 2002. Semin Perinatol 2006;30(1):8–15. (In eng). DOI: S0146-0005(06)00010-3 [pii] 10.1053/j.semperi.2006.01.009. [DOI] [PubMed] [Google Scholar]
- 8.Osterman MJK, Hamilton BE, Martin JA, Driscoll AK, Valenzuela CP. Births: Final Data for 2021. Natl Vital Stat Rep 2023;72(1):1–53. (https://www.ncbi.nlm.nih.gov/pubmed/36723449). [PubMed] [Google Scholar]
- 9.Yoder BA, Gordon MC, Barth WH Jr. Late-preterm birth: does the changing obstetric paradigm alter the epidemiology of respiratory complications? Obstet Gynecol 2008;111(4):814–22. (In eng). DOI: 111/4/814 [pii] 10.1097/AOG.0b013e31816499f4. [DOI] [PubMed] [Google Scholar]
- 10.Raju TN, Higgins RD, Stark AR, Leveno KJ. Optimizing care and outcome for late-preterm (near-term) infants: a summary of the workshop sponsored by the National Institute of Child Health and Human Development. Pediatrics 2006;118(3):1207–14. (In eng). DOI: 118/3/1207 [pii] 10.1542/peds.2006-0018. [DOI] [PubMed] [Google Scholar]
- 11.Escobar GJ, Clark RH, Greene JD. Short-term outcomes of infants born at 35 and 36 weeks gestation: we need to ask more questions. Semin Perinatol 2006;30(1):28–33. (In eng). DOI: S0146-0005(06)00006-1 [pii] 10.1053/j.semperi.2006.01.005. [DOI] [PubMed] [Google Scholar]
- 12.McIntire DD, Leveno KJ. Neonatal mortality and morbidity rates in late preterm births compared with births at term. Obstet Gynecol 2008;111(1):35–41. (In eng). DOI: 111/1/35 [pii] 10.1097/01.AOG.0000297311.33046.73. [DOI] [PubMed] [Google Scholar]
- 13.Consortium on Safe L, Hibbard JU, Wilkins I, et al. Respiratory morbidity in late preterm births. JAMA 2010;304(4):419–25. DOI: 10.1001/jama.2010.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pitkanen OM, O'Brodovich HM. Significance of ion transport during lung development and in respiratory disease of the newborn. Ann Med 1998;30(2):134–42. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9667791). [DOI] [PubMed] [Google Scholar]
- 15.Strang LB. Fetal lung liquid: secretion and reabsorption. Physiol Rev 1991;71(4):991–1016. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=1924552). [DOI] [PubMed] [Google Scholar]
- 16.Jain L, Chen XJ, Ramosevac S, Brown LA, Eaton DC. Expression of highly selective sodium channels in alveolar type II cells is determined by culture conditions. Am J Physiol Lung Cell Mol Physiol 2001;280(4):L646–58. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=11238004). [DOI] [PubMed] [Google Scholar]
- 17.Bland RD. Lung epithelial ion transport and fluid movement during the perinatal period. Am J Physiol 1990;259(2 Pt 1):L30–7. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=2200282). [DOI] [PubMed] [Google Scholar]
- 18.Bland RD. Loss of liquid from the lung lumen in labor: more than a simple "squeeze". Am J Physiol Lung Cell Mol Physiol 2001;280(4):L602–5. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=11237999). [DOI] [PubMed] [Google Scholar]
- 19.O'Brodovich H. Epithelial ion transport in the fetal and perinatal lung. Am J Physiol 1991;261(4 Pt 1):C555–64. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=1928320). [DOI] [PubMed] [Google Scholar]
- 20.O'Brodovich H, Canessa C, Ueda J, Rafii B, Rossier BC, Edelson J. Expression of the epithelial Na+ channel in the developing rat lung. Am J Physiol 1993;265(2 Pt 1):C491–6. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=7690185). [DOI] [PubMed] [Google Scholar]
- 21.O'Brodovich H, Hannam V, Rafii B. Sodium channel but neither Na(+)-H+ nor Na-glucose symport inhibitors slow neonatal lung water clearance. Am J Respir Cell Mol Biol 1991;5(4):377–84. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=1654956). [DOI] [PubMed] [Google Scholar]
- 22.O'Brodovich H, Hannam V, Seear M, Mullen JB. Amiloride impairs lung water clearance in newborn guinea pigs. J Appl Physiol 1990;68(4):1758–62. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=2161411). [DOI] [PubMed] [Google Scholar]
- 23.O'Brodovich HM. The role of active Na+ transport by lung epithelium in the clearance of airspace fluid. New Horiz 1995;3(2):240–7. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=7583165). [PubMed] [Google Scholar]
- 24.O'Brodovich HM. Immature epithelial Na+ channel expression is one of the pathogenetic mechanisms leading to human neonatal respiratory distress syndrome. Proc Assoc Am Physicians 1996;108(5):345–55. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=8902878). [PubMed] [Google Scholar]
- 25.O'Brodovich HM. Respiratory distress syndrome: the importance of effective transport. J Pediatr 1997;130(3):342–4. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9063404). [PubMed] [Google Scholar]
- 26.Gowen CW Jr., Lawson EE, Gingras J, Boucher RC, Gatzy JT, Knowles MR. Electrical potential difference and ion transport across nasal epithelium of term neonates: correlation with mode of delivery, transient tachypnea of the newborn, and respiratory rate. J Pediatr 1988;113(1 Pt 1):121–7. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=3385520). [DOI] [PubMed] [Google Scholar]
- 27.Gyamfi-Bannerman C, Thom EA, Blackwell SC, et al. Antenatal Betamethasone for Women at Risk for Late Preterm Delivery. N Engl J Med 2016;374(14):1311–20. DOI: 10.1056/NEJMoa1516783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gyamfi-Bannerman C, Thom EA . Antenatal Betamethasone for Women at Risk for Late Preterm Delivery. N Engl J Med 2023;388(18):1728. [DOI] [PubMed] [Google Scholar]
- 29.Liggins GC, Howie RN. A controlled trial of antepartum glucocorticoid treatment for prevention of the respiratory distress syndrome in premature infants. Pediatrics 1972;50(4):515–25. (https://www.ncbi.nlm.nih.gov/pubmed/4561295). [PubMed] [Google Scholar]
- 30.Effect of antenatal dexamethasone administration on the prevention of respiratory distress syndrome. Am J Obstet Gynecol 1981;141(3):276–87. (https://www.ncbi.nlm.nih.gov/pubmed/7025638). [PubMed] [Google Scholar]
- 31.Balci O, Ozdemir S, Mahmoud AS, Acar A, Colakoglu MC. The effect of antenatal steroids on fetal lung maturation between the 34th and 36th week of pregnancy. Gynecol Obstet Invest 2010;70(2):95–9. DOI: 10.1159/000295898. [DOI] [PubMed] [Google Scholar]
- 32.Garite TJ, Rumney PJ, Briggs GG, et al. A randomized, placebo-controlled trial of betamethasone for the prevention of respiratory distress syndrome at 24 to 28 weeks' gestation. Am J Obstet Gynecol 1992;166(2):646–51. (https://www.ncbi.nlm.nih.gov/pubmed/1536246). [DOI] [PubMed] [Google Scholar]
- 33.Gamsu HR, Mullinger BM, Donnai P, Dash CH. Antenatal administration of betamethasone to prevent respiratory distress syndrome in preterm infants: report of a UK multicentre trial. Br J Obstet Gynaecol 1989;96(4):401–10. (https://www.ncbi.nlm.nih.gov/pubmed/2665800). [DOI] [PubMed] [Google Scholar]
- 34.McGoldrick E, Stewart F, Parker R, Dalziel SR. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst Rev 2020;12(12):CD004454. DOI: 10.1002/14651858.CD004454.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Committee on F, Newborn, Adamkin DH. Postnatal glucose homeostasis in late-preterm and term infants. Pediatrics 2011;127(3):575–9. DOI: 10.1542/peds.2010-3851. [DOI] [PubMed] [Google Scholar]
- 36.Gyamfi-Bannerman C, Jablonski KA, Blackwell SC, et al. Evaluation of Hypoglycemia in Neonates of Women at Risk for Late Preterm Delivery: An Antenatal Late Preterm Steroids Trial Cohort Study. Am J Perinatol 2023;40(5):532–538. DOI: 10.1055/s-0041-1729561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Thompson-Branch A, Havranek T. Neonatal Hypoglycemia. Pediatr Rev 2017;38(4):147–157. DOI: 10.1542/pir.2016-0063. [DOI] [PubMed] [Google Scholar]
- 38.Zupancic JA, Dukhovny D. Resource distribution in neonatology: beyond the Pareto principle. Arch Dis Child Fetal Neonatal Ed 2015;100(6):F472–3. DOI: 10.1136/archdischild-2014-308136. [DOI] [PubMed] [Google Scholar]
- 39.Petrou S, Khan K. Economic costs associated with moderate and late preterm birth: primary and secondary evidence. Semin Fetal Neonatal Med 2012;17(3):170–8. DOI: 10.1016/j.siny.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 40.Jacob J, Lehne M, Mischker A, Klinger N, Zickermann C, Walker J. Cost effects of preterm birth: a comparison of health care costs associated with early preterm, late preterm, and full-term birth in the first 3 years after birth. Eur J Health Econ 2016. DOI: 10.1007/s10198-016-0850-x. [DOI] [PubMed] [Google Scholar]
- 41.Berard A, Le Tiec M, De Vera MA. Study of the costs and morbidities of late-preterm birth. Arch Dis Child Fetal Neonatal Ed 2012;97(5):F329–34. DOI: 10.1136/fetalneonatal-2011-300969. [DOI] [PubMed] [Google Scholar]
- 42.McLaurin KK, Hall CB, Jackson EA, Owens OV, Mahadevia PJ. Persistence of morbidity and cost differences between late-preterm and term infants during the first year of life. Pediatrics 2009;123(2):653–9. DOI: 10.1542/peds.2008-1439. [DOI] [PubMed] [Google Scholar]
- 43.Khan KA, Petrou S, Dritsaki M, et al. Economic costs associated with moderate and late preterm birth: a prospective population-based study. BJOG 2015;122(11):1495–505. DOI: 10.1111/1471-0528.13515. [DOI] [PubMed] [Google Scholar]
- 44.Aly H, Hoffman H, El-Dib M, Said L, Mohamed M. Factor affecting length of stay in late preterm infants: an US national database study. J Matern Fetal Neonatal Med 2015;28(5):598–604. DOI: 10.3109/14767058.2014.927428. [DOI] [PubMed] [Google Scholar]
- 45.Gyamfi-Bannerman C, Zupancic JAF, Sandoval G, et al. Cost-effectiveness of Antenatal Corticosteroid Therapy vs No Therapy in Women at Risk of Late Preterm Delivery: A Secondary Analysis of a Randomized Clinical Trial. JAMA Pediatr 2019;173(5):462–468. DOI: 10.1001/jamapediatrics.2019.0032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nicholson WK, Frick KD, Powe NR. Economic burden of hospitalizations for preterm labor in the United States. Obstet Gynecol 2000;96(1):95–101. (https://www.ncbi.nlm.nih.gov/pubmed/10862850). [DOI] [PubMed] [Google Scholar]
- 47.Services CfMM. Centers for Medicare & Medicaid Services Physician Fee Schedule Look-Up Tool. 2015.
- 48.Kearsey EOR, Been JV, Souter VL, Stock SJ. The impact of the Antenatal Late Preterm Steroids trial on the administration of antenatal corticosteroids. Am J Obstet Gynecol 2022;227(2):280 e1–280 e15. DOI: 10.1016/j.ajog.2022.03.037. [DOI] [PubMed] [Google Scholar]
- 49.Clapp MA, Melamed A, Freret TS, James KE, Gyamfi-Bannerman C, Kaimal AJ. US Incidence of Late-Preterm Steroid Use and Associated Neonatal Respiratory Morbidity After Publication of the Antenatal Late Preterm Steroids Trial, 2015-2017. JAMA Netw Open 2022;5(5):e2212702. DOI: 10.1001/jamanetworkopen.2022.12702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Committee on Obstetric P. Committee Opinion No. 713: Antenatal Corticosteroid Therapy for Fetal Maturation. Obstet Gynecol 2017;130(2):e102–e109. DOI: 10.1097/AOG.0000000000002237. [DOI] [PubMed] [Google Scholar]
- 51.Society for Maternal-Fetal Medicine Publications C. Implementation of the use of antenatal corticosteroids in the late preterm birth period in women at risk for preterm delivery. Am J Obstet Gynecol 2016;215(2):B13–5. DOI: 10.1016/j.ajog.2016.03.013. [DOI] [PubMed] [Google Scholar]
- 52.Effects of antenatal dexamethasone administration in the infant: long-term follow-up. J Pediatr 1984;104(2):259–67. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=6363659). [DOI] [PubMed] [Google Scholar]
- 53.Howie RN, Liggins GC The New Zealand study of antepartum glucocorticoid treatment. In: PM F, ed. Lung Development: Biological and Clinical Perspectives, II. New York: Academic Press; 1982:255–65. [Google Scholar]
- 54.Salokorpi T, Sajaniemi N, Hallback H, Kari A, Rita H, von Wendt L. Randomized study of the effect of antenatal dexamethasone on growth and development of premature children at the corrected age of 2 years. Acta Paediatr 1997;86(3):294–8. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9099320). [DOI] [PubMed] [Google Scholar]
- 55.Smolders-de Haas H, Neuvel J, Schmand B, Treffers PE, Koppe JG, Hoeks J. Physical development and medical history of children who were treated antenatally with corticosteroids to prevent respiratory distress syndrome: a 10- to 12-year follow-up. Pediatrics 1990;86(1):65–70. (In eng) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=2193304). [PubMed] [Google Scholar]
- 56.Stutchfield PR, Whitaker R, Gliddon AE, Hobson L, Kotecha S, Doull IJ. Behavioural, educational and respiratory outcomes of antenatal betamethasone for term caesarean section (ASTECS trial). Arch Dis Child Fetal Neonatal Ed 2013;98(3):F195–200. DOI: 10.1136/archdischild-2012-303157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sotiriadis A, Tsiami A, Papatheodorou S, Baschat AA, Sarafidis K, Makrydimas G. Neurodevelopmental Outcome After a Single Course of Antenatal Steroids in Children Born Preterm: A Systematic Review and Meta-analysis. Obstet Gynecol 2015;125(6):1385–1396. DOI: 10.1097/AOG.0000000000000748. [DOI] [PubMed] [Google Scholar]
- 58.Stutchfield P, Whitaker R, Russell I, Antenatal Steroids for Term Elective Caesarean Section Research T. Antenatal betamethasone and incidence of neonatal respiratory distress after elective caesarean section: pragmatic randomised trial. BMJ 2005;331(7518):662. DOI: 10.1136/bmj.38547.416493.06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Aiken CE, Fowden AL, Smith GC. Antenatal glucocorticoids prior to cesarean delivery at term. JAMA Pediatr 2014;168(6):507–8. DOI: 10.1001/jamapediatrics.2014.9. [DOI] [PubMed] [Google Scholar]
- 60.Dalziel SR, Lim VK, Lambert A, et al. Antenatal exposure to betamethasone: psychological functioning and health related quality of life 31 years after inclusion in randomised controlled trial. BMJ 2005;331(7518):665. DOI: 10.1136/bmj.38576.494363.E0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wapner RJ, Sorokin Y, Mele L, et al. Long-term outcomes after repeat doses of antenatal corticosteroids. N Engl J Med 2007;357(12):1190–8. DOI: 10.1056/NEJMoa071453. [DOI] [PubMed] [Google Scholar]
- 62.Asztalos EV, Murphy KE, Willan AR, et al. Multiple courses of antenatal corticosteroids for preterm birth study: outcomes in children at 5 years of age (MACS-5). JAMA Pediatr 2013;167(12):1102–10. DOI: 10.1001/jamapediatrics.2013.2764. [DOI] [PubMed] [Google Scholar]
- 63.Raikkonen K, Gissler M, Kajantie E. Associations Between Maternal Antenatal Corticosteroid Treatment and Mental and Behavioral Disorders in Children. JAMA 2020;323(19):1924–1933. DOI: 10.1001/jama.2020.3937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Dehaene I. Updating the balance between benefits and harms of antenatal corticosteroids requires appropriate causal inference. Am J Obstet Gynecol 2023;229(1):80–81. DOI: 10.1016/j.ajog.2023.02.004. [DOI] [PubMed] [Google Scholar]
- 65.Vidaeff AC, Belfort MA, Kemp MW, et al. Updating the balance between benefits and harms of antenatal corticosteroids. Am J Obstet Gynecol 2023;228(2):129–132. DOI: 10.1016/j.ajog.2022.10.002. [DOI] [PubMed] [Google Scholar]
- 66.Whitcomb BW, Schisterman EF, Perkins NJ, Platt RW. Quantification of collider-stratification bias and the birthweight paradox. Paediatr Perinat Epidemiol 2009;23(5):394–402. DOI: 10.1111/j.1365-3016.2009.01053.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lipsky AM, Greenland S. Causal Directed Acyclic Graphs. JAMA 2022;327(11):1083–1084. DOI: 10.1001/jama.2022.1816. [DOI] [PubMed] [Google Scholar]
- 68.Hernan MA, Hernandez-Diaz S, Robins JM. A structural approach to selection bias. Epidemiology 2004;15(5):615–25. DOI: 10.1097/01.ede.0000135174.63482.43. [DOI] [PubMed] [Google Scholar]
- 69.Raikkonen K, Gissler M, Tapiainen T, Kajantie E. Associations Between Maternal Antenatal Corticosteroid Treatment and Psychological Developmental and Neurosensory Disorders in Children. JAMA Netw Open 2022;5(8):e2228518. DOI: 10.1001/jamanetworkopen.2022.28518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ninan K, Gojic A, Wang Y, et al. The proportions of term or late preterm births after exposure to early antenatal corticosteroids, and outcomes: systematic review and meta-analysis of 1.6 million infants. BMJ 2023;382:e076035. DOI: 10.1136/bmj-2023-076035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Harding JE, Dalziel SR. Re: Antenatal corticosteroids: a time for more careful scrutiny of the indications?: The most reliable evidence is reassuring. BJOG 2016;123(10):1707–8. DOI: 10.1111/1471-0528.14062. [DOI] [PubMed] [Google Scholar]
- 72.Gyamfi-Bannerman C, Saade G, Blackwell SC. Concerns regarding the use of antenatal betamethasone in late preterm gestation: is the evidence of potential harm being overstated? BJOG 2018;125(8):923–924. DOI: 10.1111/1471-0528.14271. [DOI] [PubMed] [Google Scholar]
- 73.Smith GC, Rowitch D, Mol BW. The role of prenatal steroids at 34-36 weeks of gestation. Arch Dis Child Fetal Neonatal Ed 2017;102(4):F284–F285. DOI: 10.1136/archdischild-2016-312333. [DOI] [PubMed] [Google Scholar]
- 74.Gyamfi-Bannerman C. Neurodevelopmental outcomes after late preterm antenatal corticosteroids: the alps follow-up study. Am J Obstet Gynecol 2023;228(1):S764–S765. [DOI] [PMC free article] [PubMed] [Google Scholar]


