Abstract
The identification of regulatory networks contributing to fetal/adult gene expression switches is a major challenge in developmental biology and key to understand the aberrant proliferation of cancer cells, which often reactivate fetal oncogenes. One key example is represented by the developmental gene LIN28B, whose aberrant reactivation in adult tissues promotes tumor initiation and progression. Despite the prominent role of LIN28B in development and cancer, the mechanisms of its transcriptional regulation are largely unknown. Here, by using quantitative RT-PCR and single cell RNA sequencing data, we show that in erythropoiesis the expression of the transcription factor SOX6 matched a sharp decline of LIN28B mRNA during human embryo/fetal to adult globin switching. SOX6 overexpression repressed LIN28B not only in a panel of fetal-like erythroid cells (K562, HEL and HUDEP1; ≈92% p < 0.0001, 54% p = 0.0009 and ≈60% p < 0.0001 reduction, respectively), but also in hepatoblastoma HepG2 and neuroblastoma SH-SY5H cells (≈99% p < 0.0001 and ≈59% p < 0.0001 reduction, respectively). SOX6-mediated repression caused downregulation of the LIN28B/Let-7 targets, including MYC and IGF2BP1, and rapidly blocks cell proliferation. Mechanistically, Lin28B repression is accompanied by SOX6 physical binding within its locus, suggesting a direct mechanism of LIN28B downregulation that might contribute to the fetal/adult erythropoietic transition and restrict cancer proliferation.
Subject terms: Developmental biology, Genetics, Oncology
Introduction
Development is dictated by the timely integration of a complex network of genetic regulatory circuits. The identification of these fetal/adult switches is key to understand not only the developmental process but also the aberrant proliferation of cancer cells, which very often rely on the expression of fetal oncoproteins to sustain their proliferation1.
LIN28B is an oncofetal gene whose expression is preferentially confined to early developmental stages in various tissues. Originally identified in C. elegans, where it controls the timing of larval development2, LIN28 is a RNA-binding protein encoded in mammalians by two genes, Lin28A and Lin28B3,4. In humans, LIN28 has emerged as a Quantitative Trait Locus (QTL), influencing the timing of developmental processes,5–8 and has been implicated in cell reprogramming9, tissue repair10 and cancer, where it is upregulated in a large number of human primary cancers and tumor cell lines11–18.
A major role of LIN28B is to block the processing from precursor to mature miRNAs of Let-7 family, which promote differentiation by downregulating growth-promoting genes, including c-MYC, RAS, IGF2BP1-3 and HMGA219. This evidence suggests that the oncogenic role of LIN28B involves the de-repression of pro-proliferative Let-7 targets.
In hematopoiesis, LIN28 expression is confined to fetal HSPCs cells. Its ectopic expression in adult cells promotes a fetal-like phenotype, characterized by active self-renewal and fetal-like lymphoid and myelo-erythroid differentiation20–23. LIN28B expression in hematopoietic adult cells represses Let-724, the most upregulated miRNAs family in adult versus fetal erythroblasts25. Interestingly, LIN28B overexpression is observed in specific subtypes of pediatric leukemia26, including juvenile myelomonocytic leukemias of fetal origin (JMML), characterized by high fetal hemoglobin levels and considered to arise from a stem-progenitor cell27, possibly of fetal origin24,28,29.
SOX6 is a member of the HMG box family of Transcription Factors, preferentially expressed in adult cells30. In hematopoiesis, SOX6 is expressed in quiescent LT-HSC31 and it acts as a tumor suppressor in chronic myeloid leukemia stem cells (CML-LSC)32. In erythropoiesis, it is expressed in adult cells, where it promotes differentiation33,34 and cooperates with the BCL11A-XL in the silencing of fetal γ-globin35.
SOX6 downregulation is observed in different human cancers, including acute myeloid leukemias36, Its low expression often correlates with poor prognosis. Restoring SOX6 expression mitigates the hyper-proliferative phenotype, indicating that it can act as tumor suppressor37–41.
Here we show that SOX6 represses LIN28B in different human fetal-like erythroid cell types and in myeloid leukemia cell lines. The repression of LIN28B upon SOX6 transduction results in the repression of Let-7 targets, including c-MYC, and in arrested cell growth.
We find that the repression of LIN28B by SOX6 also occurs in tumor cell types representative of other tissues, such as SH-SY5Y neuroblastoma and HepG2 hepatoblastoma cells, where LIN28B is highly expressed13,14. In all the contexts tested, the repression of LIN28B upon SOX6 overexpression invariantly results in decreased cell growth and proliferation. Finally, we identify SOX6 consensus binding sites within the LIN28B genomic locus. ChIP-qPCR and CUT&RUN experimentally validate SOX6 physical occupancy of these sites, supporting a direct transcriptional repression of LIN28B by SOX6.
Overall, our data reveal a mechanism by which SOX6 can act as a tumor suppressor in LIN28B-positive cancer cells.
Materials and methods
Cell and cell cultures
Cell lines, culture media, supplements and conditions are listed in Supplementary table 1.
CD1 mice were housed in our animal facility with 12 h light and dark cycles and free access to water and chow. Females 8–14 week-old were mated with males from the same strain and inspected daily for vaginal plugs. The appearance of the vaginal plug was designated as day 0.5 of pregnancy. At days 11.5, 12.5 and 13.5 pregnant females were sacrificed by cervical dislocation. N = 3–5 females were sacrificed for each time point and embryos from the same litter were pooled for subsequent analysis. Bone Marrow (BM) was harvested from the sacrificed pregnant females. Mouse experiments were conducted under the approval of the Italian Ministry of Health (protocol number 358-2016/PR), in accordance with European Union (86/609/EEC) and ARRIVE guidelines.
Data analysis from publicly available single-cell RNA sequencing datasets
The single-cell RNA sequencing (scRNAseq) datasets, encompassing human fetal liver and yolk sac from ref Popescu et al.42, human neuroblastoma from Kildisiute et al.43, and human hepatocellular carcinoma from Lu et al.44, were publicly available and underwent preprocessing as outlined in the Table 1. Subsequently, Anndata objects were analyzed using Python packages Scanpy45 (v.1.9.5) and Anndata (https://github.com/scverse/anndata) (v.0.9.2), inspired by Seurat46, to visualize the expression of genes of interest. Cluster assignment of cell types was achieved utilizing cell labels and marker genes sourced from the respective papers. For the yolk sac dataset, neighborhood graphs were constructed with 10 neighbors and 40 principal components (PCs), followed by dimension reduction to two dimensions using UMAP as recommended in the Scanpy clustering pipeline.
Table 1.
Single-cell RNA sequencing data sets and processing.
| Fetal liver | Yolk sac | Neuroblastoma | Hepatocellular carcinoma | |
|---|---|---|---|---|
| Species | Human | Human | Human | Human |
| Number of subjects | 14 | 3 | 16 | 10 |
| Cell count | 113,063 | 10,071 | 13,281 | 16,498 |
| Tissue /design | Fetal liver 7–17 PCW | 4–6 PCW | Mainly pre-treated tumors, viable tumor areas | Tumor and adjacent liver, primary and relapsed tumor |
| scRNAseq Kit | 10 × 3’ v2 | CEL-seq2 | 10 × 3’ v2 | |
| Preprocessing |
Cell ranger alignment on GRCh38 (STAR), Cells filtered for > 200 detected genes and total mtCount < 20%; Genes filtered for expressed in > 3 cells |
Cell ranger alignment on GRCh38 (STAR), Cells filtered for > 200 detected genes and > 500 UMIs and total mtCount < 20%; Genes filtered for not mtGenes and not hspGenes |
Cell Ranger alignment on GRCh38, Cells filtered for > 200 detected UMIs and > 200/ < 8000 detected genes and total mtCount < 10% |
|
| Normalization | By sequencing depth scale to 10,000 counts (NormalizeData, LogNormalize method), data feature scaling, variable gene detection, PCA, Louvain graph-based clustering with a resolution of 30 with standard parameters (fetal liver only, Seurat) | By sequencing depth scaled to 10 000 counts (NormalizeData, LogNormalize method), Scaling, variable gene detection (most 2000), PCA of 2000 most variable genes, 50 first PCs were used to calculate a UMAP (resolution parameter 1) (Seurat) |
Seurat standard normalization pipeline, Shared-nearest neighbor clustering obtained a final of 53 clusters that were used to calculate a UMAP (Resolution parameter 3) (Seurat) |
|
| Download source | https://developmental.cellatlas.io/fetal-liver | http://neuroblastomacellatlas.org | http://omic.tech/scrna-hcc/ | |
| Download format | Anndata .h5ad format containing count matrix and metadata | Anndata .h5ad format count matrix and metadata | Anndata .h5ad format count matrix and metadata | |
| Reference | Popescu et al.42 (Nature) | Kildisiute et al.43 (Science Advances) | Lu et al.44 (Nature Communications) | |
| Raw data | E-MTAB-7407 (Array Express) | EGAD00001008345 | EGAC00001001616 | |
RNA isolation and real time PCR
Total RNA from ≥ 105 cells were purified with TRIzol Reagent (Euroclone) and retrotranscribed (High Capacity cDNA Reverse Transcription Kit, Applied Biosystem). RT-PCR analysis was performed using StepOne (Thermofisher). Specific PCR product accumulation was monitored by using SsoAdvanced™ Universal SYBR® Green Supermix (Bio-Rad) fuorescent dye in 12 μl reaction volume. Dissociation curves confirmed the homogeneity of PCR products. Primers, designed to amplify 150 to 300bp amplicons, are listed in the Supplementary table 3. Each experiment was done in three biological replicates and cDNAs from each replicate were analysed in technical triplicates.
Protein extracts
For whole protein extracts, cells were resuspended in RIPA buffer (20 mM Tris HCl pH 7.4, 137 mM NaCl, 10% glycerol, 0.1% SDS, 0.5% deoxycolate, 1% Triton X-100, 2mM EDTA and proteases inhibitor cocktail). Lysis was performed for 30 min in ice and, after centrifugation (15 min at 16,000×g at 4 °C) the supernatant was collected and analysed.
Immunoblotting
Protein extracts were resolved by SDS/PAGE in a 10% acrylamide gel and blotted onto Hybond-ECL Nitrocellulose membrane (GE healthcare) at 400 mA for 90 min at 4 °C (Biorad Transblot apparatus). Membranes were blocked for 1 h at RT with milk 5% in TBS-T (Tris Buffered Saline, pH 7.6 and 0,1% Tween20) and incubated with the appropriate primary antibody diluted in milk 5% TBS-T overnight at 4 °C. Membranes were washed in TBS-T and incubated with the appropriate HRP-conjugated secondary antibody (in milk 5% TBS-T) for 1 h at room temperature. Antibodies binding was detected by ECL (Millipore). The blots were cut prior to hybridisation with the indicated antibodies. Full size gels are shown in Supplementary Fig. 10.
Lentiviral particles production
The Sox6 murine cDNA was cloned in frame with a FLAG epitope immediately upstream to the IRES-eGFP cassette into the CSI vector, as in34. Packaging HEK-293T cells were transfected with the SOX6 expression vector (or the corresponding empty control Vector, EV) and with the psPAX2 and pMD-VSVG packaging vectors. 72 h upon transfection, viral pseudoparticles were concentrated by centrifugation at 4 °C. The viral pellet was resuspended in 1X PBS and aliquoted at − 80 °C. 72h upon lentiviral transduction (MOI ≥ 25) GFP + cells were scored by Flow Cytometry (FC) (CytoFlex, Beckman Coulter). For HUDEP1 and HepG2 cells where GFP positivity was about 60–70%, cells were sorted (BD FACSMelody, BD Biosciences) before further analysis. All experiments were performed on cells ≥ 90% GFP + (Supplementary Fig. 1).
CUT&RUN analysis
The detailed protocol for CUT&RUN on HUDEP1 overexpressing SOX6-Flag and for data analysis are detailed in the Supplementary file material. Sox6 peaks are listed in suppl. table 4. The CUT&RUN datasets (raw and processed files) have been deposited at ArrayExpress (https://www.ebi.ac.uk/arrayexpress/) under accession number E-MTAB-12800.
Chromatin IP
HEL cells overexpressing SOX6 were fixed with 0.4% formaldehyde for 10 min at RT. Chromatin was sonicated (Bioruptor®, Diagenode) to the size of ≈500bp. DNA immunoprecipitation was obtained by incubation with the appropriate antibodies and subsequent isolation with protein A-agarose beads (Upstate). Immunoprecipitated DNA fragments were analysed by qPCR for the regions of interest (primers are listed in Supplementary Table 3). Full size gels are shown in Supplementary Fig. 9.
Primers, antibodies and reagents
Antibodies, reagents and primers are listed in the Supplementary Tables 2 and 3.
Statistical analysis
All experiments were performed on a least of n ≥ 3 biological replicates, each analysed in technical triplicates. For Western Blots, statistics was calculated on densitometry values from 3 independent experiments obtained by using ImageJ software. Statistical analysis was performed by using GraphPad Prism version 8.0.0 for Windows, GraphPad Software (www.graphpad.com). Data were analysed by using a two-tailed unpaired t-test and are expressed as mean ± SEM (standard error of mean). In Figures, error bars represent SEM and p-values are represented as follows: *p < 0.05; **p < 0.01; ***p < 0.001. The numerical values are in Figure legends.
Figures were prepared in compliance with the Scientific Reports digital image and integrity policies.
Results
Sox6 and Lin28B show opposite expression profiles during the fetal to adult transition in erythroid cells
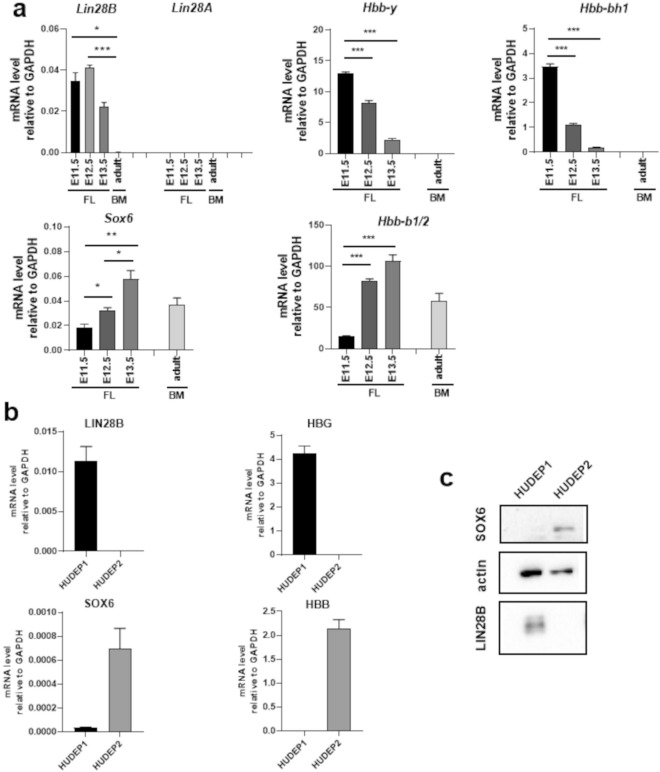
Erythropoiesis represents a model system of developmental dynamics. In particular, in the mouse fetal liver (FL), the E11.5-E13.5 timeframe corresponds to the transition from embryo/fetal to adult erythropoiesis, marked by the switching form embryo/fetal to adult globin genes expression (Fig. 1a). RT-qPCR shows that, within this developmental window, the expression of Sox6 and Lin28B inversely correlate: the highest expression of Sox6 at E13.5 coincides with the drop in Lin28B and with the establishment of the adult pattern of globin expression. Consistently, in adult bone marrow (BM) erythropoiesis where Sox6 expression persists throughout adult life, Lin28B is absent (Fig. 1a). Lin28B and Sox6 expression profiles, both at the mRNA and protein level, also inversely correlate in human HUDEP1 and HUDEP2 cells47, representative of embryo/fetal and adult erythropoiesis, respectively (Fig. 1b and c). The Lin28B paralog, Lin28A, is not expressed (Fig. 1a).
Figure 1.
Expression levels of LIN28 and SOX6 in erythropoiesis during development. (a) RT-qPCR: Sox6, Lin28B and globins expression relative to GAPDH in E11.5-E13.5 mouse fetal liver (FL) and in adult bone marrow (BM). The decline in Lin28B and the opposite increase in Sox6 parallel the transition (hemoglobin switching) from embryo/fetal (Hbb-y and Hbb-bh1) to adult (Hbb-b1/2) mouse globins. In the Figure, * = p < 0.05; ** = p < 0.01; *** = p < 0.001. Lin28B and Sox6 fold changes calculated on mean values: Lin28B fold reduction E12.5-E13.5 ≈45% p = 0.0003, Sox6 fold increase E12.5-E13.5 ≈45% p = 0.012. (b) RT-qPCR and Western Blot showing the opposite expression of LIN28B and SOX6 and human fetal γ (HBG) and adult β (HBB) globins in human fetal-like HUDEP1 and adult HUDEP2 cells. (c) Western blot showing the relative protein levels.
SOX6 and LIN28B/Let-7 downstream genes are expressed in non-overlapping cell populations during human embryo/fetal hematopoiesis
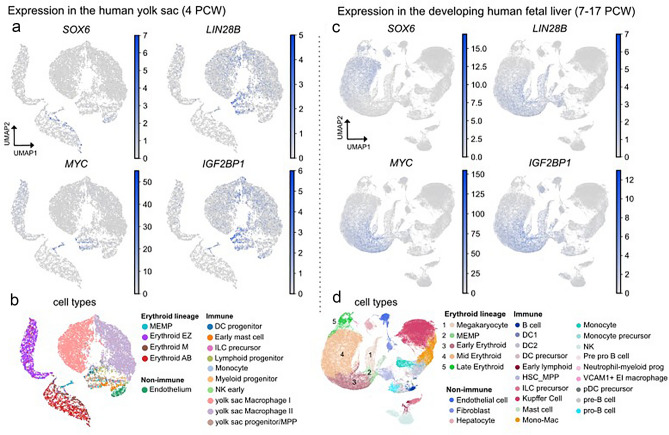
To investigate the relation between SOX6 and LIN28B expression in human early development at a single-cell resolution, we analyzed single-cell RNA sequencing (scRNA-seq) datasets obtained from yolk-sac (4 PCW, post conception weeks) and fetal liver (7 to 17 PCW) cells42. The integrated data analysis at these stages shows that the expression of SOX6 and LIN28B is confined to distinct cell populations (Fig. 2 and Supplementary Fig. 2): whereas LIN28B and the LIN28B/Let-7 downstream targets MYC and IGF2BP1 are prominently expressed in early erythroid progenitors (MEMP), SOX6 raises in mid-late erythroid cells and this is paralleled by loss of LIN28B (together with MYC and IGF2BP1). Complementing the data in Fig. 1, the expression pattern of SOX6 and LIN28B during human fetal erythropoiesis at a single cell level strongly suggests a potential switch-off mechanism of LIN28B transcription by SOX6.
Figure 2.
Single-cell RNA expression of the SOX6-LIN28B/Let-7 axis in human fetal hematopoiesis. (a) Normalized expression of SOX6, LIN28B, as well as the downstream genes MYC, and IGF2BP1 in human yolk sac cells shown as UMAP. (b) 15 UMAP clusters are associated with cell types according to the expression of distinct marker genes from Popescu et al.42. Dataset contains 10,071 yolk sac cells derived from 3 individuals at the age of 4–6 PCW. (c) Normalized expression of SOX6, LIN28B, as well as the downstream genes MYC, and IGF2BP1 in human fetal liver cells shown as UMAP (Uniform Manifold Approximation and Projection) plots. (d) 27 UMAP clusters are associated with cell types according to the expression of distinct marker genes. Dataset contains 113,063 fetal liver cells derived from 16 individuals at the age of 7–17 PCW. PCW, post conception week; MEMP, megakaryocyte-erythroid progenitors; DC, dendritic cells; HSC-MPP, hematopoietic stem cells and multipotent progenitors; ILS, induced leucocyte stem; Erythroid EZ, early (HGE expressing) erythroid; Erythoid M, mid erythroid; Erythroid AB, late (HGA and HGB expressing) erythroid.
SOX6 represses Lin28B blocking the LIN28B/Let-7 axis and cell proliferation in different erythroid cellular models
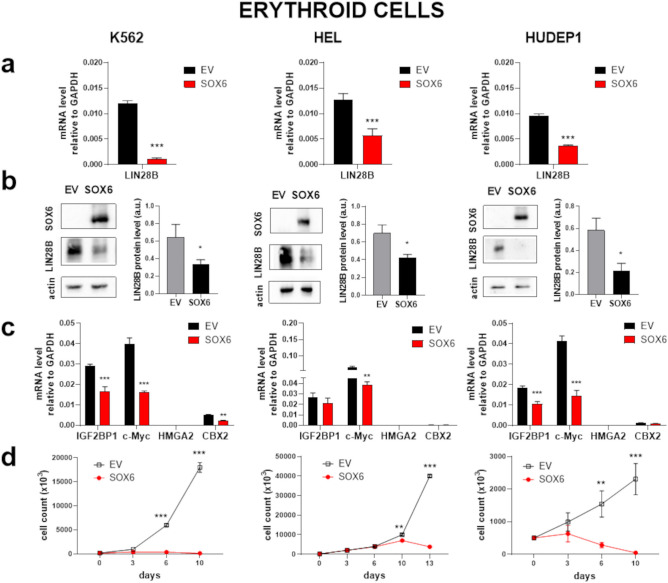
The LIN28B/Let-7 axis, which represents the main LIN28B downstream pathway, ultimately results in the control of proproliferative genes, including MYC and IGF2BP119. Our observations (Figs. 1 and 2) suggest that SOX6 may interfere on the LIN28B/Let-7 axis. To test if SOX6 is causally involved in the transcriptional regulation of LIN28B, we overexpressed it and monitor how LIN28B mRNA expression is affected as a consequence. To this aim, we selected three different “fetal-like” human erythroid cells lines: K562 and HEL erythroleukemias and HUDEP1, which model human embryonic erythropoiesis (Supplementary Fig. 3).
We infected the above cell lines with viral pseudoparticles carrying a SOX6Flag-IRES-GFP overexpressing vector (SOX6) and, in parallel, with the corresponding empty control vector (EV), (Supplementary Fig. 1). Upon SOX6 overexpression, LIN28B is significantly downregulated in all the tested cell lines, both at the mRNA and at the protein level (Fig. 3a, b). In order to evaluate the secondary impact of LIN28B repression on the LIN28B/Let-7 axis, we selected different known Let-7 target genes (Fig. 3c), including c-MYC, HMGA2, IGF2BP1 and CBX210,14,17. Among them, HMGA2 is not expressed in all the three cell lines, CBX2 is expressed at low level in K562 and HUDEP1 cells and c-MYC and IGF2BP1 are highly expressed in all three cell lines. IGF2BP1, which belongs to the family of the oncofetal IGF2 mRNA binding proteins, is highly expressed during embryogenesis and overexpressed in various tumors48,49. In erythropoiesis, IGF2BP1 is a major target of the Let-7 miRNA family1950, expressed in fetal erythroblasts25 and capable of inducing γ-globin expression51,52. As shown in Fig. 3c, c-MYC, IGF2BP1 and CBX2 (in K562) expression is reduced by SOX6 overexpression. The overexpression of SOX6 results in a marked decline in cell proliferation (Fig. 3d). We previously showed that SOX6 blocks cell proliferation in Set-2 and Uke1 myeloid cancer cells36. Here we show that SOX6 overexpression downregulates the LIN28B/Let-7 pathway also in these cells (Supplementary Fig. 4). Together, these data confirm the ability of SOX6 to repress LIN28B and the pro-proliferative genes downstream to the LIN28/Let-7 axis, pointing to a wide tumor suppressor role of SOX6.
Figure 3.
SOX6 represses LIN28B and blocks proliferation in erythroid cells. (a) Effect of SOX6 expression on LIN28B mRNA level. SOX6: cells transduced with the SOX6 overexpressing vector; Empty Vector (EV): cells transduced with the corresponding empty vector. Fold reduction calculated on the mean values: K562 ≈92% p < 0.0001, HEL ≈54% p = 0.0009, HUDEP1 ≈60% p < 0.0001. (b) Representative Western blots and quantification of the LIN28B reduction upon SOX6 overexpression at the protein level (densitometry analysis from 3 independent Western blot experiments was performed by using the ImageJ software). Fold reduction calculated on the mean values: K562 ≈48% p = 0.03, HEL ≈40% p = 0.0281, HUDEP1 ≈63% p = 0.046. (c and d) Effects of SOX6 expression on representative Let-7 downstream targets and on cell proliferation. Empty Vector (EV), in black: cells transduced with the empty vector. SOX6, in red: cells transduced with the SOX6 expressing vector. Fold reduction of targets calculated on the mean values: K562 IGF2BP1 ≈42% p = 0.0005, K562 c-MYC ≈60% p < 0.0001, K562 CBX2 ≈60% p = 0.0058, HEL, c-MYC ≈40% p = 0.0045. HUDEP1 IGF2BP1≈39% p = 0.0006, HUDEP1 c-MYC ≈64% p = 0.0008. For all RT-PCR shown, n ≥ 4, error bars: standard error of mean. *p < 0.05; **p < 0.01; ***p < 0.001.
LIN28B repression by SOX6 results in the repression of Let-7 targets in non-hematopoietic LIN28B-positive cancer cells
LIN28B is broadly expressed in early development and is reactivated in cancers17. We analyzed LIN28B expression in the large panel of comprehensively characterized Cancer Cell Line Encyclopedia (CCLE, Broad Institute, https://sites.broadinstitute.org/ccle/). Consistently with a broad role in cancer, LIN28B is found highly expressed in a wide spectrum of cancer cell lines, including myeloid, liver, and central nervous system cells, compared to other tissues (Supplementary Fig. 5a). Moreover, LIN28B is overexpressed in both primary as well as metastatic cell lines (Supplementary Fig. 5c).
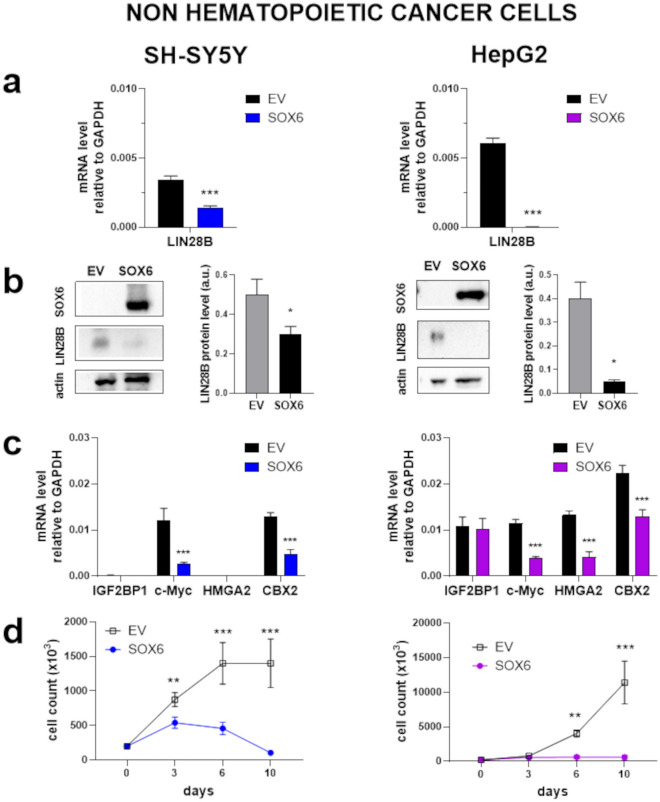
To test whether high LIN28B gene expression correlates with genetic dependency, we analyzed dependencies scores calculated by Chronos for LIN28B CRISPR in CCLE cell lines53. According to DepMap (https://depmap.org/portal/), LIN28B expression and dependency varied across cell lines. In particular, neuroblastoma cell lines, including SH-SY5Y, show high expression and significant dependency on LIN28B (Supplementary Fig. 5e, f). This is in line with the known involvement of LIN28B in high-risk neuroblastoma13,54. In addition, in neuroblastoma cell lines, SOX6 and LIN28B expression negatively correlate (Supplementary Fig. 5e, f). We therefore assessed the effect of SOX6 overexpression in human neuroblastoma SH-SY5Y cells (Fig. 4). To expand our observation, we performed the same analysis on human hepatic cancer HepG2 cells (Fig. 4) in which LIN28B was demonstrated by functional experiments to be essential for proliferation11,14.
Figure 4.
SOX6 represses LIN28B and blocks proliferation in neuroblastoma SH-SY5Y and hepatoblastoma HepG2 cells. The same analysis as in Fig. 3 was carried out in two non- hematopoietic cancer cells whose growth is sustained by LIN28B. SOX6: cells transduced with the SOX6 overexpressing vector; Empty Vector (EV): cells transduced with the corresponding empty vector. (a) mRNA fold reduction calculated on the mean values: SH-SY5Y ≈59% p < 0.0001, HepG2 ≈99.7% p < 0.0001. (b) Protein fold reduction calculated on the mean values: SH-SY5Y ≈40% p < = 0.03 HepG2 ≈88% p < 0.02. (c) Fold reduction of targets calculated on the mean values: SH-SY5Y c-MYC ≈75% p < 0.0001, SH-SY5Y CBX2 ≈62% p = 0.0004, HepG2 c-MYC ≈63% p < 0.0001, HepG2 HMGA2≈70% p = 0.0008, HepG2 CBX2 ≈41% p = 0.0007. (d) Cell proliferation curves.
In both cell types, we observed that overexpression of SOX6 represses LIN28B and ultimately results in a marked reduction of proliferation, although with slightly different kinetics. In these cells, Let-7 targets are repressed downstream to SOX6 overexpression.
Finally, we examined single-cell RNA-seq datasets obtained from neuroblastoma and hepatocellular carcinoma human biopsies (Supplementary Fig. 6). In neuroblastoma, the subset of cells expressing SOX6 shows minimal overlap with those expressing LIN28B, supporting a SOX6 mediated LIN28B downregulation in this context (Supplementary Fig. 6a, b). In hepatocellular carcinomas, the relationship between SOX6 and LIN28B expression is less clear indicating more intricate regulatory dynamics (Supplementary Fig. 6c, d).
LIN28B is a direct transcriptional target of SOX6
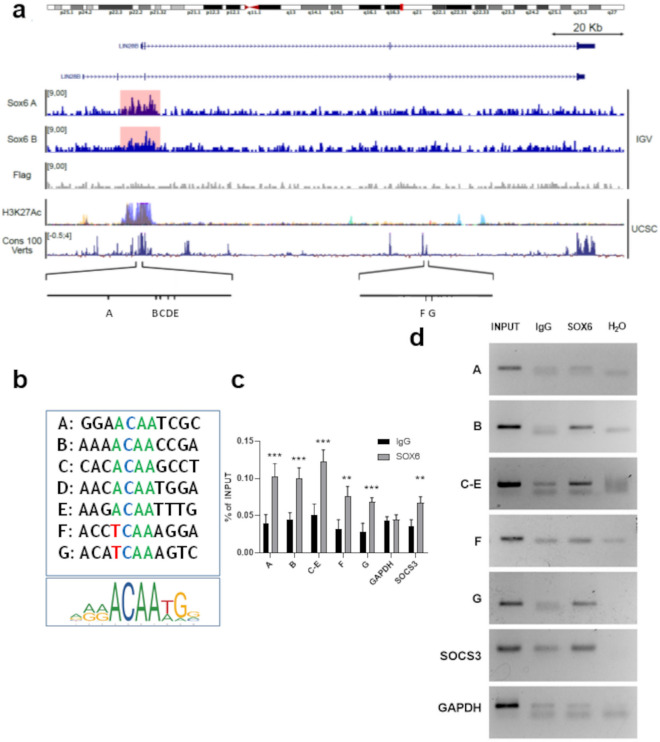
Our data points to LIN28B as a candidate gene transcriptionally repressed by SOX6. To test this hypothesis, we first searched for genomic binding sites of SOX6 across the LIN28B genomic locus. We found several SOX6 binding motifs within the LIN28B promoter and third intron (Fig. 5a and b, Supplementary Fig. 7). To test the ability of SOX6 to bind these sequences in vivo we performed a CUT&RUN analysis targeting the exogenously expressed SOX6-Flag in HUDEP1 (Supplementary Fig. 8). Untransduced cells were used as negative control. As shown in Fig. 5a, we identified reproducible signal enrichment in the vicinity of LIN28B transcriptional start site (TSS), in an evolutionarily conserved region enriched by H3K27Ac, a marker of active regulatory regions. This result constitutes robust evidence that SOX6 physically associates with this region (Fig. 5a).
Figure 5.
Identification of the SOX6 binding sites within the LIN28B locus. (a) Lin28B genomic locus visualized in Integrative Genomic Viewer (IGV) and with the UCSC genome browser (https://genome.ucsc.edu/). From the top: chromosome representation (IGV), gene transcripts (UCSC), two SOX6 CUT&RUN replicates and the respective negative Flag control in HUDEP1 cells (IGV), H3K27ac overlap (UCSC), vertebrate conservation (UCSC) and positions of the SOX6 binding sites within the promoter (A–E) and the third intron (F–G). (b) SOX6 sites compared with the SOX6 consensus Jaspar matrix 515.1. (c) Chromatin IP results obtained in HEL cells expressing SOX6. GAPDH locus: negative control; SOCS3 promoter: positive control34. Histograms represent the results of three biological replicates (n = 3, each of them in three technical replicates, as analyzed by RTqPCR (Error bars: standard error of mean; *p < 0.05; **p < 0.01; ***p < 0.001). (d) representative ChIP result. Antibodies are listed in the Supplementary table 2.
Targeted chromatin immunoprecipitation (ChIP) followed by qPCR in HEL cells (Fig. 5c and d) validates the CUT&RUN findings and provides evidence for multiple binding anchors for SOX6 within the LIN28B locus in different cell types, suggesting robust, potentially redundant regulatory mechanisms of LIN28B transcription repression by SOX6.
Discussion
LIN28B is a stemness-related oncofetal gene whose repression is required to acquire adult-specific fate in different systems20–23,53,54. Its reactivation is observed in several tumors17 and often coincide with the acquisition of fetal–like phenotypes23,24. Despite its broad role in development and cancer, the molecular basis of its transcriptional repression is largely unknown. In this work we identify the pro-differentiative, growth-restriction SOX6 “adult” transcription factor31,54 as a direct repressor of the LIN28B gene.
First we show that LIN28B and Sox6 expression is mutually exclusive in mouse and human erythropoiesis, which represents a well-defined model of development (Figs. 1 and 2) as well in several cancer cells (Figs. 3, 4 and Supplementary Figs. 5 and 6), in particular neuroblastoma and hepatocarcinoma cell lines. SOX6 binds in vivo to several consensus binding sites that we identified within the LIN28B locus (Fig. 5 and Supplementary Fig. 7), represses LIN28B transcription and cell proliferation (Figs. 3, 4).
At the molecular level, the LIN28B oncogenic potential largely relies in its ability to repress the processing of miRNAs of the Let-7 family, which in adult cells promote differentiation and repress pro-proliferative genes17. We demonstrate that SOX6 impacts on the expression of genes downstream to the LIN28B/Let-7 axis (Figs. 2, 3, 4), such as c-MYC and IGF2BP1, confirming the involvement of this pathway. Based on our observations, we propose a general model in which SOX6 acts as a tumor suppressor in cells of different tissue origin acting through the LIN28B/Let-7 pathway (Fig. 6).
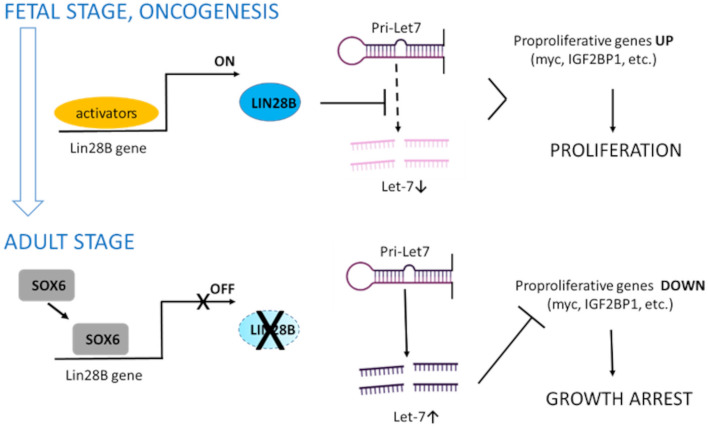
Figure 6.
Proposed model of LIN28B repression by SOX6. In fetal cells and in cancers with illegitimate LIN28B reactivation, LIN28B blocks the processing of Let-7 precursors to Let-7 miRNA. Let-7 reduction leads to the failure of silencing of proproliferative genes, including c-MYC and IGF2BP1, thus promoting cell proliferation. SOX6, by repressing LIN28B transcription, allows the maturation of Let-7 miRNAs, among the most abundant miRNAs in adult cells17. Let-7 miRNAs post-transcriptionally repress proproliferative genes inducing growth arrest (created with Biorender).
We cannot exclude that additional molecular mechanisms/pathways downstream to SOX6, independent from LIN28B downregulation concur to the observed tumor suppressive outcome and it is also likely that reduction of LIN28B expression does not exclusively impact the Let-7 axis. Nevertheless, we believe that the genetic circuit involving the direct repression of LIN28B by SOX6 represents a novel layer of regulation with relevant therapeutic implications for those tumors relying on LIN28B for their survival, often associated with high-risk malignancy.
The identification of the mechanistic link between SOX6 and LIN28B may provide novel opportunities for tumors stratification and novel therapeutic options.
Supplementary Information
Acknowledgements
This work was supported by the European Union’s Horizon 2020 Research and Innovation Program under the Marie Skłodowska Curie grant agreement No. 813091 (A.E.R.), by MUR—Ministero dell’Università e della Ricerca (PRIN: 2021-NAZ-0253) (A.E.R.) and by the European Union—NextGenerationEU through the Italian Ministry of University and Research under PNRR-M4C2-I1.3 Project PE_00000019 "HEAL ITALIA" to Antonella Ronchi (H43C22000830006), from Cancerfonden (CAN 2018/542 and 21 1572 Pj) and the Swedish Research Council (Vetenskapsrådet; 2021-03075 and 2023-01898) to C.C.. The views and opinions expressed are those of the authors only and do not necessarily reflect those of the European Union or the European Commission. Neither the European Union nor the European Commission can be HELd responsible for them. C.C. is a Wallenberg Molecular Medicine (WCMM) fellow and receives generous financial support from the Knut and Alice Wallenberg Foundation. The computations and data handling were enabled by resources provided by the National Supercomputer Centre (NSC), funded by Linköping University. Peter Münger at the National Supercomputer Centre is acknowledged for assistance concerning technical and implementational aspects in making the codes run on the Sigma resource. The sequencing was carried out at the molecular biology unit of Linköping University's core facility. We thank Gloria Barbarani and Cristina Fugazza, Agata Labedz, Cristiana Barone and Emanuele Azzoni for experimental help and for sharing preliminary results and Silvia ML Barabino and John Strouboulis for reading, discussion and suggestions.
Author contributions
V.P., G.Z., T.W. and E.C. performed experiments and contributed to the conceptualization of this work. Y.N. provided HUDEP cells. C.C. and A.E.R. conceived the experiments and wrote the paper.
Data availability
All data have been deposited in ArrayExpress under accession number E-MTAB-12800.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Valentina Pastori and Gianluca Zambanini.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-60438-3.
References
- 1.Marshall GM, Carter DR, Cheung BB, Liu T, Mateos MK, Meyerowitz JG, et al. The prenatal origins of cancer. Nat. Rev. Cancer. 2014;14(4):277–289. doi: 10.1038/nrc3679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Moss EG, Lee RC, Ambros V. The cold shock domain protein LIN-28 controls developmental timing in C. elegans and is regulated by the lin-4 RNA. Cell. 1997;88(5):637–646. doi: 10.1016/S0092-8674(00)81906-6. [DOI] [PubMed] [Google Scholar]
- 3.Moss EG, Tang L. Conservation of the heterochronic regulator Lin-28, its developmental expression and microRNA complementary sites. Dev. Biol. 2003;258(2):432–442. doi: 10.1016/S0012-1606(03)00126-X. [DOI] [PubMed] [Google Scholar]
- 4.Guo Y, Chen Y, Ito H, Watanabe A, Ge X, Kodama T, et al. Identification and characterization of lin-28 homolog B (LIN28B) in human hepatocellular carcinoma. Gene. 2006;384:51–61. doi: 10.1016/j.gene.2006.07.011. [DOI] [PubMed] [Google Scholar]
- 5.Zhu H, Shah S, Shyh-Chang N, Shinoda G, Einhorn WS, Viswanathan SR, et al. Lin28a transgenic mice manifest size and puberty phenotypes identified in human genetic association studies. Nat. Genet. 2010;42(7):626–630. doi: 10.1038/ng.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.He C, Kraft P, Chen C, Buring JE, Pare G, Hankinson SE, et al. Genome-wide association studies identify loci associated with age at menarche and age at natural menopause. Nat. Genet. 2009;41(6):724–728. doi: 10.1038/ng.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ong KK, Elks CE, Li S, Zhao JH, Luan J, Andersen LB, et al. Genetic variation in LIN28B is associated with the timing of puberty. Nat. Genet. 2009;41(6):729–733. doi: 10.1038/ng.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sulem P, Gudbjartsson DF, Rafnar T, Holm H, Olafsdottir EJ, Olafsdottir GH, et al. Genome-wide association study identifies sequence variants on 6q21 associated with age at menarche. Nat. Genet. 2009;41(6):734–738. doi: 10.1038/ng.383. [DOI] [PubMed] [Google Scholar]
- 9.Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318(5858):1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- 10.Shyh-Chang N, Zhu H, Yvanka de Soysa T, Shinoda G, Seligson MT, Tsanov KM, et al. Lin8 enhances tissue repair by reprogramming cellular metabolism. Cell. 2013;155(4):778–792. doi: 10.1016/j.cell.2013.09.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nguyen LH, Robinton DA, Seligson MT, Wu L, Li L, Rakheja D, et al. Lin28b is sufficient to drive liver cancer and necessary for its maintenance in murine models. Cancer Cell. 2014;26(2):248–261. doi: 10.1016/j.ccr.2014.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhou J, Ng SB, Chng WJ. LIN28/LIN28B: an emerging oncogenic driver in cancer stem cells. Int. J. Biochem. Cell Biol. 2013;45(5):973–978. doi: 10.1016/j.biocel.2013.02.006. [DOI] [PubMed] [Google Scholar]
- 13.Molenaar JJ, Domingo-Fernandez R, Ebus ME, Lindner S, Koster J, Drabek K, et al. LIN28B induces neuroblastoma and enhances MYCN levels via let-7 suppression. Nat. Genet. 2012;44(11):1199–1206. doi: 10.1038/ng.2436. [DOI] [PubMed] [Google Scholar]
- 14.Viswanathan SR, Powers JT, Einhorn W, Hoshida Y, Ng TL, Toffanin S, et al. Lin28 promotes transformation and is associated with advanced human malignancies. Nat. Genet. 2009;41(7):843–848. doi: 10.1038/ng.392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou J, Chan ZL, Bi C, Lu X, Chong PS, Chooi JY, et al. LIN28B activation by PRL-3 Promotes leukemogenesis and a stem cell-like transcriptional program in AML. Mol. Cancer Res. 2017;15(3):294–303. doi: 10.1158/1541-7786.MCR-16-0275-T. [DOI] [PubMed] [Google Scholar]
- 16.Manier S, Powers JT, Sacco A, Glavey SV, Huynh D, Reagan MR, et al. The LIN28B/let-7 axis is a novel therapeutic pathway in multiple myeloma. Leukemia. 2017;31(4):853–860. doi: 10.1038/leu.2016.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Balzeau J, Menezes MR, Cao S, Hagan JP. The LIN28/let-7 pathway in cancer. Front. Genet. 2017;8:31. doi: 10.3389/fgene.2017.00031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gewalt T, Noh KW, Meder L. The role of LIN28B in tumor progression and metastasis in solid tumor entities. Oncol. Res. 2023;31(2):101–115. doi: 10.32604/or.2023.028105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Busch B, Bley N, Muller S, Glass M, Misiak D, Lederer M, et al. The oncogenic triangle of HMGA2, LIN28B and IGF2BP1 antagonizes tumor-suppressive actions of the let-7 family. Nucl. Acids Res. 2016;44(8):3845–3864. doi: 10.1093/nar/gkw099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Copley MR, Babovic S, Benz C, Knapp DJ, Beer PA, Kent DG, et al. The Lin28b-let-7-Hmga2 axis determines the higher self-renewal potential of fetal haematopoietic stem cells. Nat. Cell Biol. 2013;15(8):916–925. doi: 10.1038/ncb2783. [DOI] [PubMed] [Google Scholar]
- 21.Rowe RG, Wang LD, Coma S, Han A, Mathieu R, Pearson DS, et al. Developmental regulation of myeloerythroid progenitor function by the Lin28b-let-7-Hmga2 axis. J. Exp. Med. 2016;213(8):1497–1512. doi: 10.1084/jem.20151912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang D, Tanaka-Yano M, Meader E, Kinney MA, Morris V, Lummertz da Rocha E, et al. Developmental maturation of the hematopoietic system controlled by a Lin28b-let-7-Cbx2 axis. Cell Rep. 2022;39(1):110587. doi: 10.1016/j.celrep.2022.110587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yuan J, Nguyen CK, Liu X, Kanellopoulou C, Muljo SA. Lin28b reprograms adult bone marrow hematopoietic progenitors to mediate fetal-like lymphopoiesis. Science. 2012;335(6073):1195–1200. doi: 10.1126/science.1216557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee YT, de Vasconcellos JF, Yuan J, Byrnes C, Noh SJ, Meier ER, et al. LIN28B-mediated expression of fetal hemoglobin and production of fetal-like erythrocytes from adult human erythroblasts ex vivo. Blood. 2013;122(6):1034–1041. doi: 10.1182/blood-2012-12-472308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lessard S, Beaudoin M, Orkin SH, Bauer DE, Lettre G. 14q32 and let-7 microRNAs regulate transcriptional networks in fetal and adult human erythroblasts. Hum. Mol. Genet. 2018;27(8):1411–1420. doi: 10.1093/hmg/ddy051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Helsmoortel HH, De Moerloose B, Pieters T, Ghazavi F, Bresolin S, Cave H, et al. LIN28B is over-expressed in specific subtypes of pediatric leukemia and regulates lncRNA H19. Haematologica. 2016;101(6):e240–e244. doi: 10.3324/haematol.2016.143818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cooper LJ, Shannon KM, Loken MR, Weaver M, Stephens K, Sievers EL. Evidence that juvenile myelomonocytic leukemia can arise from a pluripotential stem cell. Blood. 2000;96(6):2310–2313. doi: 10.1182/blood.V96.6.2310. [DOI] [PubMed] [Google Scholar]
- 28.Stieglitz E, Taylor-Weiner AN, Chang TY, Gelston LC, Wang YD, Mazor T, et al. The genomic landscape of juvenile myelomonocytic leukemia. Nat. Genet. 2015;47(11):1326–1333. doi: 10.1038/ng.3400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Helsmoortel HH, Bresolin S, Lammens T, Cave H, Noellke P, Caye A, et al. LIN28B overexpression defines a novel fetal-like subgroup of juvenile myelomonocytic leukemia. Blood. 2016;127(9):1163–1172. doi: 10.1182/blood-2015-09-667808. [DOI] [PubMed] [Google Scholar]
- 30.Hagiwara N. Sox6, jack of all trades: A versatile regulatory protein in vertebrate development. Dev. Dyn. 2011;240(6):1311–1321. doi: 10.1002/dvdy.22639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Forsberg EC, Prohaska SS, Katzman S, Heffner GC, Stuart JM, Weissman IL. Differential expression of novel potential regulators in hematopoietic stem cells. PLoS Genet. 2005;1(3):e28. doi: 10.1371/journal.pgen.0010028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhou M, Zhang X, Liu C, Nie D, Li S, Lai P, et al. Targeting protein lysine methyltransferase G9A impairs self-renewal of chronic myelogenous leukemia stem cells via upregulation of SOX6. Oncogene. 2021;40(20):3564–3577. doi: 10.1038/s41388-021-01799-1. [DOI] [PubMed] [Google Scholar]
- 33.Dumitriu B, Patrick MR, Petschek JP, Cherukuri S, Klingmuller U, Fox PL, et al. Sox6 cell-autonomously stimulates erythroid cell survival, proliferation, and terminal maturation and is thereby an important enhancer of definitive erythropoiesis during mouse development. Blood. 2006;108(4):1198–1207. doi: 10.1182/blood-2006-02-004184. [DOI] [PubMed] [Google Scholar]
- 34.Cantu C, Ierardi R, Alborelli I, Fugazza C, Cassinelli L, Piconese S, et al. Sox6 enhances erythroid differentiation in human erythroid progenitors. Blood. 2011;117(13):3669–3679. doi: 10.1182/blood-2010-04-282350. [DOI] [PubMed] [Google Scholar]
- 35.Xu J, Sankaran VG, Ni M, Menne TF, Puram RV, Kim W, et al. Transcriptional silencing of {gamma}-globin by BCL11A involves long-range interactions and cooperation with SOX6. Genes Dev. 2010;24(8):783–798. doi: 10.1101/gad.1897310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Barbarani G, Fugazza C, Barabino SML, Ronchi AE. SOX6 blocks the proliferation of BCR-ABL1(+) and JAK2V617F(+) leukemic cells. Sci. Rep. 2019;9(1):3388. doi: 10.1038/s41598-019-39926-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Qin YR, Tang H, Xie F, Liu H, Zhu Y, Ai J, et al. Characterization of tumor-suppressive function of SOX6 in human esophageal squamous cell carcinoma. Clin. Cancer Res. 2011;17(1):46–55. doi: 10.1158/1078-0432.CCR-10-1155. [DOI] [PubMed] [Google Scholar]
- 38.Xie Q, Chen X, Lu F, Zhang T, Hao M, Wang Y, et al. Aberrant expression of microRNA 155 may accelerate cell proliferation by targeting sex-determining region Y box 6 in hepatocellular carcinoma. Cancer. 2012;118(9):2431–2442. doi: 10.1002/cncr.26566. [DOI] [PubMed] [Google Scholar]
- 39.Guo X, Yang M, Gu H, Zhao J, Zou L. Decreased expression of SOX6 confers a poor prognosis in hepatocellular carcinoma. Cancer Epidemiol. 2013;37(5):732–736. doi: 10.1016/j.canep.2013.05.002. [DOI] [PubMed] [Google Scholar]
- 40.Li Y, Xiao M, Guo F. The role of Sox6 and Netrin-1 in ovarian cancer cell growth, invasiveness, and angiogenesis. Tumour Biol. 2017;39(5):1010428317705508. doi: 10.1177/1010428317705508. [DOI] [PubMed] [Google Scholar]
- 41.Jiang W, Yuan Q, Jiang Y, Huang L, Chen C, Hu G, et al. Identification of Sox6 as a regulator of pancreatic cancer development. J. Cell Mol. Med. 2018;22(3):1864–1872. doi: 10.1111/jcmm.13470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Popescu DM, Botting RA, Stephenson E, Green K, Webb S, Jardine L, et al. Decoding human fetal liver haematopoiesis. Nature. 2019;574(7778):365–371. doi: 10.1038/s41586-019-1652-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kildisiute G, Kholosy WM, Young MD, Roberts K, Elmentaite R, van Hooff SR, et al. Tumor to normal single-cell mRNA comparisons reveal a pan-neuroblastoma cancer cell. Sci. Adv. 2021;7(6):eabd3311. doi: 10.1126/sciadv.abd3311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lu Y, Yang A, Quan C, Pan Y, Zhang H, Li Y, et al. A single-cell atlas of the multicellular ecosystem of primary and metastatic hepatocellular carcinoma. Nat. Commun. 2022;13(1):4594. doi: 10.1038/s41467-022-32283-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wolf FA, Angerer P, Theis FJ. SCANPY: Large-scale single-cell gene expression data analysis. Genome Biol. 2018;19(1):15. doi: 10.1186/s13059-017-1382-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Butler A, Hoffman P, Smibert P, Papalexi E, Satija R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 2018;36(5):411–420. doi: 10.1038/nbt.4096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kurita R, Suda N, Sudo K, Miharada K, Hiroyama T, Miyoshi H, et al. Establishment of immortalized human erythroid progenitor cell lines able to produce enucleated red blood cells. PLoS One. 2013;8(3):e59890. doi: 10.1371/journal.pone.0059890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lederer M, Bley N, Schleifer C, Huttelmaier S. The role of the oncofetal IGF2 mRNA-binding protein 3 (IGF2BP3) in cancer. Semin. Cancer Biol. 2014;29:3–12. doi: 10.1016/j.semcancer.2014.07.006. [DOI] [PubMed] [Google Scholar]
- 49.Hammer NA, Hansen T, Byskov AG, Rajpert-De Meyts E, Grondahl ML, Bredkjaer HE, et al. Expression of IGF-II mRNA-binding proteins (IMPs) in gonads and testicular cancer. Reproduction. 2005;130(2):203–212. doi: 10.1530/rep.1.00664. [DOI] [PubMed] [Google Scholar]
- 50.Zhou J, Bi C, Ching YQ, Chooi JY, Lu X, Quah JY, et al. Inhibition of LIN28B impairs leukemia cell growth and metabolism in acute myeloid leukemia. J. Hematol. Oncol. 2017;10(1):138. doi: 10.1186/s13045-017-0507-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chambers CB, Gross J, Pratt K, Guo X, Byrnes C, Lee YT, et al. The mRNA-binding protein IGF2BP1 restores fetal hemoglobin in cultured erythroid cells from patients with beta-hemoglobin disorders. Mol. Ther. Methods Clin. Dev. 2020;17:429–440. doi: 10.1016/j.omtm.2020.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.de Vasconcellos JF, Tumburu L, Byrnes C, Lee YT, Xu PC, Li M, et al. IGF2BP1 overexpression causes fetal-like hemoglobin expression patterns in cultured human adult erythroblasts. Proc. Natl. Acad. Sci. USA. 2017;114(28):E5664–E5672. doi: 10.1073/pnas.1609552114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li Y, Kong W, Yang W, Patel RM, Casey EB, Okeyo-Owuor T, et al. Single-cell analysis of neonatal HSC ontogeny reveals gradual and uncoordinated transcriptional reprogramming that begins before birth. Cell Stem Cell. 2020;27(5):732–747e7. doi: 10.1016/j.stem.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Li Y, Magee JA. Transcriptional reprogramming in neonatal hematopoietic stem and progenitor cells. Exp. Hematol. 2021;101–102:25–33. doi: 10.1016/j.exphem.2021.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data have been deposited in ArrayExpress under accession number E-MTAB-12800.