Abstract
Aging and aging-related diseases present a global public health problem. Therefore, the development of efficient anti-aging drugs has become an important area of research. Traditional Chinese medicine is an important complementary and alternative branch of aging-related diseases therapy. Recently, a growing number of studies have revealed that traditional Chinese medicine has a certain delaying effect on the progression of aging and aging-related diseases. Here, we review the progress in research into using traditional Chinese medicine for aging and aging-related diseases (including neurodegenerative diseases, cardiovascular diseases, diabetes, and cancer). Furthermore, we summarize the potential mechanisms of action of traditional Chinese medicine and provide references for further studies on aging and aging-related diseases.
Keywords: aging and aging-related diseases, traditional Chinese medicine, potential mechanisms
Introduction
Advances in antibiotics, medicine, hygiene, housing, and lifestyle and economic development have led to an increase in human life expectancy. Aging has come to the fore and represents a worldwide focus in the life sciences. By 2050, the proportion of the world’s population aged over 60 will double to 22%, and 80% of older people will be living in low- and middle-income countries.1 All countries face major challenges to ensure that their health and social systems are adequately prepared for this demographic shift. Aging results from the effects of various types of molecular and cellular damage that accumulate over time, leading to a gradual decline in physical and mental abilities, an increased risk of disease, and eventually to death. Aging-Related Diseases (ARDs) affect human health and life, bring with them physical, psychological and economic burden, not only for older people and their families, but also for societies. However, the pathological mechanisms of ARDs have not been adequately elucidated, which greatly limits the efficacy of available treatment for these diseases in clinical practice.
Traditional Chinese medicine (TCM) has a profound theoretical basis and deep cultural heritage. Under the guidance of TCM theory, herbal medicines have been used effectively to combat disease and save lives. TCM was described as a cure for aging for the first time over 3000 years ago in Huangdi Neijing, in which the concept of health preservation was described. A classical TCM book named Shanghan Lun, written by Zhang Zhongjing, describes applications of Shenqi Wan for anti-aging. Japanese Kampo medicine advocates for Shenqi Wan as an “anti-aging recipe” for treating senile diseases that has achieved effective treatment results.2
TCM formulations used against aging feature multiple components, multiple targets and multiple pathways. Herbal medicines such as quercetin and resveratrol have been shown to extend the lifespan of model organisms.3 Sijunzi decoction has an anti-aging effect and can reduce the effects of aging of the spleen and brain in mice through the mTOR pathway.4,5 Shenqi pills protect nerves and improve memory by improving energy metabolism, reducing oxidative damage, and increasing the number and morphology of nerve cells.6 Meanwhile, Shenqi can alleviate the damage of tight junctions in senescent sertoli cells via a mechanism potentially related to the down-regulation of the p38MAPK signaling pathway.7 Notably, recent studies have shown that TCM can stimulate the oxidative stress response and attenuate the aging process,8,9 suggesting that TCM may be strongly associated with anti-aging and making the impact of TCM on aging and ARDs a promising and far-reaching topic. In this review, we discuss the close relationship between herbal medicine and ARDs by the effects of herbal medicine in anti-aging and ARDs, and the mechanisms underlying the role of herb medicine in IRDs.
TCM and Aging
Preliminary evidence from preclinical studies has revealed the anti-aging potential of TCM. Chinese herbal preparations delay intestinal aging through three pathways: anti-inflammatory, improving intestinal metabolites, and intestinal flora, which may be related to the decrease of telomerase mRNA in elderly mice.10 Herb extracts increased activity during aging and improved heart function, while increase the healthy lifespan of aging flies.11 TCM can ameliorate age-related reductions in brain performance, including memory, cognitive, and motor functions, via the preservation of redox homeostasis and telomere integrity, and inhibition of apoptosis.12 NF-κB activation is associated with age-associated pathologies and symptoms.13 Curcumin, a polyphenolic compound isolated from Curcuma longa, has been shown to prolong the mean lifespan of some aging model organisms. Curcumin has been proposed to improve immune cell chemotaxis, inhibit NF-κB signaling, and have antioxidative and anti-inflammatory biological activity.14 This implies that TCM may exert beneficial regulatory effects on immune function during aging. Additionally, mulberry leaves have been identified to increase the lifespan of Caenorhabditis elegans.8 These studies collectively support the anti-aging potential of TCM and hold the potential for better treatment regimens in the battle against aging.
TCM and Age-Related Diseases
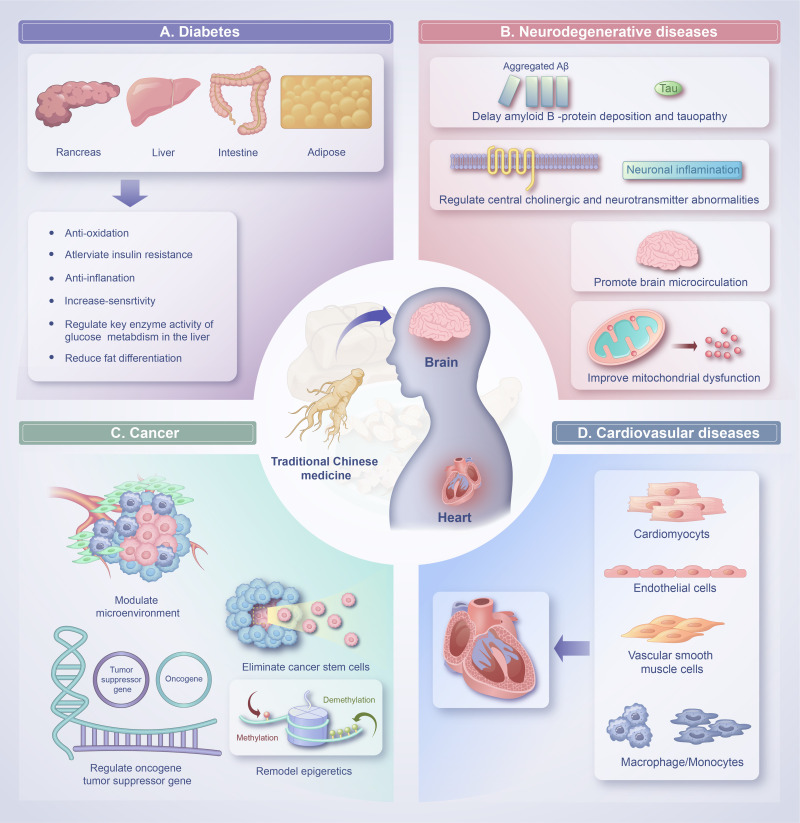
Common conditions associated with aging include diabetes, cognitive impairment, chronic obstructive pulmonary disease, diabetes, depression, dementia and cancer.1,8 As people age, they are more likely to experience several conditions at the same time. Despite ongoing research and progress in the field, cures for such chronic afflictions have not yet been found. Herbs have massive therapeutic potential in treating common conditions associated with aging and the mechanisms of action are shown in Figure 1.
Figure 1.
The effects of traditional Chinese medicine (TCM) with regards to the improvement of common conditions associated with aging and the mechanisms of action. (A) The mechanism of TCM in the treatment of diabetes is related to anti-oxidation, alleviate insulin resistance, anti-inflammation, increase insulin sensitivity, regulate key enzyme activity of glucose metabolism in the liver, reduce fat differentiation. (B) TCM can delay amyloid β-protein deposition and tauopathy, regulate central cholinergic and other neurotransmitter abnormalities, promote brain microcirculation, improve mitochondrial dysfunction. (C) Application of TCM in cancer therapy based on regulate genes (oncogenes and tumor suppressor genes), epigenetic modification (DNA and histone modification), the microenvironment, and cancer stem cells. (D) TCM have a protective effect on cardiovascular diseases by attenuate damage in cardiomyocytes, endothelial cells, vascular smooth muscle cells and macrophages/monocytes.
TCM and Neurodegenerative Diseases
As lifespans are increasing, aging is most prevalent risk factor for neurodegenerative diseases and their associated cognitive dysfunction.15 Alzheimer’s disease (AD), the most common cause of dementia, has become a global health threat to older adults. In the United States, an estimated 6.2 million people aged ≥65 years are living with Alzheimer’s disease while adults aged >70 years will experience a rapid increase of dementia; this number could grow to 13.8 million by 2060 and has become the fifth leading cause of death.16,17 Globally, AD is the leading cause of cognitive impairment, with Parkinson’s disease (PD) being second, seriously affecting the quality of life of the elderly.18,19 Amyotrophic lateral sclerosis (ALS) is an age-related neurodegenerative disease that increases in prevalence and incidence with age until the age of 70–79 years.20,21 However, effective treatment strategies for age-related degeneration are lacking, leading to its irreversible progression.
At present, there are no drugs that can effectively prevent the pathological progression of AD.22 Evidence has been presented that TCM contains multiple active ingredients that could act on multiple targets simultaneously and have good clinical efficacy in the treatment of AD.23 For example, oral administration of Polygoni Multiflori Radix Praeparata (ZhiHeShouWu) and Acori Tatarinowii Rhizoma (ShiChangPu) or their traditional combination has been reported to enhance synaptic-related proteins in mice, thereby offering the potential to alleviate cognitive deficits in AD patients.24 Chinese herbal extracts demonstrated similar alleviative effects in an AD-like rat model induced with scopolamine, which may be through modulation of the dopaminergic synapse and apoptosis signaling pathway.25 Additionally, the potential effects of TCM on ALS were explored in another study; TCM can prolong the lifespan, extend the disease duration, and prevent the loss of spinal motor neurons in mice with ALS, which was associated with reduction in atrophy of gastrocnemius muscles and improved levels of apoptosis and inflammation.26 Meanwhile, TCM can delay the decline of motor function in ALS-model mice by alleviating the degeneration of neurons.27 The results of preclinical tests and a previous clinical study support TCM as a novel therapeutic approach for AD and ALS patients. However, the potential of TCM to treat ALS should be assessed in a clinical trial.
The pathogenesis of PD is complex and is characterized as the progressive loss of dopaminergic neurons in the substantia nigra pars compacta. Oxidative stress, defective autophagy-lysosomal pathway, mitochondrial dysfunction, endoplasmic reticulum stress, microglia reactivity and neuroinflammation are recognized to be features of PD.28 However, no currently used therapeutic agent is known to slow down neuronal cell loss.
PD is closely related to mitochondrial dysfunction, which is a key factor in the occurrence and development of dopaminergic degeneration.29 A study explored the mechanisms of huangqin decoction on attenuation of mitochondrial dysfunction, finding that huangqin decoction improved mitochondrial dysfunction, alleviated metabolic abnormality of mitochondria, and improved motor coordination and muscle strength in a rotenone-induced mice model of PD.30 Andrographolide (andro) is a diterpenoid lactone isolated from Andrographis paniculata. Administration of andrographolide improved behavioral deficits and attenuated loss of dopaminergic neurons in mice with MPTP-induced PD via targeting mitochondrial fission mediated by dynamin-related protein 1.31 Duzhong fang is a TCM formula that can alleviate neuroinflammation and regulate microglia morphology and reactivity by downregulating POMC expression in PD mice model.32 Sinomenine, the main component of alkaloids, can augment the autophagy of dopaminergic neurons via inhibiting the PI3K/AKT/mTOR pathway and exerts a neuroprotective effect in PD mice.33 Zishenpingchan granules can alleviate dopaminergic neuron apoptosis in PD mice induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine by suppressing the over-activation of the c-Jun N-terminal protein kinase pathway in the substantia nigra. In addition, moxibustion can alleviate oxidative stress injury of the nigrostriatal system in mice with PD by activating the Nrf2/ARE pathway and protecting the dopamine neurons.34 In a multicenter, randomized, double-blind, placebo-controlled study, 292 participants were included and followed for 9 months, the results showed that the TCM-treated group had significantly better improvement than participants in the control group in Movement Disorder Society Unified Parkinson’s Rating Scale III motor score and its domain scores for axial symptoms, bradykinesia, rigidity, and tremor, indicating that TCM might be a viable alternative in the management of PD.35
TCM can protect dopaminergic neurons via multiple pathways. At present, great progress has been made in research into the mechanisms of TCM in the prevention and treatment of PD. However, it seems premature to conclude that TCM is associated with protective effects against deterioration of PD. There are several concerns that remain unresolved. First, the pathogenesis of PD is complex, which is the result of the mutual influence and interaction of multiple pathogenic factors, and the therapeutic advantage of TCM lies in the role of multiple components affecting multiple targets. Related studies focus on one or two molecular mechanisms, and rarely discuss the interactions between multiple molecular mechanisms from a multi-dimensional perspective. Second, in studies exploring the improvement of PD by TCM, the mechanisms of motor symptoms in model animals rarely involve non-motor symptoms. Third, there is a lack of corresponding large sample clinical studies to verify the experimental results. Therefore, extensive investigation is required to answer these questions.
TCM and Cardiovascular Diseases
At the age of 65 years or older, cardiovascular diseases (CVDs) are the leading pathological conditions, causing 40% of all deaths.36 Calcium transient amplitude decreases gradually with age, increasing the risk of arrhythmias in the older population.37 Accumulation of senescent cells within the vascular wall and heart can contribute to changes in cardiac structure and function, including progressive myocardial remodeling, left ventricular hypertrophy, decreased systolic and diastolic function, and aggravates pathophysiological processes of cardiovascular diseases, such as atherosclerosis, myocardial infarction, and cardiac fibrosis.38,39
Recent studies have confirmed that TCM has strong beneficial effects in the prevention and treatment of CVDs.40 Accumulating evidence in model organisms and humans links cardiac aging to mitochondrial regulation.41 Qishen granules promote PGC-1α-mediated mitochondrial biogenesis by coordinating mdm2-p53 mediated mitochondrial autophagy and Mitoch, and improve the degradation and protection of p53 protein in mice.42 In atherosclerotic mice, TCM can suppress the inflammatory response, ameliorate endothelial growth/function and improve blood lipid levels, leading the effect of anti-atherosclerosis.43 Similarly, berberine (BBR) in Coptis chinensis and other Berberis plants has been shown to attenuate trimethylamine/trimethylamine-N-oxide production and mitigate atherosclerotic lesion areas in mice via manipulation of the gut microbiome.44 Dehydrocorydaline, an alkaloidal compound isolated from Corydalis yanhusuo, inhibits macrophage inflammation by targeting macrophage p65- and ERK1/2-mediated pathways to ameliorate atherosclerosis in mice.45 Ethanol extracts from Schisandra chinensis fruit significantly reduced atherosclerosis in rats by regulating serum lipid levels, enhancing the antioxidant enzyme activities, and improving endothelial dysfunction.46
There are several successive and interrelated steps in the development of atherosclerosis that have served as excellent models for evaluating atherosclerotic protective drugs, and TCM acts on one or more of these steps: (1) Low-density lipoprotein (LDL) oxidation activates endothelial cells and initiates the vicious cycle of atherosclerotic plaque progression.47 Chinese herb compounds contribute to antioxidant activities and inhibit LDL oxidation.48 Oxymatrine, a quinolizidine alkaloid isolated from Sophora flavescens, may attenuate oxidized low-density lipoprotein in vitro cell model by inhibiting NLRP3 inflammasome-mediated pyroptosis via activating the SIRT1/Nrf2 signaling pathway.49 (2) Endothelial dysfunction, reduced elasticity of blood vessels and enhanced development of vascular plaque are central in the pathophysiologic processes of CVDs.50 Astragaloside IV, the primary active ingredient of the Chinese herb Huangqi, prevents endothelial dysfunction by reducing apoptosis, oxidative stress, and inflammatory response in vitro.51 (3) Vascular smooth muscle cell (VSMC) dysfunction, promoting extracellular matrix formation in atherosclerosis plaque areas, is the early form of atherosclerosis.52 BBR significantly inhibits angiotensin II and heparin-bound epidermal growth factor by delaying or partially inactivating the Akt signaling pathway, thereby inhibiting the proliferation and migration of VSMCs.53 (4) Macrophage-derived foam cell formation and inflammation is the hallmark of atherosclerosis.54 Increased ox-LDL flow and the accumulation of cholesterol esters in endometrial macrophages are the main factors. Clematichinenoside AR (AR), a major active ingredient extracted from the traditional Chinese herb Clematis chinensis, inhibits foam cell formation by increasing cholesterol efflux and reduces the production of inflammatory cytokines by passivating the activation of the NLRP3 inflammasome in macrophages.55 (5) Platelet activation and thrombus formation is the primary clinical presentation of atherothrombotic events.56 Administration of Tanshinone IIA inhibited platelet activation more effectively via downregulating the CD36 and MKK4/JNK2 signaling pathways.57 Simultaneous administration of Ginkgo biloba extract 50 and aspirin exert both synergistic and additive effects in restraining platelet aggregation, which enhance the antiplatelet effects.58 Therefore, it is reasonable to believe that there is a crucial link between TCM and age-related CVDs.
TCM and Diabetes
Diabetes is considered a disease of aging. The abundance of senescent cells in various tissues increases with age and diabetes. Aging contributes to the development of diabetes and its complications.59,60 Senescent adipocytes can induce macrophages to enter visceral adipose tissue and release chemokines, which aggravate the development of type 2 diabetes mellitus (T2DM).61 The high glucose and fat environment in diabetic patients has been identified as the main cause of cellular senescence.62 The main mechanisms of T2DM and aging are insulin resistance caused by hyperglycemia, the decrease of insulin-like growth factor⁃1 (IGF⁃1) levels, lipotoxicity and a series of senescence-associated secretory phenotype (SASP) and hyperglycemia is a key trigger of aging.63
TCM can prevent and treat diabetes through multiple targets and pathways. For example, BBR prolongs high glucose-dependent cell membrane depolarization by blocking KCNH6 currents and stimulates insulin secretion in mice with hyperglycemia induced by a high-fat diet. A Phase 1 clinical trial in humans found that compared with placebo treatment, BBR significantly promotes insulin secretion under a hyperglycemic state without side effects and did not affect basal insulin secretion.64 Tetrahydroxystilbene glucoside (TSG), a main component of Polygonatum multiflorum, can improve memory ability and prolong the lifespan via reduced levels of neural insulin or insulin-like growth factor-1 and upregulation of neural klotho in mice.65 Gegen is a Chinese herbal medicine that is often combined with other herbs to treat diabetes and other chronic metabolic diseases, and the isoflavone puerarin is the main active component of gegen. The combination of canagliflozin and puerarin can alleviate the oxidative stress and renal inflammation induced by lipotoxicity.66 In the process of aging, senescent cells secrete pro-inflammatory cytokines and other factors, collectively known as SASP. SASP secretion, by contributing to inflammation, is a key risk factor for the development of aging-related diseases. Psoralen, with prenylflavonoids (PF) as the main bioactive components, can delay aging. There is insufficient evidence to prove that these PF can be used as anti-aging drugs (via modifying SASP components), but they can block certain aging-related signaling pathways by entering cells and play an anti-inflammatory and anti-aging role (such as in diabetes).67 These results indicate that TCM can be an important entry point to treating diabetes and preventing or delaying aging. However, related research should focus on the basic mechanisms and continue to explore how best to provide precision medicine for elderly and diabetic patients.
TCM and Cancer
The global population is growing older. Malignant tumors are closely related to aging, and the incidence is much higher in the population aged over 60 years, more than double that in the younger population.68 Most elderly patients with malignant tumors are complicated with multiple chronic diseases, and the mortality rate is higher in these patients. The mechanisms of both cancer and aging are based on the time-dependent accumulation of cellular damage, and many of the characteristics of aging are shared with cancer. Recent research has shown that aging can affect the tumor microenvironment (TME) in normal cells, fibroblasts, and immune cells, which contributes to the progression and metastasis of tumors.69
Fibroblasts are the most common stromal component within tissues across the body and play a major role in tissue homeostasis. Cancer-associated fibroblasts (CAFs) are activated fibroblasts associated with cancer and play an important role in tumor growth and metastasis. Studies over the past two decades have provided important information on the ways TCM may suppress CAFs, indicating that TCM may also be effective cancer therapeutic treatment.70 Qingyihua is a TCM prescription that has an anti-cancer effect on pancreatic cancer and is related to changes in the TME. It may inhibit the tumor-promoting ability of CAFs and weaken the invasion of pancreatic cancer cells in vivo and in vitro.71 Artemisinin derivatives have been reported to inhibit CAF-induced growth and metastasis in an orthotopic model of breast cancer, possibly by inhibiting TGF-β signaling to inactivate CAFs fibroblasts and inhibit cancer metastasis.72
The interaction between tumor cells and the TME plays a decisive role in the progression of tumors. Solamargine (SM), a TCM compound, has been reported to reduce the proliferation of hepatocellular carcinoma and effectively induce hepatocellular carcinoma cell apoptosis and autophagy via inhibiting LIF/miR-192-5p/CYR61/Akt signaling pathways and eliciting an immunostimulatory tumor microenvironment in vitro and in vivo.73 Yi-Yi-Fu-Zi-Bai-Jiang-San is a CH from Synopsis of Prescriptions of the Golden Chamber that has been reported to inhibit colorectal tumorigenesis by increased immune responses in the tumor microenvironment through its hypoxia-associated anti-cancer activities in a mouse model.74 In the TME, the activation of the programmed death-1 (PD-1)–programmed death ligand-1 (PD-L1) pathway is one of the main signals of immune escape and tumor deterioration. Platycodon grandiflorum (PG), a medicine and food homology herb, can improve antitumor immunity by downregulating PD-1 expression of CD8+ T cells in the TME.75
The activation of the PD-1/PD-L1 pathway is one of the main signals of immune escape and tumor deterioration. Recently, studies have shown the importance of TCM combined with anti-PD-L1 antibodies in cancer therapy.76–78 Analyses of the relationship between the therapeutic efficacy of immune checkpoint inhibitors and TCM in patients with different cancers have found that CH could facilitate immune checkpoint blockade therapy or CAR-T cell therapy for cancer treatment. Furthermore, TCM can modulate adaptive immunity by activating T effector cells mainly through its action on SHP1/2.79 In another study, baicalein and baicalin blockade of the PD-L1/PD-1 pathway to prevent the immune evasion of tumor cells by decreasing STAT3 activity and downregulating IFN-γ subsequently restored T cell sensitivity to promote antitumor immunity.80 Neobavaisoflavone (Neo), as a traditional Chinese medicine, inhibited the growth and lung metastasis of tumors in vivo. In addition, Neo enhanced anti-PD-1 efficacy in anti-PD-1-insensitive tumors.78 The above results indicate that combining cancer immunotherapy with TCM may soon achieve better clinical results for patients.
Mechanisms of TCM Against Aging and Age-Related Diseases
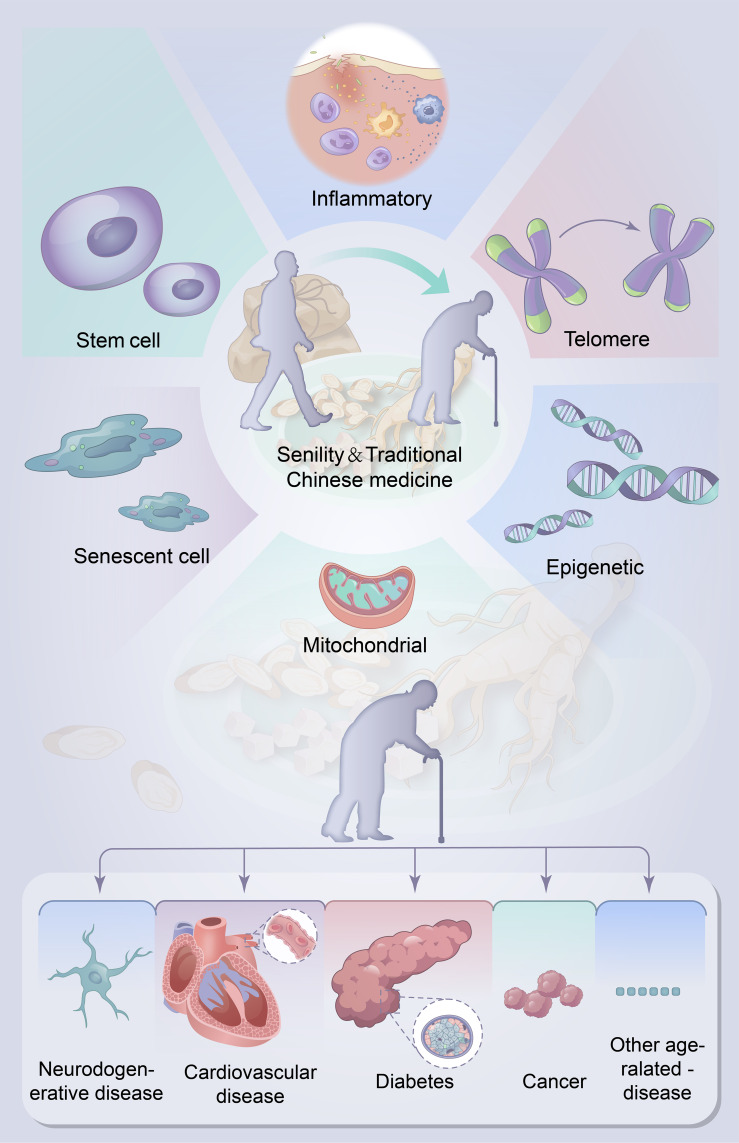
TCM has been widely used in the treatment of aging and age-related diseases and achieved satisfactory clinical effect. With the development of the modernization of TCM, researchers have explored the specific mechanisms of TCM in view of its multi-target and multi-component advantages. There are several main models based on the available evidence (Figure 2).
Figure 2.
Potential role and major mechanisms of traditional Chinese medicine on Aging and aging-related diseases. Administration with traditional Chinese medicine may be an effective project for aging and aging-related diseases. The potential possible mechanisms of traditional Chinese medicine to control the aging and aging-related diseases are (a) increase the length of telomere; (b) Modulating epigenetics; (c) Maintenance of mitochondrial homeostasis. (d) Reduce the senescent cells; (e) Maintenance of stem cell viability; (f) Improve the intestinal flora.
Epigenetic regulation is used to classify heritable changes in gene expression that are not attributable to changes in DNA sequences.81 Mounting evidence suggests that epigenetic dysregulation is a driver of cellular senescence and can accelerate the progression of age-associated disease.82,83 As a major epigenetic modification, DNA methylation is one of the important markers of aging.84,85 The role of TCM in DNA methylation has been widely reported. For example, Tanshinone IIA, the main fat‐soluble component of Salvia miltiorrhiza, can prevent rifampicin-induced liver injury through the DNA demethylation by ten-eleven translocation 2.86 Bazi bushen (BZBS), a TCM prescription, significantly restored the methylation age of aging mice and trimmed the methylation pattern to that of young mice, and the extract has a direct effect on the regulation of methylase during cell aging in vitro.87
Telomere shortening and damage are recognized causes of cellular senescence and aging. Some aging and age-related diseases associated with accelerated telomere dysfunction.88 Similarly, a range of evidence reveals the benefits of TCM for aging and age-related diseases. The effects of TCM on telomere dysfunction have been reported. BZBS can delay premature senescence by keeping redox homeostasis and telomere integrity, and inhibition of apoptosis in D-galactose and NaNO 2-induced aging mice.12
Autophagy plays important roles in regulating cell homeostasis and aging processes. Decreased autophagic capacity has been observed in animal models of aging, and some CH exert an important role in prolonging life via autophagy.89,90 An herbal drink can maintain stem cell homeostasis and provide cytoprotection.91 Dendrobium nobile var. Lindl., used as an anti-aging herb, can exert anti-aging activity via reducing oxidative stress, and by enhancing autophagy through the Sch9/Rim15/Msn2 signaling pathway.92
Senescent cells trigger a low chronic inflammatory state, leading to changes in the microenvironment, thus inducing cellular senescence in neighboring cells and playing an important role in tissue and organ aging and ARDs.14,93 With aging, stem cells show decreased self-renewal capacity and decreased function. TCM can reduce the number of senescent cells, reduce the level of cell cycle arrest protein, and maintain the cell repair ability. Studies have shown that CH play a role in maintaining the viability of stem cells, including inhibit aging, improve differentiation and promote proliferation.94 R. glutinosa, an anti-aging Chinese medicine, has been shown to reduce cell senescence and increase survival in a mouse model, possibly by maintaining the quiescence of hematopoietic stem cells and downregulating the cellular senescence-related proteins p53 and p16.95 Allicin, a chemical extracted from garlic, has been reported to alleviate the ameliorated aging phenotype in hematopoietic stem cells by up-regulating PKM2 expression.96
With aging, the gut homeostasis changes, and the imbalance of intestinal homeostasis contributes to many age-related degenerative diseases and unhealthy aging. CH can adjust the level of intestinal flora and improve the species diversity of intestinal flora and its metabolites, thereby improving the intestinal microecology. In one study, 104-week-old mice fed with ginsenoside Rb1 (GRb1) for 6 weeks showed characteristics of improved intestinal aging. GRb1 could reduce the senescent cells that are increased in the intestinal crypts of older mice. An increase in the protein levels of CLDN 2, 3, 7, 15, SIRT1, SIRT3, SIRT6 and Tert, Lgr5, mKi67, and c-Myc with changes in the composition and functions of the gut microbiota were evident. Thus, GRb1 was shown to improve intestinal aging, with an important role in regulating the expression of the sirtuin family.97
Conclusions and Perspectives
Convincing evidence has supported a strong link between TCM and aging. In terms of anti-aging, Chinese herbal has its unique advantages, and more evidence points to beneficial aspects (Table 1). Furthermore, there is a correlation between Chinese herbal and the prevention and control of age-related diseases (Table 2). TCM can enhance the ability to resist external stimuli and play a role in delaying aging through anti-inflammation and anti-oxidation. Meanwhile, TCM has shown improvement effects on cardiovascular, nervous system diseases and age-related diseases. However, there remain some deficiencies in the relevant studies on the use of TCM in anti-aging treatment. First, most of the studies on TCM remain at the level of in vitro experiments, and the relevant animal experimental studies are few, so further in vivo evaluations and clinical trials are needed to verify their reliability. In addition, although the anti-aging effect of TCM is better, research into its efficacy and safety is inevitably limited by factors such as its complex composition. Furthermore, there is a lack of support for the development of scientific research in this field, such that the public lacks a comprehensive understanding of TCM in anti-aging. In conclusion, TCM provide a vast resource that may be therapeutically useful in anti-aging. Further research on the role of TCM in anti-aging therapy is important to explore.
Table 1.
The Link Between Chinese Herbal Medicine with Age-Related Disease
| Age-Related Disease | Chinese Herbal Medicine | Composition | Potential Mechanisms | Ref. |
|---|---|---|---|---|
| Atherosclerosis | Qing-Xue-Xiao-Zhi formula | Reynoutria japonica Houtt., Rheum palmatum l., Typha angustifolia l., Curcuma longa l., Alisma plantago-aquatica Linn., Dioscorea septem loba Thunb. Artemisia capillaris Thunb., Atractylodes macrocephala Koidz., Acori tatarinowii Rhizoma, Carthamus tinctorius l. | Regulate TLR4/MyD88/NF-κB pathway | [98] |
| Parkinson’s disease | Yiguanjian decoction | Glehniae Radix, Toosendan Fructus, Angelicae Sinensis Radix, Rehmanniae Radix Praeparata, Lycii Fructus, Ophiopogon japonicus, | Regulate the PI3K-Akt pathway, modulate endocrine resistance of the central nervous | [99] |
| Osteoporosis | Qing’e Pill | Eucommia ulmoides Oliv., Psoralea corylifolia L., Juglans regia L., and Allium sativum L. | Inhibit ferroptosis and promoting cell survival via the PI3K/AKT pathway and ATM | [100] |
| Osteoarthritis | Gubitong Recipe | Davallia mariesii Moore ex bak, Epimedium brevicornu Maxim., Psoralea corylifolia Linn., Eucommia ulmoides Oliver, Lycium chinense Miller, Rhizoma Bolbostematis, Stephania tetrandra S. Moore, Spatholobus suberectus Dunn | Inhibit the NF κB pathway | [101] |
| Chronic kidney disease | Jian-Pi-Yi-Shen Formula | Astragalus membranaceus, Dioscorea opposita Thunb. Rehmannia glutinosa, Cistanche deserticola Ma, Amomum kravanh Pierre ex Gagnep. Salvia miltiorrhiza Bunge, Rheum palmatum L., Glycyrrhiza uralensis Fisch | Modulate QPRT/NAD+/SIRT3/Mitochondrial Dynamics Pathway and regulate Tryptophan Metabolism and Aryl Hydrocarbon Receptor Signaling. | [102,103] |
| Type 2 diabetes | Qijian mixture | Astragalus membranaceus, Ramulus euonymi, Coptis chinensis and Pueraria lobata, | Acte through TP53, AKT1 and PPARA proteins | [104] |
| Chronic obstructive pulmonary disease | Bufei Yishen formula | Ginseng Radix et Rhizoma, Astragali Radix, Corni Fructus, Lycii Fructus, Schisandra Chinensis Fructus, Fritillariae Thunbergii Bulbus, Perillae Fructus, Citri Reticulatae Pericarpium, Epimedii Folium, Paeoniae Rubra Radix, Pheretima, Ardisiae Japonicae Herba | Restore Th17/Treg balance via reciprocally modulating the activities of STAT3 and STAT5 | [105] |
Table 2.
Chinese Herbs and Its Anti-Aging Effects
| Chinese Herbs | Type of Bioactive Components | Potential Mechanisms | Models | Ref. |
|---|---|---|---|---|
| Ginseng | Ginsenoside Rg1 | Antioxidant enzymes GSH-Px and SO↑, telomere lengths and telomerase activity↑, the levels of proinflammatory cytokines↓ | D-galactose induced aging rats | [106] |
| R. Astragali | Astragalosides, Astragalan, TA-65 | Immunomodulatory effects↑, lengthens telomeres↑, the lifespan of daf-2 and age-1 mutants↑ | Aging and the middle-aged rats, Caenorhabditis elegans Relatively healthy cytomegalovirus-positive subjects aged 53–87 years old | [107–109] |
| Ganoderma lucidum | Total water extract of G. lucidum | Anti-oxidative stress↑, mTOR/S6K signaling pathway, | Caenorhabditis elegans | [110] |
| Astragalus membranaceus | Polysaccharides, flavonoids, and saponins | Telomerase activity↑, antioxidant, anti-inflammatory and immunoregulatory | Myocardial ischemic rat, aging rats | [111] |
| Resveratrol | NR | Prevent inflammation and oxidative stress by activating Nrf2 and SIRT1 signaling↑ | Male 18-month-old C57BL/6 mice | [112] |
| Psoralea corylifolia | Corylin | Gtr1 protein | Yeast, aging mice fed with a high-fat diet | [113] |
| Schisandra chinensis | Schisandra chinensis lignans | The mRNA expression level of p19, p53, and p21 ↓ | Aging rats | [114] |
| Dendrobium officinale | NR | Antioxidation, immunoregulatory. Serum levels of SOD, GSH-Px and T-AOC, and restored SOD levels ↑ | Aging rats | [115] |
Abbreviation: NR, Not Report.
Funding Statement
This work was supported by Henan Province COVID-19 Traditional Chinese Medicine Scientific Research Special Project (2022ZYFY02), HenanProvince Special Project of Traditional Chinese Medicine Scientific Research (2019AZB006, 2019JDZX2096, 2022JDZX154), Henan Province Key Research and Development and promotion project (No.232102311222) and the Science and Technology Research Project of HenanProvince (222102310570).
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.World Health Organization. Ageing and health; 2022. Available from: https://www.who.int/news-room/fact-sheets/detail/ageing-and-health. Accessed September 1, 2022.
- 2.Zhang M. Study on the Medication Rule of Ancient Anti-senile Prescriptions [PhD Thesis]. China Academy of Chinese Medical Sciences; 2018. [Google Scholar]
- 3.Gómez-Linton DR, Alavez S, Alarcón-Aguilar A, et al. Some naturally occurring compounds that increase longevity and stress resistance in model organisms of aging. Biogerontology. 2019;20(5):583–603. doi: 10.1007/s10522-019-09817-2 [DOI] [PubMed] [Google Scholar]
- 4.Xiao G, Xuan Z, Xiaozhen G, et al. Jiawei Sijunzi decoction delays aging of mouse spleen tissue through mTOR signaling pathway. Acad J Second Mil Med Univ. 2020;41(12):1410–1413. doi: 10.16781/j.0258-879x.2020.12.1410 [DOI] [Google Scholar]
- 5.Ying S, Xiao G, Yanqiong Q, et al. Flavored Sijunzi Delays aging of brain tissue of mice via mTOR signaling pathway. Acta Med Univ Sci Technol Huazhong. 2018;47(04):417–420. [Google Scholar]
- 6.Fan Z, Ao X, Jinyue Y, et al. Metabonomics study of Jinkui Shenqi pills on improving the memory ability of D-galactose-induced aging rats. Chin J Pharm Anal. 2020;40(12):2149–2156. doi: 10.16155/j.0254-1793.2020.12.07 [DOI] [Google Scholar]
- 7.Yan Z, Changcheng Z, Yuan Y, et al. Protective effects on shenqi pills against injury of tight junction function of sertoli cells in the testis of aging rats and its mechanism. Traditional Chin Drug Res Clin Pharmacol. 2021;32(06):783–790. doi: 10.19378/j.issn.1003-9783.2021.06.006 [DOI] [Google Scholar]
- 8.Zhu Y, Han Y, Wang W, et al. Mulberry leaves attenuate D-galactose-induced aging in vivo and in vitro. J Ethnopharmacol. 2023;311:116286. doi: 10.1016/j.jep.2023.116286 [DOI] [PubMed] [Google Scholar]
- 9.Zhu S, Jia L, Wang X, et al. Anti-aging formula protects skin from oxidative stress-induced senescence through the inhibition of CXCR2 expression. J Ethnopharmacol. 2023;318(Pt B):116996. doi: 10.1016/j.jep.2023.116996 [DOI] [PubMed] [Google Scholar]
- 10.Shenghua P, Ziqin Z, Shuyu T, et al. An integrated fecal microbiome and metabolome in the aged mice reveal anti-aging effects from the intestines and biochemical mechanism of FuFang zhenshu TiaoZhi(FTZ). Biomed Pharmacother. 2020;121:109421. doi: 10.1016/j.biopha.2019.109421 [DOI] [PubMed] [Google Scholar]
- 11.Teseo S, Houot B, Yang K, et al. G. sinense and P. notoginseng extracts improve healthspan of aging flies and provide protection in a Huntington disease model. Aging Dis. 2021;12(2):425–440. doi: 10.14336/ad.2020.0714-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li L, Zhang H, Chen B, et al. BaZiBuShen alleviates cognitive deficits and regulates Sirt6/NRF2/HO-1 and Sirt6/P53-PGC-1α-TERT signaling pathways in aging mice. J Ethnopharmacol. 2022;282:114653. doi: 10.1016/j.jep.2021.114653 [DOI] [PubMed] [Google Scholar]
- 13.Adler AS, Sinha S, Kawahara TL, et al. Motif module map reveals enforcement of aging by continual NF-kappaB activity. Genes Dev. 2007;21(24):3244–3257. doi: 10.1101/gad.1588507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zia A, Farkhondeh T, Pourbagher-Shahri AM, et al. The role of curcumin in aging and senescence: molecular mechanisms. Biomed Pharmacother. 2021;134:111119. doi: 10.1016/j.biopha.2020.111119 [DOI] [PubMed] [Google Scholar]
- 15.Culig L, Chu X, Bohr VA. Neurogenesis in aging and age-related neurodegenerative diseases. Ageing Res Rev. 2022;78:101636. doi: 10.1016/j.arr.2022.101636 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alzheimer’s Association. 2021 Alzheimer’s disease facts and figures. Alzheimers Dement. 2021;17(3):327–406. doi: 10.1002/alz.12328 [DOI] [PubMed] [Google Scholar]
- 17.Calderón-Larrañaga A, Vetrano DL, Onder G, et al. Assessing and measuring chronic multimorbidity in the older population: a proposal for its operationalization. J Gerontol a Biol Sci Med Sci. 2017;72(10):1417–1423. doi: 10.1093/gerona/glw233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Delgado-Lara DL, González-Enríquez GV, Torres-Mendoza BM, et al. Effect of melatonin administration on the PER1 and BMAL1 clock genes in patients with Parkinson’s disease. Biomed Pharmacother. 2020;129:110485. doi: 10.1016/j.biopha.2020.110485 [DOI] [PubMed] [Google Scholar]
- 19.Poewe W, Seppi K, Tanner CM, et al. Parkinson disease. Nat Rev Dis Primers. 2017;3:17013. doi: 10.1038/nrdp.2017.13 [DOI] [PubMed] [Google Scholar]
- 20.Pandya VA, Patani R. Decoding the relationship between ageing and amyotrophic lateral sclerosis: a cellular perspective. Brain. 2020;143(4):1057–1072. doi: 10.1093/brain/awz360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xu L, Liu T, Liu L, et al. Global variation in prevalence and incidence of amyotrophic lateral sclerosis: a systematic review and meta-analysis. J Neurol. 2020;267(4):944–953. doi: 10.1007/s00415-019-09652-y [DOI] [PubMed] [Google Scholar]
- 22.Lane CA, Hardy J, Schott JM. Alzheimer’s disease. Eur J Neurol. 2018;25(1):59–70. doi: 10.1111/ene.13439 [DOI] [PubMed] [Google Scholar]
- 23.Zhang Y, Lin C, Zhang L, et al. Cognitive improvement during treatment for mild Alzheimer’s disease with a Chinese Herbal Formula: a randomized controlled trial. PLoS One. 2015;10(6):e0130353. doi: 10.1371/journal.pone.0130353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ning F, Chen L, Chen L, et al. Combination of polygoni multiflori radix praeparata and acori tatarinowii rhizoma alleviates learning and memory impairment in scopolamine-treated mice by regulating synaptic-related proteins. Front Pharmacol. 2021;12:679573. doi: 10.3389/fphar.2021.679573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Huang Q, Zhang C, Qu S, et al. Chinese herbal extracts exert neuroprotective effect in Alzheimer’s disease mouse through the dopaminergic synapse/apoptosis signaling pathway. Front Pharmacol. 2022;13:817213. doi: 10.3389/fphar.2022.817213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhou Q, Wang Y, Zhang J, et al. Fingerprint analysis of Huolingshengji Formula and its neuroprotective effects in SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Sci Rep. 2018;8(1):1668. doi: 10.1038/s41598-018-19923-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gong F, Zhu W, Liao W, et al. Mechanism of the curative effect of Wen-Shen-Jian-Pi prescription in the treatment of amyotrophic lateral sclerosis. Front Aging Neurosci. 2022;14:873224. doi: 10.3389/fnagi.2022.873224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.In S, Hong CW, Choi B, et al. Inhibition of mitochondrial clearance and Cu/Zn-SOD activity enhance 6-hydroxydopamine-induced neuronal apoptosis. Mol Neurobiol. 2016;53(1):777–791. doi: 10.1007/s12035-014-9087-9 [DOI] [PubMed] [Google Scholar]
- 29.Holper L, Ben-Shachar D, Mann JJ. Multivariate meta-analyses of mitochondrial complex I and IV in major depressive disorder, bipolar disorder, schizophrenia, Alzheimer disease, and Parkinson disease. Neuropsychopharmacology. 2019;44(5):837–849. doi: 10.1038/s41386-018-0090-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gao L, Cao M, Du GH, et al. Huangqin decoction exerts beneficial effects on rotenone-induced rat model of Parkinson’s disease by improving mitochondrial dysfunction and alleviating metabolic abnormality of mitochondria. Front Aging Neurosci. 2022;14:911924. doi: 10.3389/fnagi.2022.911924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Geng J, Liu W, Gao J, et al. Andrographolide alleviates Parkinsonism in MPTP-PD mice via targeting mitochondrial fission mediated by dynamin-related protein 1. Br J Pharmacol. 2019;176(23):4574–4591. doi: 10.1111/bph.14823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li L, Fan S, Zhang W, et al. Duzhong fang attenuates the POMC-derived neuroinflammation in Parkinsonian mice. J Inflamm Res. 2021;14:3261–3276. doi: 10.2147/jir.S316314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bao X, He Y, Huang L, et al. Sinomenine exerts a neuroprotective effect on PD mouse model through inhibiting PI3K/AKT/mTOR pathway to enhance autophagy. Int J Neurosci. 2022:1–9. doi: 10.1080/00207454.2022.2100780 [DOI] [PubMed] [Google Scholar]
- 34.Liu XL, Lu J, Tian Y, et al. 艾灸激活Nrf2/ARE通路抗帕金森病模型大鼠氧化应激损伤的机制研究 [Effect and mechanism of moxibustion on oxidative stress injury in rats with Parkinson’s disease by activating Nrf2/ARE pathway]. Zhongguo Zhen Jiu. 2020;40(8):857–862. Chinese. doi: 10.13703/j.0255-2930.20190620-k0002 [DOI] [PubMed] [Google Scholar]
- 35.Gu SC, Shi R, Gaoag C, et al. Traditional Chinese medicine Pingchan granule for motor symptoms and functions in Parkinson’s disease: a multicenter, randomized, double-blind, placebo-controlled study. Phytomedicine. 2023;108:154497. doi: 10.1016/j.phymed.2022.154497 [DOI] [PubMed] [Google Scholar]
- 36.North BJ, Sinclair DA. The intersection between aging and cardiovascular disease. Circ Res. 2012;110(8):1097–1108. doi: 10.1161/circresaha.111.246876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Betts DH, Bain NT, Madan P. The p66(Shc) adaptor protein controls oxidative stress response in early bovine embryos. PLoS One. 2014;9(1):e86978. doi: 10.1371/journal.pone.0086978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen MS, Lee RT, Garbern JC. Senescence mechanisms and targets in the heart. Cardiovasc Res. 2022;118(5):1173–1187. doi: 10.1093/cvr/cvab161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yan M, Sun S, Xu K, et al. Cardiac aging: from basic research to therapeutics. Oxid Med Cell Longev. 2021;2021:9570325. doi: 10.1155/2021/9570325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wu GS, Li HK, Zhang WD. Metabolomics and its application in the treatment of coronary heart disease with traditional Chinese medicine. Chin J Nat Med. 2019;17(5):321–330. doi: 10.1016/s1875-5364(19)30037-8 [DOI] [PubMed] [Google Scholar]
- 41.Wang Y, Li Y, He C, et al. Mitochondrial regulation of cardiac aging. Biochim Biophys Acta Mol Basis Dis. 2019;1865(7):1853–1864. doi: 10.1016/j.bbadis.2018.12.008 [DOI] [PubMed] [Google Scholar]
- 42.Li W, Zhang Y, Wang X, et al. Qishen granule protects against doxorubicin-induced cardiotoxicity by coordinating MDM2-p53-mediated mitophagy and mitochondrial biogenesis. Oxid Med Cell Longev. 2022;2022:4344677. doi: 10.1155/2022/4344677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liu Y, Zhong H, Xu P, et al. Deciphering the combination mechanisms of Gualou-Xiebai herb pair against atherosclerosis by network pharmacology and HPLC-Q-TOF-MS technology. Front Pharmacol. 2022;13:941400. doi: 10.3389/fphar.2022.941400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li X, Su C, Jiang Z, et al. Berberine attenuates choline-induced atherosclerosis by inhibiting trimethylamine and trimethylamine-N-oxide production via manipulating the gut microbiome. NPJ Biofilms Microbiomes. 2021;7(1):36. doi: 10.1038/s41522-021-00205-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wen B, Dang YY, Wu SH, et al. Antiatherosclerotic effect of dehydrocorydaline on ApoE(-/-) mice: inhibition of macrophage inflammation. Acta Pharmacol Sin. 2022;43(6):1408–1418. doi: 10.1038/s41401-021-00769-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chen X, Cao J, Sun Y, et al. Ethanol extract of Schisandrae chinensis fructus ameliorates the extent of experimentally induced atherosclerosis in rats by increasing antioxidant capacity and improving endothelial dysfunction. Pharm Biol. 2018;56(1):612–619. doi: 10.1080/13880209.2018.1523933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pentikäinen MO, Oörni K, Ala-Korpela M, et al. Modified LDL - trigger of atherosclerosis and inflammation in the arterial intima. J Intern Med. 2000;247(3):359–370. doi: 10.1046/j.1365-2796.2000.00655.x [DOI] [PubMed] [Google Scholar]
- 48.Lin HH, Charles AL, Hsieh CW, et al. Antioxidant effects of 14 Chinese traditional medicinal herbs against human low-density lipoprotein oxidation. J Tradit Complement Med. 2015;5(1):51–55. doi: 10.1016/j.jtcme.2014.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jin X, Fu W, Zhou J, et al. Oxymatrine attenuates oxidized low‑density lipoprotein‑induced HUVEC injury by inhibiting NLRP3 inflammasome‑mediated pyroptosis via the activation of the SIRT1/Nrf2 signaling pathway. Int J Mol Med. 2021;48(4). doi: 10.3892/ijmm.2021.5020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cyr AR, Huckaby LV, Shiva SS, et al. Nitric Oxide and Endothelial Dysfunction. Crit Care Clin. 2020;36(2):307–321. doi: 10.1016/j.ccc.2019.12.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhu Z, Li J, Zhang X. Astragaloside IV Protects Against Oxidized Low-Density Lipoprotein (ox-LDL)-induced endothelial cell injury by reducing oxidative stress and inflammation. Med Sci Monit. 2019;25:2132–2140. doi: 10.12659/msm.912894 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bennett MR, Sinha S, Owens GK. Vascular smooth muscle cells in atherosclerosis. Circ Res. 2016;118(4):692–702. doi: 10.1161/circresaha.115.306361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lee S, Lim HJ, Park HY, et al. Berberine inhibits rat vascular smooth muscle cell proliferation and migration in vitro and improves neointima formation after balloon injury in vivo. Berberine improves neointima formation in a rat model. Atherosclerosis. 2006;186(1):29–37. doi: 10.1016/j.atherosclerosis.2005.06.048 [DOI] [PubMed] [Google Scholar]
- 54.Xu S, Ogura S, Chen J, et al. LOX-1 in atherosclerosis: biological functions and pharmacological modifiers. Cell Mol Life Sci. 2013;70(16):2859–2872. doi: 10.1007/s00018-012-1194-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Diao Y. Clematichinenoside AR alleviates foam Cell formation and the inflammatory response in Ox-LDL-Induced RAW264.7 cells by activating autophagy. Inflammation. 2021;44(2):758–768. doi: 10.1007/s10753-020-01375-x [DOI] [PubMed] [Google Scholar]
- 56.Badimon L, Padró T, Vilahur G. Atherosclerosis, platelets and thrombosis in acute ischaemic heart disease. Eur Heart J Acute Cardiovasc Care. 2012;1(1):60–74. doi: 10.1177/2048872612441582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang H, Zhong L, Mi S, et al. Tanshinone IIA prevents platelet activation and down-regulates CD36 and MKK4/JNK2 signaling pathway. BMC Cardiovasc Disord. 2020;20(1):81. doi: 10.1186/s12872-019-01289-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ke J, Li MT, Huo YJ, et al. The synergistic effect of ginkgo biloba extract 50 and aspirin against platelet aggregation. Drug Des Devel Ther. 2021;15:3543–3560. doi: 10.2147/dddt.S318515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Palmer AK, Gustafson B, Kirkland JL, et al. Cellular senescence: at the nexus between ageing and diabetes. Diabetologia. 2019;62(10):1835–1841. doi: 10.1007/s00125-019-4934-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kushner JA. The role of aging upon β cell turnover. J Clin Invest. 2013;123(3):990–995. doi: 10.1172/jci64095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xu M, Tchkonia T, Ding H, et al. JAK inhibition alleviates the cellular senescence-associated secretory phenotype and frailty in old age. Proc Natl Acad Sci U S A. 2015;112(46):E6301–E6310. doi: 10.1073/pnas.1515386112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Berlanga-Acosta JA, Guillén-Nieto GE, Rodríguez-Rodríguez N, et al. Cellular senescence as the pathogenic hub of diabetes-related wound chronicity. Front Endocrinol. 2020;11:573032. doi: 10.3389/fendo.2020.573032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Rong C, Yanmei C, Xiaofei Z, et al. Research progress in the correlation between diabetes mellitus and aging and its mechanism. Transl Med J. 2023;12(04):204–09+14. [Google Scholar]
- 64.Zhao MM, Lu J, Li S, et al. Berberine is an insulin secretagogue targeting the KCNH6 potassium channel. Nat Commun. 2021;12(1):5616. doi: 10.1038/s41467-021-25952-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zhou X, Yang Q, Xie Y, et al. Tetrahydroxystilbene glucoside extends mouse life span via upregulating neural klotho and downregulating neural insulin or insulin-like growth factor 1. Neurobiol Aging. 2015;36(3):1462–1470. doi: 10.1016/j.neurobiolaging.2014.11.002 [DOI] [PubMed] [Google Scholar]
- 66.Zhu Q, Zhou Q, Luo XL, et al. Combination of canagliflozin and puerarin alleviates the lipotoxicity to diabetic kidney in mice. Korean J Physiol Pharmacol. 2023;27(3):221–230. doi: 10.4196/kjpp.2023.27.3.221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhou YT, Zhu L, Yuan Y, et al. Effects and mechanisms of five psoralea prenylflavonoids on aging-related diseases. Oxid Med Cell Longev. 2020;2020:2128513. doi: 10.1155/2020/2128513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7–30. doi: 10.3322/caac.21442 [DOI] [PubMed] [Google Scholar]
- 69.Fane M, Weeraratna AT. How the ageing microenvironment influences tumour progression. Nat Rev Cancer. 2020;20(2):89–106. doi: 10.1038/s41568-019-0222-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Efferth T. Cancer combination therapies with artemisinin-type drugs. Biochem Pharmacol. 2017;139:56–70. doi: 10.1016/j.bcp.2017.03.019 [DOI] [PubMed] [Google Scholar]
- 71.Chen L, Qu C, Chen H, et al. Chinese herbal medicine suppresses invasion-promoting capacity of cancer-associated fibroblasts in pancreatic cancer. PLoS One. 2014;9(4):e96177. doi: 10.1371/journal.pone.0096177 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 72.Yao Y, Guo Q, Cao Y, et al. Artemisinin derivatives inactivate cancer-associated fibroblasts through suppressing TGF-β signaling in breast cancer. J Exp Clin Cancer Res. 2018;37(1):282. doi: 10.1186/s13046-018-0960-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Yin S, Jin W, Qiu Y, et al. Solamargine induces hepatocellular carcinoma cell apoptosis and autophagy via inhibiting LIF/miR-192-5p/CYR61/Akt signaling pathways and eliciting immunostimulatory tumor microenvironment. J Hematol Oncol. 2022;15(1):32. doi: 10.1186/s13045-022-01248-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhang Y, Chai N, Wei Z, et al. YYFZBJS inhibits colorectal tumorigenesis by enhancing Tregs-induced immunosuppression through HIF-1α mediated hypoxia in vivo and in vitro. Phytomedicine. 2022;98:153917. doi: 10.1016/j.phymed.2021.153917 [DOI] [PubMed] [Google Scholar]
- 75.Yang R, Pei T, Huang R, et al. Platycodon grandiflorum triggers antitumor immunity by restricting PD-1 expression of CD8(+) T cells in local tumor microenvironment. Front Pharmacol. 2022;13:774440. doi: 10.3389/fphar.2022.774440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Jiang Z, Yang Y, Yang Y, et al. Ginsenoside Rg3 attenuates cisplatin resistance in lung cancer by downregulating PD-L1 and resuming immune. Biomed Pharmacother. 2017;96:378–383. doi: 10.1016/j.biopha.2017.09.129 [DOI] [PubMed] [Google Scholar]
- 77.Song M, Qian C, Zhang T, et al. Salvia mitiorrhiza Bunge aqueous extract attenuates infiltration of tumor-associated macrophages and potentiates anti-PD-L1 immunotherapy in colorectal cancer through modulating Cox2/PGE2 cascade. J Ethnopharmacol. 2023;316:116735. doi: 10.1016/j.jep.2023.116735 [DOI] [PubMed] [Google Scholar]
- 78.Guo J, Shen Y, Hu S, et al. Neobavaisoflavone inhibits antitumor immunosuppression via myeloid-derived suppressor cells. Int Immunopharmacol. 2022;111:109103. doi: 10.1016/j.intimp.2022.109103 [DOI] [PubMed] [Google Scholar]
- 79.Lam W, Hu R, Liu SH, et al. YIV-906 enhances nuclear factor of activated T-cells (NFAT) activity of T cells and promotes immune checkpoint blockade antibody action and CAR T-cell activity. Front Pharmacol. 2022;13:1095186. doi: 10.3389/fphar.2022.1095186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ke M, Zhang Z, Xu B, et al. Baicalein and baicalin promote antitumor immunity by suppressing PD-L1 expression in hepatocellular carcinoma cells. Int Immunopharmacol. 2019;75:105824. doi: 10.1016/j.intimp.2019.105824 [DOI] [PubMed] [Google Scholar]
- 81.Dupont C, Armant DR, Brenner CA. Epigenetics: definition, mechanisms and clinical perspective. Semin Reprod Med. 2009;27(5):351–357. doi: 10.1055/s-0029-1237423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rando TA, Chang HY. Aging, rejuvenation, and epigenetic reprogramming: resetting the aging clock. Cell. 2012;148(1–2):46–57. doi: 10.1016/j.cell.2012.01.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Schroeder EA, Raimundo N, Shadel GS. Epigenetic silencing mediates mitochondria stress-induced longevity. Cell Metab. 2013;17(6):954–964. doi: 10.1016/j.cmet.2013.04.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Law PP, Holland ML. DNA methylation at the crossroads of gene and environment interactions. Essays Biochem. 2019;63(6):717–726. doi: 10.1042/ebc20190031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lowe R, Barton C, Jenkins CA, et al. Ageing-associated DNA methylation dynamics are a molecular readout of lifespan variation among mammalian species. Genome Biol. 2018;19(1):22. doi: 10.1186/s13059-018-1397-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Yang Y, Liu L, Zhang X, et al. Tanshinone IIA prevents rifampicin-induced liver injury by regulating BSEP/NTCP expression via epigenetic activation of NRF2. Liver Int. 2020;40(1):141–154. doi: 10.1111/liv.14262 [DOI] [PubMed] [Google Scholar]
- 87.Mao X, Hou Y, Fang C, et al. Bazi Bushen mitigates epigenetic aging and extends healthspan in naturally aging mice. Biomed Pharmacother. 2023;160:114384. doi: 10.1016/j.biopha.2023.114384 [DOI] [PubMed] [Google Scholar]
- 88.Rossiello F, Jurk D, Passos JF, et al. Telomere dysfunction in ageing and age-related diseases. Nat Cell Biol. 2022;24(2):135–147. doi: 10.1038/s41556-022-00842-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Hansen M, Rubinsztein DC, Walker DW. Autophagy as a promoter of longevity: insights from model organisms. Nat Rev Mol Cell Biol. 2018;19(9):579–593. doi: 10.1038/s41580-018-0033-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Pan Y, Liu Y, Fujii R, et al. Ehretiquinone from onosma bracteatum wall exhibits antiaging effect on yeasts and mammals through antioxidative stress and autophagy induction. Oxid Med Cell Longev. 2021;2021:5469849. doi: 10.1155/2021/5469849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Shibu MA, Lin YJ, Chiang CY, et al. Novel anti-aging herbal formulation Jing Si displays pleiotropic effects against aging associated disorders. Biomed Pharmacother. 2022;146:112427. doi: 10.1016/j.biopha.2021.112427 [DOI] [PubMed] [Google Scholar]
- 92.Wu E, Lian Y, Zhao S, et al. Involvement of the Sch9/Rim15/Msn2 signaling pathway in the anti-aging activity of dendrobine from Dendrobium nobile Lindl. via modification of oxidative stress and autophagy. Chin Med. 2023;18(1):111. doi: 10.1186/s13020-023-00827-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.van Deursen JM. The role of senescent cells in ageing. Nature. 2014;509(7501):439–446. doi: 10.1038/nature13193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Qin W, Chen S, Yang S, et al. The effect of traditional Chinese medicine on neural stem cell proliferation and differentiation. Aging Dis. 2017;8(6):792–811. doi: 10.14336/ad.2017.0428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bai L, Shi GY, Yang YJ, et al. Rehmannia glutinosa exhibits anti-aging effect through maintaining the quiescence and decreasing the senescence of hematopoietic stem cells. Animal Model Exp Med. 2018;1(3):194–202. doi: 10.1002/ame2.12034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Cai SZ, Zhao LN, Liu J, et al. Allicin alleviates lead-induced hematopoietic stem cell aging by up-regulating PKM2. Biosci Rep. 2019;39(7). doi: 10.1042/bsr20190243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lei Z, Chen L, Hu Q, et al. Ginsenoside Rb1 improves intestinal aging via regulating the expression of sirtuins in the intestinal epithelium and modulating the gut microbiota of mice. Front Pharmacol. 2022;13:991597. doi: 10.3389/fphar.2022.991597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Li Y, Zhang L, Ren P, et al. Qing-Xue-Xiao-Zhi formula attenuates atherosclerosis by inhibiting macrophage lipid accumulation and inflammatory response via TLR4/MyD88/NF-κB pathway regulation. Phytomedicine. 2021;93:153812. doi: 10.1016/j.phymed.2021.153812 [DOI] [PubMed] [Google Scholar]
- 99.Shen Z, Yu M, Zhang S. Network pharmacology and molecular docking analyses unveil the mechanisms of yiguanjian decoction against Parkinson’s disease from inner/outer brain perspective. Biomed Res Int. 2022;2022:4758189. doi: 10.1155/2022/4758189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hao J, Bei J, Li Z, et al. Qing`e Pill inhibits osteoblast ferroptosis via ATM Serine/Threonine Kinase (ATM) and the PI3K/AKT pathway in primary osteoporosis. Front Pharmacol. 2022;13:902102. doi: 10.3389/fphar.2022.902102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chen GY, Liu XY, Luo J, et al. Integrating network pharmacology and experimental validation to explore the key mechanism of gubitong recipe in the treatment of osteoarthritis. Comput Math Methods Med. 2022;2022:7858925. doi: 10.1155/2022/7858925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Liu X, Deng R, Chen Y, et al. Jian-Pi-Yi-Shen formula improves adenine-induced chronic kidney disease via regulating tryptophan metabolism and Aryl hydrocarbon receptor signaling. Front Pharmacol. 2022;13:922707. doi: 10.3389/fphar.2022.922707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Liu X, Deng R, Wei X, et al. Jian-Pi-Yi-Shen formula enhances perindopril inhibition of chronic kidney disease progression by activation of SIRT3, modulation of mitochondrial dynamics, and antioxidant effects. Biosci Rep. 2021;41(10). doi: 10.1042/bsr20211598 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 104.Gao K, Yang R, Zhang J, et al. Effects of Qijian mixture on type 2 diabetes assessed by metabonomics, gut microbiota and network pharmacology. Pharmacol Res. 2018;130:93–109. doi: 10.1016/j.phrs.2018.01.011 [DOI] [PubMed] [Google Scholar]
- 105.Zhao P, Li J, Tian Y, et al. Restoring Th17/Treg balance via modulation of STAT3 and STAT5 activation contributes to the amelioration of chronic obstructive pulmonary disease by Bufei Yishen formula. J Ethnopharmacol. 2018;217:152–162. doi: 10.1016/j.jep.2018.02.023 [DOI] [PubMed] [Google Scholar]
- 106.Zhu J, Mu X, Zeng J, et al. Ginsenoside Rg1 prevents cognitive impairment and hippocampus senescence in a rat model of D-galactose-induced aging. PLoS One. 2014;9(6):e101291. doi: 10.1371/journal.pone.0101291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lei H, Wang B, Li WP, et al. Anti-aging effect of astragalosides and its mechanism of action. Acta Pharmacol Sin. 2003;24(3):230–234. [PubMed] [Google Scholar]
- 108.Zhang H, Pan N, Xiong S, et al. Inhibition of polyglutamine-mediated proteotoxicity by Astragalus membranaceus polysaccharide through the DAF-16/FOXO transcription factor in Caenorhabditis elegans. Biochem J. 2012;441(1):417–424. doi: 10.1042/bj20110621 [DOI] [PubMed] [Google Scholar]
- 109.Salvador L, Singaravelu G, Harley CB, et al. A natural product telomerase activator lengthens telomeres in humans: a randomized, double blind, and placebo controlled study. Rejuvenation Res. 2016;19(6):478–484. doi: 10.1089/rej.2015.1793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Cuong VT, Chen W, Shi J, et al. The anti-oxidation and anti-aging effects of Ganoderma lucidum in Caenorhabditis elegans. Exp Gerontol. 2019;117:99–105. doi: 10.1016/j.exger.2018.11.016 [DOI] [PubMed] [Google Scholar]
- 111.Liu P, Zhao H, Luo Y. Anti-aging implications of Astragalus Membranaceus (Huangqi): a well-known Chinese tonic. Aging Dis. 2017;8(6):868–886. doi: 10.14336/ad.2017.0816 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kim EN, Lim JH, Kim MY, et al. Resveratrol, an Nrf2 activator, ameliorates aging-related progressive renal injury. Aging. 2018;10(1):83–99. doi: 10.18632/aging.101361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Wang TH, Tseng WC, Leu YL, et al. The flavonoid corylin exhibits lifespan extension properties in mouse. Nat Commun. 2022;13(1):1238. doi: 10.1038/s41467-022-28908-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Sun J, Jing S, Jiang R, et al. Metabolomics study of the therapeutic mechanism of Schisandra chinensis lignans on aging rats induced by d-galactose. Clin Interv Aging. 2018;13:829–841. doi: 10.2147/cia.S163275 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Liang CY, Liang YM, Liu HZ, et al. Effect of Dendrobium officinale on D-galactose-induced aging mice. Chin J Integr Med. 2017. doi: 10.1007/s11655-016-2631-x [DOI] [PubMed] [Google Scholar]