Abstract
Amyotrophic lateral sclerosis (ALS) is a rare, devastating disease, causing movement impairment, respiratory failure and ultimate death. A plethora of genetic, cellular and molecular mechanisms are involved in ALS signature, although the initiating causes and progressive pathological events are far from being understood. Drosophila research has produced seminal discoveries for more than a century and has been successfully used in the past 25 years to untangle the process of ALS pathogenesis, and recognize potential markers and novel strategies for therapeutic solutions. This review will provide an updated view of several ALS modifiers validated in C9ORF72, SOD1, FUS, TDP-43 and Ataxin-2 Drosophila models. We will discuss basic and preclinical findings, illustrating recent developments and novel breakthroughs, also depicting unsettled challenges and limitations in the Drosophila-ALS field. We intend to stimulate a renewed debate on Drosophila as a screening route to identify more successful disease modifiers and neuroprotective agents.
Keywords: Experimental animal models, Genetic modifiers, Neuroprotection, Therapeutics
Introduction
The rare, devastating amyotrophic lateral sclerosis (ALS) or Lou Gehrig’s disease causes movement impairment, respiratory failure and ultimate death. Epidemiology studies establish that most ALS cases (about 90%) are classified as sporadic ALS (sALS) with no clear genetic linkage [1], and that autosomal dominant (the most diffuse), autosomal recessive, and X-linked inherited forms of familial ALS (fALS) can all co-exist and show mutations in more than 20 different genes [2–4]. The relationship among the genetic, clinical phenotypes and pathological subtypes of ALS has become clearer in the past years, although the shared contribution of both genetic and environmental factors is not defined yet.
There is currently no cure for the disease, and treatments can only alleviate symptoms, prevent complications, possibly slow disease progression, and perhaps offer social and emotional support [5].
Despite much knowledge has been obtained up till now in dissecting the mechanisms of ALS [4, 6], scientists are still debating for instance about the pathogenic initiators versus the propagators of the disease, and if there is a primum movens, or simply the summation of multiple harmful events are causative of ALS. In this increasingly complex scenario, there is urgent need for genetic modifiers and efficacious therapeutics available to patients.
Drosophila responds well to this quest, thanks to its powerful genetic manipulation and forthright phenotypic impact. The reasons why the invertebrate Drosophila melanogaster has been successfully used in research for over a century are: (i) the fruit flies are easy to maintain if compared to animal models requiring controlled animal facilities; (ii) a vast progeny resource is obtained in a limited time, because the fruit fly breeds quickly and lays many eggs. In addition, Drosophila genome shares about 60% homology with the roughly 46,830 human protein- and regulatory RNA-coding genes [7]; nearly 50% of the fly protein sequences have mammalian homologs, and approximately 75% of the known human disease genes have a recognizable equivalent in Drosophila [8], thus enabling cross-investigations into genetic inheritance and pathology [9]. This adds to the enormous experimental advantages deriving from the marginal logistic and cost-effective requirements for Drosophila maintenance and, most importantly, from the avoidance of all ethical issues surrounding many animal models [10]. As such, Drosophila becomes a timely and relevant topic of discussion in the ALS field.
Experimental and clinical ALS findings
Classical ALS (Charcot type) is diagnosed after the recognition of both upper and lower motor neuron symptoms that occur concomitantly and with a worsening symptomatology, but that exclude other pathologies explaining the same symptoms. Muscle’s weakness extended to all skeletal muscles and paralysis regionally spreading over time, are universally experienced in all patients, although the rate of ALS progression can be quite variable from one person to another [11]. Upper motor neuron symptoms include hyper-reflexia, increased muscle tone and slowing of fast voluntary movements. Lower motor neuron signs comprise weakness, muscle wasting and fasciculation. In approximately 65% of patients, ALS initiates with weakness in limb muscles and consequent voluntary movement impairment (characterized by tripping, grasping difficulties, dropping things, abnormal fatigue, cramps and twitches); in the remaining 35%, with weakness in bulbar muscles and consequent disarticulation of speech, changes in vocal tone, swallowing difficulties [12]. Rarely, disease presentation includes weakness in respiratory or axial muscles [13]. In about 50% of affected individuals, the neurodegenerative course spreads to the frontal and anterior temporal lobes, causing a variable extent of executive dysfunctions, language impairments and humoral changes with uncontrollable periods of laughing or crying. Approximately 10% of patients develop frontotemporal dementia (FTD) by degeneration of cortical neurons in the frontal and anterior temporal lobes [14, 15]. Since ALS hits only motor neurons, the senses of sight, touch, hearing, taste, and smell are not affected, and for many people also, muscles of the eyes and bladder are often spared. The clinical manifestation of ALS is extremely variable in terms of site and age at onset, disease progression, proportion of upper versus lower motor neuron involvement, and rate of FTD incidence. Even in families with a monogenic cause of ALS, disease presentation is unpredictable, suggesting the existence of important disease-modifying factors [16, 17]. The adverse effects of ALS on respiratory muscles limit the survival to 2–5 years after disease onset, but many people can live 5, 10 or even more years.
In sALS and fALS, the initial onset is given by cellular and molecular changes that most likely precede the clinical onset. A motor neuron begins to degenerate when the accumulated molecular pathology exceeds a certain, still undefined, threshold. Then, symptoms are triggered by focal neuronal changes that can either occur in every motor neuron, or be limited to the affected motor neuron. Regional spread of symptoms is in turn caused by summation of degenerating neurons. ALS warning signs are characterized by damage to neuromuscular junction (NMJ), demyelination, axonal retraction, and loss of motor neuron cell bodies, in addition to the occurrence of astrogliosis, microgliosis and neuro-inflammation [18]. Ubiquitin-positive inclusions and enhanced axonal outgrowth and dendritic branching are instead observed in the surviving neurons [19]. The direct involvement of skeletal muscle is the subject of several studies, although its participation to the neuro-degenerative process is still discussed. There is compelling evidence proposing that ALS muscles suffer from oxidative stress, mitochondrial dysfunction, and bio-energetics disturbances, and the way by which different types of myofibers are affected might depend on their contractile and metabolic features [20]. Although the immune system components play a true role in early stages of ALS, more focused studies are required to determine how, when, and where the immune and inflammatory processes are crucial to disease progression [21–25].
Advantages and limitations of promoting ALS research through Drosophila
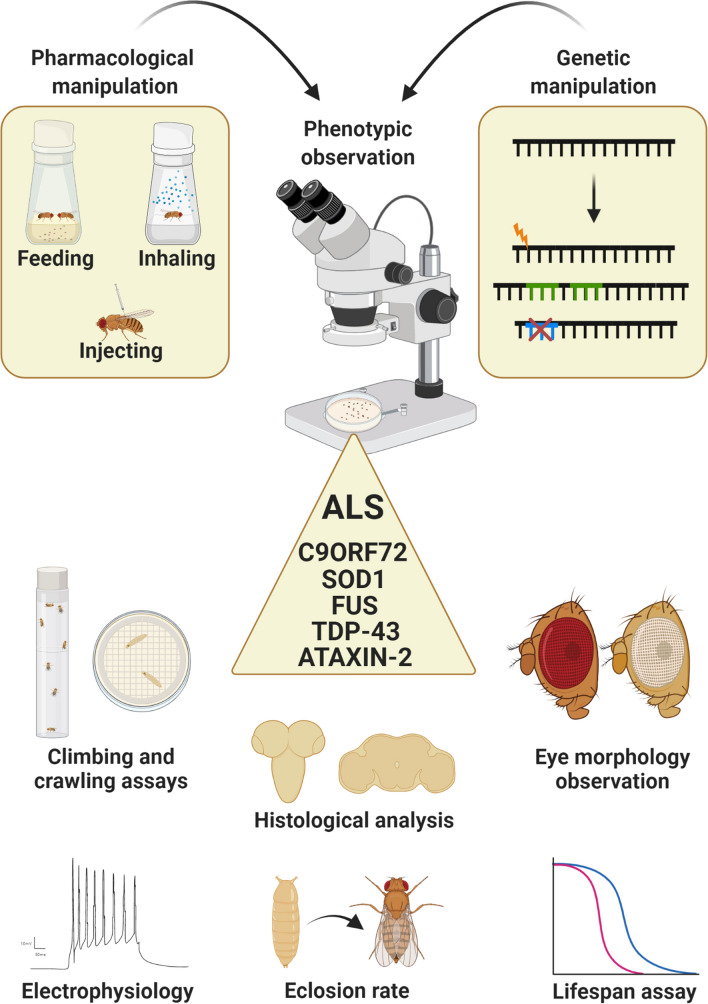
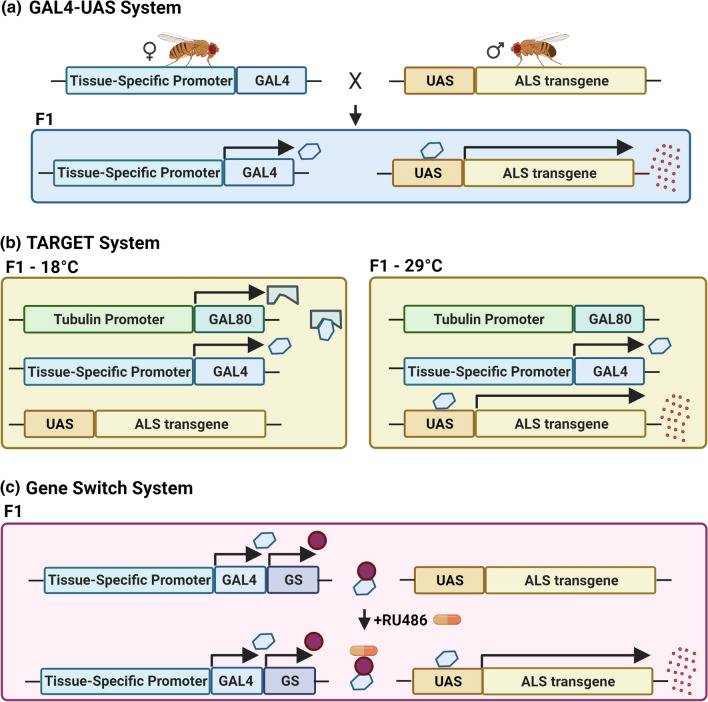
At present, Drosophila is extensively used as a genetic model for ALS [26, 27], providing valuable mechanistic information on disease insurgence and progression. What makes Drosophila an excellent ALS organism is the straightforward genetic manipulation, the multiple ways of drug delivery (by feeding, injection, inhaling), the different available modalities for testing disease progression (larval crawling and adult fly climbing tests, eclosion rate, lifespan assay, phenotypical observations, electrophysiological screenings) and, most of all, the quite faithful correspondence between what observed in ALS patients and what reproduced in flies in terms of survival, motor disabilities, motor neuron degeneration, presence of cellular inclusions, mitochondrial abnormalities and oxidative damage [28] (Fig. 1). The steps for studying ALS in Drosophila are to generate pathogenic models through available genetic techniques [29], to monitor disease progression, to screen putative disease modifiers and therapeutic compounds. Remarkably, Drosophila can further allow a precise and spatio-temporally controlled expression of human transgenes, thanks to fine-tuned transgenesis programs, such as the Gal4-Uprstream Activating Sequence (Gal4-UAS) [30], the temporal and regional gene expression targeting (TARGET), or the gene-switch systems [31] (Fig. 2).
Fig. 1.
Drosophila as a magnifying tool for dissecting ALS features. An important advantage of fly modeling is the possibility to evaluate the effectiveness of pharmacological treatments and genetic manipulation through different experimental read-outs
Fig. 2.
Graphical scheme of the available genetic systems to ectopically express human transgenes in Drosophila. Drosophila modeling allows spreading the expression of a transgene exclusively within a specific tissue, thanks to different transgenesis programs. a The bipartite Gal4-UAS system is characterized by the use of two lines of flies: the first one carrying the yeast transcription factor GAL4 under the control of a tissue-specific (or stadium-specific) promoter, and the second one carrying the transgene downstream the UAS domain (the GAL4 binding site). By crossing these two lines, a progeny (F1) expressing the transgenic protein only in the tissues where the GAL4 is active will be generated. b The temporal and regional gene expression targeting (TARGET) system permits a fine-tuned temporal control of the expression, in a more precise manner than Gal4/UAS. The ubiquitous tubulin promoter spreads the expression of a temperature-sensitive Gal80 that, at 18 °C, binds the Gal4 and blocks the transgene transcription. Shifting the temperature at 29 °C determines the Gal4 deliverance and its interaction with UAS sequences, thus allowing the transgene production. c The Gene Switch system is characterized by a hormone-inducible Gal4: feeding flies with a nutritional medium supplemented with RU486 (mifepristone) determines the Gal4 activation and consequently the initiation of the transcriptional process
Of note, the Drosophila visual system assumes a great relevance in fruit fly modeling, because about two-thirds of the vital genes in the Drosophila genome are involved in the eye development [32]. This makes the fly’s eye a powerful genetic tool to study patterning, growth, survival, neurodevelopment, neuro-degeneration and, not least, complex diseases comprising ALS. The compound eye of Drosophila consists of approximately 800 units (ommatidia), each containing eight photoreceptor neurons (R1-R8), two primary pigment cells and four lens cells. Ommatidia are separated from one another by a slight septum composed of six secondary pigment cells, three tertiary pigment cells, and three bristle complexes [33, 34]. Any subtle insult modifying the geometry of this precisely ordered structure, leads to a visible aberrant morphological phenotype, such as smaller or larger eyes, changes in ommatidia, changes in bristles, and loss of pigmentation. Consequently, the introduction of a putative genetic modifier in an altered eye genetic background may suppress or enhance the disease eye phenotype, and therefore provide interesting evidence for the involvement of a new player in the pathogenic scenario.
Despite these several advantages, we must be aware that some limitations about Drosophila modeling exist too. The anatomy of the brain and other major organs within the fruit fly are quite different from that of humans, and there is lack of adaptive immune system. Other issues can be related to behavioral studies, because in-depth cognitive abilities are absent in the fly. Not least, some drug effects are sometimes reported to be different in the fly compared to human, suggesting that future directions for preclinical drug development must also consider comparative analysis in different animal models. However, we believe that Drosophila remains an invaluable organism for performing rapid and cost-effective screening particularly of genetic modifiers and putative neuroprotective agents, and for dissecting the genetics and mechanistic pathways of ALS. The search for novel diagnostic tools and breakthrough therapeutics will surely rely on Drosophila to empower effective solutions for ALS patients.
ALS disease modifiers identified in Drosophila models
Considering that the main pathological traits of ALS may be easily recapitulated in Drosophila, several transgenic fly models have been successfully generated [27, 35] and used to screen putative genetic modifiers and molecules with a potential neuroprotective action [36]. In the next sections, we will present a detailed updated view of different ALS modifiers, able to suppress the neurodegenerative phenotype induced by the expression in Drosophila of C9ORF72, SOD1, FUS, TARDBP or Ataxin-2 transgenes, together representing about 70% of all fALS cases.
C9ORF72-ALS modifiers
Chromosome 9 Open Reading Frame 72 (C9ORF72) gene, the most frequent fALS causative gene [37, 38], is composed of 12 exons, two of which are non-coding. C9ORF72 protein has been recently described as an autophagy regulator and component of guanine nucleotide exchange factor complex [39, 40]. Within the C9ORF72 first intron, the hexa-nucleotide GGGGCC (G4C2) may be repeated from 2 to 23 times in wild-type gene [41], but its aberrant expansion reaching hundreds or thousands of repeats, has been found in ALS and FTD patients [42–44]. A consistent group of evidence shows that higher is the number of repeats above the 23-threshold, more severe is the phenotypical alteration [45–51].
The proposed and non-mutually exclusive mechanisms underlying C9ORF72-ALS pathogenesis are [52]: (i) RNA-mediated toxicity by direct sequestration of RNA-binding proteins (RBP); (ii) production through Repeat-Associated Non-ATG (RAN) translation of toxic dipeptide repeats (DPRs) that accumulate in the cytoplasm and become cytotoxic [53–56]; (iii) loss-of-function mechanisms determining a decrease of C9ORF72 protein [57, 58]. By considering that the Drosophila genome has no ortholog for C9ORF72 [59], the loss-of-function effect cannot be studied in the fly.
Concerning the RNA toxicity aspect, it is well known that expanded and GC-rich transcripts are prone to form secondary structures, such as R loops and G-quadruplexes that normally antagonize the RNA Polymerase II transcription process [60]. Recent papers highlighted the role of DRB-sensitivity-inducing factor (DSIF) and of polymerase-associated factor 1 (PAF1C) complexes. The authors showed that both facilitate the transcription machinery and promote the expression of G4C2 repeats, by resolving RNA secondary structures [48, 51, 61]. In particular, the downregulation of a core component of DSIF complex [62] partially rescues eye degeneration and increases lifespan in a fly model expressing 49 repeats of G4C2 hexa-nucleotide [48]. Likewise, reduced expression of Drosophila PAF1C components modulates toxicity at various levels: C9ORF72-flies carrying RNAi construct for PAF1C subunits show extended lifespan, better climbing performance, rescued eye phenotype and reduced presence of brain vacuoles [51]. Still focusing on the RNA toxic role, other results showed that targeted overexpression in fly eyes or motor neurons (by tissue-specific GAL4 drivers) of the RBP Purα rescues the neurodegenerative phenotype [45]. Consistently, Celona and collaborators report that the overexpression of the RBP Zfp106, suppresses hexa-nucleotide repeat expansion (HRE)-induced neurotoxicity in a Drosophila C9ORF72-ALS model expressing a (G4C2)30 construct in glutamatergic neurons [63]. Recently, also the ALS-associated RBP Matrin-3 was described as an in vivo modulator of C9ORF72-ALS pathogenesis; its overexpression, indeed, is able to rescue eye neurodegeneration, lifespan and motor performance in a fly model carrying a (G4C2) expansion [64]. Remarkably, removing the RNA-recognition-motif (RRM) domain from Matrin-3 nullifies the neuro-protective effect. Taken together, these results confirm that modulating the transcriptional process of RNA, or overexpressing its trapped interactors, may become a promising target to treat C9ORF72-ALS. In a recent paper, Jiao and co-workers reported that the enzyme Topoisomerase 2 (Top2) may be considered as a newcomer in the ALS modifier field. They showed that pharmacologically reducing Top2 expression ameliorates G4C2-induced neurotoxicity, in a fly model of C9ORF72-ALS [65], thus unveiling another potential ALS therapeutic target.
As known, G4C2 HRE may be transcribed in both sense and anti-sense directions and then translated in five toxic DPRs: poly-GA (glycine–alanine), poly-GR (glycine–arginine), poly-PR (proline–arginine), poly-PA (proline–alanine), and poly-GP (glycine–proline) [55, 56, 66]. By exploiting the Drosophila eye as screening tool, Lee and colleagues, performed an extended in vivo RNAi analysis and identified 80 suppressors and 27 enhancers of C9ORF72-induced toxicity [49], thus providing a strong evidence of DPR involvement in C9ORF72-ALS pathogenesis. Although their involvement in RAN translation mechanism is still to be clearly defined, canonical translation factors eIF4B and eIF4H were identified as modifiers of DPR-induced toxicity: their downregulation leads to a reduced production of toxic peptides in a C9ORF72-ALS fly model [67].
Several recent reports showed that DPR-induced toxicity is limited to arginine-rich DPRs [46, 47, 49, 68–71]; e.g., Mizielinska and collaborators showed that only poly-(GR)36 and poly-(PR)36 constructs, individually expressed in the Drosophila eye or motor neurons, lead to neurodegeneration, but not poly-(GA)36 and poly-(PA)36 [46]. Likewise, Freibaum and co-workers reported that poly-(GP)47 and poly-(GA)50 do not contribute to any degenerative phenotype, when ectopically expressed in flies [47]. In contrast, some recent papers highlighted that Drosophila “short repeats models” could not be totally informative about the disease mechanism, considering that ALS/FTD patients’ expansion is often greater than 500 repeats [72–74]. On this context, West and co-workers generated a Drosophila model carrying more than 1000 toxic repeats. They showed that each of the five DPRs has its unique pathological profile and contributes to neurodegeneration in a specific way. Authors revealed, moreover, that co-expressing specific DPRs determines new phenotypes not detected when pathogenic constructs are expressed one by one [75].
Arginine-rich DPRs were reported to impair Notch signaling and cause cytoplasmic aggregates [69]. Their expression in Drosophila glutamatergic neurons causes neurodegeneration and excitotoxicity with increased intracellular calcium and extracellular glutamate levels in the brain [76]. The inhibition of NMDA receptors in DPR-expressing glutamatergic neurons extends lifespan and rescues motor defects in Drosophila [76]. Considering that riluzole, one of the two approved drugs for ALS treatment [77], is an anti-glutamatergic agent, this study strongly supports the arginine-rich-DPRs/glutamatergic axis and its therapeutic value for C9ORF72-ALS treatment.
An additional cellular process increasingly involved in the C9ORF72-ALS scenario is the nucleocytoplasmic transport (NCT): consistent reports indicate the possibility of modifying C9ORF72-induced toxicity by modulating the expression of proteins belonging to this trafficking pathway. Of note, through a large-scale deficiency screening in a Drosophila model expressing a (G4C2)58 transgene in the eye, Freibaum and colleagues found that many components of NCT suppress or enhance eye ommatidia deregulation [47]. Likewise, through a RNAi screening in a C9ORF72 Drosophila model expressing a construct with 25 PRs in the eye, Boeynaems and colleagues found that modulation of importins, exportins and other nuclear pore components, improves C9ORF72-ALS-induced altered eye phenotype [78]. Consistently, Drosophila RanGAP (human RanGAP1 ortholog) overexpression, or pharmacological treatment with the nuclear export inhibitor KPT-276, rescues the neurodegeneration in a (G4C2)30 fly model [79]. The same group recently proposed that NCT disruption triggers autophagy dysfunction, leading to chronic protein stress and neuronal death. C9ORF72-mediated neurodegeneration is rescued by nuclear import of the autophagy-factor Mitf/TFEB [80]. Furthermore, increased cytosolic calcium levels have been recently reported to be crucial to regulate TDP-43 NCT and reduce its aggregation in a C9ORF72-fly model, introducing the calcium-Calpain A-Importin α3 axis as a new potential therapeutic target [81]. Interesting insights came recently from Lee and colleagues about the molecular chaperone Sigma-1 receptor, whose mutations have been already linked to fALS [82–85]. They demonstrated that Sigma-1 receptor presence at nuclear pore (it co-localizes with endogenous RanGAP and nucleoporins Nup62 and RanBP2) counterbalances G4C2-HRE toxic effect and that its overexpression rescues eye defects, aberrant motor behavior and electrophysiological deficits in a Drosophila (G4C2)30 ALS model [86].
The HRE- or DPR-mediated NCT disruption unavoidably causes an abnormal reallocation of several proteins within the nuclear and cytoplasmic compartments; recently, Ortega and collaborators identified many proteins that in C9ORF72-ALS scenario are shifted in the cytosolic fraction. Among these, the translation termination and nonsense-mediated decay (NMD) regulator eRF1 resulted to have the strongest neuroprotective effect in a (G4C2)36 fly model: the overexpression of its ortholog ETF1 rescues eye depigmentation and reduces poly-GR DPR levels [87]. This finding strengthens the NMD pathway as a potential ALS therapeutic target, as also confirmed by a recent group of evidence reporting UPF1 (the master regulator of NMD) as a strong in vivo modulator of C9ORF72-induced neurotoxicity [87–90].
Remarkably, many of the above-described results (Table 1) were validated for the first time in a fly model, confirming Drosophila as an ideal tool to dissect molecular mechanisms, identify putative genetic modifiers, and assess the contribution of different cellular patterns in a multifaceted disease like ALS.
Table 1.
Disease modifiers identified in Drosophila C9ORF72-ALS models
| Disease modifier | Fly transgenic construct | Phenotypic rescue | Refs. |
|---|---|---|---|
|
DSIF complex downregulation |
GMR-Gal4 > UAS(G4C2)49;Stp4RNAi | Eye morphology, lifespan | [48] |
|
PAF1C complex downregulation |
GMR-Gal4 > UAS(G4C2)49;dPaf1RNAi Elav-Gal4 > UAS(G4C2)49;dPaf1RNAi |
Brain vacuoles, eye morphology, motor behavior, lifespan | [51] |
|
Purα overexpression |
GMR-Gal4 > UAS-(G4C2)30;UAS-Purα | Eye morphology | [45] |
|
Zfp106 overxpression |
Oak-Gal4 > (G4C2)30;UAS-Zfp106 | Motor behavior, NMJ | [63] |
|
Matrin-3 overexpression |
GMR-Gal4 > UAS-(G4C2)30;UAS-Matr3 GMR-Gal4 > UAS-(G4C2)36;UAS-Matr3 GMR-Gal4 > UAS-(G4C2)58;UAS-Matr3 |
Eye morphology | [64] |
| ElavGS-Gal4 > UAS-(G4C2)30;UAS-Matr3 | Motor behavior, lifespan | ||
|
Topoisomerase 2 downregulation |
GMR-Gal4 > UAS-(G4C2)30;Top2RNAI | Eye morphology | [65] |
| Teniposide |
GMR-Gal4 > UAS-(G4C2)30 OK371-Gal4 > UAS-(G4C2)30 |
Motor behavior | |
| Genistein |
GMR-Gal4 > UAS-(G4C2)30 OK371-Gal4 > UAS-(G4C2)30 |
Motor behavior | |
|
eIF4B downregulation |
GMR-Gal4 > LDS(G4C2)EXP;UAS-eIF4BRNAi | Eye morphology | [67] |
|
eiF4H downregulation |
GMR-Gal4 > LDS(G4C2)EXP;UAS-eIF4HRNAi | Eye morphology | |
|
vGlut downregulation |
D42-Gal4 > UAS-GR36;vGLUTRNAi | Motor behavior, lifespan | [76] |
|
Nuclear components modulation |
GMR-Gal4 > (PR)25 | Eye morphology | [78] |
|
KPT-276 treatment |
GMR-Gal4 > UAS(G4C2)30 | Eye morphology | [79] |
|
RanGap overexpression |
GMR-Gal4 > UAS(G4C2)30;UAS-RanGap | Eye morphology | |
|
Mitf genomic duplication |
GMR-Gal4 > UAS-30R;Mitf(Dp) ElavGS > UAS-30R;Mitf(Dp) vGlut-Gal4 > UAS-30R;Mitf(Dp) |
Eye morphology, eclosion rate, motor behavior, autophagolysosomal pathway | [80] |
|
Increased cytosolic calcium concentration |
ppk1a-Gal4 > UAS-SERCARNAi; UAS-Imp a3RNAi;UAS-TBPH-Flag-HA |
TBPH aggregates | [81] |
|
Sigma-1 receptor overexpression |
GMR-Gal4 > UAS(G4C2)30;UAS-Sig-1R Elav-Gal4 > UAS(G4C2)30;UAS-Sig-1R |
Eye morphology, motor behavior | [86] |
|
eRF1/ETF1 overexpression |
GMR-Gal4 > UAS-(G4C2)36;ETF1OE | Eye morphology, Poly-GR levels | [87] |
|
UPF1 overexpression |
GMR-Gal4 > UAS-(G4C2)30;UAS-UPF1 GMR-Gal4 > UAS-(G4C2)36;UAS-UPF1 |
Eye morphology | |
|
UPF1 overexpression |
D42-Gal4 > UAS-GR36;UAS-UPF1 | Lifespan, motor behavior | [88] |
| Tranilast |
Elav-Gal4 > UAS-GR36 Elav-Gal4 > UAS-PR36 |
Lifespan, motor behavior | [88] |
|
UPF1 overexpression |
GMR-Gal4 > UAS-(G4C2)30;UAS-UPF1 | Eye morphology | [90] |
|
Phospholipase D pathway modulation |
GMR-Gal4 > UAS(G4C2)30 | Eye morphology | [91] |
SOD1-ALS modifiers
Superoxide dismutase 1 (SOD1) is an evolutionarily conserved ubiquitous protein catalyzing dismutation of superoxide into hydrogen peroxide and dioxygen. In 1993, SOD1 was identified as the first gene whose mutations were linked to ALS [92, 93]; to date, more than 200 different point mutations of the SOD1 gene have been related both to fALS and sALS [94, 95]. These mutations cause deregulation of cellular pathways by combination of loss and gain of toxic functions [96–99], all leading to the damage of motor neurons as main crucial feature. One of the most prominent pathological mechanisms of SOD1-related ALS is an above-threshold presence of oxidative stress caused by unprocessed free radicals and high production of reactive oxygen/nitrogen species [100].
During the last twenty-five years, different Drosophila transgenic models have been developed for studying pathogenic mechanisms linked to human SOD1 gene mutations [101–103], all sharing reduced climbing abilities, increased SOD1 protein aggregation and mitochondrial dysfunctions in motor neurons [102, 103].
Drosophila carrying the human SOD1 transgene with the missense mutation G85R (SOD1-G85R) [102], highlighted the potential neuroprotective effect of several different antioxidant molecules (Table 2). In detail, De Rose and collaborators demonstrated that adult flies expressing the SOD1-G85R transgene in motor neurons show higher survival rate and better motor performance if extracts of Withania somnifera (a plant with antioxidant, anti-inflammatory properties) are added to standard nutritional medium [104]. Interestingly, W. somnifera is protective in a loss-of-function TDP-43 fly model too, determining a partial rescue of climbing and walking activity [105]. Moreover, urate treatment enhances survival, attenuates motor impairments, reduces oxidative damage and increases antioxidant defense in human SOD1-G85R Drosophila [106]. Interestingly, the antioxidant α-lipoic acid exerts neuroprotection in ALS flies expressing SOD1-G85R in motor neurons, by extending survival rate, rescuing motor impairment, activating the ERK/Akt pathway and indirectly regulating the expression of antioxidant enzymes [107].
Table 2.
Disease modifiers identified in Drosophila SOD1-ALS models
| Disease modifier | Fly transgenic construct | Phenotypic rescue | Refs. |
|---|---|---|---|
| Withania somnifera extracts | D42-Gal4 > hSOD1G85R | Motor behavior, lifespan | [104] |
| Urate | D42-Gal4 > hSOD1G85R | Motor behavior, oxidative stress, lifespan | [106] |
| α-lipoic acid | D42-Gal4 > hSOD1G85R | Motor behavior, lifespan | [107] |
| Fisetin | D42-Gal4 > hSOD1G85R | Motor behavior, SOD1 aggregates | [108] |
| γ-oryzanol | D42-Gal4 > hSOD1G85R | Motor behavior, oxidative stress, lifespan | [109] |
The natural antioxidant fisetin extends lifespan, improves climbing activity and activates the ERK pathway in SOD1 mutant flies. Fisetin-treated flies have less SOD1 aggregates in brain respect to untreated flies, and the hypothesis was formulated that fisetin may regulate autophagy in ALS pathogenesis [108].
Recently, Zhang and collaborators showed that γ-oryzanol ameliorates ALS symptomatology in SOD1-G85R flies by reducing oxidative stress and free radicals damage, and sustaining lifespan and motor abilities [109].
FUS-ALS modifiers
Fused-in-Sarcoma/Translocated-in-Liposarcoma (FUS/TLS) is a multi-functional DNA/RNA-binding protein found in RNA-containing stress granules (SG) [110–112], regulating gene expression [113], RNA metabolism [114–116] and splicing [117, 118]. Only recently, FUS was described as a bicistronic gene encoding for an alternative peptide (altFUS), whose suppression is neuroprotective in a Drosophila FUS-overexpressing model [119].
ALS-linked FUS mutations were identified in 2009 [120, 121] and over the years, different FUS-related Drosophila models have been generated [122–124], all sharing ALS pathogenic hallmarks. Ectopic expression of wild-type or mutant FUS, triggered by a tissue-specific Gal4 driver, has been reported to impact on fly motor behavior, lifespan, ommatidial morphology and eclosion rate [122, 125–130]. Consistently, different mutations in Drosophila FUS ortholog, cabeza (caz), determine reduction of lifespan, locomotor abnormalities and reduced eclosion rate [126, 131, 132]; these defects are rescued by introducing a wild-type human FUS in the mutated background [126], thus highlighting the high level of conservation between caz and FUS genes.
To date, many suppressors of FUS-induced toxicity have been identified in fly models (Table 3). For instance, overexpression of ter94, fly ortholog of Valosin-containing protein (VCP) gene and ALS-causing itself [133], rescues motor neuron defects in a caz-knockdown background, whilst ter94-inactivation exacerbates the neurodegenerative phenotype [134]. Interestingly, further evidence of the interaction between FUS and VCP has been recently reported in a human iPSC cellular line obtained from VCP-mutant motor neurons, where FUS mis-localization was found [135].
Table 3.
Disease modifiers identified in Drosophila FUS-ALS models
| Disease modifier | Fly transgenic construct | Phenotypic rescue | Refs. |
|---|---|---|---|
|
altFUS suppression |
Elav-GS > UAS-altFUSØ | Motor behavior | [119] |
|
ter-94 overexpression |
GMR-Gal4 > UAS-Caz-IR363-399;UAS-ter94 | Motor behavior, lifespan | [134] |
|
Phospholipase D pathway modulation |
GMR-Gal4 > UAS-FUSR521C | Eye morphology | [91] |
|
Drosophila muscleblind downregulation |
GMR-Gal4 > FUS;mblRNAi D42-Gal4 > FUS;mblRNAi |
Eye morphology, motor behavior, NMJ | [136] |
|
Drosophila Rm62 overexpression |
GMR-Gal4 > UAS-FUS; Rm62OE GMR-Gal4 > UAS-FUSR521C; Rm62OE GMR-Gal4 > UAS-FUSR518K; Rm62OE |
Eye morphology | [143] |
|
ElavGS-Gal4 > UAS-FUS; Rm62OE ElavGS-Gal4 > UAS-FUSR521C; Rm62OE ElavGS-Gal4 > UAS-FUSR518K; Rm62OE |
Motor behavior | ||
|
Nucleoporin 154 downregulation |
CCAP-Gal4 > UAS-FUS;Nup154RNAi | FUS-induced neurotoxicity | [129] |
|
Exportin 1 downregulation |
CCAP-Gal4 > UAS-FUS;Xpo1RNAi | FUS-induced neurotoxicity | |
|
Nucleoporin 62 downregulation |
D42-Gal4 > UAS-FUSR521C; Nup62RNAi D42-Gal4 > UAS-FUSR518K; Nup62RNAi ElavGS-Gal4 > UAS-FUSP525L; Nup62RNAi |
Nuclear abnormalities, lifespan | [145] |
|
Hippo downregulation |
GMR-Gal4 > UAS-Caz-IR;hpoKS240 Elav-Gal4 > UAS-Caz-IR;hpoKS240 |
Eye morphology, motor behavior | [147] |
|
Hippo downregulation |
GMR-Gal4 > UAS-FUS;hpo GMR-Gal4 > UAS-FUS;jun |
Eye morphology | [148] |
|
Parkin overexpression |
GMR-Gal4 > UAS-FUS;UAS-parkin Elav-Gal4 > UAS-FUS;UAS-parkin |
Eye morphology, motor behavior | [149] |
Recently, Kankel and colleagues performed some independent modifier screening experiments using two different fly models expressing the fALS causing mutant transgenes FUS (missense mutation R521C) and TDP-43 (missense mutation M337V). By analyzing the eye phenotype of flies co-expressing the putative modifier together with the fALS transgenes, they identified a complex array of ALS phenotype enhancers and suppressors, many of which affecting both FUS- and TDP-43-expressing strains. Interestingly, the strongest genetic modifiers of both FUS and TDP-43 toxicity were tested on a third ALS model, expressing a (G4C2)30 construct. A cohort of genes with effects on diverse ALS models were found, opening the possibility to identify relevant genes or pathways shared by different ALS forms [91]. Of note, the Authors identified the phospholipase D pathway as one of the major modifiers of ALS phenotypes, by validating its positive effects not only in multiple fly models, but also in SOD1-G93A mice [91]. Another unbiased genetic screening highlighted muscleblind Drosophila gene as a modifier of FUS-induced toxicity: its functional inactivation in a mutant FUS background rescues ommatidia defects, improves motor abilities and recovers NMJ defects [136]. Of note, muscleblind human ortholog, MNBL1 gene, affecting RNA trafficking, splicing and processing has been previously linked to several neurodegenerative disease [137–140]. Very recently, through a RNA-sequencing approach, Fortuna and co-workers identified the RNA helicase DDX17 (DEAD-Box Helicase 17), whose activity is necessary for transcription and splicing processes [141, 142], as a new modulator of FUS-induced toxicity. In particular, the overexpression of Rm62, the DDX17 Drosophila ortholog, ameliorates eye degeneration and climbing performances in wild-type and mutant FUS expressing flies [143]. Additionally, authors unveiled the role of DDX17 in DNA damage response pathway, thus presenting DSB repair as a new potential therapeutic target for FUS-induced ALS treatment.
As for C9ORF72-ALS, mounting evidence suggests a pivotal role for NCT in FUS-ALS too [144]. Downregulation of Nucleoporin 62, Nucleoporin 154 and of Exportin 1 (key modulators of nuclear export) indeed reduces FUS-induced toxicity in a FUS-overexpressing fly model [129, 145].
A recently discovered caz modulator is Hippo, the fly ortholog of Mammalian Sterile 20-like kinase 1, whose pathway is involved in tumor suppression [146]. Caz downregulation-induced defects in motor neurons are suppressed by introducing loss-of-function mutations of Hippo [147]. Similarly, flies expressing a wild-type or mutant human FUS transgene in the eye show a rescue of neurodegeneration if components of Hippo or c-Jun N-terminal Kinase (JNK) signaling pathways are modulated [148], thus suggesting these pathways as new potential therapeutic targets for FUS-ALS treatment.
Not least, a recent result shows that the E3 ubiquitin ligase Parkin exerts a neuroprotective effect in a Drosophila model overexpressing FUS in muscle tissues: it is peculiar that Parkin expression does not directly modulate FUS protein levels, but rescues the pathological phenotype recovering mitochondrial defects caused by FUS proteinopathy [149].
TDP-43-ALS modifiers
TAR-binding protein 43 (TDP-43) is a 43 KDa RBP involved in mRNA stability [150], miRNA processing [115, 151] and splicing regulation [152]. It has been linked to ALS because it was found as a core component of neuronal inclusion bodies in ALS patients [153–155]. Over the years, 48 different point mutations in the TARDBP gene encoding for TDP-43 have been identified as ALS-causing [156]. Contextually, many Drosophila models have been generated. Targeted overexpression of wild-type or mutant human TDP-43 causes reduced lifespan, eclosion failure, impaired motor functions, axon swelling and cytoplasmic toxic aggregates [157–163]. Similar phenotypes are obtained by overexpressing TAR DNA binding homolog (TBPH) gene, TARDBP Drosophila ortholog, which indeed determines climbing defects, cytoplasmic accumulations and eclosion failure [164, 165]. Moreover, loss of TBPH causes defective motor behaviors and eclosion defects, abnormalities at NMJ and reduced lifespan: these symptoms are rescued by human TDP-43 expression [164, 166], suggesting not only the high evolutionary conservation of TDP-43 [167], but also that any positive or negative deviation from a threshold expression of TBPH might induce ALS-like features in Drosophila. Interestingly, Romano and collaborators showed that TBPH depletion either in neurons or glia impairs the organization of glutamate GluRIIA receptors at the NMJ, supporting the hypothesis of not-only-neuronal origin of TDP-43-ALS [168–170].
It is not surprising that FUS and TDP-43 induce neurodegeneration through co-incidental processes, and the effects of their overexpression in Drosophila are similar, given their functional similarity. Different studies demonstrated not only their interaction [125, 171, 172], but also TDP-43 capability to act as FUS-induced toxicity enhancer in flies [125, 126].
In the last decade, different TDP-43 fly models have been used to perform compound or genetic modifier screening (Table 4). Autophagy upregulation may be a useful therapeutic approach [173] and evidence showed that autophagy reactivation through rapamycin reduces toxic aggregation rate, improves lifespan and partially rescues motor impairments in a Drosophila model overexpressing TBPH in motor neurons [174]. Interestingly, also TBPH-deficient flies show better motor abilities and higher survival rate by powering up autophagy through overexpression of Autophagy Related 7 gene [175].
Table 4.
Disease modifiers identified in Drosophila TDP-43-ALS models
| Disease modifier | Fly transgenic construct | Phenotypic rescue | Refs. |
|---|---|---|---|
| Phospholipase D pathway modulation | GMR-Gal4 > UAS-TDP-43M337V | Eye morphology | [91] |
|
Withania somnifera extracts |
D42-Gal4 > UAS-TBPHRNAi | Motor behavior, lifespan | [105] |
| Rapamycin | D42-Gal4 > UAS-TBPH | Motor behavior, lifespan | [174] |
|
ATG7 overexpression |
Act-Gal4 > TBPHΔ23/Δ23;UAS-Atg7 | Motor behavior, lifespan | [175] |
|
Mitofusin overexpression |
Elav-Gal4 > UAS-TDP-43;UAS-Marf | Motor behavior | [179] |
|
Parkin overexpression |
Elav-Gal4 > UAS-TDP-43;UAS-Parkin | Motor behavior, lifespan | [180] |
|
PINK-1 downregulation |
Elav-Gal4 > UAS-TDP-43;PINK1RNAi | Motor behavior, lifespan | |
|
Futsch overexpression |
D42-Gal4 > UAS-TDP-43;P(EP)futschEP1419 | Motor behavior, NMJ, lifespan | [181] |
|
Cacophony overexpression |
Elav-Gal4 > UAS-Cacophony;TBPHDD96 D42-Gal4 > UAS-Cacophony;TBPHDD96 |
Motor behavior | [183] |
|
6-(3-(4-fluorobenzyl)-3-(hydroxymethyl) piperidin-1-yl)pyrazine-2-carboxamide |
D42-Gal4 > UAS-TDP-43 | Motor behavior | [185] |
|
Drosophila REF1 downregulation |
GMR-Gal4 > UAS-TDP-43; UAS-ATXN2-32Q;UAS-Ref1RNAi |
Eye morphology | [186] |
| Medium-chain fatty acids | D42-Gal4 > UAS-TDP-43 | Motor behavior | [190] |
|
Palmitoyltransferase downregulation |
D42-Gal4 > UAS-TDP-43;Cpt1RNAi D42-Gal4 > UAS-TDP-43;Cpt2RNAi |
Motor behavior | |
|
Glycolysis upregulation |
D42-Gal4 > UAS-TDP-43;UAS-Glut3 D42-Gal4 > UAS-TDP-43;UAS-PFK |
Motor behavior | [191] |
|
Drosophila Atx2 downregulation |
GMR-Gal4 > dAtx2X1 Elav-Gal4 > dAtx2X1 |
Eye morphology, lifespan | [197] |
|
PABP2 downregulation |
GMR-Gal4 > UAS-TDP-43;PABP2LOF GMR-Gal4 > UAS-TDP-43 D169G;PABP2LOF GMR-Gal4 > UAS-TDP-43 A315T;PABP2LOF |
Eye morphology (worsening) | [201] |
|
D42-Gal4 > UAS-TDP-43;PABP2LOF D42-Gal4 > UAS-TDP-43 D169G;PABP2LOF D42-Gal4 > UAS-TDP-43 A315T;PABP2LOF |
Motor behavior (worsening) | ||
|
ATXN2 PAM2 domain depletion |
GMR-Gal4 > UAS-TDP-43;UAS-ATXN2-32QΔPAM2 ElavGS-Gal4 > UAS-TDP-43;UAS-ATXN2-32QΔPAM2 |
Eye morphology, motor behavior, lifespan | [199] |
|
Poly-ADP-ribose polymerase downregulation |
GMR-Gal4 > UAS-TDP-43;PARPRNAi Elav-Gal4 > UAS-TDP-43;PARPRNAi |
Eye morphology, motor behavior, lifespan | [203] |
|
Tankyrase downregulation |
GMR-Gal4 > UAS-TDP-43;TkrsIR Elav-Gal4 > UAS-TDP-43;TkrsIR |
Eye morphology, lifespan | [202] |
Mitochondrial fragmentation is a prominent common feature of ALS [176–178]: flies pan-neuronally expressing TDP-43 show extremely small or fragmented mitochondria [179]. Mitofusin gene is a key regulator of mitochondrial fusion process and its mRNA and protein levels are reduced by TDP-43 overexpression: a reduced rate of fusion may determine mitochondrial fragmentation. Importantly, restoring mitofusin expression ameliorates spontaneous walking and climbing of wild-type or mutant TDP-43-expressing flies [179]. Recently, Sun and colleagues demonstrated that both Parkin and PINK1, master regulators of mitophagy, are deregulated by TDP-43 overexpression. Furthermore, they showed that upregulation of Parkin and downregulation of PINK1 delay climbing defects and extend lifespan in a fly model of TDP-43-ALS [180]. Taken together, these data suggest that mitochondrial dynamics, for many aspects, cover an important role in TDP-43-ALS pathogenesis and could become valuable target for therapeutic compounds.
Drosophila TBPH regulates the expression of several genes encoding for pre-synaptic terminal proteins influencing synaptic transmission, such as Futsch, Syntaxin 1A and Synapsin [166, 169]. In a Drosophila-ALS model expressing TDP-43 in motor neurons, a reduction of both futsch mRNA and protein was registered at the NMJ [181]. Remarkably, futsch ectopic expression rescues motor impairment, reduces TDP-43 aggregates, extends lifespan and recovers NMJ abnormalities [181]. An additional therapeutic target is suggested by a recent study on a Drosophila model of TDP-43 loss-of-function: cacophony is a gene directly regulated by TBPH [182] that encodes for a voltage-gated calcium channel, whose mRNA reduction is linked to TBPH loss. Restoring Cacophony levels in all neurons, or specifically in motor neurons, in a TBPH-/-background, leads to the rescue of motor disturbances caused by TBPH loss-of-function [183].
The interaction between TDP-43/TBPH and its RNA targets is crucial to determine the pathogenic effect of proteinopathy: interestingly, recent evidence demonstrates that removing TDP-43 RNA-recognition-motif (RRM) domains leads to reduction of toxic effects. Ihara and colleagues, for instance, have generated a fly ALS model expressing TDP-43 in retinal neurons, characterized by photoreceptor vacuolar degeneration, and thinning of the retina. ALS-induced altered eye phenotype is completely rescued by preventing the binding ability of TDP-43 through mutations or deletion in its RRM domains [184]. Likewise, a pharmacological approach produced the same result, thanks to in silico docking and biochemical assays, the compound 6-(3-[4-fluorobenzyl]-3-(hydroxymethyl)piperidin-1-yl)pyrazine-2-carboxamide was identified to reduce the TDP-43 ability to bind disease-linked nucleic acids and ameliorate motor capabilities in flies overexpressing wild-type or mutant TDP-43 in motor neurons [185]. Interestingly, also the downregulation of RNA export process was reported to be neuroprotective in multiple fly ALS models: for instance, the functional inactivation of Drosophila REF1, fly ortholog of human ALYREF mRNA exporting factor, is able to mitigate TDP-43, TDP-43/ATXN2-32Q and G4C2 neurotoxicity [186]. Specifically, REF1 knockdown suppresses fly eye neurodegeneration, reduces TDP-43 mRNA and protein levels, and G4C2 mRNA and poly-GA rates in their respective fly models [186].
Together with motor neuron damage, ALS patients exhibit bioenergetics deficits and hyper-metabolism [187–189]. A metabolomics study on wild-type and mutant TDP-43 overexpressing larvae presenting the common ALS phenotypical hallmarks, showed significant alteration in lipid metabolism and deficit in carnitine shuttle responsible for long-chain fatty acid import into the mitochondria, lipid beta-oxidation and ATP production [190]. Interestingly, feeding these flies with medium-chain fatty acids not needing the carnitine shuttle to reach the mitochondrial matrix, improves larval motor abilities. Moreover, downregulating the expression of the carnitine shuttle major components palmitoyltransferase 1 or 2, suppresses motor impairment [190]. The same group moreover proved that TDP-43 expressing flies show alterations in glucose metabolism. Interestingly, they demonstrated that glycolysis upregulation rescues TDP-43 proteinopathy, through overexpression of GLUT-3 glucose transporter or phosphofructokinase in motor neurons [191]. Taken together, these data provide an important link between TDP-43-induced proteinopathy and metabolic processes, highlighting a further potential therapeutic target for ALS treatment.
Further insights about TDP-43-ALS are described in the next paragraph, where some aspects of TDP-43 and Ataxin-2 interaction are also elucidated.
ATAXIN-2 as a TDP-43 modifier
Human Ataxin-2 gene (ATXN2) is the causative gene of Spinocerebellar Ataxia Type 2 (SCA2), because of a pathologic CAG repeat expansion (more than 34, respect to 22 in normal alleles) in its first exon, causing an abnormal poly-Q tract in ATXN2 protein [192, 193]. ATXN2 is involved in RNA stability, degradation and translation, and is crucial for SG assembly [194, 195]. Interestingly, through studies on Drosophila, an intermediate trinucleotide expansion (from 27 to 33 repeats [196]) has been demonstrated to be ALS-associated, confirming ATXN2 as an ALS susceptibility gene [197].
Atx2 gene, ATXN2 Drosophila ortholog, is essential for fly viability and is involved in translation control and RNP assembly [198]. Its loss causes bristle and eye defects together with motor impairments, whereas the effects of its overexpression range from locomotor deficits to lethality [194]. In a TDP-43 overexpressing ALS fly model, atx2 overexpression exacerbates ALS phenotype, further reducing lifespan and worsening eye ommatidia degeneration; conversely, atx2 functional inactivation extends lifespan and rescues eye aberrant morphology [197]. Moreover, transgenic flies carrying human ATXN2 gene with 32 CAGs show enhanced toxicity in a TDP-43-ALS background, with lower survival rate and reduced climbing performance [199]. Through PAM2 domain, human ATXN2 binds PABP protein, key regulator of SG formation [200] and whose reduction exacerbates TDP-43-induced toxicity in different ALS models [201]. Strikingly, flies expressing a domain-mutant ATXN2-32CAGs transgene encoding for ATXN2 protein without PAM2 domain, no longer modify TDP-43 toxicity pattern, indicating that PABP and SG formation may have a pivotal role in TDP-43-induced neurodegeneration [199].
In this regard, recent evidence suggests that poly-ADP-ribosylation (PARylation) plays a crucial role in regulating TDP-43 and SG dynamics: the reduction of PARylation levels suppresses SG formation and TDP-43 recruitment to SGs [202, 203]. Genetic and pharmacological inhibition of poly-ADP-ribose polymerase (PARP) rescues eye neurodegeneration, extends lifespan, and ameliorates motor performance in a TDP-43-overexpressing fly model [203]. Similarly, downregulation of poly-ADP-ribosyltransferase Tankyrase, involved in TDP-43 SG inclusion, suppresses the eye altered phenotype and fly lifespan reduction [202]. These results provide strong indications that modulation of PARylation may unveil therapeutic strategies not explored yet for ALS treatment. Further insights about RNA granules indicate that deletion of Atx2 intrinsically disordered regions domain, affecting RNP granule formation, is sufficient to rescue the neurodegenerative phenotypes of both FUS-related and C9ORF72-related fly ALS models [198], strongly suggesting that regulation of RNP granule assembly may represent an important strategy to counterbalance neurodegeneration in a wide spectrum of ALS-associated pathogenic contexts.
Conclusion
As we have detailed, Drosophila genetics has played an important role in discovering the involvement of several cellular and molecular pathways in ALS pathogenesis, and identifying potential disease modifiers. Although it may seem a paradox, great similarities in basic and network biochemistry occur and are conserved in all animals; this has allowed Drosophila being extensively studied as a powerful ALS model in which to discern components of pathways, understand disease mechanisms, investigate responses to genetic modifiers, and finally identify new therapeutics. Indeed, some exciting advances obtained through Drosophila have already facilitated our understanding of how input information is integrated and translated into an output motor response, and of how impaired molecular signaling and neuronal circuits that coordinate motor behavior can detrimentally impact on ALS pathology. Although a wealth of information has been collected so far, numerous mechanistic, basic research and clinical questions still remain unanswered. In particular, the master challenge in Drosophila-ALS research is making new genetic modifiers and therapeutic molecules safely and more rapidly available to patients, while the master limitation is of course the jump from flies to humans. Despite this insurmountable paradigm shift, genetic manipulations in Drosophila offer higher efficiency in targeting ALS disease genes than in higher organisms and provide greater proficiency in both dissecting pathological responses and screening therapeutic compounds.
We fairly anticipate and believe that Drosophila overcoming its model borders may now renew its scientific consent in ALS, and we prospect a time when research on ALS will keep sturdily investing on Drosophila.
Acknowledgements
Figures were created with BioRender.com.
Author contributions
FL and CV contributed to conceptualization; FL, SA and CV wrote and edited the manuscript; FL and SA prepared graphical contents. The authors read and approved the final manuscript.
Funding
The Italian Ministry of Health (Ricerca Corrente) is supporting current research ongoing in the authors’ laboratory.
Declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Trojsi F, D'Alvano G, Bonavita S, Tedeschi G. Genetics and sex in the pathogenesis of amyotrophic lateral sclerosis (ALS): is there a link? Int J Mol Sci. 2020 doi: 10.3390/ijms21103647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alsultan AA, Waller R, Heath PR, Kirby J. The genetics of amyotrophic lateral sclerosis: current insights. Degener Neurol Neuromuscul Dis. 2016;6:49–64. doi: 10.2147/DNND.S84956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nguyen HP, Van Broeckhoven C, van der Zee J. ALS genes in the genomic era and their implications for FTD. Trends Genet. 2018;34:404–423. doi: 10.1016/j.tig.2018.03.001. [DOI] [PubMed] [Google Scholar]
- 4.Masrori P, Van Damme P. Amyotrophic lateral sclerosis: a clinical review. Eur J Neurol. 2020 doi: 10.1111/ene.14393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Liscic RM, Alberici A, Cairns NJ, Romano M, Buratti E. From basic research to the clinic: innovative therapies for ALS and FTD in the pipeline. Mol Neurodegener. 2020;15:31. doi: 10.1186/s13024-020-00373-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schram S, Loeb JA, Song F. Disease propagation in amyotrophic lateral sclerosis (ALS): an interplay between genetics and environment. J Neuroinflammation. 2020;17:175. doi: 10.1186/s12974-020-01849-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McGurk L, Berson A, Bonini NM. Drosophila as an in vivo model for human neurodegenerative disease. Genetics. 2015;201:377–402. doi: 10.1534/genetics.115.179457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Reiter LT, Potocki L, Chien S, Gribskov M, Bier E. A systematic analysis of human disease-associated gene sequences in Drosophila melanogaster. Genome Res. 2001;11:1114–1125. doi: 10.1101/gr.169101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yamaguchi M, Yoshida H. Drosophila as a model organism. Adv Exp Med Biol. 2018;1076:1–10. doi: 10.1007/978-981-13-0529-0_1. [DOI] [PubMed] [Google Scholar]
- 10.Liguori F, Amadio S, Volonté C. Where and why modeling amyotrophic lateral sclerosis. Int J Mol Sci. 2021;22(8):3977. doi: 10.3390/ijms22083977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Longinetti E, Fang F. Epidemiology of amyotrophic lateral sclerosis: an update of recent literature. Curr Opin Neurol. 2019;32:771–776. doi: 10.1097/WCO.0000000000000730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rowland LP, Shneider NA. Amyotrophic lateral sclerosis. N Engl J Med. 2001;344:1688–1700. doi: 10.1056/NEJM200105313442207. [DOI] [PubMed] [Google Scholar]
- 13.Swinnen B, Robberecht W. The phenotypic variability of amyotrophic lateral sclerosis. Nat Rev Neurol. 2014;10:661–670. doi: 10.1038/nrneurol.2014.184. [DOI] [PubMed] [Google Scholar]
- 14.Solomon DA, Mitchell JC, Salcher-Konrad MT, Vance CA, Mizielinska S. Review: modelling the pathology and behaviour of frontotemporal dementia. Neuropathol Appl Neurobiol. 2019;45:58–80. doi: 10.1111/nan.12536. [DOI] [PubMed] [Google Scholar]
- 15.Abramzon YA, Fratta P, Traynor BJ, Chia R. The overlapping genetics of amyotrophic lateral sclerosis and frontotemporal dementia. Front Neurosci. 2020;14:42. doi: 10.3389/fnins.2020.00042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Régal L, Vanopdenbosch L, Tilkin P, et al. The G93C mutation in superoxide dismutase 1: clinicopathologic phenotype and prognosis. Arch Neurol. 2006;63:262–267. doi: 10.1001/archneur.63.2.262. [DOI] [PubMed] [Google Scholar]
- 17.Takeda T, Kitagawa K, Arai K. Phenotypic variability and its pathological basis in amyotrophic lateral sclerosis. Neuropathology. 2020;40:40–56. doi: 10.1111/neup.12606. [DOI] [PubMed] [Google Scholar]
- 18.Zhao W, Beers DR, Thonhoff JR, et al. Immunosuppressive functions of M2 macrophages derived from iPSCs of patients with ALS and healthy controls. iScience. 2020;23(6):101192. doi: 10.1016/j.isci.2020.101192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Osking Z, Ayers JI, Hildebrandt R, et al. ALS-linked SOD1 mutants enhance neurite outgrowth and branching in adult motor neurons. iScience. 2019;19:448–449. doi: 10.1016/j.isci.2019.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Scaricamazza S, Salvatori I, Giacovazzo G, et al. Skeletal-muscle metabolic reprogramming in ALS-SOD1. iScience. 2020;23:101087. doi: 10.1016/j.isci.2020.101087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Beers DR, Appel SH. Immune dysregulation in amyotrophic lateral sclerosis: mechanisms and emerging therapies. Lancet Neurol. 2019;18:211–220. doi: 10.1016/S1474-4422(18)30394-6. [DOI] [PubMed] [Google Scholar]
- 22.Lyon MS, Wosiski-Kuhn M, Gillespie R, Caress J, Milligan C. Inflammation, Immunity, and amyotrophic lateral sclerosis: I. Etiology and pathology. Muscle Nerve. 2019;59:10–22. doi: 10.1002/mus.26289. [DOI] [PubMed] [Google Scholar]
- 23.Trageser KJ, Smith C, Herman FJ, Ono K, Pasinetti GM. Mechanisms of immune activation by. Front Neurosci. 2019;13:1298. doi: 10.3389/fnins.2019.01298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Apolloni S, Amadio S, Fabbrizio P, et al. Histaminergic transmission slows progression of amyotrophic lateral sclerosis. J Cachexia Sarcopenia Muscle. 2019;10:872–893. doi: 10.1002/jcsm.12422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Volonté C, Apolloni S, Sabatelli M. Histamine beyond its effects on allergy: potential therapeutic benefits for the treatment of Amyotrophic Lateral Sclerosis (ALS) Pharmacol Ther. 2019;202:120–131. doi: 10.1016/j.pharmthera.2019.06.006. [DOI] [PubMed] [Google Scholar]
- 26.Yamaguchi M, Omori K, Asada S, Yoshida H. Epigenetic regulation of ALS and CMT: a lesson from Drosophila Models. Int J Mol Sci. 2021;22(2):491. doi: 10.3390/ijms22020491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Layalle S, They L, Ourghani S, Raoul C, Soustelle L. Amyotrophic Lateral Sclerosis Genes in Drosophila melanogaster. Int J Mol Sci. 2021;22(2):904. doi: 10.3390/ijms22020904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Azuma Y, Mizuta I, Tokuda T, Mizuno T. Amyotrophic lateral sclerosis model. Adv Exp Med Biol. 2018;1076:79–95. doi: 10.1007/978-981-13-0529-0_6. [DOI] [PubMed] [Google Scholar]
- 29.Şentürk M, Bellen HJ. Genetic strategies to tackle neurological diseases in fruit flies. Curr Opin Neurobiol. 2018;50:24–32. doi: 10.1016/j.conb.2017.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Brand AH, Perrimon N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development. 1993;118:401–415. doi: 10.1242/dev.118.2.401. [DOI] [PubMed] [Google Scholar]
- 31.McGuire SE, Mao Z, Davis RL. Spatiotemporal gene expression targeting with the TARGET and gene-switch systems in Drosophila. Sci STKE. 2004;2004(220):pl6. doi: 10.1126/stke.2202004pl6. [DOI] [PubMed] [Google Scholar]
- 32.Iyer J, Wang Q, Le T, et al. Quantitative Assessment of Eye Phenotypes for Functional Genetic Studies Using Drosophila melanogaster. G3 (Bethesda) 2016;6(5):1427–1437. doi: 10.1534/g3.116.027060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ready DF, Hanson TE, Benzer S. Development of the Drosophila retina, a neurocrystalline lattice. Dev Biol. 1976;53:217–240. doi: 10.1016/0012-1606(76)90225-6. [DOI] [PubMed] [Google Scholar]
- 34.Kumar JP. Building an ommatidium one cell at a time. Dev Dyn. 2012;241:136–149. doi: 10.1002/dvdy.23707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Casci I, Pandey UB. A fruitful endeavor: modeling ALS in the fruit fly. Brain Res. 2015;1607:47–74. doi: 10.1016/j.brainres.2014.09.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Singhal N, Jaiswal M. Pathways to neurodegeneration: lessons learnt from unbiased genetic screens in. J Genet. 2018;97:773–781. doi: 10.1007/s12041-018-0954-4. [DOI] [PubMed] [Google Scholar]
- 37.Majounie E, Renton AE, Mok K, et al. Frequency of the C9orf72 hexanucleotide repeat expansion in patients with amyotrophic lateral sclerosis and frontotemporal dementia: a cross-sectional study. Lancet Neurol. 2012;11:323–330. doi: 10.1016/S1474-4422(12)70043-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shepheard SR, Parker MD, Cooper-Knock J, et al. Value of systematic genetic screening of patients with amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry. 2021 doi: 10.1136/jnnp-2020-325014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sellier C, Campanari ML, Julie Corbier C, et al. Loss of C9ORF72 impairs autophagy and synergizes with polyQ Ataxin-2 to induce motor neuron dysfunction and cell death. EMBO J. 2016;35:1276–1297. doi: 10.15252/embj.201593350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pang W, Hu F. Cellular and physiological functions of C9ORF72 and implications for ALS/FTD. J Neurochem. 2020 doi: 10.1111/jnc.15255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Smith BN, Newhouse S, Shatunov A, et al. The C9ORF72 expansion mutation is a common cause of ALS+/-FTD in Europe and has a single founder. Eur J Hum Genet. 2013;21:102–108. doi: 10.1038/ejhg.2012.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.DeJesus-Hernandez M, Mackenzie IR, Boeve BF, et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72:245–256. doi: 10.1016/j.neuron.2011.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Renton AE, Majounie E, Waite A, et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72:257–268. doi: 10.1016/j.neuron.2011.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Beck J, Poulter M, Hensman D, et al. Large C9orf72 hexanucleotide repeat expansions are seen in multiple neurodegenerative syndromes and are more frequent than expected in the UK population. Am J Hum Genet. 2013;92:345–353. doi: 10.1016/j.ajhg.2013.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xu Z, Poidevin M, Li X, et al. Expanded GGGGCC repeat RNA associated with amyotrophic lateral sclerosis and frontotemporal dementia causes neurodegeneration. Proc Natl Acad Sci USA. 2013;110:7778–7783. doi: 10.1073/pnas.1219643110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mizielinska S, Grönke S, Niccoli T, et al. C9orf72 repeat expansions cause neurodegeneration in Drosophila through arginine-rich proteins. Science. 2014;345:1192–1194. doi: 10.1126/science.1256800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Freibaum BD, Lu Y, Lopez-Gonzalez R, et al. GGGGCC repeat expansion in C9orf72 compromises nucleocytoplasmic transport. Nature. 2015;525:129–133. doi: 10.1038/nature14974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kramer NJ, Carlomagno Y, Zhang YJ, et al. Spt4 selectively regulates the expression of C9orf72 sense and antisense mutant transcripts. Science. 2016;353:708–712. doi: 10.1126/science.aaf7791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lee KH, Zhang P, Kim HJ, et al. C9orf72 dipeptide repeats impair the assembly, dynamics, and function of membrane-less organelles. Cell. 2016;167:774–788.e717. doi: 10.1016/j.cell.2016.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Van Mossevelde S, van der Zee J, Cruts M, Van Broeckhoven C. Relationship between C9orf72 repeat size and clinical phenotype. Curr Opin Genet Dev. 2017;44:117–124. doi: 10.1016/j.gde.2017.02.008. [DOI] [PubMed] [Google Scholar]
- 51.Goodman LD, Prudencio M, Kramer NJ, et al. Toxic expanded GGGGCC repeat transcription is mediated by the PAF1 complex in C9orf72-associated FTD. Nat Neurosci. 2019;22:863–874. doi: 10.1038/s41593-019-0396-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tang X, Toro A, Sahana TG, et al. Divergence, convergence, and therapeutic implications: a cell biology perspective of C9ORF72-ALS/FTD. Mol Neurodegener. 2020;15(1):34. doi: 10.1186/s13024-020-00383-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zu T, Gibbens B, Doty NS, et al. Non-ATG-initiated translation directed by microsatellite expansions. Proc Natl Acad Sci USA. 2011;108:260–265. doi: 10.1073/pnas.1013343108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ash PE, Bieniek KF, Gendron TF, et al. Unconventional translation of C9ORF72 GGGGCC expansion generates insoluble polypeptides specific to c9FTD/ALS. Neuron. 2013;77:639–646. doi: 10.1016/j.neuron.2013.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mori K, Weng SM, Arzberger T, et al. The C9orf72 GGGGCC repeat is translated into aggregating dipeptide-repeat proteins in FTLD/ALS. Science. 2013;339:1335–1338. doi: 10.1126/science.1232927. [DOI] [PubMed] [Google Scholar]
- 56.Gendron TF, Belzil VV, Zhang YJ, Petrucelli L. Mechanisms of toxicity in C9FTLD/ALS. Acta Neuropathol. 2014;127:359–376. doi: 10.1007/s00401-013-1237-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Waite AJ, Bäumer D, East S, et al. Reduced C9orf72 protein levels in frontal cortex of amyotrophic lateral sclerosis and frontotemporal degeneration brain with the C9ORF72 hexanucleotide repeat expansion. Neurobiol Aging. 2014;35:1779.e1775–1779.e1713. doi: 10.1016/j.neurobiolaging.2014.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Burberry A, Suzuki N, Wang JY, et al. Loss-of-function mutations in the C9ORF72 mouse ortholog cause fatal autoimmune disease. Sci Transl Med. 2016 doi: 10.1126/scitranslmed.aaf6038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Iyer S, Acharya KR, Subramanian V. A comparative bioinformatic analysis of. PeerJ. 2018 doi: 10.7717/peerj.4391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hall AC, Ostrowski LA, Pietrobon V, Mekhail K. Repetitive DNA loci and their modulation by the non-canonical nucleic acid structures R-loops and G-quadruplexes. Nucleus. 2017;8:162–181. doi: 10.1080/19491034.2017.1292193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Goodman LD, Bonini NM. New roles for canonical transcription factors in repeat expansion diseases. Trends Genet. 2020;36:81–92. doi: 10.1016/j.tig.2019.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hirtreiter A, Damsma GE, Cheung AC, et al. Spt4/5 stimulates transcription elongation through the RNA polymerase clamp coiled-coil motif. Nucleic Acids Res. 2010;38:4040–4051. doi: 10.1093/nar/gkq135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Celona B, Dollen JV, Vatsavayai SC, et al. Suppression of C9orf72 RNA repeat-induced neurotoxicity by the ALS-associated RNA-binding protein Zfp106. Elife. 2017 doi: 10.7554/eLife.19032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ramesh N, Daley EL, Gleixner AM, et al. RNA dependent suppression of C9orf72 ALS/FTD associated neurodegeneration by matrin-3. Acta Neuropathol Commun. 2020;8:177. doi: 10.1186/s40478-020-01060-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Jiao B, Wang M, Feng H, et al. Downregulation of TOP2 modulates neurodegeneration caused by GGGGCC expanded repeats. Hum Mol Genet. 2021 doi: 10.1093/hmg/ddab079. [DOI] [PubMed] [Google Scholar]
- 66.Mori K, Arzberger T, Grässer FA, et al. Bidirectional transcripts of the expanded C9orf72 hexanucleotide repeat are translated into aggregating dipeptide repeat proteins. Acta Neuropathol. 2013;126:881–893. doi: 10.1007/s00401-013-1189-3. [DOI] [PubMed] [Google Scholar]
- 67.Goodman LD, Prudencio M, Srinivasan AR, et al. eIF4B and eIF4H mediate GR production from expanded G4C2 in a Drosophila model for C9orf72-associated ALS. Acta Neuropathol Commun. 2019;7:62. doi: 10.1186/s40478-019-0711-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wen X, Tan W, Westergard T, et al. Antisense proline-arginine RAN dipeptides linked to C9ORF72-ALS/FTD form toxic nuclear aggregates that initiate in vitro and in vivo neuronal death. Neuron. 2014;84:1213–1225. doi: 10.1016/j.neuron.2014.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yang D, Abdallah A, Li Z, Lu Y, Almeida S, Gao FB. FTD/ALS-associated poly(GR) protein impairs the Notch pathway and is recruited by poly(GA) into cytoplasmic inclusions. Acta Neuropathol. 2015;130:525–535. doi: 10.1007/s00401-015-1448-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Perry S, Han Y, Das A, Dickman D. Homeostatic plasticity can be induced and expressed to restore synaptic strength at neuromuscular junctions undergoing ALS-related degeneration. Hum Mol Genet. 2017;26:4153–4167. doi: 10.1093/hmg/ddx304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Fumagalli L, Young FL, Boeynaems S, et al. C9orf72-derived arginine-containing dipeptide repeats associate with axonal transport machinery and impede microtubule-based motility. Sci Adv. 2021 doi: 10.1126/sciadv.abg3013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.van Blitterswijk M, DeJesus-Hernandez M, Niemantsverdriet E, et al. Association between repeat sizes and clinical and pathological characteristics in carriers of C9ORF72 repeat expansions (Xpansize-72): a cross-sectional cohort study. Lancet Neurol. 2013;12:978–988. doi: 10.1016/S1474-4422(13)70210-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bennion Callister J, Ryan S, Sim J, Rollinson S, Pickering-Brown SM. Modelling C9orf72 dipeptide repeat proteins of a physiologically relevant size. Hum Mol Genet. 2016;25:5069–5082. doi: 10.1093/hmg/ddw327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Morón-Oset J, Supèr T, Esser J, Isaacs AM, Grönke S, Partridge L. Glycine-alanine dipeptide repeats spread rapidly in a repeat length- and age-dependent manner in the fly brain. Acta Neuropathol Commun. 2019;7:209. doi: 10.1186/s40478-019-0860-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.West RJH, Sharpe JL, Voelzmann A, et al. Co-expression of C9orf72 related dipeptide-repeats over 1000 repeat units reveals age- and combination-specific phenotypic profiles in Drosophila. Acta Neuropathol Commun. 2020;8:158. doi: 10.1186/s40478-020-01028-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Xu W, Xu J. C9orf72 dipeptide repeats cause selective neurodegeneration and cell-autonomous excitotoxicity in. J Neurosci. 2018;38:7741–7752. doi: 10.1523/JNEUROSCI.0908-18.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bensimon G, Lacomblez L, Meininger V. A controlled trial of riluzole in amyotrophic lateral sclerosis. ALS/Riluzole study group. N Engl J Med. 1994;330:585–591. doi: 10.1056/NEJM199403033300901. [DOI] [PubMed] [Google Scholar]
- 78.Boeynaems S, Bogaert E, Michiels E, et al. Drosophila screen connects nuclear transport genes to DPR pathology in c9ALS/FTD. Sci Rep. 2016;6:20877. doi: 10.1038/srep20877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhang K, Donnelly CJ, Haeusler AR, et al. The C9orf72 repeat expansion disrupts nucleocytoplasmic transport. Nature. 2015;525:56–61. doi: 10.1038/nature14973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cunningham KM, Maulding K, Ruan K, et al. TFEB/Mitf links impaired nuclear import to autophagolysosomal dysfunction in C9-ALS. Elife. 2020 doi: 10.7554/eLife.59419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Park JH, Chung CG, Park SS, et al. Cytosolic calcium regulates cytoplasmic accumulation of TDP-43 through Calpain-A and Importin α3. Elife. 2020 doi: 10.7554/eLife.60132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Luty AA, Kwok JB, Dobson-Stone C, et al. Sigma nonopioid intracellular receptor 1 mutations cause frontotemporal lobar degeneration-motor neuron disease. Ann Neurol. 2010;68:639–649. doi: 10.1002/ana.22274. [DOI] [PubMed] [Google Scholar]
- 83.Al-Saif A, Al-Mohanna F, Bohlega S. A mutation in sigma-1 receptor causes juvenile amyotrophic lateral sclerosis. Ann Neurol. 2011;70:913–919. doi: 10.1002/ana.22534. [DOI] [PubMed] [Google Scholar]
- 84.Mavlyutov TA, Epstein ML, Verbny YI, et al. Lack of sigma-1 receptor exacerbates ALS progression in mice. Neuroscience. 2013;240:129–134. doi: 10.1016/j.neuroscience.2013.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Couly S, Khalil B, Viguier V, Roussel J, Maurice T, Liévens JC. Sigma-1 receptor is a key genetic modulator in amyotrophic lateral sclerosis. Hum Mol Genet. 2020;29:529–540. doi: 10.1093/hmg/ddz267. [DOI] [PubMed] [Google Scholar]
- 86.Lee PT, Liévens JC, Wang SM, et al. Sigma-1 receptor chaperones rescue nucleocytoplasmic transport deficit seen in cellular and Drosophila ALS/FTD models. Nat Commun. 2020;11:5580. doi: 10.1038/s41467-020-19396-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ortega JA, Daley EL, Kour S, et al. Nucleocytoplasmic proteomic analysis uncovers eRF1 and nonsense-mediated decay as modifiers of ALS/FTD C9orf72 toxicity. Neuron. 2020;106:90–107.e113. doi: 10.1016/j.neuron.2020.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Xu W, Bao P, Jiang X, et al. Reactivation of nonsense-mediated mRNA decay protects against C9orf72 dipeptide-repeat neurotoxicity. Brain. 2019;142:1349–1364. doi: 10.1093/brain/awz070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sun Y, Eshov A, Zhou J, Isiktas AU, Guo JU. C9orf72 arginine-rich dipeptide repeats inhibit UPF1-mediated RNA decay via translational repression. Nat Commun. 2020;11:3354. doi: 10.1038/s41467-020-17129-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zaepfel BL, Zhang Z, Maulding K, et al. UPF1 reduces C9orf72 HRE-induced neurotoxicity in the absence of nonsense-mediated decay dysfunction. Cell Rep. 2021 doi: 10.1016/j.celrep.2021.108925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kankel MW, Sen A, Lu L, et al. Amyotrophic lateral sclerosis modifiers in. Genetics. 2020;215:747–766. doi: 10.1534/genetics.119.302985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Deng HX, Hentati A, Tainer JA, et al. Amyotrophic lateral sclerosis and structural defects in Cu, Zn superoxide dismutase. Science. 1993;261:1047–1051. doi: 10.1126/science.8351519. [DOI] [PubMed] [Google Scholar]
- 93.Rosen DR, Siddique T, Patterson D, et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature. 1993;362:59–62. doi: 10.1038/362059a0. [DOI] [PubMed] [Google Scholar]
- 94.Chattopadhyay M, Valentine JS. Aggregation of copper-zinc superoxide dismutase in familial and sporadic ALS. Antioxid Redox Signal. 2009;11:1603–1614. doi: 10.1089/ars.2009.2536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bernard E, Pegat A, Svahn J, et al. Clinical and molecular landscape of ALS patients with. Int J Mol Sci. 2020 doi: 10.3390/ijms21186807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yim MB, Kang JH, Yim HS, Kwak HS, Chock PB, Stadtman ER. A gain-of-function of an amyotrophic lateral sclerosis-associated Cu, Zn-superoxide dismutase mutant: an enhancement of free radical formation due to a decrease in Km for hydrogen peroxide. Proc Natl Acad Sci USA. 1996;93:5709–5714. doi: 10.1073/pnas.93.12.5709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Bruijn LI, Houseweart MK, Kato S, et al. Aggregation and motor neuron toxicity of an ALS-linked SOD1 mutant independent from wild-type SOD1. Science. 1998;281:1851–1854. doi: 10.1126/science.281.5384.1851. [DOI] [PubMed] [Google Scholar]
- 98.Mockett RJ, Radyuk SN, Benes JJ, Orr WC, Sohal RS. Phenotypic effects of familial amyotrophic lateral sclerosis mutant Sod alleles in transgenic Drosophila. Proc Natl Acad Sci USA. 2003;100:301–306. doi: 10.1073/pnas.0136976100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ilieva H, Polymenidou M, Cleveland DW. Non-cell autonomous toxicity in neurodegenerative disorders: ALS and beyond. J Cell Biol. 2009;187:761–772. doi: 10.1083/jcb.200908164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Barber SC, Mead RJ, Shaw PJ. Oxidative stress in ALS: a mechanism of neurodegeneration and a therapeutic target. Biochim Biophys Acta. 2006;1762:1051–1067. doi: 10.1016/j.bbadis.2006.03.008. [DOI] [PubMed] [Google Scholar]
- 101.Elia AJ, Parkes TL, Kirby K, et al. Expression of human FALS SOD in motorneurons of Drosophila. Free Radic Biol Med. 1999;26:1332–1338. doi: 10.1016/S0891-5849(98)00333-5. [DOI] [PubMed] [Google Scholar]
- 102.Watson MR, Lagow RD, Xu K, Zhang B, Bonini NM. A drosophila model for amyotrophic lateral sclerosis reveals motor neuron damage by human SOD1. J Biol Chem. 2008;283:24972–24981. doi: 10.1074/jbc.M804817200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Bahadorani S, Mukai ST, Rabie J, Beckman JS, Phillips JP, Hilliker AJ. Expression of zinc-deficient human superoxide dismutase in Drosophila neurons produces a locomotor defect linked to mitochondrial dysfunction. Neurobiol Aging. 2013;34:2322–2330. doi: 10.1016/j.neurobiolaging.2013.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.De Rose F, Marotta R, Talani G, et al. Differential effects of phytotherapic preparations in the hSOD1 Drosophila melanogaster model of ALS. Sci Rep. 2017;7:41059. doi: 10.1038/srep41059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Maccioni R, Setzu MD, Talani G, et al. Standardized phytotherapic extracts rescue anomalous locomotion and electrophysiological responses of TDP-43 Drosophila melanogaster model of ALS. Sci Rep. 2018;8:16002. doi: 10.1038/s41598-018-34452-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zhang C, Yang Y, Liang W, et al. Neuroprotection by urate on the mutant hSOD1-related cellular and Drosophila models of amyotrophic lateral sclerosis: Implication for GSH synthesis via activating Akt/GSK3β/Nrf2/GCLC pathways. Brain Res Bull. 2019;146:287–301. doi: 10.1016/j.brainresbull.2019.01.019. [DOI] [PubMed] [Google Scholar]
- 107.Wang T, Cheng J, Wang S, et al. α-Lipoic acid attenuates oxidative stress and neurotoxicity via the ERK/Akt-dependent pathway in the mutant hSOD1 related Drosophila model and the NSC34 cell line of amyotrophic lateral sclerosis. Brain Res Bull. 2018;140:299–310. doi: 10.1016/j.brainresbull.2018.05.019. [DOI] [PubMed] [Google Scholar]
- 108.Wang TH, Wang SY, Wang XD, et al. Fisetin exerts antioxidant and neuroprotective effects in multiple mutant hSOD1 models of amyotrophic lateral sclerosis by activating ERK. Neuroscience. 2018;379:152–166. doi: 10.1016/j.neuroscience.2018.03.008. [DOI] [PubMed] [Google Scholar]
- 109.Zhang C, Liang W, Wang H, et al. γ-Oryzanol mitigates oxidative stress and prevents mutant SOD1-related neurotoxicity in Drosophila and cell models of amyotrophic lateral sclerosis. Neuropharmacology. 2019 doi: 10.1016/j.neuropharm.2019.107777. [DOI] [PubMed] [Google Scholar]
- 110.Bosco DA, Lemay N, Ko HK, et al. Mutant FUS proteins that cause amyotrophic lateral sclerosis incorporate into stress granules. Hum Mol Genet. 2010;19:4160–4175. doi: 10.1093/hmg/ddq335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Gal J, Zhang J, Kwinter DM, et al. Nuclear localization sequence of FUS and induction of stress granules by ALS mutants. Neurobiol Aging. 2011;32:2323.e2327–2340. doi: 10.1016/j.neurobiolaging.2010.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Daigle JG, Krishnamurthy K, Ramesh N, et al. Pur-alpha regulates cytoplasmic stress granule dynamics and ameliorates FUS toxicity. Acta Neuropathol. 2016;131:605–620. doi: 10.1007/s00401-015-1530-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Wang X, Arai S, Song X, et al. Induced ncRNAs allosterically modify RNA-binding proteins in cis to inhibit transcription. Nature. 2008;454:126–130. doi: 10.1038/nature06992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Yang L, Embree LJ, Tsai S, Hickstein DD. Oncoprotein TLS interacts with serine-arginine proteins involved in RNA splicing. J Biol Chem. 1998;273:27761–27764. doi: 10.1074/jbc.273.43.27761. [DOI] [PubMed] [Google Scholar]
- 115.Gregory RI, Yan KP, Amuthan G, et al. The Microprocessor complex mediates the genesis of microRNAs. Nature. 2004;432:235–240. doi: 10.1038/nature03120. [DOI] [PubMed] [Google Scholar]
- 116.Tan AY, Manley JL. TLS inhibits RNA polymerase III transcription. Mol Cell Biol. 2010;30:186–196. doi: 10.1128/MCB.00884-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Lagier-Tourenne C, Polymenidou M, Hutt KR, et al. Divergent roles of ALS-linked proteins FUS/TLS and TDP-43 intersect in processing long pre-mRNAs. Nat Neurosci. 2012;15:1488–1497. doi: 10.1038/nn.3230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Rogelj B, Easton LE, Bogu GK, et al. Widespread binding of FUS along nascent RNA regulates alternative splicing in the brain. Sci Rep. 2012;2:603. doi: 10.1038/srep00603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Brunet MA, Jacques JF, Nassari S, et al. The FUS gene is dual-coding with both proteins contributing to FUS-mediated toxicity. EMBO. 2020 doi: 10.15252/embr.202050640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kwiatkowski TJ, Bosco DA, Leclerc AL, et al. Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science. 2009;323:1205–1208. doi: 10.1126/science.1166066. [DOI] [PubMed] [Google Scholar]
- 121.Vance C, Rogelj B, Hortobágyi T, et al. Mutations in FUS, an RNA processing protein, cause familial amyotrophic lateral sclerosis type 6. Science. 2009;323:1208–1211. doi: 10.1126/science.1165942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Chen Y, Yang M, Deng J, et al. Expression of human FUS protein in Drosophila leads to progressive neurodegeneration. Protein Cell. 2011;2:477–486. doi: 10.1007/s13238-011-1065-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lanson NA, Pandey UB. FUS-related proteinopathies: lessons from animal models. Brain Res. 2012;1462:44–60. doi: 10.1016/j.brainres.2012.01.039. [DOI] [PubMed] [Google Scholar]
- 124.Shahidullah M, Le Marchand SJ, Fei H, et al. Defects in synapse structure and function precede motor neuron degeneration in Drosophila models of FUS-related ALS. J Neurosci. 2013;33:19590–19598. doi: 10.1523/JNEUROSCI.3396-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lanson NA, Maltare A, King H, et al. A Drosophila model of FUS-related neurodegeneration reveals genetic interaction between FUS and TDP-43. Hum Mol Genet. 2011;20:2510–2523. doi: 10.1093/hmg/ddr150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Wang JW, Brent JR, Tomlinson A, Shneider NA, McCabe BD. The ALS-associated proteins FUS and TDP-43 function together to affect Drosophila locomotion and life span. J Clin Invest. 2011;121:4118–4126. doi: 10.1172/JCI57883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Xia R, Liu Y, Yang L, Gal J, Zhu H, Jia J. Motor neuron apoptosis and neuromuscular junction perturbation are prominent features in a Drosophila model of Fus-mediated ALS. Mol Neurodegener. 2012;7:10. doi: 10.1186/1750-1326-7-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Miguel L, Avequin T, Delarue M, et al. Accumulation of insoluble forms of FUS protein correlates with toxicity in Drosophila. Neurobiol Aging. 2012;33:1008.e1001–1015. doi: 10.1016/j.neurobiolaging.2011.10.008. [DOI] [PubMed] [Google Scholar]
- 129.Steyaert J, Scheveneels W, Vanneste J, et al. FUS-induced neurotoxicity in Drosophila is prevented by downregulating nucleocytoplasmic transport proteins. Hum Mol Genet. 2018;27:4103–4116. doi: 10.1093/hmg/ddy303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Bogaert E, Boeynaems S, Kato M, et al. Molecular dissection of FUS points at synergistic effect of low-complexity domains in toxicity. Cell Rep. 2018;24:529–537.e524. doi: 10.1016/j.celrep.2018.06.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Sasayama H, Shimamura M, Tokuda T, et al. Knockdown of the Drosophila fused in sarcoma (FUS) homologue causes deficient locomotive behavior and shortening of motoneuron terminal branches. PLoS ONE. 2012 doi: 10.1371/journal.pone.0039483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Frickenhaus M, Wagner M, Mallik M, Catinozzi M, Storkebaum E. Highly efficient cell-type-specific gene inactivation reveals a key function for the Drosophila FUS homolog cabeza in neurons. Sci Rep. 2015;5:9107. doi: 10.1038/srep09107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Johnson JO, Mandrioli J, Benatar M, et al. Exome sequencing reveals VCP mutations as a cause of familial ALS. Neuron. 2010;68:857–864. doi: 10.1016/j.neuron.2010.11.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Azuma Y, Tokuda T, Shimamura M, et al. Identification of ter94, Drosophila VCP, as a strong modulator of motor neuron degeneration induced by knockdown of Caz, Drosophila FUS. Hum Mol Genet. 2014;23:3467–3480. doi: 10.1093/hmg/ddu055. [DOI] [PubMed] [Google Scholar]
- 135.Harley J, Hagemann C, Serio A, Patani R. FUS is lost from nuclei and gained in neurites of motor neurons in a human stem cell model of VCP-related ALS. Brain. 2020 doi: 10.1093/brain/awaa339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Casci I, Krishnamurthy K, Kour S, et al. Muscleblind acts as a modifier of FUS toxicity by modulating stress granule dynamics and SMN localization. Nat Commun. 2019;10:5583. doi: 10.1038/s41467-019-13383-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kanadia RN, Johnstone KA, Mankodi A, et al. A muscleblind knockout model for myotonic dystrophy. Science. 2003;302:1978–1980. doi: 10.1126/science.1088583. [DOI] [PubMed] [Google Scholar]
- 138.Rudnicki DD, Holmes SE, Lin MW, Thornton CA, Ross CA, Margolis RL. Huntington’s disease–like 2 is associated with CUG repeat-containing RNA foci. Ann Neurol. 2007;61:272–282. doi: 10.1002/ana.21081. [DOI] [PubMed] [Google Scholar]
- 139.Daughters RS, Tuttle DL, Gao W, et al. RNA gain-of-function in spinocerebellar ataxia type 8. PLoS Genet. 2009 doi: 10.1371/journal.pgen.1000600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Sellier C, Rau F, Liu Y, et al. Sam68 sequestration and partial loss of function are associated with splicing alterations in FXTAS patients. EMBO J. 2010;29:1248–1261. doi: 10.1038/emboj.2010.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Dardenne E, Polay Espinoza M, Fattet L, et al. RNA helicases DDX5 and DDX17 dynamically orchestrate transcription, miRNA, and splicing programs in cell differentiation. Cell Rep. 2014;7:1900–1913. doi: 10.1016/j.celrep.2014.05.010. [DOI] [PubMed] [Google Scholar]
- 142.Giraud G, Terrone S, Bourgeois CF. Functions of DEAD box RNA helicases DDX5 and DDX17 in chromatin organization and transcriptional regulation. BMB Rep. 2018;51:613–622. doi: 10.5483/BMBRep.2018.51.12.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Fortuna TR, Kour S, Anderson EN, et al. DDX17 is involved in DNA damage repair and modifies FUS toxicity in an RGG-domain dependent manner. Acta Neuropathol. 2021 doi: 10.1007/s00401-021-02333-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Fallini C, Khalil B, Smith CL, Rossoll W. Traffic jam at the nuclear pore: all roads lead to nucleocytoplasmic transport defects in ALS/FTD. Neurobiol Dis. 2020 doi: 10.1016/j.nbd.2020.104835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Lin YC, Kumar MS, Ramesh N, et al. Interactions between ALS-linked FUS and nucleoporins are associated with defects in the nucleocytoplasmic transport pathway. Nat Neurosci. 2021 doi: 10.1038/s41593-021-00859-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Bao Y, Hata Y, Ikeda M, Withanage K. Mammalian Hippo pathway: from development to cancer and beyond. J Biochem. 2011;149:361–379. doi: 10.1093/jb/mvr021. [DOI] [PubMed] [Google Scholar]
- 147.Azuma Y, Tokuda T, Kushimura Y, et al. Hippo, Drosophila MST, is a novel modifier of motor neuron degeneration induced by knockdown of Caz, Drosophila FUS. Exp Cell Res. 2018;371:311–321. doi: 10.1016/j.yexcr.2018.08.001. [DOI] [PubMed] [Google Scholar]
- 148.Gogia N, Sarkar A, Mehta AS, et al. Inactivation of Hippo and cJun-N-terminal Kinase (JNK) signaling mitigate FUS mediated neurodegeneration in vivo. Neurobiol Dis. 2020 doi: 10.1016/j.nbd.2020.104837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Cha SJ, Choi HJ, Kim HJ, et al. Parkin expression reverses mitochondrial dysfunction in fused in sarcoma-induced amyotrophic lateral sclerosis. Insect Mol Biol. 2020;29:56–65. doi: 10.1111/imb.12608. [DOI] [PubMed] [Google Scholar]
- 150.Volkening K, Leystra-Lantz C, Yang W, Jaffee H, Strong MJ. Tar DNA binding protein of 43 kDa (TDP-43), 14-3-3 proteins and copper/zinc superoxide dismutase (SOD1) interact to modulate NFL mRNA stability. Implications for altered RNA processing in amyotrophic lateral sclerosis (ALS) Brain Res. 2009;1305:168–182. doi: 10.1016/j.brainres.2009.09.105. [DOI] [PubMed] [Google Scholar]
- 151.Buratti E, Baralle FE. The multiple roles of TDP-43 in pre-mRNA processing and gene expression regulation. RNA Biol. 2010;7:420–429. doi: 10.4161/rna.7.4.12205. [DOI] [PubMed] [Google Scholar]
- 152.Buratti E, Baralle FE. Characterization and functional implications of the RNA binding properties of nuclear factor TDP-43, a novel splicing regulator of CFTR exon 9. J Biol Chem. 2001;276:36337–36343. doi: 10.1074/jbc.M104236200. [DOI] [PubMed] [Google Scholar]
- 153.Arai T, Hasegawa M, Akiyama H, et al. TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun. 2006;351:602–611. doi: 10.1016/j.bbrc.2006.10.093. [DOI] [PubMed] [Google Scholar]
- 154.Neumann M, Sampathu DM, Kwong LK, et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science. 2006;314:130–133. doi: 10.1126/science.1134108. [DOI] [PubMed] [Google Scholar]
- 155.Suk TR, Rousseaux MWC. The role of TDP-43 mislocalization in amyotrophic lateral sclerosis. Mol Neurodegener. 2020;15:45. doi: 10.1186/s13024-020-00397-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Kapeli K, Martinez FJ, Yeo GW. Genetic mutations in RNA-binding proteins and their roles in ALS. Hum Genet. 2017;136:1193–1214. doi: 10.1007/s00439-017-1830-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Hanson KA, Kim SH, Wassarman DA, Tibbetts RS. Ubiquilin modifies TDP-43 toxicity in a Drosophila model of amyotrophic lateral sclerosis (ALS) J Biol Chem. 2010;285:11068–11072. doi: 10.1074/jbc.C109.078527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Li Y, Ray P, Rao EJ, et al. A Drosophila model for TDP-43 proteinopathy. Proc Natl Acad Sci USA. 2010;107:3169–3174. doi: 10.1073/pnas.0913602107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Voigt A, Herholz D, Fiesel FC, et al. TDP-43-mediated neuron loss in vivo requires RNA-binding activity. PLoS ONE. 2010 doi: 10.1371/journal.pone.0012247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Estes PS, Boehringer A, Zwick R, Tang JE, Grigsby B, Zarnescu DC. Wild-type and A315T mutant TDP-43 exert differential neurotoxicity in a Drosophila model of ALS. Hum Mol Genet. 2011;20:2308–2321. doi: 10.1093/hmg/ddr124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Miguel L, Frébourg T, Campion D, Lecourtois M. Both cytoplasmic and nuclear accumulations of the protein are neurotoxic in Drosophila models of TDP-43 proteinopathies. Neurobiol Dis. 2011;41:398–406. doi: 10.1016/j.nbd.2010.10.007. [DOI] [PubMed] [Google Scholar]
- 162.Estes PS, Daniel SG, McCallum AP, et al. Motor neurons and glia exhibit specific individualized responses to TDP-43 expression in a Drosophila model of amyotrophic lateral sclerosis. Dis Model Mech. 2013;6:721–733. doi: 10.1242/dmm.010710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Krug L, Chatterjee N, Borges-Monroy R, et al. Retrotransposon activation contributes to neurodegeneration in a Drosophila TDP-43 model of ALS. PLoS Genet. 2017 doi: 10.1371/journal.pgen.1006635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Lin MJ, Cheng CW, Shen CK. Neuronal function and dysfunction of Drosophila dTDP. PLoS ONE. 2011 doi: 10.1371/journal.pone.0020371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Diaper DC, Adachi Y, Lazarou L, et al. Drosophila TDP-43 dysfunction in glia and muscle cells cause cytological and behavioural phenotypes that characterize ALS and FTLD. Hum Mol Genet. 2013;22:3883–3893. doi: 10.1093/hmg/ddt243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Feiguin F, Godena VK, Romano G, D'Ambrogio A, Klima R, Baralle FE. Depletion of TDP-43 affects Drosophila motoneurons terminal synapsis and locomotive behavior. FEBS Lett. 2009;583:1586–1592. doi: 10.1016/j.febslet.2009.04.019. [DOI] [PubMed] [Google Scholar]
- 167.Wang HY, Wang IF, Bose J, Shen CK. Structural diversity and functional implications of the eukaryotic TDP gene family. Genomics. 2004;83:130–139. doi: 10.1016/S0888-7543(03)00214-3. [DOI] [PubMed] [Google Scholar]
- 168.Boillée S, Vande Velde C, Cleveland DW. ALS: a disease of motor neurons and their nonneuronal neighbors. Neuron. 2006;52:39–59. doi: 10.1016/j.neuron.2006.09.018. [DOI] [PubMed] [Google Scholar]
- 169.Romano G, Klima R, Buratti E, Verstreken P, Baralle FE, Feiguin F. Chronological requirements of TDP-43 function in synaptic organization and locomotive control. Neurobiol Dis. 2014;71:95–109. doi: 10.1016/j.nbd.2014.07.007. [DOI] [PubMed] [Google Scholar]
- 170.Romano G, Appocher C, Scorzeto M, et al. Glial TDP-43 regulates axon wrapping, GluRIIA clustering and fly motility by autonomous and non-autonomous mechanisms. Hum Mol Genet. 2015;24:6134–6145. doi: 10.1093/hmg/ddv330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Kim SH, Shanware NP, Bowler MJ, Tibbetts RS. Amyotrophic lateral sclerosis-associated proteins TDP-43 and FUS/TLS function in a common biochemical complex to co-regulate HDAC6 mRNA. J Biol Chem. 2010;285:34097–34105. doi: 10.1074/jbc.M110.154831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Ling SC, Albuquerque CP, Han JS, et al. ALS-associated mutations in TDP-43 increase its stability and promote TDP-43 complexes with FUS/TLS. Proc Natl Acad Sci USA. 2010;107:13318–13323. doi: 10.1073/pnas.1008227107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Menzies FM, Fleming A, Caricasole A, et al. Autophagy and neurodegeneration: pathogenic mechanisms and therapeutic opportunities. Neuron. 2017;93:1015–1034. doi: 10.1016/j.neuron.2017.01.022. [DOI] [PubMed] [Google Scholar]
- 174.Cheng CW, Lin MJ, Shen CK. Rapamycin alleviates pathogenesis of a new Drosophila model of ALS-TDP. J Neurogenet. 2015;29:59–68. doi: 10.3109/01677063.2015.1077832. [DOI] [PubMed] [Google Scholar]
- 175.Donde A, Sun M, Jeong YH, et al. Upregulation of ATG7 attenuates motor neuron dysfunction associated with depletion of TARDBP/TDP-43. Autophagy. 2020;16:672–682. doi: 10.1080/15548627.2019.1635379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Cozzolino M, Pesaresi MG, Amori I, et al. Oligomerization of mutant SOD1 in mitochondria of motoneuronal cells drives mitochondrial damage and cell toxicity. Antioxid Redox Signal. 2009;11:1547–1558. doi: 10.1089/ars.2009.2545. [DOI] [PubMed] [Google Scholar]
- 177.Tradewell ML, Yu Z, Tibshirani M, Boulanger MC, Durham HD, Richard S. Arginine methylation by PRMT1 regulates nuclear-cytoplasmic localization and toxicity of FUS/TLS harbouring ALS-linked mutations. Hum Mol Genet. 2012;21:136–149. doi: 10.1093/hmg/ddr448. [DOI] [PubMed] [Google Scholar]
- 178.Onesto E, Colombrita C, Gumina V, et al. Gene-specific mitochondria dysfunctions in human TARDBP and C9ORF72 fibroblasts. Acta Neuropathol Commun. 2016;4:47. doi: 10.1186/s40478-016-0316-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Khalil B, Cabirol-Pol MJ, Miguel L, Whitworth AJ, Lecourtois M, Liévens JC. Enhancing Mitofusin/Marf ameliorates neuromuscular dysfunction in Drosophila models of TDP-43 proteinopathies. Neurobiol Aging. 2017;54:71–83. doi: 10.1016/j.neurobiolaging.2017.02.016. [DOI] [PubMed] [Google Scholar]
- 180.Sun X, Duan Y, Qin C, et al. Distinct multilevel misregulations of Parkin and PINK1 revealed in cell and animal models of TDP-43 proteinopathy. Cell Death Dis. 2018;9:953. doi: 10.1038/s41419-018-1022-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Coyne AN, Siddegowda BB, Estes PS, et al. Futsch/MAP1B mRNA is a translational target of TDP-43 and is neuroprotective in a Drosophila model of amyotrophic lateral sclerosis. J Neurosci. 2014;34:15962–15974. doi: 10.1523/JNEUROSCI.2526-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Hazelett DJ, Chang JC, Lakeland DL, Morton DB. Comparison of parallel high-throughput RNA sequencing between knockout of TDP-43 and its overexpression reveals primarily nonreciprocal and nonoverlapping gene expression changes in the central nervous system of Drosophila. G3 (Bethesda) 2012 doi: 10.1534/g3.112.002998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Chang JC, Hazelett DJ, Stewart JA, Morton DB. Motor neuron expression of the voltage-gated calcium channel cacophony restores locomotion defects in a Drosophila, TDP-43 loss of function model of ALS. Brain Res. 2014;1584:39–51. doi: 10.1016/j.brainres.2013.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Ihara R, Matsukawa K, Nagata Y, et al. RNA binding mediates neurotoxicity in the transgenic Drosophila model of TDP-43 proteinopathy. Hum Mol Genet. 2013;22:4474–4484. doi: 10.1093/hmg/ddt296. [DOI] [PubMed] [Google Scholar]
- 185.François-Moutal L, Felemban R, Scott DD, et al. Small molecule targeting TDP-43’s RNA recognition motifs reduces locomotor defects in a. ACS Chem Biol. 2019;14:2006–2013. doi: 10.1021/acschembio.9b00481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Berson A, Goodman LD, Sartoris AN, et al. Drosophila Ref1/ALYREF regulates transcription and toxicity associated with ALS/FTD disease etiologies. Acta Neuropathol Commun. 2019;7:65. doi: 10.1186/s40478-019-0710-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Desport JC, Torny F, Lacoste M, Preux PM, Couratier P. Hypermetabolism in ALS: correlations with clinical and paraclinical parameters. Neurodegener Dis. 2005;2:202–207. doi: 10.1159/000089626. [DOI] [PubMed] [Google Scholar]
- 188.Dupuis L, Corcia P, Fergani A, et al. Dyslipidemia is a protective factor in amyotrophic lateral sclerosis. Neurology. 2008;70:1004–1009. doi: 10.1212/01.wnl.0000285080.70324.27. [DOI] [PubMed] [Google Scholar]
- 189.Joardar A, Manzo E, Zarnescu DC. Metabolic dysregulation in amyotrophic lateral sclerosis: challenges and opportunities. Curr Genet Med Rep. 2017;5:108–114. doi: 10.1007/s40142-017-0123-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Manzo E, O'Conner AG, Barrows JM, Shreiner DD, Birchak GJ, Zarnescu DC. Medium-chain fatty acids, beta-hydroxybutyric acid and genetic modulation of the carnitine shuttle are protective in a Drosophila model of ALS based on TDP-43. Front Mol Neurosci. 2018;11:182. doi: 10.3389/fnmol.2018.00182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Manzo E, Lorenzini I, Barrameda D, et al. Glycolysis upregulation is neuroprotective as a compensatory mechanism in ALS. Elife. 2019 doi: 10.7554/eLife.45114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Pulst SM, Nechiporuk A, Nechiporuk T, et al. Moderate expansion of a normally biallelic trinucleotide repeat in spinocerebellar ataxia type 2. Nat Genet. 1996;14:269–276. doi: 10.1038/ng1196-269. [DOI] [PubMed] [Google Scholar]
- 193.Sanpei K, Takano H, Igarashi S, et al. Identification of the spinocerebellar ataxia type 2 gene using a direct identification of repeat expansion and cloning technique, DIRECT. Nat Genet. 1996;14:277–284. doi: 10.1038/ng1196-277. [DOI] [PubMed] [Google Scholar]
- 194.Satterfield TF, Jackson SM, Pallanck LJ. A Drosophila homolog of the polyglutamine disease gene SCA2 is a dosage-sensitive regulator of actin filament formation. Genetics. 2002;162:1687–1702. doi: 10.1093/genetics/162.4.1687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Nonhoff U, Ralser M, Welzel F, et al. Ataxin-2 interacts with the DEAD/H-box RNA helicase DDX6 and interferes with P-bodies and stress granules. Mol Biol Cell. 2007;18:1385–1396. doi: 10.1091/mbc.e06-12-1120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.van den Heuvel DM, Harschnitz O, van den Berg LH, Pasterkamp RJ. Taking a risk: a therapeutic focus on ataxin-2 in amyotrophic lateral sclerosis? Trends Mol Med. 2014;20:25–35. doi: 10.1016/j.molmed.2013.09.001. [DOI] [PubMed] [Google Scholar]
- 197.Elden AC, Kim HJ, Hart MP, et al. Ataxin-2 intermediate-length polyglutamine expansions are associated with increased risk for ALS. Nature. 2010;466:1069–1075. doi: 10.1038/nature09320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Bakthavachalu B, Huelsmeier J, Sudhakaran IP, et al. RNP-granule assembly via ataxin-2 disordered domains is required for long-term memory and neurodegeneration. Neuron. 2018;98:754–766.e754. doi: 10.1016/j.neuron.2018.04.032. [DOI] [PubMed] [Google Scholar]
- 199.Kim HJ, Raphael AR, LaDow ES, et al. Therapeutic modulation of eIF2α phosphorylation rescues TDP-43 toxicity in amyotrophic lateral sclerosis disease models. Nat Genet. 2014;46:152–160. doi: 10.1038/ng.2853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Satterfield TF, Pallanck LJ. Ataxin-2 and its Drosophila homolog, ATX2, physically assemble with polyribosomes. Hum Mol Genet. 2006;15:2523–2532. doi: 10.1093/hmg/ddl173. [DOI] [PubMed] [Google Scholar]
- 201.Chou CC, Alexeeva OM, Yamada S, et al. PABPN1 suppresses TDP-43 toxicity in ALS disease models. Hum Mol Genet. 2015;24:5154–5173. doi: 10.1093/hmg/ddv238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.McGurk L, Gomes E, Guo L, et al. Poly(ADP-Ribose) prevents pathological phase separation of TDP-43 by promoting liquid demixing and stress granule localization. Mol Cell. 2018;71:703–717.e709. doi: 10.1016/j.molcel.2018.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Duan Y, Du A, Gu J, et al. PARylation regulates stress granule dynamics, phase separation, and neurotoxicity of disease-related RNA-binding proteins. Cell Res. 2019;29:233–247. doi: 10.1038/s41422-019-0141-z. [DOI] [PMC free article] [PubMed] [Google Scholar]