Abstract
Diabetes mellitus (DM) is an independent risk factor for atrial fibrillation (AF), which is the most common sustained arrhythmia and is associated with substantial morbidity and mortality. Advanced glycation end product and its receptor activation, cardiac energy dysmetabolism, structural and electrical remodeling, and autonomic dysfunction are implicated in AF pathophysiology in diabetic hearts. Antidiabetic drugs have been demonstrated to possess therapeutic potential for AF. However, clinical investigations of AF in patients with DM have been scant and inconclusive. This article provides a comprehensive review of research findings on the association between DM and AF and critically analyzes the effect of different pharmacological classes of antidiabetic drugs on AF.
Keywords: DPP-4, GLP-1, Metformin, SGLT2, Thiazolidinedione
Introduction
Diabetes mellitus (DM) is a complex chronic disease, and its prevalence continues to increase worldwide [1]: it was 6.4% among adults in 2010, and this rate is expected to increase to 7.7% by 2030 [2]. Accumulating evidence suggests that DM is an independent risk factor for atrial fibrillation (AF), which is the most common sustained arrhythmia [3, 4]. AF not only causes heart failure and stroke, but also increases the risk of myocardial infarction. Clinical studies showed that AF doubles the risk of myocardial infarction [5, 6]. Therefore, AF has a critical impact on human health and socioeconomic burden. The Framingham Heart Study comprising 4731 participants demonstrated that DM significantly increased the risk of AF during up to 38 years of follow-up [3]. The Women’s Health Initiative Observational Study involving 81,892 postmenopausal women reported that patients with DM had a 55% increased risk of AF over an average follow-up period of 9.8 years [7]. A meta-analysis of 29 studies that included 8,037,756 individuals reported that DM was associated with a 49% increase in the development of AF [8]. This analysis also showed that women with DM were 24% more likely to develop AF than men with DM. Moreover, an investigation on 228 patients with paroxysmal AF who had undergone catheter ablation found that patients with DM or impaired fasting glucose had a higher AF recurrence rate than did those with normal glucose metabolism [9]. A population-based case–control study (including 1410 people with new-onset AF and 2203 controls without AF in the United States) indicated that the risk of AF was significantly higher in people who had DM for over 5 years or average hemoglobin A1c (HbA1c) level above 7.0%. The estimated risk of AF increases by 3% per additional year of DM duration, supporting a causal relationship between DM and AF [10]. By contrast, a meta-analysis involving 4357 patients with paroxysmal AF, 1083 with persistent AF, and 1777 with long-standing AF revealed that DM was not an independent predictor of AF recurrence [11]. An investigation on 496 patients receiving implantable electronic devices found that DM was not a predictor of atrial high-rate episodes which are associated with an increased risk of developing AF [12]. This finding suggests that DM may not significantly contribute to the occurrence of pre-clinical AF. Therefore, the discrepancy in type, duration, or severity of DM; treatment with different antidiabetic drugs; and variable adjustment for confounding risk factors may lead to contradictory results in clinical studies regarding the link between DM and AF.
The results of the Action to Control Cardiovascular Risk in Diabetes study indicated that the incidence of new-onset AF was similar between the intensive treatment group (targeting HbA1c level < 6.0%) and the standard therapy group (targeting HbA1c level of 7.0–7.9%) [13], implying that glycemic control alone is not adequate to mitigate AF risk. Furthermore, several prospective cardiovascular outcome trials have demonstrated that treatment with either glucagon-like peptide 1 (GLP-1) receptor agonists or sodium–glucose cotransporter-2 (SGLT2) inhibitors, but not dipeptidylpeptidase-4 (DPP-4) inhibitors, reduced cardiovascular events in patients with type 2 DM and established cardiovascular disease [14], suggesting that antidiabetic drug-specific properties might have diverse effects on the cardiovascular system. Antidiabetic drugs may have varying effects on AF risk through their distinctive biological effects and pharmacological mechanisms. In this review, we evaluate the pathological mechanisms of DM in the genesis of AF and highlight the potential role of different antidiabetic drugs on the risk of AF from laboratory and clinical evidence.
Pathophysiological mechanisms of DM in the genesis of AF
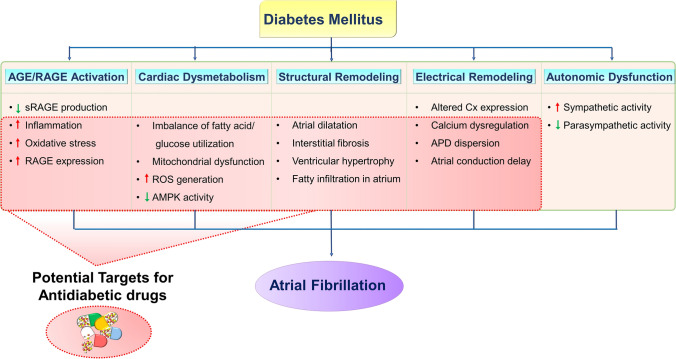
The pathogenesis through which DM promotes the development of AF remains incompletely elucidated. However, as shown in Fig. 1, advanced glycation end product (AGE) and receptor for AGE (RAGE) activation; cardiac energy dysmetabolism; structural and electrical remodeling; and autonomic dysfunction in the atria have been hypothesized to contribute to increased AF risk in patients with DM [15–17].
Fig. 1.
Schematic illustration of the proposed mechanisms underlying the effects of DM on AF arrhythmogenesis, and potential therapeutic targets for antidiabetic drugs. AGE/RAGE activation, cardiac dysmetabolism, structural and electrical remodeling, and autonomic dysfunction in the atria contribute to the development of AF in DM. AGE advanced glycation end product, AMPK adenosine monophosphate-activated protein kinase, APD action potential duration, Cx connexion, RAGE receptor for AGE, ROS reactive oxygen species, sRAGE soluble RAGE
Atrial structural remodeling in DM and AF
Chronic inflammatory and oxidative stress, activation of the renin–angiotensin system, alternation of gap junction protein expression, and upregulation of AGE/RAGE signaling all contribute to atrial structural remodeling in diabetic hearts [15, 18], which is characterized by atrial dilatation and interstitial fibrosis. A study in 2623 Framingham Heart Study participants revealed that those with higher insulin resistance and hyperglycemia had increased left ventricular mass [19]. Another retrospective analysis involving 4014 patients in Japan found that left ventricular hypertrophy was accompanied by poor left ventricular function, large left atrial size, and high prevalence of AF [20]. Compared with those from individuals without DM, atrial fibroblasts from patients with type 2 DM expressed higher levels of type 1 collagen, which may induce interatrial conduction delay and enhance AF occurrence [17, 21].
AGE/RAGE activation: signaling mechanism for DM-mediated AF
AGEs are generated and accumulated during hyperglycemia, oxidative stress, inflammation, aging, and renal failure [22, 23]. Cross-linking of AGEs and type I collagen and elastin in the extracellular matrix in diabetic hearts induces atrial fibrosis [24]. Moreover, AGEs bind to their transmembrane receptor, RAGE, eliciting inflammatory and oxidative responses, thus promoting the development of AF [25]. Administration of either an AGE-formation inhibitor or an AGE cross-link breaker suppressed DM-induced atrial fibrosis [24, 26]. Furthermore, soluble RAGE (sRAGE), the truncated isoform of the membrane-bound full-length RAGE, may act as an endogenous competitive inhibitor of RAGE in circulation [27]. Plasma AGEs and sRAGE levels were significantly higher in AF patients both with and without DM than in those who had sinus rhythm [28, 29]. By contrast, another study demonstrated that high plasma sRAGE levels were associated with low AF recurrence after catheter ablation in patients with DM [30]. In our previous studies, we found that RAGE may be downregulated by calcitriol with increased sRAGE production, which may contribute to the known effects of vitamin D on AF risk reduction through big data analysis in 20,788 osteoporosis women over a 5-year follow-up [23, 31].
Cardiac dysmetabolism in diabetic cardiomyopathy: effect on AF genesis
Fatty acids and glucose are principle substrates for myocardial energy metabolism. Fatty acid metabolism is less efficient because it consumes more oxygen to produce adenosine triphosphate (ATP) than does glucose metabolism. Therefore, fatty acid β-oxidation constitutes the major energy source in a normal heart, whereas glycolysis predominates during pathological stimuli, such as AF [32, 33]. Because of insulin resistance, the diabetic heart has increased fatty acid utilization and decreased glucose uptake; thus, it is more vulnerable to ischemic injury because of its constrained fuel substrate flexibility [32]. Furthermore, excessive fatty acid uptake in the diabetic heart results in altered mitochondrial architecture and decreased mitochondrial oxidative phosphorylation, leading to toxic lipid metabolite accumulation and reactive oxygen species (ROS) generation [34, 35]. The mitochondria in the atrial tissue of patients with DM demonstrated sharply decreased respiration capacity and increased oxidative stress [36]. Additionally, the diabetic heart is associated with an increase in epicardial adipose tissue infiltration, leading to AF arrhythmogenesis [17, 37].
Adenosine monophosphate-activated protein kinase (AMPK) is a major regulator of cardiac metabolism and is activated in response to energy depletion. AMPK activation promotes fatty acid and glucose uptake and facilitates subsequent β-oxidation and glycolysis, thus increasing ATP production in cardiomyocytes [38, 39]. When facing AF-induced metabolic stress, AMPK activation might contribute to greater resistance to AF progression by restoring atrial calcium homeostasis [40]. Moreover, mice with inactive AMPK developed spontaneous AF [41]. Our previous studies have revealed that cardiac AMPK activity was suppressed in diabetic animal models [42, 43]. Thus, AMPK activation that reduces metabolic stress may protect against the development or perpetuation of AF in DM.
Atrial electrical remodeling in DM and AF
The main features of electrical remodeling in the diabetic atrium include increased slowing and heterogeneity of electrical conduction, prolongation of action potential duration (APD), increased spatial dispersion and frequency-dependent shortening of APD, and increased incidence of APD alternans [15, 44]. Changes in Na+, K+, and Ca2+ currents may contribute to alterations in atrial action potential morphology and affect conduction velocity or susceptibility to triggered activity in diabetic hearts [15, 17]. Decreased Na+ current and reduced expression of ion channel proteins Kv4.3, Kv1.5, and Cav1.2 have been observed in the atria of diabetic animals [45–47]. An atrial action potential analysis in patients with AF indicated that L-type Ca2+ current (ICa-L) was strongly decreased [48]. Moreover, gap junctions are composed of connexin (Cx) proteins and play a crucial role in electrical conduction in the heart. Cx40 and Cx43 are predominant isoforms in the atrial myocardium [49]. Altered Cx expression in atrium was commonly found in diabetic animals [17, 44]. DM affects the expression and distribution of Cxs, resulting in atrial conduction abnormalities and ensuing AF development.
DM autonomic dysfunction in AF initiation
Autonomic dysfunction critically initiate the occurrence of AF. The importance of autonomic dysfunction in AF pathogenesis has been widely recognized [50]. Activation of either sympathetic or parasympathetic nerve activity may enhance the genesis of AF through different mechanisms [51]. An analysis of heart rate variability in patients with DM revealed that DM-increased sympathetic and reduced parasympathetic activity in the heart [52]. Autonomic dysfunction was linked to increased episodes of asymptomatic AF, which were evaluated through 48-h Holter’s ECG monitoring in patients with type 2 DM [53]. Heart rate recovery, another index of cardiac autonomic function, is reportedly an independent predictor of AF risk in patients with type 2 DM [54]. These findings suggest that autonomic remodeling plays an essential role in increased AF vulnerability in DM. However, the underlying cellular mechanisms remain largely unknown and require further investigation.
Effect of antidiabetic drugs on AF: beyond glycemic control
Glucose fluctuations stimulated ROS production, activated sympathoadrenal activity and contributed to epinephrine-induced fall in plasma levels of potassium, thereby promoting AF genesis [55–57]. A retrospective analysis in 1,509,280 Korean patients with type 2 DM aged between 30 and 75 years showed that severe hypoglycemia led to a significant increase of 10% in subsequent AF occurrence during the follow-up period of 8.5 years [58]. Therefore, antidiabetic drugs causing more hypoglycemia, such as sulfonylureas and insulin, may increase the risk of AF, and drugs causing less hypoglycemia may reduce the risk of AF.
In addition to glycemic control, multiple clinical studies have demonstrated that antidiabetic drugs may have differing effects on the risk of new-onset AF according to their distinctive biological mechanisms (Table 1).
Table 1.
Effect of antidiabetic drugs on the risk of new-onset atrial fibrillation in patients with diabetes mellitus
| Antidiabetic drug | Study type | Patient number | Subject of study | Main results |
|---|---|---|---|---|
| Metformin | Retrospective cohort study | 645,710 | NHIRD in Taiwan (1999–2010)‚ mean follow-up 5.4 years | Metformin users had a lower risk of new-onset AF as compared with nonusers (HR: 0.81) [62] |
| Sulfonylurea | Case–control study | 14,410 | NHIRD in Taiwan (2004–2013)‚ 2882 new-onset AF and 11,528 controls | No association between use of sulfonylurea and new-onset AF [67] |
| TZD | Retrospective cohort study | 108,624 | Danish nationwide registry (2000–2012)‚ patients treated with metformin or sulfonylurea as first-line drugs | TZD was associated with a reduced risk of new-onset AF as compared with other second-line antidiabetic drugs (HR: 0.76) [71] |
| Retrospective cohort study | 12,065 | NHIRD in Taiwan (2000–2007)‚ mean follow-up 5.3 years | Rosiglitazone users had a lower risk of new-onset AF as compared with nonusers (HR: 0.69) [70] | |
| DPP-4 inhibitors | Retrospective cohort study | 90,880 | NHIRD in Taiwan (2009–2012)‚ patients treated with metformin as first-line therapy‚ mean follow-up 2.4 years | DPP-4 inhibitors was associated with a decreased risk of new-onset AF as compared with other second-line antidiabetic drugs (HR: 0.65) [77] |
| SGLT2 inhibitors | Randomized controlled trial, post-hoc analysis | 17,160 | Patients with DM and high CV risk‚ median follow-up 4.2 years | Dapagliflozin was associated with a reduced risk of new-onset AF as compared with placebo (HR: 0.81) [92] |
| Insulin | Case–control study | 14,410 | NHIRD in Taiwan (2004–2013)‚ 2882 new-onset AF and 11,528 controls | Users of insulin were at a higher risk of new-onset AF than were nonusers (OR: 1.19) [67] |
AF atrial fibrillation, CV cardiovascular, DM diabetes mellitus, DPP-4 dipeptidylpeptidase-4, HR hazard ratio, NHIRD National Health Insurance Research Database, OR odds ratio, SGLT2 sodium–glucose cotransporter-2, TZD thiazolidinedione
Metformin
Metformin is currently regarded as the first-line drug treatment for type 2 DM [59, 60]. Metformin reduces hepatic glucose production and increases the insulin sensitivity of the liver, skeletal muscle, and adipose tissue [61]. Analysis of data from Taiwan’s National Health Insurance Research Database (NHIRD) revealed that treatment with metformin was associated with a 19% decrease in new-onset AF among 645,710 patients (mean age = 58.6 years) newly diagnosed with type 2 DM over a follow-up period of 13 years [62]. By contrast, a retrospective cohort study comprising 1283 patients with DM undergoing a coronary artery bypass graft or cardiac valve surgery indicated that prior use of metformin was not associated with a decreased rate of postoperative AF [63].
Metformin attenuated tachypacing-induced ROS generation and myofibril degradation in mouse atrial myocytes [62]. Metformin reversed electrophysiological changes and reduced lipid accumulation in the left atrial appendage in dogs with atrial rapid pacing [64]. Through acting as an AMPK activator [65], metformin might improve dysmetabolism in diabetic hearts. These laboratory findings have supported the beneficial effects of metformin on AF.
Sulfonylurea
Sulfonylureas bind to and close ATP-sensitive potassium channels in pancreatic β-cells, inducing the release of insulin [66]. Treatment with sulfonylureas may cause hypoglycemia which was hypothesized to result in a high AF risk. However, a nested case–control study using data from Taiwan’s NHIRD (2882 patients with DM and new-onset AF and 11,528 patients with DM without AF) indicated that the use of sulfonylureas was not associated with the risk of new-onset AF [67]. A clinical investigation involving 1310 patients with DM hospitalized for acute myocardial infarction revealed that prior use of sulfonylureas was not related to the occurrence of in-hospital AF. Among patients treated with sulfonylureas, those using gliclazide or glimepiride had fewer in-hospital arrhythmias than did those treated with glibenclamide [68]. Therefore, individual sulfonylureas have distinctive pharmacological effects on the myocardium and may explain the neutral association between sulfonylurea treatment and AF risk in clinical investigations of patients with DM.
Thiazolidinedione
Thiazolidinedione (TZD) is a synthetic peroxisome proliferator-activated receptor-γ agonist that promotes glucose and fatty acid uptake in the adipose tissue, thus ameliorating insulin resistance. Abundant clinical evidence supports the preventive effect of TZD on AF occurrence in patients with DM. A prospective observational cohort study of 150 patients with DM undergoing catheter ablation of drug-refractory paroxysmal AF reported that the treatment of participants with pioglitazone before ablation was associated with a significantly lower risk of AF recurrence compared with that of those without pioglitazone treatment [69]. Another cohort study analyzing NHIRD data demonstrated that treatment with rosiglitazone was linked to a 31% reduction in the risk of new-onset AF among 12,065 patients with DM who were followed up for an average of 63 months [70]. Similarly, a Danish nationwide study that included 108,624 patients with DM without prior AF who were treated with metformin or sulfonylurea as a first-line antidiabetic drug found that treatment with TZD, as a second-line antidiabetic drug, was correlated to a 24% decrease in the risk of AF compared with other second-line antidiabetic drugs [71]. A recent meta-analysis of seven studies investigating the potential effects of TZDs on AF (including three randomized clinical trials and four observational studies) that included 130,854 patients with DM revealed that those treated with TZD had a significantly lower risk of AF than did controls. However, the decreased AF occurrence risk caused by TZD treatment was significant only in the pooled analysis of observational studies and not in the analysis of randomized clinical trials [72]. Therefore, large-scale prospective randomized clinical trials that are designed to evaluate AF as a primary outcome are required to clarify the treatment potential of TZD in patients with DM.
Substantial experimental evidence indicates that pioglitazone prevented atrial fibrosis and inhibited AF development through its anti-inflammatory and antioxidant effects [73–75]. Pioglitazone also suppressed angiotensin II-induced ICa-L channel α1c expression and electrical remodeling in atrial myocytes [76]. These laboratory findings suggest the potential electrophysiological effects of TZD on the AF pathogenesis.
DPP-4 inhibitors
Inhibition of DPP-4 enzyme increases serum levels of GLP-1, which promotes insulin release and inhibits glucagon secretion in a glucose-dependent manner, thereby lowering blood glucose levels and rarely causing hypoglycemia. A retrospective cohort study in Taiwan involving 90,880 patients with DM taking metformin as first-line therapy indicated that patients using DPP-4 inhibitor as second-line therapy had a 35% lower risk of new-onset AF than did those treated with other antidiabetic drugs over a mean follow-up period of 2.4 years [77]. By contrast, several prospective cardiovascular outcome trials that studied alogliptin, saxagliptin, and linagliptin have not reported any significant correlation between DPP-4 inhibitors and AF occurrence, probably because these studies were not designed primarily to investigate AF incidence [78, 79].
Our previous study found that sitagliptin possessed antiarrhythmic potential, possibly through its inhibition on inflammation and RAGE expression in spontaneously hypertensive rat hearts. An increase in protein expression of Cav1.2 caused by hypertension was reversed after sitagliptin treatment [80]. Alogliptin and linagliptin reduced AF inducibility by reducing atrial interstitial fibrosis and myocyte hypertrophy, improving mitochondrial function, promoting mitochondrial biogenesis, and mitigating oxidative stress [81, 82]. Collectively, anti-inflammation, antioxidation, RAGE suppression, cardiac metabolism and calcium homeostasis modulation are involved in the potential protective effects of DPP4-inhibitors against AF.
GLP-1 receptor agonists
Treatment with DPP4-inhibitors induces a physiological increase in GLP-1 levels, whereas use of GLP-1 receptor agonists provides supraphysiological levels of GLP-1. In addition to glucose-dependent stimulation of insulin release and inhibition of glucagon secretion, these supraphysiological GLP-1 levels slow gastric emptying and suppress the appetite. Accordingly, GLP-1 receptor agonists offer superior glycemic control and weight loss compared with DPP-4 inhibitors. The weight loss benefit of GLP-1 receptor agonist treatment suggests that it can decrease the risk of AF occurrence in DM. However, clinical data have demonstrated that treatment with long-acting GLP-1 receptor agonists is associated with a modest increase in resting heart rate [83]. A meta-analysis regarding the cardiovascular safety of albiglutide, which included eight phase 3 trials and one phase 2b trial, revealed that more patients had AF in the albiglutide group than in the comparison group [84]. Although these events were not confirmed, these results have prompted further study to assess the effect of albiglutide on AF. However, a recent cardiovascular outcome trial of once-weekly exenatide reported that the incidence of AF did not differ significantly between patients who received exenatide and those who received placebo [85]. Another meta-analysis of 31 randomized controlled trials (that enrolled 17,966 patients with DM in a GLP-1 receptor agonist group and 15,305 patients in comparison groups, with a mean age of 57 years) indicated that treatment with GLP-1 receptor agonists was not associated with a significant increase in the incidence of new-onset AF [86]. Currently, clinical evidence does not support any association between GLP-1 receptor agonists and AF risk.
Exendin-4 was found to attenuate hyperglycemia-induced cardiomyocyte apoptosis by improving sarcoplasmic/endoplasmic reticulum Ca2+-ATPase 2a function [87]. Our previous study demonstrated that GLP-1 reduced ryanodine receptor S2814 phosphorylation in mouse atrial myocytes [88], which was hypothesized to reduce calcium leak and AF genesis. Therefore, GLP-1 receptor agonists might protect diabetic hearts from AF development through modulating calcium homeostasis.
SGLT2 inhibitors
SGLT2 inhibitors suppress glucose and sodium reabsorption from the renal proximal convoluted tubules, thereby promoting excretion of glucose in the urine and subsequently lowering blood glucose levels [89]. Treatment with SGLT2 inhibitors directly causes body weight loss and does not increase the risk of hypoglycemia. An observational analysis based on national registry data from Denmark, Norway, and Sweden involving 91,320 patients with DM (mean age 61 years; 25% of participants had established cardiovascular diseases at baseline) revealed that patients treated with SGLT2 inhibitors had a similar AF incidence as did those treated with other antidiabetic drugs [90]. A meta-analysis that included 35 randomized controlled trials with a total of 34,987 patients with type 2 DM found no significant difference in the occurrence of AF between SGLT2 inhibitors and placebo [91]. Although prospective cardiovascular outcome trials have demonstrated that SGLT2 inhibitors significantly reduced the risk of major adverse cardiovascular events and hospitalization for heart failure, empagliflozin and canagliflozin had no effect on the incidence of AF in patients with DM [14, 78, 79]. By contrast, a recent randomized controlled trial comprising 17,160 patients with type 2 DM who had or were at risk of atherosclerotic cardiovascular disease reported that dapagliflozin reduced AF risk and atrial flutter events by 19% over a median follow-up period of 4.2 years [92]. More clinical trials that are designed primarily to evaluate AF in well-defined populations of patients with DM are necessary for clarifying the treatment potential of SGLT2 inhibitors in DM.
Empagliflozin ameliorated atrial dilatation and fibrosis as well as improved mitochondrial function and mitochondrial biogenesis in diabetic rats [93]. Our recent study found that empagliflozin attenuated the late sodium current-induced calcium overload and arrhythmogenesis, which may potentially contribute to its anti-AF potential in diabetic hearts. Moreover, treatment with empagliflozin reversed the DM-increased sodium/hydrogen exchanger activity and oxidative stress [94]. In addition, empagliflozin simulated AMPK activation, thereby enhancing myocardial energetics and improving systolic dysfunction in a nondiabetic porcine heart failure model [95]. These findings suggest that SGLT2 inhibitors have distinctive therapeutic potential for AF in DM.
Insulin
Among antidiabetic drugs, insulin is the most efficacious in lowering blood glucose levels; however, it also poses the highest risk of inducing hypoglycemia and promoting body weight gain. Accordingly, insulin therapy was presumed to increase AF incidence in patients with DM. In a nested case–control study analyzing NHIRD data, insulin users exhibited a significant increase in the risk of new-onset AF than did nonusers (odds ratio: 1.19), even after adjusting for sex, age, DM duration, comorbidities, and concurrent medication [67]. An analysis of data from the prospective AF registry in Europe (including 1288 patients with both AF and DM, of whom 22.4% were on insulin) indicated that the adjusted hazard ratio for developing stroke or systemic embolism at 1 year was 2.61 for patients with DM on insulin therapy compared with patients with noninsulin-requiring DM [96]. By contrast, a prospective randomized controlled trial involving 12,537 patients (mean age = 63.5 years) with impaired fasting glucose, impaired glucose tolerance, or overt type 2 DM and cardiovascular risk factors reported that incidence of AF was similar between the insulin glargine and placebo groups over a median follow-up period of 6.2 years [97]. Currently, clinical findings have not clarified the pathological role of insulin therapy in the development of AF.
Multiple laboratory studies have revealed that hyperinsulinemia elicits proatherogenic responses by stimulating proliferation and migration of arterial smooth muscle cells and enhancing production of proinflammatory cytokines and endothelial adhesion molecules [98]. Insulin causes vasoconstriction in the insulin-resistant endothelium [99]. Therefore, high insulin concentrations in the blood of insulin-treated patients may provide a substrate for AF genesis in DM.
Conclusions
Antidiabetic drugs may have differing effects on AF arrhythmogenesis in patients with DM (Table 2). Sulfonylureas and insulin have a pro-AF effect through inducing hypoglycemia. Differently, clinical and laboratory evidence suggests that SGLT2 inhibitors seem to be the most promising antidiabetic drug in reducing the risk of AF in patients with DM through its multiple cardiovascular benefits. Metformin or GLP-1 receptor agonists also potentially provide cardiovascular protective effects, but their impacts on the risk of AF has not been established in patients with DM. TZD and DPP-4 inhibitors may have some anti-arrhythmogenetic potential from translational studies, but this assumption has not yet been supported by clinical investigations.
Table 2.
Potential effects of antidiabetic drugs on DM-induced AF arrhythmogenesis
| Inflammatory and oxidative stress | Cardiac dysmetabolism | AGE/RAGE signaling | Atrial structural remodeling | Electrical remodeling | Autonomic dysfunction | ||
|---|---|---|---|---|---|---|---|
| Metformin | Suppression | ↑ AMPK activity | N/A |
↓ Myofibril degradation ↓ Lipid infiltration |
N/A | N/A | |
| TZD | Suppression | N/A | N/A | ↓ Fibrosis | ↓ AngII-induced ICa-L amplification | N/A | |
| DPP-4 inhibitors | Suppression | ↑ Mitochondrial function | Suppression |
↓ Fibrosis ↓ Myocyte hypertrophy |
↓ Cav1.2 protein upregulation caused by hypertension | N/A | |
| GLP-1 receptor agonists | N/A | N/A | N/A | N/A |
↑ SERCA 2a function ↓ Ryanodine receptor phosphorylation |
N/A | |
| SGLT2 inhibitors | Suppression |
↑ AMPK activity ↑ Mitochondrial function |
N/A |
↓ Dilatation ↓ Fibrosis |
↓ INa-late-induced calcium overload ↓ Na+/H+ exchanger activity |
N/A | |
| Sulfonylurea | Promotion | N/A | N/A | N/A | N/A | N/A | |
| Insulin | Promotion | N/A | N/A | N/A | N/A | N/A | |
AngII angiotensin II, AMPK adenosine monophosphate-activated protein kinase, AGE advanced glycation end product, ICa-L L-type Ca2+ current, INa-late late sodium current, N/A not available, RAGE receptor for AGE, SERCA 2a sarcoplasmic/endoplasmic reticulum Ca2+-ATPase 2a
Acknowledgements
This work was supported by Grants from the Ministry of Science and Technology of Taiwan (MOST 107-2314-B-038 -099 -MY3) and Taipei Medical University, Wan Fang Hospital (107-wf-eva-04).
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Whiting DR, Guariguata L, Weil C, Shaw J. IDF Diabetes Atlas: global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res Clin Pract. 2011;94(3):311–321. doi: 10.1016/j.diabres.2011.10.029. [DOI] [PubMed] [Google Scholar]
- 2.Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87(1):4–14. doi: 10.1016/j.diabres.2009.10.007. [DOI] [PubMed] [Google Scholar]
- 3.Benjamin EJ, Levy D, Vaziri SM, D'Agostino RB, Belanger AJ, Wolf PA. Independent risk factors for atrial fibrillation in a population-based cohort. The Framingham Heart Study. JAMA. 1994;271(11):840–844. doi: 10.1001/jama.1994.03510350050036. [DOI] [PubMed] [Google Scholar]
- 4.Lau DH, Nattel S, Kalman JM, Sanders P. Modifiable risk factors and atrial fibrillation. Circulation. 2017;136(6):583–596. doi: 10.1161/circulationaha.116.023163. [DOI] [PubMed] [Google Scholar]
- 5.Soliman EZ, Safford MM, Muntner P, Khodneva Y, Dawood FZ, Zakai NA, Thacker EL, Judd S, Howard VJ, Howard G, Herrington DM, Cushman M. Atrial fibrillation and the risk of myocardial infarction. JAMA Intern Med. 2014;174(1):107–114. doi: 10.1001/jamainternmed.2013.11912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Violi F, Soliman EZ, Pignatelli P, Pastori D. Atrial fibrillation and myocardial infarction: a systematic review and appraisal of pathophysiologic mechanisms. J Am Heart Assoc. 2016 doi: 10.1161/jaha.116.003347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perez MV, Wang PJ, Larson JC, Soliman EZ, Limacher M, Rodriguez B, Klein L, Manson JE, Martin LW, Prineas R, Connelly S, Hlatky M, Wassertheil-Smoller S, Stefanick ML. Risk factors for atrial fibrillation and their population burden in postmenopausal women: the Women's Health Initiative Observational Study. Heart (British Cardiac Society) 2013;99(16):1173–1178. doi: 10.1136/heartjnl-2013-303798. [DOI] [PubMed] [Google Scholar]
- 8.Xiong Z, Liu T, Tse G, Gong M, Gladding PA, Smaill BH, Stiles MK, Gillis AM, Zhao J. A machine learning aided systematic review and meta-analysis of the relative risk of atrial fibrillation in patients with diabetes mellitus. Front Physiol. 2018;9:835. doi: 10.3389/fphys.2018.00835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chao TF, Suenari K, Chang SL, Lin YJ, Lo LW, Hu YF, Tuan TC, Tai CT, Tsao HM, Li CH, Ueng KC, Wu TJ, Chen SA. Atrial substrate properties and outcome of catheter ablation in patients with paroxysmal atrial fibrillation associated with diabetes mellitus or impaired fasting glucose. Am J Cardiol. 2010;106(11):1615–1620. doi: 10.1016/j.amjcard.2010.07.038. [DOI] [PubMed] [Google Scholar]
- 10.Dublin S, Glazer NL, Smith NL, Psaty BM, Lumley T, Wiggins KL, Page RL, Heckbert SR. Diabetes mellitus, glycemic control, and risk of atrial fibrillation. J Gen Intern Med. 2010;25(8):853–858. doi: 10.1007/s11606-010-1340-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.D'Ascenzo F, Corleto A, Biondi-Zoccai G, Anselmino M, Ferraris F, di Biase L, Natale A, Hunter RJ, Schilling RJ, Miyazaki S, Tada H, Aonuma K, Yenn-Jiang L, Tao H, Ma C, Packer D, Hammill S, Gaita F. Which are the most reliable predictors of recurrence of atrial fibrillation after transcatheter ablation?: a meta-analysis. Int J Cardiol. 2013;167(5):1984–1989. doi: 10.1016/j.ijcard.2012.05.008. [DOI] [PubMed] [Google Scholar]
- 12.Pastori D, Miyazawa K, Li Y, Shahid F, Hado H, Lip GYH. Inflammation and the risk of atrial high-rate episodes (AHREs) in patients with cardiac implantable electronic devices. Clin Res Cardiol. 2018;107(9):772–777. doi: 10.1007/s00392-018-1244-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fatemi O, Yuriditsky E, Tsioufis C, Tsachris D, Morgan T, Basile J, Bigger T, Cushman W, Goff D, Soliman EZ, Thomas A, Papademetriou V. Impact of intensive glycemic control on the incidence of atrial fibrillation and associated cardiovascular outcomes in patients with type 2 diabetes mellitus (from the Action to Control Cardiovascular Risk in Diabetes Study) Am J Cardiol. 2014;114(8):1217–1222. doi: 10.1016/j.amjcard.2014.07.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Home P. Cardiovascular outcome trials of glucose-lowering medications: an update. Diabetologia. 2019;62(3):357–369. doi: 10.1007/s00125-018-4801-1. [DOI] [PubMed] [Google Scholar]
- 15.Goudis CA, Korantzopoulos P, Ntalas IV, Kallergis EM, Liu T, Ketikoglou DG. Diabetes mellitus and atrial fibrillation: pathophysiological mechanisms and potential upstream therapies. Int J Cardiol. 2015;184:617–622. doi: 10.1016/j.ijcard.2015.03.052. [DOI] [PubMed] [Google Scholar]
- 16.Serban RC, Scridon A. Data linking diabetes mellitus and atrial fibrillation-how strong is the evidence? From epidemiology and pathophysiology to therapeutic implications. Can J Cardiol. 2018;34(11):1492–1502. doi: 10.1016/j.cjca.2018.08.018. [DOI] [PubMed] [Google Scholar]
- 17.Bohne LJ, Johnson D, Rose RA, Wilton SB, Gillis AM. The association between diabetes mellitus and atrial fibrillation: clinical and mechanistic insights. Front Physiol. 2019;10:135. doi: 10.3389/fphys.2019.00135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tadic M, Cuspidi C. Type 2 diabetes mellitus and atrial fibrillation: from mechanisms to clinical practice. Arch Cardiovasc Dis. 2015;108(4):269–276. doi: 10.1016/j.acvd.2015.01.009. [DOI] [PubMed] [Google Scholar]
- 19.Rutter MK, Parise H, Benjamin EJ, Levy D, Larson MG, Meigs JB, Nesto RW, Wilson PW, Vasan RS. Impact of glucose intolerance and insulin resistance on cardiac structure and function: sex-related differences in the Framingham Heart Study. Circulation. 2003;107(3):448–454. doi: 10.1161/01.cir.0000045671.62860.98. [DOI] [PubMed] [Google Scholar]
- 20.Seko Y, Kato T, Haruna T, Izumi T, Miyamoto S, Nakane E, Inoko M. Association between atrial fibrillation, atrial enlargement, and left ventricular geometric remodeling. Sci Rep. 2018;8(1):6366. doi: 10.1038/s41598-018-24875-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sedgwick B, Riches K, Bageghni SA, O'Regan DJ, Porter KE, Turner NA. Investigating inherent functional differences between human cardiac fibroblasts cultured from nondiabetic and type 2 diabetic donors. Cardiovasc Pathol. 2014;23(4):204–210. doi: 10.1016/j.carpath.2014.03.004. [DOI] [PubMed] [Google Scholar]
- 22.Lee TW, Lee TI, Chang CJ, Lien GS, Kao YH, Chao TF, Chen YJ. Potential of vitamin D in treating diabetic cardiomyopathy. Nutr Res. 2015;35(4):269–279. doi: 10.1016/j.nutres.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 23.Lee TW, Kao YH, Lee TI, Chen YJ. ADAM10 modulates calcitriol-regulated RAGE in cardiomyocytes. Eur J Clin Invest. 2017;47(9):675–683. doi: 10.1111/eci.12789. [DOI] [PubMed] [Google Scholar]
- 24.Zhang Q, Li G, Liu T. Receptor for advanced glycation end products (RAGE): novel biomarker and therapeutic target for atrial fibrillation. Int J Cardiol. 2013;168(5):4802–4804. doi: 10.1016/j.ijcard.2013.07.038. [DOI] [PubMed] [Google Scholar]
- 25.Yamagishi SI, Sotokawauchi A, Matsui T. Pathological role of advanced glycation end products (AGEs) and their receptor axis in atrial fibrillation. Mini Rev Med Chem. 2019;19(13):1040–1048. doi: 10.2174/1389557519666190311140737. [DOI] [PubMed] [Google Scholar]
- 26.Kato T, Yamashita T, Sekiguchi A, Tsuneda T, Sagara K, Takamura M, Kaneko S, Aizawa T, Fu LT. AGEs-RAGE system mediates atrial structural remodeling in the diabetic rat. J Cardiovasc Electrophysiol. 2008;19(4):415–420. doi: 10.1111/j.1540-8167.2007.01037.x. [DOI] [PubMed] [Google Scholar]
- 27.Lee TW, Kao YH, Chen YJ, Chao TF, Lee TI. Therapeutic potential of vitamin D in AGE/RAGE-related cardiovascular diseases. Cell Mol Life Sci CMLS. 2019 doi: 10.1007/s00018-019-03204-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lancefield TF, Patel SK, Freeman M, Velkoska E, Wai B, Srivastava PM, Horrigan M, Farouque O, Burrell LM. The receptor for advanced glycation end products (RAGE) is associated with persistent atrial fibrillation. PLoS ONE. 2016;11(9):e0161715. doi: 10.1371/journal.pone.0161715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Raposeiras-Roubin S, Rodino-Janeiro BK, Grigorian-Shamagian L, Seoane-Blanco A, Moure-Gonzalez M, Varela-Roman A, Alvarez E, Gonzalez-Juanatey JR. Evidence for a role of advanced glycation end products in atrial fibrillation. Int J Cardiol. 2012;157(3):397–402. doi: 10.1016/j.ijcard.2011.05.072. [DOI] [PubMed] [Google Scholar]
- 30.Yang PS, Kim TH, Uhm JS, Park S, Joung B, Lee MH, Pak HN. High plasma level of soluble RAGE is independently associated with a low recurrence of atrial fibrillation after catheter ablation in diabetic patient. Europace Eur Pacing Arrhythm Card Electrophysiol. 2016;18(11):1711–1718. doi: 10.1093/europace/euv449. [DOI] [PubMed] [Google Scholar]
- 31.Yang H-Y, Huang J-H, Chiu H-W, Lin Y-K, Hsu C-Y, Chen Y-J. Vitamin D and bisphosphonates therapies for osteoporosis are associated with different risks of atrial fibrillation in women: a nationwide population-based analysis. Medicine (Baltimore) 2018;97(43):e12947–e12947. doi: 10.1097/MD.0000000000012947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee TW, Bai KJ, Lee TI, Chao TF, Kao YH, Chen YJ. PPARs modulate cardiac metabolism and mitochondrial function in diabetes. J Biomed Sci. 2017;24(1):5. doi: 10.1186/s12929-016-0309-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Harada M, Melka J, Sobue Y, Nattel S. Metabolic Considerations in Atrial Fibrillation- Mechanistic Insights and Therapeutic Opportunities. Circ J. 2017;81(12):1749–1757. doi: 10.1253/circj.CJ-17-1058. [DOI] [PubMed] [Google Scholar]
- 34.Duncan JG. Mitochondrial dysfunction in diabetic cardiomyopathy. Biochimica et Biophysica Acta (BBA) Mol Cell Res. 2011;1813(7):1351–1359. doi: 10.1016/j.bbamcr.2011.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bugger H. Abel ED (2008) Molecular mechanisms for myocardial mitochondrial dysfunction in the metabolic syndrome. Clin Sci (London, England: 1979) 1979;114(3):195–210. doi: 10.1042/cs20070166. [DOI] [PubMed] [Google Scholar]
- 36.Anderson EJ, Kypson AP, Rodriguez E, Anderson CA, Lehr EJ, Neufer PD. Substrate-specific derangements in mitochondrial metabolism and redox balance in the atrium of the type 2 diabetic human heart. J Am Coll Cardiol. 2009;54(20):1891–1898. doi: 10.1016/j.jacc.2009.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mahajan R, Lau DH, Brooks AG, Shipp NJ, Manavis J, Wood JP, Finnie JW, Samuel CS, Royce SG, Twomey DJ, Thanigaimani S, Kalman JM, Sanders P. Electrophysiological, electroanatomical, and structural remodeling of the atria as consequences of sustained obesity. J Am Coll Cardiol. 2015;66(1):1–11. doi: 10.1016/j.jacc.2015.04.058. [DOI] [PubMed] [Google Scholar]
- 38.An D, Rodrigues B. Role of changes in cardiac metabolism in development of diabetic cardiomyopathy. Am J Physiol Heart Circ Physiol. 2006;291(4):H1489–1506. doi: 10.1152/ajpheart.00278.2006. [DOI] [PubMed] [Google Scholar]
- 39.Bairwa SC, Parajuli N. Dyck JR (2016) The role of AMPK in cardiomyocyte health and survival. Biochem Biophys Acta. 1862;12:2199–2210. doi: 10.1016/j.bbadis.2016.07.001. [DOI] [PubMed] [Google Scholar]
- 40.Harada M, Tadevosyan A, Qi X, Xiao J, Liu T, Voigt N, Karck M, Kamler M, Kodama I, Murohara T, Dobrev D, Nattel S. Atrial fibrillation activates AMP-dependent protein kinase and its regulation of cellular calcium handling: potential role in metabolic adaptation and prevention of progression. J Am Coll Cardiol. 2015;66(1):47–58. doi: 10.1016/j.jacc.2015.04.056. [DOI] [PubMed] [Google Scholar]
- 41.Harada M, Nattel SN, Nattel S. AMP-activated protein kinase: potential role in cardiac electrophysiology and arrhythmias. Circ Arrhythm Electrophysiol. 2012;5(4):860–867. doi: 10.1161/circep.112.972265. [DOI] [PubMed] [Google Scholar]
- 42.Lee TI, Kao YH, Chen YC, Tsai WC, Chung CC, Chen YJ. Cardiac metabolism, inflammation, and peroxisome proliferator-activated receptors modulated by 1,25-dihydroxyvitamin D3 in diabetic rats. Int J Cardiol. 2014;176(1):151–157. doi: 10.1016/j.ijcard.2014.07.021. [DOI] [PubMed] [Google Scholar]
- 43.Lee TI, Kao YH, Tsai WC, Chung CC, Chen YC, Chen YJ (2016) HDAC inhibition modulates cardiac PPARs and fatty acid metabolism in diabetic cardiomyopathy. PPAR Research 2016 [DOI] [PMC free article] [PubMed]
- 44.Watanabe M, Yokoshiki H, Mitsuyama H, Mizukami K, Ono T, Tsutsui H. Conduction and refractory disorders in the diabetic atrium. Am J Physiol Heart Circ Physiol. 2012;303(1):H86–95. doi: 10.1152/ajpheart.00010.2012. [DOI] [PubMed] [Google Scholar]
- 45.Liu C, Liu R, Fu H, Li J, Wang X, Cheng L, Korantzopoulos P, Tse G, Li G, Liu T. Pioglitazone attenuates atrial remodeling and vulnerability to atrial fibrillation in alloxan-induced diabetic rabbits. Cardiovasc Ther. 2017 doi: 10.1111/1755-5922.12284. [DOI] [PubMed] [Google Scholar]
- 46.Liu C, Fu H, Li J, Yang W, Cheng L, Liu T, Li G. Hyperglycemia aggravates atrial interstitial fibrosis, ionic remodeling and vulnerability to atrial fibrillation in diabetic rabbits. Anadolu kardiyoloji dergisi AKDe Anatol J Cardiol. 2012;12(7):543–550. doi: 10.5152/akd.2012.188. [DOI] [PubMed] [Google Scholar]
- 47.Fu L, Rao F, Lian F, Yang H, Kuang S, Wu S, Deng C, Xue Y. Mechanism of electrical remodeling of atrial myocytes and its influence on susceptibility to atrial fibrillation in diabetic rats. Life Sci. 2019;239:116903. doi: 10.1016/j.lfs.2019.116903. [DOI] [PubMed] [Google Scholar]
- 48.Schotten U, Verheule S, Kirchhof P, Goette A. Pathophysiological mechanisms of atrial fibrillation: a translational appraisal. Physiol Rev. 2011;91(1):265–325. doi: 10.1152/physrev.00031.2009. [DOI] [PubMed] [Google Scholar]
- 49.Gemel J, Levy AE, Simon AR, Bennett KB, Ai X, Akhter S, Beyer EC. Connexin40 abnormalities and atrial fibrillation in the human heart. J Mol Cell Cardiol. 2014;76:159–168. doi: 10.1016/j.yjmcc.2014.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chen P-S, Chen LS, Fishbein MC, Lin S-F, Nattel S. Role of the autonomic nervous system in atrial fibrillation: pathophysiology and therapy. Circ Res. 2014;114(9):1500–1515. doi: 10.1161/CIRCRESAHA.114.303772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shen MJ, Zipes DP. Role of the autonomic nervous system in modulating cardiac arrhythmias. Circ Res. 2014;114(6):1004–1021. doi: 10.1161/CIRCRESAHA.113.302549. [DOI] [PubMed] [Google Scholar]
- 52.Urbancic-Rovan V, Meglic B, Stefanovska A, Bernjak A, Azman-Juvan K, Kocijancic A. Incipient cardiovascular autonomic imbalance revealed by wavelet analysis of heart rate variability in type 2 diabetic patients. Diabet Med J Br Diabet Assoc. 2007;24(1):18–26. doi: 10.1111/j.1464-5491.2007.02019.x. [DOI] [PubMed] [Google Scholar]
- 53.Rizzo MR, Sasso FC, Marfella R, Siniscalchi M, Paolisso P, Carbonara O, Capoluongo MC, Lascar N, Pace C, Sardu C, Passavanti B, Barbieri M, Mauro C, Paolisso G. Autonomic dysfunction is associated with brief episodes of atrial fibrillation in type 2 diabetes. J Diabetes Complicat. 2015;29(1):88–92. doi: 10.1016/j.jdiacomp.2014.09.002. [DOI] [PubMed] [Google Scholar]
- 54.Negishi K, Seicean S, Negishi T, Yingchoncharoen T, Aljaroudi W, Marwick TH. Relation of heart-rate recovery to new onset heart failure and atrial fibrillation in patients with diabetes mellitus and preserved ejection fraction. Am J Cardiol. 2013;111(5):748–753. doi: 10.1016/j.amjcard.2012.11.028. [DOI] [PubMed] [Google Scholar]
- 55.Saito S, Teshima Y, Fukui A, Kondo H, Nishio S, Nakagawa M, Saikawa T, Takahashi N. Glucose fluctuations increase the incidence of atrial fibrillation in diabetic rats. Cardiovasc Res. 2014;104(1):5–14. doi: 10.1093/cvr/cvu176. [DOI] [PubMed] [Google Scholar]
- 56.Middleton TL, Wong J, Molyneaux L, Brooks BA, Yue DK, Twigg SM, Wu T. Cardiac effects of sulfonylurea-related hypoglycemia. Diabetes Care. 2017;40(5):663–670. doi: 10.2337/dc16-1972. [DOI] [PubMed] [Google Scholar]
- 57.Frier BM. Hypoglycaemia in diabetes mellitus: epidemiology and clinical implications. Nat Rev Endocrinol. 2014;10(12):711–722. doi: 10.1038/nrendo.2014.170. [DOI] [PubMed] [Google Scholar]
- 58.Ko SH, Park YM, Yun JS, Cha SA, Choi EK, Han K, Han E, Lee YH, Ahn YB. Severe hypoglycemia is a risk factor for atrial fibrillation in type 2 diabetes mellitus: nationwide population-based cohort study. J Diabetes Complicat. 2018;32(2):157–163. doi: 10.1016/j.jdiacomp.2017.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.American Diabetes Association Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(Suppl 1):S98–s110. doi: 10.2337/dc20-S009. [DOI] [PubMed] [Google Scholar]
- 60.Buse JB, Wexler DJ, Tsapas A, Rossing P, Mingrone G, Mathieu C, D'Alessio DA, Davies MJ. 2019 update to: Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) Diabetologia. 2020;63(2):221–228. doi: 10.1007/s00125-019-05039-w. [DOI] [PubMed] [Google Scholar]
- 61.Foretz M, Guigas B, Bertrand L, Pollak M, Viollet B. Metformin: from mechanisms of action to therapies. Cell Metab. 2014;20(6):953–966. doi: 10.1016/j.cmet.2014.09.018. [DOI] [PubMed] [Google Scholar]
- 62.Chang SH, Wu LS, Chiou MJ, Liu JR, Yu KH, Kuo CF, Wen MS, Chen WJ, Yeh YH, See LC. Association of metformin with lower atrial fibrillation risk among patients with type 2 diabetes mellitus: a population-based dynamic cohort and in vitro studies. Cardiovasc Diabetol. 2014;13:123. doi: 10.1186/s12933-014-0123-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Basnet S, Kozikowski A, Sun H, Troup M, Urrutia LE, Pekmezaris R. Metformin therapy and postoperative atrial fibrillation in diabetic patients after cardiac surgery. J Intensive Care. 2017;5:60. doi: 10.1186/s40560-017-0254-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bai F, Liu Y, Tu T, Li B, Xiao Y, Ma Y, Qin F, Xie J, Zhou S, Liu Q. Metformin regulates lipid metabolism in a canine model of atrial fibrillation through AMPK/PPAR-alpha/VLCAD pathway. Lipids Health Dis. 2019;18(1):109. doi: 10.1186/s12944-019-1059-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rena G, Pearson ER, Sakamoto K. Molecular mechanism of action of metformin: old or new insights? Diabetologia. 2013;56(9):1898–1906. doi: 10.1007/s00125-013-2991-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Proks P, Reimann F, Green N, Gribble F, Ashcroft F. Sulfonylurea stimulation of insulin secretion. Diabetes. 2002;51(Suppl 3):S368–376. doi: 10.2337/diabetes.51.2007.s368. [DOI] [PubMed] [Google Scholar]
- 67.Liou YS, Yang FY, Chen HY, Jong GP. Antihyperglycemic drugs use and new-onset atrial fibrillation: a population-based nested case control study. PLoS ONE. 2018;13(8):e0197245. doi: 10.1371/journal.pone.0197245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zeller M, Danchin N, Simon D, Vahanian A, Lorgis L, Cottin Y, Berland J, Gueret P, Wyart P, Deturck R, Tabone X, Machecourt J, Leclercq F, Drouet E, Mulak G, Bataille V, Cambou JP, Ferrieres J, Simon T. Impact of type of preadmission sulfonylureas on mortality and cardiovascular outcomes in diabetic patients with acute myocardial infarction. J Clin Endocrinol Metab. 2010;95(11):4993–5002. doi: 10.1210/jc.2010-0449. [DOI] [PubMed] [Google Scholar]
- 69.Gu J, Liu X, Wang X, Shi H, Tan H, Zhou L, Gu J, Jiang W, Wang Y. Beneficial effect of pioglitazone on the outcome of catheter ablation in patients with paroxysmal atrial fibrillation and type 2 diabetes mellitus. Europace Eur Pacing Arrhythm Card Electrophysiol. 2011;13(9):1256–1261. doi: 10.1093/europace/eur131. [DOI] [PubMed] [Google Scholar]
- 70.Chao TF, Leu HB, Huang CC, Chen JW, Chan WL, Lin SJ, Chen SA. Thiazolidinediones can prevent new onset atrial fibrillation in patients with non-insulin dependent diabetes. Int J Cardiol. 2012;156(2):199–202. doi: 10.1016/j.ijcard.2011.08.081. [DOI] [PubMed] [Google Scholar]
- 71.Pallisgaard JL, Lindhardt TB, Staerk L, Olesen JB, Torp-Pedersen C, Hansen ML, Gislason GH. Thiazolidinediones are associated with a decreased risk of atrial fibrillation compared with other antidiabetic treatment: a nationwide cohort study. Eur Heart J Cardiovasc Pharmacother. 2017;3(3):140–146. doi: 10.1093/ehjcvp/pvw036. [DOI] [PubMed] [Google Scholar]
- 72.Zhang Z, Zhang X, Korantzopoulos P, Letsas KP, Tse G, Gong M, Meng L, Li G, Liu T. Thiazolidinedione use and atrial fibrillation in diabetic patients: a meta-analysis. BMC Cardiovasc Disord. 2017;17(1):96. doi: 10.1186/s12872-017-0531-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Liu T, Li G. Thiazolidinediones as novel upstream therapy for atrial fibrillation in diabetic patients: a review of current evidence. Int J Cardiol. 2012;156(2):215–216. doi: 10.1016/j.ijcard.2012.01.058. [DOI] [PubMed] [Google Scholar]
- 74.Kume O, Takahashi N, Wakisaka O, Nagano-Torigoe Y, Teshima Y, Nakagawa M, Yufu K, Hara M, Saikawa T, Yoshimatsu H. Pioglitazone attenuates inflammatory atrial fibrosis and vulnerability to atrial fibrillation induced by pressure overload in rats. Heart Rhythm. 2011;8(2):278–285. doi: 10.1016/j.hrthm.2010.10.029. [DOI] [PubMed] [Google Scholar]
- 75.Xu D, Murakoshi N, Igarashi M, Hirayama A, Ito Y, Seo Y, Tada H, Aonuma K. PPAR-gamma activator pioglitazone prevents age-related atrial fibrillation susceptibility by improving antioxidant capacity and reducing apoptosis in a rat model. J Cardiovasc Electrophysiol. 2012;23(2):209–217. doi: 10.1111/j.1540-8167.2011.02186.x. [DOI] [PubMed] [Google Scholar]
- 76.Gu J, Liu X, Wang QX, Guo M, Liu F, Song ZP, Zhang DD. Beneficial effects of pioglitazone on atrial structural and electrical remodeling in vitro cellular models. J Mol Cell Cardiol. 2013;65:1–8. doi: 10.1016/j.yjmcc.2013.09.016. [DOI] [PubMed] [Google Scholar]
- 77.Chang CY, Yeh YH, Chan YH, Liu JR, Chang SH, Lee HF, Wu LS, Yen KC, Kuo CT, See LC. Dipeptidyl peptidase-4 inhibitor decreases the risk of atrial fibrillation in patients with type 2 diabetes: a nationwide cohort study in Taiwan. Cardiovasc Diabetol. 2017;16(1):159. doi: 10.1186/s12933-017-0640-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wang A, Green JB, Halperin JL, Piccini JP., Sr Atrial fibrillation and diabetes mellitus: JACC review topic of the week. J Am Coll Cardiol. 2019;74(8):1107–1115. doi: 10.1016/j.jacc.2019.07.020. [DOI] [PubMed] [Google Scholar]
- 79.Bell DSH, Goncalves E. Atrial fibrillation and type 2 diabetes: prevalence, etiology, pathophysiology and effect of anti-diabetic therapies. Diabetes Obes Metab. 2019;21(2):210–217. doi: 10.1111/dom.13512. [DOI] [PubMed] [Google Scholar]
- 80.Lee TI, Kao YH, Chen YC, Huang JH, Hsu MI, Chen YJ. The dipeptidyl peptidase-4 inhibitor-sitagliptin modulates calcium dysregulation, inflammation, and PPARs in hypertensive cardiomyocytes. Int J Cardiol. 2013;168(6):5390–5395. doi: 10.1016/j.ijcard.2013.08.051. [DOI] [PubMed] [Google Scholar]
- 81.Zhang X, Zhang Z, Zhao Y, Jiang N, Qiu J, Yang Y, Li J, Liang X, Wang X, Tse G, Li G, Liu T. Alogliptin, a dipeptidyl peptidase-4 inhibitor, alleviates atrial remodeling and improves mitochondrial function and biogenesis in diabetic rabbits. J Am Heart Assoc. 2017 doi: 10.1161/jaha.117.005945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Igarashi T, Niwano S, Niwano H, Yoshizawa T, Nakamura H, Fukaya H, Fujiishi T, Ishizue N, Satoh A, Kishihara J, Murakami M, Ako J. Linagliptin prevents atrial electrical and structural remodeling in a canine model of atrial fibrillation. Heart Vessels. 2018;33(10):1258–1265. doi: 10.1007/s00380-018-1170-0. [DOI] [PubMed] [Google Scholar]
- 83.Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8(12):728–742. doi: 10.1038/nrendo.2012.140. [DOI] [PubMed] [Google Scholar]
- 84.Fisher M, Petrie MC, Ambery PD, Donaldson J, Ye J, McMurray JJ. Cardiovascular safety of albiglutide in the Harmony programme: a meta-analysis. Lancet Diabetes Endocrinol. 2015;3(9):697–703. doi: 10.1016/s2213-8587(15)00233-8. [DOI] [PubMed] [Google Scholar]
- 85.Holman RR, Bethel MA, Mentz RJ, Thompson VP, Lokhnygina Y, Buse JB, Chan JC, Choi J, Gustavson SM, Iqbal N, Maggioni AP, Marso SP, Öhman P, Pagidipati NJ, Poulter N, Ramachandran A, Zinman B, Hernandez AF. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2017;377(13):1228–1239. doi: 10.1056/NEJMoa1612917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Monami M, Nreu B, Scatena A, Giannini S, Andreozzi F, Sesti G, Mannucci E. Glucagon-like peptide-1 receptor agonists and atrial fibrillation: a systematic review and meta-analysis of randomised controlled trials. J Endocrinol Invest. 2017;40(11):1251–1258. doi: 10.1007/s40618-017-0698-7. [DOI] [PubMed] [Google Scholar]
- 87.Younce CW, Burmeister MA, Ayala JE. Exendin-4 attenuates high glucose-induced cardiomyocyte apoptosis via inhibition of endoplasmic reticulum stress and activation of SERCA2a. Am J Physiol Cell Physiol. 2013;304(6):C508–518. doi: 10.1152/ajpcell.00248.2012. [DOI] [PubMed] [Google Scholar]
- 88.Huang JH, Chen YC, Lee TI, Kao YH, Chazo TF, Chen SA, Chen YJ. Glucagon-like peptide-1 regulates calcium homeostasis and electrophysiological activities of HL-1 cardiomyocytes. Peptides. 2016;78:91–98. doi: 10.1016/j.peptides.2016.02.007. [DOI] [PubMed] [Google Scholar]
- 89.Ferrannini E. Sodium-glucose co-transporters and their inhibition: clinical physiology. Cell Metab. 2017;26(1):27–38. doi: 10.1016/j.cmet.2017.04.011. [DOI] [PubMed] [Google Scholar]
- 90.Birkeland KI, Jorgensen ME, Carstensen B, Persson F, Gulseth HL, Thuresson M, Fenici P, Nathanson D, Nystrom T, Eriksson JW, Bodegard J, Norhammar A. Cardiovascular mortality and morbidity in patients with type 2 diabetes following initiation of sodium-glucose co-transporter-2 inhibitors versus other glucose-lowering drugs (CVD-REAL Nordic): a multinational observational analysis. Lancet Diabetes Endocrinol. 2017;5(9):709–717. doi: 10.1016/s2213-8587(17)30258-9. [DOI] [PubMed] [Google Scholar]
- 91.Usman MS, Siddiqi TJ, Memon MM, Khan MS, Rawasia WF, Talha Ayub M, Sreenivasan J, Golzar Y. Sodium-glucose co-transporter 2 inhibitors and cardiovascular outcomes: a systematic review and meta-analysis. Eur J Prev Cardiol. 2018;25(5):495–502. doi: 10.1177/2047487318755531. [DOI] [PubMed] [Google Scholar]
- 92.Zelniker TA, Bonaca MP, Furtado R, Mosenzon O, Kuder JF, Murphy SA, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Budaj A, Kiss RG, Padilla F, Gause-Nilsson I, Langkilde AM, Raz I, Sabatine MS, Wiviott SD. Effect of dapagliflozin on atrial fibrillation in patients with type 2 diabetes mellitus: insights from the DECLARE-TIMI 58 Trial. Circulation. 2020 doi: 10.1161/circulationaha.119.044183. [DOI] [PubMed] [Google Scholar]
- 93.Shao Q, Meng L, Lee S, Tse G, Gong M, Zhang Z, Zhao J, Zhao Y, Li G, Liu T. Empagliflozin, a sodium glucose co-transporter-2 inhibitor, alleviates atrial remodeling and improves mitochondrial function in high-fat diet/streptozotocin-induced diabetic rats. Cardiovasc Diabetol. 2019;18(1):165. doi: 10.1186/s12933-019-0964-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Lee TI, Chen YC, Lin YK, Chung CC, Lu YY, Kao YH, Chen YJ. Empagliflozin attenuates myocardial sodium and calcium dysregulation and reverses cardiac remodeling in streptozotocin-induced diabetic rats. Int J Mol Sci. 2019;20(7):1680. doi: 10.3390/ijms20071680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Santos-Gallego CG, Requena-Ibanez JA, San Antonio R, Ishikawa K, Watanabe S, Picatoste B, Flores E, Garcia-Ropero A, Sanz J, Hajjar RJ, Fuster V, Badimon JJ. Empagliflozin ameliorates adverse left ventricular remodeling in nondiabetic heart failure by enhancing myocardial energetics. J Am Coll Cardiol. 2019;73(15):1931–1944. doi: 10.1016/j.jacc.2019.01.056. [DOI] [PubMed] [Google Scholar]
- 96.Patti G, Lucerna M, Cavallari I, Ricottini E, Renda G, Pecen L, Romeo F, Le Heuzey JY, Zamorano JL, Kirchhof P, De Caterina R. Insulin-requiring versus noninsulin-requiring diabetes and thromboembolic risk in patients with atrial fibrillation: PREFER in AF. J Am Coll Cardiol. 2017;69(4):409–419. doi: 10.1016/j.jacc.2016.10.069. [DOI] [PubMed] [Google Scholar]
- 97.Gerstein HC, Bosch J, Dagenais GR, Diaz R, Jung H, Maggioni AP, Pogue J, Probstfield J, Ramachandran A, Riddle MC, Ryden LE, Yusuf S. Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med. 2012;367(4):319–328. doi: 10.1056/NEJMoa1203858. [DOI] [PubMed] [Google Scholar]
- 98.Madonna R, De Caterina R. Cellular and molecular mechanisms of vascular injury in diabetes—part I: pathways of vascular disease in diabetes. Vascul Pharmacol. 2011;54(3–6):68–74. doi: 10.1016/j.vph.2011.03.005. [DOI] [PubMed] [Google Scholar]
- 99.Johnson LSB, Juhlin T, Engström G, Nilsson PM. Low fasting plasma insulin is associated atrial fibrillation in men from a cohort study—the Malmö preventive project. BMC Cardiovasc Disord. 2014;14:107–107. doi: 10.1186/1471-2261-14-107. [DOI] [PMC free article] [PubMed] [Google Scholar]