Abstract
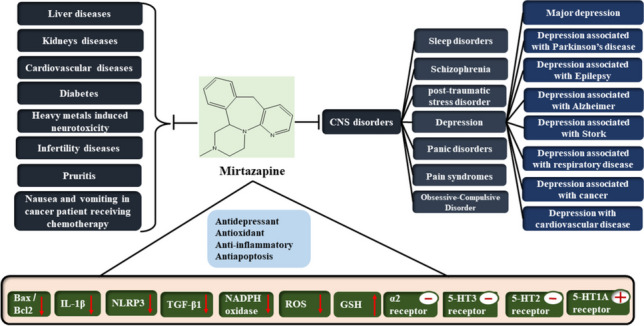
Mirtazapine (MTZ) is an antidepressant drug with an exceptional pharmacological profile. It also has an excellent safety and tolerability profile. The present review provides a pharmacological update on MTZ and summarizes the research findings of MTZ’s effects on different diseases. MTZ is hypothesized to have antidepressant effects because of the synergy between noradrenergic and serotonergic actions and is effective in treating major depressive disorder and depression associated with epilepsy, Alzheimer’s disease, stroke, cardiovascular disease, and respiratory disease. In cancer patients, MTZ significantly reduced sadness, nausea, sleep disruption, and pain and improved quality of life. Also, it has promising effects on Parkinson’s disease, schizophrenia, dysthymia, social anxiety disorder, alcohol dependency, posttraumatic stress disorder, panic disorder, pain syndromes, obsessive–compulsive disorder, and sleep disorders. Additionally, MTZ is potentially therapeutic in different situations associated with depression, such as liver, kidney, cardiovascular, respiratory, infertility, heavy metal-induced neurotoxicity, and pruritus. Potent antioxidative, anti-inflammatory, and anti-apoptotic bioactivities mediate these promising effects. These positive outcomes of the scientific investigations motivate more and more clinical trials for a golden exceptional antidepressant in different conditions.
Graphical Abstract
Keywords: Mirtazapine, Antidepressant, CNS disorders
Introduction
Mirtazapine
Chemistry
Mirtazapine (MTZ) is 5-methyl-2,5,19-triazatetracyclo [13.4.0.02,7.08,13] nonadeca-1(15),8,10,12,16,18-hexaene (Fig. 1), a noradrenergic and specific serotonergic antidepressant [1]. The empirical formula of MTZ is C17H19N3, and its molecular mass is 265.36 (Benjamin and Doraiswamy 2011). The α-2 antagonistic properties of the R(-)- and S( +)-enantiomers make them both pharmacologically active and may be a factor in their antidepressant effects, which are combined in the pill as a racemic combination (Delbressine et al. 1998). It was examined for neuropharmacological action under the name ORG-3770 by De Boer et al. (1988).
Fig. 1.
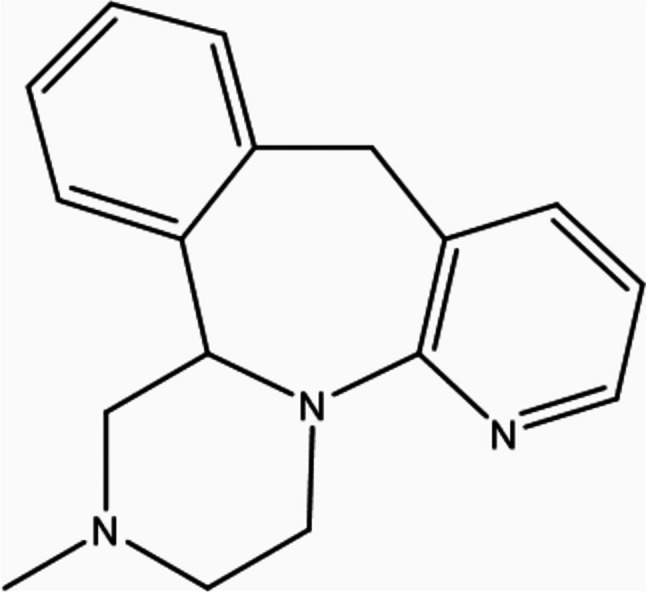
Chemical structure of MTZ
Pharmacokinetics
Absorption
MTZ is effectively absorbed from the gastrointestinal tract (Voortman and Paanakker 1995). The pharmacokinetics of MTZ are age- and sex-dependent. Adults and the elderly attain a steady state after 4 days and 6 days, respectively, after a once-daily oral dose (Voortman and Paanakker 1995; Timmer et al. 1996).
Distribution
Over a concentration range of 0.01 to 10 mcg/ml, 85% of MTZ is bound to plasma proteins. It was reported that MTZ binds to human plasma proteins in vitro at concentrations between 0.01 and 10 mg/L with an approximate 85% affinity (Van der Vorstenbosch and Delbressine 1988). Over the examined concentration range, binding was nonspecific and reversible. MTZ binds to human erythrocytes in vitro with a 40% affinity, comparable to the hematocrit of whole blood (Timmer et al. 2000).
Elimination
The liver is responsible for the majority of the biotransformation of MTZ. The main mechanisms include glucuronide conjugation, demethylation, and hydroxylation (Sandker et al. 1994). MTZ is considerably eliminated in the urine and feces after being digested. More than 75% of the prescribed dose is eliminated in the urine, leaving the remaining 20% in the feces. The amount of the dose that was excreted unchanged was less than 4% (Delbressine et al. 1998). The cytochrome P450 enzyme system is primarily involved in the oxidative pathways where MTZ is biotransformed (Dahl et al. 1997).
Pharmacodynamic of MTZ
Antidepressant
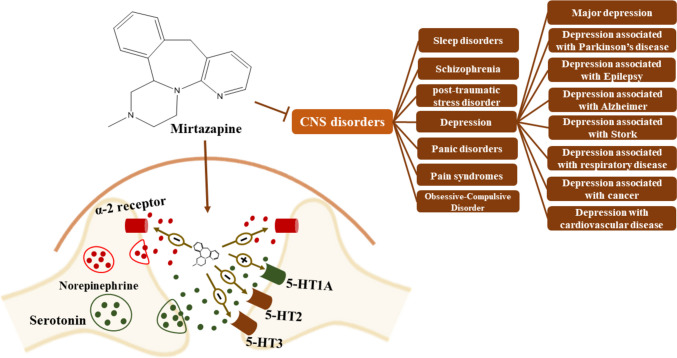
MTZ is hypothesized to have antidepressant effects because of the synergy between noradrenergic and serotonergic actions. As a racemate, MTZ has pharmacological activity in both the S-( +)- and R-( −)-enantiomers [5]. By antagonism of central α2-adrenergic heteroreceptors and autoreceptors as well as postsynaptic inhibition of 5-HT2 and 5-HT3 receptors, MTZ increases noradrenergic and 5-HT1A-mediated serotonergic neurotransmission (Fig. 2) (De Boer et al. 1995). When compared to various other types of antidepressants, MTZ differs in that it does not impede the reuptake of serotonin or noradrenaline or monoamine oxidase activity (Pinder 1997b). Theoretically, MTZ’s blocking of the 5-HT2 and 5-HT3 receptors could lessen the risk of some serotonergic side effects linked to the nonselective activation of serotonin receptors by SSRIs (Pinder 1997a, 1997b).
Fig. 2.
Effectiveness of MTZ in treating a major depressive disorder and depression associated with several conditions
MTZ has numerous additional pharmacodynamic effects in addition to its antidepressant action. Blocking 5-HT2 receptors, for instance, has been linked to better sleep (Pinder 1997b). Moreover, MTZ enhances the quality and duration of sleep and, in contrast to SSRIs, does not inhibit rapid eye movement sleep (Aslan et al. 2002; Winokur et al. 2003; Shen et al. 2006).
It is interesting to note that reuptake inhibitors can raise cortisol levels (Schule et al. 2003), and cortisol secretion is reduced by MTZ (Laakmann et al. 1999, 2004; Schüle et al. 2002; Schule et al. 2003). MTZ increases serotonergic transmission in a way that suggests that the medication may start working more quickly than selective serotonin reuptake inhibitors (SSRIs). Animal model studies, unlike SSRIs, have demonstrated that MTZ treatment increases the rate at which serotonin neurons fire (Holm and Markham 1999).
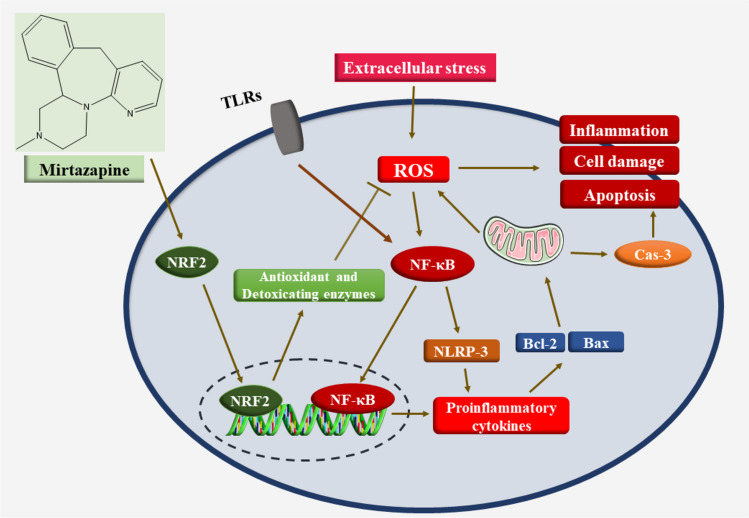
Antioxidant effect
The imbalance between the generation of free radicals and their neutralization by antioxidants is known as oxidative stress (OS). Increased ROS production and weaker antioxidant defenses lead to this redox imbalance. High levels of ROS can result in OS and cell death by causing inflammation and damage to macromolecules. As a result, OS is connected to the etiology of a variety of diseases (Rani et al. 2016; Mahmoud et al. 2017; Liguori et al. 2018). Numerous in vitro and in vivo investigations have clearly demonstrated the antioxidant and anti-inflammatory properties of MTZ (Elsisi et al. 2021; Hafez et al. 2021b). MTZ’s antioxidant function had a beneficial effect on many organs. MTZ increased the nonenzymatic antioxidant glutathione (GSH) and decreased lipid peroxidation in reproductive toxicity (El-Sisi et al. 2017). Additionally, other models of the antioxidant effect also accounted for renal ischaemia‒reperfusion injury (Tok et al. 2012) and peptic ulcers (Bilici et al. 2009). Tok et al. investigated MTZ’s impact on the OS induced by ischaemia‒reperfusion in rat kidneys. According to the findings, MTZ decreased renal MDA content and MPO enzymatic activity and GSH and glutathione-S-transferase (GST) activities. Moreover, the histopathological findings were attenuated by MTZ. Therefore, MTZ may protect against IR-induced kidney injury (Tok et al. 2012). Another study investigated the possible chemoprotective properties of MTZ against OS and DNA damage induced by cisplatin. Because of its antioxidant properties, MTZ exerts chemoprotective effects on DNA damage and OS induced by cisplatin in the rat brain, as proven by decreased MDA, MPO, and 8-OH-GUA levels and increased GSH levels (Gulec et al. 2013). Additionally, in BV2 microglia exposed to isoflurane, MTZ decreased the expression of the protein Iba1 and decreased the production of the proinflammatory mediators interleukin (IL)-1β and IL-18, which isoflurane produces by blocking the activation of the nod-like receptor family protein 3 (NLRP3) inflammasome in BV2 microglia. MTZ downregulates NADPH oxidase 4 (NOX4) expression and the production of reactive ROS. These findings led us to conclude that MTZ may be a possible intervention to stop cognitive damage induced by isoflurane exposure in clinical settings (Wang et al. 2022).
Anti-inflammatory effect
Previous research has suggested that MTZ treatment for major depressive disorders has anti-inflammatory effects and decreases tumor necrosis factor (TNF)-α levels (Gupta et al. 2016). Similarly, in a model of neuropathic pain, MTZ reduced TNF-α and nuclear factor-kappa B (NF-κB) (Zhu et al. 2008), as well as in a model of immune-mediated liver damage (Almishri et al. 2019). Inhibiting the NLRP3/caspase-1/IL-18 signal has demonstrated mechanistically anti-inflammatory effects (Hafez et al. 2021a) (Fig. 3).
Fig. 3.
Molecular mechanisms underlying the antioxidant, anti-inflammatory, and anti-apoptotic activities of MTZ
Anti-apoptotic effect
Mitochondrial membranes may be damaged by increased ROS generation, which will cause the release of cytochrome c (Maneesh and Jayalekshmi 2006). This, in turn, interacts with apoptosis protease activation factor-1 to activate caspase-9 as the initiator, which then activates caspase-3 to cause apoptosis (Sinha Hikim et al. 2003). El-Sisi et al. reported that MTZ can be used as a preventative medicine to stop the reduction in sperm count and viability caused by nitrofurazone. Additionally, both medications reduced the effects of nitrofurazone on rat testes MDA, GSH depletion, elevation of TNF-α, and decrease in c-kit levels. In terms of apoptosis, immunohistochemistry research showed that nitrofurazone-induced testicular tissue expressed caspase-3, which was decreased by chrysin and MTZ. Histopathological data further substantiated the preventive benefits of both medications. The findings revealed that OS and apoptotic tissue damage were suppressed by the cytoprotective effects of chrysin and MTZ on rat testes (El-Sisi et al. 2017). Consequently, chrysin and MTZ on rat testes are mediated by mitigating testicular oxidative injury and apoptosis. Additionally, Engel et al. showed that by lowering the expression of antiapoptotic proteins, MTZ decreased neuronal death in the hippocampus and cerebral cortex of mice by downregulating Bcl-xL, Bax, Bad, and p53 (Engel et al. 2013). Lieberknecht et al. reported that MTZ protects the brain against H2O2-induced cell death. In particular, MTZ decreased p53 mRNA expression and cell death (Lieberknecht et al. 2020).
Pharmacological update of MTZ
In our review, we used the search engines of PubMed as well as databases of Scopus, Web of Science, and Google Scholar. The pharmacological update is summarized in Table 1 and discussed as follows:
Table 1.
Summary of the pharmacological effects of MTZ
| System or organ | Disease | Main effects | Reference |
|---|---|---|---|
| CNS | Major depression | Randomized, controlled trials that compared it to placebo and other antidepressants, such as fluoxetine, paroxetine citalopram, trazodone, amitriptyline, doxepin, clomipramine, imipramine, and venlafaxine | Richou et al. (1995), Bruijn et al. (1996), Wheatley et al. (1998), Leinonen et al. (1999), Guelfi et al. (2001), Benkert et al. (2002) |
| Depression associated with Parkinson’s disease | MTZ affected PDPsy and effectively reduced hallucinations in PD | Sid-Otmane et al. (2020) | |
| MTZ effectively treated depression in PD patients as well as insomnia and psychosis | Pena et al. (2018) | ||
| Depression associated with epilepsy | MTZ is generally safe and useful for patients with epilepsy | Górska et al. (2018) | |
| Depression associated with Alzheimer’s disease |
MTZ produced a quick and long-lasting response in three individuals with AD and depression worsened by anxiety, sleeplessness, and weight loss The high frequency of these bothersome symptoms in depressed AD patients highlights the need for an antidepressant that combines beneficial effects on these symptoms and a good safety profile MTZ may play a role in treating depression that coexists with dementia, anxiety, sleeplessness, and weight loss, based on the clinical reactions of the three patients |
Lyketsos and Olin (2002), Caraci et al. (2010) | |
| MTZ exhibited promising effects for AD patients who also have sadness, sleeplessness, anxiety, and weight loss | Raji and Brady (2001) | ||
| Depression associated with stroke |
In patients with an acute stroke, MTZ dramatically decreased the incidence of poststroke depression MTZ successfully treated poststroke depression |
Niedermaier et al. (2004) | |
| Depression associated with respiratory diseases | MTZ has effects that are possibly helpful for breathlessness and elevates the mood, mostly by altering the processing and perception of afferent information in the brain MTZ’s ability to inhibit 5-HT2 and 5-HT3 receptors prevents it from sharing some of the negative effects of other regularly prescribed antidepressants, and inhibiting H1 receptors helps improve appetite and sleep for patients | Yang et al. (2020) | |
| Depression associated with cancer | In nausea and sleep disturbance, which are frequent and upsetting side effects of cancer, MTZ significantly reduced sadness, nausea, sleep disruption, pain, and quality of life in cancer patients | Kim et al. (2008) | |
| In a pilot open-label, in advanced cancer patients with pain and other disturbing symptoms, MTZ may be useful for treating numerous symptoms, depression, and quality of life in advanced cancer patients | Theobald et al. (2002) | ||
| MTZ demonstrated benefits for early, medium, and late insomnia. Research shows that MTZ can help cancer patients with insomnia, anxiety, and depressive symptoms | Cankurtaran et al. (2008) | ||
| Depression associated with cardiovascular disease | Within the MIND-IT study, a randomized controlled trial, MTZ is an effective and safe for MI who also had depression | Honig et al. (2007) | |
| A short retrospective cohort study concluded the effectiveness of MTZ for hospitalized individuals who were given MTZ following a mental consultation | Allen et al. (2020) | ||
| Sleep disorders | In patients with major depressive illness who had poor sleep quality at weeks 1 and 2 of treatment, MTZ dramatically improved sleep continuity | Winokur et al. (2000) | |
|
MTZ has a significant impact on slow-wave sleep When compared to a placebo, MTZ improved the factors connected to uninterrupted sleep. It reduced the quantity and length of awakenings while raising the sleep efficiency index Regarding characteristics related to rapid eye movement sleep, there was no discernible impact |
Aslan et al. (2002) | ||
| In alcohol abuse and social anxiety disorder, a total of 41.7% of them were categorized as respondents at the study’s conclusion. People with alcohol addiction frequently exhibit symptoms of social anxiety. On the other hand, MTZ reduced the social anxiety symptoms | Liappas et al. (2003) | ||
|
For 4–5 weeks after detoxification, a total of 54 inpatients with alcoholism were randomly assigned to undergo MTZ with psychotherapy or cognitive-behavioral treatment. The combination group significantly outperformed the control group in terms of statistically reducing social anxiety symptoms In a subsequent trial, MTZ was discovered to be beneficial during the detoxification of patients who were addicted to alcohol as opposed to venlafaxine |
Liappas et al. (2005) | ||
| Schizophrenia | Adding MTZ to haloperidol led to a statistically significant reduction in negative symptoms after 6 weeks of treatment in a double-blind, randomized, placebo-controlled study including 30 patients | Berk et al. (2001) | |
| Combination of MTZ with clozapine in an 8-week, double-blind, placebo-controlled experiment for the treatment of negative symptoms of schizophrenia. The results revealed that the treatment considerably reduced SANS scores compared to the placebo, especially on the avolition/apathy and anhedonia/asociality SANS subscales | Zoccali et al. (2004) | ||
| MTZ treated akathisia caused by neuroleptics in twenty-six schizophrenia patients were randomized to receive either MTZ 15 mg or a placebo for 5 days in a double-blind study. The MTZ group displayed noticeably higher response rates | Poyurovsky et al. (2003) | ||
| Posttraumatic stress disorder | Twenty-nine PTSD patients participated in this trial and were given MTZ or a placebo for 8 weeks. After 8 weeks of therapy, patients receiving MTZ had a response rate that was noticeably higher than those receiving a placebo, as determined. After 8 weeks of MTZ therapy, three open-label studies also discovered significant improvement rates | Connor et al. (1999), Bahk et al. (2002), Chung et al. (2004) | |
| Panic disorder | MTZ demonstrated encouraging effects in lowering depression and anxiety symptoms in panic disorders according to numerous open-label studies | Boshuisen et al. (2001), Carli et al. (2002), Sarchiapone et al. (2003) | |
| Pain syndromes | In several clinical studies, MTZ may be helpful for pain syndromes | Brannon and Stone (1999), Nutt and Law (1999), Brannon et al. (2000), Bendtsen and Jensen (2004) | |
|
Twenty-four patients with persistent tension-type headaches In a randomized, double-blind, placebo-controlled crossover trial. After 8 weeks of treatment, MTZ significantly outperformed a placebo in the preventive treatment of persistent tension-type headaches |
Bendtsen and Jensen (2004) | ||
| Obsessive–compulsive disorder | The effectiveness of MTZ in patients with OCD has been examined in two randomized, controlled trials. MTZ’s effects on OCD have been documented to be effective in OCD as a pharmacotherapy | Koran et al. (2005) | |
|
In a pilot trial, the effects of citalopram and MTZ in patients with OCD who did not also have comorbid depression When MTZ was combined with citalopram, they discovered a faster onset of responsive action in OCD symptoms and decreased undesirable side effects |
Pallanti et al. (2004) | ||
| The potential protective effects of MTZ on diseases other than CNS-related disorders | |||
| Liver diseases | High-fat diet mice model |
Obese mice treated with MTZ had reduced levels of body weight, serum TG and AST levels The treated mice had lower blood glucose levels and increased insulin sensitivity and glucose transporter 4 expression levels |
Wu et al. (2020) |
| Immune-mediated liver disease |
MTZ rapidly shifts the hepatic B-cell populations and functional cytokine signatures of mice Treatment with MTZ increases the retention of innate-like B cells that express CXCR3, producing more anti-inflammatory cytokines |
Almishri et al. (2021) | |
| IV infection with a pathogenic strain of S. aureus in mice |
MTZ affects liver innate immunity and inhibits immune-driven activation of hepatic macrophages Treatment with MTZ may have beneficial effects on sepsis, and it significantly decreases the risk of hepatic abscess formation |
Davis et al. (2020) | |
| Liver fibrosis induced by thioacetamide in mice |
MTZ treatment ameliorated TAA-induced liver fibrosis by lowering portal BP, liver procollagen I content, and α-SMA expression MTZ-treated animals had significantly less hepatic collagen accumulation. MTZ decreased the levels of protein kinase C, TGF-β1, phosphorylated Smad3, p-ERK1/2 MTZ significantly reduced oxidative stress, by declining hepatic lipid peroxidation and NADPH oxidase 1 and increased GSH content |
El-Tanbouly et al. (2017) | |
| Kidney diseases | Diabetic nephropathy in rats |
MTZ downregulated NLRP3 and caspase-1 expression as well as the renal level of IL-1β in diabetic rats MTZ may be effective in the management of DM and other metabolic illnesses |
Sahin et al. (2019) |
| Cardiovascular diseases related to CNS disorders | An in vitro human plasma-based study | The antiplatelet effects MTZ are mediated by 5-HT2A and α2-adrenergic receptors blocking | Kawano et al. (2022) |
| Diabetes | Type 1 diabetic rats | In T1DM rat models, MTZ reduced GLUT2 by changing the expression of leptin and galanin in the livers of type 1 diabetic rats with hyperglycaemia. These findings concluded that patients with T1DM can use MTZ as an antidepressant and as a medication to lower blood glucose levels | Bektur et al. (2019a) |
| Diabetes-induced hyperalgesia in rats | MTZ exhibits beneficial effects in diabetes-induced hyperalgesia is mediated by decreasing TRPV1 and ASIC1 levels | Bektur et al. (2019b) | |
| GIT | Nausea and vomiting in chemotherapy-treated cancer patients | In a study of 35 patients, when given in conjunction with platinum-based regimens to patients with thoracic cancer, adding MTZ to the recommended antiemetic regimen for CINV may be advantageous with appropriate tolerability | Kinomura et al. (2023) |
| Heavy metal-induced neurotoxicity | Cd-induced neurotoxicity in rats |
MTZ attenuates Cd-induced neurotoxicity by upregulating the transcription factor Nrf2, suppressing NF-κB/TLR4 signalling, MTZ effectively decreasing TNF-α, IL-1β, and IL-6 MTZ reduced histological abrasions in the dentate gyrus, CA1 and CA3 regions of the hippocampus, and cerebral cortex of the rat brain |
Alharthy et al. (2023) |
| Reproductive diseases | CP-induced oxidative stress in rat ovaries | CP-induced oxidative stress in rat ovaries causes infertility in female rats. MTZ could counteract this impact and safeguard fertility against CP-induced damage | Khedr (2015) |
| Cisplatin-induced infertility in rats | MTZ decreased the levels of MDA, MPO, NO, and 8-hydroxy-2 deoxyguanine and effectively increased GSH, GPx, and SOD levels | Altuner et al. (2013) | |
| Nitrofurazone-induced testicular injury in rats |
MTZ stopped the reduction in sperm count and viability caused by nitrofurazone MTZ reduced the effects of nitrofurazone on rat testes MDA, GSH depletion, elevation of TNF-α, and decrease in c-kit levels MTZ attenuated apoptosis by downregulating testicular caspase-3 |
El-Sisi et al. (2017) | |
| Four case reports of hot flushes and sweating episodes in women | MTZ exhibited promising effects on the frequency and intensity of hot flushes and sweating episodes in women, MTZ appears to significantly lessen both of these symptoms | Waldinger et al. (2000) | |
| Pruritus | Patients with chronic pruritus: a pilot study | MTZ has shown success in lowering itch severity is MTZ, a dual noradrenergic and serotonergic antidepressant with antihistaminic characteristics | Hundley and Yosipovitch (2004), Lee et al. (2016) |
CNS disorders
Major depression
MTZ is effective in treating a major depressive disorder and in depression associated with several conditions (Fig. 2) in several randomized, controlled trials that compared it to placebo and other antidepressants, such as fluoxetine, paroxetine citalopram, trazodone, amitriptyline, doxepin, clomipramine, imipramine, and venlafaxine (Richou et al. 1995, Bruijn et al. 1996, Wheatley et al. 1998, Leinonen et al. 1999, Guelfi et al. 2001, Benkert et al. 2002).
Depression associated with Parkinson’s disease
Parkinson’s disease (PD) patients frequently experience psychotic symptoms, and a set of criteria for PD with psychosis (PDPsy) has been defined. Hallucinations are one of the specific symptoms that fit this description, with visual hallucinations being the most frequent. According to research by Tagai et al., the use of MTZ in a patient with PDPsy reduced the patient’s refractory psychotic symptoms, particularly visual hallucinations, without worsening motor symptoms (Tagai et al. 2013). The effects of MTZ and other antidepressants on mild depression linked to PD were also documented by Costa et al. (2012). Sid-Otmane et al. investigated how MTZ and other antidepressants affected PDPsy and found that they effectively reduced hallucinations in PD (Sid-Otmane et al. 2020). Antidepressant medications, such as MTZ, are effective in treating depression in PD patients as well as insomnia and psychosis, according to Pena et al. (2018).
Depression associated with epilepsy
Mood issues are frequently co-occurring in people with epilepsy. The lifetime prevalence ranges from 11 to 62% (Stanisław Wiglusz et al. 2012). Depression is one of the main psychiatric conditions that has a detrimental effect on the quality of life of people with epilepsy (Mula 2017). Epilepsy and depression may be related in a bidirectional manner; having epilepsy would seem to increase the risk of depression while having depression would seem to increase the chance of epilepsy (Forsgren and Nyström, 1990). Epilepsy patients who are depressed attempt suicide four to five times more frequently than people without epilepsy (Matthews and Barabas 1981; Batzel and Dodrill 1986; Blumer 2002). According to clinical studies by Natalia et al., antidepressants, including MTZ, are generally safe for patients with epilepsy when used at therapeutic dosages (Górska et al. 2018).
Depression associated with Alzheimer’s disease
Up to 10% of Americans over the age of 65 in the USA have Alzheimer’s disease (AD), which is the most frequent cause of dementia. The main factor in the early hospitalization of these patients is frequent psychiatric and behavioral difficulties. A total of 10–30% of AD patients meet the diagnostic requirements for severe depression (Mann et al. 1989). MTZ produced a quick and long-lasting response in three individuals with AD and depression worsened by anxiety, sleeplessness, and weight loss. The high frequency of these bothersome symptoms in depressed AD patients highlights the need for an antidepressant that combines beneficial effects on these symptoms and a good safety profile. MTZ may play a role in treating depression that coexists with dementia, anxiety, sleeplessness, and weight loss, based on the clinical reactions of the three patients (Lyketsos and Olin 2002; Caraci et al. 2010). Because brain serotonergic and noradrenergic neurotransmission regulate mood, sleep, and hunger, MTZ may be beneficial in treating depressed AD patients. The selection of an antidepressant should be based on a patient’s unique medical and psychological profile due to the absence of comparative data. MTZ exhibited promising effects for AD patients who also have sadness, sleeplessness, anxiety, and weight loss (Raji and Brady 2001).
Depression associated with stroke
The most prevalent and demanding neuropsychiatric poststroke consequences are those involving depression, which can occur after a stroke (Shi et al. 2017; Taylor-Rowan et al. 2019). One of the most frequent aftereffects of a stroke is poststroke depression, which affects 20 to 40% of all patients. In patients with an acute stroke, MTZ dramatically decreased the incidence of poststroke depression. Additionally, the study showed that this antidepressant successfully treated poststroke depression. Only 5.7% (2/35) of the patients receiving MTZ treatment and 40% (14/35) of the untreated patients experienced poststroke depression. Sixteen patients in total had poststroke depression; however, 15 of them recovered after beginning MTZ therapy (Niedermaier et al. 2004).
Depression associated with respiratory diseases
Most chronic obstructive pulmonary and interstitial lung disease patients experience breathlessness as a frequent and bothersome symptom of advanced disease (Solano et al. 2006; Moens et al. 2014). MTZ has effects that are possibly helpful for breathlessness and elevates the mood, mostly by altering the processing and perception of afferent information in the brain MTZ’s ability to inhibit 5-HT2 and 5-HT3 receptors prevents it from sharing some of the negative effects of other regularly prescribed antidepressants, and inhibiting H1 receptors helps improve appetite and sleep for patients (Yang et al. 2020).
Depression associated with cancer
Kim et al. assessed the effectiveness of oral disintegrating tablets of MTZ for nausea and sleep disturbance, which are frequent and upsetting side effects of cancer. MTZ significantly reduced sadness, nausea, sleep disruption, pain, and quality of life in cancer patients (Kim et al. 2008). MTZ was tested in a pilot open-label, crossover study by Theobald et al. in advanced cancer patients with pain and other disturbing symptoms. They examined how MTZ therapy affected patients’ levels of depressive symptoms, the severity of their pain, appetites, sleep patterns, weight, and general quality of life. According to this open-label pilot trial, MTZ may be useful for treating numerous symptoms, depression, and quality of life in advanced cancer patients (Theobald et al. 2002). Cankurtaran et al. investigated the efficacy of MTZ and imipramine on depression and anxiety symptoms as well as pain, nausea, vomiting, appetite loss, and sleep disruptions. Pain, nausea, vomiting, and appetite loss did not differ significantly between the three visits in the MTZ, imipramine, or control groups. Only the MTZ group demonstrated benefits for early, medium, and late insomnia. Research shows that MTZ can help cancer patients with insomnia, anxiety, and depressive symptoms (Cankurtaran et al. 2008).
Depression associated with cardiovascular disease
Studies examining the cardiovascular effects of MTZ in various at-risk populations are available. Within the MIND-IT study, a nested randomized controlled trial examined the effectiveness and safety of MTZ administered for 24 weeks to 331 adults hospitalized with an MI who also had depression. In comparison to the placebo, MTZ did not affect blood pressure, heart rate, QT, or QRS interval (Honig et al. 2007). These results imply that, at least temporarily, MTZ may be safe for individuals who have recently experienced a cardiac episode with a history of CVD. A short retrospective cohort study examined 61 medically hospitalized individuals who were given MTZ following a mental consultation (Allen et al. 2020).
CNS disorders other than depression
Sleep disorders
The majority of people with severe depressive illness subjectively state interrupted sleep or prolonged sleep onset (Winokur and Reynolds 1994). In patients with major depressive illness who had poor sleep quality at weeks 1 and 2 of treatment, Winokur et al. found that MTZ dramatically improved sleep continuity. During the first week, MTZ considerably reduced sleep latency and significantly enhanced total sleep time and sleep efficiency. Similar results were seen following the second week. MTZ did not significantly alter rapid eye movement sleep characteristics (Winokur et al. 2000). Aslan et al. examined how healthy volunteers’ sleep was affected by a single dose of MTZ. The results indicate that the MTZ has a significant impact on slow-wave sleep. It is advised that more research be done on the effectiveness of antidepressants concerning how 5-HT2 blockage affects slow-wave sleep. When compared to a placebo, MTZ improved the factors connected to uninterrupted sleep. It reduced the quantity and length of awakenings while raising the sleep efficiency index. Regarding characteristics related to rapid eye movement sleep, there was no discernible impact (Aslan et al. 2002).
In alcohol abuse and social anxiety disorder, a total of 41.7% of them were categorized as respondents at the study’s conclusion. People with alcohol addiction frequently exhibit symptoms of social anxiety. On the other hand, MTZ reduced the social anxiety symptoms (Liappas et al. 2003). For 4–5 weeks after detoxification, a total of 54 inpatients with alcoholism were randomly assigned to undergo MTZ with psychotherapy or cognitive-behavioral treatment. The combination group significantly outperformed the control group in terms of statistically reducing social anxiety symptoms. In a subsequent trial, MTZ was discovered to be beneficial during the detoxification of patients who were addicted to alcohol as opposed to venlafaxine (Liappas et al. 2005).
Schizophrenia
Schizophrenia is a severe mental condition that can be fatal for both the sufferers and those who care for them. Anhedonia, avolition, social disengagement, poverty of thinking, and cognitive dysfunction are some of the negative symptoms of schizophrenia. Delusions, hallucinations, and thought disorders are forms of positive symptoms (Saha et al. 2005; Salavati et al. 2015; Yang and Tsai 2017). MTZ was evaluated in two randomized, double-blind, placebo-controlled trials for the management of the negative symptoms of schizophrenia (Berk et al. 2001; Zoccali et al. 2004). In both studies, MTZ was added to an antipsychotic as a supplement. Berk et al. observed that adding MTZ to haloperidol led to a statistically significant reduction in negative symptoms after 6 weeks of treatment in a double-blind, randomized, placebo-controlled study including 30 patients (Berk et al. 2001). For the treatment of negative symptoms of schizophrenia, MTZ was combined with clozapine in an 8-week, double-blind, placebo-controlled experiment. The results revealed that the treatment considerably reduced SANS scores compared to the placebo, especially on the avolition/apathy and anhedonia/asociality SANS subscales (Zoccali et al. 2004). In another study, the efficacy of MTZ in treating akathisia caused by neuroleptics was investigated. Twenty-six schizophrenia patients were randomized to receive either MTZ 15 mg or a placebo for 5 days in a double-blind study. The MTZ group displayed noticeably higher response rates (Poyurovsky et al. 2003).
Posttraumatic stress disorder
The effectiveness of MTZ in treating posttraumatic stress disorder (PTSD) is being studied in several trials, one of which is a randomized, double-blind, placebo-controlled experiment (Davidson et al. 2003). Twenty-nine PTSD patients participated in this trial and were given MTZ or a placebo for 8 weeks. After 8 weeks of therapy, patients receiving MTZ had a response rate that was noticeably higher than those receiving a placebo, as determined. After 8 weeks of MTZ therapy, three open-label studies also discovered significant improvement rates (Connor et al. 1999; Bahk et al. 2002; Chung et al. 2004).
Panic disorder
One randomized double-blind experiment examining MTZ’s effectiveness for treating panic disorder has been published thus far (Ribeiro et al. 2001). Twenty-seven outpatients were randomized to an 8-week therapy of either fluoxetine or MTZ in a flexible-dose design following a 1-week placebo run-in. At the study’s conclusion, patients from both groups demonstrated statistically significant improvement; however, no differences between the two therapy groups could be seen. MTZ demonstrated encouraging effects in lowering depression and anxiety symptoms in panic disorders according to numerous open-label studies (Boshuisen et al. 2001; Carli et al. 2002; Sarchiapone et al. 2003).
Pain syndromes
In several clinical studies, MTZ may be helpful for pain syndromes (Brannon and Stone 1999; Nutt and Law 1999; Brannon et al. 2000; Bendtsen and Jensen 2004). Twenty-four patients with persistent tension-type headaches were enrolled in the Bendtsen et al. study, which was a randomized, double-blind, placebo-controlled crossover trial. After 8 weeks of treatment, MTZ significantly outperformed a placebo in the preventive treatment of persistent tension-type headaches (Bendtsen and Jensen 2004). The results from an open-label study also point to MTZ as a potential fibromyalgia treatment (Samborski et al. 2004).
Obsessive–compulsive disorder
Many individuals with obsessive–compulsive disorder (OCD) show little improvement with serotonin reuptake medications, which are the standard of care. Through a mechanism different from reuptake inhibition, MTZ improves serotonergic activity. The effectiveness of MTZ in patients with OCD has been examined in two randomized, controlled trials. MTZ’s effects on OCD have been documented to be effective in OCD as a pharmacotherapy (Koran et al. 2005). In a pilot trial, Pallanti et al. investigated the effects of citalopram and MTZ in patients with OCD who did not also have comorbid depression. When MTZ was combined with citalopram, they discovered a faster onset of responsive action in OCD symptoms and decreased undesirable side effects. The citalopram plus MTZ group had effects earlier than the citalopram plus placebo group. Responders were higher in the citalopram + MTZ group in the fourth week of treatment (Pallanti et al. 2004).
The potential protective effects of MTZ on diseases other than CNS-related disorders
Liver diseases
In mice fed a high-fat diet, Cheng et al. inspected the effects of MTZ on metabolic parameters. The results showed that compared to untreated mice, obese mice treated with MTZ had reduced levels of body weight, serum TG, and AST levels. Additionally, the treated mice had lower blood glucose levels and increased insulin sensitivity and glucose transporter 4 expression levels. In conclusion, type 2 diabetes mellitus (DM) with hyperglycemia may be improved by MTZ treatment (Wu et al. 2020).
Treatment with MTZ quickly changes hepatic B cell populations, increasing the retention of innate-like B cells that express CXCR3 and produce a more anti-inflammatory effect. Hepatic B cell changes induced by MTZ may constitute a novel therapeutic strategy for treating immune-mediated liver disorders with B cell-driven pathology. According to research by Almishri et al., the antidepressant MTZ rapidly shifts the hepatic B cell populations and functional cytokine signatures of mice. Treatment with MTZ increases the retention of innate-like B cells that express CXCR3, producing more anti-inflammatory cytokines. B cell-driven pathology associated with immune-mediated liver illnesses may be treated uniquely by MTZ-induced hepatic B cell shifts (Almishri et al. 2021). According to Davis et al., MTZ also affects liver innate immunity and inhibits immune-driven activation of hepatic macrophages. Treatment with MTZ may have beneficial effects on sepsis, and it significantly decreases the risk of hepatic abscess formation (Davis et al. 2020).
The effects of MTZ were examined by El-Tanbouly et al. in a mouse model of liver fibrosis induced by thioacetamide. MTZ treatment ameliorated TAA-induced liver fibrosis by lowering portal BP, liver procollagen I content, and α-SMA expression. Additionally, as determined by Masson’s trichrome staining, MTZ-treated animals had significantly less hepatic collagen accumulation. MTZ decreased the levels of protein kinase C, transforming factor β1 (TGF-β1), phosphorylated Smad3 (p-Smad), and phosphorylated extracellular signal-regulated kinases 1 and 2 (p-ERK1/2). Furthermore, MTZ significantly reduced oxidative stress, as seen by declining hepatic lipid peroxidation and NADPH oxidase 1 and increased GSH content (El-Tanbouly et al. 2017).
Kidney diseases
Sahin et al. investigated the antidepressant and antioxidant effects of MTZ in diabetic nephropathy. In this work, nod-like receptor family protein 3 NLRP3 and caspase-1 expression as well as the renal level of IL-1β were decreased in diabetic rats by MTZ administration. These findings imply that MTZ may be effective in the management of DM and other metabolic illnesses (Sahin et al. 2019).
Cardiovascular diseases related to CNS disorders
According to Kawano et al., MTZ significantly suppressed both the synergistic interaction of serotonin (5-HT) and adrenaline as well as the synergistic interaction of ADP and either 5-HT or adrenaline. The antiplatelet effects MTZ are mediated by 5-HT2A and α2-adrenergic receptors blocking (Kawano et al. 2022).
Diabetes
In T1DM rat models, MTZ reduced GLUT2 by changing the expression of leptin and galanin in the livers of type 1 diabetic rats with hyperglycemia. These findings concluded that patients with T1DM can use MTZ as an antidepressant and as a medication to lower blood glucose levels (Bektur et al. 2019a). Additionally, MTZ exhibits beneficial effects in diabetes-induced hyperalgesia. Since TRPV1 and ASIC1 levels are elevated in diabetic rats, MTZ’s suppressive influence on these levels may be one of the pharmacological mechanisms behind the drug’s therapeutic effects against diabetes-induced hyperalgesia (Bektur et al. 2019b).
Nausea and vomiting in chemotherapy-treated cancer patients
In a study of 35 patients, when given in conjunction with platinum-based regimens to patients with thoracic cancer, adding MTZ to the recommended antiemetic regimen for CINV may be advantageous with appropriate tolerability (Kinomura et al. 2023).
Heavy metal-induced neurotoxicity
A recent study examined the antioxidant and anti-inflammatory properties of MTZ against Cd-induced neurotoxicity. MTZ attenuates Cd-induced neurotoxicity by upregulating the transcription factor Nrf2, suppressing NF-κB/TLR4 signalling, and effectively decreasing TNF-α, IL-1β, and IL-6. Additionally, MTZ reduced histological abrasions in the dentate gyrus, CA1 and CA3 regions of the hippocampus, and cerebral cortex of the rat brain (Alharthy et al. 2023).
Reproductive diseases
Infertility
Khedr studied MTZ and hesperidin’s protective effects against cyclophosphamide-induced infertility in rat ovaries. The findings imply that cyclophosphamide (CP)-induced oxidative stress in rat ovaries causes infertility in female rats. Hesperidin and MTZ could counteract this impact and safeguard fertility against CP-induced damage (Khedr 2015).
The effect of MTZ on cisplatin-induced infertility was explored by Altuner et al. MTZ decreased the levels of MDA, MPO, NO, and 8-hydroxy-2 deoxyguanine and effectively increased GSH, GPx, and SOD levels. In conclusion, cisplatin-induced oxidative stress in rat ovarian tissue results in infertility in female rats. This is reversed by MTZ in a dose-dependent manner (Altuner et al. 2013).
El-Sisi et al. examined whether chrysin and MTZ could protect rat testicles from nitrofurazone-induced injury. In this study, chrysin and MTZ were used as preventative medicines to reduce the increase in blood acid phosphatase enzyme activity and to stop the reduction in sperm count and viability caused by nitrofurazone. Additionally, both medications reduced the effects of nitrofurazone on rat testes MDA, GSH depletion, elevation of TNF-α, and decrease in c-kit levels. In terms of apoptosis, immunohistochemistry research showed that nitrofurazone-induced testicular tissue expressed caspase-3, which was decreased by chrysin and MTZ. Histopathological data further substantiated the preventive benefits of both medications (El-Sisi et al. 2017).
Hot flushes
Hot flushes are frequently associated with a drop in estrogen production, which is mostly brought on by menopause (Utian 1972). In the majority of patients, hormone replacement therapy (HRT) relieves 67% of women’s symptoms around menopause. HRT has been acknowledged as the first line of defense against menopausal hot flushes; however, it is contraindicated in a variety of clinical entities (Staropoli et al. 1998). According to Marcel et al. evaluations of the medication’s impact on the frequency and intensity of hot flushes and sweating episodes in women, MTZ appears to significantly lessen both of these symptoms (Waldinger et al. 2000).
Pruritus
A frequent disease that can disrupt sleep and lower quality of life is chronic pruritus. The current approach to treating chronic itching targets the root of the problem, which may be dermatologic, systemic, or psychological in origin (Dhand and Aminoff 2014). Topical emollients, topical corticosteroids, and antihistamines are frequently the first treatments used in first-line therapy. For patients with persistent pruritus, GABA receptor modulators, opioid agonists/antagonists, and phototherapy can be employed (Dhand and Aminoff 2014). Recalcitrant itch is a bothersome symptom that requires a safe and efficient treatment (Davis et al. 2003; Kaur and Sinha 2018). Recent studies have shown that oral antidepressants are effective at reducing chronic itching caused by dermatologic, systemic, and psychogenic causes (Boozalis et al. 2018, 2019). One such antidepressant that has shown success in lowering itch severity is MTZ, a dual noradrenergic and serotonergic antidepressant with antihistaminic characteristics (Hundley and Yosipovitch 2004; Lee et al. 2016).
Safety and toxicity of MTZ
MTZ has an excellent safety and tolerability profile. The overall incidence rate of adverse clinical events among patients receiving MTZ (65%) was lower than that of those receiving placebo (76%) or amitriptyline (87%) treatment (Anttila and Leinonen 2001).
Despite higher MTZ dosages, the majority of adverse effects were minor, temporary, and became progressively less intense and frequent with time. Low-dose-associated sleepiness and weight gain are often reported side effects of MTZ therapy, and they may be connected to the drug’s affinity for the antihistaminic (H1) receptor. In short-term trials, sedation or sleepiness was noted at low dosages and decreased in both intensity and frequency or progressively vanished during the titration of MTZ to higher levels (Bremner 1995; Anttila and Leinonen 2001). It is hypothesized that noradrenergic stimulation dominates the antihistaminic activity at larger doses (Barkin 1997; Nelson 1997; Fawcett and Barkin 1998), although these side effects occurred less frequently in European studies due, in part, to greater doses and were also shown to diminish over time; a much higher percentage of patients treated with low doses of MTZ experienced an increase in appetite and weight gain (Anttila and Leinonen 2001).
During the clinical development program, MTZ was often not linked to changes in laboratory parameters or cardiovascular vital signs. Some MTZ-treated patients showed a small increase in body weight. Most negative MTZ-related encounters occurred in the first few weeks of treatment (Fawcett and Barkin 1998). The psychomotor impairment caused by the initial dose of MTZ may have an impact on driving abilities; however, this does not last throughout treatment (Ramaekers et al. 1998; Ridout et al. 2003; Wingen et al. 2005).
Conclusion and future recommendation
MTZ is an FDA-approved, effective, and well-tolerated drug for treating depression. MTZ is hypothesized to have antidepressant effects because of the synergy between noradrenergic and serotonergic actions and is effective in treating major depressive disorder and depression associated with epilepsy, Alzheimer’s disease, stroke, cardiovascular disease, and respiratory disease. Moreover, studies have reported the effectiveness of MTZ on other CNS disorders, such as schizophrenia, dysthymia, social anxiety disorder, alcohol dependency, posttraumatic stress disorder, panic disorder, pain syndromes, obsessive–compulsive disorder, and sleep disorders. Additionally, MTZ is potentially therapeutic against liver, kidney, cardiovascular, respiratory, infertility, heavy metal-induced neurotoxicity, and pruritus. In cancer patients, MTZ significantly reduced sadness, nausea, sleep disruption, and pain and improved quality of life. MTZ potentially protects against chemotherapy-induced toxicities such as cyclophosphamide-induced oxidative damage and infertility in rat ovaries. MTZ appears to significantly lessen the frequency and intensity of hot flushes and sweating episodes in women. These promising effects are mediated by potent antioxidant, anti-inflammatory, and anti-apoptotic effects. These positive outcomes of the scientific investigations motivate more and more clinical trials for a golden exceptional antidepressant.
Author contributions
All authors take public responsibility for the content of the work submitted for review. The authors confirm contribution to the paper as follows: study conception and design: Emad H. M. Hassanein and Hanan S. Althagafy; draft manuscript preparation: Islam M. Ibrahim and Mohammad A. Baraka; preparation of final manuscript: Emad H. M. Hassanein, Islam M. Ibrahim, Esraa K. Abd-alhameed, and Hanan S. Althagafy. All authors reviewed and approved the final version of the manuscript. The authors confirm that no paper mill and artificial intelligence was used.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Not applicable.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Alharthy SA, Zughaibi TA, Vij P, Tabrez S, Almashjary MN, Alharthi S, Alamri T, Alghamdi BS, Harakeh S, Azhari SA (2023) Mirtazapine attenuated cadmium-induced neuronal intoxication by regulating Nrf2 and NF-κB/TLR4 signals. Toxicol Mech Methods 33:675–687 [DOI] [PubMed]
- Allen ND, Leung JG, Palmer BA. Mirtazapine's effect on the QT interval in medically hospitalized patients. Mental Health Clinician. 2020;10:30–33. doi: 10.9740/mhc.2020.01.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Almishri W, Shaheen AA, Sharkey KA, Swain MG. The antidepressant mirtazapine inhibits hepatic innate immune networks to attenuate immune-mediated liver injury in mice. Front Immunol. 2019;10:803. doi: 10.3389/fimmu.2019.00803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Almishri W, Davis RP, Shaheen AA, Altonsy MO, Jenne CN, Swain MG. The antidepressant mirtazapine rapidly shifts hepatic B cell populations and functional cytokine signatures in the mouse. Front Immunol. 2021;12:622537. doi: 10.3389/fimmu.2021.622537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altuner D, Gulaboglu M, Yapca OE, Cetin N (2013) The effect of mirtazapine on cisplatin-induced oxidative damage and infertility in rat ovaries. Sci World J 2013:327240 [DOI] [PMC free article] [PubMed]
- Anttila SA, Leinonen EV. A review of the pharmacological and clinical profile of mirtazapine. CNS Drug Rev. 2001;7:249–264. doi: 10.1111/j.1527-3458.2001.tb00198.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aslan S, Isik E, Cosar B. The effects of mirtazapine on sleep: a placebo controlled, double-blind study in young healthy volunteers. Sleep. 2002;25:677–679. [PubMed] [Google Scholar]
- Bahk WM, Pae CU, Tsoh J, Chae JH, Jun TY, Kim KS. Effects of mirtazapine in patients with post-traumatic stress disorder in Korea: a pilot study. Hum Psychopharmacol Clin Exp. 2002;17:341–344. doi: 10.1002/hup.426. [DOI] [PubMed] [Google Scholar]
- Barkin RL. Cancer pain treatment insights. Pharmacotherapy. 1997;17:397–398. [PubMed] [Google Scholar]
- Batzel LW, Dodrill CB. Emotional and intellectual correlates of unsuccessful suicide attempts in people with epilepsy. J Clin Psychol. 1986;42:699–702. doi: 10.1002/1097-4679(198609)42:5<699::aid-jclp2270420503>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- Bektur E, Sahin E, Baycu C. Mirtazapine may show anti-hyperglycemic effect by decreasing GLUT2 through leptin and galanin expressions in the liver of type 1 diabetic rats. Iran J Basic Med Sci. 2019;22:676–682. doi: 10.22038/ijbms.2019.34529.8190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bektur E, Şahin E, Ceyhan E, Donmez DB, Canbek M, Baycu C, Can OD. Beneficial effect of mirtazapine on diabetes-induced hyperalgesia: involvement of TRPV1 and ASIC1 channels in the spinal cord and dorsal root ganglion. Neurol Res. 2019;41:544–553. doi: 10.1080/01616412.2019.1580462. [DOI] [PubMed] [Google Scholar]
- Bendtsen L, Jensen R. Mirtazapine is effective in the prophylactic treatment of chronic tension-type headache. Neurology. 2004;62:1706–1711. doi: 10.1212/01.wnl.0000127282.90920.8c. [DOI] [PubMed] [Google Scholar]
- Benjamin S, Doraiswamy PM. Review of the use of mirtazapine in the treatment of depression. Expert Opin Pharmacother. 2011;12:1623–1632. doi: 10.1517/14656566.2011.585459. [DOI] [PubMed] [Google Scholar]
- Benkert O, Muller M, Szegedi A. An overview of the clinical efficacy of mirtazapine. Hum Psychopharmacol Clin Exp. 2002;17:S23–S26. doi: 10.1002/hup.397. [DOI] [PubMed] [Google Scholar]
- Berk M, Ichim C, Brook S. Efficacy of mirtazapine add on therapy to haloperidol in the treatment of the negative symptoms of schizophrenia: a double-blind randomized placebo-controlled study. Int Clin Psychopharmacol. 2001;16:87–92. doi: 10.1097/00004850-200103000-00003. [DOI] [PubMed] [Google Scholar]
- Bilici M, Ozturk C, Dursun H, Albayrak F, Saglam MB, Uyanik A, Gulaboglu M, Tekin SB. Protective effect of mirtazapine on indomethacin-induced ulcer in rats and its relationship with oxidant and antioxidant parameters. Dig Dis Sci. 2009;54:1868–1875. doi: 10.1007/s10620-008-0560-z. [DOI] [PubMed] [Google Scholar]
- Blumer D (2002) Epilepsy and suicide: a neuropsychiatric analysis. In: Trimble M, Schmitz B (Eds) The neuropsychiatry of epilepsy. Cambridge, pp 107–116
- Boozalis E, Khanna R, Kwatra SG. Selective serotonin reuptake inhibitors for the treatment of chronic pruritus. J Dermatol Treat. 2018;29:812–814. doi: 10.1080/09546634.2018.1456639. [DOI] [PubMed] [Google Scholar]
- Boozalis E, Khanna R, Zampella JG, Kwatra SG (2021) Tricyclic antidepressants for the treatment of chronic pruritus. J Dermatol Treat 32:124–126 [DOI] [PubMed]
- Boshuisen M, Slaap B, Vester-Blokland E, Den Boer J. The effect of mirtazapine in panic disorder: an open label pilot study with a single-blind placebo run-in period. Int Clin Psychopharmacol. 2001;16:363–368. doi: 10.1097/00004850-200111000-00008. [DOI] [PubMed] [Google Scholar]
- Brannon GE, Stone KD. The use of mirtazapine in a patient with chronic pain. J Pain Symptom Manage. 1999;18:382–385. doi: 10.1016/s0885-3924(99)00098-6. [DOI] [PubMed] [Google Scholar]
- Brannon GE, Rolland PD, Gary JM. Use of mirtazapine as prophylactic treatment for migraine headache. Psychosomatics. 2000;41:153–154. doi: 10.1176/appi.psy.41.2.153. [DOI] [PubMed] [Google Scholar]
- Bremner JD (1995) A double-blind comparison of Org 3770, amitriptyline, and placebo in major depression. J Clin Psychiatry 56:519–525 [PubMed]
- Bruijn JA, van den Broek W, van Hulst A, Van der Mast R, van de Wetering B, Moleman P, Mulder P. A double-blind, fixed blood-level study comparing mirtazapine with imipramine in depressed in-patients. Psychopharmacology. 1996;127:231–237. [PubMed] [Google Scholar]
- Cankurtaran ES, Ozalp E, Soygur H, Akbiyik DI, Turhan L, Alkis N. Mirtazapine improves sleep and lowers anxiety and depression in cancer patients: superiority over imipramine. Support Care Cancer. 2008;16:1291–1298. doi: 10.1007/s00520-008-0425-1. [DOI] [PubMed] [Google Scholar]
- Caraci F, Copani A, Nicoletti F, Drago F. Depression and Alzheimer's disease: neurobiological links and common pharmacological targets. Eur J Pharmacol. 2010;626:64–71. doi: 10.1016/j.ejphar.2009.10.022. [DOI] [PubMed] [Google Scholar]
- Carli V, Sarchiapone M, Camardese G, Romano L, DeRisio S. Mirtazapine in the treatment of panic disorder. Arch Gen Psychiatry. 2002;59:661–662. doi: 10.1001/archpsyc.59.7.661. [DOI] [PubMed] [Google Scholar]
- Chung MY, Min KH, Jun YJ, Kim SS, Kim WC, Jun EM. Efficacy and tolerability of mirtazapine and sertraline in Korean veterans with posttraumatic stress disorder: a randomized open label trial. Hum Psychopharmacol Clin Exp. 2004;19:489–494. doi: 10.1002/hup.615. [DOI] [PubMed] [Google Scholar]
- Connor K, Davidson J, Weisler R, Ahearn E. A pilot study of mirtazapine in post-traumatic stress disorder. Int Clin Psychopharmacol. 1999;14:29–31. doi: 10.1097/00004850-199901000-00003. [DOI] [PubMed] [Google Scholar]
- Costa FH, Rosso AL, Maultasch H, Nicaretta DH, Vincent MB. Depression in Parkinson's disease: diagnosis and treatment. Arq Neuropsiquiatr. 2012;70:617–620. doi: 10.1590/s0004-282x2012000800011. [DOI] [PubMed] [Google Scholar]
- Dahl M-L, Voortman G, Alm C, Elwin C-E, Delbressine L, Vos R, Bogaards J, Bertilsson L. In vitro and in vivo studies on the disposition of mirtazapine in humans. Clin Drug Investig. 1997;13:37–46. [Google Scholar]
- Davidson JR, Weisler RH, Butterfield MI, Casat CD, Connor KM, Barnett S, van Meter S. Mirtazapine vs. placebo in posttraumatic stress disorder: a pilot trial. Biol Psychiat. 2003;53:188–191. doi: 10.1016/s0006-3223(02)01411-7. [DOI] [PubMed] [Google Scholar]
- Davis MP, Frandsen JL, Walsh D, Andresen S, Taylor S. Mirtazapine for pruritus. J Pain Symptom Manage. 2003;25:288–291. doi: 10.1016/s0885-3924(02)00645-0. [DOI] [PubMed] [Google Scholar]
- Davis RP, Almishri W, Jenne CN, Swain MG. The antidepressant mirtazapine activates hepatic macrophages, facilitating pathogen clearance while limiting tissue damage in mice. Front Immunol. 2020;11:578654. doi: 10.3389/fimmu.2020.578654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Boer T, Maura G, Raiteri M, De Vos C, Wieringa J, Pinder R. Neurochemical and autonomic pharmacological profiles of the 6-aza-analogue of mianserin, Org 3770 and its enantiomers. Neuropharmacology. 1988;27:399–408. doi: 10.1016/0028-3908(88)90149-9. [DOI] [PubMed] [Google Scholar]
- De Boer T, Ruigt G, Berendsen H. The α2-selective adrenoceptor antagonist org 3770 (mirtazapine, Remeron®) enhances noradrenergic and serotonergic transmission. Hum Psychopharmacol Clin Exp. 1995;10:S107–S118. [Google Scholar]
- Delbressine L, Moonen M, Kaspersen F, Wagenaars G, Jacobs P, Timmer C, Paanakker J, Van Hal H, Voortman G. Pharmacokinetics and biotransformation of mirtazapine in human volunteers. Clin Drug Investig. 1998;15:45–55. doi: 10.2165/00044011-199815010-00006. [DOI] [PubMed] [Google Scholar]
- Dhand A, Aminoff MJ. The neurology of itch. Brain. 2014;137:313–322. doi: 10.1093/brain/awt158. [DOI] [PubMed] [Google Scholar]
- El-Sisi AE, El-Sayad ME, Abdelsalam NM. Protective effects of mirtazapine and chrysin on experimentally induced testicular damage in rats. Biomed Pharmacother. 2017;95:1059–1066. doi: 10.1016/j.biopha.2017.09.022. [DOI] [PubMed] [Google Scholar]
- Elsisi AEE, Sokar SS, Shalaby MF, Abu-Risha SE. Nephroprotective effects of febuxostat and/or mirtazapine against gentamicin-induced nephrotoxicity through modulation of ERK 1/2, NF-κB and MCP1. Expert Rev Clin Pharmacol. 2021;14:1039–1050. doi: 10.1080/17512433.2021.1933435. [DOI] [PubMed] [Google Scholar]
- El-Tanbouly DM, Wadie W, Sayed RH. Modulation of TGF-β/Smad and ERK signaling pathways mediates the anti-fibrotic effect of mirtazapine in mice. Toxicol Appl Pharmacol. 2017;329:224–230. doi: 10.1016/j.taap.2017.06.012. [DOI] [PubMed] [Google Scholar]
- Engel D, Zomkowski AD, Lieberknecht V, Rodrigues AL, Gabilan NH. Chronic administration of duloxetine and mirtazapine downregulates proapoptotic proteins and upregulates neurotrophin gene expression in the hippocampus and cerebral cortex of mice. J Psychiatr Res. 2013;47:802–808. doi: 10.1016/j.jpsychires.2013.02.013. [DOI] [PubMed] [Google Scholar]
- Fawcett J, Barkin RL. Review of the results from clinical studies on the efficacy, safety and tolerability of mirtazapine for the treatment of patients with major depression. J Affect Disord. 1998;51:267–285. doi: 10.1016/s0165-0327(98)00224-9. [DOI] [PubMed] [Google Scholar]
- Forsgren L, Nyström L. An incident case-referent study of epileptic seizures in adults. Epilepsy Res. 1990;6:66–81. doi: 10.1016/0920-1211(90)90010-s. [DOI] [PubMed] [Google Scholar]
- Górska N, Słupski J, Cubała WJ, Wiglusz MS, Gałuszko-Węgielnik M. Antidepressants in epilepsy. Neurol Neurochir Pol. 2018;52:657–661. doi: 10.1016/j.pjnns.2018.07.005. [DOI] [PubMed] [Google Scholar]
- Guelfi JD, Ansseau M, Timmerman L, Kørsgaard S, Group M-VS Mirtazapine versus venlafaxine in hospitalized severely depressed patients with melancholic features. J Clin Psychopharmacol. 2001;21:425–431. doi: 10.1097/00004714-200108000-00010. [DOI] [PubMed] [Google Scholar]
- Gulec M, Oral E, Dursun OB, Yucel A, Hacimuftuoglu A, Akcay F, Suleyman H. Mirtazapine protects against cisplatin-induced oxidative stress and DNA damage in the rat brain. Psychiatry Clin Neurosci. 2013;67:50–58. doi: 10.1111/j.1440-1819.2012.02395.x. [DOI] [PubMed] [Google Scholar]
- Gupta R, Gupta K, Tripathi A, Bhatia M, Gupta LK. Effect of mirtazapine treatment on serum levels of brain-derived neurotrophic factor and tumor necrosis factor-α in patients of major depressive disorder with severe depression. Pharmacology. 2016;97:184–188. doi: 10.1159/000444220. [DOI] [PubMed] [Google Scholar]
- Hafez HM, Ibrahim MA, Abdelzaher WY, Gad AA, Hafez SMNA, Abdel-Gaber SA. Protective effect of mirtazapine against acetic acid-induced ulcerative colitis in rats: role of NLRP3 inflammasome pathway. Int Immunopharmacol. 2021;101:108174. doi: 10.1016/j.intimp.2021.108174. [DOI] [PubMed] [Google Scholar]
- Hafez HM, Ibrahim MA, Yehia Abdelzaher W, Gad AA, Mohammed Naguib Abdel Hafez S, Abdel-Gaber SA. Protective effect of mirtazapine against acetic acid-induced ulcerative colitis in rats: Role of NLRP3 inflammasome pathway. Int Immunopharmacol. 2021;101:108174. doi: 10.1016/j.intimp.2021.108174. [DOI] [PubMed] [Google Scholar]
- Holm KJ, Markham A. Mirtazapine. Drugs. 1999;57:607–631. doi: 10.2165/00003495-199957040-00010. [DOI] [PubMed] [Google Scholar]
- Honig A, Kuyper AM, Schene AH, van Melle JP, De Jonge P, Tulner DM, Schins A, Crijns HJ, Kuijpers PM, Vossen H. Treatment of post-myocardial infarction depressive disorder: a randomized, placebo-controlled trial with mirtazapine. Psychosom Med. 2007;69:606–613. doi: 10.1097/PSY.0b013e31814b260d. [DOI] [PubMed] [Google Scholar]
- Hundley JL, Yosipovitch G. Mirtazapine for reducing nocturnal itch in patients with chronic pruritus: a pilot study. J Am Acad Dermatol. 2004;50:889–891. doi: 10.1016/j.jaad.2004.01.045. [DOI] [PubMed] [Google Scholar]
- Kaur R, Sinha V. Antidepressants as antipruritic agents: a review. Eur Neuropsychopharmacol. 2018;28:341–352. doi: 10.1016/j.euroneuro.2018.01.007. [DOI] [PubMed] [Google Scholar]
- Kawano Y, Obana M, Nagata M, Mano Y, Katsuyama M, Yamamoto Y, Maeda-Minami A, Negishi K, Takagi M, Shimada S, Aoyama T. The antiplatelet effect of mirtazapine is mediated by co-blocking 5-HT(2A) and α(2)-adrenergic receptors on platelets: An in vitro human plasma-based study. Eur J Pharmacol. 2022;917:174640. doi: 10.1016/j.ejphar.2021.174640. [DOI] [PubMed] [Google Scholar]
- Khedr NF. Protective effect of mirtazapine and hesperidin on cyclophosphamide-induced oxidative damage and infertility in rat ovaries. Exp Biol Med (Maywood) 2015;240:1682–1689. doi: 10.1177/1535370215576304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SW, Shin IS, Kim JM, Kim YC, Kim KS, Kim KM, Yang SJ, Yoon JS. Effectiveness of mirtazapine for nausea and insomnia in cancer patients with depression. Psychiatry Clin Neurosci. 2008;62:75–83. doi: 10.1111/j.1440-1819.2007.01778.x. [DOI] [PubMed] [Google Scholar]
- Kinomura M, Iihara H, Fujii H, Hirose C, Endo J, Yanase K, Inui T, Kaito D, Sasaki Y, Gomyo T, Sakai-Masuda C, Kawae D, Kitamura YU, Fukui M, Kobayashi R, Ohno Y, Suzuki A. Effect of mirtazapine for the prevention of nausea and vomiting in patients with thoracic cancer receiving platinum-based chemotherapy. Anticancer Res. 2023;43:1301–1307. doi: 10.21873/anticanres.16277. [DOI] [PubMed] [Google Scholar]
- Koran LM, Gamel NN, Choung HW, Smith EH, Aboujaoude EN. Mirtazapine for obsessive-compulsive disorder: an open trial followed by double-blind discontinuation. J Clin Psychiatry. 2005;66:515–520. [PubMed] [Google Scholar]
- Laakmann G, Schüle C, Baghai T, Waldvogel E. Effects of mirtazapine on growth hormone, prolactin, and cortisol secretion in healthy male subjects. Psychoneuroendocrinology. 1999;24:769–784. doi: 10.1016/s0306-4530(99)00029-3. [DOI] [PubMed] [Google Scholar]
- Laakmann G, Hennig J, Baghai T, SCHüLE C. Mirtazapine acutely inhibits salivary cortisol concentrations in depressed patients. Ann NY Acad Sci. 2004;1032:279–282. doi: 10.1196/annals.1314.038. [DOI] [PubMed] [Google Scholar]
- Lee JJ, Girouard SD, Carlberg VM, Mostaghimi A. Effective use of mirtazapine for refractory pruritus associated with carcinoma en cuirasse. BMJ Support Palliat Care. 2016;6:119–121. doi: 10.1136/bmjspcare-2014-000790. [DOI] [PubMed] [Google Scholar]
- Leinonen E, Skarstein J, Behnke K, Agren H, Helsdingen JT. Efficacy and tolerability of mirtazapine versus citalopram: a double-blind, randomized study in patients with major depressive disorder. Nordic Antidepressant Study Group. Int Clin Psychopharmacol. 1999;14:329–337. doi: 10.1097/00004850-199911000-00002. [DOI] [PubMed] [Google Scholar]
- Liappas J, Paparrigopoulos T, Tzavellas E, Christodoulou G. Alcohol detoxification and social anxiety symptoms: a preliminary study of the impact of mirtazapine administration. J Affect Disord. 2003;76:279–284. doi: 10.1016/s0165-0327(02)00094-0. [DOI] [PubMed] [Google Scholar]
- Liappas J, Paparrigopoulos T, Tzavellas E, Rabavilas A. Mirtazapine and venlafaxine in the management of collateral psychopathology during alcohol detoxification. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29:55–60. doi: 10.1016/j.pnpbp.2004.10.005. [DOI] [PubMed] [Google Scholar]
- Lieberknecht V, Engel D, Rodrigues ALS, Gabilan NH. Neuroprotective effects of mirtazapine and imipramine and their effect in pro- and anti-apoptotic gene expression in human neuroblastoma cells. Pharmacol Rep. 2020;72:563–570. doi: 10.1007/s43440-019-00009-w. [DOI] [PubMed] [Google Scholar]
- Liguori I, Russo G, Curcio F, Bulli G, Aran L, Della-Morte D, Gargiulo G, Testa G, Cacciatore F, Bonaduce D, Abete P. Oxidative stress, aging, and diseases. Clin Interv Aging. 2018;13:757–772. doi: 10.2147/CIA.S158513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyketsos CG, Olin J. Depression in Alzheimer's disease: overview and treatment. Biol Psychiatry. 2002;52:243–252. doi: 10.1016/s0006-3223(02)01348-3. [DOI] [PubMed] [Google Scholar]
- Mahmoud AM, Alexander MY, Tutar Y, Wilkinson FL, Venditti A. Oxidative Stress in Metabolic Disorders and Drug-Induced Injury: The Potential Role of Nrf2 and PPARs Activators. Oxid Med Cell Longev. 2017;2017:2508909. doi: 10.1155/2017/2508909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maneesh M, Jayalekshmi H. Role of reactive oxygen species and antioxidants on pathophysiology of male reproduction. Indian J Clin Biochem. 2006;21:80–89. doi: 10.1007/BF02912918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann DB, Laitman LB, Davis KL. Dementia with coexistent major depression. Am J Psychiatry. 1989;146:1472–1478. doi: 10.1176/ajp.146.11.1472. [DOI] [PubMed] [Google Scholar]
- Matthews WS, Barabas G. Suicide and epilepsy: a review of the literature. Psychosomatics. 1981;22:515–524. doi: 10.1016/S0033-3182(81)73498-4. [DOI] [PubMed] [Google Scholar]
- Moens K, Higginson IJ, Harding R, Brearley S, Caraceni A, Cohen J, Costantini M, Deliens L, Francke AL, Kaasa S. Are there differences in the prevalence of palliative care-related problems in people living with advanced cancer and eight non-cancer conditions? A systematic review. J Pain Symptom Manage. 2014;48:660–677. doi: 10.1016/j.jpainsymman.2013.11.009. [DOI] [PubMed] [Google Scholar]
- Mula M. Epilepsy and psychiatric comorbidities: drug selection. Curr Treat Options Neurol. 2017;19:1–11. doi: 10.1007/s11940-017-0483-0. [DOI] [PubMed] [Google Scholar]
- Nelson JC. Safety and tolerability of the new antidepressants. J Clin Psychiatry. 1997;58:26–31. [PubMed] [Google Scholar]
- Niedermaier N, Bohrer E, Schulte K, Schlattmann P, Heuser I. Prevention and treatment of poststroke depression with mirtazapine in patients with acute stroke. J Clin Psychiatry. 2004;65:1619–1623. doi: 10.4088/jcp.v65n1206. [DOI] [PubMed] [Google Scholar]
- Nutt D, Law J. Treatment of cluster headache with mirtazapine. Headache. 1999;39:586–587. doi: 10.1046/j.1526-4610.1999.t01-1-3908586.x. [DOI] [PubMed] [Google Scholar]
- Pallanti S, Quercioli L, Bruscoli M. Response acceleration with mirtazapine augmentation of citalopram in obsessive-compulsive disorder patients without comorbid depression: a pilot study. J Clin Psychiatry. 2004;65:1394–1399. doi: 10.4088/jcp.v65n1015. [DOI] [PubMed] [Google Scholar]
- Pena E, Mata M, Lopez-Manzanares L, Kurtis M, Eimil M, Martinez-Castrillo J, Navas I, Posada I, Prieto C, Ruiz-Huete C. Antidepressants in Parkinson's disease. Recommendations by the movement disorder study group of the Neurological Association of Madrid. Neurología (English Edition) 2018;33:395–402. doi: 10.1016/j.nrl.2016.02.002. [DOI] [PubMed] [Google Scholar]
- Pinder RM. Designing a new generation of antidepressant drugs. Acta Psychiatr Scand Suppl. 1997;391:7–13. doi: 10.1111/j.1600-0447.1997.tb05953.x. [DOI] [PubMed] [Google Scholar]
- Pinder RM. The pharmacologic rationale for the clinical use of antidepressants. J Clin Psychiatry. 1997;58:501–508. doi: 10.4088/jcp.v58n1108. [DOI] [PubMed] [Google Scholar]
- Poyurovsky M, Epshtein S, Fuchs C, Schneidman M, Weizman R, Weizman A. Efficacy of low-dose mirtazapine in neuroleptic-induced akathisia: a double-blind randomized placebo-controlled pilot study. J Clin Psychopharmacol. 2003;23:305–308. doi: 10.1097/01.jcp.0000084027.22282.16. [DOI] [PubMed] [Google Scholar]
- Raji MA, Brady SR. Mirtazapine for treatment of depression and comorbidities in Alzheimer disease. Ann Pharmacother. 2001;35:1024–1027. doi: 10.1345/aph.10371. [DOI] [PubMed] [Google Scholar]
- Ramaekers J, Muntjewerff N, Van Veggel L, Uiterwijk M, O'Hanlon J. Effects of nocturnal doses of mirtazapine and mianserin on sleep and on daytime psychomotor and driving performance in young, healthy volunteers. Hum Psychopharmacol Clin Exp. 1998;13:S87–S97. [Google Scholar]
- Rani V, Deep G, Singh RK, Palle K, Yadav UC. Oxidative stress and metabolic disorders: Pathogenesis and therapeutic strategies. Life Sci. 2016;148:183–193. doi: 10.1016/j.lfs.2016.02.002. [DOI] [PubMed] [Google Scholar]
- Ribeiro L, Busnello JV, Kauer-Sant'Anna M, Madruga M, Quevedo J, Busnello E, Kapczinski F. Mirtazapine versus fluoxetine in the treatment of panic disorder. Braz J Med Biol Res. 2001;34:1303–1307. doi: 10.1590/s0100-879x2001001000010. [DOI] [PubMed] [Google Scholar]
- Richou H, Ruimy P, Charbaut J, Delisle J, Brunner H, Patris M, Zivkov M. A multicentre, double-blind, clomipramine-controlled efficacy and safety study of Org 3770. Hum Psychopharmacol Clin Exp. 1995;10:263–271. [Google Scholar]
- Ridout F, Meadows R, Johnsen S, Hindmarch I. A placebo controlled investigation into the effects of paroxetine and mirtazapine on measures related to car driving performance. Hum Psychopharmacol. 2003;18:261–269. doi: 10.1002/hup.494. [DOI] [PubMed] [Google Scholar]
- Saha S, Chant D, Welham J, McGrath J. A systematic review of the prevalence of schizophrenia. PLoS Med. 2005;2:e141. doi: 10.1371/journal.pmed.0020141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin E, Bektur E, Burukoglu Donmez D, Baycu C, Can OD, Sahinturk V. Mirtazapine suppresses sterile inflammation through NLRP3-inflammasome in diabetic rat kidney. Acta Histochem. 2019;121:289–296. doi: 10.1016/j.acthis.2019.01.007. [DOI] [PubMed] [Google Scholar]
- Salavati B, Rajji TK, Price R, Sun Y, Graff-Guerrero A, Daskalakis ZJ. Imaging-based neurochemistry in schizophrenia: a systematic review and implications for dysfunctional long-term potentiation. Schizophr Bull. 2015;41:44–56. doi: 10.1093/schbul/sbu132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samborski W, Lezanska-Szpera M, Rybakowski JK. Open trial of mirtazapine in patients with fibromyalgia. Pharmacopsychiatry. 2004;37:168–170. doi: 10.1055/s-2004-827172. [DOI] [PubMed] [Google Scholar]
- Sandker G, Vos R, Delbressine L, Slooff M, Meijer D, Groothuis G. Metabolism of three pharmacologically active drugs in isolated human and rat hepatocytes: analysis of interspecies variability and comparison with metabolism in vivo. Xenobiotica. 1994;24:143–155. doi: 10.3109/00498259409043228. [DOI] [PubMed] [Google Scholar]
- Sarchiapone M, Amore M, De Risio S, Carli V, Faia V, Poterzio F, Balista C, Camardese G, Ferrari G. Mirtazapine in the treatment of panic disorder: an open-label trial. Int Clin Psychopharmacol. 2003;18:35–38. doi: 10.1097/00004850-200301000-00006. [DOI] [PubMed] [Google Scholar]
- Schüle C, Baghai T, Goy J, Bidlingmaier M, Strasburger C, Laakmann G. The influence of mirtazapine on anterior pituitary hormone secretion in healthy male subjects. Psychopharmacology. 2002;163:95–101. doi: 10.1007/s00213-002-1148-5. [DOI] [PubMed] [Google Scholar]
- Schule C, Baghai T, Rackwitz C, Laakmann G. Influence of mirtazapine on urinary free cortisol excretion in depressed patients. Psychiatry Res. 2003;120:257–264. doi: 10.1016/s0165-1781(03)00204-x. [DOI] [PubMed] [Google Scholar]
- Shen J, Chung SA, Kayumov L, Moller H, Hossain N, Wang X, Deb P, Sun F, Huang X, Novak M, Appleton D, Shapiro CM. Polysomnographic and symptomatological analyses of major depressive disorder patients treated with mirtazapine. Can J Psychiatry. 2006;51:27–34. doi: 10.1177/070674370605100106. [DOI] [PubMed] [Google Scholar]
- Shi Y, Yang D, Zeng Y, Wu W. Risk factors for post-stroke depression: a meta-analysis. Front Aging Neurosci. 2017;9:218. doi: 10.3389/fnagi.2017.00218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sid-Otmane L, Huot P, Panisset M. Effect of antidepressants on psychotic symptoms in Parkinson disease: a review of case reports and case series. Clin Neuropharmacol. 2020;43:61–65. doi: 10.1097/WNF.0000000000000384. [DOI] [PubMed] [Google Scholar]
- Sinha Hikim AP, Lue Y, Diaz-Romero M, Yen PH, Wang C, Swerdloff RS. Deciphering the pathways of germ cell apoptosis in the testis. J Steroid Biochem Mol Biol. 2003;85:175–182. doi: 10.1016/s0960-0760(03)00193-6. [DOI] [PubMed] [Google Scholar]
- Solano JP, Gomes B, Higginson IJ. A comparison of symptom prevalence in far advanced cancer, AIDS, heart disease, chronic obstructive pulmonary disease and renal disease. J Pain Symptom Manage. 2006;31:58–69. doi: 10.1016/j.jpainsymman.2005.06.007. [DOI] [PubMed] [Google Scholar]
- Stanisław Wiglusz M, Jerzy Cubała W, Gałuszko-Węgielnik M, Jakuszkowiak-Wojten K, Landowski J. Mood disorders in epilepsy–diagnostic and methodological considerations. Psychiatr Danub. 2012;24:44–50. [PubMed] [Google Scholar]
- Staropoli CA, Flaws JA, Bush TL, Moulton AW. Predictors of menopausal hot flashes. J Womens Health. 1998;7:1149–1155. doi: 10.1089/jwh.1998.7.1149. [DOI] [PubMed] [Google Scholar]
- Tagai K, Nagata T, Shinagawa S, Tsuno N, Ozone M, Nakayama K. Mirtazapine improves visual hallucinations in Parkinson's disease: a case report. Psychogeriatrics. 2013;13:103–107. doi: 10.1111/j.1479-8301.2012.00432.x. [DOI] [PubMed] [Google Scholar]
- Taylor-Rowan M, Momoh O, Ayerbe L, Evans JJ, Stott DJ, Quinn TJ. Prevalence of pre-stroke depression and its association with post-stroke depression: a systematic review and meta-analysis. Psychol Med. 2019;49:685–696. doi: 10.1017/S0033291718002003. [DOI] [PubMed] [Google Scholar]
- Theobald DE, Kirsh KL, Holtsclaw E, Donaghy K, Passik SD. An open-label, crossover trial of mirtazapine (15 and 30 mg) in cancer patients with pain and other distressing symptoms. J Pain Symptom Manage. 2002;23:442–447. doi: 10.1016/s0885-3924(02)00381-0. [DOI] [PubMed] [Google Scholar]
- Timmer C, Paanakker J, Van Hal H. Pharmacokinetics of mirtazapine from orally administered tablets: influence of gender, age and treatment regimen. Hum Psychopharmacol Clin Exp. 1996;11:497–509. [Google Scholar]
- Timmer CJ, Sitsen J, Delbressine LP. Clinical pharmacokinetics of mirtazapine. Clin Pharmacokinet. 2000;38:461–474. doi: 10.2165/00003088-200038060-00001. [DOI] [PubMed] [Google Scholar]
- Tok A, Sener E, Albayrak A, Cetin N, Polat B, Suleyman B, Akcay F, Suleyman H. Effect of mirtazapine on oxidative stress created in rat kidneys by ischemia-reperfusion. Ren Fail. 2012;34:103–110. doi: 10.3109/0886022X.2011.623499. [DOI] [PubMed] [Google Scholar]
- Utian WH. The true clinical features of postmenopause and oophorectomy, and their response to oestrogen therapy. S Afr Med J. 1972;46:732–737. [PubMed] [Google Scholar]
- Voortman G, Paanakker J. Bioavailability of mirtazapine from Remeron® tablets after single and multiple oral dosing. Hum Psychopharmacol Clin Exp. 1995;10:S83–S96. [Google Scholar]
- Van der Vorstenbosch C, Delbressine L (1988) Binding of Org 3770 to human plasma proteins and human liver proteins [Report No. 2157]. NV Organon, Oss
- Waldinger MD, Berendsen HH, Schweitzer DH. Treatment of hot flushes with mirtazapine: four case reports. Maturitas. 2000;36:165–168. doi: 10.1016/s0378-5122(00)00152-3. [DOI] [PubMed] [Google Scholar]
- Wang Q, Ma M, Yu H, Yu H, Zhang S, Li R. Mirtazapine prevents cell activation, inflammation, and oxidative stress against isoflurane exposure in microglia. Bioengineered. 2022;13:521–530. doi: 10.1080/21655979.2021.2009971. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Wheatley DP, Van Moffaert M, Timmerman L, Kremer CM. Mirtazapine: efficacy and tolerability in comparison with fluoxetine in patients with moderate to severe major depressive disorder. J Clin Psychiatry. 1998;59:306–312. [PubMed] [Google Scholar]
- Wingen M, Bothmer J, Langer S, Ramaekers JG. Actual driving performance and psychomotor function in healthy subjects after acute and subchronic treatment with escitalopram, mirtazapine, and placebo: a crossover trial. J Clin Psychiatry. 2005;66:436–443. doi: 10.4088/jcp.v66n0405. [DOI] [PubMed] [Google Scholar]
- Winokur A, Reynolds C. The effects of antidepressants on sleep physiology. Primary Psychiatry. 1994;1:22–27. [Google Scholar]
- Winokur A, Sateia MJ, Hayes JB, Bayles-Dazet W, MacDonald MM, Gary KA. Acute effects of mirtazapine on sleep continuity and sleep architecture in depressed patients: a pilot study. Biol Psychiatry. 2000;48:75–78. doi: 10.1016/s0006-3223(00)00882-9. [DOI] [PubMed] [Google Scholar]
- Winokur A, DeMartinis NA, 3rd, McNally DP, Gary EM, Cormier JL, Gary KA. Comparative effects of mirtazapine and fluoxetine on sleep physiology measures in patients with major depression and insomnia. J Clin Psychiatry. 2003;64:1224–1229. doi: 10.4088/jcp.v64n1013. [DOI] [PubMed] [Google Scholar]
- Wu C-F, Hou P-H, Mao FC, Su Y-C, Wu C-Y, Yang W-C, Lin C-S, Tsai H-P, Liao H-J, Chang G-R. Mirtazapine reduces adipocyte hypertrophy and increases glucose transporter expression in obese mice. Animals. 2020;10:1423. doi: 10.3390/ani10081423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang AC, Tsai S-J. New targets for schizophrenia treatment beyond the dopamine hypothesis. Int J Mol Sci. 2017;18:1689. doi: 10.3390/ijms18081689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang R, Gui X, Zhang Y, Xiong Y. The role of essential organ-based comorbidities in the prognosis of COVID-19 infection patients. Expert Rev Respir Med. 2020;14:835–838. doi: 10.1080/17476348.2020.1761791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J, Wei X, Feng X, Song J, Hu Y, Xu J. Repeated administration of mirtazapine inhibits development of hyperalgesia/allodynia and activation of NF-κB in a rat model of neuropathic pain. Neurosci Lett. 2008;433:33–37. doi: 10.1016/j.neulet.2007.12.037. [DOI] [PubMed] [Google Scholar]
- Zoccali R, Muscatello MR, Cedro C, Neri P, La Torre D, Spina E, Di Rosa AE, Meduri M. The effect of mirtazapine augmentation of clozapine in the treatment of negative symptoms of schizophrenia: a double-blind, placebo-controlled study. Int Clin Psychopharmacol. 2004;19:71–76. doi: 10.1097/00004850-200403000-00003. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.