Abstract
Cannabis–drug interactions have caused significant concerns, mainly due to their role in the cytochrome P450 (CYP) enzyme‐mediated metabolic pathway of numerous medications. A systematic review was conducted to gain an overview of the potential interactions of cannabis with different drug classes by extracting pertinent information from published study data. From the inception of the study to October 1, 2023, we performed a systematic search of PubMed, Scopus, clinicaltrials.gov, and Web of Science. We included 54 out of 464 articles, and a total of 20 drug classes were identified to have interactions with medicinal cannabis. The cannabis–drug interactions were assessed and classified according to their probability and severity. The analysis revealed that antiepileptics had the most evidence of interaction with cannabis, followed by clobazam (CLB), warfarin, and tacrolimus. Generally, cannabis–drug interactions result in pharmacokinetic (PK) or pharmacodynamic (PD) changes. Therefore, careful monitoring should be performed to detect any unusual elevations in plasma levels. In addition, dose titrations or treatment withdrawal could help mitigate the adverse effects attributed to cannabis–drug interactions. Nevertheless, novel drugs are constantly emerging, and more research is needed to further identify potential interactions with cannabis.
INTRODUCTION
Cannabis sativa L., widely known as cannabis, is a plant‐based psychoactive product with origins dating back to 5000 years ago. 1 Cannabis has been known to be used in both recreational and medical contexts. It has been used in folk medicine and as a source of textile fiber in ancient times. 2 This substance contains over 100 active plant compounds called cannabinoids, which include cannabidiol (CBD) and delta‐9‐tetrahydrocannabinol (THC). 3 There are numerous ways to consume cannabis, including joints, pipes, bongs, blunts, oils, edibles, and vaporizer pens.
Medical cannabis has received a considerable amount of attention from the public in recent years. In 2018, the Food and Drug Administration (FDA) approved Epidiolex®, an oral CBD‐based medication, to treat two severe forms of pediatric epilepsy, namely Dravet syndrome and Lennox–Gastaut syndrome. CBD has also been used for a variety of unofficial purposes, including inflammation, chronic pain, anxiety, muscle stiffness, and even cancer. 4 , 5 Since the 1980s, synthetic THC‐based medications have been authorized for use as an antiemetic in cancer and appetite stimulation in patients with human immunodeficiency virus (HIV). The generic medicine, dronabinol, is available in capsule form (Marinol®) and as an oral solution (Syndros®). Sativex®, a THC/CBD combination prescription product, is a buccal spray indicated as adjunctive analgesia in cancer pain, muscle stiffness, and neuropathic pain in multiple sclerosis. It has been approved in Europe, Canada, and the United Kingdom, but not in the United States (US). 6
According to estimates, 3%–5% of the world's population has used cannabis at least once recently, predominantly for non‐medical purposes and outside the parameters of prescribed use. Furthermore, approximately one in eight Americans in the United States who use cannabis do so for medical purposes. 7
The pharmacokinetics of cannabis, particularly its absorption, distribution, metabolism, and excretion (ADME), play a crucial role in understanding its interactions with other drugs. THC and CBD are cannabinoids that bind to cannabinoid receptors of the endocannabinoid system throughout the body. The endocannabinoid system plays a major role in regulating brain, endocrine, organ, and immune function. THC is metabolized by CYP2C9 and CYP3A4 enzymes, while CBD is metabolized by CYP3A4 and CYP2C19 enzymes. These cannabinoids affect the metabolism of several prescription medicines through the cytochrome P450 enzyme system, potentially resulting in drug–drug interactions (DDI). THC is also a competitive inhibitor of CYP1A2, CYP2B6, CYP2C9, and CYP2D6 enzymes, while CBD competitively inhibits enzymes CYP3A4, CYP2B6, CY92C9, CYP2D6, and CYP2E1. 4
In recent years, several countries have been reviewing their policies and regulations on medical cannabis as global trends shift toward cannabis legalization. This has spurred an increase in research involving cannabis, particularly regarding DDI. As countries consider legalizing cannabis, there is a spectrum of potential benefits and implications that warrant careful examination. For instance, legalizing cannabis allows for its therapeutic and medical use in various conditions. However, safety concerns, potential for abuse, and unknown drug interactions are significant issues. Therefore, this systematic review evaluates existing literature to provide a summary of drug interactions involving medical cannabis. It includes information detailing the backgrounds of participants, patient outcomes, and recommendations to manage different cannabis–drug interactions.
Studies that have investigated DDIs involving medical cannabis are lacking, and the majority of evidence is in the form of case reports. Therefore, by performing this systematic review, we compiled a beneficial and comprehensive list of these DDIs. Based on this list, this systematic review aims to collate succinct evidence of cannabis–drug interactions for healthcare practitioners to be aware of. This review is anticipated to serve as a reliable guide that can be referenced when necessary to determine the risk and potential management measures for possible DDIs in future drug usage. Specifically, it presents a comprehensive overview of cannabis–drug interactions by incorporating probability and severity grading, patient outcomes, and feasible recommendations. A section is also dedicated to discussing the demographic characteristics of the studies. Including demographic information allows prescribers to better understand the population profile; it will help them evaluate the generalizability of the data to their patient population (26).
METHODS
The literature sources were obtained from the following electronic databases: PubMed, Scopus, clinicaltrials.gov, and Web of Science, from the inception of the study to October 1, 2023. No geographical restrictions were implemented. Additionally, we included only articles with full‐text accessibility, English‐language literature, clinical information on medical cannabis use in humans, and completed studies with published results. The exclusion criteria were irrelevant articles, articles consisting of non‐human evidence, articles without much‐supporting evidence (theoretical interactions), and articles without specific information about cannabis–drug interactions or review articles. The search strategy included free text terms and exploded MESH headings for [(“drug–drug interactions”) AND (“cannabis” OR “medicinal cannabis” OR “medical cannabis” OR “marijuana” OR “cannabinoids”)].
In the initial screening phase of approximately, all authors separately searched the literature from databases, and appropriate articles were found using their titles and abstracts. According to the agreed‐upon inclusion and exclusion criteria, all authors independently screened the full text of selected papers (Figure 1).
FIGURE 1.
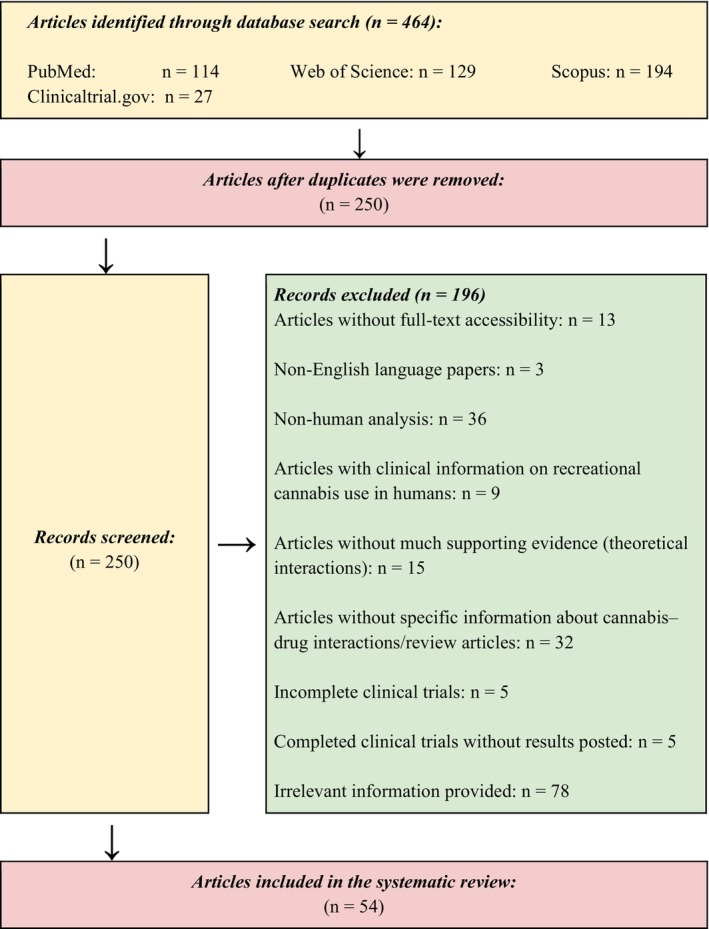
Preferred reporting items for Systematic Reviews and Meta‐Analyses (PRISMA): Flow diagram for the systematic review of cannabis–drug interaction.
Next, the collected data were organized based on study design, drugs involved, cannabis product assessed, interaction outcome, clinical relevance level (a combination of severity and probability of occurrence), mechanism of interaction, and recommendations to manage the interaction. All authors independently extracted data from eligible studies and then checked the data for clarity and consistency.
The probability was assessed and graded according to the type of supporting evidence available at the moment for each cannabis–drug interaction 4 :
Possible: The interaction was documented by results from less than three case reports.
Probable: The interaction was documented by results from at least one observational study (cohort or case–control study) or at least three case reports.
Defined: The interaction was documented by results from at least one meta‐analysis, narrative systematic review, or randomized or non‐randomized clinical trial.
The interaction severity was graded based on the degree of qualitative changes concerning drug safety or efficacy, measured through clinical parameters (Internationalized Normalized Ratio (INR), serum creatinine (SrCr), transaminases levels) or PK (plasma concentration, area under the curve (AUC)). 7 The changes were assessed by comparing the data available before and after the interaction. The following calculation was used to measure the changes in parameters associated with the safety or efficacy of the drug 4 :
where:
Par int: Parameter during the interaction (INR, SrCr, transaminases, plasma concentration, AUC).
Par: Parameter (INR, SrCr, transaminases, plasma concentration, AUC) before or after the interaction.
The severity grading was based on the degree of parameter changes. The severity grading would be irrelevant if the parameter changes did not meet the minimum 25%–100% range.
Minor (the interaction causes minimal harm to the patient): The parameter changes were between 25% and 100% (INR, SrCr, transaminases, plasma concentration, AUC).
Moderate (the interaction requires closer patient health monitoring): The parameter changes were between 100% and 400% (INR, SrCr, transaminases, plasma concentration, AUC).
Severe (the interaction causes harm or injury to the patient): The parameter changes were between 400% and more (INR, SrCr, transaminases, plasma concentration, AUC).
The system organ class (SOC) of the Medical Dictionary for Regulatory Affairs (MedDRA) was used to categorize the adverse effects reported in the studies.
RESULTS
We identified 464 articles from the database search. Out of these, 250 articles were selected for screening of titles and abstracts. After removing duplicates and checking for eligibility, we had 54 articles to include in the review (Figure 1).
Summary of drug classes involved in cannabis–drug interactions
A total of 20 drug classes were identified to have interactions with medicinal cannabis. The drugs involved were further categorized based on their indications (Table 1).
TABLE 1.
Summary of drug classes involved in cannabis–drug interactions.
| Drug classes | Number of studies | Probability | Severity | |||||
|---|---|---|---|---|---|---|---|---|
| Possible | Probable | Defined | Minor | Moderate | Severe | N/A | ||
| Antiepileptics | 14 | 2 | 12 | 3 | 2 | 9 | ||
| Benzodiazepines | 8 | 1 | 7 | 3 | 1 | 4 | ||
| Opioids | 4 | 4 | 1 | 3 | ||||
| Calcineurin inhibitors | 3 | 3 | 1 | 2 | ||||
| Anticoagulants | 2 | 2 | 2 | |||||
| Antiretrovirals | 2 | 2 | 2 | |||||
| Barbiturates | 2 | 2 | 1 | 1 | ||||
| Mechanistic target of rapamycin (mTOR) inhibitors | 2 | 2 | 1 | 1 | ||||
| Xanthines | 2 | 2 | 2 | |||||
| Alkylating agents | 1 | 1 | 1 | |||||
| Antibiotics | 1 | 1 | 1 | |||||
| Antifungals | 1 | 1 | 1 | |||||
| Proton pump inhibitors (PPIs) | 1 | 1 | 1 | |||||
| Selective serotonin reuptake inhibitors (SSRIs) | 1 | 1 | 1 | |||||
| Taxanes | 1 | 1 | 1 | |||||
| Topoisomerase I inhibitors | 1 | 1 | 1 | |||||
| Psychostimulants | 1 | 1 | 1 | |||||
| Cannabis (THC) | 1 | 1 | 1 | |||||
| Cardiovascular system treatments (not specified) | 1 | 1 | 1 | |||||
| Diuretics | 1 | 1 | 1 | |||||
| Laxatives | 1 | 1 | 1 | |||||
Interaction effects, outcomes, and recommendations
The following tables present a comprehensive overview of various drug interactions with medical cannabis. Notably, medical cannabis has the highest probability of interaction with antiepileptic drugs, specifically CLB. In contrast, medical cannabis has the lowest likelihood of interaction with alkylating agents, particularly temozolomide. Nonetheless, when combining medical cannabis and other medications of different drug classes, medical practitioners should carefully consider patient specifics, assess risks and benefits, and evaluate the clinical relevance of drug interactions before deciding on the necessary dose adjustments.
Analgesic drugs
TABLE 2.
Interaction between opioids and cannabis, based on case reports.
| Drugs | Probability | Severity | Observed effects | Patient outcomes | Recommendation | References |
|---|---|---|---|---|---|---|
| Opioids | ||||||
| Methadone (Victim—CYP3A4 and CYP2C19 inhibition) | Defined | Moderate (116.8%) | Discontinuing CBD oil led to an overall decline in the methadone serum levels. Specifically, the serum methadone level was noted to be 271 ng/mL (2 days after stopping CBD), followed by 149 ng/mL (7 days after stopping CBD) and 125 ng/mL (14 days after stopping CBD) |
A case report of a 13‐year‐old patient recorded symptoms of increased fatigue and somnolence, which worsened upon admission as the pupils were minimally reactive (~2 mm). Co‐administering methadone and CBD was believed to contribute to the adverse effects reported because an improvement in the symptoms occurred with CBD withdrawal |
Implement carer education for potential cannabis–drug interactions. Provide suggestions on other supportive therapies to manage the specific symptoms that arise during methadone treatment to avoid the use of CBD | 8 |
TABLE 3.
Interaction between opioids and cannabis, based on clinical trials.
| Drugs | Probability | Severity | Observed effects | Patient outcomes | Recommendation | References |
|---|---|---|---|---|---|---|
| Opioids | ||||||
| Fentanyl (n = 17) (Perpetrator—Unknown) | Defined | Not applicable |
Plasma CBD concentration was not significantly affected by fentanyl dosing (AUC Wilcoxon p > 0.05). Higher fentanyl dosing was associated with reduced CBD clearance (p = 0.02) In subjects on 400 mg of CBD, the mean peak urinary concentration of CBD conjugates (U max) for higher fentanyl dosing was 2 μg/L, as compared with 4.6 μg/L for lower fentanyl dosing In subjects on 800 mg of CBD, U max was 2.8 μg/L for the higher fentanyl dosing group and 3.7 μg/L for lower fentanyl dosing |
Adverse events reported were dizziness/drowsiness (n = 5), itching/rash (n = 3), abdominal discomfort (n = 2), diarrhea (n = 2), headache (n = 2), and nausea/vomiting (n = 2), but none were associated with peak concentration (C max) of CBD | Fentanyl is well tolerated and safe to be co‐administered | 9 |
| Morphine (n = 13) [Possible decreased gastrointestinal (GI) motility] | Defined | Not applicable |
Significant percentage changes in pain rating were reported (a decrease of 33.7%). C max values demonstrated a statistically significant decrease from 43.68 to 29.66 mg/mL (p = 0.003) No significant statistical change in 12‐h AUC (AUC12) was reported. THC plasma concentrations were not affected by morphine |
The analgesic effect was enhanced significantly. A subjective “high” was reported with the concomitant cannabis and morphine. All participants in the morphine/cannabis cohort (n = 10) experienced heightened stimulation and decreased hunger on Day 5 of the study | No recommendation provided | 10 |
| Oxycodone (n = 11) (Possible decreased GI motility) | Defined | Not applicable | The pain rating decreased by 21.3%. No significant changes in oxycodone kinetics were observed. THC plasma concentration was not affected by oxycodone | The analgesic effect was enhanced. A subjective “high” was reported with concomitant cannabis and oxycodone. All participants (n = 11) reported reduced anxiety on Day 5 | No recommendation provided | 10 |
Anti‐infective drugs
See Table 4.
TABLE 4.
Interaction between antibiotics, antifungals, antiretrovirals, and cannabis.
| Drugs | Probability | Severity | Observed effects | Patient outcomes | Recommendation | References |
|---|---|---|---|---|---|---|
| Antibiotics | ||||||
| Rifampicin (Perpetrator—CYP3A4 induction) | Defined | Minor (36%–87%) |
C max of THC, CBD, and THC metabolite in the form of 11‐hydroxy‐THC (11‐OH‐THC) with the use of THC/CBD oromucosal spray alone appeared to be 2.94 ng/mL, 1.03 ng/mL, and 3.38 ng/mL, respectively With the co‐administration of THC/CBD spray and rifampicin, C max of THC, CBD, and 11‐OH‐THC were reduced to 1.88 ng/mL, 0.50 ng/mL, and 0.45 ng/mL, respectively. Overall, C max reduced by 26–87% |
Adverse effects were insignificant. Headache was reported in a limited number of participants (n = 1) | Dose increments of THC/CBD spray may be needed when rifampicin is co‐administered to achieve a similar efficacy as using THC/CBD spray alone | 11 |
| Antifungals | ||||||
| Ketoconazole (Perpetrator—CYP3A4 inhibition) | Defined | Minor to moderate (27%–204%) |
Elevations in the C max of THC, CBD, and 11‐OH‐THC were seen in subjects receiving both THC/CBD oromucosal spray and ketoconazole C max of THC increased from 2.65 to 3.36 ng/mL, whereas the C max of CBD increased from 0.66 to 1.25 ng/mL. C max increased from 3.59 to 10.92 ng/mL for 11‐OH‐THC. Overall, 36%–87% of C max was increased |
The adverse effects reported mainly involved nervous system disorders (e.g., somnolence, dizziness, and malaise) and psychiatric disorders (e.g., euphoria, anxiety, and disorientation) | Reduction in the daily dose of THC/CBD spray may be required when it is co‐administered with ketoconazole. This will maintain the efficacy of THC/CBD spray and prevent the occurrence of adverse effects | 11 |
| Antiretrovirals | ||||||
| Indinavir (Victim—Possible CYP3A and CYP2C induction) | Possible | Not applicable | Decrease in C max of indinavir by 17.4% (n = 11) | PK changes were unlikely to impact antiretroviral efficacy. Long‐term consequences were likely to be negligible, especially with the increasing use of protease inhibitor (PI) boosters | Constant monitoring of serum indinavir levels is needed. Increase the frequency of follow‐ups to review treatment efficacy | 12 |
| Nelfinavir (Victim—Possible CYP3A and CYP2C induction) | Possible | Not applicable | Decrease in C max of nelfinavir by 14.1% (n = 14) | PK changes were unlikely to impact antiretroviral efficacy. Long‐term consequences were likely to be negligible, especially with the increasing use of PI boosters | Constant monitoring of serum nelfinavir levels is needed. Increase the frequency of follow‐ups to review treatment efficacy | 12 |
Cardiovascular and GI diseases
See Table 5.
TABLE 5.
Interaction between warfarin, PPIs, and cannabis.
| Drugs | Probability | Severity | Observed effects | Patient outcomes | Recommendation | References |
|---|---|---|---|---|---|---|
| Anticoagulants | ||||||
| Warfarin (Victim—CYP2C9 and CYP3A4 inhibition) | Probable | Not applicable | Not reported | A case report of a 67‐year‐old male showed adverse effects of mild dry mouth and transient dizziness. INR levels were measured at 5.2 with a home self‐test device 3 days prior. No bleeding complications were experienced | Routine home INR monitoring is indicated. Patients should receive education on the signs and symptoms of bleeding as well as the actions needed to mitigate bleeding complications | 13 |
| Probable | Not applicable | A non‐linear increase in INR was observed with the up‐titration of CBD oil. INR levels increased from 2.22 (Day 1) to 6.86 (Day 3), followed by a decrease to 4.40 (Day 4) after reducing warfarin dose. A continuous trend of decrease was observed with INR of 1.96 (Day 11) | No bleeding complications were identified in this case report of a 44‐year‐old male. Warfarin dose was reduced by 30% | Increased monitoring of INR levels is warranted during the initiation and up‐titration of CBD. Adjust warfarin dose to maintain INR within therapeutic range when necessary | 14 | |
| PPIs | ||||||
| Omeprazole (CYP2C19 inhibition) | Defined | Not applicable |
No significant difference in oral clearance (CL/F) was noted for THC/CBD spray + omeprazole as compared to THC/CBD spray alone AUC and C max differences were not significant and were affected by interindividual variability. Variation ranges (CV%) of AUC and C max% coefficient when the combination was given: 40%–67% for THC and 11‐OH‐THC; 43%–64% for CBD. CV% observed when THC/CBD spray was administered alone: 48%–69% for THC and 11‐OH‐THC; 74%–98% for CBD THC: No change of time to maximum plasma concentration (T max), similar plasma concentration, 11% decrease of half‐life (t 1/2) when taking omeprazole + THC/CBD spray in comparison with THC/CBD spray alone CBD: Marginally higher plasma concentration (not significant), 5% increase of t 1/2, 9% increase of T max when taking omeprazole + THC/CBD spray in comparison with THC/CBD spray alone 11‐OH‐THC: Marginally lower plasma concentration (not significant), 9% increase of t 1/2, 24% decrease of T max when taking omeprazole + THC/CBD spray in comparison with THC/CBD spray alone |
Adverse effects were mainly related to the nervous system. Dizziness (n = 3) was the most commonly reported symptom when combination treatment was given | No recommendation provided | 11 |
| Laxatives | ||||||
| Not specified | Probable | Not applicable | Signs of accumulation were not evident for THC, CBD, and their metabolites. This was confirmed by data analysis of blood sampling within a six‐month interval | Not reported | No recommendation provided | 15 |
Immunosuppressive drugs
See Table 6.
TABLE 6.
Interaction between calcineurin inhibitors, mTOR inhibitors, and cannabis.
| Drugs | Probability | Severity | Observed effects | Patient outcomes | Recommendation | References |
|---|---|---|---|---|---|---|
| Calcineurin inhibitors | ||||||
| Cyclosporine (CYP3A4 inhibition) | Defined | Not applicable | Stable cyclosporine levels; specific data was not reported |
Adverse effects were reported in the subjects from the cyclosporine cohort (n = 2). These included dry mouth, nausea, dizziness, drowsiness, and episodes of intermittent heat. Dizziness was reported in the female participant (n = 1) Optimal pain control was observed within the cyclosporine + CBD cohort |
Concomitant use of CBD and cyclosporine is well tolerated and can be administered together. Weekly follow‐up is advised for the first month of combination therapy, then fortnightly or monthly follow‐up as per the condition of the patient Optimal doses of CBD or cyclosporine should be titrated for each patient due to the probability of mild adverse effects |
16 |
| Tacrolimus (Victim—CYP3A4 inhibition) | Defined | Not applicable | An initial decrease in the tacrolimus level was noted (n = 1), which normalized after 1 week. Data on tacrolimus levels were not specified |
Notable adverse effects included persistent nausea (n = 1) as well as elevated tacrolimus and SrCr levels (n = 1). Doses of CBD and tacrolimus were then reduced in the respective cases. Other adverse effects reported by subjects in the tacrolimus cohort (n = 5) were dizziness, drowsiness, and a few episodes of intermittent heat Partial improvement of pain management was noted in the subjects (n = 4). There was no improvement in pain control when CBD was co‐administered in the remaining subject |
Concomitant use of CBD and tacrolimus is well tolerated and can be administered together. Weekly follow‐up is advised for the first month of combination therapy, then fortnightly or monthly follow‐up as per the patient's condition Optimal doses of CBD or tacrolimus should be titrated for each patient due to mild adverse effects |
16 |
| Defined | Minor to moderate (58.3%–241%) |
A case report of a 32‐year‐old female showed baseline tacrolimus levels of 3.9–8.4 ng/mL. On Day 164, the dose‐normalized tacrolimus level had increased by approximately three‐fold, resulting in a reading of 13.3 ng/mL SrCr level increased to 2.4 mg/dL (baseline was 1.2 mg/dL) on Day 124 of the study. Tacrolimus doses were withheld 7 days later with SrCr level decreased to 1.5 mg/dL. An increment of tacrolimus dose to maximum was trialed in which SrCr level increased on Day 282 |
Signs of tacrolimus toxicity, particularly a rise in SrCr levels | Close monitoring of tacrolimus trough levels is needed when CBD is added as an adjunctive treatment. Dose reduction is warranted when SrCr increases from baseline with close observation | 17 | |
| mTOR inhibitors | ||||||
| Everolimus (Victim—CYP3A4 inhibition) | Defined | Moderate (180%) | Median increase of everolimus level by 9.8 ng/mL as compared with baseline |
Doses of mTOR inhibitors were reduced due to the linear relationship between the drug levels and the risk of toxicity The main adverse effect reported was diarrhea (n = 10). Other adverse effects included abdominal pain, swelling ankle, drowsiness, worsening acne, worsening and severe mouth ulcers, sinusitis, and elevated levels of liver enzymes |
Close monitoring of several important parameters (e.g., mTOR inhibitors plasma levels, relevant parameters, and adverse effects) with particular care is warranted during the CBD initiation Dose reduction in CBD should be considered pre‐emptively when the patient is known to be prone to mTOR inhibitor toxicity |
18 |
| Sirolimus (Victim—CYP3A4 inhibition) | Minor (70%) | Median increase of sirolimus level by 5.1 ng/mL as compared with baseline | ||||
Neurological drugs
See Table 7.
TABLE 7.
Interaction between antiepileptics, barbiturates, benzodiazepines, SSRIs, and cannabis.
| Drugs | Probability | Severity | Observed effects | Patient outcomes | Recommendation | References |
|---|---|---|---|---|---|---|
| Antiepileptics | ||||||
| Brivaracetam (Victim—CYP2C19 inhibition) | Probable | Moderate (107%–280%) | An increase in brivaracetam levels by 107%–280% was reported (n = 7). The levels of brivaracetam were not available for the remaining three participants | Adverse effects were reported with no clear indication of brivaracetam being the causative agent | No recommendation provided | 19 |
| Moderate (95%–280%) | Brivaracetam levels increased by 95%–280% in patients receiving co‐medication of brivaracetam and CBD (n = 5) | Mild adverse effects (e.g., diarrhea and somnolence) were observed (n = 2), whereby brivaracetam dose reduction was warranted | Monitoring of brivaracetam levels should be performed when CBD is co‐administered | 19 | ||
| Eslicarbazepine (Victim—Unknown) | Defined | Not applicable |
Trough concentrations of eslicarbazepine were elevated in the adult subjects (n = 39). Pediatric patients were not involved in the eslicarbazepine treatment group The mean baseline level of CBD was 14.4 7.4 μg/mL. The first and second mean CBD levels were 16.8 7.9 μg/mL and 17.8 9.1 μg/mL respectively. The increase in CBD levels was statistically significant (p = 0.008) in the interpretation conducted for baseline and the first two serum eslicarbazepine levels |
Not reported | No recommendation provided | 20 |
| Not applicable | Co‐medication of eslicarbazepine and CBD showed an increase in the level of eslicarbazepine by 23% (n = 1). No changes were observed in the other patient | Adverse effects were reported with no clear indication of eslicarbazepine being the causative agent | No recommendation provided | 19 | ||
| Rufinamide (Victim—Unknown) | Defined | Not applicable |
The increase in trough concentrations of rufinamide was observed with the elevations of mean baseline levels (24.8 12.8 μg/mL) to mean first rufinamide level (25.6 13.6 μg/mL) and mean second rufinamide level (27.0 14.7 μg/mL) A dose reduction in rufinamide was noted in the pediatric arm (n = 1) with a mean second CBD level of 12.2 μg/mL |
Not reported | No recommendation provided | 20 |
| Stiripentol (Victim/perpetrator—CYP2C19 inhibition) | Defined | Minor (32.05%–57.56%) |
Bidirectional interactions were observed. A slight increment in stiripentol levels was noted. C max and area under the concentration‐time curve over the dosing interval (AUC tau) were elevated by 32.05% and 57.56%, respectively Minor reductions in the exposure of CBD major metabolites were shown, which included 7‐hydroxy‐cannabidiol (7‐OH‐CBD) (−29%) and 7‐carboxy‐cannabidiol (7‐COOH‐CBD) (−13%). However, CBD levels were not affected significantly |
Mild severity rashes were reported (n = 2). Dose titration was done over 3 days in one of the participants. Other adverse effects were menstrual discomfort (n = 1) and a sensation of being drunk (n = 2) | No recommendation provided | 21 |
| Minor (17%) | Co‐administration of stiripentol and CBD led to a slight increase in stiripentol levels. C max increased by 17%, whereas AUC tau increased by 30% |
Severe rashes were reported (n = 1), which resolved after the discontinuation of both CBD and stiripentol Increased alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels [without exceeding 3 times the upper limit of normal (ULN)] were also reported (n = 2). The raised liver enzymes resolved during the trial |
Close monitoring for adverse effects when stiripentol is administered concurrently with CBD | 22 | ||
| Topiramate (Possible CYP2C9 and CYP2C19 inhibition) | Defined | Not applicable |
In the analysis conducted for baseline and the first two serum topiramate levels, changes in topiramate levels in both adult and pediatric treatment groups were not significant The reported mean baseline level of topiramate was 10.3 5.9 μg/mL with elevated levels of 10.8 7.0 μg/mL for the mean first topiramate level and 11.3 8.3 μg/mL for the mean second topiramate level |
Not reported | No recommendation provided | 20 |
| Not applicable | No changes in the systemic levels of topiramate were observed | Adverse effects were reported with no clear indication of topiramate being the causative agent | No recommendation provided | 21 | ||
| Valproate (Victim—Unknown) | Defined | Not applicable | There were no significant changes in valproate levels in participants taking concomitant valproate and CBD. However, AST/ALT levels were significantly higher (p < 0.01). The mean AST and ALT in all participants co‐administered with valproate and CBD were 37.1 U/L and 35.3 U/L. In comparison, participants who were not taking valproate reported lower AST and ALT levels of 23.97 U/L and 23.7 U/L |
Valproate and CBD were discontinued in the pediatric arm (n = 4) due to elevated AST/ALT levels of >3 times ULN Discontinuation of valproate and CBD was noted as well in the adult arm (n = 1) as AST/ALT levels exceeded approximately two times ULN |
Strict monitoring of valproate levels and routine liver function test (LFT) for patients taking both valproate and CBD | 20 |
| Not applicable | PD interaction reported as AST/ALT levels were found to be >3 times ULN in the patients (n = 61), with the majority of them receiving concomitant valproate (n = 46). No PK interaction was observed | The valproate dose was reduced for most patients on both valproate and CBD. Valproate was discontinued in several patients (exact number unspecified), followed by re‐titration of CBD dose to baseline. This resolved the liver enzyme abnormalities | No recommendation provided | 23 | ||
| Not applicable | No changes were seen in the systemic levels of valproate in patients taking both valproate and CBD. AST/ALT levels were raised >3 times ULN (n = 6). There were no reports of drug‐induced liver injury due to the absence of bilirubin elevation >2 times ULN | The majority of the affected patients (n = 5) completed the trial without dose changes. Reduction in valproate dose and discontinuation of CBD were done in the remaining patient. AST/ALT levels returned to baseline in all patients | No recommendation provided | 24 | ||
| Not applicable | Elevations of AST/ALT >3 times ULN were reported (n = 28), with the majority of them on concomitant valproate (n = 22) | No reports of drug‐induced liver injury (ALT level elevation of >5 times ULN). The abnormalities of liver enzymes resolved spontaneously with treatment discontinuation or dose reduction in either CBD or valproate | No recommendation provided | 24 | ||
| Zonisamide (Victim—Possible CYP3A4 and N‐acetyltransferase inhibition) | Defined | Minor (72.09%) | Trough concentrations of zonisamide were assessed. The mean baseline CBD level was 17.2 12.2 μg/mL. Elevation in zonisamide levels was shown by the first mean CBD level of 19.3 13.0 μg/mL, followed by the mean second CBD levels of 17.2 9.3 μg/mL (dose unchanged). Dose reduction was required in the adult cohort (n = 1) with a mean second CBD level of 42.0 μg/mL | Not reported | No recommendation provided | 20 |
| Barbiturates | ||||||
| Phenobarbital (Victim—Unknown) | Defined | Minor (27%) | The phenobarbital plasma trough concentration increased from 43 to 55 mg/L, which coincided with CBD titration | The phenobarbital dose was reduced. CBD was discontinued after 6 months of treatment due to inefficacy. Intermittent vomiting was reported by the patient in the case study | No recommendation provided | 25 (Case no 20) |
| Defined | Not applicable | No significant changes in plasma phenobarbital levels were noted during co‐administration and dose titration of CBD | Not reported | No recommendation provided | 20 | |
| Benzodiazepines | ||||||
| CLB (Victim—CYP2C19 inhibition) | Defined | Minor (57%) | Increase in mean N‐desmethylclobazam (nCLB) concentrations (10%–526%) and nCLB:CLB ratios (43%–664%). No relevant changes in plasma exposure of CLB | Elevated nCLB may have contributed to the decreased seizure frequency and occurrence of adverse effects observed in patients (i.e., sedation) (n = 4) | Monitor nCLB levels and adverse effects when CBD is co‐administered | 24 |
| Defined | Minor (67.42%) | The nCLB serum levels increased linearly (p < 0.001) and CLB levels decreased linearly (p < 0.001) with rising CBD doses in both adult and pediatric groups | Sedation was the main adverse effect reported by the participants in both the adult (n = 6) and pediatric arms (n = 8). The adverse effect occurred at least once during the study, which resulted in a decrease in CLB dose in all cases | Consider adjusting the CLB dose when starting CBD in anticipation of the increase in nCLB levels and the potential association with an increased likelihood of sedation | 20 | |
| Defined | Minor (32.35%) | CLB mean levels increased by 60 ± 80% at Week 4. nCLB mean levels increased by 500 ± 300% during Week 4 | A > 50% decrease in seizures was reported in a majority of the participants (n = 9) | Monitor both CLB and nCLB levels when CBD is co‐administered | 3, 27 | |
| Defined | Not applicable |
CLB (victim) + CBD (perpetrator) subgroup: slight increase in CLB exposure with C max trough of 1.20 ng/mL and an increase in nCLB exposure, where mean C max increased by 3.39‐fold CBD (victim) + CLB (perpetrator) subgroup: slight increase in CBD exposure with C max trough of 1.34 ng/mL |
Adverse effects were reported in the CBD (victim) and CLB (perpetrator) subgroup: moderate severity of AV block first degree (n = 1) and severe rash without mucosal involvement in subjects (n = 2) | Closely monitor for adverse effects and consider CLB dose reduction when administered with CBD | 21 | |
| Defined | Not applicable |
Healthy volunteers group: small increase in exposure to steady‐state CLB (C max = 20%, AUC tau = 21%) and an increase in exposure to nCLB (C max = 3.4‐fold [239%], AUC tau = 3.4‐fold [238%]) Epilepsy volunteers group: No effects on exposure to CLB and an increase in exposure to nCLB (C max = 2.2‐fold [122%], AUC tau = 2.6‐fold [164%]) |
Concomitant use of CBD and CLB increased the incidence of somnolence in both groups | Consider reducing the CLB dose when CBD is co‐administered to reduce the risk of adverse reactions | 27 | |
| Define | Not applicable | Not reported | Pneumonia was reported in patients on CBD and CLB (n = 2) as well as the placebo group on CLB (n = 1). The CLB dose was decreased in 27% of patients | Monitor patients on concomitant CLB, and adjust doses when necessary to manage adverse effects | 28 | |
| Defined | Moderate (121.84%) | nCLB plasma concentrations increased from 2000 to 4500 ng/mL in the CBD group between Day 1 and Day 33 | More than one adverse effect was reported in patients taking CBD (n = 13) and patients taking placebo (n = 2) | Consider reducing the CLB dose when CBD is co‐administered if adverse reactions are experienced | 29 | |
| Probable | Not applicable | Concurrent use of CBD and CLB increased the risk of sedation in less than 4% of the patients. Additionally, increased alertness and improved verbal interactions were seen in 14% of patients in the CBD group and 8% of patients in the CBD + CLB group | Beneficial side effects were more substantial in the CBD monotherapy arm, as opposed to the CBD + CLB arm. However, this difference was not statistically significant | No recommendation provided | 30 | |
| SSRIs | ||||||
| Citalopram (Victim—CYP3A4 and CYP2C19 inhibition) | Possible | Not applicable | Citalopram plasma concentrations increased from baseline (42 ng/mL) to Week 8 (79 ng/mL) | Anxiety severity was reduced by 60%–83% across the 13‐week treatment (n = 5). Adverse events reported were mild | Consider therapeutic drug monitoring for citalopram or escitalopram levels when CBD is co‐administered | 31 |
| Psychostimulants | ||||||
| Methylphenidate (MPH) (Possible serine hydrolase inhibition) | Defined | Not applicable | The geometric mean ratios (GMRs) (90% CI) for the area under the plasma concentration‐time curve from time 0 to infinity (AUCinf) and C max for MPH monotherapy and CBD co‐administration were 1.09 (0.89, 1.32) and 1.08 (0.85, 1.37), respectively. The 90% CI of AUCinf and C max GMR were outside the range of 80%–125% DDI criteria. The median T max of CBD (4 h) was significantly longer than that of MPH (1.25 h) | One common adverse effect (nausea) was observed (n = 1). The subject received CBD during the 3‐day run‐in | No recommendation provided | 32 |
Oncological drugs, xanthines, and cannabis
See Table 8.
TABLE 8.
Interaction between alkylating agents, taxanes, topoisomerase I inhibitors, xanthines, and cannabis.
| Drugs | Probability | Severity | Observed effects | Patient outcomes | Recommendation | References |
|---|---|---|---|---|---|---|
| Alkylating agents | ||||||
| Temozolomide (Unknown) | Defined | N/A* | Neither temozolomide monotherapy nor temozolomide + cannabis therapy demonstrated any changes in C max, the area under the plasma concentration‐time curve to the last timepoint (AUC0−t ), and T max | Treatment‐emergent adverse events (TEAEs) were mostly related to the underlying medical condition of patients, such as glioblastoma | Condition‐based personalization of the nabiximol dose regimen should be done in patients receiving concurrent cannabis treatment and intensive temozolomide therapy | 33 |
| Taxanes | ||||||
| Docetaxel (CYP3A inhibition) | Defined | N/A* | Clearance and dose‐normalized AUC were not significantly affected by the concurrent use of cannabis. Plasma concentrations of docetaxel were similar in both monotherapy and combination therapy with cannabis |
Hematological toxicity, which is the major adverse effect of docetaxel, was not significantly noted with the concurrent administration of cannabis Other adverse events included elevated liver transaminases and bilirubin, fatigue, mild headaches, mood disturbance, and weird dreams. In addition, most patients noticed improved sleep quality |
Evaluated cannabis products can be administered together with docetaxel without any dose adjustments | 34 |
| Topoisomerase I inhibitors | ||||||
| Irinotecan (CYP3A inhibition) | Defined | N/A* |
Irinotecan: Dose‐normalized AUC and clearance were not significantly affected by cannabis co‐administration Irinotecan metabolites (SN‐38 and SN‐38‐glucuronide): Dose‐normalized AUC and clearance were not significantly altered when cannabis was co‐administered |
Similar nadir values of absolute neutrophil count (ANC) and white blood cell count (WBC) suggested that hematological toxicity (the major adverse effect) did not significantly occur with cannabis co‐administration Better sleep quality was reported by most patients, whereas some patients also reported mild headaches, mood disturbance, weird dreams, diarrhea, fatigue, nausea, and vomiting |
Evaluated cannabis products can be safely administered together with irinotecan without any dose adjustments | 34 |
| Xanthines | ||||||
| Caffeine (Victim—CYP1A2 inhibition) | Defined | Minor (caffeine: 95%; paraxanthine: 18%) |
Increments were observed in several parameters with CBD + caffeine co‐administration on Day 26, which included C max of caffeine (+15%), AUC0−t (+88%), and area under the concentration‐time curve from time zero to infinity (AUC0−∞) (+95%) Longer T max and t 1/2 were also noted when compared to a single administration of caffeine on Day 1 On Day 26, higher caffeine concentrations as well as reduced CL/F of CBD + caffeine were observed compared with Day 1 |
The most commonly reported adverse effect during CBD titration was diarrhea (n = 8) Laboratory results demonstrated elevated liver enzymes (ALT, AST, and gamma‐glutamyltransferase [GGT]) (n = 7) in subjects who were on either CBD maintenance dose or CBD + caffeine. Eosinophil percentage showed a significant rise during CBD maintenance dose (n = 1). Syncope was reported as well (n = 1) All reported elevations were resolved 9–49 days after onset, except for GGT elevations that were ongoing towards the end of the trial with 2.4 times ULN |
No recommendation provided | 35 |
| Cannabis | ||||||
| THC (Victim—Complex synergistic PK, antagonistic PD interactions) | Defined | N/A* | The mean THC peak plasma concentration of 82 ng/mL was reported in a product with a lower CBD concentration (<1 mg). In products with higher CBD concentrations of about 18 mg, THC plasma concentrations were at least 50% higher than those of lower CBD content products | Not reported | No recommendation provided | 36 |
| Diuretics | ||||||
| Not specified | Probable | N/A* | Signs of accumulation were not evident for THC, CBD, and their metabolites. This was confirmed by data analysis of blood sampling within a 6‐month interval | Not reported | No recommendation provided | 15 |
N/A implies that the severity grading is not applicable.
DISCUSSION
Co‐administering cannabis with any concomitant drug can potentially result in unilateral or bilateral drug interactions. Several mechanisms to explain the PK profiles of cannabis–drug interaction have been proposed by studies, which involve the pathways of CYP isoenzymes, UGTs, and certain transporters (e.g., P‐glycoprotein [P‐gp], breast cancer resistance protein [BCRP], and/or multi‐drug resistance‐associated protein 2 [MRP2]). Nonetheless, recent study by Cox et al. 37 revealed that neither UGTs‐ nor transporter‐mediated cannabis–drug interaction would be possible as it would require supraphysiological concentration for the systemic interaction to take place. Therefore, the evidence thus far suggests that cannabis–drug interaction is mediated via the induction or inhibition of CYP family isoenzymes, which results in the changes observed in plasma concentrations of drugs. Although existing evidence is still lacking, it is likely that there are other potential interaction targets including the efflux or uptake transporter pathway. Bardhi et al. 38 also revealed that CBD, THC, and their metabolites may exhibit reversible and time‐dependent inhibition of certain CYP enzymes as suggested by several in vitro studies. Additionally, CBD and THC are proposed to be involved in mixed‐type (competitive or non‐competitive) interaction with carboxylesterase 1 (CES1). Furthermore, PD changes associated with cannabis–drug interactions have been reported, whereby the pharmacological effect of one drug is altered by that of another drug in a combination regimen. 6 Undoubtedly, identifying these variations secondary to cannabis–drug interactions is vital as they can influence the safety and efficacy of medical cannabis or co‐administered drugs.
Out of the 20 drug classes that have been identified, most drug classes are believed to have a clear chance of interaction with medicinal cannabis. To illustrate, brivaracetam and warfarin have potential interactions with cannabis, whereas citalopram has a possible cannabis–drug interaction. Due to insufficient data, the severity grading of patient outcomes did not apply to all studies. Nevertheless, co‐administering CBD with tacrolimus was found to elevate SrCr levels significantly from the baseline; this indicates tacrolimus toxicity and could lead to more severe outcomes. Otherwise, the reported adverse effects of other drug classes were relatively minor and resolved with dose adjustments or treatment withdrawal.
Evidence
Antiepileptics
Antiepileptics are one of the drug classes that are primarily involved in DDIs with cannabis. Inconsistent results were noted for eslicarbazepine and topiramate. A trial done by Gaston et al. 20 revealed elevated eslicarbazepine levels in all adult patients, which were statistically significant (p = 0.008). Nonetheless, Klotz et al. 19 observed an increase in eslicarbazepine in only one out of two patients. It is believed that the discrepancy in outcomes may be caused by the use of different CBD formulations in the respective studies. As opposed to the study by Gaston et al. 20 which involved the use of CBD products containing sesame oil, the subjects in the study by Klotz et al. 19 were given MCT‐oil‐based synthetic formulation, which normally consists of palm or coconut oil. This minor difference in the medium administered might affect eslicarbazepine levels as sesamin (the major constituent in sesame oil) inhibits pregnane X receptor (PXR) activation. PXR is known for its function in regulating CYP3A4, glutathione S‐transferases, uridine diphosphate‐glucuronyltransferase, and sulfotransferase. This could have affected the observed results as eslicarbazepine is metabolized by glucuronidation. 39 The increased levels of rufinamide, which is metabolized by carboxyl esterase, could also be linked to the use of sesame oil in the CBD formulation. In contrast to Gaston et al., 20 Devinsky et al. 24 reported no elevation in topiramate levels; this is presumably due to the smaller sample size in the trial conducted by the latter.
In general, the interaction between antiepileptics and CBD increases antiepileptic drug levels, particularly for eslicarbazepine and brivaracetam. Although severe adverse effects were not reported in these interactions, it is recommended to monitor their plasma levels when they are co‐administered with CBD. Other notable antiepileptics that interact with CBD with antiepileptics are stiripentol and valproate. These interactions are especially crucial as they typically result in severe adverse effects, which include rashes and raised liver enzymes. Rashes were observed in subjects taking stiripentol with CBD. The severity was greatly influenced by interindividual variabilities and may warrant CBD dose reduction or withdrawal. Hence, clinicians should actively monitor for these adverse effects when patients are on both CBD and stiripentol. In addition, co‐administering CBD with stiripentol and valproate led to elevated liver enzymes (AST/ALT). Interestingly, liver enzyme elevation was observed even when valproate levels were unaffected. Some possible explanations for this PD interaction include the hepatotoxic profile of valproate and CBD, which results in a synergistic interaction between valproate and CBD. This underscores the importance of routine LFT analysis. Given the increased risk of liver damage, clinicians should consider reducing the doses of valproate/stiripentol and CBD when the levels are significantly high in reference to the published study cited. If the levels do not return to baseline, CBD or valproate may have to be discontinued due to their hepatotoxicity profile.
Anti‐infectives
An investigation by Stott et al. 11 discovered that rifampicin (a CYP3A4 inducer) decreased the concentrations of THC and CBD in 82%–100% of the participants. Consequently, patients are likely to receive suboptimal therapeutic effects from CBD, and the CBD dose needs to be up‐titrated. On the contrary, ketoconazole (a CYP3A4 inhibitor) increased THC and CBD concentrations by 63%–100% among the participants. Due to the increased risk of adverse effects such as somnolence and dizziness, the ketoconazole dose should be reduced when it is co‐administered with CBD.
PPIs
Although omeprazole has an inhibitory action on CYP2C19, the plasma levels of THC and CBD were not significantly affected by concurrent use of cannabis; this indicates that they are possibly not a substrate of CYP2C19. 11 Therefore, dose adjustment is not needed.
Tacrolimus
Tacrolimus, a calcineurin inhibitor, is commonly used to induce immunosuppression in patients who receive organ transplants to prevent rejection episodes. Inconsistencies have been noted in the interaction between tacrolimus and CBD. For example, Leino et al. 17 reported a nearly threefold increase in tacrolimus levels, whereas Cunetti et al. 16 demonstrated variable results: tacrolimus levels were shown to be elevated in two patients and reduced in two patients. One possible explanation is that tacrolimus metabolism is highly influenced by interindividual variability, which is further complicated by PK, PD, and the patient's medical condition. Hence, further studies on the interaction between cannabis and tacrolimus are needed for a more definitive conclusion.
Opioids
Manini et al. 9 found that IV fentanyl plasma concentrations were not strongly affected by cannabis when they were administered concurrently. Additionally, no adverse outcomes in the population have been noted. Thus, they can be safely administered together. Interestingly, the concurrent use of cannabis with morphine or oxycodone is known to enhance the analgesic effect of opioids without altering their kinetic parameters. Accordingly, there has been no conclusive evidence of cannabis‐oxycodone interaction. However, Abrams et al. 10 discovered that combining cannabis and morphine improves the analgesic effects of morphine. This is most likely because cannabis leads to delayed GI motility. Therefore, morphine would have a sustained release effect, and the C max of morphine would decrease. Unfortunately, this statement remains a theoretical presumption due to limited evidence available for validation. It is therefore imperative to conduct further research to establish the actual mechanisms behind these interactions.
Benzodiazepines
This review also identified significant evidence of a potential interaction between cannabis and benzodiazepines, particularly CLB. CBD has been demonstrated to inhibit the activity of CYP219 and CYP3A4, which are involved in the metabolism of nCLB, an active metabolite of CLB. As a result, CBD has a more significant impact on nCLB levels than CLB levels. This inhibition leads to the lengthening of the nCLB half‐life, resulting in its accumulation, heightened plasma exposure, and increased likelihood of adverse effects such as sedation. Several studies have confirmed this, such as those done by Geffrey et al., 26 Morrison et al., 21 Gaston et al., 20 Devinsky et al., 24 Patsalos et al., 27 and Vanlandingham et al.. 29 However, the side effects would gradually subside upon reducing the CLB dose. Consequently, it is advisable to monitor both CLB and nCLB levels in patients who are on both medical cannabis and CLB. Gaston et al. 20 provided a further recommendation to reduce the dose of CLB when starting CBD in anticipation of the rise of nCLB levels and the corresponding possibility of sedation.
Besides, the study by Porcari et al., 30 which assessed the efficacy of artisanal CBD preparations, also reported sedation as a common side effect of the concurrent use of CBD and CLB. Moreover, the study also found that using CBD alone led to better improvements in alertness and verbal interactions in seizure treatment, compared with the combined use of CBD and CLB. However, even though these benefits were more pronounced in the CBD monotherapy arm, the difference was not statistically significant. The effect of the use of artisanal products as opposed to the conventional pharmaceutical grade CBD products which follow a standardized preparation procedure should be systematically evaluated. Carvalho et al. 40 reported that CBD products prepared by artisanal techniques without pharmaceutical supervision demonstrated low CBD levels and high microbiological levels. Hence, the use of artisanal CBD in epilepsy management should be weighed prudently against the risks and benefits due to the inconsistencies of the quality of artisanal preparations in comparison to the medicinal CBD as well as the low additional benefits aforementioned.
Psychostimulants
Apart from the conventional CYP‐mediated interactions, several in vivo studies have also suggested that CBD inhibits other drug‐metabolizing enzymes in addition to the CYP isoenzymes. CES1, a serine hydrolase, is involved heavily (95%) in liver metabolism; it is involved in the deactivation and clearance of various drugs such as MPH. The clinical trial by Markowitz et al. 32 investigated the effects of CBD on the co‐administered MPH. Although the bioequivalence criteria of MPH varied between the MPH alone group and the MPH + CBD group, the changes in MPH exposure were minimal and had negligible clinical significance. Hence, it was concluded that the short‐term co‐ingestion of CBD and MPH at the doses evaluated led to minimal PK changes in MPH and insignificant DDIs. Nonetheless, the study could not assess the potential drug interactions at the steady state of CBD due to its relatively long half‐life. As such, the long‐term effects of co‐administering MPH with CBD are unclear and require further research.
SSRIs
Citalopram and escitalopram are among the most commonly prescribed antidepressants, and both drugs are categorized under the category of SSRIs. Citalopram consists of a racemic mixture of two enantiomers: S‐citalopram (escitalopram) and R‐citalopram. The metabolism of both citalopram and escitalopram primarily occurs through the CYP2C19 and CYP3A4 isoenzymes, both of which are inhibited by CBD. However, there are limited data regarding the potential interactions of CBD with citalopram or escitalopram. One study involving six participants demonstrated increased citalopram plasma concentrations when used concurrently with CBD. 31 Nonetheless, it is crucial to acknowledge that cannabis use can result in sedation and cognitive impairment, similar to the side effects associated with these antidepressants. In other words, taking SSRIs and medical cannabis concurrently may amplify these effects and increase the risk of cognitive decline. Therefore, more research on the interactions between CBD and citalopram or escitalopram is required.
Warfarin
Warfarin is an oral anticoagulant belonging to the class of vitamin K antagonists, and it is widely used to treat thromboembolic disorders. It is available in racemic mixtures of two enantiomers: S‐warfarin and R‐warfarin, with S‐warfarin being the more potent isomer. S‐warfarin is metabolized primarily by CYP2C9, while R‐warfarin is metabolized by CYP3A4. 41 CBD is a known potent inhibitor of CYP2C9 and CYP3A4 enzymes; therefore, concurrent administration of CBD and warfarin could potentially result in the accumulation of the latter. Furthermore, a study found elevated INR values with increasing CBD doses, implying a possible interaction between warfarin and CBD. In the same study, the INR values decreased after the warfarin dose was reduced. 14 Therefore, healthcare providers need to diligently monitor INR values during CBD initiation and up‐titration. Additionally, it is recommended to adjust the warfarin dose to ensure INR levels remain within the therapeutic range.
Laxatives and diuretics
Polypharmacy has become an increasingly common phenomenon in the aging population, and this practice is linked to a higher likelihood of DDIs. A recent study by Pautex et al. 15 looked into the feasibility and long‐term safety profile of CBD/THC as an off‐label treatment for patients with severe dementia, who are often polymedicated. Before the study, the participants were prescribed psychotropics, analgesics, laxatives, and diuretics. Out of all these, antipsychotics (typical and atypical), central analgesics, and antidepressants were affected by the introduction of medicinal cannabis. This is likely to be attributed to the potential inhibitory action of medicinal cannabis on the CYP1A2 and CYP2C19 enzymes, which would lead to drug accumulation. 15 For the other medications, there was no evidence indicating the accumulation of THC/CBD or their metabolites in blood sampling and phenotyping within 6 months of interval. Hence, significant cannabis–drug interactions with laxatives and diuretics were very unlikely. Nevertheless, the current evidence was not enough to substantiate a more comprehensive interaction profile for the medications in each drug class. In addition, based on individual observation, variations in dosage adjustments of medicinal cannabis are thought to influence the outcome of the interactions.
Antiretrovirals
The antiretrovirals indinavir and nelfinavir, commonly used in managing HIV infections, were also observed to have potential interaction with medical cannabis. Belonging to the PI class, these drugs are essential in halting viral replication and delaying the progression of HIV disease. One study demonstrated that adding smoked cannabis to a regimen containing indinavir and nelfinavir led to reduced PI plasma concentrations. Notably, the only statistically significant result was observed in the indinavir‐marijuana arm, with a 14% decrease in the median C max. The study's findings imply that the observed declines in indinavir and nelfinavir plasma concentrations may be associated with an induction of PI metabolism in the presence of elevated delta‐9‐THC plasma concentrations. While the specific enzymes responsible for this induction were not identified, in vitro evidence from existing literature predominantly suggests the inhibitory effects of cannabinoids on the cytochrome P450 enzyme system, particularly CYP3A and CYP2C subfamilies. Our study, however, revealed a potential induction of PI metabolism. Further research is warranted to elucidate the specific enzymes involved in this interaction and to reconcile our findings with the existing in vitro evidence on cannabis drug–drug interactions. 12
Caffeine
Thai et al. 35 demonstrated that cannabis has a major impact on the metabolism of caffeine; AUC0−t and AUC0−∞ increased to a large extent by 88% and 95%, respectively. Besides, the half‐life of caffeine also increased by 5.5 h and was associated with the CYP inhibition of cannabis. Cannabis inhibits CYP1A2, which caffeine is a substrate of. Therefore, if a large amount of caffeine is consumed with cannabis, its metabolism will be affected with an increased risk of side effects. Furthermore, one subject experienced a severe adverse effect of syncope when cannabis was administered together with caffeine. However, there is no evidence to explain the occurrence of syncope in that particular subject, suggesting that more studies are needed.
Cannabis (THC)
Interestingly, there has been evidence indicating a tendency for interaction to occur between the main components of cannabis, namely, CBD and THC. In a previous study, a higher concentration of CBD led to an increased mean peak plasma concentration of THC. There are several plausible explanations, such as CBD‐induced inhibition of THC metabolism, cyclizing of CBD into THC, and a potential CBD‐induced increase in the pulmonary uptake of THC due to the route of administering medicinal cannabis. 36 In addition, the study also reported possible PD antagonistic effects with the combination of CBD and THC due to the likelihood of negative modulatory action on CB1 cannabinoid receptors. Overall, the synergistic PK and antagonistic PD interactions between CBD and THC reflect the complex pharmacological behavior of medicinal cannabis in the human body. Therefore, further studies are needed to identify more definitive conclusions on the mechanisms and outcomes of the THC‐CBD interactions.
Challenges
Several challenges could have influenced the interpretation of the results. Firstly, this review mainly focuses on medicinal cannabis and its DDIs. Recreational cannabis was not considered due to several reasons. For instance, there is a wide variability in the type and dosage of recreational cannabis, and this would complicate the analysis of interactions with concomitant drugs. Next, the lack of a standardized dose regimen could influence the generalizability of the evidence to the larger population. The effects of cannabis–drug interaction on a patient's health will differ vastly depending on the type and quantity of CBD absorbed. Similarly, the type of cannabis formulation used in the studies is likely to affect the changes in PK and PD. For instance, the risk of other substitutions interfering with respiratory uptake and metabolism of co‐administered drugs is reduced with vaporized cannabis as opposed to marijuana cigarettes, even though both have the same concentrations of cannabis. 11 The study also mentioned that oral formulations of cannabis, such as capsules, will have a more extensive first‐pass metabolism profile in the liver. This would lead to a much lower plasma concentration of cannabis, and it may impact the metabolism of the respective drug. Thus, the influence of these factors should be considered while interpreting the outcomes of cannabis–drug interactions.
Limitations
This review has several limitations, and the findings should be carefully examined. As this study was only conducted over a few months, its short duration is the most significant limitation in this context. Besides, the reliability of the severity grading scale may be reduced by its overall generalizability across various unit measures (e.g., INR, SrCr, transaminases, plasma concentration, and AUC). The unique PK activities and disparate physiological effects exerted by each drug warrant the need of standardized laboratory parameters to quantify the changes over time for better clinical evaluation. In other words, the current severity grading scale is likely to be confounded by other health factors, which hinders direct quantitative comparison across different unit measurements. Hence, future researchers should focus on generating more robust grading tools to better analyze and represent the severity levels of cannabis–drug interactions. Methodological bias may have occurred due to the imprecise search strategy; the suboptimal inclusion and exclusion criteria could have led to relevant articles from all search results being excluded from the study.
CONCLUSION
The analysis of cannabis–drug interaction has become an important component in defining the safety profile of cannabis usage in treatment as medicinal cannabis continues to gain increasing attention from healthcare practitioners. In this systematic review, we identified 20 drug classes involved in the interaction with medicinal cannabis. The clinical significance of these interactions, particularly those involving benzodiazepines and warfarin, has been recognized and evaluated. Other potential cannabis–drug interactions have been documented, despite only limited literature existing. It is evidenced that CBD exhibits a broader spectrum of interactions beyond CYP enzymes including notable CBD's inhibitory effect on CES1 that affects drugs like MPH. This comprehensive summary underlines the multifaceted nature of CBD interactions and the importance of dose adjustments and drug level monitoring to ensure the optimal therapeutic use of cannabis. Given the large number of existing medications and the advent of new drugs, it is critical to conduct well‐designed studies to explore and identify any potential interactions with cannabis, as well as the mechanisms behind these interactions to extend the evidences in relation to the cannabis–drug interactions. Further research is also needed to enhance the current data on the association between different cannabis doses and formulations and the development of a practical severity rating scale for the purpose of analyzing the significant degree of DDIs.
AUTHOR CONTRIBUTIONS
J.J.Y.H., C.G., and C.S.A.L. wrote the manuscript, performed the research, and analyzed the data. A.B. and K.Y.N. designed the research.
ACKNOWLEDGEMENTS
Open access publishing facilitated by Monash University, as part of the Wiley ‐ Monash University agreement via the Council of Australian University Librarians.
FUNDING INFORMATION
The project was funded by the Ministry of Higher Education Malaysia (FRGS/1/2021/SKK03/MUSM/03/1).
CONFLICT OF INTEREST STATEMENT
The authors declared no competing interests for this work.
Supporting information
Table S1.
Ho JJY, Goh C, Leong CSA, Ng KY, Bakhtiar A. Evaluation of potential drug–drug interactions with medical cannabis. Clin Transl Sci. 2024;17:e13812. doi: 10.1111/cts.13812
REFERENCES
- 1. Chandra S, Lata H, ElSohly MA. Cannabis Sativa L. ‐ Botany and Biotechnology. Springer International Publishing; 2017. [Google Scholar]
- 2. Andre CM, Hausman JF, Guerriero G. Cannabis sativa: the Plant of the Thousand and one Molecules. Front Plant Sci. 2016;7:19. doi: 10.3389/fpls.2016.00019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Pellati F, Borgonetti V, Brighenti V, Biagi M, Benvenuti S, Corsi L. Cannabis sativa L. and nonpsychoactive cannabinoids: their chemistry and role against oxidative stress, inflammation, and cancer. Biomed Res Int. 2018;2018:1691428. doi: 10.1155/2018/1691428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lopera V, Rodríguez A, Amariles P. Clinical relevance of drug interactions with cannabis: a systematic review. J Clin Med. 2022;11(5):1154. doi: 10.3390/jcm11051154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Balachandran P, Elsohly M, Hill KP. Cannabidiol interactions with medications, illicit substances, and alcohol: a comprehensive review. J Gen Intern Med. 2021;36(7):2074‐2084. doi: 10.1007/s11606-020-06504-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Brown JD. Potential adverse drug events with tetrahydrocannabinol (THC) due to drug–drug interactions. J Clin Med. 2020;9(4):919. doi: 10.3390/jcm9040919 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lin LA, Ilgen MA, Jannausch M, Bohnert KM. Comparing adults who use cannabis medically with those who use recreationally: results from a national sample. Addict Behav. 2016;61:99‐103. doi: 10.1016/j.addbeh.2016.05.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Madden K, Tanco K, Bruera E. Clinically significant drug‐drug interaction between methadone and Cannabidiol. Pediatrics. 2020;145(6):e20193256. doi: 10.1542/peds.2019-3256 [DOI] [PubMed] [Google Scholar]
- 9. Manini AF, Yiannoulos G, Bergamaschi MM, et al. Safety and pharmacokinetics of Oral Cannabidiol when administered concomitantly with intravenous fentanyl in humans. J Addict Med. 2015;9(3):204‐210. doi: 10.1097/ADM.0000000000000118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Abrams DI, Couey P, Shade SB, Kelly ME, Benowitz NL. Cannabinoid–opioid interaction in chronic pain. Clin Pharmacol Therap. 2011;90(6):844‐851. doi: 10.1038/clpt.2011.188 [DOI] [PubMed] [Google Scholar]
- 11. Stott C, White L, Wright S, Wilbraham D, Guy G. A phase I, open‐label, randomized, crossover study in three parallel groups to evaluate the effect of rifampicin, ketoconazole, and omeprazole on the pharmacokinetics of THC/CBD oromucosal spray in healthy volunteers. Springerplus. 2013;2(1):236. doi: 10.1186/2193-1801-2-236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kosel BW, Aweeka FT, Benowitz NL, et al. The effects of cannabinoids on the pharmacokinetics of indinavir and nelfinavir. AIDS. 2002;16(4):543‐550. [DOI] [PubMed] [Google Scholar]
- 13. Brown GW, Bellnier TJ, Janda M, Miskowitz K. Δ‐9‐tetrahydrocannabinol dose increase leads to warfarin drug interaction and elevated INR. J Am Pharm Assoc. 2021;61(1):e57‐e60. doi: 10.1016/j.japh.2020.07.028 [DOI] [PubMed] [Google Scholar]
- 14. Grayson L, Vines B, Nichol K, Szaflarski JP. An interaction between warfarin and cannabidiol, a case report. Epilepsy Behav Case Rep. 2018;9:10‐11. doi: 10.1016/j.ebcr.2017.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Pautex S, Bianchi F, Daali Y, et al. Cannabinoids for behavioral symptoms in severe dementia: safety and feasibility in a long‐term pilot observational study in nineteen patients. Front Aging Neurosci. 2022;14:957665. doi: 10.3389/fnagi.2022.957665 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cuñetti L, Manzo L, Peyraube R, Arnaiz J, Curi L, Orihuela S. Chronic pain treatment with Cannabidiol in kidney transplant patients in Uruguay. Transplant Proc. 2018;50(2):461‐464. doi: 10.1016/j.transproceed.2017.12.042 [DOI] [PubMed] [Google Scholar]
- 17. Leino AD, Emoto C, Fukuda T, Privitera M, Vinks AA, Alloway RR. Evidence of a clinically significant drug‐drug interaction between cannabidiol and tacrolimus. Am J Transplant. 2019;19(10):2944‐2948. doi: 10.1111/ajt.15398 [DOI] [PubMed] [Google Scholar]
- 18. Ebrahimi‐Fakhari D, Agricola KD, Tudor C, Krueger D, Franz DN. Cannabidiol elevates mechanistic target of rapamycin inhibitor levels in patients with tuberous sclerosis complex. Pediatr Neurol. 2020;105:59‐61. doi: 10.1016/j.pediatrneurol.2019.11.017 [DOI] [PubMed] [Google Scholar]
- 19. Klotz KA, Grob D, Hirsch M, Metternich B, Schulze‐Bonhage A, Jacobs J. Efficacy and tolerance of synthetic Cannabidiol for treatment of drug resistant epilepsy. Front Neurol. 2019;10:1313. doi: 10.3389/fneur.2019.01313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Gaston TE, Bebin EM, Cutter GR, Liu Y, Szaflarski JP. Interactions between cannabidiol and commonly used antiepileptic drugs. Epilepsia. 2017;58(9):1586‐1592. doi: 10.1111/epi.13852 [DOI] [PubMed] [Google Scholar]
- 21. Morrison G, Crockett J, Blakey G, Sommerville K. A phase 1, open‐label, pharmacokinetic trial to investigate possible drug‐drug interactions between Clobazam, Stiripentol, or valproate and Cannabidiol in healthy subjects. Clin Pharmacol Drug Dev. 2019;8(8):1009‐1031. doi: 10.1002/cpdd.665 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ben‐Menachem E, Gunning B, Arenas Cabrera CM, et al. A phase II randomized trial to explore the potential for pharmacokinetic drug–drug interactions with Stiripentol or valproate when combined with Cannabidiol in patients with epilepsy. CNS Drugs. 2020;34(6):661‐672. doi: 10.1007/s40263-020-00726-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Szaflarski JP, Bebin EM, Comi AM, et al. Long‐term safety and treatment effects of cannabidiol in children and adults with treatment‐resistant epilepsies: expanded access program results. Epilepsia. 2018;59(8):1540‐1548. doi: 10.1111/epi.14477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Devinsky O, Patel AD, Thiele EA, et al. Randomized, dose‐ranging safety trial of cannabidiol in Dravet syndrome. Neurology. 2018;90(14):e1204‐11. doi: 10.1212/WNL.0000000000005254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Sands TT, Rahdari S, Oldham MS, Caminha Nunes E, Tilton N, Cilio MR. Long‐term safety, tolerability, and efficacy of Cannabidiol in children with refractory epilepsy: results from an expanded access program in the US. CNS Drugs. 2019;33(1):47‐60. doi: 10.1007/s40263-018-0589-2 [DOI] [PubMed] [Google Scholar]
- 26. Geffrey AL, Pollack SF, Bruno PL, Thiele EA. Drug‐drug interaction between clobazam and cannabidiol in children with refractory epilepsy. Epilepsia. 2015;56(8):1246‐1251. doi: 10.1111/epi.13060 [DOI] [PubMed] [Google Scholar]
- 27. Patsalos PN, Szaflarski JP, Gidal B, VanLandingham K, Critchley D, Morrison G. Clinical implications of trials investigating drug‐drug interactions between cannabidiol and enzyme inducers or inhibitors or common antiseizure drugs. Epilepsia. 2020;61(9):1854‐1858. doi: 10.1111/epi.16674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Thiele EA, Marsh ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox‐Gastaut syndrome (GWPCARE4): a randomised, double‐blind, placebo‐controlled phase 3 trial. Lancet. 2018;391(10125):1085‐1096. doi: 10.1016/S0140-6736(18)30136-3 [DOI] [PubMed] [Google Scholar]
- 29. VanLandingham KE, Crockett J, Taylor L, Morrison G. A phase 2, double‐blind, placebo‐controlled trial to investigate potential drug‐drug interactions between Cannabidiol and Clobazam. J Clin Pharmacol. 2020;60(10):1304‐1313. doi: 10.1002/jcph.1634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Porcari GS, Fu C, Doll ED, Carter EG, Carson RP. Efficacy of artisanal preparations of cannabidiol for the treatment of epilepsy: practical experiences in a tertiary medical center. Epilepsy Behav. 2018;80:240‐246. doi: 10.1016/j.yebeh.2018.01.026 [DOI] [PubMed] [Google Scholar]
- 31. Anderson LL, Doohan PT, Oldfield L, et al. Citalopram and Cannabidiol. J Clin Psychopharmacol. 2021;41(5):525‐533. doi: 10.1097/JCP.0000000000001427 [DOI] [PubMed] [Google Scholar]
- 32. Markowitz JS, De Faria L, Zhang Q, et al. The influence of Cannabidiol on the pharmacokinetics of methylphenidate in healthy subjects. Med Cannabis Cannabinoids. 2022;5(1):199‐206. doi: 10.1159/000527189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Twelves C, Sabel M, Checketts D, et al. A phase 1b randomised, placebo‐controlled trial of nabiximols cannabinoid oromucosal spray with temozolomide in patients with recurrent glioblastoma. Br J Cancer. 2021;124(8):1379‐1387. doi: 10.1038/s41416-021-01259-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Engels FK, de Jong FA, Sparreboom A, et al. Medicinal cannabis does not influence the clinical pharmacokinetics of Irinotecan and docetaxel. Oncologist. 2007;12(3):291‐300. doi: 10.1634/theoncologist.12-3-291 [DOI] [PubMed] [Google Scholar]
- 35. Thai C, Tayo B, Critchley D. A phase 1 open‐label, fixed‐sequence pharmacokinetic drug interaction trial to investigate the effect of Cannabidiol on the CYP1A2 probe caffeine in healthy subjects. Clin Pharmacol Drug Dev. 2021;10(11):1279‐1289. doi: 10.1002/cpdd.950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. van de Donk T, Niesters M, Kowal MA, Olofsen E, Dahan A, van Velzen M. An experimental randomized study on the analgesic effects of pharmaceutical‐grade cannabis in chronic pain patients with fibromyalgia. Pain. 2019;160(4):860‐869. doi: 10.1097/j.pain.0000000000001464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Cox EJ, Maharao N, Patilea‐Vrana G, et al. A marijuana‐drug interaction primer: precipitants, pharmacology, and pharmacokinetics. Pharmacol Ther. 2019;201:25‐38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Bardhi K, Coates S, Watson CJ, Lazarus P. Cannabinoids and drug metabolizing enzymes: potential for drug‐drug interactions and implications for drug safety and efficacy. Expert Rev Clin Pharmacol. 2022;15(12):1443‐1460. [DOI] [PubMed] [Google Scholar]
- 39. Lim YP, Ma CY, Liu CL, et al. Sesamin: a naturally occurring Lignan inhibits CYP3A4 by antagonizing the Pregnane X receptor activation. Evid Based Complement Alternat Med. 2012;2012:242810. doi: 10.1155/2012/242810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Carvalho VM, dos Santos CJ, dos Santos LMG, et al. Pharmaceutical evaluation of medical cannabis extracts prepared by artisanal and laboratory techniques. Revista Brasileira de Farmaco. 2023;33:724–735. [Google Scholar]
- 41. Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):160S‐198S. doi: 10.1378/chest.08-0670 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1.


