Dear Editor,
Deletions of the short arm of chromosome 18 encompassing the GNAL gene are associated with 18p deletion syndrome; the clinical features of this syndrome include facial dysmorphism, short stature, mental retardation, and movement disorders, particularly dystonia [1]. Heterozygous loss-of-function variants of GNAL are known to cause adult-onset cranio-cervical dystonia and/or asymmetric dystonic tremor [2]. GNAL-related dystonia (DYT-GNAL) exhibits an autosomal dominant (AD) inheritance pattern with incomplete penetrance and variable phenotypic expressivity. A limited number of cases of DYT-GNAL have been reported thus far; therefore, its clinical spectrum has yet to be fully explored [2]. In this case report, we describe a patient with dystonia-parkinsonism and evidence of nigrostriatal denervation carrying a chromosome 18p deletion encompassing the entire GNAL gene.
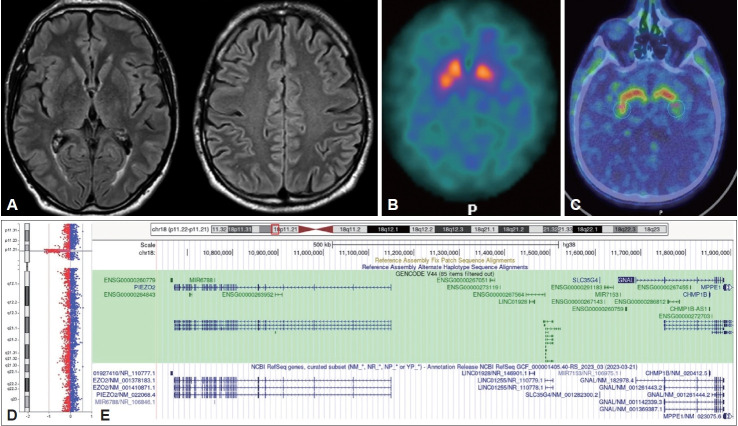
A 55-year-old man reported slowness of movement and rigidity of the right side of the body with abnormal posture of the right upper limb, which appeared simultaneously a few months previously. Neurological examination revealed rightsided hemiparkinsonism with concomitant marked dystonic posture of the right upper limb, right lower limb dystonia with slight eversion of the foot, forward trunk flexion, hypomimia and reduced right arm swing during gait (Supplementary Video 1 in the online-only Data Supplement). His past medical history was unremarkable; in particular, no history of a decreased sense of smell, depression or psychiatric disorders, autonomic dysfunction, sleep disturbances, or gastrointestinal symptoms was reported. There was no family history of Parkinson’s disease (PD) or other movement disorders. Brain magnetic resonance imaging did not reveal any significant alterations (Figure 1A), while ioflupane [123I] SPECT imaging revealed a moderate reduction in presynaptic dopaminergic uptake in the left caudate nucleus and bilateral putamen (Figure 1B). [18F] Fluorodopa PET confirmed nigrostriatal degeneration, as evidenced by reduced uptake in both putamina, which was more apparent on the left side (Figure 1C). Treatment with levodopa/ benserazide (100 mg three times a day) was started and had a subjective benefit on motor symptoms (i.e., rigidity, bradykinesia and gait). An acute levodopa challenge was administered, which slightly improved the Unified Parkinson’s Disease Rating Scale (UPDRS-III) motor subscore, particularly for items evaluating rigidity (namely, neck, right upper and lower extremities, and left upper and lower extremities) (off-med UPDRS = 25 points vs. on-med UPDRS = 19 points; improvement of 24%). Conversely, dystonia did not improve after levodopa administration. Moreover, evaluations of blood pressure and heart rate in patient with clinostatism and orthostatism (at the first and third minutes) in both the off- and on-med states did not reveal changes consistent with orthostatic hypotension. Perceptual-acoustic speech analysis was performed with the free beta version of Dysarthria Analyzer software in both the off-med and on-med conditions [3] and revealed a dysarthric pattern characterized by disfluencies, hypoarticulation of consonants and consonant clusters and excessive vowel prolongation, which did not change significantly after levodopa intake. Gait instrumental analysis [4] without medication (off-med condition) revealed a spontaneous walking speed of 53% height/second (h/s) further reduced to 40% h/s during fast walking because of increased rigidity. After levodopa intake (on-med condition), at the request for fast walking, the patient’s speed successfully increased to 72% h/s. Neuropsychological assessment revealed slight deficits in critical thinking, short-term verbal memory and bradyphrenia. The absence of nonmotor PD symptoms, the suboptimal response to levodopa and the disproportionate presence of dystonia prompted clinicians to consider alternative diagnoses to idiopathic PD and to perform genetic analyses within the Rostock International Parkinson Disease (ROPAD) study [5]. Genetic testing for PD and dystonia through an NGS gene panel [5] was performed, revealing a heterozygous 1.3 Mb genomic deletion, including the GNAL gene (NM_182978: exons 1–12). Array-CGH analysis confirmed the presence of a large chromosome 18 deletion (hg19, chr18: 10633666-11904117) involving four genes: PIEZO2, GNAL, CHMP1B, and MPPE1 (Figure 1D, E). None of these genes, except for GNAL, are known to cause AD disease when deleted.
Figure 1.
Brain MRI, dopaminergic imaging and description of genetic findings. A: Brain MRI did not show any significant alterations. B: Ioflupane [123I] SPECT imaging revealed a moderate reduction in presynaptic dopaminergic uptake in the left caudate nucleus and bilateral putamen. C: [18F] Fluorodopa PET confirmed nigrostriatal degeneration, showing reduced uptake in both putamina, which was more apparent on the left side. D: Graphical representation of the array-CGH results revealing the chromosome 18 deletion (hg19, chr18:10633666-11904117). E: Snapshot of the deleted region on chromosome 18 using the UCSC genome browser (www.genome.ucsc.edu). In addition to complete deletion of the GNAL gene, the PIEZO2 gene (left side) and CHMP1 gene were also completely deleted, while the MPPE1 gene was partially deleted. MRI, magnetic resonance imaging; SPECT, single-photon emission computed tomography; PET, positron emission tomography.
We reported a case of adult-onset dystonia-parkinsonism with nigrostriatal denervation in a patient carrying a chromosome 18p deletion encompassing GNAL. To our knowledge, degenerative parkinsonism has never been reported as a clinical manifestation of 18p deletion syndrome. Considering that no candidate PD risk genes (such as NDUFV2 [6]) were included in the deletion and that none of the deleted genes, except for GNAL, are known to cause AD diseases when deleted, we speculated that GNAL could play a significant role not only in dystonia but also in parkinsonism. GNAL encodes the alpha subunit of the heterotrimeric guanine nucleotide-binding protein [Gα(olf)]. Gα(olf) is particularly enriched in striatal medium spiny neurons and couples dopamine type 1 receptors in the direct pathway and adenosine A2A receptors in the indirect pathway for activation of adenylate cyclase type 5. Therefore, Gα(olf) plays a key role in siGNAL transduction within the basal ganglia [2]. Interestingly, Gα(olf) was found to be expressed in the dopaminergic neurons of the substantia nigra in rats and was reported to be downregulated in the postmortem brains of PD patients [7]. These preliminary observations provide a possible pathogenetic link between GNAL impairment and dopaminergic dysfunction. This unusual case may expand the clinical and pathological features associated with GNAL mutations to include dystonia-parkinsonism and dopaminergic denervation. However, a casual rather than causal co-occurrence of PD and dystonia in our patient cannot be excluded at present, and additional evidence is needed to link parkinsonism with 18p deletions. In conclusion, we described a case of dystonia-parkinsonism with nigrostriatal denervation carrying a chromosome 18p deletion encompassing GNAL, prompting further investigation of the possible link between this gene and adult-onset dystonia-parkinsonism.
Acknowledgments
None
Footnotes
Ethics Statement
The authors confirm that the approval of an institutional review board was not required for this work. Written informed consent was obtained from the patient to be videoed for publication.
Conflicts of Interest
The authors have no financial conflicts of interest.
Funding Statement
This study was partially supported by Italian Ministry of Health – Ricerca Corrente Annual Program 2023.
Author Contributions
Conceptualization: Giulia Di Rauso, Francesco Cavallieri, Franco Valzania. Data curation: all authors. Investigation: all authors. Supervision: Alessio Di Fonzo, Franco Valzania. Writing—original draft: Giulia Di Rauso, Francesco Cavallieri, Franco Valzania. Writing—review & editing: all authors.
Supplementary Material
The online-only Data Supplement is available with this article at https://doi.org/10.14802/jmd.23222.
Neurological examination revealed right-sided hemiparkinsonism with concomitant marked dystonic posture of the right upper limb, right lower limb dystonia with slight eversion of the foot, forward trunk flexion, hypomimia and reduced right arm swing during walking. The video was recorded before levodopa administration.
REFERENCES
- 1.Crosiers D, Blaumeiser B, Van Goethem G. Spectrum of movement disorders in 18p deletion syndrome. Mov Disord Clin Pract. 2019;6:70–73. doi: 10.1002/mdc3.12707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Carecchio M, Panteghini C, Reale C, Barzaghi C, Monti V, Romito L, et al. Novel GNAL mutation with intra-familial clinical heterogeneity: expanding the phenotype. Parkinsonism Relat Disord. 2016;23:66–71. doi: 10.1016/j.parkreldis.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 3. Hlavnička J, Růžičková H, Tykalová T, Novotny M, Rusz J. Dysarthria analyzer. Beta version (computer program) [Internet]. Strakonice: Dysarthria analyzer, 2022 [accessed on 2023 Apr 20]. Available at: http://www.dysan.cz/
- 4.Cavallieri F, Campanini I, Gessani A, Budriesi C, Fioravanti V, Di Rauso G, et al. Long-term effects of bilateral subthalamic nucleus deep brain stimulation on gait disorders in Parkinson’s disease: a clinical-instrumental study. J Neurol. 2023;270:4342–4353. doi: 10.1007/s00415-023-11780-5. [DOI] [PubMed] [Google Scholar]
- 5.Skrahina V, Gaber H, Vollstedt EJ, Förster TM, Usnich T, Curado F, et al. The Rostock International Parkinson’s Disease (ROPAD) study: protocol and initial findings. Mov Disord. 2021;36:1005–1010. doi: 10.1002/mds.28416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Esposito F, Addor MC, Humm AM, Vingerhoets F, Wider C. GNAL deletion as a probable cause of dystonia in a patient with the 18p- syndrome. Parkinsonism Relat Disord. 2014;20:351–352. doi: 10.1016/j.parkreldis.2013.12.005. [DOI] [PubMed] [Google Scholar]
- 7.Grison A, Zucchelli S, Urzì A, Zamparo I, Lazarevic D, Pascarella G, et al. Mesencephalic dopaminergic neurons express a repertoire of olfactory receptors and respond to odorant-like molecules. BMC Genomics. 2014;15:729. doi: 10.1186/1471-2164-15-729. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Neurological examination revealed right-sided hemiparkinsonism with concomitant marked dystonic posture of the right upper limb, right lower limb dystonia with slight eversion of the foot, forward trunk flexion, hypomimia and reduced right arm swing during walking. The video was recorded before levodopa administration.