Abstract
As a pluripotent cell, activated pancreatic stellate cells (PSCs) can differentiate into various pancreatic parenchymal cells and participate in the secretion of extracellular matrix and the repair of pancreatic damage. Additionally, PSCs characteristics allow them to contribute to pancreatic inflammation and carcinogenesis. Moreover, a detailed study of the pathogenesis of activated PSCs in pancreatic disease can offer promise for the development of innovative therapeutic strategies and improved patient prognoses. Therefore, the present study review aimed to examine the involvement of activated PSCs in pancreatic diseases and elucidate the underlying mechanisms to provide a viable therapeutic strategy for the management of pancreas-related diseases.
Keywords: PSCs, fibrosis, pancreatitis, cancer, treatment
1. Introduction
Pancreatic fibrosis is an important pathological feature of pancreas-related diseases and a key component of pancreatic injury repair, in which pancreatic stellate cells (PSCs) play a major regulatory role (1). As mesenchymal-like cells of non-endocrine nature in the pancreas, PSCs usually accumulate in the basal part of the pancreas and the lobules of the pancreas, accounting for about 4–7% of the total number of pancreatic cells (2). PSCs exist in two distinct states, namely resting and active states (3). Resting PSCs maintain their quiescent state by secreting nestin, glial fibers, acidic proteins, and waveform proteins, which play a prominent role in maintaining the normal function of the follicles and stabilizing the pancreatic pressure. However, when stimulated by various damaging factors, including trauma, inflammation, viral infection and cancer invasion, PSCs in the resting state will be converted into activated state, the myofibroblast-like phenotype. Subsequently, a significant increase in the cell volume accompanied by active proliferation, and secretion of various extracellular matrix (ECM) components, including collagen, fibronectin and laminin, is frequently observed. Moreover, various cytokines, including interleukin (IL)-1, IL-6, tumor necrosis factor-α (TNF-α), and transforming growth factor β1 (TGFβ1), chemokines and adhesion molecules are produced, promoting the chemotaxis, aggregation and adhesion of inflammatory cells, and ultimately participating in the repair and remodeling of the pancreas and pancreas-related diseases' progression (4–6). In addition, PSCs also possess stem cell characteristics and participate in the process of pancreatic tissue regeneration (7). However, persistent over-activation of PSCs will continue to synthesize and produce a large number of ECM components, thus disturbing the dynamic balance of collagen metabolism and inducing pancreatic fibrosis, destroying the structure and function of normal tissues, and ultimately leads to the development of chronic pancreatitis (CP) and pancreatic cancer (PC). The mechanism of action of targeted activated PSCs in pancreatitis and PC is expected to provide a new choice for the diagnosis and treatment of pancreatic diseases. Therefore, the present review focused on the activation mode of PSCs, including different signal transduction pathways and key molecules inside and outside cells, and systematically summarized the important role of activated PSCs in pancreatic injury repair and pancreatic diseases.
2. Regulation of PSCs' activation
Resting PSCs are often used as a sign of normal pancreatic tissue function. Once stimulated by various pathogenic factors, PSCs transform into activated myofibroblast-like cells, accompanied by changes in function and morphology (8,9). The transition from resting to activated states involves regulating various intracellular and extracellular signaling pathways, which have been comprehensively elucidated in the present review. The differences between the resting and active PSCs are summarized in Table I.
Table I.
Summary of the differences between the resting and active PSCs.
| Characteristic | Resting PSCs | Activated PSCs |
|---|---|---|
| Fat drops rich in vitamin A | Positive | Negative |
| α-SMA | Positive | Positive |
| Laminin, fibronectin, type I and III collagen | Positive | Positive |
| MMPs, TIMPs | Normal secretion | Hypersecretion |
| Cytokines, chemokines, growth factors | Normal secretion | Hypersecretion |
| Ability to proliferate and migrate | Weak | Strong |
| Metabolic change | Basically unchanged | Glucose metabolism, lipid metabolism, and amino acid metabolism increased |
PSCs, pancreatic stellate cells; α-SMA, α-smooth muscle actin; MMP, matrix metalloproteinase; TIMP, metallopeptidase inhibitor 1.
Extracellular signaling
Several studies have revealed that extracellular signaling contributes to PSCs' activation and is involved in the process of pancreatic fibrosis, including TGF-β, IL-10, oxidative stress, intracellular stress and ion channel conduction (5,6).
Cytokines. TGF-β
The TGF-β is the first identified member of the TGF-β family, which regulates the conduction of various intracellular and extracellular signals, including proliferation, apoptosis and differentiation. Additionally, it is involved in the development of inflammation, autoimmune diseases, cancer and other diseases, especially in the gastrointestinal tract (10–12). The occurrence and development of pancreatic diseases requires the participation of pancreatic fibrosis, which is inseparable from the continuous activation of PSCs, with exogenous TGF-β playing a role in this mechanism. Upon receiving effective stimulation of TGF-β, PSCs undergo activation of the myofibroblast marker α-smooth muscle actin (α-SMA). This activation triggers a continuous secretion of fibronectin and type I collagen by PSCs, thereby facilitating the deposition of ECM (13,14). Additionally, activated PSCs are overexpressed and secreted extracellularly by TGF-β1, positively regulating their activation state (15,16). TGF-β is not initially discovered in an activated form within the ECM. Instead, it is stored as a latent TGF-β binding protein and subsequently released by a mechanism involving cells. This process can be inhibited by all trans-retinoic acid (ATRA), restoring the resting state of PSCs and reducing ECM synthesis and PC progression (17). Moreover, the researchers discovered that PSCs play a key role in the fibrosis of alcoholic CP (ACP). Furthermore, the activation of PSCs is dependent on the regulation of TGF-β1. By establishing a rat ACP model, it was observed that the levels of IL-6 and TGF-β1 in the pancreas were significantly higher in the ACP group compared with the healthy control group. This increase was accompanied by the α-SMA and Col1 production. The change of TGF-β1 expression was positively correlated with IL-6 levels, and further experiments revealed that the activation of PSCs was achieved by upregulating the TGF-β1/Smad2/3 pathway, which confirmed that the activation of PSCs depends on the effective play of TGF-β (18). The efficient inhibition of PSC activation and the prevention of pancreatic disorders can be achieved through targeted intervention of TGF-β.
Connective tissue growth factor (CTGF)
The CTGF, a peptide characterized by its high cysteine content, is strongly associated with the modulation of several cellular processes, including cell differentiation, cycle and apoptosis, and adhesion. Moreover, CTGF is implicated in processes such as wound healing, tissue fibrosis and tumor progression (19,20). Previously, several studies have revealed that CTGF can promote the activation and proliferation of PSCs as well as the aggregation of ECM. These findings have garnered significant interest within the academic community. Especially in pancreatic disease, CTGF expression is associated with the severity of acute pancreatitis (AP) and CP (21,22). In PC, changes in CTGF levels directly affect the tumor stroma development (23). The aforementioned process is linked to the activation of PSCs. In necrotic tissue of pancreatic injury, activated PSCs auto-secrete CTGF and subsequently induce TGF-β and type I collagen production, which relies on the mediation of integrin α5β1 on PSCs. As a participant activated by PSCs, CTGF is continuously transmitted to other PSCs, induces their activation, and causes pancreatic fibrosis by promoting the synthesis of ECM components, ultimately leading to pancreatitis or PC (24,25). Tamura et al (26) investigated the pathogenesis of phosphatidylinositol-3-kinase (PI3K) and Hippo signaling in regulating the pathogenesis of CP. It was identified that knockdown of CEBPA in mouse CP models induced metaplasia from pancreatic acinar to duct and activation of PSCs, and this process could be mitigated by inhibiting CTGF, confirming that PI3K and Hippo signal-induced CP was achieved through the CEBPA/CTGF axis. As a member of the family of ADP-ribosylation factor-like protein 4A (ARL4), ARL4C is a small GTP-binding protein highly expressed in various types of cancer and has an important regulatory effect on tumor proliferation and drug resistance. In pancreatic tumors, cancer cells highly express ARL4C and promote paracrine CTGF, which subsequently causes activation of PSCs and promotes the progression of PC. Moreover, the aforementioned process can be reversed by silencing yes1 associated transcriptional regulator (YAP). Therefore, targeting the ARL4C-YAP-CTGF axis can prevent the progression of PC, however the in-depth regulatory mechanism remains unexplored (27).
Sonic hedgehog (SHH)
HH signaling is a protein ligand secreted by cells to regulate the proliferation and differentiation of neighboring cells. This signaling pathway has been implicated in the genesis and spread of tumors (28). Particularly in the case of highly aggressive PC cells, the secretion of the SHH protein by these cells elicits a response in the quiescent PSCs, leading to their activation. This activation, in turn, influences the behavior of PC cells in a reciprocal manner, establishing a detrimental feedback loop. Consequently, this process contributes to a significantly unfavorable prognosis and renders patients with PC resistant to chemotherapy (29–31). The SHH overexpressed in PC activates the Hh pathway in PSCs through paracrine pathways and subsequently promotes matrix metalloproteinase (MMP)-2, MMP-9, and nerve growth factor (NGF) in PSCs and proliferation of PSCs. Activated PSCs act as signaling stations for interactions between cancer cells and nerves, promoting tumor growth, metastasis and perineural invasion (32). The activation of PSCs plays a crucial role in the matrix response of PC. In the PC matrix, the level of Gremlin 1 expression increases, which is closely related to the degree of PSC activation. Cyclopamine and siGli-1 inhibit the activation of PSCs and the proliferation, invasion and epithelial-mesenchymal transition (EMT) of cancer cells. Further studies have revealed that PC cells promote their progression by secreting SHH to induce the activation of PSCs and increase the expression content of Gremlin 1 (33). The degree of proliferation of PSCs is an important manifestation of their activation level. Hwang et al (34) revealed that PC has high Hh ligand expression and low levels of Smoothened receptors, while PSCs have opposite performance. The co-cultivation of cancer cells with PSCs results in an increased rate of proliferation for both cell types. However, the administration of Hh antagonist AZD8542 effectively impedes this enhanced proliferation. In the PC model, AZD8542 inhibits tumor growth only in the presence of PSCs, confirming that cancer cells activate PSCs by secreting SHH signals to accelerate their growth. These studies suggested that PSCs promote self-activation by receiving paracrine SHH signals.
Hypoxia-inducible factor 1α (HIF-1α)
As a typical feature of the tumor microenvironment, hypoxia induces the proliferation and invasion of numerous malignant tumors, causing great obstacles to antitumor treatment. The transcription factor HIF-1α, which is widely present in hypoxic environments, is also closely related to the malignant phenotype of tumors (35,36). Previous studies have revealed that in pancreatic diseases, particularly pancreatitis and pancreatic tumors, hypoxia drives malignant progression, and an important factor is that hypoxia promotes the activation of PSCs and pancreatic fibrosis, which causes the aforementioned results (37,38). When Masamune et al (39) explored the role of hypoxia in PSCs, it was revealed that PSCs were activated by HIF-1α under hypoxic conditions, which promoted their proliferation and migration. Additionally, the activation of HIF-1α has been revealed to induce the synthesis of type I collagen and various angiogenic regulators, such as vascular endothelial growth factor, basic fibroblast growth factor, IL-8 and platelet-derived growth factor (PDGF). These molecular changes ultimately contribute to the development of pancreatic fibrosis and facilitate the metastasis of PC (39). In addition, a recent study demonstrated that hypoxia induces activation pf PSCs and produces IL-6, VEGFA, as well as stromal cell-derived factor 1 through HIF-1α to promote PC EMT. Furthermore, it inhibits apoptosis, and its efficacy was shown in a murine model of pancreatic carcinoma. However, the administration of resveratrol has the potential to reverse this outcome (40). These results demonstrated that HIF-1α in a hypoxic environment is an important source of PSCs activation, and its inhibition may be effective in alleviating pancreatic fibrosis or cancer progression.
PDGF
The PDGF, first discovered in platelets, has attracted marked attention for its ability to quickly activate the immune system at the trauma site and accelerate tissue wounds. Furthermore, PDGF has the functions of promoting angiogenesis and participating in cell division (41). Nevertheless, previous studies have revealed that PDGF is involved in the activation of PSCs and mediates the occurrence of pancreatic fibrosis. Hu et al (42) explored the pathogenesis of alcoholic pancreatitis, and it was revealed that the proliferation capacity of PDGF-treated PSCs was enhanced, and the activity of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase in cells was also increased. This finding provides confirmation that the activation and proliferation of PSCs are induced by PDGF through the stimulation of NADPH oxidase activity in quiescent PSCs. Consequently, this process leads to an accelerated production of ECM components and the promotion of pancreatic fibrosis. The effect of alcohol accelerated-process was aforementioned (42). Curcumin, as a polyphenolic compound, has powerful anti-inflammatory and antifibrotic properties. Several studies have revealed that using curcumin inhibited the expression of PDGF, IL-1β, α-SMA and collagen genes closely related to PSCs' activation, providing a new direction for treating pancreatic fibrosis and inflammation (43–45). The polyphenols in green tea extract, including epigallocatechin-3-gallate (EGCG), are the main components of natural antioxidants and have important advantages in preventing oxidative stress diseases, including cancer, cardiovascular disease and degenerative diseases. The activation and proliferation of PSCs are the key to pancreatic fibrosis, and PDGF is an important regulatory point for this process. It has been previously reported that EGCG ultimately prevents PSCs proliferation and the process of pancreatic fibrosis by inhibiting PDGF-mediated signaling. The process is achieved by inhibiting tyrosine phosphorylation of PDGF receptors and activation of extracellular regulated kinases (ERK) and PI3K/protein kinase B (AKT) pathways (46). These studies provide new insights into the development of pancreatic fibrosis and may reveal that PDGF becomes a new target for treating pancreatic fibrosis.
IL-10
The IL-10 is a cell-derived cytokine involved in a wide range of anti-inflammatory and immune responses in the body and is closely related to infection damage, tumors and cardiovascular diseases (47). Furthermore, IL-10 is secreted by T helper type 2 cells, contributes to pancreatic fibrosis and significantly lowers TGF-β1 expression in pancreatic acinar cells (PACs) (48–50). Demols et al (50) revealed that IL-10 knockout (KO) mice had more severe pancreatic tissue lesions and worsened fibrosis than the control group by establishing an AP mouse model of IL-10 KO. The reverse transcription-quantitative PCR and immunohistochemistry detected significantly elevated expression levels of TGF-β1 and activated PSCs in pancreatic ducts and mesenchymal cells in the IL-10 KO group. These findings revealed that IL-10 inhibits pancreatic collagen synthesis by reducing the activation degree of PSCs and TGF-β1 levels, thereby limiting pancreatic fibrosis and gland atrophy caused by AP. However, an additional study reported that IL-10 activation of PSCs only affected the rate of PSCs collagen synthesis but had no obvious effect on α-SMA expression or cell proliferation. However, there is no doubt that exogenous IL-10 can significantly reduce the severity of AP (51). Previous studies have demonstrated confirmed that IL-10 may be an effective target for inhibiting PSC activation.
Other inflammatory factors
It is very well established that inflammation is essential for PSCs activation, and activated PSCs secrete a series of cytokines, including IL-1β, IL-6 and TNF-α, to amplify the inflammatory response and self-activation, and further aggravate the disease (52). In numerous cell and animal models, releasing these inflammatory cytokines promoted activation of PSCs. After becoming highly activated, PSCs release cytokines including CTGF, MMP1 and others in an autocrine or paracrine manner. This vicious cycle results in significant ECM deposition and accelerates the development of pancreatic fibrosis (53–55). Additionally, these inflammatory cytokines are secreted by damaged PSCs and even cancer cells, facilitating damage repair or cancer development (56,57).
Oxidative stress
The imbalance between oxidative and non-oxidative balance induces the production of large amounts of oxidative active substances, including reactive oxygen species (ROS) and reactive nitrogen species, which ultimately lead to the aging of the body and disease development (58). Pancreatic fibrosis development is also inseparable from activation of PSCs induced by oxidative stress. Aging pancreatic β cells can induce type 2 diabetes, partly due to islet fibrosis caused by activated PSCs. In order to validate these findings, Ryu et al (59) conducted an isolation procedure on numerous resting PSCs derived from Sprague Dawley rats. Upon exposure to elevated sugar levels, the PSCs underwent a transformation into myofibroblasts and exhibited a significant increase in proliferation. Therefore, high sugar levels promote the activation of these cells by increasing oxidative stress of PSCs, consequently exacerbating the extent of islet fibrosis. To inhibit the progression of pancreatic fibrosis and prevent β cell dysfunction, Zhang et al (60) employed glutathione as a means to mitigate ROS generation in pancreatic tissue. Consequently, this intervention led to a reduction in the population of PSCs and a decrease in the severity of pancreatic fibrosis. These effects were achieved by modulation of the ROS/TGF-β/SMAD signaling pathway. The expression of fibromodulin (FMOD) and α-SMA by establishing a rat CP model has been previously determined; it was discovered that it is closely related to the degree of pancreatic fibrosis. Further experiments confirmed that FMOD upregulation increased collagen I expression and α-SMA and the proliferation and migration of PSCs, while the co-culture of pro-oxidant MND and PSCs increased FMOD levels, subsequently promoting PSCs activation (61). Therefore, reducing the production of exogenous ROS has also become the main contributor to pancreatic fibrosis.
Extracellular pressure
Mechanical stimulation plays an important role in cell growth and differentiation. It actively contributes to several cellular processes, including participating in the cell cycle, autophagy, and other processes influencing organismal homeostasis. Moreover, mechanical stimulation has been implicated in developing several pathological conditions, including heart disease, autoimmune diseases and cancer (62). Pancreatic lesions, increased tissue pressure and massive fibrosis often accompany the development of CP. To further understand the effect of extracellular hypertension on pancreatic fibrosis, the researchers induced a decrease in superoxide dismutase activity and increased ROS production in PSCs by adding compressed helium to the pressure loading device and applying pressure to PSCs, ultimately causing PSCs to produce a large amount of ECM and α-SMA, and the elimination of stress prevented the aforementioned outcome (63). Radoslavova et al (64) revealed that Ca2+ channels on the PSCs' membrane mediated the occurrence of the aforementioned behaviors. In addition to pancreatitis, the hypoxic microenvironment in which pancreatic tumors are located is a high-pressure environment, and the proliferation of tumor-associated fibroblasts is closely related to it (65). However, there are few studies on the link between extracellular hypertension and PSCs activation, and the pathway of hypertension-induced PSCs activation needs further exploration.
Calcium signaling
Previous studies have shown that the mechanical properties of the PC microenvironment can greatly promote activation of PSCs. These mechanical features include increased tissue pressure, as well as other elements previously discussed. It is plausible that this activation process is associated with the activation of relevant ion signals (66). These ion channels mainly convert mechanical forces into intracellular biological signals, acting as sensors in which calcium ions' conduction plays a key role. This process then triggers a cascade of cellular reactions, encompassing cell proliferation, migration and the secretion of relevant proteins (67). Radoslavova et al (64) revealed that extracellular high-pressure mechanical forces increase Ca2+ inflow into mouse PSCs through TRPC1 activation, whereas TRPC1 KO leads to inactivation of ERK1/2 and SMAD2 pathways and decreased Ca2+ inflow in PSCs. Finally, the proliferation and migration ability of PSCs deteriorated. The activation of PSC under hypoxia is related to the regulation of TRPC6 signaling, and calcium ion signaling appears to be involved in this activity (68). Additionally, as a free radical messenger mediating inflammation and vasodilation, NO production is closely related to the activation of calcium ion signal in PSC. Blocking the interaction between calcium ions can inhibit NO production to protect PSC and PAC from necrosis, a potential target for anti-inflammatory therapy (69). Moreover, pancreatic fibrosis induced by AMP, ADP and ATP-induced signaling depends on increased calcium ion concentration in PSC, and macrophages are involved in the important process (70). Further studies are needed to clarify the relationship between calcium ion related signals and the active state of PSC.
Intracellular signaling
The activation of PSCs is also inseparable from the conduction of intracellular signals, including PI3K/AKT, mitogen-activated protein kinase (MAPK), Smad, proliferator-activated receptor-g, and nuclear factor κB (NF-κB). A deeper understanding of these mechanisms affecting activation of PSCs is expected to provide new avenues for reversing pancreatic fibrosis-related diseases.
PI3K/AKT
The PI3K/AKT pathway is important in carcinogenesis, inflammation and other related diseases. Furthermore, it is essential for the activation of PSCs (71,72). Coenzyme Q10 (CoQ10), a coenzyme with a powerful antioxidant function, participates in electron transport in mitochondria, and regulates the inflammatory response. The CoQ10 has been shown to effectively inhibit activation of PSCs and stop the progression of pancreatic fibrosis. Simultaneously, the PI3K/AKT/mTOR signaling axis participates in this reaction, and this result can be implemented by establishing a C57BL/6 mouse model (73). Cui et al (74) additionally identified the involvement of the PI3K/AKT pathway in the activation of PSCs. In their study, they induced a rat model of CP and observed increased activation of PSCs, along with significant secretion of collagen. After the infusion of different doses of Saikosaponin d (SSd) into the aforementioned model, the utilization of SSd was documented for subsequent histological and molecular biological investigations. The findings revealed that the use of SSd inhibited PSCs activation and ECMs production, consequently mitigating pancreatic damage induced caused by inflammation. It was further reported that this phenomenon is achieved by crosstalk in the PI3K/Akt/mTOR and TGF-β1/Smads pathways. Carbon monoxide (CO) inhibits various cells as an endogenous gaseous signaling transmitter. It has been revealed that CO released by the CO-releasing molecule-2 can inhibit protein synthesis by mediating eukaryotic elongation factor 2 phosphorylation and 4E-binding protein 1 inactivation. Consequently, this leads to the downregulation of PSCs cyclin D1 and cyclin E and the arrest of the cell cycle, preventing activation of PSCs. The occurrence of these cellular activities is closely related to inhibition of PI3K/Akt/mTOR signaling (75). The PI3K/Akt/mTOR pathway is expected to provide an effective target for treating pancreatic fibrosis.
MAPK
As a key hub for communication between cells and beyond, the MAPK pathway involves physiological processes, including cell proliferation, differentiation and cycling. Moreover, it regulates the pathogenesis of several diseases, including cancer, inflammation and autoimmune diseases (76,77). Highly aggressive PDAC has a dense fibrotic matrix that acts as a barrier to immunosuppression. The formation of this matrix is primarily influenced by the interaction of PSCs with cancer-associated fibroblasts (CAF). This interaction leads to sustained activation of inflammatory signals in the tumor microenvironment (TME) through the production of inflammatory and chemokines, ultimately promoting the malignant progression of cancer. As a chaperone protein, heat shock protein (Hsp90) regulates various cellular activities between immune cells and tumors in TME. The use of Hsp90 inhibitors has been revealed to reduce PSCs' activation and α-SMA expression through Janus kinase (JAK)/signal transducers and activators of transcription (STAT) and MAPK/ERK signaling, limit PSCs/CAF growth and enhance sensitivity to PD-1 blockade (78). Oxidative stress can also use the MAPK/AP-1 signaling axis to promote FMOD overexpression in PSCs, subsequently causing upregulation of collagen I and α-SMA and proliferation and migration of PSCs, while the use of ERK and Jun N terminal kinase (JNK) inhibitors reverses the aforementioned results (61). During alcohol-induced pancreatic fibrosis, activation of PSCs is always accompanied by the loss of cytoplasmic retinol. Additionally, previous studies have confirmed that ethanol can mediate PSCs' activation through the MAPK pathway. Consequently, the question is if retinol supplementation prevents ethanol-induced PSCs' activation. This problem is explained by McCarroll et al (79), who co-cultured retinol and its metabolites ATRA and 9-RA with ethanol-treated PSCs and revealed a significant decrease in α-SMA, collagen I, fibronectin and laminin in the medium, accompanied by a decrease in activation of MAPKs (EKR1/2, p38 kinase and c-JNK). Finally, activation of PSCs was inhibited (76). The MAPK signaling pathway has also become an effective way to inhibit PSCs' activation, requiring further comprehensive investigation.
TGF-β/Smad
The TGF-β is an important regulator of fibrotic disease occurrence, which can regulate the transformation of fibroblasts to myofibroblasts. Through interaction with the downstream cytoplasmic transcription factor Smads, TGF-β induces the collagen synthesis, and is widely involved in physiological and pathological processes, including tissue fibrosis and tumor occurrence and development (80–82). Especially in pancreatic diseases, TGF-β/SMAD is crucial for pancreatic wound healing, cell growth, cell cycle regulation, angiogenesis and immune regulation (83–85). Derived from hormone-like muscle factors produced by skeletal muscle motor response, irisin is involved in various body activities, including systemic inflammation, bone balance maintenance, metabolic regulation and cancer proliferation (86). Irisin has attracted marked attention for its ability to alleviate the process of tissue fibrosis, including heart, liver and kidney fibrosis (87–89). This antifibrotic effect was also demonstrated in the mouse CP model. Continuous stimulation of inflammation causes pancreatic damage and extensive fibrosis while increasing irisin, a protective factor, significantly reduces the degree of apoptosis and necrosis of pancreatic cells. Furthermore, the immunohistochemistry results of pancreatic tissue showed a significant reduction in ECM deposition, which correlated with the oxidation-reduction and ER stress in pancreatic cells from CP mice. In vitro experiments also confirmed that these effects are mediated by downregulating kindlin-2 and inhibiting the SMAD2/3 pathway (90). Berberine (BR), extracted from the root and rhizome of various Ayurvedic and Chinese medicinal plants, has numerous pharmacological effects, including anti-inflammatory and fibrotic. In the cerulein-reduced pancreatitis model, it was revealed that BR intervention inhibited collagen deposition in pancreatic tissue, the infiltration of inflammatory cells, pancreatic vacuolization and pancreatic cell atrophy through histopathological analysis, and improved the degree of pancreatic damage. At this time, the markers of PSCs activation markers, including α-SMA, Collagen1a, collagen3a and fibronectin, were significantly downregulated, and further experiments confirmed that BP alleviated the fibrosis process of CP by activating the AMPK pathway by inhibiting TGF-β1/Smad signaling (91). In addition, Choi et al (92) confirmed that piperine, derived from black pepper, improves the degree of pancreatic fibrosis in a dose-dependent manner. The mechanism involves piperine inhibiting TGF-β-induced pSMAD2/3 activation, followed by a reduction in the expression of fibrotic mediators in PSCs, including α-SMA, collagen and fibronectin1, ultimately achieving the therapeutic purpose of CP. The targeted intervention combined with traditional Chinese medicine may be effective in stopping the process of pancreatic fibrosis.
Peroxisome proliferator-activated receptor (PPAR)
Peroxisome proliferators are widely known for their involvement in various intracellular metabolic processes, belonging to ligand-induced nuclear receptors. These receptors include three subtypes, namely PPAR-α, PPAR-β/δ, and PPAR-γ (93). Previous studies have confirmed that in addition to regulating whole-body energy homeostasis, PPAR, especially PPAR-γ, plays a key role in blocking PSCs' activation (94). Wang et al (95) used taurocholate (TLC) to treat AR42J cells to establish a CP model. They subsequently extracted exosomes secreted by acinar cells and manipulated the expression of miR-130a-3p by overexpression or knockdown techniques. Finally, they incubated PSCs with the aforementioned exosomes. In the miR-130a-3p overexpression group, expression levels of activation markers α-SMA protein and fibrotic markers collagen I and collagen III in PSCs were significantly elevated at the RNA and protein levels. The knockdown of exosome miR-130a-3p significantly inhibited PSCs' activation and fibrosis. Further experiments have confirmed that exosome-derived miR-130a-3p promotes pancreatic fibrosis by inhibiting PPAR-γ expression in PSCs, ultimately exacerbating the severity of CP. Albumin is a plasma protein primarily synthesized by liver cells. Its primary function is to support to maintain the nutrient metabolism of body and facilitate the maintenance of the internal environmental conditions necessary for tumor growth. However, PSCs extracted from the rat pancreas exhibit a significant ability to detect and regulate high levels of albumin in order to sustain their quiescent condition. However, when albumin content decreases after PSCs' activation, PPAR-γ and C/EBP-α expression levels decrease. The albumin content is increased by overexpressing PPAR-γ and C/EBP-α, whereas activated PSCs undergo a transition toward a resting state. Albumin-mediated PPAR-γ or C/EBP-α-induced phenotypic changes in PSCs provide a new strategy for treating pancreatic fibrosis (96). Cyclic AMP-response element-binding protein-binding protein (CBP)/β-catenin signaling also plays an important role in organ fibrosis. The use of CBP-β catenin antagonists has been revealed to inhibit PSCs' activation, downregulate ‘activation’ markers [α-SMA/actin alpha 2, collagen alpha1, Prolyl 4-hydroxylase, and Survivin], and reduce their proliferation. Notably, the observed effects can be counteracted by inhibiting PPAR-γ expression. Moreover, CBP/β-catenin antagonism impedes the migration of PSCs-induced PC cells, presenting a promising therapeutic approach for managing pancreatic fibrosis (97).
NF-κB
Aberrant NF-κB signaling, functioning as a multifunctional transcription factor, has been implicated in the pathogenesis of chronic inflammation, autoimmune diseases and malignancies (98). Previous studies have shown that knocking out NF-κB p65 in PACs can promote fibrosis in rain frog-induced mouse models of CP induced by rain frogs, for which the activity of NF-κB is closely related to activation of PSCs (99). Wu et al (100) conducted an in-depth discussion of this phenomenon. It was hypothesized that in pancreatic inflammation, overexpressed TGF-β1 binds to receptors on PSCs to induce TAK1 phosphorylation and activation of the NF-κB signaling pathway. The NF-κB activation further promotes MMP-1/TIMP-1 imbalance, subsequently increasing ECM production and aggravating pancreatic fibrosis. Furthermore, NF-κB activation induces PSCs to produce a large amount of MCP-1, recruiting distant macrophages to aggravate the inflammatory infiltration of pancreatic tissue. Hydrogen peroxide-induced cloning-5 (Hic-5), a LIM (Lin-11, Isl-1 and Mec-3) containing adherent scaffold proteins, is involved in various physiological activities in vivo, including cell proliferation, differentiation, aging, apoptosis and signal transduction. Previous studies have reported that Hic-5 plays an important role in fibrotic diseases, including liver fibrosis, intestinal fibrosis and glomerulosclerosis. The researchers established a mouse model of CP to confirm whether it has similar functions in pancreatic fibrosis. Compared with the untreated group, the pancreas size and weight of Hic-5 knockout mice were significantly increased. Additionally, there was a significant decrease in pancreatic tissue necrosis and inflammatory infiltrates, as well as a reduction in the extent of fibrosis. These observed effects can be attributed to inhibiting downstream NF-κB/p65/IL-6 signaling. As a diterpenoid epoxide with immunosuppressive activity, triptolide can reduce the activation ability of PSCs by inhibiting the transcriptional activity of NF-κB/p65, thereby reducing the fibrosis process of CP mice. These findings hold promise for developing novel therapeutic approaches for managing clinical CP patients (101). As a mediator of cell activation, Retinoblastoma (RB) also acts as a signal receiver to exert anti-inflammatory and antitumor effects, especially in the superphosphate state (102). Massively releasing pro-inflammatory mediators activate PSCs in pancreatic injury, which subsequently induces pancreatic fibrosis and systemic inflammation. As previously mentioned, the sequence of actions is closely connected to the involvement of hydrogen sulfide, which plays a regulatory role in the development of proteins associated with the inflammatory response through the activation of NF-κB (103). Growing evidence suggest that the RB state regulates hydrogen sulfide production, especially during RB super-phosphorylation. The large production of hydrogen sulfide induced a large increase in serum amylase, inflammatory factor TNF-α, pancreatic edema and lung injury in mice. Super-phosphorylation of RB depends on the extensive infiltration of CDK4/6. The utilization of CDK4/6 inhibitor palbociclib decreased the level of RB phosphorylation, downregulation of hydrogen sulfide production and NF-κB activation, and the reversal of pancreatic inflammation and fibrosis (104). Targeting NF-κB may effectively reduce the process of pancreatic fibrosis. However, more investigation is necessary to elucidate its efficacy fully. The molecules and signaling pathways related to converting PSCs from resting to active states are demonstrated in Fig. 1.
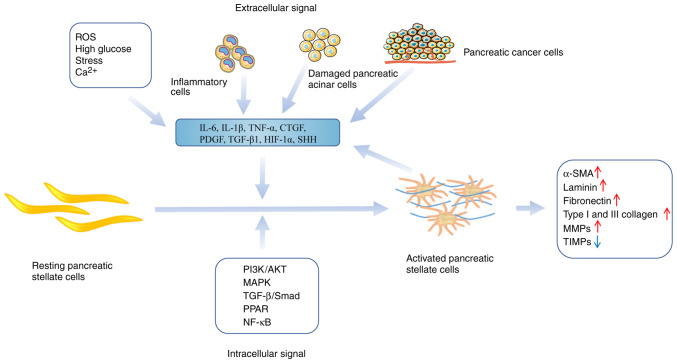
Figure 1.
The molecules and signaling pathways are related to converting PSCs from resting to active states. The figure describes the main intracellular and extracellular signals transforming PSCs from resting to active states, including high glucose, stress, oxidative stress, inflammatory factors, and the MAPK and PI3K/AKT signaling pathways. Upon activating PSCs, there is an observed increase in the secretion of α-SMA, laminin, fibronectin, and type I and III collagen associated with ECM. Additionally, there is an alteration in the equilibrium between MMPs and TIMPs, indicating an imbalance in their regulation. PSCs, pancreatic stellate cells; α-SMA, α-smooth muscle actin; ECM, extracellular matrix; MMP, matrix metalloproteinase; TIMP, metallopeptidase inhibitor 1.
3. The key role of PSCs' activation in damage repair
As one of the most important parenchymal organs in the human body, it is extremely difficult for the adult pancreas to repair itself due to slow cell renewal and low regeneration ability. Therefore, in addition to exogenous damage, pancreatic damage caused by AP, CP and PC is difficult to recover, which is closely associated with the inherent characteristics of PSCs. This section summarized the mechanism of action of PSCs and their activation in repairing pancreatic injury.
PSCs' activation promotes the secretion of ECM
The ECM is a dynamic structure outside the cell, which is essential for regulating the proliferation, migration, differentiation and other aspects of cells, and its existence provides an effective barrier for the structural and functional integrity of tissues (105,106). However, remodeling of ECM (composition, abundance and hardness) contributes to progression of diseases, including fibrosis and cancer (107,108). Concurrently, repairing pancreatic injury is strongly connected to the involvement of ECM since the creation and deposition of ECM promote fibrosis formation, which afterwards facilitates the restoration of tissue defects (109). A previous study revealed that pancreatic injury-induced activation of PSCs was followed by increased production of laminin, fibronectin, and type I and III collagen, compared with resting PSCs, which are important manifestations of ECM accumulation. Compared with normal pancreatic tissue, excessive ECM secretion and remodeling are beneficial for post-injury repair (13). Additionally, activated PSCs secrete cytokines, chemokines and growth factors, and induce an imbalance between MMP and TIMP, further promoting fibrosis (110). Therefore, activating targeted PSCs to inhibit excessive ECM secretion can effectively hinder the pancreatic fibrosis process.
Activation of PSCs enhances the EMT pathway
EMT is a complex biological process characterized by a series of sequential steps. During EMT, epithelial cells undergo a phenotypic transformation, acquiring characteristics similar to the interstitial phenotype. Several modifications, including disruption of the ECM, reconstruction of the cytoskeleton and production of ECM proteins, accompany this transformation significantly contributes to embryonic development, chronic inflammation, cancer and several fibrotic diseases (111,112). The EMT is divided into three types, of which type 2 EMT can transform epithelial cells into fibroblasts, promote the secretion of tissue ECM, and is closely related to injury repair and organ fibrosis (113). It was revealed that the activation of PSCs shared morphological and functional changes with the EMT process. After PSCs' activation, the classical regulatory molecules of EMT process, including E-cadherin, N-cadherin and vimentin can change accordingly. Furthermore, bone morphogenetic protein 7 and S100 calcium-binding protein A4 (S100A4) experienced significant protein content. The BMP signaling inhibits the progression of EMT, ultimately affecting tissue fibrosis and cancer progression. Additionally, S100A4 expression increased significantly in fibroblasts when the PSCs remained fully activated, implying an enhancement of the type 2 EMT process (114–117). Xu et al (118) also confirmed that the enhancement of EMT accompanies the activation of PSCs. It was revealed that TGF-β1 is strongly associated with pancreatic fibrosis, a result achieved by triggering activation of PSCs and promoting EMT through the PI3K-Akt signaling axis. Therefore, PSCs are considered to be essential in repairing damaged pancreatic tissue through EMT. The activation of PSCs involves transitioning from a resting state to a fibroblast-like phenotype, resembling an EMT-like process.
Crosstalk between PSC activation and mononuclear macrophages in the pancreatic microenvironment
Pancreatic injury repair is inseparable from the interaction of numerous microenvironment components (119,120). Particularly, PSCs' activation can regulate other cell functions, ultimately affecting the regeneration of pancreas (121). Infiltration of monocytes in the body is characteristic of several fibrotic diseases. Furthermore, macrophages can differentiate into different phenotypes by stimulating the microenvironment within the receptor, mainly including the M1 and M2 types, called macrophage polarization, mainly including the M1 and M2 types, which are called macrophage polarization. Among them, the M1 type is mainly manifested as pro-inflammatory, and M2 macrophages are selectively activated macrophages, which have anti-inflammatory and pro-fibrotic effects and can be directly activated by cytokines including IL-4 and IL-13 (122). Pancreatic injury can induce macrophage differentiation to M2 type by activating PSCs' production of IL-4 and other factors, further promoting the secretion of ECM in pancreatic tissues and accelerating injury healing (123). Michalski et al (124) established a monocyte and PSC co-culture system and revealed that infiltrated monocyte macrophages can promote PSCs' activation and improve autocrine capacity of PSCs, which may lead to persistent chronic inflammatory states. Another study has reported that lipopolysaccharide-stimulating macrophage activation promoted PSCs over-secretion of ECM and pancreatic fibrosis, an effect achieved by TGF-β (125). Furthermore, when the static PSC was co-cultured with macrophages, TGF-β1, fibroblast growth factor-2, TGF-α and PDGF, and tumor necrosis factor (IL-1 and IL-6) deactivate PSCs and transform them into myofibroblasts (126). Gerasimenko et al (127) revealed that a special pathway between PSC and macrophages, in which PSC secretes IL-18 in response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) receptor activation. Subsequently, IL-18 stimulates macrophages to induce the production of numerous calcium ion signals and inflammatory factors. This mechanism plays a significant role in AP outbreak and fibrosis. Further exploring the interaction between activated PSCs and macrophages may be crucial for repairing pancreatic damage.
Activated PSCs differentiate into parenchymal cells
Pancreatic parenchymal cell proliferation is important in repairing the damaged pancreas. Several studies have revealed that pancreatic stem cells exist in pancreatic acinar and islet tissues, which can control acute inflammatory response by antagonizing the release of inflammatory mediators, reducing immune response, and differentiating into PACs and endothelial cells to repair damaged pancreas (128,129). It has been established that various pancreatic mesenchymal cells have been established to have stem cell functions, including nestin-positive cells and side population cells (130,131). Moreover, it has been previously revealed that activated PSCs have biological phenotypes of pancreatic stem cells, including common markers (nestin, CK-19 and ATP-binding cassette superfamily G member 2) (132). Several studies isolated part of PSCs from pancreatic tissues of rats and measured their expression of various stem/progenitor cell markers, including CD133 and paired-like homeodomain 2 (133). Additionally, as the signaling pathways necessary for stem cell maintenance and development, β-catenin-dependent Wnt and Notch signaling participate in the cellular regulation of PSCs, indicating that PSCs have a characteristic expression profile of stem cells, clonal cell growth, long-term survival transplantation ability and regeneration process (134). The source of pancreatic stem cells is still debated. However, according to the aforementioned research analysis, the function of PSCs is limited to the secretion of ECM, and as a cell with differentiation potential, by differentiating into cells with pancreatic endocrine function and then supplementing and repairing pancreatic necrotic tissue from the cellular level when pancreatic damage, but more studies are needed to confirm it in the future. The mechanisms of action of activated PSCs in pancreatic injury repair are presented in Fig. 2.
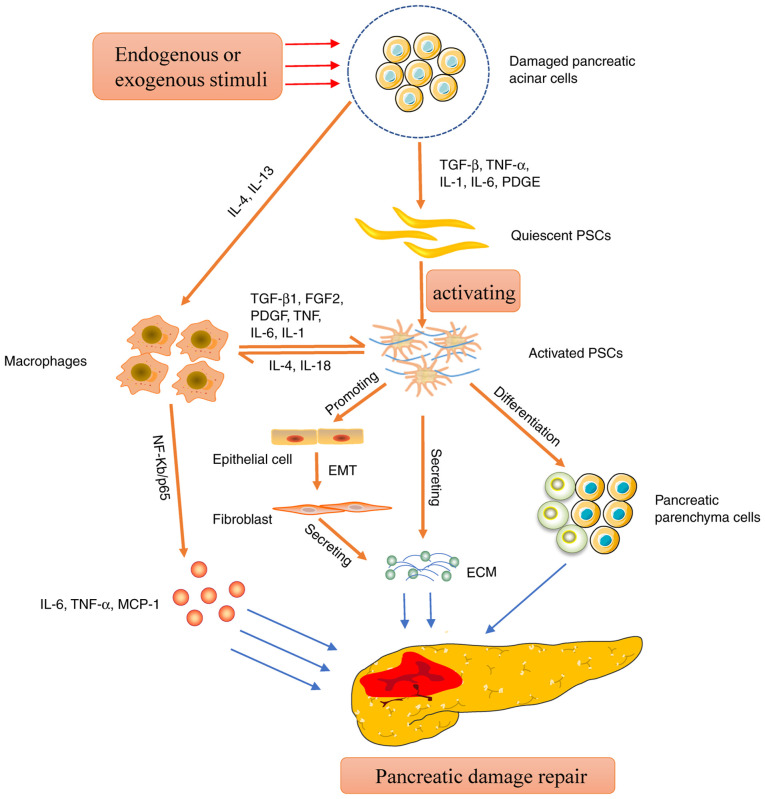
Figure 2.
Mechanism of action of activated PSCs in pancreatic injury repair. This figure mainly describes the self-secretion of ECM when resting PSCs are activated, and the secretion of ECM through the EMT pathway to promote pancreatic fibrosis. Additionally, activated PSCs promote pancreatic injury repair by recruiting monocytes for macrophage infiltration and inducing macrophages to polarize to M2 type, followed by the secretion of IL-6, TNF-α and other factors. Activated PSCs have characteristics resembling those of pancreatic stem cells, which can be guided toward differentiation into pancreatic endocrine cells, hence facilitating pancreatic repair. PSCs, pancreatic stellate cells; ECM, extracellular matrix; EMT, epithelial-mesenchymal transition.
4. PSCs in pancreatic disease
Type 2 diabetes
Type 2 diabetes is a chronic metabolic disorder characterized by fundamental pathophysiological features, including islet β cell dysfunction and insulin resistance (135). The functional status of pancreatic islets β cells is crucial for the progression of type 2 diabetes. Especially when the body develops insulin resistance, pancreatic islets β cells exhibit a heightened ability to adapt and respond to elevated metabolic demands. However, the dysfunction of pancreatic islet β cells means this glucose cannot be converted and utilized, eventually leading to a continuous increase in blood sugar (136). The causes of secretory dysfunction of pancreatic islet β cells include chronic inflammation, endoplasmic reticulum stress, glucolipid toxicity and oxidative stress (137). Furthermore, the fibrosis of islets causes irreversible damage to the β cells of islets, ultimately promoting the progression of diabetes. In this process, activated PSCs play a crucial role (138,139). Previous studies have revealed islet fibrosis obvious in rat models with type 2 diabetes. Additionally, a substantial presence of α-SMA-positive cell infiltration is observed in and around pancreatic islets, confirming that activated PSCs are closely related to islet fibrosis in type 2 diabetes. The investigation of oxidative stress as a potential contributor to the progression of islet fibrosis through the activation of PSCs deserves additional examination, given its significance as a key determinant (140). Ryu et al (59) revealed that the use of antioxidants reduced islet fibrosis by establishing an animal model of type 2 diabetes and measured that the degree of PSCs activation was reduced at this time. High-glucose feeding reversed the aforementioned results, and ROS, α-SMA, fibronectin, collagen and cytokines production in PSCs increased, confirming that high sugar increased the oxidative stress of PSCs, induced its activation and ultimately promoted islet fibrosis. Conophylline (CnP), as a natural compound, is effective in promoting the proliferation of pancreatic islet β cells and exerts antifibrotic effects. To further understand the mechanism, the researchers revealed that the medium added to CnP when PSCs were cultured in vitro decreased collagen production. The activation of PSCs was also reduced by establishing a rat model of type 2 diabetes mellitus and obtaining pancreatic pathology sections. After immunohistochemistry analysis, it was revealed that the infiltration level of pancreatic CD68-positive macrophages and PSCs in rats fed with CnP was significantly reduced. Additionally, there was a significant improvement in islet fibrosis. These findings confirmed that CnP could inhibit islet fibrosis and promote the regeneration of islet β cells by affecting the activation of PSCs (141). Targeted activation of PSCs may become an effective treatment for type 2 diabetes.
Pancreatitis
AP
This condition is defined by the self-digestion of the pancreas and its surrounding tissues, which is triggered by the excessive release of excess intracellular calcium (142). However, PSC is not only activated in PC or PC, and numerous calcium ion signals can be generated even in the normal microenvironment, which is an important pathophysiological mechanism of AP (143). Previous studies have revealed that elevated bradykinin levels in AP are associated with calcium signaling. First, bradykinin induces calcium signal storage in PSC inositol triphosphate through the B2 receptor and then releases the CRAC channel into pancreatic acinar cells under the action of the inositol triphosphate receptor. Using B2 receptor or CRAC channel inhibitors can reduce PAC necrosis induced by pancreatitis (144). However, in the experimental model of alcohol-related, bradykinin production is initially mediated by PACs. The initial injury of acinar cells includes the entry of kallikreins and other substances into the interstitial fluid, leading to an increase in bradykinin concentration, which promotes the activation of calcium ion signals in PSC and the subsequent release of a large amount of NO. This could accelerate the deterioration of acinar cells and aggravate the progression of pancreatitis. Pharmacological inhibition of NO synthase can provide a highly effective anti-necrosis effect, which was also confirmed by Gryshchenko et al (145) and Jakubowska et al (69). Unlike alcoholic pancreatitis, bile acid-induced AP has a lower degree of fibrosis, mainly due to bile acid-dependent calcium signal and NA-bile acid cotransporter-mediated eliminating effect on PSCs. Targeting NA-dependent bile acid transport provides the pharmacological basis for intervention in the acute phase of biliary pancreatitis (146). In addition to releasing bradykinin, ATP, ADP and other substances are released to activate purinergic receptors on the surface of macrophages, producing numerous calcium signals and secreting numerous inflammatory factors to aggravate pancreatic inflammatory storms. Relevant experiments showed that suramin blocking ATP can significantly reduce the levels of cytokines in AP plasma, including IL-6 and the degree of pancreatic injury (147). Furthermore, inhibition of the SARS-CoV-2 receptor to block calcium signal production of calcium signals in macrophages is expected to reduce the degree of inflammation in AP (123).
CP
When pancreatic inflammation occurs repeatedly, CP is formed by prolonged stimulation, accompanied by progressive destruction of the pancreatic acinar and massive fibrosis, eventually leading to the loss of pancreatic function, a process that currently has no means of reverse (148). Previous studies have shown that the activation of PSCs plays a crucial role in the fibrotic process of CP. The hydrogen peroxide-inducible clone 5 (Hic-5) acts as an adherent plaque scaffold protein that controls the cytoskeleton and influences vascular remodeling and disease fibrosis (149). Gao et al (150) evaluated the expression of Hic-5 in normal pancreatic tissues and CP tissues by immunohistochemistry and immunofluorescence, and it was revealed that it was highly expressed in activated PSCs in human CP tissues. When Hic-5 was knocked out in cerulein-induced CP fibrosis mice, the expression of pancreatic fibrosis-related factors, including Col1a1 and Col3:1 was significantly reduced, and the degree of PAC destruction and fibrosis was significantly reduced, confirming that Hic-5 contributes to the progression of CP fibrosis. Further research study has revealed that Hic-5 regulates the activation of PSCs and the production of ECM through the TGF-β/Smad signal axis, ultimately affecting the fibrosis process of CP. Autophagy is a dynamic process by which cells selectively eliminate dysfunctional cellular components through lysosomes. This process facilitates the timely screening of damaged organelles and other components to maintain intracellular metabolism, nutrient and energy balance (151). The aforementioned study identified that the activation of PSCs depends on autophagy and ultimately affects the CP fibrosis process. In this process, Lnc-PFAR is essential and its inhibition affects the expression of α-SMA, collagen I, collagen III and fibronectin in PSCs, which is caused by regulating the maturation of miR-141-5p. The Lnc-PFAR facilitates autophagy and exacerbates pancreatic fibrosis by reducing pre-miR-141 maturation in CP. The miR-141-5p induces dephosphorylation of Unc-51 like autophagy activating kinase 1 by binding to RB1 inducible Coiled-Coil 1, which ultimately inhibits autophagy and PSCs' activation. Furthermore, in vivo experiments also validate the accuracy of these findings. Therefore, the high expression of Lnc-PFAR reflects the severity of CP fibrosis and has also become a biomarker for early detection of CP (152). Moreover, the disorder of lipoprotein metabolism in PSCs is an important contributor to CP progression. Particularly, very low-density lipoprotein receptor overexpression promotes the accumulation of intracellular lipids, which subsequently causes the excessive release of IL-33 by PSCs. Conversely, PSCs-derived IL-33 inversely promotes the activation of PSCs, eventually causing PSCs to secrete ECM, leading to fibrosis during CP (153). Briefly, targeting the correlated factors of PSCs activation may effectively reverse the process of fibrosis during CP.
PC
PC, a malignancy mostly affecting the digestive tract, exhibits notable features, including delayed detection, heightened aggressiveness and poor prognosis, with a survival rate of <10%. PC is expected to be the second leading cause of global cancer-related death by 2030 (154,155). Tumor-interstitial interactions are considered to contribute to the progression of PC cancer progression significantly. The infiltrated collagen matrix is produced by activated PSCs, which are closely related to cancer cells and components in the microenvironment, including immune cells, endothelial cells and microorganisms, and participate in malignant biological behaviors such as tumor growth and metastasis invasion (156). As a bridge between cancer cells and the microenvironment, exosomes mediate the crosstalk of components between PSCs cells and PC cells to regulate tumor proliferation. It has been revealed that miR-5703 in PSC secreted exosomes can promote PC cells' proliferation. A previous study has demonstrated that miR-5703 ultimately promotes the progression of PC by binding to CKLF-like MARVEL transmembrane domain containing 4 and inducing its expression downregulation, which is achieved by the PAK4/PI3K/AKT signal axis. These results were verified in animal experiments, confirming that PSCs-derived exosomes mediate the malignant process of PC (157). As a member of the neurotrophic factor family, the NGF is closely related to PC proliferation, apoptosis and metastasis. Jiang et al (158) revealed a high expression of NGF by isolating PSCs in pancreatic tissues and then co-culturing PSCs with PC cells; it was revealed that PC cells' invasion and proliferation ability was significantly enhanced. This promotion is achieved by overexpressing NGF, which further binds to TrkA on the PC cell membrane and activates the PI3K/AKT/GSK signaling axis, which then promotes the secretion of MMP9, vimentin and E-cadherin, and ultimately enhances the proliferation and invasion ability of PC cells. The Wnt and tenascin C (TnC) are key regulatory mediators during PC cell interaction with PSCs, which promote tumor proliferation and metastasis by activating oncogenic β-catenin and YAP/TAZ signaling pathways in PC cells in a paracrine manner. Targeting of N-myc downstream-regulated gene-1 (NDRG1) by di-2-pyridylketone-4-cyclohexyl-4-methyl-3-thiosemicarbazone (DpC) inhibited PSCs secretion of Wnt and TnC, while weakening the activation of Wnt/β-catenin and YAP/TAZ signaling in PC cells, hindering the connection between PSCs and PC cells and PC progression. Moreover, NDRG1 inhibition affects PSCs activation mediated by PC cells through TGF-β. Furthermore, in vivo experiments have confirmed that NDRG1 and DpC inhibit Wnt/TnC-mediated interactions between PC cells and PSCs (159).
Dense fibrotic stroma is characteristic of pancreatic tumors, achieved through the interaction between PC cells and stromal cells, including PSC (5). Activation of PSC enhances malignant processes of PC, including EMT and stem expression of tumor cells (160,161). The fibrous matrix created by PSC provides the PC cells with hypoxia, nutrient deficiency, oxidative stress and an acidic environment. Cancer cells and PSCs are under constant pressure to adapt to their surroundings, and they do so by altering the expression of signaling molecules, transporters and metabolic enzymes (161). Previous studies have shown that PC tissues are significantly hypoxic compared with neighboring normal tissues, accompanied by HIF-1 activation. Hypoxia-induced upregulation of HIF-1 can promote the expression of transcription factor TWIST and enhance EMT in cancer cells (162). Additionally, HIF-1 can mediate CTCF production to protect PC from hypoxic-induced apoptosis and enhance gemcitabine resistance (163,164). Moreover, HIF-1 recruits macrophages by inducing PC cells to secrete large amounts of chemokines, including CCL2. Numerous tumor-associated macrophages promote the expression of miR-4465 and miR-616-3p in PSC, leading to increased proliferation, migration and invasion of PC (165). The hypoxia of tumor is accompanied by the mass production of superoxide anion, hydroxyl radical, hydrogen peroxide and oxidative stress reaction. To prevent excessive oxidative stress from damaging cancer cells, tumors upregulate the expression of NRF2, a transcription factor that recognizes elements of the antioxidant response in target genes (166). Increased NRF2 promotes chemotherapy resistance, EMT and metabolic remodeling of PC cells (167,168). In vivo animal models confirm that oxidative stress activates PSCs, and activated PSCs can secrete IL-6 and matter-derived factor-1α, which in turn activate PC cells to express NRF2. This results in increased cancer cell proliferation and reduced oxidative stress response, which has been confirmed in vivo in animal models (169,170). Several NRF2 inhibitors have been applied in preclinical studies, including NSC84167 and fluofuranone (171,172), and are expected to enter clinical trials for further validation. Targeting the signal axis between PSCs and PC may play an irreplaceable role in treating PC, and it is worth further exploration in the future. The role and mechanism of PSCs' activation-related factors in pancreatic diseases are summarized in Table II.
Table II.
Summary of the role and mechanism of PSCs activation-related factors in pancreatic diseases
| Related factors | Patient | Mechanism of action | In vitro/in vivo | (Refs.) |
|---|---|---|---|---|
| High glucose | Type 2 diabetes | Promotes oxidative stress in PSC and increases the expression of ROS, α-SMA, fibronectin, collagen, and cytokines. | In vitro | (140) |
| CnP | Type 2 diabetes | Reduces PSC activation, inhibits collagen synthesis and CD68-positive macrophage infiltration, and promotes islet β cell proliferation. | Both | (59) |
| Bradykinin | AP | Binding to the B2 receptor on PSC induces calcium ion signal production and NO release. | Both | (143) |
| ADP, ATP | AP | Activates purinergic receptors on the surface of macrophages and promotes PSC activation | Both | (144) |
| SARS-CoV-2 | AP | Activating PSC to secrete IL-18 promotes macrophages to produce numerous calcium ion signals and inflammatory factors. | Both | (126) |
| Hic-5 | CP | Activation of TGF-β/Smad promotes PSC activation and proliferation and increases Col1a1 and Col3:1 expression. | Both | (148) |
| Lnc-PFAR | CP | ULK1 dephosphorylation is induced by mediating the miR-141-5p/RB1CC1 axis, inhibiting autophagy and fibrosis. | Both | (151) |
| VLDLR | CP | Promotes lipoprotein accumulation and PSC secretion of IL-33. | In vitro | (152) |
| miR-5703 | PC | Binds to CMTM and Promotes cancer progression through the PAK4/PI3K/AKT signaling axis. | Both | (156) |
| NGF | PC | Binds to TrkA and activates PI3K/AKT/GSK to promote MMP9, vimentin and E-cadherin secretion. | In vitro | (157) |
| Wnt/TnC | PC | Promotes tumor proliferation and metastasis by activating β-catenin and YAP/TAZ signaling axes. | Both | (158) |
| HIF-1 | PC | Inducing the secretion of CCL2 by cancer cells recruited macrophages to promote the expression of miR-4465 and miR-616-3p by PSC. | In vitro | (164) |
| NRF2 | PC | Activated PSC promotes NFR2 expression in cancer cells and reduces oxidative stress. | Both | (168,169) |
PSCs, pancreatic stellate cells; ROS, reactive oxygen species; α-SMA, α-smooth muscle actin; AP, acute pancreatitis; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; CP, chronic pancreatitis; Col3:1, collagen type IX alpha 3 chain; ULK1, Unc-51 like autophagy activating kinase 1; RB1CC1, RB1 inducible coiled-coil 1; VLDLR, very low density lipoprotein receptor; miR, microRNA; PC, pancreatic cancer; PAK4, P21 (RAC1) activated kinase 4; NGF, nerve growth factor; TrkA, neurotrophic receptor tyrosine kinase; MMP, matrix metalloproteinase; TnC, tenascin c; YAP, yes1 associated transcriptional regulator; TAZ, WW domain containing transcription regulator 1; HIF, hypoxia-inducible factor; CCL2, C-C motif chemokine ligand 2; NRF2, NFE2 like BZIP transcription factor 2.
5. Therapeutic applications targeting PSCs
Targeting PSCs' activation
The activation of PSCs is a double-edged sword, and regulation of the transition from the resting state to the active state of PSCs depends on the state of the organ or tissue itself. When the pancreas is damaged, PSCs can be activated and produce numerous ECM components, including fibrin, to reduce body damage. Therefore, factors related to the activation of PSCs will become targets for the repair of pancreatic damage, including IL-10 and TGF-β (173). However, when pancreatic diseases occur, the activation of PSCs forms a vicious cycle. Therefore, inhibition of the activation of PSCs becomes the key to treating pancreatic diseases, and the aforementioned inhibitors of PSCs activation-related molecular targets are an effective choice. Additionally, traditional Chinese medicine plays an important role. For example, Resveratrol can inhibit intracellular ROS and reduce the fibrosis of CP by inhibiting the expression of miR-21 and the activation of PSCs (174). Rhein can mediate NF-κB and STAT3 signaling pathways to reduce the activation of PSCs in pancreatic fibrosis (175). Additionally, radiotherapy resistance against pancreatic malignancies is closely related to the massive activation of PSCs after radiation irradiation (176). The potential of combining activation inhibitors of PSCs with radiation as a means to sensitize PC requires further investigation.
Targeting proliferation of PSCs
It is well known that when PSC is activated, it will proliferate, accompanied by cell migration, increased ECM deposition and cytokine secretion. Therefore, when the activation of PSCs is uncontrolled, inhibition of PSC proliferation activity can also regulate the fibrosis process of the disease. Melatonin extracted from the pineal gland has been widely studied for its role in regulating inflammation and tumors. It is involved in regulating cell cycle and differentiation. With the participation of melatonin, the signaling pathway involved in the proliferation of PSCs (PI3K/Akt/mTOR and MAPK) is inhibited. Furthermore, mitochondrial function in PSCs is inhibited, energy generation is blocked, and proliferation of PSCs is ultimately inhibited (177). In conclusion, glucose metabolism, as the main way of energy production, will also become the direction of exploration for regulating PSCs' proliferation. Moreover, with the rise of pancreatic malignant tumors, tumor metabolism has become a focus of future research and the exploration of metabolic crosstalk between PSCs and cancer cells will be the focus of tumor treatment in the future.
Targeting of ECM
Excessive secretion of ECM is an important reason for the deterioration of pancreatic diseases. Especially for PC, the ECM is a barrier to protecting cancer cells from invasion, leading to poor therapeutic effects of molecular target drugs and chemotherapy drugs. However, tumor cells can secrete MMP to dissolve ECM to facilitate cancer cell migration and invasion. This adaptive regulation of tumors makes treatment more difficult. Moreover, under the influence of an anoxic microenvironment, the ECM structure secreted by PSCs can be remodeled to adapt to the migration of cancer cells (178). However, fibronectin in ECM can increase the resistance of PC cells to gemcitabine by activating the ERK1/2 signaling pathway, resulting in worse clinical prognosis for patients (179). Hyaluronan, one of the main components of the gelatinous matrix of tumor ECM, has been revealed to hydrolyze the important structure of the hyaluronidase skeleton of the ECM, improve the absorption of drugs at the tumor site, and recruit more T lymphocytes for antitumor effects (180). Simultaneously, if Hyaluronan is combined with an immune checkpoint, including PD-1/PD-L1, inhibitors may provide favorable results, further preclinical or clinical trials may be warranted. This provides a new option for treating pancreatic diseases, particularly PC.
6. Summary and prospect
Recently, as the function of PSCs has been gradually revealed, it is increasingly understood that PSCs play an important role in the normal repair of pancreatic injury and are also closely related to pancreatic diseases. In the present review, it was revealed that multiple intracellular and extracellular signals are closely associated with PSCs' activation, which contributes to the understanding of the critical role that activated PSCs play in pancreatic disease development and injury repair. Targeted regulation of these signals is conducive to restoring or inhibiting the activation of PSCs, and becomes an important strategy for pancreatic injury repair and treatment of pancreatic-related diseases.
Nevertheless, there are still numerous questions that have not been elucidated clearly, including how PSCs differentiate into stem cells with differentiation ability to participate in pancreatic injury repair and whether other components in the progression of pancreatic inflammation are also involved in the regulation between PSCs and acinar cells, including endothelial cells and microbiota, and how exogenous stimuli such as radiation affect the activation and proliferation of PSCs. Moreover, the activation of PSCs in chronic inflammation of the pancreas or in progressive cancer blocks the targeted penetration of drugs, leading to the occurrence of therapeutic resistance, especially in PC. Elucidating the crosstalk between stromal heterogeneity and tumor cells is an important way to address drug resistance. In addition, the development of inhibitors against key molecular targets where PSCs play a role through multi-omics technology, combined therapy such as immunotherapy, radiation therapy or the development of precision drug delivery systems will be the future direction for PC cure. Moreover, PSCs have a wide range of subgroups and functional heterogeneity due to differences in cell surface markers and functional differences. Reversing the activation of PSCs in specific subgroups can block the secretion of ECM, which is crucial for severing the link between PSCs and pancreatic diseases. In addition, the exosome-mediated information transfer and substance exchange of PSCs reshape the microenvironment of pancreatic disease development, therefore it is of great significance to further explore the key role pathways of specific exosomes and develop corresponding targeted blocking drugs for the treatment of related pancreatic diseases. In conclusion, targeting the pathways associated with PSCs activation may provide new insight for the treatment of pancreatic diseases and pancreatic injury.
Acknowledgements
Not applicable.
Glossary
Abbreviations
- PSCs
pancreatic stellate cells
- IL
interleukin
- TNF-α
tumor necrosis factor-α
- TGFβ1
transforming growth factor β1
- PC
pancreatic cancer
- α-SMA
α-smooth muscle actin
- ATRA
all trans-retinoic acid
- CP
chronic pancreatitis
- ACP
alcoholic CP
- CTGF
connective tissue growth factor
- Arl4c
ADP-ribosylation factor like-4c
- SHH
Sonic Hedgehog
- MMP
matrix metalloproteinase
- NGF
nerve growth factor
- EMT
epithelial-mesenchymal transition
- HIF
hypoxia-inducible factor
- PDGF
platelet derived growth factor
- EGCG
epigallocatechin-3-gallate
- ERK
extracellular regulated kinases
- ROS
reactive oxygen species
- FMOD
fibromodulin
- PI3K
phosphatidylinositol-3-kinase
- AKT
protein kinase B
- PAC
pancreatic acinic cell
- MAPK
mitogen-activated protein kinase
- PPAR
peroxisome proliferator-activated receptor
- NF-κB
nuclear factor κB
- CoQ10
coenzyme Q10
- SSd
Saikosaponin d
- CO
carbon monoxide
- Hsp90
heat shock protein
- JAK
Janus kinase
- STAT
signal transducers and activators of transcription
- CAF
cancer-associated fibroblasts
- JNK
c-Jun N-terminal kinase
- RB
retinoblastoma
- S100A4
S100 calcium binding protein A4
- CnP
Conophylline
- Hic-5
hydrogen peroxide-inducible clone 5
- TnC
tenascin C
- NDRG1
N-myc downstream-regulated gene-1
- DpC
di-2-pyridylketone-4-cyclohexyl-4-methyl-3-thiosemicarbazone
Funding Statement
The present study was supported by Natural Science Foundation of Gansu Province (grant no. 22JR5RA901).
Availability of data and materials
Not applicable.
Authors' contributions
ZW wrote, visualized and prepared the original draft. SD wrote, reviewed and supervised the editing process. WZ wrote and prepared the original draft, supervised and administrated this project and acquired funding. Data authentication is not applicable. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Wang Y, Li HT, Liu G, Jiang CS, Ni YH, Zeng JH, Lin X, Wang QY, Li DZ, Wang W, Zeng XP. COMP promotes pancreatic fibrosis by activating pancreatic stellate cells through CD36-ERK/AKT signaling pathways. Cell Signal. 2024;118:111135. doi: 10.1016/j.cellsig.2024.111135. [DOI] [PubMed] [Google Scholar]
- 2.Apte MV, Pirola RC, Wilson JS. Pancreatic stellate cells: A starring role in normal and diseased pancreas. Front Physiol. 2012;3:344. doi: 10.3389/fphys.2012.00344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Omary MB, Lugea A, Lowe AW, Pandol SJ. The pancreatic stellate cell: A star on the rise in pancreatic diseases. J Clin Invest. 2007;117:50–59. doi: 10.1172/JCI30082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Apte M, Pirola R, Wilson J. The fibrosis of chronic pancreatitis: New insights into the role of pancreatic stellate cells. Antioxid Redox Signal. 2011;15:2711–2722. doi: 10.1089/ars.2011.4079. [DOI] [PubMed] [Google Scholar]
- 5.Erkan M, Adler G, Apte MV, Bachem MG, Buchholz M, Detlefsen S, Esposito I, Friess H, Gress TM, Habisch HJ, et al. StellaTUM: Current consensus and discussion on pancreatic stellate cell research. Gut. 2012;61:172–178. doi: 10.1136/gutjnl-2011-301220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ulmasov B, Neuschwander-Tetri BA, Lai J, Monastyrskiy V, Bhat T, Yates MP, Oliva J, Prinsen MJ, Ruminski PG, Griggs DW. Inhibitors of Arg-Gly-Asp-Binding integrins reduce development of pancreatic fibrosis in mice. Cell Mol Gastroenterol Hepatol. 2016;2:499–518. doi: 10.1016/j.jcmgh.2016.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Masamune A, Shimosegawa T. Pancreatic stellate cells-multi-functional cells in the pancreas. Pancreatology. 2013;13:102–105. doi: 10.1016/j.pan.2012.12.058. [DOI] [PubMed] [Google Scholar]
- 8.Murray ER, Menezes S, Henry JC, Williams JL, Alba-Castellón L, Baskaran P, Quétier I, Desai A, Marshall JJT, Rosewell I, et al. Disruption of pancreatic stellate cell myofibroblast phenotype promotes pancreatic tumor invasion. Cell Rep. 2022;38:110227. doi: 10.1016/j.celrep.2021.110227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yu L, Li JJ, Liang XL, Wu H, Liang Z. PSME3 Promotes TGFB1 secretion by PC cells to induce pancreatic stellate cell proliferation. J Cancer. 2019;10:2128–2138. doi: 10.7150/jca.30235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Derynck R, Budi EH. Specificity, versatility, and control of TGF-β family signaling. Sci Signal. 2019;12:eaav5183. doi: 10.1126/scisignal.aav5183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gough NR, Xiang X, Mishra L. TGF-β signaling in liver, pancreas, and gastrointestinal diseases and cancer. Gastroenterology. 2021;161:434–452.e15. doi: 10.1053/j.gastro.2021.04.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu B, Zhang S, Guo Z, Bi Y, Zhou M, Li P, Seyedsadr M, Xu X, Li JL, Markovic-Plese S, Wan YY. The TGF-β superfamily cytokine Activin-A is induced during autoimmune neuroinflammation and drives pathogenic Th17 cell differentiation. Immunity. 2021;54:308–323.e6. doi: 10.1016/j.immuni.2020.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Apte MV, Haber PS, Darby SJ, Rodgers SC, McCaughan GW, Korsten MA, Pirola RC, Wilson JS. Pancreatic stellate cells are activated by proinflammatory cytokines: Implications for pancreatic fibrogenesis. Gut. 1999;44:534–541. doi: 10.1136/gut.44.4.534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee H, Lim C, Lee J, Kim N, Bang S, Lee H, Min B, Park G, Noda M, Stetler-Stevenson WG, Oh J. TGF-beta signaling preserves RECK expression in activated pancreatic stellate cells. J Cell Biochem. 2008;104:1065–1074. doi: 10.1002/jcb.21692. [DOI] [PubMed] [Google Scholar]
- 15.Shek FW, Benyon RC, Walker FM, McCrudden PR, Pender SL, Williams EJ, Johnson PA, Johnson CD, Bateman AC, Fine DR, Iredale JP. Expression of transforming growth factor-beta 1 by pancreatic stellate cells and its implications for matrix secretion and turnover in chronic pancreatitis. Am J Pathol. 2002;160:1787–1798. doi: 10.1016/S0002-9440(10)61125-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Apte MV, Wilson JS. Dangerous liaisons: Pancreatic stellate cells and pancreatic cancer cells. J Gastroenterol Hepatol. 2012;27((Suppl 2)):S69–S74. doi: 10.1111/j.1440-1746.2011.07000.x. [DOI] [PubMed] [Google Scholar]
- 17.Sarper M, Cortes E, Lieberthal TJ, Del Río Hernández A. ATRA modulates mechanical activation of TGF-β by pancreatic stellate cells. Sci Rep. 2016;6:27639. doi: 10.1038/srep27639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zheng M, Li H, Sun L, Brigstock DR, Gao R. Interleukin-6 participates in human pancreatic stellate cell activation and collagen I production via TGF-β1/Smad pathway. Cytokine. 2021;143:155536. doi: 10.1016/j.cyto.2021.155536. [DOI] [PubMed] [Google Scholar]
- 19.Shen YW, Zhou YD, Chen HZ, Luan X, Zhang WD. Targeting CTGF in cancer: An emerging therapeutic opportunity. Trends Cancer. 2021;7:511–524. doi: 10.1016/j.trecan.2020.12.001. [DOI] [PubMed] [Google Scholar]
- 20.Huang G, Brigstock DR. Regulation of hepatic stellate cells by connective tissue growth factor. Front Biosci (Landmark Ed) 2012;17:2495–2507. doi: 10.2741/4067. [DOI] [PubMed] [Google Scholar]
- 21.di Mola FF, Friess H, Riesle E, Koliopanos A, Büchler P, Zhu Z, Brigstock DR, Korc M, Büchler MW. Connective tissue growth factor is involved in pancreatic repair and tissue remodeling in human and rat acute necrotizing pancreatitis. Ann Surg. 2002;235:60–67. doi: 10.1097/00000658-200201000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.di Mola FF, Friess H, Martignoni ME, Di Sebastiano P, Zimmermann A, Innocenti P, Graber H, Gold LI, Korc M, Büchler MW. Connective tissue growth factor is a regulator for fibrosis in human chronic pancreatitis. Ann Surg. 1999;230:63–71. doi: 10.1097/00000658-199907000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wenger C, Ellenrieder V, Alber B, Lacher U, Menke A, Hameister H, Wilda M, Iwamura T, Beger HG, Adler G, Gress TM. Expression and differential regulation of connective tissue growth factor in pancreatic cancer cells. Oncogene. 1999;18:1073–1080. doi: 10.1038/sj.onc.1202395. [DOI] [PubMed] [Google Scholar]
- 24.Gao R, Brigstock DR. A novel integrin alpha5beta1 binding domain in module 4 of connective tissue growth factor (CCN2/CTGF) promotes adhesion and migration of activated pancreatic stellate cells. Gut. 2006;55:856–862. doi: 10.1136/gut.2005.079178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Charrier A, Chen R, Chen L, Kemper S, Hattori T, Takigawa M, Brigstock DR. Connective tissue growth factor (CCN2) and microRNA-21 are components of a positive feedback loop in pancreatic stellate cells (PSCs) during chronic pancreatitis and are exported in PSCs-derived exosomes. J Cell Commun Signal. 2014;8:147–156. doi: 10.1007/s12079-014-0220-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tamura T, Kodama T, Sato K, Murai K, Yoshioka T, Shigekawa M, Yamada R, Hikita H, Sakamori R, Akita H, et al. Dysregulation of PI3K and Hippo signaling pathways synergistically induces chronic pancreatitis via CTGF upregulation. J Clin Invest. 2021;131:e143414. doi: 10.1172/JCI143414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen X, Zhang Y, Qian W, Han L, Li W, Duan W, Wu Z, Wang Z, Ma Q. Arl4c promotes the growth and drug resistance of pancreatic cancer by regulating tumor-stromal interactions. iScience. 2021;24:103400. doi: 10.1016/j.isci.2021.103400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Qi X, Li X. Mechanistic insights into the generation and transduction of hedgehog signaling. Trends Biochem Sci. 2020;45:397–410. doi: 10.1016/j.tibs.2020.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bausch D, Fritz S, Bolm L, Wellner UF, Fernandez-Del-Castillo C, Warshaw AL, Thayer SP, Liss AS. Hedgehog signaling promotes angiogenesis directly and indirectly in pancreatic cancer. Angiogenesis. 2020;23:479–492. doi: 10.1007/s10456-020-09725-x. [DOI] [PubMed] [Google Scholar]
- 30.Tang D, Wang D, Yuan Z, Xue X, Zhang Y, An Y, Chen J, Tu M, Lu Z, Wei J, et al. Persistent activation of pancreatic stellate cells creates a microenvironment favorable for the malignant behavior of pancreatic ductal adenocarcinoma. Int J Cancer. 2013;132:993–1003. doi: 10.1002/ijc.27715. [DOI] [PubMed] [Google Scholar]
- 31.Olive KP, Jacobetz MA, Davidson CJ, Gopinathan A, McIntyre D, Honess D, Madhu B, Goldgraben MA, Caldwell ME, Allard D, et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324:1457–1461. doi: 10.1126/science.1171362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li X, Wang Z, Ma Q, Xu Q, Liu H, Duan W, Lei J, Ma J, Wang X, Lv S, et al. Sonic hedgehog paracrine signaling activates stromal cells to promote perineural invasion in pancreatic cancer. Clin Cancer Res. 2014;20:4326–4338. doi: 10.1158/1078-0432.CCR-13-3426. [DOI] [PubMed] [Google Scholar]
- 33.Yu Y, Cheng L, Yan B, Zhou C, Qian W, Xiao Y, Qin T, Cao J, Han L, Ma Q, Ma J. Overexpression of Gremlin 1 by sonic hedgehog signaling promotes pancreatic cancer progression. Int J Oncol. 2018;53:2445–2457. doi: 10.3892/ijo.2018.4573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hwang RF, Moore TT, Hattersley MM, Scarpitti M, Yang B, Devereaux E, Ramachandran V, Arumugam T, Ji B, Logsdon CD, et al. Inhibition of the hedgehog pathway targets the tumor-associated stroma in pancreatic cancer. Mol Cancer Res. 2012;10:1147–1157. doi: 10.1158/1541-7786.MCR-12-0022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Riera-Domingo C, Audigé A, Granja S, Cheng WC, Ho PC, Baltazar F, Stockmann C, Mazzone M. Immunity, Hypoxia, and Metabolism-the Ménage à trois of cancer: Implications for immunotherapy. Physiol Rev. 2020;100:1–102. doi: 10.1152/physrev.00018.2019. [DOI] [PubMed] [Google Scholar]
- 36.Liu M, Zhong J, Zeng Z, Huang K, Ye Z, Deng S, Chen H, Xu F, Li Q, Zhao G. Hypoxia-induced feedback of HIF-1α and lncRNA-CF129 contributes to pancreatic cancer progression through stabilization of p53 protein. Theranostics. 2019;9:4795–4810. doi: 10.7150/thno.30988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shi X, Wang M, Zhang Y, Guo X, Liu M, Zhou Z, Zhao Y, He R, Gao Y, Liu Y, et al. Hypoxia activated HGF expression in pancreatic stellate cells confers resistance of pancreatic cancer cells to EGFR inhibition. EBioMedicine. 2022;86:104352. doi: 10.1016/j.ebiom.2022.104352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Estaras M, Gonzalez-Portillo MR, Martinez R, Garcia A, Estevez M, Fernandez-Bermejo M, Mateos JM, Vara D, Blanco-Fernández G, Lopez-Guerra D, et al. Melatonin modulates the antioxidant defenses and the expression of proinflammatory mediators in pancreatic stellate cells subjected to hypoxia. Antioxidants (Basel) 2021;10:577. doi: 10.3390/antiox10040577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Masamune A, Kikuta K, Watanabe T, Satoh K, Hirota M, Shimosegawa T. Hypoxia stimulates pancreatic stellate cells to induce fibrosis and angiogenesis in pancreatic cancer. Am J Physiol Gastrointest Liver Physiol. 2008;295:G709–G717. doi: 10.1152/ajpgi.90356.2008. [DOI] [PubMed] [Google Scholar]
- 40.Xiao Y, Qin T, Sun L, Qian W, Li J, Duan W, Lei J, Wang Z, Ma J, Li X, et al. Resveratrol ameliorates the malignant progression of pancreatic cancer by inhibiting Hypoxia-induced pancreatic stellate cell activation. Cell Transplant. 2020;29:963689720929987. doi: 10.1177/0963689720929987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zou X, Tang XY, Qu ZY, Sun ZW, Ji CF, Li YJ, Guo SD. Targeting the PDGF/PDGFR signaling pathway for cancer therapy: A review. Int J Biol Macromol. 2022;202:539–557. doi: 10.1016/j.ijbiomac.2022.01.113. [DOI] [PubMed] [Google Scholar]
- 42.Hu R, Wang YL, Edderkaoui M, Lugea A, Apte MV, Pandol SJ. Ethanol augments PDGF-induced NADPH oxidase activity and proliferation in rat pancreatic stellate cells. Pancreatology. 2007;7:332–340. doi: 10.1159/000105499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Masamune A, Suzuki N, Kikuta K, Satoh M, Satoh K, Shimosegawa T. Curcumin blocks activation of pancreatic stellate cells. J Cell Biochem. 2006;97:1080–1093. doi: 10.1002/jcb.20698. [DOI] [PubMed] [Google Scholar]
- 44.Zhao T, Xiao D, Jin F, Sun X, Yu J, Wang H, Liu J, Cai W, Huang C, Wang X, et al. ESE3-positive PSCs drive pancreatic cancer fibrosis, chemoresistance and poor prognosis via tumour-stromal IL-1β/NF-κB/ESE3 signalling axis. Br J Cancer. 2022;127:1461–1472. doi: 10.1038/s41416-022-01927-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Guo HL, Liang XS, Zeng XP, Liu Y, Li ZS, Wang LJ, Hu LH. Pirfenidone alleviates chronic pancreatitis via suppressing the activation of pancreatic stellate cells and the M1 polarization of macrophages. Int Immunopharmacol. 2024;130:111691. doi: 10.1016/j.intimp.2024.111691. [DOI] [PubMed] [Google Scholar]
- 46.Masamune A, Kikuta K, Satoh M, Suzuki N, Shimosegawa T. Green tea polyphenol epigallocatechin-3-gallate blocks PDGF-induced proliferation and migration of rat pancreatic stellate cells. World J Gastroenterol. 2005;11:3368–3374. doi: 10.3748/wjg.v11.i22.3368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ouyang W, O'Garra A. IL-10 family cytokines IL-10 and IL-22: From basic science to clinical translation. Immunity. 2019;50:871–891. doi: 10.1016/j.immuni.2019.03.020. [DOI] [PubMed] [Google Scholar]
- 48.Zhang LJ, Zheng WD, Shi MN, Wang XZ. Effects of interleukin-10 on activation and apoptosis of hepatic stellate cells in fibrotic rat liver. World J Gastroenterol. 2006;12:1918–1923. doi: 10.3748/wjg.v12.i12.1918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Shi MN, Huang YH, Zheng WD, Zhang LJ, Chen ZX, Wang XZ. Relationship between transforming growth factor beta1 and anti-fibrotic effect of interleukin-10. World J Gastroenterol. 2006;12:2357–2362. doi: 10.3748/wjg.v12.i15.2357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Demols A, Van Laethem JL, Quertinmont E, Degraef C, Delhaye M, Geerts A, Deviere J. Endogenous interleukin-10 modulates fibrosis and regeneration in experimental chronic pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2002;282:G1105–G1112. doi: 10.1152/ajpgi.00431.2001. [DOI] [PubMed] [Google Scholar]
- 51.Mews P, Phillips P, Fahmy R, Korsten M, Pirola R, Wilson J, Apte M. Pancreatic stellate cells respond to inflammatory cytokines: Potential role in chronic pancreatitis. Gut. 2002;50:535–541. doi: 10.1136/gut.50.4.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Talukdar R, Tandon RK. Pancreatic stellate cells: New target in the treatment of chronic pancreatitis. J Gastroenterol Hepatol. 2008;23:34–41. doi: 10.1111/j.1440-1746.2007.05206.x. [DOI] [PubMed] [Google Scholar]
- 53.Marzoq AJ, Mustafa SA, Heidrich L, Hoheisel JD, Alhamdani MSS. Impact of the secretome of activated pancreatic stellate cells on growth and differentiation of pancreatic tumour cells. Sci Rep. 2019;9:5303. doi: 10.1038/s41598-019-41740-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kandikattu HK, Venkateshaiah SU, Mishra A. Chronic pancreatitis and the development of pancreatic cancer. Endocr Metab Immune Disord Drug Targets. 2020;20:1182–1210. doi: 10.2174/1871530320666200423095700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lugea A, Waldron RT. Exosome-mediated intercellular communication between stellate cells and cancer cells in pancreatic ductal adenocarcinoma. Pancreas. 2017;46:1–4. doi: 10.1097/MPA.0000000000000686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hu F, Lou N, Jiao J, Guo F, Xiang H, Shang D. Macrophages in pancreatitis: Mechanisms and therapeutic potential. Biomed Pharmacother. 2020;131:110693. doi: 10.1016/j.biopha.2020.110693. [DOI] [PubMed] [Google Scholar]
- 57.Ng B, Viswanathan S, Widjaja AA, Lim WW, Shekeran SG, Goh JWT, Tan J, Kuthubudeen F, Lim SY, Xie C, et al. IL11 Activates pancreatic stellate cells and causes pancreatic inflammation, fibrosis and atrophy in a mouse model of pancreatitis. Int J Mol Sci. 2022;23:3549. doi: 10.3390/ijms23073549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Filomeni G, De Zio D, Cecconi F. Oxidative stress and autophagy: The clash between damage and metabolic needs. Cell Death Differ. 2015;22:377–388. doi: 10.1038/cdd.2014.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ryu GR, Lee E, Chun HJ, Yoon KH, Ko SH, Ahn YB, Song KH. Oxidative stress plays a role in high glucose-induced activation of pancreatic stellate cells. Biochem Biophys Res Commun. 2013;439:258–263. doi: 10.1016/j.bbrc.2013.08.046. [DOI] [PubMed] [Google Scholar]
- 60.Zhang J, Bai J, Zhou Q, Hu Y, Wang Q, Yang L, Chen H, An H, Zhou C, Wang Y, et al. Glutathione prevents high glucose-induced pancreatic fibrosis by suppressing pancreatic stellate cell activation via the ROS/TGFβ/SMAD pathway. Cell Death Dis. 2022;13:440. doi: 10.1038/s41419-022-04894-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.An W, Zhu JW, Jiang F, Jiang H, Zhao JL, Liu MY, Li GX, Shi XG, Sun C, Li ZS. Fibromodulin is upregulated by oxidative stress through the MAPK/AP-1 pathway to promote pancreatic stellate cell activation. Pancreatology. 2020;20:278–287. doi: 10.1016/j.pan.2019.09.011. [DOI] [PubMed] [Google Scholar]
- 62.Xia B, Gao J, Li S, Huang L, Zhu L, Ma T, Zhao L, Yang Y, Luo K, Shi X, et al. Mechanical stimulation of Schwann cells promote peripheral nerve regeneration via extracellular vesicle-mediated transfer of microRNA 23b-3p. Theranostics. 2020;10:8974–8995. doi: 10.7150/thno.44912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Asaumi H, Watanabe S, Taguchi M, Tashiro M, Otsuki M. Externally applied pressure activates pancreatic stellate cells through the generation of intracellular reactive oxygen species. Am J Physiol Gastrointest Liver Physiol. 2007;293:G972–G978. doi: 10.1152/ajpgi.00018.2007. [DOI] [PubMed] [Google Scholar]
- 64.Radoslavova S, Fels B, Pethö Z, Gruner M, Ruck T, Meuth SG, Folcher A, Prevarskaya N, Schwab A, Ouadid-Ahidouch H. TRPC1 channels regulate the activation of pancreatic stellate cells through ERK1/2 and SMAD2 pathways and perpetuate their pressure-mediated activation. Cell Calcium. 2022;106:102621. doi: 10.1016/j.ceca.2022.102621. [DOI] [PubMed] [Google Scholar]
- 65.Kalli M, Papageorgis P, Gkretsi V, Stylianopoulos T. Solid stress facilitates fibroblasts activation to promote pancreatic cancer cell migration. Ann Biomed Eng. 2018;46:657–669. doi: 10.1007/s10439-018-1997-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pethő Z, Najder K, Bulk E, Schwab A. Mechanosensitive ion channels push cancer progression. Cell Calcium. 2019;80:79–90. doi: 10.1016/j.ceca.2019.03.007. [DOI] [PubMed] [Google Scholar]
- 67.Martinac B. Mechanosensitive ion channels: Molecules of mechanotransduction. J Cell Sci. 2004;117:2449–2460. doi: 10.1242/jcs.01232. [DOI] [PubMed] [Google Scholar]
- 68.Nielsen N, Kondratska K, Ruck T, Hild B, Kovalenko I, Schimmelpfennig S, Welzig J, Sargin S, Lindemann O, Christian S, et al. TRPC6 channels modulate the response of pancreatic stellate cells to hypoxia. Pflugers Arch. 2017;469:1567–1577. doi: 10.1007/s00424-017-2057-0. [DOI] [PubMed] [Google Scholar]
- 69.Jakubowska MA, Ferdek PE, Gerasimenko OV, Gerasimenko JV, Petersen OH. Nitric oxide signals are interlinked with calcium signals in normal pancreatic stellate cells upon oxidative stress and inflammation. Open Biol. 2016;6:160149. doi: 10.1098/rsob.160149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hennigs JK, Seiz O, Spiro J, Berna MJ, Baumann HJ, Klose H, Pace A. Molecular basis of P2-receptor-mediated calcium signaling in activated pancreatic stellate cells. Pancreas. 2011;40:740–746. doi: 10.1097/MPA.0b013e31821b5b68. [DOI] [PubMed] [Google Scholar]
- 71.Jiang T, Wang H, Liu L, Song H, Zhang Y, Wang J, Liu L, Xu T, Fan R, Xu Y, et al. CircIL4R activates the PI3K/AKT signaling pathway via the miR-761/TRIM29/PHLPP1 axis and promotes proliferation and metastasis in colorectal cancer. Mol Cancer. 2021;20:167. doi: 10.1186/s12943-021-01474-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Liu B, Deng X, Jiang Q, Li G, Zhang J, Zhang N, Xin S, Xu K. Scoparone improves hepatic inflammation and autophagy in mice with nonalcoholic steatohepatitis by regulating the ROS/P38/Nrf2 axis and PI3K/AKT/mTOR pathway in macrophages. Biomed Pharmacother. 2020;125:109895. doi: 10.1016/j.biopha.2020.109895. [DOI] [PubMed] [Google Scholar]
- 73.Xue R, Wang J, Yang L, Liu X, Gao Y, Pang Y, Wang Y, Hao J. Coenzyme Q10 ameliorates pancreatic fibrosis via the ROS-Triggered mTOR signaling pathway. Oxid Med Cell Longev. 2019;2019:8039694. doi: 10.1155/2019/8039694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Cui LH, Li CX, Zhuo YZ, Yang L, Cui NQ, Zhang SK. Saikosaponin d ameliorates pancreatic fibrosis by inhibiting autophagy of pancreatic stellate cells via PI3K/Akt/mTOR pathway. Chem Biol Interact. 2019;300:18–26. doi: 10.1016/j.cbi.2019.01.005. [DOI] [PubMed] [Google Scholar]
- 75.Schwer CI, Stoll P, Rospert S, Fitzke E, Schallner N, Bürkle H, Schmidt R, Humar M. Carbon monoxide releasing molecule-2 CORM-2 represses global protein synthesis by inhibition of eukaryotic elongation factor eEF2. Int J Biochem Cell Biol. 2013;45:201–212. doi: 10.1016/j.biocel.2012.09.020. [DOI] [PubMed] [Google Scholar]
- 76.Zhang P, Guan H, Yuan S, Cheng H, Zheng J, Zhang Z, Liu Y, Yu Y, Meng Z, Zheng X, Zhao L. Targeting myeloid derived suppressor cells reverts immune suppression and sensitizes BRAF-mutant papillary thyroid cancer to MAPK inhibitors. Nat Commun. 2022;13:1588. doi: 10.1038/s41467-022-29000-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zou ZL, Sun MH, Yin WF, Yang L, Kong LY. Avicularin suppresses cartilage extracellular matrix degradation and inflammation via TRAF6/MAPK activation. Phytomedicine. 2021;91:153657. doi: 10.1016/j.phymed.2021.153657. [DOI] [PubMed] [Google Scholar]
- 78.Zhang Y, Ware MB, Zaidi MY, Ruggieri AN, Olson BM, Komar H, Farren MR, Nagaraju GP, Zhang C, Chen Z, et al. Heat shock Protein-90 inhibition alters activation of pancreatic stellate cells and enhances the efficacy of PD-1 blockade in pancreatic cancer. Mol Cancer Ther. 2021;20:150–160. doi: 10.1158/1535-7163.MCT-19-0911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.McCarroll JA, Phillips PA, Santucci N, Pirola RC, Wilson JS, Apte MV. Vitamin A inhibits pancreatic stellate cell activation: Implications for treatment of pancreatic fibrosis. Gut. 2006;55:79–89. doi: 10.1136/gut.2005.064543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Roach KM, Bradding P. Ca2+ signalling in fibroblasts and the therapeutic potential of KCa3.1 channel blockers in fibrotic diseases. Br J Pharmacol. 2020;177:1003–1024. doi: 10.1111/bph.14939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Murphy-Ullrich JE, Suto MJ. Thrombospondin-1 regulation of latent TGF-β activation: A therapeutic target for fibrotic disease. Matrix Biol. 2018:68–69. 28–43. doi: 10.1016/j.matbio.2017.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Itatani Y, Kawada K, Sakai Y. Transforming Growth Factor-β signaling pathway in colorectal cancer and its tumor microenvironment. Int J Mol Sci. 2019;20:5822. doi: 10.3390/ijms20235822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kanda T, Jiang X, Yokosuka O. Androgen receptor signaling in hepatocellular carcinoma and pancreatic cancers. World J Gastroenterol. 2014;20:9229–9236. doi: 10.3748/wjg.v20.i28.9229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zhang X, Yun JS, Han D, Yook JI, Kim HS, Cho ES. TGF-β pathway in salivary gland fibrosis. Int J Mol Sci. 2020;21:9138. doi: 10.3390/ijms21239138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ren Y, Zhang J, Wang M, Bi J, Wang T, Qiu M, Lv Y, Wu Z, Wu R. Identification of irisin as a therapeutic agent that inhibits oxidative stress and fibrosis in a murine model of chronic pancreatitis. Biomed Pharmacother. 2020;126:110101. doi: 10.1016/j.biopha.2020.110101. [DOI] [PubMed] [Google Scholar]
- 86.Colaianni G, Cinti S, Colucci S, Grano M. Irisin and musculoskeletal health. Ann N Y Acad Sci. 2017;1402:5–9. doi: 10.1111/nyas.13345. [DOI] [PubMed] [Google Scholar]
- 87.Petta S, Valenti L, Svegliati-Baroni G, Ruscica M, Pipitone RM, Dongiovanni P, Rychlicki C, Ferri N, Cammà C, Fracanzani AL, et al. Fibronectin Type III Domain-containing protein 5 rs3480 A>G polymorphism, irisin, and liver fibrosis in patients with nonalcoholic fatty liver disease. J Clin Endocrinol Metab. 2017;102:2660–2669. doi: 10.1210/jc.2017-00056. [DOI] [PubMed] [Google Scholar]
- 88.Peng H, Wang Q, Lou T, Qin J, Jung S, Shetty V, Li F, Wang Y, Feng XH, Mitch WE, et al. Myokine mediated muscle-kidney crosstalk suppresses metabolic reprogramming and fibrosis in damaged kidneys. Nat Commun. 2017;8:1493. doi: 10.1038/s41467-017-01646-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chen RR, Fan XH, Chen G, Zeng GW, Xue YG, Liu XT, Wang CY. Irisin attenuates angiotensin II-induced cardiac fibrosis via Nrf2 mediated inhibition of ROS/TGFβ1/Smad2/3 signaling axis. Chem Biol Interact. 2019;302:11–21. doi: 10.1016/j.cbi.2019.01.031. [DOI] [PubMed] [Google Scholar]
- 90.Ren Y, Zhang J, Wang M, Bi J, Wang T, Qiu M, Lv Y, Wu Z, Wu R. Identification of irisin as a therapeutic agent that inhibits oxidative stress and fibrosis in a murine model of chronic pancreatitis. Biomed Pharmacother. 2020;126:110101. doi: 10.1016/j.biopha.2020.110101. [DOI] [PubMed] [Google Scholar]
- 91.Bansod S, Doijad N, Godugu C. Berberine attenuates severity of chronic pancreatitis and fibrosis via AMPK-mediated inhibition of TGF-β1/Smad signaling and M2 polarization. Toxicol Appl Pharmacol. 2020;403:115162. doi: 10.1016/j.taap.2020.115162. [DOI] [PubMed] [Google Scholar]
- 92.Choi JW, Lee SK, Kim MJ, Kim DG, Shin JY, Zhou Z, Jo IJ, Song HJ, Bae GS, Park SJ. Piperine ameliorates the severity of fibrosis via inhibition of TGF-β/SMAD signaling in a mouse model of chronic pancreatitis. Mol Med Rep. 2019;20:3709–3718. doi: 10.3892/mmr.2019.10635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mirza AZ, Althagafi, Shamshad H. Role of PPAR receptor in different diseases and their ligands: Physiological importance and clinical implications. Eur J Med Chem. 2019;166:502–513. doi: 10.1016/j.ejmech.2019.01.067. [DOI] [PubMed] [Google Scholar]
- 94.Christofides A, Konstantinidou E, Jani C, Boussiotis VA. The role of peroxisome proliferator-activated receptors (PPAR) in immune responses. Metabolism. 2021;114:154338. doi: 10.1016/j.metabol.2020.154338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wang Q, Wang H, Jing Q, Yang Y, Xue D, Hao C, Zhang W. Regulation of pancreatic fibrosis by acinar cell-Derived exosomal miR-130a-3p via targeting of stellate cell PPAR-γ. J Inflamm Res. 2021;14:461–477. doi: 10.2147/JIR.S299298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kim N, Choi S, Lim C, Lee H, Oh J. Albumin mediates PPAR-gamma or C/EBP-alpha-induced phenotypic changes in pancreatic stellate cells. Biochem Biophys Res Commun. 2010;391:640–644. doi: 10.1016/j.bbrc.2009.11.112. [DOI] [PubMed] [Google Scholar]
- 97.Che M, Kweon SM, Teo JL, Yuan YC, Melstrom LG, Waldron RT, Lugea A, Urrutia RA, Pandol SJ, Lai KKY. Targeting the CBP/β-Catenin interaction to suppress activation of Cancer-Promoting pancreatic stellate cells. Cancers (Basel) 2020;12:1476. doi: 10.3390/cancers12061476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Yu H, Lin L, Zhang Z, Zhang H, Hu H. Targeting NF-κB pathway for the therapy of diseases: Mechanism and clinical study. Signal Transduct Target Ther. 2020;5:209. doi: 10.1038/s41392-020-00312-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Treiber M, Neuhöfer P, Anetsberger E, Einwächter H, Lesina M, Rickmann M, Liang S, Kehl T, Nakhai H, Schmid RM, Algül H. Myeloid, but not pancreatic, RelA/p65 is required for fibrosis in a mouse model of chronic pancreatitis. Gastroenterology. 2011;141:1473–1485.e14857. doi: 10.1053/j.gastro.2011.06.087. [DOI] [PubMed] [Google Scholar]
- 100.Wu N, Xu XF, Xin JQ, Fan JW, Wei YY, Peng QX, Duan LF, Wang W, Zhang H. The effects of nuclear factor-kappa B in pancreatic stellate cells on inflammation and fibrosis of chronic pancreatitis. J Cell Mol Med. 2021;25:2213–2227. doi: 10.1111/jcmm.16213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chen H, Tan P, Qian B, Du Y, Wang A, Shi H, Huang Z, Huang S, Liang T, Fu W. Hic-5 deficiency protects cerulein-induced chronic pancreatitis via down-regulation of the NF-κB (p65)/IL-6 signalling pathway. J Cell Mol Med. 2020;24:1488–1503. doi: 10.1111/jcmm.14833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Carrière C, Gore AJ, Norris AM, Gunn JR, Young AL, Longnecker DS, Korc M. Deletion of Rb accelerates pancreatic carcinogenesis by oncogenic Kras and impairs senescence in premalignant lesions. Gastroenterology. 2011;141:1091–1101. doi: 10.1053/j.gastro.2011.05.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Sun HZ, Gong XY, Wu L, Wang XX, Nie YN, Shang R, Wang H, Li YC, Sun QF, Gao PF, Bi JX. Hydrogen sulfide modulates gastric acid secretion in rats via involvement of substance P and nuclear factor-κB signaling. J Physiol Pharmacol. 2018:69. doi: 10.26402/jpp.2018.3.08. doi: 10.26402/jpp.2018.3.08. [DOI] [PubMed] [Google Scholar]
- 104.Sundar V, Tamizhselvi R. Inhibition of Rb phosphorylation leads to H2S-mediated inhibition of NF-kB in acute pancreatitis and associated lung injury in mice. Pancreatology. 2020;20:647–658. doi: 10.1016/j.pan.2020.04.011. [DOI] [PubMed] [Google Scholar]
- 105.Nulali J, Zhan M, Zhang K, Tu P, Liu Y, Song H. Osteoglycin: An ECM factor regulating fibrosis and tumorigenesis. Biomolecules. 2022;12:1674. doi: 10.3390/biom12111674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Guan P, Liu C, Xie D, Mao S, Ji Y, Lin Y, Chen Z, Wang Q, Fan L, Sun Y. Exosome-loaded extracellular matrix-mimic hydrogel with anti-inflammatory property Facilitates/promotes growth plate injury repair. Bioact Mater. 2021;10:145–158. doi: 10.1016/j.bioactmat.2021.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Pietilä EA, Gonzalez-Molina J, Moyano-Galceran L, Jamalzadeh S, Zhang K, Lehtinen L, Turunen SP, Martins TA, Gultekin O, Lamminen T, et al. Co-evolution of matrisome and adaptive adhesion dynamics drives ovarian cancer chemoresistance. Nat Commun. 2021;12:3904. doi: 10.1038/s41467-021-24009-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Mosquera MJ, Kim S, Bareja R, Fang Z, Cai S, Pan H, Asad M, Martin ML, Sigouros M, Rowdo FM, et al. Extracellular matrix in synthetic Hydrogel-based prostate cancer organoids regulate therapeutic response to EZH2 and DRD2 inhibitors. Adv Mater. 2022;34:e2100096. doi: 10.1002/adma.202100096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Hwang HJ, Oh MS, Lee DW, Kuh HJ. Multiplex quantitative analysis of stroma-mediated cancer cell invasion, matrix remodeling, and drug response in a 3D co-culture model of pancreatic tumor spheroids and stellate cells. J Exp Clin Cancer Res. 2019;38:258. doi: 10.1186/s13046-019-1225-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sun L, Qu L, Brigstock DR, Li H, Li Y, Gao R. Biological and proteomic characteristics of an immortalized human pancreatic stellate cell line. Int J Med Sci. 2020;17:137–144. doi: 10.7150/ijms.36337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Tanaka S, Hosokawa M, Yonezawa T, Hayashi W, Ueda K, Iwakawa S. Induction of epithelial-mesenchymal transition and down-regulation of miR-200c and miR-141 in oxaliplatin-resistant colorectal cancer cells. Biol Pharm Bull. 2015;38:435–440. doi: 10.1248/bpb.b14-00695. [DOI] [PubMed] [Google Scholar]
- 112.Xu W, Liu H, Liu ZG, Wang HS, Zhang F, Wang H, Zhang J, Chen JJ, Huang HJ, Tan Y, et al. Histone deacetylase inhibitors upregulate Snail via Smad2/3 phosphorylation and stabilization of Snail to promote metastasis of hepatoma cells. Cancer Lett. 2018;420:1–13. doi: 10.1016/j.canlet.2018.01.068. [DOI] [PubMed] [Google Scholar]
- 113.Zou XZ, Liu T, Gong ZC, Hu CP, Zhang Z. MicroRNAs-mediated epithelial-mesenchymal transition in fibrotic diseases. Eur J Pharmacol. 2017;796:190–206. doi: 10.1016/j.ejphar.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 114.Tian L, Lu ZP, Cai BB, Zhao LT, Qian D, Xu QC, Wu PF, Zhu Y, Zhang JJ, Du Q, et al. Activation of pancreatic stellate cells involves an EMT-like process. Int J Oncol. 2016;48:783–792. doi: 10.3892/ijo.2015.3282. [DOI] [PubMed] [Google Scholar]
- 115.Ling J, Kang Y, Zhao R, Xia Q, Lee DF, Chang Z, Li J, Peng B, Fleming JB, Wang H, et al. KrasG12D-induced IKK2/β/NF-κB activation by IL-1α and p62 feedforward loops is required for development of pancreatic ductal adenocarcinoma. Cancer Cell. 2012;21:105–120. doi: 10.1016/j.ccr.2011.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Masamune A, Yoshida N, Hamada S, Takikawa T, Nabeshima T, Shimosegawa T. Exosomes derived from pancreatic cancer cells induce activation and profibrogenic activities in pancreatic stellate cells. Biochem Biophys Res Commun. 2018;495:71–77. doi: 10.1016/j.bbrc.2017.10.141. [DOI] [PubMed] [Google Scholar]
- 117.Zhang YF, Zhou YZ, Zhang B, Huang SF, Li PP, He XM, Cao GD, Kang MX, Dong X, Wu YL. Pancreatic cancer-derived exosomes promoted pancreatic stellate cells recruitment by pancreatic cancer. J Cancer. 2019;10:4397–4407. doi: 10.7150/jca.27590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Xu M, Wang G, Zhou H, Cai J, Li P, Zhou M, Lu Y, Jiang X, Huang H, Zhang Y, Gong A. TGF-β1-miR-200a-PTEN induces epithelial-mesenchymal transition and fibrosis of pancreatic stellate cells. Mol Cell Biochem. 2017;431:161–168. doi: 10.1007/s11010-017-2988-y. [DOI] [PubMed] [Google Scholar]
- 119.Xue J, Sharma V, Hsieh MH, Chawla A, Murali R, Pandol SJ, Habtezion A. Alternatively activated macrophages promote pancreatic fibrosis in chronic pancreatitis. Nat Commun. 2015;6:7158. doi: 10.1038/ncomms8158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Charrier A, Chen R, Kemper S, Brigstock DR. Regulation of pancreatic inflammation by connective tissue growth factor (CTGF/CCN2) Immunology. 2014;141:564–576. doi: 10.1111/imm.12215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Neuzillet C, Tijeras-Raballand A, Ragulan C, Cros J, Patil Y, Martinet M, Erkan M, Kleeff J, Wilson J, Apte M, et al. Inter- and intra-tumoural heterogeneity in cancer-associated fibroblasts of human pancreatic ductal adenocarcinoma. J Pathol. 2019;248:51–65. doi: 10.1002/path.5224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Orecchioni M, Ghosheh Y, Pramod AB, Ley K. Macrophage polarization: Different gene signatures in M1(LPS+) vs. Classically and M2(LPS-) vs. Alternatively activated macrophages. Front Immunol. 2019;10:1084. doi: 10.3389/fimmu.2019.01084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Dey S, Udari LM, RiveraHernandez P, Kwon JJ, Willis B, Easler JJ, Fogel EL, Pandol S, Kota J. Loss of miR-29a/b1 promotes inflammation and fibrosis in acute pancreatitis. JCI Insight. 2021;6:e149539. doi: 10.1172/jci.insight.149539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Michalski CW, Gorbachevski A, Erkan M, Reiser C, Deucker S, Bergmann F, Giese T, Weigand M, Giese NA, Friess H, Kleeff J. Mononuclear cells modulate the activity of pancreatic stellate cells which in turn promote fibrosis and inflammation in chronic pancreatitis. J Transl Med. 2007;5:63. doi: 10.1186/1479-5876-5-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Schmid-Kotsas A, Gross HJ, Menke A, Weidenbach H, Adler G, Siech M, Beger H, Grünert AC, Bachem MG. Lipopolysaccharide-activated macrophages stimulate the synthesis of collagen type I and C-fibronectin in cultured pancreatic stellate cells. Am J Pathol. 1999;155:1749–1758. doi: 10.1016/S0002-9440(10)65490-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Li N, Li Y, Li Z, Huang C, Yang Y, Lang M, Cao J, Jiang W, Xu Y, Dong J, Ren H. Hypoxia inducible factor 1 (HIF-1) recruits macrophage to activate pancreatic stellate cells in pancreatic ductal adenocarcinoma. Int J Mol Sci. 2016;17:799. doi: 10.3390/ijms17060799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Gerasimenko JV, Petersen OH, Gerasimenko OV. SARS-CoV-2 S protein subunit 1 elicits Ca2+ Influx-Dependent Ca2+ signals in pancreatic stellate cells and macrophages in situ. Function (Oxf) 2022;3:zqac002. doi: 10.1093/function/zqac002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Eberhardt M, Salmon P, von Mach MA, Hengstler JG, Brulport M, Linscheid P, Seboek D, Oberholzer J, Barbero A, Martin I, et al. Multipotential nestin and Isl-1 positive mesenchymal stem cells isolated from human pancreatic islets. Biochem Biophys Res Commun. 2006;345:1167–1176. doi: 10.1016/j.bbrc.2006.05.016. [DOI] [PubMed] [Google Scholar]
- 129.Ku HT, Zhang N, Kubo A, O'Connor R, Mao M, Keller G, Bromberg JS. Committing embryonic stem cells to early endocrine pancreas in vitro. Stem Cells. 2004;22:1205–1217. doi: 10.1634/stemcells.2004-0027. [DOI] [PubMed] [Google Scholar]
- 130.Bai C, Gao Y, Zhang X, Yang W, Guan W. Melatonin promotes self-renewal of nestin-positive pancreatic stem cells through activation of the MT2/ERK/SMAD/nestin axis. Artif Cells Nanomed Biotechnol. 2018;46:62–74. doi: 10.1080/21691401.2017.1389747. [DOI] [PubMed] [Google Scholar]
- 131.Augstein P, Loudovaris T, Bandala-Sanchez E, Heinke P, Naselli G, Lee L, Hawthorne WJ, Góñez LJ, Neale AM, Vaillant F, et al. Characterization of the human pancreas side population as a potential reservoir of adult stem cells. Pancreas. 2018;47:25–34. doi: 10.1097/MPA.0000000000000950. [DOI] [PubMed] [Google Scholar]
- 132.Zhou Y, Sun B, Li W, Zhou J, Gao F, Wang X, Cai M, Sun Z. Pancreatic stellate cells: A rising translational physiology star as a potential stem cell type for beta cell neogenesis. Front Physiol. 2019;10:218. doi: 10.3389/fphys.2019.00218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Kordes C, Sawitza I, Götze S, Häussinger D. Stellate cells from rat pancreas are stem cells and can contribute to liver regeneration. PLoS One. 2012;7:e51878. doi: 10.1371/journal.pone.0051878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Karanu FN, Yuefei L, Gallacher L, Sakano S, Bhatia M. Differential response of primitive human CD34- and CD34+ hematopoietic cells to the Notch ligand Jagged-1. Leukemia. 2003;17:1366–1374. doi: 10.1038/sj.leu.2402973. [DOI] [PubMed] [Google Scholar]
- 135.Izquierdo AG, Crujeiras AB. Role of epigenomic mechanisms in the onset and management of insulin resistance. Rev Endocr Metab Disord. 2019;20:89–102. doi: 10.1007/s11154-019-09485-0. [DOI] [PubMed] [Google Scholar]
- 136.Yoon KH, Ko SH, Cho JH, Lee JM, Ahn YB, Song KH, Yoo SJ, Kang MI, Cha BY, Lee KW, et al. Selective beta-cell loss and alpha-cell expansion in patients with type 2 diabetes mellitus in Korea. J Clin Endocrinol Metab. 2003;88:2300–2308. doi: 10.1210/jc.2002-020735. [DOI] [PubMed] [Google Scholar]
- 137.Ogihara T, Mirmira RG. An islet in distress: β cell failure in type 2 diabetes. J Diabetes Investig. 2010;1:123–133. doi: 10.1111/j.2040-1124.2010.00021.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Kim JW, Ko SH, Cho JH, Sun C, Hong OK, Lee SH, Kim JH, Lee KW, Kwon HS, Lee JM, et al. Loss of beta-cells with fibrotic islet destruction in type 2 diabetes mellitus. Front Biosci. 2008;13:6022–6033. doi: 10.2741/3133. [DOI] [PubMed] [Google Scholar]
- 139.Yang Y, Kim JW, Park HS, Lee EY, Yoon KH. Pancreatic stellate cells in the islets as a novel target to preserve the pancreatic β-cell mass and function. J Diabetes Investig. 2020;11:268–280. doi: 10.1111/jdi.13202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Yoshikawa H, Kihara Y, Taguchi M, Yamaguchi T, Nakamura H, Otsuki M. Role of TGF-beta1 in the development of pancreatic fibrosis in Otsuka Long-Evans Tokushima Fatty rats. Am J Physiol Gastrointest Liver Physiol. 2002;282:G549–G558. doi: 10.1152/ajpgi.00323.2001. [DOI] [PubMed] [Google Scholar]
- 141.Saito R, Yamada S, Yamamoto Y, Kodera T, Hara A, Tanaka Y, Kimura F, Takei I, Umezawa K, Kojima I. Conophylline suppresses pancreatic stellate cells and improves islet fibrosis in Goto-Kakizaki rats. Endocrinology. 2012;153:621–630. doi: 10.1210/en.2011-1767. [DOI] [PubMed] [Google Scholar]
- 142.Zhou W, Dong S, Chen Z, Li X, Jiang W. New challenges for microRNAs in acute pancreatitis: Progress and treatment. J Transl Med. 2022;20:192. doi: 10.1186/s12967-022-03338-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Won JH, Zhang Y, Ji B, Logsdon CD, Yule DI. Phenotypic changes in mouse pancreatic stellate cell Ca2+ signaling events following activation in culture and in a disease model of pancreatitis. Mol Biol Cell. 2011;22:421–436. doi: 10.1091/mbc.e10-10-0807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Gryshchenko O, Gerasimenko JV, Gerasimenko OV, Petersen OH. Ca(2+) signals mediated by bradykinin type 2 receptors in normal pancreatic stellate cells can be inhibited by specific Ca(2+) channel blockade. J Physiol. 2016;594:281–293. doi: 10.1113/JP271468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Gryshchenko O, Gerasimenko JV, Peng S, Gerasimenko OV, Petersen OH. Calcium signalling in the acinar environment of the exocrine pancreas: Physiology and pathophysiology. J Physiol. 2018;596:2663–2678. doi: 10.1113/JP275395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Ferdek PE, Jakubowska MA, Gerasimenko JV, Gerasimenko OV, Petersen OH. Bile acids induce necrosis in pancreatic stellate cells dependent on calcium entry and sodium-driven bile uptake. J Physiol. 2016;594:6147–6164. doi: 10.1113/JP272774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Dixit A, Cheema H, George J, Iyer S, Dudeja V, Dawra R, Saluja AK. Extracellular release of ATP promotes systemic inflammation during acute pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2019;317:G463–G475. doi: 10.1152/ajpgi.00395.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Hart PA, Conwell DL. Chronic pancreatitis: Managing a difficult disease. Am J Gastroenterol. 2020;115:49–55. doi: 10.14309/ajg.0000000000000421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Yao S, Tu Z, Yang X, Zhang L, Zhong Y, Zheng L, Wang H, Yi Z, An J, Jin H, et al. Physiological and pathological roles of Hic-5 in several organs (Review) Int J Mol Med. 2022;50:138. doi: 10.3892/ijmm.2022.5194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Gao L, Lei XF, Miyauchi A, Noguchi M, Omoto T, Haraguchi S, Miyazaki T, Miyazaki A, Kim-Kaneyama JR. Hic-5 is required for activation of pancreatic stellate cells and development of pancreatic fibrosis in chronic pancreatitis. Sci Rep. 2020;10:19105. doi: 10.1038/s41598-020-76095-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Qiang L, Yang S, Cui YH, He YY. Keratinocyte autophagy enables the activation of keratinocytes and fibroblastsand facilitates wound healing. Autophagy. 2021;17:2128–2143. doi: 10.1080/15548627.2020.1816342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Zhang T, Zhang G, Yang W, Chen H, Hu J, Zhao Z, Cheng C, Li G, Xie Y, Li Y, et al. Lnc-PFAR facilitates autophagy and exacerbates pancreatic fibrosis by reducing pre-miR-141 maturation in chronic pancreatitis. Cell Death Dis. 2021;12:996. doi: 10.1038/s41419-021-04236-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Yang X, Chen J, Wang J, Ma S, Feng W, Wu Z, Guo Y, Zhou H, Mi W, Chen W, et al. Very-low-density lipoprotein receptor-enhanced lipid metabolism in pancreatic stellate cells promotes pancreatic fibrosis. Immunity. 2022;55:1185–1199.e8. doi: 10.1016/j.immuni.2022.06.001. [DOI] [PubMed] [Google Scholar]
- 154.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33. doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 155.Sun W, Ren Y, Lu Z, Zhao X. The potential roles of exosomes in pancreatic cancer initiation and metastasis. Mol Cancer. 2020;19:135. doi: 10.1186/s12943-020-01255-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Pothula SP, Pirola RC, Wilson JS, Apte MV. Pancreatic stellate cells: Aiding and abetting pancreatic cancer progression. Pancreatology. 2020;20:409–418. doi: 10.1016/j.pan.2020.01.003. [DOI] [PubMed] [Google Scholar]
- 157.Li M, Guo H, Wang Q, Chen K, Marko K, Tian X, Yang Y. Pancreatic stellate cells derived exosomal miR-5703 promotes pancreatic cancer by downregulating CMTM4 and activating PI3K/Akt pathway. Cancer Lett. 2020;490:20–30. doi: 10.1016/j.canlet.2020.06.009. [DOI] [PubMed] [Google Scholar]
- 158.Jiang J, Bai J, Qin T, Wang Z, Han L. NGF from pancreatic stellate cells induces pancreatic cancer proliferation and invasion by PI3K/AKT/GSK signal pathway. J Cell Mol Med. 2020;24:5901–5910. doi: 10.1111/jcmm.15265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Geleta B, Tout FS, Lim SC, Sahni S, Jansson PJ, Apte MV, Richardson DR, Kovačević Ž. Targeting Wnt/tenascin C-mediated cross talk between pancreatic cancer cells and stellate cells via activation of the metastasis suppressor NDRG1. J Biol Chem. 2022;298:101608. doi: 10.1016/j.jbc.2022.101608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Kikuta K, Masamune A, Watanabe T, Ariga H, Itoh H, Hamada S, Satoh K, Egawa S, Unno M, Shimosegawa T. Pancreatic stellate cells promote epithelial-mesenchymal transition in pancreatic cancer cells. Biochem Biophys Res Commun. 2010;403:380–384. doi: 10.1016/j.bbrc.2010.11.040. [DOI] [PubMed] [Google Scholar]
- 161.Hamada S, Masamune A, Takikawa T, Suzuki N, Kikuta K, Hirota M, Hamada H, Kobune M, Satoh K, Shimosegawa T. Pancreatic stellate cells enhance stem cell-like phenotypes in pancreatic cancer cells. Biochem Biophys Res Commun. 2012;421:349–354. doi: 10.1016/j.bbrc.2012.04.014. [DOI] [PubMed] [Google Scholar]
- 162.Chen S, Chen JZ, Zhang JQ, Chen HX, Yan ML, Huang L, Tian YF, Chen YL, Wang YD. Hypoxia induces TWIST-activated epithelial-mesenchymal transition and proliferation of pancreatic cancer cells in vitro and in nude mice. Cancer Lett. 2016;383:73–84. doi: 10.1016/j.canlet.2016.02.043. [DOI] [PubMed] [Google Scholar]
- 163.Bennewith KL, Huang X, Ham CM, Graves EE, Erler JT, Kambham N, Feazell J, Yang GP, Koong A, Giaccia AJ. The role of tumor cell-derived connective tissue growth factor (CTGF/CCN2) in pancreatic tumor growth. Cancer Res. 2009;69:775–784. doi: 10.1158/0008-5472.CAN-08-0987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Maity G, Ghosh A, Gupta V, Haque I, Sarkar S, Das A, Dhar K, Bhavanasi S, Gunewardena SS, Von Hoff DD, et al. CYR61/CCN1 Regulates dCK and CTGF and Causes Gemcitabine-resistant phenotype in pancreatic ductal adenocarcinoma. Mol Cancer Ther. 2019;18:788–800. doi: 10.1158/1535-7163.MCT-18-0899. [DOI] [PubMed] [Google Scholar]
- 165.Cao W, Zeng Z, He Z, Lei S. Hypoxic pancreatic stellate cell-derived exosomal mirnas promote proliferation and invasion of pancreatic cancer through the PTEN/AKT pathway. Aging (Albany NY) 2021;13:7120–7132. doi: 10.18632/aging.202569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Yamamoto M, Kensler TW, Motohashi H. The KEAP1-NRF2 System: A Thiol-based Sensor-effector apparatus for maintaining redox homeostasis. Physiol Rev. 2018;98:1169–1203. doi: 10.1152/physrev.00023.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Ju HQ, Gocho T, Aguilar M, Wu M, Zhuang ZN, Fu J, Yanaga K, Huang P, Chiao PJ. Mechanisms of overcoming intrinsic resistance to gemcitabine in pancreatic ductal adenocarcinoma through the redox modulation. Mol Cancer Ther. 2015;14:788–798. doi: 10.1158/1535-7163.MCT-14-0420. [DOI] [PubMed] [Google Scholar]
- 168.Arfmann-Knübel S, Struck B, Genrich G, Helm O, Sipos B, Sebens S, Schäfer H. The Crosstalk between Nrf2 and TGF-β1 in the Epithelial-mesenchymal transition of pancreatic duct epithelial cells. PLoS One. 2015;10:e0132978. doi: 10.1371/journal.pone.0132978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Wu YS, Looi CY, Subramaniam KS, Masamune A, Chung I. Soluble factors from stellate cells induce pancreatic cancer cell proliferation via Nrf2-activated metabolic reprogramming and ROS detoxification. Oncotarget. 2016;7:36719–36732. doi: 10.18632/oncotarget.9165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Hamada S, Taguchi K, Masamune A, Yamamoto M, Shimosegawa T. Nrf2 promotes mutant K-ras/p53-driven pancreatic carcinogenesis. Carcinogenesis. 2017;38:661–670. doi: 10.1093/carcin/bgx043. [DOI] [PubMed] [Google Scholar]
- 171.Dai B, Augustine JJ, Kang Y, Roife D, Li X, Deng J, Tan L, Rusling LA, Weinstein JN, Lorenzi PL, et al. Compound NSC84167 selectively targets NRF2-activated pancreatic cancer by inhibiting asparagine synthesis pathway. Cell Death Dis. 2021;12:693. doi: 10.1038/s41419-021-03970-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Husain A, Khan SA, Iram F, Iqbal MA, Asif M. Insights into the chemistry and therapeutic potential of furanones: A versatile pharmacophore. Eur J Med Chem. 2019;171:66–92. doi: 10.1016/j.ejmech.2019.03.021. [DOI] [PubMed] [Google Scholar]
- 173.Sousa CM, Biancur DE, Wang X, Halbrook CJ, Sherman MH, Zhang L, Kremer D, Hwang RF, Witkiewicz AK, Ying H, et al. Pancreatic stellate cells support tumour metabolism through autophagic alanine secretion. Nature. 2016;536:479–483. doi: 10.1038/nature19084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Rauf A, Imran M, Butt MS, Nadeem M, Peters DG, Mubarak MS. Resveratrol as an anti-cancer agent: A review. Crit Rev Food Sci Nutr. 2018;58:1428–1447. doi: 10.1080/10408398.2016.1263597. [DOI] [PubMed] [Google Scholar]
- 175.Lin Z, Zheng LC, Zhang HJ, Tsang SW, Bian ZX. Anti-fibrotic effects of phenolic compounds on pancreatic stellate cells. BMC Complement Altern Med. 2015;15:259. doi: 10.1186/s12906-015-0789-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Mohapatra D, Das B, Suresh V, Parida D, Minz AP, Nayak U, Mohapatra AP, Swain RK, Senapati S. Fluvastatin sensitizes pancreatic cancer cells toward radiation therapy and suppresses radiation- and/or TGF-β-induced tumor-associated fibrosis. Lab Invest. 2022;102:298–311. doi: 10.1038/s41374-021-00690-7. [DOI] [PubMed] [Google Scholar]
- 177.Estaras M, Ortiz-Placin C, Castillejo-Rufo A, Fernandez-Bermejo M, Blanco G, Mateos JM, Vara D, Gonzalez-Cordero PL, Chamizo S, Lopez D, et al. Melatonin controls cell proliferation and modulates mitochondrial physiology in pancreatic stellate cells. J Physiol Biochem. 2022;79:235–249. doi: 10.1007/s13105-022-00930-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Sada M, Ohuchida K, Horioka K, Okumura T, Moriyama T, Miyasaka Y, Ohtsuka T, Mizumoto K, Oda Y, Nakamura M. Hypoxic stellate cells of pancreatic cancer stroma regulate extracellular matrix fiber organization and cancer cell motility. Cancer Lett. 2016;372:210–218. doi: 10.1016/j.canlet.2016.01.016. [DOI] [PubMed] [Google Scholar]
- 179.Amrutkar M, Aasrum M, Verbeke CS, Gladhaug IP. Secretion of fibronectin by human pancreatic stellate cells promotes chemoresistance to gemcitabine in pancreatic cancer cells. BMC Cancer. 2019;19:596. doi: 10.1186/s12885-019-5803-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Kim SW, Oh KT, Youn YS, Lee ES. Hyaluronated nanoparticles with pH- and enzyme-responsive drug release properties. Colloids Surf B Biointerfaces. 2014;116:359–364. doi: 10.1016/j.colsurfb.2014.01.017. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.