Abstract
Alzheimer's disease (AD), is characterised by two major hallmarks: the formation of extracellular β-amyloid (Aβ) plaques and the hyperphosphorylation of tau protein, thus leading to the formation of neurofibrillary tangles. These hallmarks cause synaptic loss, neuronal damage, and the development of neuroinflammation and oxidative stress, which promote AD progression. Thus, the goal of treating AD is eliminating these hallmarks, to prevent AD progression and decrease symptoms. However, current available therapies provide symptomatic relief rather than treating the underlying cause of the disease, because the restrictive nature of the blood brain barrier (BBB) impedes the entry of drugs, thereby affecting drug efficacy and bioavailability. Researchers are focusing on developing new therapeutic approaches to bypass the BBB, for achieving site-specific drug delivery with the highest possible bioavailability and the lowest adverse effects. Recently explored therapeutic strategies include use of biologic agents such as monoclonal antibodies. Aducanumab, a strong candidate for treating AD, has been granted accelerated Food and Drug Administration approval; however, safety concerns may hinder its future use. Thus, nanotechnological approaches have led to a new era of AD treatment. Nanoparticles (NPs), because of their small particle size, can cross the BBB, thus enhancing drug pharmacokinetic properties and enabling targeted drug delivery. Polymeric NPs have been extensively studied, because of their simple production, biodegradability, biocompatibility, and unique architecture. These NPs provide a flexible vesicle that can be easily tailored to achieve desired physicochemical features. In this review, various types of polymer-based-NPs are discussed, highlighting the properties of fabricated NPs, which have multiple benefits in AD treatment, including anti-amyloid, antioxidant, and anti-inflammatory effects.
Keywords: β-amyloid (Aβ) plaques, Alzheimer's disease (AD), Chitosan, Curcumin, Fucoxanthin, PLGA, Polyester, Polymeric nanoparticle
الملخص
تطور مرض ألزهايمر كنتيجة لتكوين علامتين رئيسيتين، والتي تتضمن تشكيل طفيليات البيتا-أميلويد خارج الخلو وفرط فسفرة بروتين الـ تاو، مما يؤدي إلى تشكل عقد عصبية متعرجة. تؤدي هاتان العلامتان إلى فقدان التآزر بين الخلايا العصبية، وتلف الخلايا العصبية، وتطوير الالتهاب العصبي والإجهاد التأكسدي، مما يعزز تقدم المرض. لذلك، يهدف علاج مرض ألزهايمر إلى القضاء على هذه العلامات الرئيسية لمنع تقدم المرض وتقليل الأعراض. ومع ذلك، فإن العلاج الحالي المتوفر في السوق يوفر تخفيفا أعراضيا بدلا من معالجة السبب الأساسي للمرض. ويعزى هذا إلى الطبيعة المقيدة لحاجز الدماغ-الدم التي تعوق دخول الأدوية، مما يؤثر على فعالية الدواء وقابليته للامتصاص. يركز الباحثون على تطوير طرق علاجية جديدة يمكن أن تتجاوز حاجز الدماغ-الدم، لتوفير توصيل الدواء إلى الموقع المحدد مع أقصى قابلية امتصاص ممكنة وأقل آثار جانبية. من بين الاستراتيجيات العلاجية الجديدة المكتشفة هي استخدام البيولوجيات كأضداد أحادية النسيلة. واقترح اداكونوماب مرشحا قويا لعلاج مرض ألزهايمر وحصل على موافقة مسرعة من إدارة الغذاء والدواء، ومع ذلك، فإن مخاوف السلامة قد تحول دون استخدامه في المستقبل. وبالتالي، تشكل التقنية النانوية عصرا جديدا لعلاج مرض ألزهايمر، نظرا للسمات المميزة التي توفرها جزيئات النانو من حيث اجتياز حاجز الدماغ-الدم بسبب حجمها الصغير، وتعزيز الخصائص الدوائية للأدوية وتوصيل الدواء المستهدف. تدرس جزيئات النانو البوليمرية بشكل موسع في الوقت الراهن، نظرا لسهولة وبساطة طريقة إنتاجها، والتحلل البيولوجي، والتوافق البيولوجي والهيكل الفريد الذي يوفر وعاء مرن يمكن تعديله بسهولة لتحقيق الخصائص الفيزيوكيميائية المرغوبة. وبالتالي، سيتم مناقشة أنواع مختلفة من جزيئات النانو القائمة على البوليمر في هذه المراجعة، مع تسليط الضوء على الخصائص التي توفرها جزيئات النانو المصنعة والتي تظهر العديد من الفوائد في علاج مرض ألزهايمر. تشمل هذه الخصائص التأثيرات المضادة للأميلويد والمضادة للأكسدة والمضادة للالتهابات.
الكلمات المفتاحية: مرض ألزهايمر, طفيليات البيتا-أميلويد, جزيئات النانو البوليمرية, الكيتوسان, البولي (لاكتيد-كو-غليكوليد), الكركمين, الفوكسانثين, البولي إستر
Introduction
The treatment of neurodegenerative diseases including Alzheimer's disease (AD) has remained a burden on the pharmaceutical industry in the past decade. This topic has received substantial research attention, because increasing numbers of people are expected to have neurodegenerative diseases by the end of the 21st century, primarily because the life expectancy of the population is increasing, and AD is typically observed with aging.1 Approximately 50% of the population is expected to have AD at some point in ther lifetimes in the future, thus making AD a leading cause of morbidity and mortality in the 21st century.2 In 2021, the World Alzheimer's Disease Report indicated that approximately 50 million people had AD globally, thus suggesting that AD is one of the most incapacitating neurodegenerative disorders. By 2030, the number of people with AD is expected to increase to 78 million worldwide, with the aging of the world's population.3 In addition, AD is a leading cause of dementia in older people: approximately 60% of cases of dementia are associated with AD, affecting 24 million people globally, and the fifth leading cause of death.4,5
Conventional oral drug therapy
AD is challenging to diagnose, because the disease progression is slow and may begin 20 years before the symptoms escalate. Currently, AD is managed with oral medications such as the Food and Drug Administration (FDA) approved cholinesterase inhibitors, including donepezil, rivastigmine, and galantamine. These drugs are indicated primarily for alleviation of symptoms rather than treating the underlying pathology.4
The main obstacle to effective and successful drug therapy is enabling the drug to reach the desired site of action with the highest possible bioavailability and at the recommended dose. A notable drawback of available therapeutic regimens is their difficulty in crossing the blood brain barrier (BBB), which is composed of a capillary network that isolates the brain from the blood circulation.2 The role of the BBB is to protect the CNS against any foreign material; hence, this barrier limits entry of drugs into the brain. The BBB contains efflux transporters, such as p-glycoprotein, that prevent the passage of foreign agents and transport them out of the brain. In addition, the BBB's high lipophilicity limits the types of drugs that can reach the brain.2 Crossing the BBB is the limiting step preventing drugs from reaching reach their sites of action. Beyond the BBB, poor drug absorption in the gastrointestinal tract and rapid metabolism also affect drugs' oral bioavailability. These factors contribute to the lack of effectiveness of drug therapy, thereby prolonging treatment and affecting patient compliance.6
Most (98%) small drug molecules cannot reach the brain. Drugs administered to the brain should be small (<500 Da) and should have suitable hydrophobicity to cross the BBB.6 No new small drug molecule has been licensed by the FDA to treat AD since 2003; therefore, the pharmaceutical industry is focusing on implementing nanotechnology and the use of biologic agents such as monoclonal antibodies (mAbs) for AD treatment and to enhance crossing of the BBB.7
Advances in AD therapy
Recently, human mAbs have been extensively studied to treat AD. Aducanumab was the first FDA approved disease-modifying mAb indicated to treat AD pathophysiology, by targeting β-amyloid (Aβ) aggregates.8 Aducanumab is the most effective mAb, according to clinical trials.
Aducanumab exerts therapeutic effects after crossing the BBB. This drug selectively binds and subsequently decreases Aβ aggregates in the brain. Aducanumab also decreases levels of phosphorylated tau protein in the cerebrospinal fluid, thereby delaying disease progression.9 However, recent studies have demonstrated the emergence of amyloid-related imaging abnormalities with high injected doses of the drug; these adverse effects are a form of the vasogenic edema that occurs with the BBB disruption that enables mAb entry to the CNS.7 In addition, a linear relationship exists between the decrease in Aβ plaques and amyloid-associated imaging abnormalities. Cerebral microhemorrhages in mouse models have been observed after administration of mAbs, thus giving rise to safety concerns regarding administration of high mAb doses. Additionally, high production costs may pose a burden on the industry.8
The FDA granted accelerated approval of aducanumab in 2021, requiring a post-approval clinical trial to ensure that the drug exerts the desired therapeutic action; however, the European Medical Agency cancelled approval of the drug in 2022.8
Importance of nanotechnology
To overcome the aforementioned limitations of the FDA-approved aducanumab and small drug molecules, researchers are implementing nanotechnology aimed at providing successful drug therapy for AD. Nanotechnology uses nanomaterials as carriers for drugs with a size range of 1–1000 nm, and has several attractive features for effective drug therapy, including the following:10, 11, 12, 13
-
○
Sustained drug release
-
○
Enhanced bioavailability and biocompatibility
-
○
Ability to deliver multiple drugs
-
○
Protection of drugs against degradation while limiting undesirable adverse effects
-
○
Flexibility of nanoparticles (NPs) to be tailored to achieve desired surface properties, such as chemical groups with positively charged surfaces that interact with the negatively charged endothelial cells in the BBB, thus facilitating cellular uptake and drug absorption in the brain, and improving drug pharmacokinetics and prolonging the half-life
-
○
High surface to volume ratio of NPs, to promote binding to Aβ fibrils and prevention of their assembly, through hydrophobic interaction or the use of target ligands
To attain these features, NP materials should be carefully chosen to provide suitable permeability, flexibility, biocompatibility, and surface charge for successful drug delivery. Thus, maintaining the hydrophobicity, surface chemistry, and ionic charge of the selected material is crucial to ensure efficient BBB crossing.11
Polymeric NPs
Finding strategies for therapeutics that can cross the BBB without disruption is the main goal in successful drug delivery. Hence, increased attention has been paid to formulating ideal NPs in which drugs are embedded and released at the target site. NPs’ small size and flexibility enable delivery of drugs across the BBB through NP retention and adsorption to the brain capillaries. This process increases transport of the loaded NPs across the endothelial cells, driven by a high concentration gradient. NPs are endocytosed by endothelial cells, thus enabling drug release. NPs can enter endothelial cells through transcytosis, or a combination of both transcytosis and endocytosis.14
NPs are classified into two main categories: organic and inorganic. Inorganic NPs are metal-based NPs that enable a more controlled size distribution; however, they are not biodegradable and may elicit toxicity.15 In contrast, organic NPs are polymeric NPs that serve as promising carriers for delivering therapeutic agents to the brain. Polymeric NP structures surround a drug core with a coating polymer. Polymeric NPs, through their flexible architecture, nano-size range, biodegradability enabling coating polymers to be easily excreted from the body, and versatility in encapsulating a wide range of drugs, enhance drug circulation times and prolong drug release. In addition, polymeric NPs have more favorable in vivo and in vitro stability, and enable better control of drug release, than liposomes.16
An additional attractive feature is that the preparation techniques for polymeric NPs are generally simple and easily scaled up.17 The methods include ionic gelation, coacervation, emulsion solvent evaporation, spontaneous emulsification, spray drying, and supercritical fluid technology of monomers as albumin or polysaccharides.12,18
Aim and objective
The purpose of this review is to highlight how the incapacitating disease of AD may affect the future of humanity. The failure of available drug therapies to treat the underlying pathophysiology of AD has motivated researchers to develop novel and smart formulations. Oral conventional drugs fail to treat AD because of their low ability to bypass the BBB and poor bioavailability. Additionally, administration of the only FDA approved mAb for treating AD poses major safety concerns that may hinder use of this therapy. Therefore, this article emphasizes recent advances in polymeric NP formulations to enhance drugs’ crossing of the BBB, to reach the brain and treat the underlying causes of AD. Effective drug delivery may be achieved by using various polymers in the NPs. To aid in identifying an ideal NP to achieve enhanced therapeutic action of the embedded drug with high safety and efficacy, this article describes diverse types of polymeric NPs, on the basis of a comparison of the literature, including their anti-AD properties, preparation methods, morphology, and surface charge that contributes to crossing the BBB, according to various characterization tests. Overall, this systematic review is aimed at determining the ability of polymeric NPs to serve as a strategy for AD treatment, and providing insights into the current state of research and gaps in the scope of AD treatment through polymeric NP approaches.
Pathophysiology
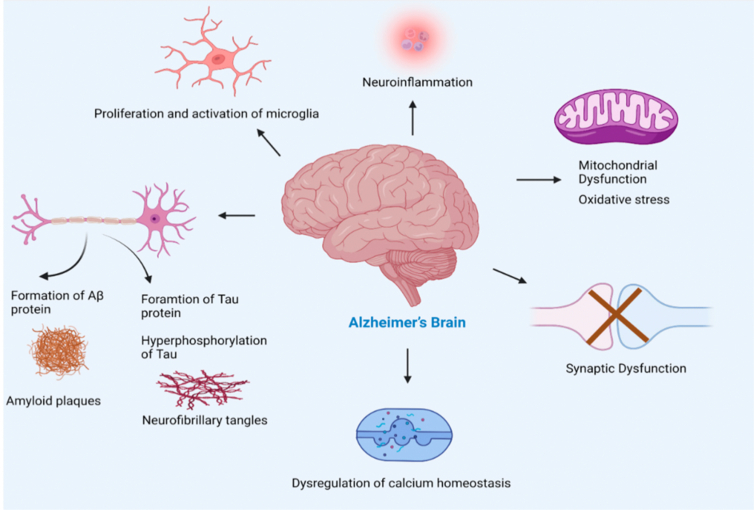
The complexity and diversity of AD pathophysiology has posed challenges in identifying AD's underlying causes, since the discovery of the disease by scientist Alois Alzheimer in 1907.3 AD is defined as an age-related neurodegenerative disease characterized primarily by chronic progressive loss of neuronal activity, neuroinflammation, and cerebral atrophy, which together lead to cognitive and memory impairment.1 People with AD experience memory loss, learning difficulties, and behavioral changes, all of which affect quality of life (see Figure 1).
Figure 1.
Graphical Abstract: Overview of proposed AD treatment strategies (constructed with biorender.com).
Although AD is a multifactorial condition, the principal factors in AD development are extracellular accumulation of β-amyloid protein (β-amyloid plaques) and abnormal intracellular accumulation of tau protein (neurofibrillary tangles; NFTs). These two hallmarks predispose individuals to a series of inflammatory reactions, as described below (Figure 2).19
-
○
Aβ plaque hypothesis: The production of extracellular Aβ plaques is a substantial hallmark in AD progression. Normally, Aβ is produced from the breakdown of a glycoprotein referred to as amyloid precursor protein (APP). APP is involved in multiple cellular processes in neuronal development and intracellular transport, including signal transduction, and the maintenance of neuronal homeostasis. APP undergoes proteolytic cleavage by beta-secretase, thereby yielding soluble APP fragments. The remaining portion of APP undergoes further cleavage by gamma-secretase, thus leading to the production of peptides that are rapidly eliminated from the brain 20. The resultant soluble Aβ fragments usually comprise a 40 amino acid sequence (Aβ1–40), which is generally considered less toxic. However, various lengths have also been identified, such as longer forms (Aβ1–42), which are prone to aggregation.21 Whereas soluble Aβ fragments have beneficial roles in synaptic plasticity, memory, and brain cell growth, in AD, these soluble fragments bind other Aβ fragments and form oligomers. These oligomers exhibit diminished clearance from the brain and ultimately aggregate into insoluble Aβ plaques, thus predisposing individuals to AD.20 Additionally, in AD, APP is broken down by beta and gamma secretase enzymes, thus producing an insoluble form of Aβ (Aβ1–42), which tends to form plaques. The accumulation of neurotoxic Aβ occurs because of an imbalance between the production and elimination of the insoluble peptide, thereby disrupting the normal levels of Aβ in the brain, and causing loss of neurons, synaptic dysfunction, and disruption of calcium homeostasis.20,22 The abnormal APP processing in AD is attributed to increased activity of beta-secretase, as well as expression and secretion of the enzyme into the blood circulation by tissues other than the brain, as reported by Nicsanu et al.23 Consequently, these disturbances lead to cognitive deficits and eventually dementia. Hence, inhibiting the accumulation of Aβ fibers to prevent the formation of toxic fibrils is the main strategy for alleviating AD progression (Figure 2).22
-
○
NFT hypothesis: Tau is a microtubule-associated protein normally found in brain neurons. Its main functions are maintaining microtubule stability and flexibility in axons, and regulating cell signaling.20,24 In healthy brains, tau protein contains several phosphate groups in its microtubule assembly domain. However, the phosphorylation of tau protein increases its tendency to aggregate in its hyperphosphorylated form, thus decreasing its affinity to bind microtubules,3 and leading to microtubule disassembly, a hallmark of AD. As a result of the accumulation of hyperphosphorylated tau protein, NFTs form through the polymerization of free tau fragments into paired helical aggregates. The appearance of NFTs in the brain results in neuron loss and cognitive impairment, by hindering nerve cells' nutrient access and leading to neuronal cell death (Figure 2).19
-
○
Neuroinflammation: The presence of Aβ aggregates and NFTs in the brain triggers a series of inflammatory reactions that contribute to AD progression. The formation of neurotoxic proteins activates the release of brain immune cells called microglial cells. Microglial cells resemble macrophages, and play crucial roles in the clearance of aggregated protein, dead neurons, and debris. Moreover, microglial cells activate the release of cytokines, such as interleukins and tumor necrosis factor-α (TNF-α), which is a major cause of neuroinflammation in AD. Consequently, inflammatory reactions increase when microglial cells fail to eliminate debris and the accumulated proteins, thus resulting in cerebral atrophy.19 Excessive inflammatory responses can cause cerebral dysfunction, which in turn contributes to disease progression. Neuronal loss has also been observed as a result of the production of reactive oxygen species and free radicals released by activated microglial cells.25 Notably, post-mitotic cells, such as neurons, exhibit high aerobic metabolism, which requires high oxygen levels to support energy production through mitochondrial respiration. This process may increase the levels of reactive oxygen species. In contrast, the levels of reactive oxygen species are kept low in healthy conditions, because of the presence of antioxidant compounds, such as glutathione, that scavenge reactive oxygen species and free radicals, and prevent oxidative stress. The activity of antioxidants decreases with aging. For instance, people with AD have low concentrations of glutathione, and thus the production of reactive oxygen species increases beyond glutathione's capture capacity. Oxidative stress may eventually lead to neurodegeneration (Figure 2). Oxidative stress is correlated with Aβ aggregation; therefore finding a therapeutic approach that combines antioxidant effects and anti-amyloid activity may provide a breakthrough in treating AD.26
-
○
Calcium dysregulation hypothesis: Given the crucial involvement of calcium signaling in several neuronal functions, such as neuronal precursor cell growth and differentiation, any alterations in calcium homeostasis may induce mitochondrial dysfunction, oxidative stress, and neuroinflammation (Figure 2). Recent studies have suggested that disturbances in calcium signaling occur during early stages of AD,27 through an underlying mechanism of increased calcium ion concentrations in the cytoplasm of neuronal cells. This elevation occurs as calcium is transported from both the extracellular environment and intracellular reservoirs, as a result of Aβ activation of calcium ion transporters. Additionally, Aβ peptide accumulation has been shown to alter calcium concentrations by activating calcium channels in cells, thus inducing the formation of membrane pores and resulting in cell death.27 The accumulation of calcium ions within neurons can trigger the generation and buildup of Aβ plaques and hyperphosphorylated tau proteins. This process contributes to the decline in cognitive ability in people with AD.28
Figure 2.
Reproduced from Sehar et al. 2022. Pathogenesis of Alzheimer's disease. The multiple factors responsible for the progression of Alzheimer's disease include amyloid beta plaques and tau neurofibrillary tangles. Subsequently, neuronal loss and activation of microglia (concentrated in the vicinity of amyloid plaques) occur, thereby leading to neuroinflammation, mitochondrial dysfunction, synaptic loss, and dysregulation of calcium homeostasis. The accumulation of Aβ in the brain in Alzheimer's disease perturbs synapsis and leads to postsynaptic hyperexcitability. Hyperexcitability of neurons causes dysregulation of calcium homeostasis and increased production of reactive oxygen species.20
Beyond these major hallmarks, diseases such as diabetes, hypertension, and obesity, as well as aging, lifestyle, and genetic factors, may predispose people to AD development. Notably, identifying risk factors may aid in the development of efficient therapies.25
Materials and Methods
This systematic literature review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses framework guidelines for analyzing selected articles. Initially, the search strategy involved use of electronic databases as PubMed, ScienceDirect, and Google Scholar. The following keywords were used: nanoparticles, anti-Alzheimer's drugs, polymeric nanoparticles, brain targeting, neurodegenerative diseases, and blood brain barrier. The relevant articles (98 articles, including research and review articles) were downloaded. In Mendeley Reference Manager software, any duplicated records were removed. Moreover, the abstracts and conclusions of the articles were screened to determine those relevant to the aim and objective of this review. The selected articles meeting the following inclusion criteria were eligible for inclusion:
-
•
Research and review articles published in the scope of health sciences and pharmaceutical innovation, emphasizing treatment of AD with polymeric NPs
-
•
Research articles published between 2010 and 2023, or review articles published between 1999 and 2023
-
•
Research articles with polymeric nanoparticles' characterization tests
-
•
Research articles focusing on AD treatment rather than managing symptoms
-
•
Research articles focusing on brain targeting with nano-drug delivery systems
The exclusion criteria comprised the following:
-
•
Studies not associated with neurodegenerative diseases
-
•
Articles focusing on various types of NPs other than polymeric NPs, such as liposomes and exosomes
-
•
Articles not using nanotechnology
Results and discussion
Over the past decade, polymeric NPs have been explored to determine their potential to treat AD with preparations of vesicles below 1000 nm in diameter, with special physicochemical properties to promote efficient drug delivery. The polymer-based NPs include poly (d,l-lactide-co-glycolide (PLGA), chitosan, polyethylene glycol (PEG), and polyester based NPs. Each NP has been assessed to determine its efficacy and effectiveness as a drug delivery carrier (summarized in Table 1).
Table 1.
Summary of the properties of the described polymeric NPs.
| Encapsulated drug | Polymer | Size (nm) | Charge (mV) | EE% | Anti-AD effect | Reference |
|---|---|---|---|---|---|---|
| Curcumin | PLGA | 150–200 | −30 to −20 | N/A∗ | Inhibited Aβ aggregation | 30 |
| Fucoxanthin | PEG-PLGA | 200 | −22.6 ± 1.2 | 48.44 |
|
33 |
| Rivastigmine | Chitosan | 185.4 ± 8.4 | 38.40 ± 2.85 | 85.3 | Intranasal administration achieved higher drug concentrations in the brain than the plasma | 39 |
| Rosiglitazone | Poloxamer stabilized mPEG-PCL | 124.6 ± 12 | −17.5 ± 5.54 | 70.65 |
|
43 |
| – | PLGA | 100 | 8 | – | Decreased Aβ aggregation and tau protein phosphorylation | 29 |
∗Not available.
PLGA-based NPs
Biodegradable polymers, such as the FDA approved PLGA, have been extensively used in the past decade as drug delivery carriers for the treatment and management of various diseases. The synthesis of PLGA involves ring opening polymerization of glycolic acid and lactic acid. According to the ratio of monomers, various forms of PLGA can be obtained. Because of its biodegradability, the ester linkage of PLGA is broken into monomers through hydrolysis in the body, the original monomers serve as physiological byproducts of various metabolic pathways that are easily eliminated through the Krebs cycle.29
Native PLGA NPs
Anand et al.29 have investigated the effects of native PLGA NPs on Aβ plaques and demonstrated that PLGA inhibits spontaneous Aβ accumulation. A series of characterization tests have been conducted to confirm the efficacy of PLGA NPs against Aβ plaques. The study revealed the following unique effects of PLGA NPs, thus highlighting the importance of PLGA in treating the underlying cause of AD—an aspect not clearly stated in any other research article:
-
1.
Decreased Aβ plaques: Transmission electron microscopy (TEM) was used to determine the size and morphology of PLGA NPs without encapsulated drug. The results indicated a stable dispersion of homogeneous spheroidal NPs with diameters of approximately 100 nm and a zeta potential of 8 mV. Thioflavin T (ThT) fluorescence assays were conducted to determine the suppression of amyloid fibril aggregation, on the basis of strong binding of ThT to Aβ deposits. Aβ peptide samples (10 μM) were added to various concentrations of PLGA NPs ranging from 1 to 50 μM, then assessed for 24 h at 37 °C. The spontaneous formation of amyloid aggregates was suppressed in the presence of PLGA NPs in a dose-dependent manner. Inhibition of amyloid formation was also confirmed by TEM and dynamic light scattering. In addition, a decrease in the size, number, and area of Aβ plaques observed after treatment of 5 × FAD mice (an animal model of AD) with PLGA NPs confirmed the ability of the NPs to spontaneously inhibit Aβ aggregates. The decreased areas of Aβ deposits correlated with the decreased levels of APP and its products in the mouse cortex, after PLGA NP treatment.
-
2.
Decreased Aβ-induced toxicity: Cytotoxicity (MTT) assays were conducted in human neuron samples derived from induced pluripotent stem cells from patients with AD, to study the effects of PLGA treatment on neuronal cells in terms of Aβ-induced toxicity. In the presence of PLGA NPs, tau protein phosphorylation in Aβ cultured neurons decreased after 24 h exposure, thus indicating increased cell viability. Additionally, the same test conducted on mouse samples supported that PLGA NP treatment protected neuronal cells against toxicity.
On the basis of the results obtained from characterization tests, native PLGA NPs have therapeutic features that not only decrease the aggregation of Aβ peptides but also protect neurons against Aβ associated toxicity. Therefore, using PLGA as a carrier for therapeutic agents may maximize the therapeutic effects against the disease through synergistic effects. Because AD is a multifactorial disease, incorporating compounds with versatile pharmacological properties, such as curcumin, could aid in AD treatment.
Curcumin loaded PLGA NPs
Curcumin is well known for its broad-spectrum biological properties, such as anti-inflammatory, antioxidant, and, most importantly, neuronal protective activity. Recently, curcumin has been demonstrated to act against the major hallmarks of AD. Curcumin is a strong candidate for AD treatment because its anti-amyloid and antioxidant properties enable it to regulate the oxidative stress and inflammatory reactions occurring during AD.30 In addition, curcumin, owing to its chemical structure, has been reported to have unique bonding properties that enable BBB permeation and binding to Aβ plaques. Moreover, curcumin's structure possesses both hydrophobic and hydrophilic moieties that facilitate BBB penetration and bind Aβ oligomers, respectively.31 The main challenges in using curcumin as an anti-AD drug are its low bioavailability and its low water solubility due to its high hydrophobicity. Therefore, encapsulating curcumin in a carrier such as PLGA could provide an ideal solution to enhance curcumin's bioavailability and biodistribution, and decrease its rapid elimination from the body.30
A recent study has investigated the effects of curcumin encapsulation in PLGA NPs (curcumin-PLGA NPs) with the conjugation of Tet-1 peptide as the targeting ligand. Tet-1 peptide interacts with motor neurons and bypasses the BBB through retrograde axonal transport, wherein the NPs are transported from the axon to the cell body of the neuron.30
Mathew et al.30 have prepared curcumin-PLGA NPs through a single emulsion evaporation method, which is optimal for encapsulating hydrophobic drugs. According to TEM images, the resultant particle size ranged from 150 to 200 nm and showed a smooth morphology because of PLGA coating of the particles. In addition, the encapsulation of curcumin in PLGA NPs resulted in full water solubility. The zeta potential of curcumin-PLGA NPs had a range of −30 to −20 mV, thus indicating the negative charge exhibited by the prepared NPs, and revealing the stability of the system supporting crossing of the BBB via clathrin-mediated endocytosis.30 This finding is in accordance with results reported by Lockman et al., in 2004,32 demonstrating that anionic NPs increase brain permeability, and that the brain penetration is attributable to the binding of anionic NPs to the BBB's low-density lipoprotein receptor.32
Effects of curcumin-PLGA NPs on AD30
-
1.
Anti-amyloid activity of curcumin-PLGA NPs
For determination of the anti-amyloid activity of the prepared NPs, Mathew et al.30 treated amyloid protein aggregates (Figure 3A) with curcumin-PLGA NPs. The results indicated that NPs attached to the aggregates, which in turn decreased in size after 12 h of incubation (Figure 3B). After 48 h of incubation, the aggregates were completely broken down (Figure 3C). The exact mechanism of anti-amyloid activity of curcumin remains unknown.30
-
2.
Antioxidant effects of curcumin-PLGA NPs
Figure 3.
Adapted from Mathew et al., 2012. Anti-amyloid activity of PLGA-curcumin nanoparticles.30
Mathew et al.30 investigated the free radical capturing capacity of the prepared NPs compared with raw curcumin, with a free radical source of 1,1′-diphenyl-2-picrylhydrazyl. The antioxidant activity was presented as percentage decrease in 1,1′-diphenyl-2-picrylhydrazyl. Raw curcumin had higher antioxidant activity, with 80% free radical capturing activity, whereas the prepared NPs had 60% antioxidant activity. This finding was attributed to the time required for curcumin to leach from the NPs. However, the encapsulation of curcumin in PLGA did not eradicate its antioxidant effects. Despite the lower antioxidant activity of the prepared NPs than raw curcumin, a potent effect was observed that could potentially ameliorate oxidative stress.
-
3.
High viability of cells treated with curcumin-PLGA NPs
Cytotoxicity testing with MTT assays was conducted to assess cell viability after curcumin-PLGA NP treatment. The experiments revealed a high safety profile of the nano-formulation: the cell viability reached 70% after exposure of cells to high concentrations of prepared NPs.30
-
4.
BBB penetration through Tet-1 conjugation
To ensure effective uptake of fabricated NPs, flow cytometry analysis was conducted to determine the in vitro uptake of fabricated NPs by glioma cells. According to the intensity of the fluorescence of the flow cytometry analysis, NPs with Tet-1 conjugation showed greater neuronal uptake than unconjugated curcumin-PLGA NPs, as indicated by the high fluorescence intensity, in which a high NP concentration was found around the cells. Therefore, the conjugation of Tet-1 peptide may serve as a promising targeting ligand for efficient drug delivery and crossing the BBB. In addition, Mathew et al.30 used confocal laser scanning microscopy to confirm the uptake of NPs by neuronal cells.
On the basis of the results of the aforementioned study, coating curcumin with PLGA to form NPs is a promising approach for AD treatment. Curcumin exhibits relatively strong antioxidant activity, which might be effective in alleviating oxidative and inflammatory stress in AD, as well as decreasing amyloid plaques that are a major disease hallmark. However, release kinetics studies must be conducted to determine the amount of drug that reaches the site of action, and the encapsulation efficiency (EE%) calculation results should be clearly stated, because this parameter is essential in assessing the efficiency of drug delivery systems but was not reported.30
Consequently, the presence of PLGA along with curcumin may maximize the anti-amyloid activity, thus further decreasing the sizes of amyloid aggregates, through its ability to interact with, and inhibit the aggregation of, Aβ, while protecting neurons against cell toxicity, thus suggesting a high safety profile. Combining these properties in a single formulation could potentiate the anti-amyloid effects and enable high drug therapy efficiency.
Fucoxanthin loaded NPs
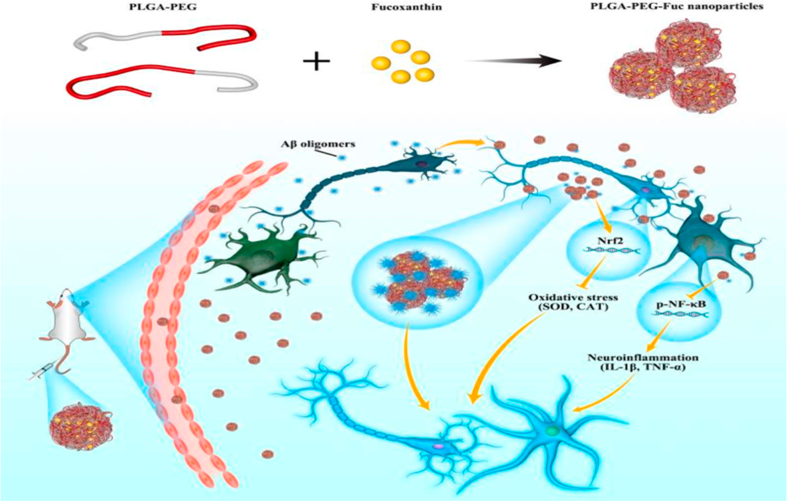
In the past decade, research has increasingly focused on examining the potential of naturally existing compounds in AD treatment, because of their high safety profile. Recently, marine algae derived fucoxanthin, which is currently used as a dietary supplement, has been studied to determine its anti-AD effects in decreasing oxidative stress and inflammatory reactions in the brain, and inhibiting Aβ aggregation. Notably, fucoxanthin has been reported to have a high safety profile, on the basis of preclinical and small population clinical studies. However, fucoxanthin's low water solubility and poor permeation of the BBB limit its use as an anti-AD drug. Encapsulating fucoxanthin in a PLGA based NPs might overcome these limitations and enhance its therapeutic effects, by achieving drug delivery into the brain with higher efficacy and bioavailability. In a study conducted by Yang and coworkers, fucoxanthin was entrapped in PLGA NPs with PEG conjugated to the surfaces of NPs, to form PEG-PLGA-fucoxanthin (PEG-PLGA-fuc) NPs (Figure 4). The purpose of PEG attachment was to enhance the surface hydrophilicity, thereby increasing the circulation time and the bioavailability of the loaded drug. These properties may potentiate the efficacy of the drug to treat AD.
Figure 4.
Reused with a permission from Yang et al., 2021. Diagram of mechanisms underlying the anti-AD neuroprotective effects of PLGA-PEG-Fuc nanoparticles.33
PEG-PLGA-fuc NPs were prepared with a solvent evaporation method to produce spherical NPs (Figure 4) with a uniform size of approximately 200 nm, which is suitable for BBB penetration. The zeta potential was measured, to determine the surface charge and stability of the NPs. PEG-PLGA-fuc NPs exhibited a negative charge of approximately −22.6 ± 1.2 mV, thus supporting BBB penetration32,33 and stable dispersion without aggregation. The EE% of the drug in the NPs was 48.44%, thus suggesting that a fair amount of drug was entrapped.33
Influence of PEG-PLGA-fuc NPs on AD33
-
1.
Prolonged drug release
A sustained release profile was observed in testing of fucoxanthin release from fabricated NPs. The release profile indicated an initial burst release followed by sustained drug release as a result of diffusion of fucoxanthin through the swollen NPs. Approximately 62.84% of the release of fucoxanthin occurred after 130 h (Figure 5A). This mechanism prolonged the presence of PEG-PLGA-fuc NPs in the blood circulation, thus enabling a high amount of the drug to penetrate the brain, and resulting in increased bioavailability and decreased metabolism in vivo.
-
2.
Inhibition of amyloid plaques
Figure 5.
Adapted with permission from Yang et al., 2021. A collection of assay techniques used and resultant data.33
In vitro ThT assays were performed to determine the ability of PEG-PLGA-fuc NPs to inhibit the formation of Aβ aggregates (Figure 5B). The human neuroblastoma cell line SH-SY5Y with Aβ oligomers was treated with PEG-PLGA-fuc NPs, and a decrease in fluorescence level was observed, indicating the prevention of Aβ fibril formation, given that ThT specifically binds Aβ fibrils. Dot blot assays were also conducted with Aβ fibrils, to compare the effects of treating the fibrils with PEG-PLGA-fuc NPs or free drug solution. Greater inhibition of Aβ aggregates (oligomers) was observed after treatment of the cells with the fabricated NPs than the free drug, thus suggesting potent anti-amyloid activity (Figure 5C). Interestingly, SH-SY5Y neuronal cells showed high viability and prevention of death of neuronal cells in MTT assays, in contrast to untreated cells and cells treated with free drug, thus indicating the protective effects of the fabricated NPs against neurotoxicity (Figure 5D). Moreover, flow cytometry testing was conducted with SH-SY5Y neuronal cells, and the fluorescence intensity was measured, to ensure efficient cellular uptake. An increase in fluorescence intensity was observed, thus indicating high cellular uptake of the fabricated NPs.
-
3.
Alleviation of oxidative stress
Oxidative stress and inflammation strongly influence the pathophysiology of AD, because of the production of reactive oxygen species and stimulation of proinflammatory cytokines, such as TNF-α and interlukin-1β (IL-1β), as a consequence of Aβ accumulation. Thus, testing the effects of prepared NPs against oxidative stress and neuroinflammation is essential. Injected PEG-PLGA-fuc NPs were speculated to prevent microglial cell stimulation in the mouse brain, thus decreasing TNF and IL-1β levels in the hippocampus and the brain cortex.
Regarding oxidative stress, PEG-PLGA-fuc NPs were tested to determine their ability to potentiate the effects of the antioxidant enzymes, such as superoxide dismutase (SOD) and catalase, in the brain. The activity of antioxidant enzymes is inhibited by the presence of Aβ oligomers in mouse brains; however, the activity of these enzymes has been found to be potentiated after treatment with PEG-PLGA-fuc NPs. Furthermore, Yang et al.33 reported that fucoxanthin inhibits the intracellular production of reactive oxygen species, on the basis of studies using 2′,7′-dichlorodihydrofluorescein staining to measure oxidative stress, on the basis of fluorescence directly correlating with the amount of reactive oxygen species.
Previous results have indicated that fucoxanthin has potent antioxidant and anti-amyloid activities, and its encapsulation in PLGA-PEG NPs facilitates its neuronal uptake, and its neuroprotective, anti-neuroinflammatory, and anti-amyloid effects. Therefore, this formulation provides a major advancement in anti-AD drug therapy. However, further in vivo studies must be conducted to validate the described effects.
Chitosan-based NPs
Chitosan, a biodegradable and biocompatible polymer derived from chitin deacetylation, is a natural mucopolysaccharide present in crustacean shells. The structure of chitosan comprises two copolymers: glucosamine and N-acetylglucosamine. Because chitosan is a biodegradable polymer, it is broken down by lysozyme in the body, thus producing safe and nonimmunogenic byproducts.34
Interestingly, chitosan is an attractive polymer for encapsulating anti-AD drugs to form NPs. Its mucoadhesive properties provide advantages in nasal drug delivery to the brain, thus enabling bypassing of the BBB and enhancing the bioavailability of orally administered drugs. Notably, chitosan bears a positive charge that leads to opening of tight junctions in the brain, thereby allowing the encapsulated drug to enter the CNS and contributing to mucoadhesive properties by facilitating binding to anionic groups present in the mucus membrane.17 The mechanism of mucus membrane binding involves absorption of water from the surrounding environment, thus causing swelling of the polymer and formation of a gel-like structure. This mechanism prolongs the residence time of NPs in the nasal cavity and limits mucociliary clearance, and consequently promotes absorption of the drug.35 Intranasal administration is a noninvasive drug delivery route to the CNS that provides several advantages including bypassing the BBB; avoiding first pass metabolism by transporting the drug through the olfactory nerve; and achieving high drug concentrations in the cerebrospinal fluid. These attractive features are associated with the rich blood supply in the nasal cavity and a large surface area promoting rapid drug absorption and enabling direct brain access within minutes.36
In contrast, chitosan has poor water solubility and its soluble only under acidic pH. The nasal mucosa has a pH range of 5.5–6.5, thus providing a suitable environment for drug delivery systems containing chitosan. This route of administration is preferred, because it does not require any modification of the polymer, and it enables direct transport to the brain 37. Therefore, chitosan is a promising candidate for intranasal administration, because of its attractive characteristics that may optimize drug delivery.
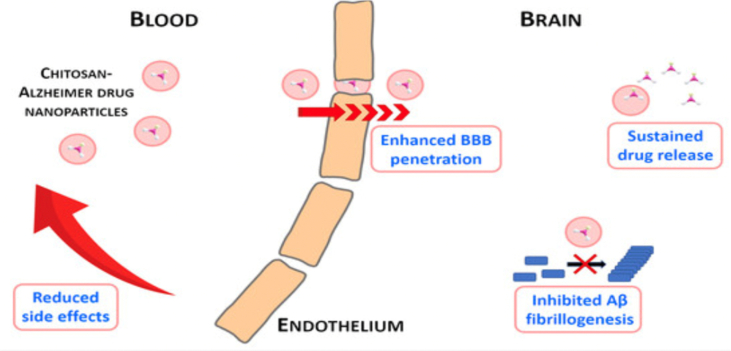
A recent study has used a strategy of encapsulating orally administered rivastigmine into chitosan NPs. Rivastigmine is an acetylcholinesterase (AChE) inhibitor widely used in AD management. This drug enhances the cholinergic functions affected by the degeneration of cholinergic neurons; its mechanism involves increasing levels of acetylcholine by hindering its hydrolysis; consequently, cognitive function is improved.38 The administration of rivastigmine requires frequent dosing, because its high hydrophilicity restricts entry to the brain. However, frequent administration of rivastigmine can cause severe cholinergic adverse effects. Thus, the encapsulation of rivastigmine enhances its delivery to the target site through the nose to brain approach, and also aids in decreasing systemic adverse effects (Figure 6).39
Figure 6.
Reproduced from Manek et al., 2020. The influence of chitosan loaded NPs in AD treatment.34
Fazil et al.39 prepared rivastigmine loaded chitosan NPs through the ionic gelation method in the presence of sodium tripolyphosphate, a crosslinking agent that interacts with chitosan electrostatically and promotes NP formation. The optimum NPs had a particle size of 185.4 ± 8.4 nm and a zeta potential of 38.40 ± 2.85 mV, thus indicating a stabilized nanosuspension with positively charged NP surfaces that cause electrostatic repulsion between particles and prevent their aggregation. Fazil et al.39 attributed the positive charge of the NPs to the residual amino groups of chitosan that were not neutralized by the negatively charged sodium tripolyphosphate. TEM images revealed smooth, spherical, and unaggregated NPs, thereby confirming the stability of system. A high EE% of the optimum NPs (85.3%) was observed, thus suggesting high drug entrapment efficiency.39
Influence of proposed NPs on AD39
-
1.
Sustained drug release
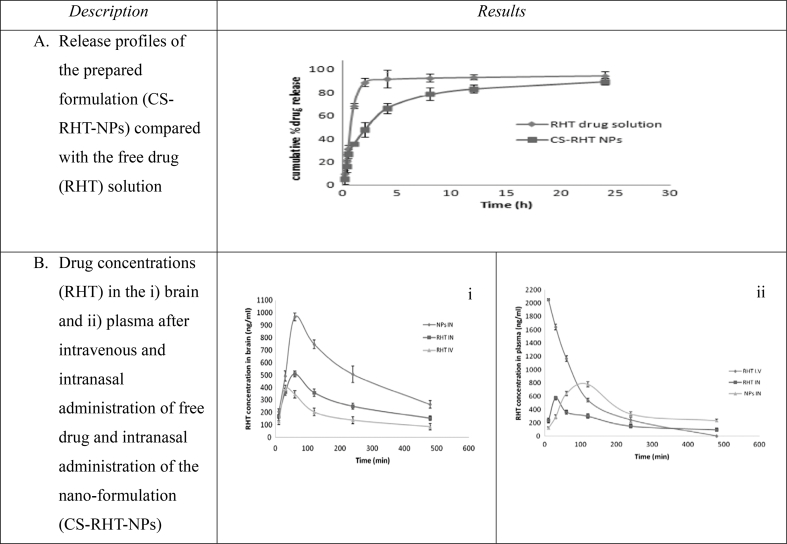
A sustained release profile was observed when the prepared NPs were subjected to an in vitro release study. The release mechanism showed an initial rapid release of the drug in the first hour and a subsequent slow release over a 24 h period, in which 89.27% of rivastigmine was released. The sustained release profile correlated with the release of the drug from the core of the NP, whereas the initial rapid release was due to the release of rivastigmine from the surfaces of the NPs (Figure 7A).
-
2.
Drug permeability through the nasal mucosa
Figure 7.
Reproduced from Fazil et al., 2012. Rivastigmine (RHT) release from chitosan nanoparticles in vitro (A) and in vivo bioavailability of RHT (B).39
In vitro permeability tests were conducted to assess the ability of the formulation to penetrate the nasal mucosa and reach the brain. The results indicated greater drug permeation with the fabricated NPs than the free drug (70.1% vs 20.3% of the drug permeating the nasal mucosa in a 24 h period). This enhanced drug permeation was associated with chitosan's ability to interact with the negatively charged cell membranes of the mucosa, thus opening the tight junctions between cells.
-
3.
Drug concentrations in the brain
In vivo studies are essential to investigate the brain targeting ability of prepared NPs and the drug concentrations in the brain. On the basis of the concentration–time curves after administration of the free drug or the prepared NPs by intravenous and intranasal routes, the rivastigmine concentration in the brain was elevated after intranasal administration of rivastigmine loaded chitosan NPs, compared with intravenous or intranasal administration of free drug, in Wistar rats (Figure 7B i). Consequently, the plasma concentration of the prepared NPs was lower after intranasal administration than intravenous administration of the free drug (Figure 7B ii). Fazil et al.39 also calculated the brain–blood ratio of each formulation (free drug solution and rivastigmine loaded chitosan NPs) with both administration routes. The prepared NPs administered intranasally achieved the highest brain–blood ratio, of 1.712. In contrast, the lowest brain–blood ratio, 0.235, was observed after intravenous administration of rivastigmine. However, when rivastigmine solution was administered intranasally, a higher value was obtained, at 0.790.
These results indicate enhanced drug uptake in the brain through direct nose to brain delivery bypassing the BBB, thus providing a major advantage in enhancing drug bioavailability. The results have been attributed to the mucoadhesive property of chitosan limiting rapid mucociliary clearance in the nose, thereby enabling higher drug concentrations to reach the target site.
Polyester based NPs
Block copolymers are defined as various repeating units of a polymer segment arranged in a distinctive manner that alters the physicochemical properties of the polymer. This composition offers several benefits over conventional polymers, such as amphiphilicity; water solubility; prolonged stability of sensitive drugs; and, most importantly, enhanced release kinetics of encapsulated drugs, thus providing a sustained release profile that can be maintained over several weeks. Moreover, the use of suitable block copolymers enables encapsulation of both hydrophilic and hydrophobic drugs. Block copolymers result in small particle sizes (below 150 nm) that increase the surface area of the resultant NPs and improve the EE%.40 Further decreases in particle size facilitate drug delivery to the brain, through more efficient BBB permeation of NPs, thus increasing the drug's ability to reach the brain and promoting effective drug delivery.41
One frequently used block copolymer is PEG-polycaprolactone (PEG-PCL). PCL, a hydrophobic polymer composed of polar ester groups and methylene groups, is effective in encapsulating hydrophobic drugs. PCL is known for its slow metabolism in the body, thus providing sustained drug release. However, PEG is hydrophilic and, when incorporated into NPs, it decreases immunogenicity by hindering phagocytosis. Conjugation of both polymers yields a block copolymer with enhanced biodegradability and circulation time.42
Sarathlal et al.43 have investigated the effects of entrapping a peroxisome proliferator-activated receptor (PPAR) agonist with insulin sensitizing effects such as rosiglitazone in methylated PEG-PCL (m-PEG-PCL) NPs. The authors have suggested that the addition of stabilizing agents such as poloxamer to the NPs, an amphiphilic triblock copolymer, enhances NP permeability in the brain by inhibiting p-glycoprotein efflux in the BBB. This study is notable for encapsulating an antidiabetic drug to treat AD. Over the past decade, several studies have reported associations between AD and disrupted glucose metabolism and insulin signaling in the brain mimicking that in type 2 diabetes mellitus.43
Moreover, insulin receptors are present in regions in the brain that affect memory generation, and decrease free fatty acid release from adipose tissue through lipase inhibition. However, under pathological conditions affecting insulin receptors, the levels of free fatty acids are elevated, thus hindering the insulin degrading enzyme that is crucial for the clearance and degradation of Aβ peptides. In addition, elevated free fatty acids trigger the accumulation of amyloid and tau fibrils. Notably, because PPAR plays a role in maintaining insulin sensitivity, PPAR agonists might have strong prospects for treating AD. Studies have confirmed that PPAR agonists, such as rosiglitazone, alleviate oxidative stress in the brain, and decrease neuroinflammation through increasing glucose levels and lipid metabolism. However, rosiglitazone has poor brain permeability and causes severe adverse effects, such as cardiac toxicity. Thus, the encapsulation of rosiglitazone into polymeric NPs might alleviate the adverse effects of the drug and potentiate its therapeutic effects by enhancing brain permeation.43 Sarathlal et al.43 have prepared rosiglitazone loaded m-PEG-PCL NPs decorated with poloxamer, by using the double emulsion solvent evaporation method. The resultant particle size of 124.6 ± 12 nm was considered optimal for the drug to reach the target site; the zeta potential of −17.5 ± 5.54 mV suggested the stability of the system; and the EE% was high, reaching 70.65%. The potential for AD treatment was assessed in several experimental assays evaluating the neuroprotective and anti-AD effects of the fabricated NPs.43
Evaluation of the activity of fabricated NPs against AD43
-
1.
Effects of nano-formulations on oxidative stress
Assessing the effects of nano-formulations on the antioxidant enzymes in the brain is essential for determining the therapeutic efficacy of a drug. Sarathlal et al.43 compared the effects of the free drug and the nano-formulation on SOD and glutathione. Increasing the dose of the free drug increased the levels of SOD and glutathione, thus confirming its antioxidant effect. However, the nano-formulation further increased the levels of SOD and glutathione beyond those observed with the free drug at the same concentration (5 mg/kg) (Figure 8 A). Thus, the nano-formulation provides greater neuroprotective effects at lower doses than the free drug.
-
2.
Effects of rosiglitazone on AChE
Figure 8.
Collection of results revealing the effects of rosiglitazone loaded poloxamer stabilized m-PEG-PCL NPs, based on the data from.43
A quantitative assay of AChE in mouse brains was conducted to determine the effect of the drug on this enzyme. AChE decreased after administration of the free drug at high doses (10 and 20 mg/kg). However, a significant decrease in enzymatic activity was observed after treatment of mouse cells with NPs at lower doses (5 mg/kg) (Figure 8B).
-
3.
Evaluation of the neuroinflammatory effects of prepared NPs
Measuring inflammatory markers such as TNF-α and IL-6 in the brain is essential to confirm the neuroprotective effects of a proposed drug and prepared NPs. After treatment with free drug at high concentrations (10 and 20 mg/kg), the levels of TNF-α and IL-6 markedly decreased However, fabricated NPs administered at a dose of 5 mg/kg markedly decreased the cytokines, to levels similar to those observed after treatment with free drug at 10 mg/kg (Figure 8C), thus indicating potent anti-inflammatory effects.
-
4.
Cell viability assays
On the basis of MTT assays, increasing the drug concentration decreases cell viability. Therefore, the safety of rosiglitazone on neuronal cells is questionable, and further cytotoxic studies with the prepared NPs and free drug solution should be conducted to evaluate whether the prepared NPs, as compared with the free drug, increase cell viability.
-
5.
Histopathological analysis
Histopathological analyses of mouse hippocampus have been performed to determine the effects of rosiglitazone loaded m-PEG-PCL NPs on neurons. The diseased cells before treatment with the prepared NPs showed low neuron density, and appeared sickle shaped and shrunken. Administration of the free drug at a dose of 5 mg/kg did not prevent cell loss, whereas, at higher concentrations, rosiglitazone successfully decreased neuronal cell loss. In contrast, at dose of 5 mg/kg, the prepared NPs, compared with the free drug, enhanced the neuronal cell density, and healthy cells were observed with spherical nuclei and clear cytoplasm.
-
6.
Effects of prepared NPs on messenger ribonucleic acid expression levels
Sarathlal et al.43 measured the expression levels of various genes and growth factors, including nerve growth factor (NGF) and PPAR, involved in cognitive functions such as memory formation. Increased messenger ribonucleic acid (mRNA) expression was observed after treatment with both the synthesized NPs and the free drug, in a dose dependent manner. Interestingly, lower doses of the nano-formulation than the free drug were required for gene upregulation. This crucial finding suggests improved cognitive function and neuroprotective effects associated with the proposed formulation (Figure 8D).
Generally, the above results confirmed that embedding the antidiabetic agent rosiglitazone into poloxamer stabilized m-PEG-PCL NPs decreases the dose required to exert the therapeutic effects mediated by PPAR activation. The formulation induced neuroprotective action through alleviating oxidative stress, and decreasing neuroinflammation by increasing antioxidant enzyme activity in the brain. In addition, the decrease in AChE activity was notable, because acetylcholine plays a key role in cognitive function. Furthermore, upregulating the levels of genes such as PPAR potentiated the neuroprotective effects of the drug. However, further experiments regarding cellular uptake and the clearance Aβ in the brain should be conducted to confirm the crossing of the BBB. Release kinetics and cytotoxicity assays should also be examined with the prepared NPs and the free drug to confirm safety in cells.
The above studies have demonstrated that polymeric NPs are strong candidates for treating AD. Such strategies should be considered in further studies, and the ideal NPs should be subjected to clinical trials.
Each study (Table 1) illustrated how the prepared formulation alleviated the hallmarks of AD, through a series of experiments demonstrating the hypothesis that the authors suggested. Size is an important factor in formulating NPs, because it controls drug passage across the BBB, a barrier limiting the entry of large molecules. Thus, smaller NP size is associated with better access to the brain. For instance, the native PLGA NPs and rosiglitazone loaded m-PEG-PCL NPs had the smallest size of therapeutics among studies (Table 1) and therefore would facilitate BBB crossing. Improvement of drug bioavailability is a critical feature of drug entrapment in NPs. A prior study has shown that fucoxanthin has poor water solubility and weak penetration of the BBB, whereas loading the drug in polymeric NPs increases its bioavailability and therapeutic efficacy by enhancing its delivery to site of action; however its encapsulation into NPs remains low (less than 50%). Additionally, the intranasal approach is a critical route of administration that should be considered, because it directly transports the drug to the brain. Fazil et al.39 have demonstrated the effects of rivastigmine loaded chitosan NPs in brain targeting after intranasal administration. The mucoadhesive properties of chitosan facilitate nose to brain delivery, and its cationic charge opens the tight junctions in the brain and thereby promotes drug delivery. Consequently, higher drug concentrations have been observed in the brain than the plasma. In addition, the highest drug release was observed when the drug was encapsulated in chitosan, on the basis of higher EE% (Table 1) than reported in the other studies described herein. Furthermore, drug repurposing is a novel approach used to develop promising treatments for various diseases. This strategy has been applied by Sarathlal et al.43 for AD treatment with rosiglitazone embedded in poloxamer stabilized m-PEG-PCL NPs. That study revealed the positive effects of the fabricated NPs on the examined cell lines, thus demonstrating the utility of this approach.
Future perspectives
As presented above, research is now being directed toward formulating ideal NPs for delivering anti-AD drugs with the highest possible efficiency. A recent study has suggested combining the benefits of two various polymers in a single NPs. In that study,44 PLGA was selected for its biocompatibility, ease of surface modification, and, more importantly, its anti-amyloid activity. Additionally, chitosan was used to decorate this nano-system, because of its mucoadhesive property and charge characteristics that facilitate nose to brain delivery of the encapsulated drug while bypassing the BBB. This approach achieved successful permeation of the nasal mucosa, thus suggesting a high distribution of the prepared NPs to the brain. In addition, the results revealed high uptake by target cells of the proposed NPs compared with PLGA loaded NPs.44 Therefore, on the basis of the experimental results, combining PLGA and chitosan provides a favorable drug carrier with optimal characteristics for intranasal delivery. Encapsulating aducanumab in polymeric NPs composed of PLGA and chitosan decorated with Aβ binding peptides—such as KLVFF peptide, which acts as an Aβ targeting ligand—has been hypothesized to enable site-specific binding of Aβ aggregates45 while limiting drug exposure at other sites of the brain. Hence, this treatment could potentially decrease the aforementioned adverse effects of the mAb, and increase the drug concentration delivered to the brain through intranasal administration. However, several characterization tests should be conducted to support this hypothesis.
Conclusion
Polymeric NPs exhibit a range of beneficial features that enhance the therapeutic action of embedded drugs and serve as promising candidates to treat the debilitating pathophysiology of AD. Several studies have demonstrated the efficacy of using a wide range of polymers to formulate stable and effective NPs to decrease AD predisposing factors, such as by alleviating amyloid plaques, oxidative stress, and neuroinflammation, as well as providing neuroprotection to ensure the safety of the prepared formulation for target cells. Bypassing the BBB is an essential feature for successful drug delivery enabled by polymeric NPs. For instance, rivastigmine's bioavailability is enhanced by its entrapment in chitosan NPs. The formulation was given intranasally to improve BBB crossing, thus increasing the drug concentrations in the brain, given the attractive physicochemical properties of chitosan. This route of administration is currently receiving attention from researchers for its ease of administration and substantial increase in drug bioavailability. Additionally, the safety profile of administered drugs is a crucial aspect of concern for researchers, given that the main goal of treating AD is to provide safe and effective therapy. Therefore, research in the past decade has focused on exploring the potential effects of herbal and natural active ingredients as anti-AD drugs. The tested therapeutic agents include fucoxanthin and curcumin, both of which have high safety profiles. When encapsulated in PLGA based NPs, both drugs exhibit potent antioxidant effects, along with an anti-amyloid action (exhibited by curcumin) and neuroprotective action (attained by fucoxanthin); consequently, they have potential as promising anti-AD agents. Notably, native PLGA NPs are strong candidates for anti-amyloid agents that, when encapsulated with drugs such as curcumin, may have synergistic effects and maximal anti-amyloid action.
Furthermore, recent studies have shown that incorporation of anti-diabetic drugs such as rosiglitazone may aid in treating AD. AD is associated with impaired insulin signaling and glucose metabolism. Loading rosiglitazone in m-PEG-PCL NPs has been found to decrease AChE enzyme activity and to increase the mRNA expression of various growth factors, thus enhancing cognitive function.
Finally, treating AD is an ongoing challenge faced by researchers throughout the past decade. However, extensive efforts have led to several nanotechnological approaches that may enhance site specific drug delivery, and achieve high bioavailability and efficacy.
Source of funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not for-profit sectors.
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
There are no ethical issues.
Authors contributions
YSME and AAE drafted, reviewed, and revised the manuscript of this review article. All authors have critically reviewed and approved the final draft, and are responsible for the content and similarity index of the manuscript.
Footnotes
Peer review under responsibility of Taibah University.
References
- 1.Sahni J.K., Doggui S., Ali J., Baboota S., Dao L., Ramassamy C. Neurotherapeutic applications of nanoparticles in Alzheimer's disease. J Contr Release. 2011;152:208–231. doi: 10.1016/j.jconrel.2010.11.033. [DOI] [PubMed] [Google Scholar]
- 2.Tosi G., Musumeci T., Ruozi B., Carbone C., Belletti D., Pignatello R., et al. The “fate” of polymeric and lipid nanoparticles for brain delivery and targeting: strategies and mechanism of blood–brain barrier crossing and trafficking into the central nervous system. J Drug Deliv Sci Technol. 2016;32:66–76. doi: 10.1016/J.JDDST.2015.07.007. [DOI] [Google Scholar]
- 3.Hu L., Tao Y., Jiang Y., Qin F. Recent progress of nanomedicine in the treatment of Alzheimer's disease. Front Cell Dev Biol. 2023;11 doi: 10.3389/FCELL.2023.1228679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kanojia N., Thapa K., Kaur G., Sharma A., Puri V., Verma N. Update on Therapeutic potential of emerging nanoformulations of phytocompounds in Alzheimer's and Parkinson's disease. J Drug Deliv Sci Technol. 2023;79 doi: 10.1016/j.jddst.2022.104074. [DOI] [Google Scholar]
- 5.Fonseca L.C., Lopes J.A., Vieira J., Viegas C., Oliveira C.S., Hartmann R.P., et al. Intranasal drug delivery for treatment of Alzheimer's disease. Drug Deliv Transl Res. 2021;11:411–425. doi: 10.1007/S13346-021-00940-7/FIGURES/3. [DOI] [PubMed] [Google Scholar]
- 6.Rabiee N., Ahmadi S., Afshari R., Khalaji S., Rabiee M., Bagherzadeh M., et al. Polymeric nanoparticles for nasal drug delivery to the brain: relevance to Alzheimer's disease. Adv Ther (Weinh) 2021;4 doi: 10.1002/ADTP.202000076. [DOI] [Google Scholar]
- 7.Pardridge W.M. Treatment of Alzheimer's disease and blood–brain barrier drug delivery. Pharmaceuticals. 2020;13:1–25. doi: 10.3390/PH13110394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rahman A., Hossen M.A., Chowdhury M.F.I., Bari S., Tamanna N., Sultana S.S., et al. Aducanumab for the treatment of Alzheimer's disease: a systematic review. Psychogeriatrics. 2023;23:512–522. doi: 10.1111/PSYG.12944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Khanna G., Bhandari R., Kuhad A., Kuhad A. Aducanumab. Drugs Future. 2023;44:115–121. doi: 10.1358/dof.2019.44.2.2895649. [DOI] [Google Scholar]
- 10.Cao Y., Zhang R. The application of nanotechnology in treatment of Alzheimer's disease. Front Bioeng Biotechnol. 2022;10 doi: 10.3389/FBIOE.2022.1042986/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kanojia N., Thapa K., Kaur G., Sharma A., Puri V., Verma N. Update on Therapeutic potential of emerging nanoformulations of phytocompounds in Alzheimer's and Parkinson's disease. J Drug Deliv Sci Technol. 2023;79 doi: 10.1016/J.JDDST.2022.104074. [DOI] [Google Scholar]
- 12.Zhang W., Mehta A., Tong Z., Esser L., Voelcker N.H. Development of polymeric nanoparticles for blood–brain barrier transfer—strategies and challenges. Adv Sci. 2021;8 doi: 10.1002/ADVS.202003937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chakraborty A., Mohapatra S.S., Barik S., Roy I., Gupta B., Biswas A. Impact of nanoparticles on amyloid β-induced Alzheimer's disease, tuberculosis, leprosy and cancer: a systematic review. Biosci Rep. 2023;43 doi: 10.1042/BSR20220324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fonseca-Santos B., Gremião M.P.D., Chorilli M. Nanotechnology-based drug delivery systems for the treatment of Alzheimer's disease. Int J Nanomed. 2015;10:4981. doi: 10.2147/IJN.S87148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sarathlal K.C.S., Kakoty V., Krishna K.V., Dubey S.K., Chitkara D., Taliyan R. Neuroprotective efficacy of Co-encapsulated rosiglitazone and vorinostat nanoparticle on streptozotocin induced mice model of Alzheimer disease. ACS Chem Neurosci. 2021;12:1528–1541. doi: 10.1021/acschemneuro.1c00022. [DOI] [PubMed] [Google Scholar]
- 16.Hanafy A.S., Farid R.M., Helmy M.W., ElGamal S.S. Pharmacological, toxicological and neuronal localization assessment of galantamine/chitosan complex nanoparticles in rats: future potential contribution in Alzheimer's disease management. Drug Delivery. 2016;23:3111–3122. doi: 10.3109/10717544.2016.1153748. [DOI] [PubMed] [Google Scholar]
- 17.Wilson B., Samanta M.K., Muthu M.S., Vinothapooshan G. Design and evaluation of chitosan nanoparticles as novel drug carrier for the delivery of rivastigmine to treat Alzheimer's disease. Ther Deliv. 2011;2:599–609. doi: 10.4155/tde.11.21. [DOI] [PubMed] [Google Scholar]
- 18.La Barbera L., Mauri E., D'Amelio M., Gori M. Functionalization strategies of polymeric nanoparticles for drug delivery in Alzheimer's disease: current trends and future perspectives. Front Neurosci. 2022;16 doi: 10.3389/FNINS.2022.939855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Moradi S.Z., Momtaz S., Bayrami Z., Farzaei M.H., Abdollahi M. Nanoformulations of herbal extracts in treatment of neurodegenerative disorders. Front Bioeng Biotechnol. 2020;8 doi: 10.3389/FBIOE.2020.00238/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sehar U., Rawat P., Reddy A.P., Kopel J., Reddy P.H. Amyloid beta in aging and Alzheimer's disease. Int J Mol Sci. 2022;23 doi: 10.3390/IJMS232112924. Page 12924 2022;23:12924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Halliday G., Robinson S.R., Shepherd C., Kril J. Alzheimer's disease and inflammation: a review of cellular and therapeutic mechanisms. Clin Exp Pharmacol Physiol. 2000;27:1–8. doi: 10.1046/J.1440-1681.2000.03200.X. [DOI] [PubMed] [Google Scholar]
- 22.Anand B., Wu Q., Nakhaei-Nejad M., Karthivashan G., Dorosh L., Amidian S., et al. Significance of native PLGA nanoparticles in the treatment of Alzheimer's disease pathology. Bioact Mater. 2022;17:506–525. doi: 10.1016/J.BIOACTMAT.2022.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nicsanu R., Cervellati C., Benussi L., Squitti R., Zanardini R., Rosta V., et al. Increased serum beta-secretase 1 activity is an early marker of Alzheimer's disease. J Alzheim Dis. 2022;87:433. doi: 10.3233/JAD-215542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Medeiros R., Baglietto-Vargas D., Laferla F.M. The role of tau in Alzheimer's disease and related disorders. CNS Neurosci Ther. 2011;17:514. doi: 10.1111/J.1755-5949.2010.00177.X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ozben T., Ozben S. Neuro-inflammation and anti-inflammatory treatment options for Alzheimer's disease. Clin Biochem. 2019;72:87–89. doi: 10.1016/J.CLINBIOCHEM.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 26.La Barbera L., Mauri E., D'Amelio M., Gori M. Functionalization strategies of polymeric nanoparticles for drug delivery in Alzheimer's disease: current trends and future perspectives. Front Neurosci. 2022;16 doi: 10.3389/FNINS.2022.939855/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Baracaldo-Santamaría D., Avendaño-Lopez S.S., Ariza-Salamanca D.F., Rodriguez-Giraldo M., Calderon-Ospina C.A., González-Reyes R.E., et al. Role of calcium modulation in the pathophysiology and treatment of Alzheimer's disease. Int J Mol Sci. 2023;24:9067. doi: 10.3390/IJMS24109067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Guan P.-P., Cao L.-L., Wang P. Elevating the levels of calcium ions exacerbate Alzheimer's disease via inducing the production and aggregation of β-amyloid protein and phosphorylated tau. Int J Mol Sci. 2021;22:5900. doi: 10.3390/IJMS22115900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Anand B., Wu Q., Nakhaei-Nejad M., Karthivashan G., Dorosh L., Amidian S., et al. Significance of native PLGA nanoparticles in the treatment of Alzheimer's disease pathology. Bioact Mater. 2022;17:506–525. doi: 10.1016/j.bioactmat.2022.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mathew A., Fukuda T., Nagaoka Y., Hasumura T., Morimoto H., Yoshida Y., et al. Curcumin loaded-PLGA nanoparticles conjugated with Tet-1 peptide for potential use in Alzheimer's disease. PLoS One. 2012;7 doi: 10.1371/journal.pone.0032616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Balasubramanian K. Molecular orbital basis for yellow curry spice curcumin's prevention of Alzheimer's disease. J Agric Food Chem. 2006;54:3512–3520. doi: 10.1021/JF0603533. [DOI] [PubMed] [Google Scholar]
- 32.Lockman P.R., Koziara J.M., Mumper R.J., Allen D. Nanoparticle surface charges alter blood–brain barrier integrity and permeability. J Drug Target. 2004;12:635–641. doi: 10.1080/10611860400015936. [DOI] [PubMed] [Google Scholar]
- 33.Yang M., Jin L., Wu Z., Xie Y., Zhang P., Wang Q., et al. PLGA-PEG nanoparticles facilitate in vivo anti-Alzheimer’s effects of fucoxanthin, a marine carotenoid derived from edible Brown algae. J Agric Food Chem. 2021;69:9764–9777. doi: 10.1021/acs.jafc.1c00569. [DOI] [PubMed] [Google Scholar]
- 34.Manek E., Darvas F., Petroianu G.A. Use of biodegradable, chitosan-based nanoparticles in the treatment of Alzheimer's disease. Molecules. 2020;25:4866. doi: 10.3390/MOLECULES25204866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Abbas H., El Sayed N.S., Youssef N.A.H.A., Gaafar P.M.E., Mousa M.R., Fayez A.M., et al. Novel luteolin-loaded chitosan decorated nanoparticles for brain-targeting delivery in a sporadic Alzheimer's disease mouse model: focus on antioxidant, anti-inflammatory, and amyloidogenic pathways. Pharmaceutics. 2022;14 doi: 10.3390/PHARMACEUTICS14051003/S1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu Z., Jiang M., Kang T., Miao D., Gu G., Song Q., et al. Lactoferrin-modified PEG-co-PCL nanoparticles for enhanced brain delivery of NAP peptide following intranasal administration. Biomaterials. 2013;34:3870–3881. doi: 10.1016/J.BIOMATERIALS.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 37.England R.J.A., Homer J.J., Knight L.C., Ell S.R. Nasal pH measurement: a reliable and repeatable parameter. Clin Otolaryngol Allied Sci. 1999;24:67–68. doi: 10.1046/J.1365-2273.1999.00223.X. [DOI] [PubMed] [Google Scholar]
- 38.Müller T. Rivastigmine in the treatment of patients with Alzheimer's disease. Neuropsychiatric Dis Treat. 2007;3:211. doi: 10.2147/NEDT.2007.3.2.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fazil M., Md S., Haque S., Kumar M., Baboota S., Sahni J.K., et al. Development and evaluation of rivastigmine loaded chitosan nanoparticles for brain targeting. Eur J Pharmaceut Sci. 2012;47:6–15. doi: 10.1016/j.ejps.2012.04.013. [DOI] [PubMed] [Google Scholar]
- 40.Revdekar A., Shende P. Block copolymers in Alzheimer's disease therapy: a perceptive to revolutionize biomaterials. J Contr Release. 2021;340:271–281. doi: 10.1016/J.JCONREL.2021.11.007. [DOI] [PubMed] [Google Scholar]
- 41.Hersh A.M., Alomari S., Tyler B.M. Crossing the blood-brain barrier: advances in nanoparticle technology for drug delivery in neuro-oncology. Int J Mol Sci. 2022;23:4153. doi: 10.3390/IJMS23084153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bhadran A., Shah T., Babanyinah G.K., Polara H., Taslimy S., Biewer M.C., et al. Recent advances in polycaprolactones for anticancer drug delivery. Pharmaceutics. 2023;15 doi: 10.3390/PHARMACEUTICS15071977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sarathlal K.C., Kakoty V., Marathe S., Chitkara D., Taliyan R. Exploring the neuroprotective potential of rosiglitazone embedded nanocarrier system on streptozotocin induced mice model of Alzheimer's disease. Neurotox Res. 2021;39:240–255. doi: 10.1007/s12640-020-00258-1. [DOI] [PubMed] [Google Scholar]
- 44.Dhas N., Mehta T. Intranasal delivery of chitosan decorated PLGA core/shell nanoparticles containing flavonoid to reduce oxidative stress in the treatment of Alzheimer's disease. J Drug Deliv Sci Technol. 2021;61 doi: 10.1016/J.JDDST.2020.102242. [DOI] [Google Scholar]
- 45.Yang X., Yang W., Xia X., Lei T., Yang Z., Jia W., et al. Intranasal delivery of BACE1 siRNA and rapamycin by dual targets modified nanoparticles for Alzheimer's disease therapy. Small. 2022;18 doi: 10.1002/SMLL.202203182. [DOI] [PubMed] [Google Scholar]