Abstract
During the coronavirus disease 2019 (COVID-19) pandemic, clinical prevention, early diagnosis, and hematological monitoring were challenging areas. This study aims to compare risk factors and hematological and biochemical data in non-survivor group patients with COVID-19 versus survivor group patients. A total of 204 patients with COVID-19 were selected as research subjects from December 2022 to January 2023. We analyzed the age, sex, time from onset to admission, and laboratory test indicators upon admission. The differences between surviving and deceased patients and mortality-related risk factors were examined. Among the 204 patients, 168 survived, whereas 36 died during hospitalization. Significant differences were observed between the two groups with COVID-19 across various factors, including age (p < 0.0001), WBC count (p < 0.0001), RBC count (p < 0.05), neutrophils (p < 0.0001), lymphocytes (p < 0.05), mean corpuscular hemoglobin concentration (MCHC) (p < 0.0001), RBC distribution width-standard deviation (RDW-SD) (p < 0.0001), RBC distribution width coefficient of variation (RDW-CV) (p < 0.0001), aspartate aminotransferase (AST) (p < 0.05), albumin (ALB) (p < 0.0001), creatinine (CR) (p < 0.0001), uric acid (UA) (p < 0.0001), blood urea nitrogen (BUN) (p < 0.0001), plasma thrombin time (TT) (p < 0.05), prothrombin time (PT) (p < 0.0001), and D-dimer (p < 0.0001). Multivariate logistic analysis revealed that older age, CR, UA, and ALB were independent factors associated with death (p < 0.05). Elderly patients with underlying diseases, abnormal routine blood test indices, and abnormal renal function and coagulation indices are at an increased worse prognosis and should be identified early. Age, UA, CR, and ALB can be used as predictors to assess the worse prognosis in the hospital.
Keywords: hematological examination, clinical chemistry, severity, risk factors, covid-19
Introduction
The novel coronavirus disease 2019 (COVID-19) [1] is an acute respiratory tract infection that was first identified in Wuhan and has caused a global pandemic since 2019. As of February 1, 2023, there have been more than 753 million confirmed cases and over 6.8 million deaths reported worldwide [2]. The main circulating strain in China was Omicron, along with mutations of the coronavirus. COVID-19 can present with a range of clinical manifestations, from asymptomatic infection to severe acute respiratory syndrome, septic shock, and acute respiratory distress syndrome (ARDS) [3]. Although the mortality rate is not as high, the increasing number of COVID-19 cases in China, with its large population base and significant aging population, poses challenges in terms of healthcare resources. Critically ill patients, especially those who are elderly or have underlying diseases, are at higher risk of death.
Recent studies in specialty medical journals have found that elderly patients or those with obesity, cardiovascular disease, diabetes, cancer, hematological malignancy, chronic kidney disease, and chronic obstructive pulmonary disease (COPD) have a higher risk of death compared to the general population [4,5].
Currently, real-time polymerase chain reaction (RT-PCR) is the best noninvasive and rapid diagnostic method for detecting the coronavirus [6,7]. However, RT-PCR has limitations in sensitivity and cannot assess the activity and severity of COVID-19 in patients [8]. Therefore, identifying blood biomarkers associated with the risk of death can be helpful in predicting clinical outcomes and monitoring treatment efficacy. Several biomarkers, including WBC count, lymphocyte count, D-dimer, RBC distribution width (RDW), and ALB have been shown in various studies to predict mortality [9-12].
The aim of this study is to analyze the clinical characteristics and prognostic risk factors of hospitalized COVID-19 patients in the early stages of the epidemic. Hematologic and biochemical abnormalities will be retrospectively analyzed in COVID-19 patients with severe and non-severe cases, providing valuable insights for the diagnosis and treatment of severe COVID-19 patients.
Materials and methods
Patients and study design
This study retrospectively analyzed the clinical records of 204 COVID-19 patients treated at prestigious hospitals in Anhui, China, from December 2022 to January 2023. The diagnosis of COVID-19 was primarily based on etiology, with RT-PCR (by Shanghai Bojie Medical Technology Co. Ltd., Pudong, China) and lung computed tomography (CT) being essential diagnostic methods. The inclusion criteria for this study were patients above 18 years old and not pregnant, according to the clinical diagnostic criteria for COVID-19. Exclusion criteria were pernicious tumor patients, radiation and chemotherapy treatment of patients, pregnancy, general systemic disease, severe infection, hematological disease, and incomplete clinical and imaging data. The patients were divided into two groups: survivors (168 patients) and non-survivors (36 patients).
Data collection
Patient demographics including age, sex, and time from onset to admission were collected, and laboratory examinations were conducted upon admission. Retrospective analysis of laboratory indexes was performed, including results from routine blood analysis (WBC count, RBC count, neutrophils, lymphocytes, hemoglobin, platelets; hematocrit (HCT); mean corpuscular volume (MCV); mean corpuscular hemoglobin (MCH); mean corpuscular hemoglobin concentration (MCHC); RBC distribution width-standard deviation (RDW-SD); RBC distribution width coefficient of variation (RDW-CV)), liver function (aspartate transaminase (AST), alanine transaminase (ALT), total bilirubin (TBIL), total protein (TP), ALB), renal function (creatinine (CR), uric acid (UA), blood urea nitrogen (BUN)), coagulation function (plasma prothrombin time (PT), activated partial thromboplastin time (APTT), plasma thrombin time (TT), fibrinogen (FIB)), and D-dimer. A comprehensive analysis was conducted to assess the differences between the survival and non-survival groups and identify worse prognosis factors.
Statistical analysis
Statistical analysis was performed using Statistical Product and Service Solutions (SPSS) (version 26; IBM SPSS Statistics for Windows, Armonk, NY). Data that followed a normal distribution were analyzed using a t-test, while non-normally distributed data were analyzed using medians and quartiles, and the Mann-Whitney U-test was employed. Binary logistic regression analysis was conducted to determine independent risk factors for mortality. A Spearman's rank correlation analysis was used to assess correlations between different independent factors. Furthermore, the diagnostic value of the risk factors in predicting death in severe patients was evaluated using a receiver operating characteristic (ROC) curve analysis. The ROC curve was generated, and the area under the curve (AUC) was calculated. The cutoff value was determined using the Youden index. Statistical significance was considered when the P value was less than 0.05.
Results
The study included a total of 204 COVID-19 patients, consisting of 66 females and 138 males, from December 2022 to January 2023. The patients were divided into two groups based on their prognosis: the survivor (168 cases) and non-survivor groups (36 cases). The median age of the survivor group ranged from 55 to 77 years old, with a mean age of 68 years old. The non-survivor group had a median age ranging from 71 to 83 years old, with a mean age of 80 years old. There was a significant difference in age distribution in the two groups (p < 0.0001), as shown in Table 1. However, there were no significant differences in sex ratio or hospital days between the two groups (p > 0.05).
Table 1. Baseline characteristics of the patients.
ALB: albumin; ALT: alanine transaminase; APTT: activated partial thromboplastin time; AST: aspartate transaminase; BUN: blood urea nitrogen; CR: creatinine; FIB: fibrinogen; HCT: hematocrit; IU: international units; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; MCV: mean corpuscular volume; M-F: male-female; PLT: platelet; PT: prothrombin time; RDW-CV: RBC distribution width coefficient of variation; RDW-SD: RBC distribution width-standard deviation; TBIL: total bilirubin; TP: total protein; TT: thrombin time; UA: uric acid
| Variable | Survivor | Non-survivor | P value |
| Age (y) | 68 (55-77) | 80 (71-83) | <0.0001 |
| Sex (M-F) | 110:58 | 28:8 | 0.152 |
| Hospital day | 11.00 (8.00-15.00) | 13.00 (7.5-17) | 0.342 |
| TP (g/L) | 66.55±7.18 | 66.29±9.00 | 0.217 |
| ALB (g/L) | 37.9 (34.55-40.5) | 32.90 (30.35-35.25) | <0.0001 |
| ALT (IU/L) | 23 (13-35) | 21.50 (14-45.5) | 0.756 |
| AST (IU/L) | 31.00 (23.75-47.25) | 40.50 (30.25-56.75) | <0.05 |
| TBIL (μmol/L) | 9.95 (5.93-13.4) | 10.65 (7.15-15.95) | 0.143 |
| BUN (mmol/L) | 5.73 (4.40-8.60) | 14.15 (9.58-20.68) | <0.0001 |
| CR (μmol/L) | 85 (59-119) | 148.00 (86.50-298.75) | <0.0001 |
| UA (μmol/L) | 278 (205.5-355) | 400.00 (292.50-540.50) | <0.0001 |
| PT (s) | 12.7 (12-13.7) | 13.70 (12.75-15.05) | <0.0001 |
| APTT (s) | 38.05 (33.58-41.3) | 38.00 (33.20-44.30) | 0.461 |
| TT (s) | 16.90 (15.80-18.30) | 17.70 (16.50-19.68) | <0.05 |
| FIB (g/L) | 4.95 (3.83-5.88) | 5.41 (4.27-6.46) | 0.129 |
| D-dimer (μg/mL) | 0.85 (0.44-1.66) | 2.07 (1.43-3.49) | <0.0001 |
| RDW-CV (%) | 13.2 (12.7-13.7) | 14.20 (13.70-15.45) | <0.0001 |
| RDW-SD | 45 (42.8-47.7) | 48.70 (45.30-52.13) | <0.0001 |
| Hemoglobin (g/L) | 123.51±19.97 | 115.19±23.98 | 0.119 |
| HCT (%) | 37.4 (32.7-40.3) | 37.30 (31.03-39.08) | 0.351 |
| MCV (fl) | 91 (87.9-94.5) | 92.20 (87.23-95.98) | 0.607 |
| MCH (pg) | 30.7 (29.4-31.9) | 30.00 (28.43-31.95) | 0.153 |
| MCHC (g/L) | 336.00 (329.00-324.00) | 326.00 (319.50-336.75) | <0.0001 |
| WBC (109/L) | 5.64 (4.25-8.07) | 9.36 (6.19-13.43) | <0.0001 |
| RBC (1,012/L) | 4.05±0.67 | 4.00±1.01 | <0.05 |
| Neutrophil (109/L) | 4.00 (2.80-6.80) | 8.25 (5.05-11.50) | <0.0001 |
| Lymphocyte (109/L) | 0.90 (0.50-1.30) | 0.65 (0.40-1.00) | <0.05 |
| PLT (109/L) | 159.00 (112.00-211.00) | 151.00 (107.25-210.00) | 0.732 |
Furthermore, various hematological, blood biochemical, coagulation function, and D-dimer parameters were evaluated. The AST, UA, CR, UA, PT, TT time, and D-dimer levels were significantly higher in the non-survivor group compared to the survivor group. Conversely, the serum ALB levels were lower in the non-survivor group. These differences were statistically significant (p < 0.05), as shown in Table 1. Additionally, the non-survivor group exhibited significantly higher WBC and neutrophil counts, as well as higher RDW-CV and RDW-SD, compared to the survivor group. Moreover, the non-survivor group had significantly lower blood cell and lymphocyte counts, as well as lower MCHC, compared to the survivor group. However, there were no significant differences in other data between the two groups.
It was determined through single-factor regression analysis that age, WBC count, neutrophil count, ALB, urea nitrogen, CR, UA, RDW-CV, RDW-SD, hemoglobin, MCH, and MCHC were independent risk factors associated with death from COVID-19 (p < 0.05). However, the other variables were not found to be significantly associated with the risk of death (p > 0.05) (Table 2). Furthermore, multiple regression analysis was performed on the factors identified through binary logistic regression. The results showed that age, low ALB levels, and elevated urea nitrogen and CR levels were the most important risk factors for death from COVID-19, as shown in Table 2.
Table 2. Univariate and multiple regression logistic regression analysis to identify risk factors associated with mortality in patients with COVID-19.
ALB: albumin; ALT: alanine transaminase; APTT: activated partial thromboplastin time; AST: aspartate transaminase; BUN: blood urea nitrogen; CI: confidence interval; CR: creatinine; FIB: fibrinogen; HCT: hematocrit; IU: international units; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; MCV: mean corpuscular volume; OR: odds ratio; PLT: platelet; PT: prothrombin time; RDW-CV: RBC distribution width coefficient of variation; RDW-SD: RBC distribution width-standard deviation; TBIL: total bilirubin; TP: total protein; TT: thrombin time; UA: uric acid
| Variables | Univariate | Multivariate | ||
| OR (95% CI) | P value | OR (95% CI) | P value | |
| Age | 1.067 (1.031,1.104) | <0.001 | 0.893 (0.845,0.943) | <0.001 |
| Hospital day | 1.040 (0.990,1.093) | 0.121 | ||
| TP (g/L) | 0.995 (0.948,1.044) | 0.848 | ||
| ALB (g/L) | 0.867 (0.808,0.931) | <0.001 | 1.152 (1.027,1.292) | <0.05 |
| ALT (IU/L) | 1.002 (0.998,1.006) | 0.365 | ||
| AST (IU/L) | 1.000 (0.999,1.022) | 0.536 | ||
| TBIL (μmol/L) | 1.010 (0.918,1.040) | 0.499 | ||
| BUN (mmol/L) | 1.019 (1.049,1.135) | <0.001 | ||
| CR (μmol/L) | 1.002 (1.000,1.003) | <0.05 | 0.997 (0.994,1.000) | <0.05 |
| UA (μmol/L) | 1.004 (1.002,1.004) | <0.001 | 0.995(0.991,1.000) | <0.05 |
| PT (s) | 1.240 (1.042,1.475) | <0.05 | ||
| APTT (s) | 1.006 (0.982,1.030) | 0.639 | ||
| TT (s) | 0.999 (0.974,1.024) | 0.913 | ||
| FIB (g/L) | 1.191 (0.981,1.444) | 0.077 | ||
| D-dimer (μg/mL) | 1.010 (0.997,1.024) | 0.124 | ||
| RDW-CV (%) | 1.841 (1.379,2.457) | <0.001 | ||
| RDW-SD | 1.136 (1.048,1.230) | <0.05 | ||
| Hemoglobin (g/L) | 0.918 (0.964,0.998) | <0.05 | ||
| HCT (%) | 0.961 (0.908,1.018) | 0.180 | ||
| MCV (fl) | 0.964 (0.917,1.014) | 0.154 | ||
| MCH (pg) | 0.821 (0.714,0.944) | <0.05 | ||
| MCHC (g/L) | 0.925 (0.891,0.961) | <0.001 | ||
| WBC (10^9/L) | 1.158 (1.065,1.259) | <0.001 | ||
| RBC (10^12/L) | 0.922 (0.567,1.499) | 0.742 | ||
| Neutrophil (10^9/L) | 1.203 (1.095,1.321) | <0.001 | ||
| Lymphocyte (10^9/L) | 1.001 (0.625,1.537) | 0.998 | ||
| PLT (10^9/L) | 1.001 (0.996,1.005) | 0.751 |
Correlation analysis was conducted using Spearman rank correlation to examine the relationship between the independent factors and other laboratory indexes. The results indicated that age, ALT, TBIL, UA, RDW-CV, RDW-SD, WBC count, and neutrophil count were positively correlated. Conversely, age showed a negative correlation with total protein and lymphocyte count (Table 3-5). In terms of ALB, it was found to be significantly associated with total protein, hemoglobin, and HCT. However, it exhibited a negative correlation with hospital day, AST, TBIL, UA, PT, APTT, FIB, RDW-CV, RDW-SD, MCV, MCH, WBC count, RBC count, and neutrophil count (Tables 3-5). Furthermore, CR showed a positive correlation with hospital day, AST, TBIL, UA, FIB, RDW-CV, RDW-SD, WBC count, and neutrophil count. However, it had a negative correlation with hemoglobin, HCT, MCHC, and RBC count (Table 3-5). The variables hospital day, total protein, AST, UA, PT, FIB, RDW-CV, RDW-SD, WBC count, neutrophil count, and lymphocyte count were positively associated with urea nitrogen. Conversely, MCV, MCH, and MCHC showed a significant negative correlation (Tables 3-5).
Table 3. The correlation coefficient between the independent factors and hospital day, liver function, and uric acid.
A P < 0.05 was considered statistically significant between the two data.
AST: aspartate aminotransferase; ALT: alanine transaminase; TBIL: total bilirubin
| Hospital day | Total protein | ALT | AST | TBIL | Uric acid | |
| Age (y) | 0.06 | -0.15a | -0.06 | 0.15a | 0.20a | 0.20a |
| Albumin (g/L) | -0.19a | 0.43a | 0.05 | -0.16a | -0.18a | -0.25a |
| Creatinine (μmol/L) | 0.18a | 0.06 | 0.01 | 0.20a | 0.16a | 0.79a |
| Urea nitrogen (μmol/L) | 0.20a | 0.14a | 0.06 | 0.18a | 0.13 | 0.62a |
Table 5. The correlation coefficient between the independent factors with routine blood test.
A P < 0.05 was considered statistically significant between the two data.
RDW-CV: RBC distribution width-coefficient of variation; RDW-SD: RBC distribution width-standard deviation; HCT: hematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; PLT: platelet
| RDW-CV | RDW-SD | Hemoglobin | HCT% | MCV | MCH | MCHC | PLT | WBC | RBC | Neutrophil | Lymphocyte | |
| Age (y) | 0.18a | 0.29a | -0.10 | -0.10 | 0.10 | 0.08 | -0.03 | -0.09 | 0.15a | -0.11 | 0.18a | -0.19a |
| Albumin (g/L) | -0.35a | -0.37a | 0.15a | 0.14a | -0.15a | -0.13 | 0.01 | -0.04 | -0.28a | 0.20a | -0.31a | 0.02 |
| Creatinine (μmol/L) | 0.18a | 0.22a | -0.42a | -0.39a | 0.04 | -0.03 | -0.14a | -0.01 | 0.20a | -0.37a | 0.24a | -0.09 |
| Urea nitrogen(μmol/L) | 0.35a | 0.14a | -0.03 | 0.01 | -0.18a | -0.24a | -0.18a | -0.08 | 0.34a | 0.09 | 0.32a | 0.20a |
Table 4. The correlation coefficient between the independent factors with blood coagulation examination.
a: P < 0.05 was considered statistically significant between the two data
PT: prothrombin time; APTT: activated partial thromboplastin time; TT: thrombin time; FIB: fibrinogen; D-D: D-dimer
| PT | APTT | TT | FIB | D-D | |
| Age (y) | 0.05 | 0.10 | 0.06 | 0.09 | 0.07 |
| Albumin (g/L) | -0.22a | -0.17a | -0.14 | -0.30a | -0.09 |
| Creatinine (μmol/L) | 0.04 | 0.14 | 0.11 | 0.21a | 0.06 |
| Urea (μmol/L) nitrogen | 0.26a | 0.10 | 0.02 | 0.23a | 0.05 |
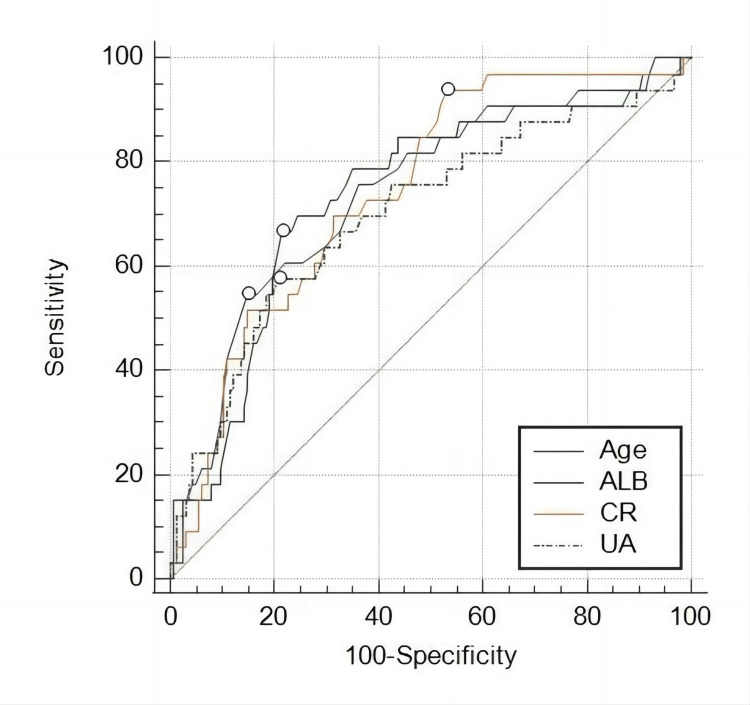
The area under the ROC curves was calculated to evaluate the predictive ability of age, ALB, CR, and BUN for mortality from COVID-19. The AUC value for age was 0.743 (95% CI= 0.675 to 0.802, P < 0.05), with a sensitivity of 54.55% and specificity of 85.19%. The cutoff value for age was determined to be 79 years old. For ALB, the AUC value was 0.734 (95% CI= 0.666 to 0.795, P < 0.05), with a cutoff value of 33.7 g/L. The sensitivity and specificity were 66.67% and 78.4%, respectively. In the case of CR, the AUC value was 0.744 (95% CI, 0.676-0.803; p < 0.05), with a sensitivity of 93.94% and specificity of 46.91%. The cutoff value for CR was 80.00 μmol/L. Moreover, the mountain curve analysis indicated that the model had a sensitivity of 57.58%, a specificity of 79.01%, and an AUC value of 0.701 (95% CI= 0.632 to 0.765, p < 0.05). The cutoff value for BUN was determined to be 373.00 μmol/L. The ROC curve and corresponding AUC values are presented in Figure 1 and Table 6.
Table 6. The ROC diagnostic model of the risk factors for death.
AUC: area under the curve; ALB: albumin; SE: serum electrolytes
| Variables | AUC (95% CI) | Sensitivity (%) | Specificity (%) | Youden index J | Cutoff value | SE |
| Age (y) | 0.743(0.675 to 0.802) | 54.55 | 85.19 | 0.40 | >79.00 | 0.049 |
| ALB (g/L) | 0.734(0.666 to 0.795) | 66.67 | 78.40 | 0.45 | ≤33.70 | 0.049 |
| Creatinine (μmol/L) | 0.744(0.676 to 0.803) | 93.94 | 46.91 | 0.41 | >80.00 | 0.044 |
| Urea nitrogen (μmol/L) | 0.701(0.632 to 0.765) | 57.58 | 79.01 | 0.37 | >373.00 | 0.055 |
Figure 1. ROC curve comparing the diagnostic validity of age, ALB, CR, and UA in the prediction of mortality among COVID-19 patients.
ALB: albumin; CR: creatinine; UA: uric acid
Discussion
Since December 2019, COVID-19 has spread worldwide, causing diverse global epidemic situations and various responses from different countries. Although the overall mortality rate of COVID-19 is not high, certain populations, particularly those with severe pneumonia, ARDS, acute kidney injury, and acute heart failure, have a higher risk of death [13]. Therefore, early detection and diagnosis of high-risk patients are crucial in reducing the mortality rate among severe cases. Analyzing changes in hematologic examinations in COVID-19 patients can help in assessing disease progression and prognosis.
In our study, we collected clinical data from 204 COVID-19 patients between December 2022 and January 2023 and divided them into survivor and non-survivor groups. The results of blood routine analysis showed that the non-survivor group had significantly higher counts of WBC, neutrophils, RDW-CV, and RDW-SD, and significantly lower levels of MCHC, RBC, and lymphocytes. These findings are consistent with previous research indicating that severe COVID-19 patients may experience more intense inflammatory reactions, leading to increased WBC and neutrophil counts [14-16]. The elevated neutrophil count may contribute to the development of cytokine storms, which can be fatal for critically ill patients [17]. Additionally, increased RDW levels have been identified as independent predictors of COVID-19 progression [18,19]. There are various reasons for increased RDW levels. First, an elevation in systemic inflammation might lead to increased RDW levels in COVID-19 patients. Furthermore, some COVID-19 patients experience disturbances in their internal environment, which might contribute to a shortened RBC survival, resulting in increased RDW. The decreased RBC count and MCHC suggest a potential worsening of anemia in COVID-19 patients. Furthermore, both survivor and non-survivor groups had lower lymphocyte counts compared to healthy individuals, indicating a close association between lymphocyte count decline and COVID-19 severity [20-22].
Coagulation disorders were more pronounced and prolonged in the non-survivor group, as evidenced by significantly elevated PT, TT, and D-dimer levels. Similar studies have indicated that PT and D-dimer levels reflect a hypercoagulable state in COVID-19 patients [23,24]. COVID-19, as an infectious disease, leads to an inflammatory response syndrome. The immune damage it causes is systemic. These coagulation disorders are associated with complex pathophysiology, which is not yet fully understood. The pathogenesis may be related to the development of circulating anticoagulants or impaired synthesis of clotting factors. Elevated D-dimer levels are associated with sepsis-induced coagulopathy and an increased risk of thromboembolism in severe COVID-19 cases [25,26]. Therefore, timely and dynamic monitoring of PT, TT, and D-dimer levels is crucial for preventing thromboembolic events [24].
Biochemical abnormalities in liver function, such as low levels of ALB and increased AST, were found to be associated with mortality. However, the changes in ALT, TP, and TBIL levels did not show significant differences between the two groups. These results are consistent with previous research indicating that elevated AST levels serve as a risk factor for COVID-19 mortality because of immune-mediated inflammatory responses and cytotoxic T cell activity [27,28]. Serum ALB, an acute phase reactant and a marker of inflammation, was significantly correlated with total protein, hemoglobin, and HCT levels, but it showed negative correlations with hospital day, AST, TBIL, UA, PT, APTT, FIB, RDW-CV, RDW-SD, MCV, MCH, WBC count, RBC count, and neutrophil count [29-31]. Serum ALB levels below 33.7 g/L were found to be a significant risk factor for mortality, as indicated by the ROC curve analysis with an AUC value of 0.73, sensitivity of 66.67%, and specificity of 78.4%.
COVID-19 can affect multiple vital organs in addition to the lungs, such as the liver, heart, and kidneys [32]. Studies have shown a significantly higher risk of death in COVID-19 patients with chronic kidney disease or acute kidney injury [4,33-35]. In line with these studies, our findings demonstrated significantly elevated levels of CR, BUN, and UA in the non-survivor group, indicating the impact of renal dysfunction on poor outcomes in COVID-19 patients. In the multivariate logistic regression analysis, older age was identified as an important risk factor for severe COVID-19 cases. Numerous studies have consistently shown that advanced age is associated with poor outcomes in COVID-19 patients [14,36-38]. The mortality risk in severe COVID-19 patients is increased when age exceeds 79 years, ALB is below 33.7 g/L, CR exceeds 80 μmol/L, and UA exceeds 373 μmol/L.
This study has some limitations, including a relatively small sample size and potential bias in the analysis because of the inclusion of different studies. Additionally, healthcare professionals may have limitations in understanding the characteristics of severe COVID-19 patients, leading to variations in treatment options. Despite these limitations, this study provides valuable insights into the mortality profile of COVID-19 patients and contributes to evidence-based decision-making in allocating limited medical resources.
Conclusions
While clinical hematological parameters may not assess the severity, our findings show that some parameters are independent predictors for evaluating patient outcomes. For example, older age, low levels of ALB, elevated CR, and increased UA are significant. Therefore, monitoring liver and kidney function, along with routine blood tests and coagulation assessments, can provide valuable information for early diagnosis and prognosis prediction. Further research is needed to validate these findings and to gain a better understanding of the factors influencing mortality in COVID-19 patients.
Acknowledgments
The authors would like to thank all the members of our team for their help to the study.
The authors have declared that no competing interests exist.
Funding Statement
This work was funded by the Natural Science Fund Project of Colleges in Anhui Province (No. KJ2020A0863) (No. KJ2019A1110), Anhui Medical College Innovation Team Research Fund Project (No. WJH202008t) (No.WJH202007t), and Teaching Team for Pathogenic Biology and Immunology (2020jxtd135).
Author Contributions
Concept and design: Shi Wei, Wu Xiaqin, Liu Liwei, Zhang Fasu, Pan Ying, Yu Furong
Acquisition, analysis, or interpretation of data: Shi Wei, Wu Xiaqin, Liu Liwei, Zhang Fasu, Tian Pingping, Yu Furong
Drafting of the manuscript: Shi Wei, Wu Xiaqin, Zhang Fasu, Tian Pingping, Yu Furong
Critical review of the manuscript for important intellectual content: Shi Wei, Wu Xiaqin, Liu Liwei, Zhang Fasu, Pan Ying
Supervision: Shi Wei, Wu Xiaqin, Liu Liwei, Zhang Fasu, Pan Ying, Tian Pingping, Yu Furong
Human Ethics
Consent was obtained or waived by all participants in this study
Animal Ethics
Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue.
References
- 1.Epidemiological and clinical characteristics of 3334 cases with prediagnosis coronavirus disease - 2019 (COVID-19) in Turkey. Simsek Yurt N, Ocak M, Yurt YC. Int J Clin Pract. 2021;75:0. doi: 10.1111/ijcp.14496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Coronavirus disease (COVID-19) epidemiological updates and monthly operational updates. February. [ May; 2024 ]. 2024. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports
- 3.Associations between hematological parameters and disease severity in patients with SARS-CoV-2 infection. Lin S, Mao W, Zou Q, Lu S, Zheng S. J Clin Lab Anal. 2021;35:0. doi: 10.1002/jcla.23604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Factors associated with COVID-19-related death using OpenSAFELY. Williamson EJ, Walker AJ, Bhaskaran K, et al. Nature. 2020;584:430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Global, regional, and national estimates of the population at increased risk of severe COVID-19 due to underlying health conditions in 2020: a modelling study. Clark A, Jit M, Warren-Gash C, et al. Lancet Glob Health. 2020;8:0–17. doi: 10.1016/S2214-109X(20)30264-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.A novel coronavirus from patients with pneumonia in China, 2019. Zhu N, Zhang D, Wang W, et al. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.The species severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. Nat Microbiol. 2020;5:536–544. doi: 10.1038/s41564-020-0695-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Benefits and limitations of serological assays in COVID-19 infection. Sidiq Z, Hanif M, Dwivedi KK, Chopra KK. Indian J Tuberc. 2020;67:0–6. doi: 10.1016/j.ijtb.2020.07.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Henry BM, de Oliveira MH, Benoit S, Plebani M, Lippi G. Clin Chem Lab Med. 2020;58:1021–1028. doi: 10.1515/cclm-2020-0369. [DOI] [PubMed] [Google Scholar]
- 10.Venous thromboembolism among hospitalized patients with COVID-19 undergoing thromboprophylaxis: a systematic review and meta-analysis. Chi G, Lee JJ, Jamil A, et al. J Clin Med. 2020;9:2489. doi: 10.3390/jcm9082489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.The role of serum inflammatory markers, albumin, and hemoglobin in predicting the diagnosis in patients admitted to the emergency department with a pre-diagnosis of COVID-19. Akman C, Bakırdöğen S. Rev Assoc Med Bras (1992) 2020;14:1037–1041. doi: 10.1590/1806-9282.67.Suppl1.20200917. [DOI] [PubMed] [Google Scholar]
- 12.Is albumin predictor of mortality in COVID-19? Violi F, Cangemi R, Romiti GF, et al. Antioxid Redox Signal. 2021;35:139–142. doi: 10.1089/ars.2020.8142. [DOI] [PubMed] [Google Scholar]
- 13.Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Lai CC, Shih TP, Ko WC, Tang HJ, Hsueh PR. Int J Antimicrob Agents. 2020;55:105924. doi: 10.1016/j.ijantimicag.2020.105924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Neutrophil-to-lymphocyte ratio as an independent risk factor for mortality in hospitalized patients with COVID-19. Zhang JJ, Cao YY, Tan G, et al. https://pubmed.ncbi.nlm.nih.gov/32662525/ Allergy. 2021;76:533–550. [Google Scholar]
- 15.Risk factors of severe cases with COVID-19: a meta-analysis. Ou M, Zhu J, Ji P, et al. Epidemiol Infect. 2020;148:0. doi: 10.1017/S095026882000179X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Qin C, Zhou L, Hu Z, et al. Clin Infect Dis. 2020;71:762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Laboratory abnormalities and risk factors associated with in-hospital death in patients with severe COVID-19. Chen X, Yan L, Fei Y, Zhang C. J Clin Lab Anal. 2020;34:0. doi: 10.1002/jcla.23467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Significance of MPV, RDW and PDW with the severity and mortality of COVID-19 and effects of acetylsalicylic acid use. Aydınyılmaz F, Aksakal E, Pamukcu HE, et al. Clin Appl Thromb Hemost. 2021;27 doi: 10.1177/10760296211048808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Diagnostic accuracy of the RDW for predicting death in COVID-19. Guaní-Guerra E, Torres-Murillo B, Muñoz-Corona C, Rodríguez-Jiménez JC, Macías AE, Scavo-Montes DA, Alvarez JA. Medicina (Kaunas) 2022;58:613. doi: 10.3390/medicina58050613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Clinical, radiological, and laboratory characteristics and risk factors for severity and mortality of 289 hospitalized COVID-19 patients. Zhang JJ, Cao YY, Tan G, et al. Allergy. 2021;76:533–550. doi: 10.1111/all.14496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Clinical characteristics of 182 pediatric COVID-19 patients with different severities and allergic status. Du H, Dong X, Zhang JJ, et al. Allergy. 2021;76:510–532. doi: 10.1111/all.14452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Prognostic value of leukocytosis and lymphopenia for coronavirus disease severity. Huang G, Kovalic AJ, Graber CJ. Emerg Infect Dis. 2020;26:1839–1841. doi: 10.3201/eid2608.201160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.COVID-19 coagulopathy, thrombosis, and bleeding. Chan NC, Weitz JI. Blood. 2020;136:381–383. doi: 10.1182/blood.2020007335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.D-dimer and prothrombin time are the significant indicators of severe Covid-19 and poor prognosis. Long H, Nie L, Xiang X, et al. Biomed Res Int. 2020;2020 doi: 10.1155/2020/6159720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pulmonary thrombosis and thromboembolism in COVID-19. Poor HD. Chest. 2021;160:1471–1480. doi: 10.1016/j.chest.2021.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.A meta-analysis of potential biomarkers associated with severity of coronavirus disease 2019 (COVID-19) Danwang C, Endomba FT, Nkeck JR, Wouna DL, Robert A, Noubiap JJ. Biomark Res. 2020;8:37. doi: 10.1186/s40364-020-00217-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Longitudinal association between markers of liver injury and mortality in COVID-19 in China. Lei F, Liu YM, Zhou F, et al. Hepatology. 2020;72:389–398. doi: 10.1002/hep.31301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.COVID-19 and the liver: little cause for concern. Bangash MN, Patel J, Parekh D. Lancet Gastroenterol Hepatol. 2020;5:529–530. doi: 10.1016/S2468-1253(20)30084-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Clinical usefulness of red cell distribution width/albumin ratio to discriminate 28-day mortality in critically ill patients with pneumonia receiving invasive mechanical ventilation, compared with lacate/albumin ratio: a retrospective cohort study. Jeong JH, Heo M, Lee SJ, Jeong YY, Lee JD, Yoo JW. Diagnostics (Basel) 2021;11:2344. doi: 10.3390/diagnostics11122344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Association between red blood cell distribution width-to-albumin ratio and prognosis of patients with aortic aneurysms. Long J, Xie X, Xu D, et al. Int J Gen Med. 2021;14:6287–6294. doi: 10.2147/IJGM.S328035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Association between red blood cell distribution width-to-albumin ratio and diabetic retinopathy. Zhao F, Liu M, Kong L. J Clin Lab Anal. 2022;36:0. doi: 10.1002/jcla.24351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Postmortem findings in Italian patients with COVID-19: a descriptive full autopsy study of cases with and without comorbidities. Falasca L, Nardacci R, Colombo D, et al. J Infect Dis. 2020;222:1807–1815. doi: 10.1093/infdis/jiaa578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Outcomes of patients with end-stage kidney disease hospitalized with COVID-19. Ng JH, Hirsch JS, Wanchoo R, et al. Kidney Int. 2020;98:1530–1539. doi: 10.1016/j.kint.2020.07.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pathophysiology of COVID-19-associated acute kidney injury. Legrand M, Bell S, Forni L, Joannidis M, Koyner JL, Liu K, Cantaluppi V. Nat Rev Nephrol. 2021;17:751–764. doi: 10.1038/s41581-021-00452-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Evidence for and against direct kidney infection by SARS-CoV-2 in patients with COVID-19. Hassler L, Reyes F, Sparks MA, Welling P, Batlle D. Clin J Am Soc Nephrol. 2021;16:1755–1765. doi: 10.2215/CJN.04560421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pre-existing traits associated with Covid-19 illness severity. Ebinger JE, Achamallah N, Ji H, et al. PLoS One. 2020;15 doi: 10.1371/journal.pone.0236240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Risk factors for Covid-19 severity and fatality: a structured literature review. Wolff D, Nee S, Hickey NS, Marschollek M. Infection. 2021;49:15–28. doi: 10.1007/s15010-020-01509-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Risk factors for disease severity, unimprovement, and mortality in COVID-19 patients in Wuhan, China. Zhang J, Wang X, Jia X, et al. Clin Microbiol Infect. 2020;26:767–772. doi: 10.1016/j.cmi.2020.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]