Abstract
Background
To systematically analyze risk factors for delayed postpolypectomy bleeding (DPPB) in colorectum.
Methods
We searched seven large databases from inception to July 2022 to identify studies that investigated risk factors for DPPB. The effect sizes were expressed by relative risk (RR) and 95% confidence interval (95% CI). The heterogeneity was analyzed by calculating I2 values and performing sensitivity analyses.
Results
A total of 15 articles involving 24,074 subjects were included in the study. The incidence of DPPB was found to be 0.02% (95% CI, 0.01–0.03), with an I2 value of 98%. Our analysis revealed that male sex (RR = 1.64), history of hypertension (RR = 1.54), anticoagulation (RR = 4.04), polyp size (RR = 1.19), polyp size ≥ 10 mm (RR = 2.43), polyp size > 10 mm (RR = 3.83), polyps located in the right semicolon (RR = 2.48) and endoscopic mucosal resection (RR = 2.99) were risk factors for DPPB.
Conclusions
Male sex, hypertension, anticoagulation, polyp size, polyp size ≥ 10 mm, polyps located in the right semicolon, and endoscopic mucosal resection were the risk factors for DPPB. Based on our findings, we recommend that endoscopists should fully consider and implement effective intervention measures to minimize the risk of DPPB.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12876-024-03251-6.
Keywords: Colorectal polyps, Postoperative bleeding, Risk factors, Meta-analysis
Background
Colonoscopy is very practical for screening and preventing colon cancer, and its importance is self-evident [1]. Endoscopic colorectal polypectomy has been proven to be an effective method to reduce the mortality of colorectal cancer [2]. With the continuous development of modern medicine and the continuous improvement of treatment technology, the safety of colorectal polypectomy has been improved to a certain extent, the number of outpatient or daytime operations has gradually increased, and the number of inpatient operations has gradually decreased. At present, the main methods of endoscopic resection of colorectal polyps include hot snare polypectomy (HSP), cold snare polypectomy (CSP), endoscopic mucosal resection (EMR), endoscopic submucosal dissection (ESD), hot biopsy (HB) and argon plasma coagulation (APC). Additionally, complications after colorectal polypectomy are still an important problem for clinicians.
Delayed postpolypectomy bleeding (DPPB) is one of the complications that can occur after colorectal polypectomy; it most often occurs after 24 h, with an incidence of 0.6%∼0.9% [3]. DPPB is difficult to detect via emergency endoscopy because the bleeding location is often hidden in the intestines covered by feces, thus increasing the burden on the doctors’ diagnosis and treatment work. Additionally, the problems caused by DPPB, such as patient discomfort, prolonged hospitalization, increased medical costs, and even increased patient mortality, can affect the harmony of the doctor-patient relationship.
There are different reports in the literature on factors related to DPPB. Some studies have shown that the incidence of DPPB is higher among patients receiving anticoagulant therapy [4–6]. Some studies also showed that the incidence of DPPB was positively correlated with the size of polyps removed [7–9]. A meta-analysis has concluded that cardiovascular disease, hypertension, polyps larger than 10 mm, and polyps located in the right colon are important risk factors for delayed bleeding [10].However, there remains a lack of clarity regarding the use of preventive measures on the wound surface during operation [11, 12], and whether such factors will affect the incidence of DPPB. Therefore, the risk factors of DPPB still need further verification. This study aims to update, comprehensively analyze, and explore the relevant risk factors for the occurrence of DPPB, to further optimize clinical response strategies and provide reference.
Methods
Our search protocol was prospectively registered with PROSPERO (CRD 42,022,375,804). (Supplementary material 1)
Literature retrieval
Three large Chinese databases were searched: CNKI database, Wan Fang database, and Wei Pu database. The databases were searched from inception to July 1, 2022. The key words were polypectomy or polypectomy or bleeding after polypectomy. Four large English databases were searched: Web of Science, PubMed, Cochrane Library, and Embase. The keywords were Polyp, Postoperative Hemorrhages, Risk Factors, and their free words. There were no language restrictions. The combination of subject words, keywords, and free words was used to search the databases and merge the search results. The search strategy is followed in Supplementary material 2.
Inclusion criteria and exclusion criteria
The inclusion criteria were as follows: [1] prospective cohort study or retrospective case-control study; [2] an independent study with complete data and more than one control group with the same research purpose; [3] the study sample was adult patients (≥ 18 years old); and [4] the content of the study was the risk factors for delayed bleeding after colorectal polypectomy.
The exclusion criteria were as follows: [1] the research content did not involve or was not related to the risk factors for delayed bleeding after colorectal polypectomy; [2] duplicate publications repeatedly; [3] animal experiments, reviews, case reports, conference abstracts, dissertations; [4] studies without a control group; [5] the same researcher published similar documents; and [6] documents with incomplete data, unclear description, unavailable data, and limited research group.
Literature screening and data extraction
All the studies were independently screened by two researchers (ZX, SL). The researchers screened the titles, abstracts, and full texts in accordance with the inclusion and exclusion criteria, and the data were extracted into an Excel spreadsheet. The extracted data included [1] general data, including first author’s name, year of publication, and type of study; [2] baseline data, including sample size, sex, and age; [3] patient factors; [4] polyp factors, including polyp number, polyp diameter, polyp shape, polyp location, and polyp pathological type; [5] operation factors; and [6] the same effect size and 95% confidence interval (CI) were obtained by multivariate regression analysis. Any disagreement was resolved through discussion, consultation with a third researcher (JX) if necessary, and discussion or arbitration.
Quality assessment
Each study was independently evaluated by two researchers (ZX, SL). The Newcastle Ottawa scale was used to grade the included documents. The case-control study and cohort study were evaluated through three blocks and eight items, including the selection of study population, comparability, exposure evaluation or result evaluation. Any differences were resolved through discussion, and if necessary, a third researcher (JX) was consulted. The maximum score is 9 stars, and studies with ≥ 5 stars were included in the analysis.
Statistical analysis
STATA 15.1 was used for data analysis, and P < 0.05 was considered statistically significant. The relative risk (RR) was used as the effect index for the secondary classification variable and the combined effect quantity, and the effect quantity was expressed by the 95% confidence interval (95% CI). I2 > 50% indicated substantial heterogeneity between studies, and in such cases, the random effects model was used to pool and analyze the data; when I2 < 50%, the fixed effects model was used to pool and analyze the data. When heterogeneity was observed, sensitivity analysis was used to further explore the source of heterogeneity. Publication bias was evaluated by a funnel chart.
Results
Literature retrieval process and results
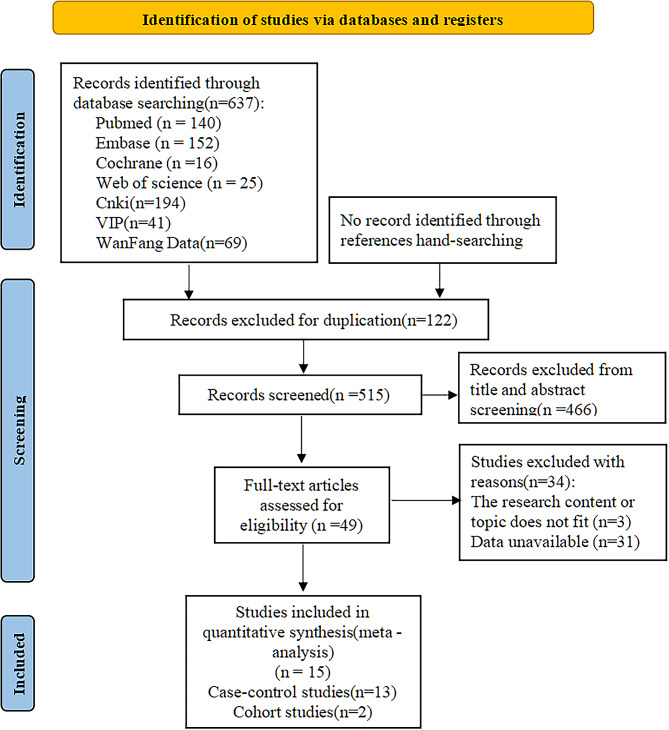
A total of 637 relevant studies were retrieved. After screening, 15 articles were ultimately included, including 13 case-control studies, 2 cohort studies, 11 English studies and 4 Chinese studies. The literature screening process is shown in Fig. 1.
Fig. 1.
Flow chart of the literature screening process
Basic characteristics of the included studies
A total of 15 studies with 24,074 study subjects were included: 897 patients were in the bleeding group, and 23,177 patients were in the control group. Patients with DPPB were included in the study, while patients with no DPPB were in the control group. The basic characteristics of the included studies are shown in Table 1 and the excluded studies are shown in Supplementary material 3.
Table 1.
Basic characteristics of the literature on risk factors for delayed postpolypectomy bleeding (DPPB).
| Researcher | Year | Nation | Study Type | Sample size(n) | Gender | Age | NOS | ||
|---|---|---|---|---|---|---|---|---|---|
| Male | Female | Bleeding Group | No Bleeding Group | ||||||
| Yoshikazu Inagaki | 2021 | Japan | case-control | 295 | 186 | 109 | 72.6 ± 8.3 | 68.6 ± 9.6 | 7 |
| Xianyi Lin | 2019 | China | case-control | 3962 | 2186 | 1776 | 51 ± 16 | 54 ± 10 | 6 |
| Peipei Li | 2019 | China | case-control | 287 | 199 | 88 | 56 ± 14 | 58 ± 12 | 6 |
| Zhe Luo | 2019 | China | case-control | 922 | 598 | 324 | 56.6 ± 12.3 | 58.8 ± 10.8 | 6 |
| Changqin Liu | 2019 | China | case-control | 709 | 468 | 241 | 62.71 ± 11.237 | 61.00 ± 9.376 | 7 |
| Peng Cheng | 2018 | China | case-control | 459 | 225 | 234 | 60.30 ± 10.66 | 58.16 ± 11.03 | 7 |
| Soo Kyung Park | 2018 | Korea | prospective cohort study | 3887 | 2661 | 1226 | 52.4 ± 12.3 | 55.8 ± 11.9 | 7 |
| Bum Su Choung | 2014 | Korea | case-control | 3788 | 2248 | 1540 | 60.21 ± 11.11 | 58.67 ± 11.40 | 7 |
| Hee Seok Moon | 2014 | Korea | case-control | 368 | 318 | 50 | 60.08 ± 13.36 | 60.62 ± 12.27 | 6 |
| Qiang Zhang | 2014 | China | case-control | 5600 | 3944 | 1656 | 47 ± 16 | 53 ± 14 | 7 |
| Jeong Ho Kim | 2013 | Korea | case-control | 210 | 155 | 55 | 58.0 ± 11.2 | 57.7 ± 11.2 | 8 |
| Xianrui Wu | 2013 | America | prospective cohort study | 120 | 62 | 58 | 69.9 ± 9.2 | 64.9 ± 12.2 | 6 |
| K. Tim Buddingh | 2011 | Netherlands | case-control | 156 | 73 | 80 | 66 ± 12 | 61 ± 12 | 8 |
| M.S. Sawhney | 2008 | America | case-control | 173 | 169 | 4 | 64.3 ± 16.7 | 65.4 ± 10.5 | 7 |
| Hirotsugu Watabe | 2006 | Japan | case-control | 3138 | 2578 | 560 | 61.4 ± 7.3 | 62.4 ± 10.1 | 6 |
NOS: Newcastle Ottawa scale
Literature quality evaluation
A total of 6 studies scored 6 stars [6, 13–17], 7 studies scored 7 stars [3, 7, 8, 18–21], and 2 studies scored 8 stars [22, 23] (Table 1).
Meta-analysis results
The incidence of DPPB
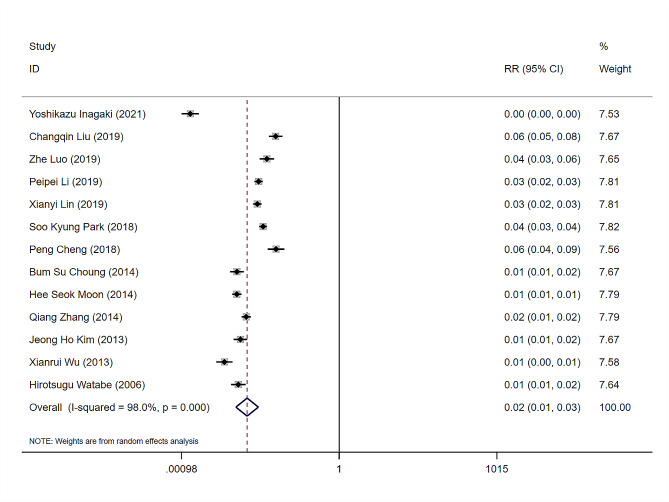
Thirteen studies [3, 6–8, 13–20, 22] have examined the incidence of DPPB (i.e., the number of DPPB cases/total number of cases), with I2 = 98%. The random effects model was used for pooled analysis, and the incidence of DPPB was 0.02, 95% CI (0.01–0.03). Among the included studies, the highest incidence of DPPB was 0.06, 95% CI (0.05–0.08), while the lowest incidence of DPPB was 0.00, 95% CI (0.00–0.00) (Fig. 2).
Fig. 2.
Forest plot of delayed postpolypectomy bleeding. RR: Relative Rate
Patient-related factors
The results showed that male sex (RR = 1.56, 95% CI: 1.34–1.81, P < 0.05), hypertension (RR = 1.27, 95% CI: 1.09–1.47, P < 0.05), cardiovascular disease (RR = 1.56, 95% CI: 1.23–1.97, P < 0.05), antithrombotic drugs (RR = 1.96, 95% CI: 1.24–3.09, P < 0.05), aspirin (RR = 1.50, 95% CI: 1.06–2.11, P < 0.05), and clopidogrel (RR = 1.89, 95% CI: 1.03–3.45, P < 0.05) (Fig.S1-6) were associated with increased risk of DPPB. Smoking, drinking, diabetes, cerebrovascular disease and warfarin were not related to the occurrence of DPPB (Table 2).
Table 2.
Single-factor logistic regression effect value meta-analysis
| Risk Factors | Study Number | Patient Number | Heterogeneity | RR 95% CI | P | ||
|---|---|---|---|---|---|---|---|
| I² (%) | P | ||||||
| Patient related factors | |||||||
| Male sex | 15 | 24,074 | 7.2 | 0.372 | 1.56(1.34–1.81) | 0.001 | |
| Smoking | 3 | 4830 | 82.4 | 0.003 | 1.36(0.50–3.66) | 0.545 | |
| Drinking | 2 | 3908 | 0.0 | 0.455 | 0.82(0.47–1.42) | 0.477 | |
| Hypertension | 11 | 14,885 | 22.8 | 0.226 | 1.27(1.09–1.47) | 0.002 | |
| Diabetes | 11 | 14,885 | 57.7 | 0.009 | 1.00(0.71–1.39) | 0.983 | |
| Cardiovascular disease | 9 | 10,539 | 0.0 | 0.549 | 1.56(1.23–1.97) | 0.001 | |
| Cerebrovascular disease | 4 | 1574 | 0.0 | 0.788 | 1.19(0.83–1.70) | 0.336 | |
| Antithrombotic drugs | 7 | 9257 | 77.0 | 0.000 | 1.96(1.24–3.09) | 0.004 | |
| Aspirin | 3 | 7923 | 46.2 | 0.156 | 1.50(1.06–2.11) | 0.021 | |
| Clopidogrel | 2 | 4082 | 0.0 | 0.363 | 1.89(1.03–3.45) | 0.038 | |
| Warfarin | 2 | 4082 | 81.7 | 0.020 | 2.70(0.57–12.68) | 0.208 | |
| Polyp-related factors | |||||||
| polyp number>3 | 2 | 7849 | 0.0 | 0.852 | 1.44(1.12–1.85) | 0.005 | |
| polyp number ≥ 3 | 2 | 746 | 98.3 | 0.000 | 6.5(0.43–98.3) | 0.176 | |
| Polyp size ≥ 10 mm | 3 | 3061 | 2.3 | 0.359 | 3.57(2.58–4.95) | 0.001 | |
| Pedunculated polyp | 3 | 4457 | 0.0 | 0.727 | 4.32(2.97–6.30) | 0.001 | |
| Polyp location | Left semicolon | 8 | 18,161 | 68.5 | 0.002 | 0.84(0.59–1.20) | 0.346 |
| Right semicolon | 8 | 18,161 | 69.1 | 0.002 | 1.14(0.80–1.62) | 0.482 | |
| Pathological type | Adenomas | 7 | 14,199 | 80.6 | 0.000 | 1.70(0.89–3.23) | 0.105 |
| Non-adenomas | 2 | 754 | 72.3 | 0.058 | 0.44(0.08–2.51) | 0.356 | |
| Serrated polyps | 3 | 9683 | 0.0 | 0.723 | 0.71(0.20–2.44) | 0.584 | |
| proliferative polyps | 4 | 10,392 | 72.3 | 0.013 | 0.67(0.30–1.49) | 0.327 | |
| malignancies | 5 | 13,445 | 0.0 | 0.933 | 2.66(1.49–4.75) | 0.001 | |
| Operational related factors | |||||||
| The way polyps removed | EMR | 6 | 13,904 | 52.2 | 0.063 | 2.34(1.44–3.82) | 0.001 |
| ESD | 2 | 919 | 55.4 | 0.134 | 3.62(0.76–17.32) | 0.107 | |
| HSP | 4 | 12,736 | 89.6 | 0.000 | 1.95(0.75–5.04) | 0.168 | |
| HB | 4 | 12,985 | 0.0 | 0.974 | 0.28(0.17–0.46) | 0.001 | |
| APC | 2 | 6309 | 99.6 | 0.000 | 3.60(0.00-52774.53) | 0.794 | |
| Preventive wound management | 2 | 3998 | 0.0 | 0.837 | 0.95(0.58–1.55) | 0.834 | |
| Endoscopists | inexperienced | 3 | 4792 | 87.9 | 0.000 | 1.55(0.42–5.69) | 0.512 |
| experienced | 3 | 4792 | 87.9 | 0.000 | 0.65(0.18–2.38) | 0.514 | |
APC: argon plasma coagulation; EMR: endoscopic mucosal resection; ESD: endoscopic submucosal dissection; HB: hot biopsy; HSP: hot snare polypectomy
Polyp-related factors
The results showed that the number of polyps > 3 (RR = 1.44, 95% CI: 1.12–1.85, P < 0.05), polyp size ≥ 10 mm (RR = 3.57, 95% CI: 2.58–4.95, P < 0.05), pedunculated polyps (RR = 4.32, 95% CI: 2.97–6.30, P < 0.05) and malignancies (RR = 2.66, 95% CI: 1.49–4.75, P < 0.05) (Fig. S7-10) were associated with an increased risk of DPPB. While polyp number ≥ 3, polyps located in left semicolon or the right semicolon, adenoma, non-adenoma, serrated polyp, and proliferative polyp were not associated with the incidence of DPPB (Table 2).
Operation-related factors
The results showed that EMR (RR = 2.34, 95% CI: 1.44–3.82, P < 0.05) (Fig.S11) was associated with an increased risk of DPPB, while HB (RR = 0.28, 95% CI: 0.17–0.46, P < 0.05) (Fig. S12) was associated with a decreased risk of DPPB. However, ESD, HSP, APC, preventive wound treatment, inexperienced endoscopists and experienced endoscopists were not associated with the occurrence of DPPB (Table 2).
Multivariate logistic regression effect value meta-analysis
The results showed male sex (RR = 1.64, 95% CI: 1.01–2.65, P < 0.05), hypertension (RR = 1.54, 95% CI: 1.15–2.07, P < 0.05), anticoagulation (RR = 4.04, 95% CI: 2.07–7.90, P < 0.05), polyp size (RR = 1.19, 95% CI: 1.10–1.30, P < 0.05), polyp size ≥ 10 mm (RR = 2.43, 95% CI: 1.80–3.29, P < 0.05), polyp size > 10 mm (RR = 3.83, 95% CI: 2.38–6.15, P < 0.05), polyp located in the right semicolon (RR = 2.48, 95% CI: 1.77–3.47, P < 0.05), and EMR (RR = 2.99, 95% CI: 1.06–8.45, P < 0.05) (Fig. S13-20) were associated with an increased risk of DPPB, while diabetes, aspirin, polyp number ≥ 3, pedunculated polyp, and HSP resection modes were not associated with the risk of DPPB (Table 3).
Table 3.
Multivariate logistic regression effect value meta-analysis
| Risk Factors | Study Number | Patient Number | Heterogeneity | RR 95% CI | P | ||
|---|---|---|---|---|---|---|---|
| I² (%) | P | ||||||
| Patient related factors | |||||||
| Male sex | 4 | 4843 | 29.5 | 0.235 | 1.64(1.01–2.65) | 0.045 | |
| Hypertension | 7 | 15,526 | 31.8 | 0.186 | 1.54(1.15–2.07) | 0.004 | |
| Diabetes | 3 | 7198 | 33.3 | 0.198 | 1.57(0.73–3.37) | 0.243 | |
| Anticoagulation | 2 | 3961 | 0.0 | 0.373 | 4.04(2.07–7.90) | 0.001 | |
| Aspirin | 3 | 8022 | 45.0 | 0.162 | 1.37(0.74–2.54) | 0.317 | |
| Polyp-related factors | |||||||
| polyp number ≥ 3 | 2 | 746 | 96.4 | 0.001 | 13.04(0.42-407.67) | 0.144 | |
| Polyp size | 4 | 4659 | 72.3 | 0.013 | 1.19(1.10–1.30) | 0.001 | |
| Polyp size | ≥ 10 mm | 5 | 7981 | 3.7 | 0.385 | 2.43(1.80–3.29) | 0.001 |
| >10 mm | 2 | 9388 | 30.4 | 0.231 | 3.83(2.38–6.15) | 0.001 | |
| Pedunculated polyp | 4 | 4630 | 81.0 | 0.001 | 1.92(0.80–4.63) | 0.147 | |
| Right semicolon | 5 | 8409 | 0.0 | 0.571 | 2.48(1.77–3.47) | 0.001 | |
| Operational related factors | |||||||
| Resection method | EMR | 4 | 9906 | 58.0 | 0.067 | 2.99(1.06–8.45) | 0.039 |
| HSP | 2 | 8738 | 0.0 | 0.539 | 1.78(0.80–3.97) | 0.157 | |
EMR: endoscopic mucosal resection; HSP: hot snare polypectomy
Publication bias evaluation
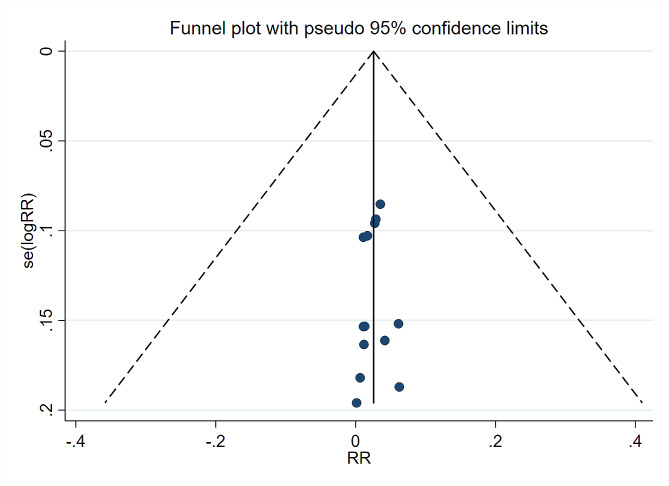
Visual inspection of the funnel plot was conducted to check for publication bias. Regarding the incidence of DPPB and the outcome with the largest number of included studies (13 articles), the funnel plot was observed to be symmetrical, indicating that there was no significant publication bias in the DPPB bleeding rate, as shown in Fig. 3.
Fig. 3.
Funnel plot with 95% confidence limits of delayed postpolypectomy bleeding. RR: Relative Rate
Sensitivity analysis
In this study, when the amount of heterogeneity for a factor was high (I2 > 50%), and the difference was statistically significant, sensitivity analyses were performed. Regarding multivariate logistic regression meta-analysis, EMR, sensitivity analysis (Fig. S20) revealed that excluding the study by Changqin Liu led to results that were outside of the 95% CI as well as a lower level of heterogeneity (I2 = 0.0%, P = 0.758) (Fig. S21). Therefore, this study may be a significant source of heterogeneity in the multivariate analysis of EMR. Regarding the polyp size and pedunculated polyp, sensitivity analysis revealed that the results remained within the 95% CI, and thus, the findings were stable. Due to the small number of included studies (2 articles), sensitivity analysis could not be performed to examine polyp number ≥ 3. Regarding single-factor logistic regression effect value meta-analysis, sensitivity analysis also revealed that the effects of smoking, diabetes, antithrombotic drugs, polyp located in left semicolon, right semicolon, adenoma, proliferative polyps, EMR, HSP, experienced and inexperienced endoscopists on the incidence of DPPB were stable. Due to the small number of included studies (2 articles), sensitivity analysis could not be performed to examine warfarin, polyp number ≥ 3, non-adenoma, ESD, and APC.
Discussion
As one of the complications that can occur after endoscopic resection of colorectal polyps, DPPB may cause hemorrhagic shock and increase the risk of mortality if it is not treated in a timely manner [24]. Moreover, most DPPB patients need to undergo colonoscopy again, thereby increasing patients’ pain and economic losses. Therefore, DPPB is an important problem for endoscopists, but its etiology and mechanism remain unclear. Domestic and foreign studies have reported that the occurrence of DPPB is related to a variety of factors. The current meta-analysis included both univariate logistic regression and multivariate logistic regression, and the results revealed that male sex, hypertension, Anticoagulation, polyp size, polyp size ≥ 10 mm, polyp located in the right semicolon, and EMR were risk factors for DPPB. The results of multivariate logistic regression meta-analysis showed a significant correlation, while the results of univariate logistic regression meta-analysis revealed polyps located in the right half colon were not associated with the risk of DPPB, which might be attributed to the correlation between it and confounding factors.
Regarding patient-related factors, this study found that male patients were more likely to develop DPPB. The reason for this association may be related to women are more likely to follow behavioral instructions [25]. Vascular endothelial cell dysfunction in patients with hypertension can seriously affect the systolic blood pressure, the diastolic function of blood vessels, and vascular sclerosis; furthermore, this dysfunction can lead to decreased blood elasticity and significantly decreased contractility of blood vessels at the broken end [26]. In addition, the effects of atherosclerosis and the elasticity of blood vessels are further weakened. Furthermore, blood pressure fluctuates greatly, and hemodynamics are unstable, which easily causes blood vessel rupture and bleeding at the cutting site [27]. This study found that oral antithrombotic drugs could increase the incidence of DPPB, which was consistent with the conclusions of Bum Su Choung et al. [19, 21] and Xianyi Lin et al. [13, 18, 21]. For the timing of antithrombotic drugs use, one research was screened for discontinuation of anticoagulants for 5 days [19], and the other research restricted the use of anticoagulants to heparin or warfarin within 1 week after polypectomy [21]. This may lead to heterogeneity and affect the results. The analysis of NOAC was not mentioned in this study because of the lack of exact data on specific NOAC in the included literature.
Previous studies have reported that polyp size was one of the important factors affecting the incidence of DPPB [13, 15, 21, 23]. Our meta-analysis also found that a larger polyp size was associated with a greater risk of DPPB, especially when the size of the removed polyp was greater than or equal to 10 mm. This association may be due to larger polyps’ size being associated with more nourishing blood vessels, larger wound caused by resection, a greater extent of damage to blood vessels, and increased difficulty with repairing the blood vessels. It remains unclear whether polyp location affects the risk of DPPB after colorectal polypectomy. Que et al. reported that the position of polyps in the right half colon or rectum was a risk factor for delayed postoperative bleeding [12]. Eleftheriadis D et al. found that delayed bleeding was more likely to occur after right half colon polypectomy [11]. However, Inagaki Y et al. found that DPPB was more likely to occur when the lesions were in the rectum [7].The results of this study showed that the removal of polyps located in the right colon increased the risk of DPPB. Previous studies [15, 18, 19, 22, 23] suggest that this association may be related to the histological variation in colon location (thinner submucosa) and the different manipulation techniques required at this site [19].
Regarding operational related factors, we found that EMR resection were more likely to develop DPPB. The reason may be attributed to the fact that EMR are primarily targeted towards polyps with a larger diameter, as these tend to have a higher vascularity. Inadequate handling of blood vessels during the procedure can potentially lead to delayed bleeding [3]. Argon plasma coagulation (APC) was used to remove smaller polyps (Diameter less than 5 mm) [20],while there is no significant association between it and DPPB was found in this study.
As for the prophylactic use of preventive endoscopic clip closure, only one of the included literatures met the criteria [19], so the relationship between preventive endoscopic clip closure and the risk of DPPB could not be analyzed, and further research is needed.
Some factors were significantly correlated with an increased risk of DPPB in the single-factor logistic regression meta-analysis but not in the multivariate logistic regression meta-analysis, including cardiovascular disease, aspirin, clopidogrel, polyp number > 3, pedunculated polyps, pathological type of malignancies, and treatment method of HB. The reason for this inconsistency may be the existence of a false correlation or indirect correlation between these factors and the occurrence of DPPB. Once other factors are added, the false correlation disappears, indicating that they may not actually be risk factors for the occurrence of DPPB. Furthermore, this inconsistency may indicate potential publication bias.
This study has some limitations. First, this meta-analysis was based on 13 case-control studies and 2 prospective cohort studies, therefore confounding is possible. Second, we found significant heterogeneity among the analyses of polyp number, polyp size, and polyp shape, which may be related to differences in study populations and study designs. Third, due to the limited number of included studies, the relationship between cold snare, preventive endoscopic clip closure and DPPB occurrence could not be analyzed.
Conclusion
In conclusion, colon colorectal polypectomy is an effective method for the prevention and treatment of colorectal cancer. However, regardless of how experienced the endoscopists are, there is always a risk of delayed postoperative bleeding. There are many risk factors for DPPB, so endoscopists should fully consider and implement effective intervention measures. Considerable attention should be devoted to patients with the following risk factors: male sex, hypertension, Anticoagulation, polyp size, polyp size ≥ 10 mm, polyp located in the right semicolon and EMR. During the operation, precise hemostasis should be performed, and drug therapy and active and rigorous follow-up should be used after surgery to form a multilink precise preventive intervention system. As it is difficult to eliminate the influence of the confounding factors examined herein, the conclusions of this study need to be further confirmed by more clinical controlled studies.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
We would like to thank the researchers and study participants for their contributions.
Abbreviations
- DPPB
Delayed postpolypectomy bleeding
- RR
Relative risk
- CI
Confidence interval
- HSP
Hot snare polypectomy
- CSP
Cold snare polypectomy
- EMR
Endoscopic mucosal resection
- ESD
Endoscopic submucosal dissection
- HB
Hot biopsy
- APC
Argon plasma coagulation
Author contributions
Xuzhen Zhang: Conceptualization, Methodology, Software, Data Curation, Writing - Original Draft, Writing - Review & Editing, Visualization, Project administration; Xiaoxing Jiang: Conceptualization, Writing - Original Draft, Writing - Review & Editing, Visualization, Project administration; Liang Shi: Conceptualization, Methodology, Investigation, Resources, Data Curation, Writing - Original Draft, Writing - Review & Editing, Project administration.All authors reviewed the manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Xirasagar S, Wu Y, Tsai MH, Zhang J, Chiodini S, de Groen PC. Colorectal cancer prevention by a CLEAR principles-based colonoscopy protocol: an observational study. Gastrointest Endosc. 2020;91(4):905–e164. doi: 10.1016/j.gie.2019.11.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dumoulin FL, Hildenbrand R. Endoscopic resection techniques for colorectal neoplasia: current developments. World J Gastroenterol. 2019;25(3):300–7. doi: 10.3748/wjg.v25.i3.300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cheng P, Bo Y, Fang J, Zhao S, Wang S, Li N, et al. Risk factors of delayed colonoscopic post-polypectomy bleeding. Chin J Dig Endoscopy. 2018;35(5):332–5. [Google Scholar]
- 4.Kishida Y, Hotta K, Imai K, Ito S, Yoshida M, Kawata N, et al. Risk analysis of colorectal post-polypectomy bleeding due to Antithrombotic Agent. Digestion. 2019;99(2):148–56. doi: 10.1159/000490791. [DOI] [PubMed] [Google Scholar]
- 5.Kubo K, Kato M, Mabe K, Harada N, Iboshi Y, Kagaya T, et al. Risk factors for delayed bleeding after therapeutic gastrointestinal endoscopy in patients receiving oral anticoagulants: a Multicenter Retrospective Study. Digestion. 2021;102(2):161–9. doi: 10.1159/000502952. [DOI] [PubMed] [Google Scholar]
- 6.Li P, Wang Q, Ni Y, Sha L, Yu L. Risk factors for postpolypectomy bleeding after endoscopic mucosal resection. J Nanjing Med University(Natural Sciences) 2019;39(3):360–474. [Google Scholar]
- 7.Inagaki Y, Yoshida N, Fukumoto K, Kassai K, Inoue K, Hirose R, et al. Risk factors of delayed bleeding after Cold Snare Polypectomy for colorectal polyps: a Multicenter Study. Dig Dis Sci. 2022;67(7):3177–84. doi: 10.1007/s10620-021-07119-7. [DOI] [PubMed] [Google Scholar]
- 8.Liu C, Wu R, Sun X, Tao C, Liu Z. Risk factors for delayed hemorrhage after colonoscopic postpolypectomy: polyp size and operative modality. JGH Open. 2019;3(1):61–4. doi: 10.1002/jgh3.12106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tsuruta S, Tominaga N, Ogata S, Tsuruoka N, Sakata Y, Shimoda R, et al. Risk factors for delayed hemorrhage after Colonic endoscopic mucosal resection in patients not on antithrombotic therapy: retrospective analysis of 3,844 polyps of 1,660 patients. Digestion. 2019;100(2):86–92. doi: 10.1159/000494455. [DOI] [PubMed] [Google Scholar]
- 10.Jaruvongvanich V, Prasitlumkum N, Assavapongpaiboon B, Suchartlikitwong S, Sanguankeo A, Upala S. Risk factors for delayed colonic post-polypectomy bleeding: a systematic review and meta-analysis. Int J Colorectal Dis. 2017;32(10):1399–406. doi: 10.1007/s00384-017-2870-0. [DOI] [PubMed] [Google Scholar]
- 11.Eleftheriadis D, Imalis C, Gerken G, Wedemeyer H, Duerig J. Risk factors for post-polypectomy bleeding; a retrospective case-control study of a high-volume colonoscopy center. Z Gastroenterol. 2022;60(10):1475–82. doi: 10.1055/a-1690-7795. [DOI] [PubMed] [Google Scholar]
- 12.Que Y, Ji F, Zhu H, Zhou X. Risk factors of delayed colorectal bleeding following endoscopic polypectomy. Zhejiang Med J. 2020;42(7):713–6. [Google Scholar]
- 13.Lin X, Chen L, Zhang H, Guo Y, Wu X, Zheng F, et al. Risk factors of post-polypectomy delayed Hemorrhage. J Sun Yat-Sen University(Medical Sciences) 2019;40(5):788–95. [Google Scholar]
- 14.Luo Z, Pu J, Wang X, Wang S, Yu L, Yan Z, et al. Clinical characteristics and risk factor analysis of delayed bleeding after endoscopic mucosal resection for colon polyps. Med J Chin People’s Liberation Army. 2019;44(9):769–73. [Google Scholar]
- 15.Moon HS, Park SW, Kim DH, Kang SH, Sung JK, Jeong HY. Only the size of resected polyps is an independent risk factor for delayed postpolypectomy hemorrhage: a 10-year single-center case-control study. Annals Coloproctology. 2014;30(4):182–5. doi: 10.3393/ac.2014.30.4.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Watabe H, Yamaji Y, Okamoto M, Kondo S, Ohta M, Ikenoue T, et al. Risk assessment for delayed hemorrhagic complication of colonic polypectomy: polyp-related factors and patient-related factors. Gastrointest Endosc. 2006;64(1):73–8. doi: 10.1016/j.gie.2006.02.054. [DOI] [PubMed] [Google Scholar]
- 17.Wu XR, Church JM, Jarrar A, Liang J, Kalady MF. Risk factors for delayed postpolypectomy bleeding: how to minimize your patients’ risk. Int J Colorectal Dis. 2013;28(8):1127–34. doi: 10.1007/s00384-013-1661-5. [DOI] [PubMed] [Google Scholar]
- 18.Park SK, Seo JY, Lee MG, Yang HJ, Jung YS, Choi KY, et al. Prospective analysis of delayed colorectal post-polypectomy bleeding. Surg Endosc. 2018;32(7):3282–9. doi: 10.1007/s00464-018-6048-9. [DOI] [PubMed] [Google Scholar]
- 19.Choung BS, Kim SH, Ahn DS, Kwon DH, Koh KH, Sohn JY, et al. Incidence and risk factors of delayed postpolypectomy bleeding: a retrospective cohort study. J Clin Gastroenterol. 2014;48(9):784–9. doi: 10.1097/MCG.0000000000000027. [DOI] [PubMed] [Google Scholar]
- 20.Zhang Q, An SL, Chen ZY, Fu FH, Jiang B, Zhi FC et al. Assessment of risk factors for delayed colonic post-polypectomy hemorrhage: a study of 15553 polypectomies from 2005 to 2013. PLoS ONE. 2014;9(10). [DOI] [PMC free article] [PubMed]
- 21.Sawhney MS, Salfiti N, Nelson DB, Lederle FA, Bond JH. Risk factors for severe delayed postpolypectomy bleeding. Endoscopy. 2008;40(2):115–9. doi: 10.1055/s-2007-966959. [DOI] [PubMed] [Google Scholar]
- 22.Kim JH, Lee HJ, Ahn JW, Cheung DY, Kim JI, Park SH, et al. Risk factors for delayed post-polypectomy hemorrhage: a case-control study. J Gastroenterol Hepatol. 2013;28(4):645–9. doi: 10.1111/jgh.12132. [DOI] [PubMed] [Google Scholar]
- 23.Buddingh KT, Herngreen T, Haringsma J, van der Zwet WC, Vleggaar FP, Breumelhof R, et al. Location in the right hemi-colon is an independent risk factor for delayed post-polypectomy hemorrhage: a multi-center case-control study. Am J Gastroenterol. 2011;106(6):1119–24. doi: 10.1038/ajg.2010.507. [DOI] [PubMed] [Google Scholar]
- 24.Chi J, He T, Wu Y, Wu K. Construction of nomograph prediction model for risk factors of delayed bleeding after endoscopic resection of colorectal polyps. Mod Digestion Intervention. 2020;25(11):1504–8. [Google Scholar]
- 25.Coordes A, Soudry J, Hofmann VM, Lenarz M. Gender-specific risk factors in post-tonsillectomy hemorrhage. European archives of oto-rhino-laryngology: official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) : affiliated with the German society for Oto-Rhino-Laryngology. Head Neck Surg. 2016;273(12):4535–41. doi: 10.1007/s00405-016-4146-7. [DOI] [PubMed] [Google Scholar]
- 26.Sang J, Zhang H, Zhou J, Xu L, Song Q, Sun X, et al. Risk factors of delayed bleeding after endoscopic polypectomy. Chin J Digestion. 2017;37(12):835–8. [Google Scholar]
- 27.Wang J, Hu C. Risk factors of delayed bleeding after colorectal polypectomy. China J Endoscopy. 2020;26(11):15–20. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.