Abstract
Purpose
To report demographic and clinical characteristics of patients who were more likely to receive proton beam therapy (PBT) than photon therapy from facilities with access to proton centers.
Materials and Methods
We utilized the national cancer database to identify the facilities with access to PBT between 2004 and 2015 and compared the relative usage of photons and PBT for demographic and clinical scenarios in breast, prostate, and nonsmall cell cancer.
Results
In total, 231 facilities with access to proton centers accounted for 168 323 breast, 39 975 lung, and 77 297 prostate cancer patients treated definitively. Proton beam therapy was used in 0.5%, 1.5%, and 8.9% of breast, lung, and prostate cases. Proton beam therapy was correlated with a farther distance traveled and longer start time from diagnosis for each site (P < .05).
For breast, demographic correlates of PBT were treatment in the west coast (odds ratio [OR] = 4.81), age <60 (OR = 1.25), white race (OR = 1.94), and metropolitan area (OR = 1.58). Left-sided cancers (OR = 1.28), N2 (OR = 1.71), non-ER+/PR+/Her2Neu− cancers (OR = 1.24), accelerated partial breast irradiation (OR = 1.98), and hypofractionation (OR = 2.35) were predictors of PBT.
For nonsmall cell cancer, demographic correlates of PBT were treatment in the south (OR = 2.6), metropolitan area (OR = 1.72), and Medicare insurance (OR = 1.64). Higher comorbid score (OR = 1.36), later year treated (OR = 3.16), and hypofractionation (not SBRT) (OR = 3.7) were predictors of PBT.
For prostate, correlates of PBT were treatment in the west coast (OR = 2.48), age <70 (OR = 1.19), white race (OR = 1.41), metropolitan area (OR = 1.25), higher income/education (OR = 1.25), and treatment at an academic center (OR = 33.94). Lower comorbidity score (OR = 1.42), later year treated (OR = 1.37), low-risk disease (OR = 1.45), definitive compared to postoperative (OR = 6.10), and conventional fractionation (OR = 1.64) were predictors of PBT.
Conclusion
Even for facilities with established referrals to proton centers, PBT utilization was low; socioeconomic status was potentially a factor. Proton beam therapy was more often used with left-sided breast and low-risk prostate cancers, without a clear clinical pattern in lung cancer.
Keywords: Proton centers, Referral patterns, Disparities
Introduction
Although its clinical merits remain widely debated, the dosimetric advantages and reduced integral dose of proton beam therapy (PBT) relative to external beam photon radiotherapy (XRT) have been consistently demonstrated for various malignancies.1 Perhaps for this reason, protons are an attractive treatment modality for many cancer patients, although utilization is quite limited due to the scarcity of centers. Indeed, for every PBT clinic in America, there are approximately 80 XRT clinics.2 Therefore, geography is a natural barrier to PBT access, and consequently so are various socioeconomic factors. Several studies indicate that PBT is disproportionately offered to affluent, white patients with the means to travel to the large metropolitan regions that typically house proton centers.3., 4., 5.
However, given the strongly confounding geographical selection bias, it can be difficult to draw conclusions regarding PBT utilization, clinical indications/referral patterns, and socioeconomic disparities. Herein, we attempted to control for geographic bias by exploring the utilization of PBT relative to XRT only from clinics with established links to proton centers. Specifically, we analyzed the clinical and demographic characteristics associated with PBT utilization from these centers for the 3 most common noncutaneous malignancies: breast, nonsmall cell lung cancer (NSCLC), and prostate cancer. Presumably, these clinics have similar access to both photon and proton therapy.
In the absence of level 1 clinical evidence to support proton use in definitive breast/lung/prostate cancer, the jury is still out as to who the appropriate candidates for PBT are. Nevertheless, PBT is the newer and more expensive technology and therefore may be more desirable for some patients. With that said, disparities in PBT utilization appear to be a reflection of the disparities in the health care system as a whole.6 Several studies have made these revelations previously, but to our knowledge, none have isolated their analyses to centers with access to photons and protons alike.
Methods
Patient selection
The national cancer database (NCDB) was queried between the years 2004-2015 to identify facilities with access to PBT, defined as institutions from which a breast, NSCLC, or prostate cancer diagnosis was made and subsequently had at least 1 patient treated with PBT. Among 1343 facilities that included radiotherapy, 231 had a referral pattern to both PBT and XRT. Within these 231 facilities, we only included nonmetastatic cases for which radiotherapy was an aspect of definitive management, yielding 287 353 breast, 52 213 NSLSC, and 123 650 prostate cases. For all disease sites, cases were then excluded if the radiation dose and timing were unknown, palliative doses, brachytherapy was involved, and nonadenocarcinoma histology (except squamous cell carcinoma for lung). Specific to breast, patients were excluded if they did not receive surgery, Tumor, Nodal, Metastasis stage unknown, receptor status unknown, or laterality unknown. Nonsmall cell lung cancer patients were excluded for unknown Tumor, Nodal, Metastasis stage and unknown laterality. Prostate patients were excluded if prostate specific antigen, Gleason score, or T stage were not available. Ultimately, complete data were available for analysis in 168 323 breast, 39 975 NSCLC, and 77 297 prostate patients, respectively.
Statistics
In order to control for the disease-specific clinical characteristics on multivariable regression analysis, we did not combine the data from each disease site and instead performed a separate analysis for breast, NSCLC, and prostate. We then compared the relative usage of PBT and XRT for various demographic and clinical scenarios via multivariable binomial regression analysis. Variables included for all sites include location, facility type, race, age, insurance, regional population, income, regional education level, distance traveled, time from diagnosis to treatment, year treated, fractionation scheme, and use of systemic therapy. Variables specific to the breast group include T/N/group stage, laterality, hormone receptor status, lumpectomy or mastectomy, and use of a boost. Variables specific to lung include T/N/group stage, laterality, histology, and definitive versus adjuvant treatment. Exclusive variables in the prostate analysis include risk group, and definitive or postoperative setting. Odds ratios (OR) were used to denote the relative utilization of PBT compared to XRT, with 95% confidence intervals (CI) to indicate statistical significance. Statistics were performed via MedCalc v 19.1.
We also investigated the proportion of payers for PBT within each geographical region. In accordance with NCDB coding, the following states were included in their corresponding region: Connecticut, Massachusetts, Maine, New Hampshire, Rhode Island, Vermont in “New England”; New Jersey, New York, Pennsylvania in “Middle Atlantic”; District of Columbia, Delaware, Florida, Georgia, Maryland, North Carolina, South Carolina, Virginia, and West Virginia in “South Atlantic”; Illinois, Indiana, Michigan, Ohio, and Wisconsin in “East North Central”; Alabama, Kentucky, Missouri, and Tennessee in “East South Central”; Iowa, Kansas, Minnesota, Montana, North Dakota, Nebraska, and South Dakota in “West North Central”; Arizona, Louisiana, Oklahoma, and Texas in “West South Central”; Arizona, Colorado, Idaho, Montana, New Mexico, Nevada, Utah, and Wyoming in “Mountain”; and Alaska, California, Hawaii, Oregon, and Washington in “Pacific.” For the purposes of multivariable analysis, however, “Middle Atlantic” and “South Atlantic” were combined to form the “Southeast,” “East North Central, East South Central, and West North Central” combined to form the “Midwest,” and “West South Central” was relabeled as “South.”
Results
Utilization
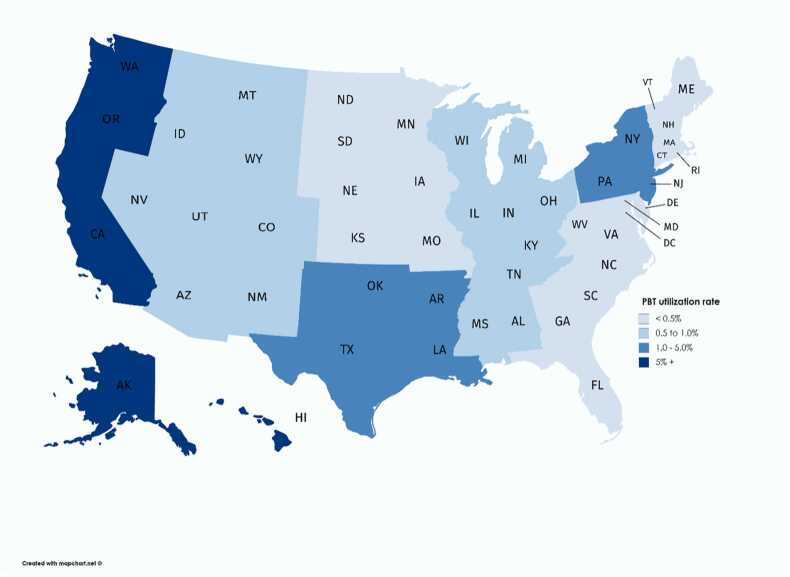
Among the 231 facilities with access to PBT, 168 323 breast, 39 975 lung, and 77 297 prostate cases were treated with radiotherapy. PBT was used in 0.5%, 1.5%, and 8.9% of breast, lung, and prostate cases, respectively. PBT use varied by primary tumor site but was consistently employed most often in the west coast, at a utilization rate of 1.5% for breast, 5.0% for NSCLC and 30.2% for prostate. A composite regional PBT utilization rate, adjusted for the total number of cases for each site, is depicted in Figure 1.
Figure 1.
Combined (weighted by disease site) proton beam therapy utilization rate by region. Abbreviation: PBT, proton beam therapy.
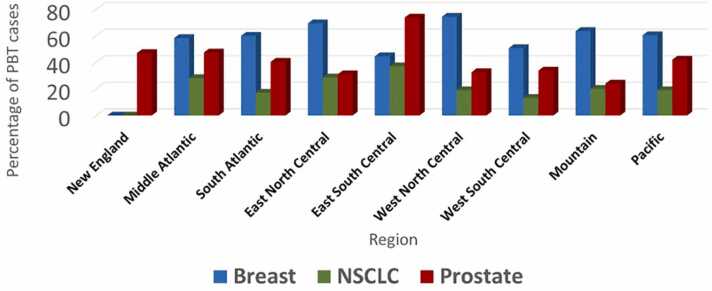
Depending on the region, private insurance covered 45% to 74% of breast, 13% to 37% of NSCLC, and 24% to 47% of prostate cases. Medicare covered virtually all remaining PBT cases. The only region where the majority of proton breast cases were covered by Medicare instead of private insurance was the “East South Central,” and the inverse was true for prostate cases (Figure 2).
Figure 2.
Proportion of proton beam therapy covered by private insurance by region and disease site. Abbreviations: NSCLC, nonsmall cell cancer; PBT, proton beam therapy.
On average, PBT was initiated 6, 13, and 9 days later from diagnosis compared to XRT for breast, lung, and prostate cases, respectively (each statistically significant on multivariable binomial regression). The median distance traveled for treatment was 9.1 miles for breast, 15.1 miles for lung, and 293 miles for prostate. For the prostate group, the distance was highly skewed by the number of patients traveling to the west coast for treatment (median 378.7 miles). If these patients were to be excluded, the median distance traveled for prostate patients was 12.8 miles. For each disease site, further distance traveled correlated with PBT utilization, OR 1.26 (95% CI 1.09-1.46), OR 1.92 (95% CI 1.57-2.34), OR 5.47 (95% CI 4.93-6.07) for breast, NSCLC, and prostate, respectively. With the exception of the west coast in prostate cancer, there was no statistically significant difference in median distance traveled for PBT between regions for any disease site.
Clinical predictors
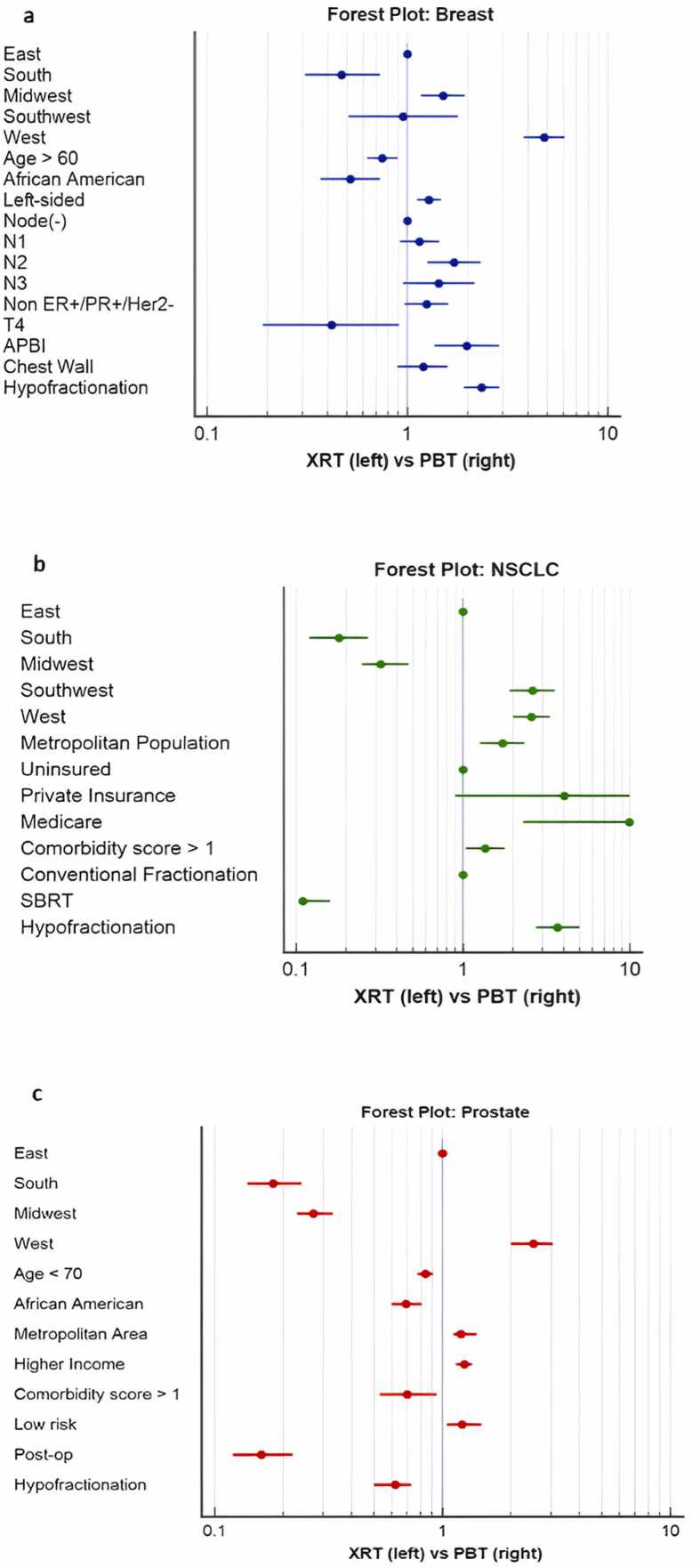
Predictors of PBT utilization for each disease site are summarized in the Table. For breast, independent predictors of PBT receipt were treatment in the west coast (OR = 4.8, 95% CI 3.81-6.07), age under 60 years (OR = 1.25, 95% CI 1.12-1.59), white race (OR = 1.94, 95% CI 1.37-2.70), metropolitan area (OR = 1.58 95% CI 1.16-2.13), left-sided tumors (OR = 1.28, 95% CI 1.12-1.47), N2 disease (OR = 1.71, 95% CI 1.26-2.32), non-ER+/PR+/Her2Neu− status (OR = 1.24, 95% CI 1.01-1.59), partial breast irradiation (OR = 1.98, 95% CI 1.37-2.87), and hypofractionation (OR = 2.35, 95% CI 1.92-2.87). There was no difference according to education level, insurance, income, lumpectomy/mastectomy, or boost/no-boost cases (Figure 3a).
Table.
Multivariable binomial regression analysis of relative proton beam use.
| Parameter | Breast cancer |
Nonsmall cell lung cancer |
Prostate cancer |
||||||
|---|---|---|---|---|---|---|---|---|---|
| XRT (%) | PBT (%) | OR (95% CI) | XRT (%) | PBT (%) | OR (95% CI) | XRT (%) | PBT (%) | OR (95% CI) | |
| Location | |||||||||
| Northeast | 22.8 | 11.8 | Reference | 19.4 | 33.0 | Reference | 19.8 | 7.6 | Reference |
| Southeast | 15.9 | 3.4 | 0.47 (0.31-0.73) | 27.8 | 6.9 | 0.18 (0.12-0.27) | 23.2 | 0.9 | 0.18 (0.14-0.24) |
| Midwest | 29.6 | 22.4 | 1.51 (1.17-1.94) | 38.8 | 17.5 | 0.32 (0.25-0.43) | 30.1 | 2.2 | 0.27 (0.22-0.33) |
| South | 7.6 | 9.5 | 2.81 (2.04-3.88) | 5.3 | 17.8 | 2.61 (1.90-3.57) | 5.9 | 2.3 | 1.00 (0.83-1.21) |
| Mountain | 2.5 | 1.3 | 0.95 (0.51-1.80) | 1.9 | 0.8 | 0.22 (0.08-0.60) | 2.5 | 0.3 | 0.31 (0.19-0.52) |
| West | 21.7 | 51.5 | 4.81 (3.81-6.07) | 6.8 | 23.9 | 2.57 (2.00-3.31) | 17.6 | 86.6 | 19.8 (17.7-22.1) |
| Facility | |||||||||
| Community | 2.7 | 3.0 | Reference | 3.9 | 4.4 | Reference | 2.7 | 0.3 | Reference |
| Comprehensive | 45.8 | 47.1 | 0.89 (0.59-1.34) | 40.1 | 37.0 | 0.90 (0.56-1.42) | 40.9 | 3.1 | 0.69 (0.42-1.12) |
| Academic | 45.1 | 48.2 | 1.03 (0.69-1.56) | 45.9 | 55.6 | 1.00 (0.62-1.60) | 48.1 | 95.5 | 33.9 (20.9-55.1) |
| Integrated | 6.5 | 1.7 | 0.55 (0.28-1.07) | 10.1 | 3.0 | 0.40 (0.20-0.78) | 8.3 | 1.2 | 1.66 (0.98-2.79) |
| Race | |||||||||
| White | 82.9 | 86.3 | Reference | 85.8 | 87.8 | Reference | 79.2 | 90.5 | Reference |
| Black | 10.5 | 4.8 | 0.52 (0.37-0.73) | 11.4 | 7.2 | 0.78 (0.55-1.11) | 15.7 | 5.9 | 0.69 (0.60-0.81) |
| Age | |||||||||
| Below mediana | 48.3 | 49.4 | Reference | 49.4 | 45.2 | Reference | 53.2 | 67.5 | Reference |
| Over median | 51.7 | 50.6 | 0.75 (0.63-0.89) | 50.6 | 54.8 | 1.01 (0.83-1.23) | 46.8 | 32.5 | 0.63 (0.58-0.68) |
| Insurance | |||||||||
| Private | 60.1 | 59.7 | Reference | 26.4 | 22.9 | Reference | 34.9 | 42.4 | Reference |
| Medicaid | 6.5 | 5.7 | 0.66 (0.34-1.28) | 8.0 | 4.0 | 0.68 (0.42-1.10) | 5.6 | 1.6 | 0.24 (0.20-0.30) |
| Medicare | 32.1 | 33.3 | 1.07 (0.57-1.99) | 61.8 | 68.2 | 1.64 (1.30-2.06) | 56.4 | 53.0 | 0.77 (0.73-0.81) |
| Population | |||||||||
| 1 million+ | 70.0 | 75.1 | Reference | 57.3 | 72.2 | Reference | 53.2 | 56.4 | Reference |
| 250k-1 million | 17.5 | 12.6 | 0.71 (0.57-0.88) | 15.3 | 10.7 | 0.58 (0.43-0.79) | 19.6 | 15.0 | 0.80 (0.71-0.89) |
| 20k-250k | 7.9 | 6.5 | 0.63 (0.47-0.86) | 13.5 | 8.4 | 0.54 (0.38-0.78) | 16.6 | 17.4 | 1.04 (0.92-1.18) |
| <20k | 4.7 | 5.9 | 1.13 (0.80-1.59) | 10.8 | 5.5 | 0.37 (0.24-0.57) | 8.1 | 7.3 | 1.10 (0.92-1.31) |
| Income | |||||||||
| Lowest quartile | 11.6 | 10.1 | Reference | 19.4 | 12.2 | Reference | 15.0 | 8.5 | Reference |
| Second lowest quartile | 16.1 | 15.5 | 0.95 (0.71-1.27) | 22.7 | 18.7 | 0.96 (0.69-1.34) | 20.1 | 18.9 | 0.89 (0.76-1.03) |
| Second highest quartile | 25.4 | 20.6 | 0.78 (0.58-1.06) | 26.3 | 26.4 | 1.04 (0.74-1.47) | 28.2 | 27.9 | 0.98 (0.84-1.13) |
| Highest quartile | 46.9 | 53.8 | 1.09 (0.79-1.50) | 30.4 | 41.3 | 0.99 (0.68-1.48) | 36.0 | 44.0 | 1.38 (1.18-1.62) |
| Year | |||||||||
| 2004-2007 | 21.5 | 30.1 | Reference | 24.4 | 15.1 | Reference | 34.1 | 33.2 | Reference |
| 2008-2011 | 31.8 | 14.4 | 0.29 (0.23-0.36) | 32.9 | 15.1 | 0.83 (0.61-1.12) | 35.0 | 36.1 | 1.13 (1.04-1.23) |
| 2012-2015 | 46.7 | 55.5 | 0.66 (0.55-0.78) | 42.7 | 60.8 | 3.16 (2.48-4.04) | 30.9 | 30.7 | 1.37 (1.25-1.50) |
| Stage/risk | |||||||||
| I (low) | 72.2 | 71.3 | Reference | 33.4 | 25.9 | Reference | 19.2 | 37.2 | Reference |
| II (intermediate) | 22.4 | 22.4 | 0.78 (0.61-1.01) | 12.6 | 15.2 | 0.93 (0.69-1.26) | 40.9 | 41.0 | 0.92 (0.78-1.09) |
| III (high) | 5.4 | 6.3 | 0.77 (0.58-1.02) | 54.0 | 58.9 | 0.86 (0.66-1.12) | 39.9 | 21.7 | 0.23 (0.21-0.26) |
| Laterality | |||||||||
| Right | 49.7 | 43.7 | Reference | 55.8 | 52.7 | Reference | - | - | - |
| Left | 50.3 | 56.3 | 1.28 (1.12-1.47) | 39.3 | 41.6 | 1.07 (0.89-1.28) | - | - | - |
| Fractionation | |||||||||
| Conventional | 55.5 | 35.6 | Reference | 73.6 | 74.4 | Reference | 92.5 | 93.3 | Reference |
| Hypofractionation | 14.8 | 26.7 | 2.35 (1.92-2.87) | 3.4 | 17.2 | 3.7 (2.74-4.99) | 4.2 | 5.8 | 0.62 (0.53-0.73) |
| SBRT/APBIb | 1.5 | 5.1 | 1.98 (1.37-2.87) | 23.0 | 3.5 | 0.10 (0.6-0.17) | 0.7 | 0.2 | 0.15 (0.11-0.21) |
Abbreviations: XRT, photon radiotherapy; PBT, proton beam therapy; OR, odds ratio; CI, confidence interval; SBRT, stereotactic body radiotherapy; APBI, accelerated partial breast irradiation.
Bold indicates statistical significance. Not every variable included in multivariable analysis is represented on this chart. Please refer to the Results section for data on additional variables.
Proportions reflected as the percentage of the particular variable within either the photon (XRT) or proton (PBT) group.
Median age for prostate and nonsmall cell lung cancer = 70 years old, breast cancer = 60 years old.
Stereotactic body radiotherapy used in nonsmall cell lung cancer and prostate cancer, accelerated partial breast irradiation used in breast cancer.
Figure 3.
(a) Forest plot depicting the odds ratios (ORs) for receiving XRT or PBT following multivariable analysis in breast cancer. (b) Forest plot depicting the ORs for receiving XRT or PBT following multivariable analysis in NSCLC. (c) Forest plot depicting the ORs for receiving XRT or PBT following multivariable analysis in prostate cancer. Abbreviations: APBI, accelerated partial breast irradiation; NSCLC, nonsmall cell cancer; PBT, proton beam therapy; SBRT, stereotactic body radiotherapy; and XRT, photon radiotherapy.
For NSCLC, treatment in the south (OR = 2.61, 95% CI 1.90-3.57), metropolitan area (OR = 1.72, 95% CI 1.27-2.33), Medicare (OR = 1.64, 95% CI 1.30-2.06), higher comorbid score (OR = 1.36, 95% CI 1.05-1.75), later year treated (OR = 3.16, 95% CI 2.48-4.04), and use of hypofractionation (but not SBRT) (OR = 3.7, 95% CI 2.74-4.99) were independent predictors of PBT usage. There was no difference according to education level, gender, race, income, laterality, or stage (Figure 3b).
For prostate, factors significantly associated with PBT receipt were treatment in the west coast (OR = 19.8, 95% CI 17.7-22.1), age under 70 years (OR = 1.58, 95% CI 1.47-1.72), white race (OR = 1.41, 95% CI 1.23-1.72), private insurance (OR = 1.30, 95% CI 1.23-1.37), metropolitan area (OR = 1.25, 95% CI 1.12-1.41), higher income/education (OR = 1.38, 95% CI 1.18-1.62), lower comorbidity score (OR = 1.42 95% CI 1.08-1.90), treatment after 2011 (OR = 1.37, 95% CI 1.25-1.50), nonhigh risk disease (OR = 4.35, 95% CI 3.84-4.76), intact prostate (OR = 6.10, 95% CI 4.76-8.13), and conventional fractionation (OR = 1.64, 95% CI 1.37-1.89) (Figure 3c).
Discussion
Regardless of location, the overall utilization for proton therapy remains quite low, even among clinics with a presumed link to proton centers. This is less true in prostate cancer, for which PBT has been a familiar and promoted modality for decades. Additionally, unlike breast cancer and NSCLC where a multimodality approach is almost always required, the majority of prostate cases can be managed solely by a radiation oncologist. Therefore, prostate patients are more likely to seek out PBT themselves rather than rely on a referring physician. That is not to suggest that PBT is necessarily underutilized, as the role of proton therapy in the oncology landscape is still subject to accumulating data and is thus far unclear. With 9 randomized controlled trials comparing photons with protons currently accruing data, the level 1 evidence is on its way.7., 8., 9., 10., 11., 12., 13., 14., 15.
The paucity of data, however, has not curtailed the demand for proton therapy. On the contrary, the number of proton centers in the United States grew from 14 to 34 in just 5 years.4., 16. This may come with some pushback from payers, as the historical cost per fraction of proton therapy is roughly twice that of intensity modulated radiotherapy.17 Perhaps, this is why the majority of prostate and NSCLC PBT cases, as reflected in this analysis, are paid for by Medicare rather than private insurance. Interestingly, the majority of breast cancer cases treated with PBT, regardless of geographic region, were covered by private insurance, although the reasons are unclear. The increasingly important economics regarding proton therapy might change in the near future given the affordability of newer cyclotrons and implementation of the oncology care model, with potentially significant implications for PBT utilization.
Of course, if level I evidence does emerge to suggest a clinical benefit for PBT, the health care system will have to facilitate increased utilization, regardless of the costs. To that end, there are some notable clinical correlations with PBT utilization observed in this study. In breast cancer, PBT patients were more likely to have greater disease burden and more aggressive receptor status. Presumably, these patients were more likely to receive comprehensive nodal irradiation, where PBT has demonstrated a 75% reduction in mean heart dose (left side) and 50% reduction in V20 of the ipsilateral lung.18 Similarly, left-sided lesions were also independent predictors of PBT utilization likely due to the dramatic reduction in mean heart dose, which can range from 1 to 7 Gy, per multiple dosimetric studies.18., 19., 20., 21.
Similar clinical patterns were not observed in NSCLC, where disease burden and laterality did not predict for PBT. Interestingly, the lone clinical predictor for PBT in lung cancer was a higher comorbidity score. This can partially be explained by the higher correlation with Medicare, and thereby older patients on average. Some physicians may also figure that any additional dose to the lungs and heart, which have shown to be lower in PBT,22 in patients who are sicker at baseline might make a difference in the ultimate outcome.
Proton beam therapy was more likely to be used in nonhigh risk disease for prostate cancer likely because the vast majority of these cases were completed at academic centers, and early PBT clinical trials largely excluded high-risk disease, but this is no longer the case for new trials.11., 23. Although most prostate patients receiving PBT were treated at an academic center, they were also less likely to receive hypofractionation, which is presumably adopted quicker among academic facilities. We speculate that these centers were waiting for the toxicity data from conventional fractionation in PBT to mature prior to converting to hypofractionated courses. However, a recent prospective trial of prostate patients undergoing PBT to 70 to 72 Gy (RBE) in 28 to 29 fractions demonstrated biochemical control rates well over 90% with <2% late grade 3 toxicity.24 This is currently being validated by NCT03561220 (COMPPARE),23 and since hypofractionation has been endorsed as the standard of care by several national guidelines with photon therapy, PBT is likely to follow suit.25
Social inequities have always existed in health care, and several studies link ethnic minorities and lower income patients with poorer cancer outcomes.6., 26., 27. Recent NCDB studies, using similar data to what we present here, also highlight racial and income inequities as it pertains to PBT utilization for breast, lung, and prostate cancer.3., 4., 5. Notably, after mitigating the geographic bias as we have done in this study, nearly all of the previously reported socioeconomic discrepancies disappear for breast cancer and NSCLC, with the exception of race in breast cancer. With the substantially greater patient population and farther distance traveled, prostate PBT patients did exhibit several socioeconomic disparities in our study, albeit to a considerably less extent than previously described. For instance, Mahal et al4 reported that the OR for PBT among black patients, those from zip code with lower income, and those from nonmetropolitan areas were 0.20, 0.67, and 0.30, respectively. By contrast, after mitigating the geographic selection bias in our study, those same parameters had an OR of 0.69, 0.72, and 0.80. If future studies do in fact reveal a benefit for PBT, our study suggests that geographic access to proton therapy can go a long way in remedying the disparities that exist currently.
Limitations
This study is novel in design and reveals data not previously reported in the literature; however, it is subject to the natural selection biases of a large retrospective dataset, regardless of the statistical tools used to control for confounding variables. Furthermore, although the investigators utilized the most up to date data from the NCDB at the time of analysis, there is a several year lag between when the data are collected and released. Thus, this study documents proton utilization up until 2016 and since that time, almost twice as many proton centers have opened in the United States. Reporting the utilization trends preceding the current era is still noteworthy; however, it will be even more important to document the trends thereafter. Additionally, facilities with access to PBT as we defined it are likely overrepresented because of patients with the means to travel far for treatment. This might appear to be especially true for prostate cancer, where the median distance traveled for PBT was significantly higher than breast or NSCLC; however, the 231 unique facilities with presumed access to both photons and protons were a consistent number among each disease site. Furthermore, the ratio of unique NCDB facilities with access to photons (1343) to the number of operational proton facilities at the time these data were collected (14) mirrors the approximately 80:1 ratio of photon to proton centers published in other studies independent of NCDB data, so that the methodology appears to be corroborated.
Conclusion
Despite limiting our PBT utilization analysis to facilities with established links to both XRT and PBT, the employment of PBT for breast and NSCLC was exceedingly low, at least in the first decade captured by the NCDB. Even though the number of proton centers in the country is growing, proton use will likely continue to trail behind conventional external beam radiation until more randomized data matures. As the debate regarding the role of proton therapy in our health care system progresses, it should be noted that the previously reported socioeconomic disparities in prostate PBT, while still present, are considerably attenuated in this study, and nearly absent in breast cancer and NSCLC. This may be related to limiting analysis only to facilities with PBT access, suggesting that geographic availability could go a long way in mitigating the well-documented disparities in cancer care.
Ethics
This project was reviewed by the authors' institutional research infrastructure and was determined to be exempt from Institutional Review Board approval.
Funding
The authors have no funding to disclose.
Author contribution
Shaakir Hasan: Conceptualization, Methodology, Formal analysis. Vivek Verma: Writing- original draft, Investigation, Resources. Ananya Raghavan: Writing- Review and editing, Visualization. Stephen Abel: Data curation, Software. R. E. Wegner: Investigation, Validation. Isabelle Choi: Supervision, Funding acquisition, Project administration. Robert Press: Resources, Investigation. Arpit Chhabra: Methodology, Resources. C. B. Simone II: Project administration, Funding acquisition.
Declaration of Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
This work was presented at the 6th Annual Conference of the Particle Therapy Cooperative Group North America: October 14-16, 2019, Miami, FL, USA.
References
- 1.Yuan T.Z., Zhan Z.J., Qian C.N. New frontiers in proton therapy: applications in cancers. Cancer Commun. 2019;39(1):61. doi: 10.1186/s40880-019-0407-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ballas L.K., Elkin E.B., Schrag D., Minsky B.D., Bach P.B. Radiation therapy facilities in the United States. Int J Radiat Oncol Biol Phys. 2006;66(4):1204–1211. doi: 10.1016/j.ijrobp.2006.06.035. [DOI] [PubMed] [Google Scholar]
- 3.Chowdhary M., Lee A., Gao S., et al. Is proton therapy a “pro” for breast cancer? A comparison of proton vs. non-proton radiotherapy using the national cancer database. Front Oncol. 2019;9 doi: 10.3389/fonc.2018.00678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mahal B.A., Chen Y.W., Efstathiou J.A., et al. National trends and determinants of proton therapy use for prostate cancer: a National Cancer Data Base study. Cancer. 2016;122(10):1505–1512. doi: 10.1002/cncr.29960. [DOI] [PubMed] [Google Scholar]
- 5.Higgins K.A., O'Connell K., Liu Y., et al. National cancer database analysis of proton versus photon radiation therapy in non-small cell lung cancer. Int J Radiat Oncol Biol Phys. 2017;97(1):128–137. doi: 10.1016/j.ijrobp.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 6.O’Keefe E.B., Meltzer J.P., Bethea T.N. Health disparities and cancer: racial disparities in cancer mortality in the United States, 2000–2010. Front Public Health. 2015;3:1–15. doi: 10.3389/fpubh.2015.00051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.ClinicalTrials.gov. Pragmatic randomized trial of proton vs. photon therapy for patients with non-metastatic breast cancer: a radiotherapy comparative effectiveness (RADCOMP) consortium trial - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT02603341. [DOI] [PMC free article] [PubMed]
- 8.ClinicalTrials.gov. Comparing photon therapy to proton therapy to treat patients with lung cancer - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT01993810.
- 9.ClinicalTrials.gov. Dose-escalated photon IMRT or proton beam radiation therapy versus standard-dose radiation therapy and temozolomide in treating patients with newly diagnosed glioblastoma - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT02179086.
- 10.ClinicalTrials.gov. Proton beam or intensity-modulated radiation therapy in preserving brain function in patients with IDH mutant grade II or III glioma - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT03180502.
- 11.ClinicalTrials.gov. Proton therapy vs. IMRT for low or intermediate risk prostate cancer - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT01617161.
- 12.ClinicalTrials.gov. Randomized trial of intensity-modulated proton beam therapy (IMPT) versus intensity-modulated photon therapy (IMRT) for the treatment of oropharyngeal cancer of the head and neck - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT01893307.
- 13.ClinicalTrials.gov. Study of proton versus photon beam radiotherapy in the treatment of head and neck cancer - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT02923570.
- 14.ClinicalTrials.gov. Radiation therapy with protons or photons in treating patients with liver cancer - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT03186898.
- 15.ClinicalTrials.gov. Comparing proton therapy to photon radiation therapy for esophageal cancer - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT03801876.
- 16.NAPT. Map of proton therapy centers. Accessed April 27, 2020. https://www.proton-therapy.org/map/.
- 17.Dvorak T., Fitzek M.M., Wazer D.E. Utilization of proton therapy: evidence-based, market-driven, or something in-between? Am J Clin Oncol. 2013;36(2):192–196. doi: 10.1097/COC.0b013e3182438d92. [DOI] [PubMed] [Google Scholar]
- 18.Patel S.A., Lu H.M., Nyamwanda J.A., et al. Postmastectomy radiation therapy technique and cardiopulmonary sparing: a dosimetric comparative analysis between photons and protons with free breathing versus deep inspiration breath hold. Pract Radiat Oncol. 2017;7(6):e377–e384. doi: 10.1016/j.prro.2017.06.006. [DOI] [PubMed] [Google Scholar]
- 19.Mast M.E., Vredeveld E.J., Credoe H.M., et al. Whole breast proton irradiation for maximal reduction of heart dose in breast cancer patients. Breast Cancer Res Treat. 2014;148(1):33–39. doi: 10.1007/s10549-014-3149-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin L.L., Vennarini S., Dimofte A., et al. Proton beam versus photon beam dose to the heart and left anterior descending artery for left-sided breast cancer. Acta Oncol. 2015;54(7):1032–1039. doi: 10.3109/0284186X.2015.1011756. [DOI] [PubMed] [Google Scholar]
- 21.Fagundes M., Hug E.B., Pankuch M., et al. Proton therapy for local-regionally advanced breast cancer maximizes cardiac sparing. Int J Part Ther. 2015;1(4):827–844. doi: 10.14338/ijpt-14-00025.1. [DOI] [Google Scholar]
- 22.Liao Z., Lee J.J., Komaki R., et al. Bayesian adaptive randomization trial of passive scattering proton therapy and intensity-modulated photon radiotherapy for locally advanced non-small-cell lung cancer. J Clin Oncol. 2018;36(18):1813–1822. doi: 10.1200/JCO.2017.74.0720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.ClinicalTrials.gov. A prospective comparative study of outcomes with proton and photon radiation in prostate cancer - Full text view. Accessed April 27, 2020. https://clinicaltrials.gov/ct2/show/NCT03561220.
- 24.Henderson R.H., Bryant C., Hoppe B.S., et al. Five-year outcomes from a prospective trial of image-guided accelerated hypofractionated proton therapy for prostate cancer. Acta Oncol. 2017;56(7):963–970. doi: 10.1080/0284186X.2017.1287946. [DOI] [PubMed] [Google Scholar]
- 25.Morgan S.C., Hoffman K., Loblaw D.A., et al. Hypofractionated radiation therapy for localized prostate cancer: an ASTRO, ASCO, and AUA evidence-based guideline. Pract Radiat Oncol. 2018;8(6):354–360. doi: 10.1016/j.prro.2018.08.002. [DOI] [PubMed] [Google Scholar]
- 26.Aboagye J.K., Kaiser H.E., Hayanga A.J. Rural-urban differences in access to specialist providers of colorectal cancer care in the United States: a physician workforce issue. JAMA Surg. 2014;149(6):537–543. doi: 10.1001/jamasurg.2013.5062. [DOI] [PubMed] [Google Scholar]
- 27.Presley C.J., Raldow A.C., Cramer L.D., et al. A new approach to understanding racial disparities in prostate cancer treatment. J Geriatr Oncol. 2013;4(1):1–8. doi: 10.1016/j.jgo.2012.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]