Abstract
A cDNA encoding the catalytic subunit of human telomerase was used to generate life-extended derivatives of primary human diploid fibroblasts. The life-extended cells supported efficient human cytomegalovirus (HCMV) replication. A subclone of the life-extended cells was generated containing the HCMV UL82 gene and used to isolate and propagate a virus that exhibited a profound growth defect after infection at a low input multiplicity.
Defining the molecular mechanisms that control human cytomegalovirus (HCMV) replication in vitro has been difficult in part due to the virus's limited host range. HCMV is typically propagated in primary cultures of human fibroblasts that yield relatively high titer stocks and produce plaques when infected with HCMV. However, primary human fibroblasts have a finite life span that limits their usefulness in studying HCMV replication. For example, it is difficult to generate stable fibroblast cell lines to complement defective HCMV strains. HCMV has been shown to replicate in a number of different cell types including human umbilical vein endothelial cells (4, 12, 13), macrophages and monocytes (5, 14), an astrocytoma cell line (3, 6), and a teratocarcinoma cell line (8). However, none of these cell lines produce high-titer stocks or plaques when infected with HCMV. Therefore, a life-extended fibroblast cell line capable of generating high-titer HCMV stocks and producing viral plaques would be a valuable tool to better understand the mechanisms of HCMV replication. It has recently been shown that constitutive expression of the catalytic subunit of human telomerase extends the life span of a number of somatic cells including human fibroblasts (2). We report here the generation of life-extended fibroblast clones that are fully permissive for HCMV replication and plaque formation. These life-extended fibroblasts have been used to generate a cell line that complements an HCMV mutant lacking the UL82 gene.
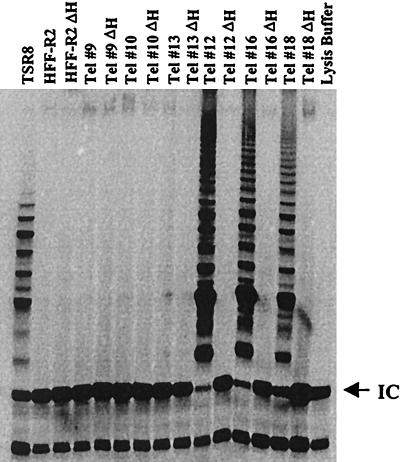
To generate life-extended human diploid foreskin fibroblasts (HFF cells), HFF-R2 cells at passage 6 were transfected with plasmid pGRN145 (2), which expresses the catalytic subunit of telomerase and a puromycin resistance marker. Stable clones were selected in puromycin (1 μg/ml) and analyzed for telomerase activity using the telomeric repeat amplification protocol (TRAP) assay (2, 7). We obtained 22 puromycin-resistant clones; 14 were negative and 8 were positive for telomerase activity (data not shown). We chose three positive and three negative clones for a more detailed analysis. Figure 1 shows that clones 12, 16, and 18 were positive and clones 9, 10, and 13 were negative for telomerase activity in the TRAP assay. The parental cells (HFF-R2) were negative for telomerase activity. As a negative control, equivalent cell lysates from all samples were incubated at 85°C for 10 min to inactivate telomerase activity (2, 7). After heat inactivation, none of the samples expressed telomerase activity.
FIG. 1.
Telomerase activity in stable HFF clones. Stable HFF clones obtained by transfection with pGRN145 expressing the catalytic subunit of telomerase (Tel) were analyzed for telomerase activity by the TRAP assay using a TRAPEZE kit (Intergen) according to the manufacturer's instructions. Two thousand cells were analyzed in all samples. TRAP products were separated by gel electrophoresis (11.25% polyacrylamide) and detected by autoradiography. As a negative control, cell lysates were incubated at 85°C for 10 min (ΔH) to inactivate telomerase. TSR8, positive control for the PCR; IC, internal control for all reactions.
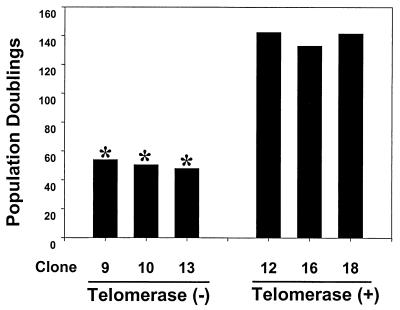
We compared the proliferative life spans of the telomerase-positive and -negative clones. Once clones were expanded to a 10-cm-diameter dish, the population doubling was set at 40. Cells were counted and passed every 5 to 7 days, and new population doublings were calculated. As shown in Fig. 2, the three telomerase-negative clones underwent senescence, i.e., failed to double within 2 weeks, between population doublings 45 and 55. All 14 telomerase-negative clones senesced after 49.4 (±3.0) population doublings (data not shown). In contrast, the three telomerase-positive clones have exceeded 135 population doublings and continue to divide like early-passage HFF cells.
FIG. 2.
Replicative life span of telomerase-positive and -negative clones. Cells were counted and passaged every 5 to 7 days, and new population doublings were calculated. Cultures were considered senescent (*) if they had undergone less than one population doubling in 2 weeks.
To determine if expression of telomerase affected the ability of HCMV to replicate and produce high-titer stocks, three telomerase-negative and three telomerase-positive clones were infected with HCMV (strain AD169) at a multiplicity of infection (MOI) of 5 PFU/cell, and virus titers were compared to those in HCMV-infected parental HFF cells. Telomerase negative clones 9, 10, and 13 were tested at population doublings 47 to 48.5, prior to undergoing senescence. HCMV replication in telomerase-positive clones 12, 16, and 18 were tested at both lower (46.5 to 51) and higher (65 to 75) population doublings, to determine if there was a difference in viral titers at different passage levels. As shown in Table 1, telomerase-positive and -negative cells were equally permissive for HCMV replication, and the three telomerase-positive clones yielded similar titers at lower or higher population doublings. There was no difference in the yield of HCMV in the parental HFF cells, telomerase-negative clones, or telomerase-positive clones.
TABLE 1.
Comparison of wild-type HCMV growth on telomerase-positive and telomerase-negative cell linesa
| Cell line | Telomerase status | No. of population doublings | PFU/ml (107) |
|---|---|---|---|
| HFF-R2 | − | 34.5 | 1.2 |
| Telomerase 9 | − | 48.5 | 3.0 |
| Telomerase 10 | − | 47 | 1.1 |
| Telomerase 13 | − | 48 | 1.1 |
| Telomerase 12 | + | 51 | 4.6 |
| 75.5 | 3.5 | ||
| Telomerase 16 | + | 46.5 | 2.9 |
| 65 | 4.9 | ||
| Telomerase 18 | + | 47 | 3.2 |
| 75 | 5.7 |
Cells were infected at an MOI of 5 PFU/cell. Virus was harvested 5 days later, and yields were determined by plaque assay on HFF-R2 cells.
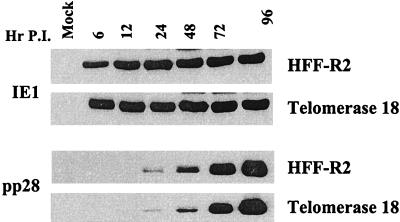
We next compared HCMV gene expression in telomerase-positive clones to that in the parental HFF cells. Expression of the immediate-early 1 (IE1) protein and the UL99 (pp28) late protein were examined in HFF and telomerase 18 cells at various times after infection. As shown in Fig. 3, the expression level and kinetics for IE1 and pp28 are identical between the two cell lines. Similar results were obtained with telomerase-positive clones 12 and 16 (data not shown). To ensure that all telomerase cells in culture were permissive for HCMV, HFF and telomerase 18 cells were infected at an MOI of 5 PFU/cell and stained 72 h postinfection for expression of the late antigen pp28. Over 95% of HFF and telomerase 18 cells stained positive for pp28 expression (data not shown).
FIG. 3.
Comparison of immediate-early and late gene expression in HFF and telomerase-positive cells. HFF or telomerase 18 cells were infected with HCMV at an MOI of 5. Cell lysates were prepared at the indicated times postinfection (P.I.), and 50 μg of protein from each sample was analyzed by Western blotting using antibodies against either IE1 (Chemicon 810) or pp28 (9, 11).
To demonstrate that the life-extended fibroblasts could be used to complement a defective HCMV mutant, we generated a derivative clone expressing the HCMV UL82 (pp71) gene product. UL82 was chosen because previous attempts to propagate a UL82 deletion mutant have been unsuccessful in our laboratory, suggesting that UL82 may be required for efficient HCMV replication (data not shown). UL82 was put under the control of the HCMV UL99 promoter to inhibit expression of UL82 in the absence of HCMV infection, thereby ameliorating a potential detrimental effect of UL82 on cell growth. The UL82 (pp71) expression vector was constructed by using oligonucleotides (5′-AGATCTGCGCCGGCGTCTCGCCGGGCATC-3′ and 5′-GCTAGCCACGTTGAGCCGGCCCAGCAGCTC-3′) for PCR amplification of the UL99 promoter from HCMV AD169 viral DNA and cloned into the pGEMT-Easy vector (Promega). A BglII/NheI fragment containing the UL99 promoter was cloned into the pCI-neo vector (Promega) that had been digested with BglII/NheI. The resulting plasmid, pWF28, replaced the HCMV immediate-early enhancer/promoter with the UL99 promoter. The UL82 cDNA containing a hemagglutinin tag was then amplified from pCGN71 (1) using oligonucleotides (5′-CTATGGCTTCTAGCTATCCTTATGAC-3′ and 5′-CTCTAGATGCGGGGTCGACTGCG), cloned into the pGEMT-Easy vector, and sequenced. The tagged cDNA was removed via EcoRI digestion and cloned into EcoRI-digested pWF28. The resulting plasmid, pWF28-71-HA, was then transfected into telomerase 12 cells via electroporation (260 V, 960 μF) at population doubling 60 and cultured in the presence of 400 μg of G418 per ml. G418-resistant clones were selected and screened for UL82 expression. A UL82 deletion virus (ADsubUL82) was then purified and propagated on one stable clone (WF28-71-HA) expressing UL82. ADsubUL82 was generated by homologous recombination using a previously described insertion cassette (10) containing the green fluorescent protein. This cassette was flanked by sequences corresponding to nucleotides 116666 to 117628 and 119206 to 120278 of the HCMV AD169 genome. This cassette containing the UL82 flanking sequences was then transfected into HFF cells together with infectious AD169 viral DNA. Green fluorescent plaques were picked, purified on WF28-71-HA cells, and screened by Southern blotting and PCR amplification to ensure that we had obtained a pure virus stock and correct recombination.
The mutant virus was then assayed for its ability to replicate on noncomplementing HFF cells compared to complementing WF28-71-HA cells. HFF or WF28-71-HA cells were infected at an MOI of 0.01 PFU/ml with either wild-type AD169 or ADsubUL82, and viral titers were determined at 21 days postinfection. As shown in Table 2, the wild-type virus gave rise to similar titers on both HFF and WF28-71-HA cells. In contrast, ADsubUL82 did not grow on the noncomplementing HFF cells but yielded near-wild-type titers on the complementing WF28-71-HA cells (Table 2).
TABLE 2.
Comparison of wild-type and ΔUL82 viral titers on complementing and noncomplementing cell linesa
| Virus | PFU/ml
|
|
|---|---|---|
| HFF-R2 | WF28-71-HA | |
| Wild type | 2.06 × 106 | 1.58 × 106 |
| ΔUL82 | NPD | 3.25 × 105 |
Cells were infected at an MOI of 0.01 PFU/cell. Virus was harvested 21 days later, and yields were determined by plaque assay on WF28-71-HA cells. NPD, no plaques detected in undiluted inoculum.
In conclusion, life-extended fibroblasts supported the efficient replication of HCMV and provided the opportunity to generate derivative cell clones expressing viral genes that are useful for the propagation of mutant viruses.
Acknowledgments
We thank Geron Corporation for pGRN145.
This work was supported by grant CA82396 from the National Cancer Institute. W.A.B. is supported by an NIH postdoctoral fellowship.
REFERENCES
- 1.Baldick C J, Jr, Marchini A, Patterson C E, Shenk T. Human cytomegalovirus tegument protein pp71 (ppUL82) enhances the infectivity of viral DNA and accelerates the infectious cycle. J Virol. 1997;71:4400–4408. doi: 10.1128/jvi.71.6.4400-4408.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bodnar A G, Ouellette M, Frolkis M, Holt S E, Chiu C P, Morin G B, Harley C B, Shay J W, Lichtsteiner S, Wright W E. Extension of life-span by introduction of telomerase into normal human cells. Science. 1998;279:349–352. doi: 10.1126/science.279.5349.349. [DOI] [PubMed] [Google Scholar]
- 3.Boeckh M, Gleaves C A, Bindra R, Meyers J D. Comparison of MRC-5 and U-373MG astrocytoma cells for detection of cytomegalovirus in shell vial centrifugation cultures. Eur J Clin Microbiol Infect Dis. 1991;10:569–572. doi: 10.1007/BF01967276. [DOI] [PubMed] [Google Scholar]
- 4.Ho D D, Rota T R, Andrews C A, Hirsch M S. Replication of human cytomegalovirus in endothelial cells. J Infect Dis. 1984;150:956–957. doi: 10.1093/infdis/150.6.956. [DOI] [PubMed] [Google Scholar]
- 5.Ibanez C E, Schrier R, Ghazal P, Wiley C, Nelson J A. Human cytomegalovirus productively infects primary differentiated macrophages. J Virol. 1991;65:6581–6588. doi: 10.1128/jvi.65.12.6581-6588.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kari B, Radeke R, Gehrz R. Processing of human cytomegalovirus envelope glycoproteins in and egress of cytomegalovirus from human astrocytoma cells. J Gen Virol. 1992;73:253–260. doi: 10.1099/0022-1317-73-2-253. [DOI] [PubMed] [Google Scholar]
- 7.Kim N W, Piatyszek M A, Prowse K R, Harley C B, West M D, Ho P L, Coviello G M, Wright W E, Weinrich S L, Shay J W. Specific association of human telomerase activity with immortal cells and cancer. Science. 1994;266:2011–2015. doi: 10.1126/science.7605428. [DOI] [PubMed] [Google Scholar]
- 8.LaFemina R, Hayward G S. Constitutive and retinoic acid-inducible expression of cytomegalovirus immediate-early genes in human teratocarcinoma cells. J Virol. 1986;58:434–440. doi: 10.1128/jvi.58.2.434-440.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nowak B, Sullivan C, Sarnow P, Thomas R, Bricout F, Nicolas J C, Fleckenstein B, Levine A J. Characterization of monoclonal antibodies and polyclonal immune sera directed against human cytomegalovirus virion proteins. Virology. 1984;132:325–338. doi: 10.1016/0042-6822(84)90039-4. [DOI] [PubMed] [Google Scholar]
- 10.Patterson C E, Shenk T. Human cytomegalovirus UL36 protein is dispensable for viral replication in cultured cells. J Virol. 1999;73:7126–7131. doi: 10.1128/jvi.73.9.7126-7131.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Romanowski M J, Garrido-Guerrero E, Shenk T. pIRS1 and pTRS1 are present in human cytomegalovirus virions. J Virol. 1997;71:5703–5705. doi: 10.1128/jvi.71.7.5703-5705.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Span A H, Mullers W, Miltenburg A M, Bruggeman C A. Cytomegalovirus induced PMN adherence in relation to an ELAM-1 antigen present on infected endothelial cell monolayers. Immunology. 1991;72:355–360. [PMC free article] [PubMed] [Google Scholar]
- 13.Waldman W J, Roberts W H, Davis D H, Williams M V, Sedmak D D, Stephens R E. Preservation of natural endothelial cytopathogenicity of cytomegalovirus by propagation in endothelial cells. Arch Virol. 1991;117:143–164. doi: 10.1007/BF01310761. [DOI] [PubMed] [Google Scholar]
- 14.Weinshenker B G, Wilton S, Rice G P. Phorbol ester-induced differentiation permits productive human cytomegalovirus infection in a monocytic cell line. J Immunol. 1988;140:1625–1631. [PubMed] [Google Scholar]