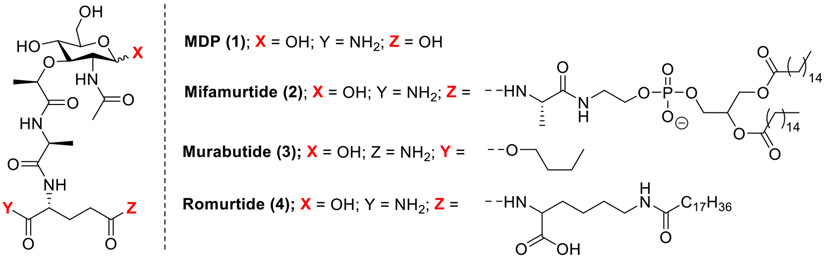Abstract
Nucleotide-binding oligomerization domain 2 (NOD2) is a receptor of the innate immune system that is capable of perceiving bacterial and viral infections. Muramyl dipeptide (MDP, N-acetyl muramyl L-alanyl-D-isoglutamine), identified as the minimal immunologically active component of bacterial cell wall peptidoglycan (PGN) is recognized by NOD2. In terms of biological activities, MDP demonstrated vaccine adjuvant activity and stimulated non-specific protection against bacterial, viral, and parasitic infections and tumors. However, MDP has certain drawbacks including pyrogenicity, rapid elimination, and lack of oral bioavailability. Several detailed structure-activity relationship (SAR) studies around MDP scaffolds are being carried out to identify better NOD2 ligands. The present review elaborates a comprehensive SAR summarizing structural aspects of MDP derivatives in relation to NOD2 agonistic activity.
Keywords: NOD2, muramyl dipeptide, MDP, vaccine adjuvant, PAMPs, innate immunity
Graphical Abstract

1. Introduction
The innate immune system is the bedrock of the body’s ability to fight early infection and comprises various families of pattern recognition receptors (PRRs) [1,2] which are emerging as promising targets for immunomodulation and vaccine adjuvant discovery [3-10]. One such family is the nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs) which include NOD2, a target of MDP, with other major families including the toll-like receptors (TLRs), and retinoic acid-inducible gene (RIG)-like receptors, and the C type lectin receptors [11]. PRRs detect invading microorganisms via recognition of pathogen-associated molecular patterns (PAMPs) [12]. PAMPs can comprise specific features in bacterial or viral DNA or RNA or cell wall components like peptidoglycan (PGN) and lipopolysaccharides (LPS) [13]. NLRs, such as NOD2, play a major role in the formation of caspase-1 activation complexes, known as inflammasomes [14].
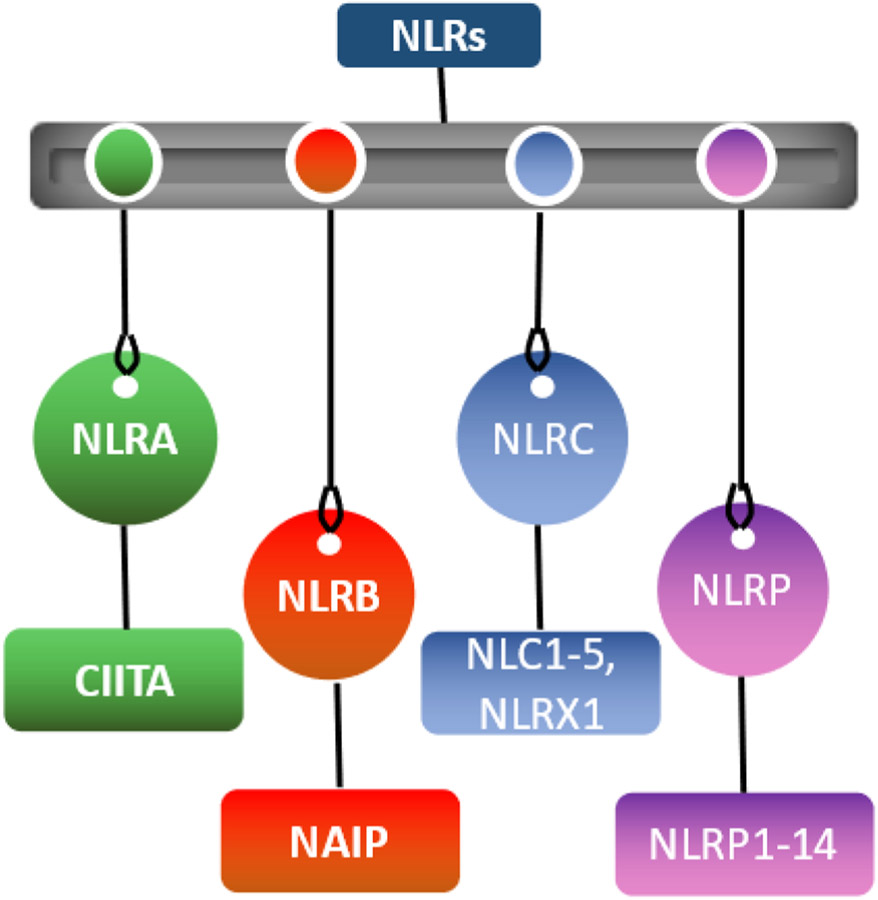
NLR family
In humans, there are 22 known NOD-like receptors (NLRs) [15,16] which are divided into four subfamilies (Fig. 1) based on the type of N-terminal domain: the acidic transactivation domain (NLRA), the baculoviral inhibitory repeat-like domain (NLRB), the caspase activation and recruitment domain-CARD (NLRC), and the pyrin domain (NLRP). The NLRA subfamily includes the MHC-II trans activator (CIITA) as the only member. Similarly, the human NLRB subfamily has only one member, NAIP. The NLRC subfamily consists of six members: NLRC1 (NOD1), NLRC2 (NOD2), NLRC3 (NOD3), NLRC4 (NOD4), NLRC5 (NOD5), and NLRX1; while the NLR-P subfamily consists of 14 members, NLRP1–14 [17][18].
Fig. 1.
Types of NLRs.
The sole member of the NLRA family, CIITA, is unique in the fact that it acts as a transcription factor [19]. Mutations in this gene are responsible for the bare lymphocyte syndrome, in which the immune system is severely compromised and cannot effectively fight infection. The NLRB subfamily consists of NAIP (NLR family, apoptosis inhibitory protein), which is responsible for triggering interleukin 1β (IL-1β) secretion in response to intracellular flagellin [12]. The most studied NLRs belong to the NLRC and NLRP families [20]. The Nucleotide-binding oligomerization domain, Leucine-Rich repeat, and Pyrin domain (NLRP) containing family, also referred to as the NALP family, play a role in apoptosis and inflammation with members of the NLRP subfamily expressing N-terminal pyrin domain and are crucial for the organization of IL-1β inflammasomes. The NLRC subfamily members display either N-terminal caspase recruitment domain (CARD) or an undefined domain that has no apparent homology with other proteins [21].
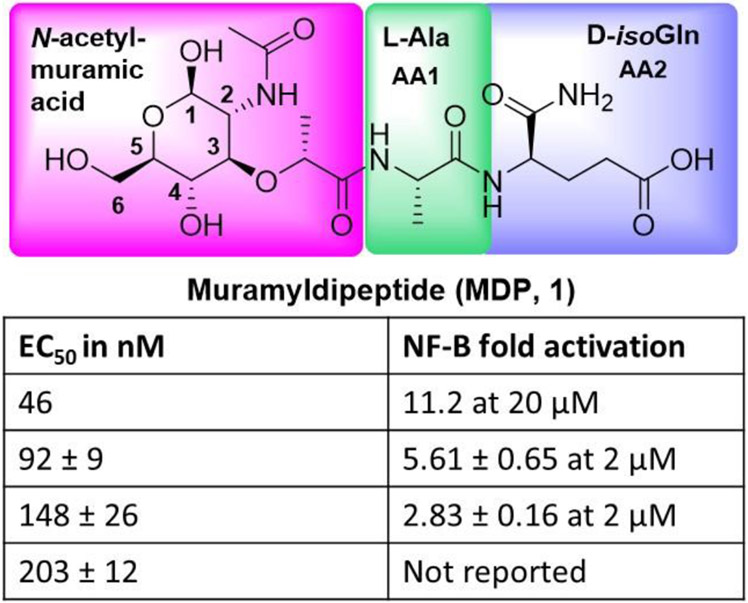
The present review focuses on NOD2, one of the receptors of the NLRC family. Among the NLRC receptors, NOD2 has two CARD domains, NOD1 and NOD4 has one, whereas NOD3, NOD5, and NLRX1 do not have an identified N-terminus domain [22]. The NOD1 and NOD2 receptors are known to recognize different bacterial and viral PAMPs [23]. NOD1 recognizes gamma-glutamyl diaminopimelic acid (iE-DAP), a peptidoglycan product of Gram-negative bacteria. Whereas, NOD2 recognizes muramyl dipeptide (MDP, 1, Fig. 2) present in the bacterial peptidoglycan which consist of N-acetyl muramic acid linked by its lactic acid moiety to a dipeptide comprising L-alanine (first amino acid, AA1) and D-isoglutamine (second amino acid, AA2) [24][25]. Recent studies have shown that NOD2 can react with viral single-stranded RNA (ssRNA), leading to type I interferon production [26][27] and activation of some inflammasomes. In humans, a frameshift mutation resulting in truncated NOD2 protein is found to be associated with susceptibility to Crohn's disease [28]. Conversely, mutations that lead to overexpression of NOD2 and excess nuclear factor-kappa B (NF-κB) activation results in Blau syndrome, an autosomal dominant disorder [29]. The EC50 (half-maximal effective concentration) or specific NF-κB activation values for Muramyl dipeptide (MDP) vary significantly based on experimental conditions, cell types used, and the assay methods employed in different studies. The fold increase in the NF-κB activation is with respect to the untreated or negative control [30-33].
Fig. 2.
Muramyl dipeptide (MDP) structure.
2. Mechanistic details of NOD2 recognition and activation
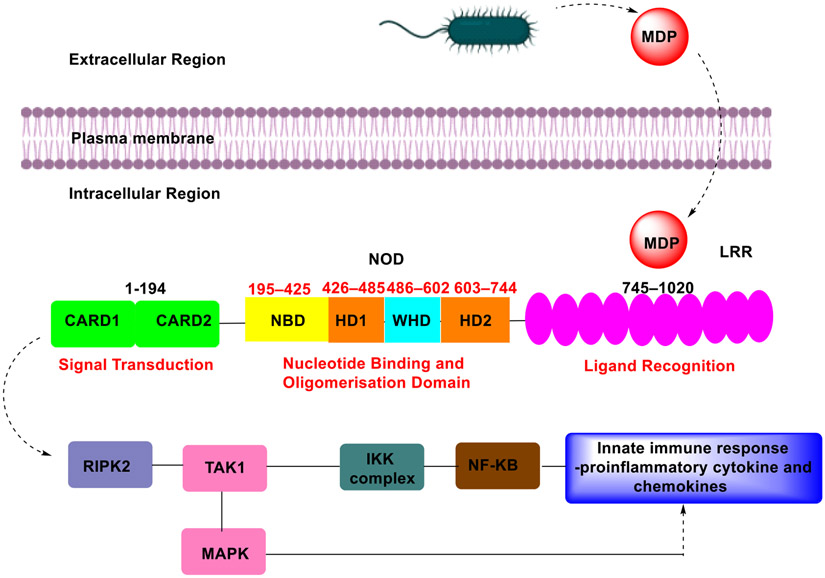
NOD2 is a bona fide receptor for MDP, a cell wall component of both Gram-positive and Gram-negative bacteria. NOD2 is known to reside in an inactivated state with the leucine-rich repeats (LRRs) folded back onto the NACHT domain (or NOD domain). On exposure to MDP, NOD2 undergoes conformational changes allowing it to self-oligomerize through the kinase receptor-interacting protein 2 (RIP2) via homophilic CARD–CARD interaction (Fig. 3). The RIP2 then mediates the recruitment and activation of the Transforming Growth Factor Beta-Activated Kinase 1 (TAK1), which is a prerequisite for activating the IKK kinase complex and mitogen-activated protein kinase (MAPK) pathway. This allows NF-κB to translocate to the nucleus and start transcription of proinflammatory genes, including cytokines, growth factors, and factors responsible for stimulating immune cells [34-40].
Fig. 3.
Structure and signalling pathway of NOD2 activation.
While the structure of human NOD2 is yet to be determined, Maekawa et al. reported the crystal structure of rabbit NOD2 in the ADP-bound state [41]. The crystal structure showed that ADP-bound NOD2 folded into a hook-shaped structure with dimensions of ~70 x 80 x 40 Å, consisting of a NOD domain (residues 195–744) and a LRR domain (residues 745–1,020). The NOD domain can be further sub-divided into the nucleotide-binding domain (NBD, residues 195-425), helical domain 1 (HD1, residues 426–485), the winged helix domain (WHD, residues 486–602), and helical domain 2 (HD2, residues 603–744) [41], Lauro et al. and others have proposed that the recognition of MDP occurs within the LRR domain [31,42,43]. However, this remains unproven as the crystal structure of NOD2 bound to MDP has not yet been reported, so the molecular details of this interaction remain poorly understood.
The mutations of NOD2 are associated with inflammatory diseases including, Blau syndrome, early-onset sarcoidosis and Crohn’s disease [44]. Apart from the role of NOD receptors in pathogen recognition and inflammation, they also play an important role in cancer progression [17]. For example, Mifamurtide (2, Fig. 4), a NOD2 agonist, has been approved for immunotherapy in combination with chemotherapy for patients with osteosarcoma, [45-47] subsequent to surgical resection of the primary tumor [17]. A synthetic derivative of MDP, murabutide (3, Fig. 4) was shown to suppress HIV-1 replication in monocyte-derived macrophages. Romurtide, a synthetic stearoyl-MDP derivative (also known under the names MDP-Lys and muroctasin) (4, Fig. 4) is being used in the treatment of leukopenia that occurs during radiotherapy [48]. Recently, another role of NOD2 in stem cell protection via recognition of commensal microbiota was highlighted by Nigro and co-workers [49].
Fig. 4.
Synthetic derivatives of MDP in clinical use.
3. Structure Activity Relationship Investigations
The NOD2 activity of MDP (1, Fig. 4. NF-κB activation: 45.6 ± 2.5 fold at 100 ng) [50] is higher than that of murabutide (MB, 3, NF-κB activation: 17.6 ± 1.2 fold at 100 ng) but has low clinical utility due to drawbacks, including pyrogenicity, rapid elimination, and lack of oral availability [51]. Identification of new NOD2 ligands with greater clinical utility will be possible with better understanding of the SAR in MDP derivatives.
3.1. Modifications at the C1 and C2 position of the N-acetyl muramyl moiety
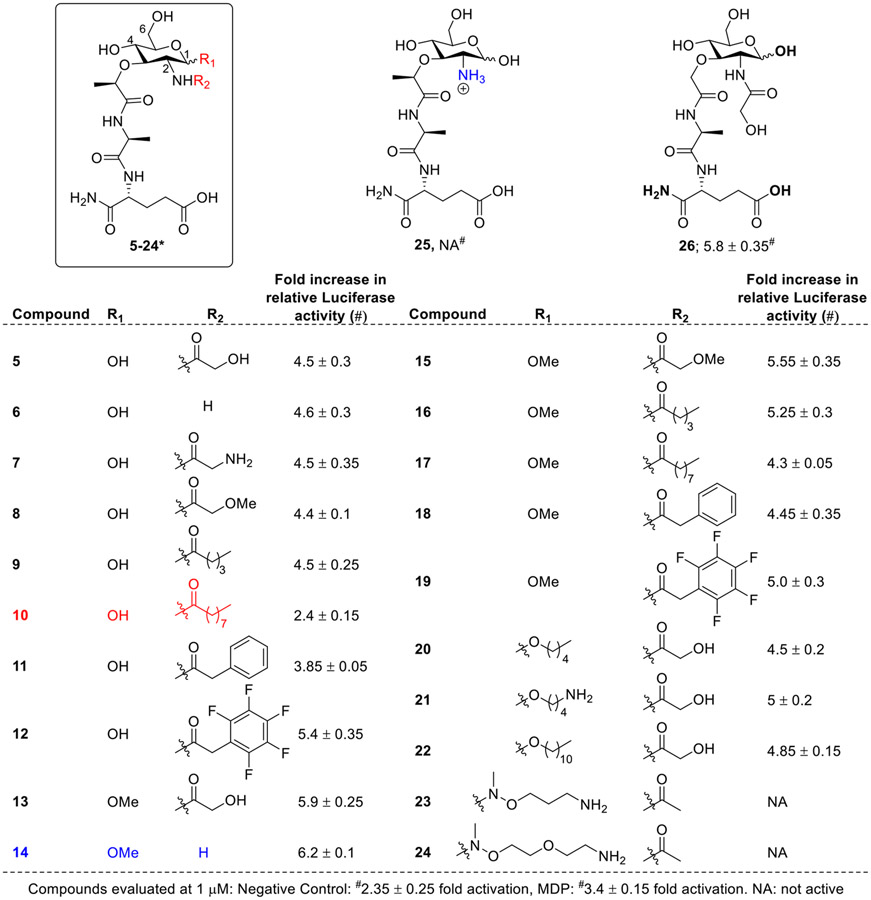
Chen and co-workers generated a library of compounds having modifications at the C1 and C2 position of MDP scaffold (5-24, Fig. 5) and evaluated at 1μM concentration in the NOD2-dependent NF-κB activation assay using HEK293T cells [52]. The first set of compounds was prepared by N-acylation at C2 position and by keeping hydroxyl functionality at the C1 position (5-12, Fig. 5). All the newly synthesized analogs were found to be potent stimulators than MDP except compound 10 confirming the non-tolerance to long N-acyl chain at C2 position. Further, analogs bearing the methoxy group at the C1 equatorial (β) position (13-19) were synthesized, and it was interesting to observe that all these analogs were more potent than their respective anomeric (axial) hydroxy analogs (5-12). Compounds bearing ether linked O-alkyl chains (C5-Cll, 20-22) at the equatorial C1 position showed similar potency to that of 5, whereas the replacement by polar methylaminoxy amines (23 and 24) at the C1 position abrogated the activity. Surprisingly, compound 14 bearing a C1 methoxy group with no substituent attached to the C2 amine group was found to be the most potent stimulator of NOD2. In contrast, the compound 25, which is the salt form of compound 6, showed diminished NOD2 agonistic activity. It was also interesting to observe that compound 26, lacking the methyl group of lactate, was a more potent activator than MDP [53][54].
Fig. 5.
Analogs of MDP having modifications at the C1 and C2 positions. *R1 and R2 modifications in compounds 5-24 are shown in tabular format.
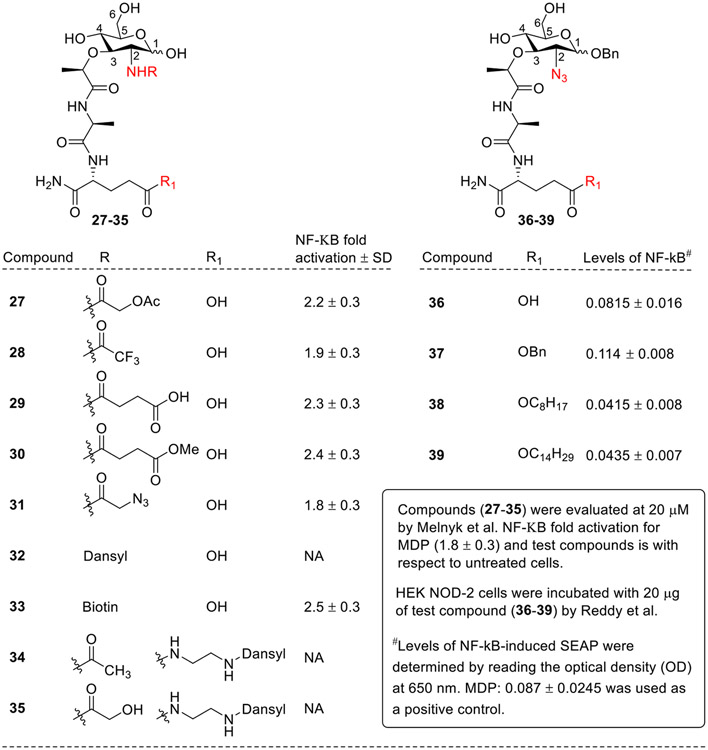
The compounds having N-acyl groups at the C2 position (27-31, Fig. 6) [55] still retained the NOD2 agonist activity two-fold greater than the untreated control. The fluorescent derivatives, bearing dansyl at the C2 position (32) or linked on the isoglutamine residue with small ethylenediamine linker (34, 35) did not activate NOD2. In contrast, compound 33, with biotin at the C2 position was still active, although to a lesser extent than other acylated derivatives. Recently, Reddy et al. synthesized the azido derivatives with O-benzyl moiety at C1 position (36-39, Fig. 6). Among the synthesized derivatives, compound 37 showed better NOD2 agonistic activity, whereas the compound 36 exhibited comparable activity to standard MDP. The compounds bearing C8 and C14 chains (38 and 39, respectively) showed minimal NOD2 activity. Also, when compound 37 was explored for in vivo studies, it showed promising IgG response, indicating its ability to recognize and bind to numerous antigens. It also promoted both T-helper cell responses and cell-mediated immune responses, expressing more CD4 (Cluster of Differentiation 4) population.
Fig. 6.
C2 modified MDP analogs by Melnyk and Reddy et al.
Overall, this SAR confirmed that most of the C1 and C2 modified analogs showed higher potency than MDP, except for compounds bearing N-decanoyl moiety at C2 position and polar methylaminoxy amines at C1 position. The compounds with equatorial C1-methoxy group were generally more potent than those with anomeric hydroxyl functionality at this position. It is noteworthy to observe that a proper-sized hydrophobic moiety at the C1 position significantly increased NOD2 agonistic activity.
3.2. Modifications at the C4 position of the N-acetyl muramyl moiety
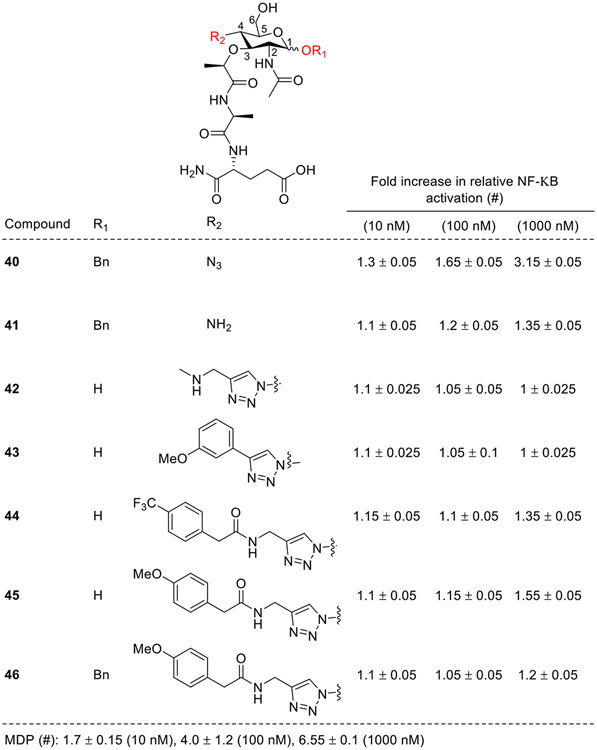
Cheng and co-workers next modified the C4 position of the carbohydrate moiety [51]. A dramatic loss of NOD2 activity was observed for 40, 41, and 46 (Fig. 7) due to the masking of the hydroxy group by the benzyl group at the C1 axial position as compared to MDP, suggesting that the orientation and substituents at the C1 position might play an important role in NOD2 activity. Also, the compounds with a substituted 1,2,3-triazole moiety (42-46) at the C4 position showed diminished activity which attributes to their weaken interactions with the NOD2 receptor. This shows that a substituted triazole moiety at the C4 position causes significant reduction of NOD2 activity.
Fig. 7.
Modifications at the C4 position of the carbohydrate moiety.
3.3. Modifications at the C6 position of the N-acetyl muramyl moiety
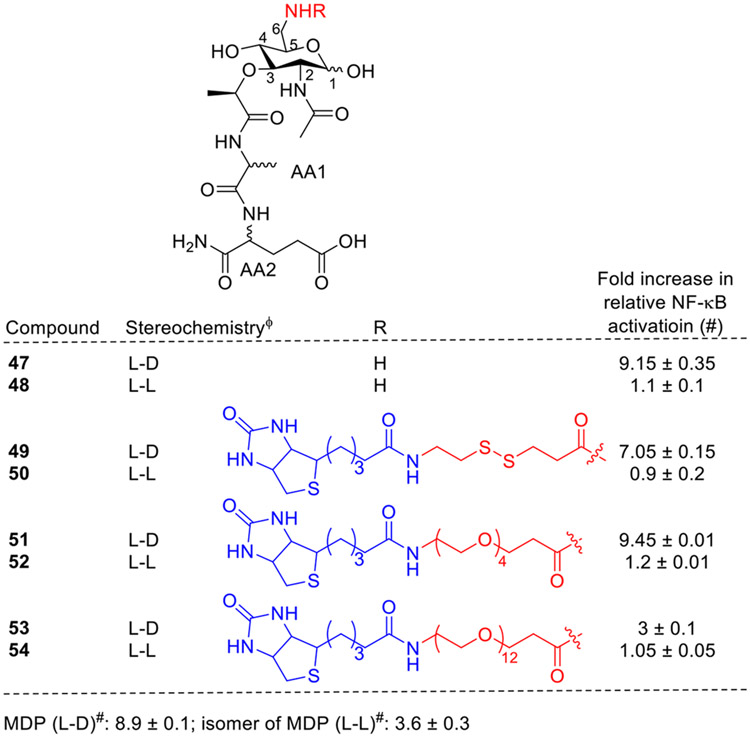
Sansonetti et al. showed that MDP activates NOD2 in a stereospecific manner. Substitution of D-isoglutamate by L-isoglutamate in MDP failed to stimulate NOD2, suggesting that the specific peptide portion of MDP is important for the ligand recognition [24,56]. To keep the recognition region of the MDP molecule intact and available for interaction, Grimes and coworkers reasoned that a modification would best be made at the C6 position of the carbohydrate, far off from the potential binding site C3. Accordingly, C6-amino versions of both MDP-L-D (47, Fig. 8) and MDP-L-L (48) were synthesized, but only L-D derivatives could activate NOD2. In addition, 6-amino MDP derivatives containing biotin with disulfide bond as a cleavable linker (49, 50) as well as with polyethyleneoxy linkers of variable lengths (51–54) were also synthesized. The synthesized analogs were tested for their ability to activate NOD2 in an established NF-κB luciferase assay. Among these compounds, 49 and 51 with a shorter linker (length 20 and 32 A, respectively) as well as with L-D configuration only were found to be the most active. Compound 53, with a longer linker (length 56 Å) was the least active in the series.
Fig. 8.
NOD2 agonists having modifications at the C6 position of MDP. ΦL-D and L-L are the stereochemistry of alanine (first amino acid AA1) and glutamine (second amino acid AA2).
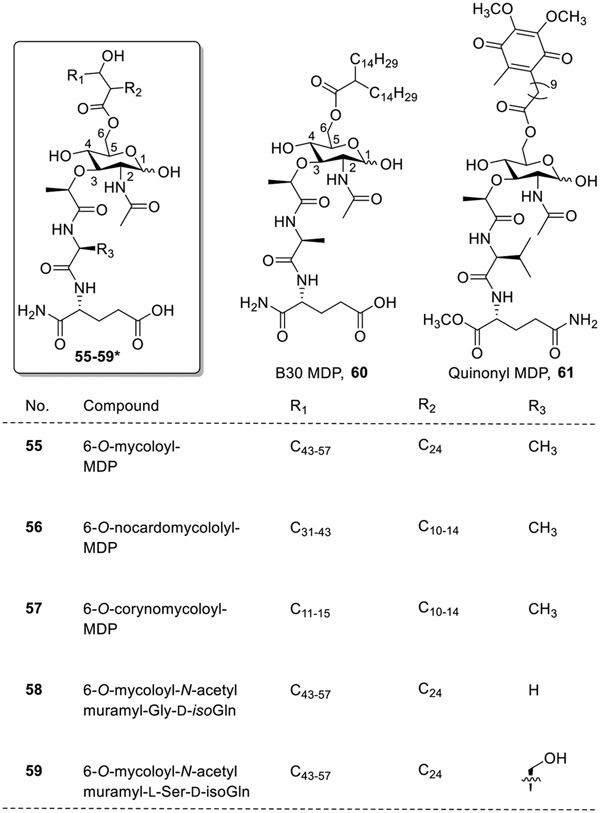
Azuma et al. also synthesized MDP analogs having modifications at the C6 position (6-O-acyl derivatives) in which the muramyl moiety was conjugated with various lipophilic molecules. These compounds were evaluated for their antitumor activities [57-63]. It was observed that administration of 100 μg each of 6-O-mycoloyl-MDP (55, Fig. 9), 6-O-nocardomycololyl (56), 6-O-corynomycoloyl-MDP (57), or 6-O-mycoloyl-N-acetylmuramyl-Gly-D-isoGln (58) in an oil-based vehicle suppressed the growth of fibrosarcoma (tumor) in mice while 6-O-mycoloyl-N-acetylmuramyl-L-Ser-D-isoGln (59) was active in terms of regression of an established line 10 hepatoma in guinea pigs. In addition to these analogs, B30-MDP (60) was also synthesized in which natural mycolic acid was substituted by a synthetic fatty acid chain of higher molecular weight resulting in stronger antitumor immunity. Further modification led to the synthesis of quinonyl-MDP-66 (61), which suppressed the growth of Meth A fibrosarcoma in mice.
Fig. 9.
MDP analogs having modifications at the C6 position. *R1, R2 and R3 modifications in compounds 55-59 are shown in tabular format.
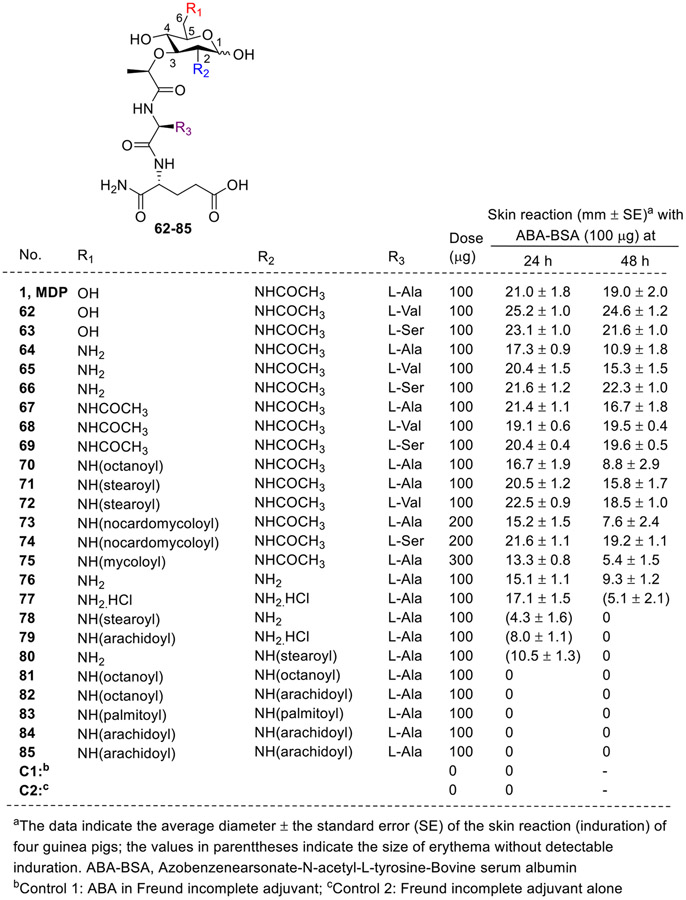
Further, Kobayashi et al. also studied the adjuvant activities of 6-amino-6-deoxy-N-acetylmuramyldipeptides (64-85) in comparison with corresponding MDP and its analogs (1, 62 and 63, Fig. 10) [57]. 6-Amino-6-deoxy-N-acetylmuramyldipeptides (64-66), 6-acetamido derivatives (67-69), and certain 6-acylamino derivatives (70-75) with long-chain fatty acids demonstrated adjuvant activity for delayed-type hypersensitivity in guinea pigs. The synthetic 6-amino-6-deoxy-muramyldipeptides (64-66) showed slightly weaker adjuvant activity compared to corresponding MDP analogs. Derivatives with mycoloyl amino modifications such as nocardomycoloylamino 73 and mycoloylamino-6-deoxy-N-acetylnuramyl-L-alanyl-D-isoglutamine 75 exhibited decreased adjuvant activity compared to acetamido derivatives. Acylation of 6-amino analogs (76 and 77) affected adjuvant activity differently than acylamino derivatives (78 and 79). Acylation with stearic or arachidic acid on certain 6-amino or 6-acetylamino groups led to decreased adjuvant activity (76 and 77) which was in contrary to 6-acylamino derivatives (67 to 75) of 6-amino-6-deoxy-N-acetylmuramyldipeptides (64 to 66), which showed potent adjuvant activity. Complete loss of adjuvant activity was observed upon acylation with octanoic acid, palmitic acid, or arachidic acid of both amino groups (82-85).
Fig. 10.
MDP analogs having modifications at the C6 position by Azuma et al.
Among the C6 modified compounds, the replacement of hydroxy group with an amino group showed improvement in NOD2 agonistic activity. It was also observed that the attachment of biotin with disulfide bond as cleavable linker retained the activity. However, variations in the linker lengths influenced the NOD2 activation; compounds with shorter linkers retained the activity and the ones with longer linkers were found to be less active. Additionally, 6-amino-6-deoxy-N-acetylmuramyldipeptides and their derivatives showed adjuvant activity, albeit slightly weaker than the corresponding MDP analogs. It was also observed that the N-acylation at the C6 position with different fatty acids drastically affected the adjuvant properties of the compounds. Overall, various C6 modified MDP analogs demonstrated promising antitumor effects, emphasizing their potential for suppressing tumor growth in different animal models, highlighting the diverse therapeutic prospects of this modification.
3.4. Modifications at the first amino acid (L-Ala)
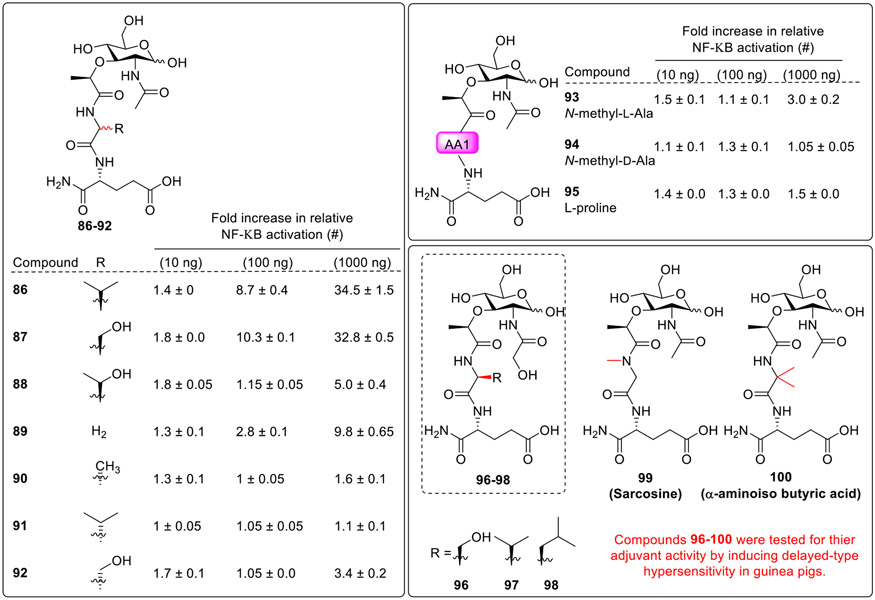
To further assess the effect of modifications at the first amino acid, L-alanine of MDP was replaced by L-valine (86, Fig. 11) or L-serine (87), [64,65][66] which showed comparable NOD2 activity. However, L-threonine (88), and glycine (90), N-methyl L-alanine (94) L-proline (95), had reduced NOD2 activity. MDP analogs 91, 92 and 94 having D-configuration of the first amino acid were utterly inactive. Further, some of these analogs were explored for their adjuvant activity by inducing delayed-type hypersensitivity in guinea pigs. Analog having proline (95), and sarcosine (99) as the first amino acid showed weak adjuvant activity, highlighting the necessity of the NH group adjacent to the lactic acid moiety for activity. Alanine substitution with other amino acids (e.g., valine 86, threonine 88, α-aminoisobutyric acid 100) increased the adjuvant activity, with the valine analog showing the most favourable effect. The N-glycolyl leucine analog 98 showed moderate activity as compared with other N-glycolyl analogs (96 and 97) showing that the size or lipophilicity of amino acids adjacent to the lactic acid affected activity. It was also observed that N-glycolylmuromyl analogs and N-acetylmuramyl analogs showed similar activity levels marking that N-glycolyl and N-acetyl group did not alter the activity.
Fig. 11.
Modifications at the first amino acid.
Overall, alanine with L-stereochemistry was found to be the best at the first amino acid of MDP. However, replacement of alanine with valine and serine showed comparable activity whereas N-methyl-alanine, threonine, proline, glycine and related amino acids with D-configuration drastically reduced the NOD2 agonistic potential.
3.5. Modifications at the second amino acid (D-isoglutamine)
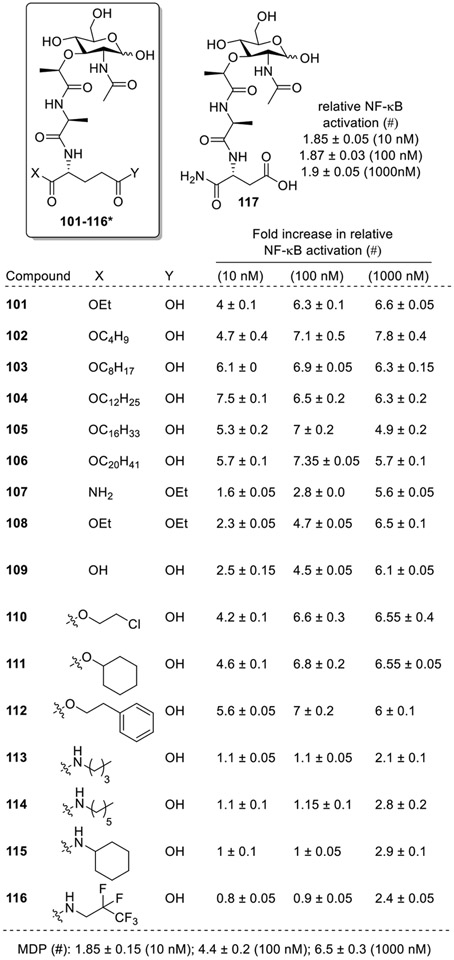
When MDP derivatives having modifications at the second amino acid were investigated, it was found that compounds having mono-ester moiety at the α-position (101-104, and 110-112, Fig. 12) were more active than MDP [50,67,68][69]. It was interesting to observe that NOD2 activity increases with increase in the chain length, with the C12 alkyl chain bearing compound 104 showing the highest NOD2 activity. Further increase in the chain 105 and 106 led to decrease in the NOD2 activity. On the other hand, compounds with mono-ester moiety at γ-position (107) exhibited less NOD2 activity than MDP. The compounds possessing di-ester moiety (108) were slightly more active than MDP, but less active than those with a mono-ester moiety (101). The compound possessing carboxylic acid moiety on both sides (109) showed similar activity to MDP. The analogs which contained a N-monosubstituted amide moiety having alkyl chains of varied lengths (113 and 114), a cyclohexyl moiety (115), and a fluorine-containing alkane (116) were inactive. The biological results also showed that the NOD2 activity was lost for the MDP analog 117, having D-isoasparagine instead of D-isoglutamine amino acid at terminal position.
Fig. 12.
MDP analogs having modification at the D-iso-glutamine residue (second amino acid) of the peptide part. *The modifications at X and Y in 101-116 are shown in tabular format.
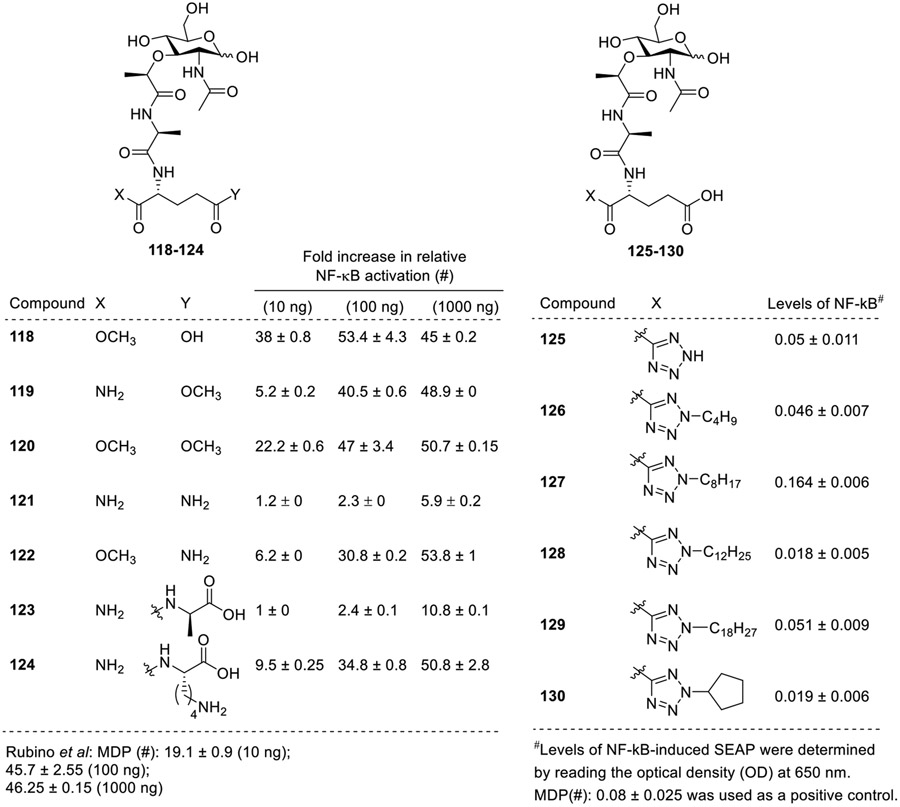
Further, few more analogs were explored by Rubino and co-workers for their NOD2 activity. Similar observations were observed that the compound with mono-ester moiety at α-position 118 (Fig. 13) showed higher activity than MDP. Surprisingly, compound 119 with methyl ester at γ-position and amino group at the α-position showed lower NOD2 agonistic activity than MDP but the compound having terminal amide group on both sides 121 was inactive. The compound with D-alanyl residue on the γ-carboxyl (123) resulted in diminished NOD2 activity, while the compound with L-lysyl residue (124) retained NOD2 activity but was lower than MDP. Further, when bioisosteric replacement of α amide of D-isoglutamine of MDP is carried out using with 2,5-substituted tetrazoles, it was observed that among the varied chain lengths 2,5-disubstituted tetrazole derivatives (125-130), [70] the tetrazole analogue 127 bearing the octyl (C8) chain showed the best NOD2 stimulation potency with reference to MDP while other analogs showed weak interaction with NOD2 receptor. Further, Rubino et al. also explored the analogs 118-124 for their adjuvant and anti-infectious activity. These compounds showed enhanced protection against bacterial infections and demonstrated strong adjuvant properties similar to MDP correlating with their robust NOD2 -dependent NF-kB activation.
Fig. 13.
MDP analogs having modification at the D-iso-glutamine residue by Rubino et al. and Mhamane et al.
Overall, it can be concluded that the mono-ester moieties at the α-position of the second amino acid (D-isoglutamine) with longer alkyl chains demonstrated higher NOD2 activity. However, the ester positioning at the γ position negatively impacted the NOD2 activity. The bioisosteric replacement of the terminal carboxamide with 2,5-substituted tetrazoles having medium sized alkyl chains were tolerated.
3.6. MDP derivatives having modifications at both the dipeptide and carbohydrate moiety
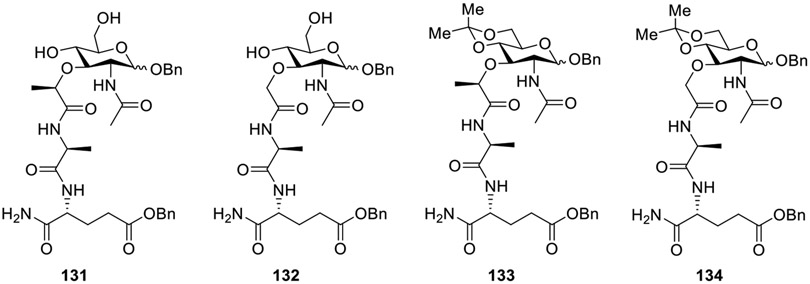
Cai et al. synthesized MDP analogues having modifications at both the dipeptide and carbohydrate moiety [71]. In the small set of compounds (131-134, Fig. 14), the dipeptide synthesized using D-isoglutamine benzyl ester and L-alanine was linked to C1 benzyl N-acetylglucosamine (GlcNAc) or its acetonide via lactic or glycolic acid. Among the four derivatives, only compound 133 was similar in activity to that of MDP. Interestingly, 133 gradually reduced TNF-α as a result of an increased concentration and induced AP1/A20 gene expression. At high doses, 133 reduced the transcription of RIP2, NOD2, and NF-κB in macrophages to a greater extent than MDP treatment. This indicates that, based on its efficacy, analog 133 is indeed an improved compound in comparison to MDP.
Fig. 14.
Modification at dipeptide and carbohydrate moiety.
3.7. Carbocyclic analogs of MDP
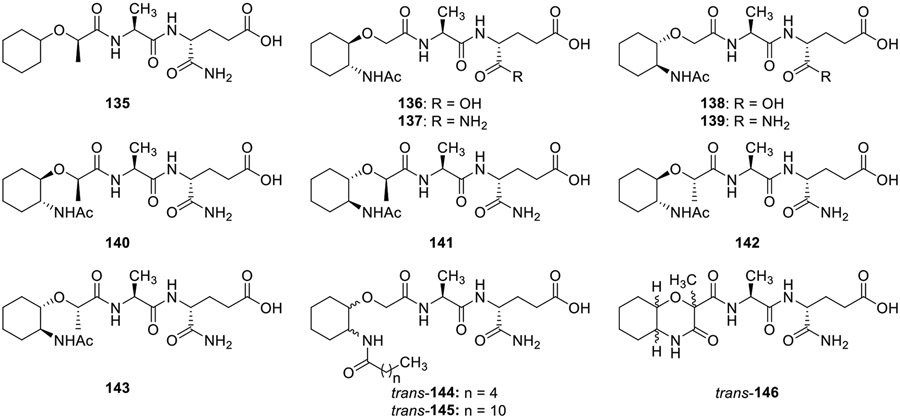
In order to understand the importance of carbohydrate moiety in MDP, the first carbocyclic analog of MDP, N-[D-2-(cyclohexyloxy)propionyl]-L-alanyl-D-isoglutamine (135, Fig. 15) was synthesized by Kikelj et al [72]. Compound 135 in which the carbohydrate moiety of MDP was replaced with cyclohexanol derivatives, was inactive as an adjuvant for the induction of delayed-type hypersensitivity to azobenzenearsonate-N-acetyl-L-tyrosine in guinea pigs. Kikelj and coworkers then synthesized a variety of carbocyclic MDP analogs (136-146) and these compounds were tested for their ability to enhance non-specific resistance in mice immunosuppressed with cyclophosphamide and infected with Candida albicans. The results were evaluated in reference to azimexon and MDP. The analogs with D-glutamic acid moiety (136 and 138) displayed significant immunorestorant activity, while the analogs with D-isoglutamine (137 and 139) were less active. Also, all the four diastereomers of N-{trans-2-[[2'-(acetylamino)cyclohexyl]oxy]propionyl}-L-alanyl-D-isoglutamine i.e., 140-143 were inactive, thus confirming the importance of D-glutamic acid. Analogs with increased lipophilicity 144 and 145 displayed activity at higher doses, being the most for 145 with a longer alkyl chain. The rigidified analog 146 showed reduced activity suggesting that rigidification is not beneficial for immunorestorative activity.
Fig. 15.
Carbocyclic analogs of MDP.
3.8. Desmuramylpeptides
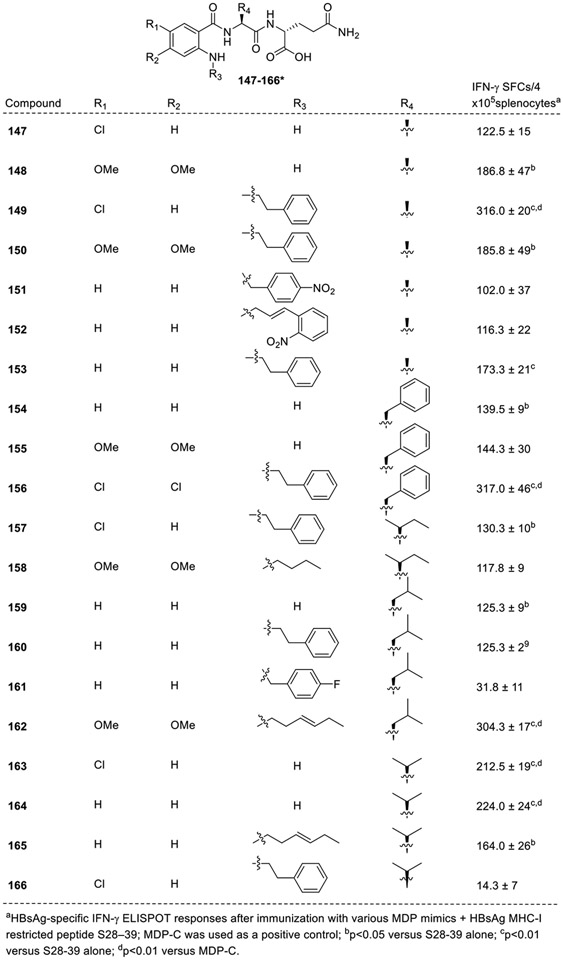
Zhao et al. synthesized a new series of analogs known as desmuramylpeptides wherein the N-acetylmuramyl moiety is replaced by a variety of aryl moieties with different spacers or linking functionalities (147-166, Fig. 16) [73]. The library was synthesized using solid phase strategy and their adjuvant activities were evaluated ex-vivo to determine the synergism with S28–39 peptide, a MHC class I binding epitope of recombinant hepatitis B surface antigen (HBsAg) for both human and mice. MDP-C (N2-[α-O-Benzyl-N-(acetylmuramyl)-L-alanyl-Disoglutaminyl]-N6-trans-(m-nitrocinnamoyl)-L-lysine,) was used as a positive control [74,75]. It is noteworthy to mention that the direct NOD2 agonistic activity of the synthesized library of compounds was not reported by the authors. In the first series, where L-alanine was used as the first amino acid, it was observed that compounds having a free amino group (147 and 148) at the C2 position did not improve the activity. Further, compound 149 having a phenethyl group at the R3 position and chlorine at R1 position was potent in regard to the enhancement of the immunological activity than the other analogs of this series. It was also observed that substitution with an electron-donating group, such as a methoxy group, at R1 and R2 positions were unable to contribute significantly to the activity of compound 150. Also, the compound with no substitution on benzene ring (151) was not effective. Further, introduction of a strong electron-withdrawing group, such as a nitro group on the substituents at the R3 position (151 and 152) did not improve the activity. Similar observations were obtained when L-phenylalanine and L-isoleucine were used as the first amino acids with N-phenethyl group at R3 position (156 and 160), showed enhancement of immunological activity. On the other hand, the analog with L-leucine as the first amino acid and phenethyl group at R3 position (157) showed moderate activity. Next, when L-valine was used as the first amino acid, only the compounds 163 and 164 showed moderate activity. Surprisingly, the analog 166 with N-phenethyl group at R3 position showed diminished activity. All the other analogs did not show any improvement in the immunological activity [73].
Fig. 16.
Substituted benzoyl containing desmuramylpeptides.
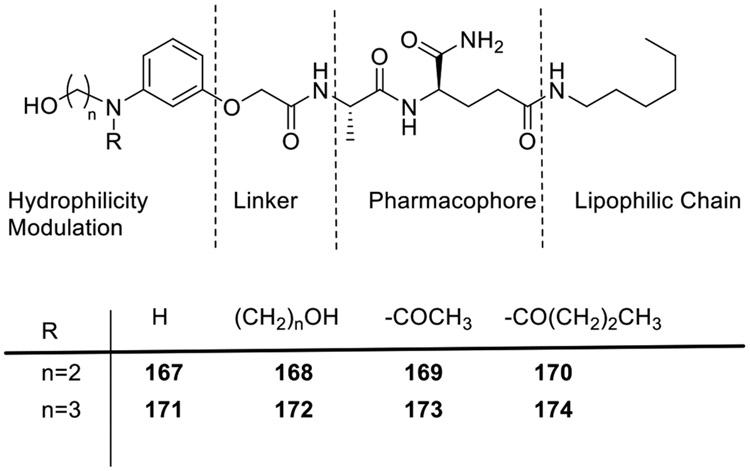
With an aim to find the appropriate surrogate for N-acetyl muramyl moiety, Khan and co-workers synthesized a new class of amphiphilic desmuramylpeptides wherein carbohydrate moiety of the parent molecule was replaced with hydrophilic arenes (Fig. 17) [76]. A lipophilic chain was also introduced at the C-terminus of dipeptide moiety while conserving L-D configuration. The 2-hydroxyethyl containing desmuramylpeptides (167-170) when used at 15 μM, showed slightly higher activity than Murabutide. Similarly, 3-hydroxypropyl containing desmuramylpeptides (171-174) showed higher activity than all 2-hydroxyethyl containing desmuramylpeptides except 169.
Fig. 17.
Desmuramylpeptides having hydrophilic arene as a substitute for carbohydrate moiety.
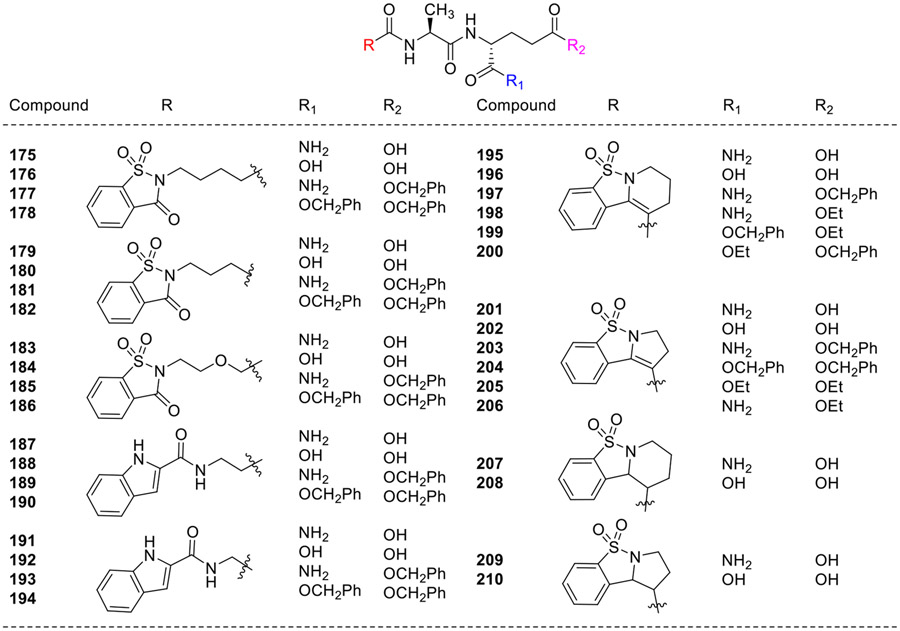
Further, Jakopin et al. synthesized a series of desmuramylpeptides, in which N-acetylmuramyl moiety was replaced by saccharin and indole heterocycles and their derivatives (175-210, Fig. 18) [77]. These compounds were screened for their ability to induce TNF-α release from naive THP-1 cells and to modulate LPS-induced TNF-α release. All these analogs, including MDP and MB, could not induce significant TNF-α release on their own, but a synergistic effect on lipopolysaccharide induced TNF-α secretion was observed. None of these analogs were as effective as MDP and MB. The MDP and MB enhanced LPS-induced TNF-α release by 87.4 and 82.6 %, respectively. All these analogs showed only weak activity, with compound 191 having an indole-scaffold, enhanced TNF-α release by 26.2%, showcasing moderate activity compared to the positive controls i.e MDP and MB. Further, to address the influence of lipophilicity, desmuramyldipeptide 195 with pyrido-fused [1,2]-benzisothiazole ring system and its corresponding benzyl (197) and ethyl esters (198) were investigated. However, the lipophilic benzyl ester (197) did not increase activity compared to the value of the parent compound 195. Similarly, the other pyrido-fused [1,2]-benzisothiazole analogs (207 and 208) were proved to be effective in enhancing the LPS-induced TNF-α release. However, immunomodulatory effects of pyrrolo-fused [1,2]-benzisothiazole derivatives 201-206 and 209-210 were not explained explicitly by the authors.
Fig. 18.
Saccharin and indole heterocycles containing desmuramylpeptides.
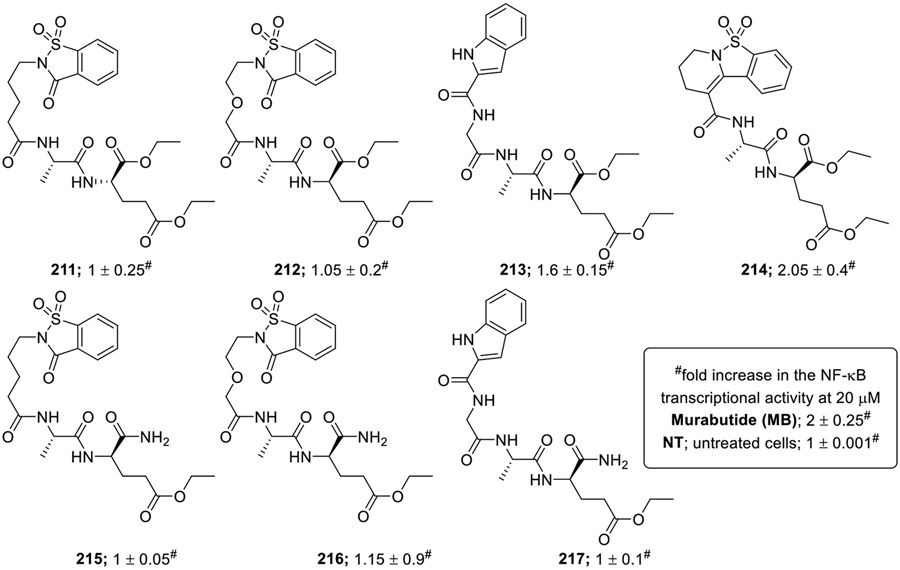
To address the lipophilicity of the saccharin and indole bearing desmuramylpeptides, the carboxylic acid functionalities of dipeptide moiety were replaced by ethyl ester functionalities (211-217, Fig. 19) [13]. These derivatives were screened for the NOD2 agonistic activity at 20 μM. The positive control MB 3 and the synthetic compounds 213 and 214 significantly increased NF-κB transcriptional activity with respect to untreated cells. However, other desmuramyldipeptides showed nearly the same activity as of untreated cells.
Fig. 19.
Novel desmuramylpeptides with ester functionalities.
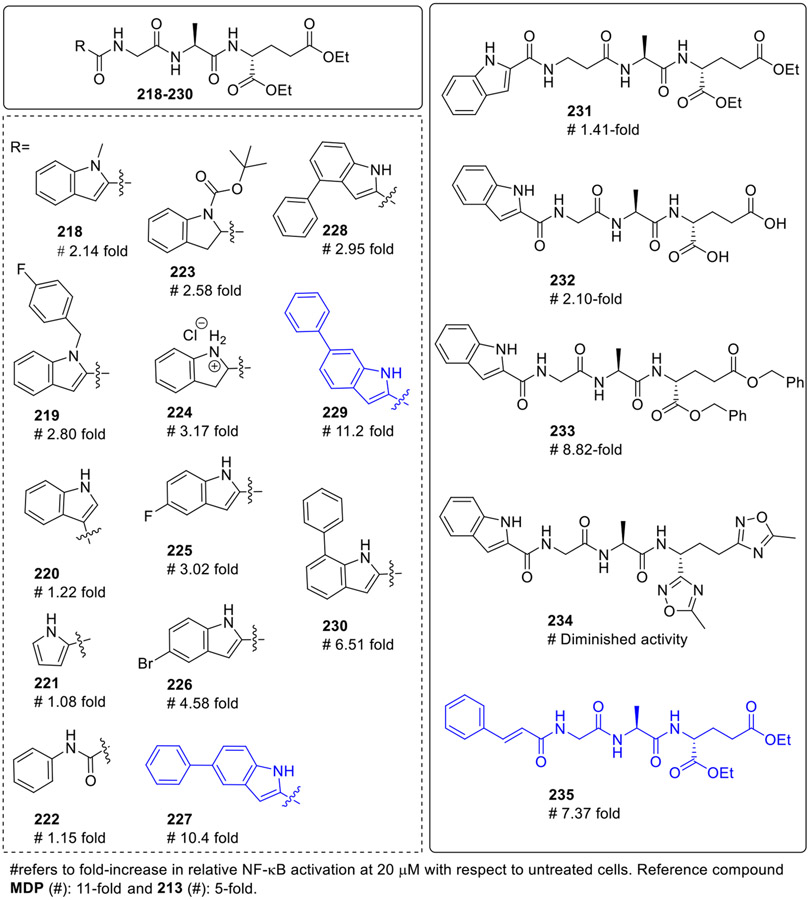
Further structural modifications to compound 213 were made involving the replacement of the indole-2-yl moiety by its closely related mimetics (218-235, Fig. 20), [30] along with the modifications in the peptide Gly-L-Ala-D-Glu moiety. The compounds derived by the methylation of secondary nitrogen of indole moiety (218) and substitution with 4-fluorobenzyl (219) demonstrated NOD2 activation, although to a lesser extent. Surprisingly, in regioisomeric derivative 220, formed by linking the peptide fragment at the C3 position of indole moiety instead of the C2 position, the NOD2 agonistic activity was completely abolished. Similarly, the analog lacking the benzene ring (221) and pyrrole ring (222) of the indole heterocycle resulted in the complete loss of activity. The partial saturation of the indole heterocycle resulted in indoline derivatives 223 and 224 with decreased NOD2 agonistic activities. It was interesting to observe that most of the 5-substituted indole derivatives were active. Introduction of electron-withdrawing group such as fluorine (225) and bromine (226), did not contribute significantly to the activity of the parent compound. Interestingly, the 5-phenyl substituted derivative 227 proved to be a potent NOD2 agonist with 10.4-fold activation. Thus, its regioisomeric derivatives 228-230 were also explored for their NOD2 agonistic activity. Among these compounds, the 6-phenylsubstituted regioisomer 229 showed slightly improved activity than 227. Elongating the glycine spacer by one methylene gave compound 231 with significantly diminished NOD2 activity. The hydrolysis of diethylester groups afforded compound 232, which exhibited decreased NOD2 activity and the bioisosteric replacement of dicarboxylic acids with 1,2,4-oxadiazoles (234) also resulted in diminished activity. As expected, the dibenzyl ester derivative 233 showed a remarkable increase in the NOD2-agonistic activity. Interestingly, the introduction of cinnamoyl moiety afforded the analog 235, which showed enhanced NOD2 stimulatory capacity suggesting that the cinnamoyl structural fragment is an important feature for NOD2 recognition.
Fig. 20.
Desmuramylpeptides and their NOD2 agonistic activity.
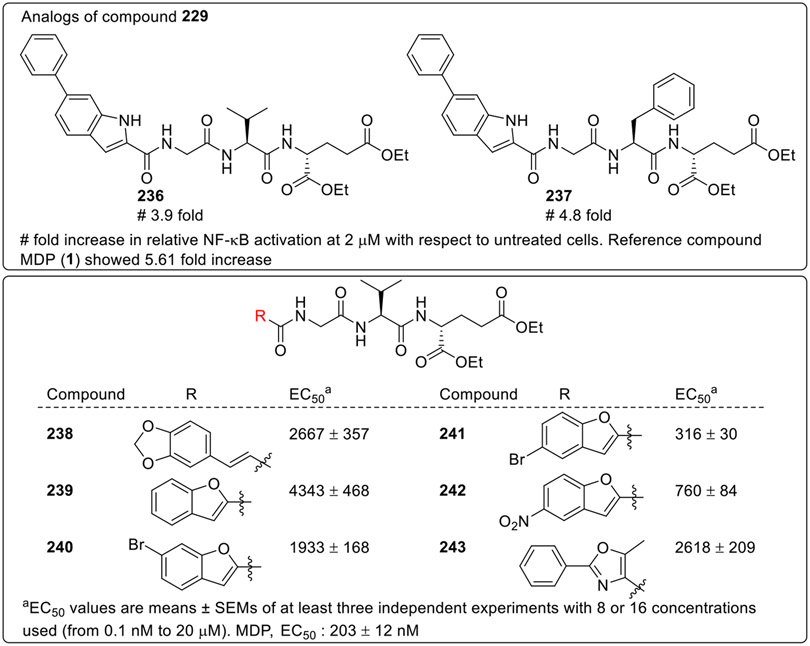
The compounds 229 and 235 were selected as the lead compounds and a detailed SAR study was then performed by Jakopin and co-workers. The synthesized compounds were evaluated for their ability to activate NOD2 in a reporter cell assay employing HEK-Blue NOD2-dependent NF-κB inducible secreted embryonic alkaline phosphatase (SEAP) reporter gene assay at 2 μM. As the compound 229 (Fig. 21) showed 11.2- fold activation, so its analogs with L-valine (236) and L-phenylalanine (237) were also explored, but none of these derivatives showed better NOD2 activation. So, this class of desmuramylpeptides were not explored further. When desmuramylpeptides having 3,4-methylenedioxy-cinnamoyl (238), benzofuran-2-carboxy (239), 6-bromobenzofuran-2-carboxy (240), and 5-methyl-2-phenyloxazole-4-carboxy (243) functionalities were explored for their NOD2 activity, the compounds showed diminished activity. However, the benzofuran derivatives having C5 bromo (241) and nitro (242) substituents showed improved activity.
Fig. 21.
Desmuramylpeptides having modifications at the C3 and C4 position along with modifications at the first amino acid.
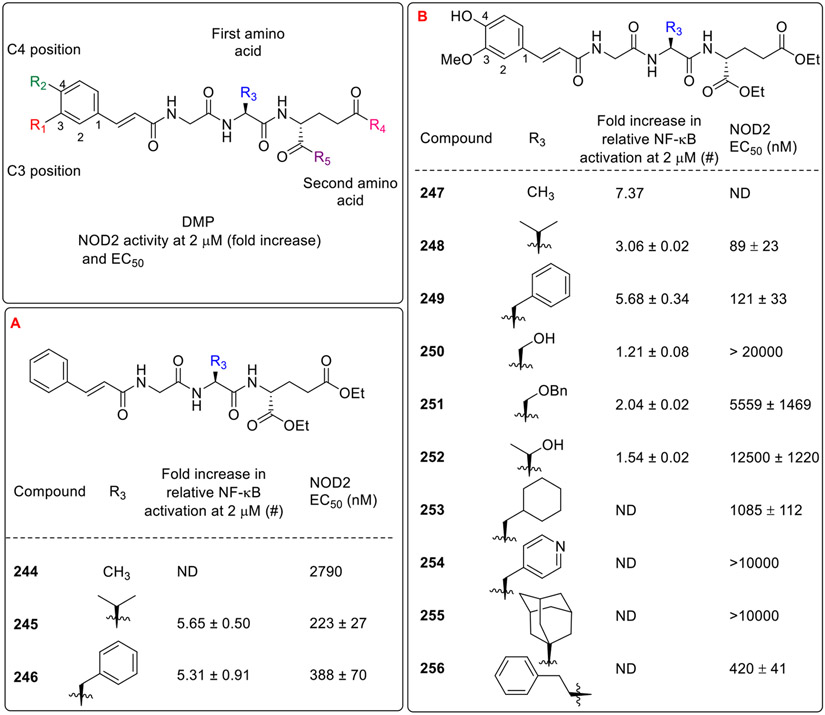
Next, the SAR study was performed on the desmuramylpeptide 235 (Fig. 22A). This SAR study analyzed the derivatization of the R1 and R2 position of cinnamoyl moiety as well as the replacement of the first (R3) and second amino acid (R4 and R5) of the peptide part. Initially, the variation on the first amino acid was carried out while keeping the cinnamoyl moiety intact. It was observed that the derivative with L-valine (245), instead of L-alanine (244) at the first amino acid showed maximum NOD2 activity followed by the L-phenylalanine derived analogue 246 [31]. Further, the cinnamoyl moiety was replaced with trans ferulic acid, and a variation of the first amino acid was also made. Among these compounds, compound 248 (Fig. 22B) with L-valine amino acid was identified as a potent NOD2 agonist. It was observed that analogs having L-serine (250), O-benzyl L-serine (251), L-threonine (252), L-cyclohexylalanine (253), L-pyridylalanine (254), (S)-adamantylglycine (255) and L-homophenylalanine (256) amino acids showed relatively poor activities while analogs bearing L-alanine (247) and L-phenylalanine (249) showed moderate NOD2 activity. Next, chemical space around the C3 and C4 positions of cinnamoyl moiety was also explored [32].
Fig. 22.
Desmuramylpeptides having cinnamoyl moiety and variation of first amino acid.
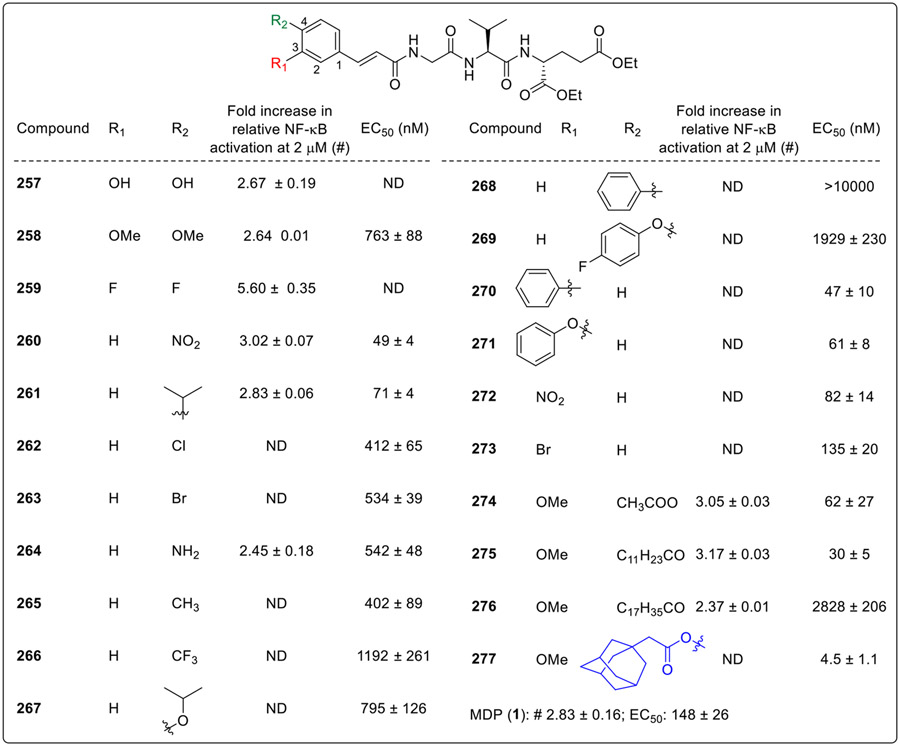
The compound 248 with R1 methoxy and R2 hydroxy group on cinnamoyl moiety was found to be a potent NOD2 agonist. On substitution with dihydroxy (257, Fig. 23) and dimethoxy (258) at R1 and R2 positions did not improve the activity, while the introduction of two fluorine atoms (259) on the phenyl ring of cinnamoyl moiety showed comparable activity as that of lead compound (248). Furthermore, the authors tested the effect of substituents at R2 by installing amino, nitro, methyl, trifluoromethyl, isopropyl, isopropoxy and phenyl groups. The 4-nitro and 4-isopropyl group bearing compounds (260 and 261), showed maximum NOD2 activity. The derivatives with functional groups such as 4-chloro (262), 4-bromo (263), 4-amino (264) and 4-methyl (265), exhibited the strongest NOD2 activity, followed by derivatives incorporating trifluoromethyl (266) or an isopropoxy (267) groups. A further increase in the size of the substituent at the R2 position to phenyl group (268) and 4-flouro phenoxy group (269) resulted in inactive analogs. In contrast, the functionalization at R1 position was better tolerated with phenyl (270) and phenoxy (271) derivatives exhibiting the same potency as lead compound 248 followed by nitro (272) and bromo (273) derivatives. Acylation of the 4-hydroxy group of the lead compound with acetyl (274) and lauroyl tail (275) on the aromatic ring retained the activity. However, increasing the C12 chain of 275 to C18 (276) resulted in markedly diminished activity. Furthermore, acylation with lipophilic adamantane-moiety produced compound 277, which was identified as the first desmuramylpeptide with NOD2 stimulating activity in the single-digit nanomolar range.
Fig. 23.
Desmuramylpeptides having C3 and C4 substituted cinnamoyl moiety.
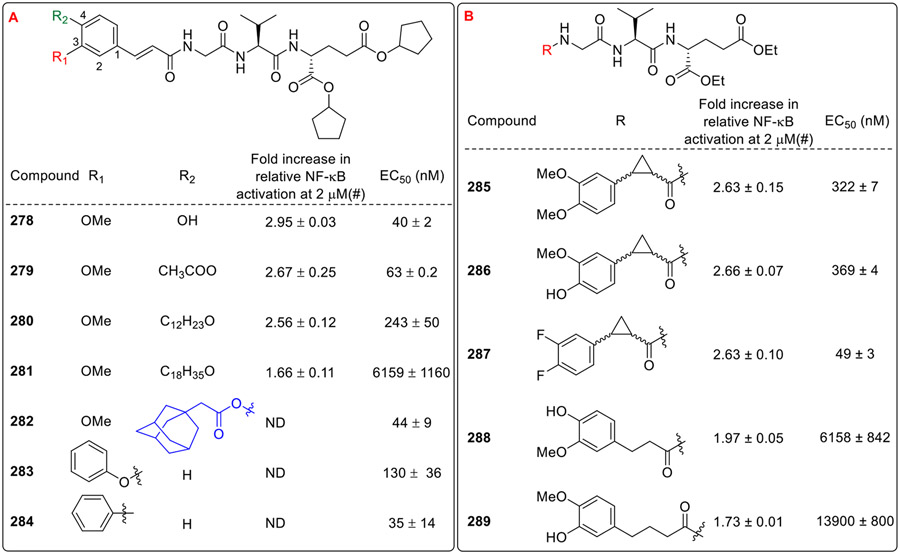
Jakopin and co-workers also synthesized cyclopentyl derivatives which served a dual role [33]. In addition to increasing the lipophilicity, analogs with cyclopentyl esters have been reported to be selectively cleaved by human carboxylesterase-1 enzyme. The cyclopentyl congener of 235 (Fig. 20) i.e., compound 278 (Fig. 24A) showed a two-fold improvement over 235. Among the O-acyl derivatives of the cinnamoyl moiety, the acetyl derivative (279) showed the maximum NOD2 activity, the lauroyl derivative (280) retained the activity and the activity was diminished by extending the lipophilic tail to C18 (281). Hence NOD2 activation by these compounds is most likely linked directly to their lipophilicity. The analog with lipophilic adamantane (282) showed maximum NOD2 activity in this series, whereas the compound 284 with phenyl ring substituted at the R1 position of cinnamoyl moiety showed moderate activity. Furthermore, the authors tested the effect of cyclopropanation of the double bond of cinnamoyl moiety (285-287, Fig. 24B). However, this approach provided only marginal improvement in the NOD2 activity. It was also observed that the reduction of the double bond (288) decreased NOD2 agonistic activity by a factor of 70. Similarly, the NOD2 agonistic activity was reduced by a factor of 156 with the spacer prolonged to a propylene group (289) which indicated that the earlier identified lead 235 provides the optimal positioning of the aromatic ring.
Fig. 24.
Desmuramylpeptides having modifications
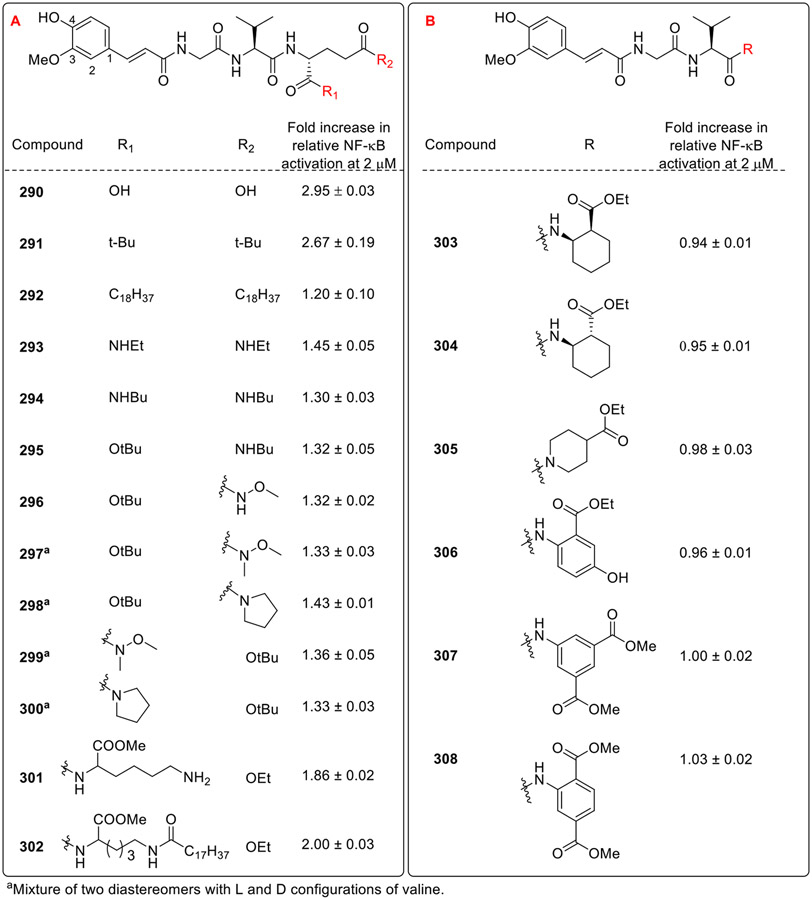
Hydrolysis of the diethyl ester groups of 235 (Fig. 20) to free carboxyl groups resulted in compound 290, with significantly diminished NOD2 activity. Similarly, when the diethyl ester groups were replaced by bulkier t-butyl ester (291, Fig. 25A), and lauroyl ester (292) resulted in markedly reduced NOD2 activation, may be because these bulky ester functionalities are considerably less hydrolyzable by human carboxylesterase-1. In addition to this, when functionalized carboxamates and amides (293-302) were evaluated as potential bioisosteric replacements of the carboxylic acid functionality, the compounds showed pronounced loss in NOD2 agonistic activity. It has been speculated that both the carboxylate groups of D-Glu form important interactions with the receptor. Thus, replacement with the rigidified moieties (303-308, Fig. 25B) completely abolished the NOD2 activity.
Fig. 25.
Desmuramylpeptides having modifications at the second amino acid. R1 and R2 are the modifications at the second amino acid (AA2) in compounds 290-302 and R is the modification at AA2 in compounds 303-308
Overall, in an attempt to replace the N-acetyl-muramyl moiety of MDP, various functionalities such as substituted benzoyl, saccharine, indole and cinnamoyl groups were explored by the research groups of Liu and Jakopin. The substituted benzoyl based desmuramylpeptides enhanced the immunological activity with meto-chloro substitution and phenylalanine as the first amino acid. Further, the systematic exploration of modifications around the indole core and cinnamoyl moiety revealed the crucial structure-activity relationships (SARs) governing NOD2 activation. Around the indole core, the electronegative groups like fluoro and bromo substituents didn't significantly impact the activity. Elongating the glycine spacer with its homologue β-alanine also resulted in loss of NOD2 activity. Interestingly, the positioning of phenyl substituent was pivotal, with the 6-phenyl substitution displaying potent activation compared to other regioisomers. Within the cinnamoyl motif, variations profoundly affected NOD2 activation. Substitutions on the phenyl ring, with methoxy at C3 position and hydroxy at C4 position enhanced the activity. Bulky and lipophilic side chains at the C4 position contributed to improved NOD2 activity, while modifications like fluoro substitution at the C3 and C4 position also retained the potency. However, alterations in the cinnamoyl moiety, including reduction of double bond and increasing the spacer length led to the significant loss in the NOD2 activity. It was also observed that the esterification of D-glutamic acid at both α as well as γ position with medium sized alcohols resulted in the improved activity. Whereas, sharp drop in the activity was observed with long chain esters as well as hydrolysis of both the esters to the carboxylic acid functionalities.
3.9. Mannosylated Desmuramylpeptides
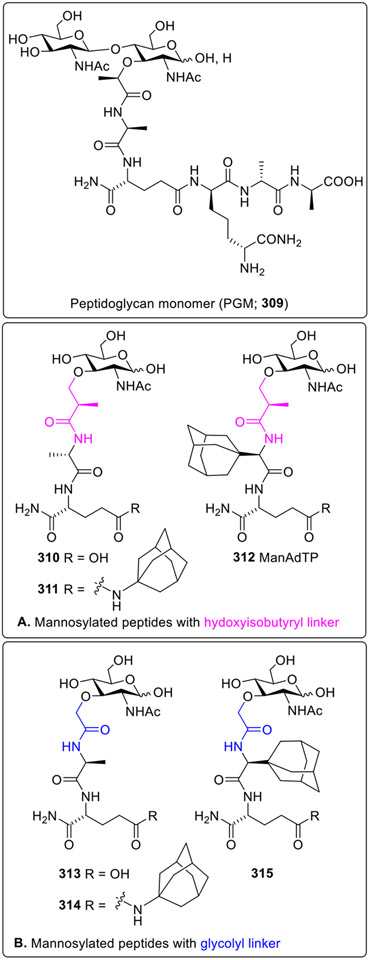
Ribic et al. synthesized two series of mannosylated desmuramylpeptides wherein mannose was coupled to dipeptides containing lipophilic adamantane on N- or C-terminus through a glycolyl or hydroxy isobutyryl linker [78,79]. The adjuvant activities of synthesized compounds were investigated in the mouse model using ovalbumin as an antigen. Their activities were compared to peptidoglycan monomer (PGM, 309, Fig. 26). In the in vivo experiments, analogs with a glycolyl linker (313-315) showed higher adjuvant activity than the analogs with a hydroxy isobutyryl linker (310-312) indicating that the introduction of the glycolyl moiety plays a significant role in the stimulation of the immune response. In particular, compound 315 was identified as the most potent adjuvant in this class of mannosylated desmuramylpeptides. It significantly enhanced the production of specific antibodies, particularly IgG, when compared to the antigen alone or commonly used adjuvants. In both series, the introduction of the bulky and lipophilic adamantane did not affect activity. Also, it was interesting to observe that the adjuvant activity changes with respect to the position of adamantane moiety. The stimulation was higher in the compounds containing the adamant-1-yl moiety at the amino acid residue in the first amino acid (312 and 315) compared to the analogs containing adamantan-1-yl-carboxymide at the γ-position of the second amino acid (311 and 314). Overall, it can be concluded that specific compounds, particularly those with a glycolyl linker significantly enhanced immune responses in mice, especially regarding antibody production against the antigen ovalbumin (OVA).
Fig. 26.
Mannosylated desmuramylpeptides.
3.10. Peptidoglycan fragment library containing two types of glycan sequence
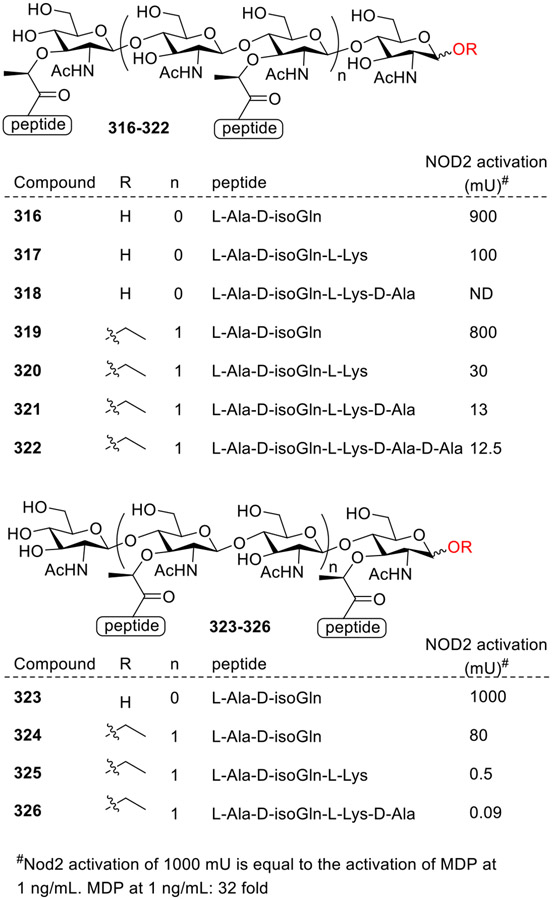
Wang et al. synthesized a peptidoglycan fragment library containing two types of glycan sequences: Mur-NAc-Glc-NAc (MG, Fig. 27) and Glc-NAc-Mur-NAc (GM) [80-82] and tested for their ability to activate hNOD2. They focused on disaccharides and tetrasaccharides with different peptide chains. Among both the sequences, the disaccharides with dipeptides, 316 and 323, showed potent activities comparable to MDP. In case of tetrasaccharides, compounds with dipeptide (L-Ala-D-isoGln) 319 and 324 exhibited stronger activities than the same glycan sequence groups with longer peptide chains. Interestingly, differences in the glycan sequence significantly influenced hNOD2 activation, with MurNAc-GlcNAc (MG) sequences showing higher potency than GlcNAc-MurNAc (GM) sequences as can 319 showed 10-fold higher activity than 324; 320 had an approximately 65-fold higher activity than 325; and 321 had an approximately 26-fold higher activity than 326. It can be concluded that the glycan sequence within PGN fragments plays a crucial role in influencing hNOD2 activation, suggesting potential implications for understanding bacterial PGN degradation and its impact on immune responses in humans.
Fig. 27.
Peptidoglycan fragment library containing two types of glycan sequences.
3.11. MDP-antigen conjugates
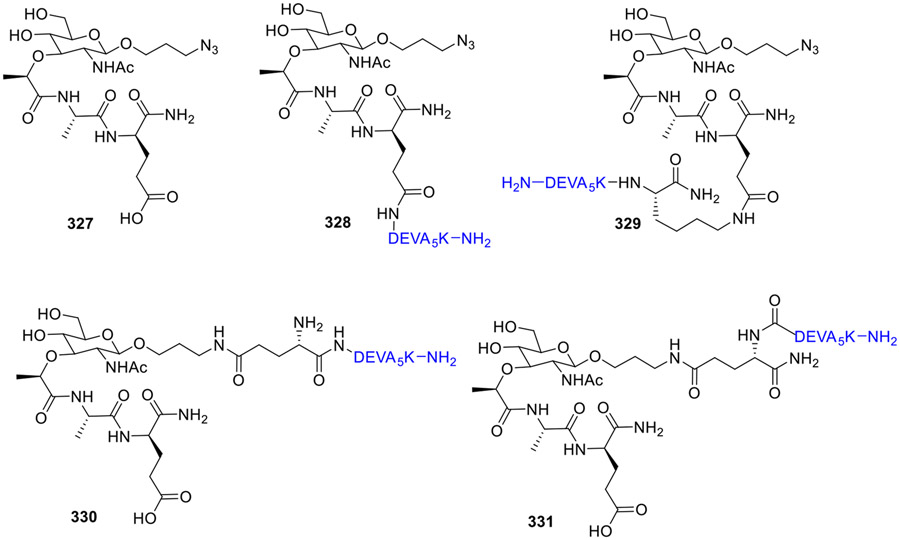
Based on the immunostimulatory properties of lipophilic MDP analogs, Williams el al. synthesized MDP-antigen conjugates [83]. The NOD2 activating capacity of these conjugates were then determined using NOD2 transfected HEK293 cells. The MDP analog 327 was used as reference, while the antigen DEVA5K [84] and the Pam3Cys-antigen conjugate were used as negative controls. The compound 327 (Fig. 28) showed a substantial amount of IL-8 production, confirming that the β-azidopropanol modification on the anomeric position is allowed. Conjugates 330 and 331 induced interleukin 8 (IL-8) production similarly to the active MDP derivative 327, indicating their ability to stimulate NOD2. However, conjugates 328 and 329 showed diminished activity. The position of attachment of the NOD2 ligand to the antigenic peptide was crucial, as conjugation at the GlcNAc anomeric center of MDP (conjugates 330 and 331) proved more favorable than attachment to the side chain of the L-isoglutamic acid residue (conjugates 328 and 329).
Fig. 28.
Muramyl dipeptide antigen conjugates.
3.12. MDP-conjugates with biomolecules and small molecule drugs
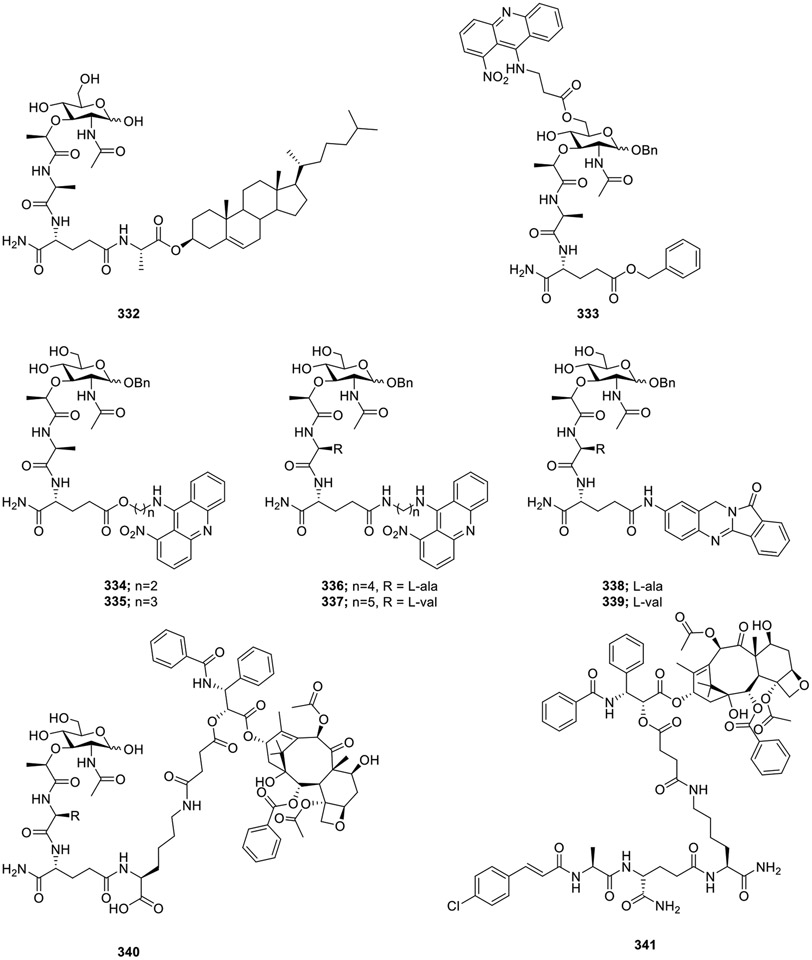
Various conjugates of MDP analogs with biomolecules and other small-molecule drugs have been synthesized. The conjugation of MDP-L-Ala with cholesterol resulted in a lipophilic derivative 332 (Fig. 29) [85][86]. The liposomal 332 with 1,2-distearoyl-sn-glycero-3-phosphocholine:polystyrene (7:3 molar ratio) induced cytotoxic activity of mouse peritoneal macrophages against P815 mastocytoma cells. It was interesting to observe that the treatment with free MDP even at 50 μg/mL concentration had no effect. Overall, 332 was about 7400-fold more active than free MDP. Dzierzbicka et al. conjugated MDP analogs with small molecule drugs like acridine and acridone derivatives (333-339) [87]. Among these, compound 333 stimulated the cytotoxic activity of NK cells, with compounds 334-337 induced cytotoxic activity against several human cell lines. Moreover, compounds 334, 336, and 337 were also active in vivo in a hollow fiber assay, and compound 335 showed in vivo activity against melanoma in mice. Several nor-MDPs were also conjugated to a heterocyclic aryl amine batracylin (338 and 339) [88]. These compounds inhibited the proliferation of melanoma cells. Furthermore, DMPs were also coupled to 3′-amino, 2′-hydroxyl, or 7-hydroxyl group of PTX forming 3′-N-MTC-01,2′-O-MTC-01, and 7-O-MTC-01. Among them, compound 2′-O-MTC-01 340 showed the most potent antitumor activity in vitro. Further experiments demonstrated the ability of compound 340 to induce expression and production of TNF-α and IL-12 [89]. To obtain analogs of MDP with antitumor and antimetastatic activities, compound 340 was further modified by replacing the muramic acid moiety by various aromatic groups, leading to MTC-220 (341) [90]. Compound 341 inhibited growth of various tumor cell lines but it was speculated that 341 might also act via TLR4 as it contains the PTX motif, which has been shown to bind to TLR4 receptors.
Fig. 29.
Muramyl dipeptide conjugates with biomolecules and small molecule drugs.
3.13. Muropeptides
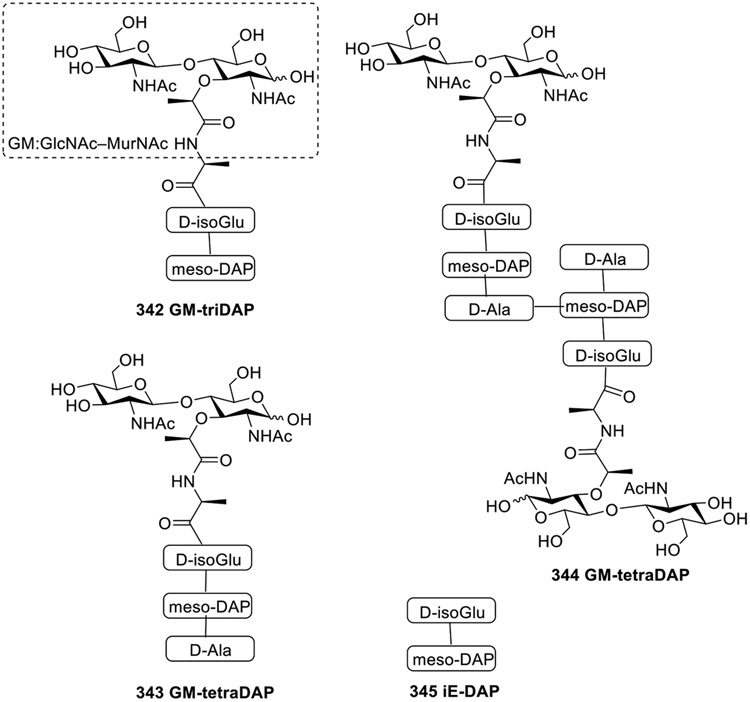
Dagil et al. had shown that there are certain muropeptides from Gram-negative bacteria that can activate both NOD1 and NOD2 receptors [91]. Various muropeptides such as GM-triDAP containing meso-DAP at the end was previously viewed as specific NOD1 agonist. This structure triggers the NOD1 receptor, leading to immune responses. So, to better understand the interactions of such meso-DAP containing compounds Dagil and coworkers developed a model using human cells with modified NOD1 and NOD2 genes, allowing the study of muropeptide reactions without interference from other receptors. The newly developed model showed that these muropeptides with meso-DAP, like GM-triDAP 342, GM-tetraDAP 343, and diGM-tetraDAP 344, can activate both NOD1 and NOD2, but they might need to be preprocessed into specific forms for activation. However, the iE-DAP 345 did not show any interaction with NOD2. Different lengths and complexities of peptide chains in NOD2-agonistic muropeptides appear to be recognized without requiring peptidase pre-processing, indicating a less stringent requirement compared to NOD1 activation. Excessive negative charge at the C-terminus interferes with NOD2 recognition, highlighting the sensitivity of NOD2 to the charge distribution within the muropeptide. Overall, it is concluded that the dual action of muropeptides on both NOD1 and NOD2 should be considered when studying immune responses or creating vaccines.
4. Conclusion
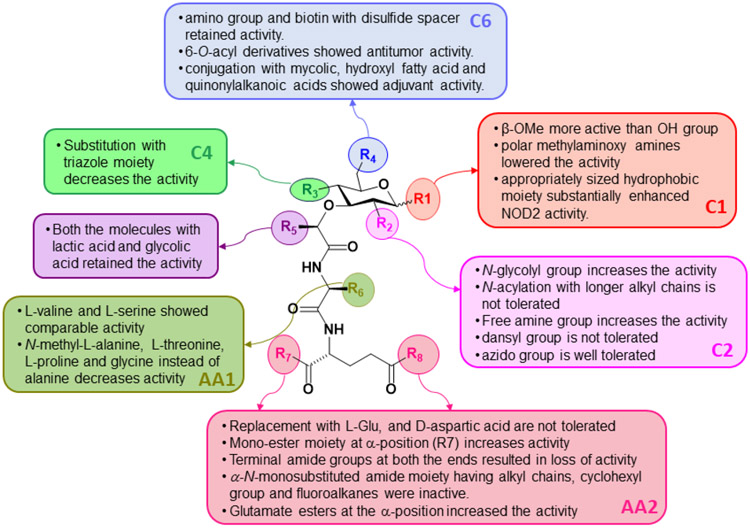
To date, a large number of MDP derivatives have been synthesized, among which several compounds showed promising activity when compared to MDP. It was observed that l-configuration of the first amino acid i.e., l-alanine and d-configuration of the glutamic acid residue of second amino acid (d-isoglutamine) are essential for maximum NOD2 activity. The modification at the C2 position of carbohydrate moiety also play an important role in NOD2 activity. Compounds with NH2 group at the C2 position along with β-OMe at C1 position of carbohydrate moiety showed potent activity, although its salt form showed no activity. Also, the replacement of N-acetyl group with N-glycolyl improved activity. Analogs in which the hydroxy group at the C1 position of carbohydrate moiety was substituted with the methoxy group had higher activity than N-acetyl MDP. Compounds with the O-benzyl moiety at the C1 axial position lost activity suggesting that the orientation and substituent at the C1 position play an important role in determining the NOD2 activity.
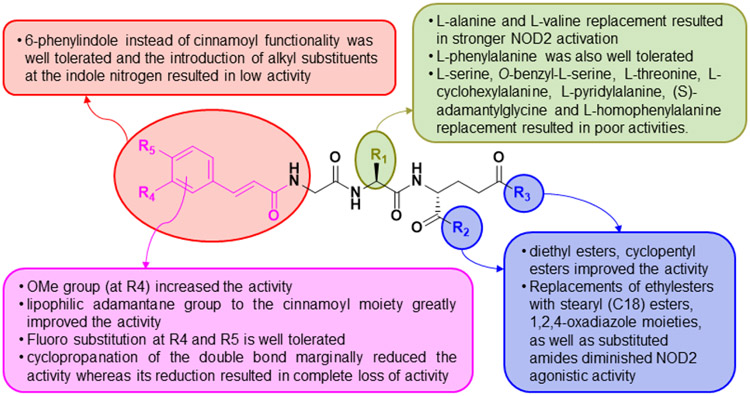
Compounds bearing successively longer alkyl chains (C5-C11) at the equatorial C1 position, showed similar activity to N-glycolyl MDP. Dramatic loss in the activity was observed for the compounds with substituted triazole moiety at the C4 position. C6 amino version of MDP also retained the activity and the compounds containing biotin moiety with disulfide bond as a cleavable linker reserved the activity. When the first amino acid was replaced by glycine, l-threonine, l-serine, l-proline, l-cyclohexylalanine, (S)-adamantyl glycine, l-homophenylalanine, and l-pyridylalanine, activity was reduced. However, substitution with valine and phenylalanine retained activity. Substituting a methyl ester for the amide of the d-isoGln residue generated Glu-MDP(D-G1u)-OCH3, which had greater activity than MDP. Substituted cinnamic acid moiety with 4-OH and 3-OMe groups could replace N-acetyl muramic acid in desmuramylpeptides without loss of activity. Introduction of two ethyl esters, a bulkier cyclopentyl ester on carboxylic acid of d-glutamic acid of desmuramylpeptide improved activity. However, two bulkier stearyl (C18) groups led to diminished activity.
Similar effects on activity were observed when phenolic hydroxyl of the ferulic acid amide moiety was functionalized with the bulkier stearyl (C18) through esterification. However, attachment of the lipophilic adamantane group to the cinnamoyl moiety greatly improved the activity. The literature clearly highlights many critical features of different substituents or derivatives of MDP which contribute to their ability to activate NOD2, which may hopefully assist in the design of further modifications to improve NOD2 agonist activity. However, a complete understanding of how these modifications affect NOD2 activity awaits the determination of their actual binding mode to the NOD2 structure, either through the determination of a crystal structure of a NOD2-MDP complex or through more detailed in silico molecular modelling studies.
Fig. 30.
Muropeptides containing meso-DAP.
Fig. 31.
SAR in muramyl dipeptides.
Fig. 32.
SAR in desmuramylpeptides.
Highlights.
Muramyl dipeptide (MDP) is the minimal peptidoglycan structure that activates NOD2
A detailed SAR on MDP analogs is elaborated
Desmuramylpeptides lacking sugar moiety are emerging as new class of NOD2 agonists
Bioconjugation approach is explored to strengthen the immune system against tumor
Acknowledgements
The project was supported by funding from National Institute of Allergy and Infectious Diseases of the National Institutes of Health under Contracts HHS-N272201400053C and HHS-N272200800039C. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. This work was also supported by Australian Government Global Connections Fund Bridging Grant 511803900. DBS and NP are thankful to MHRD’s Scheme for Promotion of Academic and Research Collaboration (SPARC/2018-2019/P385/SL) program of Govt. of India which aims to facilitate academic and research collaborations between Panjab University, Chandigarh and Flinders University, Adelaide, South Australia. DBS is thankful to Science and Engineering Research Board (SERB), New Delhi for the Core Research Grant (CRG/2021/005467) and Indian Council of Medical Research (6/9-7(299)/2022-ECD-II), New Delhi for the ad-hoc research project. MTP is thankful to DST for the award of Women Scientist Scheme-A [SR/WOS-A/CS-132/2016 (G)]. AK is thankful to UGC, New Delhi, for the award of Junior and Senior Research Fellowships.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- [1].Kaur A, Rathee J, Kanwar R, Kaushik D, Salunke DB, Mehta SK, TLR2 agonistic lipopeptide enriched PLGA nanoparticles as combinatorial drug delivery vehicle, Colloids Surfaces A Physicochem. Eng. Asp 647 (2022) 129084. 10.1016/j.colsurfa.2022.129084. [DOI] [Google Scholar]
- [2].Kaushik D, Dhingra S, Patil MT, Piplani S, Khanna V, Honda-Okubo Y, Li L, Fung J, Sakala IG, Salunke DB, Petrovsky N, BBIQ, a pure TLR7 agonist, is an effective influenza vaccine adjuvant, Hum. Vaccines Immunother 16 (2020) 1989–1996. 10.1080/21645515.2019.1710409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Kaur A, Baldwin J, Brar D, Salunke DB, Petrovsky N, Toll-like receptor (TLR) agonists as a driving force behind next-generation vaccine adjuvants and cancer therapeutics, Curr. Opin. Chem. Biol 70 (2022) 102172. 10.1016/j.cbpa.2022.102172. [DOI] [PubMed] [Google Scholar]
- [4].Singleton KL, Joffe A, Leitner WW, Review: Current trends, challenges, and success stories in adjuvant research, Front. Immunol 14 (2023) 1–9. 10.3389/fimmu.2023.1105655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Janeway CA, Medzhitov R, Innate immune recognition, Annu. Rev. Immunol 20 (2002) 197–216. 10.1146/annurev.immunol.20.083001.084359. [DOI] [PubMed] [Google Scholar]
- [6].Akira S, Uematsu S, Takeuchi O, Pathogen recognition and innate immunity, Cell. 124 (2006) 783–801. 10.1016/j.cell.2006.02.015. [DOI] [PubMed] [Google Scholar]
- [7].Medzhitov R, Recognition of microorganisms and activation of the immune response, Nature. 449 (2007) 819–826. 10.1038/nature06246. [DOI] [PubMed] [Google Scholar]
- [8].Kaushik D, Kaur A, Petrovsky N, Salunke DB, Structural evolution of toll-like receptor 7/8 agonists from imidazoquinolines to imidazoles, RSC Med. Chem 12 (2021) 1065–1120. 10.1039/d1md00031d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Kaur A, Kannan D, Mehta SK, Singh S, Salunke DB, Synthetic Toll-like receptor agonists for the development of powerful malaria vaccines: a patent review, Expert Opin. Ther. Pat 28 (2018) 837–847. 10.1080/13543776.2018.1530217. [DOI] [PubMed] [Google Scholar]
- [10].Kaur A, Kaushik D, Piplani S, Mehta SK, Petrovsky N, Salunke DB, TLR2 Agonistic Small Molecules: Detailed Structure-Activity Relationship, Applications, and Future Prospects, J. Med. Chem 64 (2021) 233–278. 10.1021/acs.jmedchem.0c01627. [DOI] [PubMed] [Google Scholar]
- [11].Nielsen AE, Hantho JD, Mancini RJ, Synthetic agonists of NOD-like, RIG-I-like, and C-type lectin receptors for probing the inflammatory immune response, Future Med. Chem 9 (2017) 1345–1360. 10.4155/fmc-2017-0101. [DOI] [PubMed] [Google Scholar]
- [12].Li D, Wu M, Pattern recognition receptors in health and diseases, Signal Transduct. Target. Ther 6 (2021) 1–24. 10.1038/s41392-021-00687-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Jakopin Ž, Gobec M, Mlinarič-Raščan I, Sollner Dolenc M, Immunomodulatory properties of novel nucleotide oligomerization domain 2 (Nod2) agonistic desmuramyldipeptides, J. Med. Chem 55 (2012) 6478–6488. 10.1021/jm300503b. [DOI] [PubMed] [Google Scholar]
- [14].Hsu LC, Ali SR, McGillivray S, Tseng PH, Mariathasan S, Humke EW, Eckmann L, Powell JJ, Nizet V, Dixit VM, Karin M, A NOD2-NALP1 complex mediates caspase-1-dependent IL-1β secretion in response to Bacillus anthracis infection and muramyl dipeptide, Proc. Natl. Acad. Sci. U. S. A 105 (2008) 7803–7808. 10.1073/pnas.0802726105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Motta V, Soares F, Sun T, Philpott DJ, Nod-like receptors: Versatile cytosolic sentinels, Physiol. Rev 95 (2015) 149–178. 10.1152/physrev.00009.2014. [DOI] [PubMed] [Google Scholar]
- [16].Zhong Y, Kinio A, Saleh M, Functions of NOD-Like Receptors in Human Diseases, Front. Immunol 4 (2013) 1–18. 10.3389/fimmu.2013.00333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Nabergoj S, Mlinarič-Rašč;an I, Jakopin Ž, Harnessing the untapped potential of nucleotide-binding oligomerization domain ligands for cancer immunotherapy, Med. Res. Rev 39 (2019) 1447–1484. 10.1002/med.21557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Schroder K, Tschopp J, The Inflammasomes, Cell. 140 (2010) 821–832. 10.1016/j.cell.2010.01.040. [DOI] [PubMed] [Google Scholar]
- [19].Li R, Guo M, Lin J, Chai T, Wei L, Molecular cloning, characterization, and anti-avian pathogenic escherichia coli innate immune response of the Cherry Valley Duck CIITA Gene, Front. Microbiol 8 (2017) 1–11. 10.3389/fmicb.2017.01629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Kim YK, Shin JS, Nahm MH, NOD-like receptors in infection, immunity, and diseases, Yonsei Med. J 57 (2016) 5–14. 10.3349/ymj.2016.57.1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Geddes K, Magalhães JG, Girardin SE, Unleashing the therapeutic potential of NOD-like receptors, Nat. Rev. Drug Discov 8 (2009) 465–479. 10.1038/nrd2783. [DOI] [PubMed] [Google Scholar]
- [22].Proell M, Riedl SJ, Fritz JH, Rojas AM, Schwarzenbacher R, The Nod-Like Receptor (NLR) family: A tale of similarities and differences, PLoS One. 3 (2008) 1–11. 10.1371/journal.pone.0002119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Bertsche U, Mayer C, Götz F, Gust AA, Peptidoglycan perception-Sensing bacteria by their common envelope structure, Int. J. Med. Microbiol 305 (2015) 217–223. 10.1016/j.ijmm.2014.12.019. [DOI] [PubMed] [Google Scholar]
- [24].Girardin SE, Boneca IG, Viala J, Chamaillard M, Labigne A, Thomas G, Philpott DJ, Sansonetti PJ, Nod2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection, J. Biol. Chem 278 (2003) 8869–8872. 10.1074/jbc.C200651200. [DOI] [PubMed] [Google Scholar]
- [25].Chamaillard M, Stephen E G., Jérôme V, Philpott D, Cellular Microbiology J - 2003 - Chamaillard - Nods Nalps and Naip intracellular regulators of bacterial-induced.pdf, (2003). https://doi.org/doi: 10.1046/j.1462-5822.2003.00304.x. [DOI] [PubMed] [Google Scholar]
- [26].Kim J, Yang YL, Jang YS, Human β-defensin 2 is involved in CCR2-mediated Nod2 signal transduction, leading to activation of the innate immune response in macrophages, Immunobiology. 224 (2019) 502–510. 10.1016/j.imbio.2019.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Sabbah A, Chang TH, Hamack R, Frohlich V, Tominaga K, Dube PH, Xiang Y, Bose S, Activation of innate immune antiviral responses by Nod2, Nat. Immunol 10 (2009) 1073–1080. 10.1038/ni.1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Yamamoto S, Ma X, Role of Nod2 in the development of Crohn’s disease, Microbes Infect. 11 (2009)912–918. 10.1016/j.micinf.2009.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Wouters CH, Maes A, Foley KP, Bertin J, Rose CD, Blau Syndrome, the prototypic auto-inflammatory granulomatous disease, Pediatr. Rheumatol 12 (2014) 1–9. 10.1186/1546-0096-12-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Gobec M, Mlinarič-Raščan I, Dolenc MS, Jakopin Z, Structural requirements of acylated Gly- l -Ala- d -Glu analogs for activation of the innate immune receptor NOD2, Eur. J. Med. Chem 116 (2016) 1–12. 10.1016/j.ejmech.2016.03.030. [DOI] [PubMed] [Google Scholar]
- [31].Gobec M, Tomašič T, Štimac A, Frkanec R, Trontelj J, Anderluh M, Mlinarič-Raščan I, Jakopin Ž, Discovery of nanomolar desmuramylpeptide agonists of the innate immune receptor nucleotide-binding oligomerization domain-containing protein 2 (NOD2) possessing immunostimulatory properties, J. Med. Chem 61 (2018) 2707–2724. 10.1021/acs.jmedchem.7b01052. [DOI] [PubMed] [Google Scholar]
- [32].Guzelj Ž, Bizjak Š, S., & , Jakopin, Discovery of Desmuramylpeptide NOD2 Agonists with Single-Digit Nanomolar Potency, ACS Med. Chem. Lett 13 (2022) 1270–1277. 10.1021/acsmedchemlett.2c00121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Guzelj S, Nabergoj S, Gobec M, Pajk S, Klančič V, Slütter B, Frkanec R, Štimac A, Šket P, Plavec J, Mlinarič-Raščan I, Jakopin Ž, Structural Fine-Tuning of Desmuramylpeptide NOD2 Agonists Defines Their In Vivo Adjuvant Activity, J. Med. Chem 64 (2021) 7809–7838. 10.1021/acs.jmedchem.1c00644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Moreira LO, El Kasmi KC, Smith AM, Finkelstein D, Fillon S, Kim YG, Núñez G, Tuomanen E, Murray PJ, The TLR2-MyD88-NOD2-RIPK2 signalling axis regulates a balanced pro-inflammatory and IL-10-mediated anti-inflammatory cytokine response to Gram-positive cell walls, Cell. Microbiol 10 (2008) 2067–2077. 10.1111/j.1462-5822.2008.01189.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Negroni L, Pierdomenico A, Cucchiara M, Stronati S, NOD2 and inflammation: current insights, J. Inflamm. Res 11 (2018) 49–60. 10.2147/JIR.S137606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Strober W, Watanabe T, NOD2, an intracellular innate immune sensor involved in host defense and Crohn’s disease, Mucosal Immunol. 4 (2011) 484–495. 10.1038/mi.2011.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Strober W, Murray PJ, Kitani A, Watanabe T, Signalling pathways and molecular interactions of NOD1 and NOD2, Nat. Rev. Immunol 6 (2006) 9–20. 10.1038/nri1747. [DOI] [PubMed] [Google Scholar]
- [38].Caruso R, Warner N, Inohara N, Núñez G, NOD1 and NOD2: Signaling, host defense, and inflammatory disease, Immunity. 41 (2014) 898–908. 10.1016/j.immuni.2014.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Wang D, NOD1 and NOD2 Are Potential Therapeutic Targets for Cancer Immunotherapy, Comput. Intell. Neurosci 2022 (2022). 10.1155/2022/2271788. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- [40].Jakopin Ž, Nucleotide-binding oligomerization domain (NOD) inhibitors: A rational approach toward inhibition of NOD signaling pathway, J. Med. Chem 57 (2014) 6897–6918. 10.1021/jm401841p. [DOI] [PubMed] [Google Scholar]
- [41].Maekawa S, Ohio U, Shibata T, Miyake K, Shimizu T, Crystal structure of NOD2 and its implications in human disease, (2016) 4–6. 10.1038/ncomms11813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Lauro ML, D’Ambrosio EA, Bahnson BJ, Grimes CL, Molecular Recognition of Muramyl Dipeptide Occurs in the Leucine-rich Repeat Domain of Nod2, ACS Infect. Dis 3 (2017) 264–270. 10.1021/acsinfecdis.6b00154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].D’Ambrosio EA, Bersch KL, Lauro ML, Grimes CL, Grimes CL, Differential Peptidoglycan Recognition Assay Using Varied Surface Presentations, J. Am. Chem. Soc 142 (2020) 10926–10930. 10.1021/jacs.0c03933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Tigno-Aranjuez J, Abbott DW, Ubiquitination and phosphorylation in the regulation of NOD2 signaling and NOD2-mediated disease, Biochim. Biophys. Acta 1823 (2012) 2022–2088. 10.1016/j.bbamcr.2012.03.017.Ubiquitination. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Hancock REW, Nijnik A, Philpott DJ, Modulating immunity as a therapy for bacterial infections, Nat. Rev. Microbiol 10 (2012) 243–254. 10.1038/nrmicro2745. [DOI] [PubMed] [Google Scholar]
- [46].Meyers PA, Muramyl tripeptide (mifamurtide) for the treatment of osteosarcoma, Expert Rev. Anticancer Ther 9 (2009) 1035–1049. 10.1586/ERA.09.69. [DOI] [PubMed] [Google Scholar]
- [47].Kager L, Pötschger, Bielack, Review of mifamurtide in the treatment of patients with osteosarcoma, Ther. Clin. Risk Manag (2010) 279. 10.2147/tcrm.s5688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Ichiro A, Development of the cytokine inducer romurtide: experimental studies and clinical application, Trends Pharmacol. Sci 13 (1992) 425–428. 10.1016/0165-6147(92)90134-R. [DOI] [PubMed] [Google Scholar]
- [49].Nigro G, Rossi R, Commere PH, Jay P, Sansonetti PJ, The cytosolic bacterial peptidoglycan sensor Nod2 affords stem cell protection and links microbes to gut epithelial regeneration, Cell Host Microbe. 15 (2014) 792–798. 10.1016/j.chom.2014.05.003. [DOI] [PubMed] [Google Scholar]
- [50].Rubino SJ, Magalhaes JG, Philpott D, Bahr GM, Blanot D, Girardin SE, Identification of a synthetic muramyl peptide derivative with enhanced Nod2 stimulatory capacity, Innate Immun. 19 (2013) 493–503. 10.1177/1753425912471691. [DOI] [PubMed] [Google Scholar]
- [51].Cheng WC, You TY, Teo ZZ, Sayyad AA, Maharana J, Guo CW, Liang PH, Lin CS, Meng FC, Further Insights on Structural Modifications of Muramyl Dipeptides to Study the Human NOD2 Stimulating Activity, Chem. - An Asian J 15 (2020) 3836–3844. 10.1002/asia.202001003. [DOI] [PubMed] [Google Scholar]
- [52].Chen KT, Huang DY, Chiu CH, Lin WW, Liang PH, Cheng WC, Synthesis of Diverse N-Substituted Muramyl Dipeptide Derivatives and Their Use in a Study of Human NOD2 Stimulation Activity, Chem. - AEur. J 21 (2015) 11984–11988. 10.1002/chem.201501557. [DOI] [PubMed] [Google Scholar]
- [53].Reddy PRS, Sambyal S, Mhamane TB, Sravanthi V, Shafi S, Khan IA, Sampath Kumar HM, Synthesis and biological evaluation of novel 2-azido muramyl dipeptide as NOD2 agonistic adjuvants, Bioorganic Med. Chem 66 (2022) 116781. 10.1016/j.bmc.2022.116781. [DOI] [PubMed] [Google Scholar]
- [54].Bertin J, Nir WJ, Fischer CM, Tayber OV, Errada PR, Grant JR, Keilty JJ, Gosselin ML, Robison KE, Wong GHW, Glucksmann MA, DiStefano PS, Human CARD4 protein is a novel CED-4/Apaf-1 cell death family member that activates NF-κB, J. Biol. Chem 274 (1999) 12955–12958. 10.1074/jbc.274.19.12955. [DOI] [PubMed] [Google Scholar]
- [55].Melnyk JE, Mohanan V, Schaefer AK, Hou CW, Grimes CL, Peptidoglycan Modifications Tune the Stability and Function of the Innate Immune Receptor Nod2, J. Am. Chem. Soc 137 (2015) 6987–6990. 10.1021/jacs.5b01607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Grimes CL, Podolsky DK, O’Shea EK, Synthesis of biologically active biotinylated muramyl dipeptides, Bioorganic Med. Chem. Lett 20 (2010) 6061–6063. 10.1016/j.bmcl.2010.08.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Saiki I, Tanio Y, Yamawaki M, Uemiya M, Kobayashi S, Fukuda T, Yukimasa H, Yamamura Y, Azuma I, Adjuvant activities of quinonyl-N-acetyl muramyl dipeptides in mice and guinea pigs, Infect. Immun 31 (1981) 114–121. 10.1128/iai.31.1.114-121.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Kataoka T, Kinomoto M, Takegawa M, Tokunaga T, Effect of a synthetic adjuvant for inducing anti-tumour immunity, Vaccine. 9 (1991) 300–302. 10.1016/0264-410X(91)90054-A. [DOI] [PubMed] [Google Scholar]
- [59].Kataoka T, Tokunaga T, A synthetic adjuvant effective in inducing antitumor immunity, Jpn. J. Cancer Res 79 (1988) 817–20. 10.1111/j.1349-7006.1988.tb00042.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Hasegawa I, Kaneda A, Amano Y, Kiso M, Azuma M, A Facile Synthesis of N-Acetylmuramyl-L-alanyl-D-isoglutamine and Its Carbohydrate Analogs, and Their Immunoadjuvant Activities., Agric. Biol. Chem 42 (1978) 2187–2189. 10.1271/bbb1961.42.2187. [DOI] [Google Scholar]
- [61].Uemiya M, Sugimura K, Kusama T, Saiki I, Yamawaki M, Azuma I, Yamamura Y, Adjuvant activity of 6-O-mycoloyl derivatives of N-acetylmuramyl-L-seryl-D-isoglutamine and related compounds in mice and guinea pigs, Infect. Immun 24 (1979) 83–89. 10.1128/iai.24.1.83-89.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Kusumoto S, Inage M, Shiba T, Azuma I, Yamamura Y, Synthesis of long chain fatty acid esters of N-acetylmuramyl-L-alanyl-D-isoglutamine in relation to antitumor activity, Tetrahedron Lett. 19 (1978) 4899–4902. 10.1016/S0040-4039(01)85765-8. [DOI] [Google Scholar]
- [63].Yoo Yung Choon, Saiki I, Sato K, Azuma I, B30-MDP, a synthetic muramyl dipeptide derivative for tumour vaccination to enhance antitumour immunity and anti metastatic effect in mice, Vaccine. 10 (1992) 792–797. 10.1016/0264-410X(92)90515-L. [DOI] [PubMed] [Google Scholar]
- [64].Parant MA, Audibert FM, Chedidl LA, Level MR, Lefrancier PL, Choay JP, Lederer E, Immunostimulant Activities of a Lipophilic Muramyl Dipeptide Derivative and of Desmuramyl Peptidolipid Analogs, 27 (1980) 826–831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [65].Lefrancier P, Derrien M, Jamet X, Choay J, Lederer E, Audibert F, Parant M, Parant F, Chedid L, Apyrogenic, Adjuvant-Active N-Acetylmuramyl-dipeptides, J. Med. Chem 25 (1982) 87–90. 10.1021/jm00343a018. [DOI] [PubMed] [Google Scholar]
- [66].Lefrancier P, Choay J, Derrien M, Lederman I, Synthesis of N-acetyl-muramyl-L-alanyl-D-isoglutamine, an adjuvant of the immune response, and of some n-acetyl-muramyl-peptide analogs. Int J Pept Protein Res. 9 (1977), 249–257. 10.1111/j.1399-3011.1977.tb03488.x [DOI] [PubMed] [Google Scholar]
- [67].Chedid L, Audibert F, Lefrancier P, Choay J, Lederer E, Modulation of the immune response by a synthetic adjuvant and analogs, Proc. Natl. Acad. Sci. U. S. A 73 (1976) 2472–2475. 10.1073/pnas.73.7.2472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Chedid L, Parant M, Parant F, Lefrancher P, Choay J, Lederer E, Enhancement of nonspecific immunity to Klebsiella pneumoniae infection by a synthetic immunoadjuvant (N-acetylmuramyl-L-alanyl-D-isoglutamine) and several analogs, Proc. Natl. Acad. Sci. U. S. A 74 (1977) 2089–2093. 10.1073/pnas.74.5.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Lefrancier P, Derrien M, Lederman I, Nief F, Choay J, Lederer E, Synthesis of some new analogs of the immunoadjuvant glycopeptide MDP (N-acetyl-muramyl-L-alanyl-D-isoglutamine), Int J Pept Protein Res. 11 (1978), 289–296. 10.1111/j.1399-3011.1978.tb02851.x. [DOI] [PubMed] [Google Scholar]
- [70].Mhamane TB, Sambyal S, Vemireddy S, Design , synthesis and biological evaluation of novel lipophilic 2 , 5-disub - stituted tetrazole analogues of muramyl dipeptide as NOD2 agonists, Bioorganic Med. Chem 88–89 (2023) 117296. 10.1016/j.bmc.2023.117296. [DOI] [PubMed] [Google Scholar]
- [71].Cai B, Panek JS, Amar S, Convergent synthesis of novel muramyl dipeptide analogues: Inhibition of porphyromonas gingivalis-induced pro-inflammatory effects by high doses of muramyl dipeptide, J. Med. Chem 59 (2016) 6878–6890. 10.1021/acs.jmedchem.6b00681. [DOI] [PubMed] [Google Scholar]
- [72].Kikelj D, Pečar S, Kotnik V, Štalc A, Wraber-Herzog B, Simčič S, Ihan A, Klamfer L, Povšič L, Grahek R, Suhadolc E, Hočevar M, Hönig H, Rogi-Kohlenprath R, N-{trans-2-[[2’-(acetylamino)cyclohexyl]oxy]acetyl}-L-alanyl-D-glutamic acid: A novel immunologically active carbocyclic muramyl dipeptide analogue, J. Med. Chem 41 (1998) 530–539. 10.1021/jm970509d. [DOI] [PubMed] [Google Scholar]
- [73].Zhao N, Ma Y, Zhang S, Fang X, Liang Z, Liu G, New muramyl dipeptide (MDP) mimics without the carbohydrate moiety as potential adjuvant candidates for a therapeutic hepatitis B vaccine (HBV), Bioorganic Med. Chem. Lett 21 (2011) 4292–4295. 10.1016/j.bmcl.2011.05.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Chen YZ, Liu G, Senju S, Wang Q, Irie A, Haruta M, Matsui M, Yasup F, Kohara M, Nishimura Y, Identification of SARS-COV spike protein-derived and HLA-A2-restricted human CTL epitopes by using a new muramyl dipeptide-derivative adjuvant, Int. J. Immunopathol. Pharmacol 23 (2010) 165–177. 10.1177/039463201002300115. [DOI] [PubMed] [Google Scholar]
- [75].Yang HZ, Xu S, Liao XY, Zhang SD, Liang ZL, Liu BH, Bai JY, Jiang C, Ding J, Cheng GF, Liu G, A novel immunostimulator, N2-[α-O-Benzyl-N-(acetylmuramyl) -L-alanyl-D-isoglutaminyl]-N6-trans-(m-nitrocinnamoyl)-L-lysine, and its adjuvancy on the hepatitis B surface antigen. Journal of Medicinal Chemistry, 48 (2005), 5112–5122. 10.1021/jm0493313 [DOI] [PubMed] [Google Scholar]
- [76].Khan FA, Ulanova M, Bai B, Yalamati D, Jiang ZH, Design, synthesis and immunological evaluation of novel amphiphilic desmuramyl peptides, Eur. J. Med. Chem 141 (2017)26–36. 10.1016/j.ejmech.2017.09.070. [DOI] [PubMed] [Google Scholar]
- [77].Jakopin Ž, Corsini E, Gobec M, Mlinarič-Raščan I, Dolenc MS, Design, synthesis and biological evaluation of novel desmuramyldipeptide analogs, Eur. J. Med. Chem 46 (2011) 3762–3777. 10.1016/j.ejmech.2011.05.042. [DOI] [PubMed] [Google Scholar]
- [78].Ribić R, Stojković R, Milković L, Antica M, Cigler M, Tomić S, Design, synthesis and biological evaluation of immunostimulating mannosylated desmuramyl peptides, Beilstein J. Org. Chem 15 (2019) 1805–1814. 10.3762/bjoc.15.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [79].Ribić R, Habjanec L, Vranešić B, Frkanec R, Tomić S, Synthesis and biological evaluation of new mannose derived immunomodulating adamantyltripeptides, Croat. Chem. Acta 84 (2011) 233–244. 10.5562/cca1827. [DOI] [Google Scholar]
- [80].Wang N, Huang CY, Hasegawa M, Inohara N, Fujimoto Y, Fukase K, Glycan Sequence-Dependent Nod2 Activation Investigated by Using a Chemically Synthesized Bacterial Peptidoglycan Fragment Library, ChemBioChem. 14 (2013) 482–488. 10.1002/cbic.201200655. [DOI] [PubMed] [Google Scholar]
- [81].Kusumoto S, Yamamoto K, Imoto M, Chemical synthesis and biological activities of two disaccharide dipeptides corresponding to the repeating units of bacterial peptidoglycan, Bull. Chem. Soc. Jpn 59 (1986) 1411–1417. 10.1246/bcsj.59.1411. [DOI] [Google Scholar]
- [82].Inamura S, Fujimoto Y, Kawasaki A, Shiokawa Z, Woelk E, Heine H, Lindner B, Inohara N, Kusumoto S, Fukase K, Synthesis of peptidoglycan fragments and evaluation of their biological activity, Org. Biomol. Chem 4 (2006) 232–242. 10.1039/b511866b. [DOI] [PubMed] [Google Scholar]
- [83].Willems MMJHP, Zom GG, Meeuwenoord N, Khan S, Ossendorp F, Overkleeft HS, Van Der Marel GA, Filippov DV, Codee JDC, Lipophilic Muramyl Dipeptide-Antigen Conjugates as Immunostimulating Agents, ChemMedChem. 11 (2016) 190–198. 10.1002/cmdc.201500196. [DOI] [PubMed] [Google Scholar]
- [84].Abehsira-Amar O, Uzan M, Audibert F, Jolivet M, Chedid L, Theze J, Covalent linkage of the synthetic adjuvant MDP to the synthetic polypeptide (T,G)-A-L changes the specificity of the immune response at the T and B cell level, Mol. Immunol 24 (1987) 945–951. 10.1016/0161-5890(87)90005-8. [DOI] [PubMed] [Google Scholar]
- [85].Yu WP, Barratt GM, Devissaguet JP, Puisieux F, Anti-metastatic activity in vivo of MDP-l-alanyl-cholesterol (MTP-Chol) entrapped in nanocapsules, Int. J. Immunopharmacol 13 (1991) 167–173. 10.1016/0192-0561(91)90095-O. [DOI] [PubMed] [Google Scholar]
- [86].Barratt G, Puisieux F, Yu WP, Foucher C, Fessi H, Devissaguet JP, Anti-metastic activity of MDP-L-alanyl-cholesterol incorporated into various types of nanocapsules, Int. J. Immunopharmacol 16 (1994) 457–461. 10.1016/0192-0561(94)90036-1. [DOI] [PubMed] [Google Scholar]
- [87].Dzierzbicka K, Kołodziejczyk AM, Synthesis and antitumor activity of conjugates of muramyldipeptide or normuramyldipeptide with hydroxyacridine/acridone derivatives, J. Med. Chem 46 (2003) 183–189. 10.1021/jm020991m. [DOI] [PubMed] [Google Scholar]
- [88].Trzonkowski P, Dzierzbicka K, Bociewicz J, Szmit E, Myśliwski A, Biological activity of conjugates of muramyl dipeptides with batracylin derivatives, Int. Immunopharmacol 5 (2005)241–251. 10.1016/j.intimp.2004.09.029. [DOI] [PubMed] [Google Scholar]
- [89].Li X, Yu J, Xu S, Wang N, Yang H, Yan Z, Cheng G, Liu G, Chemical conjugation of muramyl dipeptide and paclitaxel to explore the combination of immunotherapy and chemotherapy for cancer, Glycoconj. J 25 (2008) 415–425. 10.1007/s10719-007-9095-3. [DOI] [PubMed] [Google Scholar]
- [90].Ma Y, Zhao N, Liu G, Conjugate ( MTC-220 ) of Muramyl Dipeptide Analogue and Paclitaxel Prevents Both Tumor Growth and Metastasis in Mice, (2011) 2767–2777. [DOI] [PubMed] [Google Scholar]
- [91].Dagil YA, Arbatsky NP, Alkhazova BI, L’vov VL, Mazurov DV, Pashenkov MV, The dual NOD1/NOD2 agonism of muropeptides containing a mesodiaminopimelic acid residue, PLoS One. 11 (2016) 1–19. 10.1371/journal.pone.0160784. [DOI] [PMC free article] [PubMed] [Google Scholar]