Abstract
A man in his late 40s with no known past medical history was unresponsive for an unknown period of time. Crushed pills and white residue were found on a nearby table. On presentation he was obtunded and unresponsive to verbal commands but withdrawing to painful stimuli. The initial urine drug screen was negative, but a urine fentanyl screen was subsequently positive with a level of 137.3 ng/mL. MRI of the brain showed reduced diffusivity and fluid attenuated inversion recovery (FLAIR) hyperintensity symmetrically in the bilateral supratentorial white matter, cerebellum and globus pallidus. Alternative diagnoses such as infection were considered, but ultimately the history and workup led to a diagnosis of fentanyl-induced leukoencephalopathy. Three days after admission the patient became able to track, respond to voice and follow basic one-step commands. The patient does not recall the mechanism of inhalation. While there are case reports of heroin-induced leukoencephalopathy following inhaled heroin use and many routes of fentanyl, this is the first reported case of a similar phenomenon due to fentanyl inhalation.
Keywords: Adult intensive care, Neuroimaging, Neurological injury, Drug misuse (including addiction), Toxicology
Background
Toxic leukoencephalopathy (TLE) is an acute or chronic neurologic syndrome due to exposure to insults or toxins, resulting in damage to the cerebral white matter.1 The broader term ‘leukoencephalopathy’ refers to disorders of the white matter of the brain. The diagnosis of TLE involves clinical symptoms of white matter dysfunction that correlate with neuroradiologic white matter abnormalities. MRI is the preferred diagnostic modality as it offers better depiction of the brain parenchyma than CT. Characteristically, T2-weighted and diffusion-weighted imaging findings show diffusely increased signal intensities within the white matter.2 In patients with extensive toxin exposure, the amount of radiographic involvement is variable.
An extensive and diverse variety of signs and symptoms have been described. The most obvious clinical manifestations are neurological and behavioural changes, ranging from a mild confusion to stupor, coma and death.1
There have been numerous insults and toxins identified including chemotherapeutic agents such as carmustine and methotrexate, immunosuppressive drugs such as ciclosporin and tacrolimus, environmental toxins such as carbon monoxide, and drugs of misuse such as alcohol, cocaine and heroin.3 Glue, toluene and other volatile compounds that are inhaled have been reported to cause TLE.4–6
Prognosis and recovery generally depend on the degree of white matter injury; some patients recover fully while others may experience progressive decline.7 The mechanism of TLE is unclear but there are several proposals including endothelial injury, myelin sheath degradation or a combination of the two.1
In the context of this case with crushed pills and white residue reported, heroin-induced leukoencephalopathy also known as ‘chasing the dragon’ was considered. The inhalation of heroin vapours can lead to TLE.8 9 It is thought that the vapours lead to vacuolar degeneration of deep white matter. Numerous cases have been reported in the literature.10 This is the first case report known to document TLE secondary to fentanyl inhalation.
Case presentation
A previously healthy man in his late 40s with no significant past medical history was found unresponsive for an unknown period of time in his hotel room while attending a work conference. Unidentified crushed pills and a white residue were found on the table of his hotel room. The white powder was visible around his mouth. His only reported medications were sildenafil and vitamin D. On initial assessment his Glasgow Coma Scale was 10, he had dried vomitus around his mouth and scant red blood on his lips, but was protecting his airway. Emergency Medical Services administered naloxone without effect.
On hospital arrival, the patient was afebrile (36.7°C) with blood pressure 116/93 mmHg, heart rate 99 bpm, respiratory rate 38/min, with an oxygen saturation of 93% on a non-rebreather mask (15–20 L/min oxygen flow rate). Physical examination revealed scant dried blood on lips but no evidence of blood in the oral cavity. Pulmonary examination showed tachypnoea without signs of increased work of breathing and clear lung sounds. Neurologically, the patient’s level of consciousness was obtunded. He was non-verbal, not answering orientation questions and not following commands. Pupils were 4–5 mm bilaterally and reactive to light and there was no blink to threat bilaterally (CN II). His gaze was conjugate, oculocephalic reflex intact, and bilateral corneal reflexes intact (CN III, IV, VI). His face appeared symmetrical. He withdrew to pain in the bilateral lower extremities but not the upper extremities. He had diffusely brisk reflexes and increased tone in the bilateral upper extremities. He resisted manual manipulation of the bilateral upper extremities and briefly raised both arms antigravity. The bilateral lower extremities were withdrawn briskly to light touch. Babinski reflex was absent bilaterally as the great toes showed no movement. Sensation, coordination and gait were deferred as the patient was not following commands.
Investigations
CT head without contrast showed symmetrical hypodensities in the posterior bilateral cerebellar hemispheres, bilateral globus pallidus and questionably in the pons (figure 1), and toxic or metabolic injury were provided as the most likely diagnoses. A chest radiograph showed diffuse bilateral patchy bi-basilar ground glass opacities. Bedside point-of-care ultrasound showed no pericardial effusion, normal left ventricular function, no mitral or aortic insufficiency and the inferior vena cava was non-distended with appropriate inspiratory collapse. An ECG showed sinus tachycardia, left anterior fascicular block and prolonged QT interval (QTc 524 ms). Select admission laboratory results are shown in table 1. A respiratory viral panel (COVID-19, influenza A/B, RSV) was negative. Urine microscopy showed one white cell and two red cells with no bacteria, and a standard urine drug screen (amphetamine, barbiturates, benzodiazepines, cocaine, opiates, cannabinoids, methadone, oxycodone) was negative as were serum acetaminophen, ethanol and salicylate levels. Urine fentanyl (a separate non-facility test that returned 6 days later) was raised to 137.3 ng/mL (positive cut-off 1.0 ng/mL). No prior labs were available for baseline comparison as the patient was otherwise healthy with little prior contact with the healthcare system.
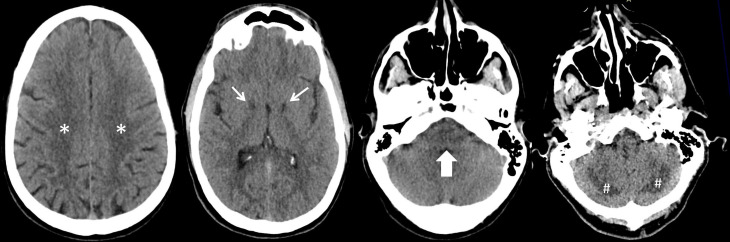
Figure 1.
Axial CT images of the brain without intravenous contrast showing symmetrical abnormal hypodensity in the bilateral globus pallidus (small arrows) and posterior cerebellar hemispheres (hash tags) corresponding to regions of reduced diffusivity and FLAIR hyperintensity on subsequent brain MRI. Apparent hypodensity in the pons (large arrow) was suspicious for an additional site of involvement but had no correlate on subsequent MRI and proved to be an artefact. Conversely, abnormality of the bilateral centrum semiovale (asterisks) was not appreciated on the CT scan and was only evident on subsequent MRI.
Table 1.
Select admission laboratory values
| Test | Patient values | Reference range |
| Creatinine | 2.37 | 0.70–1.30 mg/dL |
| Blood urea nitrogen | 43 | 6–20 mg/dL |
| Bicarbonate | 17 | 21–32 mmol/L |
| Total bilirubin | 2.9 | 0.3–1.2 mg/dL |
| Alkaline phosphatase | 41 | 53–128 U/L |
| AST | 115 | ≤39 U/L |
| ALT | 76 | ≤ 52 U/L |
| Creatine kinase | 4269 | 43–349 U/L |
| Lactic acid | 5.5 | ≤2.0 mmol/L |
| High sensitivity troponin 0 hour | 11 201 | ≤100 ng/L |
| High sensitivity troponin 2 hour | 11 660 | ≤100 ng/L |
| Ammonia | 51 | <72 μmol/L |
| Urine fentanyl | 137.3 | <1 ng/mL |
ALT, alanine aminotransferase; AST, aspartate aminotransferase.
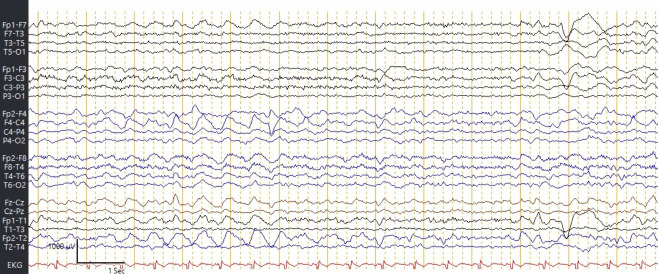
A routine electroencephalogram (EEG) with a duration of 25 min showed frontal intermittent rhythmic delta activity (FIRDA) and no epileptiform abnormalities (figure 2).
Figure 2.
Frontal intermittent rhythmic delta activity (FIRDA) indicative of a moderate encephalopathy, non-specific as to aetiology. No seizures or epileptiform discharges. FIRDA is seen in toxic or metabolic encephalopathies, degenerative diseases and deep midline structural abnormalities.
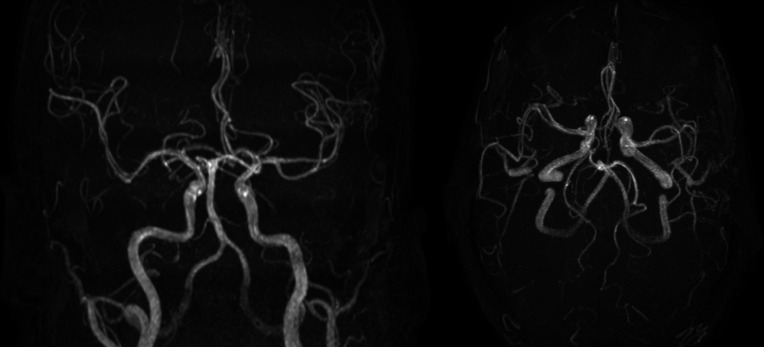
MRI of the brain with and without intravenous contrast showed symmetrical confluent reduced diffusivity and FLAIR hyperintensity in the supratentorial white matter with sparing of subcortical U-fibres, symmetrically in the bilateral globus pallidus and symmetrically in the posterior cerebellar hemisphere folia, involving the cerebellar cortex and adjacent white matter. Small foci of susceptibility and post-contrast enhancement were seen in the affected portions of the cerebellar hemispheres. The cerebral cortical grey matter, internal capsules, hippocampi and brainstem were normal. Magnetic resonance angiography (MRA) of the brain was normal. Based on the MRI and MRA findings, a toxic or metabolic process remained the leading diagnostic consideration (figures 3–6).
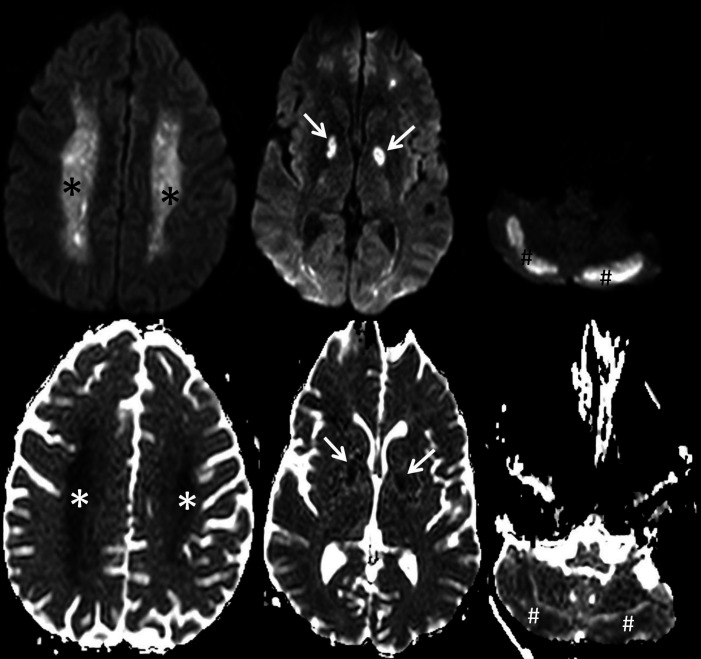
Figure 3.
Axial trace diffusion weighted images (top row) and corresponding apparent diffusion coefficient maps (bottom row) of the brain showing symmetrical confluent reduced diffusivity throughout the bilateral centrum semiovale (asterisks), globus pallidus (arrows) and posterior cerebellar hemispheres (hash tags).
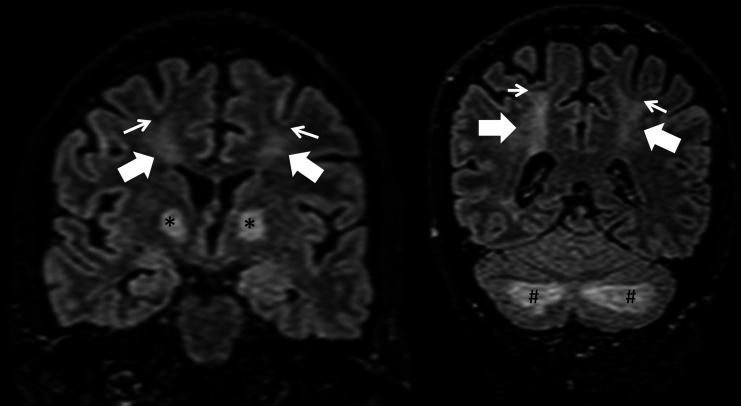
Figure 4.
Coronal T2 fluid attenuation inversion recovery images showing symmetrical hyperintense signal abnormality in the bilateral centrum semiovale (thick arrows), globus pallidus (asterisks) and cerebellar cortex/folia (hash tag). Note the distinct sparing of subcortical U-fibres (thin arrows).
Figure 5.
Coronal (left) and axial (right) maximum intensity projection MR angiogram images of the brain showing normal flow-related enhancement throughout the major intracranial arteries.
Figure 6.
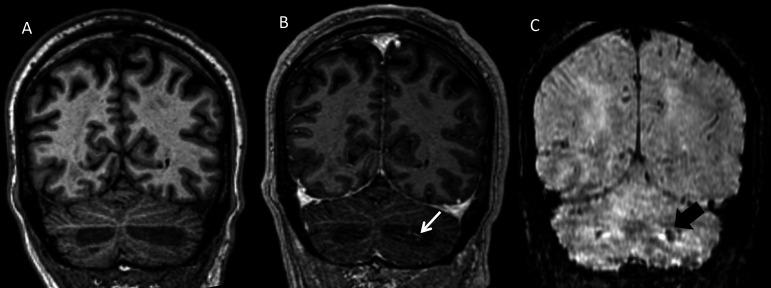
Coronal T1-weighted turbo field echo images acquired prior to (A) and following intravenous contrast administration (B) show thin linear branching foci of enhancement in the affected region of the left cerebellum (arrow) with corresponding hypointensity on susceptibility-weighted imaging (C, large arrow), favoured to represent dilated veins.
A repeat routine EEG with a duration of 21 min again revealed FIRDA and no epileptiform abnormalities. After 18 days, although the history and MRI findings supported a diagnosis of fentanyl-induced leukoencephalopathy, he continued to remain bedbound, tube feed dependent, and require restraints due to poor safety. At that time he had a thrombocytosis with platelets 946 (normal 150–400 ×109/L), which had gradually risen from his initial platelet count of 252 on admission. Given his uncertain prognosis and unexplained thrombocytosis, a lumbar puncture was performed to complete a robust work-up and rule out an infectious aetiology. Cerebrospinal fluid (CSF) laboratory analysis is shown in table 2.
Table 2.
Cerebral spinal fluid analysis
| Test | Patient values | Reference range |
| CSF appearance | Clear | |
| CSF colour | Slight haemolysis | |
| Specific gravity | 1.006 | |
| White blood cells | <1 cell/mm3 | 0–5 mm3 |
| Red blood cells | 963 cell/mm3 | <1/mm3 |
| Neutrophil % | 31% | 0–6% |
| Lymphocyte % | 49% | 40–80% |
| Monocyte % | 20% | 15–45% |
| Glucose % | 59% | 40–70% |
| Lactate dehydrogenase | 25 U/L | |
| Protein | 85 mg/dL | 14–45 mg/dL |
| Albumin | 43 mg/dL | 0–35 mg/dL |
| Albumin index oli ratio | 12.8 | 0.0–9.0 |
| IgG index ratio | 0.55 | 0.28–0.66 |
| IgG albumin ratio | 0.15 | 0.09–0.25 |
| Oligoclonal bands | 0 |
CSF, cerebrospinal fluid.
Differential diagnosis
In this case drug toxicity was considered the most likely diagnosis from early in the work-up. The crushed pills and residue found in the patient’s hotel room, the neurological examination and the radiology findings, when considered in aggregate, were highly suggestive of a drug toxicity. In general, toxic encephalopathy should be considered when brain imaging shows symmetric abnormalities involving the deep grey nuclei, white matter and cerebellum. However, there are numerous neurotoxins and each can present with a variety of imaging manifestations.
‘Chasing the dragon’ leukoencephalopathy can occur with inhalation of heroin smoke and has a well described symmetrical pattern of diffusion and FLAIR signal abnormalities involving the bilateral globus pallidus, cerebellum and supratentorial white matter,11 which fits closely with the imaging findings seen in our patient.
Cerebellar, Hippocampal and Basal Nuclei Transient Edema with Restricted diffusion (CHANTER) syndrome has been reported as a common pattern of toxic brain injury with a number of illicit drugs and shares some features with the case presented here, with the notable exception that the hippocampi were spared in our patient.12
Other toxicities including carbon monoxide and cyanide can result in bilateral globus pallidus injury. However, the symmetrical white matter and cerebellar injury seen in our case would be atypical for carbon monoxide.
Ischaemic injury can be difficult to disentangle from opiate toxicities as the two can overlap in imaging appearance and hypoxia is a frequent complication accompanying opiate-related respiratory suppression.13 It is possible that brain hypoxia may have a synergistic effect with direct toxin-mediated metabolic injury. Adult hypoxic brain injury, such as from cardiac arrest or near-drowning, will most commonly present with ischaemic (DWI) changes in both the cerebral cortical grey matter and deep grey nuclei, and possibly with a parasagittal or watershed distribution. In this patient the lack of cerebral cortical involvement and absence of a watershed pattern made isolated hypoxic brain injury unlikely.
Other vascular aetiologies (e.g., posterior reversible encephalopathy syndrome), infectious aetiologies (e.g., viral meningitis and rhombencephalitis), metabolic diseases (e.g., Leigh syndrome, pantothenate kinase-associated neurodegeneration), demyelinating disease (e.g., osmotic demyelination) and prion disease (e.g., Creutzfeldt-Jakob disease) can also result in symmetrical imaging abnormalities in the deep grey nuclei. However, these were all considered unlikely given the other features of the patient’s presentation.
Lumbar puncture with elevated protein was thought to be secondary to known CNS insult rather than a sign of a separate acute process. Raised CSF red blood cells are consistent with a traumatic tap.
Infectious aetiologies were considered given the onset of fever; however, such an acute change in cognition in the context of the patient’s history as well as the absence of laboratory abnormalities made an infectious process unlikely.
Over the course of his admission, sequelae of TLE and likely opioid withdrawal were symptomatically managed. Opioid withdrawal was a possibility given the patient’s fever, tachycardia and agitation that improved with opioids. There was no objective evidence to suggest the patient was taking other opioids prior to admission given a negative urine drug screen on admission and a lack of track marks to suggest intravenous drug use. Non-convulsive status epilepticus was considered as a potential aetiology of the patient’s symptoms given persistent encephalopathy and raised creatine kinase; however, two EEGs excluded this possibility.
Treatment
In the emergency department, given the patient’s urinary incontinence, elevated lactate and cognitive impairment, he received two doses of 2 mg lorazepam due to initial concern for seizure. He had escalating oxygen requirements requiring up to 20 L/min on a non-rebreather mask. He also received 3 L of intravenous fluid for his acute kidney injury and concern for rhabdomyolysis. Based on his chest radiograph findings, he was started on ceftriaxone and azithromycin. In the absence of ST elevations or depressions on ECG, the patient’s elevated troponins were attributed to a non-ST elevation myocardial infarction type II.
The patient was initially admitted to the medical intensive care unit for toxic metabolic encephalopathy of unclear aetiology and acute respiratory failure. In a few hours the patient was weaned to a 2 L nasal cannula and soon after to room air. He received opioids, antipsychotic medications (haloperidol, olanzapine) and benzodiazepines (lorazepam) to manage agitation, pain and suspected opioid withdrawal. A nasogastric tube was placed and tube feeds were initiated.
Supplements such as coenzyme Q10, vitamin E and vitamin C have anecdotally been shown to benefit some patients with heroin-induced spongiform leukoencephalopathy.14–19 Empirical antioxidant therapy with coenzyme Q10 was considered, but unfortunately could not be crushed to be administered through the nasogastric tube. The patient completed a course of antibiotics for a presumed aspiration pneumonia.
Outcome and follow-up
After 26 days the patient was discharged from the hospital to a skilled nursing facility. At the time of discharge the patient was oriented to self, place and general date. He was able to communicate with simple sentences and was actively participating in physical and occupational therapy. After another month of inpatient rehabilation the patient was able to perform all activities of daily life independently and was discharged to home with outpatient physical and occupational therapy. He reported that his voice sounded a little muffled and felt like he had to strain. He saw otolaryngology as an outpatient who noted a consistent dysphonia with their examination showing a right true vocal fold hypomobility and bilateral atrophy resulting in a small but persistent glottic fap. He underwent voice therapy and had subjective improvement in his voice. Otolaryngology noted improvement in the vibratory capacity of his vocal cords and improvement in his supraglottic function. He also participated in rehabilitation psychology to reduce feelings of distress. Less than a year from his hospitalisation he had returned to work full-time without accommodations. He is without neurological deficits. His expressive and receptive language functions are normal, thought process linear, and content within normal limits. Repeat imaging was not performed.
Discussion
Fentanyl and its analogues are extremely strong synthetic opioids with 50–100 times more potency than morphine.20 Fentanyl can be smoked, injected subcutaneously or intravenously, swallowed or sniffed. Overdose deaths from fentanyl outnumber deaths from other illicit substances, including methamphetamines and cocaine.21 Fentanyl typically has a short duration of action and rapid onset of effect. Importantly, fentanyl can be contaminated with emerging substances such as xylazine.22
Cases have described opioid intoxications including heroin, morphine and methadone resulting in acute TLE and delayed post-hypoxic leukoencephalopathy, distinct pathologies.23 24 Previous case reports have shown that opiates, mostly heroin, may lead to TLE. There have been prior reports of fentanyl-induced TLE. One report describes TLE following an overdose of fentanyl-contaminated fake oxycodone,25 while other cases involved the use of a fentanyl patch.26–28 There are also cases noted after oral ingestion of patches, intravenous or unknown routes of ingestion.29–32 Our case is the first to describe inhalational use of fentanyl causing TLE.
The pathophysiology of TLE is unclear, but several proposed mechanisms of injury to white matter exist including hypoxaemia, direct toxin damage to the myelin sheath or the capillary endothelium.1 Neuropathology has shown spongiform degeneration as evidenced by vacuolar changes. The degree of white matter vacuoles corresponds to the hyperintensities seen on DWI imaging.8 With regard to opioids, their lipophilicity, particularly fentanyl, allows for penetration of the blood–brain barrier.33 Furthermore, inhalation may be the fastest of all the routes as it has high bioavailability, bypassing first-pass metabolism by the liver.34 Based on the varied routes of access all resulting in the same pathology, this toxicity may have more to do with the drug class itself than the route of entry.
In our patient the initial urine drug screen was negative for opioids. A specialised fentanyl test was required to obtain the diagnosis. This case report is unique in that it describes inhalation of fentanyl causing damage to deep white matter and bilateral cerebellum without significant cortical involvement on MRI. EEG should be employed in TLE primarily for ruling out non-convulsive status epilepticus. Other white matter disorders such as posterior reversible encephalopathy syndrome (PRES) have a higher probability of seizures compared with TLE.35 Pathophysiological mechanisms such as white matter oedema versus demyelination or axonal injury are theorised as affecting EEGs differently but, overall, there is limited research and clinical evidence about what diagnostic and prognostic information it can provide.36 37
This case can help inform future clinicians to be watchful for other toxins that may not be initially identified on screening tests. Furthermore, this case illustrates the need for inclusion of fentanyl in routine urine drug screens for earlier identification and appropriate management.
Patient’s perspective.
“Well, honestly, I don’t recall much. When I came to I saw that I was in the hospital and was being attended to. From what I can remember early on is that my recovery was miraculous. Early on it was looking like I would need 24 hour care after being discharged but I focused and worked hard in my therapy session and was determined not to leave the hospital only to be checked into a group facility for ongoing care. It’s been 6 months now and I am back at home, working and feeling strong and healthy. I have regrets often about what I did to myself, my wife, and my family. I’m grateful to all the doctors, nurses, and EMTs that saved my life and the therapists that got me back to a functioning member of society.”
Learning points.
Toxic leukoencephalopathy (TLE) is a non-specific encephalopathy syndrome caused by a variety of toxic aetiologies resulting in damage to the cerebral white matter.
On MRI, symmetrical diffusion restriction in the bilateral cerebellar hemispheres, deep grey nuclei, hippocampi and supratentorial white matter should raise consideration for opioid toxicity.
While heroin has been the principal opiate associated with TLE, this case highlights the importance of recognising other opiates such as fentanyl as potentially causative.
Fentanyl is not routinely tested on all urinary drug tests; thus, providers need to be suspicious of its presence and to work with their local laboratories to have it added to the standard drug testing platforms.
Footnotes
@chrisoeden
Contributors: COE wrote the first draft of the manuscript. The following authors were responsible for drafting of the text, sourcing and editing of clinical images, investigation results, drawing original diagrams and algorithms, and critical revision for important intellectual content: COE, DSA, DRP, AJH and AHA. The following authors gave final approval of the manuscript: COE, DSA, DRP, AJH and AHA.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Consent obtained directly from patient(s).
References
- 1. Özütemiz C, Roshan SK, Kroll NJ, et al. Acute toxic leukoencephalopathy: etiologies, imaging findings, and outcomes in 101 patients. AJNR Am J Neuroradiol 2019;40:267–75. 10.3174/ajnr.A5947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Kumar Y, Drumsta D, Mangla M, et al. Toxins in brain! Magnetic resonance (MR) imaging of toxic leukoencephalopathy - a pictorial essay. Pol J Radiol 2017;82:311–9. 10.12659/PJR.901791 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Filley CM, Kleinschmidt-DeMasters BK. Toxic leukoencephalopathy. N Engl J Med 2001;345:425–32. 10.1056/NEJM200108093450606 [DOI] [PubMed] [Google Scholar]
- 4. Wen Z, Dai P, Zhou Z, et al. Case report: toxic encephalopathy caused by repeated inhalation of liquid sealant. Front Public Health 2022;10:920310. 10.3389/fpubh.2022.920310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Fornazzari L, Pollanen MS, Myers V, et al. Solvent abuse-related toluene leukoencephalopathy. J Clin Forensic Med 2003;10:93–5. 10.1016/S1353-1131(03)00035-X [DOI] [PubMed] [Google Scholar]
- 6. Mi T, Han C, Wang Y, et al. Acute toxic leukoencephalopathy in migrant workers exposed to organic solvents in construction materials. Occup Environ Med 2013;70:435–6. 10.1136/oemed-2012-101302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Macchi ZA, Carlisle TC, Filley CM. Prognosis in substance abuse-related acute toxic leukoencephalopathy: a scoping review. J Neurol Sci 2022;442:120420. 10.1016/j.jns.2022.120420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Offiah C, Hall E. Heroin-induced leukoencephalopathy: characterization using MRI, diffusion-weighted imaging, and MR spectroscopy. Clin Radiol 2008;63:146–52. 10.1016/j.crad.2007.07.021 [DOI] [PubMed] [Google Scholar]
- 9. Keogh CF, Andrews GT, Spacey SD, et al. Neuroimaging features of heroin inhalation toxicity: "chasing the dragon". AJR Am J Roentgenol 2003;180:847–50. 10.2214/ajr.180.3.1800847 [DOI] [PubMed] [Google Scholar]
- 10. Blasel S, Hattingen E, Adelmann M, et al. Toxic leukoencephalopathy after heroin abuse without heroin vapor inhalation: MR imaging and clinical features in three patients. Clin Neuroradiol 2010;20:48–53. 10.1007/s00062-010-0022-9 [DOI] [PubMed] [Google Scholar]
- 11. Alambyan V, Pace J, Miller B, et al. The emerging role of inhaled heroin in the opioid epidemic: a review. JAMA Neurol 2018;75:1423–34. 10.1001/jamaneurol.2018.1693 [DOI] [PubMed] [Google Scholar]
- 12. Jasne AS, Alsherbini KH, Smith MS, et al. Cerebellar Hippocampal and Basal Nuclei Transient Edema with Restricted Diffusion (CHANTER) syndrome. Neurocrit Care 2019;31:288–96. 10.1007/s12028-018-00666-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Kim DH. Fentanyl-induced hypoxic encephalopathy. Radiopaedia. Available: https://radiopaedia.org/cases/fentanyl-induced-hypoxic-encephalopathy [Accessed 19 Feb 2024]. [Google Scholar]
- 14. Kriegstein AR, Shungu DC, Millar WS, et al. Leukoencephalopathy and raised brain lactate from heroin vapor inhalation ("chasing the dragon"). Neurology 1999;53:1765–73. 10.1212/wnl.53.8.1765 [DOI] [PubMed] [Google Scholar]
- 15. Long H, Deore K, Hoffman RS, et al. A fatal case of spongiform leukoencephalopathy linked to “chasing the dragon". J Toxicol Clin Toxicol 2003;41:887–91. 10.1081/clt-120025358 [DOI] [PubMed] [Google Scholar]
- 16. Gacouin A, Lavoue S, Signouret T, et al. Reversible spongiform leucoencephalopathy after inhalation of heated heroin. Intensive Care Med 2003;29:1012–5. 10.1007/s00134-003-1691-5 [DOI] [PubMed] [Google Scholar]
- 17. Cordova JP, Balan S, Romero J, et al. 'Chasing the dragon': new knowledge for an old practice. Am J Ther 2014;21:52–5. 10.1097/MJT.0b013e31820b8856 [DOI] [PubMed] [Google Scholar]
- 18. Bartlett E, Mikulis DJ. Chasing "chasing the dragon" with MRI: leukoencephalopathy in drug abuse. Br J Radiol 2005;78:997–1004. 10.1259/bjr/61535842 [DOI] [PubMed] [Google Scholar]
- 19. Jee RC, Tsao WL, Shyu WC, et al. Heroin vapor inhalation-induced spongiform leukoencephalopathy. J Formos Med Assoc 2009;108:518–22. 10.1016/S0929-6646(09)60101-7 [DOI] [PubMed] [Google Scholar]
- 20. Suzuki J, El-Haddad S. A review: fentanyl and non-pharmaceutical fentanyls. Drug Alcohol Depend 2017;171:107–16. 10.1016/j.drugalcdep.2016.11.033 [DOI] [PubMed] [Google Scholar]
- 21. National Center for Health Statistics, Center for Disease Control and Prevention . U.S. overdose deaths in 2021 increased half as much as in 2020 – but are still up 15%; 2022. Available: https://www.cdc.gov/nchs/pressroom/nchs_press_releases/2022/202205.htm [Accessed 19 Feb 2024].
- 22. Ruiz-Colón K, Chavez-Arias C, Díaz-Alcalá JE, et al. Xylazine intoxication in humans and its importance as an emerging adulterant in abused drugs: a comprehensive review of the literature. Forensic Sci Int 2014;240:1–8. 10.1016/j.forsciint.2014.03.015 [DOI] [PubMed] [Google Scholar]
- 23. Rossi NP, Sathyanarayanan G, Mahmood M, et al. Toxic leukoencephalopathy versus delayed post-hypoxic leukoencephalopathy after oral morphine sulphate overdose. BMJ Case Rep 2023;16:e255291. 10.1136/bcr-2023-255291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Knudsen GH, Kermanian N, Kock-Jensen CH, et al. Opioid-induced leukoencephalopathies: a report of two cases. Case Rep Neurol 2022;14:25–30. 10.1159/000521410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Dinicu AI, Chaudhari A, Kayyal S. Diffuse subcortical white matter injury and bilateral basal ganglia neuronal loss after acute opioid overdose. Neuroradiol J 2020;33:267–70. 10.1177/1971400920927878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Yoo IS, Lee SH, Suk SH. A case of fentanyl intoxication and delayed hypoxic leuko-encephalopathy caused by incidental use of fentanyl patch in a healthy elderly man. J Neurocrit Care 2015;8:35–8. 10.18700/jnc.2015.8.1.35 [DOI] [Google Scholar]
- 27. Kim J, Hyung SW, Seo J, et al. Delayed post-hypoxic leukoencephalopathy caused by fentanyl intoxication in a healthy woman. Dement Neurocogn Disord 2020;19:170–2. 10.12779/dnd.2020.19.4.170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Foy L, Seeyave DM, Bradin SA. Toxic leukoencephalopathy due to transdermal fentanyl overdose. Pediatr Emerg Care 2011;27:854–6. 10.1097/PEC.0b013e31822c281f [DOI] [PubMed] [Google Scholar]
- 29. Lim H, Lee H, Lee YM. Toxic leukoencephalopathy by accidental oral ingestion of an infant’s fentanyl patch. Ann Child Neurol 2021;29:93–4. 10.26815/acn.2020.00262 [DOI] [Google Scholar]
- 30. Switzer AR, Beland B, Sarna JR, et al. Fentanyl overdose causing hippocampal ischaemia followed by delayed leukoencephalopathy. Can J Neurol Sci 2020;47:398–9. 10.1017/cjn.2020.33 [DOI] [PubMed] [Google Scholar]
- 31. Diaz-Segarra N, McKay O. Cognitive improvement in a patient with toxic leukoencephalopathy due to fentanyl overdose after starting donepezil and venlafaxine: a case report (abstract). PM&R 2019;11(S2). Available: https://pmrjabstracts.org/abstract/cognitive-improvement-in-a-patient-with-toxic-leukoencephalopathy-due-to-fentanyl-overdose-after-starting-donepezil-and-venlafaxine-a-case-report/ [Google Scholar]
- 32. Agarwal A, Evans M, Mogallapu R, et al. “Toxic leukoencephalopathy after "chasing the dragon" with a non-heroin opioid”. Cureus 2023;15:e45774. 10.7759/cureus.45774 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Trescot AM, Datta S, Lee M, et al. Opioid pharmacology. Pain Physician 2008;11:S133–53. [PubMed] [Google Scholar]
- 34. Lötsch J, Walter C, Parnham MJ, et al. Pharmacokinetics of non-intravenous formulations of fentanyl. Clin Pharmacokinet 2013;52:23–36. 10.1007/s40262-012-0016-7 [DOI] [PubMed] [Google Scholar]
- 35. Kamiya-Matsuoka C, Tummala S. Electrographic patterns in patients with posterior reversible encephalopathy syndrome and seizures. J Neurol Sci 2017;375:294–8. 10.1016/j.jns.2017.02.017 [DOI] [PubMed] [Google Scholar]
- 36. Mader EC, Ramos AB, Cruz RA, et al. Full recovery from cocaine-induced toxic leukoencephalopathy: emphasizing the role of neuroinflammation and brain edema. J Investig Med High Impact Case Rep 2019;7:2324709619868266. 10.1177/2324709619868266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Nunez PL, Srinivasan R, Fields RD. EEG functional connectivity, axon delays and white matter disease. Clin Neurophysiol 2015;126:110–20. 10.1016/j.clinph.2014.04.003 [DOI] [PMC free article] [PubMed] [Google Scholar]