Abstract
Cells are dynamic structures that continually generate and sustain mechanical forces within their environments. Cells respond to mechanical forces by changing their shape, moving, and differentiating. These reactions are caused by intracellular skeletal changes, which induce changes in cellular mechanical properties such as stiffness, elasticity, viscoelasticity, and adhesiveness. Interdisciplinary research combining molecular biology with physics and mechanical engineering has been conducted to characterize cellular mechanical properties and understand the fundamental mechanisms of mechanotransduction. In this review, we focus on the role of cytoskeletal proteins in cellular mechanics. The specific role of each cytoskeletal protein, including actin, intermediate filaments, and microtubules, on cellular elasticity is summarized along with the effects of interactions between the fibers.
Keywords: Cellular elasticity, Stiffness, Actin, Stress fiber, Intermediate filament, Microtubule
Introduction
Cells are continuously subjected to forces exerted both internally and externally that result in changes in the cellular mechanical properties, which are known to influence various aspects of cell structure and function. The mechanical property induces structural changes such as deformation and remodeling, and regulates cell motility, migration, and differentiation [1–3]. Loss of contractility of heart muscle cells may cause heart failure [4]. Neural cells can die when the axons of neural cells are stretched excessively, whereas cell growth is activated by moderate stretching of the axon [5]. Several researches reported that cancer cells are mechanically softened compared with healthy normal cells [6–8]. However, cancer cells that are more invasive have increased elasticity in other reports [9].
Internally, cellular mechanical properties are the result of the structural organization of subcellular components, including the nuclear envelope, cell membrane, and cytoskeleton. Among them, the cytoskeleton is a major regulator of the intracellular mechanics. The mechanical properties of a cell include elasticity, stiffness, viscoelasticity, and adhesion. Elasticity is the resistance of a material to deformation by stress [10]. The elastic materials show a linear or non-linear relationship between stress and strain, with the material returning to its initial state when the stress is removed. Since elasticity is the ratio of stress (force applied per unit area) and strain (deformation caused by stress), high elasticity indicates that large stress is required to bring about even a slight deformation. In contrast, low elasticity indicates that minimal stress can cause a large strain. Stiffness is the rigidity of a material in response to stress; it depends on the size, shape, mass, and crosslinks in the structure of a material [11, 12]. Viscoelastic materials resist shear flow and strain linearly with time (they exhibit time-dependent strain) [13].
Externally, cells are continually subjected to mechanical loads from their extracellular matrix (ECM). Cells adhere to ECM ligands through integrins, a family of transmembrane adhesion receptors [14]. Integrin-mediated adhesion between cells and the ECM is a highly regulated process associated with several critical cellular events, including gene expression, cell locomotion, and differentiation [15–17]. A cell senses and responds to its ECM by altering its structure and mechanical properties. [18–21]. On a substrate with nano-patterned grooves, cells can change shape and arrangement along the grooves [22]. Fibroblasts on a soft matrix are sitting in a small area with no detectable stress fibers [23]. As the matrix stiffens, the fibroblasts spread over a wide area and the stress fiber is formed.
Previous studies have provided valuable information on the underlying mechanism of the cellular mechanics [24–27]. However, the mechanical response to stimuli is still complicated to understand due to the intrinsic nature of cells. First, cellular mechanical properties are neither homogeneous nor isotropic [28]. Second, because cells are alive, all features of the cytoskeletal components vary as a function of time [29]. Third, the elastic property of cells is inherently nonlinear. Cytoskeletal components are semi-flexible polymers, allowing cells to resist deformation depending on the extent of stress [30, 31]. Fourth, the cytoskeletal components of actin, microtubule, and intermediate filaments interact with each other in complicated ways and are connected to other components, such as actin-binding proteins and integrins [32].
Three cytoskeletal components—actin, microtubule, and intermediate filaments—play pivotal and distinct roles in intracellular mechanics. In this work, we reviewed the literature, highlighting research that explores the role of the cytoskeleton especially in intracellular elasticity and identifies each cytoskeletal filament involved in that role. In the “Contribution of actin filaments to intracellular elasticity” section, the effects of actin filament (F-actin) contents and distribution on cell elasticity are summarized. In the “Actin-binding proteins that affects intracellular elasticity” section, the variety of F-actin-binding proteins and their contributions to intracellular elasticity are summarized. In the “Contribution of intermediate filament to intracellular elasticity” and “Non-critical role of microtubules in intracellular elasticity” sections, the roles of intermediate filaments and microtubules in intracellular elasticity are summarized, respectively. Finally, we related the complexity of cytoskeletal filaments to existing difficulties in understanding intracellular elasticity.
Contribution of actin filaments to intracellular elasticity
F-actin is a major factor in cellular elasticity
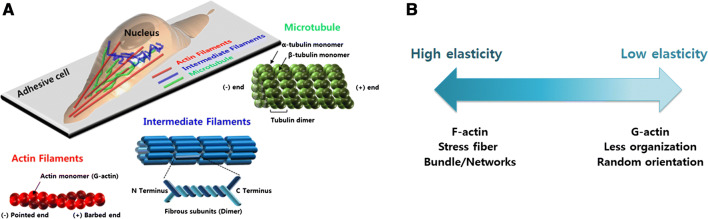
Actin is a ubiquitous and essential protein in all eukaryotic cells. It is a major constituent of the cytoskeletal network and forms many different cellular structures. Actin provides a framework, determines cell shape, and plays a role in a variety of cellular processes, including division of cytoplasm, motility, and muscle contraction [33, 34]. Actin can be present as either a globular (G) or filamentous (F) protein. The monomeric globular form of G-actin has molecular weight of 46 kDa and forms the basic unit for F-actin. The concentration of G-actin is relatively similar in most cells and it determines the polymerization rate of G-actin into F-actin [35, 36]. The polymerization of G-actin is linearly dependent on the ATP-actin concentration.
The G-actin can assemble into F-actin in vitro under the right ionic conditions; no other proteins are required to produce filaments (Fig. 1a) [37]. Several studies have been performed on various cell types to isolate the specific contributions of actin filaments on the cellular elastic property. F-actin was disrupted or stabilized by cytoskeletal drugs and the resulting changes in deformability or elasticity of cells were measured by optical stretcher or atomic force microscopy (AFM), respectively [38, 39]. The drugs cytochalasin B (Cyt-B), cytochalasin D (Cyt-D), and latrunculin A disrupt F-actin by inhibiting actin polymerization [40–42]. Jasplakinolide (Jas) promotes actin nucleation and polymerization [43].
Fig. 1.
a Main constituents of the cytoskeleton and their intracellular distribution. G-actin polymerizes to form F-actin under physiological conditions. The polymerization and de-polymerization readily occur at barbed and pointed ends of F-actin, respectively. Intermediate filaments are built from monomers that associate with each other to form dimers. Microtubules are formed by the polymerization of a dimer of two globular proteins, α- and β-tubulin. b Schematic relationships between intracellular elasticity and concentration and structure of F-actin. The intracellular elasticity is proportional to the concentration and structuring of F-actin
When cells are treated with Cyt-B, Cyt-D, or latrunculin A, long actin filaments are disrupted and their arrangement changes randomly. The disruption of F-actin leads to cell softening across multiple cell types. For example, the elastic modulus of 3T3 and NRK fibroblasts decreased by a factor of 1.4–3.2 when treated with appropriate concentrations of Cyt-B, Cyt-D, and latrunculin A [39]. Mouse embryonic fibroblast becomes more deformable by latrunculin A [44]. MCF-7 adenocarcinoma cells treated with latrunculin A were more easily deformed by optical stretching, especially at small strains (< 5%) [38].
Cellular softening by F-actin disruption depends on cell type. For example, the elastic modulus of Jurkat cells decreased by 55% (from 0.51 ± 0.06 to 0.23 ± 0.04 kPa) and that of lymphocytes decreased by 72.5% (from 1.24 ± 0.09 to 0.34 ± 0.04 kPa) when exposed to 20 μg/mL of Cyt-B [45]. The elastic modulus of chondrocytes decreased by 60–80% when treated with Cyt-D, while hepatocellular carcinoma and fibrosarcoma cells showed no apparent changes in elasticity [46]. These results suggest that F-actin may contribute to the elasticity of cancer cells differently than normal cells.
Intracellular elasticity is proportional to the amount of F-actin
F-actin content is an important factor determining intracellular elasticity. However, F-actin contents continue to fluctuate to some extent in response to internal and external environmental stimulations. When neutrophils are stimulated with bacterial-derived formyl-Met-Leu-Phe, F-actin levels increased, causing an increase in intracellular stiffness. The elasticity of neutrophils was proportional to F-actin content with a proportionality constant of 0.54 [47]. Murine fibroblasts cells (NIH3T3) also showed a strong relationship between their elastic modulus and F-actin content, with a proportionality constant of 0.023 [48]. Modeling has shown that the elastic modulus of an F-actin network is dependent on actin contents with the proportionality constant of 2.5 [49].
Irreversible changes in F-actin contents can be induced by aging and disease, which are accompanied by changes in intracellular elasticity. Skin fibroblasts isolated from young donors (< 42 years old) showed 31% less F-actin contents than fibroblasts isolated from older donors (> 60 years old) [50]. Consequently, the elastic modulus of the younger group is about 60% lower than that of old group. F-actin levels in bladder cancer cells of HTB-9, HT1376, and T24 are only 73%, 35%, and 42% that of non-malignant bladder HCV29 cells. The elastic modulus of HCV29 cells is 50 kPa and that of cancer cells is below 15 kPa—a 70% difference [8].
Intracellular elasticity depends on F-actin distribution
F-actin is organized into high-order structures within cells as bundles or three-dimensional networks. This structuring is regulated by a variety of actin-binding proteins that are closely associated with intracellular elasticity. The alignment of an F-actin bundle also affects intracellular elasticity [51]. When F-actin bundles are aligned along the long axis of a cell with well-defined stress fibers, the cells exhibit a high elastic modulus. In contrast, when actin filaments are less organized or oriented randomly, the cells have a lower elastic modulus [8, 52–54]. F-actin in ovarian epithelial cells (ISOE) is aligned in the same direction as ovarian cancer cells (OVCAR and HEY), thus ISOE has a relatively large elastic modulus [6]. Generally, an F-actin network with low density will soften a cell membrane, while dense networks with a high concentration of cross-links will stiffen a cell [55].
F-actin is bundled together via cross-linking proteins to form stress fibers [4, 56, 57], which are contractile bundles composed of approximately 10–30 actin filaments [58]. A stress fiber is a dynamic structure with non-uniform elasticity and forces, as indicated by the constantly fluctuating spacing between myosin II along the stress fiber [59–61]. The mechanical property of a stress fiber depends on the contractile level, and the elastic modulus is ~ 15 kPa [62–64]. The presence of stress fibers causes enhanced intracellular elasticity that is sensitive to spatial and temporal distribution. For example, the transient accumulation of stress fibers induced by Src proteins and their organization leads to transient increase in intracellular elasticity in a human breast cell line [48].
Actin-binding proteins that affect intracellular elasticity
Various actin-binding proteins
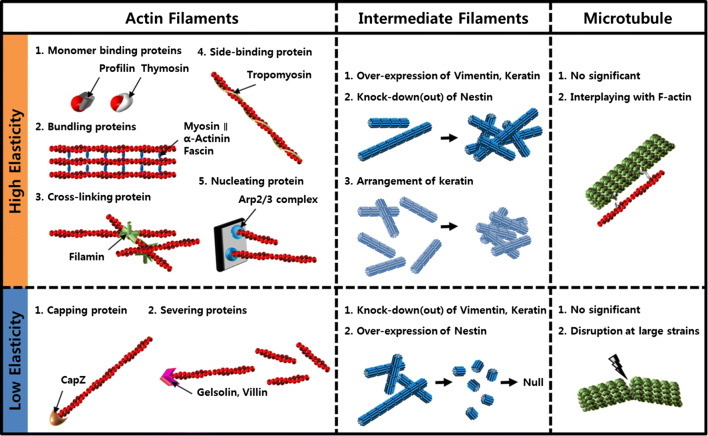
Actin-binding proteins regulate the organization and dynamics of actin filaments [65, 66]. Actin-binding proteins include monomer-binding proteins (profilin, thymosin), bundling proteins (myosin II, α-actinin, fascin), cross-linking proteins (filamin), side-binding proteins (tropomyosin), capping proteins (CapZ), severing proteins (gelsolin, villin), and nucleating protein (Arp2/3 complex). Among them, a high order of actin filament structures of bundles and networks is facilitated mainly by myosin II, α-actinin, filamin, and tropomyosin.
Actin-binding proteins increase intracellular elasticity
Actin-binding proteins are involved in the regulation of the intracellular elasticity through stabilizing F-actin structure, changing the mechanical properties of F-actin itself, and structuring bundles or networks.
α-Actinin binds to F-actin at two specific binding sites that are present in various intracellular structures that require organized actin filament polarity (Fig. 1a) [67–70]. Low concentrations of α-actinin and F-actin form orthogonal actin filament networks, but forms bundles at high concentration above a crosslinking-to-bundling threshold [71–76]. The F-actin networks demonstrate viscoelastic property with non-linear stress–strain relationship, and the property varies by binding proteins and density of crosslinking [29, 31, 77, 78]. An F-actin bundle with α-actinin behaves like a viscous fluid because each bundle can slip past other bundles [71]. Overexpression of α-actinin causes cellular stiffening. In human osteoblasts, 150% overexpression of α-actinin with a GFP fusion protein causes a 60% increase in the cell stiffness [79]. The elastic modulus of fibroblasts injected with α-actinin increased from 80 ± 20 to 190 ± 30 dyn/cm2 [80]. The cooperation of α-actinin with fascin further enhanced the elastic modulus up to 240 ± 35 dyn/cm2.
Myosin II proteins selectively bind to appropriate subsets of actin filaments via motor head domain to form an actin filament bundle structure, such as a stress fiber. In bundled F-actin networks, myosin filaments actively contract or slide antiparallel actin filaments, which causes local tension or network contraction [81–84]. Therefore, myosin can stiffen F-actin by more than two orders of magnitude [85]. The stiffened actin filament generates internal stress, which results in increased whole-cell elasticity. Approximately 300 myosin molecules must be assembled with actin filaments to generate sustained internal tension [86]. The inhibition of myosin II results in softening of cells. When myosin II was inhibited by BDM and ML-7, which inhibit myosin-light-chain-kinase, fibroblast cells softened by a factor of three in both peripheral and nuclear regions [87]. When the fibroblast was treated with the myosin inhibitor blebbistatin, the elastic modulus decreased from 20 to 8 kPa for 30–60 min [88].
Filamin is a large dimeric multidomain protein (~ 280 kDa) which is mainly localized to the leading edge and the trailing stress fibers in the cellular membrane [89, 90]. Filamin plays a role in bundling actin fibers. Actin filaments are dispersed randomly in filamin-deficient cells, while actin filaments align and form stress fibers in the presence of filamin. The formation of stress fibers can be facilitated and stabilized by filamin. Therefore, filamin-deficient cells are softer than filamin-expressing cells [91, 92]. The effects of filamin on elasticity of F-actin network are more pronounced when filamin cooperates with α-actinin [6].
Tropomyosins (Tpms) are integral components of actin filaments that play a critical role in the generation of functionally distinct F-actin structures [93–97]. Tpms stabilize F-actin structures and regulate the interaction between F-actin and cellular membranes. In red blood cell, Tpms stabilize short F-actin and strengthen the linkages between the membrane skeleton and transmembrane glycoproteins, which cause changes in morphological and mechanical properties of the red blood cells [98]. Tpms also affects the amount and organization of F-actin, with the effects depending on the Tpm isoforms [99]. Various Tpm isoforms have been identified, including Tpm 1.10, Tpm 1.11, Tpm 1,12, Tpm 1.7, Tpm 2.1, Tpm 3.1, and Tpm 4.2. By overexpressing Tpm 3.1, the F-actin/G-actin ratio increased significantly and the elastic modulus of cells increased. Overexpression of Tpm 1.10, Tpm 1,11, Tpm 1.7, and Tpm 3.1 inhibits the F-actin bundling while Tpm 1.11, Tpm 2.1, and Tpm 4.2 shows no significant effects on the F-actin bundles. The correlation between the intracellular elasticity and F-actin is summarized in Fig. 1b.
Contribution of intermediate filament to intracellular elasticity
Intermediate filaments function as tension-bearing elements to maintain cell shape and rigidity, and serve to anchor in place several organelles, including the nucleus and desmosomes [100, 101]. They are also involved in formation of the nuclear lamina, a net-like meshwork that lines the inner nuclear membrane and governs the shape of the nucleus. The intermediate filaments are flexible but stable and strong with less subunit exchange. More than 50 different intermediate filament proteins have been identified and classified into six groups based on similarities between their amino acid sequences (Table 1) [102, 103]. All intermediate filament proteins share a similar structure that is based on an N-terminal head domain, central α-helical rod domain of approximately 310 ~ 350 amino acids, and C-terminal tail domain (Fig. 1a) [101]. The head and tail domains vary in size and shape. Although intermediate filaments are essential to maintaining correct tissue structure and function, and are associated with more than 50 human diseases, much less is known about intermediate filaments compared with actin and microtubules.
Table 1.
| Class | Proteins | Size (kDa) | Cell type |
|---|---|---|---|
| I | Keratins, acidic | 40–60 | Epithelial cells |
| II | Keratins, basic | 50–70 | Epithelial cells |
| III | Vimentin | 54 | Mesenchymal cells |
| Glial fibrillary acidic protein | 51 | Astroglial cells | |
| Desmin | 53 | Muscle cells | |
| Synemin | 150 | Skeletal muscle cells | |
| Peripherin | 57 | Neurons | |
| IV | Neurofilaments H, M and L | 67, 150, 200 | Neurons |
| Internexin | |||
| Neurons | |||
| V | Lamin A, B, and C | 60–75 | All |
| VI | Filensin | 94 | Lens |
| Phakinin | 47 | Lens | |
| Nestin | 200–240 | Neuronal stem cells |
Recent studies have demonstrated that vimentin is an important regulator of intracellular elasticity among the intermediate filament proteins. Vimentin is a highly dynamic structural protein that undergoes changes in organization during various cellular processes, such as cell migration and spreading [104]. Vimentin contributes to intracellular elasticity in various cell types. For example, the elastic modulus of mouse embryonic fibroblasts expressing vimentin is 2 ~ 2.5 times higher than it is in fibroblasts that do not express vimentin [105]. The elastic modulus of chondrocytes was decreased by 2.8-fold due to vimentin disruption [106]. Vimentin-null fibroblasts also showed greater deformability than wild-type fibroblasts in response to compressive stress [107]. In addition, studies have shown that expression of vimentin is affected by the mechanical environment and is strongly related to cancer progression. Vimentin expression increased in intervertebral disc cells undergoing static compression and chondrocytes of weight-bearing cartilage [107, 108]. Vimentin is overexpressed in cancer cells and plays a critical role in metastasis [109, 110]. Although the mechanical role of vimentin is not clearly understood yet, it seems that the stiffening of cells with vimentin is related to localize subcellular components [105, 111]. Vimentin also constrains the random movement of subcellular components and traveling distance. This limitation helps stabilize subcellular components spatially, causing cellular stiffening.
Keratins are expressed in all epithelial cells and they form the structural framework of epithelial cells [112]. The keratins constitute 54 distinct proteins, 28 type I and 26 type II keratins, which are expressed in site- and differentiation-dependent manner in epithelial cells [113]. The keratins are especially abundant in epidermal keratinocytes and significantly contribute to the cellular elasticity of keratinocytes [114]. Keratinocytes are found to have relatively high Young’s modulus than other cell types due to the keratin intermediate filament [115–117]. The Young’s modulus significantly decreases in keratinocytes lacking keratin [118]. For example, the Young’s modulus of normal keratinocytes is 459 ± 31 Pa for nucleus and 752 ± 100 Pa for cell body. However, the Young’s modulus of type I keratin-deficient keratinocytes is only 343 ± 18 Pa for nucleus and 412 ± 75 Pa for cell body. When the keratin expression was decreased by immortalization in human keratinocytes, the deformability of keratinocytes was increased 2–3 times as compared to the normal keratinocytes [116]. The murine keratinocytes lacking keratins also exhibited about 60% higher deformability than wild-type cells [119].
Nestin is a class VI intermediate filament that is expressed in various tissues and stem or progenitor cells [120]. Nestin is highly expressed in high-metastatic cancer cells, such as pancreatic and prostate cancers; therefore, it is considered a biomarker for invasive phenotype [121, 122]. Nestin seems to be closely associated with the cellular elasticity and motility of metastatic cancer cells [123]. Highly metastatic mouse breast cancer cell was stiffened by nestin knockout, which may result in suppression of metastasis. When nestin was expressed normally, the cellular elasticity was recovered. Although only limited studies on nestin have been conducted to date, the change in elasticity seems to be related to the decreased vimentin–actin binding interaction caused by nestin [123].
Non-critical role of microtubules in intracellular elasticity
Microtubules are tubular polymers of tubulin and that form part of the cytoskeleton. They are straight and hollow cylinders 25 nm in diameter [124]. The length of microtubules in the cell varies between 200 nm and 25 µm depending upon the task of a particular microtubule and the stage of the cell’s life cycle [124]. Microtubules are the major components of cilia and flagella, and participate in the formation of spindle fibers during cell division [125]. They serve as conveyor belts, moving other organelles throughout the cytoplasm. Although microtubules play an important role in regulating mechanical stimuli, they show little effect on cellular elasticity compared with F-actin and intermediate filaments in most cases [45, 126]. Disruption or reorganization of the microtubules with drugs such as colchicine, nocodazole, and paclitaxel had no significant effect on intracellular elasticity [38, 127]. However, disrupted microtubules cause a decrease in elasticity in MCF-7 adenocarcinoma cells at large strains and an increase in elasticity in fibrosarcoma and hepatocellular carcinoma cells [38, 46]. Relationships between intracellular elasticity and three cytoskeletal filaments are summarized as schematic drawing (Fig. 2).
Fig. 2.
Schematic drawing of relationships between intracellular elasticity and cytoskeletal filaments. The intraelasticity of cell responds sensitively to the structure and expression level of F-actin and intermediate filaments. Role of microtubule is not significant
Concluding remarks
Cytoskeletal filaments of actin, intermediate filaments, and microtubules exhibit unique characteristics in structure, mechanics, contents, and functions (Table 2). Actin is one of the most abundant proteins in eukaryotes, accounting for 2.7 ~ 14% of cellular proteins [136, 137]. Actin forms polarized filaments that are mainly present in the cell periphery. F-actin is a highly dynamic structure that rapidly reorganizes during cell migration and morphological changes (Fig. 2a). Intermediate filaments are composed of several types of homologous proteins that are expressed in different types of cells and tissues (Fig. 2a). Intermediate filaments extend from the inner nucleus through the cytoplasm, accounting for ~ 1% of the cellular proteins in most cell types, but up to 85% in neuron and epidermal keratinocytes [138]. Intermediate filaments are relatively insoluble and stable structures with no polarity compared with actin and microtubules [102]. Microtubules are rigid hollow rods that extend throughout the cell (Fig. 2a). They are in a state of dynamic equilibrium, constantly assembling and disassembling via the addition and removal of dimers (α and β-tubulin).
Table 2.
Comparison of three cytoskeletal filaments in various aspects of structure, mechanical properties, contents, main presence region, and functions in cell
| Actin filaments | Intermediate filaments | Microtubules | Refs. | |
|---|---|---|---|---|
| Subunit | Actin monomer | Rod-like dimers (N terminus, α-helical rod domain, C terminus) | α-tubulin, β-tubulin | [37, 101, 124] |
| Subunit weight (kDa) | ~ 40 | 45 ~ 60 | ~ 50 | [128–130] |
| Subunit size (nm) | 5 | 45 (length), 2–3 (thickness) | 4 (dimer) | [130, 131] |
| Diameter (nm) | 7 | 10 | 25 | [124] |
| Persistence length (µm) |
15 ~ 20 3–10 |
~ 1.0 0.3 ~ 1.0 (vimentin) |
6000 1–8000 |
[26, 132, 133] |
| Bending stiffness (Nm2) | 7 × 10−26 | ~ 10−26 | 3 × 10−23 | [132] |
| Young’s modulus (kPa) |
1.3 × 106 ~ 2.5 × 106 ~ 2 × 106 |
~ 4 × 106 3 ~ 4 × 106 (non-stabilized filament) ~ 9 × 106 (non-stabilized filaments) |
~ 2 × 106 | [132, 134] |
| Shear modulus (kPa) | 0.283 | 0.032, 0.104 | 0.034 | [135] |
| Content (%) (actin/total cellular protein) |
7 ~ 14 (chick skin fibroblasts) 4.4 ~ 5.6 (non-tumorigenic) 2.7 ~ 3.97 (tumorigenic) |
~ 1.0 ~ 85 (epidermal keratinocyte, neurons) |
– | [102, 136, 137] |
| Region | Cell periphery | Central region | Central region | [138] |
| Function | Division of cytoplasm, motility, and muscle contraction, ear sensory cells | Stress-buffering, mechanical strength, maintenance of cell shape | Cell division, intercellular transport | [130, 134] |
In addition, the three cytoskeletal filaments are distinguished from each other by mechanical properties. A single actin filament is semi-flexible, but stiffens when bundled or networked via actin-binding proteins. Intermediate filaments exhibit a highly non-linear stress–strain relationship. For examples, vimentin and keratin are stiff at high strains, but softer than actin filaments at low strains [127, 139]. Microtubules exhibit a larger bending stiffness due to their tubular structure [46]. The stress–strain curves of the three cytoskeletal filaments with equal weight concentrations show differences in mechanical properties [140]. Actin filaments are highly resistant to deformation indicating high shear modulus that can rupture at ~ 20% strain [77]. Vimentin is easily deformed at low stresses and does not lose elasticity even under large stress and strain. Vimentin can withstand stresses of up to ~ 80% strain. Microtubules show the greatest deformability at low stress, but break down above ~ 50% strain.
Although we have much information about the F-actin, intermediate filaments, and microtubules in terms of intracellular elasticity, their properties and roles do not explain cellular elasticity quantitatively. First, because the content of F-actin is closely related to cell elasticity, the continuously or easily changed F-actin concentration makes it difficult to quantitatively characterize the mechanical properties of a cell. The concentration of F-actin can be changed by external stimuli, migration, and cell cycle processes. F-actin concentration increases by approximately 10–15% as cells enter S-phase in untransformed cells, while the concentration increases by approximately 13% during G2 + M phase in transformed cells [141]. The concentration changes not only transiently, but also permanently due to aging and diseases.
Second, although the mechanical property of single F-actin is well understood, the property changes when interacting with various binding proteins in complex ways. The degree of change depends on the variety and concentration of binding proteins. In many cases, F-actin interacts with two or more binding proteins simultaneously, making it difficult to interpret the change in elasticity clearly. For example, the elastic modulus of F-actin networks increased by twofold with 0.12 µM α-actinin and filamin separately, and by 12-fold when combining α-actinin and filamin [72]. The synergistic effect was not observed with 24 µM combining of α-actinin and filamin, but the increase of the elasticity by the filamin alone was remarkable.
Third, the three cytoskeletal filaments associate with each other, causing changes in the elastic property of both the filaments themselves and the cells as whole. Many studies have demonstrated that interactions between actin, microtubules, and intermediate filaments influence intracellular elasticity. For example, the amount of F-actin and microtubules is regulated by crosstalk [138]. Increasing densities of F-actin at the centrosome leads to reduced microtubule production. However, decrease in F-actin density due to cell adhesion and spreading activates microtubule growth. These interactions are related to intracellular elasticity. F-actin networks can be either stiffened or softened by co-polymerization with vimentin, depending on the concentrations and density of crosslinkers [142]. The length of F-actin can also change after interacting with vimentin [130]. However, the underlying mechanisms of these interactions are incompletely understood.
In conclusion, a deeper understanding of the mechanical characteristics of the cells will offer valuable insight into cellular physiology. Thanks to recent advances in biophysical techniques, the magnitudes of elastic moduli of a variety of cell types have been measured. Visualization of the structural changes in the cytoskeletal filaments of living cells during mechanical stimulation has been established using advanced imaging techniques, such as confocal microscopy and AFM. These measurements reveal remarkable elastic properties of cells adapted to environmental changes and surprising behaviors of individual cytoskeletal filaments. However, quantification of the elastic property of cells and the contribution of individual cytoskeletal filaments to the overall mechanical response of a cell remains challenging due to the complex and heterogeneous structures of the subcellular components.
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Education (Grant No. NRF-2017R1A2B2010145).
Author contributions
Sangwoo Kwon and Kyung Sook Kim conceived this article and wrote the manuscript.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gurkan UA, Akkus O. The mechanical environment of bone marrow: a review. Ann Biomed Eng. 2008;36:1978–1991. doi: 10.1007/s10439-008-9577-x. [DOI] [PubMed] [Google Scholar]
- 2.Ofek G, Willard VP, Koay EJ, Hu JC, Lin P, Athanasiou KA. Mechanical characterization of differentiated human embryonic stem cells. J Biomech Eng. 2009;131(6):061011. doi: 10.1115/1.3127262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sens P, Plastino J. Membrane tension and cytoskeleton organization in cell motility. J Phys Condens Matter. 2015;27(27):273103. doi: 10.1088/0953-8984/27/27/273103. [DOI] [PubMed] [Google Scholar]
- 4.Fung YC. Biomechanics: mechanical properties of living tissues. 2. New York: Springer; 1993. [Google Scholar]
- 5.Morrison B, 3rd, Saatman KE, Meaney DF, McIntosh TK. In vitro central nervous system models of mechanically induced trauma: a review. J Neurotrauma. 1998;15(11):911–928. doi: 10.1089/neu.1998.15.911. [DOI] [PubMed] [Google Scholar]
- 6.Xu W, Mezencev R, Kim B, Wang L, McDonald J, Sulchek T. Cell stiffness is a biomarker of the metastatic potential of ovarian cancer cells. PLoS One. 2012;7(10):e46609. doi: 10.1371/journal.pone.0046609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cross SE, Jin YS, Rao J, Gimzewski JK. Nanomechanical analysis of cells from cancer patients. Nat Nanotechnol. 2007;2(12):780–783. doi: 10.1038/nnano.2007.388. [DOI] [PubMed] [Google Scholar]
- 8.Ramos JR, Pabijan J, Garcia R, Lekka M. The softening of human bladder cancer cells happens at an early stage of the malignancy process. Beilstein J Nanotechnol. 2014;10(5):447–457. doi: 10.3762/bjnano.5.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mierke CT, Frey B, Fellner M, Herrmann M, Fabry B. Integrin α5β1 facilitates cancer cell invasion through enhanced contractile forces. J Cell Sci. 2011;124(Pt 3):369–383. doi: 10.1242/jcs.071985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Buxboim A, Ivanovska IL, Discher DE. Matrix elasticity, cytoskeletal forces and physics of the nucleus: how deeply do cells ‘feel’ outside and in? J Cell Sci. 2010;123:297–308. doi: 10.1242/jcs.041186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Samani A, Plewes D. A method to measure the hyperelastic parameters of ex vivo breast tissue samples. Phys Med Biol. 2004;49:4395–4405. doi: 10.1088/0031-9155/49/18/014. [DOI] [PubMed] [Google Scholar]
- 12.Depalle B, Qin Z, Shefelbine SJ, Buehler MJ. Influence of cross-link structure, density and mechanical properties in the mesoscale deformation mechanisms of collagen fibrils. J Mech Behav Biomed Mater. 2015;52:1–13. doi: 10.1016/j.jmbbm.2014.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Canetta Elisabetta, Duperray Alain, Leyrat Anne, Verdier Claude. Measuring cell viscoelastic properties using a force-spectrometer: influence of protein-cytoplasm interactions. Biorheology. 2005;42(5):321–333. [PMC free article] [PubMed] [Google Scholar]
- 14.García AJ, Reyes CD. Bio-adhesive surfaces to promote osteoblast differentiation and bone formation. J Dent Res. 2005;84(5):407–413. doi: 10.1177/154405910508400502. [DOI] [PubMed] [Google Scholar]
- 15.Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell. 2002;110(6):673–687. doi: 10.1016/S0092-8674(02)00971-6. [DOI] [PubMed] [Google Scholar]
- 16.Juliano RL, Haskill S. Signal transduction from the extracellular matrix. J Cell Biol. 1993;120(3):577–585. doi: 10.1083/jcb.120.3.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bernstein LR, Liotta LA. Molecular mediators of interactions with extracellular matrix components in metastasis and angiogenesis. Curr Opin Oncol. 1994;6(1):106–113. doi: 10.1097/00001622-199401000-00015. [DOI] [PubMed] [Google Scholar]
- 18.Stamenović D, Ingber DE. Tensegrity-guided self-assembly: from molecules to living cells. Soft Matter. 2009;5(6):1137–1145. doi: 10.1039/B806442C. [DOI] [Google Scholar]
- 19.Alonso JL, Goldmann WH. Cellular mechanotransduction. AIMS. Biophysics. 2016;3(1):50–62. doi: 10.3934/biophy.2016.1.50. [DOI] [Google Scholar]
- 20.Jay DH, Eric RD, Martin AS. Mechanotransduction and extracellular matrix homeostasis. Nat Rev Mol Cell Biol. 2014;15(12):802–812. doi: 10.1038/nrm3896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martino F, Perestrelo AR, Vinarský V, Pagliari S, Forte G. Cellular mechanotransduction: from tension to function. Front Physiol. 2018;9:824. doi: 10.3389/fphys.2018.00824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dunn GA, Brown AF. Alignment of fibroblasts on grooved surfaces described by a simple geometric transformation. J Cell Sci. 1986;83:313–340. doi: 10.1242/jcs.83.1.313. [DOI] [PubMed] [Google Scholar]
- 23.Yeung T, Georges PC, Flanagan LA, Marg B, Ortiz M, Funaki M, Zahir N, Ming W, Weaver V, Janmey PA. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil Cytoskeleton. 2005;60(1):24–34. doi: 10.1002/cm.20041. [DOI] [PubMed] [Google Scholar]
- 24.Trepat X, Lenormand G, Fredberg JJ. Universality in cell mechanics. Soft Matter. 2008;4(9):1750–1759. doi: 10.1039/b804866e. [DOI] [Google Scholar]
- 25.Rodriguez ML, McGarry PJ, Sniadecki NJ. Review on cell mechanics: experimental and modeling approaches. Appl Mech Rev. 2013;65(6):060801. doi: 10.1115/1.4025355. [DOI] [Google Scholar]
- 26.Moeendarbary E, Harris AR. Cell mechanics: principles, practices, and prospects. Wiley Interdiscip Rev Syst Biol Med. 2014;6(5):371–388. doi: 10.1002/wsbm.1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jansen KA, Donato DM, Balcioglu HE, Schmidt T, Danen EHJ, Koenderink GH. A guide to mechanobiology: where biology and physics meet. Biochim Biophys Acta. 2015;1853(11 Pt B):3043–3052. doi: 10.1016/j.bbamcr.2015.05.007. [DOI] [PubMed] [Google Scholar]
- 28.Nestor-Bergmann A, Johns E, Sarah Woolner, Jensen OE. Mechanical characterization of disordered and anisotropic cellular monolayers. Phys Rev E. 2018;97(5):052409. doi: 10.1103/PhysRevE.97.052409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fletcher DA, Mullins RD. Cell mechanics and the cytoskeleton. Nature. 2010;463(7280):485–492. doi: 10.1038/nature08908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pritchard RH, Huang YYS, Terentjev EM. Mechanics of biological networks: from the cell cytoskeleton to connective tissue. Soft Matter. 2014;10(12):1864–1884. doi: 10.1039/c3sm52769g. [DOI] [PubMed] [Google Scholar]
- 31.Gardel ML, Kasza KE, Brangwynne CP, Liu J, Weitz DA. Mechanical response of cytoskeletal networks. Methods Cell Biol. 2015;89:487–519. doi: 10.1016/S0091-679X(08)00619-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hohmann T, Dehghani F. The cytoskeleton-A complex interacting meshwork. Cells. 2019;8(4):362. doi: 10.3390/cells8040362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lodish H, Berk A, Zipursky SL, Matsudaira P, Baltimore D, Darnell J. Molecular cell biology. 4. New York: Freeman; 2000. [Google Scholar]
- 34.Kreis T, Vale R. Cytoskeletal and motor proteins. Oxford: Oxford University Press; 1999. [Google Scholar]
- 35.Knowles GC, McCUUOCH CAG. Simultaneous localization and quantification of relative G and F actin content: optimization of fluorescence labeling methods. J Histochem Cytochem. 1992;40(10):1605–1612. doi: 10.1177/40.10.1527379. [DOI] [PubMed] [Google Scholar]
- 36.Pollard TD. Rate constants for the reactions of ATP- and ADP-actin with the ends of actin filaments. J Cell Biol. 1986;103:2747–2754. doi: 10.1083/jcb.103.6.2747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dominguez R, Holmes KC. Actin structure and function. Annu Rev Biophys. 2011;40:169–186. doi: 10.1146/annurev-biophys-042910-155359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kubitschke H, Schnauss J, Nnetu KD, Warmt E, Stange R, Kaes J. Actin and microtubule networks contribute differently to cell response for small and large strains. New J Phys. 2017;19:093003. doi: 10.1088/1367-2630/aa7658. [DOI] [Google Scholar]
- 39.Rotsch C, Radmacher M. Drug-induced changes of cytoskeletal structure and mechanics in fibroblasts: an atomic force microscopy study. Biophys J. 2000;78(1):520–535. doi: 10.1016/S0006-3495(00)76614-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.MacLean-Fletcher S, Pollard TD. Mechanism of action of cytochalasin B on actin. Cell. 1980;20(2):329–341. doi: 10.1016/0092-8674(80)90619-4. [DOI] [PubMed] [Google Scholar]
- 41.Ping Ting-Beall H, Lee Anne S, Hochmuth Robert M. Effect of cytochalasin D on the mechanical properties and morphology of passive human neutrophils. Ann Biomed Eng. 1995;23(5):666–671. doi: 10.1007/BF02584463. [DOI] [PubMed] [Google Scholar]
- 42.Goddette DW, Frieden C. Actin polymerization. The mechanism of action of cytochalasin D. J Biol Chem. 1986;261(34):15974–15980. [PubMed] [Google Scholar]
- 43.Holzinger A. Jasplakinolide: an actin-specific reagent that promotes actin polymerization. Methods Mol Biol. 2009;586:71–87. doi: 10.1007/978-1-60761-376-3_4. [DOI] [PubMed] [Google Scholar]
- 44.Kunschmann T, Puder S, Fischer T, Steffen A, Rottner K, Mierke CT. The small GTPase Rac1 increases cell surface stiffness and enhances 3D migration into extracellular matrices. Sci Rep. 2019;9:7675. doi: 10.1038/s41598-019-43975-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cai X, Xing X, Cai J, Chen Q, Wu S, Huang F. Connection between biomechanics and cytoskeleton structure of lymphocyte and Jurkat cells: an AFM study. Micron. 2010;41(3):257–262. doi: 10.1016/j.micron.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 46.Grady ME, Composto RJ, Eckmann DM. Cell elasticity with altered cytoskeletal architectures across multiple cell types. J Mech Behav Biomed Mater. 2016;61:197–207. doi: 10.1016/j.jmbbm.2016.01.022. [DOI] [PubMed] [Google Scholar]
- 47.Saito H, Minamiya Y, Kalina U, Saito S, Ogawa J. Effect of antithrombin III on neutrophil deformability. J Leukoc Biol. 2005;78(3):777–784. doi: 10.1189/jlb.1004589. [DOI] [PubMed] [Google Scholar]
- 48.Tavares S, Vieira AF, Taubenberger AV, Araújo M, Martins NP, Brás-Pereira C, Polónia A, Herbig M, Barreto C, Otto O, Cardoso J, Pereira-Leal JB, Guck J, Paredes J, Janody F. Actin stress fiber organization promotes cell stiffening and proliferation of pre-invasive breast cancer cells. Nat Commun. 2017;8:15237. doi: 10.1038/ncomms15237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gardel ML, Shin JH, MacKintosh FC, Mahadevan L, Matsudaira P, Weitz DA. Elastic behavior of cross-linked and bundled actin networks. Science. 2004;304(5675):1301–1305. doi: 10.1126/science.1095087. [DOI] [PubMed] [Google Scholar]
- 50.Schulze C, Wetzel F, Kueper T, Malsen A, Muhr G, Jaspers S, Blatt T, Wittern KP, Wenck H, Käs JA. Stiffening of human skin fibroblasts with age. Clin Plast Surg. 2012;39(1):9–20. doi: 10.1016/j.cps.2011.09.008. [DOI] [PubMed] [Google Scholar]
- 51.Kassianidou E, Kumar S. A biomechanical perspective on stress fiber structure and function. Biochem Biophys Acta. 2015;1853(11):3065–3074. doi: 10.1016/j.bbamcr.2015.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gardel ML, et al. Elastic behavior of cross-linked and bundled actin networks. Science. 2004;304(5675):1301–1305. doi: 10.1126/science.1095087. [DOI] [PubMed] [Google Scholar]
- 53.Head DA, Levine AJ, MacKintosh FC. Deformation of cross-linked semiflexible polymer networks. Phys Rev Lett. 2003;91(10):108102. doi: 10.1103/PhysRevLett.91.108102. [DOI] [PubMed] [Google Scholar]
- 54.Head DA, Levine AJ, MacKintosh FC. Distinct regimes of elastic response and deformation modes of cross-linked cytoskeletal and semiflexible polymer networks. Phys Rev E Stat Nonlin Soft Matter Phys. 2003;68(6 Pt 1):061907. doi: 10.1103/PhysRevE.68.061907. [DOI] [PubMed] [Google Scholar]
- 55.Stricker J, Falzone T, Gardel M. Mechanics of the F-actin cytoskeleton. J Biomech. 2010;43(1):9–14. doi: 10.1016/j.jbiomech.2009.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Peterson LJ, Rajfur Z, Maddox AS, Freel CD, Chen Y, Edlund M, Otey C, Burridge K. Simultaneous stretching and contraction of stress fibers in vivo. Mol Biol Cell. 2004;15(7):3497–3508. doi: 10.1091/mbc.e03-09-0696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Weber K, Groeschel-Stewart U. Antibody to myosin: the specific visualization of myosin-containing filaments in nonmuscle cells. Proc Natl Acad Sci USA. 1974;71(11):4561–4564. doi: 10.1073/pnas.71.11.4561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cramer LP, Siebert M, Mitchison TJ. Identification of novel graded polarity actin filament bundles in locomoting heart fibroblasts: implications for the generation of motile force. J Cell Biol. 1997;136:1287–1305. doi: 10.1083/jcb.136.6.1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Adams JC. Formation of stable microspikes containing actin and the 55 kDa actin bundling protein, fascin, is a consequence of cell adhesion to thrombospondin-1: implications for the anti-adhesive activities of thrombospondin-1. J Cell Sci. 1995;108:1977–1990. doi: 10.1242/jcs.108.5.1977. [DOI] [PubMed] [Google Scholar]
- 60.Chen B, Li A, Wang D, Wang M, Zheng L, Bartles JR. Espin contains an additional actin-binding site in its N terminus and is a major actin-bundling protein of the sertoli cell-spermatid ectoplasmic specialization junctional plaque. Mol Biol Cell. 1999;10:4327–4339. doi: 10.1091/mbc.10.12.4327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wang K, Ash JF, Singer SJ. Filamin, a new high-molecular-weight protein found in smooth muscle and non-muscle cells. Proc Natl Acad Sci USA. 1975;72:4483–4486. doi: 10.1073/pnas.72.11.4483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lu L, Oswald SJ, Ngu H, Yin FC. Mechanical properties of actin stress fibers in living cells. Biophys J. 2008;95(12):6060–6071. doi: 10.1529/biophysj.108.133462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Mathur AB, Collinsworth AM, Reichert WM, Kraus WE, Truskey GA. Endothelial, cardiac muscle and skeletal muscle exhibit different viscous and elastic properties as determined by atomic force microscopy. J Biomech. 2001;34(12):1545–1553. doi: 10.1016/S0021-9290(01)00149-X. [DOI] [PubMed] [Google Scholar]
- 64.Stricker J, Falzone T, Gardel ML. Mechanics of the F-actin cytoskeleton. J Biomech. 2010;43(1):9–14. doi: 10.1016/j.jbiomech.2009.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.dos Remedios CG, Chhabra D, Kekic M, Dedova IV, Tsubakihara M, Berry DA, Nosworthy NJ. Actin binding proteins: regulation of cytoskeletal microfilaments. Physiol Rev. 2003;83(2):433–473. doi: 10.1152/physrev.00026.2002. [DOI] [PubMed] [Google Scholar]
- 66.Van Troys M, Vandekerckhove J, Ampe C. Structural modules in actin-binding proteins: towards a new classification. Biochim Biophys Acta. 1999;1448(3):323–348. doi: 10.1016/S0167-4889(98)00152-9. [DOI] [PubMed] [Google Scholar]
- 67.Blanchard A, Ohanian V, Critchley D. The structure and function of alpha-actinin. J Muscle Res Cell Motil. 1989;10:280–289. doi: 10.1007/BF01758424. [DOI] [PubMed] [Google Scholar]
- 68.Masaki T, Endo M, Ebashi S. Localization of 6S component of a alpha-actinin at Z-band. J Biochem. 1967;62:630–632. doi: 10.1093/oxfordjournals.jbchem.a128717. [DOI] [PubMed] [Google Scholar]
- 69.Bond M, Somlyo AV. Dense bodies and actin polarity in vertebrate smooth muscle. J Cell Biol. 1982;95:403–413. doi: 10.1083/jcb.95.2.403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lazarides E, Burridge K. Alpha-actinin: immunofluorescent localization of a muscle structural protein in nonmuscle cells. Cell. 1975;6:289–298. doi: 10.1016/0092-8674(75)90180-4. [DOI] [PubMed] [Google Scholar]
- 71.Wachsstock D, Schwarz WH, Pollard TD. Affinity of a-actinin for actin determines the structure and mechanical properties of actin filament gels. Biophys J. 1993;65:205–214. doi: 10.1016/S0006-3495(93)81059-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Esue Osigwe, Tseng Yiider, Wirtz Denis. α-Actinin and filamin cooperatively enhance the stiffness of actin filament networks. PLoS One. 2009;4(2):e4411. doi: 10.1371/journal.pone.0004411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Gardel ML, Nakamura F, Hartwig JH, Crocker JC, Stossel TP, Weitz DA. Prestressed F-actin networks cross-linked by hinged filamins replicate mechanical properties of cells. Proc Natl Acad Sci USA. 2006;103(6):1762–1767. doi: 10.1073/pnas.0504777103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Tseng Y, An KM, Esue O, Wirtz D. The bimodal role of filamin in controlling the architecture and mechanics of F-actin networks. J Biol Chem. 2004;279:1819–1826. doi: 10.1074/jbc.M306090200. [DOI] [PubMed] [Google Scholar]
- 75.Tseng Y, Wirtz D. Mechanics and multiple-particle tracking microheterogeneity of alpha-actinin-cross-linked actin filament networks. Biophys J. 2001;81:1643–1656. doi: 10.1016/S0006-3495(01)75818-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tseng Y, Fedorov E, McCaffery JM, Almo SC, Wirtz D. Micromechanics and microstructure of actin filament networks in the presence of the actin-bundling protein human fascin: a comparison with a-actinin. J Mol Biol. 2001;310:351–366. doi: 10.1006/jmbi.2001.4716. [DOI] [PubMed] [Google Scholar]
- 77.Storm C, Pastore JJ, MacKintosh FC, Lubensky TC, Janmey PA. Nonlinear elasticity in biological gels. Nature. 2005;435(7039):191–194. doi: 10.1038/nature03521. [DOI] [PubMed] [Google Scholar]
- 78.Pegoraro AF, Janmey P, Weitz DA. Mechanical properties of the cytoskeleton and Cells. Cold Spring Harb Perspect Biol. 2017;9(11):a022038. doi: 10.1101/cshperspect.a022038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Jackson WM, Jaasma MJ, Baik AD, Keaveny TM. Over-expression of alpha-actinin with a GFP fusion protein is sufficient to increase whole-cell stiffness in human osteoblasts. Ann Biomed Eng. 2008;36(10):1605–1614. doi: 10.1007/s10439-008-9533-9. [DOI] [PubMed] [Google Scholar]
- 80.Tseng Y, Kole TP, Lee JS, Fedorov E, Almo SC, Schafer BW, Wirtz D. How actin crosslinking and bundling proteins cooperate to generate an enhanced cell mechanical response. Biochem Biophys Res Commun. 2005;334(1):183–192. doi: 10.1016/j.bbrc.2005.05.205. [DOI] [PubMed] [Google Scholar]
- 81.Uyeda TQ, Iwadate Y, Umeki N, Nagasaki A, Yumura S. Stretching actin filaments within cells enhances their affinity for the myosin II motor domain. PLoS One. 2011;6(10):e26200. doi: 10.1371/journal.pone.0026200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Westphal M, Jungbluth A, Heidecker M, Mühlbauer B, Heizer C, Schwartz JM, Marriott G, Gerisch G. Microfilament dynamics during cell movement and chemotaxis monitored using a GFP-actin fusion protein. Curr Biol. 1997;7:176–183. doi: 10.1016/S0960-9822(97)70088-5. [DOI] [PubMed] [Google Scholar]
- 83.Asano Y, Mizuno T, Kon T, Nagasaki A, Sutoh K, Uyeda TQ. Keratocyte-like locomotion in amiB-null Dictyostelium cells. Cell Motil Cytoskeleton. 2004;59:17–27. doi: 10.1002/cm.20015. [DOI] [PubMed] [Google Scholar]
- 84.Koenderink GH, Dogic Z, Nakamura F, Bendix PM, MacKintosh FC, Hartwig JH, Stossel TP, Weitz DA. An active biopolymer network controlled by molecular motors. Proc Natl Acad Sci USA. 2009;106(36):15192–15197. doi: 10.1073/pnas.0903974106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Koenderink GH, Dogic Z, Nakamura F, Bendix PM, MacKintosh FC, Hartwig JH, Stossel TP, Weitz DA. An active biopolymer network controlled by molecular motors. Proc Natl Acad Sci USA. 2009;106(36):15192–15197. doi: 10.1073/pnas.0903974106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Stewart M, Kensler R. Arrangement of myosin heads in relaxed thick filaments from frog skeletal muscle. J Mol Biol. 1986;192:831–851. doi: 10.1016/0022-2836(86)90032-X. [DOI] [PubMed] [Google Scholar]
- 87.Schäfer A, Radmacher M. Influence of myosin II activity on stiffness of fibroblast cells. Acta Biomater. 2005;1(3):273–280. doi: 10.1016/j.actbio.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 88.Martens JC, Radmacher M. Softening of the actin cytoskeleton by inhibition of myosin II. Pflugers Arch. 2008;456(1):95–100. doi: 10.1007/s00424-007-0419-8. [DOI] [PubMed] [Google Scholar]
- 89.Langanger G, de Mey J, Moeremans M, Daneels G, de Brabander M, Small JV. Ultrastructural localization of alpha-actinin and filamin in cultured cells with the immunogold staining (IGS) method. J Cell Biol. 1984;99(4 Pt 1):1324–1334. doi: 10.1083/jcb.99.4.1324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Bloch RJ, Hall ZW. Cytoskeletal components of the vertebrate neuromuscular junction: vinculin, alpha-actinin, and filamin. J Cell Biol. 1983;97:217–223. doi: 10.1083/jcb.97.1.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kasza KE, Nakamura F, Hu S, Kollmannsberger P, Bonakdar N, Fabry B, Stossel TP, Wang N, Weitz DA. Filamin A is essential for active cell stiffening but not passive stiffening under external force. Biophys J. 2009;96(10):4326–4335. doi: 10.1016/j.bpj.2009.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kato K, Ohmori Y, Mizutani T, Haga H, Ohashi K, Ito T, Kawabata K. The role of actin-binding protein filamin a in cellular stiffness and morphology studied by wide-range scanning probe microscopy. Jpn J Appl Phys. 2006;45(1):2328. doi: 10.1143/JJAP.45.2328. [DOI] [Google Scholar]
- 93.Bryce NS, Schevzov G, Ferguson V, Percival JM, Lin JJ, Matsumura F, Bamburg JR, Jeffrey PL, Hardeman EC, Gunning P, Weinberger RP. Specification of actin filament function and molecular composition by tropomyosin isoforms. Mol Biol Cell. 2003;14(3):1002–1016. doi: 10.1091/mbc.e02-04-0244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.McMichael BK, Kotadiya P, Singh T, Holliday LS, Lee BS. Tropomyosin isoforms localize to distinct microfilament populations in osteoclasts. Bone. 2006;39:694–705. doi: 10.1016/j.bone.2006.04.031. [DOI] [PubMed] [Google Scholar]
- 95.Coulton AT, East DA, Galinska-Rakoczy A, Lehman W, Mulvihill DP. The recruitment of acetylated and unacetylated tropomyosin to distinct actin polymers permits the discrete regulation of specific myosins in fission yeast. J Cell Sci. 2010;123:3235–3243. doi: 10.1242/jcs.069971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Creed SJ, Desouza M, Bamburg JR, Gunning P, Stehn J. Tropomyosin isoform 3 promotes the formation of filopodia by regulating the recruitment of actin-binding proteins to actin filaments. Exp Cell Res. 2011;317:249–261. doi: 10.1016/j.yexcr.2010.10.019. [DOI] [PubMed] [Google Scholar]
- 97.Gunning P, O’Neill G, Hardeman E. Tropomyosin-based regulation of the actin cytoskeleton in time and space. Physiol Rev. 2008;88:1–35. doi: 10.1152/physrev.00001.2007. [DOI] [PubMed] [Google Scholar]
- 98.Sui Z, Gokhin DS, Nowak RB, Guo X, An X, Fowler VM. Stabilization of F-actin by tropomyosin isoforms regulates the morphology and mechanical behavior of red blood cells. Mol Biol Cell. 2017;28(19):2531–2542. doi: 10.1091/mbc.e16-10-0699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Jalilian I, Heu C, Cheng H, Freittag H, Desouza M, Stehn JR, Bryce NS, Whan RM, Hardeman EC, Fath T, Schevzov G, Gunning PW. Cell elasticity is regulated by the tropomyosin isoform composition of the actin cytoskeleton. PLoS One. 2015;10(5):e0126214. doi: 10.1371/journal.pone.0126214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Steinert Peter M, Jones Jonathan CR, Robert D. Goldman, intermediate filaments. J Cell Biol. 1984;99(1):22s–27s. doi: 10.1083/jcb.99.1.22s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Stewart Murray. Intermediate filament structure and assembly. Curr Opin Cell Biol. 1993;5(1):3–11. doi: 10.1016/S0955-0674(05)80002-X. [DOI] [PubMed] [Google Scholar]
- 102.Coulombe Pierre A, Ma Linglei, Yamada Soichiro, Wawersik Matthew. Intermediate filaments at a glance. J Cell Sci. 2001;114(24):4345–4347. doi: 10.1242/jcs.114.24.4345. [DOI] [PubMed] [Google Scholar]
- 103.Omary MB, Coulombe PA, McLean WH. Intermediate filament proteins and their associated diseases. N Engl J Med. 2004;351(20):2087–2100. doi: 10.1056/NEJMra040319. [DOI] [PubMed] [Google Scholar]
- 104.Guo M, Ehrlicher AJ, Mahammad S, Fabich H, Jensen MH, Moore JR, Fredberg JJ, Goldman RD, Weitz DA. The role of vimentin intermediate filaments in cortical and cytoplasmic mechanics. Biophys J. 2013;105(7):1562–1568. doi: 10.1016/j.bpj.2013.08.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lowery Jason, Kuczmarski Edward R, Herrmann Harald, Goldma Robert D. Intermediate filaments play a pivotal role in regulating cell architecture and function. Cell Mol Biol. 2015;290(28):17145–17153. doi: 10.1074/jbc.R115.640359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Haudenschild DR, Chen J, Pang N, Steklov N, Grogan SP, Lotz MK, D’Lima DD. Vimentin contributes to changes in chondrocyte stiffness in osteoarthritis. J Orthop Res. 2011;29(1):20–25. doi: 10.1002/jor.21198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Mendez MG, Restle D, Janmey PA. Vimentin enhances cell elastic behavior and protects against compressive stress. Biophys J. 2014;107(2):314–323. doi: 10.1016/j.bpj.2014.04.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Eggli PS, Hunziker EB, Schenk RK. Quantitation of structural features characterizing weight- and less-weight-bearing regions in articular cartilage: a stereological analysis of medial femoral condyles in young adult rabbits. Anat Rec. 1988;222(3):217–227. doi: 10.1002/ar.1092220302. [DOI] [PubMed] [Google Scholar]
- 109.Plodinec M, Loparic M, Suetterlin R, Herrmann H, Aebi U, Schoenenberger CA. The nanomechanical properties of rat fibroblasts are modulated by interfering with the vimentin intermediate filament system. J Struct Biol. 2011;174(3):476–484. doi: 10.1016/j.jsb.2011.03.011. [DOI] [PubMed] [Google Scholar]
- 110.Liu CY, Lin HH, Tang MJ, Wang YK. Vimentin contributes to epithelial-mesenchymal transition cancer cell mechanics by mediating cytoskeletal organization and focal adhesion maturation. Oncotarget. 2015;6(18):15966–15983. doi: 10.18632/oncotarget.3862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Guo M, Ehrlicher AJ, Mahammad S, Fabich H, Jensen MH, Moore JR, Fredberg JJ, Goldman RD, Weitz DA. The role of vimentin intermediate filaments in cortical and cytoplasmic mechanics. Biophys J. 2013;105(7):1562–1568. doi: 10.1016/j.bpj.2013.08.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Magin TM, Vijayaraj P, Leube RE. Structural and regulatory functions of keratins. Exp Cell Res. 2007;313(10):2021–2032. doi: 10.1016/j.yexcr.2007.03.005. [DOI] [PubMed] [Google Scholar]
- 113.Haines RL, Lane EB. Keratins and disease at a glance. J Cell Sci. 2012;125(Pt 17):3923–3928. doi: 10.1242/jcs.099655. [DOI] [PubMed] [Google Scholar]
- 114.Sivaramakrishnan S, DeGiulio JV, Lorand L, Goldman RD, Ridge KM. Micromechanical properties of keratin intermediate filament networks. Proc Natl Acad Sci USA. 2008;105(3):889–894. doi: 10.1073/pnas.0710728105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kuznetsova TG, Starodubtseva MN, Yegorenkov NI, Chizhik SA, Zhdanov RI. Atomic force microscopy probing of cell elasticity. Micron. 2007;38(8):824–833. doi: 10.1016/j.micron.2007.06.011. [DOI] [PubMed] [Google Scholar]
- 116.Lulevich V, Yang HY, Isseroff RR, Liu GY. Single cell mechanics of keratinocyte cells. Ultramicroscopy. 2010;110(12):1435–1442. doi: 10.1016/j.ultramic.2010.07.009. [DOI] [PubMed] [Google Scholar]
- 117.Fung CK, Xi N, Yang R, Seiffert-Sinha K, Lai KW, Sinha AA. Quantitative analysis of human keratinocyte cell elasticity using atomic force microscopy. IEEE Trans Nanobiosci. 2011;10(1):9–15. doi: 10.1109/TNB.2011.2113397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ramms L, Fabris G, Windoffer R, Schwarz N, Springer R, Zhou C, Lazar J, Stiefel S, Hersch N, Schnakenberg U, Magin TM, Leube RE, Merkel R, Hoffmann B. Keratins as the main component for the mechanical integrity of keratinocytes. Proc Natl Acad Sci USA. 2013;110(46):18513–18518. doi: 10.1073/pnas.1313491110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Kristin S, Anatol WF, Josef AK, Thomas MM. Keratins significantly contribute to cell stiffness and impact invasive behavior. PNAS. 2013;110(46):18507–18512. doi: 10.1073/pnas.1310493110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Bernal A, Arranz L. Nestin-expressing progenitor cells: function, identity and therapeutic implications. Cell Mol Life Sci. 2018;75(12):2177–2195. doi: 10.1007/s00018-018-2794-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Matsuda Y, Naito Z, Kawahara K, Nakazawa N, Korc M, Ishiwata T. Nestin is a novel target for suppressing pancreatic cancer cell migration, invasion and metastasis. Cancer Biol Ther. 2011;11:512–523. doi: 10.4161/cbt.11.5.14673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kleeberger W, Bova GS, Nielsen ME, Herawi M, Chuang AY, Epstein JI, Berman DM. Roles for the stem cell associated intermediate filament Nestin in prostate cancer migration and metastasis. Cancer Res. 2007;67:9199–9206. doi: 10.1158/0008-5472.CAN-07-0806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ayana Y, Moe S, Yuta T, Mei M, Mari M, Masumi I, Shun’ichi K, Tomoko O, Chikashi N. The structural function of nestin in cell body softening is correlated with cancer cell metastasis. Int J Biol Sci. 2019;15(7):1546–1556. doi: 10.7150/ijbs.33423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Nogales E. Structural insights into microtubule function. Annu Rev Biochem. 2000;69:277–302. doi: 10.1146/annurev.biochem.69.1.277. [DOI] [PubMed] [Google Scholar]
- 125.Avila J. Microtubule functions. Life Sci. 1992;50(5):327–334. doi: 10.1016/0024-3205(92)90433-P. [DOI] [PubMed] [Google Scholar]
- 126.Janmey PA, McCulloch CA. Cell mechanics: integrating cell responses to mechanical stimuli. Annu Rev Biomed Eng. 2007;9:1–34. doi: 10.1146/annurev.bioeng.9.060906.151927. [DOI] [PubMed] [Google Scholar]
- 127.Trickey WR, Vail TP, Guilak F. The role of the cytoskeleton in the viscoelastic properties of human articular chondrocytes. J Orthop Res. 2004;22(1):131–139. doi: 10.1016/S0736-0266(03)0150-5. [DOI] [PubMed] [Google Scholar]
- 128.PyleK SJ, Reuhl R (2010) Cytoskeletal elements in neurotoxicity, 2nd edn. In: Comprehensive toxicology, vol 13, pp 71–87
- 129.Brady S, Colman DR, Brophy P (2014) Subcellular organization of the nervous system: organelles and their functions, 3rd edn. In: From molecules to networks, pp 23–52
- 130.Shah JV, Wang LZ, Traub P, Janmey PA. Interaction of vimentin with actin and phospholipids. Biol Bull. 1998;194(3):402–405. doi: 10.2307/1543125. [DOI] [PubMed] [Google Scholar]
- 131.Feher J (2017) Cell structure, 2nd edn. In: Quantitative human physiology, pp 101–119
- 132.Nick P (2011) Mechanics of the cytoskeleton. In: Mechanical integration of plant cells and plants, pp 53–90
- 133.Mücke N, Kreplak L, Kirmse R, Wedig T, Herrmann H, Aebi U, Langowski J. Assessing the flexibility of intermediate filaments by atomic force microscopy. J Mol Biol. 2004;335(5):1241–1250. doi: 10.1016/j.jmb.2003.11.038. [DOI] [PubMed] [Google Scholar]
- 134.Guzmán C, Jeney S, Kreplak L, Kasas S, Kulik AJ, Aebi U, Forró L. Exploring the mechanical properties of single vimentin intermediate filaments by atomic force microscopy. J Mol Biol. 2006;360(3):623–630. doi: 10.1016/j.jmb.2006.05.030. [DOI] [PubMed] [Google Scholar]
- 135.Janmey PA, Euteneuer U, Traub P, Schliwa M. Viscoelastic properties of vimentin compared with other filamentous biopolymer networks. J Cell Biol. 1991;113(1):155–160. doi: 10.1083/jcb.113.1.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Gowing LR, Tellam RL, Banyard MR. Microfilament organization and total actin content are decreased in hybrids derived from the fusion of HeLa cells with human fibroblasts. J Cell Sci. 1984;69:137–146. doi: 10.1242/jcs.69.1.137. [DOI] [PubMed] [Google Scholar]
- 137.Bray D, Thomas C. The actin content of fibroblasts. Biochem J. 1975;147(2):221–228. doi: 10.1042/bj1470221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Inoue D, Obino D, Pineau J, Farina F, Gaillard J, Guerin C, Blanchoin L, Lennon-Duménil AM, Théry M. Actin filaments regulate microtubule growth at the centrosome. EMBO J. 2019;38(11):e99630. doi: 10.15252/embj.201899630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Wang N, Stamenovic D. Mechanics of vimentin intermediate filaments. J Muscle Res Cell Motil. 2002;23(5–6):535–540. doi: 10.1023/A:1023470709071. [DOI] [PubMed] [Google Scholar]
- 140.Ackbarow Theodor, Buehler Markus J. Superelasticity, energy dissipation and strain hardening of vimentin coiled-coil intermediate filaments: atomistic and continuum studies. J Mater Sci. 2007;42(21):8771–8787. doi: 10.1007/s10853-007-1719-2. [DOI] [Google Scholar]
- 141.Rao JY, Hurst RE, Bales WD, Jones PL, Bass RA, Archer LT, Bell PB, Hemstreet GP., 3rd Cellular F-actin levels as a marker for cellular transformation: relationship to cell division and differentiation. Cancer Res. 1990;50(8):2215–2220. [PubMed] [Google Scholar]
- 142.Jensen MH, Morris EJ, Goldman RD, Weitz DA. Emergent properties of composite semiflexible biopolymer networks. Bioarchitecture. 2014;4(4–5):138–143. doi: 10.4161/19490992.2014.989035. [DOI] [PMC free article] [PubMed] [Google Scholar]